Abstract
Background
This retrospective study compared the outcomes of laparoscopic complete mesocolic excision (CME) guided by superior mesenteric artery with laparoscopic conventional radical resection (CRR) performed for right-sided colon cancer.
Methods
Patients with right-sided colon cancer underwent CME (n=107; January 2011 to December 2015) or CRR (n=60; January 2008 to December 2010).
Results
The 2 groups were comparable regarding age, gender, body mass index, maximum tumor diameter, and tumor stage. In the CME group, the distances between the tumor and the high vascular tie (HVT; 12.6 cm), and between the closest bowel wall and HVT (10.4±0.9 cm) was significantly greater than that of the CRR group (11.5 cm and 9.3±1.0 cm, respectively; P<0.001). In the CME group, the number of retrieved lymph nodes (23.2) was significantly higher, and the volume of intraoperative bleeding (108.4 mL) was less than that of the CRR (14.0 and 128.7 mL; P<0.001). The length of resected bowel in the 2 groups was similar (25.8±0.7 cm and 25.5±2.1 cm; P=0.106), as was the operative time, postoperative hospitalization, time of first bowel movement, and complications. The 3-year recurrence rate of the CME group (8.4%) was significantly lower than that of the CRR (20.0%), the 3-year overall survival was significantly higher (93.5% cf. 85.0%), and the survival rates of T4 stage, N1 stage, pTNM stage II, pTNM stage III and lympho vascular invasion were significantly higher (P<0.05). The 2 groups were similar for survival rates of Tis, T1, T2, T3, N2 stage, pTNM stage I and perineural invasion (P>0.05).
Conclusion
CME for right-sided colon cancer guided by superior mesenteric artery has similar short-term outcomes, higher lymph node yield, and higher 3-year overall survival compared with CRR.
Keywords: laparoscopy, right-sided colon cancer, complete mesocolic excision
Background
Complete mesocolic excision (CME) was first proposed by the German scholar Hohenberger in 2009,1 and since has been widely used clinically as an open technique.1–3 The rationale of CME is the en bloc resection of the diseased colon with its mesocolon, and its lymphovascular supply, in an intact envelope of visceral peritoneum. Colectomy by laparoscopy is also now accepted as an alternative to conventional open surgery for colon cancer, because of less intraoperative bleeding and faster recovery.4
Bertelsen et al5 reported that laparoscopic CME can enhance the quality of surgery without affecting the incidence of postoperative complications. Subsequently, several studies have shown that laparoscopic CME is viable and safe.6–8 However, few studies have compared the outcomes of CME with laparoscopic conventional radical resection (CRR).
In our previous study, we described the spatial relationship of 3 surgical planes observed during laparoscopic CME for right colon cancer, which were identified via computed tomography (CT) reconstruction.9 Identification of these surgical planes improved the safety and effectiveness of the surgical procedure. In the current study, we retrospectively evaluated the outcomes of laparoscopic CME relative to that of laparoscopic CRR in patients with right-sided colon cancer.
Methods
The Institutional Ethics Committee of Shunde Hospital of Southern Medical University approved this study. All the participants gave written informed consent.
Patients
A retrospective analysis was conducted of patients undergoing CME or CRR (control group) for right-sided colon cancer at Shunde Hospital of Southern Medical University from January 2008 to December 2015.
All the patients analyzed in this study had histologically confirmed right-sided colon cancer confirmed on preoperative colonoscopic biopsy. The determination in these patients of postoperative pathological tumor-node-metastasis (pTNM) stages I-III (Union for International Cancer Control tumor classification, Seventh Edition) was based on histopathological examination of the resected tumors. Patients with any of the following were excluded from this study: neoadjuvant chemotherapy; colonic obstruction or perforation due to tumor; distant metastases such as liver or lung metastasis on CT/magnetic resonance imaging; past abdominal surgery with dense intraabdominal adhesions; or concurrent second malignancy.
The same group of 5 surgeons performed all the surgeries. The major surgeon was a Doctor of Medicine and chief physician, and the other surgeons were deputy chief physicians and attending physicians. These surgeons have been using laparoscopic techniques for right-sided colon cancer since 2003. Each surgeon had at least 5 years of experience in laparoscopic surgery for right-sided colon cancer and had performed ≥100 CRR and ≥20 CME laparoscopic surgeries before performing the surgeries of the study.
General surgical procedures
Preoperative preparations were similar to conventional open surgery. All the patients underwent bowel preparation. Patients were placed in the French position. Five ports were used: one 12-mm camera port just below the umbilicus; one 10 mm primary operating trocar at the anti-McBurney point in the left lower abdomen; one 5 mm auxiliary operating trocar at the McBurney point; and two 5 mm first-assistant ports along the right midclavicular line.
Laparoscopic CME
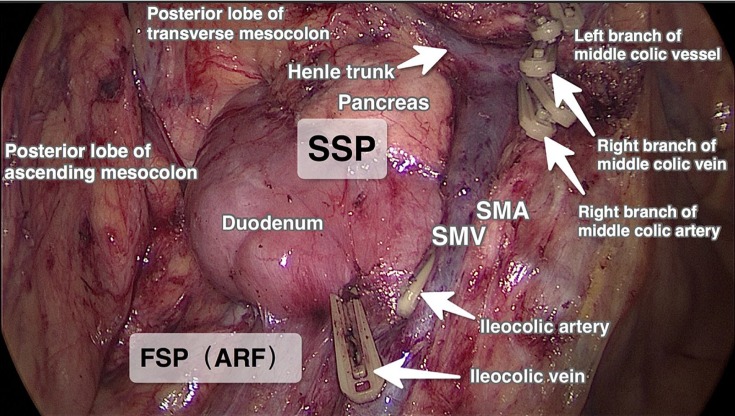
Three surgical planes were identified during laparoscopic CME, as described previously9 (Figures 1 and 2).
Figure 1.
Surgical planes in laparoscopic CME for ascending colon cancer.
Abbreviations: ARF, anterior renal fascial plane; FSP, first surgical plane; SMA, superior mesenteric artery; SMV, superior mesenteric vein; SSP, second surgical plane; CME, complete mesocolic excision.
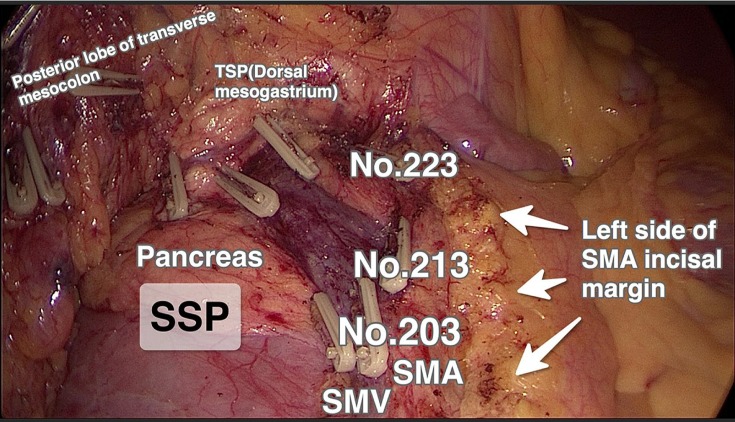
Figure 2.
Central lymph node group (N3; 203, 213, and 223) excision and the left side of SMA incisal margin in the laparoscopic CME group, for hepatic flexure colon cancer.
Abbreviations: TSP, third surgical plane; SMA, superior mesenteric artery; SMV, superior mesenteric vein; SSP, second surgical plane; CME, complete mesocolic excision.
During the laparoscopic CME, first the lower margin of the ileocolic vessels was identified and the mesentery was opened ~2 cm. This opening was extended laterally to reach the lower margin of the ascending mesocolon, and then medially to reach up to the root of the ileocolic vessels located at the right margin of the superior mesenteric vein (SMV). A sharp dissection was performed along the first surgical plane to mobilize the right colon from the retroperitoneum. On completion of the dissection up to the lower part of the duodenum, the root of the ileocolic vessels, SMV, and superior mesenteric artery (SMA) were visualized. The ileocolic vessels were transected at their roots on the right side of the SMV and the surrounding lymph nodes close to the SMV and the SMA surface (lymph nodal station 203) were dissected.
Moving cranially, the second surgical plane was opened. A sharp dissection was continued along the SMA and SMV to identify the radices of the trunk of Henle, right and middle colonic vessels, and right gastroepiploic vein. Subsequently, the right colonic artery and right colic vein were transected close to their roots and the surrounding lymph nodes (lymph nodal station 213) were dissected.
Using the right gastroepiploic vein as the posterior boundary, the third surgical plane was expanded. In tumors of the hepatic flexure (Figure 2), due to their bilateral lymphatic drainage, the middle colic artery was dissected and divided at its root on the right side of the SMA, and the nearby lymph nodes were dissected (lymph nodal station 223). In cecal and ascending colon cancers (Figure 1), lymph nodes located at station 223 were dissected and the right branch of the middle colic artery was divided, preserving the left branch of the middle colic artery. Finally, the greater omentum and the other attachments were divided to complete the CME procedure.
Laparoscopic CRR
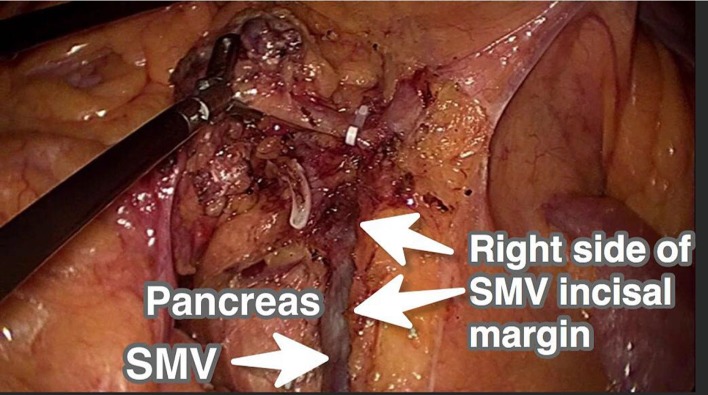
During the laparoscopic CRR, the lower margin of the ileocolic vessels was identified and the mesentery was cut open ~2 cm and extended on either side. Toldt’s space on the right was entered and the ascending mesocolon was mobilized, with the anterior fascia of the kidney as the posterior border. The ileocolic vessels were isolated and divided at their root close to the SMV, and surrounding lympho-adipose tissues were dissected. Blunt dissection was continued along the right edge of the SMV up to the radix of the trunk of Henle. The right colic vein and right colonic artery were ligated at the root to the right side of the SMV (Figure 3).
Figure 3.
Right side of SMV incisal margin in laparoscopic CRR for right-sided colon cancer.
Abbreviations: SMV, superior mesenteric vein; CRR, conventional radical resection.
Proceeding cephalad towards the radix of the middle colonic vessels, the middle colonic artery at its root was transected and the nearby lymph nodes were dissected. The ascending mesocolon was completely mobilized off the retroperitoneum by blunt dissection. The dissection was continued up to the pars horizontalis duodeni and head of the pancreas which were covered by their fascia to reach the root of the transverse mesocolon (Figure 3). Finally, the greater omentum, gastrocolic ligament, hepatocolic ligament, and right paracolic peritoneal folds were divided to complete the CRR procedure.
Specimen retrieval and anastomosis
Patients in both the CME and CRR groups received the same extent of resection, which included the terminal ileum, caecum, ascending colon, and hepatic flexure, with proximal and distal margins ≥5 cm from the lesion. Side-to-side ileocolonic anastomosis was performed in both groups using the same technique.
Adjuvant therapy
Adjuvant chemotherapy was administered based on the pathological stage of the disease, after thorough assessment by the clinical oncologists. The chemotherapy regimens were similar in both groups and in accordance with the National Comprehensive Cancer Network (NCCN) Guidelines (2008). Adjuvant therapy was given to all patients with pTNM stage III disease, and was selectively given to high-risk stage II patients as recommended in the NCCN guidelines. All of the patients in these two group were underwent the same adjuvants during the course of this research.
Follow-up
All the patients were evaluated in the outpatient department every 3 months during the first 3 years, every 6 months for the next 2 years, and then yearly afterwards. CT abdomen-pelvis, CT thorax and surveillance colonoscopy was performed within 6 months after surgery, and then annually until the patient’s condition dictated otherwise.
Statistical analysis
SPSS 13.0 software (Chicago, IL, USA) was used for statistical analyses. Continuous data with normal distribution are expressed as mean ± standard deviation, and were compared using the independent-sample t-test. The chi-squared test or Fisher’s exact probabilities test were used for categorical data. The log-rank test was used to compare the survival rates. Statistical significance was considered at P<0.05.
Results
Patient characteristics
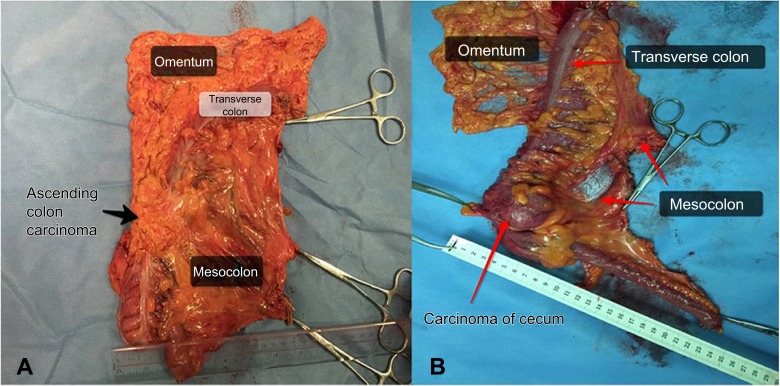
There were 107 patients in the CME group and 60 patients in the CRR group. There were no significant differences between the groups with regard to age, gender, body mass index (BMI), tumor size, lymphovascular invasion, or pTNM stage. However, the distance between the tumor and the high vascular tie (HVT) was longer in the CME group (12.6±1.3 cm) compared with the CRR group (11.5±1.1 cm, P<0.001). In addition, the distance between the closest bowel wall and the HVT was also longer in the CME group (10.4±0.9 cm) compared with the CRR group (9.3±1.0 cm, P<0.001). There was no remarkable difference in the length of the resected bowel between the CME (25.8±0.7 cm) and CRR group (25.5±2.1 cm; P=0.106; Table 1, Figure 4.)
Table 1.
Clinical characteristics and surgical data of the CME and CRR (control) groups
| CME | CRR | P | ||
|---|---|---|---|---|
| Subjects, n | 107 | 60 | ||
| Gender, n (%) | Male | 62 (57.6) | 34 (56.7) | 0.872 |
| Female | 45 (42.4) | 26 (43.3) | ||
| Age, y | 57.6±9.1 | 58.3±9.3 | 0.671 | |
| BMI, kg/m2 | 23.5±3.9 | 23.5±4.2 | 0.847 | |
| Tumor size, cm | 4.9±1.0 | 4.8±1.0 | 0.568 | |
| Tumor to HVT, cm | 12.6±1.3 | 11.5±1.1 | <0.001 | |
| Closest bowel wall to HVT, cm | 10.4±0.9 | 9.3±1.0 | <0.001 | |
| Length of the resected bowel | 25.8±0.7 | 25.5±2.1 | 0.106 | |
| pTNM, n (%) | I | 15 (14.0) | 10 (16.7) | 0.872 |
| II | 59 (55.1) | 31 (51.7) | ||
| III | 33 (30.9) | 19 (31.6) |
Figure 4.
Specimen of ascending colon cancer in laparoscopic CME. The distance between the tumor and HVT is ~13 cm.
Lymph node dissection
The mean number of lymph nodes harvested from patients in the CME group (23.2±6.8) was significantly higher than that of the control group (14.0±3.5; P<0.001). We stratified the analysis by pTNM stage, and at each stage, the CME group had significantly more lymph nodes harvested than did the CRR group. The incidence of positive lymph nodes in stage III cancer between the 2 groups did not differ significantly (P=0.473), but the number of negative lymph nodes in the CME group was significantly higher than that of the control group (P<0.001; Table 2)
Table 2.
Lymph nodes harvested in the CME and CRR groups, n
| CME | CRR | P | ||
|---|---|---|---|---|
| Subjects | 107 | 60 | ||
| Lymph nodes harvested | Stage I | 16.7±3.9 | 13.8±3.2 | <0.001 |
| Stage II | 21.4±5.1 | 13.9±3.7 | <0.001 | |
| Stage III | 24.8±5.4 | 16.2±4.1 | <0.001 | |
| Stage III lymph nodes | Positive (%) | 0.17±0.08 | 0.18±0.08 | 0.473 |
| Negative lymph nodes | Stage III | 20.6±4.6 | 13.2±3.5 | <0.001 |
Operative outcomes
Peritoneal seeding were not seen during the operation of all the patients in these two groups.
The operative time of the CME (192.4±31.4 min) and CRR (187.1±33.1 min) groups were statistically similar (P=0.306; Table 3). However, the mean blood loss in the CME group (108.4±23.9 mL) was significantly lower than that of the CRR group (128.7±25.2 mL; P<0.001). The CME and CRR groups had similar complication rates (12.1% and 16.7%, respectively; P=0.278). No postoperative deaths were observed in either group within 30 days after surgery (Table 3).
Table 3.
Operative outcomes in the CME and control groups
| CME | CRR | P | ||
|---|---|---|---|---|
| Operative time, min | 192.4±31.4 | 187.1±33.1 | 0.306 | |
| Intraoperative blood loss,mL, | 108.4±23.9 | 128.7±25.2 | <0.001 | |
| First flatus, d | 2.9±1.4 | 3.1±1.3 | 0.472 | |
| First stool, d | 3.8±1.1 | 4.1±1.0 | 0.066 | |
| Postoperative hospital stay, d | 6.44±2.1 | 7.29±2.9 | 0.077 | |
| Complications | Overall | 13 (12.1%) | 10 (16.7%) | 0.278 |
| Intestinal obstruction | 5 (4.7%) | 2 (3.3%) | 0.510 | |
| Wound infection | 3 (2.8%) | 2 (3.3%) | 0.592 | |
| Fat liquefaction of wound | 2 (1.9%) | 1 (1.7%) | 0.707 | |
| Anastomotic leakage | 1 (0.9%) | 3 (5.0%) | 0.133 | |
| Pulmonary infection | 1 (0.9%) | 1 (1.7%) | 0.519 | |
| Urinary tract infection | 1 (0.9%) | 1 (1.7%) | 0.519 |
The time from operation to first passage of flatus or first passage of stool in the CME group were each less than that of the control group, although the differences did not reach significance. Moreover, the postoperative hospital stays of the CME group (6.44±2.1 d) and CRR group (7.2±2.9 d; P=0.077) were statistically similar (Table 3).
The patients in both groups with postoperative ileus were successfully managed by gastrointestinal decompression, nutritional support, and fluid therapy. Cases of wound infection and fat liquefaction of the wound in the 2 groups were treated by wound dressing. For pulmonary and urinary tract infection, appropriate antibiotics, anti-inflammatory drugs, and symptomatic treatment were provided. In cases of anastomotic leakage in both groups, emergent redo-laparoscopy was conducted. Thorough peritoneal lavage with primary suturing of the leakage and proximal covering loop ileostomy was performed.
Oncologic 3-year outcomes
The median follow-up was 18 months for both the CME and CRR groups. Six and 9 patients in the CME and CRR groups, respectively, were lost to follow-up. Nine patients (8.4%) experienced cancer recurrence in the CME group: local recurrence in 3 patients, liver metastases in 2 patients, lung metastases in 2 patients, peritoneal carcinomatosis in one patient, and systemic multiple metastases in one patient. Twelve patients (20.0%) were found to have cancer recurrence in the CRR group: local recurrence in 4 patients; liver metastases in 3 patients; lung metastases in 2 patients; peritoneal carcinomatosis in 2 patients; and systemic multiple metastases in one patient.
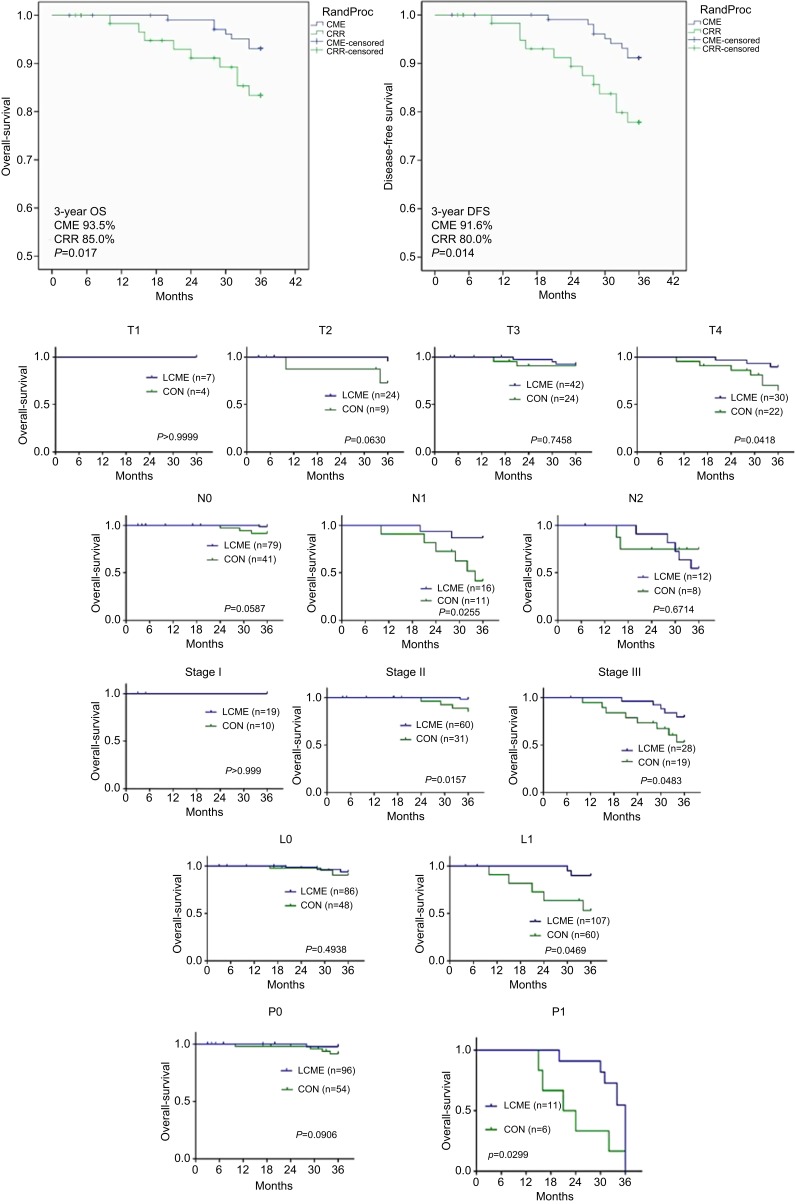
The 3-year overall survival and disease-free survival rates were significantly higher in the CME group (93.5% and 91.6%, respectively) than in the CRR group (85.0% and 80.0%; Figure 5). The 3-year survival rates of T4 stage, N1 stage, pTNM stage II, pTNM stage III and lympho vascular invasion were significantly higher (P<0.05, all). The 2 groups were similar for survival rates of Tis, T1, T2, T3, N2 stage, pTNM stage I and perineural invasion (P>0.05, all).
Figure 5.
Kaplan Meier curves representing disease-free survival and overall survival of the 2 groups.
Discussion
The rationale underlying CME is complete removal of the colon and intact mesocolon, along with its surrounding visceral peritoneum and fascia, high ligation of the vascular supply at its origin, and dissection of the central lymph nodes. Laparoscopic technique for performing colectomy is safe, reliable, and minimally invasive.10 Laparoscopic CME is meant to combine the advantages of laparoscopic colonic resection and CME to achieve better results. We began performing laparoscopic CME for right-sided colon cancer in the year 2010. Storli et al6 verified the advantages of laparoscopic CME relative to open surgery for colon cancer. In the present study, we demonstrated that laparoscopic CME to treat right-sided colon cancer has better oncological outcomes compared with laparoscopic CRR.
In our previous study, we reported that the posterior surface of the right-side colon comprises 3 surgical planes.9 By performing the laparoscopic CME procedure according to these planes, the integrity of the colonic mesocolon is maintained, which probably prevents tumor spread across the fascial planes. It also facilitates exposure and isolation of the root of the mesentery for ligation of the colonic blood vessels, and dissection of the central group of lymph nodes. These key principles of laparoscopic CME were probably responsible, in the present study, for the better harvest of lymph nodes and superior survival outcomes in the CME group relative to the CRR group.
A study by Galizia et al11 found that CME could help achieve better lymph node clearance compared with conventional surgery, by making hidden areas accessible. Similarly, Eiholm and Ovesen3 found that CME can not only improve lymph node dissection, but also improve the 5-year survival rate. In the present study, the number of dissected lymph nodes in the CME group was considerably higher than that of the CRR group (Table 2). This may be related to the dissection of the central group of lymph nodes. In laparoscopic CME, dissection is conducted close to the SMA, with high ligation of the nutrient vessels and excision of adjoining lymphatic tissue, including the central lymph node (N3) group (station no. 203, 213, and 223; Figure 2). Thus, CME reduces the possibility of residual positive lymph nodes. In contrast, conventional right colectomy is close to the right edge of the SMV and does not involve more lymph node dissection (Figure 3).
Lymph node metastasis is a risk factor for poor prognosis in colon cancer, and the number of dissected lymph nodes is closely related to prognosis and the adjuvant treatment protocol.12–14 In the present study, significantly more lymph nodes were dissected in the CME group than in the CRR group (Table 2). This was likely due to the larger area of the resected mesentery in the CME group, as suggested by the greater distance of the tumor to the HVT, and the greater distance between the closest bowel wall and HVT (Figure 4). Moreover, the yield of lymph nodes without metastasis in the patients with stage III colon cancer was significantly higher in comparison with the CRR group, although there was no significant difference in the detection rates of positive lymph nodes between the groups. This finding could be due to the small number of stage III patients with nodal disease in this study, and we will increase the sample size to verify these findings in the future.
Hohenberger et al1 and Pramateftakis14 reported rates of surgical complications of 19.7% and 13.9%, respectively, in patients who underwent CME. Similarly, Bertelsen et al15 observed a 14.7% rate of postoperative complications. Shin et al16 found that the complication rate after laparoscopic CME surgery was ~17.8%. For the present study, the rate of postoperative complications in the laparoscopic CME group was 12.1%. In addition, in our study there were no remarkable differences in operative time or complications between the CME and CRR groups. However, intraoperative blood loss was significantly lower in the CME group (Table 3). This suggests that, by dissecting according to the correct surgical planes, less bleeding occurs, outcomes are improved, and the rate of complications is no worse.
In the present study, the first passage of flatus occurred earlier in the CME group than in the CRR, and postoperative hospital stay was shorter, although these differences were not statistically significant. This may be related to the better surgical technique and less bleeding in the CME group.
With regard to long-term outcomes, the retrospective analysis of Hohenberger et al1 found that, in 1329 cases of colon cancer, use of the CME technique was associated with a decrease in the 5-y local recurrence rate of 6.9–3.6%, and the 5-year overall survival rate increased from 82.1–89.1%. Similarly, other studies have reported better local recurrence rate, disease free survival, and overall survival after CME compared with conventional radical resection for right-sided colon cancer.17–19 Our present long-term results are in agreement with these reports, with lower 3-year recurrence and higher 3-year overall survival rates associated with laparoscopic CME compared with laparoscopic CRR (Figure 5).
We also found that the rates of 3-year survival of patients with T4 stage, N1 stage, pTNM stage II, pTNM stage III and lympho vascular invasion in the CME group were higher than that of the CRR group (Figure 5). Lymphovascular invasion is closely related to lymph node metastasis, tumor recurrence, and survival rate in colorectal cancer, and can serve as a powerful complement to TNM staging. While the 2 groups had similar lymphovascular invasion statuses, the CME group experienced a significantly better 3-year survival rate. This may be due to the better harvest of lymph nodes and greater area of resected mesentery in the CME group, leading to a reduction in recurrence and improved survival.
This study is limited, first, because the laparoscopic CME procedures were performed in the later phase of the study. Thus, greater surgical experience may have influenced the outcomes of the CME group. Of note, the follow-up period of 3 years is too short for proving the superiority of laparoscopic CME over laparoscopic CRR, with regard to oncological outcomes. In addition, while there were no obese patients in the current study (Table 1), laparoscopic CME or CRR may be difficult in such patients (BMI >30 kg/m2).
Conclusion
In summary, the present study suggests that laparoscopic CME compared with laparoscopic CRR yields better lymph node clearance, with less recurrence during a 3-year follow-up, and without increasing the risk of postoperative complications. The results of this study warrant further investigations with prospective randomized trials.
Abbreviations
BMI, body mass index; CME, mesocolic excision complete; CRR, conventional radical resection; FSP, first surgical plane; HVT, high vascular tie; SMA, superior mesenteric artery; SMV, superior mesenteric vein; SSP, second surgical plane; TNM, tumor-node-metastasis; TSP, third surgical plane; LVI, lympho vascular invasion; PNI, perineural invasion.
Acknowledgments
This study was financially supported by the Science and Technology Planning Project of Guangdong Province (No. 2014A020212053, No. 2014A020212636, No. 2016A020215128, No. 2017A030223006), the Medical Scientific Research Fund Project of Guangdong Province (No. B2013374, No. A2014718, No. A2017160), the Science and Technology Planning Project of Guangzhou City (No. 20170402077), the Science and Technology Bureau of Foshan City (No. 201308247), and the 13th Five-Year Key Specialty Project of Foshan City (FSGSPZD135051).
Availability of data and material
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The Institutional Ethics Committee of Shunde Hospital of Southern Medical University approved this study. All participants in this study gave written informed consent. This study is conducted in accordance with the Declaration of Helsinki.
Author contributions
All authors contributed to data analysis, drafting or revising the article, gave final approval of the version to be published, and agree to be accountable for all aspects of the work.
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Hohenberger W, Weber K, Matzel K, Papadopoulos T, Merkel S. Standardized surgery for colonic cancer: complete mesocolic excision and central ligation–technical notes and outcome. Colorectal Dis. 2009;11:354–364. discussion 364–355. doi: 10.1111/j.1463-1318.2008.01735.x. [DOI] [PubMed] [Google Scholar]
- 2.West NP, Hohenberger W, Weber K, Perrakis A, Finan PJ, Quirke P. Complete mesocolic excision with central vascular ligation produces an oncologically superior specimen compared with standard surgery for carcinoma of the colon. J Clin Oncol. 2010;28:272–278. doi: 10.1200/JCO.2009.24.1448 [DOI] [PubMed] [Google Scholar]
- 3.Eiholm S, Ovesen H. Total mesocolic excision versus traditional resection in right-sided colon cancer - method and increased lymph node harvest. Dan Med Bull. 2010;57:A4224. [PubMed] [Google Scholar]
- 4.Nelson H, Sargent DJ, Wieand HS, Fleshman J, Anvari M, Stryker SJ, Beart RW Jr, Hellinger M, Flanagan R Jr, Peters W, Ota D. A comparison of laparoscopically assisted and open colectomy for colon cancer. N Engl J Med. 2004;350:2050–2059. doi: 10.1056/NEJMoa032651. [DOI] [PubMed] [Google Scholar]
- 5.Bertelsen CA, Bols B, Ingeholm P, Jansen JE, Neuenschwander AU, Vilandt J. Can the quality of colonic surgery be improved by standardization of surgical technique with complete mesocolic excision? Colorectal Dis. 2011;13:1123–1129. doi: 10.1111/j.1463-1318.2010.02474.x [DOI] [PubMed] [Google Scholar]
- 6.Storli KE, Sondenaa K, Furnes B, Eide GE. Outcome after introduction of complete mesocolic excision for colon cancer is similar for open and laparoscopic surgical treatments. Dig Surg. 2013;30:317–327. doi: 10.1159/000354580 [DOI] [PubMed] [Google Scholar]
- 7.Gouvas N, Pechlivanides G, Zervakis N, Kafousi M, Xynos E. Complete mesocolic excision in colon cancer surgery: a comparison between open and laparoscopic approach. Colorectal Dis. 2012;14:1357–1364. doi: 10.1111/j.1463-1318.2012.03019.x [DOI] [PubMed] [Google Scholar]
- 8.Feng B, Sun J, Ling TL, et al. Laparoscopic complete mesocolic excision (CME) with medial access for right-hemi colon cancer: feasibility and technical strategies. Surg Endosc. 2012;26:3669–3675. doi: 10.1007/s00464-012-2435-9 [DOI] [PubMed] [Google Scholar]
- 9.Zhu DJ, Chen XW, OuYang MZ, Lu Y. Three surgical planes identified in laparoscopic complete mesocolic excision for right-sided colon cancer. World J Surg Oncol. 2016;14:7. doi: 10.1186/s12957-015-0758-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ohtani H, Tamamori Y, Arimoto Y, Nishiguchi Y, Maeda K, Hirakawa K. A meta-analysis of the short- and long-term results of randomized controlled trials that compared laparoscopy-assisted and open colectomy for colon cancer. J Cancer. 2012;3:49–57. doi: 10.7150/jca.3621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Galizia G, Lieto E, De Vita F, et al. Is complete mesocolic excision with central vascular ligation safe and effective in the surgical treatment of right-sided colon cancers? A prospective study. Int J Colorectal Dis. 2014;29:89–97. doi: 10.1007/s00384-013-1766-x [DOI] [PubMed] [Google Scholar]
- 12.Chen SL, Bilchik AJ. More extensive nodal dissection improves survival for stages I to III of colon cancer: a population-based study. Ann Surg. 2006;244:602–610. doi: 10.1097/01.sla.0000237655.11717.50 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schumacher P, Dineen S, Barnett C Jr., Fleming J, Anthony T. The metastatic lymph node ratio predicts survival in colon cancer. Am J Surg. 2007;194:827–831. discussion 831–822. doi: 10.1016/j.amjsurg.2007.08.030. [DOI] [PubMed] [Google Scholar]
- 14.Pramateftakis MG. Optimizing colonic cancer surgery: high ligation and complete mesocolic excision during right hemicolectomy. Tech Coloproctol. 2010;14(Suppl 1):S49–S51. doi: 10.1007/s10151-010-0609-9 [DOI] [PubMed] [Google Scholar]
- 15.Bertelsen CA, Neuenschwander AU, Jansen JE, et al. Short-term outcomes after complete mesocolic excision compared with ‘conventional’ colonic cancer surgery. Br J Surg. 2016;103:581–589. doi: 10.1002/bjs.10083 [DOI] [PubMed] [Google Scholar]
- 16.Shin JW, Amar AH, Kim SH, et al. Complete mesocolic excision with D3 lymph node dissection in laparoscopic colectomy for stages II and III colon cancer: long-term oncologic outcomes in 168 patients. Tech Coloproctol. 2014;18:795–803. doi: 10.1007/s10151-014-1134-z [DOI] [PubMed] [Google Scholar]
- 17.Bertelsen CA, Neuenschwander AU, Jansen JE, et al. Disease-free survival after complete mesocolic excision compared with conventional colon cancer surgery: a retrospective, population-based study. Lancet Oncol. 2015;16:161–168. doi: 10.1016/S1470-2045(14)71168-4 [DOI] [PubMed] [Google Scholar]
- 18.Kanemitsu Y, Komori K, Kimura K, Kato T. D3 lymph node dissection in right hemicolectomy with a no-touch isolation technique in patients with colon cancer. Dis Colon Rectum. 2013;56:815–824. doi: 10.1097/DCR.0b013e3182919093 [DOI] [PubMed] [Google Scholar]
- 19.Storli KE, Sondenaa K, Furnes B, et al. Short term results of complete (D3) cf. standard (D2) mesenteric excision in colon cancer shows improved outcome of complete mesenteric excision in patients with TNM stages I-II. Tech Coloproctol. 2014;18:557–564. doi: 10.1007/s10151-013-1100-1 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used or analyzed during the current study are available from the corresponding author on reasonable request.