Abstract
Background
Human pancreatic β-cells are heterogeneous. This has been known for a long time and is based on various functional and morphological readouts. β-Cell heterogeneity could reflect fixed subpopulations with distinct functions. However, recent pseudotime analysis of large-scale RNA sequencing data suggest that human β-cell subpopulations may rather reflect dynamic interchangeable states characterized by low expression of genes involved in the unfolded protein response (UPR) and low insulin gene expression, low UPR and high insulin expression or high UPR and low insulin expression.
Scope of review
This review discusses findings obtained by single-cell RNA sequencing combined with pseudotime analysis that human β-cell heterogeneity represents dynamic interchangeable functional states. The physiological significance and potential implications of β-cell heterogeneity in the development and progression of diabetes is highlighted.
Major conclusions
The existence of dynamic functional states allow β-cells to transition between periods of high insulin production and UPR-mediated stress recovery. The recovery state is important since proinsulin is a misfolding-prone protein, making its biosynthesis in the endoplasmic reticulum a stressful event. The transition of β-cells between dynamic states is likely controlled at multiple levels and influenced by the microenvironment within the pancreatic islets. Disturbances in the ability of the β-cells to transition between periods of high insulin biosynthesis and UPR-mediated stress recovery may contribute to diabetes development. Diabetes medications that restore the ability of the β-cells to transition between the functional states should be considered.
Keywords: Human β-cell, Heterogeneity, Diabetes, Pancreatic islet, Insulin
1. Introduction
Insulin plays an essential role for the regulation of blood glucose levels and is secreted from the pancreatic β-cells [1]. It has been known for a long time that β-cells show functional and morphological differences and have been proposed to provide complementarities in their roles [2], [3], [4]. This evidence was mainly derived from studies using rodent islets. In recent years, the increased access to human islets coupled with development of novel single-cell technologies has spurred a renewed interest in β-cell heterogeneity. Despite this renewed interest, it remains to be defined whether β-cell heterogeneity represents subpopulations with fixed or dynamic interchangeable functional states. In this review, we focus on this aspect of human β-cell heterogeneity and discuss its implications for β-cell health and diabetes development.
2. Heterogeneity in human β-cell function and islet architecture
Evidence for functional heterogeneity in the human β-cell population is summarized in Table 1. This has been detected as cell specific patterns of intracellular calcium signaling and insulin secretory responses to glucose stimulation. Interestingly, some β-cells are glucose non-responsive and require additional stimuli to secrete insulin. The sensitivity to these stimuli varies between the β-cells [5]. Even among the β-cells responding to glucose stimulation, it has been shown that a small proportion secretes the majority of the insulin [5]. Heterogeneity has also been observed in the rate of insulin biosynthesis, and the percentage of biosynthetically active β-cells increases with the glucose concentration. Noteworthy, even at high glucose concentrations, a population of β-cells remain biosynthetically inactive [6]. It remains to be determined whether these inactive cells require additional stimuli to produce insulin.
Table 1.
Functional heterogeneity observed in human β-cells.
| Functional Heterogeneity | Observation | References |
|---|---|---|
| Insulin Secretory Response | • Glucose-dependent β-cell recruitment was shown by hemolytic plaque assay | [5] |
| • Insulin secretion of β-β and β-α cell pairs was higher than single β-cells | ||
| • A large proportion of non-glucose responding cells secreted insulin in response to other secretagogues | ||
| • In-situ imaging revealed that GIP and GLP-1 recruit a highly coordinated network of β-cells to augment GSIS | [48] | |
| • β-cells respond to increased glucose concentrations with discrete bursts of insulin secretion | [9] | |
| • β-cells formed distinct regional clusters of synchronized activity | ||
| • Clusters of β-cells within islets display asynchroneous secretory burst patterns | ||
| • The order in which β-cells show exocytotic responses change over time | ||
| • Glucose-dependent increases in insulin biosynthesis in β-cells | [6] | |
| Calcium Oscillations | • The response was reported to be synchronomous throughout the islet and with the oscillatory pattern modulated by increasing glucose concentrations | [49] |
| • Ca2+ signals were observed to be synchronized among β-cells grouped in clusters within the islet, but not to be coordinated throughout the entire β-cell population | [7] | |
| • Differences in the Ca2+ response were found for cells differentially co-expressing INS and NKX6.1 marker genes | [50] | |
| • Ca2+ imaging showed β-cell oscillatory activity not to be coordinated throughout the islet. Oscillations were only found when smaller islet regions were analyzed | [8] | |
| • Computational modeling revealed that β-cell hubs likely dictate the coordinated Ca2+ response in human islets | [51] |
Abbreviations: GIP, glucose-dependent insulinotropic polypeptide; GLP-1, glucagon-like peptide-1; GSIS, glucose stimulated insulin secretion.
An important part of the insulin secretory response is the rise of cytosolic Ca2+ levels after a glucose challenge. The pattern and amplitude of the Ca2+ responses vary among β-cells. Some studies suggest a synchronization among β-cells within small regions of the islets [7], [8]. Interestingly, this pattern of cluster synchronization translates into insulin secretion [9]. These data suggest that neighboring β-cells communicate within the human islet clusters. It is not known if there is communication between the clusters and if a specific subpopulation of β-cells initiates the functional responses, similarly to what has been proposed for mouse β-cells [10].
Heterogeneity in human islet architecture and composition is summarized in Table 2. Although human islets have been a focus of study for many years, there is still no consensus regarding their architectural organization. A recent study by Bonner-Weir and colleagues [11] has shown that the architecture of the human islet is not that different from the mouse counterpart. In particular, small human islets resemble those in mice and are characterized by a β-cell rich core surrounded by a mantle of non-β endocrine cells. This is different from larger human islets, which appear to consist of multiple clusters, and each cluster has a resemblance to that of small islets with a distinct mantle-core arrangement. Thus, the majority of human islets have clear, non-random clustering of β-cells and that the whole islet (small) or sub-islet (large) architecture is not so different between the species (11). This raises important questions about how human islets evolve and function throughout life and how this might relate to the pathogenesis of diabetes.
Table 2.
Heterogeneity observed in the human islet microenvironment.
| Heterogeneity in the islet microenvironment | Observation | References |
|---|---|---|
| Islet Architecture | • β-cells do not appear to cluster in the islet and show common paracrine interactions with other endocrine cells | [8], [52] |
| • 3D analysis showed that β-cells are intercalated between α-cells with a subset contacting blood vessels | [53] | |
| • β-cells cluster in a non-random manner, with small islets resembling mouse islets and larger islets consisting of clusters of cell aggregates with a mantle-core arrangement | [11] | |
| • Innervation was not observed to be widespread in the islet | [54] | |
| • Vascular perfusion was observed to follow a core to mantle direction | [55] | |
| • Blood vessels were reported to penetrate the β-cell core of the islet | [11] | |
| Islet Composition | • The human pancreas can be partitioned in PP-rich and PP-poor lobes | [13] |
| • PP-cells are enriched at the uncinate process and head of the pancreas | [12] | |
| • Variability in cell composition among islets of different pancreatic regions is reported | [8] | |
| • Diabetes influences the islet endocrine cell composition | [56] | |
| • While islet composition and architecture are reported to be similar, the head region was found to be enriched with PP-cells. Preferential loss of β-cells is observed in the head region in patients with T2D | [57] | |
| • Islet endocrine cell composition varies with size, with larger islets containing a lower proportion of β-cells and consisting of multiple subunits of cell aggregates | [11] | |
| • Variability in β-cell mass was found within different regions of the pancreas and among individuals | [14] |
Abbreviations: PP, pancreatic polypeptide.
An additional level of complexity relates to differences in human islet cell composition across the pancreas. Islets enriched in PP-cells are found in the head region, whereas islets enriched in α-cells are found in the neck region of the pancreas [8], [12], [13]. Although less distinct, the number of β-cells per islet seems to increase from the head to the tail region [14]. Several studies have shown that islet cell composition, circulation, innervation and cell-cell interactions can influence the insulin secretory response. We refer the reader to recent reviews for detailed accounts of how the islet architecture influences β-cell heterogeneity and function [15], [16], [17].
3. Rna sequencing identifies human β-cell subpopulations
Recent advances in single-cell RNA sequencing technologies have provided unprecedented insights into the gene expression of human β-cells [18], [19], [20], [21], [22], [23], [24], [25], [26], [27]. Figure 1 exemplifies the workflow for large-scale single human islet cell RNA sequencing and analysis. Human β-cells express at least 18,000 genes [21]. Additional genes are likely to be detected once more sensitive sequencing methods become available. Unbiased clustering of individual transcriptomic profiles revealed four human β-cell subpopulations (Figure 2). Table 3 summarizes the five published single human β-cell RNA sequencing projects of sufficient scale to identify human β-cell heterogeneity. Interestingly, unbiased clustering of the individual transcriptomic profiles across the initial four studies revealed comparable subpopulations [21]. Thus, similar human β-cell subpopulations were identified despite differences in islet isolation, processing and handling, and in the sequencing platforms. Importantly, the results are not influenced by differences in donor age, sex, ethnicity, ischemia time and cause of death. These data suggest that single-cell RNA sequencing is a powerful approach for understanding human β-cell heterogeneity.
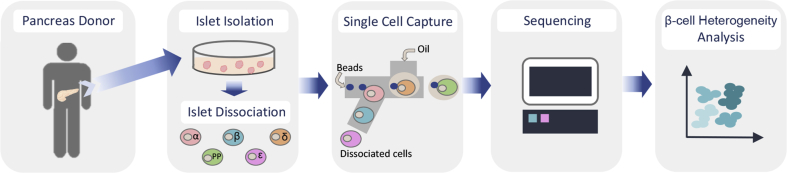
Figure 1.
Schematic depicting the workflow of large-scale single cell RNA sequencing of human pancreatic islet cells. Pancreatic islets were obtained from cadaver organ donors. Islets were cultured and subsequently dissociated into single cells, which were then loaded into a single cell instrument for their individual capture and preparation of RNAseq libraries. Sequencing was performed to the appropriate specifications. Single cell sequencing data were processed (demultiplexing, alignment, filtering and UMI counting). Further analysis to assess cell heterogeneity can be performed using a variety of software packages that provide tools to identify cell clusters, cell-type subpopulations and cluster enriched genes among others.
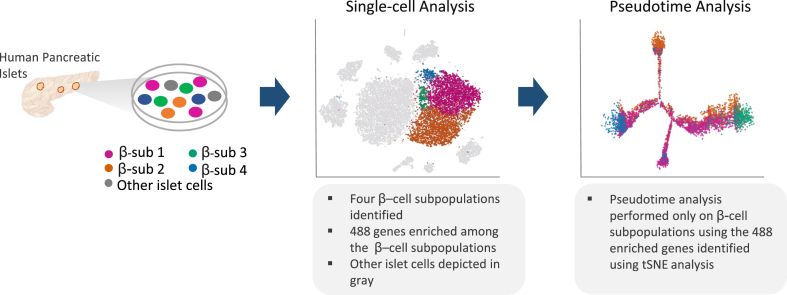
Figure 2.
Workflow of human β-cell heterogeneity analysis. Single cell RNA sequencing data from pancreatic islet cells was used to perform single-cell analysis. Here, four different β-cell subpopulations were found with 488 enriched genes in total (enriched genes in all four subpopulations). Each dot represents a cell, with each color depicting the four distinct subpopulations. Other islet cells are shown in gray for reference. Pseudotime analysis was performed encompassing 6,241 β-cells and using the 488 enriched genes originated in the single-cell analysis. Here, the color of each cell emphasizes the distribution of cells from the four subpopulations observed in the single-cell analysis along the pseudotime trajectory [21], [47]. Adapted from Ref. Xin et al. [21].
Table 3.
Single cell RNA sequencing studies identifying human β-cell heterogeneity.
| Molecular Heterogeneity | |||||
|---|---|---|---|---|---|
| Donors | Islet cell number | β-cell number | Sequencing Platform | Observation | References |
| 12 | 19,174 | 7,361 | 10X Genomics | • Four different β-cell states characterized by differential INS expression and UPR mediated recovery activation | [21] |
| 4 | 8,569 | 2,498 | in-Drop | • Heterogeneity in the β-cell population characterized by UPR activation | [18] |
| 4 | 2,409 | NA | Sort Seq | • β-cell heterogeneity found to be dictated by genes implicated in response to ER and oxidative stress | [20] |
| 10 | 2,209 | 270 | Smart-Seq | • Five sub-clusters of β-cells with differential gene expression | [19] |
| 9 | 28,026 | 9,966 | Drop-Seq | • β-cell heterogeneity characterized by sub-populations that are mostly individual specific | [27] |
Abbreviations: ER, endoplasmic reticulum; NA, not available; UPR, uncoupled protein response.
4. Human β-cells transition between states of activity and stress recovery
The RNA sequencing data discussed above suggest that human β-cell heterogeneity is observed across studies using donors of different backgrounds. The heterogeneity persists over time and is independent of islet isolation, handling and treatment conditions. Heterogeneity of this kind could play an important role for human β-cell biology. Interestingly, the gene expression signatures characterizing the subpopulations relate to insulin biosynthesis, endoplasmic reticulum (ER) stress and unfolded protein response (UPR) activation [21]. While clustering approaches can be a useful tool to explore the existence of cell subpopulations, these can only be observed as static sub-clusters. To better understand the biological significance of the β-cell subpopulations, we performed pseudotime analysis (Figure 2). This analysis projects each cell onto a trajectory, and the ordered sequence of the cells was used to study the changes in gene expression [21]. This provides a higher resolution view of the gene expression landscape and helps understand the biological processes governing β-cell function in each of the subpopulations. Figure 3 shows the principles of pseudotime cell ordering. The main advantage of the pseudotime analysis is that it provides an opportunity to identify and characterize dynamic functional states. No a-priori knowledge of marker genes is needed. However, pseudotime analysis does allow the performance of semi-supervised analysis using a set of relevant marker genes when a particular biological process is of interest. A limitation of the approach is that pseudotime represents a relative measure of progress and not physical time. Also orthogonal methods to confirm accuracy of the pseudotime trajectory are not always available. We identified three major β-cell states characterized by low UPR and low insulin expression, low UPR and high insulin expression or high UPR and low insulin expression. The ordering of the human β-cells according to their pseudotime and the expression levels of insulin and UPR genes for each state is shown in Figure 4A–C. These data suggest that human β-cells undergo periods with high insulin biosynthetic activity and periods with UPR activation to cope with the ER stress. Insulin biosynthesis accounts for >10% of total protein production under basal conditions and increases up to 50% in the stimulated state [28], [29]. Proinsulin is known to be a misfolding-prone protein, with 20% of synthesized proinsulin failing to reach its mature configuration [30], [31], [32], [33]. Once proinsulin is misfolded it can either be refolded or degraded. Under conditions of high insulin demand, misfolded proinsulin can accumulate in the ER and lead to cellular stress. To counteract ER stress, the UPR mechanism is activated enhancing protein folding activity, reducing ER workload and promoting the clearance of misfolded proteins [34]. In this scenario, a proportion of β-cells would be responsible for fulfilling the body's need for insulin, while another proportion would be granted time to recover from the stress of its biosynthetic activity.
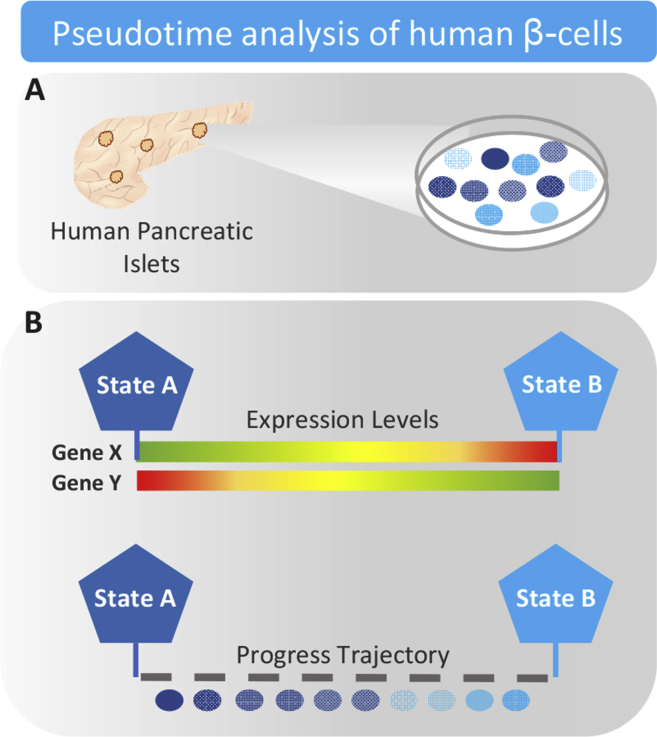
Figure 3.
Schematic illustrating the principles of pseudotime cell ordering. (A) Cells display asynchronous activity within distinct functional states. Purifying cells with similar level of activity at distinct functional stages is difficult or impossible. (B) As cells move between functional states they undergo transcriptional re-configuration (some genes being silenced, others activated). Through machine learning techniques, the sequence of gene expression changes is determined and the trajectory that best fits the data is constructed. Based on the molecular profile of each human β-cell pseudotime analysis infers the order of gene activation events and assigns each cell a value of progress within the trajectory.
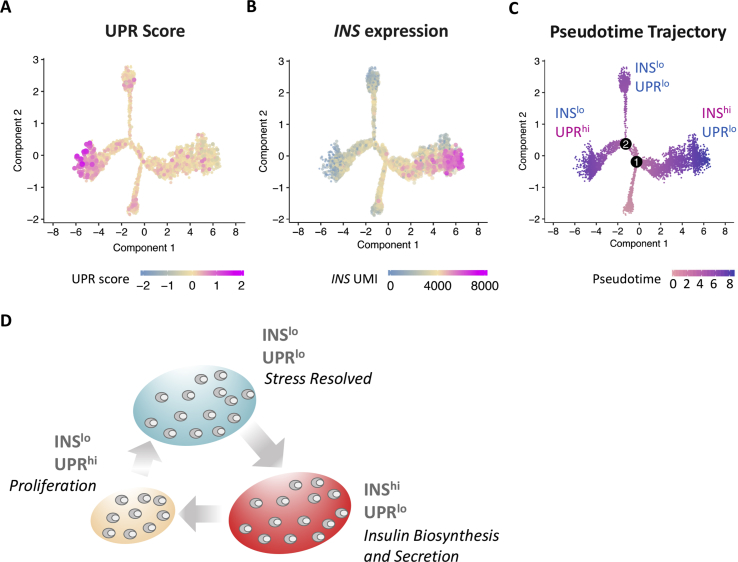
Figure 4.
Pseudotime analysis shows dynamic states of INS expression and stress recovery. (A) An UPR score was calculated using a gene set obtained from IPA Ingenuity. Briefly, the score is the average of scaled UMI of all genes in the gene set. The distribution of the score was calculated by random selection of the genes for the specific gene set with 1,000 iterations. The empirical P value was calculated against the distribution of the score. The score value of each cell was plotted into pseudotime ordering. (B) INS expression pattern is shown in pseudotime ordering. Each dot represents a cell and the color highlights the level of composite score or gene expression. (C) Pseudotime trajectory where each dot represents a cell and the grayscale color highlights the trajectory ranging from 0 to 8.9 [21]. (D) Human β-cells undergoing active insulin biosynthesis and secretion (INShiUPRlo) are likely to become stressed, transitioning to a period of recovery encompassing UPR activation and low INS expression (INSloUPRhi). Following recovery, β-cells transition to a state characterized by low INS expression and reduced UPR activation (INSloUPRlo), where they are nearly ready to become actively secreting again. Among these states, proliferating β-cells were primarily found in the state of low INS expression and high UPR activation. Adapted from Ref. Xin et al. [21].
It is unknown if the proportion of β-cells in the states remains constant during periods of high insulin demand and how many times a β-cell cycles through the states in its lifetime. A study by Szabat and colleagues [35] has shed light on some of the dynamic processes that take place in the β-cells. They used a lentivirus dual reporter for the transcription factor Pdx1 and Ins to track the dynamics of β-cell subpopulations [35]. Two main β-cell populations were identified expressing high insulin (Pdx1+/Ins+) or low insulin (Pdx1+/Inslow). Cells in the Pdx1+/Inslow population showed a continuum of INS expression and it was possible to calculate that it takes 27 h to transition from the Pdx1+/Inslow to the Pdx1+/Ins+ stage. These data suggest that the transit time between β-cell subpopulations could be relatively fast and would occur multiple times over the course of the lifespan of the human β-cell. It is tempting to speculate that a number of factors regulate the transition between the functional states including glucose, insulin, incretins and other hormones and paracrine mechanisms. As we learn more about the β-cell states, it will be interesting to understand their origin and whether their proportions are set during development or in the postnatal period.
5. Only human β-cells reveal a subpopulation of stressed cells
Large-scale RNA sequencing provides data for all islet cell types. It was therefore surprising that a subpopulation of stressed cells was not detected for the other endocrine cell types despite originating from the same donors and processed and analyzed together. A potential explanation could stem from the fact that insulin is prone to misfolding coupled with the high biosynthetic load. β-cells are metabolically active and rely on oxidative phosphorylation for ATP generation [36]. This generates reactive oxygen species and can result in oxidative stress. ER stress and oxidative stress can potentiate each other since protein misfolding results in the production of reactive oxygen species, and these can perturb the ER redox state and cause damage to nascent proteins [37]. Additionally, β-cells have low antioxidant defense increasing their susceptibility to stress [38], [39]. To our knowledge, comparable properties on hormone misfolding and oxidative stress have not been described for the other islet endocrine cell types. Thus, it appears that within the human islet and at the transcriptomic level, the β-cell represents a unique example of heterogeneity to adapt efficiently to environmental challenges and reduce its vulnerability to insults.
6. Heterogeneity identified by marker genes
Being able to identify β-cell subpopulations using enriched marker genes is an attractive and sensitive approach and provides a convenient tool to enrich them for their study. Among the first marker genes reported to be enriched in human β-cell subsets were DKK3 and VMAT2 [40], [41]. Of these, VMAT2 expression was not found in 10% of the β-cells. Unfortunately, the functional significance of these markers and their subpopulations was not identified. In 2016, Dorrell and colleagues [42] identified four subpopulations of human β-cells with differential expression of ST8SIA1 and CD9. These subpopulations were observed to have differential gene expression, as well as distinct basal and stimulated insulin secretory responses. Their potential relevance during disease was addressed, showing the proportion of the subpopulations to be altered in donors with type 2 diabetes. Of note, differences in the expression of previous markers of heterogeneity (DKK3 and VMAT2) were explored, but no differences were found. During the same year, a study using an alternative strategy that relied on the use of previous biologic knowledge was published [43]. This approach employed mass cytometry and 24 marker genes including endocrine hormones, signaling factors, apoptosis and proliferation markers, and previously identified heterogeneity markers [43]. Three subpopulations of human β-cells were identified with KI67 segregating into two of them. A more recent study by Aguayo and colleagues [44] found heterogeneity in the expression of senescence markers (IGF1R and P53BP1). Expression of these marker genes positively correlated with age.
Thus, marker genes have been used to separate human β-cells into subpopulations with distinct properties. Unfortunately, some of the markers were not consistently identified between the studies and were not enriched in the subpopulations identified using unbiased RNA sequencing. This suggest that human β-cell heterogeneity might be defined differently depending on the readouts and sensitivity of the applied technologies. This should be a focus for further investigation.
7. Stressed human β-cells proliferate
Our analysis of heterogeneity revealed that proliferating human β-cells were enriched in the state characterized by low insulin expression and high UPR activation [21]. Both conditions have been reported to influence cell division. Activation of the UPR was shown by Sharma and colleagues [45] to induce human β-cell proliferation. Additionally, the EndoC-BH2 human β-cell line has enhanced functionality when undergoing cell cycle arrest [46]. Thus, it could be speculated that UPR activation acts as an indicator of low insulin expression and provides an opportunity for cell proliferation. Understanding the processes that trigger proliferation during high UPR and low insulin expression could provide important information for the development of novel diabetes therapies with the potential to increase the β-cell mass.
8. Conclusions and perspectives
The study of human β-cell heterogeneity has gained significant attention in recent years, taking advantage of new technologies and increased access to human donor tissue. Cellular heterogeneity has been observed at the architectural, functional and molecular level. While we have acquired valuable information regarding the existence of heterogeneity at different levels, we are still only beginning to understand their significance. Among the questions that remain to be addressed are the number of subpopulations that exist and whether it is finite. It is important to determine the biological information unique to each subpopulation. We also need to understand if heterogeneity arises during pancreatic development or in the postnatal period owing to human β-cell plasticity and changes in islet architecture. Further studies addressing heterogeneity at the translational and post-translational level will contribute to our understanding of these questions. Furthermore, in-situ RNA/protein analysis may allow us to explore the current knowledge of β-cell heterogeneity in the context of the islet environment.
Our analysis of heterogeneity revealed that ER stress from insulin production and UPR-mediated recovery play an important function for the human β-cell (Figure 4D). During disease conditions, we speculate that the proportion of β-cells in each state will shift as the disease progresses. During insulin resistance, chronic high insulin demand will force a large proportion of β-cells to be actively secreting, thus having less time to recover from stress. As these conditions persist, chronic cellular stress will lead to increased susceptibility to apoptosis. Type 2 diabetes will develop when a substantially reduced β-cell population is unable to sustain the necessary balance between activity and rest resulting in diminished insulin secretion.
Although single-cell RNA sequencing provides a snapshot in time, it cannot alone provide definitive evidence for transitions of β-cells between stages. Future experiments will be needed to examine stage transitions, for example, time-course studies with reporter genes. Furthermore, additional experiments addressing the dynamics of the β-cell states during disease conditions will be of importance to understand the effects of current diabetes therapies. Rapidly advancing new technologies coupled with a deeper understanding of the biological significance of β-cell heterogeneity could help in the design of novel therapies promoting the timely transition of the β-cells between periods of activity and rest. This would ensure optimal secretory capacity and human β-cell health and could prevent disease progression.
Author contributions
J.G., Y.X. and G.D.G wrote the manuscript.
Guarantor statement
J.G., Y.X. and G.D.G. are the guarantors of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Prior presentation of information
None.
Funding
The studies were funded by: Exonics Therapeutics, Inc.
This article is part of a supplement entitled ‘Biomarkers of Beta-Cell Health and Dysfunction: Towards Personalised Diabetes Care. Proceedings of the 20th Servier-IGIS Symposium’. The Symposium and the publication of the Supplement have been made possible by an unrestricted educational grant from Institut la Conférence Hippocrate – Servier Group.
Conflict of interest
J.G. and G.D.G. are employees and shareholders of Exonics Therapeutics, Inc.
Y.X. is an employee and shareholder of Regeneron Pharmaceuticals, Inc.
References
- 1.Fu Z., R Gilbert E., Liu D. Regulation of insulin synthesis and secretion and pancreatic beta-cell dysfunction in diabetes. Current Diabetes Reviews. 2013;9:25–53. [PMC free article] [PubMed] [Google Scholar]
- 2.Gutierrez G.D., Gromada J., Sussel L. Heterogeneity of the pancreatic beta cell. Frontiers in Genetics. 2017;8 doi: 10.3389/fgene.2017.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Pipeleers D.G. Heterogeneity in pancreatic beta-cell population. Diabetes. 1992;41:777–781. doi: 10.2337/diab.41.7.777. [DOI] [PubMed] [Google Scholar]
- 4.Nasteska D., Hodson D.J. The role of beta cell heterogeneity in islet function and insulin release. Journal of Molecular Endocrinology. 2018;61:R43–R60. doi: 10.1530/JME-18-0011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wojtusciszyn A., Armanet M., Morel P., Berney T., Bosco D. Insulin secretion from human beta cells is heterogeneous and dependent on cell-to-cell contacts. Diabetologia. 2008;51:1843–1852. doi: 10.1007/s00125-008-1103-z. [DOI] [PubMed] [Google Scholar]
- 6.Ling Z., Pipeleers D.G. Prolonged exposure of human beta cells to elevated glucose levels results in sustained cellular activation leading to a loss of glucose regulation. Journal of Clinical Investigation. 1996;98:2805–2812. doi: 10.1172/JCI119108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Quesada I., Todorova M.G., Alonso-Magdalena P., Beltrá M., Carneiro E.M., Martin F. Glucose induces opposite intracellular Ca2+ concentration oscillatory patterns in identified α- and β-cells within intact human islets of langerhans. Diabetes. 2006;55:2463–2469. doi: 10.2337/db06-0272. [DOI] [PubMed] [Google Scholar]
- 8.Cabrera O., Berman D.M., Kenyon N.S., Ricordi C., Berggren P.-O., Caicedo A. The unique cytoarchitecture of human pancreatic islets has implications for islet cell function. Proceedings of the National Academy of Sciences. 2006;103:2334–2339. doi: 10.1073/pnas.0510790103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Almaça J., Liang T., Gaisano H.Y., Nam H.G., Berggren P.-O., Caicedo A. Spatial and temporal coordination of insulin granule exocytosis in intact human pancreatic islets. Diabetologia. 2015;58:2810–2818. doi: 10.1007/s00125-015-3747-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Johnston N., Mitchell R., Haythorne E., Pessoa M., Semplici F., Ferrer J. Beta cell hubs dictate pancreatic islet responses to glucose. Cell Metabolism. 2016;24:389–401. doi: 10.1016/j.cmet.2016.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bonner-Weir S., Sullivan B.A., Weir G.C. Human islet morphology revisited. Journal of Histochemistry and Cytochemistry. 2015;63:604–612. doi: 10.1369/0022155415570969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Gersell D.J., Gingerich R.L., Greider M.H. Regional distribution and concentration of pancreatic polypeptide in the human and canine pancreas. Diabetes. 1978;28:11–15. [PubMed] [Google Scholar]
- 13.Malaisse-Lagae F., Stefan Y., Cox J., Perrelet A., Orci L. Identification of a lobe in the adult human pancreas rich in pancreatic polypeptide. Diabetologia. 1979;17:361–365. doi: 10.1007/BF01236270. [DOI] [PubMed] [Google Scholar]
- 14.Olehnik S.K., Fowler J.L., Avramovich G., Hara M. Quantitative analysis of intra- and inter-individual variability of human beta-cell mass. Scientific Reports. 2017;7 doi: 10.1038/s41598-017-16300-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Benninger R.K., Hodson D.J. New understanding of β-cell heterogeneity and in situ islet function. Diabetes. 2018;67:537–547. doi: 10.2337/dbi17-0040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Benninger R.K., Piston D.W. Cellular communication and heterogeneity in pancreatic islet insulin secretion dynamics. Trends in Endocrinology and Metabolism. 2014;25:399–406. doi: 10.1016/j.tem.2014.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Roscioni S.S., Migliorini A., Gegg M., Lickert H. Impact of islet architecture on β-cell heterogeneity, plasticity and function. Nature Reviews Endocrinology. 2016;12:695–709. doi: 10.1038/nrendo.2016.147. [DOI] [PubMed] [Google Scholar]
- 18.Baron M., Veres A., Wolock S., Faust A., Gaujoux R., Vetere A. A single-cell transcriptomic map of the human and mouse pancreas reveals inter- and intra-cell population structure. Cell Systems. 2016;3(4):346–360. doi: 10.1016/j.cels.2016.08.011. e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Segerstolpe A., Palasantza A., Eliasson P., Andersson E.-M., Andréasson A.-C., Sun X. Single-cell transcriptome profiling of human pancreatic islets in health and type 2 diabetes. Cell Metabolism. 2016;24:593–607. doi: 10.1016/j.cmet.2016.08.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Muraro M., Dharmadhikari G., Grün D., Groen N., Dielen T., Jansen E. A single-cell transcriptome atlas of the human pancreas. Cell Systems. 2016;3:385–394. doi: 10.1016/j.cels.2016.09.002. e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xin Y., Dominguez Gutierrez G., Okamoto H., Kim J., Lee A.-H., Adler C. Pseudotime ordering of single human β-cells reveals states of insulin production and unfolded protein response. Diabetes. 2018;67:1783–1794. doi: 10.2337/db18-0365. [DOI] [PubMed] [Google Scholar]
- 22.Lawlor N., George J., Bolisetty M., Kursawe R., Sun L., Sivakamasundari V. Single-cell transcriptomes identify human islet cell signatures and reveal cell-type–specific expression changes in type 2 diabetes. Genome Research. 2017;27:208–222. doi: 10.1101/gr.212720.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang Y.J., Schug J., Won K.-J., Liu C., Naji A., Avrahami D. Single-cell transcriptomics of the human endocrine pancreas. Diabetes. 2016;65:3028–3038. doi: 10.2337/db16-0405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Li J., Klughammer J., Farlik M., Penz T., Spittler A., Barbieux C. Single-cell transcriptomes reveal characteristic features of human pancreatic islet cell types. European Molecular Biology Organization Reports. 2016;17:178–187. doi: 10.15252/embr.201540946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Xin Y., Kim J., Okamoto H., Ni M., Wei Y., Adler C. RNA sequencing of single human islet cells reveals type 2 diabetes genes. Cell Metabolism. 2016;24:608–615. doi: 10.1016/j.cmet.2016.08.018. [DOI] [PubMed] [Google Scholar]
- 26.Wang Y.J., Kaestner K.H. Single-cell rna-seq of the pancreatic islets-a promise not yet fulfilled? Cell Metabolism. 2019;29:539–544. doi: 10.1016/j.cmet.2018.11.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Fang Z., Weng C., Li H., Tao R., Mai W., Liu X. Single-cell heterogeneity analysis and CRISPR screen identify key β-cell-specific disease genes. Cell Reports. 2019;26:3132–3144. doi: 10.1016/j.celrep.2019.02.043. e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Scheuner D., Kaufman R.J. The unfolded protein response: a pathway that links insulin demand with β-cell failure and diabetes. Endocrine Reviews. 2008;29:317–333. doi: 10.1210/er.2007-0039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schuit F.C., In't Veld P.A., Pipeleers D.G. Glucose stimulates proinsulin biosynthesis by a dose-dependent recruitment of pancreatic beta cells. Proceedings of the National Academy of Sciences of the United States of America. 1988;85:3865–3869. doi: 10.1073/pnas.85.11.3865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Liu M., Haataja L., Wright J., Wickramasinghe N.P., Hua Q.-X., Phillips N.F. Correction: mutant INS-gene induced diabetes of youth: proinsulin cysteine residues impose dominant-negative inhibition on wild-type proinsulin transport. Public Library of Science one. 2010;5(10) doi: 10.1371/journal.pone.0013333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Liu M., Li Y., Cavener D., Arvan P. Proinsulin disulfide maturation and misfolding in the endoplasmic reticulum. Journal of Biological Chemistry. 2005;280:13209–13212. doi: 10.1074/jbc.C400475200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wang J., Chen Y., Yuan Q., Tang W., Zhang X., Osei K. Control of precursor maturation and disposal is an early regulative mechanism in the normal insulin production of pancreatic β-cells. Public Library of Science one. 2011;6:e19446. doi: 10.1371/journal.pone.0019446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang J., Osei K. Proinsulin maturation disorder is a contributor to the defect of subsequent conversion to insulin in β-cells. Biochemical and Biophysical Research Communications. 2011;411:150–155. doi: 10.1016/j.bbrc.2011.06.119. [DOI] [PubMed] [Google Scholar]
- 34.Fonseca S.G., Gromada J., Urano F. Endoplasmic reticulum stress and pancreatic β-cell death. Trends in Endocrinology and Metabolism. 2011;22(7):266–274. doi: 10.1016/j.tem.2011.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Szabat M., Pourghaderi P., Soukhatcheva G., Verchere C.B., Warnock G.L., Piret J.M. Kinetics and genomic profiling of adult human and mouse β-cell maturation. Islets. 2011;3:175–187. doi: 10.4161/isl.3.4.15881. [DOI] [PubMed] [Google Scholar]
- 36.Schuit F., De Vos A., Farfari S., Moens K., Pipeleers D., Brun T. Metabolic fate of glucose in purified islet cells. Journal of Biological Chemistry. 1997;272:18572–18579. doi: 10.1074/jbc.272.30.18572. [DOI] [PubMed] [Google Scholar]
- 37.Hasnain S.Z., Prins J.B., McGuckin M.A. Oxidative and endoplasmic reticulum stress in β-cell dysfunction in diabetes. Journal of Molecular Endocrinology. 2016;56:R33–R54. doi: 10.1530/JME-15-0232. [DOI] [PubMed] [Google Scholar]
- 38.Lenzen S., Drinkgern J., Tiedge M. Low antioxidant enzyme gene expression in pancreatic islets compared with various other mouse tissues. Free Radical Biology and Medicine. 1996;20:463–466. doi: 10.1016/0891-5849(96)02051-5. [DOI] [PubMed] [Google Scholar]
- 39.Tiedge M., Lortz S., Drinkgern J., Lenzen S. Relation between antioxidant enzyme gene expression and antioxidative defense status of insulin-producing cells. Diabetes. 1997;46:1733–1742. doi: 10.2337/diab.46.11.1733. [DOI] [PubMed] [Google Scholar]
- 40.Hermann M., Untergasser G., Pirkebner D., Draxl A., Berger P., Margreiter R. Dickkopf-3 is expressed in a subset of adult human pancreatic beta-cells. Histochemistry and Cell Biology. 2007;127(5):513–521. doi: 10.1007/s00418-007-0278-6. [DOI] [PubMed] [Google Scholar]
- 41.Saisho Y., Harris P.E., Butler A.E., Galasso R., Gurlo T., Rizza R.A. Relationship between pancreatic vesicular monoamine transporter 2 (VMAT2) and insulin expression in human pancreas. Journal of Molecular Histology. 2008;39:543–551. doi: 10.1007/s10735-008-9195-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dorrell C., Schug J., Canaday P.S., Russ H.A., Tarlow B.D., Grompe M.T. Human islets contain four distinct subtypes of β cells. Nature Communications. 2016;7:11756. doi: 10.1038/ncomms11756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang Y., Golson M., Schug J., Traum D., Liu C., Vivek K. Single-cell mass cytometry analysis of the human endocrine pancreas. Cell Metabolism. 2016;24:616–626. doi: 10.1016/j.cmet.2016.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Aguayo-Mazzucato C., van Haaren M., Mruk M., Lee T.B., Crawford C., Hollister-Lock J. β cell aging markers have heterogeneous distribution and are induced by insulin resistance. Cell Metabolism. 2017;25:898–910. doi: 10.1016/j.cmet.2017.03.015. e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sharma R.B., O'Donnell A.C., Stamateris R.E., Ha B., McCloskey K.M., Reynolds P.R. Insulin demand regulates β cell number via the unfolded protein response. Journal of Clinical Investigation. 2015;125:3831–3846. doi: 10.1172/JCI79264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Scharfmann R., Pechberty S., Hazhouz Y., von Bülow M., Bricout-Neveu E., Grenier-Godard M. Development of a conditionally immortalized human pancreatic β cell line. Journal of Clinical Investigation. 2014;124:2087–2098. doi: 10.1172/JCI72674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Qiu X., Hill A., Packer J., Lin D., Ma Y.-A., Trapnell C. Single-cell mRNA quantification and differential analysis with census. Nature Methods. 2017;14:309–315. doi: 10.1038/nmeth.4150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hodson D.J., Mitchell R.K., Bellomo E.A., Sun G., Vinet L., Meda P. Lipotoxicity disrupts incretin-regulated human β cell connectivity. Journal of Clinical Investigation. 2013;123:4182–4194. doi: 10.1172/JCI68459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Martin F., Soria B. Glucoseminduced [Ca2+]i oscillations in single human pancreatic islets. Cell Calcium. 1996;20:409–414. doi: 10.1016/s0143-4160(96)90003-2. [DOI] [PubMed] [Google Scholar]
- 50.Kenny J.H.R., Melton D.A. Testing pancreatic islet function at the single cell level by calcium influx with associated marker expression. Public Library of Science one. 2015;10 doi: 10.1371/journal.pone.0122044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lei C.-L., Kellard J.A., Hara M., Johnson J.D., Rodriguez B., Briant L.J. Beta-cell hubs maintain Ca2+ oscillations in human and mouse islet simulations. Islets. 2018;10:151–167. doi: 10.1080/19382014.2018.1493316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Brissova M., Fowler M.J., Nicholson W.E., Chu A., Hirshberg B., Harlan D.M. Assessment of human pancreatic islet architecture and composition by laser scanning confocal microscopy. Journal of Histochemistry and Cytochemistry. 2005;53:1087–1097. doi: 10.1369/jhc.5C6684.2005. [DOI] [PubMed] [Google Scholar]
- 53.Bosco D., Armanet M., Morel P., Niclauss N., Sgroi A., Muller Y.D. Unique arrangement of alpha and beta cells in human islets of langerhans. Diabetes. 2010;59:1202–1210. doi: 10.2337/db09-1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Rodriguez-Diaz R., Abdulreda M., Formoso A., Gans I., Ricordi C., Berggren P.-O. Innervation patterns of autonomic axons in the human endocrine pancreas. Cell Metabolism. 2011;14:45–54. doi: 10.1016/j.cmet.2011.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Stagner J.I., Samols E. The vascular order of islet cellular perfusion in the human pancreas. Diabetes. 1992;41:93–97. doi: 10.2337/diab.41.1.93. [DOI] [PubMed] [Google Scholar]
- 56.Kilimnik G., Zhao B., Jo J., Periwal V., Witkowski P., Misawa R. Altered islet composition and disproportionate loss of large islets in patients with type 2 diabetes. Public Library of Science one. 2011;6:e27445. doi: 10.1371/journal.pone.0027445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wang X., Misawa R., Zielinski M.C., Cowen P., Jo J., Periwal V. Regional differences in islet distribution in the human pancreas -preferential beta-cell loss in the head region in patients with type 2 diabetes. Public Library of Science one. 2013;8:e67454. doi: 10.1371/journal.pone.0067454. [DOI] [PMC free article] [PubMed] [Google Scholar]