Abstract
Analysis of liquid–liquid phase separation in biological systems shows that this process is similar to the phase separation observed in aqueous two-phase systems formed by nonionic polymers, proteins, and polysaccharides. The emergence of interfacial tension is a necessary condition of phase separation. The situation in this regard is similar to that of phase separation in mixtures of partially miscible solvents. It is suggested that the evaluation of the effects of biological macromolecules on the solvent properties of aqueous media and the measurement of the interfacial tension as a function of these solvent properties may be more productive for gaining insights into the mechanism of liquid–liquid phase separation than the study of structural details of proteins and RNAs engaged in the process.
Keywords: aqueous two-phase system, interfacial tension, liquid–liquid phase separation, phase-forming polymer, solvent features of water
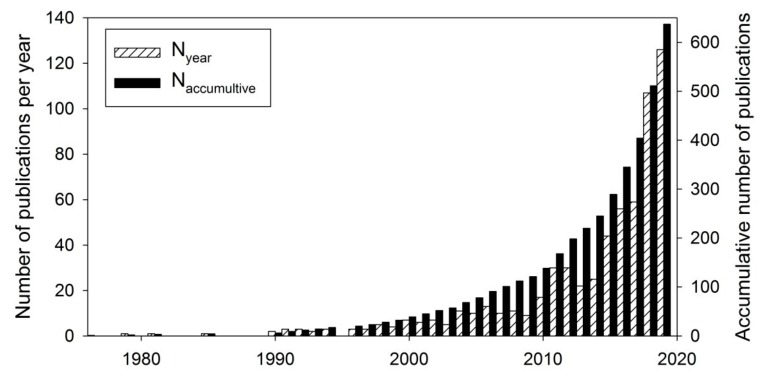
The liquid–liquid phase separation (LLPS) observed in the cytoplasm, nucleoplasm, mitochondrial matrix, and stroma of chloroplasts [1,2,3,4,5,6,7] is currently the research focus of multiple groups. The role of LLPS in the organization of cell biochemistry and its possible role [8] in fast cellular responses to external stimuli have attracted many researchers from different fields. The publications in the field are numerous and exponentially growing (see Figure 1). The majority of these publications are focused on the analysis of macromolecules (proteins and RNA/DNA) enriched in membrane-less organelles and their structural features [7,9,10,11,12,13,14,15,16,17], as well as on the analysis of model systems capable to isolate protein-rich phases under certain conditions in vitro as well as in vivo. Typically, the model systems used in such studies include different phase-separating proteins, such as tau-protein [18], elastin-like polypeptides [19], fused in sarcoma (FUS) [20], transactive response element (TAR) DNA-binding protein of 43 kDa (TDP-43) [21], structural γ-crystallins [22], polyQ-protein Whi3 [23], proteasomal shuttle factor UBQLN2 [24], nucleophosmin (NPM1) [25], Numb/Pon complex from Drosophila neuroblast [26], C9orf72 dipeptide repeat proteins [27], multivalent signaling proteins [28], and many others. There are several reports [29,30] indicating that the solvent properties of membrane-less organelles and protein-rich phases are different from those of the environment from which these phases originate.
Figure 1.
Increase in the number of publications dealing with liquid–liquid phase separation in protein solutions: Number of publications per annum (white crossed bars); accumulative number of publications (black bars). Data are based on the results of a PubMed search on August 19, 2019, using the search terms “liquid-liquid phase separation” and “protein”.
It is well known that different nonionic polymers, proteins, and polysaccharides in their aqueous mixtures may form systems with two or more phases [31,32,33,34,35]. It is also known that the LLPS occurring in such mixtures when the concentrations of phase-forming polymers exceed certain thresholds typically leads to the phase-forming macromolecules being highly concentrated in different phases. If we ignore the solvent, aqueous in both phases, from the physicochemical point of view, the LLPS in such mixtures appears similar to that observed in mixtures of partially miscible solvents, such as water and butanol or methyl ethyl ketone. As an example, aliphatic alcohols such as butanol or pentanol are quite miscible with water until a particular concentration, beyond which the two mutually saturated solvents separate into two phases. The saturation concentration of water in n-butanol is 20.3%wt, and the saturation concentration of butanol in water is 7.4%wt. Any further addition of either solvent leads to an increasing volume of the corresponding phase, while the properties of the phases remain the same.
The situation is different for aqueous mixtures of two phase-forming polymers (including proteins and polysaccharides). The concentrations of the polymers in the two phases continues to change above the threshold concentration of phase separation, resulting in the formation of two-phase systems with properties different from those at lower concentrations of the polymers. Hence, the mixture of the same two phase-forming polymers in water can form a large variety of two-phase systems with properties dependent on the polymers concentrations.
It is important to note that for liquid–liquid phase separation to occur in water, as well as in any solvent mixture, the necessary and sufficient condition is the emergence of interfacial tension. The interfacial tension between organic solvent and water biphasic systems is well known [36] to increase with increasing dissimilarity of the two solvents. However, in any of these organic solvent–water biphasic systems, the value of interfacial tension is not affected by the increase in the organic solvent concentration. On the other hand, in aqueous two-phase systems formed by two polymers, it is well established experimentally [37,38,39,40] that the interfacial tension increases with increasing concentrations of the polymers, i.e., with increasing difference between the polymer concentrations in the two phases.
According to the recently reported [41] model of phase separation in aqueous mixtures of two polymers, the phase diagram may be described in terms of the polymers’ effects on the solvent features of water. The different properties of the coexisting phases in aqueous two-phase systems are successfully described by different solvent properties of water in the phases [42,43,44]. It should be emphasized that the Flory–Huggins theory of the incompatibility of polymers in solution considers the solvent solely as a diluent of unfavorable contacts between polymers and cannot be used to explain the LLPS in aqueous media. This is unsurprising, as the theory was stated [45] as inapplicable to polar systems. Therefore, attempts to use this theory for explanation of LLPS in biological systems seem to be counterproductive.
All the above considerations allow us to hypothesize that the emergence of interfacial tension and resulting LLPS in biological systems may be the consequence of different effects of phase-forming biopolymers (proteins, RNAs) on the properties of water in the cytoplasm, nucleoplasm, mitochondrial matrix, or stroma of chloroplasts. We suggest that studies of interfacial tension might be more productive for gaining deeper insights into the molecular mechanism of LLPS in biology than studies of unarguably important structural details of the macromolecules participating in and/or driving such LLPS.
Acknowledgments
We thank Elizabeth K. Mann for useful discussions and suggestions and for careful reading and editing of this manuscript.
Author Contributions
Conceptualization, B.Y.Z. and V.N.U.; Validation, L.A.F.; Investigation, B.Y.Z., L.A.F., and V.N.U.; Data Curation, L.A.F.; Writing-Original Draft Preparation, B.Y.Z. and V.N.U.; Writing-Review & Editing, L.A.F.; Visualization, V.N.U.; Supervision, B.Y.Z. and V.N.U.; Project Administration, B.Y.Z.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Chong P.A., Forman-Kay J.D. Liquid–liquid phase separation in cellular signaling systems. Curr. Opin. Struct. Boil. 2016;41:180–186. doi: 10.1016/j.sbi.2016.08.001. [DOI] [PubMed] [Google Scholar]
- 2.Shin Y., Brangwynne C.P. Liquid phase condensation in cell physiology and disease. Science. 2017;357 doi: 10.1126/science.aaf4382. [DOI] [PubMed] [Google Scholar]
- 3.Uversky V.N. Intrinsically disordered proteins in overcrowded milieu: Membrane-less organelles, phase separation, and intrinsic disorder. Curr. Opin. Struct. Boil. 2017;44:18–30. doi: 10.1016/j.sbi.2016.10.015. [DOI] [PubMed] [Google Scholar]
- 4.Uversky V.N. Protein intrinsic disorder-based liquid–liquid phase transitions in biological systems: Complex coacervates and membrane-less organelles. Adv. Colloid Interface Sci. 2017;239:97–114. doi: 10.1016/j.cis.2016.05.012. [DOI] [PubMed] [Google Scholar]
- 5.Wheeler R.J., Hyman A.A. Controlling compartmentalization by non-membrane-bound organelles. Philos. Trans. R. Soc. B Boil. Sci. 2018;373:20170193. doi: 10.1098/rstb.2017.0193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Turoverov K.K., Kuznetsova I.M., Fonin A.V., Darling A.L., Zaslavsky B.Y., Uversky V.N. Stochasticity of Biological Soft Matter: Emerging Concepts in Intrinsically Disordered Proteins and Biological Phase Separation. Trends Biochem. Sci. 2019;44:716–728. doi: 10.1016/j.tibs.2019.03.005. [DOI] [PubMed] [Google Scholar]
- 7.Gomes E., Shorter J. The molecular language of membraneless organelles. J. Biol. Chem. 2019;294:7115–7127. doi: 10.1074/jbc.TM118.001192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yoo H., Triandafillou C., Drummond D.A. Cellular sensing by phase separation: Using the process, not just the products. J. Boil. Chem. 2019;294:7151–7159. doi: 10.1074/jbc.TM118.001191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Amaya J., Ryan V.H., Fawzi N.L. The SH3 domain of Fyn kinase interacts with and induces liquid–liquid phase separation of the low-complexity domain of hnRNPA2. J. Boil. Chem. 2018;293:19522–19531. doi: 10.1074/jbc.RA118.005120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bouchard J.J., Otero J.H., Scott D.C., Szulc E., Martin E.W., Sabri N., Granata D., Marzahn M.R., Lindorff-Larsen K., Salvatella X., et al. Cancer Mutations of the Tumor Suppressor SPOP Disrupt the Formation of Active, Phase-Separated Compartments. Mol. Cell. 2018;72:19–36.e8. doi: 10.1016/j.molcel.2018.08.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chong P.A., Vernon R.M., Forman-Kay J.D. RGG/RG Motif Regions in RNA Binding and Phase Separation. J. Mol. Boil. 2018;430:4650–4665. doi: 10.1016/j.jmb.2018.06.014. [DOI] [PubMed] [Google Scholar]
- 12.Posey A.E., Holehouse A.S., Pappu R.V. Phase Separation of Intrinsically Disordered Proteins. Methods Enzym. 2018;611:1–30. doi: 10.1016/bs.mie.2018.09.035. [DOI] [PubMed] [Google Scholar]
- 13.Ruff K.M., Roberts S., Chilkoti A., Pappu R.V. Advances in Understanding Stimulus-Responsive Phase Behavior of Intrinsically Disordered Protein Polymers. J. Mol. Boil. 2018;430:4619–4635. doi: 10.1016/j.jmb.2018.06.031. [DOI] [PubMed] [Google Scholar]
- 14.Wang J., Choi J.-M., Holehouse A.S., Lee H.O., Zhang X., Jahnel M., Maharana S., Lemaitre R., Pozniakovsky A., Drechsel D., et al. A molecular grammar governing the driving forces for phase separation of prion-like RNA binding proteins. Cell. 2018;174:688–699.e16. doi: 10.1016/j.cell.2018.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Boeynaems S., Holehouse A.S., Weinhardt V., Kovacs D., Van Lindt J., Larabell C., Bosch L.V.D., Das R., Tompa P.S., Pappu R.V., et al. Spontaneous driving forces give rise to protein—RNA condensates with coexisting phases and complex material properties. Proc. Natl. Acad. Sci. USA. 2019;116:7889–7898. doi: 10.1073/pnas.1821038116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bracha D., Walls M.T., Wei M.-T., Zhu L., Kurian M., Avalos J.L., Toettcher J.E., Brangwynne C.P. Mapping Local and Global Liquid Phase Behavior in Living Cells Using Photo-Oligomerizable Seeds. Cell. 2019;176:407. doi: 10.1016/j.cell.2018.12.026. [DOI] [PubMed] [Google Scholar]
- 17.Gallego-Iradi M.C., Strunk H., Crown A.M., Davila R., Brown H., Rodriguez-Lebron E., Borchelt D.R. N-terminal sequences in matrin 3 mediate phase separation into droplet-like structures that recruit TDP43 variants lacking RNA binding elements. Lab. Investig. 2019;99:1030–1040. doi: 10.1038/s41374-019-0260-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Majumdar A., Dogra P., Maity S., Mukhopadhyay S. Liquid–Liquid Phase Separation Is Driven by Large-Scale Conformational Unwinding and Fluctuations of Intrinsically Disordered Protein Molecules. J. Phys. Chem. Lett. 2019;10:3929–3936. doi: 10.1021/acs.jpclett.9b01731. [DOI] [PubMed] [Google Scholar]
- 19.Mackay J.A., Callahan D.J., Fitzgerald K.N., Chilkoti A. A quantitative model of the phase behavior of recombinant pH-responsive elastin-like polypeptides. Biomacromolecules. 2010;11:2873–2879. doi: 10.1021/bm100571j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Chen C., Ding X., Akram N., Xue S., Luo S.-Z. Fused in Sarcoma: Properties, Self-Assembly and Correlation with Neurodegenerative Diseases. Molecules. 2019;24:1622. doi: 10.3390/molecules24081622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sun Y., Medina Cruz A., Hadley K.C., Galant N.J., Law R., Vernon R.M., Morris V.K., Robertson J., Chakrabartty A. Physiologically important electrolytes as regulators of tdp-43 aggregation and droplet-phase behavior. Biochemistry. 2019;58:590–607. doi: 10.1021/acs.biochem.8b00842. [DOI] [PubMed] [Google Scholar]
- 22.Bierma J.C., Roskamp K.W., Ledray A.P., Kiss A.J., Cheng C.-H.C., Martin R.W. Controlling Liquid–Liquid Phase Separation of Cold-Adapted Crystallin Proteins from the Antarctic Toothfish. J. Mol. Boil. 2018;430:5151–5168. doi: 10.1016/j.jmb.2018.10.023. [DOI] [PubMed] [Google Scholar]
- 23.Langdon E.M., Qiu Y., Ghanbari Niaki A., McLaughlin G.A., Weidmann C.A., Gerbich T.M., Smith J.A., Crutchley J.M., Termini C.M., Weeks K.M., et al. Mrna structure determines specificity of a polyq-driven phase separation. Science. 2018;360:922–927. doi: 10.1126/science.aar7432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dao T.P., Kolaitis R.-M., Kim H.J., O’Donovan K., Martyniak B., Colicino E., Hehnly H., Taylor J.P., Castañeda C.A. Ubiquitin Modulates Liquid-Liquid Phase Separation of UBQLN2 via Disruption of Multivalent Interactions. Mol. Cell. 2018;69:965–978.e6. doi: 10.1016/j.molcel.2018.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mitrea D.M., Cika J.A., Stanley C.B., Nourse A., Onuchic P.L., Banerjee P.R., Phillips A.H., Park C.-G., Deniz A.A., Kriwacki R.W. Self-interaction of NPM1 modulates multiple mechanisms of liquid–liquid phase separation. Nat. Commun. 2018;9:842. doi: 10.1038/s41467-018-03255-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shan Z., Tu Y., Yang Y., Liu Z., Zeng M., Xu H., Long J., Zhang M., Cai Y., Wen W. Basal condensation of Numb and Pon complex via phase transition during Drosophila neuroblast asymmetric division. Nat. Commun. 2018;9:737. doi: 10.1038/s41467-018-03077-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Boeynaems S., Bogaert E., Kovacs D., Konijnenberg A., Timmerman E., Volkov A., Guharoy M., De Decker M., Jaspers T., Ryan V.H., et al. Phase Separation of C9orf72 Dipeptide Repeats Perturbs Stress Granule Dynamics. Mol. Cell. 2017;65:1044–1055.e5. doi: 10.1016/j.molcel.2017.02.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Li P., Banjade S., Cheng H.-C., Kim S., Chen B., Guo L., Llaguno M., Hollingsworth J.V., King D.S., Banani S.F., et al. Phase Transitions in the Assembly of Multi-Valent Signaling Proteins. Nature. 2012;483:336–340. doi: 10.1038/nature10879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nakashima K.K., Vibhute M.A., Spruijt E. Biomolecular Chemistry in Liquid Phase Separated Compartments. Front. Mol. Biosci. 2019;6:21. doi: 10.3389/fmolb.2019.00021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Feric M., Vaidya N., Harmon T.S., Mitrea D.M., Zhu L., Richardson T.M., Kriwacki R.W., Pappu R.V., Brangwynne C.P. Coexisting liquid phases underlie nucleolar sub-compartments. Cell. 2016;165:1686–1697. doi: 10.1016/j.cell.2016.04.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tolstoguzov V. Phase behaviour of macromolecular components in biological and food systems. Food/Nahrung. 2000;44:299–308. doi: 10.1002/1521-3803(20001001)44:5<299::AID-FOOD299>3.0.CO;2-9. [DOI] [PubMed] [Google Scholar]
- 32.Tolstoguzov V. Compositions and phase diagrams for aqueous systems based on proteins and polysaccharides. Int. Rev. Cytol. 2000;192:3–31. doi: 10.1016/s0074-7696(08)60520-3. [DOI] [PubMed] [Google Scholar]
- 33.Ding P., Wolf B., Frith W., Clark A., Norton I., Pacek A., Frith W. Interfacial Tension in Phase-Separated Gelatin/Dextran Aqueous Mixtures. J. Colloid Interface Sci. 2002;253:367–376. doi: 10.1006/jcis.2002.8572. [DOI] [PubMed] [Google Scholar]
- 34.Zaslavsky B.Y., Ferreira L.A., Darling A.L., Uversky V.N. The solvent side of proteinaceous membrane-less organelles in light of aqueous two-phase systems. Int. J. Boil. Macromol. 2018;117:1224–1251. doi: 10.1016/j.ijbiomac.2018.06.030. [DOI] [PubMed] [Google Scholar]
- 35.Zaslavsky B.Y., Uversky V.N. In Aqua Veritas: The Indispensable yet Mostly Ignored Role of Water in Phase Separation and Membrane-less Organelles. Biochemistry. 2018;57:2437–2451. doi: 10.1021/acs.biochem.7b01215. [DOI] [PubMed] [Google Scholar]
- 36.Boudh-Hir M.-E., Mansoori G. Theory for interfacial tension of partially miscible liquids. Phys. A: Stat. Mech. its Appl. 1991;179:219–231. doi: 10.1016/0378-4371(91)90060-P. [DOI] [Google Scholar]
- 37.Atefi E., Mann J.A., Tavana H. Ultralow Interfacial Tensions of Aqueous Two-Phase Systems Measured Using Drop Shape. Langmuir. 2014;30:9691–9699. doi: 10.1021/la500930x. [DOI] [PubMed] [Google Scholar]
- 38.Bamberger S., Seaman G.V., Sharp K., E Brooks D. The effects of salts on the interfacial tension of aqueous dextran poly(ethylene glycol) phase systems. J. Colloid Interface Sci. 1984;99:194–200. doi: 10.1016/0021-9797(84)90100-0. [DOI] [Google Scholar]
- 39.Forciniti D., Hall C., Kula M. Interfacial tension of polyethyleneglycol-dextran-water systems: influence of temperature and polymer molecular weight. J. Biotechnol. 1990;16:279–296. doi: 10.1016/0168-1656(90)90042-A. [DOI] [Google Scholar]
- 40.Ryden J., Albertsson P.-Å. Interfacial tension of dextran—polyethylene glycol—water two—phase systems. J. Colloid Interface Sci. 1971;37:219–222. doi: 10.1016/0021-9797(71)90283-9. [DOI] [Google Scholar]
- 41.Ferreira L., Uversky V., Zaslavsky B. Modified binodal model describes phase separation in aqueous two-phase systems in terms of the effects of phase-forming components on the solvent features of water. J. Chromatogr. A. 2018;1567:226–232. doi: 10.1016/j.chroma.2018.07.005. [DOI] [PubMed] [Google Scholar]
- 42.Da Silva N.R., Ferreira L.A., Madeira P.P., Teixeira J.A., Uversky V.N., Zaslavsky B.Y. Analysis of partitioning of organic compounds and proteins in aqueous polyethylene glycol-sodium sulfate aqueous two-phase systems in terms of solute–solvent interactions. J. Chromatogr. A. 2015;1415:1–10. doi: 10.1016/j.chroma.2015.08.053. [DOI] [PubMed] [Google Scholar]
- 43.Ferreira L.A., Madeira P.P., Uversky V.N., Zaslavsky B.Y. Analyzing the effects of protecting osmolytes on solute-water interactions by solvatochromic comparison method: I. Small organic compounds. Rsc. Adv. 2015;5:59812–59822. doi: 10.1039/C5RA08610H. [DOI] [Google Scholar]
- 44.Madeira P.P., Bessa A., Teixeira M.A., Álvares-Ribeiro L.M., Aires-Barros M.R., Rodrigues A.E., Zaslavsky B.Y. Study of organic compounds–water interactions by partition in aqueous two-phase systems. J. Chromatogr. A. 2013;1322:97–104. doi: 10.1016/j.chroma.2013.10.085. [DOI] [PubMed] [Google Scholar]
- 45.Tompa H. Polymer Solutions. Butterworth Science Publications; London, UK: 1956. [Google Scholar]