Abstract
Anogeissus leiocarpus (DC.) Guill. & Perr. (Combretaceae) has a long history of use by folk populations for the management of multiple human ailments. Based on the published literature, there has been no attempt to conduct a comparative assessment of the biological activity and the phytochemical profiles of the leaves and stem bark of A. leiocarpus extracted using methanol, ethyl acetate, and water. By high-performance liquid chromatography with electrospray ionization mass spectrometric detection (HPLC-ESI-MSn) analysis, quinic, shikimic, gallic, and protocatechuic acids were tentatively identified from all the extracts, while chlorogenic, caffeic, ferulic, and dodecanedioic acids were only characterised from the leaves extracts. Additionally, a pharmacological study was carried out to evaluate potential protective effects that are induced by the extracts in rat colon and colon cancer HCT116 cell line. In general, the methanol and water extracts of A. leiocarpus leaves and stem bark showed potent radical scavenging and reducing properties. It was noted that the stem bark extracts were more potent antioxidants as compared to the leaves extracts. The methanol extract of A. leiocarpus leaves showed the highest acetyl (4.68 mg galantamine equivalent/g) and butyryl (4.0 mg galantamine equivalent/g) cholinesterase inhibition. Among ethyl acetate extracts, the pharmacological investigation suggested stem bark ethyl acetate extracts to be the most promising. This extract revealed ability to protect rat colon from lipopolysaccharide-induced oxidative stress, without exerting promoting effects on HCT116 cell line viability and migration. As a conclusion, A. leiocarpus represents a potential source of bioactive compounds in the development of novel therapeutic agents.
Keywords: Anogeissus, bioactive compounds, antioxidant, enzyme inhibition, ulcerative colitis
1. Introduction
Anogeissus leiocarpus (DC.) Guill. & Perr. (Combretaceae), also known as chewing stick or axlewood tree, has a long history of traditional use for the management of multiple human ailments. The leaves of A. leiocarpus are used in the treatment of skin diseases, fever, diarrhoea, malaria, and stomach infections [1]. A. leiocarpus is used by the Yoruba people in Nigeria to treat bacterial infections and the roots and twigs of the plant are used as chewing sticks for dental hygiene. Various parts of the plant (roots, leaves, stem bark, and twigs) are used in the management of gonorrhoea, cough, wounds, acute respiratory tract infections, stomach infections, fever, tuberculosis, dysentery, giardiasis, malaria, trypanosomiasis, yellow fever, jaundice, and pathogenic microbial infections [2]. The water extract of A. leiocarpus stem bark was recently found to combat erectile dysfunction in paroxetine-induced sexually impaired male Wistar rats [3]. A spontaneous decrease in serum glucose level in alloxan-induced diabetic rats administered with the aqueous extract of A. leiocarpus leaves [4] was linked to the α-amylase and α-glucosidase inhibitory action of the extract [5]. The aqueous extract of A. leiocarpus trunk bark was reported to exert significant antihypertensive effects in NG-nitro-L-arginine methyl ester (L-NAME)-induced hypertensive rats [6]. The methanolic and ethyl acetate extracts of A. leiocarpus leaves exhibited antioxidant and antibacterial properties [7]. The stem bark methanolic extract of A. leiocarpus demonstrated antitrypanosomal activity against four Trypanosoma strains [8] and leishmanicidal activity [9]. The methylene chloride extract of A. leiocarpus (IC50 value of 3.8 μg/mL) showed in vitro antiplasmodial activity against Plasmodium falciparum, the protozoan parasite that is responsible for malaria in human [10]. Lately, a group of researchers investigated the effect of A. leiocarpus methanolic extract on the liver function in mice that were infected with Plasmodium berghei [11].
From the literature, several studies attempted to investigate the biological activity, mainly, the antibacterial properties, of different extracts of A. leiocarpus. However, as far as our literature search could ascertain, no study was focused on the comparative evaluation of the phytochemical profiles of the methanol, ethyl acetate, and water extracts of the leaves and stem bark of A. leiocarpus. Additionally, in the present study, the authors present the antioxidant and inhibitory action of A. leiocarpus extracts on key enzymes that are related to diabetes type II, Alzheimer’s disease, and skin hyperpigmentation. Finally, while considering the potential antiproliferative effects that are exerted by A. latofolia on colon cancer cells [12], the antiproliferative effects of A. leiocarpus extracts were tested on human colon cancer HCT116 cell line. Additionally, the same extracts were tested for their putative antioxidant/anti-inflammatory effects on isolated rat colon specimens that were exposed to E. coli lipopolysaccharide (LPS), in order to reproduce the burden of oxidative stress and inflammation occurring in ulcerative colitis [13]. To this regard, selected biomarkers of oxidative stress/inflammation, including prostaglandin (PG)E2, 8-iso-PGF2α, and serotonin (5-HT) were selected. It is expected that detailed phytochemical profiles of the different extracts will enable tentative identification of phytochemical/s, which might be responsible for the observed biological activity.
2. Materials and Methods
2.1. Plant Material and Preparation of Extracts
The sampling of the plant species was done in Gontougo region (Sandegue) of Ivory Coast in the year 2018. Botanical authentication of the plant was done by the botanist Dr. Kouadio Bene (Laboratoire de Botanique et Phytothérapie, Université Nangui Abrogoua, Abidjan, Ivory Coast). The leaves and stem barks were dried at room temperature (in shade, about 10 days). These materials were then powdered by using a laboratory mill.
Methanol and ethyl acetate extracts were prepared through maceration techniques (five grams of plant samples were mixed with one hundred ml of each solvents for 24 h). After maceration, the extracts were subjected to filtration and evaporation in vacuo at 40 °C. Traditional infusion was selected to prepare the water extract (five grams of plant samples were infused with one hundred mL of boiling water for 20 min.). After preparation, the water extract was subjected to filtration and freeze drying. Finally, the extracts were stored at 4 °C until phytochemical and pharmacological analysis.
2.2. Profile of Bioactive Compounds
Total phenols, flavonoids, phenolic acids, and flavonols were assayed through spectrophotometric assays [14,15], The extract concentrations of phenolics, flavonoids, phenolic acids, flavonols and tannins, and saponins were determined through spectrophotometric assays [14,15], and were expressed as equivalents of gallic acid (mg GAE/g dry extract), rutin (mg RE/g dry extract), caffeic acid (mg CAE/g dry extract), catechin (mg CE/g dry extract), and quillaja (mg QE/g dry extract), respectively.
The qualitative analysis of A. santonicum extracts (5 mg/mL) was carried out according the protocol that was described by Zengin et al. [16].
An high performance liquid chromatography (HPLC)-fluorimetric analysis was carried out in order to quantify the selected phenolic compounds, in A. santonicum extracts (5 µg/mL). To this regard, an HPLC apparatus (MOD. 1525, Waters Corporation, Milford, MA, USA) coupled to fluorimetric detector (MOD. 2475, Waters Corporation, Milford, MA, USA) and a C18 reversed-phase column (Phenomenex Kinetex, Torrance, CA, USA, 150 mm × 4.6 mm i.d., 2.6 µm) were used. The HPLC gradient conditions were selected, as previously mentioned by Rodrıguez-Delgado and coworkers [17]. In agreement with the same authors, λex = 278 nm and λem = 360 nm were selected in order to analyze the following phenolic compounds: gallic acid, catechin, and epicatechin.
2.3. Determination of Antioxidant and Enzyme Inhibitory Effects
The evaluation of anti-α-amylase, anti-α-glucosidase, anti-cholinesterases, and anti-tyrosinase activities was carried out as previously described by Uysal and coworkers [18]. The enzyme inhibitory results were evaluated in terms of standard equivalents; galatamine for cholinesterase (mg GALAE/g dry extract); kojic acid for tyrosinase (mg KAE/g dry extract), acarbose for amylase, and glucosidase (mmol ACAE/g dry extract). According to the same paper [18], the antiradical activity of the extracts was measured through the use of ferric reducing antioxidant power (FRAP), 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid) (ABTS) cupric reducing antioxidant capacity (CUPRAC), 2,2-diphenyl-1-picrylhydrazyl (DPPH), phosphomolydenum, and metal chelating tests. The antioxidant results were explained as equivalents of trolox (mg TE/g dry extract) and ethylenediaminetetraacetic acid (EDTA) (in metal chelating assay) (mg EDTAE/g dry extract). One-way ANOVA, followed by Tukey’s post hoc test, were applied for comparing the samples in terms of bioactive compounds content and biological activities. The MCA (multiple correspondence analysis) and Clustering Image Map were performed for the discrimination between the samples based on their chemical compositions and Venn graph was built to identify the chemical profile differences among those samples. Before, the data of the chemical composition were attributed to classes with two modalities (e.g., + for presence and – for the absence of compounds in extracts). Afterwards, Multiple datasets supervised analysis, namely DIABLO, was achieved to find out the key factor (parts and solvents) that is responsible for variation in datasets. Subsequently, the correlation between the bioactive compounds and biological activities were estimated. All of the statistical tests were conducted by using R 3.5.1 software environment.
2.4. Pharmacological Assays
2.4.1. Allelopathy Assay
As previously described [19], the allelopathy bioassay was carried out in 90 mm diameter Petri dishes, which represented the substrate for the germination of seeds, whereas A. leiocarpus extracts (0.1–10 mg/mL) were dissolved in imbibition water. During the incubation period (three days at 4 °C), seeds were monitored in order to evaluate their uniform size and integrity. To this regard, lettuce could be considered as one of the most suitable dicotyledon for allelopathy assay. This is due to both fast germination rate and high sensitivity. After the third day of treatment, a root length ≥ 1 mm was the condition to consider positive the germination of seeds [19]. The experiments were carried out in triplicate and means ± SEM were determined through the use of GraphPad Prism software (version 5.01).
2.4.2. Brine Shrimp Lethality Assay
Artemia salina lethality bioassay was performed, as previously reported [13]. The larvae of brine shrimps were exposed to the extracts (0.01–10 mg/mL) at 25–28 °C for 24 h. At the end of the incubation period, brine shrimp lethality was evaluated with the equation ((T – S)/T) × 100, being T and S the total number of larvae that were exposed to extracts and living nauplii, respectively. The experiments were carried out in triplicate.
2.5. In Vitro Studies
HCT116 cell line (ATCC® CCL-247™) culture and differentiation were carried out as previously described in our published paper [13]. To evaluate the biocompatibility of A. leiocarpus extracts (0.1 mg/mL), the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) viability test was carried out, as recently described [13]. The effects of extracts (0.1 mg/mL) on HCT116 cell viability was evaluated in comparison to the untreated control group 24 h after treatment. Finally, the effects of extracts on HCT116 cell spontaneous migration, through the use of wound healing test, as recently reported. Briefly, the cells were challenged with A. leiocarpus extracts (0.1 mg/mL), and spontaneous migration was monitored at different time points (0 and 24 h). The Image-J software (NIH) was used to quantify the scratch area, whereas GraphPad software was employed to calculate mean data at 0 and 24 h and express them as percentage variation with reference to relative 100% of that at 0 h.
2.6. Ex Vivo Studies
Male adult Sprague-Dawley rats (200–250 g) were sacrificed by CO2 inhalation, and colon specimens were immediately stimulated with Escherichia coli lipopolysaccharide (LPS) 10 µg/mL for 4 h (incubation period), as recently described [13]. Italian Health Minstry (authorization N. F4738.N.XTQ, delivered on 11th Novembre 2018) approved the experimental procedures.
During the incubation period, the colon specimens were also treated with A. leiocarpus extracts (0.1 mg/mL). Subsequently, extraction and chromatographic quantification of 5-HT (ng/mg wet tissue) was carried out in colon homogenate, as previously reported [20,21]. Additionally, colon homogenate was assayed for measuring PGE2 and 8-iso-PGF2α via radioimmunoassay [22,23].
2.7. Statistical Analysis
Data were means ± SEM and analyzed by one-way analysis of variance (ANOVA), followed by Newman-Keuls post hoc test (GraphPad Prism version 5.01 for Windows, GraphPad Software, San Diego, CA, USA). The data were considered to be significant for p values less than 0.05. With the aim to apply 3Rs (Reduction, Refinement and Reduction) approach to the ex vivo procedures, the number of animals was determined through the “Resource Equation” N = (E + T)/T (10 ≤ E ≤ 20) [24]), where N, T, and E represent the number of animals, pharmacological treatments, and degree freedom in ANOVA, respectively.
3. Results
3.1. Phytochemical Profile
Table 1 presents phytochemical evaluations of the different extracts of A. leiocarpus leaves and stem bark. Quantitative determination showed that the stem barks extracts of A. leiocarpus (water extract = methanol extract > ethyl acetate extract) possessed significant amounts of phenolics when compared to their respective leaves extracts. The water extract of A. leiocarpus leaves showed the highest flavonoid (89.0 mg RE/g) and phenolic acid (14 mg CAE/g) contents. Highest tannin (77.0 mg CE/g), flavanol (79 mg CE/g), and saponin (438 mg QE/g) contents were recorded from the methanol extract of A. leiocarpus leaves. Phenolic acids, such as, protocatechuic acid, chlorogenic acid, caffeic acid, and ferulic acid, were previously reported to be soluble in polar protic solvents, like methanol, except gallic acid, which was readily soluble in water [25]. From Table 1, it is noted that no phenolic acid was recorded in the ethyl acetate extracts of A. leiocarpus leaves and stem bark. Caffeic acid, a phenolic acid, was found to be minimally soluble in ethyl acetate [26].
Table 1.
Quantitative phytochemical determinations of A. leiocarpus leaves and stem bark extracts.
| Samples | Total Phenolic Content (mg GAE/g) |
Total Flavonoid Content (mg RE/g) |
Total Flavonol (mg CE/g) |
Total Phenolic acid (mg CAE/g) |
Total Tannin Content (mg CE/g) |
Total Saponin Content (mgQE/g) |
|---|---|---|---|---|---|---|
| Leaves-EA | 49 ± 1e | 35.0 ± 0.6c | 6.0 ± 0.1d | nd | 6.0 ± 0.4e | 190 ± 17bc |
| Leaves-MeOH | 223 ± 2c | 54.0 ± 0.6b | 79 ± 3a | 8 ± 1b | 77.0 ± 0.7a | 438 ± 54a |
| Leaves-Water | 257 ± 3b | 89.0 ± 0.2a | 3.46 ± 0.02e | 14 ± 1a | 18 ± 4c | 200 ± 30bc |
| Stem barks-EA | 207 ± 2d | 16 ± 0.3f | 14.0 ± 0.3c | nd | 17 ± 0.5c | 171 ± 24bc |
| Stem barks-MeOH | 271 ± 1a | 27.0 ± 0.3e | 28.0 ± 0.4b | nd | 33 ± 1b | 230 ± 34b |
| Stem barks-Water | 274 ± 2a | 33.0 ± 0.2d | 2.09 ± 0.01e | 7.0 ± 0.6c | 10.0 ± 0.1d | 163 ± 28c |
Values expressed are means ± SD of three parallel measurements. GAE: Gallic acid equivalent; RE: Rutin equivalent. CE: Catechin equivalent; CAE: Caffeic acid equivalent; QE: Quillaja equivalent. nd: not detected. Superscripts in the same column indicate significant difference in the tested extracts (p < 0.05).
HPLC-fluorimeter analysis that was performed on selected phenolic compounds (i.e., gallic acid, catechin and epicatechin), revealed that leaf extract could have, overall, a higher content of these metabolites than stem bark extract (Table 2). This is consistent with the observed content of the total phenolic acids and flavanols.
Table 2.
Phenol content of A. leiocarpus leaves and stem bark ethyl acetate (EA), methanol (MeOH) and water extracts.
| Phenolic Compound | Leaves-EA mg/g | Leaves-MeOH mg/g | Leaves-Water Extract mg/g | Stem Barks-EA mg/g | Stem Barks MeOH mg/g | Stem Barks-Water mg/g |
|---|---|---|---|---|---|---|
| Gallic acid | 226 ± 21 | 89 ± 28 | 30 ± 3 | 65 ± 6 | 38 ± 2 | 37 ± 1 |
| Catechin | 7.0 ± 0.8 | 3.0 ± 0.2 | 9 ± 1 | 1.0 ± 0.1 | 0.29 ± 0.02 | 0.28 ± 0.02 |
| Epicatechin | 0.29 ± 0.02 | 2.0 ± 0.1 | 0.27 ± 0.02 | 0.27 ± 0.02 | 0.18 ± 0.01 | 1.0 ± 0.1 |
Detailed analysis of the chemical composition of the ethyl acetate, methanol, and water extracts of A. leiocarpus leaves and stem bark by HPLC-ESI-MSn, while using both positive and negative ionisation modes, was also conducted. The detailed results are given as supplemental materials (Tables S1–S6). The results are also summarized in Table 3.
Table 3.
Chemical composition of A. leiocarpus extracts.
| No. | Name | Formula | [M + H]+ | [M – H]– | Leaves-EA | Leaves-MeOH | Leaves-Water | Stem Bark-EA | Stem Bark-MeOH | Stem Bark-Water |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Quinic acid | C7H12O6 | 19,105,557 | + | + | + | + | + | + | |
| 2 | Hexahydroxydiphenoylhexose | C20H18O14 | 48,106,184 | – | – | – | + | + | + | |
| 3 | Shikimic acid | C7H10O5 | 17,304,500 | + | + | + | + | + | + | |
| 4 | Galloylquinic acid isomer 1 | C14H16O10 | 34,306,653 | – | – | – | – | + | + | |
| 5 | Galloylhexose isomer 1 | C13H16O10 | 33,106,653 | – | – | – | + | + | + | |
| 6 | Galloylhexose isomer 2 | C13H16O10 | 33,106,653 | – | – | – | + | + | + | |
| 71 | Gallic acid (3,4,5-Trihydroxybenzoic acid) | C7H6O5 | 16,901,370 | + | + | + | + | + | + | |
| 8 | Galloylhexose isomer 3 | C13H16O10 | 33,106,653 | – | – | – | + | + | + | |
| 9 | Galloylquinic acid isomer 2 | C14H16O10 | 34,306,653 | – | – | – | – | + | + | |
| 10 | Galloylquinic acid isomer 3 | C14H16O10 | 34,306,653 | – | – | – | – | + | + | |
| 11 | Gallocatechin | C15H14O7 | 30,506,613 | – | + | + | + | + | + | |
| 12 | Protocatechuic acid (3,4-Dihydroxybenzoic acid) | C7H6O4 | 15,301,879 | + | + | + | + | + | + | |
| 13 | 3-Hydroxybenzaldehyde | C7H6O2 | 12,304,461 | + | + | + | – | – | – | |
| 14 | Procyanidin B isomer 1 | C30H26O12 | 57,713,460 | – | – | – | + | + | + | |
| 15 | Punicalagin | C48H28O30 | 108,305,872 | + | + | + | + | + | + | |
| 16 | Kynurenic acid | C10H7NO3 | 19,005,042 | – | + | + | – | – | – | |
| 17 | Procyanidin B isomer 2 | C30H26O12 | 57,713,460 | – | – | – | + | + | + | |
| 181 | Catechin | C15H14O6 | 28,907,121 | + | + | + | + | + | + | |
| 191 | Epigallocatechin | C15H14O7 | 30,506,613 | – | + | + | + | + | + | |
| 20 | Casuarinin | C41H28O26 | 93,507,906 | + | + | + | + | + | + | |
| 21 | Chlorogenic acid (3-O-Caffeoylquinic acid) | C16H18O9 | 35,510,291 | + | + | + | – | – | – | |
| 22 | Caffeic acid | C9H8O4 | 17,903,444 | + | + | + | + | + | + | |
| 23 | Cornusiin B or isomer | C48H30O30 | 108,507,437 | + | + | + | + | + | – | |
| 24 | Ampelopsin (Dihydromyricetin) | C15H12O8 | 31,904,540 | + | + | + | + | + | + | |
| 25 | Tellimagrandin I or isomer | C34H26O22 | 78,508,375 | + | + | + | + | + | + | |
| 26 | Coumaroylquinic acid | C16H18O8 | 33,709,235 | + | + | + | – | – | – | |
| 27 | Corilagin or isomer | C27H22O18 | 63,307,279 | + | + | + | – | – | – | |
| 28 | Caffeoylshikimic acid | C16H16O8 | 33,507,670 | – | + | + | – | – | – | |
| 29 | Procyanidin B isomer 3 | C30H26O12 | 57,713,460 | – | – | – | + | + | – | |
| 30 | Digalloylhexose | C20H20O14 | 48,307,749 | – | – | – | + | + | + | |
| 311 | Epigallocatechin-3-O-gallate (Teatannin II) | C22H18O11 | 45,707,709 | – | + | + | + | + | + | |
| 321 | Epicatechin | C15H14O6 | 28,907,121 | – | + | + | + | + | + | |
| 33 | Punicacortein C or D | C48H28O30 | 108,305,872 | – | – | – | + | + | + | |
| 34 | Trigalloylhexose isomer 1 | C27H24O18 | 63,508,844 | – | – | – | + | + | + | |
| 35 | Trigalloylhexose isomer 2 | C27H24O18 | 63,508,844 | – | – | – | + | + | + | |
| 36 | Di-O-methylcoruleoellagic acid | C16H10O10 | 36,101,958 | – | – | – | + | – | – | |
| 37 | Mangiferin | C19H18O11 | 42,107,709 | – | – | – | + | + | + | |
| 38 | Trigalloylhexose isomer 3 | C27H24O18 | 63,508,844 | – | – | – | + | + | + | |
| 391 | Taxifolin (Dihydroquercetin) | C15H12O7 | 30,305,048 | + | + | + | + | + | + | |
| 40 | Ferulic acid | C10H10O4 | 19,305,009 | + | + | + | – | – | – | |
| 41 | Tetragalloylhexose | C34H28O22 | 78,709,940 | – | – | – | + | + | – | |
| 421 | Epicatechin-3-O-gallate | C22H18O10 | 44,108,218 | + | + | + | + | + | + | |
| 43 | Chebulagic acid | C41H30O27 | 95,308,963 | + | + | + | – | – | – | |
| 44 | Ellagic acid O-glucuronide | C20H14O14 | 47,703,054 | – | – | – | + | + | + | |
| 45 | Ellagic acid O-hexoside isomer 1 | C20H16O13 | 46,305,127 | + | + | + | + | + | + | |
| 46 | Ellagic acid O-hexoside isomer 2 | C20H16O13 | 46,305,127 | + | + | + | + | + | + | |
| 47 | Coumaroylshikimic acid | C16H16O7 | 31,908,178 | + | + | + | – | – | – | |
| 48 | O–Methylellagic acid O-hexoside isomer 1 | C21H18O13 | 47,706,692 | – | – | – | + | + | + | |
| 49 | Ellagic acid C-hexoside isomer 1 | C20H16O13 | 46,506,692 | – | – | – | + | + | + | |
| 50 | Myricetin-O-hexoside | C21H20O13 | 47,908,257 | + | + | + | + | + | + | |
| 51 | Quercetin-O-galloylhexoside | C28H24O16 | 61,509,862 | + | + | + | – | – | – | |
| 52 | Vitexin (Apigenin-8-C-glucoside) | C21H20O10 | 43,311,348 | – | – | – | + | + | + | |
| 53 | Pentagalloylhexose | C41H32O26 | 93,911,036 | – | – | – | + | + | – | |
| 54 | Di-O-methylflavellagic acid O-hexoside | C22H20O14 | 50,707,749 | – | – | – | + | + | + | |
| 55 | Theaflavin or isomer | C29H24O12 | 56,513,461 | – | + | – | – | + | – | |
| 56 | O-Methylellagic acid O-hexoside isomer 2 | C21H18O13 | 47,706,692 | – | – | – | + | + | + | |
| 57 | Aromadendrin (Dihydrokaempferol) | C15H12O6 | 28,705,557 | + | + | + | + | + | – | |
| 58 | 3,3′-Di-O-methylellagic acid-4-O-glucoside | C22H20O13 | 49,108,257 | + | + | + | + | + | + | |
| 59 | Quercetin-3-O-glucuronide | C21H18O13 | 47,706,692 | + | + | + | – | – | – | |
| 60 | Isoquercitrin (Hirsutrin, Quercetin-3-O-glucoside) | C21H20O12 | 46,308,765 | + | + | + | – | – | – | |
| 61 | Rutin (Quercetin-3-O-rutinoside) | C27H30O16 | 61,116,122 | + | + | + | – | – | – | |
| 62 | Luteolin-7-O-glucoside (Cynaroside) | C21H20O11 | 44,709,274 | – | – | – | – | – | + | |
| 63 | Luteolin-O-deoxyhexosylhexoside | C27H30O15 | 59,315,065 | – | – | – | – | – | + | |
| 64 | Isovitexin (Apigenin-6-C-glucoside) | C21H20O10 | 43,311,348 | – | – | – | + | + | + | |
| 65 | Coatline A isomer | C21H24O10 | 43,512,913 | – | + | + | + | + | – | |
| 66 | Ellagic acid O-pentoside | C19H14O12 | 43,304,071 | – | – | – | + | + | + | |
| 67 | Ellagic acid C-hexoside isomer 2 | C20H16O13 | 46,506,692 | – | – | – | + | + | + | |
| 68 | Eschweilenol C (Ellagic acid-4-O-rhamnoside) | C20H16O12 | 44,705,636 | + | + | + | + | + | + | |
| 69 | Reinutrin (Quercetin-3-O-xyloside) | C20H18O11 | 43,307,709 | + | + | + | – | – | – | |
| 70 | Ellagic acid | C14H6O8 | 30,099,845 | + | + | + | + | + | + | |
| 71 | Avicularin (Quercetin-3-O-arabinoside) | C20H18O11 | 43,307,709 | + | + | + | – | – | – | |
| 721 | Myricetin (3,3′,4′,5,5′,7-Hexahydroxyflavone) | C15H10O8 | 31,702,974 | + | + | + | + | + | + | |
| 73 | Guaijaverin (Quercetin-3-O-arabinoside) | C20H18O11 | 43,307,709 | + | + | + | – | – | – | |
| 74 | Isorhamnetin-O-glucuronide isomer 1 | C22H20O13 | 49,108,257 | – | + | – | – | – | – | |
| 75 | Quercitrin (Quercetin-3-O-rhamnoside) | C21H20O11 | 44,709,274 | + | + | + | – | – | – | |
| 76 | Di-O-methylflavellagic acid O-pentoside | C21H18O13 | 47,706,692 | – | – | – | + | – | – | |
| 77 | Ducheside A (3-O-Methylellagic acid-4′-O-xyloside) | C20H16O12 | 44,705,636 | – | – | – | + | + | + | |
| 78 | Eriodictyol | C15H12O6 | 28,705,557 | + | + | – | + | + | + | |
| 79 | Isorhamnetin-3-O-glucoside | C22H22O12 | 47,710,330 | + | + | + | – | – | – | |
| 80 | Dimethoxy-tetrahydroxy(iso)flavone-O-hexoside | C23H24O13 | 50,711,387 | – | + | + | – | – | – | |
| 81 | Isorhamnetin-O-glucuronide isomer 2 | C22H20O13 | 49,108,257 | – | + | – | – | – | – | |
| 82 | Isorhamnetin-O-glucuronide | C22H20O13 | 49,108,257 | – | – | + | – | – | – | |
| 83 | Isorhamnetin-3-O-rutinoside (Narcissin) | C28H32O16 | 62,316,122 | + | + | + | – | – | – | |
| 84 | 4-Methoxycinnamic acid | C10H10O3 | 17,907,082 | – | – | – | – | + | – | |
| 85 | Di-O-methylellagic acid-O-pentoside | C21H18O12 | 46,107,200 | + | + | + | + | + | + | |
| 86 | 3,3′,4-Tri-O-methylflavellagic acid-4-O-glucoside | C23H22O14 | 52,109,314 | + | + | + | + | + | + | |
| 87 | Quercetin | C15H10O7 | 30,103,483 | + | + | + | – | – | – | |
| 88 | 3-O-Methylellagic acid | C15H8O8 | 31,501,410 | – | + | + | + | + | + | |
| 89 | Di-O-methylellagic acid-O-deoxyhexoside | C22H20O12 | 47,508,766 | – | – | – | + | + | + | |
| 901 | Naringenin | C15H12O5 | 27,106,065 | + | + | + | + | + | + | |
| 91 | Di-O-methylflavellagic acid isomer 1 | C16H10O9 | 34,502,466 | – | – | – | + | + | + | |
| 921 | Luteolin (3′,4′,5,7-Tetrahydroxyflavone) | C15H10O6 | 28,503,991 | + | + | + | – | + | – | |
| 93 | 3,3′-Di-O-methylellagic acid | C16H10O8 | 32,902,975 | + | + | + | + | + | + | |
| 94 | Kaempferol (3,4′,5,7-Tetrahydroxyflavone) | C15H10O6 | 28,705,556 | + | + | + | – | – | – | |
| 95 | Isorhamnetin (3′-Methoxy-3,4′,5,7-tetrahydroxyflavone) | C16H12O7 | 31,505,048 | + | + | + | – | – | – | |
| 96 | Dimethoxy-trihydroxy(iso)flavone | C17H14O7 | 32,906,613 | + | + | – | – | – | – | |
| 97 | Di-O-methylflavellagic acid isomer 2 | C16H10O9 | 34,502,466 | – | – | – | + | + | + | |
| 98 | Tetra-O-methylflavellagic acid isomer 1 | C18H14O9 | 37,305,596 | – | – | – | + | + | + | |
| 99 | Apigenin (4′,5,7-Trihydroxyflavone) | C15H10O5 | 26,904,500 | – | + | – | – | – | + | |
| 100 | 3,3′,4-Tri-O-methylellagic acid | C17H12O8 | 34,304,540 | + | + | + | + | + | + | |
| 101 | Undecanedioic acid | C11H20O4 | 21,512,834 | + | + | + | – | – | – | |
| 102 | Tetra-O-methylflavellagic acid isomer 2 | C18H14O9 | 37,305,596 | – | – | – | + | + | + | |
| 103 | 3,3′,4-Tri-O-methylflavellagic acid | C17H12O9 | 35,904,031 | + | + | + | + | + | + | |
| 104 | Dihydroxy-dimethoxy(iso)flavone | C17H14O6 | 31,307,122 | + | + | – | – | – | – | |
| 105 | Pinocembrin (5,7-Dihydroxyflavanone) | C15H12O4 | 25,506,573 | + | + | – | – | + | – | |
| 106 | Dihydroxy-trimethoxy(iso)flavone | C18H16O7 | 34,308,178 | + | + | + | – | – | – | |
| 107 | Dodecanedioic acid | C12H22O4 | 22,914,399 | + | + | + | – | – | – | |
| 108 | Hexadecanedioic acid | C16H30O4 | 28,520,659 | + | + | – | – | – | – |
1 Confirmed by standard.
3.2. Phenolic Acids
Quinic ([M – H]– at m/z 191), shikimic ([M – H]– at m/z 173), gallic ([M – H]– at m/z 169), and protocatechuic ([M – H]– at m/z 153) acids were tentatively identified from all the extracts of A. leiocarpus leaves and stem bark. Chlorogenic ([M – H]+ at m/z 355), caffeic ([M – H]– at m/z 179), ferulic ([M – H]– at m/z 193), and dodecanedioic ([M – H]– at m/z 229) acids were tentatively identified from the leaves extracts of A. leiocarpus only.
3.3. Flavonoids
Several compounds belonging to the flavonoid family were identified from A. leiocarpus extracts. The compound suffering deprotonation at m/z 269 [M – H]– and fragment ions at m/z 225, 151, 149, and 117 was characterised as apigenin and was present in the methanol extract of A. leiocarpus leaves and water extract of A. leiocarpus stem bark. C-glucosides of apigenin ([M – H]+ at m/z 433), namely, vitexin and isovitexin, only occurred in the stem bark extracts of A. leiocarpus. Luteolin, suffering deprotonation at m/z 285 [M – H]– and fragment ions at m/z 217, 199, 175, 151, and 133 was tentatively characterised. Other flavonoids, such as, catechin ([M – H]– at m/z 289), naringenin ([M – H]– at m/z 271), myricetin ([M – H]– at m/z 317), taxifolin ([M – H]– at m/z 303), and pinocembrin ([M – H]– at m/z 255) were also identified.
3.4. Tannins Derivatives
Some tannin derivatives were tentatively characterised. Casuarinin with deprotonation at m/z 935 [M – H]– and fragment ions at m/z 917, 783, 633, 300, and 275, was tentatively characterized in all the extracts. Hydrolysable tannins, namely chebulagic acid and punicalagin [27], were characterized at [M – H]– m/z 953 and 1083, respectively. While punicalagin was identified in all of the extracts, chebulagic acid was only characterised in the leaves extracts.
3.5. Antioxidant Activities
Phytochemicals, which are ubiquitously present in plants, have been identified to possess antioxidant activity and they are capable of managing oxidative stress related diseases [28]. In this study, three types of antioxidant mechanisms were used, namely, radical scavenging, reducing power, and metal chelating. Table 4 presents the ability of A. leiocarpus extracts to scavenge DPPH and ABTS radicals. DPPH, which is a stable radical, is widely used to assess the free radical scavenging abilities of plant extract. By proton transfer, there is the DPPH change in the non-radical form, characterized by a yellow chromophore [29]. On the other side, the ABTS method is based on the monitoring of electron or hydrogen transfer-induced ABTS radical-cation decay, which is characterized by the disappearance of the corresponding blue-green radical [30]. From Table 4, it is observed that, in general, the methanol and water extracts of A. leiocarpus leaves and stem bark showed potent radical scavenging properties compared to the ethyl acetate extracts of A. leiocarpus leaves and stem bark. Besides, it was noted that the stem bark extracts were more potent radical scavengers when compared to the leaves extracts. Furthermore, the strong radical scavenging activities of the extracts was related to their high phenolic contents. Reducing power, as described by electron transfer ability, is considered to be one of the key indicators of antioxidant capacity of plant extracts [28]. In this study, the reducing power of A. leiocarpus extracts was assessed using FRAP and CUPRAC assays, which are characterized by the reduction of Fe3+ to Fe2+ and Cu2+ to Cu+, respectively [30,31]. Comparable to the free radical scavenging assessment, the methanol and water extracts of A. leiocarpus showed potent reducing properties as compared to the ethyl acetate extracts. The total antioxidant capacity that was determined by the phosphomolybdenum assay demonstrated that methanol and water extracts of A. leiocarpus leaves and stem bark were more potent antioxidants as compared to the ethyl acetate extracts. This finding is in line with radical scavenging and reducing power evaluations. The ability of A. leiocarpus extracts to chelate metal was also evaluated and is presented in Table 4. Given the recognised role of iron in oxidative stress, which is understood as an increase in oxygen radical intermediates concentration, eventually leading to dysregulation, the development of metal chelators having the ability to restore metal homeostasis and oxidative status appears to be a valuable challenge, particularly if the chelators possess other important biological activities that might mitigate other diseases [32]. Data that were gathered from this study revealed that the water extract of A. leiocarpus leaves, possessing the highest flavonoid content, was the most activity metal chelator. It has previously been proposed that flavonoids were potent chelators of iron [33]. Naringenin, quercetin, luteolin, and catechin compounds, belonging to the flavonoid family, were identified in the water extract of A. leiocarpus leaves and they were reported to possess metal chelating abilities [33,34]. For instance, quercetin was reported to form different complexes with Fe2+ through its 5-OH and 4-carbonyl groups [35].
Table 4.
Antioxidant properties of A. leiocarpus extracts.
| Samples | DPPH (mmol TE/g) |
ABTS (mmol TE/g) |
CUPRAC (mmol TE/g) |
FRAP (mmol TE/g) |
Metal Chelating (mg EDTAE/g) |
Phosphomolybdenum (mmol TE/g) |
|---|---|---|---|---|---|---|
| Leaves-EA | 30 ± 0.01f | 0.26 ± 0.02e | 0.50 ± 0.04e | 0.26 ± 0.03d | 10.0 ± 0.8f | 2.0 ± 0.1e |
| Leaves-MeOH | 5.0 ± 0.1d | 3.0 ± 0.4c | 7.0 ± 0.2c | 4.0 ± 0.1c | 47.0 ± 0.8c | 4.0 ± 0.1d |
| Leaves-Water | 5.0 ± 0.1c | 4.0 ± 0.3b | 7.0 ± 0.2b | 6.0 ± 0.4b | 79.0 ± 0.9a | 4.0 ± 0.1d |
| Stem barks-EA | 3.0 ± 0.1e | 2.0 ± 0.1d | 5.0 ± 0.2d | 4.0 ± 0.1c | 30.0 ± 0.5e | 4.0 ± 0.1c |
| Stem barks-MeOH | 6.0 ± 0.1a | 5.0 ± 0.1a | 8.0 ± 0.2a | 6.0 ± 0.3a | 45.0 ± 0.6d | 6.0 ± 0.2a |
| Stem barks-Water | 5.0 ± 0.1b | 4.0 ± 0.4b | 8.0 ± 0.1b | 6.0 ± 0.2a | 61.0 ± 0.4b | 5.0 ± 0.1a |
Values expressed are means ± S.D. of three parallel measurements. DPPH: 2-diphenyl-1-picrylhydrazyl; ABTS: 2,2’-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid; CUPRAC: cupric reducing antioxidant capacity; FRAP: ferric reducing antioxidant power; TE: Trolox equivalent; EDTAE: ethylenediaminetetraacetic acid equivalent. Superscripts in the same column indicate significant difference in the tested extracts (p < 0.05).
3.6. Enzyme Inhibitory Activities
While the antioxidant activity of plant extract is often linked to the phenolic content, the enzyme inhibitory properties of extracts mainly involves the interaction of phytochemicals with the enzyme or enzyme-substrate complex. To the best of our knowledge, this is the first report on the assessment of the inhibitory potential of A. leiocarpus leaves and stem bark on enzymes related to Alzheimer’s disease and skin hyperpigmentation. A previous study has appraised the amylase and glucosidase inhibitory action of A. leiocarpus leaves [5]. However, no comparison has been made with the stem bark extract of the plant and the possible effect of different extraction solvents. Among the five food drug administration (FDA)-approved Alzheimer’s disease treatments, four are acetyl cholinesterase inhibitors [36]. Cholinesterase inhibitors designed for the management of Alzheimer’s disease stem from the cholinergic hypothesis, which is the leading theory proposed to explain the pathogenesis of Alzheimer’s disease [37]. It has been recognised that cholinergic neurons loss in brain area that is responsible for cognition and behaviour was the hallmark of Alzheimer’s disease. While the role of acetyl cholinesterase has been clearly claimed, the exact mechanism that involves butyryl cholinesterase remains elusive. Butyryl cholinesterase, previously underestimated in the pathogenesis of Alzheimer’s disease, was found to be up-regulated in advanced stages of the condition and plays a key role in the disease maintenance and progression [38]. From this perspective, it can be stated that cholinesterase inhibitors targeting both acetyl and butyryl cholinesterases are in need. Table 5 reports the acetyl cholinesterase inhibitory activity of the different extracts of A. leiocarpus leaves and stem bark ranging from 3.51 to 4.68 mg GALAE/g. With regards to the butyryl cholinesterase inhibitory action, the values ranged from 0.45 to 4.0 mg GALAE/g. Interestingly, the water extract of A. leiocarpus leaves only inhibited acetyl cholinesterase. It can be suggested that this extract might be targeted at the initial stage of the disease, when butyryl cholinesterase activity is not pronounced. The methanol extract of A. leiocarpus leaves (4.68 and 4.0 mg GALAE/g) showed potent inhibition against both cholinesterases. Over the past decades, there has been an emerging trend of naturally derived cosmetic products. This shift has encouraged researchers to find new cosmeceuticals and the focus has geared towards plants. Plant extracts have witnessed increased global demand for de-pigmenting agents due to their safety and compatibility with all skin types [39]. The inhibitory action of A. leiocarpus extracts on tyrosinase, a copper-containing enzyme responsible for the biosynthesis of melanin [40], was investigated. The data collected showed potent tyrosinase inhibition with values ranging from 113.0 to 155.26 mg KAE/g, the highest values was recorded for methanol extract of A. leiocarpus stem bark. Pinocembrin, shikimic acid, and vitexin, tentatively identified in the methanol extract of A. leiocarpus stem bark, were previously reported to inhibit tyrosinase [41,42,43]. A group of researchers [5] have reported the amylase (IC50 value of 242.17 μg/mL) and glucosidase (IC50 value of 196.35 μg/mL) inhibitory activity of A. leiocarpus leaves water extract. In the present investigation and, as opposed to the previous study, low inhibition was recorded against amylase, while no inhibitory action was observed against glucosidase in the presence of the water extract of A. leiocarpus leaves. The different activity that was recorded in our study might be related to the geographical location along with the environmental conditions of the studied A. leiocarpus plants. In this study, it was observed that the different A. leiocarpus extracts were poor inhibitors of amylase, with values that ranged from 0.19 to 1.13 mmol ACAE/g. Only ethyl acetate extracts inhibited glucosidase and the values were higher as compared to amylase. The inhibition of glucosidase is considered as strategic in the management of diabetes type II. Indeed, it has been advocated that the inhibition of glucosidase reduced post-prandial glucose rise and it was associated to less side effects.
Table 5.
Enzyme inhibitory properties of A. leiocarpus extracts.
| Samples | AChE Inhibition (mg GALAE/g) | BChE Inhibition (mg GALAE/g) |
Tyrosinase Inhibition (mg KAE/g) |
Amylase Inhibition (mmol ACAE/g) |
Glucosidase Inhibition (mmol ACAE/g) |
|---|---|---|---|---|---|
| Leaves-EA | 4.0 ± 0.2d | 3.0 ± 0.2b | 131.0 ± 0.2d | 0.79 ± 0.04b | 15.0±0.1b |
| Leaves-MeOH | 4.68 ± 0.02a | 4.0 ± 0.1a | 154.0 ± 0.2b | 1.0 ± 0.1a | nd |
| Leaves-Water | 4.19 ± 0.04c | nd | 113 ± 1e | 0.53 ± 0.04c | nd |
| Stem barks-EA | 4.0 ± 0.1bc | 2.0 ± 0.2c | 152.0 ± 0.6c | 1.0 ± 0.1b | 15.0 ± 0.1a |
| Stem barks-MeOH | 4.0 ± 0.1b | 1.0 ± 0.3c | 155.26 ± 0.04a | 0.85 ± 0.03b | nd |
| Stem barks-Water | 4.0 ± 0.1e | 0.5 ± 0.1d | 113.0 ± 0.6e | 0.19 ± 0.01d | nd |
Values expressed are means ± S.D. of three parallel measurements. AChE: Acetylcholinesterase; BChE: Butyrylcholinesterase; GALAE: Galatamine equivalent; KAE: Kojic acid equivalent; ACAE: Acarbose equivalent. nd: not detected. Superscripts in the same column indicate significant difference in the tested extracts (p <0.05).
3.7. Multivariate Analysis
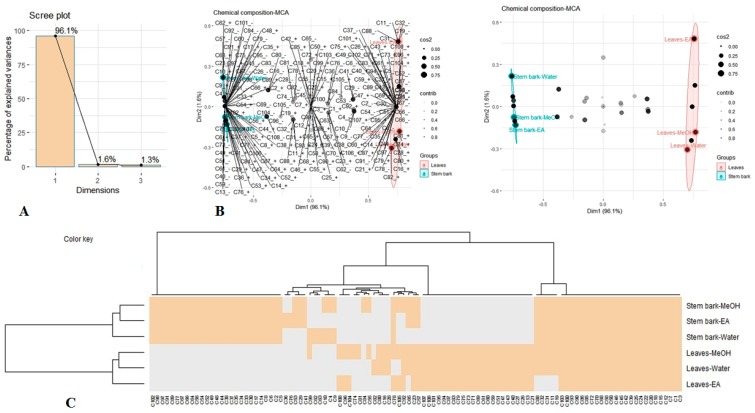
An unsupervised multiple correspondence analysis (MCA), a Heatmap clustering approach, and Venn graph were applied on the chemical composition of A. leiocarpus samples to obtain a typology of the samples and to characterize the chemical profile differences among those samples. MCA is commonly used qualitative variables to examine a set of observations described by a set of nominal variables. Figure 1A displays the proportion of explained inertia per component and the projection of the samples and chemical compounds on the first two dimensions. Two components were required to summarize approximately 97.7% of the variance. The first component explained 96,1% of variance, while the second accounted for 1, 6%. As we could notice, the samples were predominantly separated by the first component of MCA, with the extracts of stem bark being grouped on the negative side of the factors and the leaves extract on the positive side (Figure 1B). In agreement with MCA, Heatmap separated and categorized the samples into two groups, with each group being divided into two sub-clusters (Figure 1C). On the other hand, we observed that the stem bark extracts were more homogeneous than the leaves extracts. Indeed, ethyl acetate extract of leaves was clearly separated from the other two extracts (methanol and water), which were close (Figure 1B). This indicates that solvent used have a large influence on the leaves secondary metabolite extraction than that of stem bark. Moreover, when cross-checking a list of 108 compounds that were identified in all the leaves extracts against those were recognized in all stem bark extracts, we observed that a total of 38 were commonly detected in the two organs, whereas there were 38 and 32 unique compounds found in stem bark and leaves, respectively (Figure 2).
Figure 1.
Multivariate analysis using multiple correspondence analysis (MCA) and Heatmap clustering analysis of chemical composition in A. leiocarpus extracts. (A): Percentage of explained variance per component. (B): projection of extracts and chemical compounds into the subspace spanned by the first two components of MCA. (C): Clustered Image Map (Euclidean Distance, Ward linkage). Gray colour: Absence, Wheat colour: Presence.
Figure 2.
Venn diagram representing the overlap of compounds on the two organs.
The analysis showed the variation of chemical composition of A. leiocarpus depending to part used, as well as the influence of solvent on the extraction of compounds, with a more pronounced effect on the leaves. In agreement with this observation, we decided to ascertain whether the plant parts and solvent types used had any statistically significant effect on both total bioactive compounds content and biological activities of A. leiocarpus. Thus, multivariate methods for integrative large biological data sets, namely DIABLO, was applied to total bioactive compounds content and biological activities data-sets. DIABLO is a highly flexible supervised multivariate method that enables to classify in an optimal and reliable manner the studied samples and to construct a predictive multi-omics model that can be used to classify new samples. Figure 3 shows the multivariate analysis results. From Figure 3A,B, it is clear that there were parts and solvents effects on the total bioactive compounds content and biological activities of A. leiocarpus. In fact, as we could observe in Figure 3A, the factorial plan discriminated leaves parts with the stem bark parts effectively in both bioactive compounds and biological activities data sets. A similar outcome was provided with the second studied factor, in short, a clear segregation between the solvents was achieved (Figure 3B). Furthermore, a better separation of solvents was found while using biological activities data than when using the bioactive compounds data. The different extracts of the stem bark were relatively close as we have seen on the plots. As well by observing the samples plot using bioactive compounds content data (Figure 3A,B, Block: Bioactive compounds), this view was echoed with a consolidation of stem bark extracts contrast to a high variability between the leaves extracts. Accordingly, the extraction solvent, by extension the change of polarity, greatly influenced the bioactive compounds content of leaves than those of stem bark. The present result indicated that A. leiocarpus leaves, unlike stem bark, contain chemical molecules with varying polarity and solubility that are sensitive to the variation of solvent.
Figure 3.
N-integration across multiple datasets analysis on Anogeissus leiocarpus bioactive compounds content and biological activities according to two factors (solvents and parts). (A,B): Sample plot with confidence ellipse according to the parts of plant and the extracting solvent as factor, respectively. (C,F): The model performance per component for Centroids Distance using 5-fold CV repeated 10 times. (D,G): the global overview of the relationship between the two datasets at the two first component level. (E,H): AUC (area under the curve) average and ROC (Receiver Operating Characteristic Curve) curve using one-vs-all comparisons.
Figure 3D,G shows that the first three and two components, respectively, of bioactive compounds dataset were positively correlated to biological activities dataset, which allowed for us to say that DIABLO analysis was able to model a good agreement between our datasets. Subsequently, to compare DIABLO models that include/exclude the repeated measures experimental design, we examined the ROC assay (Receiver Operating Characteristic Curve). As we could observe in Figure 3E, the AUC (area under the curve) for the first three component for bioactive compounds and biological activities were 0.89 and 0.77, respectively. As for the second model, the AUC for the first two component were 1 for both bioactive compounds and biological activities (Figure 3H). Finally, the performance of each model was evaluated by estimating the classification error rate. Centroids distance was used as prediction distance and 10 × 5-fold CV as repeated stratified cross-validation. Thus, by observing Figure 3C,F, the best performance was obtained for 3 and 2 component, respectively, which suggests a satisfactory result on our model.
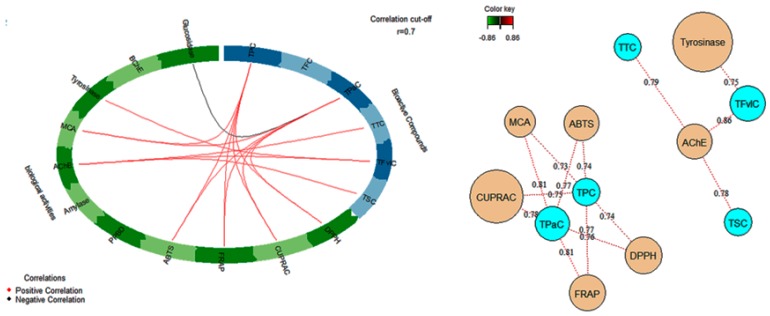
Circos plot and network were carried out to analyze the correlative relationships between total bioactive compound contents of each extract with their biological activities (Figure 4). The analysis revealed that AChE had a positive correlation with total tannin content (TTC) (r = 0.79), total saponin content (TSC) (r = 0.78) and total flavonol content (TFvLC) (r = 0.86), whereas tyrosinase was correlated with TFvLC (r = 0.75). Likewise total phenolic content (TPC) and total phenolic acid (TPaC) were positively related to Radical Scavenging Activity ABTS (r = 0.74; r = 0.77) and DPPH (r = 0.74; r = 0.77), Reducing Power ability FRAP (r = 0.77; r = 0.81) and CUPRAC (r = 0.75; r = 0.78) and Ferrous ion Chelation (MCA) (r = 0.73; r = 0.81). Accordingly, it was obvious that the phenolic compounds especially phenolic acid compounds were mostly responsible for the antioxidant activities and metal chelating ability of A. leiocarpus extracts.
Figure 4.
Circos-plot and network showing the relationship between total bioactive compounds and evaluated biological activities (cut-off: r = 0.7).
3.8. Pharmacological Studies
With the aim of investigating extract biological activity, EA, MeOH, and water extracts of A. leiocarpus leaves and stem barks were assayed through the allelopathy test. To this regard, the seeds of the commercial Lollo bionda lettuce cultivar were exposed to scalar extract concentrations (0.1–10 mg/mL), and the seedling germination and growth were monitored. After incubation of seeds with extracts, we observed a null effect on the seedling germination (Figure 5), thus obtaining a preliminary index of the extract biocompatibility.
Figure 5.
Effects of A. leiocarpus leaf (L) and stem bark (SB) extracts (0.1–10 mg/mL) on Lollo bionda lettuce root elongation rate. Data, expressed as mean length distribution of germinated seeds, are means ± SD of three experiments performed in triplicate. After exposing lettuce roots to the extracts, a null effect on seedling germination was observed. EA: Ethyl acetate; MeOH: Methanol.
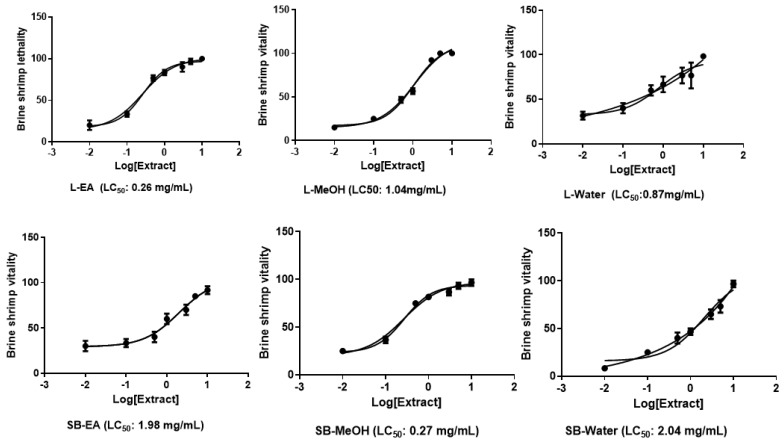
As a further approach to evaluate potential toxicity, A. leiocarpus extracts, in the concentration range 0.01–10 mg/mL, were tested on brine shrimp lethality assay, performed on the brine shrimp Artemia salina Leach, which is recognized as a valuable tool to predict potential cytotoxicity related to plant extracts [44]. The experimental procedure was conducted in agreement with a previous published paradigm [45]. The results of this assay indicated LC50 values in the range 0.26–2.04 mg/mL (Figure 6), which were indicatory to choose the extract concentration for the in vitro and ex vivo investigations in order to elucidate putative protective effects, in the colon.
Figure 6.
Effects of A. leiocarpus leaf (L) and stem bark (SB) extracts (0.01–10 mg/mL) on Artemia salina Leach lethality (Brine shrimp lethality test). Data are means ± SD of three experiments performed in triplicate. After exposing brine shrimps to the extracts, LC50 values in the range 0.26–2.04 mg/mL were recorded. EA: Ethyl acetate; MeOH: Methanol.
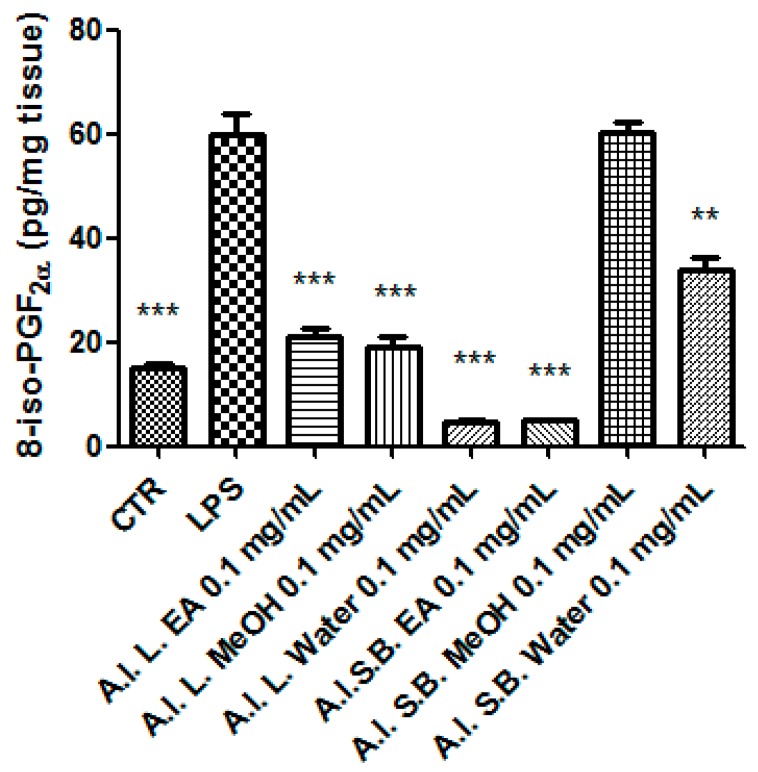
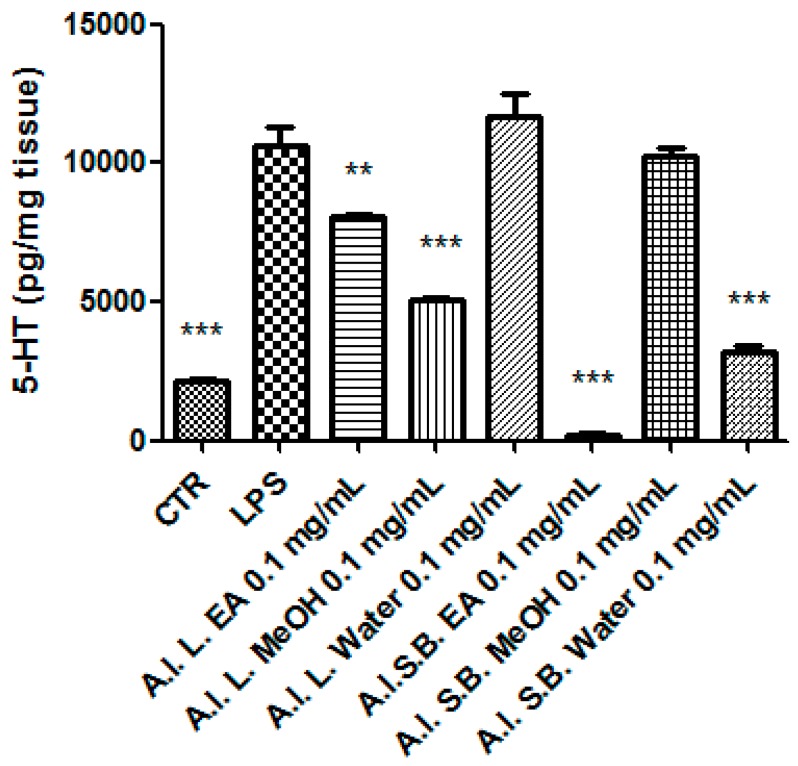
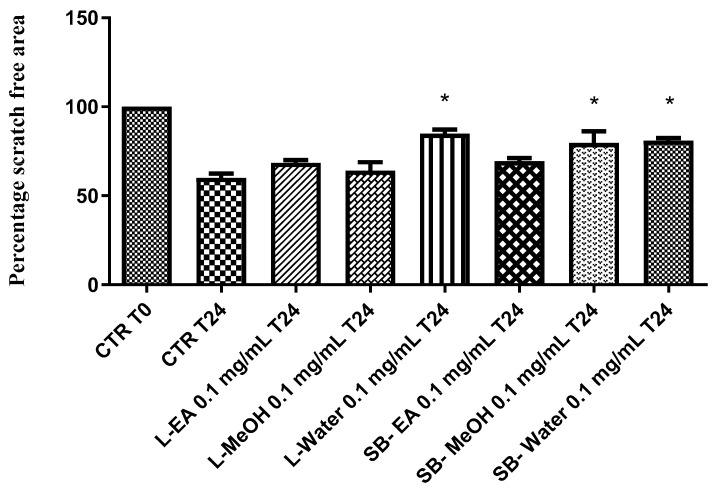
Particularly, we selected the concentration 0.1 mg/mL that was at least two-fold lower than LC50 and in agreement with previous investigations that demonstrated the antioxidant effects on isolated porcine tissue [46]. While considering these findings, we assayed extract effects on rat colon stimulated with LPS, ex vivo, in order reproduce the burden of oxidative stress and inflammation that characterize ulcerative colitis [20,47,48]. All extracts, with the only exception of stembark methanol extract, revealed effective in reducing LPS-induced 8-iso-PGF2α level (Figure 7). On the other hand, all of the extracts blunted LPS-stimulated PGE2 colon level (Figure 8), whereas leaf water and stem bark methanol extracts failed to reduce 5-HT concentration (Figure 9). Finally, when the extracts were tested on colon cancer HCT116 cell line, only stem bark ethyl acetate extract revealed biocompatibility, exerting a null effect on cell proliferation (Figure 10 and Figure 11). Conversely, the other extracts displayed stimulatory effects on either viability or spontaneous migration of HCT116 cells.
Figure 7.
Effect A. leiocarpus leaf (L) and stem bark (SB) extracts (0.01 mg/mL) on lipopolysaccharide (LPS)-induced 8-iso-prostaglandin(PG)F2α level in isolated rat colon. EA: Ethyl acetate; MeOH: Methanol. ANOVA, p < 0.0001; post hoc, **p < 0.01, ***p < 0.001 vs. LPS.
Figure 8.
Effect A. leiocarpus leaf (L) and stem bark (SB) extracts (0.01 mg/mL) on lipopolysaccharide (LPS)-induced prostaglandin(PG)E2 level in isolated rat colon. EA: Ethyl acetate; MeOH: Methanol. ANOVA, p < 0.0001; post hoc, ***p < 0.001 vs. LPS.
Figure 9.
Effect A. leiocarpus leaf (L) and stem bark (SB) extracts (0.01 mg/mL) on lipopolysaccharide (LPS)-induced serotonin (5-HT) level in isolated rat colon. EA: Ethyl acetate; MeOH: Methanol. ANOVA, p < 0.0001; post hoc, **p < 0.01, ***p < 0.001 vs. LPS.
Figure 10.
Effect A. leiocarpus leaf (L) and stem bark (SB) extracts (0.01 mg/mL) on human colon cancer HCT116 cell line viability (MTT assay). EA: Ethyl acetate; MeOH: Methanol. ANOVA, p < 0.0001; post hoc, *p < 0.05, ***p < 0.001 vs. CTR (Control group).
Figure 11.
Effect A. leiocarpus leaf (L) and stem bark (SB) extracts (0.01 mg/mL) on human colon cancer HCT116 cell line spontaneous migration (wound healing assay). EA: Ethyl acetate; MeOH: Methanol. ANOVA, p < 0.01; post hoc, *p < 0.05 vs. CTR T24 (Control group).
4. Discussion
Oxidative stress is characterized by the overproduction of reactive oxygen/nitrogen (ROS/RNS) species that could drive to lipid peroxidation [49], which displays a key role in the pathogenesis of ulcerative colitis [50]. 8-iso-PGF2α, deriving from ROS/RNS peroxidation of membrane arachidonic acid, represents a stable marker of lipid peroxidation and tissue damage, in vivo [51], whereas the blunting effects on 8-iso-PGF2α production that are induced by herbal extracts were related to protective effects [48,52]. Consistently with the reported antioxidant effects and the findings by Belemnaba et al. [46], the tested extracts blunted LPS-induced 8-iso-PGF2α production, with the only exception being represented by stem bark MeOH extract. On the other hand, stem bark MeOH extract showed the ability to reduce the level of malondialdehyde (MDA), which is another key marker of lipid peroxidation, in vivo [53]. This discrepancy could depend on more than one speculation. On one side, the differences in the employed experimental ex vivo and in vivo paradigms that were chosen by us and Akanbi and colleagues [53], respectively, could be crucial. On the other side, we should also consider that isoprostanes derive only by arachidonic acid peroxidation, whereas MDA could originate from various polyunsaturated acids [54], and this could represent a limit in the evaluation of lipid peroxidation.
The effects of A. leiocarpus extracts on LPS-induced levels of colon PGE2 were investigated as well. PGE2 is a cyclooxygenase (COX)-2-derived pro-inflammatory mediator, whose upregulation has been long involved in colon inflammation and damage, whereas the antioxidants were revealed to be effective in blunting the colon levels of this prostaglandin [47,55]. All of the tested extracts proved able in down-regulating LPS-induced PGE2 level (Figure 8), consistently with the reported antioxidant effects.
A blunting effect on LPS-induced 5-HT level was observed after treating the colon specimens with A. leiocarpus extract, as well. 5-HT pro-inflammatory role in ulcerative colitis was previously suggested [56], which possibly involved the activation of 5-HT3 receptors [57]. With the only exception of leaf water and stem bark MeOH extracts, the A. leiocarpus extracts displayed a significant inhibition of 5-HT steady state level, in the colon. This could be, albeit partially, related to decreased neurotransmitter synthesis and release, in the colon tissue [58,59].
Collectively, all of the tested A. leiocarpus extracts could play a noteworthy anti-inflammatory role, as indicated by their blunting effects on LPS-stimulated PGE2 level. On the other hand, the lack of efficacy that was exerted by the leaf water and stem bark MeOH extracts on LPS-induced levels of 5-HT and 8-iso-PGF2α suggests that the lower quantitative profile of gallic acid, catechin, and epicatechin could limit the antioxidant potency, as compared to the other tested extracts.
Finally, the extracts were tested for their putative anti-proliferative role against the human colon HCT116 cell line, which was previously found to be sensitive to different polarity extracts from A. latifolia [12]. In the present study, the anti-proliferative effects were investigated through validated in vitro tests, including MTT and wound healing assays. A different pattern of effects on cell viability was observed after exposing HCT116 cells to A. leiocarpus extracts. On one side, stem bark extracts displayed a null effect on cell viability (Figure 10), which resulted in the range of biocompatibility (>70% and <130% as compared to control group). On the other side, leaf extract increased significantly HCT116 cell viability (>140% as compared to control group). Actually, the stimulating effect on HCT116 cell viability induced by leaf extracts could be related to the their higher qualitative content of metabolites that are related to quercetin, which was found to exert protective effects on this cell line [60]. Additionally, water leaf and water and MeOH stem bark extracts induced spontaneous HCT116 cell migration in wound healing assay (Figure 11), thus also indicating a potential stimulating-effect on invasion capacity. Conversely, stem bark EA and leaf EA and MeOH extracts did not exert any influence on spontaneous HCT116 cell migration, in the 24 h following experimental lesion induced on cell monolayer. Actually, the different effects that were showed by the tested extracts in the wound healing paradigm could be related, at least in part, to the different content of gallic acid, which was found to inhibit spontaneous cell migration [61]. As a conclusive note of the pharmacological investigation, it resulted of particular interest the antioxidant/anti-inflammatory profile that was exerted by the stem bark EA extract, together with its null effect on colon cancer cell proliferation. The highest inhibitory effect that is exerted by this extract on colon 5-HT level could be one of the main causes leading to the null effect on HCT116 viability and spontaneous migration [62].
5. Conclusions
Data that are presented in this study highlighted the key role of solvent choice in the quest for novel bioactive compounds from plants. It was demonstrated that water and methanol were good solvents for the extraction of phytochemicals having antioxidant properties. The methanol extract of A. leiocarpus leaves was also an active cholinesterase inhibitor, while the methanol extract of the stem bark inhibited tyrosinase. On the other hand, the ethyl acetate extract of A. leiocarpus leaves and stem bark showed potent inhibition against α-glucosidase. The pharmacological study that was carried out on isolated colon and HCT116 cell line further deepened the spectrum of potential application of the present extracts. Noteworthy interest derives from the stem bark EA extract that, besides exerting the best antioxidant/anti-inflammatory profile, was the only one that was unable to stimulate the proliferation of human colon cancer HCT116 cell line, thus supporting potential application in the prevention of the oxidative stress-induced tissue damage occurring in ulcerative colitis. As a conclusion, A. leiocarpus stem bark EA extract represents a potential source of bioactive compounds for the development of novel therapeutic agents.
Acknowledgments
This work was supported by grants from the Italian Ministry of University (FAR 2016 granted to Claudio Ferrante; FAR 2018 granted to Giustino Orlando; FAR 2016 granted to Luigi Menghini).
Supplementary Materials
The following are available online at https://www.mdpi.com/2076-3921/8/9/343/s1. Table S1: Chemical composition of A. leiocarpus leaves ethyl acetate extract. Table S2: Chemical composition of A. leiocarpus leaves methanol extract. Table S3: Chemical composition of A. leiocarpus leaves water extract. Table S4: Chemical composition of A. leiocarpus stem bark ethyl acetate extract. Table S5: Chemical composition of A. leiocarpus stem bark methanol extract. Table S6: Chemical composition of A. leiocarpus stem bark water extract.
Author Contributions
Conceptualization, C.F. and G.Z.; methodology, G.O.; L.M.; software, L.M.; validation, C.F.; G.Z.; L.M.; G.O.; formal analysis, C.F.; G.Z.; investigation, K.I.S.; K.B.; A.D.; J.J.; L.R.; A.C.; S.L.; Z.C.; S.D.S.; C.M.N.P.-A.; resources, C.F.; G.O.; L.M.; data curation, C.F.; G.Z.; writing—original draft preparation, M.F.M.; writing—review and editing, C.F., G.Z.; G.O.; L.M.; visualization, L.B.; supervision, L.B.; project administration, C.F.; L.M.; G.O.; G.Z.; funding acquisition, C.F.; G.O.; L.M.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Chaabi M., Benayache S., Benayache F., N’Gom S., Koné M., Anton R., Weniger B., Lobstein A. Triterpenes and polyphenols from Anogeissus leiocarpus (Combretaceae) Biochem. Syst. Ecol. 2008;36:59–62. doi: 10.1016/j.bse.2007.05.007. [DOI] [Google Scholar]
- 2.Salih E.Y.A., Kanninen M., Sipi M., Luukkanen O., Hiltunen R., Vuorela H., Julkunen-Tiitto R., Fyhrquist P. Tannins, flavonoids and stilbenes in extracts of African savanna woodland trees Terminalia brownii, Terminalia laxiflora and Anogeissus leiocarpus showing promising antibacterial potential. S. Afr. J. Bot. 2017;108:370–386. doi: 10.1016/j.sajb.2016.08.020. [DOI] [Google Scholar]
- 3.Ademosun A.O., Adebayo A.A., Oboh G. Anogeissus leiocarpus attenuates paroxetine-induced erectile dysfunction in male rats via enhanced sexual behavior, nitric oxide level and antioxidant status. Biomed. Pharmacother. 2019;111:1029–1035. doi: 10.1016/j.biopha.2019.01.022. [DOI] [PubMed] [Google Scholar]
- 4.Etuk E., Mohammed B. Informant consensus selection method: A reliability assessment on medicinal plants used in north western Nigeria for the treatment of diabetes mellitus. Afr. J. Pharm. Pharmacol. 2009;3:496–500. [Google Scholar]
- 5.Adefegha S.A., Oboh G., Omojokun O.S., Jimoh T.O., Oyeleye S.I. In vitro antioxidant activities of African birch (Anogeissus leiocarpus) leaf and its effect on the α-amylase and α-glucosidase inhibitory properties of acarbose. J. Taibah Univ. Med. Sci. 2016;11:236–242. doi: 10.1016/j.jtumed.2016.03.001. [DOI] [Google Scholar]
- 6.Belemnaba L., Nitiema M., Ilboudo S., Ouedraogo N., Ouedraogo G.G., Belemlilga M.B., Ouedraogo S., Guissou I.P. O8 Study on antihypertensive activity of an aqueous extract of Anogeissus leiocarpus (AEAL) DC Guill et Perr bark of trunk in L-NAME-induced hypertensive rats. Biochem. Pharmacol. 2017;139:112. doi: 10.1016/j.bcp.2017.06.073. [DOI] [Google Scholar]
- 7.Victor B.Y., Grace A. Phytochemical studies, in-vitro antibacterial activities and antioxidant properties of the methanolic and ethyl acetate extracts of the leaves of Anogeissus leiocarpus. Int. J. Biochem. Res. Rev. 2013;3:137. doi: 10.9734/IJBCRR/2013/3349. [DOI] [Google Scholar]
- 8.Shuaibu M.N., Wuyep P.T., Yanagi T., Hirayama K., Ichinose A., Tanaka T., Kouno I. Trypanocidal activity of extracts and compounds from the stem bark of Anogeissus leiocarpus and Terminalia avicennoides. Parasitol. Res. 2008;102:697–703. doi: 10.1007/s00436-007-0815-1. [DOI] [PubMed] [Google Scholar]
- 9.Shuaibu M.N., Pandey K., Wuyep P.A., Yanagi T., Hirayama K., Ichinose A., Tanaka T., Kouno I. Castalagin from Anogeissus leiocarpus mediates the killing of Leishmania in vitro. Parasitol. Res. 2008;103:1333–1338. doi: 10.1007/s00436-008-1137-7. [DOI] [PubMed] [Google Scholar]
- 10.Vonthron-Sénécheau C., Weniger B., Ouattara M., Bi F.T., Kamenan A., Lobstein A., Brun R., Anton R. In vitro antiplasmodial activity and cytotoxicity of ethnobotanically selected Ivorian plants. J. Ethnopharmacol. 2003;87:221–225. doi: 10.1016/S0378-8741(03)00144-2. [DOI] [PubMed] [Google Scholar]
- 11.Akanbi O.M., Omonkhua A.A., Cyril-Olutayo C.M. Effect of crude methanolic extract of Anogeissus leiocarpus on the liver function of P. berghei infected mice. Int. J. Infect. Dis. 2014;21:196. doi: 10.1016/j.ijid.2014.03.830. [DOI] [Google Scholar]
- 12.Diab K.A., Guru S.K., Bhushan S., Saxena A.K. In vitro anticancer activities of Anogeissus latifolia, Terminalia bellerica, Acacia catechu and Moringa oleiferna Indian plants. Asian Pac. J. Cancer Prev. 2015;16:6423–6428. doi: 10.7314/APJCP.2015.16.15.6423. [DOI] [PubMed] [Google Scholar]
- 13.Ferrante C., Recinella L., Ronci M., Menghini L., Brunetti L., Chiavaroli A., Leone S., Di Iorio L., Carradori S., Tirillini B., et al. Multiple pharmacognostic characterization on hemp commercial cultivars: Focus on inflorescence water extract activity. Food Chem. Toxicol. 2019;125:452–461. doi: 10.1016/j.fct.2019.01.035. [DOI] [PubMed] [Google Scholar]
- 14.Zengin G., Aktumsek A. Investigation of antioxidant potentials of solvent extracts from different anatomical parts of Asphodeline anatolica E. Tuzlaci: An endemic plant to Turkey. Afr. J. Tradit. Complement. Altern. Med. 2014;11:481–488. doi: 10.4314/ajtcam.v11i2.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zengin G., Llorent-Martínez E.J., Fernández-de Córdova M.L., Bahadori M.B., Mocan A., Locatelli M., Aktumsek A. Chemical composition and biological activities of extracts from three Salvia species: S. blepharochlaena, S. euphratica var. leiocalycina, and S. verticillata subsp. amasiaca. Ind. Crops Prod. 2018;111:11–21. doi: 10.1016/j.indcrop.2017.09.065. [DOI] [Google Scholar]
- 16.Zengin G., Uysal A., Diuzheva A., Gunes E., Jekő J., Cziáky Z., Picot-Allain C.M.N., Mahomoodally M.F. Characterization of phytochemical components of Ferula halophila extracts using HPLC-MS/MS and their pharmacological potentials: A multi-functional insight. J. Pharm. Biomed. Anal. 2018;160:374–382. doi: 10.1016/j.jpba.2018.08.020. [DOI] [PubMed] [Google Scholar]
- 17.Rodrıguez-Delgado M., Malovana S., Perez J., Borges T., Montelongo F.G. Separation of phenolic compounds by high-performance liquid chromatography with absorbance and fluorimetric detection. J. Chromatogr. A. 2001;912:249–257. doi: 10.1016/S0021-9673(01)00598-2. [DOI] [PubMed] [Google Scholar]
- 18.Uysal S., Zengin G., Locatelli M., Bahadori M.B., Mocan A., Bellagamba G., De Luca E., Mollica A., Aktumsek A. Cytotoxic and enzyme inhibitory potential of two Potentilla species (P. speciosa L. and P. reptans Willd.) and their chemical composition. Front. Pharmacol. 2017;8:290. doi: 10.3389/fphar.2017.00290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mahmoodzadeh H., GhasemI M., Zanganeh H. Allelopathic effect of medicinal plant Cannabis sativa L. on Lactuca sativa L. seed germination. Acta Agric. Slov. 2015;105:233–239. doi: 10.14720/aas.2015.105.2.06. [DOI] [Google Scholar]
- 20.Brunetti L., Orlando G., Ferrante C., Recinella L., Leone S., Chiavaroli A., Di Nisio C., Shohreh R., Manippa F., Ricciuti A., et al. Peripheral chemerin administration modulates hypothalamic control of feeding. Peptides. 2014;51:115–121. doi: 10.1016/j.peptides.2013.11.007. [DOI] [PubMed] [Google Scholar]
- 21.Ferrante C., Orlando G., Recinella L., Leone S., Chiavaroli A., Di Nisio C., Shohreh R., Manippa F., Ricciuti A., Vacca M., et al. Central inhibitory effects on feeding induced by the adipo-myokine irisin. Eur. J. Pharmacol. 2016;791:389–394. doi: 10.1016/j.ejphar.2016.09.011. [DOI] [PubMed] [Google Scholar]
- 22.Chiavaroli A., Brunetti L., Orlando G., Recinella L., Ferrante C., Leone S., Di Michele P., Di Nisio C., Vacca M. Resveratrol inhibits isoprostane production in young and aged rat brain. J. Biol. Regul. Homeost. Agents. 2010;24:441. [PubMed] [Google Scholar]
- 23.Locatelli M., Macchione N., Ferrante C., Chiavaroli A., Recinella L., Carradori S., Zengin G., Cesa S., Leporini L., Leone S. Graminex pollen: Phenolic pattern, colorimetric analysis and protective effects in immortalized prostate cells (PC3) and rat prostate challenged with LPS. Molecules. 2018;23:1145. doi: 10.3390/molecules23051145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.National Centre for Replacement Refinement& Reduction of Animals in Research. [(accessed on 16 August 2019)]; Available online: https://www.nc3rs.org.uk/experimental-designstatistics.
- 25.Galanakis C.M., Goulas V., Tsakona S., Manganaris G.A., Gekas V. A Knowledge Base for the Recovery of Natural Phenols with Different Solvents. Int. J. Food Prop. 2013;16:382–396. doi: 10.1080/10942912.2010.522750. [DOI] [Google Scholar]
- 26.Ji W., Meng Q., Ding L., Wang F., Dong J., Zhou G., Wang B. Measurement and correlation of the solubility of caffeic acid in eight mono and water+ethanol mixed solvents at temperatures from (293.15 to 333.15) K. J. Mol. Liq. 2016;224:1275–1281. doi: 10.1016/j.molliq.2016.10.110. [DOI] [Google Scholar]
- 27.Lin L.-T., Chen T.-Y., Chung C.-Y., Noyce R.S., Grindley T.B., McCormick C., Lin T.-C., Wang G.-H., Lin C.-C., Richardson C.D. Hydrolyzable Tannins (Chebulagic Acid and Punicalagin) Target Viral Glycoprotein-Glycosaminoglycan Interactions To Inhibit Herpes Simplex Virus 1 Entry and Cell-to-Cell Spread. J. Virol. 2011;85:4386–4398. doi: 10.1128/JVI.01492-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Perera H.D.S.M., Samarasekera J.K.R.R., Handunnetti S.M., Weerasena O.V.D.S.J. In vitro anti-inflammatory and anti-oxidant activities of Sri Lankan medicinal plants. Ind. Crops Prod. 2016;94:610–620. doi: 10.1016/j.indcrop.2016.09.009. [DOI] [Google Scholar]
- 29.Holtz R.W. Chapter 13—In Vitro Methods to Screen Materials for Anti-aging Effects. In: Dayan N., editor. Skin Aging Handbook. William Andrew Publishing; Norwich, NY, USA: 2009. pp. 329–362. [Google Scholar]
- 30.Cerretani L., Bendini A. Chapter 67—Rapid Assays to Evaluate the Antioxidant Capacity of Phenols in Virgin Olive Oil. In: Preedy V.R., Watson R.R., editors. Olives and Olive Oil in Health and Disease Prevention. Academic Press; San Diego, CA, USA: 2010. pp. 625–635. [Google Scholar]
- 31.Özyürek M., Güçlü K., Tütem E., Başkan K.S., Erçağ E., Celik S.E., Baki S., Yıldız L., Karaman Ş., Apak R. A comprehensive review of CUPRAC methodology. Anal. Methods. 2011;3:2439–2453. doi: 10.1039/c1ay05320e. [DOI] [Google Scholar]
- 32.Puntarulo S. Iron, oxidative stress and human health. Mol. Asp. Med. 2005;26:299–312. doi: 10.1016/j.mam.2005.07.001. [DOI] [PubMed] [Google Scholar]
- 33.Mira L., Fernandez M.T., Santos M., Rocha R., Florencio M.H., Jennings K.R. Interactions of flavonoids with iron and copper ions: A mechanism for their antioxidant activity. Free Radic. Res. 2002;36:1199–1208. doi: 10.1080/1071576021000016463. [DOI] [PubMed] [Google Scholar]
- 34.Mandel S., Amit T., Reznichenko L., Weinreb O., Youdim M.B. Green tea catechins as brain-permeable, natural iron chelators-antioxidants for the treatment of neurodegenerative disorders. Mol. Nutr. Food Res. 2006;50:229–234. doi: 10.1002/mnfr.200500156. [DOI] [PubMed] [Google Scholar]
- 35.Liu Y., Guo M. Studies on Transition Metal-Quercetin Complexes Using Electrospray Ionization Tandem Mass Spectrometry. Molecules. 2015;20:8583–8594. doi: 10.3390/molecules20058583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Santos M.A., Chand K., Chaves S. Recent progress in multifunctional metal chelators as potential drugs for Alzheimer’s disease. Coord. Chem. Rev. 2016;327:287–303. doi: 10.1016/j.ccr.2016.04.013. [DOI] [Google Scholar]
- 37.Ragab H.M., Teleb M., Haidar H.R., Gouda N. Chlorinated tacrine analogs: Design, synthesis and biological evaluation of their anti-cholinesterase activity as potential treatment for Alzheimer’s disease. Bioorg. Chem. 2019;86:557–568. doi: 10.1016/j.bioorg.2019.02.033. [DOI] [PubMed] [Google Scholar]
- 38.de Andrade P., Mantoani S.P., Gonçalves Nunes P.S., Magadán C.R., Pérez C., Xavier D.J., Hojo E.T.S., Campillo N.E., Martínez A., Carvalho I. Highly potent and selective aryl-1,2,3-triazolyl benzylpiperidine inhibitors toward butyrylcholinesterase in Alzheimer’s disease. Bioorg. Med. Chem. 2019;27:931–943. doi: 10.1016/j.bmc.2018.12.030. [DOI] [PubMed] [Google Scholar]
- 39.Liyanaarachchi G.D., Samarasekera J.K.R.R., Mahanama K.R.R., Hemalal K.D.P. Tyrosinase, elastase, hyaluronidase, inhibitory and antioxidant activity of Sri Lankan medicinal plants for novel cosmeceuticals. Ind. Crops Prod. 2018;111:597–605. doi: 10.1016/j.indcrop.2017.11.019. [DOI] [Google Scholar]
- 40.Wang R., Wang G., Xia Y., Sui W., Si C. Functionality study of lignin as a tyrosinase inhibitor: Influence of lignin heterogeneity on anti-tyrosinase activity. Int. J. Biol. Macromol. 2019;128:107–113. doi: 10.1016/j.ijbiomac.2019.01.089. [DOI] [PubMed] [Google Scholar]
- 41.Chen Y.H., Huang L., Wen Z.H., Zhang C., Liang C.H., Lai S.T., Luo L.Z., Wang Y.Y., Wang G.H. Skin whitening capability of shikimic acid pathway compound. Eur. Rev. Med. Pharmacol. Sci. 2016;20:1214–1220. [PubMed] [Google Scholar]
- 42.Mapunya M.B., Hussein A.A., Rodriguez B., Lall N. Tyrosinase activity of Greyia flanaganii (Bolus) constituents. Phytomedicine. 2011;18:1006–1012. doi: 10.1016/j.phymed.2011.03.013. [DOI] [PubMed] [Google Scholar]
- 43.Yao Y., Cheng X., Wang L., Wang S., Ren G. Mushroom tyrosinase inhibitors from mung bean (Vigna radiatae L.) extracts. Int. J. Food Sci. Nutr. 2012;63:358–361. doi: 10.3109/09637486.2011.629177. [DOI] [PubMed] [Google Scholar]
- 44.Ohikhena F.U., Wintola O.A., Afolayan A.J. Toxicity Assessment of Different Solvent Extracts of the Medicinal Plant, Phragmanthera capitata (Sprengel) Balle on Brine Shrimp (Artemia salina) Int. J. Pharmacol. 2016;12:701–710. [Google Scholar]
- 45.Taviano M.F., Marino A., Trovato A., Bellinghieri V., Melchini A., Dugo P., Cacciola F., Donato P., Mondello L., Güvenç A. Juniperus oxycedrus L. subsp. oxycedrus and Juniperus oxycedrus L. subsp. macrocarpa (Sibth. & Sm.) Ball. “berries” from Turkey: Comparative evaluation of phenolic profile, antioxidant, cytotoxic and antimicrobial activities. Food Chem. Toxicol. 2013;58:22–29. doi: 10.1016/j.fct.2013.03.049. [DOI] [PubMed] [Google Scholar]
- 46.Belemnaba L., Ouédraogo S., Nitiéma M., Chataigneau T., Guissou I.P., Schini-Kerth V.B., Bucher B., Auger C. An aqueous extract of the Anogeissus leiocarpus bark (AEAL) induces the endothelium-dependent relaxation of porcine coronary artery rings involving predominantly nitric oxide. J. Basic Clin. Physiol. Pharmacol. 2018;29:599–608. doi: 10.1515/jbcpp-2017-0084. [DOI] [PubMed] [Google Scholar]
- 47.Locatelli M., Ferrante C., Carradori S., Secci D., Leporini L., Chiavaroli A., Leone S., Recinella L., Orlando G., Martinotti S. Optimization of aqueous extraction and biological activity of Harpagophytum procumbens root on ex vivo rat colon inflammatory model. Phytother. Res. 2017;31:937–944. doi: 10.1002/ptr.5821. [DOI] [PubMed] [Google Scholar]
- 48.Menghini L., Ferrante C., Leporini L., Recinella L., Chiavaroli A., Leone S., Pintore G., Vacca M., Orlando G., Brunetti L. An hydroalcoholic chamomile extract modulates inflammatory and immune response in HT29 cells and isolated rat colon. Phytother. Res. 2016;30:1513–1518. doi: 10.1002/ptr.5655. [DOI] [PubMed] [Google Scholar]
- 49.Uttara B., Singh A.V., Zamboni P., Mahajan R. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 2009;7:65–74. doi: 10.2174/157015909787602823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Achitei D., Ciobica A., Balan G., Gologan E., Stanciu C., Stefanescu G. Different profile of peripheral antioxidant enzymes and lipid peroxidation in active and non-active inflammatory bowel disease patients. Dig. Dis. Sci. 2013;58:1244–1249. doi: 10.1007/s10620-012-2510-z. [DOI] [PubMed] [Google Scholar]
- 51.Praticò D. Alzheimer’s disease and oxygen radicals: New insights. Biochem. Pharmacol. 2002;63:563–567. doi: 10.1016/S0006-2952(01)00919-4. [DOI] [PubMed] [Google Scholar]
- 52.Brunetti L., Menghini L., Orlando G., Recinella L., Leone S., Epifano F., Lazzarin F., Chiavaroli A., Ferrante C., Vacca M. Antioxidant effects of garlic in young and aged rat brain in vitro. J. Med. Food. 2009;12:1166–1169. doi: 10.1089/jmf.2008.0176. [DOI] [PubMed] [Google Scholar]
- 53.Akanbi O.M., Omonkhua A.A., Cyril-Olutayo C.M., Fasimoye R.Y. The antiplasmodial activity of Anogeissus leiocarpus and its effect on oxidative stress and lipid profile in mice infected with Plasmodium bergheii. Parasitol. Res. 2012;110:219–226. doi: 10.1007/s00436-011-2472-7. [DOI] [PubMed] [Google Scholar]
- 54.Tsikas D. Assessment of lipid peroxidation by measuring malondialdehyde (MDA) and relatives in biological samples: Analytical and biological challenges. Anal. Biochem. 2017;524:13–30. doi: 10.1016/j.ab.2016.10.021. [DOI] [PubMed] [Google Scholar]
- 55.Yu L., Yan J., Sun Z. D-limonene exhibits anti-inflammatory and antioxidant properties in an ulcerative colitis rat model via regulation of iNOS, COX-2, PGE2 and ERK signaling pathways. Mol. Med. Rep. 2017;15:2339–2346. doi: 10.3892/mmr.2017.6241. [DOI] [PubMed] [Google Scholar]
- 56.Regmi S.C., Park S.-Y., Ku S.K., Kim J.-A. Serotonin regulates innate immune responses of colon epithelial cells through Nox2-derived reactive oxygen species. Free Radic. Biol. Med. 2014;69:377–389. doi: 10.1016/j.freeradbiomed.2014.02.003. [DOI] [PubMed] [Google Scholar]
- 57.Mousavizadeh K., Rahimian R., Fakhfouri G., Aslani F., Ghafourifar P. Anti-inflammatory effects of 5-HT3 receptor antagonist, tropisetron on experimental colitis in rats. Eur. J. Clin. Investig. 2009;39:375–383. doi: 10.1111/j.1365-2362.2009.02102.x. [DOI] [PubMed] [Google Scholar]
- 58.Bungo T., Shiraishi J.-i., Yanagita K., Ohta Y., Fujita M. Effect of nociceptin/orphanin FQ on feeding behavior and hypothalamic neuropeptide expression in layer-type chicks. Gen. Comp. Endocrinol. 2009;163:47–51. doi: 10.1016/j.ygcen.2009.03.009. [DOI] [PubMed] [Google Scholar]
- 59.Clark K.A., MohanKumar S.M., Kasturi B.S., MohanKumar P.S. Effects of central and systemic administration of leptin on neurotransmitter concentrations in specific areas of the hypothalamus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006;290:R306–R312. doi: 10.1152/ajpregu.00350.2005. [DOI] [PubMed] [Google Scholar]
- 60.Salem I.B., Boussabbeh M., Graiet I., Rhouma A., Bacha H., Essefi S.A. Quercetin protects HCT116 cells from Dichlorvos-induced oxidative stress and apoptosis. Cell Stress Chaperones. 2016;21:179–186. doi: 10.1007/s12192-015-0651-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wang X., Liu K., Ruan M., Yang J., Gao Z. Gallic acid inhibits fibroblast growth and migration in keloids through the AKT/ERK signaling pathway. Acta Biochim. Biophys. Sin. 2018;50:1114–1120. doi: 10.1093/abbs/gmy115. [DOI] [PubMed] [Google Scholar]
- 62.Sui X., Zhu J., Tang H., Wang C., Zhou J., Han W., Wang X., Fang Y., Xu Y., Li D., et al. p53 controls colorectal cancer cell invasion by inhibiting the NF-κB-mediated activation of Fascin. Oncotarget. 2015;6:22869–22879. doi: 10.18632/oncotarget.5137. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.