Abstract
At central synapses, a large and fast spike of neurotransmitter efficiently activates postsynaptic receptors. However, low concentrations of transmitter can escape the cleft and activate presynaptic and postsynaptic receptors. We report here that low concentrations of GABA reduce IPSCs in hippocampal neurons by preferentially desensitizing rather than opening GABAAchannels. GABA transporter blockade also caused desensitization by locally elevating GABA to ∼1 μm. Recovery of the IPSC required several seconds, mimicking recovery of the channel from slow desensitization. These results indicate that low levels of GABA can regulate the amplitude of IPSCs by producing a slow form of receptor desensitization. Accumulation of channels in this absorbing state allows GABAA receptors to detect even a few molecules of GABA in the synaptic cleft.
Keywords: synaptic inhibition, desensitization, mIPSC, hippocampus, GABA transporters, ligand-gated ion channel, uptake blocker, postsynaptic
After activation, ligand-gated channels can enter long-lived closed states (i.e., they desensitize). For example, GABAA receptors rapidly desensitize in response to brief exposures of high GABA concentrations. Fast desensitization prolongs GABAergic responses by detaining receptors in a bound conformation from which channels can reopen (Maconochie et al., 1994; Jones and Westbrook, 1995; Galarreta and Hestrin, 1997;Mellor and Randall, 1997; Zhu and Vicini, 1997; Zhu et al., 1998). More prolonged accumulation of receptors in desensitized states can also modulate the amplitude of sequential synaptic responses during repetitive firing (Turecek and Trussell, 2000). Although GABAA receptors desensitize in response to exogenous agonist exposure, and manipulations that modulate macroscopic desensitization in patches also modulate synaptic currents (Jones and Westbrook, 1997; Mellor and Randall, 1997; Zhu and Vicini, 1997; Zhu et al., 1998), the role of GABAA receptor desensitization in response to endogenously released GABA is still not well defined.
Under what conditions might one expect to observe desensitization at synapses? Although fusion of a synaptic vesicle yields a brief and high transmitter concentration profile at the release site (Clements et al., 1992; Clements, 1996), lower concentrations of glutamate and GABA activate receptors and transporters outside the cleft (Isaacson et al., 1993; Asztely et al., 1997; Bergles and Jahr, 1997, 1998; Bergles et al., 1997, 1999; Scanziani et al., 1997; Rossi and Hamann, 1998;Scanziani, 2000). Simulations of glutamate clearance also predict that micromolar levels can persist for relatively long periods at locations distant from the release site (Clements, 1996; Otis et al., 1996a;Barbour and Häusser, 1997; Rusakov and Kullmann, 1999). The consequences of sustained, subsaturating transmitter exposure (Hill et al., 1998) are not necessarily limited to receptor activation. For example, prolonged low concentrations of GABA result in pronounced desensitization of GABAA receptors (Celentano and Wong, 1994; Orser et al., 1994; Berger et al., 1998), possibly because of the presence of a singly bound desensitized state with slow entry and exit rates (Jones and Westbrook, 1995). Thus low levels of GABA could reduce synaptic receptor availability by shuttling receptors into such a slow desensitized state.
We tested this possibility in cultured hippocampal neurons and in brain slices by examining the reduction of GABAAreceptor-mediated currents produced by low levels of GABA or after block of GABA uptake. Our results suggest that although brief and large GABA transients at the release site lead to efficient channel opening, low concentrations of GABA reduce the availability of synaptic GABAA channels. Because the concentration profile is reduced and prolonged as GABA diffuses away from the release site, slow desensitization may provide local spatial control of inhibition as receptors at the site of release open, whereas those at neighboring locations desensitize.
MATERIALS AND METHODS
Tissue culture recordings. Hippocampal neurons from postnatal day 0–1 Sprague Dawley rats were grown on microdot islands (Bekkers and Stevens, 1991). Whole-cell recordings were made 2–4 weeks after plating using borosilicate glass pipettes containing (in mm): 144 KCl, 1 CaCl2, 3.45 BAPTA, 10 HEPES, 5 Mg2ATP; pH 7.2, 315 mOsm, 2–5 mΩ resistance. In some experiments, 1.1 mm EGTA and 0.1 mm CaCl2 replaced BAPTA and 1 mm CaCl2, which reduced current rundown but had no other noticeable effects. The extracellular solution contained (in mm): 140 NaCl, 2.8 KCl, 1 MgCl2, 1.5 CaCl2, 10 HEPES, 10 d-glucose; pH 7.4, and adjusted to 325 mOsm with sucrose). Strychnine (1 μm) and 6-cyano-7-nitroquinoxaline-2,3-dione (CNQX; 10 μm) were included in the extracellular solution. After the initial experiments to establish the GABAB receptor independence of the effects of uptake inhibition, 2-hydroxysaclofen (200 μm) or CGP55845 (1 μm) were included in the whole-cell extracellular solution to block GABAB receptors. All experiments were performed at room temperature.
Cells and patches were voltage-clamped at −60 mV (Warner PC 501) and perfused continuously with extracellular solutions via local perfusion pipes (Vitro Dynamics, Rockaway, NJ) mounted on a piezoelectric bimorph (Vernitron, Bedford, OH). The recording chamber was also perfused at 1–2 ml/min. Autaptic IPSCs were evoked every 20 sec by application of voltage steps (to 0 mV, 0.2–0.5 msec duration). Alternating control and drug solutions were applied at ∼20 sec intervals throughout the experiment. The whole-cell solution exchange rate was determined by the change in GABA-mediated current after a jump into low NaCl-containing extracellular solution (10–90% rise time <200 msec). Membrane currents were acquired with pClamp6 (Axon Instruments) and analyzed off-line using Axograph 3.6 (Axon Instruments). Currents were sampled at 10 kHz and filtered at 2 kHz using an eight-pole Bessel filter (Frequency Devices, Haverhill, MA). Whole-cell series resistance (<20 mΩ) was monitored by delivering brief voltage steps and was partially compensated (50–80%) in some experiments.
Data are presented as mean ± SEM. Unless noted, statistical significance was determined using a two-tailed paired t test at the p < 0.05 level. All drugs were dissolved in water or DMSO (final concentration <0.3%) and added to the local perfusion solution. CNQX, 2-hydroxysaclofen, and CGP55845 were purchased from Tocris Cookson (Ballwin, MO). 1-(2-(((Diphenylmethylene)imino)oxy)ethyl)-1,2,5,6-tetrahydro-3-pyridine-carboxylic acid (NO-711) and SR95531 were purchased from Research Biochemicals (Natick, MA). Tiagabine was a gift from Abbott Laboratories (North Chicago, IL). GABA, gamma-vinyl GABA, strychnine, and TTX were purchased from Sigma (St. Louis, MO).
Patch current recordings. GABA-evoked patch currents were recorded from outside-out patches pulled from the soma of neurons grown for 2–4 weeks in microdot (Bekkers and Stevens, 1991) or monolayer (Forsythe and Westbrook, 1988) cultures. Rapid application of solutions was accomplished as described previously (Jones et al., 1998). The solution exchange time for patch experiments (<1 msec) was measured after each experiment by monitoring the change in liquid junction current at the pipette tip after patch rupture. Membrane currents were acquired with pClamp6 (Axon Instruments) and analyzed off-line using Axograph 3.6 (Axon Instruments). Currents were filtered at less than half the sampling frequency (250 Hz–10 kHz) using an eight-pole Bessel filter (Frequency Devices). In the GABA preequilibration experiments, a saturating GABA test pulse was used to assess the maximal available patch current (Imax).Imax was proportional to the total number of channels (Ftotal) that were distributed between three “states” during preequilibration: open (Fopen), unbound (Favailable), and desensitized (Fdesensitized), such that Ftotal =Fopen +Favailable +Fdesensitized. GABA preequilibration directly activated a small current (Ilow) that was proportional toFopen. The current produced by a subsequent test pulse (Itest, measured fromIlow) indicated the fractional channel availability (Itest/Imax= Favailable) after the preequilibration. The fraction of desensitized channels (i.e., bound but closed; Fdesensitized) was calculated by Imax − (Itest +Ilow).
Hippocampal slice recordings. Whole-cell recordings were made from granule cells in transverse hippocampal slices (400 μm) from 13- to 16-d-old Sprague Dawley rats. During recordings, slices were continuously perfused with an extracellular solution containing (in mm): 125 NaCl, 25 NaHCO3, 2.5 KCl, 1.25 NaH2PO4, 2 CaCl2, 1 MgCl2, and 25d-glucose, bubbled with 95% O2 and 5% CO2. Patch pipettes were filled with (in mm): 140 KCl, 10 EGTA, 10 HEPES, and 2 Mg2ATP, adjusted to pH 7.3 and 310 mOsm. Visually identified granule cells were voltage-clamped at −60 mV using an Axopatch 200B amplifier (Axon Instruments) and maintained at room temperature or 34 ± 1°C using an in-line heater (Warner Instrument Corp.). Records were filtered at 2 kHz and sampled at 10 kHz. Series resistance was <10 mΩ before compensation (∼80–90%), and data were discarded if substantial increases were observed. Miniature IPSCs (mIPSCs) were isolated by adding 0.5–1 μm TTX, 100 μm AP5, and 5–10 μm CNQX to the extracellular solution. mIPSCs were detected with the template matching procedure of Axograph 4.0 (Axon Instruments), using the sum of one rising (τ = 200 μsec at 34°C or τ = 400 μsec at room temperature) and one falling (τ = 10 msec at 34°C or τ = 20 msec at room temperature) exponential as a template. Detected mIPSCs were extracted and aligned by the time of maximum agreement with the template. Data are presented as average median ± SEM, and statistical significance was determined using a Mann–Whitney Utest.
RESULTS
Low concentrations of GABA desensitize synaptic GABAA receptors
We first tested the effect of a low concentration of exogenous GABA on GABAA receptor-mediated synaptic transmission. In the presence of either 2-hydroxysaclofen (200 μm) or CGP55845 (1 μm) to block GABAB receptors, preequilibration of a hippocampal autapse with GABA (1 μm, 20 sec) reduced the IPSC amplitude to 77 ± 3% of control. GABA also activated an inward current (118 ± 27 pA, n = 11) that quickly returned to baseline within 1.5 sec after washout. However, the peak of the IPSC (75 ± 3% of control at 1.5 sec after removal of GABA,n = 5) (Fig.1a) recovered much more slowly (τ = 8.0 sec) (Fig. 1b). There was a correlation between the peak amplitude of the GABA-activated current and the degree of IPSC depression (r = 0.7, p = 0.026; data not shown). The decay phase of the IPSC was unaffected by the preequilibration (Fig. 1a), indicating that the gating of the underlying channels was unchanged but that fewer channels were activated during the IPSC. The paired-pulse ratio, a common test for a presynaptic site of action [but see Frerking et al. (1999)], was unaffected by GABA preequilibration (0.88 ± 0.06 in control, 0.84 ± 0.04 in GABA, n = 7).
Fig. 1.

Preequilibration with 1 μm GABA reduced the IPSC amplitude. a, Top panel, Schematic diagram of the protocol used to measure the effect of GABA on the IPSC. Autaptic IPSCs were evoked by voltage steps (Vc) applied before GABA application (a), and at intervals from 1.5 to 27.5 sec after solution exchange (b). Bottom panel, Averaged IPSC evoked at 1.5 sec after solution exchange (b) overlaid on the control IPSC (a). GABA reduced the amplitude with no change in the time course. b, Slow recovery of the IPSC after GABA exposure. Each trace is the average of four to seven IPSCs, normalized to the amplitude of the control IPSC. The inward current produced by GABA application is illustrated at the end of the control trace. The time course of recovery of the IPSC amplitude was fit with a single exponential function. Each point represents the mean ± SEM from five cells.
To examine the possibility that postsynaptic GABAA receptor desensitization was responsible for the decrease in the IPSC, we made rapid applications of GABA to outside-out patches using protocols similar to that used for IPSCs. Preequilibration with a low concentration of GABA reduced the current produced by a saturating agonist test pulse (Fig.2a). Fractional availability decreased with increasing duration and concentration of the GABA preequilibration, with a time course similar to macroscopic desensitization (Jones and Westbrook, 1995). The ratio of receptor desensitization/opening (Fdesensitized/Fopen; see Materials and Methods) provides a measure of the preference of bound channels for desensitized or open states. This ratio was >1 for GABA preequilibrations <10 μm regardless of the exposure duration, whereas the highest concentration tested (10 μm) favored channel opening only at the shortest exposure duration (10 msec). These results indicate that low concentrations or long exposure times promote desensitization, whereas only brief and high GABA transients preferentially favor channel opening.
Fig. 2.
Preequilibration with low concentrations of GABA desensitized GABAA receptors. a, Top panel, Schematic diagram of the protocol used to measure desensitization by GABA in outside-out patches. Maximal available GABAA currents were evoked by brief saturating GABA applications (10 mm, 20 msec duration, upward deflection) delivered before and after low concentrations of GABA (0.4–10 μm, 10 msec–20 sec). An example of desensitization produced by preequilibration with 2 μm GABA is illustrated (seven traces overlaid). Each trace is normalized to the peak amplitude of the control current. Bottom panel, Fractional availability declined as a function of preequilibration duration and GABA concentration. Each symbol is the mean ± SEM of currents from three to nine patches. The onset of desensitization was fitted with a single exponential function and a constant (for 0.4 μm GABA) or the sum of two exponential functions and a constant (dotted lines). For 0.4 μm GABA, τ = 3.2 sec (14% of total amplitude). For 1 μmGABA, τfast = 53 msec (6%) and τslow = 3.5 sec. For 2 μm GABA, τfast = 41 msec (15%) and τslow= 2.9 sec. For 5 μm GABA, τfast = 85 msec (50%) and τslow = 2.1 sec. For 10 μm GABA, τfast = 42 msec (57%) and τslow = 2.7 sec. b, Top panel, Schematic diagram of the protocol used to measure recovery from desensitization in outside-out patches. GABA test pulses (10 mm, 20 msec, upward deflection) were delivered at intervals from 0.1 to 34 sec after GABA exposure (1 or 5 μm, 20 sec duration). An example of recovery from exposure to 5 μm GABA is illustrated. Six traces are overlaid, normalized to the peak amplitude of the control current.Bottom panel, The recovery from desensitization produced by 1 μm GABA was best fit by a single exponential, whereas an additional faster component was present at 5 μm. Each symbol is the mean ± SEM from six to eight patches.
The time course of patch current recovery after preequilibration with 5 μm GABA is illustrated in Figure 2b (top panel). Saturating GABA test pulses were applied at intervals from 0.1 to 34 sec after a 20 sec preequilibration. For 5 μm GABA, recovery had two components (τfast = 740 msec, 24%; τslow = 15.8 sec, 76%), whereas the recovery from 1 μm GABA had only a slow component (τ = 11.5 sec, 33% total amplitude), similar to that observed for the IPSC recovery (Fig. 1b). This suggests that after exposure to the lower GABA concentration, desensitized receptors primarily reside in a slow desensitized state, whereas exposure to 5 μm GABA drives ∼25% of the desensitized receptors into a desensitized state with a faster exit rate. Thus the patch results are consistent with the idea that exogenous GABA reduced the IPSC by causing slow desensitization of synaptic GABAA receptors.
Block of GABA transport promotes slow desensitization
GABA transporters remove GABA from the extracellular space. We therefore tested whether inhibition of GABA transport would result in slow desensitization by allowing local accumulation of GABA. Consistent with this hypothesis, the GABA transport inhibitor NO711 (100 μm) reduced IPSCs to 51 ± 3% of control (n = 33) (Fig.3a) but had no effect on autaptic EPSCs (89 ± 6% of control, n = 8; 95 ± 5% of control in 1 μm CGP55845,n = 6). The IPSC reduction was not caused by activation of presynaptic GABAB autoreceptors (Oh and Dichter, 1994) because it was not blocked by 2-hydoxysaclofen (200 μm; 55 ± 5%, n = 7) or CGP55845 (53 ± 6%, n = 7) (Fig. 3a,right panel). To exclude a presynaptic action of NO711, we measured the mean2/variance of the peak IPSC in the absence and presence of NO711 or baclofen. As expected for a purely presynaptic action (Malinow and Tsien, 1990), the mean2/variance in baclofen was reduced proportionally to the mean current, but this was not true for NO711 (Fig. 3b). Similarly, NO711 did not affect the paired-pulse ratio in either the absence (0.78 ± 0.06 vs 0.83 ± 0.10,n = 14) or presence of CGP55845 (0.65 ± 0.09 vs 0.67 ± 0.10, n = 5) (Fig. 3a). In contrast, baclofen (1 μm) reduced the IPSC to 41 ± 9% and increased the paired-pulse ratio (0.91 ± 0.03 vs 1.16 ± 0.1, p = 0.06, n = 7). Neither 2-hydoxysaclofen nor CGP55845 alone altered the amplitude of IPSCs (99 ± 6%, n = 7 and 94 ± 8%,n = 5, respectively) or the paired-pulse ratio, suggesting that there was not appreciable activation of GABAB receptors under control conditions. Furthermore, NO711 reduced the median amplitude of mIPSCs recorded in TTX (by 14.9 ± 4%, paired t test, n = 5). The reduction was only significant in 3 of 5 cells (Mann–WhitneyU test, average event frequency ∼0.8 Hz), possibly because of the very low frequency of events in the remaining two cells (∼0.2 Hz). Taken together, these data indicate that NO711 reduced the IPSC amplitude by a postsynaptic mechanism.
Fig. 3.
The GABA uptake inhibitor NO711 reduced the IPSC amplitude. a, NO711 (100 μm) reduced the IPSC amplitude in the presence of the GABAB receptor antagonists 2-hydroxysaclofen (200 μm) or CGP55845 (1 μm). Asterisks indicate a significant difference between the amplitude of IPSCs in NO711 and the corresponding control. The reduction of IPSC amplitude produced by NO711 was not accompanied by a change in the paired-pulse ratio. In control experiments, 2-hydroxysaclofen (200 μm) attenuated the reduction in IPSC produced by baclofen (1 μm, 41 ± 9% of control in baclofen, 90 ± 9% in baclofen + 2-hydroxysaclofen; n = 7).b, The mean2/variance was reduced proportionally to the mean amplitude for IPSCs recorded in baclofen (1 μm, ▵), but not for IPSCs recorded in NO711 (●, in CGP55845; ♦, in 2-hydroxysaclofen). The mean and mean2/variance were computed for each condition and normalized to control values. The average slope of the lines connecting each point to the control point was 0.06 ± 0.23 for NO711 (thick line) and 1.03 ± 0.07 for baclofen (thin line). The nonoverlapping 95% confidence limits (dotted lines) indicate that the slope for NO711 was not significantly different from zero, suggesting a postsynaptic site of action. c, NO711 (100 μm) reduced the amplitude of patch currents activated by 100 μm GABA, but this direct effect cannot account for the reduction of the IPSC by NO711. Inset, The recovery from the direct action of NO711 on GABA-activated patch currents was very rapid (τ = 56 msec; n = 2–10).
NO711 had no effect on GABAA receptor-mediated currents activated by a concentration of GABA similar to that in the synaptic cleft (1 mm) (Jones and Westbrook, 1995; Mozrzymas et al., 1999). However, NO711 did reduce currents activated by a lower concentration of GABA (100 μm) in patches from cultured neurons (to 79.8 ± 2.3% of control,n = 4) (Fig. 3c). This direct action on GABAA receptors could not account for the reduction of the IPSC by NO711 because the magnitude was small and the recovery was fast. For example, at 100 msec after removal of NO711, the amplitude of GABA-activated patch currents had already returned to control levels (98 ± 2%, n = 9) (Fig.3c, inset). Another uptake inhibitor, tiagabine (100 μm), also had a direct effect on patch currents activated by 100 μm GABA (reduced to 77 ± 7% of control, n = 4). Neither uptake inhibitor altered the decay time course of 5 or 500 msec GABA-activated currents.
The recovery time course of the IPSC after removal of NO711 was slow (τ = 10.0 sec) (Fig.4a), similar to the recovery of GABA-activated patch currents after desensitization (Fig.2b) and the recovery of IPSCs after GABA application (Fig.1b). Tiagabine (100 μm) also reduced IPSCs in 11 of 13 cells (to 62 ± 8% of control,n = 13), and demonstrated a slow recovery time course (Fig. 4c). Both uptake inhibitors prolonged the decay of the IPSC (111 ± 2% of control for NO711; 140 ± 7% of control for tiagabine) (Fig. 4b). The effect of tiagabine on the IPSC amplitude recovered with a much slower time course (τ = 13.3 sec) than did the effect on IPSC decay (τ = 1.3 sec) (Fig.4c), indicating that they were mediated by separate mechanisms. Similarly, the prolongation of the IPSC decay produced by NO711 recovered by 1.5 sec, whereas the amplitude was still significantly reduced at 7.5 sec (data not shown). The fast recovery of the IPSC decay implies that transporter inhibitors dissociated quickly, whereas the slow recovery of the IPSC amplitude reflects the kinetics of recovery from slow desensitization.
Fig. 4.

Recovery of the IPSC after inhibition of uptake.a, Recovery of the IPSC amplitude after NO711 was fit with a single exponential function (same protocol as Fig. 1). Recovery from NO711 (τ = 10.0 sec, n = 4–7 cells per time point) was similar to recovery from GABA (Figs. 1,2b). Recovery of the IPSC decay was much faster (see Results). b, Tiagabine (100 μm) reduced the amplitude and prolonged the decay of IPSCs. The decay of the IPSC was fit with the sum of either two or three exponential functions, and the weighted decay was calculated by the equationA1τ1 +A2τ2 +A3τ3, whereA is the relative amplitude of each component and τ is its time constant. Inset, Both uptake inhibitors prolonged the weighted decay. For NO711, τ = 46 ± 7 msec in control and 50 ± 7 msec in NO711 (n = 7). For tiagabine, τ = 57 ± 9 msec in control and 79 ± 13 msec in tiagabine (n = 8). The uptake inhibitors increased the amplitude of the slow component of decay without altering the time constants. c, The recovery of the IPSC decay to control values after removal of tiagabine (●, n = 4–9 cells per time point) was faster than the recovery of the peak amplitude (○, n = 3–6 cells per time point) in the same population of cells. The recovery of the amplitude and decay were fit with single exponential functions (τamplitude = 13.3 sec, τdecay = 1.3 sec).
If the reduction in the IPSC amplitude by NO711 is caused by increases in ambient GABA with subsequent receptor desensitization, it should be possible to reduce this effect by reducing the total time each receptor spends in the GABA-bound state (Celentano and Wong, 1994; Jones and Westbrook, 1995). We used a competitive antagonist to test this possibility. At equilibrium, a competitive antagonist is continually binding and unbinding such that the total time each receptor spends bound by the accumulating GABA will be reduced by competition with the antagonist. We added the high-affinity competitive GABAA receptor antagonist SR95531 at a concentration near its IC50 (200 nm) to the control and the NO711 solutions. For each cell tested, NO711 had a smaller effect on the IPSC in the presence of SR95531 (to 80 ± 4% in SR95531 vs 56 ± 2% in control, n = 4) (Fig. 5b). The simplest explanation for this result is that SR95531 reduced the occupancy of synaptic receptors by GABA, thereby decreasing the fraction of receptors that were desensitized by GABA, although more complicated scenarios are possible. Together these data indicate that NO711 caused an accumulation of endogenous GABA, thus promoting slow desensitization of synaptic GABAA receptors.
Fig. 5.
A competitive antagonist attenuated the effect of NO711. An example of the effect of SR95531 on the reduction of the IPSC amplitude produced by NO711. Top panel, NO711 (100 μm) reduced the IPSC by 44% under control conditions.Bottom panel, In the presence of SR95531 (200 nm) the IPSC was 56 ± 9% of control, and NO711 reduced the amplitude further by only 23% (same cell as top panel). SR95531 significantly attenuated the reduction of the IPSC produced by NO711 (n = 4). In each cell the effect of NO711 was measured in the presence and absence of SR95531. SR95531 had no effect on the paired-pulse ratio.
Slow desensitization of GABAA receptors provides a tool to estimate the average concentration of GABA in the synaptic cleft after inhibition of GABA transport. As shown in Figure6a, the fractional availability of patch receptors after a 20 sec GABA preequilibration was replotted as a function of GABA concentration. The reduction of the IPSC produced by uptake inhibitors, measured 1.5 sec after removal, predicts an average cleft GABA concentration of ∼1 μm. This value may be an underestimate if NO711 is acting as a very weak competitive antagonist, thus protecting receptors from desensitization in the same manner as SR95531. Furthermore, it is likely that 1 μm GABA produced a slightly greater reduction in patch currents compared with synaptic currents because transporter activity maintained the synaptic GABA concentration below 1 μm during the whole-cell GABA application (Zorumski et al., 1996). Interestingly, exogenous application of 1 μm GABA produced a robust inward current in all cells tested (∼100 pA), whereas NO711 produced a significant current in only 2 of 9 cells examined (∼10 pA,n = 2) (Fig. 6b). Tiagabine produced an inward current of ∼10 pA in 3 of 13 cells, such that the average current across all cells was 3.6 ± 1.5 pA (n = 13). The weak antagonist action of the uptake inhibitors did not completely mask a whole-cell current, because NO711 only blocked the current activated by 1 μm GABA by 42 ± 9% (n = 4). The lack of a robust current in uptake inhibitors suggests that exogenous GABA activated channels over the entire cell surface, whereas the GABA accumulation produced by uptake inhibitors was limited to the synapse.
Fig. 6.

Uptake inhibitors increased GABA in the synaptic cleft. a, The fractional availability of patch receptors measured after 20 sec of GABA preequilibration and the peak current activated during preequilibration were plotted against the concentration of GABA. These data were fit with the Hill equation (dotted lines, n = 1.3 for both fits) and predict the reduction in IPSC amplitude caused by application of 1 μm GABA to the synapse. The reduction in IPSC amplitude after NO711 was consistent with desensitization induced by elevation of GABA to an average concentration of ∼1 μmat the synapse. b, Whole-cell application of GABA (1 μm, top trace) produced an inward current, but NO711 did not (100 μm, bottom trace). However, both reduced the IPSC amplitude to a similar extent after a 20 sec equilibration (test IPSCs were evoked 1.5 sec after GABA or NO711 washout). Thus, the NO711-induced elevation in GABA appears to be confined to the synapse.
Block of GABA transport leads to desensitization in hippocampal slices
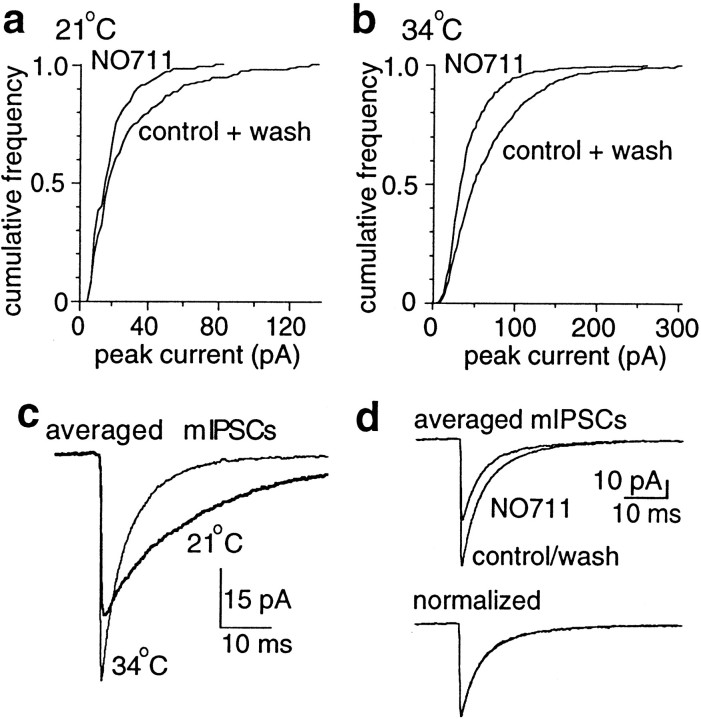
Accumulation of GABA at synapses is likely to depend on many factors, including synaptic geometry, the location, density and kinetics of transporters, diffusional barriers, and temperature. Therefore, to determine the generality of the results obtained at cultured synapses, we tested the effect of transport inhibition on synaptic currents recorded in dentate granule cells of hippocampal slices at two temperatures. We measured mIPSC amplitudes because they should be proportional to postsynaptic receptor availability without contamination by presynaptic effects produced by GABA accumulation. Bath application of NO711 reduced mIPSC amplitudes in every cell tested (Mann–Whitney U test) (Fig.7), both at room temperature (19–23°C; average median amplitude 22 ± 3 pA in control vs 15 ± 2 pA in NO711; n = 5) and near physiological temperature (34°C, average median amplitude 45 ± 4 pA vs 26 ± 3 pA;n = 3). Consistent with an accumulation of GABA, NO711 produced an increase in the mean holding current (12 ± 4 pA,n = 6) that was prevented by 5 μm SR95531 (3 ± 2 pA, n = 4) and enhanced by slice pretreatment with the GABA transaminase inhibitor, γ-vinyl GABA (400 μm, 56 ± 15 pA, n = 6). As expected, mIPSC amplitudes were larger, and rise and decay times were significantly faster at 34°C (n = 5) (Fig. 7c). At room temperature, NO711 reduced the mIPSC frequency in 4 of 5 cells (to 79 ± 18% of control). This was predicted to occur as some of the smallest events became undetectable (Zhou et al., 2000). At 34°C, NO711 reduced the mIPSC frequency in 2 of 3 cells (to 84 ± 7% of control). GABAB receptor antagonists were not present in the experiments at 34°C, hence this reduction may reflect activation of presynaptic GABAB receptors. NO711 did not prolong the decay of the ensemble average mIPSC at either temperature (Fig. 7d), consistent with previous findings that uptake inhibitors prolong large evoked responses but not miniature events (Thompson and Gähwiler, 1992; Isaacson et al., 1993). The direct effect of NO711 on GABA-evoked currents from granule cell patches was not different from the direct effect on patches from cultured neurons (to 73 ± 8% of control, n = 5, 100 μm GABA), but NO711 had no effect on patch currents activated by 1 mm GABA (100 ± 10%, n = 6). These findings indicate that the direct effect of NO711 is unlikely to contribute significantly to the reduction of mIPSCs. Rather, these results suggest that inhibition of GABA transporters in slices can also lead to an accumulation of GABA and subsequent desensitization of synaptic GABAAreceptors.
Fig. 7.
Block of GABA transport in hippocampal slices reduced mIPSCs.a, b, The amplitude of mIPSCs recorded in dentate granule cells was reduced by NO711 (100 μm) at room temperature and at 34°C. Data from individual experiments are shown, with events from the control and wash periods combined. c, Averaged mIPSCs recorded in the same cell at room temperature and 34°C. In five cells, increasing the temperature increased the median frequency of events (3.2 ± 0.6 Hz at 34°C vs 0.8 ± 0.2 Hz at room temperature) and the median amplitude (35 ± 5 pA vs 24 ± 2 pA) and reduced the median 20–80% rise time (0.28 ± 0.04 msec vs 0.58 ± 0.04 msec) and weighted decay time constant (8.5 ± 0.8 msec vs 25.7 ± 2.3 msec). d, NO711 had no effect on the time course of decay of the ensemble average mIPSC at either temperature.
DISCUSSION
Ligand-gated channels desensitize in response to micromolar concentrations of agonist (Trussell and Fischbach, 1989; Colquhoun et al., 1992; Sather et al., 1992; Lester and Dani, 1995; Zorumski et al., 1996; Berger et al., 1998) that are near the extracellular concentrations measured in vivo (Lerma et al., 1986). For excitatory synapses, increases in ambient glutamate can modulate synaptic transmission (Trussell and Fischbach, 1989; Zorumski et al., 1996), as can delayed clearance of synaptically released glutamate at synapses with specialized morphology (Otis et al., 1996a,b; Kinney et al., 1997). In our experiments, the concentration of GABA at hippocampal inhibitory synapses rose into the micromolar range when uptake was blocked, sufficient to reduce the IPSC by driving GABAA channels into a slow desensitized state. This postsynaptic mechanism for reduced inhibition therefore operates over a range of ambient GABA concentrations similar to that of the more commonly studied presynaptic GABABreceptor-mediated depression (Yoon and Rothman, 1991; Sodickson and Bean, 1996). However, recovery from slow desensitization is an order of magnitude slower than the GABAB receptor-mediated action (Davies et al., 1990; Isaacson et al., 1993; Sodickson and Bean, 1996; Dittmann and Regehr, 1997), thus providing a different temporal profile for the regulation of inhibition.
Multiple effects of GABA transport inhibition
GABA uptake is susceptible to both physiological and pharmacological regulation (Corey et al., 1994; Quick et al., 1997;Berstein and Quick, 1999). Previous studies have often emphasized the prolongation of the evoked IPSC/P decay as the primary consequence of transport inhibition (Dingledine and Korn, 1985; Roepstorff and Lambert, 1992, 1994; Thompson and Gähwiler, 1992; Draguhn and Heinemann, 1996). However, reductions in the evoked IPSC amplitude produced by uptake inhibitors have also been noted (Dingledine and Korn, 1985; Deisz and Prince, 1989; Roepstorff and Lambert, 1992; Oh and Dichter, 1994; Draguhn and Heinemann, 1996). Elevated GABA levels can activate presynaptic GABAB receptors, thereby reducing transmitter release (Deisz and Prince, 1989; Yoon and Rothman, 1991; Isaacson et al., 1993; Oh and Dichter, 1994; Scanziani, 2000). In our experiments, however, GABAB receptor antagonists had no effect, whereas uptake blockers mimicked the postsynaptic desensitizing action of GABA. Therefore, slight elevations of GABA may modulate inhibitory transmission via both presynaptic and postsynaptic mechanisms. The relative contribution of presynaptic and postsynaptic mechanisms is likely influenced by the spatial extent of GABA accumulation, because GABAB receptors may be localized at extrasynaptic locations (Fritschy et al., 1999). The extent of GABA accumulation after uptake inhibition is presumably determined at least in part by the intactness of the preparation, consistent with our finding that NO711 produced more robust currents in slices compared with microdot cultures. We also noted that tiagabine produced a more variable reduction but slightly larger prolongation of the evoked IPSC than NO711, whereas both had similarly small effects on isolated GABAA receptors. The reason for these differences remains unclear.
Diethyl-lactam, another experimental anticonvulsant, prolongs the IPSC decay, although it only potentiates responses to low concentrations of GABA (<30 μm), leading to the conclusion that subsaturating GABA concentrations contribute to the generation of IPSCs (Hill et al., 1998). Our results indicate that such a subsaturating component of the GABA transient will promote channel desensitization. This further suggests that the prolongation of evoked synaptic currents produced by uptake inhibitors may be the result of enhancing the amplitude of a slow component of the GABA transient from desensitizing levels into a range that shifts the balance from desensitization toward channel opening. The lack of effect of uptake inhibitors on the decay of mIPSCs (Thompson and Gähwiler, 1992; this study) and small evoked IPSCs (Isaacson et al., 1993) implies that uptake blockers elevate the putative slow component of the GABA transient by allowing interactions between GABA released from multiple sites (Roepstorff and Lambert, 1994; Scanziani, 2000).
Our results suggest that tonic GABA accumulation during uptake blockade in culture is limited to the synapse itself, although the source of the GABA is unknown. One possibility is that spontaneously released vesicles (mIPSCs) contribute to the tonic level of GABA (Brickley et al., 1996; Wall and Usowicz, 1997; Leao et al., 2000). However, under our recording conditions the mIPSC frequency was low (<1 Hz), and there was no obvious correlation between mIPSC frequency and the efficacy of NO711. Another possible source is reversal of GABA transport (Schwartz, 1987; Attwell et al., 1993). Although NO711 and tiagabine are not reported to be transport substrates, even a minimal amount of heteroexchange (Solis and Nicoll, 1992; Gaspary et al., 1998) would be sufficient to generate a cleft concentration of 1 μm GABA. This concentration is equivalent to only a few GABA molecules in the small volume of the synaptic cleft. A third source may be nonvesicular release that is independent of transporters (Wall and Usowicz, 1997). Such release may result from the efflux of GABA (or another GABAA receptor agonist such as taurine) via volume-sensitive osmolyte/anion channels (for review, seeStrange et al., 1996). Thus, our results may be analogous to the discovery that blockade of glutamate uptake with a nonsubstrate inhibitor results in accumulation of extracellular glutamate of nonvesicular origin (Jabaudon et al., 1999).
Distinct functions of fast and slow desensitization
A direct experimental demonstration of the role of desensitization in neural processing is still lacking. However, there is now sufficiently detailed information about fast and slow desensitization kinetics to make testable predictions. We expect that slow desensitization of synaptic GABAA receptors (Dslow) has computational implications that are distinct from those of fast desensitization (Dfast). First, large and fast GABA transients that occur during vesicular release activate receptors with high efficiency and tend to promote entry intoDfast. Conversely,Dslow is particularly well suited to respond to small and slow transients because it is a strongly absorbing, and possibly monoliganded, state (Jones and Westbrook, 1995). Slow transients are thus relatively ineffective at activating current but rather reduce the ability of the synapse to respond to subsequent stimuli. The presence of two desensitization mechanisms with distinct concentration and time course selectivities suggests that information carried by fluctuations in the GABA concentration may be transduced into distinct behaviors of the receptor, depending on their frequency content. For example, slow desensitization may serve as a high-pass temporal filter with respect to the synaptic GABA concentration time course, and also as a slow negative feedback adjustment of synaptic gain. In contrast, when synapses are repeatedly activated, accumulation in fast desensitized states may reduce the amplitude of subsequent IPSCs, thus acting as a low-pass filter with respect to IPSC frequency. Fast desensitization also acts as a low-pass filter on the shape of each response by virtue of its ability to prolong GABAA receptor deactivation (Jones and Westbrook, 1995).
In addition to a high-pass function in the time domain, slow desensitization is likely to confer a high-pass character in the spatial domain as well. GABA can diffuse between inhibitory synapses to activate GABAA receptors (Rossi and Hamann, 1998) and to extrasynaptic sites to activate GABABreceptors (Scanziani, 2000). GABA can also diffuse to excitatory terminals where it activates presynaptic GABABreceptors (Isaacson et al., 1993; Dittmann and Regehr, 1997). The amplitude and time course of GABA cross-talk will depend on many factors, including the distance between GABAergic synapses and the frequency of nearby release events. However, during intersynaptic diffusion, the GABA transient reaching distant sites will be small and slowly rising (Barbour and Häusser, 1997; Bergles et al., 1999;Rusakov and Kullmann, 1999), which our data suggest will result predominantly in slow desensitization, rather than activation, of GABAA receptors at neighboring synapses. One might therefore expect intersynaptic communication mediated by GABA to reduce the efficiency of neighboring synapses, whether inhibitory or excitatory, as a microscopic analog of the well known macroscopic “surround inhibition” phenomenon (Rodieck, 1965).
Footnotes
This work was supported by National Institutes of Health Grants RO1 NS26494 (G.L.W.) and T32 DA07262 (L.S.O.) and a grant from the Human Frontiers Science Program Organization (G.L.W.). We thank Dr. Pascale Chavis for technical advice and Dr. Laurence Trussell for comments on this manuscript.
Correspondence should be addressed to Linda S. Overstreet, Vollum Institute, Oregon Health Sciences University, L474, 3181 SW Sam Jackson Park Road, Portland, OR 97201. E-mail:overstre@ohsu.edu.
REFERENCES
- 1.Asztely F, Erdemli G, Kullmann DM. Extrasynaptic glutamate spillover in the hippocampus: dependence on temperature and the role of active glutamate uptake. Neuron. 1997;18:281–293. doi: 10.1016/s0896-6273(00)80268-8. [DOI] [PubMed] [Google Scholar]
- 2.Attwell D, Barbour B, Szatkowski M. Nonvesicular release of neurotransmitter. Neuron. 1993;11:401–407. doi: 10.1016/0896-6273(93)90145-h. [DOI] [PubMed] [Google Scholar]
- 3.Barbour B, Häusser M. Intersynaptic diffusion of neurotransmitter. Trends Neurosci. 1997;20:377–384. doi: 10.1016/s0166-2236(96)20050-5. [DOI] [PubMed] [Google Scholar]
- 4.Bekkers JM, Stevens CF. Excitatory and inhibitory autaptic currents in isolated hippocampal neurons maintained in cell culture. Proc Natl Acad Sci USA. 1991;88:7834–7838. doi: 10.1073/pnas.88.17.7834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Berger T, Schwarz C, Kraushaar U, Monyer H. Dentate gyrus basket cell GABAA receptors are blocked by Zn2+ via changes of their desensitization kinetics: an in situ patch-clamp and single-cell PCR study. J Neurosci. 1998;18:2437–2448. doi: 10.1523/JNEUROSCI.18-07-02437.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bergles DE, Jahr CE. Synaptic activation of glutamate transporters in hippocampal astrocytes. Neuron. 1997;19:1297–1308. doi: 10.1016/s0896-6273(00)80420-1. [DOI] [PubMed] [Google Scholar]
- 7.Bergles DE, Jahr CE. Glial contribution to glutamate uptake at Schaffer collateral–commissural synapses in the hippocampus. J Neurosci. 1998;18:7709–7716. doi: 10.1523/JNEUROSCI.18-19-07709.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bergles DE, Dzubay JA, Jahr CE. Glutamate transporter currents in Bergmann glial cells follow the time course of extrasynaptic glutamate. Proc Natl Acad Sci USA. 1997;94:14821–14825. doi: 10.1073/pnas.94.26.14821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bergles DE, Diamond JS, Jahr CE. Clearance of glutamate inside the synapse and beyond. Curr Opin Neurobiol. 1999;9:293–298. doi: 10.1016/s0959-4388(99)80043-9. [DOI] [PubMed] [Google Scholar]
- 10.Berstein EM, Quick MW. Regulation of gamma-aminobutyric acid (GABA) transporters by extracellular GABA. J Biol Chem. 1999;274:889–895. doi: 10.1074/jbc.274.2.889. [DOI] [PubMed] [Google Scholar]
- 11.Brickley SG, Cull-Candy SG, Farrant M. Development of a tonic form of synaptic inhibition in rat cerebellar granule cells resulting from persistent activation of GABAA receptors. J Physiol (Lond) 1996;497:753–759. doi: 10.1113/jphysiol.1996.sp021806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Celentano JJ, Wong RKS. Multiphasic desensitization of the GABAA receptor in outside-out patches. Biophys J. 1994;66:1039–1050. doi: 10.1016/S0006-3495(94)80885-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Clements JD. Transmitter timecourse in the synaptic cleft: its role in central synaptic function. Trends Neurosci. 1996;19:163–171. doi: 10.1016/s0166-2236(96)10024-2. [DOI] [PubMed] [Google Scholar]
- 14.Clements JD, Lester RA, Tong G, Jahr CE, Westbrook GL. The time course of glutamate in the synaptic cleft. Science. 1992;258:1498–1501. doi: 10.1126/science.1359647. [DOI] [PubMed] [Google Scholar]
- 15.Colquhoun D, Jonas P, Sakmann B. Action of brief pulses of glutamate on AMPA/kainate receptors in patches from different neurons of rat hippocampal slices. J Physiol (Lond) 1992;458:261–287. doi: 10.1113/jphysiol.1992.sp019417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Corey JL, Davidson N, Lester HA, Brecha N, Quick MW. Protein kinase C modulates the activity of a cloned gamma-aminobutyric acid transporter expressed in Xenopus oocytes via regulated subcellular redistribution of the transporter. J Biol Chem. 1994;269:14759–14769. [PubMed] [Google Scholar]
- 17.Davies CH, Davies SN, Collingridge GL. Paired-pulse depression of monosynaptic GABA-mediated inhibitory postsynaptic responses in rat hippocampus. J Physiol (Lond) 1990;424:513–531. doi: 10.1113/jphysiol.1990.sp018080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Deisz RA, Prince DA. Frequency-dependent depression of inhibition in guinea-pig neocortex in vitro by GABAB receptor feed-back on GABA release. J Physiol (Lond) 1989;412:513–541. doi: 10.1113/jphysiol.1989.sp017629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dingledine R, Korn SJ. γ-aminobutyric acid uptake and the termination of inhibitory synaptic potentials in the rat hippocampal slice. J Physiol (Lond) 1985;366:387–409. doi: 10.1113/jphysiol.1985.sp015804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Dittmann JS, Regehr WG. Mechanism and kinetics of heterosynaptic depression at a cerebellar synapse. J Neurosci. 1997;17:9048–9059. doi: 10.1523/JNEUROSCI.17-23-09048.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Draguhn A, Heinemann U. Different mechanisms regulate IPSC kinetics in early postnatal and juvenile hippocampal granule cells. J Neurophysiol. 1996;76:3983–3993. doi: 10.1152/jn.1996.76.6.3983. [DOI] [PubMed] [Google Scholar]
- 22.Forsythe ID, Westbrook GL. Slow excitatory postsynaptic currents mediated by N-methyl-d-aspartate receptors on cultured mouse central neurones. J Physiol (Lond) 1988;396:515–533. doi: 10.1113/jphysiol.1988.sp016975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Frerking M, Petersen CCH, Nicoll RA. Mechanisms underlying kainate receptor-mediated disinhibition in the hippocampus. Proc Natl Acad Sci USA. 1999;96:12917–12922. doi: 10.1073/pnas.96.22.12917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fritschy JM, Meskenaite V, Weinmann O, Honer M, Benke D, Mohler H. GABAB-receptor splice variants GB1a and GB1b in rat brain: developmental regulation, cellular distribution and extrasynaptic localization. Eur J Neurosci. 1999;11:761–768. doi: 10.1046/j.1460-9568.1999.00481.x. [DOI] [PubMed] [Google Scholar]
- 25.Galarreta M, Hestrin S. Properties of GABAA receptors underlying inhibitory synaptic currents in neocortical pyramidal neurons. J Neurosci. 1997;17:7220–7227. doi: 10.1523/JNEUROSCI.17-19-07220.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gaspary HL, Wang W, Richerson GB. Carrier-mediated GABA release activates GABA receptors on hippocampal neurons. J Neurophysiol. 1998;80:270–281. doi: 10.1152/jn.1998.80.1.270. [DOI] [PubMed] [Google Scholar]
- 27.Hill MW, Reddy PA, Covey DF, Rothman SM. Contribution of subsaturating GABA concentrations to IPSCs in cultured hippocampal neurons. J Neurosci. 1998;18:5103–5111. doi: 10.1523/JNEUROSCI.18-14-05103.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Isaacson JS, Solis JM, Nicoll RA. Local and diffuse synaptic actions of GABA in the hippocampus. Neuron. 1993;10:165–175. doi: 10.1016/0896-6273(93)90308-e. [DOI] [PubMed] [Google Scholar]
- 29.Jabaudon D, Shimamoto K, Yasuda-Kamatani Y, Scanziani M, Gähwiler BH, Gerber U. Inhibition of uptake unmasks rapid extracellular turnover of glutamate of nonvesicular origin. Proc Natl Acad Sci USA. 1999;96:8733–8738. doi: 10.1073/pnas.96.15.8733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jones MV, Westbrook GL. Desensitized states prolong GABAA channel responses to brief agonist pulses. Neuron. 1995;15:181–191. doi: 10.1016/0896-6273(95)90075-6. [DOI] [PubMed] [Google Scholar]
- 31.Jones MV, Westbrook GL. Shaping of IPSCs by endogenous calcineurin activity. J Neurosci. 1997;17:7626–7633. doi: 10.1523/JNEUROSCI.17-20-07626.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jones MV, Sahara Y, Dzubay JA, Westbrook GL. Defining affinity with the GABAA receptor. J Neurosci. 1998;18:8590–8604. doi: 10.1523/JNEUROSCI.18-21-08590.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kinney GA, Overstreet LS, Slater NT. Prolonged physiological entrapment of glutamate in the synaptic cleft of cerebellar unipolar brush cells. J Neurophysiol. 1997;78:1320–1333. doi: 10.1152/jn.1997.78.3.1320. [DOI] [PubMed] [Google Scholar]
- 34.Leao RM, Mellor JR, Randall AD. Tonic benzodiazepine-sensitive GABAergic inhibition in cultured rodent cerebellar granule cells. Neuropharmacology. 2000;39:990–1003. doi: 10.1016/s0028-3908(99)00177-x. [DOI] [PubMed] [Google Scholar]
- 35.Lerma J, Herranz AS, Herreras O, Abraira V, Martin Del Rio R. In vivo determination of extracellular concentration of amino acids in the rat hippocampus. A method based on brain dialysis and computerized analysis. Brain Res. 1986;384:145–155. doi: 10.1016/0006-8993(86)91230-8. [DOI] [PubMed] [Google Scholar]
- 36.Lester RA, Dani JA. Acetylcholine receptor desensitization induced by nicotine in rat medial habenula neurons. J Neurophysiol. 1995;74:195–206. doi: 10.1152/jn.1995.74.1.195. [DOI] [PubMed] [Google Scholar]
- 37.Maconochie DJ, Zempel JM, Steinbach JH. How quickly can GABAA receptors open? Neuron. 1994;12:61–71. doi: 10.1016/0896-6273(94)90152-x. [DOI] [PubMed] [Google Scholar]
- 38.Malinow R, Tsien RW. Presynaptic enhancement shown by whole-cell recordings of long-term potentiation in hippocampal slices. Nature. 1990;346:177–180. doi: 10.1038/346177a0. [DOI] [PubMed] [Google Scholar]
- 39.Mellor JR, Randall AD. Frequency-dependent actions of benzodiazepines on GABAA receptors in cultured murine cerebellar granule cells. J Physiol (Lond) 1997;503:353–369. doi: 10.1111/j.1469-7793.1997.353bh.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mozrzymas JW, Barberis A, Michalak K, Cherubini E. Chlorpromazine inhibits miniature GABAergic currents by reducing the binding and by increasing the unbinding rate of GABAA receptors. J Neurosci. 1999;19:2474–2488. doi: 10.1523/JNEUROSCI.19-07-02474.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Oh DJ, Dichter MA. Effect of a gamma-aminobutyric acid uptake inhibitor, NNC-711, on spontaneous postsynaptic currents in cultured rat hippocampal neurons: implications for antiepileptic drug development. Epilepsia. 1994;35:426–430. doi: 10.1111/j.1528-1157.1994.tb02455.x. [DOI] [PubMed] [Google Scholar]
- 42.Orser BA, Wang L-Y, Pennefather PS, MacDonald JF. Propofol modulates activation and desensitization of GABAA receptors in cultured murine hippocampal neurons. J Neurosci. 1994;14:7747–7760. doi: 10.1523/JNEUROSCI.14-12-07747.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Otis TS, Wu Y-C, Trussell LO. Delayed clearance of transmitter and the role of glutamate transporters at synapses with multiple release sites. J Neurosci. 1996a;16:1634–1644. doi: 10.1523/JNEUROSCI.16-05-01634.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Otis TS, Zhang S, Trussell LO. Direct measurement of AMPA receptor desensitization induced by glutamatergic synaptic transmission. J Neurosci. 1996b;16:7496–7504. doi: 10.1523/JNEUROSCI.16-23-07496.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Quick MW, Corey JL, Davidson N, Lester HA. Second messengers, trafficking-related proteins, and amino acid residues that contribute to the functional regulation of the rat brain GABA transporter GAT1. J Neurosci. 1997;17:2967–2979. doi: 10.1523/JNEUROSCI.17-09-02967.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rodieck RW. Quantitative analysis of cat retinal ganglion cell response to visual stimuli. Vision Res. 1965;5:583–601. doi: 10.1016/0042-6989(65)90033-7. [DOI] [PubMed] [Google Scholar]
- 47.Roepstorff A, Lambert JDC. Comparison of the effect of the GABA uptake blockers, tiagabine and nipecotic acid, on inhibitory synaptic efficacy in hippocampal CA1 neurones. Neurosci Lett. 1992;146:131–134. doi: 10.1016/0304-3940(92)90060-k. [DOI] [PubMed] [Google Scholar]
- 48.Roepstorff A, Lambert JDC. Factors contributing to the decay of the stimulus-evoked IPSC in rat hippocampal CA1 neurons. J Neurophysiol. 1994;72:2911–2926. doi: 10.1152/jn.1994.72.6.2911. [DOI] [PubMed] [Google Scholar]
- 49.Rossi DJ, Hamann M. Spillover-mediated transmission at inhibitory synapses promoted by high affinity alpha6 subunit GABA(A) receptors and glomerular geometry. Neuron. 1998;20:783–795. doi: 10.1016/s0896-6273(00)81016-8. [DOI] [PubMed] [Google Scholar]
- 50.Rusakov DA, Kullmann DM, Stewart MG. Hippocampal synapses: do they talk to their neighbors? Trends Neurosci. 1999;22:382–388. doi: 10.1016/s0166-2236(99)01425-3. [DOI] [PubMed] [Google Scholar]
- 51.Sather W, Dieudonné S, MacDonald JF, Ascher P. Activation and desensitization of N-methyl-d-aspartate receptors in nucleated outside-out patches from mouse neurones. J Physiol (Lond) 1992;450:643–672. doi: 10.1113/jphysiol.1992.sp019148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Scanziani M. GABA spillover activates postsynaptic GABAB receptors to control rhythmic hippocampal activity. Neuron. 2000;25:673–681. doi: 10.1016/s0896-6273(00)81069-7. [DOI] [PubMed] [Google Scholar]
- 53.Scanziani M, Salin PA, Bogt KE, Malenka RC, Nicoll RA. Use-dependent increases in glutamate concentration activate presynaptic metabotropic glutamate receptors. Nature. 1997;385:630–634. doi: 10.1038/385630a0. [DOI] [PubMed] [Google Scholar]
- 54.Schwartz EA. Depolarization without calcium can release gamma-aminobutyric acid from a retinal neuron. Science. 1987;238:350–355. doi: 10.1126/science.2443977. [DOI] [PubMed] [Google Scholar]
- 55.Sodickson DL, Bean BP. GABAB receptor-activated inwardly rectifying potassium current in dissociated hippocampal CA3 neurons. J Neurosci. 1996;16:6374–6385. doi: 10.1523/JNEUROSCI.16-20-06374.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Solis J, Nicoll RA. Postsynaptic action of endogenous GABA released by nipecotic acid in the hippocampus. Neurosci Lett. 1992;147:16–20. doi: 10.1016/0304-3940(92)90764-x. [DOI] [PubMed] [Google Scholar]
- 57.Strange K, Emma F, Jackson PS. Cellular and molecular physiology of volume-sensitive anion channels. Am J Physiol. 1996;270:C372–381. doi: 10.1152/ajpcell.1996.270.3.C711. [DOI] [PubMed] [Google Scholar]
- 58.Thompson SM, Gähwiler BH. Effects of the GABA uptake inhibitor tiagabine on inhibitory synaptic potentials in rat hippocampal slice cultures. J Neurophysiol. 1992;67:1698–1701. doi: 10.1152/jn.1992.67.6.1698. [DOI] [PubMed] [Google Scholar]
- 59.Trussell LO, Fischbach GD. Glutamate receptor desensitization and its role in synaptic transmission. Neuron. 1989;3:209–218. doi: 10.1016/0896-6273(89)90034-2. [DOI] [PubMed] [Google Scholar]
- 60.Turecek R, Trussell LO. Control of synaptic depression by glutamate transporters. J Neurosci. 2000;20:2054–2063. doi: 10.1523/JNEUROSCI.20-05-02054.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wall MJ, Usowicz MM. Development of action potential-dependent and independent spontaneous GABAA receptor-mediated currents in granule cells of postnatal rat cerebellum. Eur J Neurosci. 1997;9:533–548. doi: 10.1111/j.1460-9568.1997.tb01630.x. [DOI] [PubMed] [Google Scholar]
- 62.Yoon K-W, Rothman SM. The modulation of rat hippocampal synaptic conductances by baclofen and γ-aminobutyric acid. J Physiol (Lond) 1991;442:377–390. doi: 10.1113/jphysiol.1991.sp018798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zhou Q, Petersen CCH, Nicoll RA. Effects of reduced vesicular filling on synaptic transmission in rat hippocampal neurons. J Physiol (Lond) 2000;525:195–206. doi: 10.1111/j.1469-7793.2000.t01-1-00195.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zhu WJ, Vicini S. Neurosteroid prolongs GABAA channel deactivation by altering kinetics of desensitized states. J Neurosci. 1997;17:4022–4031. doi: 10.1523/JNEUROSCI.17-11-04022.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Zhu WJ, Wang JF, Corsi L, Vicini S. Lanthanum-mediated modification of GABAA receptor deactivation, desensitization and inhibitory synaptic currents in rat cerebellar neurons. J Physiol (Lond) 1998;511:647–661. doi: 10.1111/j.1469-7793.1998.647bg.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zorumski CF, Mennerick S, Qui J. Modulation of excitatory synaptic transmission by low concentrations of glutamate in cultured rat hippocampal neurons. J Physiol (Lond) 1996;494:465–477. doi: 10.1113/jphysiol.1996.sp021506. [DOI] [PMC free article] [PubMed] [Google Scholar]