Abstract
Objective:
To assess the impact of adjuvant volumetric modulated arc therapy (VMAT) compared with three-dimensional conformal radiation therapy (3DCRT) in terms of toxicity and local control (LC) in patients with soft tissue sarcoma of the extremities.
Methods:
From 2004 to 2016, 109 patients were treated, initially using 3DCRT and subsequently with VMAT. Clinical outcome was evaluated by contrast-enhanced MRI, thoracic and abdominal CT 3 months after treatments and then every 6 months. Toxicity was evaluated with Common Terminology Criteria for Adverse Events scale v. 4.3.
Results:
Patients presented Stage III soft tissue sarcoma disease (77%), localized tumor (95%) at the lower extremity (87%), adipocytic histotype (46%). Surgical resection was performed in all patients, followed by adjuvant 3DCRT in 38, and VMAT in 71. The median total dose was 66 Gy/33 fractions (range 60–70 Gy;25–35 fractions). More successful bone sparing was recorded using VMAT (p < 0.001). Median follow-up was 61 months, 93 and 58 months for 3DCRT and VMAT group, respectively. The 2- and 5 year LC were 95.3±2.1%, and 87.4±3.4% for the whole cohort, 92.0±4.5%, 82.9±6.4% for 3DCRT, 97.1±2.0%, 89.6±4.1% for VMAT (p = 0.150). On univariate and multivariate analysis the factors recorded as conditioning LC were the status of the surgical resection margins (p = 0.028) and the total dose delivered (p = 0.013).
Conclusion:
The availability of modern radiotherapy technique permit a better conformity on the target with maximum sparing of normal tissue and acceptable side-effects. VMAT is a safe and feasible treatment with limited rate of toxicity, compared to 3DCRT. Results on LC of VMAT are encouraging.
Advances in knowledge:
Soft tissue sarcoma of the extremities can benefit from the use of VMAT, with a reduction of the high dose to bones to avoid radiation osteonecrosis. An adequate total dose of at least 66 Gy and a radical surgical margin allow a good local control.
Introduction
Soft tissue sarcomas (STS) are rare tumors, representing about 1% of adult malignancies, and occurring in about half of patients at the lower limb.1,2 Radical surgical resection represents the mainstay of treatment. Surgery is followed by adjuvant radiation therapy (RT) in case of deeply located Grades II–III STS, and/or tumors larger than 5 cm, and/or positive resection margins.3 Using this approach, a long-term local control (LC) is obtained in more than 80–90% of cases treated with satisfactory functional preservation.4 The prescription of adequate RT therapeutic doses is mandatory.5 Several series investigated the role of the optimal effective dose, suggesting that at least 64 Gy should be delivered.6,7 Unfortunately, concerning the large volumes that conventionally need to be treated, the delivery of high doses to the whole target is not always safe and feasible, because of the high risk of side-effects. The most frequent complications are represented by radiodermatitis, edema, subcutaneous fibrosis, lymphedema, bone fractures, osteomyelitis and joint stiffness.8 To date, the big question is whether a more precise and selective RT technique could be able to obtain a satisfactory local control minimizing toxicities. Three-dimensional conformal radiation therapy (3DCRT) is the main RT modality used; however, 3DCRT is unable to modulate the dose to the tumor, and consequently to spare adequately the normal tissue. More recently, intensity modulated radiation therapy (IMRT) or volumetric modulated arc therapy (VMAT) have been employed.9 These techniques allow to achieve a high dose conformity with maximum avoidance of normal structures. Few studies have been published assessing the role of VMAT for extremity STS, and none of these compared 3DCRT and VMAT.10,11 In our department, this setting of STS patients were treated using 3DCRT at first, and with VMAT at a second time. Based on the poorness of literature data about this issue, we retrospectively analyzed the role of these different RT modalities in terms of toxicities and local control rate. Disease free survival and patients overall survival were evaluated as well.
methods and materials
Patients and procedures
The present retrospective study includes patients with newly diagnosed STS of the extremities treated at Humanitas Cancer Center. All patients were treated in agreement with the Helsinki Declaration. This study was based on a retrospective analysis of treatment charts and received approval by local Ethical Committee. All patients signed at admission an informed consent to the use of their data for scientific scope. To define the appropriate therapy, each patient was evaluated by a multidisciplinary team including surgeons, oncologists and radiation oncologists. All patients underwent limb-sparing surgery followed by adjuvant RT, and/or chemotherapy (CHT). Surgery was performed to obtain an en-bloc gross tumor resection paying attention to preserve nerve and vascular integrity, whenever feasible. Complete surgical resection (R0) was defined as the absence of microscopical invasion of the resection margin in the histological specimen; incomplete surgical as the presence of microscopically (R1) or macroscopically (R2) positive margins. In cases of R2, a surgical re-exploration was evaluated but not always performed. Neoadjuvant and/or adjuvant CHT was administered in case of metastatic disease at diagnosis, and/or Grade III STS. The schedule of CHT more frequently used, was epirubicin 60 mg/m2 per day (short infusion, days 1 and 2) plus ifosfamide 3 g/m2 per day (days 1, 2, and 3), repeated every 21 days for 3–4 cycles. RT was performed within 8 weeks from surgery, or within 4 weeks from the last CHT cycle. The median total dose prescribed was 66 Gy (range 60–70 Gy) in 33 daily fractions (range 25–35).
Radiation therapy treatment procedures
Simulation contrast-enhanced CT scan and preoperative MRI sequences were used to define the target volume and organs at risk (OARs). Two different target volumes were delineated: the clinical target volume (CTV) corresponded to the surgical cavity including the surgical scar, and the planning target volume (PTV) generated by adding an isotropic margin of 10 mm from CTV. OARs defined were: bone, bladder, colon-rectum, and brachial plexus, according to the treated regions. Among bone, every single structures was outlined. Osteonecrosis was identified using basal T1 and T2 MRI images, and defined as the presence of hypointensity areas surrounded by slight area of hyperintensity at T2 image. From 2004 to 2010 patients were treated with 3DCRT using multiple coplanar fixed gantry beams shaped with the multi leaf collimator. Since 2011 to 2016, patients were treated with VMAT modality (in the RapidArc form, Varian Medical System, Palo Alto, CA) using single or multiple, full or partial, coplanar or non-coplanar arcs based on an individualized plan optimization. The quantitative evaluation of the treatment plans was performed by means of standard dose–volume histogram (DVH) analysis. A number of relevant metrics were scored and assessed for all target volumes and OARs. These included, among others, V95%, V107% for the target volumes, together with a number of appropriate DXccm (dose received by at most X cm3 of the delineated structure volume) related to the bone. For the risk of osteonecrosis, a maximum dose Dmax <55 Gy was observed according to internal guidelines. Mean values of the selected parameters for the two cohorts of patients (3DCRT and VMAT) were considered. A cone beam CT was performed for all patients every day with the aim to assure a correct patient positioning and isocenter accuracy.
Outcome evaluation
Clinical outcome was evaluated by physical examination, muscloskeletal MRI and total body CT scan performed 3 months after the RT, every 3 months thereafter for the first year, and every 6 months afterwards. Local control (LC) was defined as the absence of recurrence at the site of treatment. Toxicities were assessed according to the Common Toxicity Criteria CTCAE v. 4.03,12 and the RTOG/EORTC late morbidity scoring system.13
Statistical analysis
Standard descriptive statistics (mean standard deviation and cross-tabulation analysis) was used to describe the data general behavior. The Pearson χ2 test were used to analyze the differences between qualitative toxicity data. The survival curves were plotted by the Kaplan–Meier method evaluating the time elapsed from the surgical act to the last follow-up (censored data) or the specific event, and compared using the log-rank test. Overall survival (OS) evaluated the deaths from any causes, while the disease-free survival (DFS) events were local, nodal or distant progression, or death from any causes. Multivariate Cox regression analysis included the variable with p-value less than 0.05 by univariate analysis. All data were analyzed using the SPSS statistical software package (v. 21, IBM Corporation, NY).
Results
Patients and treatments
From June 2004 to August 2016, 109 patients with STS of the extremity were treated, 38 using 3DCRT and 71 VMAT technique. The majority of the patients had Stage III disease (77%), localized tumor (95%) at the lower extremity (87%), and adipocytic histotype (46%). Surgical resection was performed in all patients, the majority had radical resection (76%). Chemotherapy was administered in about half of the patients, both in pre- and post-operative setting. RT was started within 8 weeks from surgery or within 4 weeks since the last CHT cycle. The mean PTV volume was 1160 ± 80 cm3 (range 66–3443 cm3). All patients completed the planned radiation treatment. Patient, tumor and treatment characteristics of the whole cohort and stratified for the RT treatment modalities (3DCRT vs VMAT) are shown in Table 1.
Table 1. .
Patient characteristics
| All patients | 3DCRT | VMAT | ||
| Nb patients | 109 | 38 (34.9%) | 71 (65.1%) | |
| Follow-up | Median [range] months | 61 [6–152] | 93 [6–152] | 58 [15–89] |
| Age (at diagnosis) | Median [range] years | 55 [15–84] | 57 [19–81] | 55 [15–84] |
| Gender | Male | 63 (57.8%) | 22 (57.9%) | 41 (57.7%) |
| Female | 46 (42.2%) | 16 (42.1%) | 30 (42.3%) | |
| Location | Lower extremity | 95 (87.2%) | 28 (73.7%) | 67 (94.4%) |
| Upper extremity | 14 (12.8%) | 10 (26.3%) | 4 (5.6%) | |
| Histology | Adypocytic | 50 (45.9%) | 14 (36.8%) | 36 (50.7%) |
| Fibroblastic / Myofibroblastic | 23 (21.1%) | 9 (23.7%) | 14 (19.7%) | |
| Smooth muscle | 13 (11.9%) | 6 (15.8%) | 7 (9.9%) | |
| Other histologiesa | 23 (21.1%) | 9 (23.7%) | 14 (19.7%) | |
| PTV volume | Mean ± Std Error of mean (cm3) | 1160 ± 80 | 1031 ± 146 | 1228 ± 95 |
| Grade | I–II | 25 (22.9%) | 11 (28.9%) | 14 (19.7%) |
| III | 84 (77.1%) | 27 (71.1%) | 57 (80.3%) | |
| Stage | Localized | 104 (95.4%) | 36 (94.7%) | 68 (95.8%) |
| Metastatic | 5 (4.6%) | 2 (5.3%) | 3 (4.2%) | |
| Surgical margins | Radical, R0 | 83 (76.1%) | 30 (78.9%) | 53 (74.6%) |
| Positive microscopic margin, R1-R2 | 26 (28.3%) | 8 (21.1%) | 18 (25.4%) | |
| Chemotherapy | No | 56 (51.4%) | 26 (68.4%) | 30 (42.3%) |
| Neoadjuvant | 31 (28.4%) | 5 (13.2%) | 26 (36.6%) | |
| Adjuvant | 18 (16.5%) | 4 (10.5%) | 14 (19.7%) | |
| Neo- and adjuvant | 4 (3.7%) | 3 (7.9%) | 1 (1.4%) | |
| Prescribed dose | <66 Gy | 13 (11.9%) | 11 (28.9%) | 2 (2.8%) |
| ≥66 Gy** | 96 (88.1%) | 27 (71.1%) | 69 (97.2%) |
** 66–70 Gy in 33–35 fractions, or 66.5 Gy in 25 fractions
Histologies with uncertain differentiation: Synovial, Clear Cell, Rhabdomiosarcoma, other rare histologies
From the dosimetric viewpoint, the target coverage (V95%) was better in the VMAT cohort, although not significant (p = 0.207), while the improved bone sparing was highly significant (p < 0.001). Dose distributions for a 3DCRT (on the left) and a VMAT (on the right) patients are shown in Figure 1. A dosimetric comparative analysis is shown in Table 2.
Figure 1. .
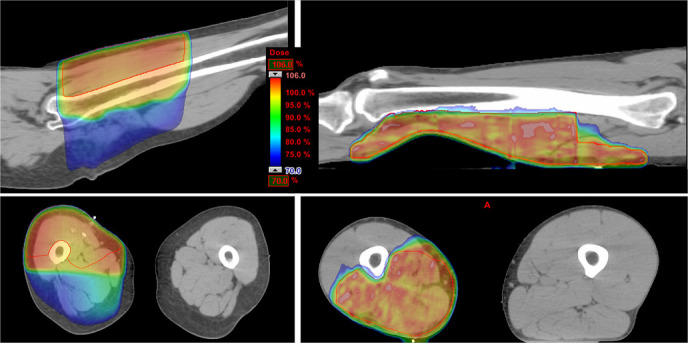
Dose distribution for a 3DCRT and a VMAT patient. 3DCRT,three-dimensional conformal radiation therapy; VMAT, volumetric modulated arctherapy.
Table 2. .
Dosimetric analysis summary, as mean values ± standard error of the mean, and range in brackets
| Structure | Parameter | All patients | 3DCRT | VMAT | p |
| PTV | Volume (cm3) [range] | 1160 ± 80 [66–3443] |
1031 ± 144 [66, 3443] |
1228 ± 94 [98, 3396] |
0.262 |
| Meana (Gy) | 65.7 ± 0.3 | 64.6 ± 0.5 | 66.3 ± 0.3 | 0.003 | |
| V95% (%) | 90.9 ± 1.3 | 87.9 ± 3.4 | 92.5 ± 0.9 | 0.207 | |
| V107% (%) | 0.80 ± 0.31 | 1.9 ± 0.8 | 0.2 ± 0.1 | 0.063 | |
| Bone | D1ccm (Gy) | 60.6 ± 0.9 | 66.9 ± 0.5 | 57.3 ± 1.2 | <0.001 |
| D5ccm (Gy) | 58.8 ± 1.0 | 66.1 ± 0.6 | 55.0 ± 1.3 | <0.001 | |
| D10ccm (Gy) | 57.5 ± 1.1 | 65.4 ± 0.8 | 53.3 ± 1.4 | <0.001 | |
| Dmax (Gy)b | 63.5 ± 0.8 | 67.7 ± 0.5 | 61.3 ± 1.2 | <0.001 |
3DCRT, three-dimensional conformal radiation therapy; PTV, planning target volume; VMAT, volumetric modulated arc therapy.
corrected for equivalence to 2 Gy/fraction
Dmax is the maximum point dose
Toxicity
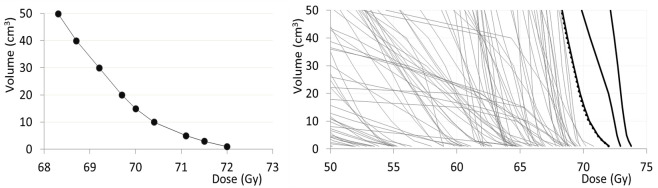
Grade 2–3 acute skin toxicity occurred in 58 patients: 53.2% of the whole cohort, 60.5 and 49.3% in the 3DCRT and VMAT groups, respectively. Grade 1, G1, late fibrosis occurred in 17 (15.6%) patients. A G2 fibrosis was observed in 11 patients, 7.6% in the 3DCRT group (n = 3), 11.3% of patients of the VMAT group (n = 8). None of the 109 patients experienced G3 soft tissue and subcutaneous toxicity. G4 late ulceration occurred in one patient of the whole cohort (0.9%). Three patients had G4 late osteonecrosis (2.8%). The bone toxicity was investigated in detail: in the VMAT group, there were no cases of osteonecrosis. The three patients presenting G4 osteonecrosis were treated using 3DCRT (7.6%) within 1 year from the end of radiation treatment, 2 for lower and 1 for upper extremity STS; all these patients were symptomatic, presenting pain and functional limitations, and the osteonecrosis was radiologically confirmed; none of them experienced a bone fracture. In one of the patients, the necrosis was so extended that required surgical excision; in that case a histologic confirmation was also obtained. All these patients received >70 Gy to a bone volume ≥15 cm3. A more detailed analysis of the dose–volume parameters for the three patients having had G4 osteonecrosis is reported in Figure 2. All and only the three patients with G4 bone toxicity received doses by a volume from 1 to 50 cm3 higher than the points depicted in the left side of the figure. The DVHs of the bone in the range 0–50 cm3 of volume for all the patients are reported in the right plot of the same figure, with the G4 patients shown with black bold lines (dotted DVH reports the plot on the right side).
Figure 2. .
DVH of bone. On the left: tolerance DVH for G4 toxicity; on the right: DVH of the whole patient cohort, in black bold the patients who had G4 toxicity, in dotted line the tolerance DVH as plot on the left. DVH,dose–volume histogram.
For the whole series, 3DCRT patients received higher doses to the bone compared to those treated with VMAT: the D15ccm mean values in the 3DCRT and VMAT groups were 64.9 ± 5.6 and 51.9 ± 11.9 Gy, respectively (p < 0.001).
LC, DFS and OS analysis
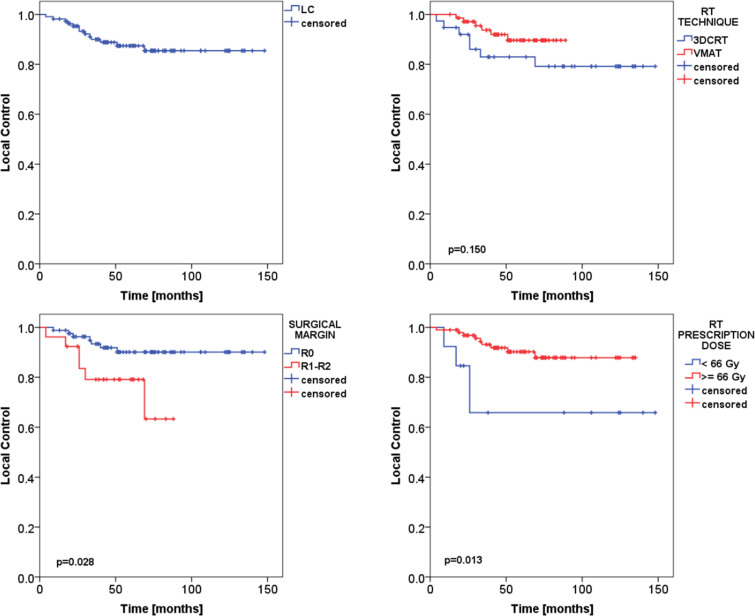
The median follow-up was 61 months for the entire cohort, 93 and 58 months for the 3DCRT and VMAT groups, respectively. The 2- and 5 year LC rates were 95.3±2.1%, and 87.4±3.4% for the whole cohort, 92.0±4.5% and 82.9±6.4% for the 3DCRT group, 97.1±2.0% and 89.6±4.1% for the VMAT group, with no significant differences (p = 0.150), as shown in Figure 3. No age, gender, histological subtype, tumor grading, target volume or RT modalities employed were recorded as significantly impacting on LC. On univariate and multivariate analysis the factors recorded as conditioning LC were the status of the resection margins and the total dose delivered. In case of R0 surgery compared to R1–R2, the 5 year LC rate was 90.1±3.6% and 79.1±8.4%, respectively (p = 0.028). For patients who received a total dose ≥66 Gy compared to <66 Gy, the 5 year LC rate was 90.2±3.3% and 65.8±14.1%, respectively (p = 0.013).
Figure 3. .
Kaplan–Meier plot of the local lontrol for the whole patient cohort (on the upper left), stratified by RT technique (upper right), by surgical margin (lower left), and by prescription dose (lower right). 3DCRT,three-dimensional conformal radiation therapy; RT,radiation therapy; VMAT, volumetric modulated arctherapy.
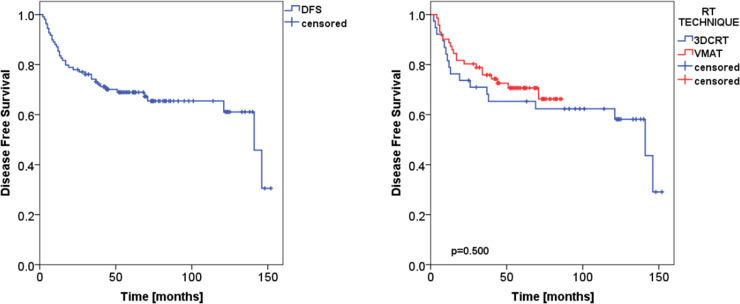
The median, 2- and 5 year DFS were 141 ± 12 months, 78.0±4.0% and 68.9±4.5% for the whole cohort; 73.7±7.1% and 59.6±8.1% for the 3DCRT group, 80.3±4.7% and 70.3±5.6% for the VMAT group (p = 0.500) as presented in Figure 4.
Figure 4. .
Kaplan–Meier plot of the disease free survival for the whole patient cohort (on the left), and stratified by RT technique (on the right). 3DCRT,three-dimensional conformal radiation therapy; RT, radiation therapy; VMAT, volumetricmodulated arc therapy.
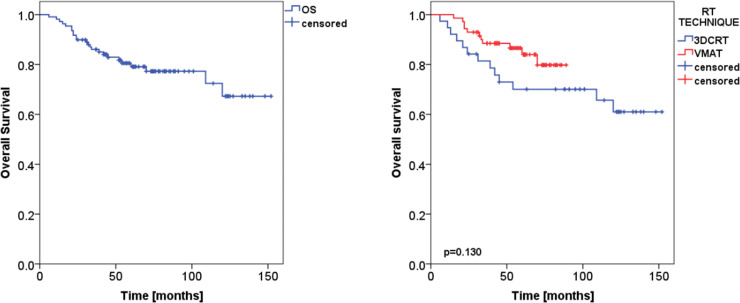
The 2- and 5 year OS were 89.9±2.9% and 79.1±4.1% for the whole series; 84.2±5.9% and 70.1±7.6% for the 3DCRT group, 93.0±3.0% and 84.0±4.8% for the VMAT group (p = 0.130), as shown in Figure 5. On univariate analysis, the OS correlated with patient age (p = 0.004), the disease stage (localized or metastatic, p < 0.001), and the prescription dose (p = 0.022), while the surgical resection only presented a tendence to significance (p = 0.063). On multivariate Cox analysis, only the patient age remained significant (p = 0.006).
Figure 5. .
Kaplan–Meier plot of the overall survival for the whole patient cohort (on the left), and stratified by RT technique (on the right). 3DCRT,three-dimensional conformal radiation therapy; RT, radiation therapy; VMAT, volumetricmodulated arc therapy.
Discussion
The standard of care for STS of the extremities is the surgical resection followed by adjuvant RT. The results in terms of local control and quality of life are highly satisfactory, with more than 80% of patients free from long-term recurrence. The use of a precise RT technique, able to obtain an adequate target coverage with maximum sparing of normal structures, is mandatory. The 3DCRT is the most frequent RT modality employed, but it can be burdened by high risk of late irreversible toxicities such as edema, joint weakness, fibrosis, osteonecrosis and bone fracture. The recent availability of IMRT and VMAT permits an optimization of RT treatment. Unfortunately, few studies compared 3DCRT and IMRT/VMAT, and to date no consolidate evidence exists about the optimal RT techniques to utilize.8,9,14 Preliminary reports conducted on large surgical series seem to show a real advantage of IMRT compared to 3DCRT. Folkert et al analyzed data of 319 adult patients with STS of extremities, undergoing 3DCRT (154 patients) or IMRT (165 patients). A significantly lower incidence of Grade ≥ 2 toxicities was reported in the IMRT group compared to 3DCRT, in terms of radiation dermatitis (31.5% vs 48.7%), edema (7.9% vs 14.9%), and fracture incidence (4.8% vs 9.1%). The high doses delivered to the normal structures, using 3DCRT, seem to be the critical factor conditioning the occurrence of severe side-effect, particularly bone fractures.11 Dickie et al, in a retrospective study, evaluated 691 lower extremity STS patients, with the aim to correlate bone fractures with dosimetric features. They reported an incidence of 4.5% with a higher risk in case of maximum doses ≥ 59 Gy, a mean dose ≥37 Gy, and more than half of normal bone structures receiving 40 Gy.15 Considering the availability at our institution of VMAT technique, our aim was to evaluate feasibility and safety of this RT modality in patients treated for STS of extremities. In addition, we compared outcome of patients treated using 3DCRT at first and VMAT in later times in terms of treatment-related toxicities, LC rate and OS. All VMAT patients received the total dose prescribed without discontinuation for severe toxicity. No Grade 3–4 late toxicity to soft tissue or skin was recorded. Literature data showed osteoradionecrosis as the main and most severe side-effect related to the irradiated volume and RT modality employed. Arthus et al,16 evaluating dosimetric differences between 3DCRT and IMRT reported a maximum dose to bone lower with 3DCRT than with IMRT, in contrast with our findings. In our study, higher dose was recorded using 3DCRT respect to VMAT, allowing a greater sparing of normal bone with the advanced technique. This translated in no bone G3-4 toxicity reported in our serie using VMAT, while osteonecrosis occurred in three patients who underwent 3DCRT (p < 0.001). The risk of bone toxicity can emerge with 3DCRT to deliver a therapeutic effective dose. We choose to pursue LC at the expense of higher chances of side-effects. Some publications assessed the role of the RT total dose delivered on LC, underlying the necessity to administer high RT doses aiming to obtain an adequate disease control. Zagars et al stated a cut-off value of at least 64 Gy above all in case of positive surgical margins (p < 0.001).6 Similarly, Delaney et al7 retrospectively reviewed data on 105 patients treated for extremities STS; a 5 year LC of 85% was shown in case of a dose >64 Gy compared to 66% for lower ones. Our data confirmed these previous observations with a significant impact of dose delivered on LC. In our series, the 5 year LC rate was 89.6% for patients who received a total dose ≥66 Gy and 71.4% for total doses < 66 Gy. Other prognostic factors, eventually impacting outcome, were investigated. The status of resection margins, the tumor volume, and the technique employed have shown to affect the outcome. In the same serie by Zagars, a LC rate of 88% was showed in case of R0 margins and 64% in cases of R1–R2.6 In addition, the tumor size was also outlined as factor conditioning LC with a threshold identified at 10 cm. In our analysis, the positive surgical margins were recorded as one of the main factor influencing LC, while the tumor volume at diagnosis has not proven to be a determinant factor. This could be due to the fact that only 30% of patients had a maximum diameter larger than 10 cm. Unfortunately, few investigators assessed the role of different RT in the adjuvant setting and, to our knowledge, no one concerning VMAT mode. Among these, Folkert et al compared 3DCRT or IMRT and a 5 year LC rate of 85 and 92% was detected respectively (p = 0.05).11 We observed a favorable trend employing VMAT, although without significant differences, with a LC rate in about 90% of cases treated. In our experience, the use of VMAT modality compare to 3DCRT has proven to be effective on LC with maximum reduction of toxicities.
With the VMAT technology, it is important to assess the low dose to the surrounding structures due to the nature of the technique, based on arcs instead of a limited number of fixed beams. In the STS treatment, a concern is of course the irradiation of the contralateral limb. To evaluate this difference, the mean dose to the contralateral limb for lower extremities, in a volume long as the PTV of the involved limb, was computed in our patient cohort, resulting in 2.4 Gy (range 0.1–8.5 Gy) and 4.1 Gy (range 0.2–8.4 Gy) for 3DCRT and VMAT, respectively. The difference in the low dose bath is present in average; however, the contralateral limb dose depends on the patient anatomy and on the arc geometry choice: the dose range is indeed the same for both techniques. Particular attention has, however, to be paid to the contralateral structures during VMAT optimization.
Limitations in our study were the retrospective nature, the different sample of 3DCRT and VMAT group, and the shorter follow-up time in VMAT patients. However, few studies are available regarding the role of different RT modalities for the treatment of extremities STS, and our experience can be useful for future prospective studies.
Conclusion
Our data show that VMAT is a safe and feasible treatment with limited rate of toxicity, compared to 3DCRT. The high dose conformity and the maximum sparing of bone structures allow to obtain an adequate LC with a negligible incidence of side-effects. Preserving functional outcome and quality of life is fundamental in this group of young and active patients. These data can help to exploit VMAT technique in all the other critical site of STS origin.
Contributor Information
Lucia Di Brina, Email: lucia.di_brina@humanitas.it.
Antonella Fogliata, Email: Antonella.Fogliata@humanitas.it.
Pierina Navarria, Email: pierina.navarria@humanitas.it.
Giuseppe D'Agostino, Email: giuseppe.dagostino@humanitas.it.
Ciro Franzese, Email: ciro.franzese@humanitas.it.
Davide Franceschini, Email: davide.franceschini@humanitas.it.
Fiorenza De Rose, Email: fiorenza.de_rose@humanitas.it.
Tiziana Comito, Email: tiziana.comito@humanitas.it.
Alexia Bertuzzi, Email: alexia.bertuzzi@humanitas.it.
Andrea Marrari, Email: andrea.marrari@humanitas.it.
Piergiuseppe Colombo, Email: piergiuseppe.colombo@humanitas.it.
Vittorio Quagliuolo, Email: vittorio.quagliuolo@humanitas.it.
Armando Santoro, Email: armando.santoro@humanitas.it.
Marta Scorsetti, Email: marta.scorsetti@humanitas.it.
REFERENCES
- 1.Stiller CA, Trama A, Serraino D, Rossi S, Navarro C, Chirlaque MD, et al. Descriptive epidemiology of sarcomas in Europe: report from the RARECARE project. Eur J Cancer 2013; 49: 684–95. doi: 10.1016/j.ejca.2012.09.011 [DOI] [PubMed] [Google Scholar]
- 2.Lawrence W, Donegan WL, Natarajan N, Mettlin C, Beart R, Winchester D. Adult soft tissue sarcomas. A pattern of care survey of the American College of surgeons. Ann Surg 1987; 205: 349–59. doi: 10.1097/00000658-198704000-00003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.National Comprehensive Cancer Network Soft Tissue Sarcoma (Version 1.2014)... Available from: http://www.nccn.org/professionals/physician_gls/pdf/softtissuesarcoma.pdf.. [DOI] [PubMed]
- 4.Lack EE, Steinberg SM, White DE, Kinsella T, Glatstein E, Chang AE, et al. Extremity soft tissue sarcomas: analysis of prognostic variables in 300 cases and evaluation of tumor necrosis as a factor in stratifying higher-grade sarcomas. J Surg Oncol 1989; 41: 263–73. doi: 10.1002/jso.2930410414 [DOI] [PubMed] [Google Scholar]
- 5.Koshy M, Rich SE, Mohiuddin MM. Improved survival with radiation therapy in high-grade soft tissue sarcomas of the extremities: a seer analysis. Int J Radiat Oncol Biol Phys 2010; 77: 203–9. doi: 10.1016/j.ijrobp.2009.04.051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zagars GK, Ballo MT. Significance of dose in postoperative radiotherapy for soft tissue sarcoma. Int J Radiat Oncol Biol Phys 2003; 56: 473–81. doi: 10.1016/S0360-3016(02)04573-X [DOI] [PubMed] [Google Scholar]
- 7.Delaney TF, Kepka L, Goldberg SI, Hornicek FJ, Gebhardt MC, Yoon SS, et al. Radiation therapy for control of soft-tissue sarcomas resected with positive margins. Int J Radiat Oncol Biol Phys 2007; 67: 1460–9. doi: 10.1016/j.ijrobp.2006.11.035 [DOI] [PubMed] [Google Scholar]
- 8.Fein DA, Lee WR, Lanciano RM, Corn BW, Herbert SH, Hanlon AL, et al. Management of extremity soft tissue sarcomas with limb-sparing surgery and postoperative irradiation: do total dose, overall treatment time, and the surgery-radiotherapy interval impact on local control? Int J Radiat Oncol Biol Phys 1995; 32: 969–76. doi: 10.1016/0360-3016(95)00105-8 [DOI] [PubMed] [Google Scholar]
- 9.Teoh M, Clark CH, Wood K, Whitaker S, Nisbet A. Volumetric modulated Arc therapy: a review of current literature and clinical use in practice. Br J Radiol 2011; 84: 967–96. doi: 10.1259/bjr/22373346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Alektiar KM, Brennan MF, Healey JH, Singer S. Impact of intensity-modulated radiation therapy on local control in primary soft-tissue sarcoma of the extremity. J Clin Oncol 2008; 26: 3440–4. doi: 10.1200/JCO.2008.16.6249 [DOI] [PubMed] [Google Scholar]
- 11.Folkert MR, Singer S, Brennan MF, Kuk D, Qin L-X, Kobayashi WK, et al. Comparison of local recurrence with conventional and intensity-modulated radiation therapy for primary soft-tissue sarcomas of the extremity. J Clin Oncol 2014; 32: 3236–41. doi: 10.1200/JCO.2013.53.9452 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0 Published: May 28, 2009 (v4.03: June 14, 2010) U.S. Department of health and human services National Institutes of health National cancer Institute...
- 13. RTOG/EORTC late radiation Morbity scoring schema. RTOG Foundation Inc.. accessed December 2018.
- 14.Hong L, Alektiar KM, Hunt M, Venkatraman E, Leibel SA. Intensity-Modulated radiotherapy for soft tissue sarcoma of the thigh. Int J Radiat Oncol Biol Phys 2004; 59: 752–9. doi: 10.1016/j.ijrobp.2003.11.037 [DOI] [PubMed] [Google Scholar]
- 15.Dickie CI, Parent AL, Griffin AM, Fung S, Chung PWM, Catton CN, et al. Bone fractures following external beam radiotherapy and limb-preservation surgery for lower extremity soft tissue sarcoma: relationship to irradiated bone length, volume, tumor location and dose. Int J Radiat Oncol Biol Phys 2009; 75: 1119–24https://doi.org/10. doi: 10.1016/j.ijrobp.2008.12.006 [DOI] [PubMed] [Google Scholar]
- 16.Arthurs M, Gillham C, O'Shea E, McCrickard E, Leech M. Dosimetric comparison of 3-dimensional conformal radiation therapy and intensity modulated radiation therapy and impact of setup errors in lower limb sarcoma radiation therapy. Pract Radiat Oncol 2016; 6: 119–25. doi: 10.1016/j.prro.2015.03.008 [DOI] [PubMed] [Google Scholar]