Abstract
Objective:
Adequate assessment of fluid responsiveness in shock necessitates correct interpretation of hemodynamic changes induced by preload challenge. This study evaluates the accuracy of point-of-care Doppler ultrasound assessment of the change in carotid corrected flow time (ccFT) induced by a passive leg raise (PLR) maneuver as a predictor of fluid responsiveness. Noninvasive cardiac output monitoring (NICOM™) was the comparison standard.
Design:
Prospective, non-interventional study.
Setting:
Intensive care unit at a large academic center
Patients:
Patients with new, undifferentiated shock and vasopressor requirements despite fluid resuscitation were included. Patients with significant cardiac disease and conditions that precluded adequate passive leg raising were excluded.
Intervention:
ccFT was measured via ultrasound before and after a PLR maneuver. Predicted fluid responsiveness was defined as >10% increase in stroke volume on NICOM™ following PLR. Images and measurements were reanalyzed by a second, blinded physician. The accuracy of ΔccFT to predict fluid responsiveness was evaluated using ROC analysis.
Results:
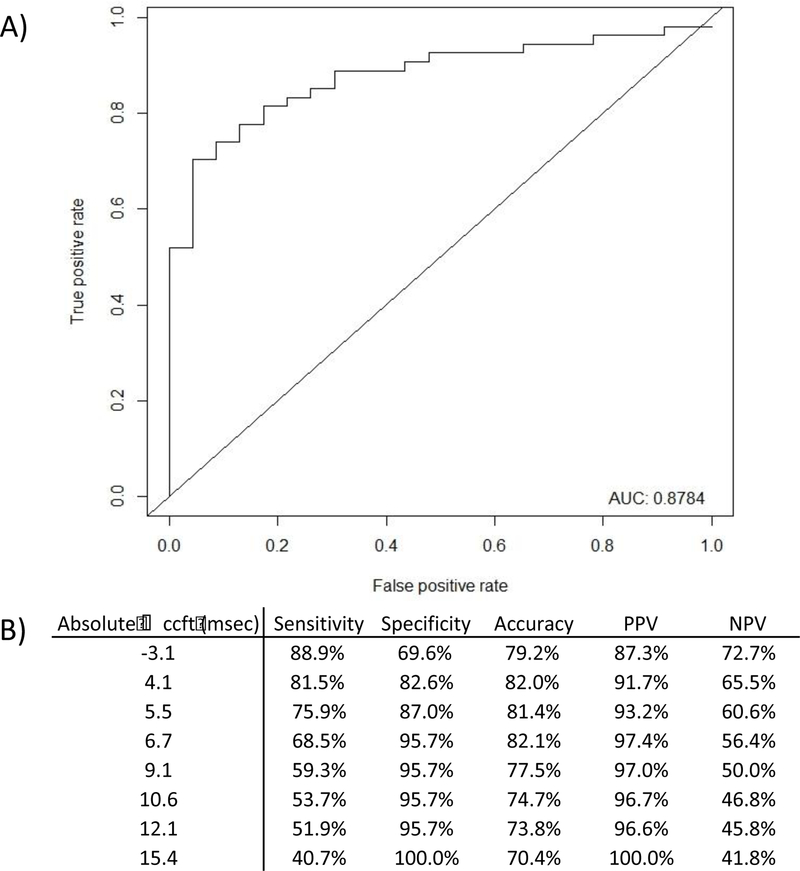
Seventy-seven subjects were enrolled with 54 (70.1%) classified as fluid responders by NICOM™. The average ΔccFT after PLR for fluid responders was 14.1 ± 18.7 msec vs. −4.0 ± 8 msec for non-responders (P<0.001). ROC analysis demonstrated that ΔccFT is an accurate predictor of fluid responsiveness status (AUC 0.88, 95% CI 0.80–0.96) and a 7 msec increase in ccFT post PLR was shown to have a 97% positive predictive value and 82% accuracy in detecting fluid responsiveness using NICOM™ as a reference standard. Mechanical ventilation, respiratory rate, and high PEEP had no significant impact on test performance. Post-hoc blinded evaluation of bedside acquired measurements demonstrated agreement between evaluators.
Conclusions:
ΔccFT can predict fluid responsiveness status after a PLR maneuver. Using point-of-care ultrasound to assess ΔccFT is an acceptable and reproducible method for non-invasive identification of fluid responsiveness in critically ill patients with undifferentiated shock.
Keywords: Fluid responsiveness, corrected flow time, ultrasound, shock
Introduction:
Fluid responsiveness assessment is defined as an increase in cardiac output (CO) in response to preload augmentation, and is used in resuscitation from shock [1, 2]. Temporary intravascular fluid shift maneuvers such as the passive leg raise (PLR) test [3] transiently increase venous return, thus enabling the assessment of CO change with an intervention that mimics fluid administration. Unfortunately, CO monitoring technology is expensive, not widely available, and imprecise. New technologies to assess the hemodynamic response to PLR are needed.
Flow time (FT), or left ventricular ejection time, reflects the duration of systole, and is measured from the beginning of the upstroke to the trough of the incisural notch on a pulse waveform analysis [4]. Corrected for the heart rate variability, it is called corrected FT and the change in its duration may reflect changes in stroke volume. Point-of-care ultrasound is noninvasive and increasingly available in critical care settings [5, 6], and the assessment of corrected FT via Doppler ultrasound [7–9] is a safe and simple method which does not require extensive ultrasonographic expertise by the operator.
In this study, we hypothesize that the change in carotid corrected FT (ΔccFT) induced by a PLR maneuver may predict fluid-responsive status in early, undifferentiated shock. The non-invasive bioreactance CO monitoring (NICOM™, Cheetah Medical, Newton Center, MA) system was used as the reference standard as it has been validated in the assessment of fluid responsiveness in combination with PLR [10–14].
Materials and Methods:
This prospective, non-interventional study was conducted in a single academic quaternary care center. Adult patients with early (<24 hours duration), undifferentiated shock, who were admitted to a medical or surgical intensive care unit with persistent vasopressor requirements despite pre-enrollment fluid resuscitation of >1 liter of IV fluids were enrolled after informed consent. Patients were excluded if they presented with a history of left or right heart failure, pulmonary hypertension, cardiac rhythm other than sinus, significant peripheral vascular disease, suspected or known increased intracranial pressure, recent abdominal surgery, recent history of venous thromboembolism, and body-mass-index (BMI) <15 or >40 kg/m2. Enrollment period was from May 2016 to April 2017. Approval for this study was granted by the University of California, Los Angeles Institutional Review Board (IRB#15–001768).
Fluid responsiveness assessment:
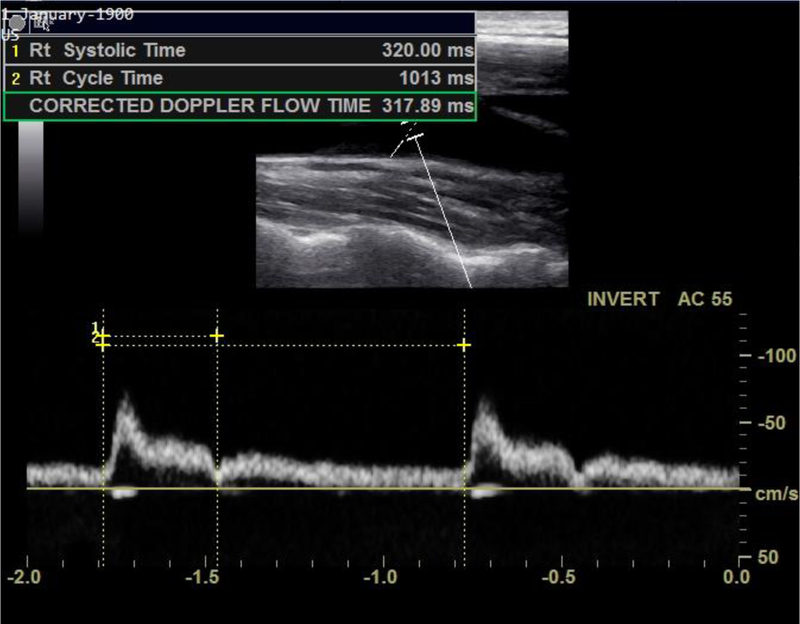
Measurements of ccFT were made at an increment of a tenth of a milliseconds (msec) and were obtained analyzing Doppler images of common carotid artery pulse waveforms (LOGIQ e, GE Healthcare, Wauwatosa, WI) by a trained physician sonographer (Figure 1). A linear array probe was used to obtain and record Doppler images of the vessel in long-axis view. Patients were evaluated using Ultrasound and NICOM™ simultaneously. Measures we obtained at baseline (prior to PLR, with the patient in a semi-recumbent position with 45° head of bed elevation for at least 10 minutes), and during the PLR maneuver performed using NICOM™ manufacturer’s protocol (patient in supine position for three minutes with legs passively supported by an inflated wedge at 45° elevation, Supplemental Figure 1). ccFT measurements were captured after 120 seconds of the PLR maneuver. Fluid-responsive status was defined as ≥10% increase in SV via NICOM™ [15]. Systolic and cycle times were analyzed by the bedside operator’s interpretation of ultrasound-captured images and ccFT values were calculated using Wodey’s formula, , which has been shown to better correct for fast heart rates in comparison to widely used Bazett’s formula () [16, 17]. A second, blinded investigator reevaluated unprocessed bedside images to avoid treatment bias and assessinter-user variability.
Figure 1.
Carotid Doppler waveform with markings: 1) flow time (FT), 2) cycle time. ccFT = FT + 1.29 × (HR-60)
Statistical Methods:
The NICOM™ and carotid Doppler measures were compared by response status using the two-sample t-test. The accuracy of ΔccFT as a predictor of fluid responsive status was assessed using ROC analysis. The best threshold of ΔccFT to detect fluid responsiveness was chosen to maximize the sensitivity for a target specificity of at least 96%. The accuracy of ΔccFT as a predictor of fluid response status was evaluated by the following potential covariates: mechanical ventilation, passive breathing on mechanical ventilation, and positive end-expiratory pressure (PEEP) > 5 among the mechanically ventilated subset of subjects. The AUCs were compared by level of each specified covariate [18]. The agreement between the bedside and blinded ccFT measures was evaluated using the Bland-Altman plot, a plot of the differences vs. the means. Accuracy was calculated as an average value of specificity and sensitivity of the test. P values < 0.05 were considered statistically significant. Data are presented as mean +/− SD or median [IQR].
Results:
79 patients were enrolled in the study. Two of the enrolled patients (2.5%) developed complications during PLR and did not complete the protocol. One of this pair developed atrial fibrillation, and the other had a significant decrease in the oxygen saturation of hemoglobin as measured by pulse oximetry (Supplemental Figure 2). Baseline characteristics of the 77 patients who completed the full PLR protocol are displayed in Table 1. These patients were grouped according to their stroke volume response to a PLR as measured by NICOM. “Fluid responders” included patients that had a stroke volume increase ≥10% by NICOM after a PLR. “Non-responders” included patients demonstrating a stroke volume increase <10% by NICOM after a PLR. The majority of the patients (70.1%) were designated as fluid responsive based on these criteria.
Table 1.
Patient baseline characteristics
| Patient Characteristic | Total (N=77) |
|---|---|
| Age, (mean ± SD), years | 60.6 ± 17 |
| Female, (%) | 51% |
| Body-mass-index, (mean ± SD), kg/m2 | 24 ± 8 |
| Hematocrit, (mean ± SD), % | 29.4 ± 7 |
| End-stage renal disease or dialysis, (%) | 42% |
| Total fluids received, (mean ± SD), L | 8 ± 5 |
| Mechanical ventilation (%) | 59% |
| – Passive ventilation, (% of ventilated patients) | 47% |
| PEEP >5 mmHg | 25% |
| Pressor Used | |
| – Norepinephrine, (%) | 71% |
| – Dopamine, (%) | 5% |
| – Vasopressin, (%) | 3% |
| – Phenylephrine, (%) | 6% |
| – Combination, (%) | 14% |
| APACHE II, (mean ± SD) | 24.5 ± 10 |
PEEP, positive-end-expiratory-pressure; SD, standard deviation; APACHE II, Acute Physiology And Chronic Health Evaluation II.
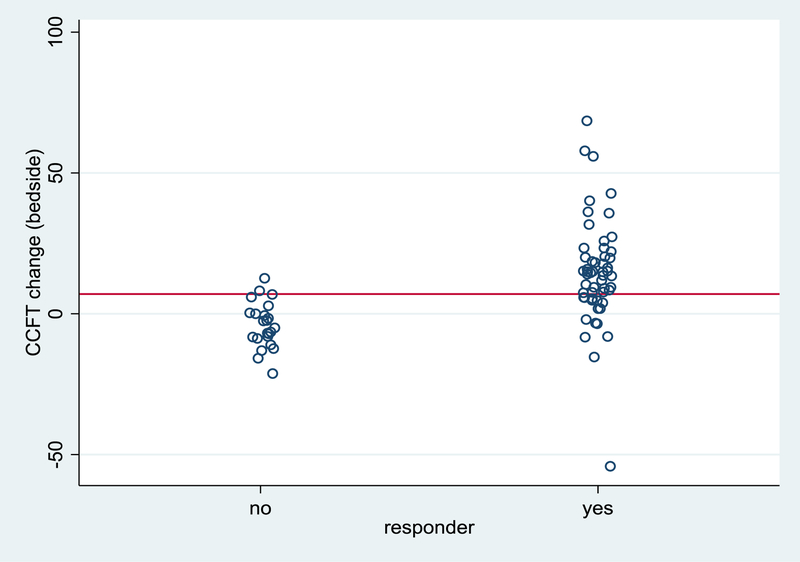
Fluid responsive patients had a greater increase in ccFT after PLR than non-responsive patients [14.1 ± 19 (SD) vs −4.0 ± 8 msec, p<0.001, Table 2]. The percentage increase from baseline in ccFT was also higher among responders than non-responders [+4.8 ± (SD) 6.4 vs −1.4 ± 2.9%, p<0.001]. Dot-plot analysis presented in Figure 2 demonstrates the differences in ΔccFT between NICOM-defined fluid responders and non-responders. Receiver operating characteristic curve analysis for ΔccFT ability to predict fluid responsiveness is presented in Figure 3, and we show that using a cut-off value of 7 msec as a ΔccFT to define fluid responsiveness had a specificity of 96%, sensitivity of 68%, positive predictive value of 97% and 82% accuracy. Additional sub-group analyses found that mechanical ventilation, respiratory rate, and positive-end-expiratory pressure (PEEP) >5cmH2O had no significant impact on the test performance (Supplemental Table 1). Blinded vs. bedside-obtained results were compared via Bland-Altman plot showing a mean difference score of 0 at baseline (non-significant, 95% limits of agreement were –6.7 and +6.6) and a mean difference score of −0.2 (non-significant, 95% limits of agreement were −6.6 and +6.4) after PLR, showing good agreement between investigators (Supplemental Figure 3).
Table 2.
ccFT and NICOM™ results pre and post PLR
| Parameter | Total, N=77 | Responders, N=54 (70.1%) |
Non-Responders, N=23 (29.9%) |
P=value | |
|---|---|---|---|---|---|
| Mean arterial pressure, (mean ± SD), mmHg | 60 ± 8 | 61 ± 8 | 68 ± 7 | 0.15 | |
| Heart rate, (mean ± SD), beats/min | 103 ± 24 | 101 ± 25 | 108 ± 21 | 0.28 | |
| NICOM™ | Baseline cardiac index, L/min/m2 | 3.7 ± 5.1 | 4.0 ± 6.1 | 3.1 ± 0.9 | 0.51 |
| Post PLR Cardiac index, L/min/m2 | 4.3 ± 3.7 | 4.8 ± 4.3 | 3.0 ± 0.8 | 0.06 | |
| Baseline SV, ml | 64.1 ± 24.7 | 65.0 ±24.5 | 62.0 ± 25.6 | 0.63 | |
| Post PLR SV, ml | 77.7 ± 32.2 | 85.9 ± 31.7 | 59.1 ± 25 | 0.01 | |
| ΔSV, % | 24.7 ± 23.6 | 33.9 ± 22.1 | 3.1 ± 6.9 | ||
| Carotid Doppler | Baseline ccFT, msec | 301 ± 33 | 300 ± 32 | 302 ± 35 | 0.86 |
| Post PLR ccFT, msec | 310 ± 37 | 315 ± 36 | 298 ± 37 | 0.067 | |
| ΔccFT, msec | 8.7 ± 18 | 14.1 ± 19 | −4.0 ± 8 | <0.001 | |
| ΔccFT, % | 3.0 ± 6.3 | 4.8 ± 6.4 | −1.4 ± 2.9 | <0.001 | |
ccFT, corrected carotid flow time; PLR, passive leg raise; SV, stroke volume
Figure 2.
Dot plot analysis of ΔccFT by fluid responder status
Figure 3.
A) Receiver operating characteristic curve analysis for ΔccFT ability to predict fluid responsiveness; B) ΔccFT test characteristics when cut-off values are used to predict fluid responsiveness
Discussion:
The results suggest that ΔccFT induced by a PLR maneuver can determine fluid responsiveness in a selected population of patients with early undifferentiated shock. The area under the receiver operator curve suggests that ΔccFT can be used in place of the reference method, which was a 10% increase in stroke volume measured by NICOM. A cutoff of a 7 msec increase in ΔccFT gave excellent positive predictive value and accuracy. Moreover, the PLR protocol was well tolerated and able to be completed in 97.5% of patients.
There is no consensus about the best way to predict fluid responsiveness [19]. In contrast to other novel hemodynamic monitoring systems, point-of-care ultrasound is widely present in ICU settings [20], and the number of indications for its use continue to grow [21]. Ultrasonographic measures such as respiratory change in inferior vena cava diameter [22], respiratory change in peak aortic velocity [23], or change in echocardiography-measured end-diastolic area of left ventricle [24] have been used for hemodynamic evaluation of a patient in shock [25]. The use of ultrasound to measure Doppler velocity time integral (VTI) of large arteries following PLR maneuver can predict fluid responsiveness assessment [26–28], although variability in the angle of insonation between measurements limits its precision [29].
Despite conflicting data from earlier studies [15, 30, 31], there is an increasing body of published evidence showing the usefulness of corrected flow time evaluation in fluid management [32–38]. However, there are a number of limitations of using the absolute value of corrected flow time. First, it is not a simple metric of preload but also depends on heart rate, inotropy and afterload conditions [4, 39, 40]. Second, its absolute duration does not correlate with the SV [41]. Instead, the change in duration of ccFT can identify changes in left ventricular stroke volume due to altered loading conditions. In order to determine if a change in preload leads to a change in the duration of flow time, the heart’s afterload and contractility must be constant and the flow time must be corrected for a heart rate [17]. Accordingly, ΔccFT decreases with fluid or blood removal [36, 42, 43], and increases with fluid administration in volume-depleted patients [38, 43, 44]. A number of pilot studies show that ΔccFT also increases after intravenous fluid bolus challenge [33, 35] or PLR [36, 38] in fluid responsive patients. Using ultrasonographic CO monitoring [35], pulse contour based analysis [37], or the more widely used, esophageal Doppler-based assessment of SV index to define preload responsiveness [33, 34], fluid responders show significantly higher ΔccFT in comparison to non-responders. Combining Doppler evaluation of ΔccFT with a well-validated PLR maneuver [10–14] offers several advantages in comparison to other methods of fluid responsiveness assessment. Measuring ΔccFT is less subject to artifact that many other measures of stroke volume, it is almost universally applicable and, based on results presented here, there is an excellent agreement between bedside and blinded investigator measurements.
There are several strengths of this study. This is the largest study to our knowledge evaluating ΔccFT after PLR as a predictor of fluid responsiveness in shock. All patients were on fixed vasopressor support during the test, thus minimizing the alteration of systemic vascular resistance during the evaluation. The study included patients spontaneously breathing and on passive mechanical ventilation. Positive pressure ventilation had no significant impact on the ability of ΔccFT to predict fluid responsiveness.
Our study has several limitations. We did not assess fluid responsiveness directly. Instead, we compared ΔccFT to a reference method. NICOM™ was used as a reference standard for fluid responsiveness because it has acceptable agreement with other CO monitoring systems [11, 13, 19, 45, 46], is easy to apply and has been studied in both spontaneously breathing and mechanically ventilated patients with shock in combination with PLR [13, 47, 48].
The exclusion of patients with congestive heart failure reduces the generalizability of conclusions, although post-hoc analysis of echocardiographic results obtained within the same hospital stay indicated that left ventricular ejection fraction was reduced in at least 13.7% of cases (10/73). The same applies for excluding conditions which can potentially lead to a suboptimal, or potentially harmful PLR – lower extremity thromboembolism, recent abdominal surgery or hip fractures, suspected elevated intracranial pressure or significant peripheral vascular disease. Broadening the inclusion criteria in the future should help understand better the general applicability of this method. Despite good interrater agreement, manual measurement can lead to misinterpretation of results, both due to measurement bias, or skill of the operator. Additionally, ccFT slightly varies throughout the respiratory cycle, and random averaging of the ccFT between the three beats [41] may not be able to sufficiently correct potential inaccuracy in interpretation of ccFT measurements. Automated identification of pulse waveform components combined with respiratory tracing may improve accuracy of ΔccFT interpretation in the future. More importantly, appreciating complex relationship between hemodynamic determinants and understanding the limits of currently used dynamic parameters which act as surrogates for SV change, future critical care research may lean toward utilization of composite measures of fluid responsiveness capable of predicting fluid responsiveness with better accuracy [49]. We can speculate that one such example could be combining carotid Doppler-derived parameters ΔVTI and ΔccFT.
Conclusion:
In patients with early, undifferentiated shock, ΔccFT induced by a PLR maneuver was able to predict fluid responsiveness. It compares favorably with NICOM™, with an AUC of 0.88 suggesting that it is an alternative to other methods. Its effectiveness is not affected by mechanical ventilation, respiratory rate, or PEEP >5mmH2O. Further studies focused on clarification of the role of ΔccFT in the assessment of fluid responsiveness are warranted.
Supplementary Material
Supplemental Figure 1. A) The patient is maintained in a semi-recumbent (45o angle of the bed) position with semi-extended neck and head partially rotated away from the operator at least 10 minutes before the measurement and PLR maneuver. The operator, using vascular probe of an ultrasound, localizes carotid artery in a transverse view. B) and C) Maintaining the view of carotid artery, the operator rotates the probe to establish a longitudinal view of the artery. D) Using Doppler mode in the established longitudinal view (upper part of the screen image), the operator then obtains adequate pulse waveform velocities (lower part of the screen image). When it is the appropriate time make measurements, the operator saves the image at the end of patient’s exhalation, and then carefully measures the duration of a cardiac cycle (beginning of systole to the end of diastole) and the systolic time (beginning of systole to the end of systole). The measurements are recorded, and ccFT is calculated using Wodey’s formula,. The system used in our study had preset parameters to identify “Systolic Time” and “Cycle Time” and a pre-programmed calculation to deliver the value of “Corrected Doppler Flow Time”. E) After completion of the baseline measurement, the assessment and measurement is repeated during the PLR maneuver as the patient lays in the in a supine position with legs being passively elevated at 45o angle and supported with a premade inflated wedge pillow. Upon initiation of the PLR, the operator will again identify the carotid artery, repeat the steps described above and, then captures a snapshot at the end of exhalation 2 minutes after the initiation of PLR. F) The measurements of systolic and cycle duration times are repeated, and the difference between ccFTBaseline and ccFTPLR represents the ΔccFT.
Supplemental Figure 2. Consort Diagram
Supplemental Figure 3. Bland-Altman plot for bedside and blinded correlation
Supplemental Table 1. The effect of mechanical ventilation and high PEEP on the test ability to predict fluid responsiveness: there is no difference in the test applicability based on Invasive positive pressure mechanical ventilation vs. negative pressure ventilation, passive positive pressure ventilation in comparison to spontaneously breathing patients or positive end-expiratory pressure >5cmH2O vs. ≤5cmH2O
Take home:
The change in carotid corrected flow time (ΔccFT) induced by a passive leg raise maneuver can predict fluid responsiveness in patients in early, undifferentiated shock. The ability to use this parameter, together with other ultrasonographic measures which reflect the dynamic changes induced by preload challenge, make point-of-care ultrasound an attractive tool in routine assessment of fluid responsiveness in shock
Tweet (Mandatory):
The change in carotid corrected flow time measured by Doppler sonography after a passive leg raise can predict fluid responsiveness in shock
Acknowledgements:
We would like to thank GE Healthcare for the financial and logistical support to the study. We would also like to thank our patients who participated the study, nursing teams, and UCLA Clinical and Translational Science Institute Research Associates Program as without them the study could not be completed successfully.
Sources of Funding:
This study was financially supported by GE Healthcare, General Electric Company, Wauwatosa, WI, USA. MC is a consultant for Edwards Lifesciences and Masimo Corp. SYC is consultant for La Jolla Pharmaceuticals. IB is a consultant for GE Healthcare.
Footnotes
Conflicts of interest
The remaining authors have disclosed that they have no relevant financial conflicts of interest.
References:
- 1.Cecconi M, De Backer D, Antonelli M, et al. : Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med 2014, 40(12):1795–1815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rhodes A, Evans LE, Alhazzani W, et al. : Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Intensive Care Med 2017, 43(3):304–377. [DOI] [PubMed] [Google Scholar]
- 3.Monnet X, Marik PE, Teboul JL: Prediction of fluid responsiveness: an update. Ann Intensive Care 2016, 6(1):111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hassan S, Turner P: Systolic time intervals: a review of the method in the non-invasive investigation of cardiac function in health, disease and clinical pharmacology. Postgrad Med J 1983, 59(693):423–434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rupp SM, Apfelbaum JL, Blitt C, et al. : Practice guidelines for central venous access: a report by the American Society of Anesthesiologists Task Force on Central Venous Access. Anesthesiology 2012, 116(3):539–573. [DOI] [PubMed] [Google Scholar]
- 6.Frankel HL, Kirkpatrick AW, Elbarbary M, et al. : Guidelines for the Appropriate Use of Bedside General and Cardiac Ultrasonography in the Evaluation of Critically Ill Patients-Part I: General Ultrasonography. Crit Care Med 2015, 43(11):2479–2502. [DOI] [PubMed] [Google Scholar]
- 7.Koito H, Spodick DH: Optimal Doppler measurement of left ventricular ejection time. Am J Cardiol 1989, 63(3):257–259. [DOI] [PubMed] [Google Scholar]
- 8.Tournadre JP, Muchada R, Lansiaux S, et al. : Measurements of systolic time intervals using a transoesophageal pulsed echo-Doppler. Br J Anaesth 1999, 83(4):630–636. [DOI] [PubMed] [Google Scholar]
- 9.Rothendler JA, Schick EC Jr., Ryan TJ: Derivation of systolic time intervals from Doppler measurement of temporal arterial blood flow . Am J Cardiol 1981, 47(1):68–72. [DOI] [PubMed] [Google Scholar]
- 10.Benomar B, Ouattara A, Estagnasie P, et al. : Fluid responsiveness predicted by noninvasive bioreactance-based passive leg raise test. Intensive Care Med 2010, 36(11):1875–1881. [DOI] [PubMed] [Google Scholar]
- 11.Squara P, Rotcajg D, Denjean D, et al. : Comparison of monitoring performance of Bioreactance vs. pulse contour during lung recruitment maneuvers. Crit Care 2009, 13(4):R125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Raval NY, Squara P, Cleman M, et al. : Multicenter evaluation of noninvasive cardiac output measurement by bioreactance technique. J Clin Monit Comput 2008, 22(2):113–119. [DOI] [PubMed] [Google Scholar]
- 13.Berlin DA, Peprah-Mensah H, Manoach S, et al. Agreement of Bioreactance Cardiac Output Monitoring With Thermodilution During Hemorrhagic Shock and Resuscitation in Adult Swine. Crit Care Med 2017, 45(2):e195–e201. [DOI] [PubMed] [Google Scholar]
- 14.Keren H, Burkhoff D, Squara P: Evaluation of a noninvasive continuous cardiac output monitoring system based on thoracic bioreactance. Am J Physiol Heart Circ Physiol 2007, 293(1):H583–589. [DOI] [PubMed] [Google Scholar]
- 15.Cecconi M, Parsons AK, Rhodes A: What is a fluid challenge? Curr Opin Crit Care 2011, 17(3):290–295. [DOI] [PubMed] [Google Scholar]
- 16.Mohammadinejad P, Hossein-Nejad H: Calculation of corrected flow time: Wodey’s formula vs. Bazett’s formula. J Crit Care 2017, 44:154–155. [DOI] [PubMed] [Google Scholar]
- 17.Wodey E, Carre F, Beneux X, et al. : Limits of corrected flow time to monitor hemodynamic status in children. J Clin Monit Comput 2000, 16(3):223–228. [DOI] [PubMed] [Google Scholar]
- 18.DeLong ER, DeLong DM, Clarke-Pearson DL: Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics 1988, 44(3):837–845. [PubMed] [Google Scholar]
- 19.Mehta Y, Arora D: Newer methods of cardiac output monitoring. World J Cardiol 2014, 6(9):1022–1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zieleskiewicz L, Muller L, Lakhal K, et al. : Point-of-care ultrasound in intensive care units: assessment of 1073 procedures in a multicentric, prospective, observational study. Intensive Care Med 2015, 41(9):1638–1647. [DOI] [PubMed] [Google Scholar]
- 21.Kwon SH, Gopal AS: 3D and 4D Ultrasound: Current Progress and Future Perspectives. Curr Cardiovasc Imaging Rep 2017, 10(12):43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Feissel M, Michard F, Faller JP, et al. : The respiratory variation in inferior vena cava diameter as a guide to fluid therapy. Intensive Care Med 2004, 30(9):1834–1837. [DOI] [PubMed] [Google Scholar]
- 23.Feissel M, Michard F, Mangin I, et al. : Respiratory changes in aortic blood velocity as an indicator of fluid responsiveness in ventilated patients with septic shock. Chest 2001, 119(3):867–873. [DOI] [PubMed] [Google Scholar]
- 24.Swenson JD, Harkin C, Pace NL, et al. : Transesophageal echocardiography: an objective tool in defining maximum ventricular response to intravenous fluid therapy. Anesth Analg 1996, 83(6):1149–1153. [DOI] [PubMed] [Google Scholar]
- 25.Beaulieu Y: Bedside echocardiography in the assessment of the critically ill. Crit Care Med 2007, 35(5 Suppl):S235–249. [DOI] [PubMed] [Google Scholar]
- 26.Lamia B, Ochagavia A, Monnet X, et al. : Echocardiographic prediction of volume responsiveness in critically ill patients with spontaneously breathing activity. Intensive Care Med 2007, 33(7):1125–1132. [DOI] [PubMed] [Google Scholar]
- 27.Marik PE, Levitov A, Young A, et al. : The use of bioreactance and carotid Doppler to determine volume responsiveness and blood flow redistribution following passive leg raising in hemodynamically unstable patients. Chest 2013, 143(2):364–370. [DOI] [PubMed] [Google Scholar]
- 28.Wong LS, Yong BH, Young KK, et al. : Comparison of the USCOM ultrasound cardiac output monitor with pulmonary artery catheter thermodilution in patients undergoing liver transplantation. Liver Transpl 2008, 14(7):1038–1043. [DOI] [PubMed] [Google Scholar]
- 29.de Waal KA: The methodology of Doppler-derived central blood flow measurements in newborn infants. Int J Pediatr 2012, 2012:680162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Monnet X, Rienzo M, Osman D, et al. : Esophageal Doppler monitoring predicts fluid responsiveness in critically ill ventilated patients. Intensive Care Med 2005, 31(9):1195–1201. [DOI] [PubMed] [Google Scholar]
- 31.Vallee F, Fourcade O, De Soyres O, et al. : Stroke output variations calculated by esophageal Doppler is a reliable predictor of fluid response. Intensive Care Med 2005, 31(10):1388–1393. [DOI] [PubMed] [Google Scholar]
- 32.Mackenzie DC, Noble VE: Assessing volume status and fluid responsiveness in the emergency department. Clin Exp Emerg Med 2014, 1(2):67–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yang SY, Shim JK, Song Y, et al. : Validation of pulse pressure variation and corrected flow time as predictors of fluid responsiveness in patients in the prone position. Br J Anaesth 2013, 110(5):713–720. [DOI] [PubMed] [Google Scholar]
- 34.Lee JH, Kim JT, Yoon SZ, et al. : Evaluation of corrected flow time in oesophageal Doppler as a predictor of fluid responsiveness. Br J Anaesth 2007, 99(3):343–348. [DOI] [PubMed] [Google Scholar]
- 35.Su BC, Luo CF, Chang WY, et al. : Corrected flow time is a good indicator for preload responsiveness during living donor liver donation. Transplant Proc 2014, 46(3):672–674. [DOI] [PubMed] [Google Scholar]
- 36.Antiperovitch P, Iliescu E, Chan B: Carotid systolic flow time with passive leg raise correlates with fluid status changes in patients undergoing dialysis. J Crit Care 2017, 39:83–86. [DOI] [PubMed] [Google Scholar]
- 37.Jalil B, Thompson P, Cavallazzi R, et al. : Comparing Changes in Carotid Flow Time and Stroke Volume Induced by Passive Leg Raising. Am J Med Sci 2018, 355(2):168–173. [DOI] [PubMed] [Google Scholar]
- 38.Shokoohi H, Berry GW, Shahkolahi M, et al. : The diagnostic utility of sonographic carotid flow time in determining volume responsiveness. J Crit Care 2017, 38:231–235. [DOI] [PubMed] [Google Scholar]
- 39.Singer M: The FTc is not an accurate marker of left ventricular preload. Intensive Care Med 2006, 32(7):1089; author reply 1091. [DOI] [PubMed] [Google Scholar]
- 40.Singer M, Bennett ED: Noninvasive optimization of left ventricular filling using esophageal Doppler. Crit Care Med 1991, 19(9):1132–1137. [DOI] [PubMed] [Google Scholar]
- 41.Ma IWY, Caplin JD, Azad A, et al. : Correlation of carotid blood flow and corrected carotid flow time with invasive cardiac output measurements. Crit Ultrasound J 2017, 9(1):10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mackenzie DC, Khan NA, Blehar D, et al. : Carotid Flow Time Changes With Volume Status in Acute Blood Loss. Ann Emerg Med 2015, 66(3):277–282 e271. [DOI] [PubMed] [Google Scholar]
- 43.Hossein-Nejad H, Mohammadinejad P, Lessan-Pezeshki M, et al. : Carotid artery corrected flow time measurement via bedside ultrasonography in monitoring volume status. J Crit Care 2015, 30(6):1199–1203. [DOI] [PubMed] [Google Scholar]
- 44.Blehar DJ, Glazier S, Gaspari RJ: Correlation of corrected flow time in the carotid artery with changes in intravascular volume status. J Crit Care 2014, 29(4):486–488. [DOI] [PubMed] [Google Scholar]
- 45.Marque S, Cariou A, Chiche JD, et al. : Comparison between Flotrac-Vigileo and Bioreactance, a totally noninvasive method for cardiac output monitoring. Crit Care 2009, 13(3):R73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Spiess BD, Patel MA, Soltow LO, et al. : Comparison of bioimpedance versus thermodilution cardiac output during cardiac surgery: evaluation of a second-generation bioimpedance device. J Cardiothorac Vasc Anesth 2001, 15(5):567–573. [DOI] [PubMed] [Google Scholar]
- 47.Duus N, Shogilev DJ, Skibsted S, et al. : The reliability and validity of passive leg raise and fluid bolus to assess fluid responsiveness in spontaneously breathing emergency department patients. J Crit Care 2015, 30(1):217 e211–2s15. [DOI] [PubMed] [Google Scholar]
- 48.Marik PE: Noninvasive cardiac output monitors: a state-of the-art review. J Cardiothorac Vasc Anesth 2013, 27(1):121–134. [DOI] [PubMed] [Google Scholar]
- 49.Barjaktarevic I, Chiem A, Cannesson M: Time to correct the flow of corrected flow time. Crit Ultrasound J 2017, 9(1):18. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Figure 1. A) The patient is maintained in a semi-recumbent (45o angle of the bed) position with semi-extended neck and head partially rotated away from the operator at least 10 minutes before the measurement and PLR maneuver. The operator, using vascular probe of an ultrasound, localizes carotid artery in a transverse view. B) and C) Maintaining the view of carotid artery, the operator rotates the probe to establish a longitudinal view of the artery. D) Using Doppler mode in the established longitudinal view (upper part of the screen image), the operator then obtains adequate pulse waveform velocities (lower part of the screen image). When it is the appropriate time make measurements, the operator saves the image at the end of patient’s exhalation, and then carefully measures the duration of a cardiac cycle (beginning of systole to the end of diastole) and the systolic time (beginning of systole to the end of systole). The measurements are recorded, and ccFT is calculated using Wodey’s formula,. The system used in our study had preset parameters to identify “Systolic Time” and “Cycle Time” and a pre-programmed calculation to deliver the value of “Corrected Doppler Flow Time”. E) After completion of the baseline measurement, the assessment and measurement is repeated during the PLR maneuver as the patient lays in the in a supine position with legs being passively elevated at 45o angle and supported with a premade inflated wedge pillow. Upon initiation of the PLR, the operator will again identify the carotid artery, repeat the steps described above and, then captures a snapshot at the end of exhalation 2 minutes after the initiation of PLR. F) The measurements of systolic and cycle duration times are repeated, and the difference between ccFTBaseline and ccFTPLR represents the ΔccFT.
Supplemental Figure 2. Consort Diagram
Supplemental Figure 3. Bland-Altman plot for bedside and blinded correlation
Supplemental Table 1. The effect of mechanical ventilation and high PEEP on the test ability to predict fluid responsiveness: there is no difference in the test applicability based on Invasive positive pressure mechanical ventilation vs. negative pressure ventilation, passive positive pressure ventilation in comparison to spontaneously breathing patients or positive end-expiratory pressure >5cmH2O vs. ≤5cmH2O