Abstract
Purpose:
Serum platinum is measurable for years after completion of cisplatin-based chemotherapy (CBC). We report the largest investigation of serum platinum levels to date of 1,010 testicular cancer survivors (TCS) assessed 1-35 years after CBC and evaluate genetic contributions to these levels.
Experimental Design:
Eligible TCS given 300 or 400 (±15) mg/m2 cisplatin underwent extensive audiometric testing, clinical examination, completed questionnaires and had crude serum platinum levels measured. Associations between serum platinum and various risk factors and toxicities were assessed after fitting a bi-exponential model adjusted for follow-up time and cumulative cisplatin dose. A genome-wide association study (GWAS) was performed using the serum platinum residuals of the dose and time-adjusted model.
Results:
Serum platinum levels exceeded the reference range for approximately 31 years, with a strong inverse relationship with creatinine clearance at follow-up (age-adjusted p = 2.13×10−3). We observed a significant, positive association between residual platinum values and luteinizing hormone (age-adjusted p=6.58×10−3). Patients with high residual platinum levels experienced greater Raynaud’s phenomenon than those with medium or low levels (age-adjusted ORhigh/low = 1.46; p = 0.04), as well as a higher likelihood of developing tinnitus (age-adjusted ORhigh/low = 1.68, p = 0.07). GWAS identified one single nucleotide polymorphism (SNP) meeting genome-wide significance rs1377817 (p=4.6×10−8, a SNP intronic to MYH14).
Conclusions:
This study indicates that residual platinum values are correlated with several cisplatin-related toxicities. One genetic variant is associated with these levels.
Introduction
Advances in chemotherapy have markedly improved overall cancer survival, enabling more patients to live decades after completion of chemotherapeutic treatments. However, survivors often suffer from severe off-target toxicities due to chemotherapy. These toxicities can limit clinical use, compromise efficacy, and many can permanently impact survivors’ quality of life. As the number of long-term cancer survivors has continued to increase, the health and financial burdens associated with chemotherapy-induced adverse sequelae have become increasingly prevalent (1–3).
This clinical problem is exemplified by cisplatin, a widely used platinating agent that is effective against several adult-onset and pediatric malignancies. High five-year relative survival rates follow cisplatin-based therapy for a number of tumors (4), including testicular cancer (95%), hepatoblastoma (> 80%), medulloblastoma (70-80%), and osteosarcoma (60-80%). Unfortunately, cisplatin also elicits a number of debilitating side effects, including ototoxicity, neurotoxicity, nephrotoxicity, cardiometabolic toxicities, and secondary malignancies (5, 6). These toxicities can be progressive and irreversible, leading to chronic health conditions in young cancer survivors. For example, approximately 18% of adults (7) and 7-22% of children (8) are left with severe to profound of hearing loss. In addition, 56.2% of testicular cancer survivors given a median dose of 400 mg/m2 report symptoms of sensory neuropathy at a median of 5 years after treatment, with 12.5% reporting severe symptoms (9). Reduced renal function is also detected in 25-35% of patients after one cisplatin dose (10). While the majority of patients fully recover from the initial onset of nephrotoxicity, both progressive and irreversible nephrotoxicity have been reported (11, 12).
Several studies have shown detectable tissue platinum (13) and platinum-DNA adducts (14) years after cisplatin therapy. A 13-28 year follow-up study demonstrated that circulating platinum persisted for decades after treatment (15). Serum platinum levels have been shown to remain up to 1,000 times higher than normal for 20 years after completion of therapy (15–18). One study demonstrated that about 10% of circulating serum platinum remains reactive (19).
Circulating platinum has been evaluated for its contribution to the severity and persistence of cisplatin-induced neurotoxicity (paresthesias, neuropathy, and Raynaud’s phenomenon), nephrotoxicity, and ototoxicity (hearing loss and tinnitus). Inconsistent associations have been found between serum platinum levels and cisplatin-induced toxicities. Sprauten et al. (20) identified significant associations between crude serum platinum levels (defined as total platinum concentration) and symptoms of paresthesias, Raynaud’s phenomenon, and tinnitus in 169 testicular cancer survivors (TCS). Hjelle et al. (17) found crude serum platinum levels to be significantly associated with tinnitus and elevated concentrations of luteinizing hormone (LH), but not with paresthesias and Raynaud’s phenomenon in 292 TCS after adjusting for cisplatin dose. A study that longitudinally assessed 77 TCS regarding declines in crude serum platinum levels and toxicities counter-intuitively showed larger declines in platinum levels related to worsening of tinnitus and hand paresthesias (15), perhaps because larger declines are correlated with less time since treatment. Importantly, none of these studies accounted for the kinetics of metabolism and clearance by accounting for variance in time since treatment. A longitudinal pharmacokinetic study serially measuring serum and urine platinum found associations of area-under-the-platinum-exposure-curve with neuropathy, but not tinnitus or Raynaud’s phenomenon, and noted new associations with hypertension, hypercholesterolemia, and hypogonadism (21). This study, however, had a relatively small sample size (n = 99 TCS) and did not compare platinum exposure to an unexposed control group.
In this study, we evaluate clinical correlates with serum platinum levels after constructing a dose- and follow-up time-adjusted pharmacokinetic model in a cohort of 1,010 TCS characterized for variables on diagnosis, treatment, medical history, lifestyle and behavioral factors, and comorbidities over a wide range of follow-up periods (1-35 years). We then use the model to examine the association between serum platinum levels and late adverse events associated with cisplatin toxicity. In addition, we perform a genome-wide association study (GWAS) to assess genetic contributions to our phenotype derived from serum platinum levels.
Patients and Methods
Patients and Data Collection.
All patients were enrolled in The Platinum Study, a cross-sectional study including eight cancer centers in the United States and Canada (5). Eligibility criteria were previously described (9, 22). Briefly, during routine follow-up, eligible TCS underwent physical examination/phlebotomy and completed questionnaires as well as blood collection for serum platinum levels. Data relating to germ cell tumor diagnosis and treatment were abstracted from medical records using standardized forms described previously (23). All abstractors participated in centralized, in-person training (23). Study procedures were approved by the Human Subjects Review Board at each institution, and all patients provided written consent for participation in study procedures, including genetic analyses. The studies were conducted in accordance with recognized ethical guidelines (U.S. Common Rule).
Serum Platinum Quantification.
The concentration of serum platinum was quantified in two batches. For the first batch, 50 μL of serum at 10x dilution were aliquoted into 96-well plates, and a low-volume autosampler (Teledyne Technologies, Thousand Oaks, CA) introduced aliquots into an Agilent 7900 for inductively coupled plasma mass spectrometry [ICP-MS] (Agilent Technologies, Santa Clara, CA). For the second set, samples were diluted 10x and measured on the NexION 2000C for ICP-MS. Both batches utilized an iridium internal standard for calibration. Seven non-zero calibration standards ranging from 0.01 ng/L to 100 ng/L of Pt, spiked with 20 ng/L iridium for internal standardization, were analyzed every 10 samples with weighted linear regression (1/standard deviation of triplicate sample readings as weight-factor). Linearity of calibrations was 0.9999 ± 0.0001. Method detection limit for platinum (NexIon 2000C) was 0.006 ng/L following Long and Winefordner (24). Lower limit of quantification (LLOQ) was 0.010 ng/L translating to an absolute LLOQ of 1 pg Pt. Carry-over was measured from blanks analyzed post-analysis of 100 ng/L standard (Greater Limit of Quantification) and a 35-s rinse with measured blanks below method detection limit. All samples were analyzed in a single day so no inter-day variation is noted. Within day analytical accuracy was determined by comparison of measured replicates of human serum (NIST 909c) spiked with a known amount platinum (5 ng/L) resulting in % error of 0.02. Precision, based on standard deviation of triplicate analyses of samples and standards was better than 0.025 (relative standard deviation, 0.17%).
To evaluate inter-batch consistency, 50 samples were measured using both methods and produced a correlation coefficient of 0.94. Batches were therefore combined for analysis and the mean value of duplicates was taken following data normalization. The greater limit of quantification of both methods was used (5 ng/L), and the nonparametric reference interval for serum platinum that was previously established based on 147 non-platinum treated patient samples was used (25). This was determined as the 2.5th-97.5th percentile of 147 non-platinum treated serum samples (22).
Pharmacokinetic Modeling.
Given the cross-sectional study design, we constructed a bi-exponential model accounting for time since treatment completion and cumulative cisplatin dose. Most patients received a cumulative cisplatin dose of 300 or 400 mg/m2. Adding a 15 mg/m2 margin enabled the inclusion of an additional 56 patients (5.5%). Therefore, we treated dose as a dichotomous variable of 300±15 or 400±15 mg/m2 and excluded remaining patients (n=55). Prior to model fitting, cumulative cisplatin dose was taken into account by multiplying the serum platinum levels of 400±15 mg/m2 patients by 0.75, enabling normalization to those who received 300±15 mg/m2. We fit the following bi-exponential model to serum platinum levels:
where A, α, B, and ß are parameters to be estimated in the bi-exponential model, and t is years since treatment completion. Multiplicative residuals were calculated by dividing the observed serum platinum values by values expected from the fitted bi-exponential model, which were then log-transformed for normalization. These residuals are referred to as the residual platinum value. We also generated an ordinal version of residual platinum values by stratifying values based on their deviation from the mean to create three levels: “medium” (regression residuals = 0 ± 1 standard deviation [SD]), “low” (residuals < −1 SD), and “high” (residuals > 1 SD).
Patient-Reported Outcomes and Medical Records Data Abstraction.
As previously described (7, 23), patients completed questionnaires ascertaining neurotoxic and other symptoms, lifestyle habits, comorbidities, and medication use. Participant responses to questionnaires were mapped to individual adverse health outcomes and graded according to severity on a 0 to 4-point scale using a modified version of the National Cancer Institute Common Terminology Criteria for Adverse Events (version 4.03) as in prior studies (23, 26, 27). The cumulative burden of morbidity (CBM) score for cisplatin-induced toxicities was discerned by using adverse health outcomes previously related to cisplatin exposure (i.e., peripheral sensory neuropathy, hearing damage, tinnitus, and kidney disease) using a modified version described by Kerns et al (23) that removed autonomic neuropathy.
Peripheral sensory neuropathy was evaluated as previously described using nine items in the EORTC-CIPN20. Briefly, an ordinal (0-3) scale was constructed after taking the mean of symptom severity: 0 for “none”, 1 for “a little”, 2 for “quite a bit”, and 3 for “very much.” Groups 2 and 3 were combined. Raynaud’s phenomenon and tinnitus were evaluated with the validated Scale for Chemotherapy-Induced Neurotoxicity as previously described (28). Hearing loss was measured by extensive quantitative audiometry as previously described (7). Briefly, measured hearing thresholds in frequencies that showed statistically significant dose-response relationships with cumulative cisplatin dose (4-12 kHz) were geometrically averaged and rank normalized from age matched controls reported by Engdahl et al., (29) before testing for genetic associations (22). Hypogonadism was defined as testosterone levels ≤ 3 ng/mL based on crude measurement or whether the patient was on testosterone therapy. All patients who had testosterone levels > 3 ng/mL and were not on testosterone therapy were labeled as normal or high, and were grouped together as controls for the linear regression analysis, as previously described (30).
We defined lifestyle habits based on patient responses to The Platinum Study questionnaire, as previously described (9). Briefly, alcohol consumption was assessed as the response to the question “During the past year, how many drinks of alcoholic beverage have you consumed on average? (1 drink = 12 oz. beer [1 can or bottle], 4 oz. glass of wine, 1 mixed drink or shot of liquor)” with the following options: Rarely/never (0), 1-3/month (1), 1/week (2), 2-4/week (3), 5-6/week (4), 1/day (5), 2-3/day (6), 4-5/day (7), and 6+/day (8). Tobacco use was assessed as the response to the following two questions: “Have you ever smoked cigarettes?” with “Yes” (1) and “No” (0) options and “Do you currently smoke cigarettes?”
Statistical Analysis.
The residual platinum value was treated both continuously and ordinally (low, medium, and high, based on the deviation from the mean). Simple and multiple linear regressions were used to evaluate associations between the continuous residual platinum values (dependent variable) and comorbidities and risk factors. Simple and age-adjusted multinomial logistic regressions were used when residual platinum values were specified ordinally, with low residual platinum values designated as the reference group. When conceptually appropriate, age at diagnosis was added as a covariate. All statistical models were fit using R version 3.3.2 (http://www.R-project.org/). Statistical significance was set at p < 0.05.
Genotyping and Genome-Wide Association Study.
DNA was extracted from peripheral blood. Genotyping was performed on the HumnaOmniExpressExome chip (Set 1 was genotyped on the HumanOmniExpressExome-8v1-2_A chip; set 2 was genotyped on the InfiniumOmniExpressExome-8v1-3_A chip; Illumina, San Diego, CA) at the RIKEN Center (Yokohama, Japan) as previously described (9, 22). Sample-level quality control (QC) criteria included: sample call rate > 0.99, pairwise identity by descent < 0.125, coefficient of inbreeding F < 6 standard deviations from the mean, and genetically European as determined by principal component analysis (performed using SMARTPCA). Single nucleotide polymorphism (SNP)-level QC included: call rate > 0.99, and Hardy-Weinberg equilibrium (Chi-squared P < 1 × 10−6). Imputation was done on the University of Michigan Imputation Server. SNPs and samples passing QC criteria comprised the input set for imputation with EAGLE phasing, performed on the Michigan Imputation Server using the Haplotype Reference Consortium (31–33). GWAS assumed linear additive SNP effects and was adjusted for age at diagnosis and the first 10 genetic principal components. Significance was set to p ≤ 5 × 10-8. Following GWAS, coding genes (n = 18,741) were annotated with the p-value of the most significant SNP in the gene or within 25 kilobases (kb) using gencode v18. SNPs with imputation R2 < 0.8, MAF < 0.01, and INFO scores > 1.05 or < 0.3 were excluded. Only subjects who passed QC were included in the GWAS (Supplemental Figure 1). Narrow-sense heritability was estimated with a genetic relationship matrix variance component model as implemented by GCTA with the same covariates as GWAS. The GWAS included 909 subjects with 7,305,641 SNPs.
Results
Cohort Characteristics.
Demographic and clinical characteristics for the entire cohort of TCS, as well as for each residual platinum value category, are provided in Table 1 and Supplemental Table 1. The median age at diagnosis and evaluation was 31 (range: 15-54) years and 37 (range: 18-75) years, respectively. Patients were treated with the following regimens: BEP (bleomycin, etoposide, and cisplatin; 54.0%), EP (etoposide and cisplatin; 30%), VIP (etoposide, ifosfamide, and cisplatin; 2.5%), VeIP (vinblastine, ifosfamide, and cisplatin; 0.1%), and other (unspecified cisplatin-based chemotherapy; 13.4%). Of the 1,010 TCS, 436 and 574 (43.2% and 56.8%, respectively) were treated with 300±15 and 400±15 mg/m2 cisplatin, respectively. Median follow-up time was 4.5 years (range 1-35 years).
Table 1.
Clinical and Sociodemographic Characteristics for 1,010 Testicular Cancer Survivors According to Residual Platinum Values.
| Residual Platinum Value | ||||
|---|---|---|---|---|
| Characteristic | All Patients (n=1,010) | Low (n=118) | Medium (n=785) | High (n=107) |
| Age at GCT diagnosis (years) | ||||
| Median (range) | 31 (15-54) | 30 (15-53) | 31 (15-54) | 35 (16-50) |
| <20 | 70 (6.9%) | 16 (13.6%) | 49 (6.2%) | 5 (4.7%) |
| 20-29 | 390 (38.6%) | 52 (44.1%) | 311 (39.6%) | 27 (25.2%) |
| 30-39 | 351 (34.8%) | 32 (27.1%) | 273 (34.8%) | 46 (43.0%) |
| 40-55 | 199 (19.7%) | 18 (15.3%) | 152 (19.4%) | 29 (27.1%) |
| Time from diagnosis to clinical evaluation (years) | ||||
| Median (range) | 4.5 (0.6-35.3) | 12.2 (0.6-30.3) | 4.3 (0.8-30.6) | 3.4 (1.1-35.3) |
| ≤1 | 4 (0.3%) | 2 (1.7%) | 2 (0.3%) | 0 (0%) |
| >1 and ≤5 | 539 (53.4%) | 35 (29.7%) | 437 (55.7%) | 67 (62.6%) |
| >5 and ≤10 | 243 (24.1%) | 14 (11.9%) | 206 (26.2%) | 23 (21.5%) |
| >10 and ≤20 | 181 (17.9%) | 48 (40.7%) | 122 (15.5%) | 11 (10.3%) |
| >20 | 43 (4.3%) | 19 (16.1%) | 18 (2.3%) | 6 (5.6%) |
| Treatment regimena,b | ||||
| BEP | 545 (54.0%) | 72 (61.0%) | 429 (54.7%) | 44 (41.1%) |
| EP | 303 (30.0%) | 29 (24.6%) | 232 (29.6%) | 42 (39.3%) |
| VIP | 25 (2.5%) | 6 (5.1%) | 15 (1.9%) | 4 (3.7%) |
| VeIP | 1 (0.1%) | 0 (0%) | 1 (0.1%) | 0 (0%) |
| Other (includes cisplatin) | 135 (13.4%) | 11 (9.3%) | 107 (13.6%) | 17 (15.9%) |
| Cumulative cisplatin dose (mg/m2) | ||||
| Median (range) | 400 (286-414) | 400 (292-406) | 400 (286-414) | 400 (297-400) |
| <300 | 20 (2.0%) | 1 (0.9%) | 18 (2.3%) | 1 (0.9%) |
| 300 | 406 (40.2%) | 47 (39.8%) | 317 (40.4%) | 42 (39.3%) |
| >300 and <400 | 28 (2.8%) | 5 (4.2%) | 21 (2.7%) | 2 (1.9%) |
| 400 | 548 (54.3%) | 64 (54.2%) | 422 (53.8%) | 62 (57.9%) |
| >400 | 8 (0.8%) | 1 (0.9%) | 7 (0.9%) | 0 (0%) |
| Audiometrically assessed hearing lossc,d | ||||
| None | 125 (17.1%) | 15 (16.0%) | 98 (17.6%) | 12 (15.4%) |
| Mild | 162 (22.2%) | 17 (18.1%) | 127 (22.8%) | 18 (23.1%) |
| Moderate | 111 (15.2%) | 18 (19.1%) | 81 (14.5%) | 12 (15.4%) |
| Moderately severe | 154 (21.1%) | 17 (18.1%) | 122 (21.9%) | 15 (19.2%) |
| Severe/profound | 178 (24.4%) | 27 (28.7%) | 130 (23.3%) | 21 (26.9%) |
| Tinnituse | ||||
| Yes | 355 (37.2%) | 38 (33.0%) | 271 (36.7%) | 46 (45.5%) |
| No | 599 (62.8%) | 77 (67.0%) | 467 (63.3%) | 55 (54.5%) |
| Peripheral sensory neuropathyf,g | ||||
| None | 429 (43.1%) | 52 (44.1%) | 343 (45.3%) | 9 (9.5%) |
| A little | 472 (47.4%) | 57 (48.3%) | 361 (47.6%) | 69 (72.6%) |
| Quite a bit/very much | 95 (9.5%) | 9 (7.6%) | 54 (7.1%) | 17 (17.9%) |
| Raynaud’s phenomenong,h | ||||
| None | 634 (64.0%) | 80 (67.8%) | 497 (64.5%) | 57 (55.3%) |
| A little | 205 (20.7%) | 27 (22.9%) | 154 (20.0%) | 24 (23.3%) |
| Quite a bit/very much | 152 (15.3%) | 11 (9.3%) | 119 (15.5%) | 22 (21.4%) |
Abbreviations: BEP: bleomycin, etoposide, and cisplatin; EP: etoposide and cisplatin; VIP: etoposide, ifosfamide, and cisplatin; VeIP: vinblastine, ifosfamide, and cisplatin
BEP category includes patients who received only bleomycin, etoposide, and cisplatin; EP includes patients who received only etoposide and cisplatin; VIP includes patients who received only etoposide, ifosfamide, and cisplatin; VeIP includes patients who received only vinblastine, ifosfamide, and cisplatin. The other category includes patients who received an unspecified cisplatin-based treatment regimen.
1 participant was missing data for treatment regimen.
280 participants did not have hearing assessed audiometrically.
ASHA criteria defined hearing loss severity as the following: mild: 21 to 40 dB; moderate: 41 to 55 dB; moderately severe: 56 to 70 dB; severe: 71 to 90 dB; and profound: > 90 dB; for at least one tested frequency for either ear (https://www.asha.org/public/hearing/Degree-of-Hearing-Loss).
56 participants did not report tinnitus status.
14 participants did not report peripheral sensory neuropathy status.
Following conversion of the Likert scale: “none, a little, quite a bit, very much” to a 0-3 numeric scale, each individual was attributed a summary statistic for the sensory subscale (Cronbach α = 0.88) and the motor subscale (α = 0.78) by taking the mean of the response in the subscale: none (mean = 0), mild (0 < mean ≤ 1), severe (mean > 1), as in Dolan et al (9).
19 participants did not report Raynaud’s phenomenon status.
Pharmacokinetic Analysis.
Median serum platinum concentration was 309 ng/L (range 7.2-8,252 ng/L; 259 ng/L 6.7-8,252 ng/L after normalization). Only 62 survivors (6.1%) had serum platinum concentrations below 47 ng/L, the upper limit of the normal range determined from non-platinum-treated patient samples. The median follow-up time for this group of survivors was 19.5 years (range 6-31 years, 19.1 years for 300±15 mg/m2 and 20.3 years for 400±15 mg/m2, respectively). Using bi-exponential regression, we estimated Cmax (concentration at time 0) to be 4,720 ng/L. We fit a bi-exponential model to the normalized serum platinum data (Figure 1A). Attempts to use a tri-exponential model resulted in a singular gradient error. When simulating the progression of the tri-exponential model fitting, the estimates for the tri-exponential parameters became unreasonably high, indicating that addition of the tri-exponential parameters produced an overfitted model (data not shown). Consequently, it appeared that the two-compartment model was sufficient to model the decay of serum platinum over time.
Figure 1. Population Pharmacokinetic Modeling of Long-Term Serum Platinum Levels.
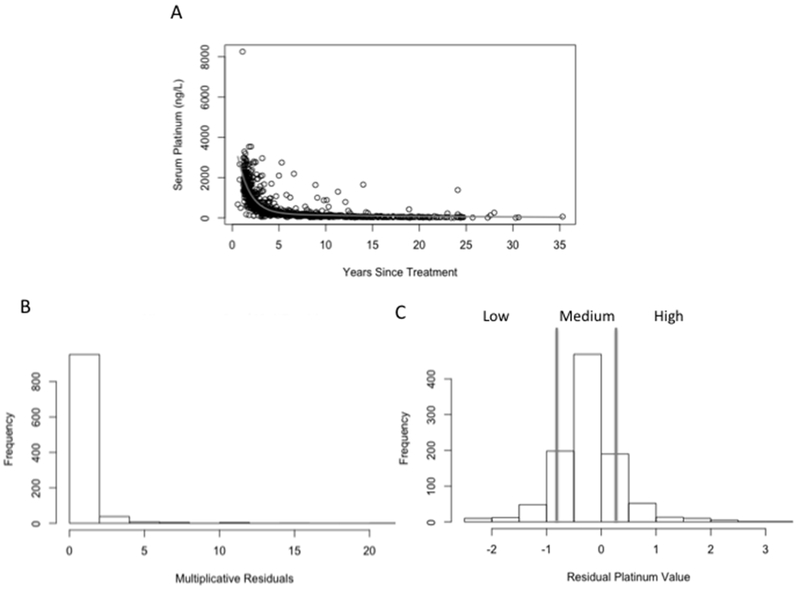
A) Serum platinum levels from 1,010 TCS were fitted to a bi-exponential model in which years since treatment completion was taken into account. Cumulative cisplatin dose was taken into account by multiplying the serum platinum levels of 400 ± 15 mg/m2 patients by 0.75, enabling normalization to 300 ± 15 mg/m2 patients prior to model fitting. B) Histogram of multiplicative residuals from the bi-exponential model. C) Residuals were log-transformed to fit a near normal distribution in order to examine the extent to which residual platinum values associate with cisplatin-induced toxicities. The bars denote groups of low, medium and high.
The estimated time to reach the upper limit of the reference range was strikingly high (31 years), and took nearly seven half-lives. Interestingly, the bi-exponential model never reached the seventh half-life of decay (serum platinum = 36.9 ng/L) over the course of 35 years (the length of the longest follow-up period), indicating that patients are likely still at the upper limits of the reference interval nearly four decades after completion of cisplatin-based chemotherapy. Because we could not compute concentration-time AUC with cross-sectional data, we used the log-transformed multiplicative residuals from the fit of the bi-exponential model to the normalized serum platinum data as the exposure phenotype and categorized survivors into low (n=118, 11.7%), medium (n=785, 77.7%), and high (n=107, 10.6%) residual platinum values (Figure 1B and C). Importantly, residual platinum values were highly associated with normalized serum platinum levels obtained from the 1,010 patients (p < 2×10−16), which was comparable to years since treatment completion (p < 2×10−16), and more statistically significant than cumulative cisplatin dose (p = 0.09), weight (p = 0.23), BMI (p = 0.01), age at diagnosis (p = 0.05), age at clinical examination (p = 6.92×10−15), or creatinine clearance (p = 0.04). Therefore, residual platinum values appear to be an accurate predictor of cisplatin clearance.
Predictors, Risk Factors, and Comorbidities.
We found a significant positive association between residual platinum values and age at diagnosis (p = 3.13×10−9), and a strong negative correlation between residual platinum values and creatinine clearance at follow-up (p = 1.99×10−5) (Figure 2A and B, Table 2). This association appears to persist years after cisplatin-based chemotherapy has been completed, as the association between residual platinum values and creatinine clearance remains statistically significant when only evaluating patients who last received cisplatin more than 15 years prior to clinical examination (n = 100; p = 0.01). We also found a significant positive association with LH levels (p = 6.07×10−3; Figure 2C, Table 2). Creatinine clearance and LH levels remained statistically significant after incorporating age at diagnosis as a covariate (Table 2). Hypogonadism was not significantly associated with residual platinum values (β = 0.07, p = 0.22, age-adjusted p = 0.75; Table 2). Hypertension, smoking, excess alcohol, and extent of physical activity were also not significantly associated with residual platinum values (p > 0.05, data not shown).
Figure 2. Linear Regression of Residual Platinum Value and Continuous Variables.
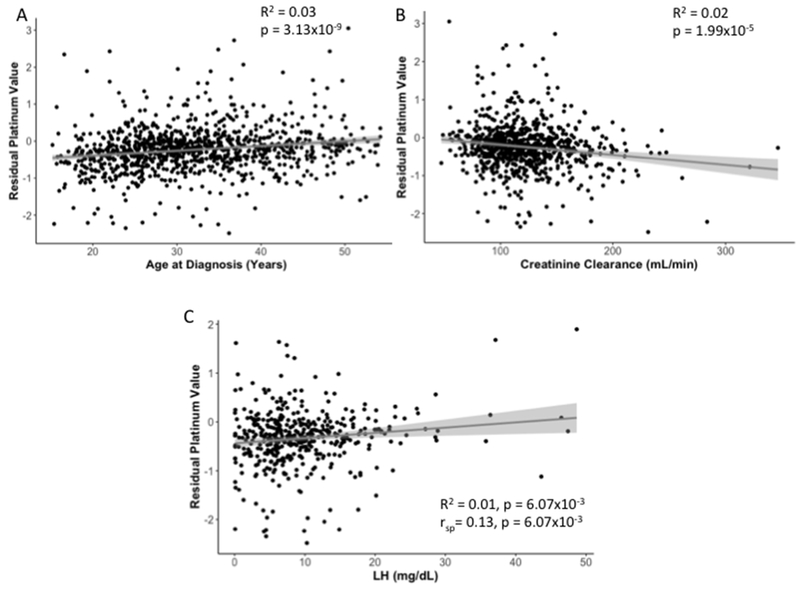
Simple linear regression results are presented for A) age at diagnosis, B) creatinine clearance, and C) LH levels. R2 and p-values for linear regression are reported for all phenotypes. A Spearman rank correlation test (rsp) was also performed on LH levels due to its positive skew distribution, and results are shown in panel C. Fitted linear regression lines are highlighted in gray and 95% confidence intervals are indicated by the light gray shaded regions.
Table 2.
Linear Regression of the Association Between Residual Platinum Values as a Continuous Variable and Phenotypes Relevant to Cisplatin-Based Chemotherapy.
| Linear Regression | Linear Regression with Age at Diagnosis | |||||
|---|---|---|---|---|---|---|
| Variable Type | n | β (95% CI) | p-value | β (95% CI) | p-value | |
| Risk Factors/Comorbidities | ||||||
| Age at Diagnosis | continuous | 1,010 | 0.01 (8.68×10−3, 0.02) | 3.13×10−9 | N/A | N/A |
| Creatinine clearancea | continuous | 769 | −2.67×10−3 (−3.89×10−3, −1.45×10−3) | 1.99×10−5 | −1.95×10−3 (−3.20×10−3, −7.09×10−4) | 2.13×10−3 |
| LDL (mg/dL) | continuous | 580 | 9.12×10−3(−3.61×10−4, 2.18×10−3) | 0.16 | 7.52×10−4 (−5.00×10−4, 2.00×10−3) | 0.24 |
| LH (mg/dL) | continuous | 437 | 0.01 (3.7×10−3, 0.02) | 6.07×10−3 | 0.01 (2.91×10-3, 0.02) | 6.58×10−3 |
| Hypogonadismb,c | categorical | 428 | 0.07 (−0.04, 0.19) | 0.22 | 0.02 (−0.10, 0.13) | 0.75 |
| Testosterone (ng/mL) | continuous | 441 | −2.28×10−3(−7.66×10−3, 3.10×10−3) | 0.41 | −1.68×10−3 (−6.97×10−3, 3.60×10−3) | 0.53 |
| Cisplatin-Induced Toxicities | ||||||
| CBM score for cisplatin-induced toxicitiesd | categorical | 1003 | 0.04 (8.80×10−3, 0.08) | 0.01 | 0.02 (−0.02, 0.05) | 0.28 |
| Peripheral sensory neuropathye | categorical | 999 | 0.08 (0.02, 0.14) | 7.52×10−3 | 0.04 (−0.02, 0.10) | 0.18 |
| Raynaud phenomenone | categorical | 994 | 0.07 (0.02, 0.12) | 9.84×10−3 | 0.05 (4.53×10-3, 0.10) | 0.03 |
| Hearing Loss (rnGM412)f | continuous | 751 | 0.03 (−0.02, 0.08) | 0.21 | −0.05 (−0.11, 6.78×10−5) | 0.05 |
| Tinnitus | categorical | 957 | 0.07 (5.44×10-3, 0.15) | 0.07 | 0.07 (−7.19×10−3, 0.15) | 0.07 |
Abbreviations: CBM: cumulative burden of morbidity; LDL: low-density lipoprotein cholesterol; LH: luteinizing hormone
For the linear regression model, the continuous version of residual platinum values was the dependent variable. The other phenotypes were classified as independent variables, and age at diagnosis was included as a covariate where indicated. Bold indicates p ≤ 0.05; italics indicates 0.05 < p < 0.10.
Was calculated by using the following formula: creatinine clearance = (140- age at clinical examination)*weight (kg) /(72* serum creatinine (mg/dL).
Excluded 54 patients who had measured LH values, but received testosterone therapy.
Defined as testosterone levels ≤ 3 ng/mL based on crude measurement or whether the patient was on testosterone therapy. All patients who had testosterone levels > 3 ng/mL and were not on testosterone therapy were labeled as normal or high, and were grouped together as controls for the linear regression analysis, as in Abu Zaid et al (30).
Calculated by using selected adverse health outcomes previously related to cisplatin exposure (i.e., peripheral sensory neuropathy, hearing damage, tinnitus, and kidney disease), using a modified version of Kerns et al. (23) by removing autonomic neuropathy.
Following conversion of the Likert scale: “none, a little, quite a bit, very much” to a 0-3 numeric scale, each individual was attributed a summary statistic for the sensory subscale (Cronbach α = 0.88) and the motor subscale (α = 0.78) by taking the mean of the response in the subscale: none (mean = 0), mild (0 < mean ≤ 1), severe (mean > 1), as in Dolan et al (9).
Defined by the rank normalized geometric mean of air conduction thresholds measured at 4, 6, 8, 10, and 12 kHz, as in Frisina et al (7).
We found significant positive associations between residual platinum values and several examined drug-induced toxicities, including the CBM score for cisplatin-induced toxicities (β = 0.04, p = 0.01), peripheral sensory neuropathy (β = 0.08, p = 7.52×10−3), and Raynaud’s phenomenon (β = 0.07, p = 9.84×10−3; Table 2). The association with Raynaud’s phenomenon remained statistically significant after adjusting for age at diagnosis (β = 0.05, p = 0.03), while the associations with CBM score (β = 0.02, p = 0.28) and peripheral sensory neuropathy (β = 0.04, p = 0.18) were no longer statistically significant. The association with Raynaud’s phenomenon also remained statistically significant after adjusting for both age and cumulative cisplatin dose (β = 0.05, p = 0.04). Tinnitus demonstrated a marginally significant association with residual platinum values (β = 0.07, p = 0.07, age-adjusted p = 0.07, age and cisplatin dose-adjusted p = 0.07), but audiometric hearing thresholds were not statistically significant (β = 0.03, p = 0.21). Although audiometric hearing thresholds became statistically significant after adjusting for age at diagnosis (β = −0.05, p = 0.05), the β-value changed from positive to negative and by more than 10%, indicating that age at diagnosis is a negative confounder for the association.
We categorized patients into low, medium, or high residual platinum values to assess whether high levels of serum platinum were more associated with cisplatin-induced toxicities than medium or low levels (Supplemental Tables 2 and 3). In considering the severity of the toxicity, a higher proportion of patients who experienced more severe toxicity was demonstrated in the high residual platinum value group when compared to the low or medium groups for CBM score (Figure 3A), peripheral sensory neuropathy (Figure 3B) and Raynaud’s phenomenon (Figure 3C). In addition, patients with high residual platinum values were significantly more likely to have a higher CBM score (ORhigh/low = 1.26, p = 0.05), and peripheral sensory neuropathy (ORhigh/low = 1.61; p = 0.02, Supplemental Table 2), but not after adjusting for age (ORhigh/low = 1.13, p = 0.32 and ORhigh/low = 1.34; p = 0.17, respectively, Supplemental Table 3). Patients with high residual platinum values were significantly more likely to have Raynaud’s phenomenon (age-adjusted ORhigh/low = 1.46; p = 0.04, age and cisplatin dose-adjusted ORhigh/low = 1.45; p = 0.04). In regards to ototoxicity, patients with high residual platinum values had a higher likelihood of developing tinnitus (ORhigh/low = 1.69, p = 0.06), which remained marginally significant after adjusting for age at diagnosis (ORhigh/low = 1.68, p = 0.07), as well as age at diagnosis and cumulative cisplatin dose (ORhigh/low = 1.69, p = 0.07). Audiometric hearing thresholds were not significantly associated with high residual platinum values (ORhigh/low = 1.03, p = 0.86).
Figure 3. Distributions of Cisplatin-Induced Toxicities in Testicular Cancer Survivors Based on Residual Platinum Value.
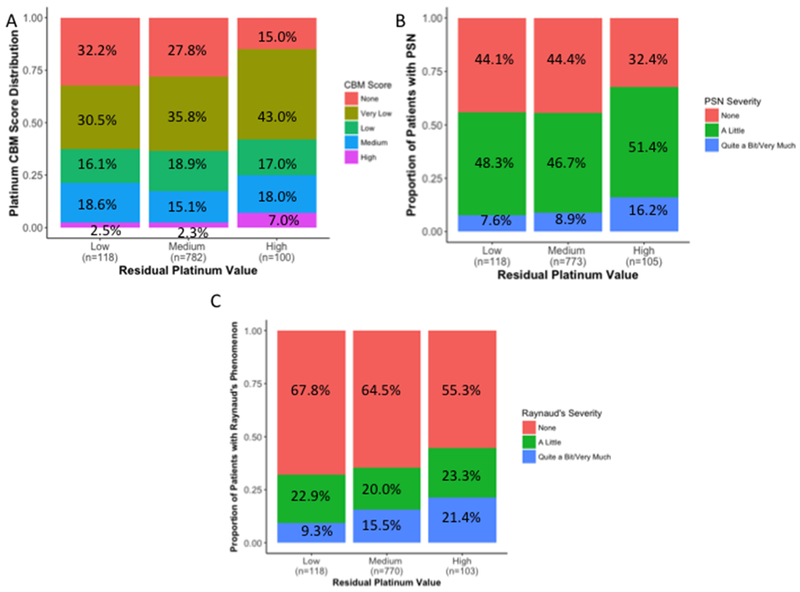
The overall distribution of A) CBM score for cisplatin-induced toxicities (p = 0.06), B) peripheral sensory neuropathy (PSN; p = 0.02), and C) Raynaud’s phenomenon (p = 0.02) in TCS based on having low, medium, and high residual platinum values is provided. Low, medium, and high groups reflect ordinal stratifications of residual platinum values based on their deviation from the mean: “medium” (regression residuals = 0 ± 1 standard deviation [SD]), “low” (residuals < −1 SD), and “high” (residuals > 1 SD). All three toxicities are divided into different degrees of severity, as indicated in the legend, with associated percentages provided in each panel. Sample sizes for each group are indicated within each panel on the x-axis. Differences between the proportions of toxicity severity observed for the low, medium, and high residual platinum value groups were evaluated for statistical significance through the Cochran-Armitage-Mantel 1df chi-square trend test (54).
Genome-Wide Association Study.
GWAS of residual platinum values as a continuous variable identified one SNP that met genome-wide significance: rs1377817 (p = 4.6×10−8; Figure 4A). This SNP is intronic to MYH14, which encodes for a heavy chain of nonmuscle myosin 2. The SNP is in linkage disequilibrium with rs58754699 and rs113890379 (R2 = 1.0, p <0.0001), the next two most statistically significant genetic variants (p = 5.0×10−8 and 1.6×10−7, respectively). The SNP, rs1377817, explained 3.2% of the phenotype’s variance (p = 7.11×10−8). The Q-Q plot in Figure 4B indicates that the observed p-values of associated SNPs deviate significantly from the expected distribution (null hypothesis of no association) of p-values, suggesting that multiple SNPs are associated with the residual platinum value phenotype. Supplemental Table 4 lists all SNPs associated with residual platinum values with p<0.0001. Using GCTA’s linear mixed model approach (34), we found that additive SNP effects did not explain phenotypic variance (h2 = 0.04 ± 0.38, p = 0.45).
Figure 4. Genome-Wide Association Study of Residual Platinum Value as a Continuous Variable.
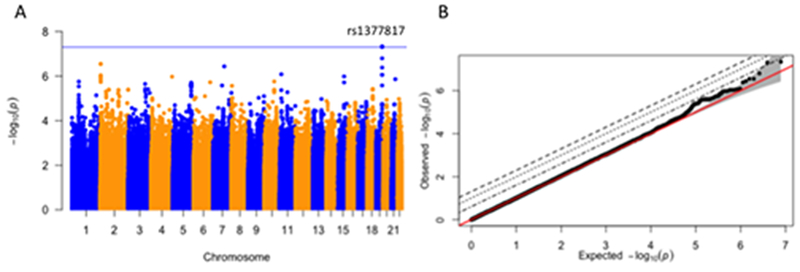
A) Manhattan plot of GWAS results reveals one locus meeting genome-wide significance (p ≤ 5 × 10−8): rs1377817 (p = 4.6 × 10−8). Covariates in the analysis include age at diagnosis and 10 genetic principal components accounting for population substructure. B) Quantile-Quantile plot of GWAS results.
Discussion
In this study, we uniquely interrogate exposure of serum platinum levels in a cohort of 1,010 TCS following near uniform treatment with cisplatin-based chemotherapy and compare this with a platinum-unexposed control group. After normalizing serum platinum concentration, we fit a bi-exponential model with follow-up time as a variable. We define our measure of inter-patient variability in serum platinum as the observed serum platinum value divided by the expected value at the observed follow-up time derived from this model for each patient, followed by log-transformation. We postulate that this phenotype is correlated to the area under the concentration-time curve (AUC), which cannot be directly computed due to the cross-sectional study design. Our analytical method allows the interrogation of time-dependent data without serial sampling. Although the AUC is not directly estimated, the multiplicative residual is theoretically proportional to dose-adjusted AUC, and thus is a plausible marker of cisplatin exposure. From this, we find that serum platinum levels exceed the reference range for approximately 31 years. This is consistent with previous findings indicating long-term detection of serum platinum, but to our knowledge this is the first study to provide an estimated time-to-reference range. We interrogate the association of platinum with toxicities beyond crude serum concentration measurements, the predominant analytical strategy previously used in studies evaluating the relationship of serum platinum levels and toxicities (15, 17, 20). We do so because associations between serum platinum levels and toxicological traits are likely confounded by follow-up time, the strongest predictor of serum platinum concentration. Our data suggest that patients with high serum platinum levels are more susceptible to developing tinnitus and Raynaud’s phenomenon than those with medium or low serum platinum levels, but does not provide robust associations with other toxicities, including cumulative burden of morbidity scores, hearing loss, and peripheral sensory neuropathy. We additionally perform a GWAS to determine the genetic variants associated with residual platinum values.
As expected, we found a strong negative association between chronic serum platinum exposure and creatinine clearance. Cisplatin is excreted through the renal route by glomerular filtration with some tubular secretion, and is also nephrotoxic (35). Therefore, we expect that lower pre-treatment creatinine clearance would contribute to higher chronic serum concentrations of platinum, as found by Boer et al. (21). In addition, we expect that ongoing exposure to higher chronic serum platinum levels could contribute to additional nephrotoxicity. Our significant association with follow-up creatinine clearance values may therefore be due to low renal function (possibly due to the SNP identified in our GWAS intronic to MYH14) raising serum platinum concentrations as well as high serum platinum concentrations lowering renal function. One limitation of our study is the lack of baseline renal function assessments.
In univariate analysis, the serum platinum phenotype was found to be significantly associated with the cumulative morbidity profile and certain toxicities, but robust associations were not detected. Boer et al. (21) identified a significant association with neuropathy; however, our dataset indicates that when age is considered, the association with neuropathy is no longer statistically significant. Taking age into account, Sprauten et al. (20) reported that both paresthesias and Raynaud’s phenomenon were increased two to four-fold in the highest quartile of crude serum platinum concentrations. In evaluating categorized patients (low, medium, or high residual platinum values), patients with high residual platinum values were significantly more likely to have Raynaud’s phenomenon even after adjusting for age.
Interestingly, hearing loss and tinnitus appeared to have contrasting levels of association with residual platinum values. Although the extent of hearing loss did not appear to be associated with residual platinum values, tinnitus did demonstrate a marginal association that was independent of age at diagnosis. Further, multinomial regression indicated that patients with high levels of serum platinum were more likely to develop tinnitus, suggesting these individuals are at an increased risk of developing this off-target toxicity. These data are in accord with Hjelle et al. (17) and Sprauten et al. (20) who both found crude serum platinum levels to be significantly associated with tinnitus after adjusting for cumulative cisplatin dose. Nevertheless, the notable difference in statistical association for hearing loss and tinnitus is surprising in light of the fact that concentrations of platinum in the cochlea decline much slower than in serum, as cisplatin binding to the cochlea is largely irreversible (36). It is plausible that there are differences in the pathophysiology underlying cisplatin-induced hearing loss and tinnitus, as recent studies have indicated the development of tinnitus is more dependent on disruptions in the balance of excitatory and inhibitory nerve transmission within central auditory structures than pathology in the cochlea (37). However, most investigations of cisplatin-associated ototoxicity have focused on hair cell damage in the organ of corti that induces hearing loss (38), and the mechanisms underlying cisplatin-induced tinnitus are still poorly understood. Therefore, further investigation will be needed to discern potential differences regarding the importance of platinum clearance in cisplatin-induced hearing loss and tinnitus.
Our study relied in large part on patient-reported outcomes in the quantification of platinum toxicities. Patient-reported outcomes are increasingly recognized as valid and important and enable a broad interrogation of symptoms across conditions in long-term cancer survivors (39). Additionally, adverse effects like peripheral neuropathy may not be fully captured with objective assessments (40) and may be confounded by physician interpretation on physician-graded scales (41). An NCI Clinical Trials Planning Meeting (42) recently agreed that self-report for neuropathy is superior to exam and recommended EORTC-CIPN20. Self-reported data may also more adequately quantify the perceived impact on quality of life.
Residual platinum values were also associated with increased LH levels, a finding that has been previously noted when assessing either crude serum platinum levels (17) or cumulative cisplatin dose (43). Interestingly, residual platinum values were not associated with decreased testosterone levels or hypogonadism as in Boer et al. (21), which are often observed in TCS due to disturbed endocrine gonadal function that impairs testosterone production (17, 43). However, disturbance of endocrine gonadal function in TCS is also mediated by other factors such as orchiectomy and testicular dysgenesis syndrome (43). Further, the study that initially reported an association between crude serum platinum and LH levels reported no association with testosterone levels (17). Therefore, it is plausible that unaccounted variables we have not considered and different phenotype definitions explain this discrepancy.
In GWAS, we identified a prominent signal in chromosome 19 in which rs1377817 (p=4.6×10−8), a SNP intronic to MYH14, met criteria for genome-wide significance, and had several other top SNPs in linkage disequilibrium. Importantly, MYH14 encodes for a heavy chain that is an integral component of nonmuscle myosin 2, a protein essential for kidney development and function (44, 45). When evaluated in mice, Myh14 was expressed throughout most segments of the renal tubules, and was implicated in the regulation of the renal epithelial transport process (46). Since cisplatin is excreted from the body predominantly through renal clearance, the importance of genetic architecture surrounding kidney development and function is apparent, and could markedly influence long-term platinum kinetics and circulation in serum. Interestingly, a mutation in MYH14 has previously been associated with an autosomal dominant disorder of peripheral neuropathy, myopathy, hoarseness, and hearing loss (47), indicating that the gene could also potentially influence two prominent cisplatin-induced toxicities (peripheral neuropathy and hearing loss).
It is important to note that interpretation of genetic results is especially difficult for serum platinum, as it is a highly complex phenotype. Contributions of DNA damage-related apoptotic pathways, organ development and regeneration, renal function, protein-binding, as well as the pharmacokinetic pathways of cisplatin (absorption, distribution, metabolism, and excretion) are likely all contributing to serum platinum concentrations, but are difficult to measure to enable interpretation of biomolecular mechanisms. A strategy of multiple-tissue sampling and measurement of platinum concentration has been evaluated in rodent models and has generated important insights (36), but this would be difficult in humans.
To our knowledge, at this point, there is no candidate agent that could significantly reduce toxicity without impacting antitumor activity for all cancers treated with cisplatin. Plausible detoxifying agents include antioxidants (48, 49) although potential risks must always be balanced against benefits. In addition, transporter-mediated uptake can be of importance in decreasing off-target toxicities of platinum derivatives and therefore can provide protective intervention, without compromising anticancer efficacy (50–52). Sodium thiosulfate was recently approved to protect against cisplatin-induced hearing loss in children with localized hepatoblastoma (49) and did not hamper therapeutic efficacy in localized hepatoblastoma. However, administration of this compound has been shown to reduce overall survival in pediatric patients with metastatic cancer, which may greatly hamper its clinical applicability (53). Reducing the severity of toxicities and comorbidities associated with cisplatin treatment is an important goal, particularly for children and young adults with many decades of subsequent life.
Conclusion
Our study demonstrates the need to adjust for time since treatment and confirms previous observations that platinum follows a slow elimination and remains in circulation at exceptionally high levels for years after cisplatin treatment. Importantly, residual platinum values were associated with several cisplatin-induced toxicities and associated comorbidities. In addition, our GWAS identified rs1377817 (p=4.6×10−8), a SNP intronic to MYH14, to be associated with residual platinum values, suggesting that genetic variation may predispose certain patients to high residual platinum values for years after treatment has been completed. The genetic association we report requires replication and validation before mechanistic insights can be surmised.
Supplementary Material
Translational Relevance.
Although curative in many settings, cisplatin can elicit severe, often irreversible toxicities that impact long-term survivors’ quality of life. Platinum is detectable in the serum for decades after completion of cisplatin-based chemotherapy and it has been hypothesized that circulating platinum contributes to the severity and persistence of cisplatin-induced toxicities. Results of previous studies have been inconsistent partially due to study design, lack of consideration of time since treatment in the modeling, and the use of relatively small patient cohorts. Here, we report the largest study to date of serum platinum levels in 1,010 testicular cancer survivors, and perform a genome-wide association study to assess genetic contributions to these levels. We find that, on average, it takes decades for patients to reach normal levels of serum platinum. We find modest associations between platinum exposure and toxicities, including tinnitus and Raynaud’s phenomenon. If confirmed, our results suggest that reducing circulating platinum levels in testicular cancer survivors might hold value as a potential strategy to mitigate selected toxicities, and subsequently improve the long-term quality of life in this growing population of survivors.
Acknowledgement.
This work was supported by the National Institutes of Health Genetic Susceptibility and Biomarkers of Platinum‐related Toxicities grant (R01 CA157823; L.Travis, ME Dolan), Pharmacogenomics Research Network-RIKEN Global Alliance (PGRN-RIKEN), which is supported by the RIKEN Center for Integrative Medical Science and the NIH Pharmacogenomics Research Network (GM115370; K. Giacomini, PI; MJ Ratain, ME Dolan) and the University of Chicago Cancer Research Foundation’s Women’s Board.
Abbreviations list:
- BEP
bleomycin, etoposide, and cisplatin
- CBC
cisplatin-based chemotherapy
- CBM
cumulative burden of morbidity
- EP
etoposide and cisplatin
- GWAS
genome-wide association study
- LDL
low-density lipoprotein cholesterol
- LH
luteinizing hormone
- QC
quality control
- SNP
single nucleotide polymorphism
- TCS
testicular cancer survivors
- VeIP
vinblastine, ifosfamide, and cisplatin
- VIP
etoposide, ifosfamide, and cisplatin
- AUC
area under the concentration-time curve
Footnotes
Disclosure of Potential Conflicts of Interest:
The authors declare no potential conflicts of interest.
References
- 1.Hastert TA, Young GS, Pennell ML, Padamsee T, Zafar SY, DeGraffinreid C, Naughton M, Simon M, Paskett ED. Financial burden among older, long-term cancer survivors: Results from the LILAC study. Cancer Med. 2018;7(9):4261–72. 10.1002/cam4.1671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Altice CK, Banegas MP, Tucker-Seeley RD, Yabroff KR. Financial Hardships Experienced by Cancer Survivors: A Systematic Review. J Natl Cancer Inst. 2017;109(2). Epub 2016/10/22. doi: 10.1093/jnci/djw205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yabroff KR, Lawrence WF, Clauser S, Davis WW, Brown ML. Burden of illness in cancer survivors: findings from a population-based national sample. J Natl Cancer Inst. 2004;96(17):1322–30. Epub 2004/09/02. doi: 10.1093/jnci/djh255. [DOI] [PubMed] [Google Scholar]
- 4.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018;68(1):7–30. Epub 2018/01/10. doi: 10.3322/caac.21442. [DOI] [PubMed] [Google Scholar]
- 5.Fung C, Sesso HD, Williams AM, Kerns SL, Monahan P, Abu Zaid M, Feldman DR, Hamilton RJ, Vaughn DJ, Beard CJ, Kollmannsberger CK, Cook R, Althouse S, Ardeshir-Rouhani-Fard S, Lipshultz SE, Einhorn LH, Fossa SD, Travis LB, Platinum Study G. Multi-Institutional Assessment of Adverse Health Outcomes Among North American Testicular Cancer Survivors After Modern Cisplatin-Based Chemotherapy. J Clin Oncol. 2017;35(11):1211–22. Epub 2017/02/28. doi: 10.1200/JCO.2016.70.3108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chovanec M, Abu Zaid M, Hanna N, El-Kouri N, Einhorn LH, Albany C. Long-term toxicity of cisplatin in germ-cell tumor survivors. Ann Oncol. 2017;28(11):2670–9. Epub 2017/10/19. doi: 10.1093/annonc/mdx360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Frisina RD, Wheeler HE, Fossa SD, Kerns SL, Fung C, Sesso HD, Monahan PO, Feldman DR, Hamilton R, Vaughn DJ, Beard CJ, Budnick A, Johnson EM, Ardeshir-Rouhani-Fard S, Einhorn LH, Lipshultz SE, Dolan ME, Travis LB. Comprehensive Audiometric Analysis of Hearing Impairment and Tinnitus After Cisplatin-Based Chemotherapy in Survivors of Adult-Onset Cancer. J Clin Oncol. 2016;34(23):2712–20. Epub 2016/06/30. doi: 10.1200/JCO.2016.66.8822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Knight KR, Chen L, Freyer D, Aplenc R, Bancroft M, Bliss B, Dang H, Gillmeister B, Hendershot E, Kraemer DF, Lindenfeld L, Meza J, Neuwelt EA, Pollock BH, Sung L. Group-Wide, Prospective Study of Ototoxicity Assessment in Children Receiving Cisplatin Chemotherapy (ACCL05C1): A Report From the Children’s Oncology Group. J Clin Oncol. 2017;35(4):440–5. Epub 2016/12/13. doi: 10.1200/JCO.2016.69.2319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dolan ME, El Charif O, Wheeler HE, Gamazon ER, Ardeshir-Rouhani-Fard S, Monahan P, Feldman DR, Hamilton RJ, Vaughn DJ, Beard CJ, Fung C, Kim J, Fossa SD, Hertz DL, Mushiroda T, Kubo M, Einhorn LH, Cox NJ, Travis LB, Platinum Study G. Clinical and Genome-Wide Analysis of Cisplatin-Induced Peripheral Neuropathy in Survivors of Adult-Onset Cancer. Clin Cancer Res. 2017;23(19):5757–68. Epub 2017/06/15. doi: 10.1158/1078-0432.CCR-16-3224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Han X, Yue J, Chesney RW. Functional TauT protects against acute kidney injury. J Am Soc Nephrol. 2009;20(6):1323–32. Epub 2009/05/09. doi: 10.1681/ASN.2008050465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Santoso JT, Lucci JA, 3rd, Coleman RL, Schafer I, Hannigan EV. Saline, mannitol, and furosemide hydration in acute cisplatin nephrotoxicity: a randomized trial. Cancer Chemother Pharmacol. 2003;52(1):13–8. Epub 2003/04/30. doi: 10.1007/s00280-003-0620-1. [DOI] [PubMed] [Google Scholar]
- 12.Taguchi T, Nazneen A, Abid MR, Razzaque MS. Cisplatin-associated nephrotoxicity and pathological events. Contrib Nephrol. 2005;148:107–21. Epub 2005/05/25. doi: 10.1159/000086055. [DOI] [PubMed] [Google Scholar]
- 13.Tothill P, Klys HS, Matheson LM, McKay K, Smyth JF. The long-term retention of platinum in human tissues following the administration of cisplatin or carboplatin for cancer chemotherapy. Eur J Cancer. 1992;28A(8–9):1358–61. Epub 1992/01/01. [DOI] [PubMed] [Google Scholar]
- 14.Poirier MC, Reed E, Litterst CL, Katz D, Gupta-Burt S. Persistence of platinum-ammine-DNA adducts in gonads and kidneys of rats and multiple tissues from cancer patients. Cancer Res. 1992;52(1):149–53. Epub 1992/01/01. [PubMed] [Google Scholar]
- 15.Hjelle LV, Gundersen PO, Oldenburg J, Brydoy M, Tandstad T, Wilsgaard T, Fossa SD, Bremnes RM, Haugnes HS. Long-term platinum retention after platinum-based chemotherapy in testicular cancer survivors: a 20-year follow-up study. Anticancer Res. 2015;35(3):1619–25. Epub 2015/03/10. [PubMed] [Google Scholar]
- 16.Gelevert T, Messerschmidt J, Meinardi MT, Alt F, Gietema JA, Franke JP, Sleijfer DT, Uges DR. Adsorptive voltametry to determine platinum levels in plasma from testicular cancer patients treated with cisplatin. Ther Drug Monit. 2001;23(2):169–73. Epub 2001/04/11. [DOI] [PubMed] [Google Scholar]
- 17.Hjelle LV, Bremnes RM, Gundersen POM, Sprauten M, Brydoy M, Tandstad T, Wilsgaard T, Fossa SD, Oldenburg J, Haugnes HS. Associations between long-term serum platinum and neurotoxicity and ototoxicity, endocrine gonadal function, and cardiovascular disease in testicular cancer survivors. Urol Oncol. 2016;34(11):487 e13–e20. Epub 2016/08/16. doi: 10.1016/j.urolonc.2016.06.012. [DOI] [PubMed] [Google Scholar]
- 18.Gietema JA, Meinardi MT, Messerschmidt J, Gelevert T, Alt F, Uges DR, Sleijfer DT. Circulating plasma platinum more than 10 years after cisplatin treatment for testicular cancer. Lancet. 2000;355(9209):1075–6. Epub 2000/04/01. [DOI] [PubMed] [Google Scholar]
- 19.Brouwers EE, Huitema AD, Beijnen JH, Schellens JH. Long-term platinum retention after treatment with cisplatin and oxaliplatin. BMC Clin Pharmacol. 2008;8:7 Epub 2008/09/18. doi: 10.1186/1472-6904-8-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sprauten M, Darrah TH, Peterson DR, Campbell ME, Hannigan RE, Cvancarova M, Beard C, Haugnes HS, Fossa SD, Oldenburg J, Travis LB. Impact of long-term serum platinum concentrations on neuro- and ototoxicity in Cisplatin-treated survivors of testicular cancer. J Clin Oncol. 2012;30(3):300–7. Epub 2011/12/21. doi: 10.1200/JCO.2011.37.4025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Boer H, Proost JH, Nuver J, Bunskoek S, Gietema JQ, Geubels BM, Altena R, Zwart N, Oosting SF, Vonk JM, Lefrandt JD, Uges DR, Meijer C, de Vries EG, Gietema JA. Long-term exposure to circulating platinum is associated with late effects of treatment in testicular cancer survivors. Ann Oncol. 2015;26(11):2305–10. Epub 2015/09/09. doi: 10.1093/annonc/mdv369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wheeler HE, Gamazon ER, Frisina RD, Perez-Cervantes C, El Charif O, Mapes B, Fossa SD, Feldman DR, Hamilton RJ, Vaughn DJ, Beard CJ, Fung C, Kollmannsberger C, Kim J, Mushiroda T, Kubo M, Ardeshir-Rouhani-Fard S, Einhorn LH, Cox NJ, Dolan ME, Travis LB. Variants in WFS1 and Other Mendelian Deafness Genes Are Associated with Cisplatin-Associated Ototoxicity. Clin Cancer Res. 2017;23(13):3325–33. Epub 2017/01/01. doi: 10.1158/1078-0432.CCR-16-2809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kerns SL, Fung C, Monahan PO, Ardeshir-Rouhani-Fard S, Abu Zaid MI, Williams AM, Stump TE, Sesso HD, Feldman DR, Hamilton RJ, Vaughn DJ, Beard C, Huddart RA, Kim J, Kollmannsberger C, Sahasrabudhe DM, Cook R, Fossa SD, Einhorn LH, Travis LB, Platinum Study G. Cumulative Burden of Morbidity Among Testicular Cancer Survivors After Standard Cisplatin-Based Chemotherapy: A Multi-Institutional Study. J Clin Oncol. 2018;36(15):1505–12. Epub 2018/04/05. doi: 10.1200/JCO.2017.77.0735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Long GL WJ. Limit of detection: A closer look at the IUPAC definition. Anal Chem. 1983;55:712A–724A. [Google Scholar]
- 25.Strathmann FG, Travis LB, Ardeshirrouhanifard S, Fossa SD, Moody S, Clarke D, Law CL. Residual Platinum Concentrations in Post-Cancer Chemotherapy and Healthy Control Populations Using an Automated, 96-Well Plate Method and Inductively Coupled Plasma Mass Spectrometry2016;1(2):143–51. doi: 10.1373/jalm.2016.020081 %J The Journal of Applied Laboratory Medicine: An AACC Publication. [DOI] [PubMed] [Google Scholar]
- 26.Geenen MM, Cardous-Ubbink MC, Kremer LC, van den Bos C, van der Pal HJ, Heinen RC, Jaspers MW, Koning CC, Oldenburger F, Langeveld NE, Hart AA, Bakker PJ, Caron HN, van Leeuwen FE. Medical assessment of adverse health outcomes in long-term survivors of childhood cancer. JAMA. 2007;297(24):2705–15. Epub 2007/06/28. doi: 10.1001/jama.297.24.2705. [DOI] [PubMed] [Google Scholar]
- 27.Oeffinger KC, Mertens AC, Sklar CA, Kawashima T, Hudson MM, Meadows AT, Friedman DL, Marina N, Hobbie W, Kadan-Lottick NS, Schwartz CL, Leisenring W, Robison LL, Childhood Cancer Survivor S. Chronic health conditions in adult survivors of childhood cancer. N Engl J Med. 2006;355(15):1572–82. Epub 2006/10/13. doi: 10.1056/NEJMsa060185. [DOI] [PubMed] [Google Scholar]
- 28.Oldenburg J, Fossa SD, Dahl AA. Scale for chemotherapy-induced long-term neurotoxicity (SCIN): psychometrics, validation, and findings in a large sample of testicular cancer survivors. Qual Life Res. 2006;15(5):791–800. Epub 2006/05/25. doi: 10.1007/s11136-005-5370-6. [DOI] [PubMed] [Google Scholar]
- 29.Engdahl B, Tambs K, Borchgrevink HM, Hoffman HJ. Screened and unscreened hearing threshold levels for the adult population: results from the Nord-Trondelag Hearing Loss Study. Int J Audiol. 2005;44(4):213–30. Epub 2005/07/14. [DOI] [PubMed] [Google Scholar]
- 30.Zaid MA, Gathirua-Mwangi WG, Fung C, Monahan PO, El-Charif O, Williams AM, Feldman DR, Hamilton RJ, Vaughn DJ, Beard CJ, Cook R, Althouse SK, Ardeshir-Rouhani-Fard S, Dinh PC, Sesso HD, Einhorn LH, Fossa SD, Travis LB, Platinum Study G. Clinical and Genetic Risk Factors for Adverse Metabolic Outcomes in North American Testicular Cancer Survivors. J Natl Compr Canc Netw. 2018;16(3):257–65. Epub 2018/03/11. doi: 10.6004/jnccn.2017.7046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Das S, Forer L, Schonherr S, Sidore C, Locke AE, Kwong A, Vrieze SI, Chew EY, Levy S, McGue M, Schlessinger D, Stambolian D, Loh PR, Iacono WG, Swaroop A, Scott LJ, Cucca F, Kronenberg F, Boehnke M, Abecasis GR, Fuchsberger C. Next-generation genotype imputation service and methods. Nat Genet. 2016;48(10):1284–7. Epub 2016/08/30. doi: 10.1038/ng.3656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Loh PR, Danecek P, Palamara PF, Fuchsberger C, Y AR, H KF, Schoenherr S, Forer L, McCarthy S, Abecasis GR, Durbin R, A LP. Reference-based phasing using the Haplotype Reference Consortium panel. Nat Genet. 2016;48(11):1443–8. Epub 2016/10/28. doi: 10.1038/ng.3679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.McCarthy S, Das S, Kretzschmar W, Delaneau O, Wood AR, Teumer A, Kang HM, Fuchsberger C, Danecek P, Sharp K, Luo Y, Sidore C, Kwong A, Timpson N, Koskinen S, Vrieze S, Scott LJ, Zhang H, Mahajan A, Veldink J, Peters U, Pato C, van Duijn CM, Gillies CE, Gandin I, Mezzavilla M, Gilly A, Cocca M, Traglia M, Angius A, Barrett JC, Boomsma D, Branham K, Breen G, Brummett CM, Busonero F, Campbell H, Chan A, Chen S, Chew E, Collins FS, Corbin LJ, Smith GD, Dedoussis G, Dorr M, Farmaki AE, Ferrucci L, Forer L, Fraser RM, Gabriel S, Levy S, Groop L, Harrison T, Hattersley A, Holmen OL, Hveem K, Kretzler M, Lee JC, McGue M, Meitinger T, Melzer D, Min JL, Mohlke KL, Vincent JB, Nauck M, Nickerson D, Palotie A, Pato M, Pirastu N, McInnis M, Richards JB, Sala C, Salomaa V, Schlessinger D, Schoenherr S, Slagboom PE, Small K, Spector T, Stambolian D, Tuke M, Tuomilehto J, Van den Berg LH, Van Rheenen W, Volker U, Wijmenga C, Toniolo D, Zeggini E, Gasparini P, Sampson MG, Wilson JF, Frayling T, de Bakker PI, Swertz MA, McCarroll S, Kooperberg C, Dekker A, Altshuler D, Willer C, Iacono W, Ripatti S, Soranzo N, Walter K, Swaroop A, Cucca F, Anderson CA, Myers RM, Boehnke M, McCarthy MI, Durbin R, Haplotype Reference C. A reference panel of 64,976 haplotypes for genotype imputation. Nat Genet. 2016;48(10):1279–83. Epub 2016/08/23. doi: 10.1038/ng.3643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yang J, Lee SH, Goddard ME, Visscher PM. GCTA: a tool for genome-wide complex trait analysis. Am J Hum Genet. 2011;88(1):76–82. Epub 2010/12/21. doi: 10.1016/j.ajhg.2010.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Miller RP, Tadagavadi RK, Ramesh G, Reeves WB. Mechanisms of Cisplatin nephrotoxicity. Toxins (Basel). 2010;2(11):2490–518. Epub 2011/11/10. doi: 10.3390/toxins2112490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Breglio AM, Rusheen AE, Shide ED, Fernandez KA, Spielbauer KK, McLachlin KM, Hall MD, Amable L, Cunningham LL. Cisplatin is retained in the cochlea indefinitely following chemotherapy. Nat Commun. 2017;8(1):1654 Epub 2017/1½3. doi: 10.1038/s41467-017-01837-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Henry JA, Roberts LE, Caspary DM, Theodoroff SM, Salvi RJ. Underlying mechanisms of tinnitus: review and clinical implications. J Am Acad Audiol. 2014;25(1):5–22; quiz 126. Epub 2014/03/14. doi: 10.3766/jaaa.25.1.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Paken J, Govender CD, Pillay M, Sewram V. Cisplatin-Associated Ototoxicity: A Review for the Health Professional. J Toxicol. 2016;2016:1809394 Epub 2017/0½5. doi: 10.1155/2016/1809394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.LeBlanc TW, Abernethy AP. Patient-reported outcomes in cancer care - hearing the patient voice at greater volume. Nat Rev Clin Oncol. 2017;14(12):763–72. Epub 2017/10/05. doi: 10.1038/nrclinonc.2017.153. [DOI] [PubMed] [Google Scholar]
- 40.Hershman DL, Weimer LH, Wang A, Kranwinkel G, Brafman L, Fuentes D, Awad D, Crew KD. Association between patient reported outcomes and quantitative sensory tests for measuring long-term neurotoxicity in breast cancer survivors treated with adjuvant paclitaxel chemotherapy. Breast Cancer Res Treat. 2011;125(3):767–74. Epub 2010/12/04. doi: 10.1007/s10549-010-1278-0. [DOI] [PubMed] [Google Scholar]
- 41.Wolf SL, Barton DL, Qin R, Wos EJ, Sloan JA, Liu H, Aaronson NK, Satele DV, Mattar BI, Green NB, Loprinzi CL. The relationship between numbness, tingling, and shooting/burning pain in patients with chemotherapy-induced peripheral neuropathy (CIPN) as measured by the EORTC QLQ-CIPN20 instrument, N06CA. Support Care Cancer. 2012;20(3):625–32. Epub 2011/04/12. doi: 10.1007/s00520-011-1141-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Dorsey SG, Kleckner IR, Barton D, Mustian K, O’Mara A, St Germain D, Cavaletti G, Danhauer SC, Hershman D, Hohmann AG, Hoke A, Hopkins JO, Kelly KP, Loprinzi CL, McLeod HL, Mohile S, Paice J, Rowland JH, Salvemini D, Segal RA, Lavoie Smith E, McCaskill Stevens W, Janelsins MC. NCI Clinical Trials Planning Meeting for prevention and treatment of chemotherapy-induced peripheral neuropathy. J Natl Cancer Inst. 2019. Epub 2019/02/05. doi: 10.1093/jnci/djz011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sprauten M, Brydoy M, Haugnes HS, Cvancarova M, Bjoro T, Bjerner J, Fossa SD, Oldenburg J. Longitudinal serum testosterone, luteinizing hormone, and follicle-stimulating hormone levels in a population-based sample of long-term testicular cancer survivors. J Clin Oncol. 2014;32(6):571–8. Epub 2014/01/15. doi: 10.1200/JCO.2013.51.2715. [DOI] [PubMed] [Google Scholar]
- 44.Recuenco MC, Ohmori T, Tanigawa S, Taguchi A, Fujimura S, Conti MA, Wei Q, Kiyonari H, Abe T, Adelstein RS, Nishinakamura R. Nonmuscle Myosin II Regulates the Morphogenesis of Metanephric Mesenchyme-Derived Immature Nephrons. J Am Soc Nephrol. 2015;26(5):1081–91. Epub 2014/08/30. doi: 10.1681/ASN.2014030281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Haque F, Kaku Y, Fujimura S, Ohmori T, Adelstein RS, Nishinakamura R. Non-muscle myosin II deletion in the developing kidney causes ureter-bladder misconnection and apical extrusion of the nephric duct lineage epithelia. Dev Biol. 2017;427(1):121–30. Epub 2017/05/10. doi: 10.1016/j.ydbio.2017.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Otterpohl KL, Hart RG, Evans C, Surendran K, Chandrasekar I. Nonmuscle myosin 2 proteins encoded by Myh9, Myh10, and Myh14 are uniquely distributed in the tubular segments of murine kidney. Physiol Rep. 2017;5(23). Epub 2017/12/07. doi: 10.14814/phy2.13513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Choi BO, Kang SH, Hyun YS, Kanwal S, Park SW, Koo H, Kim SB, Choi YC, Yoo JH, Kim JW, Park KD, Choi KG, Kim SJ, Zuchner S, Chung KW. A complex phenotype of peripheral neuropathy, myopathy, hoarseness, and hearing loss is linked to an autosomal dominant mutation in MYH14. Hum Mutat. 2011;32(6):669–77. Epub 2011/04/12. doi: 10.1002/humu.21488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pace A, Giannarelli D, Galie E, Savarese A, Carpano S, Della Giulia M, Pozzi A, Silvani A, Gaviani P, Scaioli V, Jandolo B, Bove L, Cognetti F. Vitamin E neuroprotection for cisplatin neuropathy: a randomized, placebo-controlled trial. Neurology. 2010;74(9):762–6. Epub 2010/03/03. doi: 10.1212/WNL.0b013e3181d5279e. [DOI] [PubMed] [Google Scholar]
- 49.Brock PR, Maibach R, Childs M, Rajput K, Roebuck D, Sullivan MJ, Laithier V, Ronghe M, Dall’Igna P, Hiyama E, Brichard B, Skeen J, Mateos ME, Capra M, Rangaswami AA, Ansari M, Rechnitzer C, Veal GJ, Covezzoli A, Brugieres L, Perilongo G, Czauderna P, Morland B, Neuwelt EA. Sodium Thiosulfate for Protection from Cisplatin-Induced Hearing Loss. N Engl J Med. 2018;378(25):2376–85. Epub 2018/06/21. doi: 10.1056/NEJMoa1801109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sprowl JA, van Doorn L, Hu S, van Gerven L, de Bruijn P, Li L, Gibson AA, Mathijssen RH, Sparreboom A. Conjunctive therapy of cisplatin with the OCT2 inhibitor cimetidine: influence on antitumor efficacy and systemic clearance. Clin Pharmacol Ther. 2013;94(5):585–92. Epub 2013/07/19. doi: 10.1038/clpt.2013.145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ciarimboli G, Deuster D, Knief A, Sperling M, Holtkamp M, Edemir B, Pavenstadt H, Lanvers-Kaminsky C, am Zehnhoff-Dinnesen A, Schinkel AH, Koepsell H, Jurgens H, Schlatter E. Organic cation transporter 2 mediates cisplatin-induced oto- and nephrotoxicity and is a target for protective interventions. Am J Pathol. 2010;176(3):1169–80. Epub 2010/01/30. doi: 10.2353/ajpath.2010.090610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sprowl JA, Lancaster CS, Pabla N, Hermann E, Kosloske AM, Gibson AA, Li L, Zeeh D, Schlatter E, Janke LJ, Ciarimboli G, Sparreboom A. Cisplatin-induced renal injury is independently mediated by OCT2 and p53. Clin Cancer Res. 2014;20(15):4026–35. Epub 2014/06/12. doi: 10.1158/1078-0432.CCR-14-0319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Freyer DR, Chen L, Krailo MD, Knight K, Villaluna D, Bliss B, Pollock BH, Ramdas J, Lange B, Van Hoff D, VanSoelen ML, Wiernikowski J, Neuwelt EA, Sung L. Effects of sodium thiosulfate versus observation on development of cisplatin-induced hearing loss in children with cancer (ACCL0431): a multicentre, randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2017;18(1):63–74. Epub 2016/12/05. doi: 10.1016/S1470-2045(16)30625-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Mantel N Chi-square tests with one degree of freedom: extensions of the Mantel–Haenszel procedure. Journal of the American Statistical Association. 1963;58: 690–700. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


