Abstract
Rationale:
Benign fibrous histiocytoma with pleural involvement in spinal region is a highly unusual disease with no standard curative managements yet. The objective of this study is to report an extremely rare case of a giant benign fibrous histiocytoma with pleural involvement in spinal region successfully operated by posterior spinal surgery. The management of these unique cases has yet to be well-documented.
Patient concerns:
A 23-year-old man presented with a 2-month history of continuous and progressive back pain. A giant, expanding lesion of the T7 vertebral and paraspinal region with pleural involvement was identified.
Diagnoses:
Computed tomography scan and magnetic resonance imaging of spine showed expanding lesion of the T7 vertebral and paraspinal region involving the right thoracic cavity, which presented as a solid tumor. Postoperative pathology confirmed the diagnosis of thoracic benign fibrous histiocytoma.
Interventions:
The patient underwent thoracic spinal canal decompression, complete tumor resection, pleural defect repair, and T4 to T10 internal fixation procedure via a posterior approach.
Outcomes:
The patient's symptom improved significantly after the surgery, and the postoperative period was uneventful at the 2-year follow-up visit. There were no complications associated with the operation during the follow-up period.
Lessons:
In summary, the tumor's clinical features, imaging results, and pathological characteristics are unique. Combined efforts of specialists from orthopedics, thoracic surgery, neurosurgery, pathology, and medical oncology led to the successful diagnosis and management of this patient. Giant benign fibrous histiocytoma with pleural involvement in spinal region, although rare, should be part of the differential diagnosis when the patient presents with back pain and radiculopathy. We recommend the posterior or 1-stage anterior–posterior combined approach for complete resection of the giant thoracic benign fibrous histiocytoma when the tumor has caused severe symptoms or neurological deficits.
Keywords: benign fibrous histiocytoma, diagnosis, pleural involvement, spinal tumor resection, surgical treatment, thoracic spine
1. Introduction
Benign fibrous histiocytoma (BFH) is an extremely rare primary bone tumor that arises from primitive mesenchymal cells, being capable of multi-directional differentiation, accounting for less than 1% of all benign bone tumors.[1–3] BFH usually manifests as soft tissue mass with progressive enlargement, which rarely arises in or from spinal region.[1] It is less aggressive with lower rates of local recurrence and metastasis, compared with malignant forms of fibrous histiocytomas.[1–4] The vast majority of BFH cases occur in the long bones such as the femur, tibia, and humerus.[1–3] BFH with spinal and pleural involvement is exceedingly rare, and the thoracic spine is a very uncommon site of localization. Spinal BFH usually produces nonspecific pain or discomfort as a primary presenting symptom, similar to other spinal tumors, and may cause neurologic symptoms secondary to nerve roots and spinal cord compression.[1–3] Herein, we describe a rare case of BFH in a 23-year-old man referred for surgical treatment of a large mass in spinal region, manifesting rapidly progressive back pain. We performed a surgical exploration, total resection of T7 vertebral and paravertebral tumor, spinal canal decompression, and T4 to T10 internal fixation procedure. In the short term, the patient's conditions improved significantly postoperatively. After reviewing pertinent literature, we discussed common perioperative considerations in patients with a giant BFH of the thoracic spine and management considerations for these patients. Our focus is to emphasize the importance of considering BFH as a diagnosis and guiding to the optimum surgical treatment.
2. Case report
In June of 2017, a 23-year-old man with progressive back pain was presented to our hospital. In his history of present illness, the patient stated he has been experiencing a paroxysmal and severe back pain for approximately 2 months. The pain in his back could reach 7 points using visual analog scale (VAS) and could not be alleviated with rest and hot compresses. Initially, the patient attributed the pain to his overwork and thus did not seek medical attention. The patient denied experiencing any other constitutional symptoms. Upon further questioning, he denied history of any injury or underlying diseases. No pertinent family history was identified, including, hypertension, cancer, and congenital birth difficulties.
On physical examination, the patient showed pressure pain and percussion pain in his thoracic region, normal sensation to pin-prick and fine-touch of bilateral lower extremities and exhibited a 5/5 strength in bilateral lower extremities. Deep tendon reflexes revealed normal for both knee jerk and Achilles tendon reflexes bilaterally. Ataxia was absent. Cranial nerves, mini-mental, and the rest of the neurological exam showed no abnormalities. Routine laboratory tests were ordered, including electrolytes, liver and kidney function tests, tumor markers, and complete blood count. The results of the laboratory studies were almost within normal range. Preoperative assessments included electrocardiogram, echocardiogram, and chest X-ray. Plain radiographs of the spine showed an evident soft tissue mass sized 7.3 × 5.2 cm in thoracic spinal region penetrating into the right thoracic cavity and compressing lower lobe of the right lung (Fig. 1A and B). Chest enhanced computed tomography was performed to assess pleural involvement of the giant mass and revealed the density of soft tissue measuring 7.5 × 7.0 × 5.5 cm, obvious bony destruction in the left pedicle of T7, and pleural involvement caused by the thoracic mass. Spinal magnetic resonance imaging (MRI) was ordered to visualize the thoracic lesions, to assess the stability of the vertebral column and pleural involvement, and to aid in the formulation of the surgical approach. The internal multiple calcifications were observed on computed tomography (CT) scan, and the enhanced CT scan revealed that the solid part of the tumor was slightly enhanced (Fig. 2A–H). MRI of the thoracolumbar spine revealed widespread abnormal and irregular signals at T7 caused by the component of the mass, with increased marrow infiltration of the lateral vertebral body and the 7th rear rib, as well as mild bony destruction secondary to the epidural component of the large mass in the left T7 vertebral and paravertebral regions, which presented as a solid tumor (Fig. 3A–H). Tumor infiltrated through the T7 vertebral bodies into the right pedicles and posterior elements (Fig. 4A–H). Extraosseous spread into the right aspect of the epidural space extending posteriorly, resulting nerve root compression (Fig. 4A–H). Bone scintigraphy indicated the skeletal abnormality of right side of the T7 vertebrae and the right 7th posterior rib, with high suspicion of benign tumor (Fig. 5).
Figure 1.
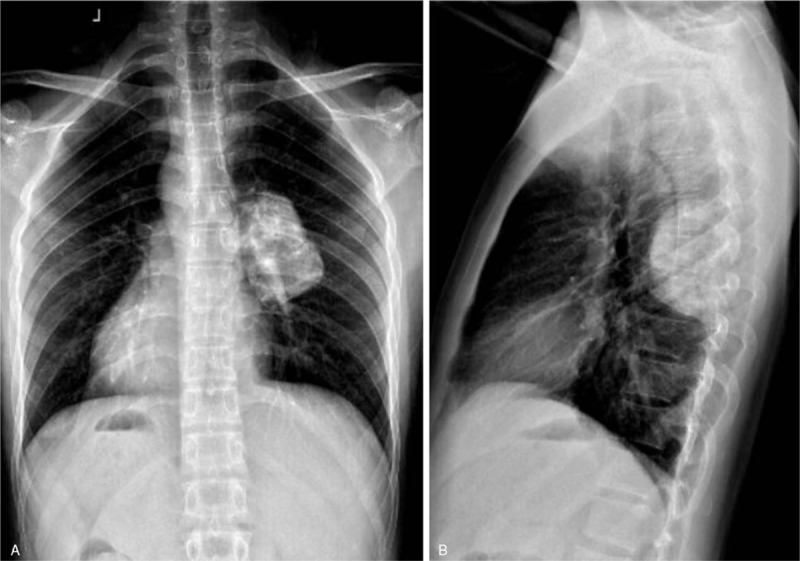
(A, B) Preoperative X-rays revealing thoracic lesions with high suspicion of spinal soft tissue tumors.
Figure 2.
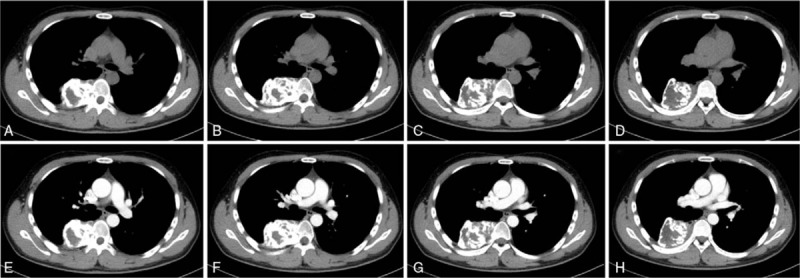
(A–H) Preoperative transverse CT scan revealing the density of soft tissue measuring 7.5 × 7.0 × 5.5 cm, obvious bony destruction in the T7, and pleural involvement caused by thoracic mass. CT = computed tomography.
Figure 3.
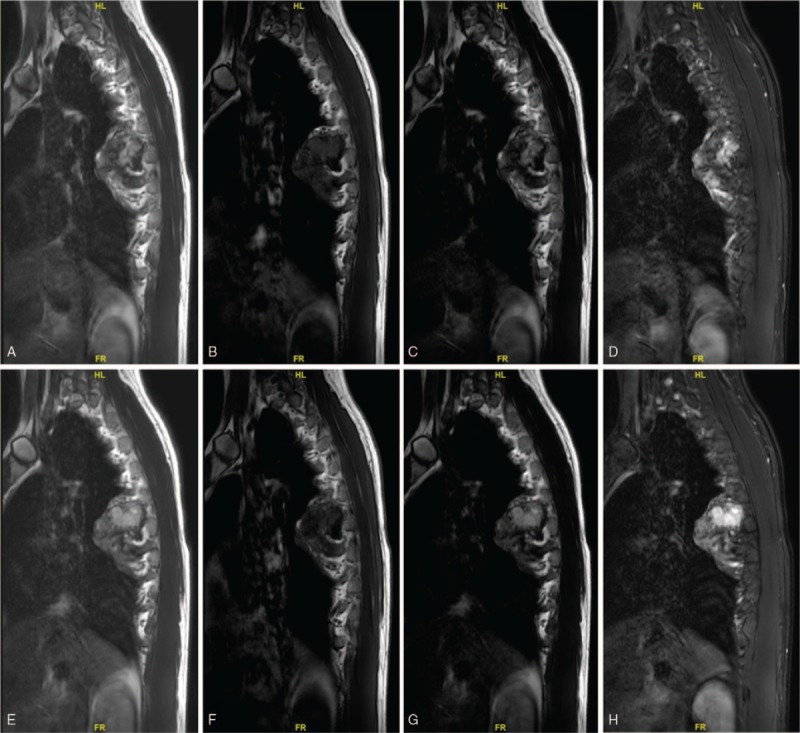
(A–H) Preoperative sagittal MRI showing the thoracic BFH. BFH = benign fibrous histiocytoma, MRI = magnetic resonance imaging.
Figure 4.
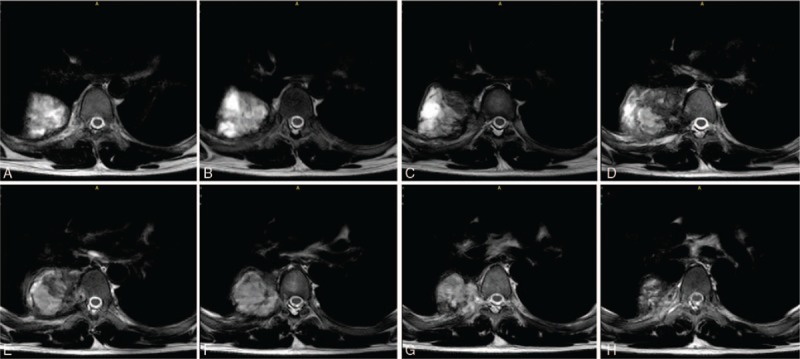
(A–H) Preoperative transverse MRI showing the thoracic BFH. BFH = benign fibrous histiocytoma, MRI = magnetic resonance imaging.
Figure 5.
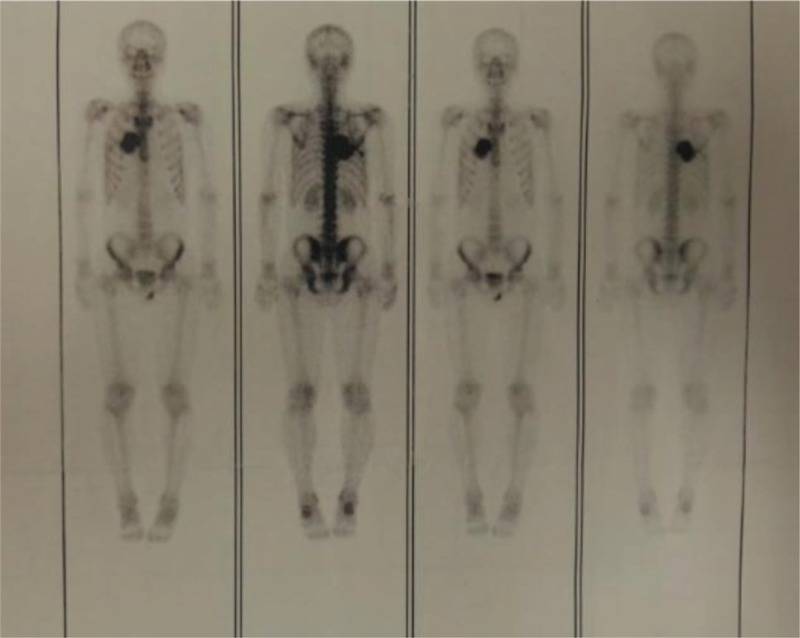
Bone scan revealed high intake in the thoracic region, with high suspicion of a solid tumor.
After a detailed assessment, posterior thoracic spinal canal decompression, complete tumor resection, pleural defect repair, and T4 to T10 internal fixation were performed to alleviate the symptoms caused by the giant tumor and subsequently stabilize the vertebral spine. Because of the size and location of the giant lesion and the extent of the involvement, the risk of surgical intervention is exceedingly high.
After successful anesthesia, the patient was placed in a prone position for dorsal access to the thoracic spine. In brief, posterior circumferential decompression, tumor resection, and T4 to T10 internal fixation were performed. For the posterior approach, the paraspinal muscles were detached gently on each side after a midline longitudinal incision was made over the spinous processes from T4 to T10. The pedicle entry points were exposed by step-by-step bilateral dissection. At first, the pedicles of T4, T5, T9, T10, and the right pedicles of T7 were fixed by routine screw preparation and placement needles. C-arm fluoroscopy showed that the location of pedicles was accurate and the position was satisfactory. Then, the pedicle screws were placed bilaterally, followed by pedicle screw insertion. The heads and proximal region of the right 7th rib were exposed by dissection and resected. The tumor was located at the right paravertebral region of T7, involving the vertebral body and the right pedicle of T7. During the operation, the root of right 7th rib, the right transverse costal process joint, and the right paravertebral region were involved by the tumor. The tumor was oval in shape and hard in texture, sizing 7.5 × 7.0 × 5.5 cm (Fig. 6A and B). Moreover, the tumor invaded the parietal and visceral pleura and adhered to the adjacent pulmonary lobe. T6 to T8 spinous processes and interspinous ligaments were removed, then the right laminae of T6, T7, and T8 vertebrae were resected. Subsequently, the dural sac was exposed and protected. The proximal segments of the right 6, 7, 8, and 9 ribs were resected and the intercostal vessels and nerves were ligated. The transverse costal process joints and the right vertebral body of T7 were all resected. The tumor was then turned up and the adhesion of the tumor, the pleura, and the right lung was carefully separated. Finally, the giant tumor was completely removed. There was a partial pleural defect and a local air leak in the right lung, thus lung repair and visceral pleura repair were performed by a thoracic surgeon. There was no air leakage in the expansion process of the lung, and the repair was satisfactory. Because the patient did not exhibit hemodynamic instability to the placement of the pedicle screws, fixation using a screw-rod system was employed. Visual inspection using the intraoperative fluoroscopy showed optimal position of all pedicle screws. After spinal operation, 1 thoracic tube was placed between the 5th and 6th ribs of the right axillary midline, and closed drainage bottle was connected to the right thoracic cavity. The incision was closed. Intraoperative blood loss was approximately 2940 mL, thus we used erythrocyte 6 U and plasma 800 mL. Postoperatively, the patient was referred to the intensive care unit and transferred to general ward the next day. An x-ray after the surgery confirmed the correct positioning of the implants and no signs of displacement of the screws and rods (Fig. 7A and B). The postoperative pathology report confirmed a BFH with negative margin. Pathological result was positive for CD68 indicating epithelial origin. Biopsy samples were positive for S-100, smooth muscle actin (SMA), with 1% Ki-67 positive nuclei (Fig. 8A–I).
Figure 6.
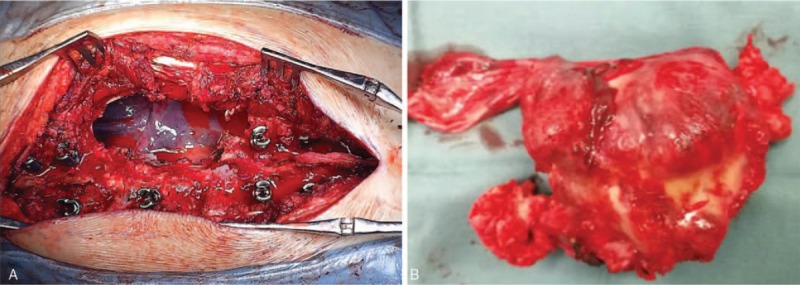
(A) Intraoperative photography depicting the exposed spinal cord. (B) Intraoperative photography depicting entirely resected tumor.
Figure 7.
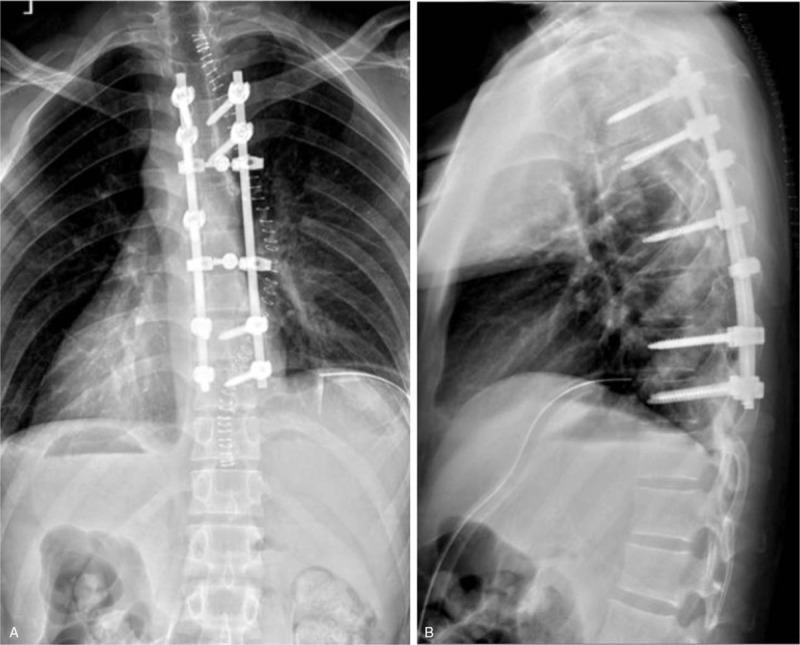
(A, B) Posteroanterior and lateral x-ray images of the thoracic spine obtained postoperatively.
Figure 8.
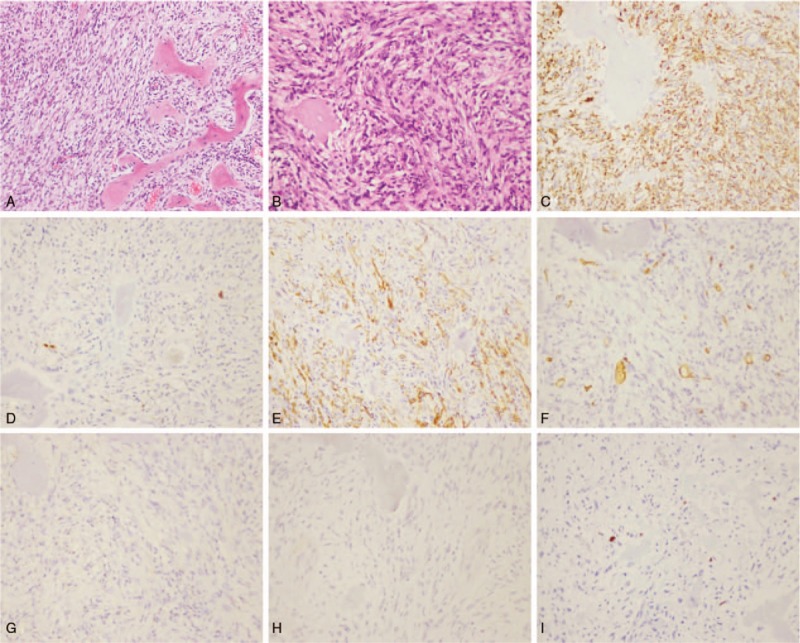
Pathologic histology of spinal tumors. (A, B) Microphotography showing characteristic nests of tumor cells separated by vascular septa (Zellballen) with cells showing a stroma of spindle-shaped fibrohistiocytic cells (H&E, original magnification 100× and 200×). (C) CD68 immunostaining is strongly positive in the tumor cells. (D) S-100 immunostaining shows positive staining in the tumor cells. (E) SMA immunostaining shows positive staining in the tumor cells. (F–H) CD34, CD99, Desmin immunostaining shows positive staining in the tumor cells. (I) Ki-67 immunostaining shows 1% Ki-67 positive cells. Ki-67 staining is localized in the tumor nuclei. SMA = smooth muscle actin.
One week after the operation, the VAS score of his back pain improved to 0-1 points compared to the preoperative status, 7 points. Following wound healing and removal of thoracic drainage tube, the patient underwent rehabilitation therapy and was monitored as an outpatient. The postoperative 2-year follow-up visit showed no tumor progression and no new symptoms.
3. Discussion
BFH is an extraordinarily rare bone tumor which may exhibit benign differentiation, accounting for less than 1% of all benign bone tumors.[1,2] The most prevalent sites of BFHs are the skin and eyeballs, followed by the pharynx, larynx, trachea, and lungs.[5,6] The incidence of BFH in males is approximately 2 times of that in females.[1–3,7] BFH grows slowly, recurs locally, and may metastasize later, thus it can be difficult to diagnose and may result in devastating consequences if mismanaged.[8–10] It is a relatively rare tumor, and BFH with involvement of the thoracic spine is exceedingly rare. Clinically the tumor may be asymptomatic but usually presents with symptoms related to compression or involvement of adjacent structures and organs, including the trachea, esophagus, pleura cavity, and spinal cord.[1,2,9–12] The characteristics of thoracic BFHs have not yet been fully clarified. Moreover, there is still no defined treatment protocol for thoracic BFHs due to the paucity of these patients.
In literature, it is a rarity of BFH in the thoracic region presenting with pleural involvement.[1,2,13–15] Clinical studies looking at BFHs to the mobile spine is lacking due to the extremely low incidence rate. Based on our review of the case reports on PubMed, the location of the spinal lesion determines the neurological deficits, and there is a great deal of variability.[1–22] Compression of the cervical vertebra often exhibit symptoms of paresthesia and weakness in the upper extremities, while those located in the thoracic or lumbar regions usually show symptoms of back pain, lower extremity paresthesia, and weakness.[21,23] In our case, the patient sought medical attention after experiencing serious back pain, which is consistent with previous case reports. In previous case reports, MRI images of thoracic BFH appear as inhomogenous lesion, iso-hyperintense on T1-weighted image and iso-hyperintense on T2-weighted image, and indistinguishable from other primary or metastatic spinal lesions.[1–22] Imaging studies including CT, MRI, bone scan, and positron emission tomography/CT are nonspecific, making it difficult to differentiate thoracic BFHs from other common spinal lesions.[1–3] However, imaging studies play a crucial role in the surgical intervention decision making. Imaging studies can demonstrate consecutive spinal stenosis, spinal cord, or nerve root compression, compression or involvement of adjacent structures, and pathological vertebrae fractures.[1,23,24] Radiographically, vertebral destruction is associated with the mediastinal mass because of the bony origin of the tumor. Lesions are usually lytic with calcifications within the tumor mass.[1–3] CT scan of the chest and MRI of the spine are mandatory to evaluate the extent of both the tumor and involvement of adjacent organs. To date, the “gold-standard” diagnosis of BFHs relies on pathological findings. The differential diagnosis between benign and malignant fibrous histiocytomas depends entirely on the presence of the cytological atypia, high mitotic rate, and necrosis. Histopathologically, BFHs are characterized by an architecture of a stroma of spindle-shaped fibrohistiocytic cells arranged in a whorled stroriform pattern, among which a variable number of small, multinucleated osteoclast-type giant cells are scattered.[25,26] Foam cells are frequently found among the stromal cells.[25,26] Generally, BFHs are commonly immunoreactive for CD68, S-100, SMA.[25,26] A histological examination of our case was positive for CD68, S-100, SMA, which confirmed the diagnosis of BFH. Spinal BFHs should be distinguished from other tumors, such as eosinophilic granuloma, nonossifying fibroma, malignant fibrous histiocytoma, giant cell tumor, and other primary or metastatic spinal tumors via evaluating the clinical symptoms, age of onset, prevalent location, and radiological and histological features.[24–26] For our reported case, we could not exactly rule out malignancy before surgery since histological examination is mandatory and the gold standard for a correct diagnosis and plan of treatment for spinal BFHs. Histologically, BFH consists of a stroma of spindle-shaped fibrohistiocytic cells, among which a variable number of small, multinucleated osteoclast-type giant cells are scattered. Foam cells can be frequently seen among the stromal cells.[24–26]
Currently, no treatment guidelines exist because of the variability in treatment modalities and reported outcomes.[1–11] Surgical resection is the mainstay of treatment for BFH. We recommend surgical management of the spinal BFHs when the tumor has caused neurological deficits, spinal cord compression, destruction of spinal stability, or severe compression or involvement of adjacent structures. The best treatment for metastatic spinal BFHs is posterior decompression, total tumor resection, and internal fixation.[1–22] This protocol accomplishes 2 objectives: it alleviates the neurological deficits by decompressing the stenosis or relieve the oppression; and at the same time it provides histopathological specimens for diagnosis, which is valuable in cases where the patient presents with atypical clinical and radiological findings.[1–22] If possible, complete or grossly complete surgical resection of the primary tumor remains the treatment of the first choice. To date, surgical management of thoracic BFH still remains under evaluation with no standard criteria. The more accepted approach is aggressive surgery to resect the tumor as far as possible and prevent dissemination into the surrounding tissues.[1–4] Thoracoscopic surgery combined with posterior spinal surgery might be a useful strategy to achieve rapid and sustained neurological improvements for patients with thoracic BFHs involving the pleural cavity. Completion of surgical removal of the mediastinal component is directly related to its location and expansion. Recurrence after surgical resection is a common feature and is considered to be related to the residual tumor margins at surgical time.[1–22] Due to the rarity of spinal BFHs, the recurrence rate of BFH is reported to be low but needs to be further clarified. This procedure does have the benefit aiming at controlling residual tumor and is recommended for most patients with suspicion of BFH.[1–11,27]
This patient has the following unique clinical features: First, from chest CT and spinal MRI examination, we can see that the lesion involved the thoracic spine, and protruded into the right pleural cavity. The margin of the tumor was relatively clear and the calcification was significant within the tumor. Second, we need to be fully aware of the transformation of lesions from BFH to malignant fibrous histiocytoma, which can directly affect the course of disease and prognosis of patients. It is reported that the malignant transformation of the BFHs may be closely related to the local progression, incomplete surgical resection, long course of disease, and the decline of the body's immunity. Finally, for BFH, radical resection is the best and preferred treatment option. Primary BFH in the thoracic region is rare, but its prognosis is reported to be relatively good.
In conclusion, we present an extremely unusual occurrence of a giant thoracic BFH that was successfully managed by surgical treatment, which has not been previously well reported in literature. Our focus is to emphasize the importance of considering thoracic BFH as a diagnosis and guiding the proper management strategy upon surgical treatment. The giant thoracic BFHs are truly rare neoplasms, particularly compared to other primary or metastatic lesions of the spine. Although uncommon, it should be part of the differential diagnosis when the patient presents with back pain or neurological deficits. To date, surgery is the mainstay of treatment in patients with good performance status before symptom onset and should be strongly considered in patients with the suspicion of BFH. With a multidisciplinary team approach, proper planning, and adequate perioperative medical management, thoracic BFHs can be managed effectively.
Acknowledgments
We would like to thank our colleagues at the Department of Orthopaedic Surgery, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College.
Author contributions
Conceptualization: Shuzhong Liu, An Song, Yipeng Wang, Yong Liu.
Funding acquisition: Shuzhong Liu, Yipeng Wang, Yong Liu.
Investigation: Shuzhong Liu, Xi Zhou, An Song, Siyuan Yao, Yong Liu.
Methodology: Shuzhong Liu, Yong Liu.
Resources: Shuzhong Liu, Xi Zhou, Zhen Huo, Yong Liu.
Supervision: Yipeng Wang, Yong Liu.
Writing – original draft: Shuzhong Liu, Xi Zhou, An Song.
Writing – review and editing: Shuzhong Liu, Yipeng Wang, Yong Liu.
Footnotes
Abbreviations: CT = computed tomography, MRI = magnetic resonance imaging, SMA = smooth muscle actin, VAS = visual analog scale.
How to cite this article: Liu S, Zhou X, Song A, Huo Z, Yao S, Wang Y, Liu Y. Management of giant benign fibrous histiocytoma in the spinal region with pleural involvement. Medicine. 2019;98:39(e17144).
Written informed consent was obtained from the patient for publication of this article, a copy of which is available for review from the editors of Medicine. Because this article does not involve any human or animal trials, it did not require institutional ethical review and approval.
This study was supported by National Natural Science Foundation of China (Project No. 81871746; Grant recipient: YW), and Peking Union Medical College Graduate Student Innovation Fund (2018) (Project No. 2018-1002-02-08; Grant recipient: SL). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
The authors have no conflicts of interest to disclose.
References
- [1].Skunda R, Puckett T, Martin M, et al. 14-year-old boy with mild antecedent neck pain in setting of acute trauma: a rare case of benign fibrous histiocytoma of the spine. Am J Orthop (Belle Mead NJ) 2016;45:E148–52. [PubMed] [Google Scholar]
- [2].Donati F, Proietti L, Burrofato A, et al. Intraspinal extradural benign fibrous histiocytoma of the lumbar spine in a pediatric patient. Case report and literature review. Childs Nerv Syst 2016;32:1549–53. [DOI] [PubMed] [Google Scholar]
- [3].Khor YM, Yan X. Benign fibrous histiocytoma of the thoracic spine as the cause of pyrexia of unknown origin identified by positron emission tomography/computed tomography. Spine J 2015;15:1691–2. [DOI] [PubMed] [Google Scholar]
- [4].Yang M, Wang XB, Li J, et al. Surgical treatment of large abdominally involved primary dumbbell tumor in the lumbar region. J Spinal Disord Tech 2014;27:E268–75. [DOI] [PubMed] [Google Scholar]
- [5].Kim SB, Jang JS, Lee SH, et al. Surgical treatment of benign fibrous histiocytoma as a form of intraspinal extradural tumor at lumbar spine. Asian Spine J 2010;4:132–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Demiralp B, Kose O, Oguz E, et al. Benign fibrous histiocytoma of the lumbar vertebrae. Skeletal Radiol 2009;38:187–91. [DOI] [PubMed] [Google Scholar]
- [7].Morales Alba NA. Posterior placement of an expandable cage for lumbar vertebral body replacement in oncologic surgery by posterior simple approach: technical note. Spine (Phila Pa 1976) 2008;33:E901–5. [DOI] [PubMed] [Google Scholar]
- [8].Balasubramanian C. Benign fibrous histiocytoma of the pterygopalatine fossa with intracranial extension. Acta Neurochir (Wien) 2007;149:100. [DOI] [PubMed] [Google Scholar]
- [9].Kuruvath S, O’Donovan DG, Aspoas AR, et al. Benign fibrous histiocytoma of the thoracic spine: case report and review of the literature. J Neurosurg Spine 2006;4:260–4. [DOI] [PubMed] [Google Scholar]
- [10].Balasubramanian C, Rajaraman G, Singh CS, et al. Benign fibrous histiocytoma of the sacrum–diagnostic difficulties facing this rare bone tumor. Pediatr Neurosurg 2005;41:253–7. [DOI] [PubMed] [Google Scholar]
- [11].van Giffen NH, van Rhijn LW, van Ooij A, et al. Benign fibrous histiocytoma of the posterior arch of C1 in a 6-year-old boy: a case report. Spine (Phila Pa 1976) 2003;28:E359–63. [DOI] [PubMed] [Google Scholar]
- [12].Li Z, Zhu N, Su J, et al. Benign fibrous histiocytoma of the frontal bone. J Craniofac Surg 2016;27:e302–4. [DOI] [PubMed] [Google Scholar]
- [13].Liu S, Zhou X, Song A, et al. Successful treatment of Gorham-Stout syndrome in the spine by vertebroplasty with cement augmentation: a case report and literature review. Medicine (Baltimore) 2018;97:e11555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Grohs JG, Nicolakis M, Kainberger F, et al. Benign fibrous histiocytoma of bone: a report of ten cases and review of literature. Wien Klin Wochenschr 2002;114:56–63. [PubMed] [Google Scholar]
- [15].Pimentel J, Fernandes A, Távora L, et al. Benign isolated fibrohistiocytic tumor arising from the central nervous system. Considerations about two cases. Clin Neuropathol 2002;21:93–8. [PubMed] [Google Scholar]
- [16].Stull DE, Kneisl JS. Incidental neoplasm in iliac crest autograft procured for anterior cervical fusion. Spine (Phila Pa 1976) 2002;27:E221–3. [DOI] [PubMed] [Google Scholar]
- [17].Peicha G, Seibert FJ, Bratschitsch G, et al. Pathologic odontoid fracture and benign fibrous histiocytoma of bone. Eur Spine J 1999;8:161–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Hoeffel JC, Boman-Ferrand F, Tachet F, et al. So-called benign fibrous histiocytoma: report of a case. J Pediatr Surg 1992;27:672–4. [DOI] [PubMed] [Google Scholar]
- [19].Wuisman P, Roessner A, Härle A, et al. Case report 503: giant cell tumor of sacrum; fibrous histiocytoma of ischium (benign); fibroushistiocytoma of tibia (benign). Skeletal Radiol 1989;17:592–7. [DOI] [PubMed] [Google Scholar]
- [20].Puopolo A, Newmarch W, Casserly B, et al. Deep benign fibrous histiocytoma of the anterior mediastinum mimicking malignancy. Lung 2017;195:503–6. [DOI] [PubMed] [Google Scholar]
- [21].Pattamparambath M, Sathyabhama S, Khatri R, et al. Benign fibrous histiocytoma of mandible: a case report and updated review. J Clin Diagn Res 2016;10:ZD24–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Doyle LA, Mariño-Enriquez A, Fletcher CD, et al. ALK rearrangement and overexpression in epithelioid fibrous histiocytoma. Mod Pathol 2015;28:904–12. [DOI] [PubMed] [Google Scholar]
- [23].Liu S, Song A, Zhou X, et al. Malignant pheochromocytoma with multiple vertebral metastases causing acute incomplete paralysis during pregnancy: literature review with one case report. Medicine (Baltimore) 2017;96:e8535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Yang L, Feng Y, Yan X, et al. Benign fibrous histiocytoma of parietal bone: case report and review of the literature. World J Surg Oncol 2015;13:177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Shimizu J, Emori M, Okada Y, et al. Arthroscopic resection for benign fibrous histiocytoma in the epiphysis of the femur. Case Rep Orthop 2018;2018:8030862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Ali-Ridha A, Brownstein S, O’Connor M, et al. Benign solitary fibrous histiocytoma of xanthomatous subtype of the perilimbal conjunctiva and adjacent sclera in a youth. Ocul Oncol Pathol 2018;4:341–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Liu S, Zhou X, Song A, et al. Successful treatment of malignant pheochromocytoma with sacrum metastases: a case report. Medicine (Baltimore) 2018;97:e12184. [DOI] [PMC free article] [PubMed] [Google Scholar]


