Key Points
Question
What is the association between oral contraceptive use and concurrent depressive symptoms in adolescents and young women?
Findings
In this cohort study of 1010 adolescents followed up for 9 years, 16-year-old oral contraceptive users showed higher concurrent depressive symptom scores compared with their counterparts not using oral contraceptives. Oral contraceptive users particularly reported more crying, eating problems, and hypersomnia compared with nonusers.
Meaning
It is important to monitor for depressive symptoms in adolescents who are using oral contraceptives, as it may affect their quality of life and put them at risk for nonadherence.
Abstract
Importance
Oral contraceptives have been associated with an increased risk of subsequent clinical depression in adolescents. However, the association of oral contraceptive use with concurrent depressive symptoms remains unclear.
Objectives
To investigate the association between oral contraceptive use and depressive symptoms and to examine whether this association is affected by age and which specific symptoms are associated with oral contraceptive use.
Design, Setting, and Participants
Data from the third to sixth wave of the prospective cohort study Tracking Adolescents’ Individual Lives Survey (TRAILS), conducted from September 1, 2005, to December 31, 2016, among females aged 16 to 25 years who had filled out at least 1 and up to 4 assessments of oral contraceptive use, were used. Data analysis was performed from March 1, 2017, to May 31, 2019.
Exposure
Oral contraceptive use at 16, 19, 22, and 25 years of age.
Main Outcomes and Measures
Depressive symptoms were assessed by the DSM-IV–oriented affective problems scale of the Youth (aged 16 years) and Adult Self-Report (aged 19, 22, and 25 years).
Results
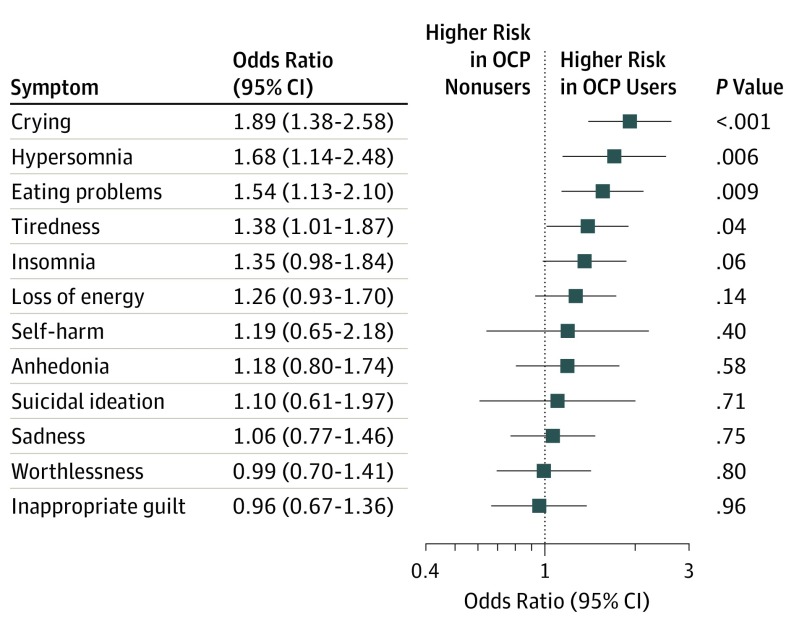
Data from a total of 1010 girls (743-903 girls, depending on the wave) were analyzed (mean [SD] age at the first assessment of oral contraceptive use, 16.3 [0.7]; (mean [SD] age at the final assessment of oral contraceptive use, 25.6 [0.6] years). Oral contraceptive users particularly differed from nonusers at age 16 years, with nonusers having a higher mean (SD) socioeconomic status (0.17 [0.78] vs –0.15 [0.71]) and more often being virgins (424 of 533 [79.5%] vs 74 of 303 [24.4%]). Although all users combined (mean [SD] ages, 16.3 [0.7] to 25.6 [0.6] years) did not show higher depressive symptom scores compared with nonusers, adolescent users (mean [SD] age, 16.5 [0.7] years) reported higher depressive symptom scores compared with their nonusing counterparts (mean [SD] age, 16.1 [0.6] years) (mean [SD] score, 0.40 [0.30] vs 0.33 [0.30]), which persisted after adjustment for age, socioeconomic status and ethnicity (β coefficient for interaction with age, –0.021; 95% CI, –0.038 to –0.005; P = .0096). Adolescent contraceptive users particularly reported more crying (odds ratio, 1.89; 95% CI, 1.38-2.58; P < .001), hypersomnia (odds ratio, 1.68; 95% CI, 1.14-2.48; P = .006), and more eating problems (odds ratio, 1.54; 95% CI, 1.13-2.10; P = .009) than nonusers.
Conclusions and Relevance
Although oral contraceptive use showed no association with depressive symptoms when all age groups were combined, 16-year-old girls reported higher depressive symptom scores when using oral contraceptives. Monitoring depressive symptoms in adolescents who are using oral contraceptives is important, as the use of oral contraceptives may affect their quality of life and put them at risk for nonadherence.
This cohort study uses data from the Tracking Adolescents’ Individual Lives Survey to investigate the association between oral contraceptive use and depressive symptoms and examine the association of age and which specific symptoms are associated with oral contraceptive use.
Introduction
Oral contraceptive pills (OCPs) are often used by women to prevent pregnancies or diminish menstrual symptoms. Successful fertility control engenders many social and economic benefits for women, such as better education and personal autonomy.1 However, 32% to 60% of women discontinue OCP use within 6 months for varying reasons, including mood changes.2,3,4 Research findings regarding OCP use and concurrent mood changes are inconsistent, and range from improved mood or having fewer mood swings5,6,7,8,9,10,11 to worsened mood12,13,14,15,16,17 or having no effect at all.18,19,20,21,22,23,24,25 These results suggest that some women may benefit from OCP use whereas others do not, or are even negatively affected.
The above-described heterogeneity in findings may be related to differences in study populations, as previous research showed that the incidence of adverse OCP effects may depend on age. In a large Swedish population-based observational study, adolescents were more likely than adult women to subsequently start using psychotropic drugs after initiation of OCP use.26 A comparable pattern was found in a Danish observational study that showed that adolescents were more prone than adult women to seek psychiatric help when using OCPs.12 This finding suggests that the first onset of clinical depression associated with OCP use is particularly salient during early adolescence. Whether this phenomenon also holds true for the concurrent presence of depressive symptoms is unknown. Alternatively, the inconsistent results might also be because of the use of diverse (unvalidated) mood questionnaires.27 Assessment of separate symptoms with validated questionnaires is of interest, as a randomized clinical trial revealed that OCP use increased mood swings and irritability symptoms, but improved depressive symptoms.28
Therefore, we aimed to examine the associations between OCP use and concurrent depressive symptoms, test whether these associations are affected by adolescence, and determine which particular symptoms—if any—are associated with OCP use. We used data from a large longitudinal survey of adolescents in the Netherlands, including 1010 adolescent girls whose depressive symptoms were assessed using validated and sensitive questionnaires for depressive symptoms in up to 4 waves for 9 years.29
Methods
Study Population
Data were derived from the Dutch population survey TRAILS (Tracking Adolescents’ Individual Lives Survey), conducted from September 1, 2005, to December 31, 2016. TRAILS is a large prospective cohort designed to investigate the psychological, social, and physical development of adolescents. Children were recruited from primary schools in 2001 and 2002.30 Exclusion criteria were serious health or language problems that would hamper full participation in the study. In total, 2230 children (mean [SD] age, 11.1 [0.6] years; 1137 girls) were enrolled in the study. After baseline assessment, children were followed up at the median ages of 13 (T2), 16 (T3), 19 (T4), 22 (T5), and 25 (T6) years.31 All follow-up assessments had good retention rates (the proportion of the baseline sample) of 96.4% (2149 of 2230; T2), 81.4% (1816 of 2230; T3), 84.3% (1881 of 2230; T4), 79.7% (1778 of 2230; T5), and 72.6% (1618 of 2230; T6). The present study included girls and young women who had filled out at least 1 and up to 4 assessments of oral contraceptive use and depressive symptoms from wave T3 to T6 (ages, 16-25 years). Oral contraceptive pill use was assessed at waves T3 to T6 with the question: “Do you use an oral contraceptive pill?” and as part of a medication list provided by the participants. Fourteen data points were excluded owing to discrepancies between the OCP use question and the medication list. An additional 37 observations were excluded as participants were using sex steroids other than OCPs or were pregnant at that point. These exclusions resulted in a final sample of 3317 data points from 1010 girls and young women (mean, 3.3 observations per participant). Excluded participants (T3, 107 of 952 [11.2%]; T4, 80 of 983 [8.1%]; T5, 111 of 937 [11.8%]; T6, 49 of 792 [6.2%]) more often had a non-Dutch ancestry (percentile difference, 5.3% at age 16 years; 8.2% at age 19 years; 8.4% at age 22 years; 9.3% at age 25 years) and a lower socioeconomic status (SES) (difference in SES score, 0.34 at age 16 years; 0.39 at age 19 years; 0.42 at age 22 years; 0.40 at age 25 years) than included girls at all waves (eTable 1 in the Supplement). The national ethical Central Committee on Research Involving Human Subjects approved the study design. At baseline (T1) parents provided written consent and children provided oral assent. At wave T2 and T3, both parents and children provided written consent and assent for participation in the new wave. From T4 to T6, children were at least 18 years of age and provided written consent.
Depressive Symptoms
Depressive symptom scores at age 16 years were assessed with the DSM-IV–oriented affective problems scale of the Youth Self-Report.32 This self-report version of the Child Behavior Checklist consists of items on crying, eating, sleeping, suicidal ideation, self-harm, feelings of worthlessness and guilt, energy, activity, sadness, and anhedonia. Items were rated as 0 = not true, 1 = a little or sometimes true, or 2 = very or often true for the preceding 6 months. At ages 19, 22, and 25 years, depressive symptoms were assessed with the affective problems scale of the Adult Self-Report,33 the adult version of the Youth Self-Report. This scale includes the above-mentioned items plus 2 items on decision-making and feelings of not being able to succeed. The scale scores represent the mean item scores (range, 0-2), with a score of more than 1 corresponding closely to the DSM-IV criteria for major depressive disorder.34 Cronbach α in our samples were 0.78 at age 16 years, 0.84 at age 19 years, 0.85 at age 22 years, and 0.87 at age 25 years.
Statistical Analysis
Statistical analysis was performed from March 1, 2017, to May 31, 2019. Population characteristics of OCP users and nonusers were compared by mean values of χ2 tests or t tests, depending on the type of variable. First, the association between OCP use and depressive symptoms was examined with a linear mixed model considering OCP use as a fixed factor. A random intercept and random slope were added to the model as they lowered the Akaike information criterion. The random intercept accounted for between-participant variability of depressive symptoms and the random slope (modeled by age) allowed for individual slopes for depressive symptoms over time. Mixed models with different covariance structures were fitted using restricted maximum likelihood estimation and, in this case, variance components was chosen based on the Akaike information criterion. The final model was estimated with maximum likelihood estimation. Second, the interaction term age × OCP use was added to the model to investigate whether age modified the association. Stratified analyses were performed to examine the association per age category using linear regression analyses. Third, associations between OCP use and specific depressive symptoms were determined using binary logistic regression analyses in the age groups that showed significance in the interaction analysis.
Exploratory Analyses
Women who experience psychological adverse effects are more likely to discontinue their OCP use (the “healthy survivor effect”).26 As this healthy survivor effect would underestimate the association between OCP use and depressive symptoms, the same mixed model was repeated in girls who had never used OCP before, in comparison with never users. As an alternative approach, duration of use (0, no OCP use; 1, 1 month-3 years; 2, 3-6 years; 3, 6-9 years, and 4, 9-12 years, based on the number of subsequent assessments with reported OCP use) was added to the model as a covariate. To explore whether the potential association between OCP use and depressive symptoms could be bidirectional, we adjusted the association between OCP use and depressive symptoms at age 16 years for depressive symptom scores at age 13 years (T2).
All analyses were conducted with and without adjustment for age (time varying), SES, and ethnicity. Age was centered and divided by 3 to ease interpretation for the effect per each 3-year increase in age. Socioeconomic status was assessed on a continuous scale based on the educational level, job, and income of the parents at study entry, with higher values indicating a better social and economic position. Ethnicity was categorized as having a Dutch or non-Dutch ancestry.
Data were analyzed using IBM SPSS Statistics, version 24 (IBM Corp) using 2-sided tests. P < .01 for the main analyses and P < .05 for the exploratory analyses were considered statistically significant.
Results
Data from 743 to 903 girls, depending on the wave, were used for the analyses. Girls had a mean (SD) age of 16.3 (0.7) years at the first assessment of OCP use (T3) and 25.6 (0.6) years at the final assessment (T6). Descriptive statistics of OCP users and nonusers are shown in the Table. Oral contraceptive pill users differed particularly from nonusers at age 16 years. During this assessment, nonusers had a higher mean (SD) SES than users (0.17 [0.78] vs –0.15 [0.71]), were more likely to be of non-Dutch ethnicity (76 of 536 [14.2%] vs 24 of 309 [7.8%]), and were more likely to be virgins (424 of 533 [79.5%] vs 74 of 303 [24.4%]).
Table. Sociodemographic and Clinical Characteristics of the Participants per Wave.
| Characteristic | 16 y of Age | 19 y of Age | 22 y of Age | 25 y of Age | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| No OCP Use (n = 536) | OCP Use (n = 309) | P Valuea | No OCP Use (n = 290) | OCP Use (n = 613) | P Valuea | No OCP Use (n = 283) | OCP Use (n = 543) | P Valuea | No OCP Use (n = 398) | OCP Use (n = 345) | P Valuea | |
| Age, mean (SD), y | 16.1 (0.6) | 16.5 (0.7) | <.001 | 18.9 (0.6) | 19.0 (0.6) | .01 | 22.3 (0.6) | 22.2 (0.7) | .004 | 25.6 (0.6) | 25.6 (0.6) | .55 |
| Dutch ethnicity, No./total No. (%) | 460/536 (85.8) | 285/309 (92.2) | .006 | 235/290 (81.0) | 564/613 (92.0) | <.001 | 235/283 (83.0) | 501/543 (92.3) | <.001 | 352/398 (88.4) | 317/345 (91.9) | .14 |
| SES, mean (SD)b | 0.17 (0.78) | −0.15 (0.71) | <.001 | 0.11 (0.77) | 0.03 (0.76) | .15 | 0.03 (0.78) | 0.12 (0.74) | .13 | 0.07 (0.77) | 0.16 (0.74) | .09 |
| BMI, mean (SD) | 21.5 (3.1) | 22.0 (3.1) | .01 | 23.3 (4.3) | 23.0 (3.7) | .26 | 24.1 (4.7) | 23.6 (4.2) | .12 | 24.1 (4.7) | 24.0 (4.8) | .58 |
| Virgin, No./total No. (%) | 424/533 (79.5) | 74/303 (24.4) | <.001 | 157/287 (54.7) | 85/613 (13.9) | <.001 | 54/283 (19.1) | 44/542 (8.1) | <.001 | 25/395 (6.3) | 17/344 (4.9) | .43 |
| Age at menarche, mean (SD), y | 13.0 (1.2) | 12.6 (1.1) | <.001 | 12.9 (1.2) | 12.9 (1.1) | .97 | 12.8 (1.2) | 12.9 (1.1) | .50 | 12.8 (1.1) | 13.0 (1.1) | .01 |
| Antidepressant use, No./total No. (%) | 0/536 (0.0) | 1/308 (0.3) | .37 | 4/290 (1.4) | 6/613 (1.0) | .74 | 11/283 (2.0) | 11/543 (2.0) | .17 | 16/398 (4.0) | 15/345 (4.3) | .86 |
| Outcome measures, No./total No. (%) | ||||||||||||
| Depressive Symptom Score, mean (SD) | 0.33 (0.30) | 0.40 (0.30) | .001 | 0.36 (0.34) | 0.36 (0.32) | .96 | 0.39 (0.34) | 0.37 (0.32) | .29 | 0.45 (0.37) | 0.42 (0.34) | .19 |
| Sadness | 184/535 (34.4) | 196/304 (35.5) | .76 | 106/290 (36.6) | 231/613 (37.7) | .77 | 100/283 (35.3) | 178/542 (32.8) | .49 | 182/398 (45.7) | 146/345 (42.3) | .37 |
| Anhedonia | 89/534 (16.7) | 63/306 (20.6) | .16 | 39/290 (13.4) | 99/613 (16.2) | .32 | 46/283 (16.3) | 72/542 (13.3) | .25 | 91/398 (22.9) | 53/345 (15.4) | .01 |
| Worthlessness | 129/534 (24.2) | 72/306 (23.5) | .87 | 80/289 (27.7) | 139/613 (22.7) | .11 | 84/283 (29.7) | 137/543 (25.2) | .19 | 140/398 (35.2) | 101/345 (29.3) | .10 |
| Inappropriate guilt | 132/533 (24.8) | 70/306 (22.9) | .56 | 60/290 (20.7) | 110/613 (17.9) | .36 | 60/283 (21.2) | 100/542 (18.5) | .35 | 109/398 (27.4) | 72/345 (20.9) | .04 |
| Indecisiveness | NA | NA | NA | 150/290 (51.7) | 286/613 (46.7) | .18 | 131/283 (46.3) | 266/542 (49.1) | .46 | 225/398 (56.5) | 181/345 (52.5) | .27 |
| Tiredness | 277/534 (51.9) | 183/305 (60.0) | .03 | 121/290 (41.7) | 301/613 (49.1) | .04 | 142/283 (50.2) | 321/542 (59.2) | .02 | 229/398 (57.2) | 217/345 (62.9) | .15 |
| Loss of energy | 242/535 (45.2) | 160/306 (52.3) | .05 | 123/290 (42.4) | 285/613 (46.5) | .25 | 150/283 (53.0) | 261/542 (48.2) | .19 | 224/398 (56.3) | 203/345 (58.8) | .50 |
| Insomnia | 181/536 (33.8) | 120/305 (39.3) | .12 | 96/290 (33.1) | 214/613 (34.9) | .60 | 139/282 (49.3) | 214/542 (39.5) | .008 | 158/398 (39.7) | 142/345 (41.2) | .71 |
| Hypersomnia | 77/535 (14.4) | 73/304 (24.0) | <.001 | 77/290 (26.6) | 177/613 (28.9) | .48 | 71/282 (25.2) | 149/541 (27.5) | .51 | 118/398 (29.6) | 115/345 (33.3) | .30 |
| Eating problems | 225/536 (42.0) | 144/305 (47.2) | .15 | 119/290 (41.0) | 257/613 (41.9) | .83 | 120/283 (42.4) | 216/543 (39.8) | .50 | 198/398 (49.7) | 175/345 (50.7) | .83 |
| Not succeeding | NA | NA | NA | 101/290 (34.8) | 171/613 (27.9) | .04 | 95/283 (33.6) | 146/542 (26.9) | .05 | 175/398 (44.0) | 109/345 (31.6) | <.001 |
| Self-harm | 32/536 (6.0) | 21/306 (6.9) | .66 | 15/290 (5.2) | 16/613 (2.6) | .05 | 9/283 (3.2) | 11/543 (2.0) | .34 | 11/398 (2.8) | 6/345 (1.7) | .46 |
| Suicidal ideation | 37/536 (6.9) | 22/305 (7.2) | .89 | 16/290 (5.5) | 26/613 (4.2) | .40 | 13/283 (4.6) | 21/543 (3.9) | .71 | 29/398 (7.3) | 13/345 (3.8) | .04 |
| Crying | 179/534 (33.5) | 146/304 (48.0) | <.001 | 104/290 (35.9) | 274/613 (44.7) | .01 | 126/283 (44.5) | 268/543 (49.4) | .21 | 171/398 (43.0) | 142/345 (41.2) | .66 |
Abbreviations: BMI, body mass index (calculated as weight in kilograms divided by height in meters squared); NA, not applicable; OCP, oral contraceptive pill; SES, socioeconomic status.
Based on 2-tailed χ2 tests for dichotomous variables or t tests for continuous variables. All variables were assessed at every wave, except for SES, ethnicity, and menarche; the first 2 were determined at study entry (T1) and the latter at age 16 years (T3).
Continuous scale based on the educational level, job, and income of the parents.
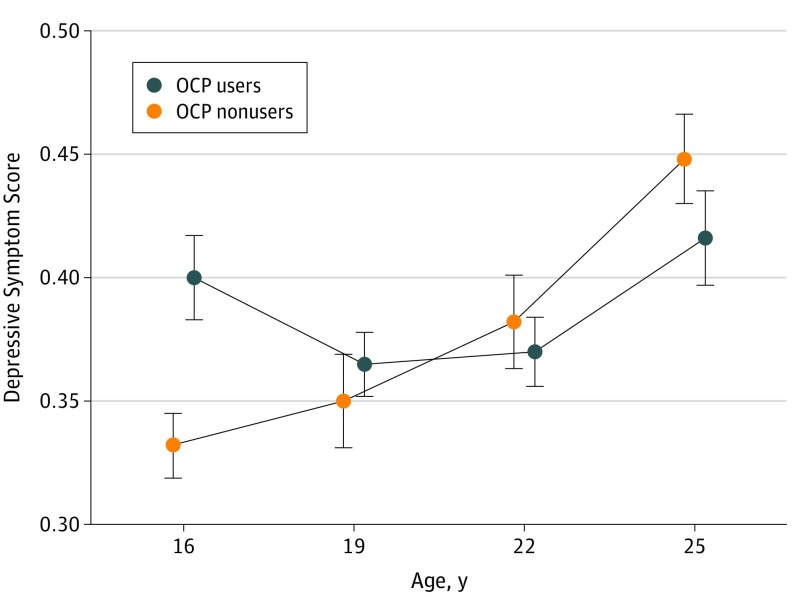
For the cohort as a whole, OCP use was not associated with higher adjusted mean scores on depressive symptoms (β coefficient, 0.006; 95% CI, –0.013 to 0.025; P = .52) (eTable 2 in the Supplement). As depicted in Figure 1, age significantly affected the association (β coefficient for age × OCP use, –0.021; 95% CI, –0.038 to –0.005; P = .0096); this association was driven by differences in 16-year-old girls. At this age, girls who used OCP had higher concurrent depressive symptom scores than their nonusing counterparts (mean [SD] score, 0.40 [0.30] vs 0.33 [0.30]; β coefficient, 0.075; 95% CI, 0.033-0.120; P < .001). The depressive symptom item scores at age 16 years are depicted in Figure 2. Use of OCPs was associated with more crying (odds ratio, 1.89; 95% CI, 1.38-2.58; P < .001), eating problems (odds ratio, 1.54; 95% CI, 1.13-2.10; P = .009), and hypersomnia (odds ratio, 1.68; 95% CI, 1.14-2.48; P = .006) compared with nonusers.
Figure 1. Oral Contraceptive Pill (OCP) Use and Depressive Symptom Scores by Age Groups.
Data are (adjusted) mean values with 1 SE for the depressive symptom score through multilevel analysis (ie, mixed models). Analyses were adjusted for age, ethnicity, and socioeconomic status (β coefficient for interaction = −0.021 [95% CI, −0.038 to −0.005; P = .0096]).
Figure 2. Depressive Item Scores According to Oral Contraceptive Pill (OCP) Use in Youngest Age Group.
Data are (adjusted) odds ratio with 95% CIs for severe and moderate symptoms vs the reference group (no symptoms) through binary logistic regression analysis. The mean (SD) age in the youngest age group was 16.3 (0.7) years.
Exploratory Analyses
Excluding girls who used OCP previously (regardless of whether they continued or discontinued use) strengthened the association between OCP use and depressive symptoms for the whole cohort (β coefficient for first-time OCP use, 0.021; 95% CI, –0.005 to 0.046; P = .11) (eTable 2 in the Supplement). Also, long-term OCP use (up to 3, 6, 9, and 12 years) decreased the likelihood of higher depressive symptoms scores, although this finding was not significant (β coefficient, –0.007; 95% CI, –0.024 to 0.009; P = .39). Adjustment for depressive symptom scores before OCP use weakened the association in 16-year-old girls (β coefficient for OCP use, 0.040; 95% CI, 0.001-0.079; P = .046).
Post Hoc Analyses
To investigate whether the age-specific association may be explained by residual confounding or preexisting differences, we explored which differences between OCP users and nonusers were already present at the age of 13 years, when OCP use was unlikely. At age 13 years, the 16-year-old user group was more often sexually active, experienced more stressful events, and more often had menstrual-related pain and acne than the 16-year-old nonuser group (eTable 3 in the Supplement). In subsequent analyses, these factors were added one by one to the adjusted model (that already included age, ethnicity, and SES) to examine whether they individually affected the association. All 4 factors weakened the association (change in β coefficient for OCP use: virginity, –9.3%; stressful events, –26.7%; menstrual-related pain, –21.3%; acne, –17.3%). However, none of the factors diminished the association between OCP use and depressive symptoms at age 16 years.
Discussion
In this large cohort study of adolescents, we did not find support for an overall association between OCP use and depressive symptoms among young women. However, 16-year-old girls using OCPs did report higher concurrent depressive symptom scores compared with their nonusing counterparts. When individual depressive symptoms were examined, 16-year-old girls using OCPs particularly reported more crying, hypersomnia, and eating problems, while the required symptoms for the diagnosis of depression—anhedonia and sadness—were unaffected. Adjusting the association between OCP use and depressive symptoms for pre-OCP use depressive symptoms in 16-year-old girls reduced the magnitude of the association, but did not eliminate the significance of the findings.
Earlier clues of a possible association between OCP use in adolescence and vulnerability for depressive symptoms come from 2 large population-based studies.12,26 These studies showed that adolescent girls using OCP were the most likely to subsequently start using psychotropic drugs or be admitted to a psychiatric hospital compared with nonusers. Our study provides important evidence that this vulnerable group is the most likely to report more concurrent depressive symptoms.
This age-specific vulnerability is complex and likely bidirectional. Important emotion-related regions of the brain, such as the amygdala, prefrontal cortex, and hippocampus, are still maturing during adolescence and therefore may be particularly sensitive to sex hormone–related changes at age 16 years.35 In addition, ages 15 to 18 years may be a critical time for the development of depression in girls, as the sex gap in the incidence of depression dramatically increases during this period.36 Neurobiological studies support this idea, as sex hormones are capable of directly influencing gene expression in the cell nuclei in the brain.37 Furthermore, the use of OCPs affects sex hormone levels, including androgens and stress hormones.38 Although almost nothing is known about the effect of OCP use on the maturation of the brain,35 these findings confirm its potential capability to do so.
In addition to the possibility that OCP use leads to more depressive symptoms, the weakened strength of the association after adjustment for pre-OCP use depressive symptoms in 16-year-old girls also points toward an association in the reverse direction. A previous study demonstrated that mood worsening among individuals using OCPs was more likely in users with a history of depression.25 This finding might suggest that a subset of women with depression may be at risk for OCP-associated mood deterioration because they are particularly sensitive to interactions between cycling gonadal steroids and affect. Also, treatment with OCPs is standard care for cycle-related mood problems39 and for polycystic ovarian syndrome, which is associated with depressive symptoms.40 Therefore, mood-related physical problems may also be involved in the association between OCP use and depressive symptoms. Hence, some girls in our sample may have initiated OCP use because they experienced mood problems.
Residual confounding could have led to overestimation of the association. Differences between OCP users and nonusers that were likely present before the initiation of OCP use weakened the strength of the association at age 16 years, although none of the differences completely diminished the association. However, future studies should replicate and consider the importance of these factors. In contrast, the healthy survivor effect may have led to underestimation of the association between OCP use and depressive symptoms. In support of this possibility, comparing first-time users with never-users yielded a stronger association. This finding is in line with findings from a Danish study in which the highest relative risks (RRs) for a diagnosis of depression were found after 6 months (RR, 1.5) of use, whereas RRs were smaller when individuals had used OCP for longer periods (RR, 1.2 for use of 1-3 years; RR, 0.9 for use of 4-6 years; RR, 0.8 for use of 7-9 years).12 Our finding, combined with these data, suggests that the absence of an association in older individuals may be owing to the healthy survivor effect. The possibility of an underestimation in our younger participants cannot be excluded either, because information on OCP use before the age of 16 years was not available and thus could not be explored.
One may wonder why hypersomnia, eating problems, and increased crying, rather than core symptoms of depression such as sadness, were reported more commonly among adolescent OCP users. These symptoms fit well in symptom profiles of adolescent depression, where, in contrast to adult depression, the emphasis is more on vegetative or physical disturbances (eg, loss of energy, changes in weight, and appetite and sleep changes) than on anhedonia.41 The symptom-specific findings are also in line with results from a randomized clinical trial, which showed that OCP use worsened mood swings and irritability symptoms, but improved depressive symptoms such as sadness,28 suggesting that associations between OCP use and mood may be symptom specific.
Strengths and Limitations
Strengths of this study are the availability of multiple time points when depressive symptoms were assessed with the use of well-validated instruments. Also, we were able to adjust for important covariates such as SES and ethnicity. The assessment of depressive symptoms provides a complementary, more inclusive perspective on the association between OCP use and depression across the severity spectrum to existing studies using proxies such as incident clinical depression. More important, subclinical depressive symptoms may also cause distress in adolescents, and many adolescents with psychiatric problems in the clinical range do not seek help.42 Moreover, the concurrent examination of OCP use and depressive symptom severity over several years contributes to a better insight into the real-time risks associated with OCP use.
This study has some limitations. Use of observational data precludes any causal inference. Also, this longitudinal analysis does not provide information about specific OCPs. Because very few girls specified OCP use as medication use, we were unable to conduct separate analyses for different kinds of OCPs. However, we checked which OCPs were used in a comparable cohort of girls born in the same year and postal code as the girls included in this study (InterActive DataBase [IADB] cohort).43 In that cohort, most of the girls used either monophasic (81.1%) or triphasic ethinyl estradiol and levonorgestrel (2.5%), followed by ethinyl estradiol and drospirenone (4.6%) and ethinyl estradiol and desogestrel (1.8%). Hence, testing for specific components was not useful, as probably almost all participants were using the same type of progestin. Finally, generalizability of results to other countries might be limited owing to differences in the acceptability of and access to contraception across societies.44 For example, unlike Dutch teenagers, not all teenagers in the United States have access to no-cost contraception.45,46 Such differences have implications for group characteristics of girls using OCPs and, hence, associated outcomes.
Although we cannot be conclusive about the direction of the association, awareness of the possible existence of mood problems among OCP-using adolescents is critical to the health and development of adolescents. Depressive symptoms may reduce quality of life47 and OCP use adherence,48 because, for example, girls may attribute these depressive symptoms to the use of OCPs, potentially resulting in unwanted teenage pregnancies. To lower the probability of an unintended conception, long-acting reversible contraceptives are recommended as a first-line option.49,50 However, those are only used by a minority of teenagers in both the Netherlands and the United States.51,52 We do not suggest limiting OCP use to counterbalance this risk for depressive symptoms. In fact, previous research has shown benefits of OCP use such as improvement of dysmenorrhea and premenstrual syndrome49; additionally, OCP use is much safer than pregnancy and associated postpartum depression risks.50
Conclusions
In this study, 16-year-old OCP users showed higher concurrent depressive symptom scores compared with their nonusing counterparts. Overall, adolescent girls are more likely to report increased crying, hypersomnia, and eating problems while taking OCPs, but these symptoms seem to diminish once they enter adulthood. Monitoring these symptoms is important, as they may affect their quality of life and put them at risk for nonadherence.
eTable 1. Sociodemographic and Clinical Characteristics of Included and Excluded Girls Per Wave
eTable 2. Associations Between OCP Use and Depressive Symptom Scores During 9 Years of Follow-up
eTable 3. Post Hoc Comparison of Sociodemographic and Clinical Variables Among OCP Users and Nonusers at Age 16 Years and at Age 13 Years When OCP Use Was Unlikely
References
- 1.Sonfield A, Hasstedt K, Kavanaugh M, Anderson R. The Social and Economic Benefits of Women’s Ability to Determine Whether and When to Have Children. New York, NY: Guttmacher Institute; 2013. [Google Scholar]
- 2.Westhoff CL, Heartwell S, Edwards S, et al. Oral contraceptive discontinuation: do side effects matter? Am J Obstet Gynecol. 2007;196(4):412.e1-412.e6. doi: 10.1016/j.ajog.2006.12.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sanders SA, Graham CA, Bass JL, Bancroft J. A prospective study of the effects of oral contraceptives on sexuality and well-being and their relationship to discontinuation. Contraception. 2001;64(1):51-58. doi: 10.1016/S0010-7824(01)00218-9 [DOI] [PubMed] [Google Scholar]
- 4.Rosenberg MJ, Waugh MS. Oral contraceptive discontinuation: a prospective evaluation of frequency and reasons. Am J Obstet Gynecol. 1998;179(3, pt 1):577-582. doi: 10.1016/S0002-9378(98)70047-X [DOI] [PubMed] [Google Scholar]
- 5.Kristjánsdóttir J, Olsson GI, Sundelin C, Naessen T. Self-reported health in adolescent girls varies according to the season and its relation to medication and hormonal contraception—a descriptive study. Eur J Contracept Reprod Health Care. 2013;18(5):343-354. doi: 10.3109/13625187.2013.821107 [DOI] [PubMed] [Google Scholar]
- 6.Toffol E, Heikinheimo O, Koponen P, Luoto R, Partonen T. Further evidence for lack of negative associations between hormonal contraception and mental health. Contraception. 2012;86(5):470-480. doi: 10.1016/j.contraception.2012.02.014 [DOI] [PubMed] [Google Scholar]
- 7.Sucato GS, Land SR, Murray PJ, Cecchini R, Gold MA. Adolescents’ experiences using the contraceptive patch versus pills. J Pediatr Adolesc Gynecol. 2011;24(4):197-203. doi: 10.1016/j.jpag.2011.02.001 [DOI] [PubMed] [Google Scholar]
- 8.Ott MA, Shew ML, Ofner S, Tu W, Fortenberry JD. The influence of hormonal contraception on mood and sexual interest among adolescents. Arch Sex Behav. 2008;37(4):605-613. doi: 10.1007/s10508-007-9302-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.O’Connell K, Davis AR, Kerns J. Oral contraceptives: side effects and depression in adolescent girls. Contraception. 2007;75(4):299-304. doi: 10.1016/j.contraception.2006.09.008 [DOI] [PubMed] [Google Scholar]
- 10.Coffee AL, Sulak PJ, Kuehl TJ. Long-term assessment of symptomatology and satisfaction of an extended oral contraceptive regimen. Contraception. 2007;75(6):444-449. doi: 10.1016/j.contraception.2007.01.014 [DOI] [PubMed] [Google Scholar]
- 11.Kutner SJ, Brown WL. Types of oral contraceptives, depression, and premenstrual symptoms. J Nerv Ment Dis. 1972;155(3):153-162. doi: 10.1097/00005053-197209000-00001 [DOI] [PubMed] [Google Scholar]
- 12.Skovlund CW, Mørch LS, Kessing LV, Lidegaard Ø. Association of hormonal contraception with depression. JAMA Psychiatry. 2016;73(11):1154-1162. doi: 10.1001/jamapsychiatry.2016.2387 [DOI] [PubMed] [Google Scholar]
- 13.Gingnell M, Engman J, Frick A, et al. Oral contraceptive use changes brain activity and mood in women with previous negative affect on the pill—a double-blinded, placebo-controlled randomized trial of a levonorgestrel-containing combined oral contraceptive. Psychoneuroendocrinology. 2013;38(7):1133-1144. doi: 10.1016/j.psyneuen.2012.11.006 [DOI] [PubMed] [Google Scholar]
- 14.Hall KS, White KO, Rickert VI, Reame N, Westhoff C. Influence of depressed mood and psychological stress symptoms on perceived oral contraceptive side effects and discontinuation in young minority women. Contraception. 2012;86(5):518-525. doi: 10.1016/j.contraception.2012.04.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wiebe ER, Brotto LA, MacKay J. Characteristics of women who experience mood and sexual side effects with use of hormonal contraception. J Obstet Gynaecol Can. 2011;33(12):1234-1240. doi: 10.1016/S1701-2163(16)35108-8 [DOI] [PubMed] [Google Scholar]
- 16.Kulkarni J. Depression as a side effect of the contraceptive pill. Expert Opin Drug Saf. 2007;6(4):371-374. doi: 10.1517/14740338.6.4.371 [DOI] [PubMed] [Google Scholar]
- 17.Herzberg BN, Johnson AL, Brown S. Depressive symptoms and oral contraceptives. BMJ. 1970;4(5728):142-145. doi: 10.1136/bmj.4.5728.142 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cheslack-Postava K, Keyes KM, Lowe SR, Koenen KC. Oral contraceptive use and psychiatric disorders in a nationally representative sample of women. Arch Womens Ment Health. 2015;18(1):103-111. doi: 10.1007/s00737-014-0453-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Berenson AB, Odom SD, Breitkopf CR, Rahman M. Physiologic and psychologic symptoms associated with use of injectable contraception and 20 microg oral contraceptive pills. Am J Obstet Gynecol. 2008;199(4):351.e1-351.e12. doi: 10.1016/j.ajog.2008.04.048 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rosenthal SL, Cotton S, Ready JN, Potter LS, Succop PA. Adolescents’ attitudes and experiences regarding levonorgestrel 100 mcg/ethinyl estradiol 20 mcg. J Pediatr Adolesc Gynecol. 2002;15(5):301-305. doi: 10.1016/S1083-3188(02)00196-1 [DOI] [PubMed] [Google Scholar]
- 21.Oinonen KA, Mazmanian D. Effects of oral contraceptives on daily self-ratings of positive and negative affect. J Psychosom Res. 2001;51(5):647-658. doi: 10.1016/S0022-3999(01)00240-9 [DOI] [PubMed] [Google Scholar]
- 22.Oddens BJ. Women’s satisfaction with birth control: a population survey of physical and psychological effects of oral contraceptives, intrauterine devices, condoms, natural family planning, and sterilization among 1466 women. Contraception. 1999;59(5):277-286. doi: 10.1016/S0010-7824(99)00034-7 [DOI] [PubMed] [Google Scholar]
- 23.Rubino-Watkins MF, Doster JA, Franks S, et al. Oral contraceptive use: implications for cognitive and emotional functioning. J Nerv Ment Dis. 1999;187(5):275-280. doi: 10.1097/00005053-199905000-00002 [DOI] [PubMed] [Google Scholar]
- 24.Walker A, Bancroft J. Relationship between premenstrual symptoms and oral contraceptive use: a controlled study. Psychosom Med. 1990;52(1):86-96. doi: 10.1097/00006842-199001000-00007 [DOI] [PubMed] [Google Scholar]
- 25.Joffe H, Cohen LS, Harlow BL. Impact of oral contraceptive pill use on premenstrual mood: predictors of improvement and deterioration. Am J Obstet Gynecol. 2003;189(6):1523-1530. doi: 10.1016/S0002-9378(03)00927-X [DOI] [PubMed] [Google Scholar]
- 26.Zettermark S, Perez Vicente R, Merlo J. Hormonal contraception increases the risk of psychotropic drug use in adolescent girls but not in adults: a pharmacoepidemiological study on 800 000 Swedish women. PLoS One. 2018;13(3):e0194773. doi: 10.1371/journal.pone.0194773 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Böttcher B, Radenbach K, Wildt L, Hinney B. Hormonal contraception and depression: a survey of the present state of knowledge. Arch Gynecol Obstet. 2012;286(1):231-236. doi: 10.1007/s00404-012-2298-2 [DOI] [PubMed] [Google Scholar]
- 28.Lundin C, Danielsson KG, Bixo M, et al. Combined oral contraceptive use is associated with both improvement and worsening of mood in the different phases of the treatment cycle—a double-blind, placebo-controlled randomized trial. Psychoneuroendocrinology. 2017;76:135-143. doi: 10.1016/j.psyneuen.2016.11.033 [DOI] [PubMed] [Google Scholar]
- 29.Ormel J, Oldehinkel AJ, Sijtsema J, et al. The TRacking Adolescents’ Individual Lives Survey (TRAILS): design, current status, and selected findings. J Am Acad Child Adolesc Psychiatry. 2012;51(10):1020-1036. doi: 10.1016/j.jaac.2012.08.004 [DOI] [PubMed] [Google Scholar]
- 30.de Winter AF, Oldehinkel AJ, Veenstra R, Brunnekreef JA, Verhulst FC, Ormel J. Evaluation of non-response bias in mental health determinants and outcomes in a large sample of pre-adolescents. Eur J Epidemiol. 2005;20(2):173-181. doi: 10.1007/s10654-004-4948-6 [DOI] [PubMed] [Google Scholar]
- 31.Oldehinkel AJ, Rosmalen JGM, Buitelaar JK, et al. Cohort profile update: the tracking adolescents’ individual lives survey (TRAILS). Int J Epidemiol. 2015;44(1):76-76n. doi: 10.1093/ije/dyu225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Achenbach TM, Rescorla LA. Manual for the ASEBA School-Age Forms & Profiles. Burlington, VT: University of Vermont, Research Center for Children, Youth, & Families; 2001. [Google Scholar]
- 33.Achenbach TM, Rescorla LA. Manual for the ASEBA Adult Forms & Profiles. Burlington, VT: University of Vermont, Research Center for Children, Youth, & Families; 2003. [Google Scholar]
- 34.Achenbach TM, Dumenci L, Rescorla LA Ratings of relations between DSM-IV diagnostic categories and items of the Adult Self-Report (ASR) and Adult Behavior Checklist (ABCL). https://aseba.org/wp-content/uploads/2019/02/dsm-adultratings.pdf. Published February 13, 2003. Accessed January 1, 2018.
- 35.Cahill L. How does hormonal contraception affect the developing human adolescent brain? Curr Opin Behav Sci. 2018;23:131-135. doi: 10.1016/j.cobeha.2018.06.015 [DOI] [Google Scholar]
- 36.Hankin BL, Abramson LY, Moffitt TE, Silva PA, McGee R, Angell KE. Development of depression from preadolescence to young adulthood: emerging gender differences in a 10-year longitudinal study. J Abnorm Psychol. 1998;107(1):128-140. doi: 10.1037/0021-843X.107.1.128 [DOI] [PubMed] [Google Scholar]
- 37.Heberden C. Sex steroids and neurogenesis. Biochem Pharmacol. 2017;141:56-62. doi: 10.1016/j.bcp.2017.05.019 [DOI] [PubMed] [Google Scholar]
- 38.Eisenhofer G, Peitzsch M, Kaden D, et al. Reference intervals for plasma concentrations of adrenal steroids measured by LC-MS/MS: impact of gender, age, oral contraceptives, body mass index and blood pressure status. Clin Chim Acta. 2017;470:115-124. doi: 10.1016/j.cca.2017.05.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Casper RF, Yonkers KA Treatment of premenstrual syndrome and premenstrual dysphoric disorder. https://www.uptodate.com/contents/treatment-of-premenstrual-syndrome-and-premenstrual-dysphoric-disorder. Accessed January 1, 2019.
- 40.Martin KA, Anderson RR, Chang RJ, et al. Evaluation and treatment of hirsutism in premenopausal women: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2018;103(4):1233-1257. doi: 10.1210/jc.2018-00241 [DOI] [PubMed] [Google Scholar]
- 41.Rice F, Riglin L, Lomax T, et al. Adolescent and adult differences in major depression symptom profiles. J Affect Disord. 2019;243:175-181. doi: 10.1016/j.jad.2018.09.015 [DOI] [PubMed] [Google Scholar]
- 42.Jörg F, Visser E, Ormel J, Reijneveld SA, Hartman CA, Oldehinkel AJ. Mental health care use in adolescents with and without mental disorders. Eur Child Adolesc Psychiatry. 2016;25(5):501-508. doi: 10.1007/s00787-015-0754-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Visser ST, Schuiling-Veninga CC, Bos JH, de Jong-van den Berg LT, Postma MJ. The population-based prescription database IADB.nl: its development, usefulness in outcomes research and challenges. Expert Rev Pharmacoecon Outcomes Res. 2013;13(3):285-292. doi: 10.1586/erp.13.20 [DOI] [PubMed] [Google Scholar]
- 44.Higgins JA, Smith NK. The sexual acceptability of contraception: reviewing the literature and building a new concept. J Sex Res. 2016;53(4-5):417-456. doi: 10.1080/00224499.2015.1134425 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zorginstituut Nederland Anticonceptiemiddelen (Zvw). https://www.zorginstituutnederland.nl/Verzekerde+zorg/a/anticonceptiemiddelen-zvw. Accessed May 1, 2019.
- 46.Guttmacher Institute Insurance Coverage of Contraceptives. https://www.guttmacher.org/state-policy/explore/insurance-coverage-contraceptives. Accessed May 1, 2019. [Google Scholar]
- 47.Rogers J, Hengartner MP, Angst J, Ajdacic-Gross V, Rössler W. Associations with quality of life and the effect of psychopathology in a community study. Soc Psychiatry Psychiatr Epidemiol. 2014;49(9):1467-1473. doi: 10.1007/s00127-014-0841-0 [DOI] [PubMed] [Google Scholar]
- 48.Hall KS, Moreau C, Trussell J, Barber J. Role of young women’s depression and stress symptoms in their weekly use and nonuse of contraceptive methods. J Adolesc Health. 2013;53(2):241-248. doi: 10.1016/j.jadohealth.2013.02.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.ACOG Practice Bulletin No ACOG Practice Bulletin No. 110: noncontraceptive uses of hormonal contraceptives. Obstet Gynecol. 2010;115(1):206-218. doi: 10.1097/AOG.0b013e3181cb50b5 [DOI] [PubMed] [Google Scholar]
- 50.Committee on Adolescence Contraception for adolescents. Pediatrics. 2014;134(4):e1244-e1256. doi: 10.1542/peds.2014-2299 [DOI] [PubMed] [Google Scholar]
- 51.Kavanaugh ML, Jerman J. Contraceptive method use in the United States: trends and characteristics between 2008, 2012 and 2014. Contraception. 2018;97(1):14-21. doi: 10.1016/j.contraception.2017.10.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.de Graaf H, van den Borne M, Nikkelen S, Twisk D, Meijer S. Seksuele gezondheid van jongeren in Nederland anno 2017. Delft, The Netherlands: Rutgers and Soa Aids Nederland; 2017. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eTable 1. Sociodemographic and Clinical Characteristics of Included and Excluded Girls Per Wave
eTable 2. Associations Between OCP Use and Depressive Symptom Scores During 9 Years of Follow-up
eTable 3. Post Hoc Comparison of Sociodemographic and Clinical Variables Among OCP Users and Nonusers at Age 16 Years and at Age 13 Years When OCP Use Was Unlikely