Abstract
Stressed eukaryotic cells store mRNAs in protein-rich condensates called stress granules. Using single-molecule tracking techniques to examine how mRNAs enter stress granules, a new study shows that mRNAs make transient contacts with the granule surface before stable association, and become largely immobile after entry.
Stress granules (SGs) are RNA–protein condensates that form in the cytoplasm of cells exposed to acute stress. Following arsenite treatment or heat shock, translation initiation is blocked. Ribosomes complete elongation and release thousands of polysome-free messenger (m)RNAs unable to initiate further rounds of translation. The stalled translation initiation complexes, including translation initiation factors and RNA-binding proteins, coalesce to form SGs1. Sequencing analyses of the stress granule transcriptomes from yeast and mammalian cells have revealed that >99% of mRNA species enter SGs. Most mRNAs, however, enter in low numbers, with only 10% of bulk mRNA stably located in SGs2. Many studies have analysed the dynamics of proteins during SG assembly, but how mRNAs enter the granules had not been addressed until now.
In this issue of Nature Cell Biology, Moon et al. adapted the recently developed nascent chain tracking technique to simultaneously visualize the translation and dynamic state of single mRNA molecules in the cytoplasm of stressed cells3. The cells also expressed a Ras-GAP SH3 domain-binding protein (G3BP)–GFP fusion to mark SGs. The authors tracked three mRNAs of differing sizes: H2B (375 nt), KDM5B (4632 nt) and p300 (8625 nt). mRNA–SG interactions could be described by a two-state model, with a fast state lasting only a few seconds and a slow state lasting several minutes. Three parameters affect the frequency of each state: translation status of the RNA, size of the RNA and size of the granule3. RNAs undergoing translation were only rarely observed in stable interactions. This observation is consistent with bulk measurements that reveal an anti-correlation between ribosome occupancy and SG enrichment2. Ribosome-free status, therefore, is likely to be a prerequisite for granule entry. Most interactions between ribosome-free mRNAs and SGs, however, were short-lived with only a few resulting in stable entry. Entry into SGs is an inefficient process, which explains why, for most mRNA species, the majority of mRNA molecules remain in the cytoplasm.
The frequency of stable interactions increased for longer mRNAs and larger SGs, consistent with bulk measurements showing an enrichment for longer mRNAs in the SG transcriptome2. The size dependency suggests that stable interactions require binding events that scale with the length of the RNA and the size of the granule. What mediates those binding events? The authors observed two cases where a KDM5B mRNA molecule engaged in a stable interaction with an SG, with one end extended beyond the granule border for a few seconds before snapping back into place. In situ hybridization experiments confirmed that a long mRNA is more likely than a shorter mRNA to have ends that extend beyond the granule boundary3. An intriguing possibility is that transiently extended mRNAs interact with other mRNAs at the granule surface and pull them in (Fig. 1). Several lines of evidence have suggested that RNA–RNA interactions contribute to RNA granule assembly4. RNA secondary structure has been shown to influence sorting of mRNAs into different RNA granules in a syncytial fungus5, and various RNA species have been reported to aggregate in vitro without the help of any protein4. Most remarkably, precipitation of total yeast RNA with the molecular crowder PEG, yields a precipitate whose composition resembles the SG transcriptome4. In cells, untranslated mRNAs adopt a more compact configuration than translated mRNAs6,7, with RNA compaction proceeding from the 5’ to 3’ end6. These observations suggest that ribosome run-off releases naked RNA molecules that fold through self-interactions.
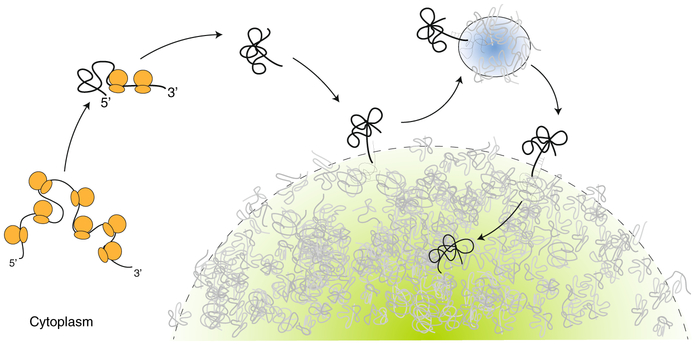
Fig. 1 ∣. A model for mRNA entry into stress granules.
mRNA (black line) undergoing translation in a stressed cell is unable to reinitiate translation and condense as ribosomes (orange) run off. The condensed mRNA diffuses freely in the cytoplasm until a chance encounter with a stress granule (green). The mRNA makes transient contact with the granule surface and visits a P-body (blue) before entering the stress granule. Contacts are mediated by transient pairing interactions with resident granule mRNAs (grey), whose ends extend past the granule surface.
mRNAs in SGs were more than 1,000 times less dynamic than free mRNAs in the cytoplasm; three KDM5B molecules trapped in the same SG were observed to maintain the same relative positions for at least 10 minutes3. These observations suggest that, once in the granules, RNAs experience a stable, non-dynamic environment. Some proteins in SGs exchange rapidly with the cytoplasm (such as G3BP1), while others exhibit a significant immobile fraction (such as IGF2BP1, YB1 and HUR)8. Protein density across SGs is uneven, with dense regions (cores) surrounded by more dilute and dynamic regions (shells)1. One possibility is that, while most proteins interact reversibly with SGs, RNAs make so many interactions with other RNAs as to become kinetically trapped.
Other types of cytoplasmic RNA granules exist in eukaryotic cells, including P-bodies, neuronal granules and germ granules. The current view is that all these condensates behave like viscous liquids: resident proteins are dynamic in vivo and spontaneously phase-separate into liquid droplets in vitro1. Moon et al. observed that mRNAs interact with P-bodies in similar ways to SGs: they form many transient interactions and fewer stable interactions that are enhanced by RNA length and granule size. In cells starved for amino acids, it has been estimated that 60% of mRNA molecules held in P-bodies do not exchange with cytoplasm9. In the germ granules of Drosophila, mRNAs reside in homotypic clusters arranged in stereotypical locations, suggesting a stable organization that is inconsistent with a liquid medium10,11. Kinetically trapped mRNAs, therefore, may be a common feature of RNA granules. If so, how do RNA granules disassemble? SGs contain many ATPases, including RNA helicases, protein chaperones and AAA-ATPases12. In vitro, the N-terminal disordered domain of the helicase Ddx4 forms phase-separated droplets that melt DNA duplexes and selectively exclude certain oligonucleotides13. One possibility is that restoration of ATP levels during stress recovery stimulates the dynamic protein environment to melt RNA duplexes, releasing mRNAs to the cytoplasm where they are free to resume translation.
Several neurodegenerative diseases have been associated with cytoplasmic aggregates that contain RNA-binding proteins also found in SGs. It has been proposed that the long lifespan and high metabolism of neurons make them prone to accumulating stress granule remnants that seed stable aggregates14. Repeat-expansion mRNAs, such as C9orf72 (associated with amyotrophic lateral sclerosis), can form toxic RNA foci in cells4. Understanding how healthy cells control RNA aggregation will be an important goal for future studies. Fortunately, the exciting new tools of single-molecule imaging hold great promise for moving this field forward at a brisk pace15.
Footnotes
Competing interests
The authors declare no competing interests.
References
- 1.Fay MM & Anderson PJ J. Mol. Biol 430, 4685–4701 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Khong A et al. Mol. Cell 68, 808–820 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Moon S et al. Nat. Cell Biol 10.1038/s41556-018-0263-4 (2019). [DOI] [PMC free article] [PubMed]
- 4.Van Treeck B & Parker R Cell 174, 791–802 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Langdon EM et al. Science 360, 922–927 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Khong A & Parker RJ Cell Biol. 217, 4124–4140 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Adivarahan S et al. Mol. Cell 72, 727–738e5 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bley N et al. Nucleic Acid. Res 43, e26 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Aizer A et al. J. Cell. Sci 127, 4443–4456 (2014). [DOI] [PubMed] [Google Scholar]
- 10.Trcek T et al. Nat. Commun 6, 7962 (2015). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Eagle WVI, Yeboah-Kordieh DK, Niepielko MG & Gavis ER Development 145, dev164657 (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Jain S et al. Cell 164, 487–498 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nott TJ, Craggs TD & Baldwin AJ Nat. Chem 8, 569–575 (2016). [DOI] [PubMed] [Google Scholar]
- 14.Dobra I et al. Curr. Neurol. Neurosci. Rep 18, 107 (2018). [DOI] [PubMed] [Google Scholar]
- 15.Pichon X, Lagha M, Mueller F & Bertrand E Mol. Cell 71, 468–480 (2018). [DOI] [PubMed] [Google Scholar]