Abstract
Glycyrrhiza, commonly known as licorice, is a herbal medicine that has been used for thousands of years. Licorice contains multiple flavonoids, which possess a variety of biological activities. On the basis of the anti-inflammatory effects of licorice flavonoids, the potential mechanism of action was investigated via a plasma metabolomics approach. A total of 9 differential endogenous metabolites associated with the therapeutic effect of licorice flavonoids were identified, including linoleic acid, sphingosine, tryptophanamide, corticosterone and leukotriene B4. Besides classical arachidonic acid metabolism, metabolism of sphingolipids, tryptophan and fatty acids, phospholipids synthesis, and other pathways were also involved. The multi-pathway integrated adjustment mechanism of licorice flavonoid action may reduce side effects in patients, along with any anti-inflammatory functions, which provides a foundation for identifying and developing novel, high-potential natural drugs with fewer side effects for clinical application.
Keywords: licorice flavonoids, anti-inflammatory activity, multi-pathway, mechanism
Introduction
Inflammation is considered to be a primary physiological defense mechanism and is associated with the protection of the body against burns, infections, toxic chemicals, allergens and other harmful stimuli. These stimuli may induce local and systemic effects, signs of which may include pain, redness, swelling, heat, and loss of function in the affected area (1,2). The mechanisms and mediators that contribute to the induction and exacerbation of inflammation are complex, and may involve enzyme activation, mediator release, extravasation of fluid, cell migration, tissue breakdown and tissue repair (3). Inflammation may become a causative factor for other chronic diseases, including rheumatoid arthritis (4,5) and atherosclerosis (6,7), if not controlled. Current treatments for inflammation include non-steroidal anti-inflammatory drugs and steroids; although these treatments have merits, potential risks and side effects (particularly to the gastrointestinal tract) are widely reported, and restrict clinical application (8,9). Numerous traditional Chinese medicines (TCMs) have been widely used as preventive or therapeutic strategies in modern medicine. TCMs boast antioxidant, anti-inflammatory, anti-apoptotic and autophagic regulatory functions, of which the anti-inflammatory activities have been extensively reported (10–12).
Glycyrrhiza comes from the roots and rhizomes of Glycyrrhiza uralensis Fisch., Glycyrrhiza inflata Bat. or Glycyrrhiza glabra L. (13). Use of Glycyrrhiza was first reported in ‘Shen Nong's Herbal Classic’ in around 2100 BC and recorded in each version of ‘Chinese Pharmacopoeia’. Glycyrrhiza is a widely used herbal medicine native to southern Europe and regions of Asia. In China it is widespread in Inner Mongolia, Gansu, Heilongjiang, Ningxia, Qinghai and a number of other provinces (14). Owing to Glycyrrhiza's diverse chemical composition and its wide range of pharmacological effects, it has been documented as having various therapeutic applications (15). The main components of licorice are triterpenoids and flavonoids (16,17). Licorice flavonoids contain licochalcone A, liquiritigenin, glabridin, glycyrol and numerous other monomer components that are known to have biological activities, which include anti-inflammatory, antidepressant, and antitumor effects (18–21).
In the present study, a metabolomic approach was applied to investigate the anti-inflammatory effect of licorice flavonoids in formaldehyde-injected mice, and to identify the metabolic pathways and potential biomarkers associated with inflammation. The present study may aid the understanding of the underlying mechanisms of action of licorice flavonoids in the suppression of inflammation, providing a theoretical foundation for the development of licorice flavonoids to be applied clinically.
Materials and methods
Reagents and materials
Aspirin enteric-coated tablets were purchased from Bayer (Shanghai, China). Formaldehyde was purchased from Longhai Dandong Reagent Factory (Dandong, China). Potassium hydroxide was purchased from Tianjin Kaixin Chemical Industry Co., Ltd. (Tianjin, China). High-performance liquid chromatography (HPLC)-grade acetonitrile was purchased from J. T. Baker (Thermo Fisher Scientific, Inc., Waltham, MA, USA). HPLC-grade formic acid was purchased from Fluka (Merck & Co., Inc., Whitehouse Station, NJ, USA). Ultrapure water (18.2 MSZ) was prepared with a Milli-Q water purification system (Merck KGaA, Darmstadt, Germany).
Licorice was collected in Tongliao, the Inner Mongolia Autonomous Region in China at locations between 42°15′N-45°41′N and 119°15′E-123°43′E. In the Chinese Pharmacopoeia (2015 edition) the liquiritigenin and glycyrrhizic acid content is required to be >0.50 and >2.0%, respectively (22). The licorice used in this experiment contained 1.84 and 2.57% of liquiritigenin and glycyrrhizic acid, respectively, in accordance with the relevant provision of Chinese Pharmacopoeia. Licorice flavonoids were prepared as previously described (23). The average paste rate was 5.40%; the purity of total flavonoids reached up to 90%, as detected by HPLC method (24), primarily contained 1.1695% liquiritin apioside, 1.8402% liquiritin, 0.2087% isoliquiritin apioside, 0.2042% ononin, 0.4023% isoliquiritin, 0.6443% liquiritigenin, 0.1548% calycosin and 0.0726% isoliquiritigenin.
Animals
A total of 54 male Kunming mice weighing 18–22 g were provided by Liaoning Biological Technology Co., Ltd. (Liaoning, China). The mice were bred in a specific pathogen-free environment and had free access to a standard diet and tap water. The animals were housed in stainless steel metabolic cages under standard humidity (50±10%), temperature (25±2°C) and light (12/12 h light/dark cycle). All animal treatments were conducted in strict accordance with the National Institutes of Health Guide to the Care and Use of Liaoning University of Traditional Chinese Medicine, and the present study was approved by Attitude of the Animal Core and Welfare Committee of Liaoning University of Traditional Chinese Medicine.
Animal handling
Mice were divided randomly into 6 groups (n=9). These groups were: The control group (mice injected with 0.05 ml saline into the sub-plantar region of the right hind paw), the model group (mice injected with 0.05 ml 2.5% formaldehyde solution into the sub-plantar region of the right hind paw), the licorice flavonoid high-dose group (treated with 0.65 g/kg of licorice flavonoids), the middle-dose group (0.21 g/kg of licorice flavonoids), the low-dose group (0.07 g/kg of licorice flavonoids) and the aspirin group (0.04 g/kg of aspirin). All mice were orally administered the active group solution twice daily (control and model groups were administered saline) for 3 days. The mice were prohibited any food for 24 h before the final day of the experiment, but were allowed ad libitum access to water. At 1 h after the last administration, the paw volume of mice in each group prior to inducing inflammation was measured with a self-made foot volume-measuring device. Subsequently, mice were injected with 0.05 ml of 2.5% formaldehyde solution into the sub-plantar region of the right hind paw except control group (25). Following inducing inflammation 24 h, the paw volume was measured with a self-made foot volume-measuring device again; the ratio of paw edema (%) and the inhibitory rate of paw edema (%) were taken as indexes of detection. Mice were then anesthetized and sacrificed, blood was collected into Na-heparin tubes and plasma was obtained following centrifugation (1,006.2 × g, 4°C for 15 min), and stored at −80°C until metabolomics analysis were performed. The inflammatory paws of the mice from their right ankle were removed, weighed, and soaked for 1 h with 3 ml normal saline (precooled to <4°C). Following centrifugation (1,006.2 × g, 4°C for 15 min) of the solution, 2 ml supernatant was absorbed and mixed with 2 ml of 0.15 mol l−1 KOH-CH4O solution, samples were incubated in water bath for 20 min at 50°C, then the content of prostaglandin E2 (PGE2) was detected with a UV spectrophotometer at a wavelength of 278 nm once the liquid had cooled.
Sample preparation
The blood samples were thawed prior to analysis and 100-µl aliquots of bloods were added to 400 µl methanol. The mixture was vortexed for 3 min and centrifuged at 1,006.2 × g for 15 min at 4°C. The supernatant was then transferred to auto-sampler vials. A pooled quality control (QC) sample was made by mixing 20-µl aliquots of each sample, using the same method as that used for each of the other samples. The pooled QC sample was analyzed randomly through the analytical run to monitor instrument stability. In addition, a random sample was divided into 6 and was treated in the same way. These 6 samples were continuously analyzed to validate the repeatability of the sample preparation method.
HPLC-quadrupole-time of flight (Q-TOF)-mass spectrometry (MS) conditions
Metabolic profiling of the blood serum was conducted using an Agilent-1260 LC system coupled with an Agilent-6530 Q-TOF mass spectrometer (Agilent Technologies, Inc., Santa Clara, CA, USA). Plasma chromatographic separation was performed on an Agilent Poroshell 120 SB-C18 column (2.7 µm, 4.6×100 mm; Agilent Technologies, Inc.) with the temperature of the column set at 45°C. The flow rate was 1.0 ml/min and the mobile phase was ultrapure water with 1‰ formic acid and 1‰ acetonitrile The gradient elution of acetonitrile was performed as follows: 5–30% acetonitrile at 0–4 min; 30–67% acetonitrile at 4–6 min; 67–98% acetonitrile at 6–11 min; 98–100% acetonitrile at 11–17 min; followed by a 7-min re-equilibration step. The volume of sample injected was 1 µl. The parameters of mass detection were set as followed: In positive mode, the flow rate of the dry gas of MS mode was 9 l/min, Vcap 4000 V; the nebulizer pressure was 45 psig; the fragmentation voltage was 175 V; the Sheath Gas Temp was 350°C; the Sheath gas velocity was 12 l/min; the acquisition rate was 25 Spectra/sec; MS data were acquired in full-scan mode from m/z 50 to 1,050 amu; and the collision energy of MS/MS date acquisition was set at 20 eV. The corrected mixed solution (Agilent Technologies, Inc.; m/z=121.050873, 149.02332, 922.009798) was used as the lock mass.
Data processing
The raw MS data were exported by Agilent Mass Hunter Qualitative Analysis software (vB.04.00; Agilent Technologies, Inc.) for peak detection, alignment and filtering (Agilent Technologies, Inc.). Integrated information of the retention time and molecular mass were then outputted from the Agilent Mass Profiler software (vB.02.00; Agilent Technologies, Inc.). Finally, principal components analysis (PCA), the cluster analysis used for multivariate analysis and one-way analysis of variance (ANOVA) used for expressing the significance of data were performed with Mass Profiler Professional (MPP) 12.6 software (Agilent Technologies, Inc.), as described previously (26). The metabolites were preliminarily identified in METLIN (https://metlin.scripps.edu/landing_page.php?pgcontent=mainPage) and PUBCHEM (https://www.ncbi.nlm.nih.gov/pccompound/) databases, and biochemical reactions involving the identified metabolites were obtained through KEGG (http://www.kegg.jp/), HMDB (http://www.hmdb.ca/) and LIPID MAPS (http://www.lipidmaps.org/) databases.
Statistical analysis
SPSS 19.0 statistical software (IBM SPSS, Armonk, NY, USA) was used for statistical analysis, and one-way ANOVA was used to design the group data followed by Student-Newman-Keuls test. Student's t-test was used to analyze the different metabolites between the groups with MPP software. All quantitative data are expressed as the mean ± standard deviation, as indicated. P<0.01 was considered to indicate a statistically significant difference.
Results
Effects of licorice flavonoids on formalin-induced inflammation in mice
The inhibitory effects of different doses of groups on formalin-induced paw edema in mice are shown in Figs. 1 and 2. The degree of paw edema in the model group was substantially higher compared with that in control group (no inflammation), indicating that the acute inflammation model was successfully established (data not shown). Compared with the model group, the ratio of paw edema [(the volume of paw after inflammation-the volume of paw before inflammation)/the volume of paw after inflammation)] in each administered group was significantly lower (P<0.01), among which aspirin group, the inhibition rate of the high- and middle-dose licorice flavonoid groups reached >50% (Fig. 2), indicating that licorice flavonoids inhibited the increase of inflammatory mediators caused by formalin stimuli. In addition, the content of PGE2 in the model group significantly increased when compared with the control group (Fig. 3), which indicated that the inflammation in the paws of the mice caused by formalin is able to raise the PGE2 level in vivo. The content of PGE2 in the licorice flavonoid dose groups was significantly decreased compare to that of model group (P<0.01), suggesting that licorice flavonoid can inhibit inflammation though regulating the level of PGE2.
Figure 1.

Effect of licorice flavonoids on formalin-induced mice paw edema. Data are expressed as the mean ± standard deviation (n=9). The ratio of paw edema (%) = [(volume of edema (injected)-volume of edema (normal)]/volume of edema (normal) ×100. *P<0.01 compared with the model group. LFL, low dosage group of licorice flavonoids; LFM, middle dosage group of licorice flavonoids; LFH, high dosage group of licorice flavonoids.
Figure 2.

Inhibitory rate of paw edema in different dose groups. Data are expressed as means (n=9). Inhibitory rate (%) = [(Volume of edema (model)-Volume of edema (test)]/Volume of edema (model) ×100. *P<0.01 vs. model group. LFL, low dosage group of licorice flavonoids; LFM, middle dosage group of licorice flavonoids; LFH, high dosage group of licorice flavonoids.
Figure 3.
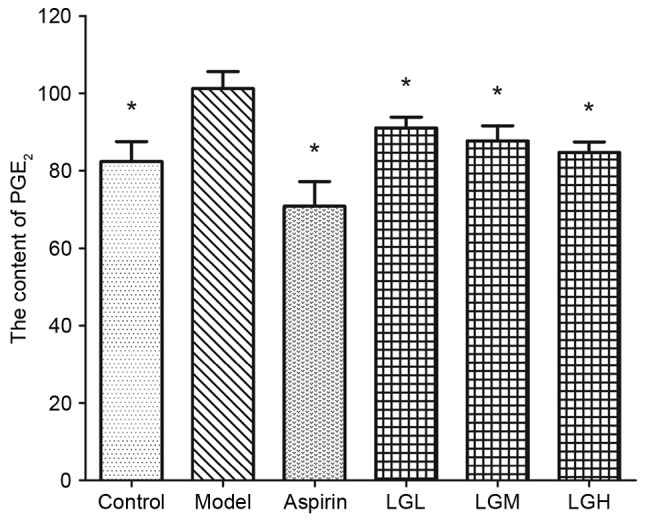
Content of PGE2 in different groups on formalin-induced mice paw edema. Data are expressed as the mean ± standard deviation (n=9). *P<0.01 vs. model group. PGE2, prostaglandin E2; LFL, low dosage group of licorice flavonoids; LFM, middle dosage group of licorice flavonoids; LFH, high dosage group of licorice flavonoids.
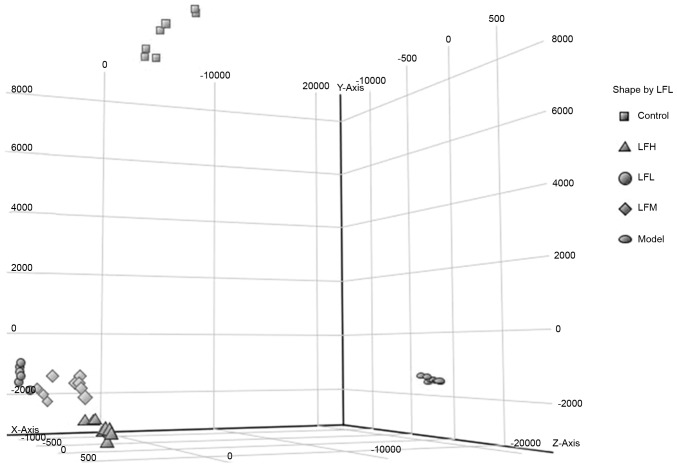
PCA and cluster analysis of metabolites
PCA and hierarchical clustering analysis were used to classify the metabolic phenotypes and identify the different metabolites. On the observation of 3-dimensional plots, samples in the same group were evidently clustered and the model group differed significantly from others (Fig. 4), indicating that the endogenous metabolites in the formalin-induced paw edema in the model group differed significantly from the drug-treated groups. The spatial position of the licorice flavonoid groups was close to that of the control group, demonstrating that the plasma metabolite composition of inflammation mice has a tendency to return to normal following treatment. Clustering analysis of metabolomics data also revealed distinct segregation between the control, model and licorice flavonoid dose groups (Fig. 5).
Figure 4.
Principal component analysis of each group. Each colored point represents a sample. The first, second and third principal components are displayed on the x-, y- and z-axis, respectively. These three components represent the largest fraction of the overall variability. LFL, low dosage group of licorice flavonoids; LFM, middle dosage group of licorice flavonoids; LFH, high dosage group of licorice flavonoids.
Figure 5.

Clustering analysis of the endogenous metabolites in inflammation. The difference in the gray level represents the difference of content. Light gray represents the normal level, dark gray and black represents the content of compound increased or decreased. LFL, low dosage group of licorice flavonoids; LFM, middle dosage group of licorice flavonoids; LFH, high dosage group of licorice flavonoids.
Identification of endogenous metabolites with differing levels
Small molecule metabolites whose levels appeared to differ significantly (P<0.05) between licorice flavonoids, model and control groups were analyzed using MPP software. According to the MS/MS fragments of differing endogenous metabolites, compared with METLIN, PUBCHEM and other associated databases, 9 metabolites associated with inflammation were preliminary identified (Table I).
Table I.
Identified endogenous metabolites with difference and metabolic pathway between groups.
| Identity | RT, min | Molecular mass, Da | Fragment ions | Molecular formula | Regulation (vs. model group) | Metabolic pathway |
|---|---|---|---|---|---|---|
| Tryptophanamide | 1.44 | 203.1059 | 187, 159 | C11H13N3O | Down | Tryptophan metabolism |
| Acetylcarnitine | 1.70 | 203.1172 | 85, 60 | C9H17NO4 | Down | Fatty acid metabolism |
| Linoleic acid | 6.17 | 280.2406 | 69, 83, 97 | C18H32O2 | Up | Linoleic acid metabolism |
| PE [19:0/22:6 (4Z, 7Z, 10Z, 13Z, 16Z, 19Z)] | 6.88 | 805.5621 | 184 | C46H80NO8P | Down | Phospholipid synthesis |
| N-linoleoyl taurine | 7.29 | 387.2443 | 117, 207, 149 | C20H37NO4S | Up | Taurine and hypotaurine metabolism |
| LysoPE [0:0/20:3 (8Z, 11Z, 14Z] | 7.68 | 525.2855 | 89, 184, 327, 526 | C27H44NO7P | Up | Phospholipid synthesis |
| Leukotriene B4 | 8.06 | 336.4656 | 189, 119, 105 | C20H32O4 | Down | AA metabolism |
| Corticosterone | 10.22 | 346.2152 | 347, 97, 109 | C21H30O4 | Up | Steroid hormone biosynthesis |
| Sphingosine | 13.92 | 299.2824 | 282, 252 | C18H37NO2 | Down | Sphingolipid metabolism |
AA, arachidonic acid; LysoPE, Lysophosphatidylethanolamine; PE, phosphatidylethanolamines; RT, retention time.
Pathway analysis
The pathways associated with endogenous metabolites and anti-inflammatory effects of licorice flavonoids are listed in Table I and Fig. 6. These metabolites were identified to be involved in pathways, including in arachidonic acid (AA) metabolism, tryptophan metabolism, sphingolipid metabolism, according to HMDB, KEGG, LIPID MAPS and other backend knowledge databases.
Figure 6.
The metabolite pathways involved in licorice flavonoids intervention on inflammation. The association between metabolites in groups is depicted in Table 1. The metabolites presented in upward facing trapeziums indicate an increase in levels, whilst those in downward facing trapeziums indicate a decrease in levels following treatment with licorice flavonoids when compared with the model group.
Discussion
AA is a polyunsaturated fatty acid that is present in mammalian systems. Cyclooxygenase (COX) and lipoxygenase (LOX) pathways are the two major enzymatic pathways involved in AA metabolism, and are associated with inflammation (27). In the present study, licorice flavonoids regulated a variety of endogenous metabolites associated with AA metabolism to mediate its anti-inflammatory effect. In metabolic networks, lipid metabolism and inflammation are associated (28,29) and there is a mutual transformation association between linoleic acid and AA (30): As the level of AA decreases, linoleic acid levels increase. The results of the present study indicated that the content of linoleic acid in the plasma of the mice administered licorice flavonoids was higher compared with that in the model group, which may influence the AA metabolism level and alleviate inflammation (Table I; Fig. 6). In a process catalyzed by sphingosine kinases 1 and 2, sphingosine-1-phosphate (S1P) is formed by the phosphorylation of sphingosine (31), and is recognized as an important intracellular and extracellular messenger, participating in inflammatory signaling pathways (32–34). Nuclear factor-κ-light-chain-enhancer of activated B cells (NF-κB) are reported be responsible for the transcriptional activation of a number of inflammatory mediators (35–37), which regulate the expression of a variety of different genes and enzymes, including prostaglandin-endoperoxide synthase 2 (COX-2), interleukin (IL)-1, IL-6, IL-8, chemokines, and tumor necrosis factor (TNF) (38–40). Following the oral administration of licorice flavonoids, the content of sphingosine decreased in the serum of mice, then downregulated S1P, which transcriptionally regulates the transient expression of COX-2 and PGE2 (41,42), from AA metabolism and NF-κB metabolism pathway to reduce the production of inflammatory factors.
The monoamine 5-hydroxytryptophan (5-HT, also known as serotonin), serves an important role in evaluating the inflammatory condition of the body (43). When cells are stimulated by environment triggers, 5-HT is released from cell particles into the bloodstream, resulting in an increase in vascular permeability, which leads to inflammation. Tryptophanamide is a substrate of tryptophan aminopeptidase and is converted to L-tryptophan by enzymatic hydrolysis. In the presence of tryptophan hydroxylase, L-tryptophan generates 5-HT (44). In the present study, licorice flavonoids also downregulated the content of tryptophanamide, resulting in a decrease in the content of 5-HT and thus the indirect downregulation of PGE2 (45). The lower concentration of PGE2 in the inflamed mouse tissues verified the accuracy of the results. Additionally, the level of an important inflammatory mediator, leukotriene B4, in the mice administered licorice flavonoids was decreased, which demonstrated its anti-inflammatory action from the LOXs pathway.
Taurine has been associated with potent anti-inflammatory effects in a variety of models of systemic inflammation (46). In the body, taurine is chlorinated to form taurine chloramine, which inhibits the generation of macrophage inflammatory mediators, including NO, TNF, and PGE2 (47). The present study demonstrated that licorice flavonoids upregulate the level of N-linoleoyl taurine, a derivative of taurine, which may serve an indirect anti-inflammatory role (Table I; Fig. 6).
Corticosterone is the metabolite of cortisol, which is a corticosteroid hormone produced by the adrenal cortex. The anti-inflammatory effects of corticosterone have been reported previously (48,49). In the present study, the content of corticosterone in the mice administered with licorice flavonoids was higher compared with that in the model group, indicating that flavonoids may alleviate inflammation through the steroid hormone biosynthesis pathway.
L-acetylcarnitine is an acetic acid ester of carnitine that facilitates the movement of acetyl-coenzyme A into the matrices of the mammalian mitochondria during the oxidation of fatty acids. It promotes the production of pro-inflammatory cytokines, including TNF, IL-6 and IL-1β (50). In the serum of licorice flavonoids groups, the level of L-acetylcarnitine was decreased, indicating that flavonoids may influence fatty acid metabolism to serve anti-inflammatory roles.
Phospholipids are components of cell membranes; their metabolites participate in phospholipid maintenance of normal physiological function. Abnormalities in phospholipid metabolism may promote systemic inflammation (51). Lysophosphatidylethanolamine (LysoPE) and phosphatidylethanolamines (PE) are important phospholipid synthetic pathway intermediates; licorice flavonoids may influence the phospholipid synthetic pathway to alleviate inflammation.
Licorice flavonoids possess the same mechanism of regulating AA metabolism and the NF-κB metabolism pathway to relieve inflammation as aspirin. Additionally, they may also influence phospholipid synthesis and steroid hormone biosynthesis to promote anti-inflammatory effects, revealing a multiple pathway regulation function of licorices flavonoids (52,53).
In the present study, on the basis of prominent anti-inflammatory effect, the mechanism of action of licorice flavonoids was assessed via metabolomic analysis, using HPLC-Q-TOF/MS. The results of pharmacodynamics analysis demonstrated that licorice flavonoids inhibited formaldehyde-induced mice paw edema and relieved inflammation, presenting a remarkable dose-effect association. Furthermore, it has been speculated that licorice flavonoids is able to regulate the levels of endogenous metabolites in mice, including linoleic acid, sphingosine, tryptophanamide, N-linoleoyl taurine, corticosterone, L-acetylcarnitine, leukotriene B4, PE[19:0/22:6(4Z,7Z,10Z,13Z,16Z,19Z)] and LysoPE[0:0/20:3(8Z,11Z,14Z)], involving the metabolism of AAs, sphingolipid, tryptophans, fatty acids, taurine, hypotaurine and linoleic acid, and the synthesis of phospholipids and steroid hormones. Understanding of the multi-pathway integrated adjustment mechanism of licorice flavonoids may aid in the reduction of side effects along with their anti-inflammatory functions, and provides a foundation for identifying and developing novel, high-potential natural drugs with fewer side effects for clinical use.
Acknowledgements
The authors wish to thank Dr Nannan Li, Mr Hechen Wang, Mrs Lulu Tian and Mrs Yanan Zhang (Liaoning University of Traditional Chinese Medicine, Dalian, China) from our research team for their work on the present study.
Funding
The present study was supported by grants from the National Natural Science Foundation of China (grant no. 81241111), the Project of Institutions to Support Higher Learning Talents in Liaoning Province (grant no. LR2013044) and Eleventh Five-Year Plan, Significant New Medicines Creation, Science and Technology Major Special Project (grant no. 2010ZX09401-304-201).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
XY, XM and GY conceived and designed the experiments. XY, XC, WX and YB performed the experiments. TL, SW and TB analyzed the data. XY wrote the paper.
Ethics approval and consent to participate
Ethics approval was granted by the Attitude of the Animal Core and Welfare Committee of Liaoning University of Traditional Chinese Medicine (reference no. 20150702).
Patient consent for publication
Not applicable.
Competing interests
The authors confirm that no conflicts of interest exist.
References
- 1.Kumar V, Abbas AK, Fausto N. 7th. Elsevier Saunders; Philadelphia: 2004. Robbins and Cotran Pathologic Basis of Disease. [Google Scholar]
- 2.Henson PM, Murphey RC. Elsevier; Amsterdam: 1989. Mediators of the Inflammatory Process (Handbook Of Inflammation) [Google Scholar]
- 3.Vane JR, Botting RM. New insights into the mode of action of anti-inflammatory drugs. Inflamm Res. 1995;44:1–10. doi: 10.1007/BF01630479. [DOI] [PubMed] [Google Scholar]
- 4.Lane MA, Mcdonald JR, Zeringue AL, Caplan L, Curtis JR, Ranganathan P, Eisen SA. TNF-α antagonist use and risk of hospitalization for infection in a national cohort of veterans with rheumatoid arthritis. Medicine (Baltimore) 2011;90:139–145. doi: 10.1097/MD.0b013e318211106a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mcinnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Eng J Med. 2011;365:2205–2219. doi: 10.1056/NEJMra1004965. [DOI] [PubMed] [Google Scholar]
- 6.Saely CH, Drexel H, Sourij H, Aczel S, Jahnel H, Zweiker R, Langer P, Marte T, Hoefle G, Benzer W, Wascher TC. Key role of postchallenge hyperglycemia for the presence and extent of coronary atherosclerosis: An angiographic study. Atherosclerosis. 2008;2:317–322. doi: 10.1016/j.atherosclerosis.2007.11.020. [DOI] [PubMed] [Google Scholar]
- 7.Devaraj S, Rogers J, Jialal I. Statins and biomarkers of inflammation. Curr Atheroscler Rep. 2007;9:33–41. doi: 10.1007/BF02693938. [DOI] [PubMed] [Google Scholar]
- 8.Mullarkey C. Soothing a sore throat: The efficacy and safety of steroids in acute pharyngitis. Ir J Med Sci. 2011;180:837–840. doi: 10.1007/s11845-011-0719-z. [DOI] [PubMed] [Google Scholar]
- 9.Gaddi A, Cicero AF, Pedro EJ. Clinical perspectives of anti-inflammatory therapy in the elderly: The lipoxigenase (LOX)/cycloxigenase (COX) inhibition concept. Arch Gerontol Geriatr. 2004;38:201–202. doi: 10.1016/j.archger.2003.10.001. [DOI] [PubMed] [Google Scholar]
- 10.Tao J, Hou Y, Ma X, Liu D, Tong Y, Zhou H, Gao J, Bai G. An integrated global chemomics and system biology approach to analyze the mechanisms of the traditional Chinese medicinal preparation Eriobotrya japonica-Fritillaria usuriensis dropping pills for pulmonary diseases. BMC Complement Altern Med. 2016;16:4. doi: 10.1186/s12906-015-0983-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ni LJ, Wang NN, Zhang LG, Guo YZ, Shi WZ. Evaluation of the effects of active fractions of Chinese medicine formulas on IL-1β, IL-6, and TNF-α release from ANA-1 Murine Macrophages. J Ethnopharmacol. 2015;179:420–431. doi: 10.1016/j.jep.2015.12.052. [DOI] [PubMed] [Google Scholar]
- 12.Lin B, Sun LN, Xin HL, Nian H, Song HT, Jiang YP, Wei ZQ, Qin LP, Han T. Anti-inflammatory constituents from the root of Litsea cubeba in LPS-induced RAW 264.7 macrophages. Pharm Biol. 2016;5:1741–1747. doi: 10.3109/13880209.2015.1126619. [DOI] [PubMed] [Google Scholar]
- 13.Beijing: China Medical Science Press; 2015. The State Pharmacopoeia Committee of China. The Pharmacopoeia of the People's Republic of China. Part1; pp. 80–81. [Google Scholar]
- 14.Flora reipublicae popularis sinicae, corp-author. First. Science Press; Beijing: 2004. (In Chinese) [Google Scholar]
- 15.Fiore C, Eisenhut M, Ragazzi E, Zanchin G, Armanini D. A history of the therapeutic use of liquorice in Europe. J Ethnopharmacol. 2005;99:317–324. doi: 10.1016/j.jep.2005.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shin YW, Bae EA, Lee B, Lee SH, Kim JA, Kim YS, Kim DH. In vitro and in vivo antiallergic effects of Glycyrrhiza glabra and its components. Planta Med. 2007;73:257–261. doi: 10.1055/s-2007-967126. [DOI] [PubMed] [Google Scholar]
- 17.Yokota T, Nishio H, Kubota Y, Mizoguchi M. The inhibitory effect of glabridin from licorice extracts on melanogenesis and inflammation. Pigment Cell Res. 1998;11:355–361. doi: 10.1111/j.1600-0749.1998.tb00494.x. [DOI] [PubMed] [Google Scholar]
- 18.Li J, Tu Y, Tong L, Zhang W, Zheng J, Wei Q. Immunosuppressive activity on the murine immune responses of glycyrol from Glycyrrhiza uralensis via inhibition of calcineurin activity. Pharm Biol. 2010;48:1177–1184. doi: 10.3109/13880200903573169. [DOI] [PubMed] [Google Scholar]
- 19.Armanini D, Fiore C, Mattarello M, Bielenberg J, Palermo M. History of the endocrine effects of licorice. Exp Clin Endocrinol Diabetes. 2002;110:257–261. doi: 10.1055/s-2002-34587. [DOI] [PubMed] [Google Scholar]
- 20.Zhao Z, Wang W, Guo H, Zhou D. Antidepressant-like effect of liquiritin from Glycyrrhiza uralensis in chronic variable stress induced depression model rats. Behav Brain Res. 2008;194:108–113. doi: 10.1016/j.bbr.2008.06.030. [DOI] [PubMed] [Google Scholar]
- 21.Yamamoto S, Aizu E, Jiang H, Nakadate T, Kiyoto I, Wang JC, Ryuichi K. The potent anti-tumor-promoting agent isoliquiritigenin. Carcinogenesis. 1991;12:317–323. doi: 10.1093/carcin/12.2.317. [DOI] [PubMed] [Google Scholar]
- 22.Pharmacopoeia Commission of PRC, corp-author. 1st. Chemical Industry Press; Beijing: 2015. Pharmacopoeia of the People's Republic of China. [Google Scholar]
- 23.Jiang HH, Meng XS, Kang TG, Bao YR. Study on extraction and purification of Licorice Flavonoids components. Chin Med Herald. 2010;7:54–56. (In Chinese) [Google Scholar]
- 24.Wu YP, Meng XS, Bao YR, Wang S, Kang TG. Simultaneous quantitative determination of nine active chemical compositions in traditional Chinese medicine Glycyrrhiza by RP-HPLC with full-time five-wavelength fusion method. Am J Chin Med. 2013;41:211–219. doi: 10.1142/S0192415X13500158. [DOI] [PubMed] [Google Scholar]
- 25.Chen Q. Research methodology of pharmacology of Chinese materia medica. 1st. People's Medical Publishing House; Beijing: 1993. [Google Scholar]
- 26.Tianjiao L, Shuai W, Xiansheng M, Yongrui B, Shanshan G, Bo L, Lu C, Lei W, Xiaorong R. Metabolomics coupled with multivariate data and pathway analysis on potential biomarkers in gastric ulcer and intervention effects of Corydalis Yanhusuo alkaloid. PLoS One. 2014;9:e82499. doi: 10.1371/journal.pone.0082499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Agarwal S, Reddy GV, Reddanna P. Eicosanoids in inflammation and cancer: The role of COX-2. Expert Rev Clin Immunol. 2009;5:45–65. doi: 10.1586/1744666X.5.2.145. [DOI] [PubMed] [Google Scholar]
- 28.Kubala L, Schmelzer KR, Klinke A, Kolarova H, Baldus S, Hammock BD, Eiserich JP. Modulation of arachidonic and linoleic acid metabolites in myeloperoxidase-deficient mice during acute inflammation. Free Radic Biol Med. 2010;48:1311–1320. doi: 10.1016/j.freeradbiomed.2010.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mathers L, Bailey MJ. Enzyme deletions and essential fatty acid metabolism in cultured cells. J Biol Chem. 1975;250:1152–1153. [PubMed] [Google Scholar]
- 30.Stratz C, Anakwue J, Bhatia H, Pitz S, Fiebich BL. Anti-inflammatory effects of 5-HT3 receptor antagonists in interleukin-1beta stimulated primary human chondrocytes. Int Immunopharmacol. 2014;22:160–166. doi: 10.1016/j.intimp.2014.06.003. [DOI] [PubMed] [Google Scholar]
- 31.Pyne S, Pyne NJ. Translational aspects of sphingosine-1-phosphate biology. Trends Mol Med. 2011;17:463–472. doi: 10.1016/j.molmed.2011.03.002. [DOI] [PubMed] [Google Scholar]
- 32.Cuvillier O, Pirianov G, Kleuser B, Vanek PG, Coso OA, Gutkind S. Suppression of ceramide-mediated programmed cell death by sphingosine-1-phosphate. Nature. 1996;381:800–803. doi: 10.1038/381800a0. [DOI] [PubMed] [Google Scholar]
- 33.Olivera A, Spiegel S. Sphingosine-1-phosphate as second messenger in cell proliferation induced by PDGF and FCS mitogens. Nature. 1993;365:557–560. doi: 10.1038/365557a0. [DOI] [PubMed] [Google Scholar]
- 34.Prager B, Spampinato SF, Ransohoff RM. Sphingosine-1-phosphate signaling at the blood-brain barrier. Trends Mol Med. 2015;21:354–363. doi: 10.1016/j.molmed.2015.03.006. [DOI] [PubMed] [Google Scholar]
- 35.Chung HY, Kim HJ, Kim JW, Yu BP. The inflammation hypothesis of aging: Molecular modulation by calorie restriction. Ann N Y Acad Sci. 2001;928:327–335. doi: 10.1111/j.1749-6632.2001.tb05662.x. [DOI] [PubMed] [Google Scholar]
- 36.Shea LM, Beehler C, Schwartz M, Shenkar R, Tuder R, Abraham E. Hyperoxia activates NF-kappaB and increases TNF-alpha and IFN-gamma gene expression in mouse pulmonary lymphocytes. J Immunol. 1996;157:3902–3908. [PubMed] [Google Scholar]
- 37.Tak PP, Firestein GS. NF-kappaB: A key role in inflammatory diseases. J Clin Invest. 2001;107:7–11. doi: 10.1172/JCI11830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sarkar D, Fisher PB. Molecular mechanisms of aging-associated inflammation. Cancer Lett. 2006;236:13–23. doi: 10.1016/j.canlet.2005.04.009. [DOI] [PubMed] [Google Scholar]
- 39.Helenius M, Hänninen M, Lehtinen SK, Salminen A. Aging-induced up-regulation of nuclear binding activities of oxidative stress responsive NF-κB transcription factor in mouse cardiac muscle. J Mol Cell Cardiol. 1996;28:487–498. doi: 10.1006/jmcc.1996.0045. [DOI] [PubMed] [Google Scholar]
- 40.Helenius M, Hanninen M, Lehtinen SK, Salminen A. Changes associated with aging and replicative senescence in the regulation of transcription factor nuclear factor-kappa B. Biochem J. 1996;318:603–608. doi: 10.1042/bj3180603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Cheng SE, Lee IT, Lin CC, Wu WL, Hsiao LD, Yang CM. ATP mediates NADPH oxidase/ROS generation and COX-2/PGE2 expression in A549 cells: Role of P2 receptor-dependent STAT3 activation. PLoS One. 2013;8:e54125. doi: 10.1371/journal.pone.0054125. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 42.Nodai A, Machida T, Izumi S, Hamaya Y, Kohno T, Igarashi Y, Iizuka K, Minami M, Hirafuji M. Sphingosine-1-phosphate induces cyclooxygenase-2 via Ca2+-dependent, but MAPK-independent mechanism in rat vascular smooth muscle cells. Life Sci. 2007;80:1768–1776. doi: 10.1016/j.lfs.2007.02.008. [DOI] [PubMed] [Google Scholar]
- 43.Lim SS, Shin KH, Ban HS, Kim YP, Jung SH, Kim YJ, Ohuchi K. Effect of the essential oil from the flowers of Magnolia sieboldii on the lipopolysaccharide-induced production of nitric oxide and prostaglandin E2 by rat peritonealmacrophages. Planta Med. 2002;68:459–462. doi: 10.1055/s-2002-32085. [DOI] [PubMed] [Google Scholar]
- 44.Walther DJ, Bader M. A unique central tryptophan hydroxylase isoform. Biochem Pharmacol. 2003;66:1673–1680. doi: 10.1016/S0006-2952(03)00556-2. [DOI] [PubMed] [Google Scholar]
- 45.Yao W, Zhang L, Hua Y, Ji P, Li P, Li J, Zhong L, Zhao H, Wei Y. The investigation of anti-inflammatory activity of volatile oil of Angelica sinensis by plasma metabolomics approach. Int Immunopharmacol. 2015;29:269–277. doi: 10.1016/j.intimp.2015.11.006. [DOI] [PubMed] [Google Scholar]
- 46.de Rienzo-Madero B, Coffeen U, Simón-Arceo K, Mercado F, Jaimes O, Magis-Weinberg L, Contreras B, Pellicer F. Taurine enhances antinociception produced by a COX-2 inhibitor in an inflammatory pain model. Inflammation. 2013;36:658–664. doi: 10.1007/s10753-012-9589-4. [DOI] [PubMed] [Google Scholar]
- 47.Marcinkiewicz J, Grabowska A, Bereta J, Bryniarski K, Nowak B. Taurine chloramine down-regulates the generation of murine neutrophil inflammatory mediators. Immunopharmacology. 1998;40:27–38. doi: 10.1016/S0162-3109(98)00023-X. [DOI] [PubMed] [Google Scholar]
- 48.del Rey A, Wolff C, Wildmann J, Randolf A, Hahnel A, Besedovsky HO, Straub RH. Disrupted joint-immune-brain communication during experimental arthritis. Arthritis Rheum. 2008;58:3090–3099. doi: 10.1002/art.23869. [DOI] [PubMed] [Google Scholar]
- 49.Li A, Zhang RX, Wang Y, Zhang H, Ren K, Berman BM, Tan M, Lao L. Corticosterone mediates electroacupuncture-produced anti-edema in a rat model of inflammation. BMC Complement Altern Med. 2007;7:27. doi: 10.1186/1472-6882-7-27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Altun Z, Olgun Y, Ercetin P, Aktas S, Kirkim G, Serbetcioglu B, Olgun N, Guneri EA. Protective effect of acetyl-l-carnitine against cisplatin ototoxicity: Role of apoptosis-related genes and pro-inflammatory cytokines. Cell Prolif. 2014;47:72–80. doi: 10.1111/cpr.12080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ding X, Hu J, Li J, Zhang Y, Shui B, Ding Z, Yao L, Fan Y. Metabolomics analysis of collagen-induced arthritis in rats and interventional effects of oral tolerance. Anal Biochem. 2014;458:49–57. doi: 10.1016/j.ab.2014.04.035. [DOI] [PubMed] [Google Scholar]
- 52.Basselin M, Ramadan E, Chen M, Rapoport SI. Anti-inflammatory effects of chronic aspirin on brain arachidonic acid metabolites. Neurochem Res. 2011;36:139–145. doi: 10.1007/s11064-010-0282-4. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 53.Liu Y, Fang S, Li X, Feng J, Du J, Guo L, Su Y, Zhou J, Ding G, Bai Y, et al. Aspirin inhibits LPS-induced macrophage activation via the NF-κB pathway. Sci Rep. 2017;7:11549. doi: 10.1038/s41598-017-10720-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.