Abstract
In neurons, a limited number of mRNAs have been identified in dendritic processes, whereas other transcripts are restricted to the cell soma. Here we have investigated the molecular mechanisms underlying extrasomatic localization of mRNAs encoding microtubule-associated protein 2 (MAP2) in primary neuronal cultures. Vectors expressing recombinant mRNAs were introduced into hippocampal and sympathetic neurons using DNA transfection and microinjection protocols, respectively. Chimeric mRNAs containing the entire 3′ untranslated region of MAP2 transcripts fused to a nondendritic reporter mRNA are detected in dendrites. In contrast, RNAs containing MAP2 coding and 5′ untranslated regions or tubulin sequences are restricted to the cell soma. Moreover, 640 nucleotides from the MAP2 3′ untranslated region (UTR) are both sufficient and essential for extrasomatic localization of chimeric mRNAs in hippocampal and sympathetic neurons. Thus, a cis-acting dendritic targeting element that is effective in two distinct neuronal cell types is contained in the 3′ UTR of MAP2 transcripts. The observation of RNA granules in dendrites implies that extrasomatic transcripts seem to assemble into multimolecular complexes that may function as transport units.
Keywords: dendritic targeting element, RNA localization into dendrites, subcellular transport, extrasomatic protein synthesis, neuronal cytoskeleton/microtubule-associated protein, primary cultures of neurons, microinjection, transfection
Neurons possess distinct cellular compartments that are highly diverse with respect to their protein repertoire. Most likely a differential molecular composition of dendritic microdomains and postsynaptic structures is not only attributable to selective protein sorting but also reflects local synthesis of specific proteins (Steward, 1994, 1997; Kindler et al., 1997; Kuhl and Skehel, 1998; Tiedge et al., 1999). This hypothesis first arose with the ultrastructural detection of polyribosomes in dendritic shafts at the base of dendritic spines (Steward and Levy, 1982; Steward and Reeves, 1988) and was subsequently supported by the identification of distinct mRNAs in dendrites. Dendritic mRNAs encode, among other proteins, the microtubule-associated protein 2 (MAP2) (Garner et al., 1988; Bruckenstein et al., 1990; Kleiman et al., 1990), the α subunit of the Ca2+/calmodulin-dependent protein kinase II (α-CaMKII) (Burgin et al., 1990), the product of an activity-regulated gene (arg3.1) (Link et al., 1995), also described as activity-regulated cytoskeleton-associated protein, arc (Lyford et al., 1995), the neuropeptides vasopressin and oxytocin (Mohr et al., 1995), the inositol 1,4,5-triphosphate receptor type 1 (Furuichi et al., 1993), neurogranin (Landry et al., 1994), amino acid receptors (Miyashiro et al., 1994), the cAMP response element binding protein (CREB) (Crino et al., 1998), and dendrin (Herb et al., 1997). The noncoding RNA BC1 (Tiedge et al., 1991), ribosomal RNAs (Kleiman et al., 1993), and tRNAs (Tiedge and Brosius, 1996) are also present in dendrites. Dendritic tran-scripts were detected in synaptosome preparations (Chicurel et al., 1993; Rao and Steward, 1993), and isolated dendrites are capable of protein synthesis (Torre and Steward, 1992; Crino and Eberwine, 1996). Moreover, specific forms of synaptic plasticity in the rat hippocampus seem to depend on translation in dendrites (Kang and Schuman, 1996). These findings imply that extrasomatic protein synthesis influences the protein composition in dendritic compartments and contributes to modulations of synaptic function (for review, see Steward, 1994, 1997; Kindler et al., 1997;Kuhl and Skehel, 1998; Tiedge et al., 1999).
Modulation of extrasomatic protein composition and synaptic transmission seems to partially rely on dendritic targeting of selected transcripts and their regulated decentralized translation. This idea is supported by the observation that arc/arg3.1 mRNA and protein specifically accumulate in dendritic layers in which synapses have previously been stimulated (Steward et al., 1998). However, the molecular mechanisms directing selective mRNA targeting to dendrites and local protein synthesis are poorly understood. In other cell systems, such as Drosophila embryos and Xenopusoocytes, regulated interactions between mRNA signal sequences andtrans-acting factors govern cytoplasmic transport and site-specific translation of various transcripts (St. Johnston, 1995). Our aim was to functionally characterize cis-acting dendritic targeting sequences in MAP2 mRNAs. This was accomplished by the expression of chimeric transcripts containing various MAP2 mRNA fragments in two primary neuronal cell systems and an analysis of their subcellular distribution. A 640 nucleotide element contained in the 3′ untranslated region (3′ UTR) of MAP2 transcripts was found to be both sufficient and essential to mediate efficient dendritic mRNA localization in two different neuronal cell types.
MATERIALS AND METHODS
Construction of eukaryotic expression vectors. The basic vector pNE expresses a nuclear location signal (NLS)/enhanced green fluorescent protein (EGFP) fusion protein driven by the β-actin promoter from chicken. It was derived from the plasmid βact-16 (Fregien and Davidson, 1986) provided with additional cloning sites and the GFP cDNA sequence (pβact-hCGFP; courtesy of A. Matus, Friedrich Miescher Institute, Basel, Switzerland). TheNcoI–NotI fragment was replaced by the corresponding EGFP cDNA fragment from pEGFP-N1 (Clontech Laboratories, Palo Alto, CA). A sequence encoding an NLS (MGPKKKRKVGS) was introduced at the NcoI site upstream of the EGFP coding region using two oligonucleotides (5′-CATGGGGCCCAAGAAGAAACGCAAAGTGGGAAG-3′ and 5′-CATGCTTCCCACTTTGCGTTTCTTCTTGGGCCC-3′).
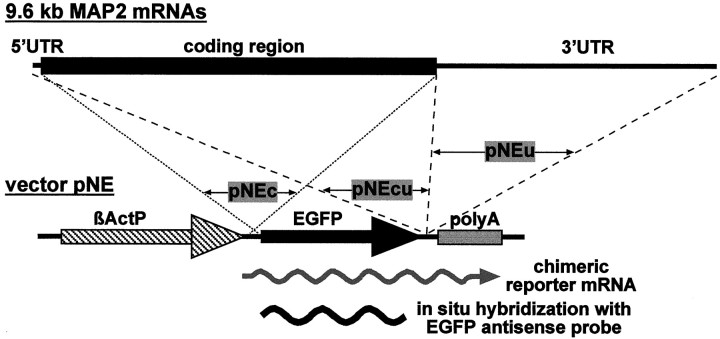
Vectors that express EGFP-encoding mRNAs fused to parts of MAP2 mRNAs contain the following cDNA sequences (GenBank/EMBL Data Bank accession numbers U30937, X51842, and U30938): pNEc (nucleotides 60–5549,X51842); pNEcu (198–367, U30937; 60–5552, X51842; 4–176,U30938); pNEu (5383–5552, X51842; 4–3720, U30938); pNEuΔ2436–3071 (5383–5552, X51842; 4–2435 and 3072–3720, U30938); the numbers in pNEu181–1963, pNEu1586–3274, pNEu2804–3728, pNEu1586–2435, pNEu2432–3274, pNEu2432–2807, pNEu2804–3274, pNEu2432–3071, pNEu2632–3274, and pNEu2632–3071 refer to the 5′ and 3′ ends of the inserted MAP2 cDNA fragments, respectively, with nucleotide numbering according to the 3′ UTR sequence (U30938); α-tubulin vector pNEtub (GenBank/EMBL Data Bank accession number V01227): nucleotides 1–1617.
Preparation and transfection of primary hippocampal neurons. Cultures of hippocampal neurons were prepared from embryonic day 21 (E21) rat embryos as described (Goslin and Banker, 1991). Briefly, cells were grown at a density of 400 cells/mm2 on 18 mm poly-l-lysine (Sigma-Aldrich Chemie, Deisenhofen, Germany)-coated glass coverslips face down above a confluent layer of glial cells in serum-free medium with N2 supplements (Goslin and Banker, 1991) at 37°C and 5% CO2. One day after plating, the cells were transfected. Thirty minutes before transfection, the coverslips were transferred into fresh N2 medium containing 1% (v/v) fetal calf serum (FCS) with cells facing up. Fifteen micrograms of pUC19 carrier DNA prepared over two CsCl gradients (Sambrook et al., 1989) and 5 μg of vector DNA (purified over affinity columns; Qiagen, Hilden, Germany) were mixed in a final volume of 100 μl in 0.25 m CaCl2. One hundred microliters of 2 × BBS (50 mm BES, 280 mm NaCl, 1.5 mmNa2HPO4, pH 6.96) were slowly added while air was continuously blown into the solution with a Pasteur pipette. Twenty minutes later, the mixture was distributed dropwise onto the cells (100 μl per coverslip) while the dish was gently swirled. After 6 hr at 37°C, 5% CO2, cells were washed twice with HBSS (Gibco BRL/Life Technologies, Eggenstein, Germany) buffered with 10 mm HEPES, pH 7.4, and placed back onto the glial cells. After 14–18 d, neurons were analyzed by in situ hybridization or immunocytochemistry. Transfection rates, as judged by the amount of cells exhibiting a positive in situ hybridization signal, were typically between 0.1 and 0.5%.
Preparation and microinjection of primary sympathetic neurons. Primary cultures of superior cervical ganglia (SCG) neurons were generated from E21 rat embryos as described by Higgins et al. (1991). Cells were seeded on poly-d-lysine (Sigma-Aldrich Chemie) and laminin (Gibco BRL/Life Technologies)-coated coverslips at a density of 5–10 cells/mm2in serum-free medium. Three days after plating, dendritic growth was induced by supplementing the medium with matrigel (75 μg/ml; Roche Diagnostics, Heidelberg, Germany). Non-neuronal cell proliferation was discouraged by adding cytosine β-d-arabinofuranoside (2 μm; Sigma-Aldrich Chemie). After 14–21 d in culture, differentiated neurons were injected with vector DNA at a concentration of 200 ng/μl using the Eppendorf microinjector 5242 (Eppendorf, Hamburg, Germany). Cells were placed into Leibovitz L-15 medium (Gibco BRL/Life Technologies) and viewed with an Axiovert 135 microscope (Zeiss, Oberkochen, Germany) at 400× magnification. During the injection of nuclei with Femto-tips (Eppendorf), the pressure was constantly kept at about 100 hPa. The injection time was restricted to 1 hr per coverslip. Afterward the cells were incubated for 4 hr at 37°C, 5% CO2 in normal SCG cell culture medium, fixed, and analyzed by in situ hybridization or immunocytochemistry.
In situ hybridization analysis. TheKpnI–XbaI EGFP cDNA fragment from pEGFP-N1 (Clontech Laboratories) was subcloned into pBluescript-SKII(−) (Stratagene, Heidelberg, Germany). The resulting plasmid pBS-EGFP was linearized with SmaI to transcribe a digoxigenin-labeled antisense EGFP RNA probe with T3 RNA polymerase according to the manufacturer’s description (Roche Diagnostics, Mannheim, Germany). For in situ hybridization, cells were briefly washed with PBS (5 mmNaH2PO4, 5 mmNa2HPO4, 2.7 mm KCl, 137 mm NaCl, pH 7.4) containing 4% (w/v) sucrose and fixed for 15 min in 4% (w/v) paraformaldehyde, 2 mmMgCl2, 5 mm EGTA, and 4% (w/v) sucrose in PBS at room temperature, followed by washing in PBS containing 4% (w/v) sucrose (three times, 5 min each). Subsequently, coverslips were irradiated in a UV cross-linker (Stratagene) set to 120 mJ. Cells were permeabilized for 3 min in PBS containing 0.1% (v/v) Triton X-100 and washed three times for five min each in PBS containing 2 mm MgCl2. Air-dried neurons were prehybridized for 2 hr at 50°C in 50% (v/v) deionized formamide, 5 × SSC, 5 × Denhardt’s solution, 0.2% (w/v) SDS, 50 μg/ml heparin, 100 μg/ml poly(A) homopolymer, 250 μg/ml denatured herring sperm DNA, and 250 μg/ml yeast tRNA. Hybridization was performed overnight at 50°C in the same solution but included ∼500 ng/ml of the in vitro synthesized digoxigenin-labeled riboprobe. Coverslips were washed twice in 1 × SSC and 0.1% (w/v) SDS at room temperature for 5 min each, and then in 0.2 × SSC, 0.1% (w/v) SDS at 65°C (twice, 10 min each). The hybridized probe was detected immunocytochemically using a sheep anti-digoxigenin antibody coupled to alkaline phosphate (Roche Diagnostics) and nitro blue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate as a substrate according to the protocol provided by the manufacturer. Cells were photographed on a Leitz Aristoplan microscope (Ernst Leitz Wetzlar, Wetzlar, Germany). To evaluate the maximal dendritic transport length of mRNA granules, pictures taken with a video camera were analyzed using the NIH image software (developed at National Institutes of Health, Bethesda, MD, and available on the Internet at http://rsb.info.nih.gov/nih-image/). For each transfected neuron the distance between the cell body and the distal-most dendritic signal was measured using the program’s segmented line tool.
Immunofluorescence microscopy of primary neuronal cultures. Hippocampal and sympathetic neurons were prepared and grown on coverslips as described above, washed in a physiological salt solution [0.9% (w/v) NaCl, 100 mm sodium phosphate buffer, pH 7.4], fixed with 4% paraformaldehyde in high-salt PBS (450 mm NaCl, 20 mm sodium phosphate buffer, pH 7.4) at room temperature for 15 min, and blocked with 5% (v/v) FCS in high-salt PBS for 30 min. For immunofluorescence microscopy, coverslips were incubated overnight with monoclonal antibodies against MAP2 (1:200 dilution; Chemicon International, Temecula, CA), tau (1:200 dilution; Roche Diagnostics) or EGFP (1:500 dilution; Clontech Laboratories) at 4°C in 2% (v/v) FCS in high-salt PBS. After three washes in high-salt PBS, coverslips were incubated overnight at 4°C with goat anti-mouse IgG antibodies conjugated with either fluorescein, Cy3, or Cy2 (Jackson ImmunoResearch Laboratories, West Grove, PA). Secondary antibodies were diluted 1:100 in 2% (v/v) FCS in high-salt PBS. After three washes in high-salt PBS, cells were mounted and photographed as described above, or a Zeiss laser-scanning microscope (Zeiss) was used.
RESULTS
Polar differentiation of cultured sympathetic and hippocampal neurons
For the characterization of molecular determinants involved in dendritic mRNA targeting, primary neurons derived from embryonic rat hippocampi and superior cervical ganglia were grown in culture. Phase-contrast micrographs presented in Figure1A,Bshow that the given cell culture conditions promote a polar differentiation of both sympathetic and hippocampal neurons as well as the formation of an extended network of processes. Visualization of a recombinant EGFP in individual neurons indicates that hippocampal cells form a slightly more complex neurite network than sympathetic neurons (Fig. 1C,D). The nature of these cell processes was further determined by immunocytochemical staining of cultures with antibodies against the dendritic and axonal marker proteins MAP2 and tau, respectively. MAP2 antibodies lead to a weak staining of neuronal cell bodies and a strong labeling of several tapered, often brancheddendrites per cell (Fig.1E,F). In contrast, tau antibodies stain a very dense meshwork of thin and long axonal processes often running in bundles (Fig.1G,H). Thus, both sympathetic and hippocampal neurons develop dendrites and axons that can be reliably distinguished on the basis of morphological and immunocytochemical criteria. This feature and the fact that in cell culture both neuronal cell types have been shown to localize endogenous MAP2 transcripts to dendrites (Bruckenstein et al., 1990; Kleiman et al., 1990) make these cell systems well suited for functional studies on the molecular components underlying dendritic mRNA targeting.
Fig. 1.
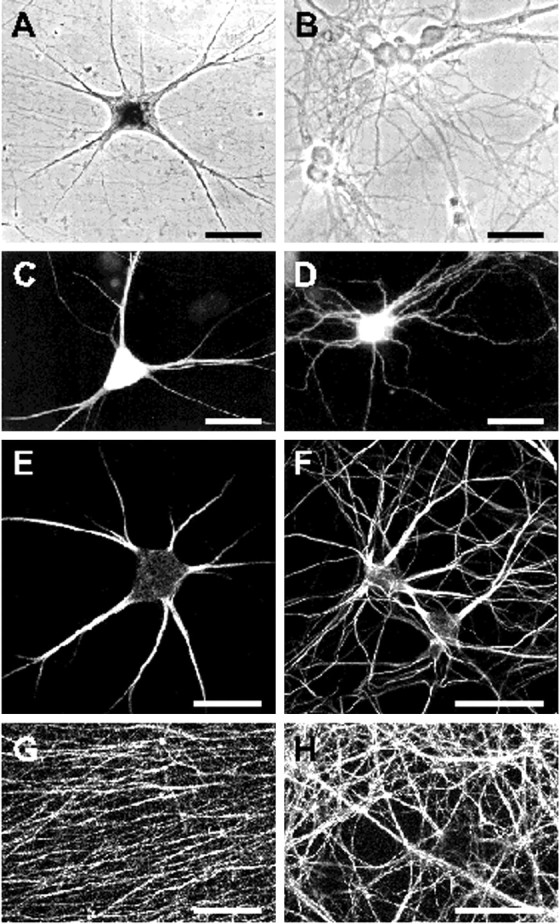
Polar morphology and protein compartmentalization in primary neurons. Phase-contrast (A, B) and fluorescent (C–H) micrographs of cultured sympathetic (A, C, E, G) and hippocampal neurons (B, D, F, H) after 2 weeks in culture. Phase-contrast images shown in A and Breveal an elaborated neuronal network present in both primary cell cultures. C, D, Neurons were microinjected and transfected, respectively, with pNE vector DNA leading to the expression of EGFP. The recombinant protein was either directly visualized by autofluorescence (D) or detected through immunocytochemistry with an anti-EGFP antibody and a Cy2-coupled secondary antibody (C).E–H, Neurons immunostained with mouse monoclonal antibodies against the cytoskeletal proteins MAP2 (E, F) and tau (G, H) and secondary goat anti-mouse antibodies coupled to Cy3. Neuronal somata and dendrites are intensely stained with anti-MAP2 antibodies (E, F). Dendrites taper over their length and often form secondary branches. In hippocampal cultures, dendrites are longer relative to the cell body diameter and branch more frequently than processes of sympathetic neurons. G, H,Anti-tau antibodies primarily label the finely meshed axonal network consisting of thin processes that course in and out of the field of view. Processes often assemble to thicker axon bundles.A–D represent scanned prints of pictures taken with a conventional Leitz fluorescent microscope. Micrographs shown inE–H were captured with a laser-scanning microscope. Scale bars, 50 μm.
cis-acting sequences direct dendritic targeting of MAP2 mRNAs
Observations on mRNA localization in various non-neuronal cell systems indicate that localized transcripts typically carrycis-acting transport signals that interact withtrans-acting proteins to mediate cytoplasmic mRNA targeting. Matus and colleagues (Marsden et al., 1996) have suggested an alternative mechanism in which a translational complex is localized by virtue of a targeting signal encoded on the nascent protein leading to passive co-transport of the corresponding transcripts. To investigate whether MAP2 mRNA localization to dendrites depends on targeting signals contained in transcripts or polypeptides, we have introduced vectors into primary neurons that lead to the expression of chimeric mRNAs and proteins. In the basic vector pNE, the avian β-actin promoter initiates transcription of mRNAs encoding an EGFP variant carrying an N-terminal NLS. Previous experiments have shown that somatically injected EGFP transcripts and α-tubulin mRNAs transcribed from eukaryotic expression vectors remain restricted to the somata of sympathetic neurons (Muslimov et al., 1997; Prakash et al., 1997). To investigate whether individual MAP2 mRNA or protein segments are capable of imparting extrasomatic localization competence on an exogenous reporter mRNA, different cDNA fragments corresponding to coding and noncoding regions of MAP2 mRNAs were inserted next to the EGFP cDNA in the vector pNE (Fig. 2). As a control, the cDNA of the entire α-tubulin transcript was inserted downstream of the EGFP sequence. The subcellular localization of chimeric mRNAs expressed from these vectors was monitored by in situ hybridization with a digoxygenin-labeled antisense EGFP RNA. Thus, all recombinant transcripts are detected with the same sensitivity level and distinguished from endogenous MAP2 mRNAs. In hybridization experiments with a sense EGFP probe, no significant signals were observed (see Fig.4I,J).
Fig. 2.
Schematic representation of the expression and detection of recombinant transcripts in primary neurons to identifycis-acting dendritic targeting signals in MAP2 mRNAs. Different MAP2 cDNA fragments are inserted adjacent to the EGFP cDNA in vector pNE. From pNEc a MAP2/EGFP fusion protein encoding mRNA is transcribed, whereas in pNEcu and pNEu MAP2 sequences are inserted downstream of the EGFP stop codon. Subcellular localization of chimeric transcripts in primary neurons is determined by nonradioactivein situ hybridizations with a digoxygenin-labeled RNA probe complementary to EGFP sequences. βActP, β-actin promoter; EGFP, enhanced green fluorescent protein; polyA, polyadenylation signal.
Fig. 4.
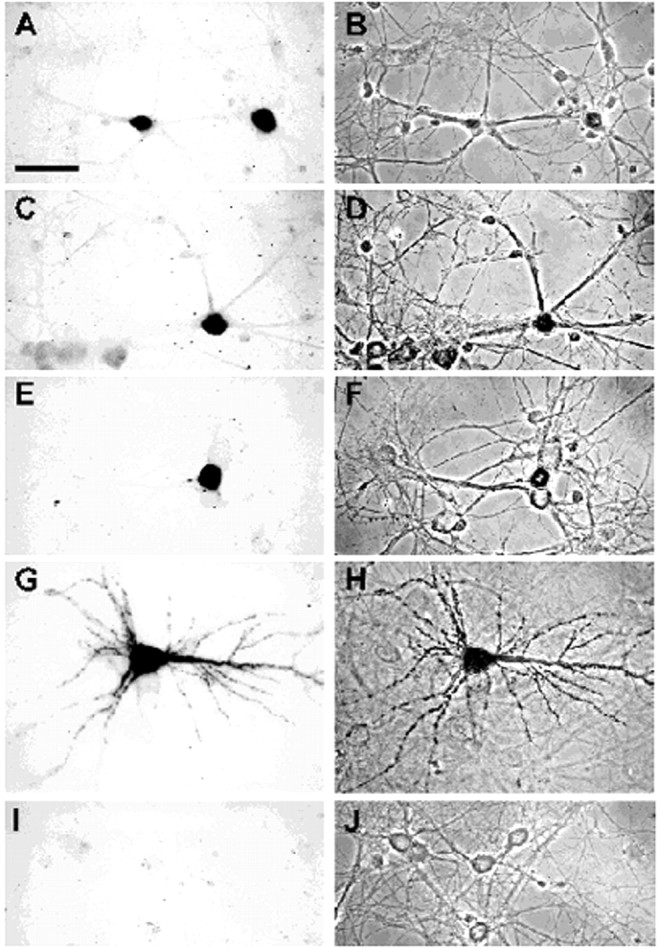
Detection of chimeric transcripts in hippocampal neurons after transfection with vector DNA. Bright-field (A, C, E, G, I) and phase-contrast (B, D, F, H, J) micrographs of neurons transfected with pNEtub (A, B, I, J), pNEcu (C, D), pNEc (E, F), and pNEu (G, H) are shown. In A–H, neurons were hybridized with a digoxigenin-labeled antisense EGFP probe. After incubation with alkaline phosphatase-conjugated sheep anti-digoxigenin antibody, the addition of nitro blue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate resulted in a dark-colored reaction product in a small fraction of the cells. Hybridization with a sense EGFP probe never resulted in significant cell staining (I, J). Recombinant transcripts with sequences of the MAP2 coding region (C–F) or α-tubulin mRNA (A, B) are only detected in the cell soma. In contrast, chimeric mRNAs possessing the entire 3′ UTR of MAP2 transcripts are present in dendrites (G, H). Cells were photographed with a video camera attached to a Leitz microscope. Scale bar, 50 μm.
In SCG cultures, vectors were microinjected into the nuclei of fully differentiated neurons. The subcellular localization of reporter mRNAs was monitored 4 hr after injection. In contrast, undifferentiated hippocampal neurons were transfected with vector DNAs the day after plating. Analysis of transcript distribution was performed ∼2 weeks after transfection. During this time neurons had developed axons, dendrites, and synapses (Fig. 1).
Interestingly, despite these methodological differences, data obtained with both cell systems are very similar. Control transcripts consisting of the EGFP coding region fused to the entire α-tubulin mRNA were restricted to cell somata and proximal-most dendritic segments of sympathetic (Fig. 3A) and hippocampal neurons (Fig.4A). This is consistent with the observation that in both primary cultures endogenous α-tubulin transcripts are found only in neuronal cell bodies and the basal parts of dendrites (Bruckenstein et al., 1990; Kleiman et al., 1990). Next we examined two chimeric transcripts in which sequences from the MAP2 coding region are fused to the EGFP mRNA. In the first, the 5′ UTR and the entire coding region of MAP2 transcripts are located downstream of the EGFP sequence such that the MAP2 sequences are not translated (vector pNEcu). These reporter mRNAs remained in the cell soma (Figs. 3C, 4C). In the second, the MAP2 coding region is situated upstream and in frame with the EGFP sequence (vector pNEc). These transcripts were also retained in the cell soma (Figs. 3E, 4E). The ability of the latter mRNAs to express a MAP2/EGFP fusion protein was confirmed by transfecting the vector into HEK293 cells. As expected, an autofluorescent protein decorated the microtubules of transfected cells (data not shown). In contrast to the above results, reporter RNAs carrying the entire MAP2 3′ UTR downstream of the EGFP sequence were found in dendrites of the majority of microinjected sympathetic and transfected hippocampal neurons (vector pNEu) (Figs. 3G,4G). The findings described above are summarized in Figure5. Labeled cells were grouped into two classes: (1) those exhibiting a “somatic” RNA distribution pattern for which the in situ hybridization signal was restricted to cell bodies and proximal-most sections of dendrites and (2) cells showing “dendritic” localization of chimeric transcripts to parts of at least one cell process that were farther away from the soma than one cell body diameter as measured from the base of the dendrite. On the basis of these criteria, chimeric mRNAs containing the MAP2 3′ UTR were dendritically localized in ∼60% of the evaluated sympathetic and hippocampal neurons. In contrast, recombinant reporter transcripts containing tubulin sequences or MAP2 5′ untranslated and coding regions almost exclusively exhibited a somatic distribution pattern (>96%). In none of the microinjected or transfected cells were reporter transcripts ever detected in axons. Taken together, these findings show that MAP2 mRNAs are targeted selectively and specifically to dendrites of sympathetic and hippocampal neurons. Dendritic localization of MAP2 transcripts is directed by cis-acting targeting elements situated in the 3′ UTR of the mRNAs and is not indirectly mediated by an inherent dendritic targeting signal of the nascent MAP2 polypeptide chain.
Fig. 3.
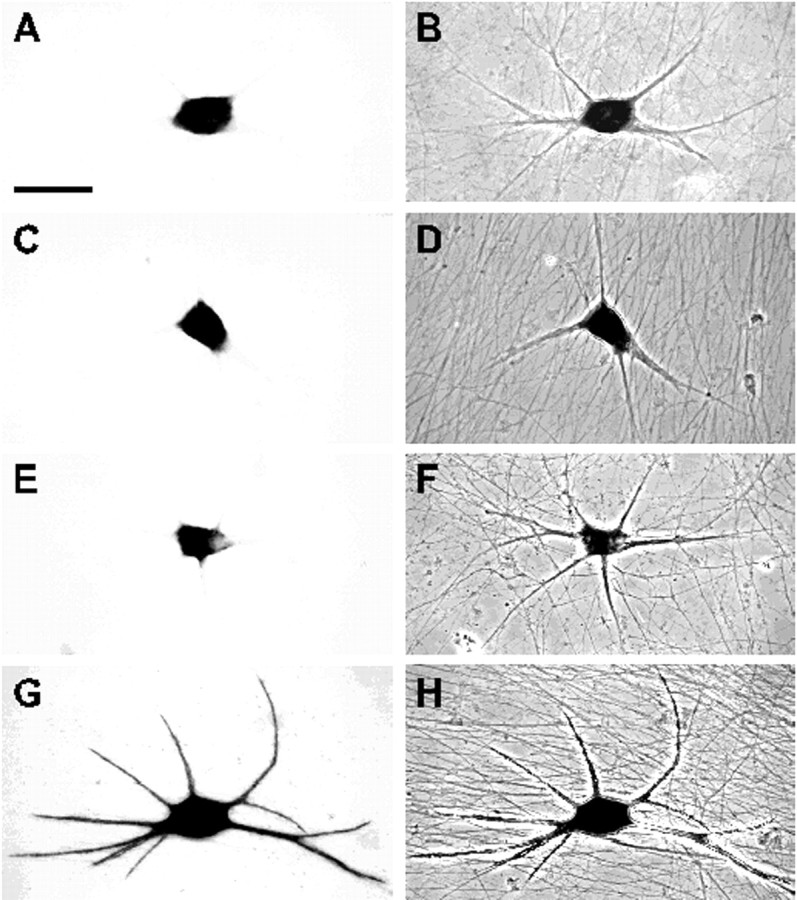
In situ hybridization analysis of the subcellular distribution of chimeric mRNAs in sympathetic neurons after vector DNA injection into the nucleus. Bright-field (A, C, E, G) and phase-contrast (B, D, F, H) micrographs of cells injected with pNEtub (A, B), pNEcu (C, D), pNEc (E, F), and pNEu (G, H). In neurons shown in A–H, reporter mRNAs were immunocytochemically detected using a digoxigenin-labeled antisense EGFP probe and an alkaline phosphatase-conjugated sheep anti-digoxigenin antibody. After the addition of nitro blue tetrazolium and 5-bromo-4-chloro-3-indolyl phosphate, a dark-colored reaction product was detectable. Chimeric RNAs containing α-tubulin sequences (A, B) or the MAP2 coding region (C, D, E, F) are restricted to cell somata, whereas transcripts carrying the entire 3′ UTR of MAP2 mRNAs are found in dendrites (G, H). Images were collected with a video camera connected to a Leitz microscope. Scale bar, 50 μm.
Fig. 5.
Subcellular distribution of chimeric transcripts in sympathetic and hippocampal neurons. In the left panel, the molecular structure of 9.6 kb MAP2 transcripts is schematically indicated with black bars andlines representing coding and noncoding regions, respectively. Below, MAP2 sequences that are present in recombinant mRNAs derived from the corresponding vectors are indicated asheavy black bars. From pNEc, a MAP2/EGFP fusion protein is synthesized (indicated by a black arrow), whereas the MAP2 coding region in pNEcu transcripts is not translated into a protein. The right panel lists the relative amount of cells showing dendritic localization of chimeric transcripts in sympathetic and hippocampal neurons and the total number of evaluated cells per construct. In both types of transgenic primary neurons, only recombinant mRNAs containing the MAP2 3′ UTR are efficiently localized to dendrites.
The cis-acting dendritic targeting element resides in a subregion of the 3′ untranslated region
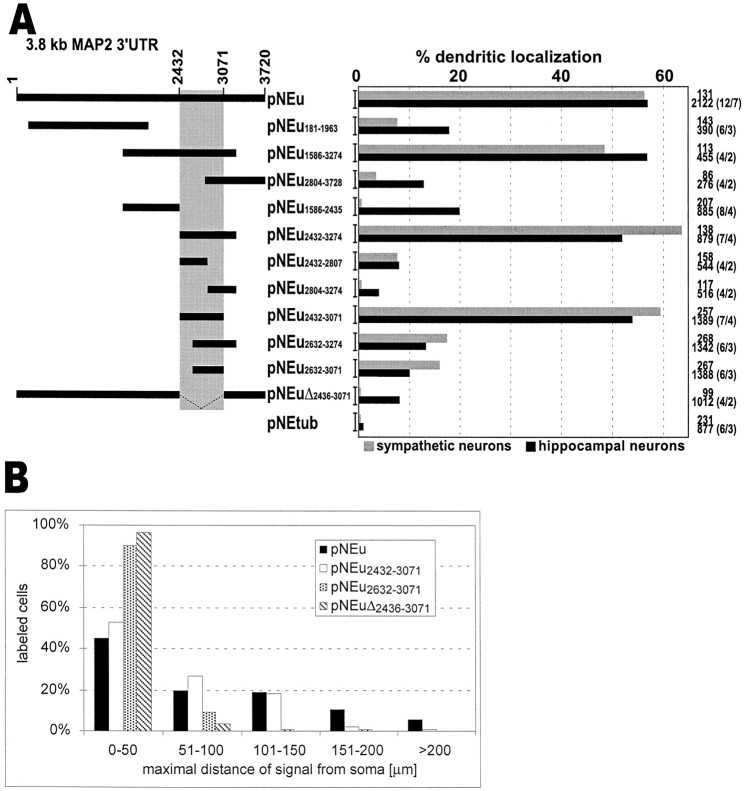
Because the 3′ UTR of MAP2 mRNAs comprises ∼3.74 kb (Kindler et al., 1996), it seems likely that this region mediates additional functions besides transcript localization, such as RNA stability and translation. To further delineate the cis-acting dendritic targeting element (DTE) in this region, we have expressed a number of chimeric transcripts containing subfragments of the 3′ UTR in both sympathetic and hippocampal neurons. Data obtained with both primary cell systems are summarized in Figure6A.
Fig. 6.
A, Identification of a 640 nucleotide cis-acting DTE in the 3′ UTR of MAP2 transcripts. In the left panel, regions of the MAP2 3′ UTR included in chimeric mRNAs transcribed from the corresponding vectors are shown as heavy black bars. In theright panel, the percentage of primary neurons exhibiting dendritic mRNA localization patterns and the total number of analyzed cells are shown. In parentheses, the total number of analyzed coverslips/independent transfections of hippocampal neurons are shown. Only chimeric transcripts containing a 640-nucleotide-long sequence highlighted in gray are detected with high frequency in dendrites of sympathetic and hippocampal neurons. Deletion of this targeting element omits dendritic localization. B, Dendritic mRNA localization patterns of several hybrid mRNAs. The histogram displays the relative amount of cells that show their distal-most in situ staining in the indicated ranges of distance from the soma. For each construct, 200 cells from two independent transfections of hippocampal neurons were analyzed. In cells that exhibit a low frequency of dendritic mRNA localization (pNEu2632–3071 and pNEuΔ2436–3071 transfected cells), recombinant transcripts on average traveled less far into the cell processes than in neurons with a high percentage of dendritic labeling (pNEu and pNEu2432–3071).
As a first step to further characterize the DTE, three overlapping mRNA regions spanning most of the 3′ UTR [Fig. 6A, vectors pNEu181–1963, pNEu1586–3274, pNEu2804–3728 (nucleotide numbering according to the entire 3′ UTR sequence)] were tested for their ability to mediate dendritic localization of reporter transcripts. Interestingly, only mRNAs containing nucleotides 1586–3274 were targeted into dendrites with approximately the same frequency as transcripts possessing the entire 3′ UTR. In contrast, for mRNAs comprising nucleotides 181–1963 and 2804–3728 (pNEu181–1963 and pNEu2804–3728), the relative amount of cells showing dendritic mRNA targeting was dramatically decreased. Results obtained with sympathetic and hippocampal neurons are similar, with a slightly higher frequency of dendritic localization observed in hippocampal cells (Fig. 6A). Thus, in MAP2 transcripts, essential signals for dendritic mRNA localization seem to reside in the middle part of the 3′ UTR (nucleotides 1586–3274). The relatively low, but in comparison to EGFP/α-tubulin transcripts significantly higher, frequency of dendritic localization of mRNAs transcribed from vectors pNEu181–1963 and pNEu2804–3728 (Fig. 6A) may indicate that the corresponding subregions of the MAP2 3′ UTR contribute to transcript targeting, although to a minor extent.
To further outline DTE sequences, the targeting capacities of the 5′ and 3′ parts of the RNA fragment spanning nucleotides 1586–3274 were individually analyzed. Transcripts derived from pNEu2432–3274 were localized to dendrites with high frequency, whereas mRNAs transcribed from pNEu1586–2435 were predominantly restricted to cell bodies of hippocampal and sympathetic neurons. When thecis-active mRNA fragment spanning nucleotides 2432–3274 was further divided into two halves (pNEu2432–2807and pNEu2804–3274) or 200 nucleotide deletions were made from either the 5′ end (pNEu2632–3274) or from both ends (pNEu2632–3071), all four resulting subfragments were unable to mediate significant dendritic mRNA targeting. In contrast, deleting 200 nucleotides from the 3′ end (pNEu2432–3071) did not interfere with the extrasomatic localization capacity of the resulting fragment (Figs.6A,7A,B,E,F). Taken together, these data imply that a 640 nucleotide region containing nucleotides 2432–3071 of the MAP2 3′ UTR contains allcis-acting signals sufficient for dendritic mRNA localization. Further deletions from either the 5′ (pNEu2632–3071) or 3′ end (pNEu2432–2807) of this DTE dramatically interferes with its extrasomatic targeting capacity, indicating that the entire 640 nucleotide region is required to mediate transcript localization into dendrites (Fig. 6A). The central role of the DTE in dendritic mRNA targeting was further confirmed by its deletion from the entire 3′ UTR (pNEuΔ2436–3071). In contrast to dendritically localized reporter transcripts containing the entire 3′ UTR (pNEu), mRNAs transcribed from pNEuΔ2436–3071 are primarily restricted to cell somata (Figs. 6A, 7C,D,G,H). Thus, a 640 nucleotide DTE situated in the 3′ UTR of MAP2 mRNAs appears to be sufficient and essential for their efficient localization into dendrites of both sympathetic and hippocampal neurons. Cells expressing pNEuΔ2436–3071 transcripts exhibited a slightly lower frequency of dendritic mRNA localization than neurons that were transfected with the constructs pNEu181–1963 and pNEu1586–2435. This is a surprising observation because all MAP2 mRNA sequences found in pNEu181–1963 and pNEu1586–2435 transcripts that may induce a low-frequency dendritic localization are also present in pNEuΔ2436–3071 mRNAs. It is possible that the 640 nucleotide DTE deletion in pNEuΔ2436–3071transcripts induces a change in the secondary structure of the remaining 3′ UTR parts, thereby inhibiting a minor dendritic localization capacity of these sequences.
Fig. 7.
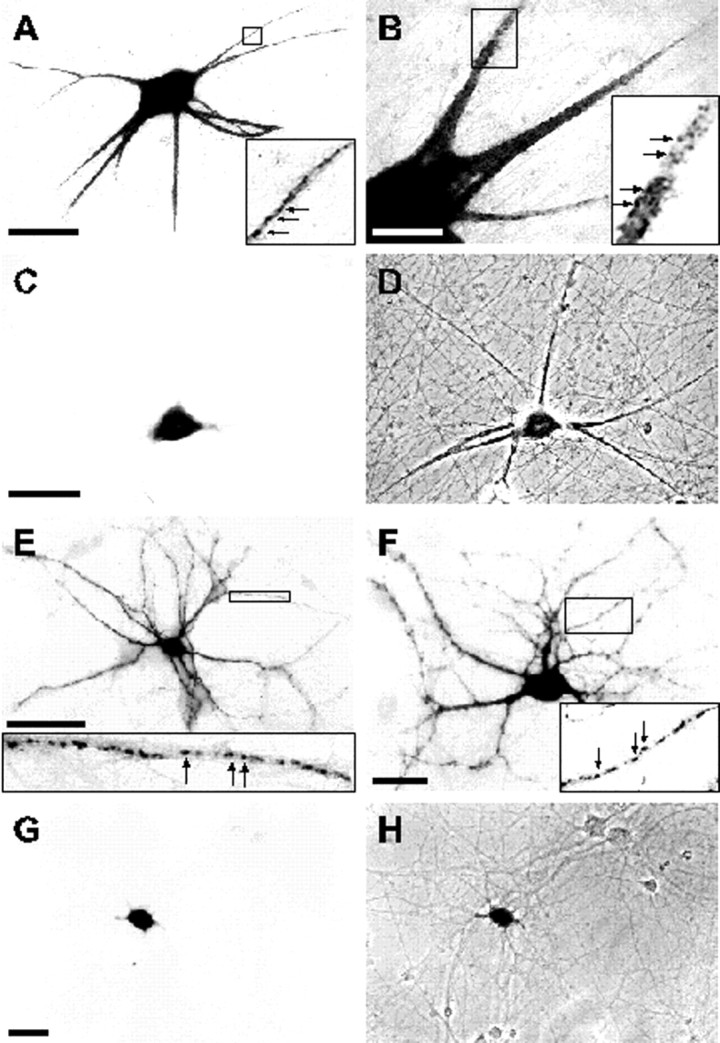
In situ hybridization analysis of the subcellular distribution of chimeric mRNAs in sympathetic (A–D) and hippocampal neurons (E–H). Bright-field (A–C, E–G) and phase-contrast (D, H) micrographs of cells expressing recombinant transcripts from pNEu2432–3071(A, B, E, F) and pNEuΔ2436–3071(C, D, G, H) are shown. The 640 nucleotide DTE in the MAP2 3′ UTR is sufficient to mediate extrasomatic mRNA localization in both neuronal cell types (A, B, E, F). Higher magnification images shown as insets depict the particulate nature of the color-reaction product of the nonradioactivein situ hybridization in dendrites (arrows). Deletion of the DTE from the 3′ UTR omits the dendritic targeting capacity (C, D, G, H). Images were captured either with a video camera (A–D) or on conventional film (E–H). Cand G are bright-field images of areas shown as phase-contrast micrographs in D and H, respectively. Scale bars: A, C,E, 50 μm; B,F, G, 20 μm.
To compare the dendritic distribution patterns of recombinant transcripts exhibiting a low versus high frequency of dendritic localization, we have measured the distance between the base of the dendrite and the distal-most RNA granule in hippocampal neurons transfected with four different constructs. The data summarized in Figure 6B indicate that recombinant transcripts that lead to a low dendritic localization frequency (pNEu2632–3071 and pNEuΔ2436–3071 mRNAs) tend to migrate less far into dendrites than transcripts inducing a high percentage of dendritic labeling (pNEu and pNEu2432–3071). This finding shows that the DTE determines not only the amount of cells exhibiting extrasomatic mRNA localization but also the mean dendritic transport distance.
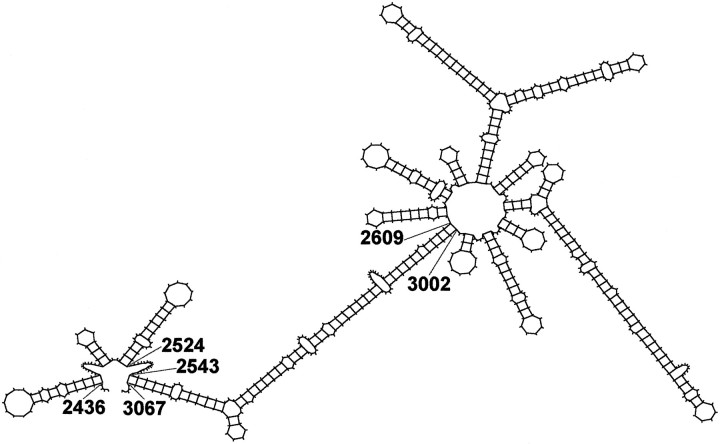
Several lines of evidence suggest that a specific secondary RNA structure may serve as a localization signal in different cell systems (Macdonald, 1990; Chartrand et al., 1999). The mfold program (Zuker, 1989) was used to predict an optimal secondary structure for the DTE on the basis of free energy minimization (Fig.8). The predicted structure consists of two stem-loop clusters (nucleotides 2436–2524 and 2610–3000 of the 3′ UTR) that are connected via a long double-stranded region (nucleotides 2544–2609 and 3002–3067). Future experiments will be necessary to identify elements within this structure that are essential for dendritic RNA localization.
Fig. 8.
Predicted secondary structure of the dendritic targeting element in rat MAP2 mRNAs. The structure was determined using the mfold program 2.3 developed by Zuker (1989) included in the Wisconsin Package (Genetics Computer Group, Madison, WI). The free energy of the structure shown is −160.7 kcal/mol.
Dendritic mRNA granules
Microinjection of fluorescently labeled transcripts in cultured oligodendrocytes (Ainger et al., 1993) and labeling of endogenous RNAs in cultured hippocampal neurons with a fluorescent dye (Knowles et al., 1996) imply that transport of mRNAs into cell processes occurs in the form of distinct multimolecular complexes. Here we show that thein situ hybridization signal in cell bodies of transfected and microinjected primary neurons mostly exhibits a uniform distribution. In contrast, dendritic labeling typically appears in the form of granules scattered along the length of cell processes of both sympathetic (Fig. 7A,B) and hippocampal neurons (Fig.7E,F). The patchy in situ hybridization signal observed in dendrites may reflect a discontinuous nature of the extrasomatic mRNA delivery apparatus, with distinct moving mRNA granules, or alternatively, with transcripts preferentially docking to particular binding sites along dendritic shafts.
DISCUSSION
We have used two assay systems based on primary neurons in culture to analyze the molecular components involved in dendritic targeting of MAP2 mRNAs. Our results show that dendritic translocation is a selective process that depends on MAP2 mRNA sequences. Thesecis-elements specifically mediate dendritic but not axonal mRNA targeting. Control transcripts containing α-tubulin and EGFP sequences were seen to be almost exclusively restricted to somata. The data presented here demonstrate that neurons possess mechanisms to specifically target selected mRNAs into dendritic processes.
Our observation that the coding region of MAP2 mRNAs does not impart extrasomatic localization capacity on nondendritictranscripts shows that MAP2 transcript targeting into dendrites is not mediated indirectly by an inherent dendritic targeting signal of the nascent MAP2 polypeptide chain as has been proposed by Marsden et al. (1996). Similarly, Wallace et al. (1998) showed that an inhibition of protein synthesis with cycloheximide does not disrupt dendritic localization of arc/arg3.1 transcripts in the intact rat brain. Here we show that chimeric mRNAs containing the entire 3′ UTR of MAP2 transcripts localize to dendrites with high efficiency. This finding indicates that this subregion of MAP2 transcripts contains cis-acting signals for extrasomatic mRNA targeting. This is consistent with the observation that in transgenic mice that express MAP2c transcripts missing the 3′ noncoding region, recombinant mRNAs were found to be restricted to hippocampal cell bodies (Marsden et al., 1996). Moreover, most cis-acting targeting sequences that have so far been identified on mRNAs that undergo cytoplasmic localization in non-neuronal cell systems are situated in 3′ noncoding regions (St. Johnston, 1995; Bassell and Singer, 1997; Steward, 1997; Kuhl and Skehel, 1998). Thus, in addition to regulating transcript stability and translation, for example, 3′ UTR sequences may direct the spatial distribution of mRNAs in different cell types.
To date, a limited number of studies to functionally characterizecis-acting targeting sequences in dendritic mRNAs have been performed. Data obtained with a microinjection assay comparable to what we have used here indicate that regions involved in the localization of vasopressin transcripts into dendrites are redundant encompassing sequences of the coding region and potentially the 3′ UTR (Prakash et al., 1997). Conversely, using a transgenic approach, the 3′ UTR of α-CaMKII mRNAs has been shown to impart extrasomatic translocation competence on normally nondendritic β-galactosidase transcripts (Mayford et al., 1996). Subsequent studies to further delineate the exact sequence elements involved in dendritic mRNA localization have not yet been performed. Here we present a study to functionally identify a cis-acting DTE. Our results indicate that 640 nucleotides from the 3′ UTR of MAP2 mRNAs are sufficient to mediate efficient dendritic localization of chimeric reporter transcripts. Most of this region seems to be essential for correct subcellular mRNA localization because further deletions from both ends of the element disrupt the targeting capacity. Interestingly, the DTE in MAP2 transcripts does not share any striking similarity with the 3′ UTR sequence of α-CaMKII mRNAs (A. Blichenberg, S. Kindler, and D. Richter, unpublished observations) or the DTEs of vasopressin transcripts (Rehbein et al., 1986; Prakash et al., 1997). Although the existence of rather short conserved sequence stretches that perform a specific cellular function cannot be excluded, different mRNA species may use slightly distinct molecular means to couple to a dendritic transport system. This idea is supported by the observation that MAP2 and α-CaMKII mRNAs exhibit diverse subcellular distribution patterns in dendrites. In hippocampal neurons in brain, the relative amount of MAP2 transcripts seems to decrease from proximal to distal regions (Garner et al., 1988), whereas α-CaMKII mRNAs appear to be evenly distributed throughout the dendritic arbor (Burgin et al., 1990).
Using microinjection of radioactively labeled RNAs into sympathetic neurons, the extrasomatic targeting sequences in the short, noncoding BC1 RNA have been restricted to its 5′ part containing approximately 62 nucleotides (Muslimov et al., 1997). This is the shortest sequence element described so far that is capable of mediating dendritic targeting of chimeric RNAs. Interestingly, the MAP2 DTE does not possess any obvious sequence similarity with this region. The extent of structural conservation within DTEs may become clearer when additional localization elements of other extrasomatic RNAs will be characterized in the future. In this context it seems important to point out that the sequence of a DTE per se is probably not the most important feature for dendritic RNA localization. In contrast, secondary and higher-order RNA structures may represent more essential determinants for extrasomatic transcript localization (Macdonald, 1990; Chartrand et al., 1999). A computer-based prediction indicates that the DTE in MAP2 mRNAs has the ability to form a complex secondary structure. Specific secondary structures of other RNA regions have indeed been shown to perform critical roles in specifying RNA–protein interactions and thus specific cellular functions (Draper, 1995; Ferrandon et al., 1997; Conn and Draper, 1998).
The DTE of MAP2 mRNAs functions in two distinct neuronal cell types, namely primary sympathetic and hippocampal neurons. This finding implies that the dendritic mRNA transport machinery is identical in different neuronal cell types. Small divergences in the results obtained with both primary cell systems either may indicate minor differences in the molecular system underlying dendritic mRNA localization or may be related to differences in methodology, such as the developmental time point of vector DNA introduction into neurons.
In both cultured sympathetic and hippocampal neurons, chimeric mRNAs exhibit a granular pattern in dendrites. This is in accordance with several other studies using primary neuronal cell culture systems. In cortical neurons, Knowles et al. (1996) visualized individual moving granules in neurites after fluorescent labeling of the entire RNA population. In sympathetic neurons, chimeric transcripts containing sequences of dendritically localized BC1 or vasopressin RNAs were observed to assemble into clusters along dendritic shafts (Muslimov et al., 1997; Prakash et al., 1997). The endogenous β-actin mRNA was found to assemble into complexes in growth cones of early differentiating hippocampal neurons (Bassell et al., 1998). Moreover, after microinjection into cultured oligodendrocytes, fluorescently labeled myelin basic protein mRNA forms granules that undergo anterograde movement into cell processes (Ainger et al., 1993). Taken together these observations imply that in distinct neuronal and non-neuronal cells, mRNA molecules are delivered to cytoplasmic subregions in the form of multimolecular transport complexes.
Footnotes
This research was supported by the Deutsche Forschungsgemeinschaft (Ri191-19-1, Ri192-21-5). This work forms part of a thesis (A.B.).
Correspondence should be addressed to Stefan Kindler, Institute for Cell Biochemistry and Clinical Neurobiology, University of Hamburg, D-20246 Hamburg, Germany.
REFERENCES
- 1.Ainger K, Avossa D, Morgan F, Hill SJ, Barry C, Barbarese E, Carson JH. Transport and localization of exogenous myelin basic protein mRNA microinjected into oligodendrocytes. J Cell Biol. 1993;123:431–441. doi: 10.1083/jcb.123.2.431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bassell G, Singer RH. mRNA and cytoskeletal filaments. Curr Opin Cell Biol. 1997;9:109–115. doi: 10.1016/s0955-0674(97)80159-7. [DOI] [PubMed] [Google Scholar]
- 3.Bassell GJ, Zhang H, Byrd AL, Femino AM, Singer RH, Taneja KL, Lifshitz LM, Herman IM, Kosik KS. Sorting of β-actin mRNA and protein to neurites and growth cones in culture. J Neurosci. 1998;18:251–265. doi: 10.1523/JNEUROSCI.18-01-00251.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bruckenstein DA, Lein PJ, Higgins D, Fremeau RT., Jr Distinct spatial localization of specific mRNAs in cultured sympathetic neurons. Neuron. 1990;5:809–819. doi: 10.1016/0896-6273(90)90340-l. [DOI] [PubMed] [Google Scholar]
- 5.Burgin KE, Waxham MN, Rickling S, Westgate SA, Mobley WC, Kelly PT. In situ hybridization histochemistry of Ca2+/calmodulin-dependent protein kinase in developing rat brain. J Neurosci. 1990;10:1788–1798. doi: 10.1523/JNEUROSCI.10-06-01788.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chartrand P, Meng XH, Singer RH, Long RM. Structural elements required for the localization of ASH1 mRNA and of a green fluorescent protein reporter particle in vivo. Curr Biol. 1999;9:333–336. doi: 10.1016/s0960-9822(99)80144-4. [DOI] [PubMed] [Google Scholar]
- 7.Chicurel ME, Terrian DM, Potter H. mRNA at the synapse: analysis of a synaptosomal preparation enriched in hippocampal dendritic spines. J Neurosci. 1993;13:4054–4063. doi: 10.1523/JNEUROSCI.13-09-04054.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Conn GL, Draper DE. RNA structure. Curr Opin Struct Biol. 1998;8:278–285. doi: 10.1016/s0959-440x(98)80059-6. [DOI] [PubMed] [Google Scholar]
- 9.Crino PB, Eberwine J. Molecular characterization of the dendritic growth cone: regulated mRNA transport and local protein synthesis. Neuron. 1996;17:1173–1187. doi: 10.1016/s0896-6273(00)80248-2. [DOI] [PubMed] [Google Scholar]
- 10.Crino PB, Khodakhah K, Becker K, Ginsberg S, Hemby S, Eberwine J. Presence and phosphorylation of transcription factors in developing dendrites. Proc Natl Acad Sci USA. 1998;95:2313–2318. doi: 10.1073/pnas.95.5.2313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Draper DE. Protein-RNA recognition. Annu Rev Biochem. 1995;64:593–620. doi: 10.1146/annurev.bi.64.070195.003113. [DOI] [PubMed] [Google Scholar]
- 12.Ferrandon D, Koch I, Westhof E, Nusslein-Volhard C. RNA-RNA interaction is required for the formation of specific bicoid mRNA 3′UTR-STAUFEN ribonucleoprotein particles. EMBO J. 1997;16:1751–1758. doi: 10.1093/emboj/16.7.1751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fregien N, Davidson N. Activating elements in the promoter region of the chicken β-actin gene. Gene. 1986;48:1–11. doi: 10.1016/0378-1119(86)90346-x. [DOI] [PubMed] [Google Scholar]
- 14.Furuichi T, Simon-Chazottes D, Fujino I, Yamada N, Hasegawa M, Miyawaki A, Yoshikawa S, Guenet JL, Mikoshiba K. Widespread expression of inositol 1,4,5-trisphosphate receptor type 1 gene (Insp3r1) in the mouse central nervous system. Receptors Channels. 1993;1:11–24. [PubMed] [Google Scholar]
- 15.Garner CC, Tucker RB, Matus A. Selective localization of messenger RNA for cytoskeletal protein MAP2 in dendrites. Nature. 1988;336:674–677. doi: 10.1038/336674a0. [DOI] [PubMed] [Google Scholar]
- 16.Goslin K, Banker G. Rat hippocampal neurons in low density culture. In: Banker G, Goslin K, editors. Culturing nerve cells. MIT; Cambridge, MA: 1991. pp. 251–281. [Google Scholar]
- 17.Herb A, Wisden W, Catania M, Marechal D, Dresse A, Seeburg P. Prominent dendritic localization in forebrain neurons of a novel mRNA and its product, dendrin. Mol Cell Neurosci. 1997;8:367–374. doi: 10.1006/mcne.1996.0594. [DOI] [PubMed] [Google Scholar]
- 18.Higgins D, Lein PJ, Osterhout DJ, Johnson MI. Tissue culture of mammalian autonomic neurons. In: Banker G, Goslin K, editors. Culturing nerve cells. MIT; Cambridge, MA: 1991. pp. 177–205. [Google Scholar]
- 19.Kang H, Schuman E. A requirement for local protein synthesis in neurotrophin-induced hippocampal synaptic plasticity. Science. 1996;273:1402–1406. doi: 10.1126/science.273.5280.1402. [DOI] [PubMed] [Google Scholar]
- 20.Kindler S, Müller R, Chung WJ, Garner CC. Molecular characterization of dendritically localized transcripts encoding MAP2. Mol Brain Res. 1996;36:63–69. doi: 10.1016/0169-328x(95)00237-m. [DOI] [PubMed] [Google Scholar]
- 21.Kindler S, Mohr E, Richter D. Quo vadis: extrasomatic targeting of neuronal mRNAs. Mol Cell Endocrinol. 1997;128:7–10. doi: 10.1016/s0303-7207(97)04015-x. [DOI] [PubMed] [Google Scholar]
- 22.Kleiman R, Banker G, Steward O. Differential subcellular localization of particular mRNAs in hippocampal neurons in culture. Neuron. 1990;5:821–830. doi: 10.1016/0896-6273(90)90341-c. [DOI] [PubMed] [Google Scholar]
- 23.Kleiman R, Banker G, Steward O. Subcellular distribution of rRNA and poly(A) RNA in hippocampal neurons in culture. Brain Res Mol Brain Res. 1993;20:305–312. doi: 10.1016/0169-328x(93)90057-v. [DOI] [PubMed] [Google Scholar]
- 24.Knowles RB, Sabry JH, Martone ME, Deerinck TJ, Ellisman MH, Bassell GJ, Kosik KS. Translocation of RNA granules in living neurons. J Neurosci. 1996;16:7812–7820. doi: 10.1523/JNEUROSCI.16-24-07812.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kuhl D, Skehel P. Dendritic localization of mRNAs. Curr Opin Neurobiol. 1998;8:600–606. doi: 10.1016/s0959-4388(98)80087-1. [DOI] [PubMed] [Google Scholar]
- 26.Landry CF, Watson JB, Kashima T, Campagnoni AT. Cellular influences on RNA sorting in neurons and glia: an in situ hybridization histochemical study. Brain Res Mol Brain Res. 1994;27:1–11. doi: 10.1016/0169-328x(94)90178-3. [DOI] [PubMed] [Google Scholar]
- 27.Link W, Konietzko U, Kauselmann G, Krug M, Schwanke B, Frey U, Kuhl D. Somatodendritic expression of an immediate early gene is regulated by synaptic activity. Proc Natl Acad Sci USA. 1995;92:5734–5738. doi: 10.1073/pnas.92.12.5734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lyford GL, Yamagata K, Kaufmann WE, Barnes CA, Sanders LK, Copeland NG, Gilbert DJ, Jenkins NA, Lanahan AA, Worley PF. Arc, a growth factor and activity-regulated gene, encodes a novel cytoskeleton-associated protein that is enriched in neuronal dendrites. Neuron. 1995;14:433–445. doi: 10.1016/0896-6273(95)90299-6. [DOI] [PubMed] [Google Scholar]
- 29.Macdonald PM. Bicoid mRNA localization signal: phylogenetic conservation of function and RNA secondary structure. Development. 1990;110:161–171. doi: 10.1242/dev.110.1.161. [DOI] [PubMed] [Google Scholar]
- 30.Marsden KM, Doll T, Ferralli J, Botteri F, Matus A. Transgenic expression of embryonic MAP2 in adult mouse brain: implications for neuronal polarization. J Neurosci. 1996;16:3265–3273. doi: 10.1523/JNEUROSCI.16-10-03265.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mayford M, Baranes D, Podsypanina K, Kandel ER. The 3′ untranslated region of CaMKII alpha is a cis-acting signal for the localization and translation of mRNA in dendrites. Proc Natl Acad Sci USA. 1996;93:13250–13255. doi: 10.1073/pnas.93.23.13250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Miyashiro K, Dichter M, Eberwine J. On the nature and differential distribution of mRNAs in hippocampal neurites: implications for neuronal function. Proc Natl Acad Sci USA. 1994;91:10800–10804. doi: 10.1073/pnas.91.23.10800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mohr E, Morris JF, Richter D. Differential subcellular mRNA targeting: deletion of a single nucleotide prevents the transport to axons but not to dendrites of rat hypothalamic magnocellular neurons. Proc Natl Acad Sci USA. 1995;92:4377–4381. doi: 10.1073/pnas.92.10.4377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Muslimov IA, Santi E, Homel P, Perini S, Higgins D, Tiedge H. RNA transport in dendrites: a cis-acting targeting element is contained within neuronal BC1 RNA. J Neurosci. 1997;17:4722–4733. doi: 10.1523/JNEUROSCI.17-12-04722.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Prakash N, Fehr S, Mohr E, Richter D. Dendritic localization of rat vasopressin mRNA: ultrastructural analysis and mapping of targeting elements. Eur J Neurosci. 1997;9:523–532. doi: 10.1111/j.1460-9568.1997.tb01629.x. [DOI] [PubMed] [Google Scholar]
- 36.Rao A, Steward O. Evaluation of RNAs present in synaptodendrosomes: dendritic, glial, and neuronal cell body contribution. J Neurochem. 1993;61:835–844. doi: 10.1111/j.1471-4159.1993.tb03594.x. [DOI] [PubMed] [Google Scholar]
- 37.Rehbein M, Hillers M, Mohr E, Ivell R, Morley S, Schmale H, Richter D. The neurohypophyseal hormones vasopressin and oxytocin. Precursor structure, synthesis and regulation. Biol Chem. 1986;367:695–704. doi: 10.1515/bchm3.1986.367.2.695. [DOI] [PubMed] [Google Scholar]
- 38.Sambrook J, Fritsch EF, Maniatis T. Molecular cloning: a laboratory manual. Cold Spring Harbor Laboratory; Cold Spring Harbor, NY: 1989. [Google Scholar]
- 39.St. Johnston D. The intracellular localization of messenger RNAs. Cell. 1995;81:161–170. doi: 10.1016/0092-8674(95)90324-0. [DOI] [PubMed] [Google Scholar]
- 40.Steward O. Dendrites as compartments for macromolecular synthesis. Proc Natl Acad Sci USA. 1994;91:10766–10768. doi: 10.1073/pnas.91.23.10766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Steward O. mRNA localization in neurons: a multipurpose mechanism? Neuron. 1997;18:9–12. doi: 10.1016/s0896-6273(01)80041-6. [DOI] [PubMed] [Google Scholar]
- 42.Steward O, Levy WB. Preferential localization of polyribosomes under the base of dendritic spines in granule cells of the dentate gyrus. J Neurosci. 1982;2:284–291. doi: 10.1523/JNEUROSCI.02-03-00284.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Steward O, Reeves TM. Protein-synthetic machinery beneath postsynaptic sites on CNS neurons: association between polyribosomes and other organelles at the synaptic site. J Neurosci. 1988;8:176–184. doi: 10.1523/JNEUROSCI.08-01-00176.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Steward O, Wallace CS, Lyford GL, Worley PF. Synaptic activation causes the mRNA for the IEG Arc to localize selectively near activated postsynaptic sites on dendrites. Neuron. 1998;21:741–751. doi: 10.1016/s0896-6273(00)80591-7. [DOI] [PubMed] [Google Scholar]
- 45.Tiedge H, Brosius J. Translational machinery in dendrites of hippocampal neurons in culture. J Neurosci. 1996;16:7171–7181. doi: 10.1523/JNEUROSCI.16-22-07171.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tiedge H, Fremeau RT, Jr, Weinstock PH, Aranchio O, Brosius J. Dendritic localization of neural BC1 RNA. Proc Natl Acad Sci USA. 1991;88:2093–2097. doi: 10.1073/pnas.88.6.2093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Tiedge H, Bloom FE, Richter D. RNA, whither goest thou? Science. 1999;283:186–187. doi: 10.1126/science.283.5399.186. [DOI] [PubMed] [Google Scholar]
- 48.Torre ER, Steward O. Demonstration of local protein synthesis within dendrites using a new cell culture system that permits the isolation of living axons and dendrites from their cell bodies. J Neurosci. 1992;12:762–772. doi: 10.1523/JNEUROSCI.12-03-00762.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wallace CS, Lyford GL, Worley PF, Steward O. Differential intracellular sorting of immediate early gene mRNAs depends on signals in the mRNA sequence. J Neurosci. 1998;18:26–35. doi: 10.1523/JNEUROSCI.18-01-00026.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zuker M. On finding all suboptimal foldings of an RNA molecule. Science. 1989;244:48–52. doi: 10.1126/science.2468181. [DOI] [PubMed] [Google Scholar]