Abstract
In the tumor microenvironment, various stromal and immune cells accumulate and interact with cancer cells to contribute to tumor progression. Among stromal players, nerves have recently been recognized as key regulators of tumor growth. More neurotransmitters, such as catecholamines and acetylcholine (ACh), are present in tumors, as the cells that secrete neurotransmitters accumulate by the release of neurotrophic factors from cancer cells. In this short review, we focus on the role of nerve signaling in gastrointestinal (GI) cancers. Given that muscarinic acetylcholine receptor signaling seems to be a dominant regulator of GI stem cells and cancers, we review the function and mechanism of the muscarinic ACh pathway as a regulator of GI cancer progression. Accumulating evidence suggests that ACh, which is secreted from nerves and tuft cells, stimulates GI epithelial stem cells and contributes to cancer progression via muscarinic receptors.
Keywords: muscarinic acetylcholine receptor, tuft cell, gastric cancer, colon cancer
1. Interaction between Nerves and Cancers
There has been accumulating evidence on the neural regulation of tissue stem cells and the promoting role of nerves and neurotransmitters in cancer initiation and progression [1,2,3,4,5,6,7,8,9]. For instance, Brownell et al. reported that a nerve-dependent microenvironment creates a molecularly and phenotypically distinct population of hair follicle stem cells via the Sonic hedgehog (SHH) pathway [1]. Hanoun et al. reported that the sympathetic nervous system promotes the infiltration of leukemic cells into bone marrow in an MLL-AF9 AML model [2]. Katayama et al. reported that hematopoietic stem and progenitor cells in the bone marrow are regulated by the sympathetic nervous system, which attracts hematopoietic stem cells to their niche [3]. Magnon et al. reported that the autonomic nerve fibers, both adrenergic and cholinergic, in the prostate gland regulate prostate cancer development and dissemination in mouse models [4]. The authors later revealed that during this process, adrenergic signals regulate angiogenesis in prostate cancer tissues via metabolic changes in endothelial cells [10,11]. Mendez-Ferrer et al. reported that Nestin-expressing mesenchymal stem cells (MSCs) constitute an essential hematopoietic stem cell niche via adrenergic receptor signaling [5]. Peterson et al. reported that Gli1-expressing progenitors within mechanosensory touch dome epithelia are activated by sensory nerves via Hedgehog signaling in normal touch domes and contribute to tumorigenesis, while denervation attenuates these tumors [6]. Stopczynski et al. reported that neurotrophic factors and sensory nerves are increased during pancreatic cancer development, and metastatic tumor cells could be found along with the celiac and sensory ganglia in the spinal cord [7]. Furthermore, Renz et al. demonstrated that adrenergic signals promote pancreatic cancer progression, while cholinergic signals inhibit pancreatic cancer progression [12,13]. Venkatesh et al. reported that the synaptic protein neuroligin-3 (NLGN3) activates the PI3K-mTOR pathway and induces the feedforward expression of NLGN3 in glioma cells, and NLGN3 promotes robust high-grade glioma cell proliferation [8]. Zhao et al. reported that the vagus nerve contributes to gastric tumorigenesis via muscarinic acetylcholine receptor 3-mediated Wnt signaling in the gastric stem cells [9]. Thus, nerves and neuronal signals widely regulate cancer progression within the tumor microenvironment.
Neurite outgrowth from nerves is caused by the neurotrophic factors secreted from cancer cells. On the other hand, nerves, which can be adrenergic, cholinergic, or of sensory origin, release various neurotransmitters from their endings and stimulate stromal cells, immune cells, and cancer cells, resulting in the promotion of cancer initiation, progression, and metastasis [14]. Therefore, there is a bidirectional interaction between nerves, cancer cells, and other stromal cells, and nerve signaling plays a central role in the complex tumor microenvironment.
2. Muscarinic Acetylcholine (ACh) Receptors
Muscarinic ACh receptors are members of the G protein-coupled receptors (GPCRs) that activate 5′-phosphate (G) proteins. GPCRs regulate a variety of biological processes by modulating the activation of adenylyl cyclase, the turnover of phosphatidylinositol lipid, and the status of ion channels [15,16,17]. Like other GPCRs, muscarinic ACh receptors have seven transmembrane helical domains that are connected by three extracellular and three intracellular loops. To date, five muscarinic ACh receptor subtypes have been identified and termed as CHRM1–5. These receptors regulate various cellular functions through different signaling pathways [17,18,19]. In general, CHRM1, CHRM3, and CHRM5 activate the phospholipid turnover and change the cellular calcium concentration. CHRM2 and CHRM4 inhibit the activity of adenylyl cyclase and reduce levels of cyclic adenosine monophosphate (cAMP). Muscarinic ACh receptors are widely expressed throughout the body, including in the gut, brain, eye, heart, vasculature, etc. [20,21,22,23,24,25,26,27,28]. In particular, CHRM1 and CHRM3 are robustly expressed in the gastrointestinal (GI) tract. In the stomach, ACh is secreted from vagus nerve endings, and gastric acid secretion from parietal cells is caused by CHRM3 [29,30,31,32], while pepsinogen secretion from gastric chief cells is promoted by CHRM1 and CHRM3 [33,34,35,36]. In addition, a low expression of CHRM5 has been reported in the gastric epithelium [9,37]. CHRM1 and CHRM3 appear to be expressed in normal colonic epithelia as well [17,38]. The influence of muscarinic receptors is highly diversified, depending on the cellular environment and proteome [39].
3. ACh Signaling and Gastric Cancer
In a recent study involving three mouse models of gastric cancer (genetically engineered, chemically induced, and Helicobacter pylori-induced), surgical vagotomy significantly attenuated gastric tumorigenesis. In addition, pharmacological denervation using botulinum toxin A or a blockade of muscarinic acetylcholine receptor 3 (M3R) inhibited tumorigenesis [9,40]. Interestingly, Lgr5+ gastric antral stem cells highly express Chrm3, the gene encoding M3R, suggesting that Lgr5+ stem cell function is modulated by M3R signaling [9]. Indeed, denervation inhibits Wnt signaling in gastric stem cells and suppresses stem cell expansion during carcinogenesis. In humans, tumor stage is correlated with nerve density in gastric cancer tissues, whereas vagotomy reduces the risk of gastric cancer recurrence [9].
Like other GPCRs, the activation of M3R leads to a variety of biochemical and electrophysiological responses, but the resulting physiological effects depend on the cell type and/or tissue type. M3R has been suggested to activate mitogen-activated protein kinase (MAPK) [41,42,43,44], Akt [25,43,45], or ras homolog family member A (RhoA) [46,47], thereby contributing to tumor growth in various cancers [48] (Figure 1). Subsequent studies have shown that M3R signaling promotes cellular proliferation and inhibits apoptosis via the epidermal growth factor receptor (EGFR) and Akt pathways in human gastric cancer cell lines and xenografts [49,50].
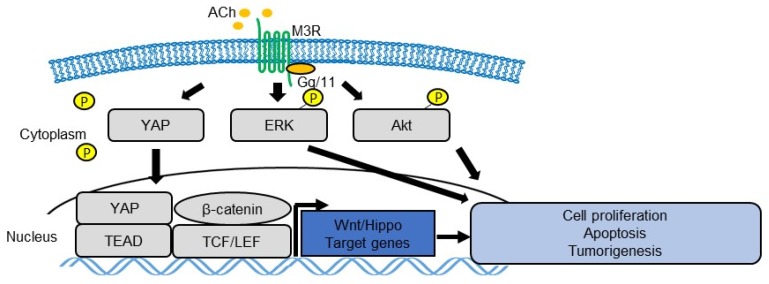
Figure 1.
Schema of ACh-muscarinic acetylcholine receptor 3 (M3R) signaling pathway in gastrointestinal (GI) tumor cells. ACh-M3R signaling regulates cell proliferation, survival, and tumorigenesis by activating various signaling pathways, such as mitogen-activated protein kinase (MAPK), Akt, yes-associated protein (YAP), and Wnt.
The yes-associated protein (YAP), a downstream effector of the Hippo pathway, regulates various cellular functions, such as proliferation, survival, stemness, and pluripotency [51,52]. In the Apc-dependent gastrointestinal tumors, YAP is highly upregulated and activated, and YAP activation appears to be required for the growth of Apc/β-catenin-dependent tumors [53,54,55]. Indeed, YAP regulates tissue regeneration and tumorigenesis in multiple organs, including the stomach and intestines, by activating tissue stem cells [56,57,58]. It was also suggested that types of GPCRs that activate G12/13, Gq/11, or Gi/o can activate YAP by suppressing its phosphorylation, whereas types of GPCRs that mainly activate Gs signaling can phosphorylate and, therefore, inhibit the YAP pathway [59]. Zhou et al. reported that SRY-Box9 (SOX9), which is known to be upregulated in gastric tissues during carcinogenesis [60], regulates the expression and phosphorylation of YAP in gastric cancer cell lines [61]. They also reported that SOX9 enhanced the proliferation, invasion, and migration of gastric cancer cells, and SOX9 may enhance the epithelial–mesenchymal transition (EMT) in gastric cancer cells via the Hippo-YAP pathway [61], supporting the notion that the Hippo-YAP pathway plays a critical role in gastric cancer progression. Similarly, our group demonstrated that M3R activates YAP signaling in gastric cancer cells, similar to the actions of other Gq/11 family receptors, and regulates the subsequent activation of Wnt in gastric tumors [48]. Thus, M3R and YAP could be a potential therapeutic target of nerve-dependent cancer development [51,62] (Figure 1).
4. Tuft Cells as a Source of ACh
Results of studies involving vagotomy have suggested that the vagus nerve is the main source of ACh secretion in the GI tract, but also that an alternative source of ACh exists in the GI epithelium. It has been reported that differentiated tuft cells, which commonly express doublecortin like kinase 1 (Dclk1), also express choline acetyltransferase (ChAT) and act as an epithelial source of ACh [48,63]. During carcinogenesis, tuft cells expand earlier and nerves expand later within cancerous tissues, resulting in the increase in ACh concentration in tissues. In particular, tuft cells expand dramatically in inflammation-associated cancer models and contribute to early cancer growth and the remodeling of the peritumoral neural microenvironment [48]. Furthermore, mouse models suggest that cholinergic stimulation of the gastric epithelium, in part, through ACh secretion from tuft cells, induces nerve growth factor (NGF) expression. NGF overexpression in the gastric epithelium, in turn, expands axonal growth from nerves in the lamina propria, and eventually promotes carcinogenesis by activating gastric stem cells [48]. Indeed, the ablation of Dclk1+ tuft cells and blockade of NGF/Trk signaling inhibit epithelial regeneration and tumorigenesis in an M3R-dependent manner, highlighting the importance of tuft cells as a component of the cancer niche. This feedforward ACh–NGF axis offers a compelling target for tumor treatment and prevention [48] (Figure 2).
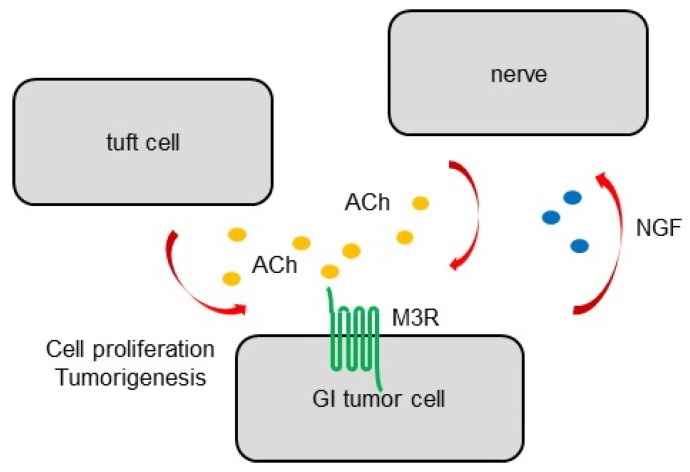
Figure 2.
Schema of GI cancer cells and ACh sources. The nerve is the main source of ACh secretion. Dclk1+ tuft cells act as an alternative epithelial source of ACh. Nerves and tuft cells in the stomach and intestines expand at discrete times during carcinogenesis, leading to an increased ACh concentration in cancerous tissues. Cholinergic stimulation of the gastric epithelium induces nerve growth factor (NGF) expression, and, in turn, NGF overexpression in the gastric epithelium expands enteric nerves and promotes carcinogenesis.
5. ACh Signaling and Colon Cancer
Evidence suggests that ACh-M3R signaling also plays a role in colonic carcinogenesis [64]. In ApcMin mice carrying the Apc gene mutation, the simultaneous knockout of the Chrm3 gene leads to the reduction in intestinal tumor load. β-catenin nuclear staining is attenuated in ApcMin/+ Chrm3−/− mice compared to in ApcMin/+ Chrm3+/+ mice with tumors, suggesting a similar regulatory mechanism of Wnt activation by M3R signaling [65]. Another study demonstrated that M3R-deficient mice have an attenuated cell proliferation, tumor number, and tumor size in chemically induced colon tumor models [38]. In addition, treatment with bethanechol, a muscarinic agonist, increases colonic proliferation by upregulating selected matrix metalloproteinase (Mmp) genes and Wnt target genes such as Myc and cyclin D1 [66]. Collectively, these findings support important roles for M3R expression and activation in the progression of colon neoplasia [67]. M3R and its downstream signaling pathway may be a promising therapeutic target of colon cancer [68].
While the relationship between M3R and YAP signaling has been suggested in gastric cancers [48], it remains undetermined whether such a clear connection between these two pathways also exists in intestinal cancers. Several studies have suggested that YAP signaling generally promotes epithelial regeneration and tumor development in intestines, primarily by stimulating tissue stem cells and progenitors [56,69,70]. The YAP pathway also plays a role in the regulation of colonic cell death/apoptosis, invasion, or metastasis [71,72]. Nevertheless, given that the opposite effects by YAP signaling on intestinal tumor development have been reported [62,73], further studies would be required to understand the precise function of this pathway in nerve-dependent tumorigenesis.
During inflammation-associated colon cancer development, vagus nerves also mediate anti-inflammatory reflux in the spleen [74]. Vagus stimuli promote the secretion of trefoil factor 2 (TFF2), an anti-inflammatory peptide, from memory T cells, which leads to the suppression of myeloid cells and the alteration of the inflammatory microenvironment in colonic tumors. Myeloid cells are also known as a source of Wnt ligands in the intestines and contribute to epithelial regeneration [75]. Therefore, vagus nerve signaling and ACh signaling are involved in colonic carcinogenesis in a complicated manner.
As demonstrated in the stomach, Dclk1+ tuft cells contribute to colonic regeneration and homeostasis, at least, in part, through ACh/M3R signaling. The ablation of Dclk1+ tuft cells in the colon leads to a significant reduction in epithelial proliferation, and exaggerated epithelial damage, after dextran sodium sulfate (DSS)-induced colitis [76]. In contrast, the overexpression of NGF and subsequent increased nerve signaling promoted epithelial repair following colonic injury, whereas M3R knockout in the colonic epithelium worsened the outcome, in a DSS-induced colitis model [48]. Interestingly, the conditional knockdown of Apc in Dclk1+ tuft cells is insufficient to drive colonic carcinogenesis under normal conditions; however, DSS-induced colitis induces the development of poorly differentiated colonic adenocarcinoma in mice specifically lacking the Apc gene in Dclk1+ tuft cells [76], suggesting that Dclk1+ tuft cells also act as cancer-initiating cells under specific conditions. In addition, in vitro co-culture experiments showed that nerves fail to support organoid growth in the absence of Dclk1+ tuft cells, suggesting that Dclk1+ cells are involved in the integration of neuronal signals in the epithelium [76]. More recently, Goto et al. identified interleukin 17 receptor B (IL17RB) as a cell-specific marker of tuft cells, and showed that IL17RB regulates tuft cell-derived cancer stem cell function and could be a therapeutic target [77].
6. Conclusions
M3R signaling regulates cell proliferation, survival, and tumorigenesis by activating various signaling pathways, such as MAPK, Akt, YAP, Wnt, and NGF, in GI cancers. In addition to nerves, tuft cells serve as sources of ACh in the GI tract and contribute to regeneration and homeostasis. Components and involved signaling pathways in nerve–tumor interactions may be a promising target for the treatment of cancers of the GI tract.
Funding
Y.H. is supported by the KAKENHI Grant-in-Aid for Scientific Research, 17K09347 and 17H05081, P-CREATE from AMED, the Pharmacological Research Foundation, the research grant of Bristol-Myers Squibb, the Kowa Life Science Foundation, the Senshin Medical Research Foundation, the Yokoyama Clinical Pharmacological Research Foundation, the Kanae Foundation of the Promotion of Medical Science, the Inoue Science Research Award, the Takeda Science Foundation Visionary Research Grant, the Princess Takamatsu Cancer Research Fund, and the Advanced Research and Development Programs for Medical Innovation (PRIME). M.K. is supported by a research grant from the Institute for Adult Diseases, Asahi Life Foundation.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- 1.Brownell I., Guevara E., Bai C.B., Loomis C.A., Joyner A.L. Nerve-derived sonic hedgehog defines a niche for hair follicle stem cells capable of becoming epidermal stem cells. Cell Stem Cell. 2011;8:552–565. doi: 10.1016/j.stem.2011.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hanoun M., Zhang D., Mizoguchi T., Pinho S., Pierce H., Kunisaki Y., Lacombe J., Armstrong S.A., Duhrsen U., Frenette P.S. Acute myelogenous leukemia-induced sympathetic neuropathy promotes malignancy in an altered hematopoietic stem cell niche. Cell Stem Cell. 2014;15:365–375. doi: 10.1016/j.stem.2014.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Katayama Y., Battista M., Kao W.M., Hidalgo A., Peired A.J., Thomas S.A., Frenette P.S. Signals from the sympathetic nervous system regulate hematopoietic stem cell egress from bone marrow. Cell. 2006;124:407–421. doi: 10.1016/j.cell.2005.10.041. [DOI] [PubMed] [Google Scholar]
- 4.Magnon C., Hall S.J., Lin J., Xue X., Gerber L., Freedland S.J., Frenette P.S. Autonomic nerve development contributes to prostate cancer progression. Science. 2013;341:1236361. doi: 10.1126/science.1236361. [DOI] [PubMed] [Google Scholar]
- 5.Mendez-Ferrer S., Michurina T.V., Ferraro F., Mazloom A.R., Macarthur B.D., Lira S.A., Scadden D.T., Ma’ayan A., Enikolopov G.N., Frenette P.S. Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature. 2010;466:829–834. doi: 10.1038/nature09262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Peterson S.C., Eberl M., Vagnozzi A.N., Belkadi A., Veniaminova N.A., Verhaegen M.E., Bichakjian C.K., Ward N.L., Dlugosz A.A., Wong S.Y. Basal cell carcinoma preferentially arises from stem cells within hair follicle and mechanosensory niches. Cell Stem Cell. 2015;16:400–412. doi: 10.1016/j.stem.2015.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stopczynski R.E., Normolle D.P., Hartman D.J., Ying H., DeBerry J.J., Bielefeldt K., Rhim A.D., DePinho R.A., Albers K.M., Davis B.M. Neuroplastic changes occur early in the development of pancreatic ductal adenocarcinoma. Cancer Res. 2014;74:1718–1727. doi: 10.1158/0008-5472.CAN-13-2050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Venkatesh H.S., Johung T.B., Caretti V., Noll A., Tang Y., Nagaraja S., Gibson E.M., Mount C.W., Polepalli J., Mitra S.S., et al. Neuronal Activity Promotes Glioma Growth through Neuroligin-3 Secretion. Cell. 2015;161:803–816. doi: 10.1016/j.cell.2015.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhao C.M., Hayakawa Y., Kodama Y., Muthupalani S., Westphalen C.B., Andersen G.T., Flatberg A., Johannessen H., Friedman R.A., Renz B.W., et al. Denervation suppresses gastric tumorigenesis. Sci. Transl. Med. 2014;6:250ra115. doi: 10.1126/scitranslmed.3009569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zahalka A.H., Arnal-Estape A., Maryanovich M., Nakahara F., Cruz C.D., Finley L.W.S., Frenette P.S. Adrenergic nerves activate an angio-metabolic switch in prostate cancer. Science. 2017;358:321–326. doi: 10.1126/science.aah5072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hayakawa Y., Wang T.C. Nerves switch on angiogenic metabolism. Science. 2017;358:305–306. doi: 10.1126/science.aaq0365. [DOI] [PubMed] [Google Scholar]
- 12.Renz B.W., Takahashi R., Tanaka T., Macchini M., Hayakawa Y., Dantes Z., Maurer H.C., Chen X., Jiang Z., Westphalen C.B., et al. β2 Adrenergic-Neurotrophin Feedforward Loop Promotes Pancreatic Cancer. Cancer Cell. 2018;33:75–90. doi: 10.1016/j.ccell.2017.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Renz B.W., Tanaka T., Sunagawa M., Takahashi R., Jiang Z., Macchini M., Dantes Z., Valenti G., White R.A., Middelhoff M.A., et al. Cholinergic Signaling via Muscarinic Receptors Directly and Indirectly Suppresses Pancreatic Tumorigenesis and Cancer Stemness. Cancer Discov. 2018;8:1458–1473. doi: 10.1158/2159-8290.CD-18-0046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Faulkner S., Jobling P., March B., Jiang C.C., Hondermarck H. Tumor Neurobiology and the War of Nerves in Cancer. Cancer Discov. 2019;9:702–710. doi: 10.1158/2159-8290.CD-18-1398. [DOI] [PubMed] [Google Scholar]
- 15.Gilman A.G. G proteins and dual control of adenylate cyclase. Cell. 1984;36:577–579. doi: 10.1016/0092-8674(84)90336-2. [DOI] [PubMed] [Google Scholar]
- 16.Malbon C.C. G proteins in development. Nat. Rev. Mol. Cell Biol. 2005;6:689–701. doi: 10.1038/nrm1716. [DOI] [PubMed] [Google Scholar]
- 17.Von Rosenvinge E.C., Raufman J.P. Muscarinic receptor signaling in colon cancer. Cancers. 2011;3:971–981. doi: 10.3390/cancers3010971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bonner T.I., Buckley N.J., Young A.C., Brann M.R. Identification of a family of muscarinic acetylcholine receptor genes. Science. 1987;237:527–532. doi: 10.1126/science.3037705. [DOI] [PubMed] [Google Scholar]
- 19.Kruse A.C., Li J., Hu J., Kobilka B.K., Wess J. Novel insights into M3 muscarinic acetylcholine receptor physiology and structure. J. Mol. Neurosci. 2014;53:316–323. doi: 10.1007/s12031-013-0127-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Abrams P., Andersson K.E., Buccafusco J.J., Chapple C., De Groat W.C., Fryer A.D., Kay G., Laties A., Nathanson N.M., Pasricha P.J., et al. Muscarinic receptors: Their distribution and function in body systems, and the implications for treating overactive bladder. Br. J. Pharm. 2006;148:565–578. doi: 10.1038/sj.bjp.0706780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pronin A.N., Wang Q., Slepak V.Z. Teaching an Old Drug New Tricks: Agonism, Antagonism, and Biased Signaling of Pilocarpine through M3 Muscarinic Acetylcholine Receptor. Mol. Pharm. 2017;92:601–612. doi: 10.1124/mol.117.109678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wang Z., Shi H., Wang H. Functional M3 muscarinic acetylcholine receptors in mammalian hearts. Br. J. Pharm. 2004;142:395–408. doi: 10.1038/sj.bjp.0705787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kovacevic I., Muller M., Kojonazarov B., Ehrke A., Randriamboavonjy V., Kohlstedt K., Hindemith T., Schermuly R.T., Fleming I., Hoffmeister M., et al. The F-BAR Protein NOSTRIN Dictates the Localization of the Muscarinic M3 Receptor and Regulates Cardiovascular Function. Circ. Res. 2015;117:460–469. doi: 10.1161/CIRCRESAHA.115.306187. [DOI] [PubMed] [Google Scholar]
- 24.Radu B.M., Osculati A.M.M., Suku E., Banciu A., Tsenov G., Merigo F., Di Chio M., Banciu D.D., Tognoli C., Kacer P., et al. All muscarinic acetylcholine receptors (M1–M5) are expressed in murine brain microvascular endothelium. Sci. Rep. 2017;7:5083. doi: 10.1038/s41598-017-05384-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.He Y., Lin G., Han D., Shi D., Liu T., Gao Y., Guan W., Cheng G. Aclidinium Bromide holds promising inhibitory effects in A549 lung cancer cells potentials by regulating PI3K/AKT signaling pathway. J. Buon. 2019;24:560–565. [PubMed] [Google Scholar]
- 26.Fryer A.D., Jacoby D.B. Muscarinic receptors and control of airway smooth muscle. Am. J. Respir. Crit. Care Med. 1998;158:S154–S160. doi: 10.1164/ajrccm.158.supplement_2.13tac120. [DOI] [PubMed] [Google Scholar]
- 27.Wei W., Wang M., Li Y., Meng Q., Tang Y., Lu H., Yu W., Cheng Q., Xu L., Jian S., et al. Muscarinic cholinergic signaling and overactive bladder-like symptoms associated with invasive bladder cancer. Oncol. Lett. 2018;16:775–784. doi: 10.3892/ol.2018.8715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kitazawa T., Hirama R., Masunaga K., Nakamura T., Asakawa K., Cao J., Teraoka H., Unno T., Komori S., Yamada M., et al. Muscarinic receptor subtypes involved in carbachol-induced contraction of mouse uterine smooth muscle. Naunyn. Schmiedebergs Arch. Pharm. 2008;377:503–513. doi: 10.1007/s00210-007-0223-1. [DOI] [PubMed] [Google Scholar]
- 29.Ecknauer R., Dial E., Thompson W.J., Johnson L.R., Rosenfeld G.C. Isolated rat gastric parietal cells: Cholinergic response and pharmacology. Life Sci. 1981;28:609–621. doi: 10.1016/0024-3205(81)90124-7. [DOI] [PubMed] [Google Scholar]
- 30.Soll A.H. Specific inhibition by prostaglandins E2 and I2 of histamine-stimulated [14C] aminopyrine accumulation and cyclic adenosine monophosphate generation by isolated canine parietal cells. J. Clin. Investig. 1980;65:1222–1229. doi: 10.1172/JCI109777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Aihara T., Fujishita T., Kanatani K., Furutani K., Nakamura E., Taketo M.M., Matsui M., Chen D., Okabe S. Impaired gastric secretion and lack of trophic responses to hypergastrinemia in M3 muscarinic receptor knockout mice. Gastroenterology. 2003;125:1774–1784. doi: 10.1053/j.gastro.2003.09.018. [DOI] [PubMed] [Google Scholar]
- 32.Kajimura M., Reuben M.A., Sachs G. The muscarinic receptor gene expressed in rabbit parietal cells is the m3 subtype. Gastroenterology. 1992;103:870–875. doi: 10.1016/0016-5085(92)90019-U. [DOI] [PubMed] [Google Scholar]
- 33.Raufman J.P., Sutliff V.E., Kasbekar D.K., Jensen R.T., Gardner J.D. Pepsinogen secretion from dispersed chief cells from guinea pig stomach. Am. J. Physiol. 1984;247:G95–G104. doi: 10.1152/ajpgi.1984.247.1.G95. [DOI] [PubMed] [Google Scholar]
- 34.Sutliff V.E., Rattan S., Gardner J.D., Jensen R.T. Characterization of cholinergic receptors mediating pepsinogen secretion from chief cells. Am. J. Physiol. 1989;257:G226–G234. doi: 10.1152/ajpgi.1989.257.2.G226. [DOI] [PubMed] [Google Scholar]
- 35.Xie G., Drachenberg C., Yamada M., Wess J., Raufman J.P. Cholinergic agonist-induced pepsinogen secretion from murine gastric chief cells is mediated by M1 and M3 muscarinic receptors. Am. J. Physiol. Gastrointest. Liver Physiol. 2005;289:G521–G529. doi: 10.1152/ajpgi.00105.2004. [DOI] [PubMed] [Google Scholar]
- 36.Tobin G., Giglio D., Lundgren O. Muscarinic receptor subtypes in the alimentary tract. J. Physiol. Pharm. 2009;60:3–21. [PubMed] [Google Scholar]
- 37.Aihara T., Nakamura Y., Taketo M.M., Matsui M., Okabe S. Cholinergically stimulated gastric acid secretion is mediated by M3 and M5 but not M1 muscarinic acetylcholine receptors in mice. Am. J. Physiol. Gastrointest. Liver Physiol. 2005;288:G1199–G1207. doi: 10.1152/ajpgi.00514.2004. [DOI] [PubMed] [Google Scholar]
- 38.Raufman J.P., Samimi R., Shah N., Khurana S., Shant J., Drachenberg C., Xie G., Wess J., Cheng K. Genetic ablation of M3 muscarinic receptors attenuates murine colon epithelial cell proliferation and neoplasia. Cancer Res. 2008;68:3573–3578. doi: 10.1158/0008-5472.CAN-07-6810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ockenga W., Kuhne S., Bocksberger S., Banning A., Tikkanen R. Non-neuronal functions of the m2 muscarinic acetylcholine receptor. Genes. 2013;4:171–197. doi: 10.3390/genes4020171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rabben H.L., Zhao C.M., Hayakawa Y., Wang T.C., Chen D. Vagotomy and Gastric Tumorigenesis. Curr. Neuropharmacol. 2016;14:967–972. doi: 10.2174/1570159X14666160121114854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kodaira M., Kajimura M., Takeuchi K., Lin S., Hanai H., Kaneko E. Functional muscarinic m3 receptor expressed in gastric cancer cells stimulates tyrosine phosphorylation and MAP kinase. J. Gastroenterol. 1999;34:163–171. doi: 10.1007/s005350050238. [DOI] [PubMed] [Google Scholar]
- 42.Said A.H., Hu S., Abutaleb A., Watkins T., Cheng K., Chahdi A., Kuppusamy P., Saxena N., Xie G., Raufman J.P. Interacting post-muscarinic receptor signaling pathways potentiate matrix metalloproteinase-1 expression and invasion of human colon cancer cells. Biochem. J. 2017;474:647–665. doi: 10.1042/BCJ20160704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Sales M.E., Espanol A.J., Salem A.R., Martinez P.P., Sanchez Y., Sanchez F. Role of muscarinic acetylcholine receptors in Breast Cancer. Design of metronomic chemotherapy. Curr. Clin. Pharm. 2018 doi: 10.2174/1574884714666181203095437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Felton J., Hu S., Raufman J.P. Targeting M3 Muscarinic Receptors for Colon Cancer Therapy. Curr. Mol. Pharm. 2018;11:184–190. doi: 10.2174/1874467211666180119115828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Song P., Sekhon H.S., Lu A., Arredondo J., Sauer D., Gravett C., Mark G.P., Grando S.A., Spindel E.R. M3 muscarinic receptor antagonists inhibit small cell lung carcinoma growth and mitogen-activated protein kinase phosphorylation induced by acetylcholine secretion. Cancer Res. 2007;67:3936–3944. doi: 10.1158/0008-5472.CAN-06-2484. [DOI] [PubMed] [Google Scholar]
- 46.Belo A., Cheng K., Chahdi A., Shant J., Xie G., Khurana S., Raufman J.P. Muscarinic receptor agonists stimulate human colon cancer cell migration and invasion. Am. J. Physiol. Gastrointest. Liver Physiol. 2011;300:G749–G760. doi: 10.1152/ajpgi.00306.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Kuol N., Stojanovska L., Apostolopoulos V., Nurgali K. Role of the nervous system in cancer metastasis. J. Exp. Clin. Cancer Res. 2018;37:5. doi: 10.1186/s13046-018-0674-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hayakawa Y., Sakitani K., Konishi M., Asfaha S., Niikura R., Tomita H., Renz B.W., Tailor Y., Macchini M., Middelhoff M., et al. Nerve Growth Factor Promotes Gastric Tumorigenesis through Aberrant Cholinergic Signaling. Cancer Cell. 2017;31:21–34. doi: 10.1016/j.ccell.2016.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wang L., Zhi X., Zhang Q., Wei S., Li Z., Zhou J., Jiang J., Zhu Y., Yang L., Xu H., et al. Muscarinic receptor M3 mediates cell proliferation induced by acetylcholine and contributes to apoptosis in gastric cancer. Tumour. Biol. 2016;37:2105–2117. doi: 10.1007/s13277-015-4011-0. [DOI] [PubMed] [Google Scholar]
- 50.Yu H., Xia H., Tang Q., Xu H., Wei G., Chen Y., Dai X., Gong Q., Bi F. Acetylcholine acts through M3 muscarinic receptor to activate the EGFR signaling and promotes gastric cancer cell proliferation. Sci. Rep. 2017;7:40802. doi: 10.1038/srep40802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Calses P.C., Crawford J.J., Lill J.R., Dey A. Hippo Pathway in Cancer: Aberrant Regulation and Therapeutic Opportunities. Trends Cancer. 2019;5:297–307. doi: 10.1016/j.trecan.2019.04.001. [DOI] [PubMed] [Google Scholar]
- 52.Chen Y.A., Lu C.Y., Cheng T.Y., Pan S.H., Chen H.F., Chang N.S. WW Domain-Containing Proteins YAP and TAZ in the Hippo Pathway as Key Regulators in Stemness Maintenance, Tissue Homeostasis, and Tumorigenesis. Front Oncol. 2019;9:60. doi: 10.3389/fonc.2019.00060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Azzolin L., Panciera T., Soligo S., Enzo E., Bicciato S., Dupont S., Bresolin S., Frasson C., Basso G., Guzzardo V., et al. YAP/TAZ incorporation in the β-catenin destruction complex orchestrates the Wnt response. Cell. 2014;158:157–170. doi: 10.1016/j.cell.2014.06.013. [DOI] [PubMed] [Google Scholar]
- 54.Cai J., Maitra A., Anders R.A., Taketo M.M., Pan D. β-Catenin destruction complex-independent regulation of Hippo-YAP signaling by APC in intestinal tumorigenesis. Genes Dev. 2015;29:1493–1506. doi: 10.1101/gad.264515.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rosenbluh J., Nijhawan D., Cox A.G., Li X., Neal J.T., Schafer E.J., Zack T.I., Wang X., Tsherniak A., Schinzel A.C., et al. β-Catenin-driven cancers require a YAP1 transcriptional complex for survival and tumorigenesis. Cell. 2012;151:1457–1473. doi: 10.1016/j.cell.2012.11.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Gregorieff A., Liu Y., Inanlou M.R., Khomchuk Y., Wrana J.L. Yap-dependent reprogramming of Lgr5+ stem cells drives intestinal regeneration and cancer. Nature. 2015;526:715–718. doi: 10.1038/nature15382. [DOI] [PubMed] [Google Scholar]
- 57.Imajo M., Ebisuya M., Nishida E. Dual role of YAP and TAZ in renewal of the intestinal epithelium. Nat. Cell Biol. 2015;17:7–19. doi: 10.1038/ncb3084. [DOI] [PubMed] [Google Scholar]
- 58.Jiao S., Wang H., Shi Z., Dong A., Zhang W., Song X., He F., Wang Y., Zhang Z., Wang W., et al. A peptide mimicking VGLL4 function acts as a YAP antagonist therapy against gastric cancer. Cancer Cell. 2014;25:166–180. doi: 10.1016/j.ccr.2014.01.010. [DOI] [PubMed] [Google Scholar]
- 59.Yu F.X., Zhao B., Panupinthu N., Jewell J.L., Lian I., Wang L.H., Zhao J., Yuan H., Tumaneng K., Li H., et al. Regulation of the Hippo-YAP pathway by G-protein-coupled receptor signaling. Cell. 2012;150:780–791. doi: 10.1016/j.cell.2012.06.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Serizawa T., Hirata Y., Hayakawa Y., Suzuki N., Sakitani K., Hikiba Y., Ihara S., Kinoshita H., Nakagawa H., Tateishi K., et al. Gastric Metaplasia Induced by Helicobacter pylori is Associated with Enhanced SOX9 Expression via Interleukin-1 Signaling. Infect. Immun. 2015;84:562–572. doi: 10.1128/IAI.01437-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhou H., Li G., Huang S., Feng Y., Zhou A. SOX9 promotes epithelial-mesenchymal transition via the Hippo-YAP signaling pathway in gastric carcinoma cells. Oncol. Lett. 2019;18:599–608. doi: 10.3892/ol.2019.10387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Barry E.R., Morikawa T., Butler B.L., Shrestha K., Rosa R.D.L., Yan K.S., Fuchs C.S., Magness S.T., Smits R., Ogino S., et al. Restriction of intestinal stem cell expansion and the regenerative response by YAP. Nature. 2013;493:106–110. doi: 10.1038/nature11693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Schutz B., Jurastow I., Bader S., Ringer C., Von Engelhardt J., Chubanov V., Gudermann T., Diener M., Kummer W., Krasteva-Christ G., et al. Chemical coding and chemosensory properties of cholinergic brush cells in the mouse gastrointestinal and biliary tract. Front Physiol. 2015;6:87. doi: 10.3389/fphys.2015.00087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cheng K., Xie G., Khurana S., Heath J., Drachenberg C.B., Timmons J., Shah N., Raufman J.P. Divergent effects of muscarinic receptor subtype gene ablation on murine colon tumorigenesis reveals association of M3R and zinc finger protein 277 expression in colon neoplasia. Mol. Cancer. 2014;13:77. doi: 10.1186/1476-4598-13-77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Raufman J.P., Shant J., Xie G., Cheng K., Gao X.M., Shiu B., Shah N., Drachenberg C.B., Heath J., Wess J., et al. Muscarinic receptor subtype-3 gene ablation and scopolamine butylbromide treatment attenuate small intestinal neoplasia in Apcmin+ mice. Carcinogenesis. 2011;32:1396–1402. doi: 10.1093/carcin/bgr118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Peng Z., Heath J., Drachenberg C., Raufman J.P., Xie G. Cholinergic muscarinic receptor activation augments murine intestinal epithelial cell proliferation and tumorigenesis. BMC Cancer. 2013;13:204. doi: 10.1186/1471-2407-13-204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Cheng K., Shang A.C., Drachenberg C.B., Zhan M., Raufman J.P. Differential expression of M3 muscarinic receptors in progressive colon neoplasia and metastasis. Oncotarget. 2017;8:21106–21114. doi: 10.18632/oncotarget.15500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Tolaymat M., Larabee S.M., Hu S., Xie G., Raufman J.P. The Role of M3 Muscarinic Receptor Ligand-Induced Kinase Signaling in Colon Cancer Progression. Cancers. 2019;11:308. doi: 10.3390/cancers11030308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Hayakawa Y., Tsuboi M., Asfaha S., Kinoshita H., Niikura R., Konishi M., Hata M., Oya Y., Kim W., Middelhoff M., et al. BHLHA15-Positive Secretory Precursor Cells Can Give Rise to Tumors in Intestine and Colon in Mice. Gastroenterology. 2019;156:1066–1081. doi: 10.1053/j.gastro.2018.11.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Yui S., Azzolin L., Maimets M., Pedersen M.T., Fordham R.P., Hansen S.L., Larsen H.L., Guiu J., Alves M.R.P., Rundsten C.F., et al. YAP/TAZ-Dependent Reprogramming of Colonic Epithelium Links ECM Remodeling to Tissue Regeneration. Cell Stem Cell. 2018;22:35–49. doi: 10.1016/j.stem.2017.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Blanchard T.G., Lapidus R., Banerjee V., Bafford A.C., Czinn S.J., Ahmed H., Banerjee A. Upregulation of RASSF1A in Colon Cancer by Suppression of Angiogenesis Signaling and Akt Activation. Cell. Physiol. Biochem. 2018;48:1259–1273. doi: 10.1159/000492012. [DOI] [PubMed] [Google Scholar]
- 72.Wang L., Shi S., Guo Z., Zhang X., Han S., Yang A., Wen W., Zhu Q. Overexpression of YAP and TAZ is an independent predictor of prognosis in colorectal cancer and related to the proliferation and metastasis of colon cancer cells. PLoS ONE. 2013;8:e65539. doi: 10.1371/journal.pone.0065539. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 73.Ou C., Sun Z., Li S., Li G., Li X., Ma J. Dual roles of yes-associated protein (YAP) in colorectal cancer. Oncotarget. 2017;8:75727–75741. doi: 10.18632/oncotarget.20155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Dubeykovskaya Z., Si Y., Chen X., Worthley D.L., Renz B.W., Urbanska A.M., Hayakawa Y., Xu T., Westphalen C.B., Dubeykovskiy A., et al. Neural innervation stimulates splenic TFF2 to arrest myeloid cell expansion and cancer. Nat. Commun. 2016;7:10517. doi: 10.1038/ncomms10517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Saha S., Aranda E., Hayakawa Y., Bhanja P., Atay S., Brodin N.P., Li J., Asfaha S., Liu L., Tailor Y., et al. Macrophage-derived extracellular vesicle-packaged WNTs rescue intestinal stem cells and enhance survival after radiation injury. Nat. Commun. 2016;7:13096. doi: 10.1038/ncomms13096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Westphalen C.B., Asfaha S., Hayakawa Y., Takemoto Y., Lukin D.J., Nuber A.H., Brandtner A., Setlik W., Remotti H., Muley A., et al. Long-lived intestinal tuft cells serve as colon cancer-initiating cells. J. Clin. Investig. 2014;124:1283–1295. doi: 10.1172/JCI73434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Goto N., Fukuda A., Yamaga Y., Yoshikawa T., Maruno T., Maekawa H., Inamoto S., Kawada K., Sakai Y., Miyoshi H., et al. Lineage tracing and targeting of IL17RB+ tuft cell-like human colorectal cancer stem cells. Proc. Natl. Acad. Sci. USA. 2019;116:12996–13005. doi: 10.1073/pnas.1900251116. [DOI] [PMC free article] [PubMed] [Google Scholar]