1.0. Introduction
Four decades following the earliest acquisition of magnetic resonance (MR) derived images of the brain, MR technology has developed into an essential diagnostic tool across a diverse range of clinical applications. Central to this evolution has been the development of human MR scanners with increasingly greater magnetic fields, most recently those operating at field strengths of 7 tesla (T) or more (up to 11.7T) [1]. These ultrahigh field MR scanners have the potential to permit neuroimaging with unprecedented detail, leading to better characterization of normal tissue and pathologic lesions and improved planning of treatment and monitoring of response. As of early 2019, at least 76 whole-body MRI systems of 7T or greater field strength have been installed around the world according to a database developed by Renzo Huber of the layer fMRI blog. The 7T Terra (Siemens Healthineers, Erlangen, Germany) system has become the first ultrahigh field system to receive 510(k) clearance for clinical imaging.
Signal-to-noise (SNR) and tissue contrast, two considerations central to the utility of MRI, both scale proportionally with field strength (see Table 1). Previous comparative studies across high (3T) and ultrahigh field MRI (7T and above) have empirically demonstrated a potentially supralinear relationship between field strength and SNR [2, 3]. A consequence of this greater SNR compared to conventional scanners is that images at ultrahigh field can typically be acquired with greater resolution and better differentiation of fine anatomical structures [4]. This greater sensitivity of ultrahigh field imaging may help to establish imaging of biomarkers for diseases like brain tumors, Alzheimer’s disease [5], neuropsychiatric disorders like depression, post-traumatic stress and schizophrenia and neurological disorders like epilepsy and multiple sclerosis. As a leading-edge technology ultrahigh field imaging is not without its technical challenges. Realizing the clinical potential of ultrahigh field neuroimaging requires addressing issues such as greater B0 and B1 inhomogeneity and specific absorption rates (SAR).
Table 1:
The relationship between various MRI/MRS/MRSI related parameters and field strength (B0)
| Imaging Parameter | Relationship to Field Strength | Reference |
|---|---|---|
| Signal-to-Noise Ratio (SNR) | SNR ∝ B0 | [6–8] |
| Spatial Resolution | Resolution ∝ SNR1/3 ∝ B01/3 | [9] |
| T1 | T1 = 1/(.35 + .64*B0−0.7) | [6] |
| T2* | T2* ∝ 1/(7 + 3.5*B0) | [6, 10, 11] |
| Apparent T2 | T2 ∝ 1/B0 | [10–13] |
| Spectral Separation | δ ∝ B0 | [14, 15] |
| Susceptibility | Δφ ∝ TE * B0 | [16] |
2.0. Parametric benefits of Ultrahigh field Imaging
Table 1 shows a list of the relationship between main field strength (B0) and various imaging-related parameters which may ultimately enjoy a benefit from scanning at ultrahigh field. While parameters such as higher SNR or greater spectral separation are generally beneficial across a range of imaging applications, other parameters such as greater susceptibility and faster relaxation times may be exploited towards a benefit or act as a limitation depending on the particular imaging sequence.
2.1. T1 and T2 weighted structural Imaging
The currency of greater SNR provided by higher field strength can be for other performance improvements including higher spatial resolution and faster scan time. Ultrahigh field MRI benefitting from greater tissue contrast and higher resolution has facilitated the visualization of finer structures and improved differentiation of lesions and other regions of interest from the surrounding tissue. These improvements have advanced post-processing towards more precise segmentation and volumetric quantification of smaller structures such as the subfields of the hippocampus [17–19] and the nuclei of the amygdala [20]. Other small features of clinical interest with the potential for improved characterization with ultrahigh field MRI include tissue microbleeds [21], polymicrogyria [22] and subcortical features [23, 24].
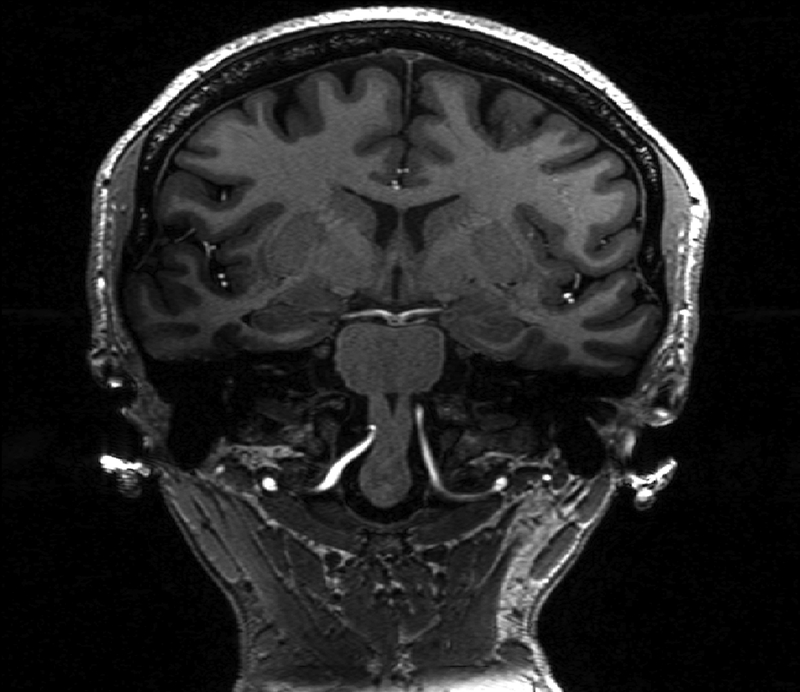
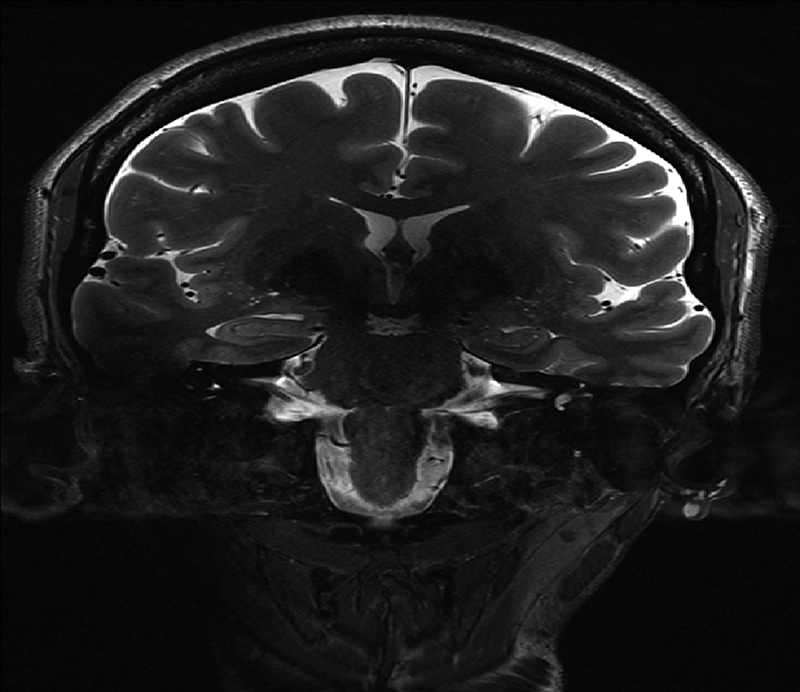
The SNR benefit of ultrahigh field may be counterbalanced in the context of the T2-weighted turbo spin echo (TSE) sequence, which relies on trains of 180° radio frequency (RF) pulses. The greater SAR at ultrahigh field may limit the echo train lengths (ETL) or minimum repetition time (TR) of these RF-intensive techniques, resulting in longer scan times. Figure 1 demonstrates a coronal-oblique T1-weighted MPRAGE and T2-weighted TSE acquisition showing an in-plane resolution of 0.4×0.4 mm with a slice thickness of 2 mm. Figure 2 shows a fast gray matter acquisition T1 inversion recovery (FGATIR) sequence employing an inversion pulse to null the signal contribution from white matter and improve anatomical contrast in deep gray matter structures. All images shown in this chapter were acquired using a Nova Medical 32-channel receive head coil on a Siemens 7T MAGNETOM MRI scanner.
Figure 1:
T1-weighted MPRAGE (right) and showing coronal-oblique section of a healthy volunteer. Spatial resolution of the T1-weighted MPRAGE is 0.7 mm3 isotropic.
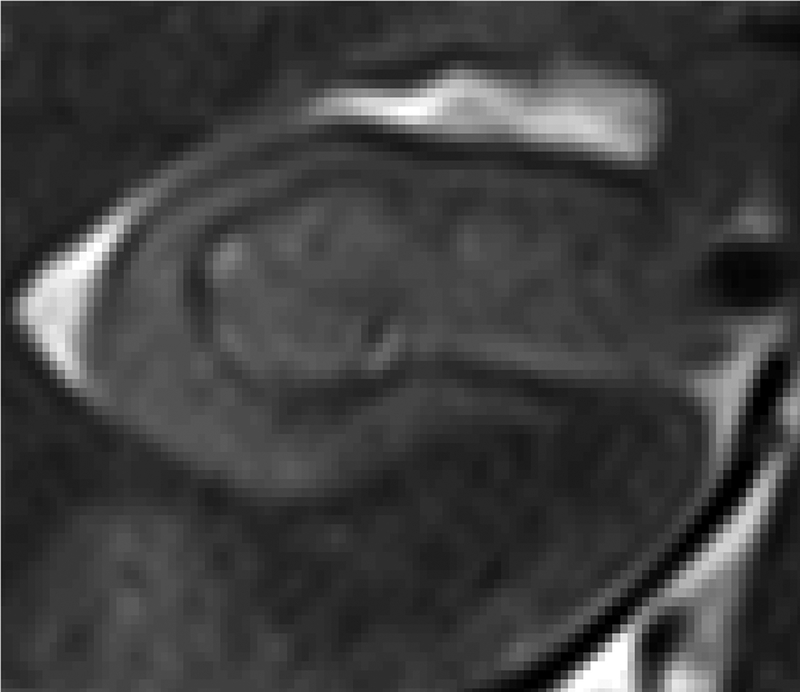
Figure 2:
Top: T2-weighted TSE (left) from the same volunteer as figure 1 with a spatial resolution of 0.4×0.4×2.0 mm3. Bottom: Zoomed-in inset of right hippocampus from T2-TSE.
2.2. Functional and Vascular Imaging
Contrast mechanisms based on magnetic susceptibility, including susceptibility weighted imaging (SWI), quantitative susceptibility mapping (QSM), blood oxygen level-dependent (BOLD) imaging and flow-dependent imaging such as time-of-flight (TOF) each benefit from the proportionally greater susceptibility effect at ultrahigh field. TOF imaging also benefits from longer T1 relaxation times at ultrahigh field, yielding a near-doubling of observed contrast-to-noise ratio (CNR) [25]. Likewise, the arterial spin labeling (ASL) technique benefits from both the higher SNR at higher field and the longer T1, with signal decay described as:
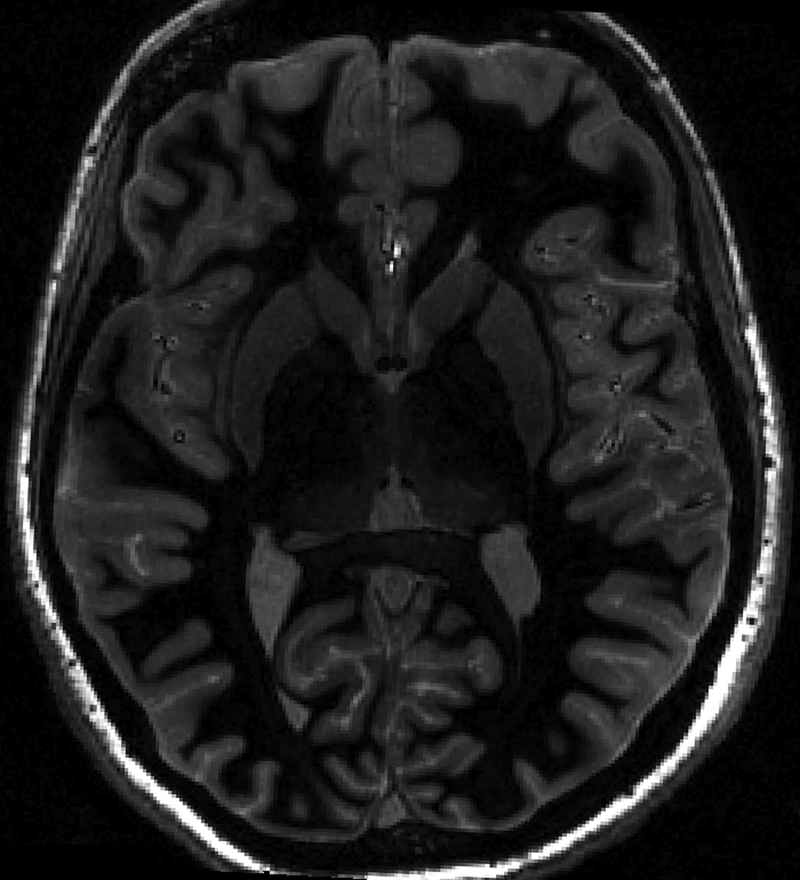
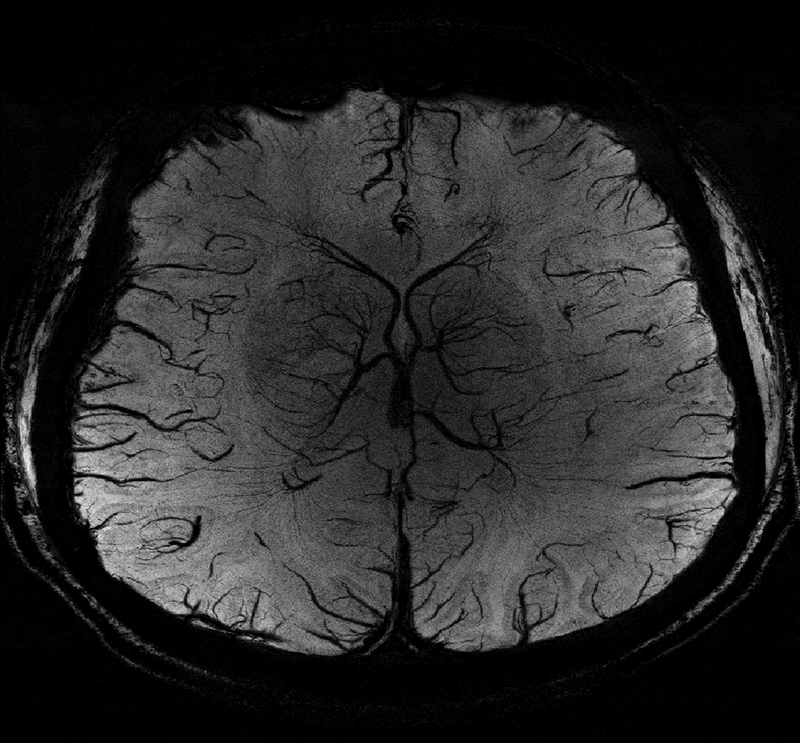
The SNR advantages at ultrahigh field could lead to reliable detection of smaller arteries than is achievable at lower fields, with potential applications in detection of occlusions in acute ischemic stroke as well as in neurosurgical planning. Figure 3 shows a high-resolution axial SWI image acquired from a healthy volunteer at 7T with 0.2 mm2 in-plane resolution and 1.5 mm slice thickness.
Figure 3 (right):
Minimum intensity projection (mip) of an axial susceptibility weighted imaging scan from a healthy volunteer with 0.2 mm2 in-plane resolution and 1.5 mm slice thickness.
Resting-state functional magnetic resonance imaging (fMRI) studies demonstrate sensitivity benefits from greater SNR at ultra-high field, which may be leveraged towards higher spatial resolution. A recent comparison study between 3T and 7T by Morris et al. [26] demonstrates up to a 300% improvement in SNR and resting-state functional connectivity coefficients at the higher field strength. This greater sensitivity may allow improved characterization of subtle neuropsychiatric disorders including major depressive disorder (MDD).
2.3. Diffusion Weighted Imaging
Although the chief contrast mechanism of diffusion weighted imaging (DWI) and diffusion tensor imaging (DTI) is modulated by the magnitude of the applied directional gradient, diffusion-based sequences still benefits from the higher SNR at ultrahigh field. As with anatomical imaging, these SNR gains can be leveraged towards smaller voxel sizes, and therefore higher resolution while maintaining adequate signal quality. DWI/DTI studies typically rely on an echo-planar (EP) readout for fast spatial encoding, which is prone to susceptibility artifacts at tissue interfaces, particularly at ultrahigh field, and may furthermore be limited in duration by T2* relaxation. Both limitations may be alleviated by a segmented EP readout method such as the one proposed by Heidemann et al. [27].
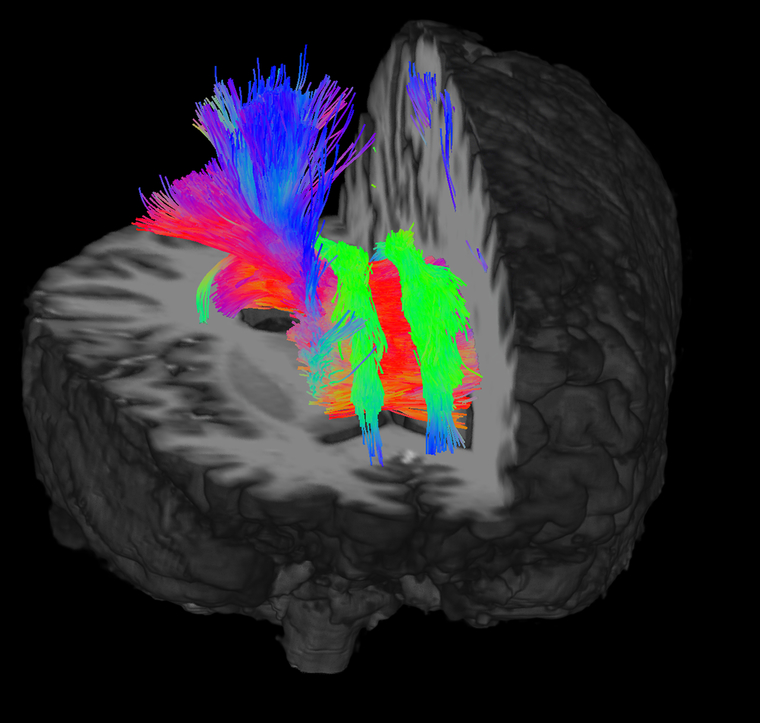
Higher resolution and SNR may also assist the accuracy of probabilistic tractography approaches popular in DTI post-processing to resolve crossing fibers and probe the connectomic properties of brain structures. Figure 4 demonstrates tractography from the whole brain generated from a 64-direction multi-band DTI sequence acquired at 7T with an isotropic voxel resolution of 1.05 mm3. Figure 5 shows tractography between the left and right hippocampus, amygdala and thalamus from a separate DTI study acquired with identical scan parameters at 7T. The left and right sides of the image show fiber tracts which pass through the hippocampus, amygdala and thalamus.
Figure 4:
3D map showing whole brain tractography. Data was acquired using a 64-direction DTI sequence acquired at 7T with 1.05 mm3 isotropic resolution and b = 1200s/mm2. [47, 51]. Image courtesy of John W. Rutland.
Figure 5:
Tractography of left and right hippocampus-amygdala-thalamus from 64-direction DTI sequence acquired at 7T with 1.05 mm3 isotropic resolution and b = 1200s/mm2. Shown are fiber tracts which pass through the hippocampus, amygdala and thalamus. Image courtesy of Stephanie SG Brown.
2.4. Spectroscopy & Spectroscopic Imaging
Magnetic Resonance Spectroscopy and Spectroscopic imaging (MRS/MRSI) [28, 29] and chemical exchange spectroscopy (CEST) [30] sequences derive a double benefit from scanning at ultrahigh field, with both proportionally greater SNR and chemical shift dispersion. These advantages result in the detection and quantification of metabolites with greater specificity and sensitivity than possible with equivalent sequences at lower fields [31–33]. Recent studies have attempted to leverage this improved performance to probe the role of low-concentration metabolites like gamma aminobutyric acid (GABA), glutamate (Glu) and glutathione (GSH) in neurological [34] and neuropsychiatric disorders [35, 36].
Gains in spectral separation at ultrahigh field may be partially mitigated by broader line-widths due to greater B0 inhomogeneity, yet these limitations can be overcome by advancements in B0 shimming algorithms and the implementation of higher order dedicated shim coils. Greater potential for water contamination due to B0-inhomogeneity also prompted the implementation of variable power and optimized relaxation delays (VAPOR) water suppression by Tkáć et al [28, 37]. MRSI sequences at ultrahigh field may benefit from adiabatic radio frequency (RF) pulses such as the semi-localized by adiabatic selective refocusing (semi-LASER) [38, 39] pulse sequence to reduce signal loss from B1 inhomogeneity. The typically-desirable greater spectral dispersion available at ultrahigh field may exacerbate chemical shift localization (CSL) errors endemic to most MRSI techniques, [40] yet may be alleviated by high spatial bandwidth technique such as the spatial-spectral adiabatic spectroscopic imaging (SASSI) sequence [40].
The two-dimensional (2D) correlation spectroscopy (COSY) technique benefits from the greater spectral dispersion of ultrahigh field along both its directly acquired (F2) dimension and the indirectly sampled (F1). This is not the case for the 2D J-resolved spectroscopy technique, as the J-coupling effects it measures are independent of the magnetic field.
2.5. Non-proton and multinuclear Imaging
While greater spectral dispersion may be beneficial to ultrahigh field imaging, it also contributes to greater B0 and B1 inhomogeneity for proton imaging applications. This is not so for non-proton applications whose gyromagnetic ratios (γ) tend to be smaller than the 42.58 MHz/T value for 1H. These include sodium (23Na) (11.26 MHz/T), carbon-13 (13C) (10.71) and phosphorous-31 (31P) (17.24) imaging. As an example, the resonance frequency of sodium at 7T is only 78.82 MHz, comparable to the dispersion of 1H at 1.5T (63.87 Hz). Furthermore, sodium imaging also receives SNR benefits from the longer T1 relaxation values at ultrahigh field. Sodium T1 relaxation rates for brain, blood, cartilage and muscle have been observed to be in the range between 10–55 ms [41], necessitating short TEs for most imaging applications. The tendency for longer T1 relaxation with increasing field strength, as shown in Table 1, makes the fast relaxation properties of sodium less of a limitation at ultrahigh field.
Because the MR-visible isotopes of those atomic nuclei occur at much lower natural concentrations than proton-containing molecules, their imaging applications tend to be relatively SNR-starved, and thus benefit greatly from the higher sensitivity of ultrahigh field. Non-proton imaging applications are limited by the availability of dedicated RF coils tuned to their particular resonance frequencies, particularly at ultrahigh field. Early development of non-proton ultrahigh field hardware was dominated by custom-built coils by dedicated RF hardware laboratories, but commercial availability of these coils has increased in recent years as manufacturers such as Nova Medical have entered the space.
3.0. Clinical Applications
The added resolution and signal quality of ultrahigh resolution imaging may be used to improve detection, characterization, treatment planning and monitoring for a range of clinical applications.
3.1. Brain Tumor
The greater sensitivity and spatial resolution of ultrahigh field imaging may help elucidate smaller or fainter lesions under contrast enhanced T1-weighted MRI, T2-weighted fluid-attenuated inversion recovery (FLAIR) imaging or proton density (PD) weighted imaging. The greater contrast and resolution offered by ultrahigh field may facilitate reliable detection of smaller or less well-differentiated lesions. Vessel imaging applications such as SWI or arterial imaging applications such as ASL or TOF may help to characterize tumor vasculature, detect microbleeds induced by radiation therapy [42], identify angiogenesis or identify major arteries near the tumor in the context of imaging-guided surgical planning.
Abnormal metabolism in brain tumors and the surrounding parenchyma may be non-invasively detected by MRSI. With greater SNR and spectral separation, MRSI at ultrahigh field may help in detection of the tumor-specific biomarkers of increasing interest in the era of personalized medicine. For example, specialized spectroscopy techniques including spectral editing and 2D MRS may enable reliable detection of the “oncometabolite” 2-hydroxyglutarate [43, 44], a highly-specific marker for mutation in isocitrate dehydrogenase (IDH1/IDH2, found in a majority of WHO Grade II/III gliomas). Other detected metabolic abnormalities may be markers for tumor activity including lactate, which is a product of anaerobic metabolism, and choline-containing metabolites, particularly phosphor-choline, which are markers for membrane turnover. A sensitive and reliable MRSI technique may be used to non-invasively characterize tumor both before and after treatment, such as differentiation of recurrent tumor from pseudo-progression or treatment necrosis.
3.2. Epilepsy
In focal epilepsy, high resolution structural MRI obtained at ultrahigh field may facilitate the detection of small epileptic lesions that are difficult to detect at lower field strengths [45]. Even in non-lesional epilepsy these high-resolution scans may reveal abnormalities in the cortex such as polymicrogyria or abnormalities in white matter such as dilated peri-ventricular spaces (PVSs), which may yield clues as to which cortical structures are pathologically involved [46, 47].
As deep-brain stimulation (DBS), particularly of the thalamus, has gained interest as a potential treatment strategy for non-lesional epilepsy, there is potential for ultrahigh field imaging to assist in the planning of electrode placement. High-resolution structural MRI and DTI may be able to elucidate small subcortical structures and probe their associated cortical connections to highlight potential targets for DBS.
3.3. Psychiatric Disorders
Higher SNR and spatial resolution of structural imaging may facilitate more reliable measurement of potential imaging biomarkers like cortical thickness and volume. Automatic segmentation packages like FreeSurfer and ASHS may leverage high resolution ultrahigh field imaging to more accurately parcellate fine structures such as the subfields of the hippocampus or the nuclei of the amygdala and thalamus. Higher resolution diffusion weighted imaging may also enable more reliable whole-brain tractography including resolution of crossing fibers to establish connectomic imaging markers for MDD, which may correlate with increasing severity of symptoms [48–51].
MRSI at ultrahigh field may more reliably detect gamma-aminobutyric acid (GABA), the brain’s chief parasympathetic neurotransmitter and glutathione (GSH), a marker for oxidative stress. Reductions in GSH and GABA concentration have been implicated in psychiatric disorders such as MDD and schizophrenia [35, 36, 52–54].
3.4. Alzheimer’s Disease
A principal goal in imaging applications of Alzheimer’s Disease (AD) is the direct or indirect detection of plaque-like pathology by “MR microscopy.” [55] Even in applications where this direct characterization may not be possible, high-resolution ultrahigh field MRI may help to identify subtle volumetric loss in cortical or subcortical structures [56] to gain a better understanding of the nature of volume losses and cognitive deficits associated with the disease. Various imaging modalities at ultrahigh field, including T2-weighted imaging, SWI and QSM, may be used to facilitate mapping or iron associated with amyloid disease [57]. MRS may also assist in the characterization of AD through the detection and quantification of brain metabolites, particularly N-acetyl aspartate and GABA [31].
3.5. Multiple Sclerosis (MS)
Ultrahigh field SWI and QSM applications have attempted to characterize multiple sclerosis (MS) lesions, by detecting the contributions of iron and myelin to their magnetic susceptibility [58]. Lesion subtype may have predictive power in characterizing physical and cognitive deficits in patients with MS [59]. The greater SNR and resolution of structural MRI at ultrahigh field may also help to reliably detect MS lesions too small or faint to detect at lower fields [60, 61]. Better identification may also improve the accuracy and reliability of automated segmentation techniques for detection and quantification of MS lesions [62]. The enhanced anatomical contrast at ultrahigh field may also help to detect MS lesions within the gray matter, where contrast is typically poorer [63].
3.6. Stroke
Signal quality of arterial imaging applications such as ASL and TOF is modulated by both the main magnetic field and the T1 relaxation time, and therefore enjoys a double benefit at ultrahigh field. This greater signal quality may lead to the improved characterization of the microvasculature and the identification of small occlusions. Comparative studies at ultrahigh field have demonstrated better identification of microbleeds [21] and microinfarcts [64].
4.0. Technical Challenges and Engineering Solutions
There are several physical, technical and clinical challenges involved in scanning at ultrahigh field, with the potential to affect the performance, sensitivity, reliability and safety characteristics of the acquired sequences. Various software and hardware based engineering solutions are in development to meet these challenges including coils with multiple transmit elements to better shape the excitation field or adiabatic RF pulses [45, 65] with greater insensitivity to B1 inhomogeneity.
4.1. Parametric limitations of Ultrahigh-field Imaging
4.2. B0 Inhomogeneity
Inhomogeneity of the main magnetic field (B0 inhomogeneity) scales proportionally with field strength, resulting in both spatial distortions and alterations in signal intensity and contrast. In MRS/MRSI applications, B0 inhomogeneity may manifest both as a loss of signal and a mis-positioned spectral shift. This shifting may complicate the suppression of competing water and lipid signal and broaden the observed line-widths of metabolite peaks. The resulting overlap with other peaks or water/lipid background may complicate reliable quantification of metabolite signal through MRS. In large voxels, B0 may contribute partial volume effects to the acquired signal and an overall loss in SNR.
4.3. B1 Inhomogeneity
Scanning at ultrahigh field can be subject to greater inhomogeneity of the applied radio frequency field (B1 inhomogeneity) resulting in varying signal intensity and tissue contrast in the acquired image. These artifacts are believed to be modulated by the dielectric properties of the scanned tissue, like conductivity and permittivity. For typical brain permittivity, half-wavelengths of the proton resonance are on the order of 10 cm, smaller than the cross-sectional size of the head. This can create standing wave patterns of constructive (bright) and destructive (dark) interference within the FOV. Furthermore, skin-depth effects and eddy currents opposing the applied B1 field can cause reduction in the effective B1 in the peripheral tissue.
Various types of adiabatic RF pulses, including hyperbolic secant pulses [70] and adiabatic Shinnar-Le Roux pulses [45, 71] have demonstrated reduction of B1-inhomogeneity effects. In the context of MRS/MRSI, the semi-LASER class of pulses is a commonly-used strategy for B1-insensitivity. Because adiabatic excitation and refocusing pulses tend to be higher bandwidth than their conventional counterparts, sequences employing these pulses may introduce additional SAR to the scans. This can be problematic as scanning at ultrahigh field is already subject to higher SAR independent of these pulses meaning their implementation may impose other limitations to the scan parameters, such as longer scan durations.
Parallel transmission (PTx) imaging with segmented readouts may be a hardware-based approach to the problem of B1 inhomogeneity [72]. Along with a B1 field mapping approach multiple independent coil transmit elements allow for more degrees of freedom in dealing with the complex interference patterns observed at ultrahigh field. PTx approaches have thus far been limited by safety concerns related to the complex SAR profiles from multiple transmit elements [73]. These interactions will need to be understood and modeled for the individual transmit arrays before ultrahigh field pulse sequences can fully take advantage of this emerging technology for clinical use.
4.4. Increased chemical shift localization artifacts
Because chemical shift dispersion is proportional to B0 field strength, chemical shift localization artifacts also tend to become proportionally more severe at higher field. These artifacts occur when the effective excitation volume for a given metabolite of interest is displaced from the nominal excitation volume in proportion to its chemical shift and are a major challenge in reliable MRSI. Acquisition techniques with high spatial bandwidths, such as the spatial-spectral pulses employed by the MR-SASSI technique [40], have demonstrated mitigation of chemical shift localization errors.
4.5. Specific Absorption Rate
RF deposition tends to scale with the square of the B0 field strength, with RF power requirements for 7T shown to be roughly 1.8–2.0 times higher than needed for 4T. FDA-designated SAR guidelines (3.2 W/kg over 10 or minutes for head imaging) pose a significant limitation at ultrahigh field, limiting the minimum repetition times of RF-intensive techniques, reducing flip angles or spatial coverage, and potentially mandating the introduction of time delays between acquisitions. This is of particular concern in sequences requiring echo trains of multiple 180° RF refocusing pulses such as the turbo spin-echo (TSE) / fast spin-echo (FSE) sequence. Adiabatic pulses sequences tend to deposit more RF energy than their conventional counterparts - for example, in the implementation of 180° refocusing pulse pairs in semi-LASER. This further complicates their implementation as potential solutions to address the B1-inhomogeneity issue, and may ultimately lead to longer scan duration.
4.6. Lack of Body Transmit RF coil
At the current time of writing, no human 7T MR system ships with a body transmit coil, making it necessary to integrate RF transmit elements along with the receive elements in an RF head coil. Hardware manufacturers like Nova Medical have introduced 7T head coils with 32 receive elements and either a single transmit channel or eight parallel transmit channels.
4.7. Unconventional Contrasts and Relaxation Properties
Relaxation behavior differs between conventional and ultrahigh field strengths, including longer T1 values, shortened T2* values for most brain tissues and shorter apparent T2 values. This may complicate direct comparisons to tissue contrast at lower fields and introduce unreliability in automated contrast-based segmentation methods. This may introduce some T1-weighting to short TR spectroscopy sequences, also causing a slight loss in SNR.
5.0. Patient Experience
Though FDA guidelines state that main magnetic field strengths up to 8 tesla (8T) are not “significant risks”, there remain some basic considerations for patient safety and comfort at ultrahigh field. Magnet resonance safety studies have thus far not demonstrated adverse effects beyond transient phenomenon such as nausea, vertigo, balance difficulty and metallic taste in the mouth [74]. Among the most widely-reported of these is the sensation of dizziness experienced as the scanning bed moves into and out of the scanner core. As a consequence, the early generation of ultrahigh field scanners was deployed with manually-moving scanner tables and these tables tend to require slow movement to minimize patient discomfort.
Human studies involving 1-hour exposure to high magnetic field have not demonstrated significant changes to physiological parameters such as body temperature, heart rate or blood pressure [75]. Though RF deposition is greater at higher field strengths, scanning at ultrahigh fields conforms to the same safety guidelines on SAR as established for lower field strengths, and these levels may be achieved through adjustments in scan parameters including repetition time, flip angle or the introduction of specifically-designed lower-SAR RF pulses. Ultrahigh field scanners also adhere to the same peripheral nerve stimulation limits initially established by regulatory bodies like the FDA for lower field scanners. This stimulation is modulated by the speed of gradient switching rather than the strength of the main magnetic field, and therefore not observed to behave differently at higher field strengths.
Ultrahigh field MR scanners adhere to the same limits for peak unweighted sound pressure (140 dB) and weighted root mean square (rms) sound pressure with hearing protection in place (99 dB) as scanners at lower field. Noise due to rapidly switching field gradients is primarily modulated by the speed of that gradient switching, and therefore not expected to be higher at higher field strengths. Noise dampening, insulation, hardware and sequence design can all play a role in maintaining sound levels within guidelines for patient safety and comfort. Ultrahigh field scanners tend to have longer bores than their lower-field counterparts and currently lack the wider bores seen in newer generations of 3T MRI scanners. This may exacerbate feelings of claustrophobia when inside the bore. Overall, 7T scanners have been used extensively for human studies and are reported to be well tolerated by patients in research studies.
6.0. Conclusions and Future directions
Despite ongoing technical challenges, the promise of higher signal quality, greater resolution and more reliable quantification has spurred the accelerating adoption of ultrahigh field scanners. As the first generation 7T human scanners enter clinical use, the technology may enable the detection of more subtle structural, physiological and metabolic abnormalities than is detectable at lower field strengths. These sensitivity enhancements may lead to the reliable detection of smaller arteries in stroke, fainter lesions in MS, more peri-ventricular spaces in epilepsy or more subtle metabolic imbalances in depression. Technological advancements such as parallel transmit coils and adiabatic RF pulses may further improve the quality and reliability of ultrahigh field imaging and sophisticated post-processing algorithms such as automatic segmentation and probabilistic tractography may tease out still further detail from these high-quality images. The combination of these technologies will push ultrahigh field scanners into improving the detection, assessment, monitoring and treatment of a range of diseases and disorders.
Table 2:
The relationship between various MRI/MRS-related limiting parameters and field strength (B0).
6.1. Acknowledgements
The authors would like to acknowledge the contributions of Stephanie S.G. Brown, Rebecca E. Feldman, Laurel S. Morris and John W. Rutland to this work. The authors would like to acknowledge funding from NIH R00 NS070821 (PB), NIH R01 MH109544 (PB) and R01 CA202911–01A1 (PB).
Disclosures:
The authors would like to acknowledge funding from NIH R00 NS070821 (PB), NIH R01 MH109544 (PB) and R01 CA202911–01A1 (PB).
PB is a named inventor on patents relating to magnetic resonance imaging (MRI) and RF pulse design. The patents have been licensed to GE Healthcare, Siemens AG, and Philips International. PB receives royalty payments relating to these patents.
Footnotes
Conflicts of Interest and Disclosures
PB is a named inventor on patents relating to magnetic resonance imaging (MRI) and RF pulse design. The patents have been licensed to GE Healthcare, Siemens AG, and Philips International. PB receives royalty payments relating to these patents.
References
- 1.Vedrine P, et al. , Iseult/INUMAC whole body 11.7 T MRI magnet status. IEEE Transactions on Applied Superconductivity, 2010. 20(3): p. 696–701. [Google Scholar]
- 2.Pohmann R, Speck O, and Scheffler K, Signal-to-noise ratio and MR tissue parameters in human brain imaging at 3, 7, and 9.4 tesla using current receive coil arrays. Magnetic resonance in medicine, 2016. 75(2): p. 801–809. [DOI] [PubMed] [Google Scholar]
- 3.Pfrommer A and Henning A. On the superlinear increase of the ultimate intrinsic signal-to-noise ratio with regard to main magnetic field strength in a spherical sample in 2017 International Conference on Electromagnetics in Advanced Applications (ICEAA). 2017. IEEE. [Google Scholar]
- 4.Van Der Kolk A, et al. , Imaging the intracranial atherosclerotic vessel wall using 7T MRI: initial comparison with histopathology. American Journal of Neuroradiology, 2015. 36(4): p. 694–701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kerchner GA, Ultra-high field 7T MRI: a new tool for studying Alzheimer’s disease. Journal of Alzheimer’s disease, 2011. 26(s3): p. 91–95. [DOI] [PubMed] [Google Scholar]
- 6.Duyn JH, The future of ultra-high field MRI and fMRI for study of the human brain. Neuroimage, 2012. 62(2): p. 1241–1248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ugurbil K, et al. , Ultrahigh field magnetic resonance imaging and spectroscopy. Magn Reson Imaging, 2003. 21(10): p. 1263–1281. [DOI] [PubMed] [Google Scholar]
- 8.Collins CM and Smith MB, Signal-to-noise ratio and absorbed power as functions of main magnetic field strength, and definition of “90°” RF pulse for the head in the birdcage coil. Magnetic Resonance in Medicine: An Official Journal of the International Society for Magnetic Resonance in Medicine, 2001. 45(4): p. 684–691. [DOI] [PubMed] [Google Scholar]
- 9.Gruber S, Mlynárik V, and Moser E, High-resolution 3D proton spectroscopic imaging of the human brain at 3 T: SNR issues and application for anatomy-matched voxel sizes. Magnetic Resonance in Medicine: An Official Journal of the International Society for Magnetic Resonance in Medicine, 2003. 49(2): p. 299–306. [DOI] [PubMed] [Google Scholar]
- 10.Peters AM, et al. , T2* measurements in human brain at 1.5, 3 and 7 T. Magnetic resonance imaging, 2007. 25(6): p. 748–753. [DOI] [PubMed] [Google Scholar]
- 11.Uludağ K, Müller-Bierl B, and Uğurbil K, An integrative model for neuronal activity-induced signal changes for gradient and spin echo functional imaging. Neuroimage, 2009. 48(1): p. 150–165. [DOI] [PubMed] [Google Scholar]
- 12.Bartha R, et al. , In vivo 1H2O T measurement in the human occipital lobe at 4T and 7T by Carr-Purcell MRI: Detection of microscopic susceptibility contrast. Magnetic Resonance in Medicine: An Official Journal of the International Society for Magnetic Resonance in Medicine, 2002. 47(4): p. 742–750. [DOI] [PubMed] [Google Scholar]
- 13.Michaeli S, et al. , Proton T2 relaxation study of water, N-acetylaspartate, and creatine in human brain using Hahn and Carr-Purcell spin echoes at 4T and 7T. Magnetic resonance in medicine, 2002. 47(4): p. 629–633. [DOI] [PubMed] [Google Scholar]
- 14.Kittel C, On the gyromagnetic ratio and spectroscopic splitting factor of ferromagnetic substances. Physical Review, 1949. 76(6): p. 743. [Google Scholar]
- 15.Kibble B, A measurement of the gyromagnetic ratio of the proton by the strong field method, in Atomic masses and fundamental constants 5. 1976, Springer; p. 545–551. [Google Scholar]
- 16.Gati JS, et al. , Experimental determination of the BOLD field strength dependence in vessels and tissue. Magnetic resonance in medicine, 1997. 38(2): p. 296–302. [DOI] [PubMed] [Google Scholar]
- 17.Thomas BP, et al. , High-resolution 7T MRI of the human hippocampus in vivo. Journal of Magnetic Resonance Imaging: An Official Journal of the International Society for Magnetic Resonance in Medicine, 2008. 28(5): p. 1266–1272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wisse L, et al. , Subfields of the hippocampal formation at 7 T MRI: in vivo volumetric assessment. Neuroimage, 2012. 61(4): p. 1043–1049. [DOI] [PubMed] [Google Scholar]
- 19.Wisse LE, et al. , Automated hippocampal subfield segmentation at 7T MRI. American Journal of Neuroradiology, 2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Solano-Castiella E, et al. , Parcellation of human amygdala in vivo using ultra high field structural MRI. Neuroimage, 2011. 58(3): p. 741–748. [DOI] [PubMed] [Google Scholar]
- 21.Conijn MM, et al. , Visualization of cerebral microbleeds with dual-echo T2*-weighted magnetic resonance imaging at 7.0 T. Journal of Magnetic Resonance Imaging, 2010. 32(1): p. 52–59. [DOI] [PubMed] [Google Scholar]
- 22.De Ciantis A, et al. , Ultra-high-field MR imaging in polymicrogyria and epilepsy. American Journal of Neuroradiology, 2015. 36(2): p. 309–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Keuken MC, et al. , Ultra-high 7T MRI of structural age-related changes of the subthalamic nucleus. Journal of Neuroscience, 2013. 33(11): p. 4896–4900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Keuken MC, et al. , Quantifying inter-individual anatomical variability in the subcortex using 7T structural MRI. NeuroImage, 2014. 94: p. 40–46. [DOI] [PubMed] [Google Scholar]
- 25.von Morze C, et al. , Intracranial time-of-flight MR angiography at 7T with comparison to 3T. Journal of Magnetic Resonance Imaging: An Official Journal of the International Society for Magnetic Resonance in Medicine, 2007. 26(4): p. 900–904. [DOI] [PubMed] [Google Scholar]
- 26.Morris LS, et al. , Ultra-high field MRI reveals mood-related circuit disturbances in depression: A comparison between 3-Tesla and 7-Tesla. Translational Psychiatry, 2019. 9(1): p. 94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Heidemann RM, et al. , Diffusion imaging in humans at 7T using readout-segmented EPI and GRAPPA. Magnetic Resonance in Medicine, 2010. 64(1): p. 9–14. [DOI] [PubMed] [Google Scholar]
- 28.Tkáč I, et al. , In vivo 1H NMR spectroscopy of the human brain at 7 T. Magnetic resonance in medicine, 2001. 46(3): p. 451–456. [DOI] [PubMed] [Google Scholar]
- 29.Wijtenburg SA, et al. , Reproducibility of brain spectroscopy at 7T using conventional localization and spectral editing techniques. Journal of Magnetic Resonance Imaging, 2013. 38(2): p. 460–467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Singh A, et al. , On B1 inhomogeneity correction of in vivo human brain glutamate chemical exchange saturation transfer contrast at 7T. Magnetic resonance in medicine, 2013. 69(3): p. 818–824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stephenson MC, et al. , Applications of multi-nuclear magnetic resonance spectroscopy at 7T. World journal of radiology, 2011. 3(4): p. 105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Verma G, et al. , Implementation of two-dimensional L-COSY at 7 tesla: An investigation of reproducibility in human brain. Journal of Magnetic Resonance Imaging, 2013. [DOI] [PubMed] [Google Scholar]
- 33.Terpstra M, et al. , Test-retest reproducibility of neurochemical profiles with short-echo, single-voxel MR spectroscopy at 3T and 7T. Magnetic resonance in medicine, 2016. 76(4): p. 1083–1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Prinsen H, et al. , Reproducibility measurement of glutathione, GABA, and glutamate: towards in vivo neurochemical profiling of multiple sclerosis with MR spectroscopy at 7T. Journal of Magnetic Resonance Imaging, 2017. 45(1): p. 187–198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Houtepen LC, et al. , Acute stress effects on GABA and glutamate levels in the prefrontal cortex: A 7T 1H magnetic resonance spectroscopy study. NeuroImage: Clinical, 2017. 14: p. 195–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Schür RR, et al. , Brain GABA levels across psychiatric disorders: A systematic literature review and meta-analysis of 1H-MRS studies. Human brain mapping, 2016. 37(9): p. 3337–3352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tkac I, et al. , In Vivo 1H NMR Spectroscopy of Rat Brain at 1 ms Echo Time. Magnetic resonance in medicine, 1999. 41: p. 649–656. [DOI] [PubMed] [Google Scholar]
- 38.Scheenen TW, Heerschap A, and Klomp DW, Towards 1H-MRSI of the human brain at 7T with slice-selective adiabatic refocusing pulses. Magnetic Resonance Materials in Physics, Biology and Medicine, 2008. 21(1–2): p. 95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Scheenen TW, et al. , Short echo time 1H-MRSI of the human brain at 3T with minimal chemical shift displacement errors using adiabatic refocusing pulses. Magnetic Resonance in Medicine: An Official Journal of the International Society for Magnetic Resonance in Medicine, 2008. 59(1): p. 1–6. [DOI] [PubMed] [Google Scholar]
- 40.Feldman RE and Balchandani P, A semiadiabatic spectral-spatial spectroscopic imaging (SASSI) sequence for improved high-field MR spectroscopic imaging. Magnetic resonance in medicine, 2016. 76(4): p. 1071–1082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Madelin G and Regatte RR, Biomedical applications of sodium MRI in vivo. Journal of Magnetic Resonance Imaging, 2013. 38(3): p. 511–529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sanson M, et al. , Isocitrate dehydrogenase 1 codon 132 mutation is an important prognostic biomarker in gliomas. Journal of Clinical Oncology, 2009. 27(25): p. 4150–4154. [DOI] [PubMed] [Google Scholar]
- 43.Verma G, et al. , Non-invasive detection of 2-hydroxyglutarate in IDH-mutated gliomas using two-dimensional localized correlation spectroscopy (2D L-COSY) at 7 Tesla. Journal of Translational Medicine, 2016. 14(1): p. 274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ganji SK, et al. , In vivo detection of 2-hydroxyglutarate in brain tumors by optimized point-resolved spectroscopy (PRESS) at 7T. Magnetic resonance in medicine, 2017. 77(3): p. 936–944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Balchandani P, Pauly J, and Spielman D, Designing adiabatic radio frequency pulses using the Shinnar–Le Roux algorithm. Magnetic resonance in medicine, 2010. 64(3): p. 843–851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Feldman RE, et al. , Quantification of perivascular spaces at 7 T: A potential MRI biomarker for epilepsy. Seizure, 2018. 54: p. 11–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Rutland JW, et al. , Subfield-specific tractography of the hippocampus in epilepsy patients at 7 Tesla. Seizure, 2018. 62: p. 3–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lener MS, et al. , Cortical abnormalities and association with symptom dimensions across the depressive spectrum. Journal of affective disorders, 2016. 190: p. 529–536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zou K, et al. , Alterations of white matter integrity in adults with major depressive disorder: a magnetic resonance imaging study. Journal of psychiatry & neuroscience: JPN, 2008. 33(6): p. 525. [PMC free article] [PubMed] [Google Scholar]
- 50.Cole J, et al. , White matter abnormalities and illness severity in major depressive disorder. The British Journal of Psychiatry, 2012. 201(1): p. 33–39. [DOI] [PubMed] [Google Scholar]
- 51.Rutland JW, et al. , Hippocampal subfield-specific connectivity findings in major depressive disorder: A 7 Tesla diffusion MRI study. Journal of Psychiatric Research, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tayoshi SY, et al. , GABA concentration in schizophrenia patients and the effects of antipsychotic medication: a proton magnetic resonance spectroscopy study. Schizophrenia research, 2010. 117(1): p. 83–91. [DOI] [PubMed] [Google Scholar]
- 53.Wong T, et al. , GABA, γ-hydroxybutyric acid, and neurological disease. Annals of neurology, 2003. 54(S6): p. S3–S12. [DOI] [PubMed] [Google Scholar]
- 54.Lydiard RB, The role of GABA in anxiety disorders. The Journal of clinical psychiatry, 2002. 64: p. 21–27. [PubMed] [Google Scholar]
- 55.Nakada T, et al. , In vivo visualization of senile-plaque-like pathology in Alzheimer’s disease patients by MR microscopy on a 7T system. Journal of Neuroimaging, 2008. 18(2): p. 125–129. [DOI] [PubMed] [Google Scholar]
- 56.Wisse LE, et al. , Hippocampal subfield volumes at 7T in early Alzheimer’s disease and normal aging. Neurobiology of aging, 2014. 35(9): p. 2039–2045. [DOI] [PubMed] [Google Scholar]
- 57.Langkammer C, et al. , MRI for iron mapping in Alzheimer’s disease. Neurodegenerative Diseases, 2014. 13(2–3): p. 189–191. [DOI] [PubMed] [Google Scholar]
- 58.Li X, et al. , Magnetic susceptibility contrast variations in multiple sclerosis lesions. Journal of magnetic resonance imaging, 2016. 43(2): p. 463–473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nielsen AS, et al. , Contribution of cortical lesion subtypes at 7T MRI to physical and cognitive performance in MS. Neurology, 2013: p. 10.1212/WNL.0b013e3182a08ce8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Tallantyre EC, et al. , 3 Tesla and 7 Tesla MRI of multiple sclerosis cortical lesions. Journal of Magnetic resonance imaging, 2010. 32(4): p. 971–977. [DOI] [PubMed] [Google Scholar]
- 61.Mainero C, et al. , In vivo imaging of cortical pathology in multiple sclerosis using ultra-high field MRI. Neurology, 2009. 73(12): p. 941–948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Shiee N, et al. , A topology-preserving approach to the segmentation of brain images with multiple sclerosis lesions. NeuroImage, 2010. 49(2): p. 1524–1535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kilsdonk ID, et al. , Multicontrast MR imaging at 7T in multiple sclerosis: highest lesion detection in cortical gray matter with 3D-FLAIR. American Journal of Neuroradiology, 2013. 34(4): p. 791–796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Van Veluw SJ, et al. , In vivo detection of cerebral cortical microinfarcts with high-resolution 7T MRI. Journal of Cerebral Blood Flow & Metabolism, 2013. 33(3): p. 322–329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Balchandani P, Pauly J, and Spielman D, Interleaved narrow-band PRESS sequence with adiabatic spatial-spectral refocusing pulses for 1H MRSI at 7T. Magnetic Resonance in Medicine: An Official Journal of the International Society for Magnetic Resonance in Medicine, 2008. 59(5): p. 973–979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Shellock FG, Schaefer DJ, and Kanal E, Physiologic responses to an MR imaging procedure performed at a specific absorption rate of 6.0 W/kg. Radiology, 1994. 192(3): p. 865–868. [DOI] [PubMed] [Google Scholar]
- 67.Ibrahim T, et al. , Analysis of B1 field profiles and SAR values for multi-strut transverse electromagnetic RF coils in high field MRI applications. Physics in Medicine & Biology, 2001. 46(10): p. 2545. [DOI] [PubMed] [Google Scholar]
- 68.Goelman G, et al. , Chemical-shift artifact reduction in hadamard-encoded MR spectroscopic imaging at high (3T and 7T) magnetic fields. Magnetic Resonance in Medicine: An Official Journal of the International Society for Magnetic Resonance in Medicine, 2007. 58(1): p. 167–173. [DOI] [PubMed] [Google Scholar]
- 69.Farahani K, et al. , Effect of field strength on susceptibility artifacts in magnetic resonance imaging. Computerized Medical Imaging and Graphics, 1990. 14(6): p. 409–413. [DOI] [PubMed] [Google Scholar]
- 70.Wrede KH, et al. , Caudal image contrast inversion in MPRAGE at 7 Tesla: problem and solution. Academic radiology, 2012. 19(2): p. 172–178. [DOI] [PubMed] [Google Scholar]
- 71.Balchandani P and Qiu D, Semi-adiabatic Shinnar–Le Roux pulses and their application to diffusion tensor imaging of humans at 7T. Magnetic resonance imaging, 2014. 32(7): p. 804–812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Gumbrecht R, et al. E, A. Fast high-flip pTx pulse design to mitigate B1+ inhomogeneity using composite pulses at 7T. in Proc Intl Soc Mag Reson Med. 2010. [Google Scholar]
- 73.Zelinski AC, et al. , Specific absorption rate studies of the parallel transmission of inner-volume excitations at 7T. Journal of Magnetic Resonance Imaging: An Official Journal of the International Society for Magnetic Resonance in Medicine, 2008. 28(4): p. 1005–1018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Hu X and Norris DG, Advances in high-field magnetic resonance imaging . Annu. Rev. Biomed. Eng, 2004. 6: p. 157–184. [DOI] [PubMed] [Google Scholar]
- 75.Kangarlu A, et al. , Cognitive, cardiac, and physiological safety studies in ultra high field magnetic resonance imaging. Magnetic resonance imaging, 1999. 17(10): p. 1407–1416. [DOI] [PubMed] [Google Scholar]