Abstract
BACKGROUND
Nonvariceal upper digestive bleeding (NVUDB) represents a severe emergency condition and is associated with significant morbidity and mortality. Despite a decrease in the incidence due to the widespread use of potent therapy with proton pump inhibitors as well as the implementation of modern endoscopic techniques, the mortality rate associated with NVUDB is still high.
AIM
To identify the clinical, biological, and endoscopic parameters associated with a poor outcome in patients with NVUDB to allow the stratification of risk, which will lead to the implementation of the most accurate management.
METHODS
We performed a retrospective study including patients who were admitted to the Gastroenterology Department of Clinical Emergency County Hospital Timisoara, Romania, with a diagnosis of NVUDB between 1 January 2008 and 31 December 2016. All the data were collected from the patient’s records, including demographic data, medication history, hemodynamic status, paraclinical tests, and endoscopic features as well as the methods of hemostasis, rate of rebleeding, need for surgery and death; we also assessed the Rockall score of the patients, length of hospitalization and associated comorbidities. All these parameters were evaluated as potential risk factors associated with rebleeding and death in patients with NVUDB.
RESULTS
We included a batch of 1581 patients with NVUDB, including 523 (33%) females and 1058 (67%) males with a median age of 66 years. The main cause of NVUDB was peptic ulcer (73% of patients). More than one-third of the patients needed endoscopic treatment. Rebleeding rate was 7.72%; surgery due to failure of endoscopic hemostasis was needed in 3.22% of cases; the in-hospital mortality rate was 8.09%, and the bleeding-episode-related mortality rate was 2.97%. Although our predictive models for rebleeding and death had a low sensitivity, the specificity was very high, suggesting a better discriminative capacity for identifying patients with better outcomes. Our results showed that the Rockall score was associated with both rebleeding and death; comorbidities such as respiratory conditions, liver cirrhosis and sepsis increased significantly the risk of in-hospital mortality (OR of 3.29, 2.91 and 8.03).
CONCLUSION
Our study revealed that the Rockall score, need for endoscopic therapy, necessity of transfusion and sepsis were risk factors for rebleeding. Moreover, an increased Rockall score and the presence of comorbidities were predictive factors for in-hospital mortality.
Keywords: Nonvariceal upper digestive bleeding, Risk factors, Rebleeding, Death, Outcome
Core tip: Because the rate of morbidity and mortality in patients with nonvariceal upper digestive bleeding (NVUDB) remains high, our retrospective study aims to identify clinical and paraclinical parameters associated with the risk of rebleeding and death in these patients. Our data showed that the Rockall score was associated with both rebleeding and death. The presence of comorbidities was associated with an increased risk of in-hospital mortality; among them, sepsis was associated with the highest risk. Identification of the risk factors for poor outcomes in patients with NVUDB proved to be associated with an improvement in management and, subsequently, in patient outcomes.
INTRODUCTION
Nonvariceal upper digestive bleeding (NVUDB) represents a severe condition commonly encountered in patients who are admitted in emergency units and is associated with significant morbidity and mortality. Despite a decrease in the incidence due to the widespread use of potent antisecretory therapy with proton pump inhibitors (PPIs) as well as the implementation of modern endoscopic hemostatic techniques, the mortality rate associated with NVUDB is still high (approximately 5%-10%, as revealed by literature data)[1-3]. Current studies reveal an incidence of NVUDB ranging from 20 to 60 per 100 000 inhabitants of North America and Europe[4,5], with an increasing proportion of cases in elderly patients presenting with multiple and severe comorbid diseases.
Aim of the study
As a result of these worrisome data, the main objective of our study is to detect the clinical, biological, and endoscopic parameters associated with poor outcomes in patients with NVUDB to allow the stratification of risk. Hence, we focus on identifying the variables associated with rebleeding and in-hospital mortality. Subsequently, the assessment of poor outcome predictors will lead to the implementation of the most accurate management. The algorithm will help to identify the high-risk patients who may need intensive care and prolonged hospitalization, implying the use of more medical resources, and the low-risk patients who may be safely discharged earlier; in addition to achieving better clinical results, this attitude is associated with cost savings.
MATERIALS AND METHODS
Inclusion of the patients
We performed a retrospective study that included the patients who were admitted to the Gastroenterology Department of Clinical Emergency County Hospital Timisoara, Romania, with a diagnosis of NVUDB between 1 January 2008 and 31 December 2016. Patient data were obtained by reviewing medical records and collecting information regarding demographic data, medication history (aspirin, non-steroidal anti-inflammatory drugs (NSAIDs), and antiaggregant/anticoagulant consumption), hemodynamic status at admission, laboratory tests, and endoscopic features (including the description of the type of the lesion and the method of endoscopic hemostasis performed along with their efficiency), which were translated into the rate of rebleeding, need for surgical intervention or death. Moreover, we assessed the Rockall score of the patients, length of hospitalization and associated comorbidities. All the parameters collected were assessed as potential risk factors associated with the rebleeding and death of patients with NVUDB.
All the patients presenting with NVUDB received antisecretory treatment with PPIs started at the time of admission with the administration of high doses of PPI, i.e., 80 mg IV bolus, followed by 8 mg/h continuous infusion for 72 h in all the cases that required endoscopic hemostatic treatment. All the endoscopic interventions were performed in emergency settings within 12 h after admission, after the patient was stabilized hemodynamically; we have to mention that the endoscopic unit worked in a round-the-clock manner.
Definitions
Initial failure of endoscopic treatment was considered as subsequent active bleeding despite performing endoscopic hemostasis or onset of active digestive bleeding represented by hematemesis, melena or hematochezia and the presence of hemodynamic shock [systolic blood pressure (SBP) < 100 mmHg and/or pulse > 100 beats per minute] within 12 h of initial endoscopic hemostasis.
Rebleeding was considered as the recurrence of active digestive hemorrhage (hematemesis, melena or hematochezia), hemodynamic instability, or a decrease in the hemoglobin level of more than 2 g/dL within 24 h of the first endoscopic procedure associated with the endoscopic visualization of active bleeding at the site of the previously treated lesion.
In-hospital mortality refers to deaths occurring during hospitalization in patients diagnosed and treated for NVUDB.
Bleeding episode-related mortality refers to deaths determined by irreversible hypovolemic shock.
Unidentified cause of bleeding means the endoscopic visualization of blood in the stomach without demonstrating a source of bleeding (causative lesion).
Ethical considerations
All patients included in this study or their relatives had signed informed consent forms for the diagnostic and therapeutic procedures needed. This study was approved by the University Ethical Committee.
Statistical analysis
Because continuous variables were skewed, data are presented as medians and interquartile ranges (IQRs), and categorical variables are presented as percentages. Statistical differences between groups were assessed by the Mann-Whitney U-test and Chi-square test where appropriate. As a secondary analysis, we estimated the associations of rebleeding and death with clinical and endoscopic parameters using odds ratios (OR) and 95% confidence intervals (CI) in logistic regression models. The receiver operator characteristic (ROC) curve for each model of the best combination of predictors was plotted, and the area under the curve was calculated. The P values for all hypothesis tests were two-sided, and we set statistical significance at P < 0.05. All analyses were conducted with Stata version 15.1 (Statacorp, Texas, United States).
RESULTS
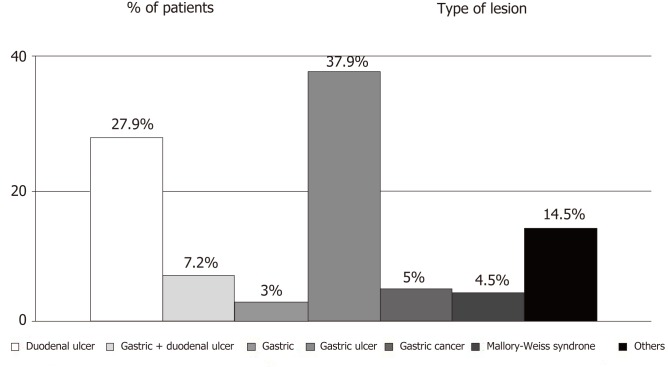
We assessed a batch of 1581 patients admitted with the diagnosis of NVUDB to the Department of Gastroenterology, Clinical Emergency County Hospital Timisoara, Romania, between 1 January 2008 and 31 December 2016, which included 523 (33%) females and 1058 (67%) males with a median age 66 years (IQR 55–76). Mean values of important clinical and laboratory parameters are presented in Table 1. The main cause of NVUDB was peptic ulcer, which was diagnosed in 1153 (73%) patients. Other important etiologies were gastritis, gastric cancer and Mallory-Weiss syndrome (Figure 1).
Table 1.
Clinical and laboratory parameters of the study population (median values)
| Parameters (n = 1581) | Median value | IQ range | |
| Age | 66 | 55 | 76 |
| Rockall score | 5 | 3 | 6 |
| Hb | 8 | 7 | 10 |
| Hospitalization length | 5 | 3 | 7 |
Hb: Hemoglobin value; IQ range: Interquartile range.
Figure 1.
Etiology of non-variceal upper digestive bleeding. Others include the following lesions: Dieulafoy lesions, duodenitis, dicumarinic overdose, pancreatic tumor invasions, telangiectasia, reflux and post-caustic esofagitis, esophageal ulcers, esophageal neoplasm, hiatal hernia, portal gastropathy, malignant ampuloma and unknown causes of bleeding.
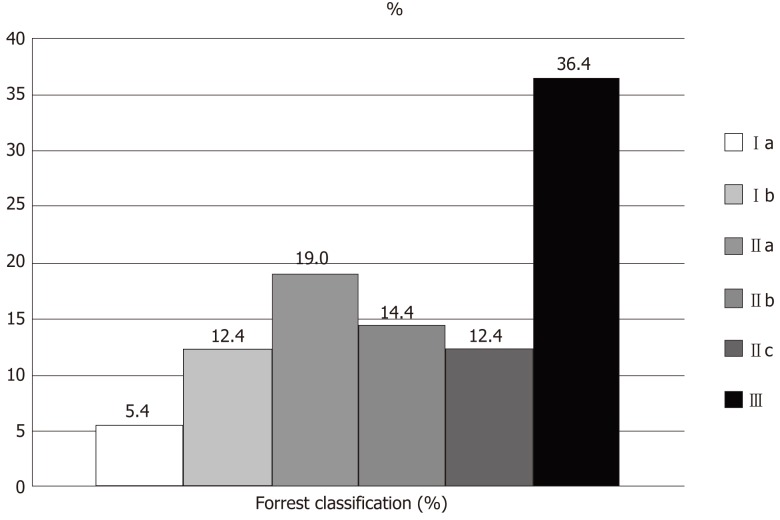
In the case of peptic ulcers, according to Forrest classification, 17.8% of patients presented with Forrest type I, 45.8% with type II, and 36.4 with type III (Figure 2).
Figure 2.
Endoscopic Forrest classification of upper digestive bleeding from peptic ulcers. Ia: Active spurting; Ib: Active oozing: IIa: Non-bleeding visible vessel; IIb: Adherent clot; IIc: Flat pigmented spot; III: Clean ulcer base.
More than one-third of the patients (548 patients, 34.64%) needed endoscopic treatment, most often combined with hemostatic procedures, while the rest of the 1033 patients (65.36%) did not receive any endoscopic therapy. The most frequently used technique of simple therapy was epinephrine injection, while the most frequently used combined hemostatic method was epinephrine injection plus hemoclip mounting (Table 2).
Table 2.
Type of endoscopic treatment applied to the study population
| Endoscopic therapy | No. of patients | % out of entire batch |
| Simple therapy | 174 | 11.01 |
| Epinephrine injection | 62 | 3.92 |
| Bipolar coagulation | 18 | 1.14 |
| Hemoclip | 74 | 4.68 |
| Banding1 | 7 | 0.44 |
| Argon plasma coagulation | 13 | 0.83 |
| Combined therapy | 375 | 23.72 |
| Epinephrine injection + bipolar coagulation | 126 | 7.97 |
| Epinephrine injection + hemoclip | 206 | 13.04 |
| Epinephrine injection + bipolar coagulation + hemoclip | 35 | 2.21 |
| Bipolar coagulation + hemoclip | 4 | 0.25 |
| Epinephrine injection + argon plasma coagulation + hemoclip | 1 | 0.06 |
| Epinephrine injection + banding | 2 | 0.15 |
| Epinephrine injection + argon plasma coagulation | 1 | 0.06 |
banding was used for endoscopic hemostasis in 4 cases of Mallory-Weiss syndrome and 3 cases of Dieulafoy lesion.
Rebleeding was encountered in 122/1581 patients (7.72%). Surgery due to failure of endoscopic hemostasis was needed in 51/1581 patients (3.22%). The in-hospital mortality rated included 128/1581 patients (8.09%), while the bleeding-episode related mortality included 47/1581 patients (2.97%).
Regarding the presence of comorbidities, we detected the following: 977/1581 patients (61.8%) had cardiovascular diseases, 333/1581 patients (21%) had renal conditions, 296/1581 patients (18.7%) had diabetes mellitus, 256/1581 patients (16.2%) had neoplasias, 216/1581 patients (13.7%) had neurological diseases, 208/1581 patients (13.2%) had respiratory conditions, 143/1581 patients (9%) had liver cirrhosis, 134/1581 patients (8.5%) had obesity, 49/1581 patients (3%) had sepsis, 8/1581 patients (0.5%) had acute pancreatitis, and 6/1581 patients (0.4%) had an acute abdomen.
Parameters associated with rebleeding
Univariate analysis revealed that the clinical and endoscopic parameters that were statistically significantly associated with rebleeding were Rockall score, need for endoscopic therapy (simple or combined), and presence of hemorrhagic shock at admission (systemic blood pressure < 100 mmHg and/or pulse rate > 100 beats per minutes) (P < 0.001). Among the comorbidities, the presence of sepsis and acute abdomen were significantly associated with rebleeding (P < 0.05) (Table 3).
Table 3.
Clinical and endoscopic features of the study population: Association with rebleeding
| Parameter | No-rebleeding (no. pts, %) | Rebleeding, (no. pts, %) | Total no. patients, % | Total (% out of entire batch) | P value |
| Rockall score | 1435 (92.16) | 122 (7.84) | 1.557 (100) | 100 | < 0.001 |
| Simple therapy | 143 (82.18) | 31 (17.82) | 174 (100) | 11.01 | < 0.001 |
| Combined therapy | 318 (84.8) | 57 (15.2) | 375 (100) | 23.72 | < 0.001 |
| SBP (mmHg) < 100 | 240 (83.33) | 48 (16.67) | 288 (100) | 18.22 | < 0.001 |
| Pulse (beats per minute) > 100 | 404 (87.26) | 59 (12.74) | 463 (100) | 29.32 | < 0.001 |
| Cardiovascular comorbidities | 901 (92.32) | 75 (7.68) | 976 (100) | 61.73 | 0.857 |
| Diabetes mellitus | 272 (91.89) | 24 (8.11) | 296 (100) | 18.72 | 0.815 |
| Respiratory comorbidities | 184 (89.32) | 22 (10.68) | 206 (100) | 13.03 | 0.096 |
| Renal comorbidities | 303 (91.82) | 27 (8.18) | 330 (100) | 20.87 | 0.759 |
| Liver cirrhosis | 128 (90.78) | 13 (9.22) | 141 (100) | 8.92 | 0.504 |
| Neoplasias | 232 (90.63) | 24 (9.38) | 256 (100) | 16.19 | 0.298 |
| Neurologic comorbidities | 202 (94.39) | 12 (5.61) | 214 (100) | 13.54 | 0.202 |
| Obesity | 125 (93.28) | 9 (6.72) | 134 (100) | 8.48 | 0.631 |
| Sepsis | 38 (80.85) | 9 (19.15) | 47 (100) | 2.97 | 0.003 |
| Acute pancreatitis | 6 (75) | 2 (25) | 8 (100) | 0.51 | 0.068 |
| Acute abdomen | 4 (66.67) | 2 (33.33) | 6 (100) | 0.38 | 0.019 |
SBP: Systolic blood pressure.
Using multivariate analysis, parameters that were markedly associated with an increased risk of rebleeding were Rockall score (OR: 1.41; 95%CI: 1.25-1.58, P < 0.001), need for simple therapy (OR: 3.18; 95%CI: 1.92-5.28, P < 0.001) or combined therapy (OR: 1.75; 95%CI: 1.31-2.33, P < 0.001), number of blood transfusions (OR: 1.46; 95%CI: 1.32-1.62, P < 0.001), and sepsis (OR: 2.95; 95%CI: 1.34-7.29, P = 0.008) (Table 4).
Table 4.
Logistic regression for rebleeding
| Variables | Odds ratio | 95% Confidence interval | P value | |
| Age | 0.98 | 0.96 | 0.99 | 0.030 |
| No. of blood units | 1.46 | 1.32 | 1.62 | < 0.001 |
| Sepsis | 2.95 | 1.26 | 6.94 | 0.008 |
| Rockall score | 1.41 | 1.25 | 1.58 | < 0.001 |
| Simple therapy | 3.18 | 1.92 | 5.28 | < 0.001 |
| Combined therapy | 1.75 | 1.31 | 2.33 | < 0.001 |
Based on these results, we performed a predictive model for rebleeding. The model presented with a sensitivity (Se) of 10.66%, specificity (Sp) of 99.3%, positive predictive value (PPV) of 56.52% and negative predictive value (NPV) of 92.89%. The accuracy of our model was 92.35%. Although the predictive model had a low sensitivity, the specificity was very high. Therefore, it had a very powerful discriminative capacity for identifying patients with NVUDB who would not develop rebleeding and hence was associated with a better outcome.
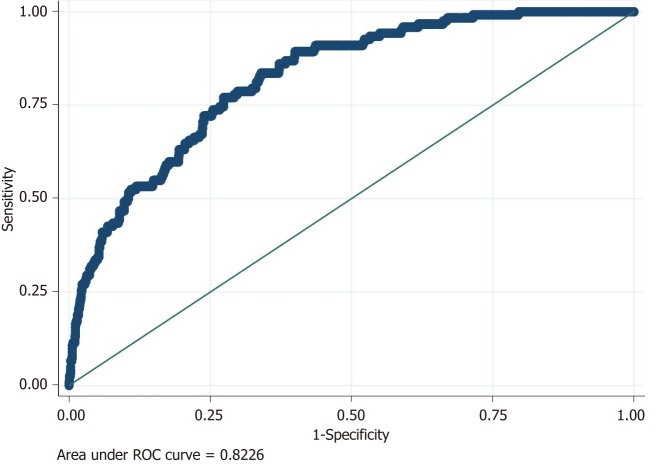
Using the area under the ROC curve, our predictive model for rebleeding obtained an AUROC value of 0.82 (Figure 3).
Figure 3.
Logistic model for rebleeding: Accuracy of the predictive model for rebleeding using area under ROC curve (AUROC).
Parameters associated with death due to bleeding episode
Statistical analysis of our data showed that patients who died due to a bleeding episode were significantly older (P = 0.012), had a higher Rockall score, had a lower level of hemoglobin and died quite soon after admission (median hospitalization length of 1 d) (P < 0.001) (Table 5).
Table 5.
Clinical and laboratory parameters of the study population: Association with deaths due to bleeding
| Parameters | Deaths due to bleeding | Median (IQ range) | P value |
| Age (yr) | No | 65 (55-76) | 0.012 |
| Yes | 72 (57-82) | ||
| Rockall score | No | 5 (3-6) | < 0.001 |
| Yes | 7 (7-8) | ||
| Hb (g/dL) | No | 8 (7-11) | < 0.001 |
| Yes | 7 (5-8) | ||
| Hospitalization length (d) | No | 5 (3-7) | < 0.001 |
| Yes | 1 (1-2) |
Hb: Hemoglobin value; IQ range: Interquartile range.
Using univariate analysis, variables independently and extremely significantly associated with death due to a bleeding episode were Rockall score, hemorrhagic shock at admission (systemic blood pressure < 100 mmHg and/or pulse rate > 100 beats per minutes), rebleeding, number of blood units (P < 0.001), and the presence of respiratory comorbidities. Comorbidities such as sepsis and acute abdomen were also significantly associated with death (P < 0.05) (Table 6).
Table 6.
Clinical and endoscopic features of the study population: Association with deaths due to bleeding
| Parameter | Survivors (no. pts) | Deaths due to bleeding (no. pts) | P value |
| Gender | 1534 | 47 | 0.989 |
| Lesion | 1534 | 47 | 0.140 |
| Rockall score | 1514 | 43 | < 0.001 |
| Simple therapy | 167 | 7 | 0.196 |
| Combined therapy | 363 | 12 | 0.966 |
| SBP < 100 mmHg | 252 | 36 | < 0.001 |
| Pulse > 100 beats per minute | 429 | 34 | < 0.001 |
| Rebleeding | 113 | 10 | < 0.001 |
| No. blood units | 1532 | 47 | < 0.001 |
| Aspirin | 310 | 6 | 0.451 |
| NSAIDs | 240 | 5 | 0.350 |
| Surgery | 51 | 0.204 | |
| Antiaggregants | 104 | 2 | 0.179 |
| Anticoagulants | 93 | 7 | 0.110 |
| Cardiovascular comorbidities | 0.221 | ||
| Diabetes mellitus | 189 | 12 | 0.224 |
| Respiratory comorbidities | 189 | 17 | < 0.001 |
| Renal comorbidities | 320 | 10 | 0.945 |
| Liver cirrhosis | 134 | 7 | 0.145 |
| Neoplasias | 248 | 8 | 0.876 |
| Neurologic comorbidities | 209 | 5 | 0.556 |
| Obesity | 133 | 1 | 0.113 |
| Sepsis | 42 | 5 | 0.002 |
| Acute pancreatitis | 4 | 4 | 0.620 |
| Acute abdomen | 5 | 1 | 0.048 |
SBP: Systolic blood pressure; NSAIDs: Non-steroidal anti-inflammatory drugs.
Parameters associated with in-hospital death
As in the case of death due to a bleeding episode, statistical analysis showed that patients who died during hospitalization were significantly older, had a higher Rockall score, had a lower level of hemoglobin and died in a short period after admission (median hospitalization length of 2 d) (P < 0.001) (Table 7).
Table 7.
Clinical and laboratory parameters of the study population: Association with deaths
| Parameters | Deaths | Median (IQ range) | P value |
| Age (yr) | No | 65 (55-76) | 0.001 |
| Yes | 70 (58-80) | ||
| Rockall score | No | 5 (3-6) | < 0.001 |
| Yes | 7 (6-8) | ||
| Hb (g/dL) | No | 8 (7-11) | < 0.001 |
| Yes | 7 (6-9) | ||
| Hospitalization length (d) | No | 5 (3-7) | < 0.001 |
| Yes | 2 (1-4) |
Hb: Hemoglobin value; IQ range: Interquartile range.
Using univariate analysis, variables independently and extremely significantly associated with death were type of lesion (gastric ulcer was associated with the highest number of deaths - 41 cases, meaning 32.03% of all deaths), Rockall score, hemorrhagic shock at admission (systemic blood pressure < 100 mmHg and/or pulse rate > 100 beats per minutes), hospitalization length and number of blood units (P < 0.001). Comorbidities such as respiratory and renal diseases, sepsis, and acute pancreatitis as well as liver cirrhosis were also extremely significantly associated with death (P < 0.001). Moreover, consumption of aspirin and NSAIDs, need for surgery, and existence of neoplasia were significantly associated with death (P < 0.05) (Table 8).
Table 8.
Clinical and endoscopic features of the study population: Association with deaths
| Parameter | Survivors (no. pts, %) | Deaths (no. pts, %) | Total no. patients, % | Total (% out of entire batch) | P value |
| Lesion | 1453 (91.90) | 128 (8.10) | 1581 (100) | 100 | 0.001 |
| Rockall score | 1442 (92.61) | 115 (7.39) | 1557 (100) | 100 | < 0.001 |
| Simple therapy | 160 (91.95) | 14 (8.05) | 174 (100) | 11.01 | 0.069 |
| Combined therapy | 355 (94.66) | 20 (5.34) | 375 (100) | 23.72 | 0.209 |
| SBP < 100 mmHg | 216 (75) | 72 (25) | 288 (100) | 18.22 | < 0.001 |
| Pulse > 100 beats per minute | 392 (84.67) | 71 (15.53) | 463 (100) | 29.32 | < 0.001 |
| Rebleeding | 109 (88.62) | 14 (11.38) | 123 (100) | 7.78 | 0.164 |
| Hospitalization length | 1449 (91.94) | 127 (8.06) | 1576 (100) | 100 | < 0.001 |
| No. blood units | 1452 (91.96) | 127 (8.04) | 1579 (100) | 100 | < 0.001 |
| Aspirin | 301 (95.56) | 14 (4.44) | 315 (100) | 19.92 | 0.028 |
| NSAIDs | 237 (96.73) | 8 (3.27) | 245 (100) | 15.50 | 0.003 |
| Surgery | 51 (100) | 0.00 (0.00) | 51 (100) | 3.23 | 0.031 |
| Cardiovascular comorbidities | 895 (91.70) | 81 (8.30) | 976 (100) | 61.73 | 0.707 |
| Diabetes mellitus | 268 (90.54) | 28 (9.46) | 296 (100) | 18.72 | 0.340 |
| Respiratory comorbidities | 156 (75.73) | 50 (24.27) | 206 (100) | 13.03 | < 0.001 |
| Renal comorbidities | 285 (86.36) | 45 (13.64) | 330 (100) | 20.87 | < 0.001 |
| Liver cirrhosis | 115 (81.56) | 26 (18.44) | 141 (100) | 8.92 | < 0.001 |
| Neoplasias | 225 (87.89) | 31 (12.11) | 256 (100) | 16.19 | 0.010 |
| Neurologic comorbidities | 193 (90.19) | 21 (9.81) | 214 (100) | 13.54 | 0.322 |
| Obesity | 127 (94.78) | 7 (5.22) | 134 (100) | 8.48 | 0.203 |
| Sepsis | 25 (53.19) | 22 (46.81) | 47 (100) | 2.97 | < 0.001 |
| Acute pancreatitis | 4 (50) | 4 (50) | 8 (100) | 0.51 | < 0.001 |
| Acute abdomen | 5 (83.33) | 1 (16.67) | 6 (100) | 0.38 | 0.441 |
SBP: Systolic blood pressure; NSAIDs: Non-steroidal anti-inflammatory drugs.
Multivariate analysis demonstrated that the variables significantly associated with death were Rockall score (OR: 1.73; 95%CI: 1.53-1.96, P < 0.001), respiratory comorbidities (OR: 3.29; 95%CI: 2.06-5.25, P < 0.001), liver cirrhosis (OR: 2.91; 95%CI: 1.64-5.15, P < 0.001), sepsis (OR: 8.03; 95%CI: 3.81-16.93, P < 0.001) and acute pancreatitis (OR: 6.58; 95%CI: 1.04-41.63, P < 0.05) (Table 9).
Table 9.
Logistic regression for death
| Death | Odds ratio | 95% Confidence interval | P value | |
| Rockall score | 1.73 | 1.53 | 1.96 | < 0.001 |
| Respiratory comorbidities | 3.29 | 2.06 | 5.25 | < 0.001 |
| Liver cirrhosis | 2.91 | 1.64 | 5.15 | < 0.001 |
| Sepsis | 8.03 | 3.81 | 16.93 | < 0.001 |
| Acute pancreatitis | 6.58 | 1.04 | 41.63 | 0.045 |
Based on these results, we performed a predictive model for death. The model presented Se of 19.13%, Sp of 99.24%, PPV of 66.67% and NPV of 93.90%. The accuracy of our model was 93.32%. Although the predictive model had a low sensitivity, the specificity was very high. Therefore, it had a very powerful discriminative capacity for identifying patients who would not die, hence identifying NVUDB patients with a favorable prognosis.
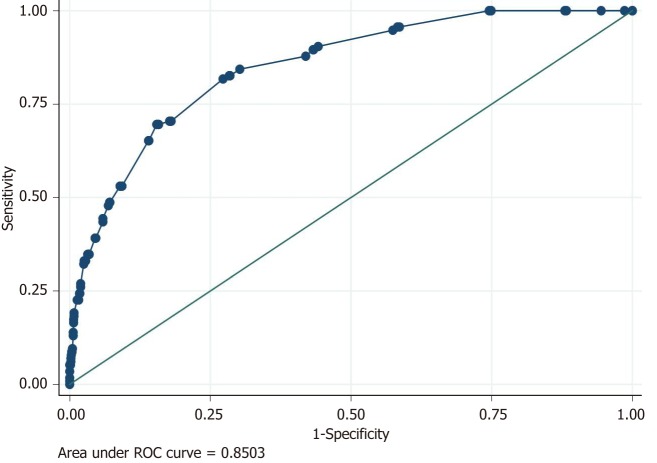
Using the area under the ROC curve, our predictive model for death obtained an AUROC value of 0.85 (Figure 4).
Figure 4.
Logistic model for death: Accuracy of the predictive model for death using area under ROC curve (AUROC).
DISCUSSION
Many studies have been performed in order to identify predictors of negative outcomes in NVUDB (rebleeding, surgery, and death) on which to decide the timing of discharge in these patients and select the proper management according to risk stratification[6].
Our study reveals both differences and similarities compared to other similar research (Table 10)[1-17]. The age of patients may be the result of the age distribution of different populations[1,14,18]. In conformity with the literature, our results revealed that peptic ulcer represents the most common etiology of NVUDB (73% of cases)[7]. In our analysis, rebleeding was encountered in 7.72% of cases, and surgery was needed in 3.22% of patients. The differences of rebleeding rate among studies may be the result of various proportions of patients receiving combined endoscopic hemostasis vs pharmacologic therapy[9,18-22]. Similar to other studies, rebleeding proved to be an independent predictor of death due to a bleeding episode in our research (P < 0.001).
Table 10.
Studies regarding predictors of rebleeding and mortality in patients with upper digestive bleeding (nonvariceal/all causes)
| Study | Yr | Condi-tion |
Type of study |
No. of patients | Mean age (yr) | Mor-tality rate, (%) | Re-bleeding rate (%) | Predic-tive factors for mor-tality | Odds ratio, (OR) | Predic-tive factors for re-bleeding | Odds ratio, (OR) | Surgery (%) | |
| Prospective/retrospec-tive | Uni-/multicentric | ||||||||||||
| Barkun et al[1] | 2004 | NVUDB | R | M | 869 | 66 ± 17 | 5.4 | 14.1 | (1) PPI use; (2) Endo-scopic therapy | (1) 0.18; (2) 0.31 | (1) PPI use; (2) Endo-scopic hemostasis in patients with high risk stigmata | (1) 0.53; (2) 0.39 | 6.5 |
| Travis et al[6] | 2008 | NVUDB | R | M | 236 | 67 | NA | 7.1%, 16.4%, 37.0%, 75.0% and 100% for zero, one, two, three or four risk factors | NA | NA | (1) Use of PPI post-proce-dure; (2) Endo-scopically demon-strated bleeding; (3) Hemo-stasis with epineph-rine mono-therapy; (4) Post-proce-dure i.v or LMWH use; (5) Mode-rate/severe liver disease; (6) Peptic ulcer as the source of bleeding | (1) 0.25; (2) 2; (3) 3.35; (4) 8.09; (5) 4.92 | NA |
| Sung et al[7] | 2010 | PUB | P | U | 10428 | 61.0 (sur-vivors) | 6.23 | 2.93 | (1) Use of NSAIDs/ aspirin; (2) Active bleeding ulcer; (3) Cloth/vessel at the base of ulcer; (4) Hemodymamic shock (bleeding-related death) | (1) 3.70; (2) 12.96; (3) 12.29; (4) 3.75 | NA | NA | 2.8 (deaths during surgery) |
| 72.5 (deaths) | |||||||||||||
| Zhang et al[8] | 2010 | NVUDB | R | U | 223 | NA | NA | 19.3 (failure of endo-scopic treat-ment) | (1) No. of comorbidities > 1; (2) Spurting of blood | (1) 9.580; (2) 9.971 | (1) Shock; (2) History of GI bleeding; (3) PLT 100 x 109/L; (4) Active spurting of blood; (5) Large lesion size | (1) 3.058; (2) 2.809; (3) 0.067; (4) 10.390; (5) 7.111 | NA |
| González-González et al[9] | 2011 | NVUDB | P | U | 1077 | 58.8 ± 18.9 | 10.2 | 3.4 | (1) No. of comorbidities/patient; (2) Serum albumin level < 2.6 g/dL; (3) Re-bleeding; (4) Rockall score pre-endo-scopy; (5) Lengths of hospital stay | (1) 1.6; (2) 4.9; (3) 6.5; (4) 1.3; (5) 1.04 | NA | NA | 1.5 |
| Morales Uribe et al[10] | 2011 | UDB | P | M | 464 | 59.7 | 9.9 | 17.4 | (1) Bleeding site (in-hospital vs outpatients); (2) Comorbidities | (1) 2.4; (2) 2.5 | NA | NA | 2.2 |
| Nahon et al[11] | 2012 | UDB | P | M | 3298 | 63 ± 18 | 8.3 | 9.9 | (1) Rockall score; (2) Comorbidities; (3) SBP < 100 mmHg | (1) 2.8; (2) 3.6 (for each additio-nal comorbidity); (3) 2.1 | (1) Need for transfu-sions; (2) Hb < 10 g/dL; (3) Rockall score; (4) SBP < 100 mmHg; (5) Signs of recent bleeding | (1) 19.1; (2) 1.7; (3) 1.4 (for each point score increase); (4) 1.9; (5) 2.4 | NA |
| Del Piano et al[12] | 2013 | NVUDB | P | M | 1413 | (1) -66.5 ± 15.8 male; (2) -74.2 ± 14.6 female | 5.4 | 4 | NA | NA | (1) Female sex; (2) Neo-plasia; (3) Multiple comorbidities; (4) Shock at admis-sion; (5) Early re-bleeding | (1) 2.19; (2) 2.7; (3) 5.04; (4) 4.55; (5) 1.47 | 14.3 (of early rebleed-ers) |
| Taha et al[13] | 2014 | UDB | R | U | 2669 | NA | 7.1 | (1) Age; (2) Charlson score; (3) Rockall score; (4) Units of blood trans-fused | (1) 1.020; (2) 1.291; (3) 1.274; (4) 1.085 | NA | NA | 2.1 | |
| Marmo et al[14,15] | 2014 | NVUDB | P | M | 2317 | 67.9 ± 16.7 | 4.573 | 5.61 | (1) Hemodynamic insta-bility on presen-tation; (2) ASA class 3 or 4; (3) Low-dose aspirin use; (4) History of peptic ulcer; (5) Re-bleeding; (6) Failed endo-scopic treatment | (1) 7.311; 2.312; (2) 6.721; 3.892; (3) 0.121; 0.252; (4) 3.181; 1.542; (5) 5.222; 14.292 | NA | NA | (1) 1.51; (2) 22 |
| 3.42 | |||||||||||||
| Lee et al[16] | 2016 | NVUDB | P | U | 184 | 59.81 | 8.73 | 14.73 | (1) Diabetes mellitus; (2) Meta-static malignancy; (3) Age ≥ 65 yr; (4) Hypoten-sion | (1) 12.67; (2) 29.24; (3) 5.06; (4) 16.63 | NA | NA | NA |
| Hwang et al[17] | 2016 | NVUDB | P | M | 1584 | 65 | 3.43 | 7.3 | (1) Age > 65 yr; (2) Hemodynamic insta-bility; (3) Serum BUN levels > 40 mg/dL; (4) Active bleeding at endo-scopy; (5) Transfusions; (6) Comorbidities; (7) Re-bleeding | (1) 2.627; (2) 2.217; (3) 1.895; (4) 2.434; (5) 3.811; (6) 3.481; (7) 10.581 | NA | NA | 2.8 (surgery/ percutaneous embolisa-tion) |
bleeding occurs in patients already hospitalized for another condition;
in-hospital bleeding; 3 at 30-d from the bleeding episode. NA: Not available; NVUDB: Nonvariceal upper digestive bleeding; UDB: Upper digestive bleeding (all causes); PUB: Peptic ulcer bleeding; R: Retrospective study; P: Prospective study; U: Unicentric study; M: Multicentric study; PPI: Proton pump inhibitor; LMWH: Low molecular weight heparin; GI: Gastrointestinal; PLT: Platelets.
A multidisciplinary team is needed for the initial assessment of patients with NVUDB and for hemodynamic stabilization before endoscopic hemostasis. Currently, the literature reveals that endoscopic hemostasis can be achieved in over 95% of patients with upper digestive bleeding[23].
Independent parameters identified in our study to be significantly associated with rebleeding were Rockall score, need for endoscopic therapy, presence of hemorrhagic shock at admission, and comorbidities such as sepsis and acute abdomen. Using logistic regression, parameters significantly associated with an increased risk of rebleeding were Rockall score (OR: 1.41), need for therapy [simple (OR: 3.18) or combined (OR: 1.75)], number of blood units received (OR: 1.46), and sepsis (OR: 2.95).
The widespread use of potent PPIs, detection and treatment of Helicobacter pylori infection in the case of peptic ulcer disease, and the use of modern endoscopic devices and hemostatic techniques have contributed to a change in the management of NVUDB from predominantly surgical to endoscopic. On the other hand, in recent years, there has been an increasing proportion of elderly patients[24] with the presence of multiple comorbidities, leading to an increased consumption of NSAIDs and antiplatelet treatment. In this context, NVUDB still continues to be associated with significant morbidity and mortality as well as with high costs for the health care systems.
Many publications in recent years have compared the efficacy of various scoring systems as a risk stratification method[4,25-34]. Patients with a low score usually do not need any clinical intervention and may be safely discharged very early with elective endoscopy performed later[35,36]. In contrast, a high-risk score is associated with frequent need for clinical intervention, such as administration of blood transfusions and hemostatic endoscopic treatment. Therefore, high risk scores, as significant predictors of adverse outcome for NVUDB, should be associated with more aggressive endoscopic management, more prolonged utilization of intravenous PPIs, or even additional days of hospitalization[37].
The study of Wang et al[38] demonstrated a significant positive linear correlation between clinical Rockall scores and patient outcomes defined as rebleeding, surgery and mortality rate, and high clinical Rockall scores (> 3 points) were associated with adverse outcomes in NVUDB patients.
Because NVUDB represents a dynamic manifestation that mirrors the changing pattern of the comorbidities and treatments of diverse diseases, a strategy to reduce the mortality rate should concentrate on managing coexisting diseases[39,40].
In our study, independent variables significantly associated with death were type of lesion, Rockall score, hemorrhagic shock at admission, hospitalization length, number of blood units transfused, and comorbidities (such as respiratory and renal diseases, sepsis, liver cirrhosis, neoplasias, and severe acute pancreatitis) as well as consumption of aspirin and NSAIDs and need for surgery. Logistic regression demonstrated that variables significantly associated with death were Rockall score (OR: 1.73) and comorbidities such as respiratory conditions (OR: 3.29), liver cirrhosis (OR: 2.91), sepsis (OR: 8.03) and acute pancreatitis (OR: 6.58). Sung et al[7] reported that most of the patients died due to comorbidities leading to multiorgan failure; furthermore, only 18.3% of deaths were due to ulcer-related bleeding.
Our study has certain limitations. First, it was a retrospective study implying potential data bias, and it was a single center study. However, because the Gastroenterology Department represents a referral tertiary center covering the western region of the country, the Emergency Unit receives many critical cases with NVUDB from all the neighboring counties. A second limitation was encountered because we were able to identify only patients who died in the hospital, not after discharge. On the other hand, the main strengths of our study were that it was comprehensive research conducted over a long period of time and included a large number of patients on which we were able to assess the particularities of our pool of patients presenting with NVUDB and the risk factors for rebleeding and death. Moreover, we had the availability of detailed patient records, including clinical parameters, results of paraclinical and endoscopic investigations and outcomes. Additionally, we used standardized methods, and we had the possibility to check the data.
We performed a predictive model for rebleeding associated with a Se of only 10.66% but a Sp of 99.3% and an accuracy of 92.35%. Furthermore, we were able to perform a predictive model for death with a Se of 19.13%, a Sp of 99.24%, and an accuracy of 93.32%. Although the predictive models had a low sensitivity, the specificity was very high, proving a better discriminative capacity for identifying patients with NVUDB with favorable outcomes who will not develop rebleeding and will not die.
In conclusion, our results showed that the Rockall score was significantly associated with both rebleeding and death. Also, the need for endoscopic hemostasis, the number of blood units transfused and presence of sepsis were correlated with the risk of rebleeding. Furthermore, the presence of comorbidities was a predictive factor for in-hospital mortality; among them, sepsis proved to be associated with the highest relative risk for death, increasing by 8 times the in-hospital mortality risk. A thorough identification of the risk factors starting from admission of patients and further stratification of patients with NVUDB will lead to an improvement in patient outcomes and reduced costs. The approach of these patients should focus also on the management of coexisting conditions and sepsis control.
ARTICLE HIGHLIGHTS
Research background
Nonvariceal upper digestive bleeding (NVUDB) is a severe condition, associated with significant morbidity, mortality and health care resource use. Despite the progress in the treatment of NVUDB by using potent proton pump inhibitors and modern endoscopic devices, its mortality remains high. In recent years, considerable effort has been made to the identification of predictive factors for unfavorable outcome, defined by rebleeding, need of surgery and death.
Research motivation
The identification of prognostic factors of poor outcome and implementation of risk stratification systems helps to improve clinical management of the patients with NVUDB by optimal allocation of health care resources. This stratification allows an early discharge or outpatient management in low-risk patients, whereas high-risk patients benefit from an intensive therapeutic approach, needing endoscopic hemostasis and hospitalization.
Research objectives
The main objective of our study is to describe the particularities of patients with NVUDB, including demographic characteristics, clinical and endoscopic findings, treatment used, as well as to identify predictive factors of rebleeding and in-hospital mortality in patients with NVUDB admitted in an emergency hospital from western Romania.
Research methods
Our retrospective study included patients with NVUDB admitted in the Gastroenterology Department of Emergency County Hospital Timisoara, Romania, during 2008-2016. On this batch we analyzed the demographic data, medication history, clinical and biological parameters, endoscopic findings, type of endoscopic hemostasis used, the Rockall score of the patients, length of hospitalization and associated comorbidities. Also, we assessed the rate of unfavorable outcome in patients with NVUDB (rebleeding, surgery, death). Furthermore, we evaluated the potential risk factors associated with rebleeding and death in patients with NVUDB.
Research results
We assessed a batch of 1581 patients with NVUDB, 523 (33%) females and 1058 (67%) males, median age of 66 years. Peptic ulcer represented the most common etiology of NVUDB. The rate of rebleeding was 7.72%, surgery was performed in 3.22% of patients; the in-hospital mortality rate was 8.09%, whereas bleeding-episode-related mortality was 2.97%. Parameters significantly associated with an increased risk of rebleeding (using logistic regression) were Rockall score (OR: 1.41), need for therapy [simple (OR: 3.18) or combined (OR: 1.75)], number of blood units transfused (OR: 1.46), and sepsis (OR: 2.95). Logistic regression demonstrated that parameters significantly associated with death were Rockall score (OR: 1.73) and the following comorbidities: respiratory diseases (OR: 3.29), liver cirrhosis (OR: 2.91), sepsis (OR: 8.03) and acute pancreatitis (OR: 6.58). We performed a predictive model for rebleeding associated with an accuracy of 92.35%, and a predictive model for death, with an accuracy of 92.35%. Because the predictive models have a low sensitivity and a very high specificity, they provide a better discriminative capacity for identifying patients with NVUDB with favorable outcomes (no evolution towards rebleeding or death).
Research conclusions
Our study revealed that the risk factors for rebleeding were the Rockall score, need for endoscopic therapy, number of blood units transfused, and presence of sepsis. Our results showed that patients who died during hospitalization were significantly older, had a higher Rockall score, and a more severe anemia. The existence of severe comorbididies such as respiratory conditions, cirrhosis, sepsis, and acute pancreatitis were also risk factors for death in patients with NVUDB.
Research perspectives
In the near future we expect to be able to implement better strategies of risk stratification of NVUDB patients in our daily practice, using the clinical and endoscopic findings demonstrated as predictive parameters of poor outcome in our specific pool of patients. The continuous use of the risk stratification algorithms for the patients with NVUDB will lead to an improved management of these patients, a better quality of care and cost-efficiency of health resource use, and finally to a better prognosis of the patients diagnosed with this severe condition. The therapeutic approach of patients with NVUDB should involve a multidisciplinary team for the initial assessment of patients and for hemodynamic stabilization. The involvement of an experienced endoscopist and availability of modern endoscopic devices and techniques are essential for the proper management, prevention of rebleeding, reduction of hospitalization length, and minimization of morbidity and mortality. A strategy for improving the outcome of the patients should focus on managing coexisting diseases. Multidisciplinary teams for the management of NVUDB patients should include experienced nurses, gastroenterologists, surgeons, and other clinicians who can deal with comorbidities.
Footnotes
Institutional review board statement: This study was reviewed and approved by the Ethics Committee of the University of Medicine and Pharmacy “Victor Babeş”, Timişoara.
Informed consent statement: Patients were not required to give informed consent for the study because the analysis was retrospective, and it used anonymous clinical data that were obtained after each patient agreed to treatment by written consent.
Conflict-of-interest statement: The authors have no conflict of interest to report.
Data sharing statement: No additional data are available.
Manuscript source: Invited manuscript
Peer-review started: June 25, 2019
First decision: July 20, 2019
Article in press: August 26, 2019
Specialty type: Medicine, Research and Experimental
Country of origin: Romania
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Karatapanis S, Guerra I, Thomopoulos K S-Editor: Dou Y L-Editor: A E-Editor: Liu JH
Contributor Information
Daniela Cornelia Lazăr, Department of Internal Medicine I, University Medical Clinic, University of Medicine and Pharmacy “Victor Babeş”, Timişoara 300041, Timiş County, Romania. lazar.daniela@umft.ro.
Sorin Ursoniu, Department of Public Health and Health Management, University of Medicine and Pharmacy “Victor Babeş”, Timişoara 300041, Timiş County, Romania.
Adrian Goldiş, Department of Gastroenterology and Hepatology, University of Medicine and Pharmacy “Victor Babeş”, Timişoara 300041, Timiş County, Romania.
References
- 1.Barkun A, Sabbah S, Enns R, Armstrong D, Gregor J, Fedorak RN, Rahme E, Toubouti Y, Martel M, Chiba N, Fallone CA RUGBE Investigators. The Canadian Registry on Nonvariceal Upper Gastrointestinal Bleeding and Endoscopy (RUGBE): Endoscopic hemostasis and proton pump inhibition are associated with improved outcomes in a real-life setting. Am J Gastroenterol. 2004;99:1238–1246. doi: 10.1111/j.1572-0241.2004.30272.x. [DOI] [PubMed] [Google Scholar]
- 2.Barkun AN, Martel M, Toubouti Y, Rahme E, Bardou M. Endoscopic hemostasis in peptic ulcer bleeding for patients with high-risk lesions: a series of meta-analyses. Gastrointest Endosc. 2009;69:786–799. doi: 10.1016/j.gie.2008.05.031. [DOI] [PubMed] [Google Scholar]
- 3.Marmo R, Koch M, Cipolletta L, Capurso L, Pera A, Bianco MA, Rocca R, Dezi A, Fasoli R, Brunati S, Lorenzini I, Germani U, Di Matteo G, Giorgio P, Imperiali G, Minoli G, Barberani F, Boschetto S, Martorano M, Gatto G, Amuso M, Pastorelli A, Torre ES, Triossi O, Buzzi A, Cestari R, Della Casa D, Proietti M, Tanzilli A, Aragona G, Giangregorio F, Allegretta L, Tronci S, Michetti P, Romagnoli P, Nucci A, Rogai F, Piubello W, Tebaldi M, Bonfante F, Casadei A, Cortini C, Chiozzini G, Girardi L, Leoci C, Bagnalasta G, Segato S, Chianese G, Salvagnini M, Rotondano G. Predictive factors of mortality from nonvariceal upper gastrointestinal hemorrhage: a multicenter study. Am J Gastroenterol. 2008;103:1639–47; quiz 1648. doi: 10.1111/j.1572-0241.2008.01865.x. [DOI] [PubMed] [Google Scholar]
- 4.Sung JJ, Chiu PW, Chan FKL, Lau JY, Goh KL, Ho LH, Jung HY, Sollano JD, Gotoda T, Reddy N, Singh R, Sugano K, Wu KC, Wu CY, Bjorkman DJ, Jensen DM, Kuipers EJ, Lanas A. Asia-Pacific working group consensus on non-variceal upper gastrointestinal bleeding: an update 2018. Gut. 2018;67:1757–1768. doi: 10.1136/gutjnl-2018-316276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Barkun AN, Bardou M, Kuipers EJ, Sung J, Hunt RH, Martel M, Sinclair P International Consensus Upper Gastrointestinal Bleeding Conference Group. International consensus recommendations on the management of patients with nonvariceal upper gastrointestinal bleeding. Ann Intern Med. 2010;152:101–113. doi: 10.7326/0003-4819-152-2-201001190-00009. [DOI] [PubMed] [Google Scholar]
- 6.Travis AC, Wasan SK, Saltzman JR. Model to predict rebleeding following endoscopic therapy for non-variceal upper gastrointestinal hemorrhage. J Gastroenterol Hepatol. 2008;23:1505–1510. doi: 10.1111/j.1440-1746.2008.05594.x. [DOI] [PubMed] [Google Scholar]
- 7.Sung JJ, Tsoi KK, Ma TK, Yung MY, Lau JY, Chiu PW. Causes of mortality in patients with peptic ulcer bleeding: a prospective cohort study of 10,428 cases. Am J Gastroenterol. 2010;105:84–89. doi: 10.1038/ajg.2009.507. [DOI] [PubMed] [Google Scholar]
- 8.Zhang JY, Wang Y, Zhang J, Ding SG, Zhou LY, Lin SR. [Risk factors associated with failure from endoscopic therapy in acute non-variceal upper gastrointestinal bleeding] Beijing Da Xue Xue Bao Yi Xue Ban. 2010;42:703–707. [PubMed] [Google Scholar]
- 9.González-González JA, Vázquez-Elizondo G, García-Compeán D, Gaytán-Torres JO, Flores-Rendón ÁR, Jáquez-Quintana JO, Garza-Galindo AA, Cárdenas-Sandoval MG, Maldonado-Garza HJ. Predictors of in-hospital mortality in patients with non-variceal upper gastrointestinal bleeding. Rev Esp Enferm Dig. 2011;103:196–203. doi: 10.4321/s1130-01082011000400005. [DOI] [PubMed] [Google Scholar]
- 10.Morales Uribe CH, Sierra Sierra S, Hernández Hernández AM, Arango Durango AF, López GA. Upper gastrointestinal bleeding: risk factors for mortality in two urban centres in Latin America. Rev Esp Enferm Dig. 2011;103:20–24. doi: 10.4321/s1130-01082011000100004. [DOI] [PubMed] [Google Scholar]
- 11.Nahon S, Hagège H, Latrive JP, Rosa I, Nalet B, Bour B, Faroux R, Gower P, Arpurt JP, Denis J, Henrion J, Rémy AJ, Pariente A Groupe des Hémorragies Digestives Hautes de l’ANGH. Epidemiological and prognostic factors involved in upper gastrointestinal bleeding: results of a French prospective multicenter study. Endoscopy. 2012;44:998–1008. doi: 10.1055/s-0032-1310006. [DOI] [PubMed] [Google Scholar]
- 12.Del Piano M, Bianco MA, Cipolletta L, Zambelli A, Chilovi F, Di Matteo G, Pagliarulo M, Ballarè M, Rotondano G Prometeo study group of the Italian Society of Digestive Endoscopy (SIED) The "Prometeo" study: online collection of clinical data and outcome of Italian patients with acute nonvariceal upper gastrointestinal bleeding. J Clin Gastroenterol. 2013;47:e33–e37. doi: 10.1097/MCG.0b013e3182617dcc. [DOI] [PubMed] [Google Scholar]
- 13.Taha AS, Saffouri E, McCloskey C, Craigen T, Angerson WJ. Falling mortality when adjusted for comorbidity in upper gastrointestinal bleeding: relevance of multi-disciplinary care. Frontline Gastroenterol. 2014;5:243–248. doi: 10.1136/flgastro-2014-100453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Marmo R, Koch M, Cipolletta L, Bianco MA, Grossi E, Rotondano G PNED 1 and PNED 2 Investigators. Predicting mortality in patients with in-hospital nonvariceal upper GI bleeding: a prospective, multicenter database study. Gastrointest Endosc. 2014;79:741–749.e1. doi: 10.1016/j.gie.2013.10.009. [DOI] [PubMed] [Google Scholar]
- 15.Marmo R, Koch M, Cipolletta L, Capurso L, Grossi E, Cestari R, Bianco MA, Pandolfo N, Dezi A, Casetti T, Lorenzini I, Germani U, Imperiali G, Stroppa I, Barberani F, Boschetto S, Gigliozzi A, Gatto G, Peri V, Buzzi A, Della Casa D, Di Cicco M, Proietti M, Aragona G, Giangregorio F, Allegretta L, Tronci S, Michetti P, Romagnoli P, Piubello W, Ferri B, Fornari F, Del Piano M, Pagliarulo M, Di Mitri R, Trallori G, Bagnoli S, Frosini G, Macchiarelli R, Sorrentini I, Pietrini L, De Stefano S, Ceglia T, Chiozzini G, Salvagnini M, Di Muzio D, Rotondano G Italian registry on upper gastrointestinal bleeding (Progetto Nazionale Emorragie Digestive--PNED 2) Predicting mortality in non-variceal upper gastrointestinal bleeders: validation of the Italian PNED Score and Prospective Comparison with the Rockall Score. Am J Gastroenterol. 2010;105:1284–1291. doi: 10.1038/ajg.2009.687. [DOI] [PubMed] [Google Scholar]
- 16.Lee YJ, Min BR, Kim ES, Park KS, Cho KB, Jang BK, Chung WJ, Hwang JS, Jeon SW. Predictive factors of mortality within 30 days in patients with nonvariceal upper gastrointestinal bleeding. Korean J Intern Med. 2016;31:54–64. doi: 10.3904/kjim.2016.31.1.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hwang S, Jeon SW, Kwon JG, Lee DW, Ha CY, Cho KB, Jang B, Park JB, Park YS Daegu-Gyengbuk Gastrointestinal Study Group (DGSG) The Novel Scoring System for 30-Day Mortality in Patients with Non-variceal Upper Gastrointestinal Bleeding. Dig Dis Sci. 2016;61:2002–2010. doi: 10.1007/s10620-016-4087-4. [DOI] [PubMed] [Google Scholar]
- 18.Jurado Hernández AM, de Teresa Galván J, Ruiz-Cabello Jiménez M, Pinel Julián LM. [Evolution in the epidemiology of non-variceal upper digestive hemorrhage from 1985 to 2006] Rev Esp Enferm Dig. 2008;100:273–277. doi: 10.4321/s1130-01082008000500005. [DOI] [PubMed] [Google Scholar]
- 19.Enestvedt BK, Gralnek IM, Mattek N, Lieberman DA, Eisen G. An evaluation of endoscopic indications and findings related to nonvariceal upper-GI hemorrhage in a large multicenter consortium. Gastrointest Endosc. 2008;67:422–429. doi: 10.1016/j.gie.2007.09.024. [DOI] [PubMed] [Google Scholar]
- 20.Hearnshaw SA, Logan RF, Lowe D, Travis SP, Murphy MF, Palmer KR. Use of endoscopy for management of acute upper gastrointestinal bleeding in the UK: results of a nationwide audit. Gut. 2010;59:1022–1029. doi: 10.1136/gut.2008.174599. [DOI] [PubMed] [Google Scholar]
- 21.Bianco MA, Rotondano G, Marmo R, Piscopo R, Orsini L, Cipolletta L. Combined epinephrine and bipolar probe coagulation vs. bipolar probe coagulation alone for bleeding peptic ulcer: a randomized, controlled trial. Gastrointest Endosc. 2004;60:910–915. doi: 10.1016/s0016-5107(04)02232-1. [DOI] [PubMed] [Google Scholar]
- 22.Bjorkman DJ, Zaman A, Fennerty MB, Lieberman D, Disario JA, Guest-Warnick G. Urgent vs. elective endoscopy for acute non-variceal upper-GI bleeding: an effectiveness study. Gastrointest Endosc. 2004;60:1–8. doi: 10.1016/s0016-5107(04)01287-8. [DOI] [PubMed] [Google Scholar]
- 23.Cipolletta L, Cipolletta F, Granata A, Ligresti D, Barresi L, Tarantino I, Traina M. What Is the Best Endoscopic Strategy in Acute Non-variceal Gastrointestinal Bleeding? Curr Treat Options Gastroenterol. 2018;16:363–375. doi: 10.1007/s11938-018-0192-0. [DOI] [PubMed] [Google Scholar]
- 24.Yachimski PS, Friedman LS. Gastrointestinal bleeding in the elderly. Nat Clin Pract Gastroenterol Hepatol. 2008;5:80–93. doi: 10.1038/ncpgasthep1034. [DOI] [PubMed] [Google Scholar]
- 25.Kim BJ, Park MK, Kim SJ, Kim ER, Min BH, Son HJ, Rhee PL, Kim JJ, Rhee JC, Lee JH. Comparison of scoring systems for the prediction of outcomes in patients with nonvariceal upper gastrointestinal bleeding: a prospective study. Dig Dis Sci. 2009;54:2523–2529. doi: 10.1007/s10620-008-0654-7. [DOI] [PubMed] [Google Scholar]
- 26.Chen IC, Hung MS, Chiu TF, Chen JC, Hsiao CT. Risk scoring systems to predict need for clinical intervention for patients with nonvariceal upper gastrointestinal tract bleeding. Am J Emerg Med. 2007;25:774–779. doi: 10.1016/j.ajem.2006.12.024. [DOI] [PubMed] [Google Scholar]
- 27.Rockall TA, Logan RF, Devlin HB, Northfield TC. Risk assessment after acute upper gastrointestinal haemorrhage. Gut. 1996;38:316–321. doi: 10.1136/gut.38.3.316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Trawick EP, Yachimski PS. Management of non-variceal upper gastrointestinal tract hemorrhage: controversies and areas of uncertainty. World J Gastroenterol. 2012;18:1159–1165. doi: 10.3748/wjg.v18.11.1159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gu L, Xu F, Yuan J. Comparison of AIMS65, Glasgow-Blatchford and Rockall scoring approaches in predicting the risk of in-hospital death among emergency hospitalized patients with upper gastrointestinal bleeding: a retrospective observational study in Nanjing, China. BMC Gastroenterol. 2018;18:98. doi: 10.1186/s12876-018-0828-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Stanley AJ, Dalton HR, Blatchford O, Ashley D, Mowat C, Cahill A, Gaya DR, Thompson E, Warshow U, Hare N, Groome M, Benson G, Murray W. Multicentre comparison of the Glasgow Blatchford and Rockall Scores in the prediction of clinical end-points after upper gastrointestinal haemorrhage. Aliment Pharmacol Ther. 2011;34:470–475. doi: 10.1111/j.1365-2036.2011.04747.x. [DOI] [PubMed] [Google Scholar]
- 31.Pang SH, Ching JY, Lau JY, Sung JJ, Graham DY, Chan FK. Comparing the Blatchford and pre-endoscopic Rockall score in predicting the need for endoscopic therapy in patients with upper GI hemorrhage. Gastrointest Endosc. 2010;71:1134–1140. doi: 10.1016/j.gie.2010.01.028. [DOI] [PubMed] [Google Scholar]
- 32.Chandra S, Hess EP, Agarwal D, Nestler DM, Montori VM, Song LM, Wells GA, Stiell IG. External validation of the Glasgow-Blatchford Bleeding Score and the Rockall Score in the US setting. Am J Emerg Med. 2012;30:673–679. doi: 10.1016/j.ajem.2011.03.010. [DOI] [PubMed] [Google Scholar]
- 33.Dicu D, Pop F, Ionescu D, Dicu T. Comparison of risk scoring systems in predicting clinical outcome at upper gastrointestinal bleeding patients in an emergency unit. Am J Emerg Med. 2013;31:94–99. doi: 10.1016/j.ajem.2012.06.009. [DOI] [PubMed] [Google Scholar]
- 34.Chaikitamnuaychok R, Patumanond J. Upper Gastrointestinal Hemorrhage: Validation of the Severity Score. Gastroenterology Res. 2013;6:56–62. doi: 10.4021/gr540w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Le Jeune IR, Gordon AL, Farrugia D, Manwani R, Guha IN, James MW. Safe discharge of patients with low-risk upper gastrointestinal bleeding (UGIB): can the use of Glasgow-Blatchford Bleeding Score be extended? Acute Med. 2011;10:176–181. [PubMed] [Google Scholar]
- 36.Soncini M, Triossi O, Leo P, Magni G, Bertelè AM, Grasso T, Ferraris L, Caruso S, Spadaccini A, Brambilla G, Verta M, Muratori R, Attinà A, Grasso G. Management of patients with nonvariceal upper gastrointestinal hemorrhage before and after the adoption of the Rockall score, in the Italian Gastroenterology Units. Eur J Gastroenterol Hepatol. 2007;19:543–547. doi: 10.1097/MEG.0b013e3281532b89. [DOI] [PubMed] [Google Scholar]
- 37.Shrestha UK, Sapkota S. Etiology and adverse outcome predictors of upper gastrointestinal bleeding in 589 patients in Nepal. Dig Dis Sci. 2014;59:814–822. doi: 10.1007/s10620-013-2946-9. [DOI] [PubMed] [Google Scholar]
- 38.Wang CY, Qin J, Wang J, Sun CY, Cao T, Zhu DD. Rockall score in predicting outcomes of elderly patients with acute upper gastrointestinal bleeding. World J Gastroenterol. 2013;19:3466–3472. doi: 10.3748/wjg.v19.i22.3466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Crooks CJ, West J, Card TR. Comorbidities affect risk of nonvariceal upper gastrointestinal bleeding. Gastroenterology. 2013;144:1384–1393, 1393.e1-2; quiz e18-9. doi: 10.1053/j.gastro.2013.02.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Crooks CJ, Card TR, West J. Excess long-term mortality following non-variceal upper gastrointestinal bleeding: a population-based cohort study. PLoS Med. 2013;10:e1001437. doi: 10.1371/journal.pmed.1001437. [DOI] [PMC free article] [PubMed] [Google Scholar]