Abstract
Recent studies on mice with null mutation of the angiotensin type 2 receptor (AT2R) gene have implicated the involvement of AT2R in regulating adipocyte size and obesity, a major risk factor for metabolic syndrome. However, the outcome from these studies remains inconclusive. Therefore, current study was designed to test whether pharmacological activation of AT2R regulates adiposity and lipid metabolism. Male mice (5-weeks old) were pre-treated with vehicle or AT2R agonist (C21, 0.3 mg/kg, i.p., daily, for 4 days) and fed normal diet (ND). Then these animals were subdivided into ND and high-fat diet (HFD) regimen and concomitantly treated with vehicle or C21 through day 14. Vehicle-treated HFD-fed mice demonstrated an increase in epididymal white adipose tissue (eWAT) weight and adipocyte size, which were associated with increased eWAT expression of the lipogenic regulators, fatty acid binding protein and fatty acid synthase, decreased expression of adipose triglyceride lipase and increased expression of hormone-sensitive lipase. Interestingly, C21 pre-treatment altered HFD-induced changes in lipogenic and lipolytic regulators. C21 pre-treatment prevented decrease in expression of uncoupler protein-1 in brown adipose in HFD-fed mice, which was associated with increased core temperature. In addition, C21 pre-treatment ameliorated plasma free fatty acids, triglycerides, insulin and tumor necrosis factor-α in HFD fed mice. Ex-vivo study in isolated primary epididymal adipocytes revealed that C21 inhibits long chain fatty acid transporter, via a nitric oxide synthase/guanylate cyclase/protein kinase G-dependent pathway. Collectively, we propose pharmacological activation of AT2R regulates fatty acid metabolism and thermogenesis and prevents HFD-induced adiposity in mice.
Keywords: Adipocyte, AT2R, fatty acid transporter (FATP), lipid metabolism, adiposity
Introduction
Obesity, characterized by increase in body weight and adiposity, is a major risk factor for the development of dyslipidemia, atherosclerosis, insulin resistance, diabetes and hypertension [1–5]. The incidence of obesity has been increasing worldwide and is now considered a global epidemic. According to the international guidelines, the lifestyle modification (decreased caloric intake and increased physical exercise) is a primary modality of body weight management [6]. However, the long-term effects of diet and exercise on weight have been mostly disappointing [7]. Further strategies for body weight management include pharmacological intervention and bariatric surgery [8]. But most anti-obesity drugs are associated with serious side effects [9]. Thus, there has been a pressing need for the discovery of newer anti-obesity drugs.
Lipogenesis, lipolysis and thermogenesis are some determinants of body weight gain and adiposity. Under fed condition, the surplus or extra energy from lipid is stored in adipocytes as triglycerides (TAG) for future use [10]. As and when the energy demand increases, the stored TAGs are broken down to free fatty acids. Fatty acid binding protein-4 (FABP4) and fatty acid synthase (FASN) (lipogenic), adipocyte triglyceride lipase (ATGL) and hormone sensitive lipase (HSL) (lipolytic) and uncoupling protein-1 (UCP1) (thermogenic) are some of the enzymic regulators of these processes. Fatty acid transporter protein (FATP) is a family of plasma membrane transporters regulating long chain fatty acids (LCFA) uptake into adipocytes [11]. Knockdown of FATP1, which is an insulin sensitive LCFA transporter, prevents HFD-induced resistance in skeletal muscle [12]. Also, the FATP4 expression in human adipose tissue positively correlates with obesity and insulin resistance [13].
Although renin angiotensin system (RAS) is mostly known for its role in cardiovascular function and blood pressure regulation, it has been recently implicated to have a role in lipid metabolism and adiposity [14–16]. Adipocytes are known to have all the components of the RAS. In high-fat diet (HFD)-induced obesity, an increased formation of angiotensin-II (ang-II) in adipose tissue has been reported. Ang-II is thus considered as a trophic factor for the adipose tissue growth. It acts via angiotensin type 1 and type 2 receptors (AT1R and AT2R). The physiologic effects of these receptors generally are antagonistic [17, 18]. While the blocking of AT1R decreases adipocyte size in mice [19], the role of AT2R in adiposity is not yet established. Recent studies demonstrated that the genetic deletion of AT2R resulted in a decrease in adiposity in mice [20, 21]. On the contrary, deficiency of AT2R Apolipoprotein E-knockout (KO) mice (a model of atherosclerosis) increased adipose tissue weight in mice [22]. Thus, the role of AT2R on adiposity in knockout studies has remained inconclusive. Therefore, we designed the current study to investigate the effects of the AT2R agonist C21, a novel non-peptide and orally active compound, on lipid metabolism and adiposity.
Materials and methods
Animals and experimental protocols
Twenty four male C57BL/6 mice (5-week-old) were obtained from Harlan (Indianapolis, IN). The mice were housed at the University of Houston’s animal care facility with free access to food and water and maintained under a 12-hr light/dark cycle. The animal experimental protocols were approved by Institutional Animal Care and Use Committee and performed in accordance with the National Institutes of Health Guidelines.
Mice were randomized and pre-treated with ‘Compound 21’ (C21) at a dose of 0.3 mg/kg [23, 24] or equivolume of normal saline (i.p., daily, days 1–4 at 10 am) and fed normal diet (29% protein, 56% carbohydrate and 15% fat) (referred as ND, 7022). Thereafter the mice on C21 and vehicle were further sub-divided into two groups: ND or high-fat diet (18.4% protein, 21.3% carbohydrate and 60.3% fat) (referred as HFD, TD.06414) for the next 10 days along with concurrent treatment with saline or C21. Sodium salt of C21 (PubChem CID: 9804984) was provided as a gift from Vicore Pharma, Sweden. Food intake and body weight of the mice were measured as reported previously [21]. Core temperature was measured by mice rectal probe (RET-3) and thermocouple meter (WD-35627–00) (Kent Scientific corp., Torrington, CT) on terminal days 12–14 in morning immediately after drug administration. At the end of day 14, mice were euthanized in non-fasting state by cervical dislocation under isoflurane anesthesia. Plasma and brown adipose tissue (BAT) were collected and stored at −80°C until further use. Epididymal white adipose tissue (eWAT) pads were removed, patted dry, weighed and a part of the tissue was preserved in buffered formalin for histology. The remaining eWAT was stored frozen at −80°C for biochemical measurements. Details on various assay methods are available in the online-only Data Supplement.
Statistical Analysis
The data were analyzed using GraphPad Prism (GraphPad software, San Diego, CA). In vitro results are subjected to one-way ANOVA with Newman-Keuls post-hoc test and unpaired Student’s t test. To determine diet*treatment interaction, in vivo results are subjected to two-way ANOVA followed by Fisher’s LSD test at p < 0.05 to be considered statistically significant. Data are presented as mean±SEM.
Results
Calorie intake and body weight:
Food intake was calculated in terms of total kilo calorie (Kcal) consumed during 2 weeks of treatment period. As shown in Table 1, HFD-fed mice had significantly higher Kcal intake in 2 weeks (ND 64±4.0 vs HFD 130±5.7 Kcal) compared with ND group. The C21 pre-treatment did not affect the Kcal intake under ND or HFD conditions in these mice. Body weight gain during the treatment period remained same in all the groups (Table 1). We did not observe adverse events related to animals’ health during the study.
Table 1.
Metabolic and hormonal parameters and rectal body temperature of control and C21-treated 5 week old male C57BL/6 mice fed with ND or HFD.
| Parameters/Mice groups | ND | ND-C21 | HFD | HFD-C21 | p value | ||
|---|---|---|---|---|---|---|---|
| Diet | Treatment | Interaction | |||||
| Total Kcal intake (2 weeks) | 64±4 | 55±5 | 130±6* | 133±7 | 0.0001 | n.s. | n.s. |
| Body weight gain (g) (terminal-initial) | 3.43±0.95 | 3.38±0.73 | 4.13±0.95 | 3.38±0.53 | n.s. | n.s. | n.s. |
| Initial body weight (g) | 17.00±1.33 | 16.88±1.19 | 16.00±1.30 | 16.75±0.56 | n.s. | n.s. | n.s. |
| Terminal body weight (g) | 20.43±0.57 | 20.29±0.81 | 20.13±0.44 | 20.13±0.44 | n.s. | n.s. | n.s. |
| Plasma FFA (nmol/µL) | 2.30±0.20 | 2.30±0.18 | 5.24±0.36* | 3.27±0.19# | 0.0001 | 0.0002 | 0.0002 |
| Plasma TAG (nmol/µL) | 1.96±0.23 | 2.20±0.34 | 4.03±0.25* | 3.30±0.18# | 0.0001 | n.s. | 0.0408 |
| Plasma insulin (ng/mL) | 0.78±0.06 | 0.73±0.06 | 2.34±0.09* | 1.33±0.19# | 0.0001 | 0.0001 | 0.0004 |
| Plasma TNF-α (pg/mL) | 35±3 | 34±4 | 72±16* | 43±4# | 0.0147 | n.s. | n.s. |
| Plasma adiponectin (ng/mL) | 4.41±0.19 | 3.60±0.55 | 2.68±0.43* | 5.24±0.68# | n.s. | n.s. | 0.0029 |
| Rectal body temperature days 12–14 (°C) | 37.01±0.19 | 37.29±0.18 | 37.39±0.13 | 38.04±0.13# | 0.0021 | 0.0087 | n.s. |
ND - Normal Diet, ND-C21 - Normal Diet on C21, HFD - High-Fat Diet, HFD-C21 - High- Fat Diet on C21
-significantly different from ND
-significantly different from HFD; data were analyzed using two-way ANOVA with Fisher’s LSD test at p<0.05.
Epididymal WAT weight/body weight ratio and epididymal adipocyte size:
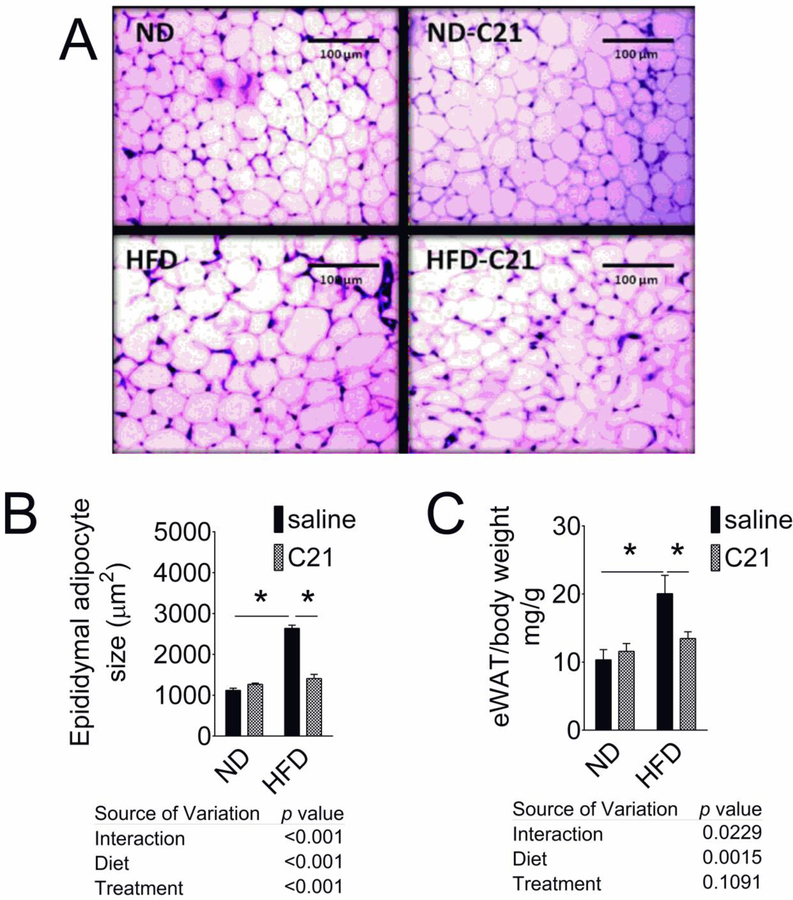
The H&E staining of the eWAT sections from control and C21-treated mice on ND and HFD is shown in Fig. 1A. Compared with ND, HFD caused an increase in epididymal adipocyte size (ND 1118±54 vs HFD 2636±79 μm2), which was reduced by C21-treatment (1407±102 μm2) (Fig. 1B). C21 treatment of ND-fed mice had no effect on the weight ratio or the adipocyte size. High fat diet caused significant increase only in the eWAT weight/body weight ratio (Fig. 1C), while other fat depots weight remained unchanged among treatment groups (SI Table 1). C21-treatment reduced the HFD-induced increase in eWAT and eWAT weight/body weight ratio (by 34%).
Fig. 1. Epididymal WAT weight-to-body weight ratio and adipocyte size of control and C21-treated mice fed with ND or HFD.
(A) Epididymal WAT weight-to-body weight ratio; N=6–8 in each group. (B) Representative micrograph of epididymal adipose tissue (magnification, 10X). (C) Epididymal adipocyte size was expressed as cell area (µm2); N=3 (3 sections per slide from 3 mice). Results are mean+SEM; *significantly different compared with ND, #significantly different compared with HFD. Data were analyzed using two-way ANOVA followed by Fisher’s LSD test at p < 0.05. (ND- Normal Diet, ND-C21- Normal Diet on C21, HFD- High-Fat Diet, HFD-C21- High-Fat Diet on C21).
Plasma FFA and TAG:
The HFD fed mice had significantly increased levels of plasma FFA levels (ND 2.30±0.20 vs HFD 5.24±0.36 nmol/μL), which were reduced by C21-treatment (HFD-C21 3.27±0.19 nmol/μL) (Table 1). Similarly, HFD-feeding increased the plasma TAG levels, which were attenuated by C21 treatment (HFD 4.03±0.25 vs HFD-C21 3.3±0.18 nmol/μL). Both TAG and FFA in ND-fed mice were not affected by C21 treatment (Table 1).
Plasma insulin, TNF-α and adiponectin:
The plasma insulin level (non-fasting) was nearly three-fold higher in HFD fed mice (ND 0.78±0.06 vs HFD 2.34±0.09 ng/mL). This increase in plasma insulin in HFD-fed mice was attenuated (HFD-C21 1.33±0.19 ng/mL) by C21 treatment, which had no effect in ND-fed mice (Table 1). The HFD feeding significantly increased plasma TNF-α levels (ND 35±2.6 vs HFD 72±15.8 pg/mL), which were significantly reduced (HFD-C21 43±4.5 pg/mL) by C21 treatment (Table 1). On the other hand, plasma adiponectin was significantly decreased by HFD feeding compared with ND (ND 4.41±0.19 vs HFD 2.68±0.43 ng/mL). The C21 treatment significantly increased the adiponectin levels (HFD-C21 5.24±0.68 ng/mL) under HFD condition but had no effect in the ND-fed mice (Table 1).
Lipogenic regulators’ protein expression in eWAT:
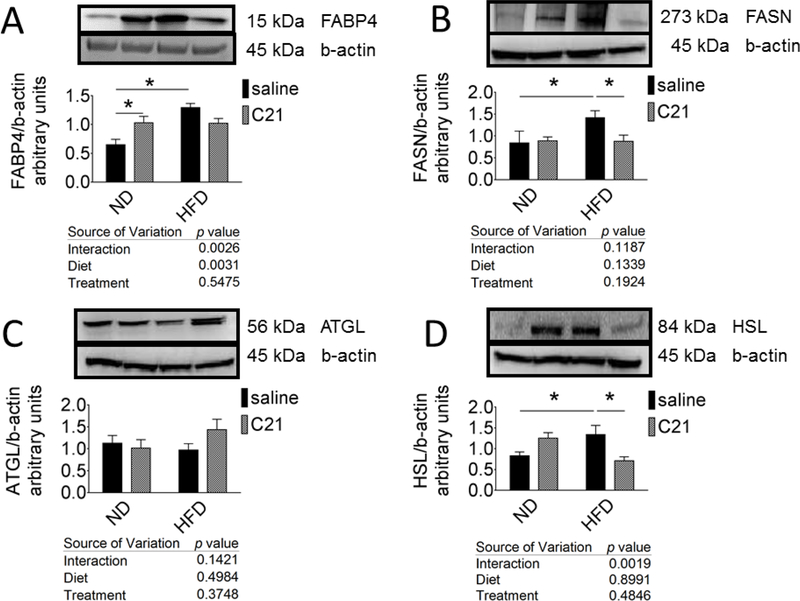
Western blotting revealed the presence of the lipogenic regulators fatty acid binding protein-4 (FABP4) (15 KDa) and fatty acid synthase (FASN) (273 KDa) protein expression in the eWAT. Densitometric analysis of the bands suggested that FABP4 (99%) and FASN (68%) expression in eWAT in HFD were significantly greater than ND (Fig. 2A and 2B respectively). C21-treatment under HFD condition reduced the expression of both the proteins - FABP4 (by 26%) and FASN (by 62%).
Fig. 2. Protein expression of lipogenic and lipolytic regulators in the epididymal WAT of control and C21-treated mice fed with ND or HFD.
Representative western blots for (A) FABP4, (B) FASN, (C) ATGL and (D) HSL with loading control β-actin. The bar graphs represent the ratios of densities of FABP4, FASN, ATGL and HSL normalized with β-actin protein bands. *significantly different compared with ND, #significantly different compared with HFD. Results are mean+SEM; Data were analyzed using two-way ANOVA followed by Fisher’s LSD test at p < 0.05; N=4–6 in each group. (ND- Normal Diet, ND-C21- Normal Diet on C21, HFD- High-Fat Diet, HFD- C21- High-Fat Diet on C21).
Lipolytic regulators’ protein expression in eWAT:
Distinct bands for adipose triglyceride lipase (ATGL) and hormone sensitive lipase (HSL) were detected approximately at 56 KDa and 84 KDa, respectively, in the eWAT by western blotting. Densitometric analysis of the bands revealed that compared with ND, HFD fed mice expressed reduced ATGL (16%) and increased HSL (62%) proteins (Fig. 2C and 2D, respectively). C21 treatment of HFD fed mice caused an increase in the ATGL expression (47%) and decreased the HSL expression (52%). Surprisingly, C21 treatment under ND condition significantly increased the protein expression of HSL (50%) (Fig. 2D).
Fatty acid-uptake by isolated primary epididymal adipocytes:
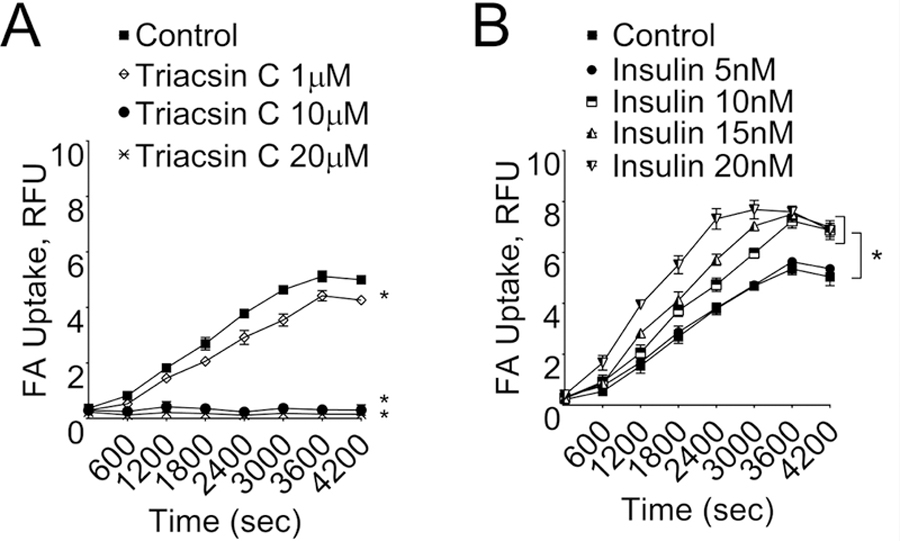
With the increasing number of epididymal adipocytes in the assay tube, there was an increase in the FA-uptake, which plateaued off at ~3600 seconds after initiating the assay. After conducting preliminary experiments, we chose 40,000 cells/assay for further experiments and terminated the assay at 4200 sec. The FA-uptake was inhibited by the FATP inhibitor, triacsin C (Fig. 3A) in a concentration (1–20 μM) dependent manner suggesting the involvement of FATP. Further, FA-uptake was stimulated by insulin in a dose-dependent manner (Fig. 3B), with a significant increase at ≥10 nM concentrations of insulin.
Fig. 3. Fatty acid uptake in isolated primary epididymal adipocytes assessed using fluorescence assay.
(A) In absence or presence of different concentrations (1, 10 and 20 μM) of triacsin C, the FATP-inhibitor and (B) in absence or presence of different concentrations (5, 10, 15 and 20 nM) of insulin, the FATP-activator. *significantly different control adipocytes (without triacsin C treatment) (A), *-significantly different control adipocytes (without insulin treatment) (B). Results are mean+SEM; Data were analyzed using one-way ANOVA followed by Neuman-Keuls post hoc test and Student’s t test, p<0.05; N=3 in each group.
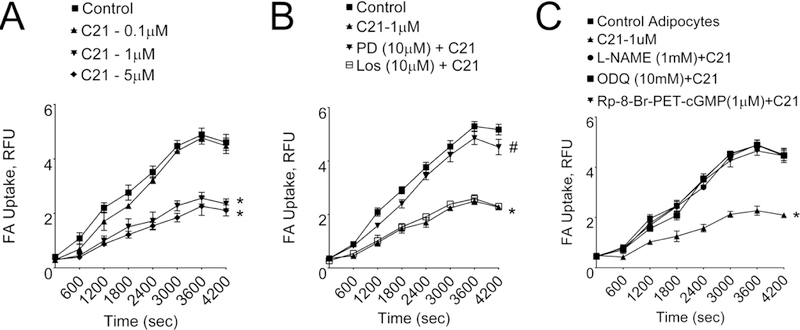
Adipocyte FA-uptake:
Figure 4 depicts FA-uptake was significantly decreased by C21 at 1 μM concentration. A lower dose (0.1 μM) of C21 was not effective and a higher dose (5 μM) did not cause any further reduction in FA uptake (Fig. 4A). In subsequent experiments, we used 1 μM of C21. The C21-induced decrease in FA uptake was blocked by the AT2R antagonist PD123319 (10 μM), but not by the AT1R antagonist losartan (1 μM) (Fig. 4B) suggesting the involvement of AT2R. In order to understand the potential role of NO/cGMP signaling pathway in AT2R-mediated inhibition of FA-uptake in adipocytes, we utilized specific inhibitors. The C21 inhibition of FA uptake was blocked by the NO synthase (NOS) inhibitor L-NAME (1 mM), the guanylyl cyclase (GC) inhibitor ODQ (10 mM) or the cGMP-dependent protein kinase-G (PKG)-inhibitor Rp-8-Br-PET-cGMPS (1 μM) (Fig. 4C).
Fig. 4. Fatty acid uptake in isolated primary epididymal adipocytes assessed using fluorescence assay.
(A) in absence or presence of different concentrations (0.1, 1.0 and 5.0 μM) of C21, the AT2R agonist, (B) in absence or presence of C21 (AT2R agonist), presence of PD (PD123,319 - AT2R antagonist)+C21 and presence of losartan (AT1R antagonist)+C21, and (C) in absence and presence of C21 (AT2R agonist), presence of L-NAME (NO synthase inhibitor)+C21, presence of ODQ (guanylyl cyclase inhibitor)+C21 and presence of Rp-8-Br-PET-Cgmps (PKG-inhibitor)+C21. *significantly different control adipocytes (without C21 treatment), #significantly different from C21 treatment group. Results are mean+SEM; Data were analyzed using one-way ANOVA followed by Neuman-Keuls post hoc test and Student’s t test, p<0.05; N=3 in each group. L-NAME-L-NG-Nitroarginine Methyl Ester; ODQ-1H-[1,2,4]Oxadiazolo[4,3-a]quinoxalin-1-one; PKG-I-PKG-inhibitor / Rp-8-Br-PET-cGMPS.
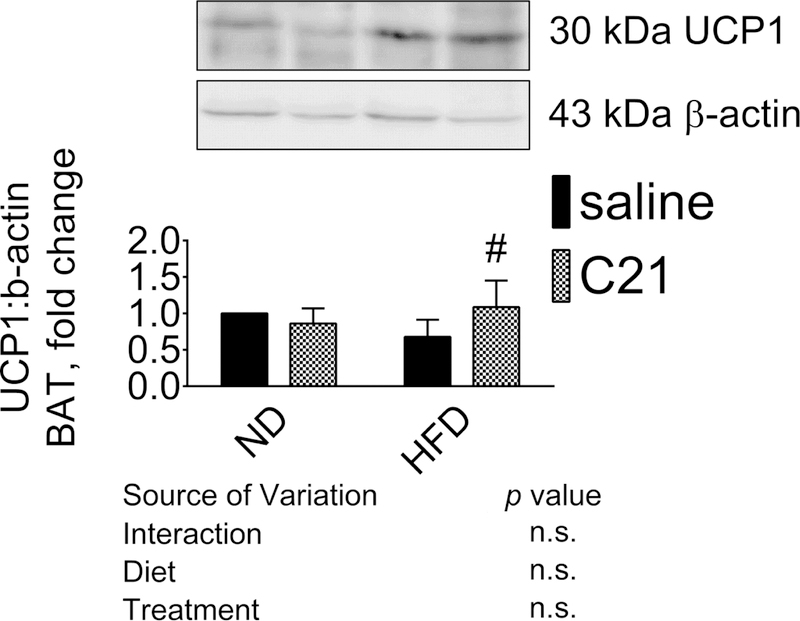
Core temperature and expression of UCP1 in BAT:
The treatment of AT2R agonist C21 significantly increased body temperature in HFD fed mice, but not in ND-fed mice (Table 1). HFD-feeding did not affect body temperature as compared to ND-fed mice. The expression of UCP1 was detected at 30 KDa in BAT (Fig. 5). The BAT expression of UCP1 was lower in HFD-fed mice (~30%) and C21 treatment of these mice attenuated the decrease in UCP-1 expression.
Fig. 5. Protein expression of uncoupling protein 1 (UCP1) in brown adipose tissue of control and C21-treated mice fed with ND or HFD.
Representative western blots of UCP1 with loading control β-actin. The bar graphs represent the ratios of densities of UCP1 normalized with β-actin protein bands. *significantly different compared with ND; #significantly different compared with HFD at n=13, 10% alpha error level and 50% beta error level. Results are mean+SEM; Data were analyzed using two-way ANOVA followed by Fisher’s LSD test at p < 0.05; N=6 in each group. (ND-Normal Diet, ND- C21- Normal Diet on C21, HFD- High-Fat Diet, HFD-C21- High-Fat Diet on C21).
Discussion
This is a short-term study where 2-weeks HFD regimen, as expected, had no effect on body weight gain yet could initiate metabolic changes [25]. Numerous studies including ours suggest that 6–8 weeks HFD regimen is needed to cause a significant shift in body weight [21]. However, this regimen clearly shows various metabolic parameters were affected by HFD and the AT2R agonist C21 treatment significantly prevented those changes. Particularly findings revealed that AT2R agonist treatment of HFD fed mice prevented increase in adipocyte size, improved plasma indices of dyslipidemia, which were associated with the changes in expression of lipogenic (FABP4 and FASN), lipolytic (ATGL and HSL) and thermogenic (UCP1) enzymes. Additionally, the AT2R agonist C21 inhibits primary adipocyte FA uptake via NO/cGMP pathway, suggesting adipose tissue as a potential site of AT2R action affecting FA transport and metabolism. The size of the adipocyte is built-up by the process of lipogenesis. In the postprandial phase, the FAs are first transported inside the adipocytes by the FATPs [26]. Once inside the aqueous cytosol, FFAs are then bound to the FABP4 for cytosolic transportation [27]. These FFAs along with the de-novo synthesized FFAs from the Kreb’s cycle (by FASN) are packed and stored as TAG in the lipid droplets of adipocytes [28]. An earlier study has reported that AT2R-KO mice fed HFD had smaller adipocytes and decreased expression of FABP4 and FASN gene [20]. In the present study we observed, HFD significantly increased the protein expression of the eWAT lipogenic regulators (FABP4 and FASN) explaining the basis of adipocyte size increase under HFD condition.
The adipocyte size is also regulated by lipolytic enzymes (ATGL and HSL). Recently, it has been reported that the ATGL-KO mice show increased adipose mass and adipocyte size [29]. In contrast to ATGL-KO mice, the HSL-KO mice do not have increased fat deposition, body weight and adipocyte size [30, 31]. A non-coordinated regulation of ATGL and HSL mRNA as well as protein expressions have been reported during inflammatory conditions, exercise, weight loss [32] or during acute lypolytic phase, i.e. in the lipid mobilization phase and the protein breakdown phase such as fasting [33]. In concurrence with these observations, in the present study we observed that along with the increased eWAT weight, HFD dysregulated lipolysis by decreasing the ATGL expression which might have led to increased HSL expression in adipocytes, suggesting a differential regulation of these enzymes by the HFD and plausibly accounts for the increased levels of plasma insulin and TNF-α and reduced adiponectin [34]. However, the AT2R agonist treatment significantly altered the protein expression of both the epididymal WAT lipolytic regulators. The increased level of ATGL by C21 under HFD condition might be critical in regularizing the imbalanced energy homeostasis state in obesity. However, the significant increase in protein expression of HSL by C21 treatment under ND condition plausibly be due to the body’s physiological increase of the protein levels to compensate the reduction of FA uptake and restore the lipid metabolism under ND condition. Since this study measures only the protein expression, which is not a direct reflection of the enzyme activities, this can be considered a limitation of these measurements.
The lipogenic and lipolytic regulators are governed by the function of the FA transport into the adipocytes [35]. In adipocytes, plasma membrane FATP facilitates the LCFA uptake [36]. Modulation of FATP function results in alteration of energy homeostasis and insulin sensitivity [37]. Treatment with angiotensin-II alone and with PD123319 (AT2R blocker) has been shown to increase the uptake of palmitic acid (LCFA) in immortalized cardiomyocytes [38]; this indicates that the AT2R is involved in the FA uptake. To test whether the AT2R regulates FA transport, we performed an ex-vivo study using isolated mouse primary epididymal adipocytes. Our data demonstrate that the FA uptake was reduced by the AT2R agonist C21 in the primary epididymal adipocytes. Moreover, the C21-induced decrease in FA uptake was blocked by PD123319, indicating involvement of AT2R. Furthermore at the molecular level, studies have shown AT2R increases NOS activity [39]. Also it has been shown that the overexpression of NOS prevents adiposity [39]. So, we further explored whether the pharmacological activation of AT2R reduces fatty acid uptake via NOS-dependent pathway. We observed that the pre-treatment of primary epididymal adipocytes with inhibitors of NOS/GC/PKG blocked the AT2R-mediated decrease in FA uptake indicating the involvement of NOS/GC/PKG pathway. Whether these enzyme inhibitors alone would have their own effects (increase) on FA uptake and alter the interpretation of C21 effects are not clear thus limiting the study. Moreover, further studies are needed to explore whether NOS/GC/PKG pathway is involved in AT2R-medaited anti-obesity effects. Given that insulin is a potent stimulator of FA uptake (Fig. 3), these inhibitors alone would have a modest, if any, increase in FA uptake, thus unlikely to affect the net outcome on their ability to markedly attenuate AT2R-mediated inhibition of FA uptake inhibition. This reduction in FA transport possibly as a consequence prevents the HFD-induced changes in lipogenic and lipolytic regulators and thus prevents epididymal adipocyte size and eWAT weight increase under HFD. FATP1 is a major insulin sensitive LCFA transporter in adipose and skeletal muscle [11, 12, 40]. Inactivation of FATP1 prevents HFD-induced resistance in skeletal muscle [12]. In addition, another report indicates that the FATP4 expressions in human adipose tissue positively correlate with obesity and insulin resistance [13]. In accordance, we observed that the AT2R agonist treatment inhibits FA uptake in adipocytes, and reduces plasma FA and TAG levels under HFD.
Thermogenesis could increase energy expenditure and reduce adiposity. Tsukuda et al. (2016) recently reported that depletion of AT1R, a counter-regulator of AT2R, resulted in increase in thermogenic regulators in adipose tissue [41]. Conversely, current results show that pharmacological activation of AT2R also increased core temperature and prevented a decrease in expression of thermogenic regulator UCP1 in BAT. Similarly, chronic administration of AT2R agonist C21 resulted in upregulation of adipose UCP1 [42]. Our findings are in accordance with other study [43] showing that activation of AT2R by C21 increases browning of adipose tissue and increases body surface temperature.
Increased adiposity is associated with hyperlipidemia, hyperinsulinemia and increased inflammation. The larger adipocytes under HFD not only have impaired lipid metabolism but also triggered the pro-inflammatory state of obesity leading to insulin resistance [44–46]. In postprandial phase, the presence of diet stimulates the increase in plasma insulin to facilitate the uptake of glucose in cells. However, in obesity as adipocytes become resistant to insulin, the plasma FFA and TAG levels increase. The larger adipocytes also trigger the inflammation resulting in increased plasma TNF-α, a pro-inflammatory cytokine and decreased plasma adiponectin, an anti-inflammatory and insulin-sensitizing cytokine. The AT2R agonist C21 treatment improved insulin signaling and sensitivity that was associated with increase in adipose adiponectin [42]. The AT2R-KO mice developed insulin resistance with decreased adiponectin [20] while the AT2R-KO atherosclerotic mice have increased plasma FFA [22]. Pharmacological stimulation of AT2R with CGP42112A (a peptide AT2R agonist) has also been reported to increase plasma FFA and TAG in rats [47], albeit these experiments were conducted in ND-fed rats. Another study described opposite role of AT2R in regulation of thermogenic capacity of adipose tissue, however these researchers employed the mice model exhibiting transgenic activation of brain RAS and partial AT2R agonist CGP42112a [48]. There is some evidence showing that AT2R activation may have opposite effects in lean vs obese rats. For example, we have reported opposing effects of AT2R agonist on inflammation in lean and obese rats i.e., anti-inflammatory effects in obese vs inflammatory in lean rats [49]. In the present study we observed that C21 treatment in HFD condition prevented all the HFD-induced biochemical changes in young (5-wk age) as well as old (12-wk age) male mice (SI Table 2). Thus, C21 treatment not only prevents adiposity but also protects against hyperlipidemia, hyperinsulinemia and increased inflammation under HFD condition regardless of age. However, a direct role of AT2R in reducing inflammation and improving insulin resistance cannot be ruled out. We and others have reported that the AT2R exerts anti-inflammatory effects and also protects against insulin resistance [50–52].
Overall, there is a diet*treatment interaction regarding biochemical indices of metabolism (insulin, adiponectin, FFA and triglyceride), expression of lipogenic (FABP4) and lipolytic (HSL) enzymes, eWAT cell size and body weight gain indicating a dependency of the effects of AT2R agonist on these indices upon intake of type of diet. Our in vivo finding showing increase in expression of FABP4 in ND-fed mice does not correlate with finding showing acute inhibition of FA uptake in vitro. However, this discrepancy could be related to AT2R’s facilitation of initial steps of differentiation of pre-adipocytes [51]. Literature suggest that AT2R stimulation reduces the size of large lipid droplets once adipocytes mature and gain full capacity to store lipids [51] and that ND-fed mice treated with AT2R agonist C21 for 12 weeks did not affect body weight gain [16, 51]. Therefore, we speculate that increase in expression of FABP4 in WAT upon C21 treatment of ND-fed mice may help maintain basal fat mass but would not promote weight gain. Our earlier study reported that AT2R activation produces anti-adiposity effects and that beneficial effect is estrogen independent in female mice [16], whereas the current study shows that anti-adiposity effects of AT2R may be related to improvement in fat metabolism in terms of transport, synthesis and break-down. Additionally, we have reported that liver steatosis, not measured in the present study, caused by HFD in females is attenuated by AT2R agonist treatment [16]. The effects of AT2R agonist were independent of estrogen i.e., ovary-intact and ovariectomized females responded to C21 treatment in regard to the plasma metabolic markers and adipocyte size [16]. Since the effects of C21 are estrogen-independent [16], it can be argued that the soy-based diet which might contain traces of phytoestrogens may not have any bearing on the outcome of this study. Since obesity is connected to cardiovascular disease, affects kidney function in terms of fluid retention and is a major cause of hypertension and diabetes, long-term study may shed light as to the beneficial effects of AT2R agonist treatment in obesity. We have shown that AT2R agonist treatment for 2-weeks reduces high salt-induced blood pressure only in obese Zucker and not in obese rats per se [53]. Moreover, 2-weeks HFD regimen does not increase blood pressure and it is unlikely C21 treatment will have any effect on blood pressure in HFD fed animals.
Conclusions
Adiposity and obesity are metabolic disorders which are intricately connected to various cardiovascular diseases, hypertension and diabetes. Present study is an attempt to identify AT2R as new target to combat adiposity and obesity. Our results provide evidence that pharmacological activation of AT2R prevents HFD-induced adiposity, dyslipidemia and inflammation and insulin resistance. Inhibition of fatty acid uptake by adipose and thermogenesis could be possible mechanisms responsible for these beneficial effects of the AT2R agonist. Collectively, studies indicate AT2R activation as a potential therapeutic approach for controlling obesity and obesity-associated disorders such as insulin resistance and inflammation.
Supplementary Material
Acknowledgments
Funding: This work was supported by the National Institutes of Health grant R0I DK-61578 to TH and institutional funds.
Footnotes
This Author Accepted Manuscript is a PDF file of an unedited peer-reviewed manuscript that has been accepted for publication but has not been copyedited or corrected. The official version of record that is published in the journal is kept up to date and so may therefore differ from this version.
Conflict of interest: None
REFERENCES
- 1.Hubert HB, Feinleib M, McNamara PM and Castelli WP (1983) Obesity as an independent risk factor for cardiovascular disease - a 26-year follow-up of participants in the Framingham Heart Study. Circulation 67:968–977 [DOI] [PubMed] [Google Scholar]
- 2.Barrett-Connor EL (1985) Obesity, atherosclerosis, and coronary artery disease. Ann Intern Med 103:1010–1019 [DOI] [PubMed] [Google Scholar]
- 3.Bonadonna RC, Leif G, Kraemer N, Ferrannini E, Del Prato S and DeFronzo RA (1990) Obesity and insulin resistance in humans: a dose-response study. Metabolism 39:452–459 [DOI] [PubMed] [Google Scholar]
- 4.Elmarakby AA and Imig JD (2010) Obesity is the major contributor to vascular dysfunction and inflammation in high-fat diet hypertensive rats. Clin Sci 118:291–301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mathieu P, Lemieux I and Despres JP (2010) Obesity, inflammation, and cardiovascular risk. Clin Pharmacol Ther 87:407–16 [DOI] [PubMed] [Google Scholar]
- 6.Fodor GJ, McInnis NH, Helis E, Turton P and Leenen FH (2009) Lifestyle changes and blood pressure control: a community-based cross-sectional survey (2006 Ontario Survey on the Prevalence and Control of Hypertension). J Clin Hypertens 11:31–5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jansson SP, Engfeldt P, Magnuson A, Lohse G and Liljegren G (2013) Interventions for lifestyle changes to promote weight reduction, a randomized controlled trial in primary health care. BMC Res Notes 6:213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bloom SR, Kuhajda FP, Laher I, Pi-Sunyer X, Ronnett GV, Tan TM and Weigle DS (2008) The obesity epidemic - pharmacological challenges. Mol Interventions 8:82–98 [DOI] [PubMed] [Google Scholar]
- 9.Rodgers RJ, Tschop MH and Wilding JP (2012) Anti-obesity drugs: past, present and future. Dis. Model. Mech 5:621–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Greqoir FM, Smas CM and Sul HS (1998) Understanding adipocyte differentiation. Physiol Rev 78:783–809 [DOI] [PubMed] [Google Scholar]
- 11.Wu Q, Ortegon AM, Tsang B, Doege H, Feingold KR and Stahl A (2006) FATP1 is an insulin-sensitive fatty acid transporter involved in diet-induced obesity. Mol Cell Biol 26:3455–67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kim JK, Gimeno RE, Higashimori T, Kim H-J, Choi H, Punreddy S, Mozell RL, Tan G, Stricker-Krongrad A, Hirsch DJ, Fillmore JJ, Liu Z-X, Dong J, Cline G, Stahl A, Lodish HF and Shulman GI (2004) Inactivation of fatty acid transport protein 1 prevents fat-induced insulin resistance in skeletal muscle. J. Clin. Invest 113:756–763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gertow K, Pietilainen KH, Yki-Jarvinen H, Kaprio J, Rissanen A, Eriksson P, Hamsten A and Fisher RM (2004) Expression of fatty-acid-handling proteins in human adipose tissue in relation to obesity and insulin resistance. Diabetologia 47:1118–25 [DOI] [PubMed] [Google Scholar]
- 14.Yvan-Charvet L and Quignard-Boulange A (2011) Role of adipose tissue renin-angiotensin system in metabolic and inflammatory diseases associated with obesity. Kidney Int 79:162–8 [DOI] [PubMed] [Google Scholar]
- 15.Kalupahana NS and Moustaid-Moussa N (2012) The renin-angiotensin system: a link between obesity, inflammation and insulin resistance. Obes Rev 13:136–49 [DOI] [PubMed] [Google Scholar]
- 16.Nag S, Khan MA, Samuel P, Ali Q and Hussain T (2015) Chronic angiotensin AT2R activation prevents high-fat diet-induced adiposity and obesity in female mice independent of estrogen. Metabolism 64:814–25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hakam AC and Hussain T (2005) Renal angiotensin II type-2 receptors are upregulated and mediate the candesartan-induced natriuresis/diuresis in obese Zucker rats. Hypertens 45:270–5 [DOI] [PubMed] [Google Scholar]
- 18.Jones ES, Black MJ and Widdop RE (2012) Influence of angiotensin II subtype 2 receptor (AT(2)R) antagonist, PD123319, on cardiovascular remodelling of aged spontaneously hypertensive rats during chronic angiotensin II subtype 1 receptor (AT(1)R) blockade. Int J Hypertens 2012:543062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Putnam K, Batifoulier-Yiannikouris F, Bharadwaj KG, Lewis E, Karounos M, Daugherty A and Cassis LA (2012) Deficiency of angiotensin type 1a receptors in adipocytes reduces differentiation and promotes hypertrophy of adipocytes in lean mice. Endocrinology 153:4677–86 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yvan-Charvet L, Even P, Bloch-Faure M, Guerre-Millo M, Moustaid-Moussa N, Ferre P and Quignard-Boulange A (2005) Deletion of the angiotensin type 2 receptor (AT2R) reduces adipose cell size and protects from diet-induced obesity and insulin resistance. Diabetes 54:991–999 [DOI] [PubMed] [Google Scholar]
- 21.Samuel P, Khan MA, Nag S, Inagami T and Hussain T (2013) Angiotensin AT(2) receptor contributes towards gender bias in weight gain. PLoS One 8:e48425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Iwai M, Tomono Y, Inaba S, Kanno H, Senba I, Mogi M and Horiuchi M (2009) AT2 receptor deficiency attenuates adipocyte differentiation and decreases adipocyte number in atherosclerotic mice. Am J Hypertens 22:784–91 [DOI] [PubMed] [Google Scholar]
- 23.Namsolleck P, Boato F, Schwengel K, Paulis L, Matho KS, Geurts N, Thone-Reineke C, Lucht K, Seidel K, Hallberg A, Dahlof B, Unger T, Hendrix S and Steckelings UM (2013) AT2-receptor stimulation enhances axonal plasticity after spinal cord injury by upregulating BDNF expression. Neurobiol Dis 51:177–91 [DOI] [PubMed] [Google Scholar]
- 24.Matavelli LC, Huang J and Siragy HM (2011) Angiotensin AT(2) receptor stimulation inhibits early renal inflammation in renovascular hypertension. Hypertens 57:308–13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pellegrinelli V, Carobbio S and Vidal-Puig A (2016) Adipose tissue plasticity: how fat depots respond differently to pathophysiological cues. Diabetologia 59:1075–88 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schaffer JE and Lodisht HF (1994) Expression cloning and characterization of a novel adipocyte long chain fatty acid transport protein. Cell 79:427–436 [DOI] [PubMed] [Google Scholar]
- 27.Thompson BR, Mazurkiewicz-Munoz AM, Suttles J, Carter-Su C and Bernlohr DA (2009) Interaction of adipocyte fatty acid-binding protein (AFABP) and JAK2: AFABP/aP2 as a regulator of JAK2 signaling. J Biol Chem 284:13473–80 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wakil SJ and Abu-Elheiga LA (2009) Fatty acid metabolism: target for metabolic syndrome. J Lipid Res 50 Suppl:S1 38–43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Haemmerle G, Lass A, Zimmermann R, Gorkiewicz G, Meyer C, Rozman J, Heldmaier G, Maier R, Theussl C, Eder S and Kratky D (2006) Defective lipolysis and altered energy metabolism in mice lacking adipose triglyceride lipase. Science 312:734–737 [DOI] [PubMed] [Google Scholar]
- 30.Osuga JI, Ishibashi S, Oka T, Yagyu H, Tozawa R, Fujimoto A, Shionoiri F, Yahagi N, Kraemer FB, Tsutsumi O and Yamada N (2000) Targeted disruption of hormone-sensitive lipase results in male sterility and adipocyte hypertrophy, but not in obesity. Proc Natl Acad Sci U S A 97:787–792 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sekiya M, Osuga J, Okazaki H, Yahagi N, Harada K, Shen WJ, Tamura Y, Tomita S, Iizuka Y, Ohashi K, Okazaki M, Sata M, Nagai R, Fujita T, Shimano H, Kraemer FB, Yamada N and Ishibashi S (2004) Absence of hormone-sensitive lipase inhibits obesity and adipogenesis in Lep ob/ob mice. J Biol Chem 279:15084–90 [DOI] [PubMed] [Google Scholar]
- 32.Schoiswohl G, Stefanovic-Racic M, Menke MN, Wills RC, Surlow BA, Basantani MK, Sitnick MT, Cai L, Yazbeck CF, Stolz DB, Pulinilkunnil T, O’Doherty RM and Kershaw EE (2015) Impact of Reduced ATGL-Mediated Adipocyte Lipolysis on Obesity-Associated Insulin Resistance and Inflammation in Male Mice. Endocrinology 156:3610–24 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bertile F and Raclot T (2011) ATGL and HSL are not coordinately regulated in response to fuel partitioning in fasted rats. J. Nutr. Biochem 22:372–9 [DOI] [PubMed] [Google Scholar]
- 34.Li L, Yang G, Shi S, Yang M, Liu H and Boden G (2009) The adipose triglyceride lipase, adiponectin and visfatin are downregulated by tumor necrosis factor-alpha (TNF-alpha) in vivo. Cytokine 45:12–9 [DOI] [PubMed] [Google Scholar]
- 35.Glatz JF, Luiken JJ and Bonen A (2010) Membrane fatty acid transporters as regulators of lipid metabolism: implications for metabolic disease. Physiol Rev 90:367–417 [DOI] [PubMed] [Google Scholar]
- 36.Schaffer JE and Lodish HF (1995) Molecular mechanism of long-chain fatty acid uptake. Trends Cardiovasc Med 5:218–224 [DOI] [PubMed] [Google Scholar]
- 37.Gimeno RE (2007) Fatty acid transport proteins. Curr Opin Lipidol 18:271–276 [DOI] [PubMed] [Google Scholar]
- 38.Alfarano C, Sartiani L, Nediani C, Mannucci E, Mugelli A, Cerbai E and Raimondi L (2008) Functional coupling of angiotensin II type 1 receptor with insulin resistance of energy substrate uptakes in immortalized cardiomyocytes (HL-1 cells). Br J Pharmacol 153:907–14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Taguchi K, Matsumoto T, Kamata K and Kobayashi T (2012) Angiotensin II type 2 receptor-dependent increase in nitric oxide synthase activity in the endothelium of db/db mice is mediated via a MEK pathway. Pharmacol Res 66:41–50 [DOI] [PubMed] [Google Scholar]
- 40.Holloway GP, Chou CJ, Lally J, Stellingwerff T, Maher AC, Gavrilova O, Haluzik M, Alkhateeb H, Reitman ML and Bonen A (2011) Increasing skeletal muscle fatty acid transport protein 1 (FATP1) targets fatty acids to oxidation and does not predispose mice to diet-induced insulin resistance. Diabetologia 54:1457–1467 [DOI] [PubMed] [Google Scholar]
- 41.Tsukuda K, Mogi M, Iwanami J, Kanno H, Nakaoka H, Wang XL, Bai HY, Shan BS, Kukida M, Higaki A, Yamauchi T, Min LJ and Horiuchi M (2016) Enhancement of Adipocyte Browning by Angiotensin II Type 1 Receptor Blockade. PLoS One 11:e0167704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Quiroga DT, Munoz MC, Gil C, Pffeifer M, Toblli JE, Steckelings UM, Giani JF and Dominici FP (2018) Chronic administration of the angiotensin type 2 receptor agonist C21 improves insulin sensitivity in C57BL/6 mice. Physiol. Rep 6:e13824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Than A, Xu S, Li R, Leow M-S, Sun L and Chen P (2017) Angiotensin type 2 receptor activation promotes browning of white adipose tissue and brown adipogenesis. Signal Transduct Target Ther 2:e17022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hajer GR, van Haeften TW and Visseren FL (2008) Adipose tissue dysfunction in obesity, diabetes, and vascular diseases. Eur Heart J 29:2959–71 [DOI] [PubMed] [Google Scholar]
- 45.Guilherme A, Virbasius JV, Puri V and Czech MP (2008) Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat Rev Mol Cell Biol 9:367–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Gustafson B, Gogg S, Hedjazifar S, Jenndahl L, Hammarstedt A and Smith U (2009) Inflammation and impaired adipogenesis in hypertrophic obesity in man. Am J Physiol Endocrinol Metabol 297:E999–E1003 [DOI] [PubMed] [Google Scholar]
- 47.Ran J, Hirano T and Adachi M (2005) Angiotensin II infusion increases hepatic triglyceride production via its type 2 receptor in rats. J Hypertens 23:1525–1530 [DOI] [PubMed] [Google Scholar]
- 48.Littlejohn NK, Keen HL, Weidemann BJ, Claflin KE, Tobin KV, Markan KR, Park S, Naber MC, Gourronc FA, Pearson NA, Liu X, Morgan DA, Klingelhutz AJ, Potthoff MJ, Rahmouni K, Sigmund CD and Grobe JL (2016) Suppression of Resting Metabolism by the Angiotensin AT2 Receptor. Cell Rep 16:1548–60 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sabuhi R, Ali Q, Asghar M, Al-Zamily NR and Hussain T (2011) Role of the angiotensin II AT2 receptor in inflammation and oxidative stress: opposing effects in lean and obese Zucker rats. Am J Physiol Renal Physiol 300:F700–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ohshima K, Mogi M, Jing F, Iwanami J, Tsukuda K, Min LJ, Ogimoto A, Dahlof B, Steckelings UM, Unger T, Higaki J and Horiuchi M (2012) Direct angiotensin II type 2 receptor stimulation ameliorates insulin resistance in type 2 diabetes mice with PPARgamma activation. PLoS One 7:e48387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Shum M, Pinard S, Guimond MO, Labbe SM, Roberge C, Baillargeon JP, Langlois MF, Alterman M, Wallinder C, Hallberg A, Carpentier AC and Gallo-Payet N (2013) Angiotensin II type 2 receptor promotes adipocyte differentiation and restores adipocyte size in high-fat/high-fructose diet-induced insulin resistance in rats. Am J Physiol Endocrinol Metabol 304:E197–210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dhande I, Ali Q and Hussain T (2013) Proximal tubule angiotensin AT2 receptors mediate an anti-inflammatory response via interleukin-10: role in renoprotection in obese rats. Hypertens 61:1218–26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ali Q, Patel S and Hussain T (2015) Angiotensin AT2 receptor agonist prevents salt-sensitive hypertension in obese Zucker rats. Am J Physiol Renal Physiol 308:F1379–85 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.