Abstract
The transport systems for metals play crucial roles in both the physiological functions of essential metals and the toxic effects of hazardous metals in mammals and plants. In mammalian cells, Zn transporters such as ZIP8 and ZIP14 have been found to function as the transporters for Mn(II) and Cd(II), contributing to the maintenance of Mn homeostasis and metallothionein-independent transports of Cd, respectively. In rice, the Mn transporter OsNramp5 expressed in the root is used for the uptake of Cd from the soil. Japan began to cultivate OsNramp5 mutant rice, which was found to accumulate little Cd, to prevent Cd accumulation. Inorganic trivalent arsenic (As(III)) is absorbed into mammalian cells via aquaglyceroporin, a water and glycerol channel. The ortholog of aquaporin in rice, OsLsi1, was found to be an Si transporter expressed in rice root, and is responsible for the absorption of soil As(III) into the root. Since rice is a hyperaccumulator of Si, higher amounts of As(III) are incorporated into rice compared to other plants. Thus, the transporters of essential metals are also utilized to incorporate toxic metals in both mammals and plants, and understanding the mechanisms of metal transports is important for the development of mitigation strategies against food contamination.
Keywords: Cadmium, Manganese, Arsenic, Silicon, Zinc, Transporter
ROLES OF METAL TRANSPORTERS IN TOXICOMETALLOMICS
Proteins, nucleic acids, and lipids are essential for the lives of all organisms. From the viewpoint of elemental composition, however, they are composed of a limited number of elements such as C, H, O, N, S, and P. On the other hand, all prokaryotes and eukaryotes require a variety of metals such as Ca, Mg, Na, K, Fe, Zn, Cu, Mn, and so on, for survival. These metals constitute important components or cofactors of versatile biomolecules, and influence the functions of proteins, nucleic acids, and lipids. Metallomics is a field of science that aims to elucidate all features of the actions, interactions, structures, transports and roles of metals in biological systems (1).
Although organisms can biosynthesize most proteins, nucleic acids, and lipids, they cannot create metal elements within cells. They must incorporate metals from the extracellular environment via transporters. Therefore, metal transporters are essential to maintaining appropriate amounts of metals within tissues, cells, and organelles for the survival of all organisms. For example, human cells contain 24 types of Zn transporters, which regulate the influx and efflux of Zn at the plasma membrane and organelles, and the expression of each Zn transporter is distinctly regulated in a tissue-, cell-, and organelle-specific manner (2). Dysfunctions of metal transporters may lead to disturbances in metal homeostasis, resulting in the syndromes of metal deficiency or excess.
It has long been known that metals such as Hg, As, Cd, and Pb play no essential role in organisms, and incorporation of excess amounts of these toxic metals causes a variety of diseases in plants, animals, and humans. During the process of evolution, organisms did not develop any specific transporters for cellular incorporation of these toxic metals. Consequently, when toxic metals enter the cells of animals and plants, they utilize the transporters developed for other essential metals. This can happen because most metal transporters have a broad spectrum of affinities for multiple metals. To elucidate the mechanisms of cellular accumulation of toxic metals, the identification and characterization of the transporters involved in the transport of both toxic and essential metals are required. From a viewpoint of toxicometallomics, which is a part of metallomics and focuses on the actions, metabolisms, and the effects of chemical forms of toxic metals as well as the interactions of toxic metals with essential metals, elucidation of the transport mechanisms of both toxic and essential metals is crucial for understanding the accumulation modes of toxic metals and for solving the metal contaminations in plants, animals, and humans.
In this review, we briefly describe the roles of metal transporters involved in the transport of Cd and Mn, as well as those involved in the transport of As, water, and Si in mammals and plants.
TRANSPORTERS FOR CELLULAR INCORPORATION OF Cd AND Mn IN MAMMALS
It has long been considered that metallothionein (MT), a cysteine-rich metal-binding protein, plays several crucial roles in the transport and accumulation of Cd in mammalian cells (3). MT can efficiently bind Cd, as well as other metals such as Zn, Cu, Bi, Hg, and Ag, and protect against cytotoxicity of these metals. In the kidney, which is the target organ of chronic toxicity of Cd, the Cd bound to MT in the blood circulation is readily filtered through the glomerulus, due to its low molecular weight (about 7,000), and then reabsorbed from the lumen to the proximal tubule epithelial cells (PTECs) via megalin-dependent endocytosis (4,5). Since the Cd ions released from the absorbed MT protein within PTECs may continuously induce the synthesis of MT, leading to the binding of Cd ions to the newly synthesized MT protein, the biological half-life of Cd in the kidney was thought to be very long (6). However, the modes of cellular incorporation of Cd via pathways other than Cd-MT endocytosis have not been fully elucidated.
To identify non-MT factors, including non-MT transport systems involved in Cd resistance, we have developed Cd-resistant cell lines from mouse embryonic fibroblast (MEF) cells obtained from MT-I, II knockout mice via stepwise increases in Cd concentrations in the media (7). The MT-null Cd-resistant cells showed a markedly reduced accumulation of Cd, primarily caused by extremely reduced Cd uptake rates (7). Since there is no specific transporter for Cd uptake in mammalian cells, it is assumed that expression of the transporter that functions for the uptake of other essential metals, and also has an affinity for Cd, was suppressed in the Cd-resistant MT-null cells. To identify which metal was involved in this suppression of metal uptake, we applied a multi-tracer technique (8). Among 20 metals examined by this technique, Mn(II) showed a markedly lower uptake rate in Cd-resistant MT-null cells, suggesting that Cd(II) and Mn(II) share the same pathway for cellular incorporation (8).
The transport of Cd(II) and Mn(II) by the same transporter was also supported by competitive inhibition of the uptakes of Cd(II) and Mn(II) (8,9). Subsequent microarray analysis revealed that the expression of ZIP8, a Zn transporter coded by Slc39a8, was suppressed in Cd-resistant MT-null cells compared with parental MT-null cells (10). The role of ZIP8 in Cd transport was confirmed by the reduced uptake of Cd(II) in the parental MT-null cells transfected with shRNA of ZIP8 (11). The suppressed expression of ZIP8 in Cd-resistant MT-null cells was not due to the mutation of Slc39a8 but to epigenetic hypermethylation of the Slc39a8 gene, because the treatment of Cd-resistant MT-null cells with 5-aza-cytidine, an inhibitor of DNA methyltransferase, led to the recovery of ZIP8 expression, the enhanced uptake of Cd, and consequently, the enhanced cytotoxicity of Cd (12).
To further examine the role of ZIP8 in Cd resistance, we also established Cd-resistant cell lines from MT-expressing MEF cells and from rat basophilic leukemia RBL-2H3 cells. The latter showed extremely high sensitivity to Cd. Both Cd-resistant cell lines showed suppressed expression of ZIP8 with lowered uptake rates of Cd (13,14). Interestingly, both Cd-resistant cell lines showed cross-resistance to MnCl2 due to the lowered uptake rates of Mn(II). As the parental RBL-2H3 cells showed higher expression of ZIP8 and higher sensitivity to MnCl2 cytotoxicity than the other rat cell lines examined (15), we also developed Mn-resistant cells from RBL-2H3 cells. As expected, the Mn-resistant RBL-2H3 cells showed a cross-resistance to Cd, accompanied by the suppression of ZIP8 expression and lower uptake rates of both Mn(II) and Cd(II) (14). Collectively, the results obtained from Cd-resistant and Mn-resistant cells indicate that ZIP8 plays an important role in the transport of Cd(II) and Mn(II) in addition to Zn(II), and that the lack of ZIP8 expression results in the reduced accumulation and cytotoxicity of Cd and Mn.
In addition to ZIP8, ZIP14, which has the highest similarity to ZIP8 in its amino acid sequence among ZIP family transporters, was also shown to be involved in the transport of Cd(II) and Mn(II) (Fig. 1) in our studies and those of other groups (16,17). The affinities of ZIP8 and two isoforms of ZIP14, ZIP14A and ZIP14B, for Cd(II), Mn(II), and Zn(II) have been compared in the mammalian cells and Xenopus oocytes in which these transporters were ectopically expressed (17). The Km values for the uptake of Cd(II) (0.62 μM, 0.10 μM, and 0.14 μM for ZIP8, ZIP14A, and ZIP14B, respectively) were lower than those of Mn(II) (2.2 μM, 18 μM, and 4.4 μM for ZIP8, ZIP14A, and ZIP14B, respectively) in mammalian cells, whereas the Km values for the uptake of Cd(II) (0.48 μM and 0.46 μM for ZIP8 and ZIP14A, respectively) were roughly similar to those of Zn (0.26 μM and 0.38 μM for ZIP8 and ZIP14A, respectively) in Xenopus oocytes (17). These data indicate the high affinity of ZIP8 and ZIP14 for Cd(II).
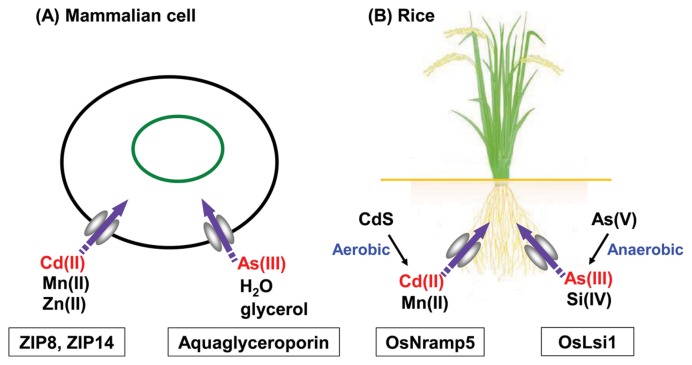
Fig. 1.
Schematic illustrations of Cd and As(III) uptake via metal transporters in mammalian cells (A) and rice root (B). (A): Among the ZIP family members of Zn transporters, ZIP8 and ZIP14 participate in the transports of Cd(II) and Mn(II) as well as that of Zn(II). The functionalities of ZIP8 and ZIP14 for the transports of Cd(II) and Mn(II) have been recognized in a variety of mammalian cells including hepatic (15), renal (21,23), cardiac (15), neuronal (16), intestinal (19), and uterine (8) cells. Cellular uptake of As(III) in mammalian cells is mediated by aquaglyceroporin, a water channel used also for the uptake of glycerol (34–37). (B): In the root of rice, the uptake of Cd(II) is mediated by the Mn(II) transporter, OsNramp5 (29), and that of As(III) is mediated by the Si(IV) transporter OsLsi1 (44), which is an ortholog of aquaporin in mammals. Since rice is a hyperaccumulator of Si (42), higher amounts of As(III) is accumulated in rice than other plants. The microenvironment of soil and water surrounding the rice root affect the release of soluble Cd(II) and As(III) from the soil in opposite ways. Under aerobic conditions, the insoluble CdS in soil, which is stable under anaerobic conditions, is oxidized to CdSO4, leading to the release of soluble Cd(II). In contrast, under anaerobic conditions, the complex of As(V)-Fe(III), which is stable under aerobic conditions, is reduced to As(III) and Fe(II), leading to the release of soluble As(III). To achieve the mitigation of Cd and As contaminations in rice by the management of irrigation water, this trade-off problem should be solved (46–49).
A divalent metal transporter 1 (DMT1) also has affinities not only for Fe(II) but also for Mn(II) and Cd(II) (18). Since the expression of DMT1 is high in the intestine (11,18,19), and that of ZIP14 is high in the liver (20), these transporters may play tissue-specific roles in the transport of Cd(II) and Mn(II) in different tissues of the body. We found high expression of ZIP8 in the S3 region (straight part) of kidney proximal tubules (21–23). Since it is known that Cd-MT in the glomerular filtrate is mainly reabsorbed in the S1 and S2 regions of proximal tubules (4,5), where megalin-dependent endocytosis largely contributes to the reabsorption of low-molecular-weight proteins, ZIP8 may play a different role in the reabsorption of Cd not bound to MT in the lumen (24).
To explore the possibility that ZIP8 is involved in the reabsorption of Cd(II) from the apical side of PTECs, especially at the S3 region, we carried out an in vitro experiment using immortalized S1, S2, and S3 cells derived from each region of mouse proximal tubules (23). When Cd(II) was added to the apical side of monolayer S1, S2, and S3 cells cultured on the membrane of the trans-well, the highest uptake of Cd(II) was detected in S3 cells, and the uptake of Cd(II) from the apical side of S3 cells was competitively inhibited by Mn(II) and Zn(II) (23). This suggests that ZIP8 plays a role in the reabsorption of luminal Cd(II) in the S3 region of proximal tubules. However, further in vivo studies are needed to clarify the roles of ZIP8 in renal handling and accumulation of Cd.
Recently, accumulating evidence has shown the role of ZIP8 as an Mn(II) transporter in humans. In Germany and Egypt, congenital glycosylation dysfunctions in infants were found to be related to mutations of human SLC39A8 coding ZIP8 (25,26). Blood Mn levels of these infants were extremely low or undetectable. Since the galactosyl transferase, which is essential for the glycosylation process in the body, is highly dependent on Mn as a cofactor, disturbed metabolism of Mn might have led to the whole-body disorder of glycosylation. Since the excretion rate of Mn from the liver into bile is markedly high, the role of ZIP8 in the epithelial cells of the bile ducts in reabsorbing the Mn excreted to the bile has been considered (27). The ZIP8 expressed in the S3 region of renal proximal tubules may also be involved in the renal reabsorption of Mn (23), but the physiological roles of ZIP8 in the regulation of whole-body Mn homeostasis remain to be elucidated.
Recently, the results of the genome-wide association study (GWAS) showed the links between the SNP of ZIP8 causing amino acid change (p.Ala391Thr) and a variety of conditions such as hypertension, schizophrenia, and other common diseases (28). Future studies will be needed to clarify whether disordered Mn metabolism is involved in these diseases. Thus, greater attention is currently being paid to the roles of ZIP8 not only in Cd transport but also in Mn transport in humans.
ABSORPTION OF SOIL Cd IN RICE VIA A Mn TRANSPORTER
For Asian people who eat large amounts of rice as a staple food, the contamination of rice with toxic metals such as Cd or As is an important issue, and the scientists in the fields of plant physiology and soil sciences are attempting to mitigate the metal contamination of rice. Since plants cannot move like animals, they evolved a variety of transport systems for acquiring necessary metals from the soils. One plant physiology study revealed that OsNramp5 located in the outer membrane of rice root is the transporter responsible for the uptake of Mn into rice (29). Intriguingly, OsNramp5 also has an affinity for Cd (Fig. 1), and the knockout of the OsNramp5 gene resulted in almost complete loss of Cd absorption into the roots and consequently grains of rice (29). Since Mn constitutes the catalytic centers of the water-oxidizing complex of photo-system II in plants, it is inevitable that rice will absorb significant amounts of soil Cd via the transporter for Mn. Thus, the transport systems for Mn play an important role in Cd incorporation in rice as well as in mammals.
Recently in Japan, mutant rice, in which the function of OsNramp5 is lost and therefore Cd is not accumulated, was developed and has begun to be cultivated as a counter-measure against Cd contamination of rice. This mutant rice was developed using an ion-beam irradiation that can generate a variety of mutations with high specificity in plants (30). Approximately 3,000 rice seeds were irradiated with an ion beam, and three mutants obtained in the next generation seeds were found to produce rice that accumulated no detectable levels of Cd in their grains even when they were cultivated in Cd-contaminated soil. All three mutant rice varieties were found to have a mutation in the OsNramp5 gene (30). The mutant rice also showed low levels of Mn, but did not show apparent Mn deficiency symptoms such as growth defects, possibly because of the compensatory uptake of Mn by other pathways.
TRANSPORTERS FOR CELLULAR INCORPORATION OF As IN MAMMALS
Human As poisonings have been extensively reported in Asian countries, especially in Bangladesh, India, and China, where groundwater is contaminated by inorganic As (iAs) from the earth’s crust (31). On the other hand, arsenic trioxide is clinically used for the treatment of acute promyelocytic leukemia (32). Among As compounds, iAs such as arsenite (As(III)) and arsenate (As(V)) are highly toxic compared with the methylated forms of As. Therefore, elucidation of the transport systems for iAs is important for understanding the metabolic fates and pharmacological or toxicological effects of As compounds.
The search for the transport systems for As(III) in eukaryotes was triggered by a report showing that the Saccharomyces cerevisiae strain that lacks the expression of glycerol transporter Fps1p exhibited a resistance to As(III) (33). The yeast Fps1p is a protein homologous to the mammalian aquaglyceroporins AQP7 and AQP9. Microinjection of mammalian AQP7 or AQP9 into Xenopus oocytes (34) and that of AQP9 into mouse hepatocytes (35) resulted in enhanced uptake of As(III). AQP3 and AQP10 have also been shown to transport As(III) in mammalian cells (36,37). Aquaglyceroporins (AQP3, 7, 9, and 10) are members of the aquaporin superfamily, which is basically a water channel. However, aquaglyceroporins can also permeate electrically neutral small molecules such as glycerol and urea in addition to water molecules. Since AQP9 is highly expressed in the liver, AQP9 may play an important role in the absorption of As(III) into the liver, the major organ for the metabolism of ingested As compounds (38).
The expression of AQP9 is also detected in human leukemia cell lines such as HL-60 (39). Recently, we found that treatment of HL-60 cells with all-trans retinoic acid, the first-line drug for treatment of acute promyelocytic leukemia, significantly increased the expression of AQP9, leading to an enhanced cellular uptake of As(III) (39). This suggests that co-treatment of leukemic cells with all-trans retinoic acid and arsenic trioxide may enhance the therapeutic efficacy of arsenic trioxide.
As(V), another form of iAs, is absorbed by phosphate transporters because arsenate has physicochemical properties similar to those of phosphate. In particular, it has been suggested that the intestinal phosphate transporter NaPiIIb is involved in the absorption of As(V) into the body (40).
ABSORPTIONS OF As VIA Si TRANSPORTER IN RICE AND ITS TRADE-OFF PROBLEM WITH Cd
In areas where As poisoning is prevalent, not only groundwater for drinking and cooking but also the rice cultivated with irrigation water derived from As-contaminated groundwater is the source of As ingestion in humans (41). Since As is not an essential element for plants, the absorption of As into rice is mediated by other essential elements, similar to Cd absorption via the Mn transporter. The rice accumulates higher amounts of As than other plants because of its specific nature as a hyperaccumulator of Si. The content of Si in rice is as high as 10% of the total weight, and Si deficiency causes detrimental effects on the rigidity and stress resistance of rice (42). The absorption of Si in the form of Si(OH)4 from the soil to rice root is mediated by the OsLsi1 transporter expressed in rice root (43). Intriguingly, OsLsi1 is an ortholog of mammalian AQP, which is involved in As(III) uptake. It was found that OsLsi1 has the ability to transport As(III) in addition to Si into rice root (Fig. 1) (44). Thus, rice, as a hyperaccumulator of Si inevitably becomes a hyperaccumulator of As(III). Actually, 60–80% of As in rice grains is in the form of As(III) (45). Since OsLsi1 plays an essential role in the absorption of Si, its inactivation by mutation or knockout rice could not be achieved.
The solubilities of As(III) and As(V) from soil are affected by the oxidizing/reducing conditions of the microenvironment of soil and water around the rice root (46–48). For cultivation of rice, the management of the irrigation water supply is important; water is supplied to (flooding) or drained from (non-flooding) the paddy field depending on the growth periods of rice. The “flooding” causes reducing (anaerobic) conditions in the microenvironment of soils and water surrounding the rice roots due to the blockage of air, while the “non-flooding” causes oxidizing (aerobic) conditions. Under the oxidizing conditions, As and Fe form a stable and insoluble complex in the form of As(V)-Fe(III)(OH)3, whereas under the reducing conditions the reductions of Fe(III) to Fe(II) and As(V) to As(III) occur, leading to the release of soluble As(III) from the soil. Thus, the flooding of rice fields facilitates the release of the soluble form of As(III), and therefore the non-flooding of fields is preferable to minimize the absorption of As(III) into rice, especially at harvest time (45).
Unfortunately, however, the release of soluble Cd from insoluble CdS in the soil via the formation of CdSO4 is facilitated under the oxidizing conditions and suppressed under the reducing conditions (47,49). This opposite behavior of As and Cd depending on environmental conditions produces a trade-off problem in the mitigation strategy against metal contaminations of rice. Especially in Japan, flooding rice fields at harvest time is recommended as a water management strategy for reducing Cd accumulation in rice in areas where there is soil contamination of Cd (49,50); this strategy may, however, increase As accumulation in rice. Since the Codex Alimentarius Commission of FAO/WHO has recommended that iAs concentration in polished rice grains be less than 0.2 mg/kg (51), a solution for this trade-off problem is urgently needed, especially in areas where soil Cd contamination is detected. The utilization of the low-Cd rice cultivar (30,52) may be one of the warranted solutions for this problem.
CONCLUSIONS
In this review, recent advances in the study of the transport systems for Cd and As in mammals and plants are briefly summarized. In both mammals and plants, transporters originally evolved for the uptake of essential elements such as Mn, Zn, and Si are used for the uptake of toxic metals such as Cd and As. To facilitate the mitigation of metal contamination in plants and animals, a more detailed understanding of the whole profiles of transporting systems for essential and toxic metals is needed.
ACKNOWLEDGMENTS
This research was partly supported by JSPS KAKENHI Grant Numbers 16H05834 (to SH), 19K12346 (to DS), and 18K06646 (to HF).
Footnotes
CONFLICT OF INTEREST
The authors have no conflicts of interest to declare.
REFERENCES
- 1.Haraguchi H. Metallomics: the history over the last decade and a future outlook. Metallomics. 2017;9:1001–1013. doi: 10.1039/C7MT00023E. [DOI] [PubMed] [Google Scholar]
- 2.Hara T, Takeda TA, Takagishi T, Fukue K, Kambe T, Fukada T. Physiological roles of zinc transporters: molecular and genetic importance in zinc homeostasis. J Physiol Sci. 2017;67:283–301. doi: 10.1007/s12576-017-0521-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Klaassen CD, Liu J,, Diwan BA. Metallothionein protection of cadmium toxicity. Toxicol Appl Pharmacol. 2009;238:215–220. doi: 10.1016/j.taap.2009.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sabolić I, Breljak D, Škarica M, Herak-Kramberger CM. Role of metallothionein in cadmium traffic and toxicity in kidneys and other mammalian organs. Biometals. 2010;23:897–926. doi: 10.1007/s10534-010-9351-z. [DOI] [PubMed] [Google Scholar]
- 5.Wolff NA, Abouhamed M, Verroust PJ, Thévenod F. Megalin-dependent internalization of cadmium-metallothionein and cytotoxicity in cultured renal proximal tubule cells. J Pharmacol Exp Ther. 2006;318:782–791. doi: 10.1124/jpet.106.102574. [DOI] [PubMed] [Google Scholar]
- 6.Elinder CG, Lind B, Kjellstrom T, Linnman L, Friberg L. Cadmium in kidney cortex, liver, and pancreas from Swedish autopsies. Estimation of biological half time in kidney cortex, considering calorie intake and smoking habits. Arch Environ Health. 1976;31:292–302. doi: 10.1080/00039896.1976.10667239. [DOI] [PubMed] [Google Scholar]
- 7.Yanagiya T, Imura N, Kondo Y, Himeno S. Reduced uptake and enhanced release of cadmium in cadmium resistant metallothionein null fibroblasts. Life Sci. 1999;65:PL177–PL182. doi: 10.1016/S0024-3205(99)00393-8. [DOI] [PubMed] [Google Scholar]
- 8.Yanagiya T, Imura N, Enomoto S, Kondo Y, Himeno S. Suppression of a high-affinity transport system for manganese in cadmium-resistant metallothionein-null cells. J Pharmacol Exp Ther. 2000;292:1080–1086. [PubMed] [Google Scholar]
- 9.Himeno S, Yanagiya T, Fujishiro H. The role of zinc transporters in cadmium and manganese transport in mammalian cells. Biochimie. 2009;91:1218–1222. doi: 10.1016/j.biochi.2009.04.002. [DOI] [PubMed] [Google Scholar]
- 10.Fujishiro H, Okugaki S, Nagao S, Satoh M, Himeno S. Characterization of gene expression profiles of metallothionein-null cadmium-resistant cells. J Health Sci. 2006;52:292–299. doi: 10.1248/jhs.52.292. [DOI] [Google Scholar]
- 11.Fujishiro H, Okugaki S, Kubota K, Fujiyama T, Miyataka H, Himeno S. The role of ZIP8 down-regulation in cadmium-resistant metallothionein-null cells. J Appl Toxicol. 2009;29:367–373. doi: 10.1002/jat.1419. [DOI] [PubMed] [Google Scholar]
- 12.Fujishiro H, Okugaki S, Yasumitsu S, Enomoto S, Himeno S. Involvement of DNA hypermethylation in down-regulation of the zinc transporter ZIP8 in cadmium-resistant metallothionein-null cells. Toxicol Appl Pharmacol. 2009;241:195–201. doi: 10.1016/j.taap.2009.08.015. [DOI] [PubMed] [Google Scholar]
- 13.Fujishiro H, Kubota K, Inoue D, Inoue A, Yanagiya T, Enomoto S, Himeno S. Cross-resistance of cadmium-resistant cells to manganese is associated with reduced accumulation of both cadmium and manganese. Toxicology. 2011;280:118–125. doi: 10.1016/j.tox.2010.12.002. [DOI] [PubMed] [Google Scholar]
- 14.Fujishiro H, Ohashi T, Takuma M, Himeno S. Suppression of ZIP8 expression is a common feature of cadmium-resistant and manganese-resistant RBL-2H3 cells. Metallomics. 2013;5:437–444. doi: 10.1039/c3mt00003f. [DOI] [PubMed] [Google Scholar]
- 15.Fujishiro H, Doi M, Enomoto S, Himeno S. High sensitivity of RBL-2H3 cells to cadmium and manganese: an implication of the role of ZIP8. Metallomics. 2011;3:710–718. doi: 10.1039/c1mt00020a. [DOI] [PubMed] [Google Scholar]
- 16.Fujishiro H, Yoshida M, Nakano Y, Himeno S. Interleukin-6 enhances manganese accumulation in SH-SY5Y cells: Implications of the up-regulation of ZIP14 and the down-regulation of ZnT10. Metallomics. 2014;6:944–949. doi: 10.1039/C3MT00362K. [DOI] [PubMed] [Google Scholar]
- 17.He L, Wang B, Hay EB, Nebert DW. Discovery of ZIP transporters that participate in cadmium damage to testis and kidney. Toxicol Appl Pharmacol. 2009;238:250–257. doi: 10.1016/j.taap.2009.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gunshin H, Mackenzie B, Berger UV, Gunshin Y, Romero MF, Boron WF, Nussberger S, Gollan JL, Hediger MA. Cloning and characterization of a mammalian proton-coupled metal-ion transporter. Nature. 1997;388:482–488. doi: 10.1038/41343. [DOI] [PubMed] [Google Scholar]
- 19.Fujishiro H, Hamao S, Tanaka R, Kambe T, Himeno S. Concentration-dependent roles of DMT1 and ZIP14 in cadmium absorption in Caco-2 cells. J Toxicol Sci. 2017;42:559–567. doi: 10.2131/jts.42.559. [DOI] [PubMed] [Google Scholar]
- 20.Liuzzi JP, Lichten LA, Rivera S, Blanchard RK, Aydemir TB, Knutson MD, Ganz T, Cousins RJ. Interleukin-6 regulates the zinc transporter Zip14 in liver and contributes to the hypozincemia of the acute-phase response. Proc Natl Acad Sci USA. 2005;102:6843–6848. doi: 10.1073/pnas.0502257102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fujishiro H, Yano Y, Takada Y, Tanihara M, Himeno S. Roles of ZIP8, ZIP14, and DMT1 in transport of cadmium and manganese in mouse kidney proximal tubule cells. Metallomics. 2012;4:700–708. doi: 10.1039/c2mt20024d. [DOI] [PubMed] [Google Scholar]
- 22.Fujishiro H, Himeno S. Gene expression profiles of immortalized S1, S2, and S3 cells derived from each segment of mouse kidney proximal tubules. Fundam Toxicol Sci. 2019;6:117–123. doi: 10.2131/fts.6.117. [DOI] [PubMed] [Google Scholar]
- 23.Fujishiro H, Hamao S, Isawa M, Himeno S. Segment-specific and direction-dependent transport of cadmium and manganese in immortalized S1, S2, and S3 cells derived from mouse kidney proximal tubules. J Toxicol Sci. 2019;44:611–619. doi: 10.2131/jts.44.611. [DOI] [PubMed] [Google Scholar]
- 24.Himeno S, Fujishiro H. In: Roles of metal transporters in cellular cadmium transport in mammals in Cadmium Toxicity New Aspects in Human Disease, Rice Contamination, and Cytotoxicity. Himeno S, Aoshima K, editors. Springer; Singapore: 2019. pp. 163–178. [Google Scholar]
- 25.Park JH, Hogrebe M, Fobker M, Brackmann R, Fiedler B, Reunert J, Rust S, Tsiakas K, Santer R, Grüneberg M, Marquardt T. SLC39A8 deficiency: Biochemical correction and major clinical improvement by manganese therapy. Genet Med. 2018;20:259–268. doi: 10.1038/gim.2017.106. [DOI] [PubMed] [Google Scholar]
- 26.Park JH, Hogrebe M, Grüneberg M, Duchesne I, Von Der Heiden AL, Reunert J, Schlingmann KP, Boycott KM, Beaulieu CL, Mhanni AA, Innes AM, Hörtnagel K, Biskup S, Gleixner EM, Kurlemann G, Fiedler B, Omran H, Rutsch F, Wada Y, Tsiakas K, Santer R, Nebert DW, Rust S, Marquardt T. SLC39A8 deficiency: a disorder of manganese transport and glycosylation. Am J Hum Genet. 2015;97:894–903. doi: 10.1016/j.ajhg.2015.11.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lin W, Vann DR, Doulias PT, Wang T, Landesberg G, Li X, Ricciotti E, Scalia R, He M, Hand NJ, Rader DJ. Hepatic metal ion transporter ZIP8 regulates manganese homeostasis and manganese-dependent enzyme activity. J Clin Invest. 2017;127:2407–2417. doi: 10.1172/JCI90896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fujishiro H, Himeno S. New insights into the roles of ZIP8, a cadmium and manganese transporter, and its relation to human diseases. Biol Pharm Bull. 2019;42:1076–1082. doi: 10.1248/bpb.b18-00637. [DOI] [PubMed] [Google Scholar]
- 29.Sasaki A, Yamaji N, Yokosho K, Ma JF. Nramp5 is a major transporter responsible for manganese and cadmium uptake in rice. Plant Cell. 2012;24:2155–2167. doi: 10.1105/tpc.112.096925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ishikawa S, Ishimaru Y, Igura M, Kuramata M, Abe T, Senoura T, Hase Y, Arao T, Nishizawa NK, Nakanishi H. Ion-beam irradiation, gene identification, and marker-assisted breeding in the development of low-cadmium rice. Proc Natl Acad Sci USA. 2012;109:19166–19171. doi: 10.1073/pnas.1211132109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yamauchi H, Sun G. Arsenic Contamination in Asia Biological Effects and Preventive Measures. Springer; Singapore: 2019. [DOI] [Google Scholar]
- 32.Zhang XW, Yan XJ, Zhou ZR, Yang FF, Wu ZY, Sun HB, Liang WX, Song AX, Lallemand-Breitenbach V, Jeanne M, Zhang QY, Yang HY, Huang QH, Zhou GB, Tong JH, Zhang Y, Wu JH, Hu HY, de Thé H, Chen SJ, Chen Z. Arsenic trioxide controls the fate of the PML-RARα oncoprotein by directly binding PML. Science. 2010;328:240–243. doi: 10.1126/science.1183424. [DOI] [PubMed] [Google Scholar]
- 33.Wysocki R, Chéry CC, Wawrzycka D, Van Hulle M, Cornelis R, Thevelein JM, Tamás MJ. The glycerol channel Fps1p mediates the uptake of arsenite and antimonite in Saccharomyces cerevisiae. Mol Microbiol. 2001;40:1391–1401. doi: 10.1046/j.1365-2958.2001.02485.x. [DOI] [PubMed] [Google Scholar]
- 34.Liu Z, Shen J, Carbrey JM, Mukhopadhyay R, Agre P, Rosen BP. Arsenite transport by mammalian aquaglyceroporins AQP7 and AQP9. Proc Natl Acad Sci USA. 2002;99:6053–6058. doi: 10.1073/pnas.092131899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shinkai Y, Sumi D, Toyama T, Kaji T, Kumagai Y. Role of aquaporin 9 in cellular accumulation of arsenic and its cytotoxicity in primary mouse hepatocytes. Toxicol Appl Pharmacol. 2009;237:232–236. doi: 10.1016/j.taap.2009.03.014. [DOI] [PubMed] [Google Scholar]
- 36.Lee TC, Ho IC, Lu WJ, Huang JD. Enhanced expression of multidrug resistance-associated protein 2 and reduced expression of aquaglyceroporin 3 in an arsenic-resistant human cell line. J Biol Chem. 2006;281:18401–18407. doi: 10.1074/jbc.M601266200. [DOI] [PubMed] [Google Scholar]
- 37.Calatayud M, Barrios JA, Vélez D, Devesa V. In vitro study of transporters involved in intestinal absorption of inorganic arsenic. Chem Res Toxicol. 2012;25:446–453. doi: 10.1021/tx200491f. [DOI] [PubMed] [Google Scholar]
- 38.Tsukaguchi H, Shayakul C, Berger UV, Mackenzie B, Devidas S, Guggino WB, Van Hoek AN, Hediger MA. Molecular characterization of a broad selectivity neutral solute channel. J Biol Chem. 1998;273:24737–24743. doi: 10.1074/jbc.273.38.24737. [DOI] [PubMed] [Google Scholar]
- 39.Sumi D, Suzukawa K, Himeno S. Arsenic trioxide augments all-trans retinoic acid-induced differentiation of HL-60 cells. Life Sci. 2016;149:42–50. doi: 10.1016/j.lfs.2016.02.054. [DOI] [PubMed] [Google Scholar]
- 40.Villa-Bellosta R, Sorribas V. Arsenate transport by sodium/phosphate cotransporter type IIb. Toxicol Appl Pharmacol. 2010;247:36–40. doi: 10.1016/j.taap.2010.05.012. [DOI] [PubMed] [Google Scholar]
- 41.Meharg AA, Rahman M. Arsenic contamination of Bangladesh paddy field soils: implications for rice contribution to arsenic consumption. Environ Sci Technol. 2003;37:229–234. doi: 10.1021/es0259842. [DOI] [PubMed] [Google Scholar]
- 42.Yamamoto T, Nakamura A, Iwai H, Ishii T, Ma JF, Yokoyama R, Nishitani K, Satoh S, Furukawa J. Effect of silicon deficiency on secondary cell wall synthesis in rice leaf. J Plant Res. 2012;125:771–779. doi: 10.1007/s10265-012-0489-3. [DOI] [PubMed] [Google Scholar]
- 43.Jian FM, Tamai K, Yamaji N, Mitani N, Konishi S, Katsuhara M, Ishiguro M, Murata Y, Yano M. A silicon transporter in rice. Nature. 2006;440:688–691. doi: 10.1038/nature04590. [DOI] [PubMed] [Google Scholar]
- 44.Jian FM, Yamaji N, Mitani N, Xu XY, Su YH, McGrath SP, Zhao FJ. Transporters of arsenite in rice and their role in arsenic accumulation in rice grain. Proc Natl Acad Sci USA. 2008;105:9931–9935. doi: 10.1073/pnas.0802361105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xu XY, McGrath SP, Meharg AA, Zhao FJ. Growing rice aerobically markedly decreases arsenic accumulation. Environ Sci Technol. 2008;42:5574–5579. doi: 10.1021/es800324u. [DOI] [PubMed] [Google Scholar]
- 46.Arao T, Kawasaki A, Baba K, Mori S, Matsumoto S. Effects of water management on cadmium and arsenic accumulation and dimethylarsinic acid concentrations in Japanese rice. Environ Sci Technol. 2009;43:9361–9367. doi: 10.1021/es9022738. [DOI] [PubMed] [Google Scholar]
- 47.Moreno-Jiménez E, Meharg AA, Smolders E, Manzano R, Becerra D, Sánchez-Llerena J, Albarrán Á, López-Piñero A. Sprinkler irrigation of rice fields reduces grain arsenic but enhances cadmium. Sci Total Environ. 2014;485–486:468–473. doi: 10.1016/j.scitotenv.2014.03.106. [DOI] [PubMed] [Google Scholar]
- 48.Hu P, Huang J, Ouyang Y, Wu L, Song J, Wang S, Li Z, Han C, Zhou L, Huang Y, Luo Y, Christie P. Water management affects arsenic and cadmium accumulation in different rice cultivars. Environ Geochem Health. 2013;35:767–778. doi: 10.1007/s10653-013-9533-z. [DOI] [PubMed] [Google Scholar]
- 49.Arao T. In: Mitigation strategies for cadmium and arsenic in rice in Cadmium Toxicity New Aspects in Human Disease, Rice Contamination, and Cytotoxicity. Himeno S, Aoshima K, editors. Springer; Singapore: 2019. pp. 125–138. [DOI] [Google Scholar]
- 50.Horiguchi H. In: Cadmium exposure and its effects on the health status of rice farmers in Akita prefecture in Cadmium Toxicity New Aspects in Human Disease, Rice Contamination, and Cytotoxicity. Himeno S, Aoshima K, editors. Springer; Singapore: 2019. pp. 75–83. [DOI] [Google Scholar]
- 51.Codex Alimentarius Commission. Distribution of the report of the eighth session of the Codex Committee on contaminants in foods (REP14/CF) FAO/WHO; 2014. [Google Scholar]
- 52.Ishikawa S, Makino T, Ito M, Harada K, Nakada H, Nishida I, Nishimura M, Tokunaga T, Shirao K, Yoshizawa C, Matsuyama M, Abe T, Arao T. Low-cadmium rice (Oryza sativa L.) cultivar can simultaneously reduce arsenic and cadmium concentrations in rice grains. Soil Sci Plant Nutr. 2016;62:327–339. doi: 10.1080/00380768.2016.1144452. [DOI] [Google Scholar]