Abstract
Many neurons with cell bodies in laminae III or IV of the spinal dorsal horn possess the neurokinin 1 receptor and have dorsal dendrites that arborize in the superficial dorsal horn. We have performed a confocal microscopic study to determine whether these cells receive inputs from substance P-containing primary afferents. All neurons of this type received contacts from substance P-immunoreactive axons, and in most cases the contacts onto dorsal dendrites were very numerous. A great majority (90–100%) of substance P-immunoreactive varicosities in contact with these cells were also immunoreactive with antibody to calcitonin gene-related peptide, indicating that they were of primary afferent origin. The density of contacts from substance P-immunoreactive varicosities onto these cells was significantly higher than that seen on cholinergic neurons in lamina III (which do not possess the receptor). Electron microscopy revealed that synapses were present at points of contact between substance P-immunoreactive boutons and dorsal dendrites of cells with the neurokinin 1 receptor. Some cells of this type belong to the spinothalamic tract, and we therefore examined neurons with cell bodies in laminae III or IV that possessed the neurokinin 1 receptor and were labeled retrogradely after thalamic injection of cholera toxin B subunit. These cells also received contacts from substance P-immunoreactive axons on their dorsal dendrites. The results of this study indicate that neurons of this type are a major target for substance P-containing primary afferents.
Keywords: substance P, substance P receptor, tachykinins, confocal microscopy, electron microscopy, spinothalamic tract
The primary afferent input to the spinal dorsal horn is arranged in a highly ordered manner, with fine-diameter fibers terminating mainly in the superficial part (laminae I–II), whereas larger afferents reach the deeper laminae (Light and Perl, 1979; Sugiura et al., 1987). Many fine afferents, including some with unmyelinated (C) and small myelinated (Aδ) axons, contain the neuropeptide substance P (McCarthy and Lawson, 1989), and these arborize principally in lamina I and the dorsal part of lamina II. Because various types of peripheral noxious stimulus can release substance P within the spinal cord, it is likely that many substance P-containing afferents are nociceptors (Kantner et al., 1985; Duggan et al., 1988). Another major group consists of unmyelinated afferents that do not contain neuropeptides but that possess a fluoride-resistant acid phosphatase and have axons that end in the ventral half of lamina II as the central terminals of synaptic glomeruli (Ribeiro-da-Silva et al., 1986).
Although C fibers terminate mainly in laminae I and II, neurons with cell bodies in deeper laminae (III–V) frequently respond to C fiber input, and two mechanisms are thought to underlie this phenomenon. Activity may be relayed by neurons in superficial laminae with axons that pass ventrally (Light and Kavookjian, 1988); in addition, some cells in deeper laminae have dendrites that pass dorsally (Szentagothai, 1964; Surmeier et al., 1988) and receive monosynaptic input from primary afferents in laminae I and II (Todd, 1989; De Koninck et al., 1992; Ma et al., 1996).
Antibodies recently have been raised against the neurokinin 1 (NK1) receptor on which substance P acts (Vigna et al., 1994) and have been used to examine its distribution within rat spinal cord. The NK1 receptor is present on many spinal neurons, including a population of large cells in laminae III and IV with prominent dorsal dendrites that enter the superficial laminae (Bleazard et al., 1994; Liu et al., 1994;Brown et al., 1995; Littlewood et al., 1995; Mantyh et al., 1995). In this study we have investigated the possibility that the dorsal dendrites of these neurons are a major target for substance P-containing primary afferents by using immunofluorescence and confocal microscopy to search for contacts between substance P-immunoreactive axons and dorsal dendrites of NK1 receptor-immunoreactive neurons. In the rat all substance P-containing primary afferents are thought to contain calcitonin gene-related peptide (CGRP) (Ju et al., 1987), and CGRP in dorsal horn appears to be derived exclusively from primary afferents (Chung et al., 1988). We therefore have used antibodies against both peptides to identify substance P-containing primary afferents. To determine whether substance P-containing primary afferents form synapses onto the dorsal dendrites of cells in laminae III and IV with the NK1 receptor, we have developed a method for combining confocal and electron microscopy. Some neurons of this type belong to the spinothalamic tract (Marshall et al., 1996), and so we also have searched for contacts formed by substance P-containing axons onto spinothalamic neurons.
MATERIALS AND METHODS
Immunofluorescence. Seven adult albino Swiss rats (either sex, 220–310 gm) were anesthetized deeply and perfused with a fixative containing 4% freshly depolymerized formaldehyde in 0.1 m phosphate buffer (PB). Lumbar spinal cord segments were removed, stored in fixative for at least 4 hr, rinsed in PB, and cut into transverse or parasagittal sections (60 or 70 μm thick) with a vibratome. The sections were treated with 50% ethanol for 30 min to enhance antibody penetration (Llewellyn-Smith and Minson, 1992) and then incubated in 10% donkey serum for 1 hr and subsequently in a mixture of primary antibodies consisting of rabbit antiserum to NK1 receptor (Vigna et al., 1994) (diluted 1:10,000), monoclonal antibody to substance P raised in rat (diluted 1:100–200; Biogenesis, Poole, UK), and in some cases either sheep antiserum to CGRP (diluted 1:5000; Affiniti, Exeter, UK) or affinity-purified goat antibody to choline acetyltransferase (ChAT; diluted 1:100; Chemicon, Temecula, CA), for 1–3 d. After rinsing, the sections were incubated overnight in a mixture of the following species-specific secondary antibodies: biotinylated anti-rabbit IgG (diluted 1:500), anti-rat IgG conjugated to Cy5 (diluted 1:100), and (if primary antibody to CGRP or ChAT was used) anti-goat IgG conjugated to lissamine rhodamine (diluted 1:100). The secondary antibodies all were raised in donkey and were obtained from Jackson ImmunoResearch (West Grove, PA). Finally, sections were incubated overnight in fluorescein conjugated to avidin (diluted 1:10,000; Vector Laboratories, Peterborough, UK), rinsed, and mounted between glass coverslips in antifade medium (Vectashield, Vector Laboratories). All antibody solutions were made up in PBS containing 0.3% Triton X-100 and 5% donkey serum. In control sections pretreatment of the primary antibody cocktail with substance P or CGRP (each 10−6m; Sigma, Poole, UK) or omission of the NK1 receptor antibody selectively abolished the corresponding type of fluorescent staining.
Sections were examined with a Bio-Rad (Hemel Hempstead, UK) MRC 1024 confocal laser scanning microscope equipped with a krypton–argon laser. They were viewed initially with epifluorescence, and NK1 receptor-immunoreactive neurons with cell bodies in laminae III or IV and dendrites that entered the superficial dorsal horn were identified. Because laminar boundaries cannot be identified accurately in material prepared in this way, only cells that were at least 150 μm below the dorsal white matter were studied. Each cell initially was scanned with a 20× objective lens and the 488 nm line of the krypton–argon laser to excite fluorescein (NK1 receptor-like immunoreactivity). Then the immunoreactive neurons were scanned with all three lines of the laser with a 40× or 60× oil immersion objective lens, and an extensive search was made for contacts formed onto the dendrites and cell bodies by varicosities that were substance P-immunoreactive (see Fig. 1). In sections reacted with CGRP antiserum, the dendrites and cell bodies of the NK1 receptor-immunoreactive neurons were scanned sequentially with the three lines of the laser to distinguish varicosities that contained only substance P immunoreactivity from those that were also CGRP-immunoreactive. At least 90 cells were examined in this part of the study, and over one-half of these were from sections that had been reacted with the CGRP antiserum.
Fig. 1.
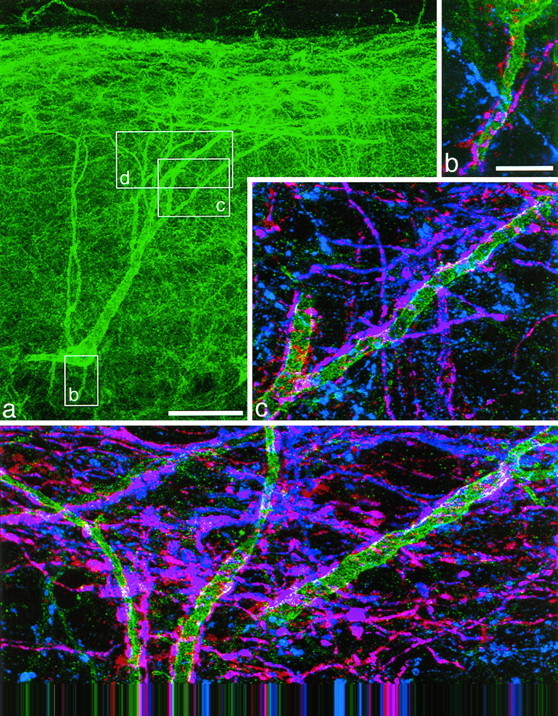
An NK1 receptor-immunoreactive neuron with the cell body 290 μm below the dorsal white matter, which received numerous contacts from substance P-immunoreactive varicosities. This cell is number 10 in Figure 3 and Table 1. NK1 receptor immunoreactivity is shown in green(a–d), substance P immunoreactivity inblue (b–d), and CGRP immunoreactivity inred (b–d). a, A low-magnification view of the cell body and dendritic tree revealed with antibody to the NK1 receptor. The boxes show the regions represented in b–d. b–d, Three regions of the dendritic tree shown at higher magnification. In each region axons with only substance P (blue) or CGRP (red) immunoreactivity are visible, but many show both types of peptide immunoreactivity and appear purple. Many varicosities with both substance P and CGRP immunoreactivity are in contact with the dendrites of the NK1 receptor-immunoreactive neuron; where these overlap with the NK1 receptor immunoreactivity, this appears white. a was obtained from 35 optical sections 1 μm apart, b and dfrom four and five sections, respectively, at 0.7 μm, andc from nine sections at 0.5 μm apart. Scale bars:a, 65 μm; b–d, 10 μm.
Quantitative analysis. A detailed analysis of the density of contacts from substance P-immunoreactive varicosities was performed on 12 NK1 receptor-immunoreactive neurons (four each from three rats) taken from sections reacted with substance P and CGRP antibodies and, for comparison, on six ChAT-immunoreactive neurons in lamina III (two each from three rats). These cells were selected at random from sagittal sections in which penetration of the peptide immunostaining apparently was complete. Complete series of scans at different focal planes (z series) initially were made through the cell with the 20× objective lens to reveal the dendritic arborization (immunostained with NK1 receptor or ChAT antibodies). Projections of these z series were used to reconstruct the cells with the aid of a computer drawing program. The boundary between lamina I and the overlying white matter and the ventral limit of the plexus of substance P-/CGRP-immunoreactive axons were indicated on the drawings. Then the cells were reexamined with the confocal microscope as described above, and the positions of all peptide-immunoreactive varicosities that contacted them were plotted onto these drawings (see Figs. 2, 3, 4).
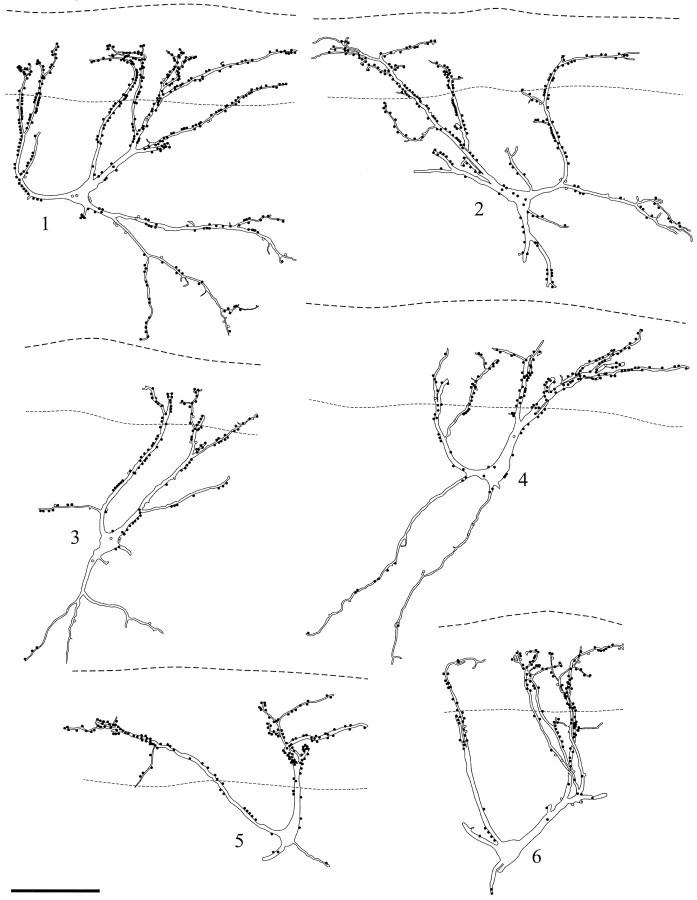
Fig. 2.
Drawings of 6 of the 12 NK1 receptor-immunoreactive neurons for which a detailed analysis of the contacts from substance P-immunoreactive varicosities was performed. The cells are all from parasagittal sections. In each case theupper dashed line represents the border between lamina I and the dorsal white matter, whereas the lower lineindicates the ventral limit of the dense plexus of peptide-immunoreactive axons. Filled circles show contacts from varicosities with both substance P and CGRP immunoreactivities; open circles are those with only substance P immunoreactivity. Scale bar, 100 μm.
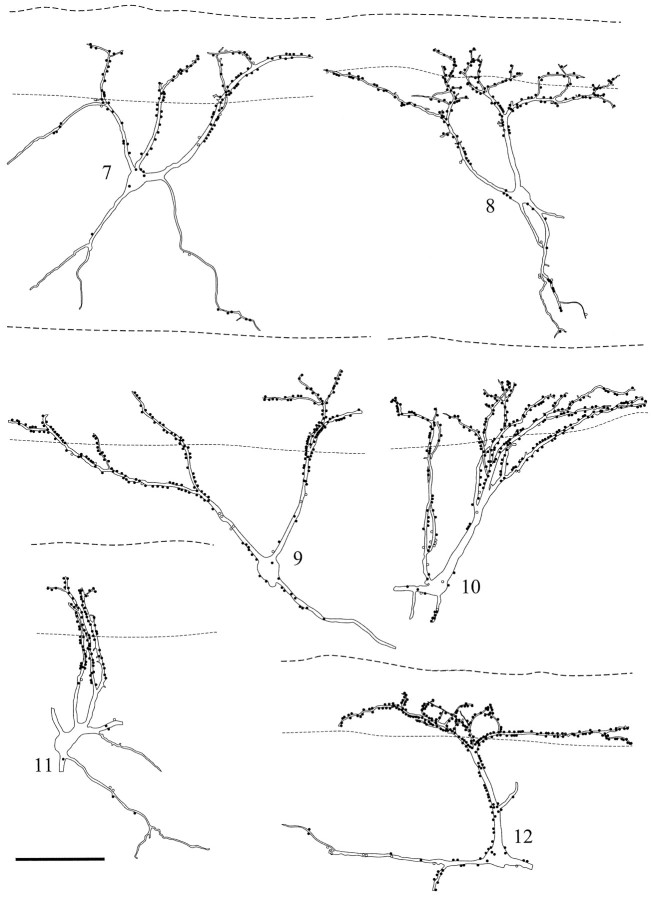
Fig. 3.
Drawings of the other six cells for which a detailed analysis of contacts was performed. The linesand symbols are the same as those used in Figure 2. Scale bar, 100 μm.
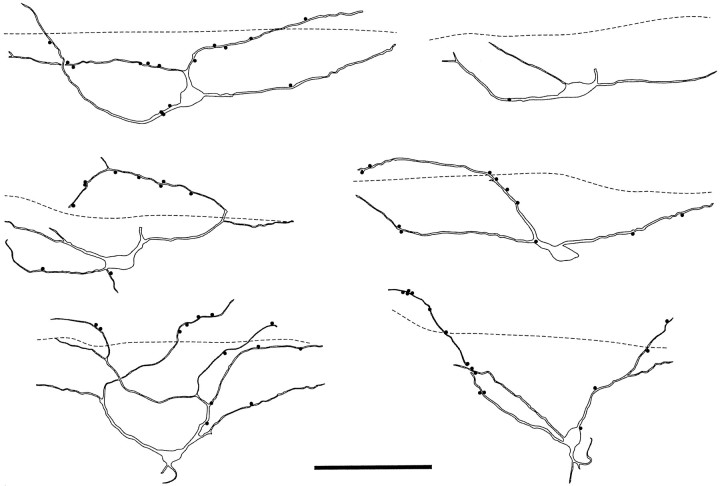
Fig. 4.
Drawings of six ChAT-immunoreactive neurons seen in parasagittal sections. In each case the dashed line represents the ventral limit of the plexus of substance P-containing axons, and filled circles indicate contacts from substance P-immunoreactive varicosities onto the cells. Scale bar, 100 μm.
To estimate the density of contacts onto different parts of the dendritic trees of the NK1 receptor-immunoreactive neurons, we divided segments of the dendrites of these cells into three groups: superficial dorsal dendrites (which lay within the plexus of peptide-immunoreactive axons in lamina I and II); deep dorsal dendrites (which lay deep to this plexus), and ventrally directed dendrites (which were defined as those terminating ventral to the cell body). The ChAT-immunoreactive neurons did not have significant ventrally directed dendrites, although in five of six cases, dendrites did extend into the plexus of substance P-immunoreactive axons. For these cells portions of the dendrites within the plexus and those parts that lay below it were analyzed separately.
Because two-dimensional views of the neurons underestimate the length of dendrites that are passing obliquely through the section, measurements of the depth at different points on the dendritic tree within the vibratome section were determined from the z series. For each cell these measurements were made at points at which dendrites originated from the soma, terminated, left the section, or branched and also where the gradient changed significantly. It was assumed that in between these points dendrites traveled in a straight line and the “real” length of the intervening dendritic segment was, therefore, determined as the square root of the sum of the squares of measured length and difference in depths. Lengths were measured with an image analysis program (KS400, Kontron Electronik, Munich, Germany).
Electron microscopy. To combine confocal and electron microscopy, we made some modifications to the immunofluorescence procedure described above. Three male albino Swiss rats (270–350 gm) were anesthetized and perfused with a fixative consisting of 0.2% glutaraldehyde and 4% formaldehyde in PB. Parasagittal sections (70 μm) were cut with a vibratome, treated with 50% ethanol and then with 1% sodium borohydride, each for 30 min, and rinsed extensively. Then they were incubated for 3 d in the antibodies to NK1 receptor, substance P, and CGRP at the same concentrations as described above, rinsed, and incubated overnight in the following secondary antibodies: anti-rabbit IgG conjugated to fluorescein, anti-goat IgG conjugated to lissamine rhodamine, and anti-rat IgG conjugated to Cy5 (all raised in donkey, diluted 1:100; Jackson ImmunoResearch). The diluent for all antibodies consisted of PBS containing 5% donkey serum, but without Triton X-100. After rinsing, the sections were mounted in Vectashield antifade mounting medium. Sections were viewed with the confocal microscope as described above, and from each of the three rats a single NK1 receptor-immunoreactive neuron with a cell body in lamina III or IV and dorsal dendrites that received numerous contacts from substance P-immunoreactive varicosities was selected. For each of these cells z series were obtained with the 488 nm line and 20× objective to reveal the dendritic tree, and then a selected region of the dendritic tree was scanned sequentially with each of the three laser lines and the 60× objective to record the positions of contacts from substance P- and CGRP-immunoreactive varicosities.
Then the vibratome sections that contained these neurons were processed to reveal NK1 receptor and substance P immunoreactivities with diaminobenzidine (DAB). The sections were removed from the coverslips, rinsed in PBS, and incubated for 3 d in a mixture of species-specific biotinylated secondary antibodies (donkey anti-rabbit IgG and donkey anti-rat IgG, diluted 1:200 in PBS with 5% donkey serum; Jackson ImmunoResearch). After further rinsing, they were incubated for 3 d in extravidin–peroxidase (diluted 1:1000 in PBS; Sigma), and peroxidase activity was revealed with DAB in the presence of hydrogen peroxide. The sections were treated with 1% osmium tetroxide for 30 min, dehydrated in acetone, and flat-embedded in Durcupan between glass coverslips. They were examined with the light microscope, and the selected parts of the NK1 receptor-immunoreactive neurons were identified and, in two cases, photographed. Then one of the coverslips was removed, and the sections were mounted onto blocks of cured resin. Ultrathin sections through the region of interest were cut with a diamond knife, collected on single-slot grids coated with Formvar, stained with lead citrate, and viewed on a Philips CM100 transmission electron microscope.
To identify the immunoreactive structures seen with the electron microscope, we printed the electron micrographs so that the final magnification was exactly equal to that of the corresponding confocal image. Photographs of the immunoreactive dendrite taken with the light microscope were printed at this magnification also. By a comparison of these images it was possible to determine the corresponding fluorescent profile for each DAB-labeled structure.
Labeling of spinothalamic neurons. Because we wanted to identify spinothalamic tract neurons, three adult male albino Swiss rats (280–320 gm) received stereotaxic injections of 4 μl of 1% cholera toxin B subunit (CTb) into the left thalamus. The procedure was performed exactly as described previously (Marshall et al., 1996), except that it was performed under halothane anesthesia. After a 3 or 4 d survival, the rats were reanesthetized and perfused with 4% formaldehyde. The lumbar spinal cord was removed and cut into 70 μm parasagittal sections with a vibratome. After treatment with 50% ethanol, sections were incubated for 1–3 d in a mixture of the following primary antibodies: goat antiserum to CTb (diluted 1:5000; List Biological Laboratories, Campbell, CA), rabbit antiserum to NK1 receptor (1:10,000), and rat monoclonal antibody to substance P (1:200). After incubation overnight in secondary antibodies (biotinylated anti-rabbit, rhodamine anti-goat, and Cy5 anti-rat, as described above) and then in avidin–fluorescein (1:10,000; Vector), they were mounted and viewed with epifluorescence. Antibodies were made up in PBS containing 0.3% Triton X-100 and 5% donkey serum. A search was made for spinothalamic neurons with cell bodies in laminae III or IV, NK1 receptor immunoreactivity, and dendrites that entered the superficial dorsal horn. Then these cells were examined with the confocal microscope, and a search was made for contacts formed by substance P-immunoreactive axons. The brains from these rats were cut into 100 μm coronal sections that were reacted with goat antibody to CTb (1:40,000) and processed by an immunoperoxidase method (Marshall et al., 1996) to reveal the spread of tracer at the injection site.
Antibody specificity. The NK1 receptor antibody was raised against a synthetic peptide corresponding to the 15 amino acid residues at the C terminus of the rat NK1 receptor, which was coupled to bovine thyroglobulin (Vigna et al., 1994). This antibody has been shown to recognize a protein band of 80–90 kDa on Western blots of membranes from cells transfected with the NK1 receptor, and immunostaining can be blocked by addition of the immunizing peptide (Liu et al., 1994; Vigna et al., 1994; Brown et al., 1995).
The monoclonal antibody raised against substance P (Cuello et al., 1979) recognizes the C-terminal part of the peptide and therefore does not distinguish between substance P and the related tachykinins neurokinin A and neurokinin B. Substance P and neurokinin A both are derived from the same gene (preprotachykinin I), and although alternative splicing can result in the production of three different tachykinin-encoding mRNAs, each of these encodes substance P (Helke et al., 1990). Substance P is considerably more abundant than neurokinin A in both dorsal root ganglia and dorsal horn (Ogawa et al., 1985;Moussaoui et al., 1992), and Dalsgaard et al. (1985) have demonstrated that all dorsal root ganglion cells in the rat that possess neurokinin A also contain substance P. Although neurokinin B (which is derived from a different gene, preprotachykinin II) is present in the dorsal horn, it is not detectable in primary afferents and is thought to originate from neurons within the spinal cord (Ogawa et al., 1985;Warden and Young, 1988; Too and Maggio, 1991; Moussaoui et al., 1992). Therefore, although the substance P antibody will detect all of the tachykinins, those boutons that showed both CGRP and substance P-like immunoreactivity (and were therefore of primary afferent origin) will all have contained substance P. For convenience, this type of immunostaining will be referred to as substance P immunoreactivity. The CGRP antiserum was raised in sheep against synthetic rat α-CGRP conjugated to bovine serum albumin with glutaraldehyde.
RESULTS
Confocal microscopy
Many large NK1 receptor-immunoreactive neurons were seen at depths between 150 and 300 μm below the dorsal white matter, and in most cases dorsal dendrites of these cells could be followed into the superficial laminae, where they often branched extensively. With confocal microscopy these branches could be followed even when they entered the dense plexus of immunoreactive dendrites, which is present in lamina I and the dorsal part of lamina II. The dendrites of these cells often extended for long distances in rostral and caudal directions but appeared to be more compact in the mediolateral axis, and the neurons therefore were seen best in parasagittal sections (Figs. 1–3) although, even in these, most cells had some dendrites that left the section. The dendritic morphology of the cells was variable, but, as reported by Brown et al. (1995), some of them had a pyramidal cell body and gave off a dorsal dendrite that passed up into the superficial dorsal horn, whereas others were multipolar. In many cases dendrites that arborized in the deeper laminae could be identified, and these were often as long as the dorsally directed branches. Although dendritic spines were seen occasionally on these cells, they were never numerous.
Immunostaining with the substance P and CGRP antibodies was similar to that reported previously in the dorsal horn. An extensive plexus of immunoreactive fibers and varicosities was present in the superficial part (laminae I and II), and these structures were also present at a lower density throughout the remainder of the dorsal horn. Many axons possessed both types of peptide immunoreactivity; however, axons showing only substance P or only CGRP immunoreactivity also were present (Fig. 1). Although penetration of immunostaining with the substance P and CGRP antibodies appeared to be complete in transverse sections (because approximately equal numbers of immunoreactive structures were seen throughout the depth of the vibratome section), the penetration of peptide immunostaining in parasagittal sections was more variable, and in some cases there was a clear reduction in the density of immunoreactive profiles away from the cut surfaces of the section.
All of the dorsal dendrites belonging to these neurons that could be followed up into the superficial dorsal horn received contacts from substance P-immunoreactive varicosities (Figs. 1, 2, 3). These contacts were often very numerous, particularly on the large primary and secondary dendrites as they passed through lamina III and lamina II, where often it was possible to follow individual substance P-immunoreactive axons that ran along the length of the dorsal dendrites and on which the varicosities formed many contacts. Substance P-immunoreactive varicosities also formed contacts onto the cell bodies and ventrally directed dendrites of most of these neurons, although these contacts were much less numerous than those on the dorsal dendrites. In sections reacted with antibodies to both peptides, a great majority of substance P-immunoreactive varicosities that contacted all parts of these neurons were also CGRP-immunoreactive (Figs. 1, 2, 3), although varicosities that possessed only substance P immunoreactivity also were seen in contact with most of the cells. In addition, the cells received some contacts from varicosities with CGRP, but not substance P, immunoreactivity.
Quantitative analysis
The 12 NK1 receptor-immunoreactive neurons analyzed quantitatively are illustrated in Figures 2 and 3. The parts of the cells that were included in the vibratome sections received contacts from between 100 and 434 substance P-immunoreactive varicosities. The proportion of these varicosities that were also CGRP-immunoreactive varied from 90.2 to 100% (Table 1). The superficial parts of dorsal dendrites of these neurons received between 13.5 and 26.9 contacts from substance P-containing varicosities per 100 μm (mean = 18.8 ± 4.0 SD), whereas the density of contacts per 100 μm on deeper parts of the dorsal dendrites was between 7.7 and 20.1 (mean 13.2 ± 3.9 SD) and on ventral dendrites was between 1.3 and 15.7 (mean 5.5 ± 4.6 SD).
Table 1.
The numbers of contacts formed by varicosities with substance P immunoreactivity onto the 12 NK1 receptor-immunoreactive neurons illustrated in Figures 2 and 3
| Cell number | Total SP contacts | Substance P + CGRP | Substance P only | Percentage of substance P also CGRP |
|---|---|---|---|---|
| 1 | 434 | 414 | 20 | 95.4 |
| 2 | 252 | 247 | 5 | 98 |
| 3 | 135 | 123 | 12 | 91.1 |
| 4 | 188 | 181 | 7 | 96.3 |
| 5 | 160 | 160 | 0 | 100 |
| 6 | 164 | 148 | 16 | 90.2 |
| 7 | 137 | 127 | 10 | 92.7 |
| 8 | 201 | 194 | 7 | 96.5 |
| 9 | 232 | 222 | 10 | 95.7 |
| 10 | 285 | 269 | 16 | 94.4 |
| 11 | 100 | 94 | 6 | 94 |
| 12 | 280 | 272 | 8 | 97.1 |
The six ChAT-immunoreactive neurons analyzed are shown in Figure4. None of these neurons showed NK1 receptor immunoreactivity. Although all of the cells were contacted by substance P-immunoreactive varicosities, the density of contacts was much lower than that seen on the NK1 receptor-immunoreactive neurons (Fig. 4). The portions of dendrites of these cells that lay within the plexus of substance P-containing axons received between 0.9 and 6.9 contacts per 100 μm from substance P-immunoreactive varicosities (mean 3.8 ± 2.3 SD), whereas parts of the dendrites that lay below the plexus received between 0.3 and 2.5 contacts per 100 μm (mean 1.3 ± 0.9 SD).
The differences between the densities of contacts from substance P-immunoreactive varicosities onto the dorsal dendrites of NK1 receptor-immunoreactive or ChAT-immunoreactive neurons were highly significant, both for dendrites within the plexus of substance P-immunoreactive axons (p < 0.0001, unpairedt test) and for those that lay below the plexus (p < 0.0001, unpaired t test).
Electron microscopy
In sections reacted with biotinylated anti-rabbit and anti-rat IgG followed by avidin–peroxidase, profiles that had been labeled with fluorescein (NK1 receptor-immunoreactive) or Cy5 (substance P-immunoreactive) were stained with DAB and could be identified with both light and electron microscopy (Figs.5-7). For each confocal image an approximately corresponding ultrathin section could be identified, although because the ultrathin sections were thinner than the “optical sections” obtained with the confocal microscope and the two may not have been perfectly parallel, not all of the profiles seen on a particular confocal image were present in a single ultrathin section and vice versa. However, by comparing the positions of DAB-labeled profiles in a series of ultrathin sections with profiles that were labeled with Cy5 in the confocal images, it was possible to identify the substance P-immunoreactive profiles that were in contact with the NK1 receptor-immunoreactive dendrites (Fig.6b,d). Twenty-four substance P-immunoreactive varicosities that contacted these dendrites were identified with the electron microscope (six to ten from each of the three rats), and 22 of these were also CGRP-immunoreactive. At all of these contacts the substance P-immunoreactive varicosity was presynaptic to the immunoreactive dendrite at an asymmetrical synapse (Fig.7).
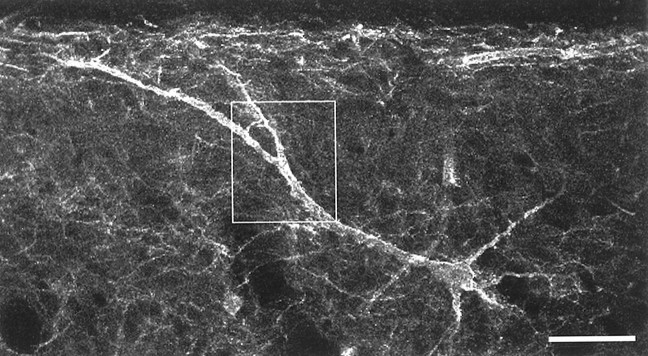
Fig. 5.
One of the NK1 receptor-immunoreactive neurons that was used for the electron microscopic analysis, seen in a parasagittal section. The cell body is 150 μm below the dorsal white matter, and one of the primary dendrites passes obliquely toward the superficial dorsal horn. This image was constructed from three optical sections at 1.5 μm intervals and shows the NK1 receptor immunoreactivity. The boxed area shows the region represented in Figure 6. Scale bar, 50 μm.
Fig. 6.
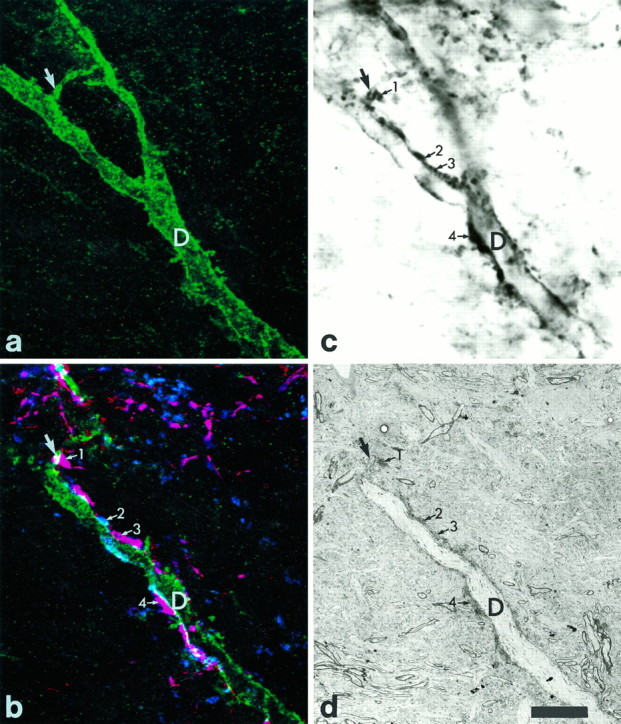
Combined confocal and electron microscopy of part of the dorsal dendrite of the cell shown in Figure 5. a, NK1 receptor immunoreactivity in a series of seven optical sections taken at 0.5 μm intervals. The arrow shows a small branch given off from the main dendritic shaft (D). b, A confocal image obtained from a single optical section, which was taken from the series used to generate a. In this case all three types of immunoreactivity are shown. Green represents NK1 receptor immunoreactivity, whereas blue andred represent substance P and CGRP immunoreactivity, respectively. Many structures show both types of peptide immunoreactivity and appear purple. Several substance P-immunoreactive varicosities are in contact with the NK1 receptor-immunoreactive dendrite (D) or its small branch (large arrow), and four of these are indicated with the numbered small arrows. The varicosity numbered2 shows only substance P immunoreactivity, whereas those numbered 1, 3, and 4 have both substance P and CGRP immunoreactivity. c, The corresponding region seen with light microscopy after the immunoperoxidase reaction and at a focal depth approximately equivalent to the confocal image in b. The main dendritic shaft (D) and part of its small branch (large arrow) are seen clearly, whereas most of the right-hand branch is out of focus. Many of the substance P-immunoreactive varicosities are also visible, including the ones indicated in b (numbered small arrows).d, A low-magnification electron micrograph of the corresponding region. This section is also at a depth nearly equivalent to the confocal image in b. The main dendritic shaft (D) and its small branch (large arrow) can be seen. The substance P-immunoreactive varicosities indicated in b are visible again (numbered small arrows). Scale bar, 10 μm.
Fig. 7.
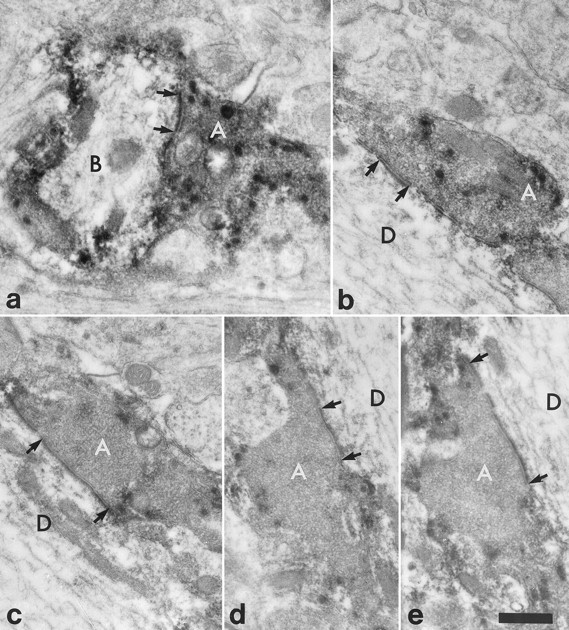
High-magnification electron micrographs to show synapses between the substance P-immunoreactive varicosities indicated with numbered arrows in Figure6b–d and the NK1 receptor-immunoreactive dendrite belonging to the cell illustrated in Figure 5. The micrographs are taken either from the ultrathin section illustrated in Figure6d (b, d) or else from nearby sections in the series (a, c, e).a, In a nearby section the substance P-immunoreactive axon (A; arrow 1 in Fig.6b–d) forms a synapse onto the small branch (B), which was given off from the main dendritic shaft. b–d, The substance P-immunoreactive axonal boutons (A) (numbered 2–4 in Fig.6b–d, respectively) form synapses onto the dendrite (D) of the NK1 receptor-immunoreactive neuron. e, The synapse shown in d is seen more clearly in a nearby ultrathin section. In each case an asymmetrical synaptic specialization is visible (between arrows). Scale bar, 0.5 μm.
Spinothalamic tract neurons
The spread of tracer resulting from injection of CTb into the thalamus was similar in all three experiments, and one example is illustrated in Figure 8. In each case CTb immunoreactivity virtually filled the left thalamus, but there was no spread of tracer to the hypothalamus or midbrain. The appearance and distribution of labeled spinothalamic neurons in the lumbar spinal cord were the same as those reported previously (Marshall et al., 1996). Ten neurons with cell bodies in laminae III or IV on the right side of the lumbar spinal cord (contralateral to the thalamic injection), which showed CTb and NK1 receptor immunoreactivity and had dendrites that entered the superficial dorsal horn, were identified (between two and five from each rat). All of these neurons received contacts from substance P-immunoreactive axons, and in five cases the frequency of contacts was similar to that seen on the NK1 receptor-immunoreactive neurons illustrated in Figures 2 and 3 (Fig. 9). These cells were analyzed quantitatively (as described above), and the densities of contacts from substance P-immunoreactive varicosities were found to vary between 18.2 and 24.7 per 100 μm (mean = 20.7 ± 2.6 SD) for dendrites in the plexus of substance P-immunoreactive axons, between 9.5 and 16.7 per 100 μm (mean = 13.9 ± 3.0 SD) for dorsal dendrites below the plexus, and between 2.7 and 9.4 per 100 μm (mean = 5.0 ± 2.5 SD) for ventrally directed dendrites. These values are within the range observed for the 12 NK1 receptor-immunoreactive cells illustrated in Figures 2 and 3.
Fig. 8.
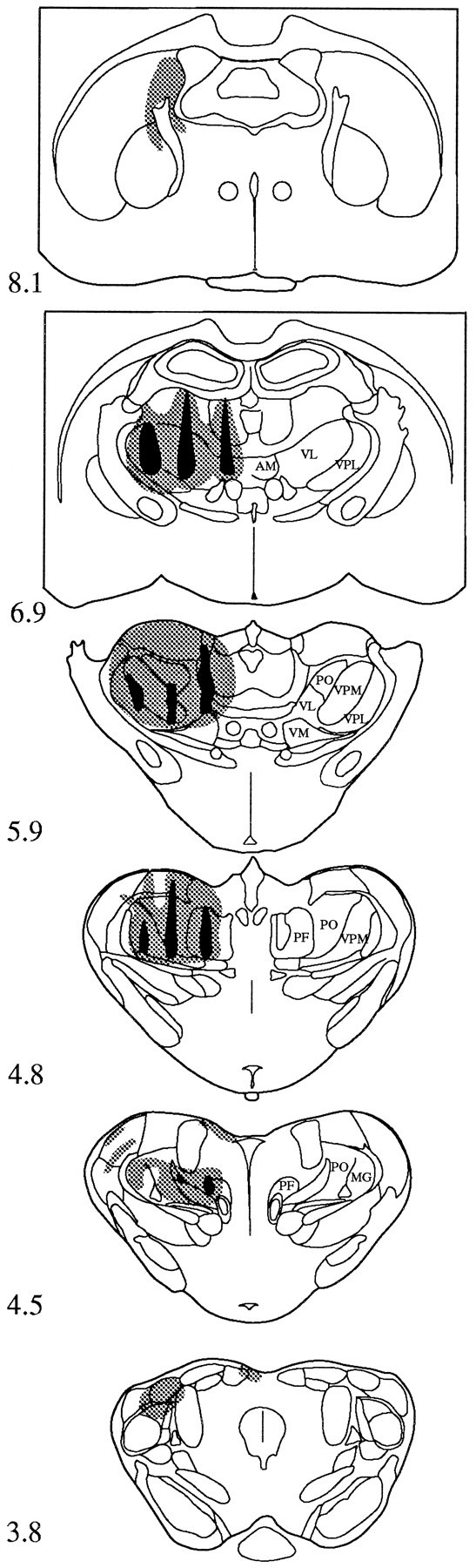
Drawings of the thalamic injection sites in one animal. The area of damage caused by the injection is shown inblack, and the spread of tracer is indicated byshading. The numbers associated with each section refer to the distance in millimeters anterior to the ear bar.AM, Anteromedial thalamic nucleus;MG, medial geniculate nucleus; PF, parafascicular thalamic nucleus; PO, posterior thalamic nuclear group; VM, ventromedial thalamic nucleus;VL, ventrolateral thalamic nucleus; VPM, ventroposterior thalamic nucleus (medial); VPL, ventroposterior thalamic nucleus (lateral).
Fig. 9.
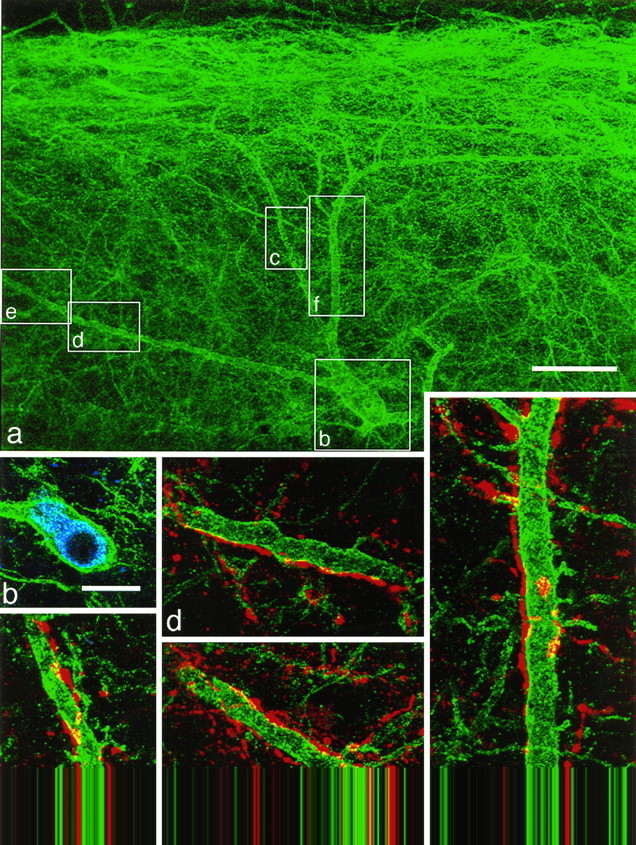
Substance P-immunoreactive contacts onto a spinothalamic neuron with the NK1 receptor. NK1 receptor immunoreactivity is shown in green(a–f), CTb immunoreactivity inblue (b), and substance P immunoreactivity in red (c–f).a, A low-magnification confocal image showing only the NK1 receptor immunoreactivity. The cell body is located 235 μm below the dorsal white matter, and dendrites extend up into the superficial dorsal horn. Boxes indicate the regions illustrated in b–f. b, NK1 receptor and CTb immunoreactivity in a single optical section through the cell body. c–f, Confocal images showing NK1 receptor and substance P immunoreactivity. The dendrites of this neuron receive numerous contacts from substance P-immunoreactive varicosities.a was obtained from nine optical sections 1.5 μm apart, c and e–f from four sections, andd from seven optical sections, each 0.5 μm apart. Scale bars: a, 50 μm; b, 20 μm;c–f, 10 μm.
On the remaining five spinothalamic neurons, fewer contacts were observed; however, these cells were present in sections in which the penetration of substance P immunostaining clearly was incomplete.
DISCUSSION
The main finding of this study was that neurons with cell bodies in lamina III or IV that possess the NK1 receptor and have dendrites that penetrate the superficial dorsal horn receive numerous contacts from substance P-immunoreactive boutons. Although most of these contacts are located in laminae I and II (where substance P-containing axons are common), many of the cells also received contacts on the proximal parts of these dendrites and also on dendrites that arborize in the deeper laminae. A great majority of these boutons are apparently of primary afferent origin because they also contain CGRP, and, from the sample examined with electron microscopy, it appears that most or all of them form asymmetrical synapses onto the dendrites.
To confirm that this relationship was specific, we also examined the distribution of contacts formed by substance P-immunoreactive axons onto another population of lamina III neurons: those that contained ChAT. These were chosen because the ChAT immunoreactivity gives a Golgi-like filling of the neurons and therefore allows contacts on all parts of the dendritic tree to be identified. Although these cells did receive some contacts from substance P-immunoreactive boutons, the density of contacts was significantly lower than that on the dendrites of the NK1 receptor-immunoreactive neurons in the equivalent part of the dorsal horn.
Technical considerations
Immunofluorescence and confocal microscopy offered advantages over other techniques for the present study, in particular the possibility of distinguishing three fluorochromes (Brelje et al., 1993) and the ability to follow the dorsal dendrites of the NK1 receptor-immunoreactive neurons even when they entered the dense plexus of immunoreactive dendrites in the superficial dorsal horn. The high spatial resolution and narrow depth of focus of the confocal microscope meant that contacts from peptide-immunoreactive varicosities onto NK1 receptor-immunoreactive dendrites could be identified reliably. However, to confirm that synapses were present at points of contact, we found it necessary to use electron microscopy. We found that in sections prepared for confocal microscopy, although the primary antibodies already were labeled with fluorescent secondary antibodies, it was still possible to reveal them with DAB by applying biotinylated secondary antibodies and avidin–peroxidase. Therefore, minor modification of the immunostaining protocol (by including glutaraldehyde in the fixative and avoiding the use of Triton X-100) meant that electron microscopy could be performed on the tissue after examination with the confocal microscope. Although both NK1 receptor and substance P immunoreactivity were revealed with DAB, comparison of electron micrographs with confocal images meant that the two types of immunoreactivity could be distinguished easily (Fig. 6b,d) and synapses between substance P-immunoreactive boutons and NK1 receptor-immunoreactive dendrites could be identified readily (Fig.7).
Cells in laminae III and IV with the NK1 receptor
Neurons with cell bodies in laminae III and IV of the dorsal horn and dendrites that penetrate the superficial laminae initially were observed after Golgi impregnation. Although cells of this type were illustrated by Ramón y Cajal (1909), Szentagothai (1964) first drew attention to their significance as a possible output of the substantia gelatinosa (which he regarded as a closed system).Todd (1989) combined Golgi impregnation with dorsal rhizotomy and found that cells of this morphological type received many synapses from degenerating primary afferents in laminae I and II. Basbaum and coworkers (Liu et al., 1994; Brown et al., 1995) first demonstrated that cells with this pattern of dendritic branching possessed the NK1 receptor, and because most neurons in lamina II did not have the receptor and the dendrites of lamina I neurons generally remain within lamina I, they suggested that the dorsal dendrites of the deep cells were likely to represent the major target of substance P-containing primary afferents that terminate in lamina II (Brown et al., 1995). The results of the present study confirm that suggestion, and although it is now clear that the substantia gelatinosa is not a closed system (Light and Kavookjian, 1988), they also support the view that these neurons form one of the major outputs from the superficial dorsal horn (Szentagothai, 1964).
De Koninck et al. (1992) and Ma et al. (1996) have combined intracellular recording and injection with electron microscopic immunocytochemistry to examine the synaptic input from substance P-containing axons to physiologically characterized neurons in cat dorsal horn. They have recorded from various neurons with somata located in laminae II–V and wide-dynamic-range receptive field properties. These cells all showed a slow prolonged EPSP in response to noxious stimuli, which was thought to be mediated by substance P (De Koninck and Henry, 1991), and it is therefore likely that they possessed NK1 receptors. The population included two neurons in lamina V with long dorsal dendrites that reached lamina I, a cell in lamina IV with a small part of its dendritic arbor extending into lamina II, a cell in lamina III with dorsal dendrites that entered the superficial dorsal horn, and another located on the border between laminae II and III. All of these cells received numerous contacts from substance P-immunoreactive axons, many of which formed synapses. Ma et al. (1996) performed postembedding immunocytochemistry on ultrathin sections from one of these neurons and found that 30% of the substance P-immunoreactive boutons were also CGRP-immunoreactive.
NK1 receptor-immunoreactive neurons with cell bodies in lamina V and dendrites that reach the superficial laminae are seen occasionally in rat dorsal horn (Brown et al., 1995), and these may correspond to the lamina V cells reported by De Koninck et al. (1992). The lamina III cell of De Koninck et al. probably belongs to the same population as the NK1 receptor-immunoreactive neurons seen in this study; however, it had numerous dendritic spines on the dendrites that remained in lamina III, whereas spines rarely were seen on any parts of the dendrites of the neurons that we examined. A more significant difference between the findings of Ma et al. (1996) and those reported here is that we found that a much higher proportion of the substance P-immunoreactive profiles contacting lamina III/IV cells also showed CGRP immunoreactivity. As discussed by Ma et al., this probably reflects the lower sensitivity of the postembedding method for detecting CGRP.
Because many substance P-containing primary afferents are thought to be nociceptors (Kantner et al., 1985; Duggan et al., 1988; McCarthy and Lawson, 1989), it is likely that cells of the type reported here receive a significant nociceptive input. Mantyh et al. (1995) have shown that after acute noxious stimulation the dorsal dendrites of these neurons showed internalization of the NK1 receptor, presumably because of its activation by substance P released from primary afferents in response to the stimulus. Although substance P is likely to diffuse from its site of release and act at nonsynaptic locations by “volume” transmission, the density of innervation of these neurons by substance P-containing afferents suggests that their dendrites will be exposed to particularly high concentrations of the peptide after its release. The presence of synapses between substance P-containing primary afferents and dorsal dendrites of NK1 receptor-immunoreactive neurons is probably more significant for glutamatergic transmission. Substance P-containing primary afferents also are enriched with glutamate, which they are thought to use as a fast transmitter (De Biasi and Rustioni, 1988), and this action presumably occurs at the asymmetrical synapses that we observed here. In many cases it was possible to follow a single peptide-immunoreactive axon along the dorsal dendrite of a NK1 receptor-immunoreactive neuron, and it was common for numerous varicosities apparently derived from the same axon to form contacts onto one of these dendrites. This suggests that certain substance P-containing primary afferents may have a very secure synaptic linkage onto neurons with cell bodies in the deeper laminae.
We have reported previously that some cells of this type belong to the spinothalamic tract (Marshall et al., 1996), and the present results show that these cells also receive contacts from substance P-containing axons. For five of the ten spinothalamic cells examined, the density of contacts appeared to be relatively low, and therefore it is possible that some of these neurons receive a less dense innervation from substance P-containing axons. However, because the sections in which these cells were located showed incomplete penetration of substance P immunoreactivity, this may represent a false-negative result. In either case the presence of a dense innervation of some spinothalamic cells by substance P-containing axons (Fig. 9), many of which presumably function as nociceptors, is likely to be important because of the probable involvement of this tract in pain mechanisms (Willis and Coggeshall, 1991).
Footnotes
This work was supported by grants from the Wellcome Trust and the Robertson Trust, which are gratefully acknowledged. We thank Dr. S. Vigna for donating antiserum to the neurokinin-1 receptor and Mr. M. Neilson for technical assistance.
Correspondence should be addressed to Dr A. J. Todd, Laboratory of Human Anatomy, University of Glasgow, Glasgow G12 8QQ, U.K.
REFERENCES
- 1.Bleazard L, Hill RG, Morris R. The correlation between the distribution of NK1 receptor and the actions of tachykinin agonists in the dorsal horn of the rat indicates that substance P does not have a functional role on substantia gelatinosa (lamina II) neurons. J Neurosci. 1994;14:7655–7664. doi: 10.1523/JNEUROSCI.14-12-07655.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Brelje TC, Wessendorf MW, Sorenson RL. Multicolor laser scanning confocal immunofluorescence microscopy: practical application and limitation. In: Matsumoto B, editor. Cell biological applications of confocal microscopy. Academic; San Diego: 1993. pp. 98–181. [Google Scholar]
- 3.Brown JL, Liu H, Maggio JE, Vigna SR, Mantyh PW, Basbaum AI. Morphological characterization of substance P receptor-immunoreactive neurons in rat spinal cord and trigeminal nucleus caudalis. J Comp Neurol. 1995;356:327–344. doi: 10.1002/cne.903560302. [DOI] [PubMed] [Google Scholar]
- 4.Chung K, Lee WT, Carlton SM. The effects of dorsal rhizotomy and spinal cord isolation on calcitonin gene-related peptide-labeled terminals in the rat lumbar dorsal horn. Neurosci Lett. 1988;90:27–32. doi: 10.1016/0304-3940(88)90781-1. [DOI] [PubMed] [Google Scholar]
- 5.Cuello AC, Galfre G, Milstein C. Detection of substance P in the central nervous system by a monoclonal antibody. Proc Natl Acad Sci USA. 1979;76:3532–3536. doi: 10.1073/pnas.76.7.3532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dalsgaard CJ, Haegerstrand A, Theodorsson-Norheim E, Brodin E, Hökfelt T. Neurokinin A-like immunoreactivity in rat primary sensory neurons: coexistence with substance P. Histochemistry. 1985;83:37–39. doi: 10.1007/BF00495297. [DOI] [PubMed] [Google Scholar]
- 7.De Biasi S, Rustioni A. Glutamate and substance P coexist in primary afferent terminals in the superficial laminae of the spinal cord. Proc Natl Acad Sci USA. 1988;85:7820–7824. doi: 10.1073/pnas.85.20.7820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.De Koninck Y, Henry JL. Substance P-mediated slow EPSP in vivo in dorsal horn neurons elicited by noxious stimulation. Proc Natl Acad Sci USA. 1991;88:11344–11348. doi: 10.1073/pnas.88.24.11344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.De Koninck Y, Ribeiro-da-Silva A, Henry JL, Cuello AC. Spinal neurons exhibiting a specific nociceptive response receive abundant substance P-containing synaptic contacts. Proc Natl Acad Sci USA. 1992;89:5073–5077. doi: 10.1073/pnas.89.11.5073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Duggan AW, Hendry IA, Morton CR, Hutchison WD, Zhao ZQ. Cutaneous stimuli releasing immunoreactive substance P in the dorsal horn of the cat. Brain Res. 1988;451:261–273. doi: 10.1016/0006-8993(88)90771-8. [DOI] [PubMed] [Google Scholar]
- 11.Helke CJ, Krause JE, Mantyh PW, Couture R, Bannon MJ. Diversity of tachykinin peptidergic neurons: multiple peptides, receptors, and regulatory mechanisms. FASEB J. 1990;4:1606–1615. [PubMed] [Google Scholar]
- 12.Ju G, Hökfelt T, Brodin E, Fahrenkrug J, Fischer JA, Frey P, Elde RP, Brown JC. Primary sensory neurons of the rat showing calcitonin gene-related peptide immunoreactivity and their relation to substance P-, somatostatin-, galanin-, vasoactive intestinal polypeptide-, and cholecystokinin-immunoreactive ganglion cells. Cell Tissue Res. 1987;247:417–431. doi: 10.1007/BF00218323. [DOI] [PubMed] [Google Scholar]
- 13.Kantner RM, Kirby ML, Goldstein BD. Increase in substance P in the dorsal horn during a chemogenic nociceptive stimulus. Brain Res. 1985;338:196–199. doi: 10.1016/0006-8993(85)90268-9. [DOI] [PubMed] [Google Scholar]
- 14.Light AR, Kavookjian AM. Morphology and ultrastructure of physiologically identified substantia gelatinosa (lamina II) neurons with axons that terminate in deeper dorsal horn (laminae III–V). J Comp Neurol. 1988;267:172–189. doi: 10.1002/cne.902670203. [DOI] [PubMed] [Google Scholar]
- 15.Light AR, Perl ER. Reexamination of the dorsal root projection to the spinal dorsal horn, including observations on the differential termination of coarse and fine fibers. J Comp Neurol. 1979;186:117–131. doi: 10.1002/cne.901860202. [DOI] [PubMed] [Google Scholar]
- 16.Littlewood NK, Todd AJ, Spike RC, Watt C, Shehab SAS. The types of neuron in spinal dorsal horn which possess neurokinin-1 receptors. Neuroscience. 1995;66:597–608. doi: 10.1016/0306-4522(95)00039-l. [DOI] [PubMed] [Google Scholar]
- 17.Liu H, Brown JL, Jasmin L, Maggio JE, Vigna SR, Mantyh PW, Basbaum AI. Synaptic relationship between substance P and the substance P receptor: light and electron microscopic characterization of the mismatch between neuropeptides and their receptors. Proc Natl Acad Sci USA. 1994;91:1009–1013. doi: 10.1073/pnas.91.3.1009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Llewellyn-Smith IJ, Minson JB. Complete penetration of antibodies into vibratome sections after glutaraldehyde fixation and ethanol treatment: light and electron microscopy for neuropeptides. J Histochem Cytochem. 1992;40:1741–1749. doi: 10.1177/40.11.1431060. [DOI] [PubMed] [Google Scholar]
- 19.Ma W, Ribeiro-da-Silva A, De Koninck Y, Radhakrishnan V, Henry JL, Cuello AC. Quantitative analysis of substance P-immunoreactive boutons on physiologically characterized dorsal horn neurons in the cat lumbar spinal cord. J Comp Neurol. 1996;376:45–64. doi: 10.1002/(SICI)1096-9861(19961202)376:1<45::AID-CNE3>3.0.CO;2-O. [DOI] [PubMed] [Google Scholar]
- 20.Mantyh PW, DeMaster E, Malhotra A, Ghilardi JR, Rogers SD, Mantyh CR, Liu H, Basbaum AI, Vigna SR, Maggio JE, Simone DA. Receptor endocytosis and dendrite reshaping in spinal neurons after somatosensory stimulation. Science. 1995;268:1629–1632. doi: 10.1126/science.7539937. [DOI] [PubMed] [Google Scholar]
- 21.Marshall GE, Shehab SAS, Spike RC, Todd AJ. Neurokinin-1 receptors on lumbar spinothalamic neurons in the rat. Neuroscience. 1996;72:255–263. doi: 10.1016/0306-4522(95)00558-7. [DOI] [PubMed] [Google Scholar]
- 22.McCarthy PW, Lawson SN. Cell type and conduction velocity of rat primary sensory neurons with substance P-like immunoreactivity. Neuroscience. 1989;28:7745–753. doi: 10.1016/0306-4522(89)90019-5. [DOI] [PubMed] [Google Scholar]
- 23.Moussaoui SM, LePrado N, Bonici B, Faucher DC, Cuiné F, Laduron PM, Garret C. Distribution of neurokinin B in rat spinal cord and peripheral tissues: comparison with neurokinin A and substance P and effects of neonatal capsaicin treatment. Neuroscience. 1992;48:969–978. doi: 10.1016/0306-4522(92)90285-a. [DOI] [PubMed] [Google Scholar]
- 24.Ogawa T, Kanazawa I, Kimura S. Regional distribution of substance P, neurokinin α, and neurokinin β in rat spinal cord, nerve roots, and dorsal root ganglia and the effects of dorsal root section or spinal transection. Brain Res. 1985;359:152–157. doi: 10.1016/0006-8993(85)91423-4. [DOI] [PubMed] [Google Scholar]
- 25.Ramón y Cajal S. Histologie du système nerveux de l’homme et des vertébrés, Vol I. Maloine; Paris: 1909. [Google Scholar]
- 26.Ribeiro-da-Silva A, Castro-Lopes JM, Coimbra A. Distribution of glomeruli with fluoride-resistant acid phosphatase (FRAP)-containing terminals in the substantia gelatinosa of the rat. Brain Res. 1986;377:323–329. doi: 10.1016/0006-8993(86)90875-9. [DOI] [PubMed] [Google Scholar]
- 27.Sugiura Y, Lee CL, Perl ER. Central projections of identified, unmyelinated (C) afferent fibers innervating mammalian skin. Science. 1987;234:358–361. doi: 10.1126/science.3764416. [DOI] [PubMed] [Google Scholar]
- 28.Surmeier DJ, Honda CN, Willis WD. Natural groupings of spinothalamic neurons based upon cutaneous stimulation. Physiological and anatomical features. J Neurophysiol. 1988;59:833–860. doi: 10.1152/jn.1988.59.3.833. [DOI] [PubMed] [Google Scholar]
- 29.Szentagothai J. Neuronal and synaptic arrangement in the substantia gelatinosa Rolandi. J Comp Neurol. 1964;122:219–239. doi: 10.1002/cne.901220207. [DOI] [PubMed] [Google Scholar]
- 30.Todd AJ. Cells in laminae III and IV of rat spinal dorsal horn receive monosynaptic primary afferent input in lamina II. J Comp Neurol. 1989;289:676–686. doi: 10.1002/cne.902890411. [DOI] [PubMed] [Google Scholar]
- 31.Too HP, Maggio JE. Immunocytochemical localization of neuromedin K (neurokinin B) in rat spinal ganglia and cord. Peptides. 1991;12:431–443. doi: 10.1016/0196-9781(91)90081-y. [DOI] [PubMed] [Google Scholar]
- 32.Vigna SR, Bowden JJ, McDonald DM, Fisher J, Okamoto A, McVey DC, Payan DG, Bunnett NW. Characterization of antibodies to the rat substance P (NK-1) receptor and to a chimeric substance P receptor expressed in mammalian cells. J Neurosci. 1994;14:834–845. doi: 10.1523/JNEUROSCI.14-02-00834.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Warden MK, Young WS. Distribution of cells containing mRNAs encoding substance P and neurokinin B in the rat central nervous system. J Comp Neurol. 1988;272:90–113. doi: 10.1002/cne.902720107. [DOI] [PubMed] [Google Scholar]
- 34.Willis WD, Coggeshall RE. Sensory mechanisms of the spinal cord. Plenum; New York: 1991. [Google Scholar]