Abstract
D1 receptors located on striatonigral neurons and D2 receptors located, together with A2Areceptors, on striatopallidal neurons are known to interact functionally. Using in situ hybridization, we examined the effects of D1 and D2 agonists and of an A2A antagonist on c-fos mRNA in identified striatal neurons and in globus pallidus. The full D1agonist, SKF 82958 (1 mg/kg), induced a homogenous increase ofc-fos mRNA in the striatum. This increase occurred to a similar extent in D1 and D2 receptor-containing striatal neurons. Conversely, the D2 agonist, quinelorane (2 mg/kg), decreased c-fos mRNA in these populations but increased it in globus pallidus. The adenosine A2A receptor antagonist, SCH 58261 (5 mg/kg), also decreased c-fosmRNA in D2 receptor-containing neurons in striatum but did not affect pallidal c-fos mRNA. Concomitant administration of either D1 plus D2 agonists or D1 agonist plus A2A antagonist caused a potentiation of c-fos mRNA in striatal neurons expressing the D1 receptor and in globus pallidus. However, only the combination of D1 and D2 agonists modified the c-fos mRNA expression to a “patchy” distribution. Our data show that (1) c-fos expression can be activated through D1 and inhibitedthrough A2A or D2 receptors in both striatal output pathways in normal rats, and (2) D2 receptor stimulation as well as A2A receptor blockade can interact with D1 receptor activation to potentiatec-fos expression in the striatum and the globus pallidus. The data also suggest that the topological alteration ofc-fos expression after coadministration of D1 and D2 agonists involves D2receptors located on interneurons or presynaptically on dopaminergic nerve terminals.
Keywords: In situ hybridization, phenotypical characterization, immediate early gene, dopamine–adenosine interactions, synergistic effects, striatal output pathways, globus pallidus
The basal ganglia are involved in the integration of sensorimotor, associative, and limbic information to produce motor behaviors. The central component of these structures, the striatum, integrates excitatory glutamatergic inputs from cortex, thalamus, and limbic areas, with dopaminergic inputs from mesencephalon. It is composed of a large proportion of medium-sized spiny output neurons (95%) and of interneurons (5%). Striatal output neurons are GABAergic and project to either substantia nigra (pars reticulata) or globus pallidus and differ in their neuropeptide content: the striatonigral pathway contains substance P/dynorphin and the striatopallidal enkephalin (for review, see Graybiel, 1990; Gerfen and Wilson, 1996).
Dopamine regulates striatal neurotransmission via two types of receptor families, D1-type (D1 and D5) and D2-type (D2, D3, D4) receptors, which have distinct pharmacological profiles and mechanisms of transduction (Creese et al., 1983; Jaber et al., 1996). It has been suggested that dopamine differentially regulates the two striatal output pathways and that a balanced control is essential for the proper function of the extrapyramidal motor system (for review, see Alexander and Crutcher, 1990; Gerfen, 1992). Accordingly, several anatomical studies have demonstrated a segregation of D1 and D2receptors, respectively, in striatonigral/substance P and striatopallidal/enkephalin neurons (Gerfen et al., 1990; Le Moine et al., 1990a, 1991; Hersch et al., 1995; Le Moine and Bloch, 1995, 1996;Yung et al., 1996). However, many physiological data indicate synergistic effects after coactivation of D1- and D2-type receptors (for review, see Waddington and Daly, 1993; White and Hu, 1993).
In the basal ganglia A2A receptors are restricted to striatopallidal/D2-containing neurons and, in contrast to D2 receptors, are not present on dopaminergic nerve terminals and are virtually absent from cholinergic interneurons (Schiffmann et al., 1991; Fink et al., 1992; Augood and Emson, 1994;Svenningsson et al., 1997). An alternative way to investigate how D1/D2 interactions occur is to study how adenosine modulates neurotransmission via adenosine A2Areceptors and how they can be involved in interactions with D1 receptor-mediated effects. Indeed, it has been shown that dopamine acting on D2 receptors and adenosine acting on A2A receptors have opposing actions on neurotransmitter release, gene expression, and several motor behaviors (for review, seeFerré et al., 1992; Ongini and Fredholm, 1996). Accordingly, selective A2A antagonists share with D2agonists the ability to potentiate motor effects induced by D1 receptor agonists as well as D1-inducedc-fos expression in dopamine-depleted striatum (Jiang et al., 1993; Pinna et al., 1996; Pollack and Fink, 1996).
In this context, detailed analysis of the modulation of D1 or D2 agonist-mediated effects by an A2A antagonist may help to elucidate the D1/D2 interactions in the basal ganglia. We therefore used sensitive in situ hybridization with riboprobes to examine how pharmacological treatments involving dopamine or adenosine receptors might up- or downregulate the expression ofc-fos in the basal ganglia. In particular, c-fosexpression was studied in phenotypically identified striatal neurons, with double-labeling, after challenges with selective compounds acting at D1, D2, and A2Areceptors given alone or in combination.
MATERIALS AND METHODS
Pharmacological manipulations and tissue preparation.All experiments have been performed in accordance with the guidelines of the French Agriculture and Forestry Ministry (decree 87849, license 01499) and with the Centre National de la Recherche Scientifique approval. Adult male Sprague Dawley rats (200–280 gm) (Iffa Credo, France) were maintained in standard housing conditions several days before the experiments. Animals were treated with systemic injections of saline (NaCl 0.9%); ±SKF-82958 (Research Biochemicals, Natick, MA), a full dopamine receptor agonist that has a 200-fold selectivity for D1 over D2 receptors (Andersen and Jansen, 1990); quinelorane or LY-163,502 (Research Biochemicals), a dopamine receptor agonist that conversely shows at least a 50-fold selectivity for D2 over D1 receptors (Bymaster et al., 1986; Andersen and Jansen, 1990); or SCH-58261 (Schering-Plough, Milan, Italy), an adenosine receptor antagonist that is 60-fold selective for A2A over A1 receptors (Zocchi et al., 1996). All rats had been handled the day before the injection and had received two injections. The different treatment groups were as follows: saline plus saline (n = 5), quinelorane 2 mg/kg plus saline (n = 4), SKF-82958 0.5 mg/kg plus saline (n = 3), SKF-82958 1 mg/kg plus saline (n = 5), SKF-82958 2 mg/kg plus saline (n = 2), SCH-58261 5 mg/kg plus saline (n = 4), SKF-82958 1 mg/kg plus quinelorane 2 mg/kg (n = 5), SKF-82958 1 mg/kg plus SCH-58261 5 mg/kg (n = 4), or quinelorane 2 mg/kg plus SCH-58261 5 mg/kg (n = 5). SKF-82958 and quinelorane were dissolved in saline, whereas SCH-58261 was dissolved in saline/5% Tween 80 after careful sonication. Drugs were injected intraperitoneally, 0.5 ml per injection, and the rats were decapitated 1 hr after the injections. The brains were dissected out, frozen over liquid nitrogen, and then sectioned into 10 μm sections, collected on gelatin-coated slides, and stored at −80°C until used.
Probe synthesis. 35S-labeled cRNA probes were prepared by in vitro transcription from cDNA clones corresponding to fragments of the rat c-fos cDNA (Curran et al., 1987) (a gift from Dr. T. Curran, Roche Institute of Molecular Biology, Nutley, NJ), rat D1 and D2 dopamine receptor cDNAs (Monsma et al., 1989, 1990) (a gift from Dr. D. Sibley, National Institute of Health, NINDS, Bethesda, MD), and rat μ-opioid receptor cDNA (Thompson et al., 1993) (a gift from Dr. S. J. Watson, University of Michigan, Ann Arbor, MI). Transcriptions were performed from 50 ng of linearized plasmid, using either 35S-UTP (>1000 Ci/mmol; DuPont de Nemours, Les Ulis, France) or digoxigenin-11-UTP (Boehringer Mannheim, Meylan, France) and SP6, T3, or T7 RNA polymerases as described by Le Moine and Bloch (1995). After alkaline hydrolysis to obtain 250 bp cRNA fragments, the35S-labeled probes were purified on G50-Sephadex. The35S-labeled probes and the digoxigenin-labeled probes were precipitated in 3 m sodium acetate/absolute ethanol (0.1:2.5, v/v), pH 5.
Single detection of c-fos mRNA on cryostat sections.Sections were hybridized as described by Le Moine and Bloch (1995,1996) with minor modifications. Cryostat sections were post-fixed in 4% paraformaldehyde (PFA) for 5 min at room temperature, rinsed twice in 4× SSC, and placed into 0.25% acetic anhydride in 0.1m triethanolamine/4× SSC, pH 8, for 10 min at room temperature. After dehydration, the sections were hybridized overnight at 55°C with 106 cpm of 35S-labeled cRNA probe in 50 μl of hybridization solution (20 mmTris-HCl, 1 mm EDTA, 300 mm NaCl, 50% formamide, 10% dextran sulfate, 1× Denhardt’s, 250 μg/ml yeast tRNA, 100 μg/ml salmon sperm DNA, 100 mm DTT, 0.1% SDS, and 0.1% sodium thiosulfate). After 20 min of RNase A treatment (20 mg/ml), the sections were washed with 2× SSC (5 min, twice), 1× SSC (5 min), 0.5× SSC (5 min) at room temperature, and rinsed in 0.1× SSC at 65°C (30 min, twice) before dehydration (the latter SSC washes contained 1 mm DTT). Sections either were exposed on x-ray films (Kodak BIOMAX, Rochester, NY) for 3–6 d or dipped into Ilford K5 emulsion, exposed for 7 weeks, developed, and stained with toluidine blue.
Simultaneous detection of c-fos mRNA with D1 or D2 mRNAs on cryostat sections. Two combinations of probes were used for the simultaneous detection of two mRNAs on a single section: a 35S-labeled c-fosprobe in combination with digoxigenin-labeled D1 or D2 probes. Cryostat sections were pretreated as mentioned above. After dehydration the sections were hybridized overnight at 55°C with a combination of 35S- and digoxigenin-labeled probes (106 cpm of 35S-labeled probe and 10–20 ng of digoxigenin-labeled probe in 50 μl of hybridization solution). After 20 min of RNase A treatment at 37°C (20 μg/ml), the slides were washed in various concentrations of SSC as mentioned above, but without DTT. After washing, the sections were put in 0.1× SSC at room temperature and then processed directly for detection of the digoxigenin signal. The sections were rinsed twice for 5 min in buffer A (1 m NaCl, 0.1 m Tris, and 2 mm MgCl2, pH 7.5) and then for 30 min in buffer A containing 3% normal goat serum and 0.3% Triton X-100. After 5 hr of incubation at room temperature with alkaline phosphatase-conjugated anti-digoxigenin antiserum (Boehringer Mannheim; 1:1000 in buffer A, 3% normal goat serum, and 0.3% Triton X-100), the sections were rinsed in buffer A (5 min, twice) and then for 10 min twice in STM buffer (1 m NaCl, 0.1 m Tris, and 5 mm MgCl2, pH 9.5), and finally for 10 min twice in 0.1 m STM buffer, pH 9.5 (0.1 mNaCl, 0.1 m Tris, and 5 mmMgCl2, pH 9.5). Then the sections were incubated overnight in the dark at room temperature in 0.1 m STM buffer, pH 9.5, containing 0.34 mg/ml nitroblue tetrazolium and 0.18 mg/ml bromo-chloro-indolylphosphate. They were rinsed in 0.1m STM buffer, pH 9.5, and then in 1× SSC, dried, and dipped into Ilford K5 emulsion (diluted 1:3 in 1× SSC). After being exposed for 10 weeks in the dark, the sections were developed and mounted without counterstaining.
Counting of labeled neurons. Labeled neurons both from single-labeling and double-labeling experiments (exposure times: 7 weeks for single in situ hybridization and 10 weeks for double in situ hybridization) were counted as previously described on similar material (Le Moine and Bloch, 1995). Accordingly, a labeled neuron corresponded to a density of silver grains at least twofold higher than background. One section per animal was analyzed for counting in single in situ hybridization, and one section per animal was counted for the double labeling. The densities ofc-fos mRNA-containing neurons were studied in the striatum (+1 mm from bregma) and globus pallidus (−0.8 mm from bregma) according to Swanson (1992). The areas examined were 2–4 mm2 for the caudate putamen and 1.5–2 mm2 for the globus pallidus. The labeled neurons were counted using an image analyzer system for cartography (HISTO 200, Biocom, Les Ulis, France). For double in situ hybridization, quantification was performed only on the sections with simultaneous detection of c-fos and D2 mRNAs, and thec-fos mRNA-labeled neurons were divided into two populations: the D2 mRNA-positive (+) and D2mRNA-negative (−) neurons. The densities ofc-fos-expressing neurons (number of c-fosmRNA-positive neurons per mm2) were pooled and averaged for each group, and statistical analysis was performed by a two-way ANOVA, followed by post hoc t tests corrected for the experiment-wise α level by the Bonferroni correction.
RESULTS
Effects of D1 and D2 agonists onc-fos expression in the striatum and in the globus pallidus
Under control conditions (i.e., saline-treated rats), neurons containing c-fos mRNA were observed in several cortical areas, especially the endopiriform and piriform cortices, in the septum and in the caudate putamen and nucleus accumbens (Fig.1). The densities ofc-fos-positive neurons (mean ± SEM) were 35.25 ± 4.34 per mm2 in the caudate putamen and 24.2 ± 3.4 per mm2 in the globus pallidus (Table1).
Fig. 1.

D1/D2 and D1/A2A receptor interactions onc-fos expression. Dark-field photomicrographs afterin situ hybridization with a 35S-labeled riboprobe show the localization of c-fos mRNA-containing neurons in the striatum after saline (A), D1 agonist SKF-82958 (B), D1 agonist SKF-82958 + A2A antagonist SCH-58261 (C), and D1 agonist SKF-82958 + D2 agonist quinelorane (D). Under basal conditions (A) c-fosmRNA-containing neurons are few and scattered in the caudate putamen (cp) and the nucleus accumbens (acb).c-fos is induced after the D1 agonist both in the caudate putamen and the nucleus accumbens (B). As compared with D1 agonist + A2A antagonist (C), the combined treatment with D1 + D2 agonists potentiates the D1-induced expression of c-fos with a heterogeneous “patchy” pattern (arrowheads inD). Cortical expression of c-fos in layer VIb is seen clearly after D1 agonist alone or in combination with either A2A antagonist or plus D2 agonists (arrows in B–D). Magnification, 11×.
Table 1.
Density of neurons containing c-fos mRNA after D1 or/and D2 agonists and A2Aantagonist, alone or in combination
| Treatment group | n | Caudate putamen | Globus pallidus |
|---|---|---|---|
| Saline (a) | 6 | 35.25 ± 4.34 | 24.20 ± 3.40 |
| Quinelorane (b) | 5 | 8.55 ± 1.70*a | 42.20 ± 4.00*a |
| SKF-82958 (c) | 4 | 132.75 ± 15.30*a | 12.50 ± 1.60 |
| SCH-58261 (d) | 4 | 16.70 ± 1.50*a | 18.25 ± 4.20 |
| SKF-82958 + quinelorane | 5 | 156.20 ± 6.50*b,ns,c | 122.30 ± 17.60*b,c |
| SKF-82958 + SCH-58261 | 4 | 183.90 ± 6.30*c,d | 69.80 ± 13.90*c,d |
| Quinelorane + SCH-58261 | 5 | 17.35 ± 1.39*b,ns,d | 60.30 ± 8.10*d,ns,b |
Rats were treated with saline (NaCl 0.9%), with the D2 agonist quinelorane (2 mg/kg), with the D1agonist SKF-82958 (1 mg/kg), with the A2A antagonist SCH 58261 (5 mg/kg), or various combinations: SKF-82958 (1 mg/kg) + quinelorane (2 mg/kg), SCH 58261 (5 mg/kg) + SKF-82958 (1 mg/kg), and quinelorane (2 mg/kg) + SCH 58261 (5 mg/kg). c-fos mRNA was detected with single in situ hybridization (exposure times, 7 weeks). Values represent the mean ± SEM of the number of c-fos mRNA-containing neurons per mm2. Two-way ANOVA, followed by post hoc t tests corrected for the experiment-wise alpha level (Bonferroni correction). The results of the global ANOVA were for quinelorane/SKF-82958 interaction:F(1,16) = 11.48, p < 0.005 for caudate putamen (CP) and F(1,16) = 23.87,p < 0.001 for globus pallidus (GP); for SKF-82958/SCH-58261 interaction: F(1,14) = 19.02, p < 0.001 for CP andF(1,14) = 19.84, p < 0.001 for GP; for quinelorane/SCH-58261 interaction:F(1,16) = 21.27, p < 0.001 for CP and F(1,16) = 5.163, p < 0.05 for GP. For the multiple post hoc t tests Bonferroni correction, an asterisk indicates relevant significant differences between indicated groups (p < 0.05).
One hour after administration of the D1 agonist SKF-82958 at the dose of 1 mg/kg, the number of c-fos mRNA-containing neurons dramatically increased in the caudate putamen (+277%) and the nucleus accumbens (Figs. 1, 2, Table 1). An increase also was found in the cortex (with a particularly high concentration in layer VIb) and in the septum (Fig. 1). By contrast, the number of c-fos mRNA-containing neurons tended to decrease (by 48%, p = 0.08) in the globus pallidus (Fig. 3, Table 1). In all of the examined areas, the effects of SKF-82958 were similar over the dose range tested (0.5–2 mg/kg; data not shown).
Fig. 2.
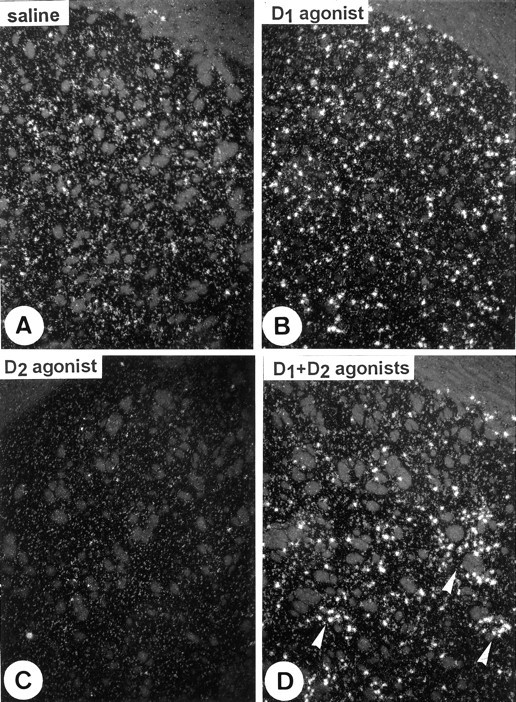
D1- and D2-mediated regulation of c-fos expression in the caudate putamen. Dark-field photomicrographs from single in situhybridization with a 35S-labeled riboprobe showc-fos mRNA after treatments with D1 and D2 agonists alone or in combination. The D1agonist SKF-82958 increases the number of c-fos-positive neurons (B), whereas the D2 agonist quinelorane decreases it (C), as compared with saline-treated rats (A). Association of D1 and D2 agonists changes the D1-induced c-fos expression into a heterogeneous “patchy” pattern (arrowheads inD). Quantitative data are listed in Table 1. Magnification, 40×.
Fig. 3.

D1- and D2-mediated regulation of c-fos expression in the globus pallidus. Dark-field photomicrographs from single in situhybridization with a 35S-labeled riboprobe showc-fos mRNA after treatments with D1 and D2 agonists alone or in combination. The level ofc-fos mRNA observed under basal conditions inA is increased after the D2 agonist (C), whereas it tends to decrease with the D1 agonist (B). Combined treatment with both D1 and D2 agonists potentiated the D2-mediated induction of c-fos in the globus pallidus (D). Stars indicate the internal capsule. Quantitative data are listed in Table 1. Magnification, 40×.
Conversely, the D2 agonist quinelorane, at the dose of 2 mg/kg, caused a decrease in the number of c-fosmRNA-containing neurons in the caudate putamen (−75%, Table 1). Detection of such a decrease is directly related to our ability to consistently detect and quantify c-fos mRNA in basal conditions by using sensitive riboprobes (Fig. 2). In contrast, the density of labeled neurons in the globus pallidus was increased after treatment with the D2 agonist (+74%) (Fig. 3, Table1).
When quinelorane (2 mg/kg) was coadministered with SKF-82958 (1 mg/kg), the density of c-fos-labeled neurons in the caudate putamen and the nucleus accumbens was increased to the same extent as after SKF-82958 alone (Table 1). However, as shown in Figures 1 and4, the homogenous distribution of thec-fos mRNA-containing neurons after SKF-82958 treatment was heterogeneous (“patchy”) after coadministration of the two drugs. Comparison on adjacent sections shows that the distribution ofc-fos mRNA after D1 plus D2 agonists was parallel to the distribution of μ-opioid receptor mRNA (Fig. 4). At the same time, in the globus pallidus, the coadministration of both SKF-82958 (1 mg/kg) and quinelorane (2 mg/kg) increased by 190% the density of c-fos-labeled neurons as compared with quinelorane alone (Fig. 3, Table 1).
Fig. 4.

Striatal c-fos expression in patches after combined treatment with D1 and D2agonists. Dark-field photomicrographs after in situhybridization with 35S-labeled riboprobes on adjacent sections show that the “patches” of c-fosmRNA-containing neurons (arrowheads in A) correspond to patches of μ-opioid receptor mRNA expression in the striatum (arrowheads in B). Also note the concomitant expression of c-fos and μ-mRNA in the subcallosal patch. cc, Corpus callosum. Magnification, 23×.
Effects of an A2A antagonist alone or in combination with a D1 agonist on c-fos expression in the striatum and in the globus pallidus
The adenosine A2A antagonist SCH-58261 had similar effects to the D2 agonist quinelorane in the striatum. Treatment with SCH-58261 at a dose of 5 mg/kg induced adecrease in the density of c-fos-labeled neurons in the caudate putamen (−53%). In contrast to quinelorane, it had no effect on the density of labeled neurons in the globus pallidus (Fig.5, Table 1). The coadministration of SKF-82958 (1 mg/kg) and SCH-58261 (5 mg/kg) induced a further increase in the density of c-fos mRNA-containing neurons in the caudate putamen (+38%) as compared with SKF-82958 alone (Fig. 5, Table1). The distribution pattern of the c-fos-labeled neurons after the coadministration was homogeneous in the striatum and not patchy, as seen after D1 plus D2 agonists (Figs. 1, 2, 4, 5).
Fig. 5.

Effect of the A2A antagonist alone or in combination with the D1 agonist on c-fosexpression in the caudate putamen (A–C) and the globus pallidus (D–F). Dark-field photomicrographs from single in situ hybridization with a 35S-labeled riboprobe show the basal levels ofc-fos mRNA in the caudate putamen (A) and in the globus pallidus (D). The A2A antagonist SCH-58261 alone decreases the number ofc-fos-positive neurons in the caudate putamen (B) but has no effect on the globus pallidus (E). Coadministration of the D1agonist, together with the A2A antagonist, inducesc-fos both in the caudate putamen (C) and in the globus pallidus (F) with a synergistic effect, as compared with the D1 agonist alone (see also Table 1).Stars indicate the internal capsule. Quantitative data are listed in Table 1. Magnification, 40×.
In the globus pallidus the coadministration of SKF-82958 with SCH-58261 induced a dramatic increase in the density of labeled neurons as compared with the saline-treated rats (+188%) but also as compared with SKF-82958 alone (+458%) (Fig. 5, Table 1).
Effects of an A2A antagonist alone or in combination with a D2 agonist on c-fos expression in the striatum and in the globus pallidus
As mentioned above, the D2 receptor agonist quinelorane (2 mg/kg) decreased the density of c-fosmRNA-containing neurons in the caudate putamen and increased it in the globus pallidus, whereas the A2A receptor antagonist SCH 58261 (5 mg/kg) affected c-fos mRNA expression only in the caudate putamen, where it caused a decrease in the density of labeled neurons (Table 1). The coadministration of D2agonist and A2A antagonist significantly counteracted the decrease induced by quinelorane in the caudate putamen (from −75 to −52%). No synergistic effect of the two drugs on c-fosexpression was found in the globus pallidus as compared with quinelorane alone (Table 1).
Phenotypical identification of the c-fosmRNA-containing neurons in the caudate putamen after D1 and D2 agonists, given alone or in combination
To examine in which type of striatal neurons the above-mentioned changes in c-fos expression occurred, we used double-labeling experiments with probes for either D1 or D2 receptor mRNA, together with a probe forc-fos mRNA. Because the results, analyzed in two separate experiments (as illustrated in Fig. 6), were identical, quantitative data were generated only fromc-fos plus D2 mRNAs simultaneous detection (Table 2). Therefore, in the following, D2 mRNA-negative (−) neurons are referred to as D1 mRNA-positive (+) neurons on the basis of both experiments and previously published data (Le Moine and Bloch, 1995,1996).
Fig. 6.

Phenotypical characterization of the striatal neurons expressing c-fos after D1 and D2 agonists, alone or in combination. Double in situ hybridization detects D1 or D2receptor mRNA with digoxigenin-labeled riboprobe (stained cells), together with c-fos mRNA, with a 35S-labeled riboprobe (silver grains). A and D show that c-fos mRNA is present both in D1mRNA-containing (A) and D2mRNA-containing (D) neurons under basal conditions. The D1 agonist SKF-82958 increasesc-fos expression both in D1 mRNA-containing neurons (arrowheads in B) and in D2 mRNA-containing neurons (arrowhead inE). As compared with the D1 agonist alone, coadministration of D1 and D2 agonists potentiates the increase of c-fos expression in D1 mRNA-containing neurons (arrowheads inC) and decreases it in D2 mRNA-containing neurons. Quantitative data are listed in Table 2. Magnification, 640×.
Table 2.
Density of D1− or D2 striatal neurons expressing c-fos mRNA after D1 or/and D2 agonists and A2A antagonist, alone or in combination
| Treatment group | n | Fos+/D2− neurons | Fos+/D2+ neurons |
|---|---|---|---|
| Saline (a) | 5 | 34.2 ± 4.6 | 26.7 ± 3.7 |
| Quinelorane (b) | 4 | 5.3 ± 1.7*a | 0.75 ± 0.4*a |
| SKF-82958 (c) | 5 | 84.2 ± 14.8*a | 73.0 ± 10.4*a |
| SCH-58261 (d) | 4 | 22.5 ± 3.8ns,a | 10.8 ± 3.1*a |
| SKF-82958 + quinelorane | 5 | 233.5 ± 25.3*b,c | 22.4 ± 2.5*b,c |
| SKF-82958 + SCH-58261 | 4 | 166.6 ± 18.0*c,d | 65.6 ± 7.7*d,ns,c |
| Quinelorane + SCH-58261 | 5 | 10.8 ± 2.3*b,ns,d | 1.5 ± 0.5*ns,b,d |
Rats were treated with saline (NaCl 0.9%), with the D2 agonist quinelorane (2 mg/kg), with the D1agonist SKF-82958 (1 mg/kg), with the A2A antagonist SCH 58261 (5 mg/kg), or various combinations: SKF-82958 (1 mg/kg) + quinelorane (2 mg/kg), SCH 58261 (5 mg/kg) + SKF-82958 (1 mg/kg), and quinelorane (2 mg/kg) + SCH 58261 (5 mg/kg). c-fos mRNA was detected with double in situ hybridization (exposure times, 10 weeks). Values represent the mean ± SEM of the number ofc-fos mRNA-containing neurons per mm2. Two-way ANOVA, followed by post hoc t tests corrected for the experiment-wise alpha level (Bonferroni correction). The results of the global ANOVA were for quinelorane/SKF-82958 interaction:F(1,15) = 31.55, p < 0.001 for D2-negative neurons and F(1,15) = 4.155 for D2-positive neurons; for SKF-82958/SCH-58261 interaction: F(1,14) = 15.45, p< 0.001 for D2-negative neurons andF(1,14) = 0.35 for D2-positive neurons. For the multiple post hoc t tests Bonferroni correction, an asterisk indicates relevant significant differences between indicated groups (p < 0.05).
Figure 6 shows that administration of the D1 agonist SKF-82958 (1 mg/kg) increased the number of both D1 and D2 mRNA-containing neurons that express c-fosmRNA (Table 2). Conversely, the D2 agonist quinelorane (2 mg/kg) decreased the density of c-fos-labeled neurons both for D1 and D2 mRNA-containing neurons (Table2). The coadministration of D1 and D2 agonists had opposite effects on c-fos expression in these two populations because it induced an increase in the density ofc-fos-labeled neurons containing D1 mRNA and a decrease in the density of c-fos-labeled neurons containing D2 mRNA, as compared with the D1 agonist alone (Fig. 6, Table 2). Indeed, in SKF-82958 treated rats 53% of thec-fos expressing neurons were D1 mRNA-positive, whereas in rats treated by SKF-82958 plus quinelorane, the proportion of these neurons reached 91% (Table 2). Note here and below that the relative changes observed in the density of c-fos-labeled neurons in the caudate putamen are comparable to what was observed in the single-labeling experiments and summarized in Table 1.
Phenotypical identification of the c-fosmRNA-containing neurons in the caudate putamen after A2A antagonist and D1 agonist, given alone or in combination
Similar experiments, performed with the A2A antagonist SCH 58261 (5 mg/kg), showed a decrease in the density ofc-fos-labeled neurons and in D2 mRNA-containing neurons, but not in D1 mRNA-containing neurons (Table 2). As mentioned above, the D1 agonist SKF-82958 increased the density of c-fos-labeled neurons both in D1 and D2 mRNA-positive neurons (Table 2). The coadministration of the D1 agonist and the A2A antagonist potentiated the increase in the density of c-fos-labeled neurons that were positive for D1 mRNA but had no effect on the density of c-fos labeled in D2mRNA-containing neurons, as compared with the D1 agonist alone (Table 2).
Phenotypical identification of the c-fosmRNA-containing neurons in the caudate putamen after A2A antagonist and D2 agonist, given alone or in combination
The density of D2 mRNA-positive neurons that expressc-fos mRNA was lower in SCH-58261 (−60%) and quinelorane-treated animals (−97%) as compared with saline (Table 2). At the same time, quinelorane—and not SCH-58261—induced a reduction of c-fos in neurons positive for D1 mRNA (−84.5%). When SCH-58261 and quinelorane were coadministered, there was no synergistic effect on c-fos expression in the D1-containing nor in the D2-containing neurons (Table 2).
DISCUSSION
Individual and synergistic effects of dopamine D1 and D2 receptor agonists and of an adenosine A2Areceptor antagonist on c-fos expression were analyzed in the striatum and globus pallidus. Our data, summarized in Figure7, show that (1) c-fosexpression can be either activated through D1 andinhibited through A2A or D2receptors in the two striatal output pathways in normal rats, and (2) D2 receptor stimulation as well as A2A receptor blockade can interact with D1, but not D2, receptor activation to potentiatec-fos expression in both the striatum and the globus pallidus.
Fig. 7.

Schematic representation of the interactions in the basal ganglia after treatments with D1 and D2 agonists or combined treatment with D1 + D2 agonist or D1 agonist + A2Aantagonist. The variations of expression of c-fos mRNA as compared with basal conditions are indicated inside the structure or the neuronal populations that we have studied. Dark arrows represent excitatory pathways, and white arrows represent inhibitory pathways. Thethickness of the arrows changes according to the supposed neuronal activity in the different pathways.ST, Striatum; GP, globus pallidus;SNc, substantia nigra pars compacta;SNr/EPN, substantia nigra pars reticulata/entopeduncular nucleus; STN, subthalamic nucleus; fos,c-fos mRNA.
Effect of D2 and D1 agonists given alone onc-fos expression in the striatum
Selective activation of D2 receptors by the D2 agonist produced a significant decrease in the number of striatal neurons expressing c-fos in the caudate putamen. The decrease was found in both D1- and D2-positive neurons. In D2-containing neurons this decrease may be explained by the fact that dopamine is likely to have an inhibitory action on striatopallidal neurons via postsynaptic D2 receptors (Gerfen et al., 1990). Conversely, the D2 agonist effect on c-fos in D1-containing neurons might be related to activation of presynaptic D2 autoreceptors located on dopaminergic terminals, because this strongly decreases striatal dopamine release (Imperato et al., 1988; Suaud-Chagny et al., 1991) and thereby the D1-mediated activity in striatonigral neurons. Decreases of mRNA coding for the immediate early gene NGFI-A (zif 268) have been described after treatment with drugs acting on D2or A2A receptors (Gerfen et al., 1995; Svenningsson et al., 1995), but we describe here for the first time the D2-mediated inhibition of c-fos expression in the two striatal output neurons.
The full D1 agonist SKF-82958 increased c-fosexpression in the striatum in normal rats, as previously reported byWang and McGinty (1996). A strong induction of c-fosexpression in the D1 rich cortical layer VIb (Gaspar et al., 1995) also was found. Interestingly, c-fos mRNA increased to a similar extent in D1- and D2-containing neurons in the striatum. The stimulation ofc-fos expression in D1-positive neurons was expected, because many studies have demonstrated that the dopamine-mediated induction of striatal Fos is dependent on D1 activation [see Hughes and Dragunow (1995) and references therein]. The increased number of D2-positive neurons expressing c-fos after SKF-82958 was unexpected. In previous studies, using the partial D1 agonist SKF-38393, researchers observed c-fos induction only in the D1 receptor-containing striatonigral neurons (Robertson et al., 1990; Gerfen et al., 1995). However, these studies were performed in animals with nigrostriatal lesions, and we therefore suggest thatc-fos induction by the D1 agonist in striatopallidal neurons requires intact nigrostriatal neurons. We hypothesize that the D1 agonist, when injected systemically, acts on D1 receptors located on striatonigral terminals (Caillé et al., 1996) and stimulates GABA release (Cameron and Williams, 1993), which in turn inhibits nigrostriatal neurons and decreases the extracellular striatal dopamine level (Suaud-Chagny et al., 1992). This effect would be indirectly responsible for an increase of c-fos in striatopallidal neurons. Nevertheless, cholinergic interneurons expressing D5 (C. Le Moine, unpublished results) in addition to D2 receptors (Le Moine et al., 1990b) and corticostriatal glutamatergic neurons (Gaspar et al., 1995) also may be involved in this D1-dependent c-fos activation in the D2-containing neurons (Berretta et al., 1992).
Effect of combined D1 and D2 agonists onc-fos expression in the striatum
Thus, the effects of D1 or D2 agonists probably can be attributed to both direct postsynaptic effects and indirect effects mediated by the mesencephalic dopamine neurons. However, when these drugs are combined, the effects of endogenous dopamine are likely to be masked. Indeed, in the striatum, combined treatment with D1 and D2 agonists potentiatedc-fos expression in D1-containing neurons but inhibited it in D2-containing neurons. The fact that the combined treatment induces c-fos at 92% in D1-containing neurons is consistent with data obtained in conditions that enhance extracellular dopamine concentration (Graybiel et al., 1990; Young et al., 1991; Moratalla et al., 1993; Jaber et al., 1995; Wang et al., 1995; Chergui et al., 1996).
Effect of an A2A antagonist alone or in combination with D1 or D2 agonists in the striatum
A2A and D2 receptors regulate pallidal GABA release in an opposite manner (Ferré et al., 1993; Mayfield et al., 1993, 1996) and are colocalized in striatopallidal neurons, but not in interneurons nor on nigrostriatal terminals (Schiffmann et al., 1991; Fink et al., 1992; Augood and Emson, 1994; Svenningsson et al., 1997). Therefore, studying the effects of A2A receptors on striatal neurotransmission may be of interest not only to better understand adenosinergic modulation but also to delineate effects specifically related to an altered activity of striatopallidal neurons. We show here that the A2A antagonist SCH-58261 shared with the D2 agonist the ability to decrease c-fosexpression in the striatum. This decrease occurred only in D2-containing neurons, suggesting that this effect is mainly postsynaptic. Indeed, unlike the D2 agonist, the A2A antagonist does not affect dopamine release (Ferré et al., 1993). This supports the idea that endogenous adenosine acting at A2A receptors regulates the constitutive expression of immediate early genes in the striatum (Svenningsson et al., 1995).
Coadministration of the A2A antagonist with the D1 agonist potentiated the D1-induced increase in c-fos expression in D1-containing neurons, like treatment with D1 and D2 agonists. However, this combination, unlike the D1 plus D2 combination, caused no inhibition of D1-mediated c-fos induction in D2-containing neurons. This suggests that regulation ofc-fos by dopamine is more potent than A2A-mediated effects on these neurons in our conditions. Whereas the D1/D2 combined treatment produced a change of the initial homogeneous striatal expression ofc-fos into a “patchy” pattern, as previously described (Paul et al., 1992; Wang and McGinty, 1996), the pattern ofc-fos expression after the D1/A2A combination was homogeneous in the striatum. These results suggest that D2 receptors located postsynaptically on striatopallidal neurons, like the A2A receptors, are involved in the quantitative enhancement of c-fos mRNA in striatal neurons, whereas D2receptors located presynaptically or on interneurons might be involved more specifically in differential dopaminergic regulations between the patch/matrix compartments.
D1/D2 and D1/A2A interactions in the globus pallidus
In accordance with previous immunohistochemical studies (Robertson et al., 1992; Marshall et al., 1993), we show here an increase ofc-fos expression in the globus pallidus after administration of the D2 agonist. A strong tendency for a decrease ofc-fos expression was found after D1 agonist treatment, although not significant in our statistical conditions. This tendency might be attributable to the D1-mediatedc-fos expression in striatopallidal neurons. Taken together, these data suggest that stimulation of D1 and D2 receptors has opposite effects on pallidal neurons also.
The combined treatment with D1 and D2 agonists potentiated the increase in c-fos expression induced by the D2 agonist alone, as previously shown (Paul et al., 1992,1995; Marshall et al., 1993). This agrees with electrophysiological data showing that the D1 plus D2 coactivation is required for the maximal excitatory effect, demonstrating a potentiated effect mediated by D1 receptors on D2 receptor-activated responses (Walters et al., 1987). There was also a strong induction of c-fos expression after combined treatment, using the A2A antagonist together with the D1 agonist. Interestingly, coadministration of A2A antagonist together with the D2 agonist had no synergistic effects on c-fos expression in the globus pallidus. This implies that, despite their coexpression and their well established interactions (Ferré et al., 1992, 1993), the D2 and A2A receptors are not solely the key for adenosine/dopamine interactions in the basal ganglia. Instead, our findings suggest that the most important functional interactions may be between drugs that affect A2A and dopamine receptors in distinct neuronal populations. This conclusion also has implications for our understanding of the D1/D2interactions.
Disinhibition of striatopallidal neurons is one of the mechanisms whereby c-fos is induced in globus pallidus. However, ifc-fos expression can correlate with the activity of striatopallidal neurons, these neurons are likely to be stimulated rather than inhibited by combined treatments with D1 plus D2 agonists or D1 agonist plus A2Aantagonist. Thus, the increase in pallidal c-fos expression may be attributable to the involvement of additional inputs to the globus pallidus. This may be attributable to an increased activity in the excitatory input from subthalamic nucleus. It has been found that NMDA receptor antagonists inhibit the induction of pallidal Fos immunoreactivity after combined administration of D1 and D2 agonists (Paul et al., 1992, 1995). Thus, it might turn out that concomitant stimulation of an excitatory input and inhibition of striatopallidal neurons act in synergy to increase c-fosin globus pallidus.
Conclusion
Although c-fos generally is used as a neuronal activation marker, we demonstrate here that basal c-fosexpression is upregulated by a D1 agonist butdownregulated by a D2 agonist or an A2A antagonist. This suggests that c-fos mRNA levels may be used as an indicator of inhibition as well as activation of a neuronal pathway. Synergistic effects have been observed in the striatal output pathways after coadministration of D1 plus D2 agonists or D1 agonist plus A2A antagonist, providing evidence for important interactions between these parallel pathways. This work gives a basis for further investigations to elucidate the mechanisms whereby these synergistic effects occur, especially in the globus pallidus.
Footnotes
P.S. was the recipient of a travel grant from the Swedish Medical Research Council. This study was supported in part by the Swedish Medical Research Council and the Institute for Scientific Information on Coffee (to B.B.F). We thank Dr. Ongini for providing us with the A2A antagonist, Drs. M. Jaber and F. Gonon for helpful discussions, and C. Vidauporte for expert photographic artwork.
P.S. and C.L.M. contributed equally to this work.
Correspondence should be addressed to Dr. C. Le Moine, Laboratoire d’Histologie-Embryologie, Centre National de la Recherche Scientifique Unité Mixte de Recherche 5541, Université Victor Segalen Bordeaux II, Bat. 3B, zone Nord, 146 Rue Léo Saignat, 33076 Bordeaux Cedex, France.
REFERENCES
- 1.Alexander GE, Crutcher MD. Functional architecture of basal ganglia circuits: neural substrates of parallel processing. Trends Neurosci. 1990;13:266–271. doi: 10.1016/0166-2236(90)90107-l. [DOI] [PubMed] [Google Scholar]
- 2.Andersen PH, Jansen JA. Dopamine receptor agonists: selectivity and dopamine D1 receptor efficacy. Eur J Pharmacol. 1990;188:335–347. doi: 10.1016/0922-4106(90)90194-3. [DOI] [PubMed] [Google Scholar]
- 3.Augood SJ, Emson PC. Adenosine A2A receptor mRNA is expressed by enkephalin cells but not by somatostatin cells in the rat striatum: a co-expression study. Mol Brain Res. 1994;22:204–210. doi: 10.1016/0169-328x(94)90048-5. [DOI] [PubMed] [Google Scholar]
- 4.Berretta S, Robertson HA, Graybiel AM. Dopamine and glutamate agonists stimulate neuron-specific expression of Fos-like protein in the striatum. J Neurophysiol. 1992;68:767–777. doi: 10.1152/jn.1992.68.3.767. [DOI] [PubMed] [Google Scholar]
- 5.Bymaster FP, Reid LR, Nichols CL, Kornfeld EC, Wong DT. Elevation of acetylcholine levels in striatum of the rat brain by LY163502, trans-(−)5,5a,6,7,8,9a,10-octahydro-6-propylpyrimido<4,5-g>quinolin-2-amine dihydrochloride, a potent and stereospecific dopamine (D2) agonist. Life Sci. 1986;38:317–322. doi: 10.1016/0024-3205(86)90078-0. [DOI] [PubMed] [Google Scholar]
- 6.Caillé I, Dumartin B, Bloch B. Ultrastructural localization of D1 dopamine receptor immunoreactivity in the rat striatonigral neurons and its relation with dopaminergic innervation. Brain Res. 1996;730:17–31. doi: 10.1016/0006-8993(96)00424-6. [DOI] [PubMed] [Google Scholar]
- 7.Cameron DL, Williams JT. Dopamine D1 receptors facilitate transmitter release. Nature. 1993;366:344–347. doi: 10.1038/366344a0. [DOI] [PubMed] [Google Scholar]
- 8.Chergui K, Nomikos GG, Mathé JM, Gonon F, Svensson TH. Burst stimulation of the medial forebrain bundle selectively increases Fos-like immunoreactivity in the limbic forebrain of the rat. Neuroscience. 1996;72:141–156. doi: 10.1016/0306-4522(95)00513-7. [DOI] [PubMed] [Google Scholar]
- 9.Creese I, Sibley DR, Hamblin MW, Leff SE. The classification of dopamine receptors: relationship to radioligand binding. Annu Rev Neurosci. 1983;6:43–71. doi: 10.1146/annurev.ne.06.030183.000355. [DOI] [PubMed] [Google Scholar]
- 10.Curran T, Gordon MB, Rubino KL, Sambucetti LC. Isolation and characterization of the c-fos (rat) cDNA and analysis of post-translational modification in vitro. Oncogene. 1987;2:79–84. [PubMed] [Google Scholar]
- 11.Ferré S, Fuxe K, von Euler G, Johansson B, Fredholm BB. Adenosine–dopamine interactions in the brain. Neuroscience. 1992;51:501–512. doi: 10.1016/0306-4522(92)90291-9. [DOI] [PubMed] [Google Scholar]
- 12.Ferré S, O’Connor WT, Fuxe K, Ungerstedt U. The striatopallidal neuron: a main locus for adenosine–dopamine interactions in the brain. J Neurosci. 1993;13:5402–5406. doi: 10.1523/JNEUROSCI.13-12-05402.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fink JS, Weaver DR, Rivkees SA, Peterfreund RA, Pollack AE, Adler EM, Reppert SM. Molecular cloning of the rat A2 adenosine receptor: selective co-expression with D2 dopamine receptors in rat striatum. Mol Brain Res. 1992;14:186–195. doi: 10.1016/0169-328x(92)90173-9. [DOI] [PubMed] [Google Scholar]
- 14.Gaspar P, Bloch B, Le Moine C. D1 and D2 receptor gene expression in the rat frontal cortex: cellular localization in different classes of efferent neurons. Eur J Neurosci. 1995;7:1050–1063. doi: 10.1111/j.1460-9568.1995.tb01092.x. [DOI] [PubMed] [Google Scholar]
- 15.Gerfen CR. The neostriatal mosaic: multiple levels of compartmental organization in the basal ganglia. Annu Rev Neurosci. 1992;15:285–320. doi: 10.1146/annurev.ne.15.030192.001441. [DOI] [PubMed] [Google Scholar]
- 16.Gerfen CR, Wilson CJ. The basal ganglia. In: Swanson LW, Björklund A, Hökfelt T, editors. Handbook of chemical neuroanatomy, Vol 12, Integrated systems of the CNS, Pt III. Elsevier; Amsterdam: 1996. pp. 371–468. [Google Scholar]
- 17.Gerfen CR, Engber TM, Maham LC, Susel Z, Chase TN, Monsma FJ, Sibley DR. D1 and D2 dopamine receptor-regulated gene expression of striatonigral and striatopallidal neurons. Science. 1990;250:1429–1432. doi: 10.1126/science.2147780. [DOI] [PubMed] [Google Scholar]
- 18.Gerfen CR, Keefe KA, Gauda EB. D1 and D2 dopamine receptor function in the striatum: coactivation of D1 and D2 dopamine receptors on separate populations of neurons results in potentiated immediate early gene response in D1-containing neurons. J Neurosci. 1995;15:8167–8176. doi: 10.1523/JNEUROSCI.15-12-08167.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Graybiel AM. Neurotransmitters and neuromodulators in the basal ganglia. Trends Neurosci. 1990;13:244–253. doi: 10.1016/0166-2236(90)90104-i. [DOI] [PubMed] [Google Scholar]
- 20.Graybiel AM, Moratalla R, Robertson HA. Amphetamine and cocaine induce drug-specific activation of the c-fos gene in striosome-matrix compartments and limbic subdivisions of the striatum. Proc Natl Acad Sci USA. 1990;87:6912–6916. doi: 10.1073/pnas.87.17.6912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hersch SM, Ciliax BJ, Gutekunst CA, Rees HD, Heilman CJ, Yung KKL, Bolam JP, Ince E, Yi H, Levey AI. Electron microscopic analysis of D1 and D2 dopamine receptor proteins in the dorsal striatum and their synaptic relationships with motor corticostriatal afferents. J Neurosci. 1995;15:5222–5237. doi: 10.1523/JNEUROSCI.15-07-05222.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Hughes P, Dragunow M. Induction of immediate early genes and the control of neurotransmitters-regulated gene expression within the nervous system. Pharmacol Rev. 1995;47:133–178. [PubMed] [Google Scholar]
- 23.Imperato A, Tanda G, Frau R, Di Chiara G. Pharmacological profile of dopamine receptor agonists as studied by brain dialysis in behaving rats. J Pharmacol Exp Ther. 1988;245:257–264. [PubMed] [Google Scholar]
- 24.Jaber M, Cador M, Dumartin B, Normand E, Stinus L, Bloch B. Acute and chronic amphetamine treatments differently regulate messenger RNA levels and Fos immunoreactivity in rat striatal neurons. Neuroscience. 1995;65:1041–1050. doi: 10.1016/0306-4522(94)00537-f. [DOI] [PubMed] [Google Scholar]
- 25.Jaber M, Robinson SW, Missale C, Caron MG. Dopamine receptors and brain function. Neuropharmacology. 1996;35:1503–1519. doi: 10.1016/s0028-3908(96)00100-1. [DOI] [PubMed] [Google Scholar]
- 26.Jiang H, Jackson-Lewis V, Muthane U, Dollison A, Ferreira M, Espinosa A, Parsons B, Przedborski S. Adenosine receptor antagonists potentiate dopamine receptor agonist-induced rotational behaviour in 6-hydroxydopamine-lesioned rats. Brain Res. 1993;613:347–351. doi: 10.1016/0006-8993(93)90925-d. [DOI] [PubMed] [Google Scholar]
- 27.Le Moine C, Bloch B. D1 and D2 dopamine receptor gene expression in the rat striatum: sensitive cRNA probes demonstrate prominent segregation of D1 and D2 mRNAs in distinct neuronal populations of the dorsal and ventral striatum. J Comp Neurol. 1995;355:418–426. doi: 10.1002/cne.903550308. [DOI] [PubMed] [Google Scholar]
- 28.Le Moine C, Bloch B. Expression of the D3 dopamine receptor in peptidergic neurons of the nucleus accumbens: comparison with D1 and D2 dopamine receptors. Neuroscience. 1996;73:131–143. doi: 10.1016/0306-4522(96)00029-2. [DOI] [PubMed] [Google Scholar]
- 29.Le Moine C, Normand E, Guitteny AF, Fouque B, Teoule R, Bloch B. Dopamine receptor gene expression by enkephalin neurons in rat forebrain. Proc Natl Acad Sci USA. 1990a;87:230–234. doi: 10.1073/pnas.87.1.230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Le Moine C, Tison F, Bloch B. D2 dopamine receptor gene expression by cholinergic neurons in the rat striatum. Neurosci Lett. 1990b;117:248–252. doi: 10.1016/0304-3940(90)90671-u. [DOI] [PubMed] [Google Scholar]
- 31.Le Moine C, Normand E, Bloch B. Phenotypical characterization of the rat striatal neurons expressing the D1 dopamine receptor gene. Proc Natl Acad Sci USA. 1991;88:4205–4209. doi: 10.1073/pnas.88.10.4205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Marshall JF, Cole BN, LaHoste GJ. Dopamine D2 receptor control of pallidal fos expression: comparisons between intact and 6-hydroxydopamine-treated hemispheres. Brain Res. 1993;632:308–313. doi: 10.1016/0006-8993(93)91166-p. [DOI] [PubMed] [Google Scholar]
- 33.Mayfield RD, Suzuki F, Zahniser NR. Adenosine A2A receptor modulation of electrically evoked endogenous GABA release from slices of rat globus pallidus. J Neurochem. 1993;60:2334–2337. doi: 10.1111/j.1471-4159.1993.tb03526.x. [DOI] [PubMed] [Google Scholar]
- 34.Mayfield RD, Larson G, Orona RA, Zahniser NR. Opposing actions of adenosine A2A and dopamine D2 receptor activation on GABA release in the basal ganglia: evidence for an A2A/D2 receptor interaction in globus pallidus. Synapse. 1996;22:132–138. doi: 10.1002/(SICI)1098-2396(199602)22:2<132::AID-SYN6>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 35.Monsma FJ, McVittie LD, Gerfen CR, Maham LC, Sibley DR. Multiple D2 dopamine receptors produced by alternative RNA splicing. Nature. 1989;342:926–929. doi: 10.1038/342926a0. [DOI] [PubMed] [Google Scholar]
- 36.Monsma FJ, Maham LC, McVittie LD, Gerfen CR, Sibley DR. Molecular cloning and expression of a D1 dopamine receptor linked to adenylate cyclase activation. Proc Natl Acad Sci USA. 1990;87:6723–6727. doi: 10.1073/pnas.87.17.6723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Moratalla R, Vickers EA, Robertson HA, Cochran BH, Graybiel AM. Coordinate expression of c-fos and jun B is induced in the rat striatum by cocaine. J Neurosci. 1993;13:423–433. doi: 10.1523/JNEUROSCI.13-02-00423.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ongini E, Fredholm BB. Pharmacology of adenosine A2A receptors. Trends Pharmacol Sci. 1996;17:364–372. [PubMed] [Google Scholar]
- 39.Paul ML, Graybiel AM, David JC, Robertson HA. D1-like and D2-like dopamine receptors synergistically activate rotation and c-fos expression in the dopamine-depleted striatum in a rat model of Parkinson’s disease. J Neurosci. 1992;12:3729–3742. doi: 10.1523/JNEUROSCI.12-10-03729.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Paul ML, Currie RW, Robertson HA. Priming of a D1 dopamine receptor behavioural response is dissociated from striatal immediate-early gene activity. Neuroscience. 1995;66:347–359. doi: 10.1016/0306-4522(94)00582-p. [DOI] [PubMed] [Google Scholar]
- 41.Pinna A, Di Chiara G, Wardas J, Morelli M. Blockade of A2A adenosine receptors positively modulates turning behaviour and c-fos expression induced by D1 agonists in dopamine denervated rats. Eur J Neurosci. 1996;8:1176–1181. doi: 10.1111/j.1460-9568.1996.tb01285.x. [DOI] [PubMed] [Google Scholar]
- 42.Pollack AE, Fink JS. Synergistic interaction between an adenosine antagonist and a dopamine D1 agonist on rotational behaviour and striatal c-fos induction in 6-hydroxydopamine-lesioned rats. Brain Res. 1996;743:124–130. doi: 10.1016/s0006-8993(96)01036-0. [DOI] [PubMed] [Google Scholar]
- 43.Robertson GS, Vincent SR, Fibiger HC. Striatonigral projections neurons contain D1 dopamine receptor-activated c-fos. Brain Res. 1990;523:288–290. doi: 10.1016/0006-8993(90)91498-6. [DOI] [PubMed] [Google Scholar]
- 44.Robertson GS, Vincent SR, Fibiger HC. D1 and D2 dopamine receptors differentially regulate c-fos expression in striatonigral and striatopallidal neurons. Neuroscience. 1992;49:285–296. doi: 10.1016/0306-4522(92)90096-k. [DOI] [PubMed] [Google Scholar]
- 45.Schiffmann SN, Jacobs OP, Vanderhaegen JJ. Striatal restricted adenosine A2 receptor (RDC8) is expressed by enkephalin but not substance P neurons: an in situ hybridization study. J Neurochem. 1991;57:1062–1067. doi: 10.1111/j.1471-4159.1991.tb08257.x. [DOI] [PubMed] [Google Scholar]
- 46.Suaud-Chagny MF, Ponec J, Gonon F. Presynaptic autoinhibition of the electrically evoked dopamine release studied in the rat olfactory tubercle by in vivo electrochemistry. Neuroscience. 1991;45:641–652. doi: 10.1016/0306-4522(91)90277-u. [DOI] [PubMed] [Google Scholar]
- 47.Suaud-Chagny MF, Chergui K, Chouvet G, Gonon F. Relationship between dopamine release in the rat nucleus accumbens and the discharge activity of dopaminergic neurons during local in vivo application of amino acids in the ventral tegmental area. Neuroscience. 1992;49:63–72. doi: 10.1016/0306-4522(92)90076-e. [DOI] [PubMed] [Google Scholar]
- 48.Surmeier DJ, Reiner A, Levine MS, Ariano MA. Are neostriatal dopamine receptors co-localized? Trends Neurosci. 1993;16:299–305. doi: 10.1016/0166-2236(93)90103-s. [DOI] [PubMed] [Google Scholar]
- 49.Svenningsson P, Nomikos G, Fredholm BB. Biphasic changes in locomotor behavior and in expression of mRNA for NGFI-A and NGFI-B in rat striatum following acute caffeine administration. J Neurosci. 1995;15:7612–7624. doi: 10.1523/JNEUROSCI.15-11-07612.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Svenningsson P, Le Moine C, Kull B, Sunahara R, Bloch B, Fredholm BB. Cellular expression of adenosine A2A receptor mRNA in the rat central nervous system with special reference to dopamine innervated areas. Neuroscience. 1997;80:1171–1185. doi: 10.1016/s0306-4522(97)00180-2. [DOI] [PubMed] [Google Scholar]
- 51.Swanson LW. Brain maps: structure of the rat brain. Elsevier; Amsterdam: 1992. [Google Scholar]
- 52.Thompson RC, Mansour A, Akil H, Watson SJ. Cloning and pharmacological characterization of a rat μ opioid receptor. Neuron. 1993;11:903–913. doi: 10.1016/0896-6273(93)90120-g. [DOI] [PubMed] [Google Scholar]
- 53.Waddington JL, Daly SA. Regulation of unconditioned motor behaviour by D1:D2 interactions. In: Waddington JL, editor. Neuroscience and psychopharmacology: D1:D2 dopamine receptor interactions. Academic; San Diego: 1993. pp. 51–78. [Google Scholar]
- 54.Walters JR, Bergstrom DA, Carlson JH, Chase TN, Braun AR. D1 dopamine receptor activation required for postsynaptic expression of D2 agonist effects. Science. 1987;236:719–722. doi: 10.1126/science.2953072. [DOI] [PubMed] [Google Scholar]
- 55.Wang JQ, McGinty JF. Scopolamine augments c-fos and zif/268 messenger RNA expression induced by the full D1 dopamine receptor agonist SKF-82958 in the intact rat striatum. Neuroscience. 1996;72:601–616. doi: 10.1016/0306-4522(95)00597-8. [DOI] [PubMed] [Google Scholar]
- 56.Wang JQ, Smith AJW, McGinty JF. A single injection of amphetamine or methamphetamine induces dynamic alterations in c-fos, zif 268, and preprodynorphin messenger RNA expression in rat forebrain. Neuroscience. 1995;68:83–95. doi: 10.1016/0306-4522(95)00100-w. [DOI] [PubMed] [Google Scholar]
- 57.White FJ, Hu XT. Electrophysiological correlates of D1:D2 interactions. In: Waddington JL, editor. Neuroscience and psychopharmacology: D1:D2 dopamine receptor interactions. Academic; San Diego: 1993. pp. 79–114. [Google Scholar]
- 58.Young ST, Porrino LJ, Iadarola MJ. Cocaine induces striatal c-Fos immunoreactive proteins via dopaminergic D1 receptors. Proc Natl Acad Sci USA. 1991;88:1291–1295. doi: 10.1073/pnas.88.4.1291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Yung KKL, Smith AD, Levey AI, Bolam JP. Synaptic connections between spiny neurons of the direct and indirect pathways in the neostriatum of the rat: evidence from dopamine receptor and neuropeptide immunostaining. Eur J Neurosci. 1996;8:861–869. doi: 10.1111/j.1460-9568.1996.tb01573.x. [DOI] [PubMed] [Google Scholar]
- 60.Zocchi C, Ongini E, Conti A, Monopoli A, Negretti A, Baraldi PG, Dionisotti S. The non-xanthine heterocyclic compound SCH 58261 is a new potent and selective A2A adenosine receptor antagonist. J Pharmacol Exp Ther. 1996;276:398–404. [PubMed] [Google Scholar]


