Abstract
Cancer stem cells (CSCs) are involved in metastatic colorectal cancer recurrence, but no effective therapy targeting these cells is currently available. Because trifluridine (FTD)/tipiracil therapy is used for refractory colorectal cancer, we sought to determine whether FTD is effective against CSC-like cells. CD44+CD133+ high-expressing and other populations of human DLD-1 colon cancer cells were separately isolated through fluorescence-activated cell sorting. The sphere-forming activity of each population and the anti-sphere-forming effects of FTD and fluorouracil (5-FU) on CD44+CD133+ cells were then measured. CD44+CD133+ DLD-1 cells formed substantially more spheres than other cells. Moreover, treating CD44+CD133+ DLD-1 cells with subtoxic concentrations of FTD (1 µM) inhibited sphere formation, and this was superior to the effect of subtoxic concentrations (1 µM) of 5-FU. The associated inhibition rates for FTD and 5-FU were 58.2% and 26.1%, respectively. Further, CD44+CD133+ DLD-1 cells expressed higher levels of thymidine kinase 1, which is responsible for FTD phosphorylation, than DLD-1 cells, and FTD was incorporated into the DNA of CD44+CD133+ DLD-1 cells. Thus, our data show that FTD treatment is effective against CSC-like cells and might be applied as CSC-targeting chemotherapy for tumor subtypes with high CD44 and CD133 expression.
Subject terms: Chemotherapy, Cancer stem cells
Introduction
Cancer stem cells (CSCs) represent a subpopulation of cells that displays stem cell characteristics and this subset influences tumorigenesis1; further, CSCs exhibit diverse cancer-promoting properties such as self-renewal2, chemoresistance3, and metastatic potential4,5. Accordingly, it is considered that CSCs adversely affect patient outcome, but active anti-cancer agents targeting these cells are not currently available in clinical settings.
For the identification of CSCs, one of the first markers of stemness used was the transmembrane glycoprotein CD133, also known as prominin-11,2, for which expression strongly predicts poorer prognosis; specifically high CD133 levels are inversely correlated with the 5-year overall survival and disease-free survival rates in patients with cancers6 including colorectal cancer (CRC). Another putative CSC marker is the cell-surface glycoprotein CD447, which was reported to be an adhesion molecule expressed in cancer stem-like cells8. The combined analysis of these putative co-CSC markers, namely CD133 and CD44, was found to improve the discrimination of low- and high-risk cases of CRC, as compared to that with single-marker analyses9. Moreover, patients with CD44- and CD133-positive gastric cancer were found to have a poorer survival rate than patients with CD44- and CD133-negative disease10.
For CRC, the standard chemotherapy used globally is fluoropyrimidine-, oxaliplatin (L-OHP)-, and irinotecan-based combination regimens with anti-EGFR and anti-VEGF treatment for first-line and second-line settings11. In the third- or later-line settings, regorafenib was approved and has been used12 in the United States. In addition, The Food and Drug Administration (FDA) has approved pembrolizumab or nivolumab for CRC patients with microsatellite instability-high (MSI-H) tumours and larotrectinib for CRC patients with tumours with neurotrophic tropomyosin receptor kinase (NTRK) fusions in the US. Trifluridine (FTD)/tipiracil (TPI), also known as TAS-102, was also tested for its ability to treat patients with metastatic CRC who were previously treated with chemotherapy based on drugs such as fluoropyrimidine, L-OHP, and irinotecan, among others, and this therapy was found to improve overall survival13. After receiving the results of this clinical trial, FTD/TPI was approved and has been used globally in regions such as Japan, the EU, and the US in third- or later-line settings. FTD/TPI is an oral combination drug consisting of FTD, a thymidine analogue, and TPI, a thymidine phosphorylase inhibitor that improves the bioavailability of FTD14. Despite the initial success of this agent, its effect on CSC populations was not previously known. Because FTD/TPI is used for refractory CRC, which is considered a CSC-enriched phenotype15, we hypothesised that FTD might effectively target CSC-like cells. Here, we investigated whether this drug is effective against CD44- and CD133-highly-expressing (CD44+CD133+) CRC cells that possess CSC-like properties.
Results
Isolation of CD44+ CD133+, CD44+ CD133−, CD44− CD133+, and CD44− CD133− cells and examination of their sphere-formation ability
To delineate CD44 and CD133 expression in the DLD-1 CRC cell line, we used flow cytometry to measure the expression of these surface molecules after double-staining with anti-CD44-FITC and anti-CD133-PE antibodies. Figure 1a shows DLD-1 flow cytometry data collected after staining and sorting. These cells showed a distribution of CD44 and CD133 expression, with a CD44+ CD133+ population of 11.3%, a CD44+ CD133− population of 65.3%, a CD44− CD133+ population of 2.1%, and a CD44− CD133− population of 21.3%. Cells were isolated using FACS and then transferred into serum-free DMEM/F-12; the isolated CD44+ CD133+, CD44+ CD133−, CD44− CD133+, and CD44− CD133− cells were then used for sphere-formation assays to evaluate their stem cell-like properties. Sphere formation in suspension culture is recognised as the prominent characteristic of CSCs and can be used for in vitro measurements of CSC function. Here, culturing these cells in serum-free medium in low-adhesion 96-well plates revealed that sphere formation ability was considerably higher in the CD44+ CD133+ population compared to that in the CD44− CD133−, CD44+ CD133−, and CD44− CD133+ populations (fold-changes for CD44+ CD133+ sphere numbers relative to that with other populations were 3.7, 2.5, and 12.1 respectively; Fig. 1c). In addition, sphere sizes were larger in the CD44+ CD133+ population than in other populations (Fig. 1b). The results indicate that CD44+ CD133+ populations exhibit the most stem cell-like properties compared to other populations.
Figure 1.
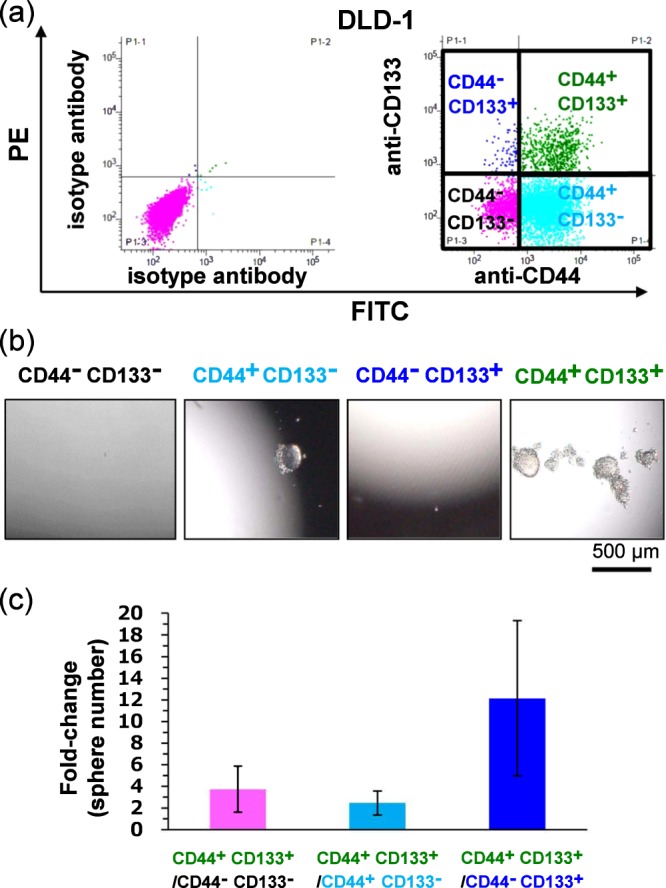
Formation of stem cell spheres after seeding sorted CD44+ CD133+, CD44− CD133−, CD44− CD133+, and CD44+ CD133− cells of the colorectal cancer (CRC) DLD-1 cell line. (a) Left column shows isotype control and right column shows anti-CD44-FITC and anti-CD133-PE antibody double-staining of DLD-1 cells. The quadrants comprising each population are defined as CD44− CD133+, CD44+ CD133+, CD44− CD133−, and CD44+ CD133−, respectively. (b) Representative sphere images of CD44− CD133− cells, CD44− CD133+ cells, CD44+ D133− cells, and CD44+ CD133+ cells are shown from the left to right column, respectively. (c) Sphere numbers determined for CD44− CD133−, CD44− CD133+, CD44+ D133−, and CD44+ CD133+ DLD-1 cells. Data points represent means ± SD (n = 6).
Anti-proliferative effect of FTD on isolated CD44+ CD133+ cells
We next investigated whether FTD was effective against CSC-like CD44+ CD133+ DLD-1 cells. The antiproliferative effect of FTD on these cells was investigated by performing cytotoxicity tests with crystal violet staining on CD44+ CD133+ (depicted in Fig. 1a) and unsorted DLD-1 cells. After 72 h of treatment, FTD was effective against both cell populations, with the calculated IC50 values being 10.7 and 8.9 μM, respectively (Fig. 2a). In contrast, resistance toward 5-FU was higher for CD44+ CD133+ DLD-1 cells (IC50 = 5.5 μM) than for unsorted DLD-1 cells (IC50 = 2.5 μM); the fold-change in IC50 was 2.2 for 5-FU and 1.2 for FTD (Fig. 2b). These results indicate that FTD is effective against a CD44+ CD133+ CSC-like population.
Figure 2.
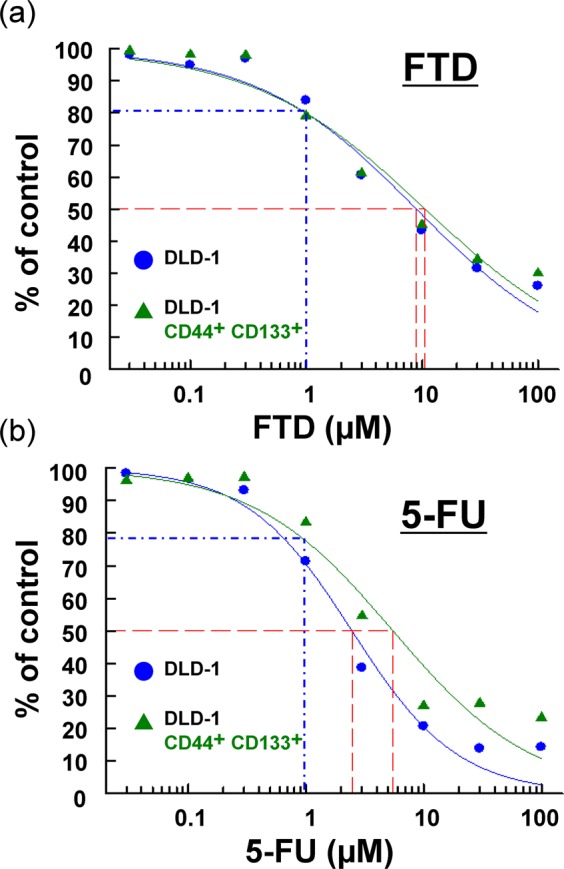
Antiproliferative effect of trifluridine (FTD) on isolated CD44+ CD133+ cells. Sorted CD44+ CD133+ cells (shown in Fig. 1) and unsorted DLD-1 cells shown here were cultured with various concentrations of FTD (a) and fluorouracil (5-FU) (b). Cell viability was determined using crystal violet staining based on at least three independent experiments. Data points represent means ± SD (n = 3). Blue dashed line represents estimated viability determined for 1 µM FTD and 5-FU; values were estimated using a fitting curve in the logistic model. Red dashed line represents estimated IC50 values.
FTD treatment and sphere-formation activity
Next, to investigate the effect of FTD treatment on the sphere-forming capacity of CD44+ CD133+ DLD-1 cells, we performed sphere-formation assays on cells treated with FTD and 5-FU (Fig. 3a,b). As compared to the number of spheres in control samples (DMSO treatment), fewer spheres were present in cells treated with FTD at 1 μM, but not in those treated with 5-FU at 1 μM (Fig. 3b). Both drug concentrations used in this study were sub-toxic (estimated viability determined in the presence of FTD and 5-FU based on cytotoxicity assays with crystal violet staining at 72 h was 79.9% and 77.6%, respectively; both values were estimated based on a fitting curve in the logistic model). Thus, the efficacy of FTD was greater than that of 5-FU in terms of sphere-formation activity, although the cytotoxic effects of both drugs at 1 μM were comparable.
Figure 3.
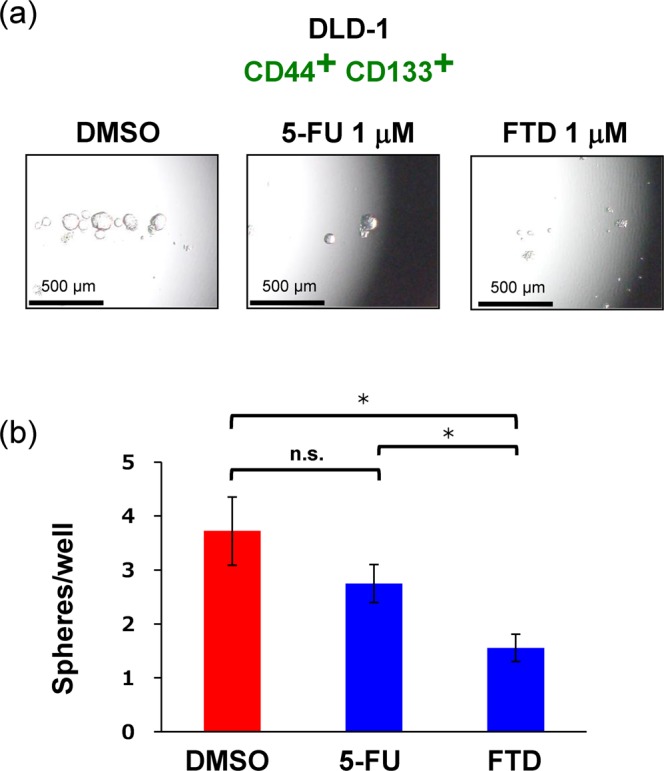
FTD efficacy toward sphere-forming activity of CD44+ CD133+ cells. Sphere culture was performed using the CD44+ CD133+ population of the DLD-1 cell line. (a) Images of cell spheres treated with DMSO (Control), 1 μM 5-FU, and 1 μM FTD; (b) Respective sphere numbers. Data points represent means ± SD (n = 6); Student’s t-test, *P < 0.05, n.s.: not significant.
Thymidine kinase 1 expression in CD44+ CD133+ cells
FTD was previously reported to be phosphorylated by thymidine kinase 1 (TK1) and incorporated into DNA16. In addition, FTD incorporation into DNA is the main mechanism underlying its anti-tumour effects17. Thus, to further investigate the inhibitive effect of FTD on the sphere-forming capacity of CD44+ CD133+ DLD-1 cells, we performed real-time PCR analysis to determine the expression of TK1 using CD44+ CD133+ DLD-1 cells and unsorted cells. As compared to that in unsorted DLD-1 cells, CD44+ CD133+ DLD-1 cells exhibited 2.3-fold increased TK1 expression (Fig. 4a).
Figure 4.
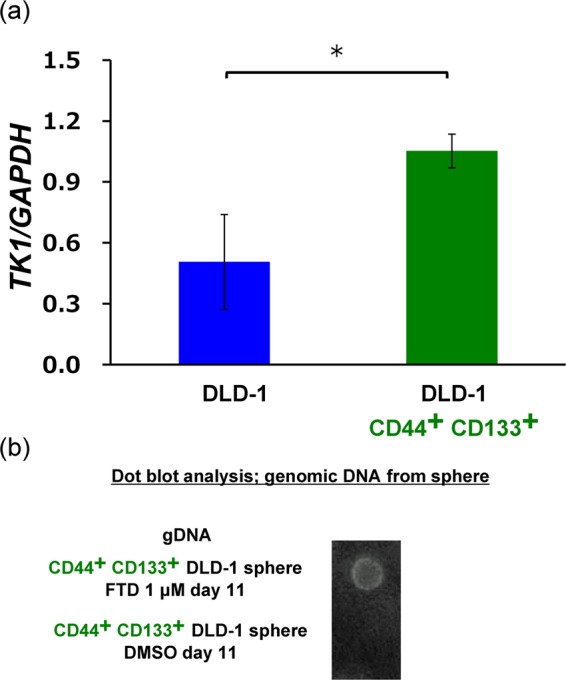
Thymidine kinase (TK1) expression and FTD incorporation in CD44+ CD133+ DLD-1 cells. (a) TK1 expression in CD44+ CD133+ cells. TK1 levels in DLD-1 cells and CD44+ CD133+ DLD-1 cells were assayed using qRT-PCR and normalized against GAPDH levels. Data points represent means ± SD of triplicate determinations; Student’s t-test, *P < 0.05. (b) Dot plot analysis of FTD incorporation into the genomic DNA of CD44+ CD133+ DLD-1 spheres. CD44+CD133+ DLD-1 cells were cultured in the presence of 1 μM FTD or DMSO for 11 days in sphere-formation assay conditions and genomic DNA was purified. Purified DNA (1 ng) was denatured with an alkaline solution (0.1 N NaOH), spotted onto a Hybond-N+ membrane, and blotted with anti-BrdU antibodies.
FTD incorporation into the DNA of CD44+ CD133+ spheres
Finally, because TK1 was highly expressed in CD44+ CD133+ DLD-1 cells, we hypothesised that FTD could be incorporated into the DNA of these cells to inhibit their sphere-forming activity. Hence, we investigated the incorporation of FTD into the DNA of CD44+ CD133+ DLD-1 spheres. Detectable spheres were formed approximately 10 days after cell seeding. Further, after sorting CD44+ CD133+ DLD-1 cells, the expression of both markers was maintained for 14 days (data not shown). Therefore, the genomic DNA of CD44+ CD133+ DLD-1 spheres treated with 1 µM FTD was isolated after 11 days, and this compound was detected by dot plot analysis using an FTD-cross-reacting anti-BrdU antibody18. Indeed, FTD was detected in the DNA of treated CD44+ CD133+ DLD-1 spheres but not in the DNA of DMSO (control)-treated cells (Fig. 4b). This result indicates the contribution of FTD DNA-incorporation to its inhibitory effect on sphere-forming activity.
Discussion
In this study, we showed that CD44+ CD133+ DLD-1 cells exhibit the highest sphere-forming activity compared to CD44− CD133−, CD44+ CD133−, and CD44− CD133+ DLD-1 cells, as reported previously for HCT-116 cells19. Here, isolated CD44+ CD133+ DLD-1 cells also exhibited increased resistance to 5-FU but not FTD as compared to that in unsorted DLD-1 cells based on a sensitivity test (Fig. 2a,b); moreover, FTD but not 5-FU treatment inhibited sphere-forming activity against these cells at a subtoxic concentration (Fig. 3a,b). These results indicated that FTD is effective against CSC-like cells, unlike 5-FU.
In a previous study, a CSC-like CD44-stably expressing MKN28 gastric cancer cell line was reported to have resistance to cisplatin and docetaxel8 and CSC-like CD133-highly expressing primary colon cancer cells were reported to exhibit resistance to 5-FU and L-OHP3. Among DLD-1 cells, a CD133-expressing population was also found to be more resistant to 5-FU treatment compared to that with unsorted cells20. Further, 5-FU treatment was found to induce an increase in CD44 and CD133 expression, and CD44- and CD133-highly-expressing cells were reported to exhibit stem cell-like properties and resistance to 5-FU15. In CSC-like cells, one drug resistance mechanism, with respect to 5-FU and other drugs, was reported to be partially dependent on TRAIL-induced death cell, which was inhibited by the expression of anti-apoptotic proteins3. CSC-like CD133+ cells are also enriched in anti-apoptotic proteins and are resistant to 5-FU21. Our study showed that unlike 5-FU, FTD was effective against CD44+CD133+ cells. Hence, another mechanism underlying the response of CSC-like cells (such as CD44+CD133+ cells) to FTD treatment, compared to that associated with 5-FU, is suggested. Poorly-differentiated human hepatocellular carcinoma cells, which present an overlapping stem cell-like phenotype with CSCs, have been reported to show the upregulated expression of pyrimidine metabolism-associated rate-limiting enzymes including TK122. In addition, CD44 expression is positively correlated with TK1 expression in triple negative breast cancer23. Moreover, TK1 mRNA was found to be upregulated in CD133-expressing human embryonic stem cells24. Because TK1 catalyses FTD phosphorylation16, it might be readily incorporated into the DNA in TK1-upregulated CSC-like populations. In this study, TK1 mRNA expression was higher in CD44+ CD133+ DLD-1 cells than in other populations (Fig. 4a), and indeed, FTD was incorporated into the DNA of CD44+ CD133+ DLD-1 spheres (Fig. 4b). FTD incorporation into the DNA of CSC-like cells might explain the anti-sphere forming activity of FTD toward 5-FU-resistant CSC-like spheres. Although CSC-like cells could be enriched in anti-apoptotic proteins, incorporation of the nucleotide analogue FTD into the DNA might provoke the interruption of DNA replication and delay the self-renewal of CSC-like cells. As a consequence, a preferential antiproliferative effect might be sustained in CSC-like cells (Fig. 5).
Figure 5.
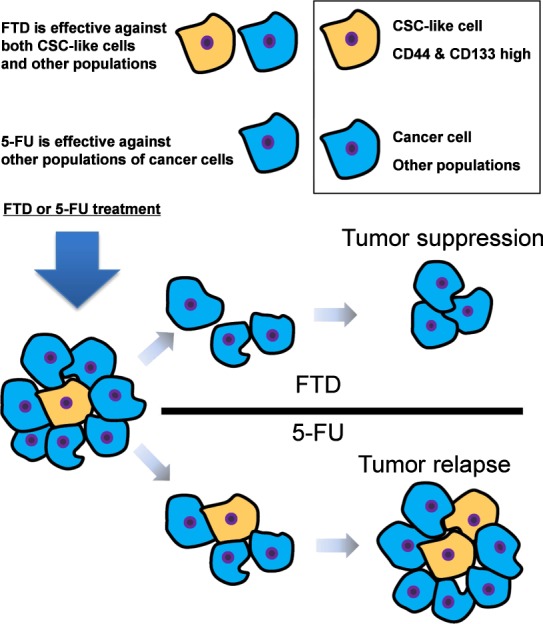
Schematic view of the efficacy of FTD treatment on CD44+ CD133+ cells. FTD is effective against cancer stem cell (CSC)-like (CD44+ CD133+) cells, but CSC-like cells show resistance toward 5-FU. FTD could be readily incorporated into the DNA of CSC-like cells. Consequently, FTD might exert an anti-sphere-forming effect on CSC-like cells, unlike 5-FU.
There are several limitations to this study. First, we used only DLD-1 cells, which were associated with the highest sorting efficacy for both CD44− CD133− and CD44+ CD133+ populations among three cell lines tested, also including HCT-116 and RKO cells (data not shown), to determine the efficacy of FTD toward CD44+ CD133+ CRC cells. Moreover, our study showed that FTD exerted anti-sphere-forming activity toward CSC-like CD44+ CD133+ cells. However, these results are based only on in vitro experiments and therefore must be confirmed using in vivo assays and clinical studies. Accordingly, to investigate FTD efficacy in CSC-like CD44+ CD133+ cells, we are planning an in vivo serial transplantation study to evaluate whether it can decrease tumorigenic capacity.
Despite the limitations, our study indicated for the first time that unlike other anti-cancer drugs, FTD might be effective against CSC-like cells expressing high levels of CD44 and CD133. These effects on CSC-like cells might explain the observed improvement in overall survival in patients treated with FTD/TPI in the clinical setting13. For gastric cancer, FTD/TPI has been used to treat patients with heavily-pre-treated metastatic gastric/gastroesophageal junction (GEJ) cancers who have progressed or are intolerant to previous lines of therapy, and this was found to improve overall survival25. In gastric cancer, CD44 and CD133 were also reported to be relevant to chemoresistance26. Therefore, similarly, the improvement in overall survival could be due to the efficacy of FTD against CSC-like cells exhibiting high CD44 and CD133 expression and might be applicable for GEJ, as well as CRC. In addition, a similar response rate between FTD/TPI + bevacizumab and capecitabine + bevacizumab was confirmed in the TASCO-1 clinical trial conducted based on a first-line setting27; thus, the possibility that this drug might also be useful in an adjuvant setting (stem-like cell killing effect) should also be considered. Furthermore, this study suggests a potential role for FTD/TPI in chemo-preventive therapy due to its efficacy against drug-resistant (or at least 5-FU-resistant) CD44+ CD133+ CSC-like cells at subtoxic, low doses (see Figs 2 and 3). Although further investigation is required to clarify the efficacy of this drug for tumors exhibiting CSC-like properties in the clinical setting, our study indicates that FTD-based treatment might lead to improved clinical outcomes for patients with tumours exhibiting high CD44 and CD133 expression.
Materials and Methods
Cell lines and culture conditions
The DLD-1 human colon cancer cell line was obtained from the American Type Culture Collection and grown in Dulbecco’s modified Eagle’s medium (DMEM; Sigma-Aldrich, Tokyo, Japan) supplemented with 10% fetal bovine serum (Life Technologies, Carlsbad, CA, USA), 100 U/mL penicillin, and 100 µg /mL streptomycin (Life Technologies). Cells were grown at 37 °C in a humidified atmosphere with 5% CO2. Cell lines were maintained as mentioned, and all lines were verified based on short tandem repeats before the study; all experimental procedures were performed using exponentially growing cells.
Chemicals
FTD, 5-FU were purchased from Tokyo Chemical Industry (Tokyo, Japan).
Cytotoxicity assay
Cell lines were seeded at a density of 2000 cells/well in 96-well plates, precultured for 24 h, and then exposed to FTD (0.03, 0.1, 0.3. 1, 3, 10, 30, 100, 300 μM) or 5-FU (0.03, 0.1, 0.3. 1, 3, 10, 30, 100, 300 μM) for 72 h. The in vitro cytotoxic effects of FTD and 5-FU were evaluated based on crystal violet staining. For this, cells were fixed with 2% glutaraldehyde for 20 min, stained with 0.05% crystal violet (Wako, Tokyo, Japan) in 20% methanol for 20 min, and rinsed with tap water; subsequently, the plates were dried on paper for 1 h and then 100 μL of a 1:1 mixture of ethanol and 0.1 M sodium dihydrogen phosphate was added to each well. Absorbance was measured at 595 nm by using an EnSpire system (PerkinElmer, Waltham, MA, USA). The FTD and 5-FU concentration that inhibited cell growth by 50% (IC50) was calculated from the generated regression lines.
Fluorescence-activated cell sorting (FACS) analysis of cells
To analyse and sort based on the expression of CD44 and CD133, cells were routinely cultured, harvested, digested with Accutase (Life Technologies), and resuspended in staining buffer (1 × 107 cells in 1 mL), and then incubated with antibodies (mouse anti-human CD44-FITC (fluorescein isothiocyanate) and mouse anti-human CD133-PE (Phycoerythrin); Miltenyi Biotec, Bergisch Gladbach, NW, Germany) for 10 min according to the manufacturer’s procedures. After staining, cells were analysed and sorted by flow cytometry using a BD FACSARIA cell sorter and a BD FACSMelody Cell Sorter (Becton Dickinson, Franklin Lakes, NJ, USA).
Sphere-formation assay
Cells were seeded into 96-well flat-bottom ultra-low-attachment culture dishes (Corning, Corning, NY, USA) at 100–500 cells/well in DMEM/F-12 serum-free medium (Life Technologies) containing 20 ng/mL h-EGF, 20 ng/mL h-bFGF (Sigma-Aldrich), 2% B27 Supplement, and 1% N-2 Supplement (Life Technologies) with or without FTD and 5-FU. After 14 to 40 days, the size of spheroid colonies was measured under a microscope, and colonies with a diameter exceeding 50 μm were counted.
Real-time quantitative reverse transcription-polymerase chain reaction (qPCR)
To assay mRNA expression, cDNA was synthesized from extracted total RNA, and then qPCR was performed using the synthesized cDNA products, ReverTra Ace qPCR RT Master Mix (TOYOBO, Tokyo, Japan), and the SYBR qRT-PCR Kit (Clontech, Mountain View, CA); cDNA was amplified using primers specific for TK1 (forward: 5ʹ-AGAGTACTCGGGTTCGTGAACTT-3ʹ; reverse: 5ʹ-CACTTGTACTGAGCAATC-3ʹ). GAPDH was used as a normalisation control (primers, forward: 5ʹ-GAGTCAACGGATTTGGTCGT-3ʹ; reverse: 5ʹ-TTGATTTTGGAGGGATCTCG-3ʹ). Relative expression was calculated using the CT-based calibrated standard-curve method. The calculated values were then normalised against GAPDH expression.
Dot plot analysis
Genomic DNA was purified from CD44+ CD133+DLD-1 spheres cultured in the presence of 1 μM FTD for 11 days using the NucleoSpin® Tissue kit (MACHEREY-NAGEL, Düren, NN, Germany). Equal amounts (1 ng: DLD-1 in 2 μL) of denatured (0.1 N NaOH for 5 min at room temperature (RT)) genomic DNA were spotted onto a Hybond-N+ blotting membrane (GE Healthcare Life Sciences, Little Chalfont, Chicago, IL, USA). The membranes were baked at 80 °C for 2 h, blocked with Blocking-One solution (Nacalai Tesque, Kyoto, Japan), and blotted with diluted anti-BrdU antibody 3D4 (Cat#555627) from BD Biosciences (San Jose, CA, USA). Chemiluminescent signals were captured using the ChemiDoc™ Touch system (BIO-RAD, Hercules, CA, USA).
Statistical analysis
Statistically significant differences were identified using the Student’s t-test as appropriate. All statistical analyses were performed using JMP Pro 9 software (SAS Institute, Cary, NC, USA).
Acknowledgements
We thank the members of our laboratories for their helpful discussion. This work received financial support from grants-in-aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology (to M.M. and H.I.; grant nos. 17H04282, 17K19698, 16K15615, and 15H05791) and Taiho Pharmaceutical Co., Ltd. (to Osaka University, Japan).
Author contributions
H.I. and M.M. conceived this study; K.T. conducted the experiments; K.T., M.K., J.K., T.K., and A.A. analysed and interpreted the data; K.T. wrote the manuscript; N.H., K.M., T.T., Y.D., and M.M. supervised the project. All authors reviewed the manuscript.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.
Competing interests
This study was funded by Taiho Pharmaceutical Co., Ltd (Tokyo, Japan), Evidence Based Medical Research Center INC. (Osaka, Japan), Unitech Co., Ltd. (Chiba, Japan), IDEA Consultants, Inc. (Tokyo, Japan), and Kinshu-kai Medical Corporation (Osaka, Japan). K.T., K.M., T.K., and T.T., are employees of Taiho Pharmaceutical Co. Ltd (Tokyo, Japan).
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Masaki Mori, Email: mmori@gesurg.med.osaka-u.ac.jp.
Hideshi Ishii, Email: hishii@gesurg.med.osaka-u.ac.jp.
References
- 1.O’Brien CA, Pollett A, Gallinger S, Dick JE. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature. 2007;445:106–110. doi: 10.1038/nature05372. [DOI] [PubMed] [Google Scholar]
- 2.Ricci-Vitiani L, et al. Identification and expansion of human colon-cancer-initiating cells. Nature. 2007;445:111–115. doi: 10.1038/nature05384. [DOI] [PubMed] [Google Scholar]
- 3.Todaro M, et al. Colon cancer stem cells dictate tumor growth and resist cell death by production of interleukin-4. Cell Stem Cell. 2007;1:389–402. doi: 10.1016/j.stem.2007.08.001. [DOI] [PubMed] [Google Scholar]
- 4.Zeuner A, Todaro M, Stassi G, De Maria R. Colorectal cancer stem cells: From the crypt to the clinic. Cell Stem Cell. 2014;15:692–705. doi: 10.1016/j.stem.2014.11.012. [DOI] [PubMed] [Google Scholar]
- 5.Batlle E, Clevers H. Cancer stem cells revisited. Nat. Med. 2017;23:1124–1134. doi: 10.1038/nm.4409. [DOI] [PubMed] [Google Scholar]
- 6.Chen S, et al. CD133 Expression and the prognosis of colorectal cancer: A systematic review and meta-analysis. PLoS One. 2013;8:1–9. doi: 10.1371/annotation/dd945f7c-c50b-461d-ab38-15e8b0966458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kreso A, Dick JE. Evolution of the cancer stem cell model. Cell Stem Cell. 2014;14:275–291. doi: 10.1016/j.stem.2014.02.006. [DOI] [PubMed] [Google Scholar]
- 8.Ishimoto T, et al. CD44 variant regulates redox status in cancer cells by stabilizing the xCT subunit of system xc- and thereby promotes tumor growth. Cancer Cell. 2011;19:387–400. doi: 10.1016/j.ccr.2011.01.038. [DOI] [PubMed] [Google Scholar]
- 9.Horst D, Kriegl L, Engel J, Kirchner T, Jung A. Prognostic significance of the cancer stem cell markers CD133, CD44, and CD166 in colorectal cancer. Cancer Invest. 2009;27:844–850. doi: 10.1080/07357900902744502. [DOI] [PubMed] [Google Scholar]
- 10.Wakamatsu Y, et al. Expression of cancer stem cell markers ALDH1, CD44 and CD133 in primary tumor and lymph node metastasis of gastric cancer. Pathol. Int. 2012;62:112–119. doi: 10.1111/j.1440-1827.2011.02760.x. [DOI] [PubMed] [Google Scholar]
- 11.Yoshino T, et al. TAS-102 monotherapy for pretreated metastatic colorectal cancer: A double-blind, randomised, placebo-controlled phase 2 trial. Lancet Oncol. 2012;13:993–1001. doi: 10.1016/S1470-2045(12)70345-5. [DOI] [PubMed] [Google Scholar]
- 12.Eng C, et al. Atezolizumab with or without cobimetinib versus regorafenib in previously treated metastatic colorectal cancer (IMblaze370): a multicentre, open-label, phase 3, randomised, controlled trial. Lancet Oncol. 2019;102:1–13. doi: 10.1016/S1470-2045(19)30027-0. [DOI] [PubMed] [Google Scholar]
- 13.Mayer RJ, et al. Randomized trial of TAS-102 for refractory metastatic colorectal cancer. N. Engl. J. Med. 2015;372:1909–1919. doi: 10.1056/NEJMoa1414325. [DOI] [PubMed] [Google Scholar]
- 14.Fukushima M, et al. Structure and activity of specific inhibitors of thymidine phosphorylase to potentiate the function of antitumor 2′-deoxyribonucleosides. Biochem. Pharmacol. 2000;59:1227–1236. doi: 10.1016/S0006-2952(00)00253-7. [DOI] [PubMed] [Google Scholar]
- 15.Dallas NA, et al. Chemoresistant colorectal cancer cells, the cancer stem cell phenotype, and increased sensitivity to insulin-like growth factor-I receptor inhibition. Cancer Res. 2009;69:1951–1957. doi: 10.1158/0008-5472.CAN-08-2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sakamoto K, et al. Crucial roles of thymidine kinase 1 and deoxyUTPase in incorporating the antineoplastic nucleosides trifluridine and 2′-deoxy-5-fluorouridine into DNA. Int. J. Oncol. 2015;46:2327–2334. doi: 10.3892/ijo.2015.2974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Peters GJ. Therapeutic potential of TAS-102 in the treatment of gastrointestinal malignancies. Ther. Adv. Med. Oncol. 2015;7:340–356. doi: 10.1177/1758834015603313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kitao H, et al. The antibodies against 5-bromo-2′-deoxyuridine specifically recognize trifluridine incorporated into DNA. Sci. Rep. 2016;6:1–12. doi: 10.1038/s41598-016-0001-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhou J-Y, et al. Role of CD44high/CD133high HCT-116 cells in the tumorigenesis of colon cancer. Oncotarget. 2016;7:7657–7666. doi: 10.18632/oncotarget.7084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Deng YH, et al. 5 Fluorouracil upregulates the activity of Wnt signaling pathway in CD133positive colon cancer stemlike cells. Chin. J. Cancer. 2010;29:810–815. doi: 10.5732/cjc.010.10134. [DOI] [PubMed] [Google Scholar]
- 21.Ma S, Lee TK, Zheng B, Chan KW, Guan X. CD133+ HCC cancer stem cells confer chemoresistance by preferential expression of the Akt/PKB survival pathway. Oncogene. 2008;27:1749–1758. doi: 10.1038/sj.onc.1210811. [DOI] [PubMed] [Google Scholar]
- 22.Yeh H-W, et al. Pyrimidine metabolic rate limiting enzymes in poorly-differentiated hepatocellular carcinoma are signature genes of cancer stemness and associated with poor prognosis. Oncotarget. 2017;8:77734–77751. doi: 10.18632/oncotarget.20774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Weagel EG, et al. Membrane expression of thymidine kinase 1 and potential clinical relevance in lung, breast, and colorectal malignancies. Cancer Cell Int. 2018;18:135. doi: 10.1186/s12935-018-0633-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kim CG, et al. Profiling of differentially expressed genes in human stem cells by cDNA microarray. Mol. Cells. 2006;21:343–355. [PubMed] [Google Scholar]
- 25.Shitara K, et al. Trifluridine/tipiracil versus placebo in patients with heavily pretreatedmetastatic gastric cancer (TAGS): a randomised, double-blind, placebo-controlled,phase 3 trial. Lancet Oncol. 2018;19:1437–1448. doi: 10.1016/S1470-2045(18)30739-3. [DOI] [PubMed] [Google Scholar]
- 26.Bekaii-Saab T, El-Rayes B. Identifying and targeting cancer stem cells in the treatment of gastric cancer. Cancer. 2017;123:1303–1312. doi: 10.1002/cncr.30538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Van Cutsem E, et al. Abstracts. Annals of Oncol. 2018;29(suppl_5):O–022. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated during and/or analysed during the current study are available from the corresponding author upon reasonable request.


