Abstract
Epithelial-mesenchymal transition (EMT) has been shown to exert promoting effects on the progression of a number of cancer types, including endometrial carcinoma (EC). MicroRNA (miRNA or miR)-195 has been shown to function as a tumor suppressor. This study aimed to explore the potential role of miR-195 in the EMT process of EC. miR-195 overexpression (Mimics) and mimics control (Mock) vectors were constructed and transfected into human endometrial cancer cells (AN3-CA and Hec1A) using Lipofectamine 2000, and cell viability was detected using the Cell Counting kit-8 (CCK-8). The invasive and migratory capacities of the cells transfected with the Mimics or Mock vectors were assessed by Transwell and wound healing assays. Relative mRNA and protein levels were analyzed by reverse transcription-quantitative polymerase chain reaction (RT-qPCR) and western blot analysis, respectively. Using TargetScan prediction, the potential target of miR-195 was identified and was further verified by dual-luciferase reporter assay. Following transfection with miR-195 mimics, the viability of the AN3-CA and Hec1A cells decreased in a time-dependent manner, specifically at 24 h. The wound closure rate and the number of invaded cells in the Mimics group were much lower than those in the Control and Mock groups (P<0.01). miR-195 overexpression significantly upregulated the mRNA and protein levels of tissue inhibitor of metalloproteinase 2 (TIMP-2), while it downregulated the expression levels of matrix metalloproteinase (MMP)-2 and MMP-9. Moreover, the phosphorylation levels of PI3K and AKT were also notably decreased (P<0.01). G protein-coupled estrogen receptor 1 (GPER) was identified as a target of miR-195. On the whole, the findings of this study indicate that the inhibitory effects of miR195 on EC cell migration and invasion are associated with the PI3K/AKT signaling pathway and GPER expression.
Keywords: metastasis, cell viability, dual-luciferase reporter assay, PI3K/AKT signaling pathway
Introduction
Endometrial carcinoma (EC) is one of three most common types of malignant tumors of the female reproductive tract worldwide, accounting for 20–30% of the total number of female malignant tumors of the genital tract (1). Over the past few years, the incidence of EC has been increasing. In 2014, there were 52,630 new EC cases and 8,590 EC-related deaths, while in 2017, an estimated number of 61,380 would be diagnosed with EC and more than one-sixth of these would perhaps succumb to the disease (2,3). Currently, radical surgery is one of the most effective treatments for early-stage EC; however, 40% of cases receiving surgery still have a risk of recurrence and metastasis (4). Previous studies have indicated that the main cause of the high mortality rate among patients with EC is recurrence and metastasis (5,6). Epithelial-mesenchymal transition (EMT) is closely associated with tumor invasion and metastasis; EMT promotes the disengagement of epithelial cells from cell-cell adhesion and cell polarity, thus promoting cell migration and invasion, as well as the development of an aggressive phenotype (7–9). Previous research has revealed the molecular mechanisms of common EMT in EC, including the downregulation of the E-cadherin level, and other molecular alterations consistent with the mesenchymal phenotype (10). In addition, it has been indicated that EMT affects EC through estrogen signaling and microRNAs (miRNAs or miRs) (11).
G protein-coupled estrogen receptor (GPER or GPR30) is a member of the 7-transmembrane spanning G protein-coupled receptor (GPCR) superfamily, structurally unrelated to the nuclear estrogen receptors (nERs) (12). Currently, high levels of GPER have been detected and it exists at least 4 types of human tumor cells, including breast cancer (13), thyroid cancer (14), ovarian cancer (15) and EC (16) cells. Moreover, increasing evidence has indicated that GPER is significantly associated with tumor progression, migration, infiltration and prognosis (17,18). A previous study demonstrated that the abnormal expression of GPER was mainly found in advanced tumors and that GPER was associated with a poor survival of patients with EC; in that study, only 65.2% of patients with EC with GPER overexpression survived, whereas a 100% survival rate was noted in patients with EC with normal GPER levels (19). These findings indicated GPER may be a promising biomarker for the progression of EC. Moreover, GPER has also been reported to be associated with the activation of several signaling pathways; for example, GPER binding to estradiol can rapidly activate the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway through non-genomic effects, promoting cell proliferation (20,21).
miRNAs are a class of non-coding short RNAs (21–25 bp in length), which can modulate the expression of a large number of genes by binding to the 3′-UTR of their transcripts (22). In 2014, by comparative analysis of endometrioid EC (EEC) tissue, normal endometrium (NE) tissue and blood cells of the same patient, miR-195 was demonstrated as one of the key miRNAs of EC pathogenesis (23). In this study, we investigated the effects of miR-195 on the EMT process in EC.
Materials and methods
Cells and cell culture
Human endometrial cancer cell lines (AN3-CA and Hec1A) were obtained from the American Type Culture Collection (ATCC). The cells was curtured with Eagle's minimum essential medium (EMEM, #30-2003, ATCC) with 10% (v/v) fetal bovine serum (FBS, #30-2020, ATCC), and grown at 37°C under 5% CO2 atmosphere.
Cell transfection
miR-195 mimics (5′-UAGCAGCACAGAAAUAUUGGC-3′) and negative control (5′-UUCUCCGAACGUGUCACGUTT-3′) were synthesized by GenePharma. The AN3-CA or Hec1A cells were embedded in 6-well plates (5×104 cells/well) containing serum-free medium, and the cells were then cultured for 3 h at 37°C. miR-195 mimics or Mock vectors (50 nM) were added to 250 µl EMEM medium, supplemented with Lipofectamine 2000 reagent (Invitrogen), keeping the mixture at 37°C in 5% CO2 for 6 h. The transfected cells were harvested and cultured with new medium for 24 h at 37°C. After 24 h, transfected cells were collected using EDTA (0.25%)-Trypsin (Sigma-Aldrich; Merck KGaA), for the assessment of the transfection efficiencies.
Cell Counting Kit-8 (CCK-8) assay
The CCK-8 assay (Dojindo Laboratories) was used for the analysis of cell viability. In brief, the AN3-CA or Hec1A cells were embedded in 6-well plates at a cell density of 1×105 per well and cultured in a 5% CO2 humidified atmosphere at 37°C. The cells were harvested at 3 time points (12, 24 and 48 h), and the harvested cells were then cultured with 10% CCK-8 reagent, respectively. Following incubation for 1 h at 37°C, the absorbance at 450 nm was quantified using a Microplate Reader (Multiskan FC, Thermo Fisher Scientific).
Wound healing assay
The AN3-CA or Hec1A cells were embedded in 6-cm culture dishes and cultured at 37°C in 5% CO2 humidified atmosphere until the cells reached 90% confluence. A sterile pipette tip was used to create a cell-free line. At 24 h after the scratch was made, the condition of wound healing was captured using a Nikon ECLIPSE Ts2 light microscope (Nikon Corporation), and the migration rates were calculated by comparing the width of the wound.
Transwell assay
The invasive ability of the AN3-CA and Hec1A cells was assessed by Transwell chamber assay. Briefly, 5×105 AN3-CA or Hec1A cells or transfected cells were seeded into the 8-µm pore size of 6-well Matrigel invasion chambers (BD Biosciences). The top chamber was loaded with 200 µl serum-free EMEM, while the bottom chamber was loaded with 600 µl EMEM medium containing 20% FBS. The chamber was maintained at 37°C in a 5% CO2 humidified incubator for 24 h, and the cells on the upper surface of the membrane were removed using a cotton swab, and the cells that had invaded into the bottom chamber were fixed with 4% paraformaldehyde for 15 min, followed by staining with 0.1% crystal violet solution for 20 min at 37°C. Five random field views were selected for counting.
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
miRNAs from each experimental group were isolated using the miRcute miRNA Isolation kit (Tiangen), and then reverse transcribed using the miScript II RT kit (Qiagen GmbH) according to the instructions of the manufacturer. All samples were reacted at 37°C for 60 min, heated at 95°C for 5 min, finally held at 4°C. The miRNA levels were analyzed using the miScript SYBR-Green PCR kit (Qiagen GmbH). The 20 µl volume of qPCR reaction consisted of 2 µl cDNA, 1X QuantiTect SYBR-Green PCR Master Mix (Qiagen GmbH) and 0.5 mM of each primer. The relative miR-195 levels were normalized to U6. RNAs from AN3-CA or Hec1A cells were extracted using TRIzol reagent (Invitrogen). A total of 2 µl total RNAs were used for cDNA generation using the Prime Script RT reagent kit (Takara). The reaction protocol was as follows: 5 min at 65°C, then 6 min at 30°C and 60 min at 50°C. SYBR-Green detection was used for the assessment of the relative mRNA levels. The ABI 7500 real-time PCR system was used to carry out the miRNA and mRNA level analysis, using the following reaction parameters: Pre-degeneration at 95°C for 15 min, followed by 40 cycles at 95°C for 5 sec, and annealing at 60°C for 30 sec. Fold changes were calculated using the relative quantification (2−ΔΔCq) method (24) and normalized against GAPDH. All primers used are listed in Table I.
Table I.
Sequences of primers used for RT-qPCR.
| Gene name | Primer sequences |
|---|---|
| miR-195 | Forward: 5′-GGGGAGCCAAAAGGGTCATCATCT-3′ |
| Reverse: 5′-GAGGGGCCATCCACAGTCTTCT-3′ | |
| TIMP-2 | Forward: 5′-CCAAAGCAGTGAGCGAGAA-3′ |
| Reverse: 5′-CATCCAGAGGCACTCATCC-3′ | |
| MMP-2 | Forward: 5′-GGAGGCACGATTGGTCTG-3′ |
| Reverse: 5′-TTGGTTTCCGCATGGTCT-3′ | |
| MMP-9 | Forward: 5′-TTGACAGCGACAAGAAGTGG-3′ |
| Reverse: 5′-GGCACAGTAGTGGCCGTAG-3′ | |
| GPER | Forward: 5′-TCACGGGCCACATTGTCAACCTC-3′ |
| Reverse: 5′-GCTGAACCTCACATCTGACTGCTC-3′ | |
| U6 | Forward: 5′-CTCGCTTCGGCAGCACA-3′ |
| Reverse: 5′-AACGCTTCACGAATTTGCGT-3′ | |
| GAPDH | Forward: 5′-ACCACAGTCCATGAAATCAC-3′ |
| Reverse: 5′-AGGTTTCTCCAGGCGGCATG-3′ |
TIMP-2, tissue inhibitor of metalloproteinase 2; MMP, matrix metalloproteinase.
Western blot analysis
Subconfluent AN3-CA or Hec1A cells were suspended in RIPA buffer (Beyotime). The concentration of the cell lysates was quantified by BCA protein assay (Pierce). Proteins (15 µg) were separated on sodium dodecyl sulfate-polyacrylamide gels and electrophoretically transferred onto polyvinylidene fluoride (PVDF) membranes (Pall Inc.). After being blocked with 5% non-fat milk overnight at 4°C, the membranes were incubated with several types of primary antibodies at 4°C overnight. All antibodies were obtained from Abcam, including anti-GAPDH (rabbit, 1:1,000, cat. no. ab8245, 37 kDa), anti-tissue inhibitor of metalloproteinase 2 (TIMP-2, rabbit, 1:1,000, cat. no. ab180630, 24 kDa), anti-matrix metalloproteinase (MMP)-2, (rabbit, 1:1,000, cat. no. ab37150, 72 kDa), anti-MMP-9 (rabbit, 1:1,000, cat. no. ab73734, 95 kDa), PI3K (mouse, 1:2,000, cat. no. ab140307, 127 kDa), phospho-PI3K (p-PI3K, rabbit, 1:1,000, cat. no. ab138364, 85 kDa), AKT (rabbit, 1:1,000, cat. no. ab18785, 55 kDa), p-AKT (rabbit, 1:1,000, cat. no. ab38449, 56 kDa) and GPER (rabbit, 1:1,000, cat. no. ab137479, 42 kDa). After washing with PBS, secondary antibodies, such as goat anti-mouse IgG, HRP-linked antibody (1:2,000, cat. no. ab205719, Abcam) and goat anti-rabbit IgG H&L (HRP) (1:2,000, cat. no. ab205718) (both from Abcam) were incubated with the membranes at room temperature for 2 h. The membranes were stained with enhanced chemiluminescence solution (Pierce), visualized using a laser densitometer GeneGnome XRQ and analyzed using GeneSys image acquisition software version 3.0 (SynGene).
Luciferase reporter assay
GPER was a potential target of miR-195 based on the prediction algorithm of TargetScan (http://www.targetscan.org/vert_72/). Dual-luciferase reporter assay system (E1910; Promega) was performed to further verify this predicted target. In brief, miR-195 was inserted into the GV272 plasmid (GV272-miR-195; Shanghai Genepharma Co., Ltd.), and the 3′-UTR of GPER containing miR-195-binding site was inserted into the GV268 plasmid (GV268-GPER-3′UTR; Shanghai Genepharma Co., Ltd.). The QuikChangeH Site-Directed Mutagenesis kit (Agilent Technologies) was used to create a mutant GPER 3′-UTR containing a mutant sequence in the miR-195 binding site, and the mutant was also cloned into GV268 (GV268-GPER-3′-UTR mut). GV272-miR-195 was co-transfected with GV268-GPER-3′-UTR or GV268-GPER-3′-UTR mut into 293T cells (#CRL-3216, ATCC) by Lipofectamine 2000, and the control was transfected with GV268-GPER-3′-UTR alone. The cells were lysed and separated under a 5 min centrifugation at 15,000 × g at 4°C. The prepared dual luciferase reporter mixture was added into the supernatant of lysed cells, and the relative Firefly and Renilla luciferase activity was then immediately detected using a Sinergy 2 luminometer (Biotek Instruments).
Statistical analysis
All tests were carried out using SPSS 15.0 software (SPSS Inc.). The values are reported as the means ± SEM. One-way analysis of variance (ANOVA) followed by a post-hoc Tukey's test was used for comparisons between various groups. Differences were considered statistically significant at P<0.05.
Results
Overexpression of miR-195 exerts an inhibitory effect on AN3-CA and Hec1A cell viability
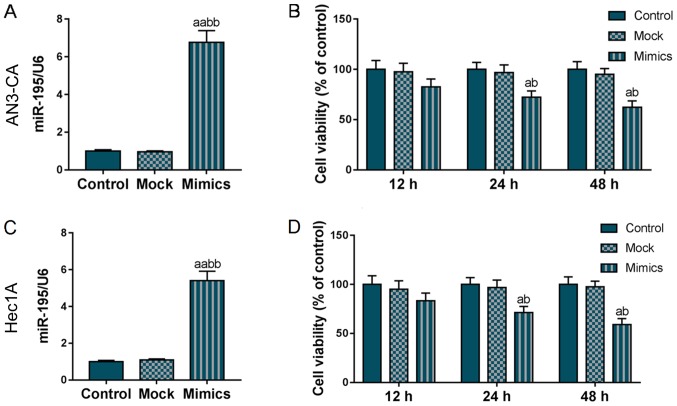
To examine the effects of miR-195 on EC development, a miR-195 overexpression AN3-CA and Hec1A cell model was constructed and the cell viability was detected using the CCK-8 kit. As shown in Fig. 1A and C, miR-195 was overexpressed in the Mimics group, indicating that the miR-195 overexpression vector was stably expressed in the AN3-CA and Hec1A cells. The results of CCK-8 kit assay revealed that the viability of both the AN3-CA and Hec1A cells gradually decreased with the increasing incubation time and from the 24-h time point, the overexpression of miR-195 significantly reduced the viability of the AN3-CA and Hec1A cells (P<0.05, Fig. 1B and D). These findings suggested that miR-195 overexpression notably inhibited AN3-CA and Hec1A cell viability.
Figure 1.
Overexpression of miR-195 exerts an inhibitory effect on AN3-CA and Hec1A cell viability. To examine the effects of miR-195 on EC development, the miR-195 mimics vector was transfected into human EC (AN3-CA and Hec1A) cells. (A and C) The transfection efficiency was determined by RT-qPCR. (B and D) Cell viabilities at 12, 24 and 48 h of each experimental group were detected using the CCK-8 kit. Each value represents the mean ± SEM (n=3). U6 was considered as an internal control. aP<0.05, aaP<0.01 vs. Control group; bP<0.05, bbP<0.01 vs. Mock group. EC, endometrial carcinoma.
Overexpression of miR-195 suppresses the migration and invasion of AN3-CA and Hec1A cells
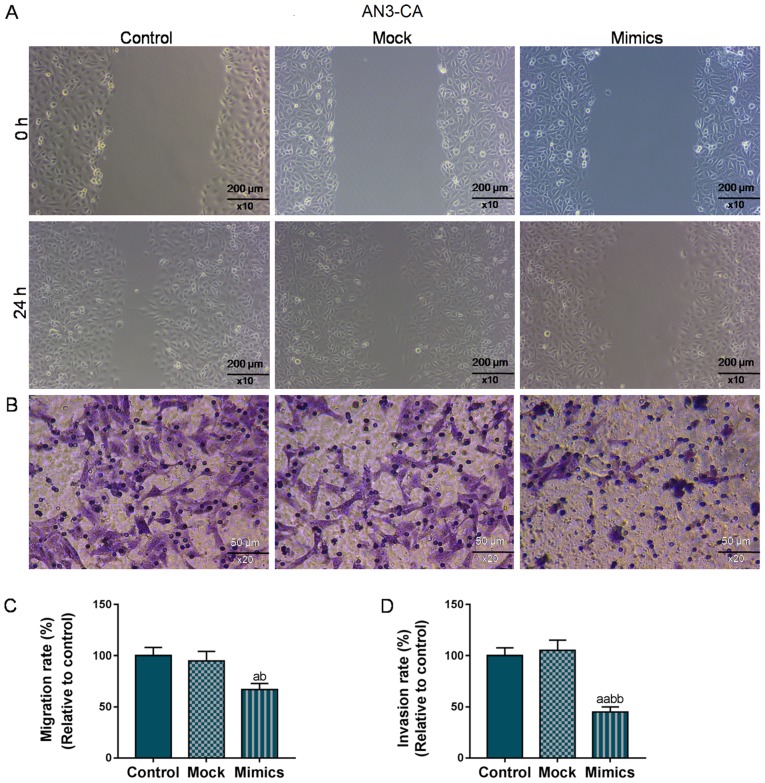
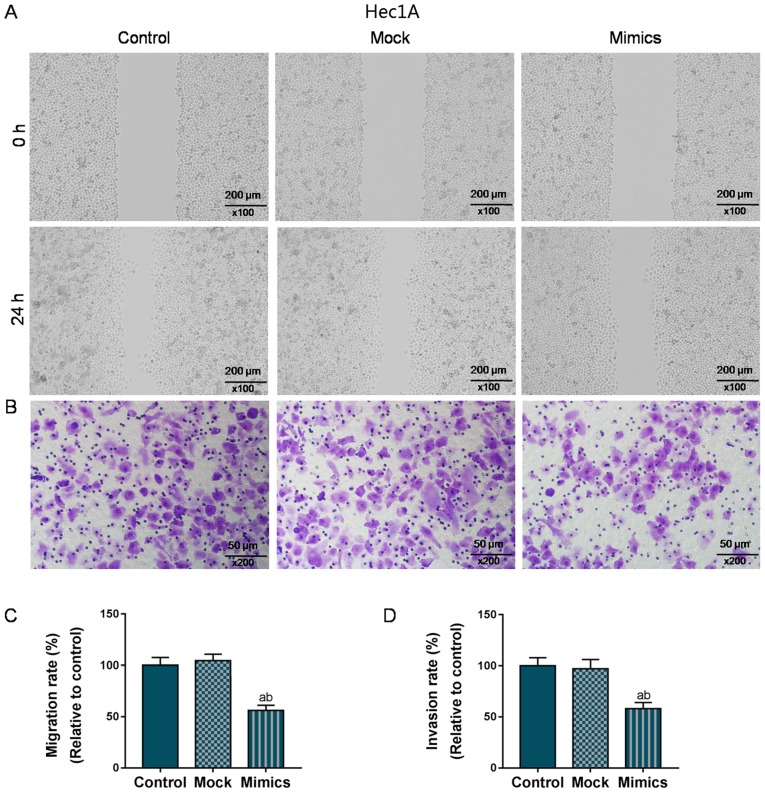
As shown in Fig. 2A and C, the wound closure rate in the AN3-CA cells in the Mimics group significantly decreased to 70% (from 100%), compared with the Control and Mock groups (P<0.05). At the same time, the number of invaded cells in the Mimics group was also markedly decreased (50 vs. 100%, P<0.01; Fig. 2B and D). Similarly, in the Hec1A cells, the overexpression of miR-195 significantly suppressed migration and invasion (P<0.05; Fig. 3). Taken together, the migratory and invasive capacities of the AN3-CA or Hec1A EC cells were effectively suppressed in the cells in which miR-195 was overexpressed.
Figure 2.
Overexpression of miR-195 exerts an inhibitory effect on the migration and invasion of AN3-CA cells. Wound healing and Transwell assays were performed to determine whether the overexpression of miR-195 affects the migratory and invasive capacities of AN3-CA cells. (A and C) Effects of miR-195 overexpression on the migration of AN3-CA cells were assessed using wound healing assay, and the wound width was observed under a microscope (magnification, ×10). (B and D) The invasive capacity of AN3-CA cells was detected by Transwell assay, and the number of invaded cells was detected under an optical microscope (magnification, ×20). Each value represents mean ± SEM (n=3). aP<0.05, aaP<0.01 vs. Control group; bP<0.05, bbP<0.01 vs. Mock group.
Figure 3.
Overexpression of miR-195 exerts an inhibitory effect on the migration and invasion of Hec1A cells. (A and C) Effects of miR-195 overexpression on the migration of Hec1A cells were assessed using wound healing assay, and the wound width was observed under a microscope (magnification, ×10). (B and D) The invasive capacity of Hec1A cells was detected by Transwell assay, and the number of invaded cells was detected under an optical microscope (magnification, ×20). Each value represents mean ± SEM (n=3). aP<0.05 vs. Control group; bP<0.05 vs. Mock group.
Overexpression of miR-195 inhibits the expression of metastasis-associated genes and the activation of the PI3K/AKT signaling pathway
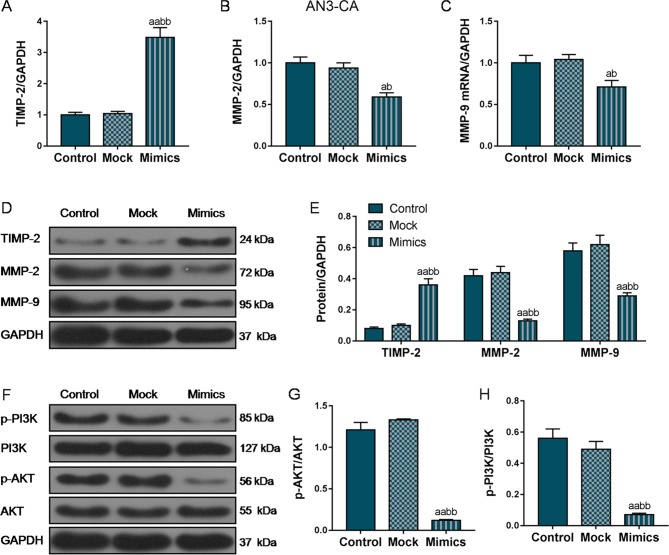
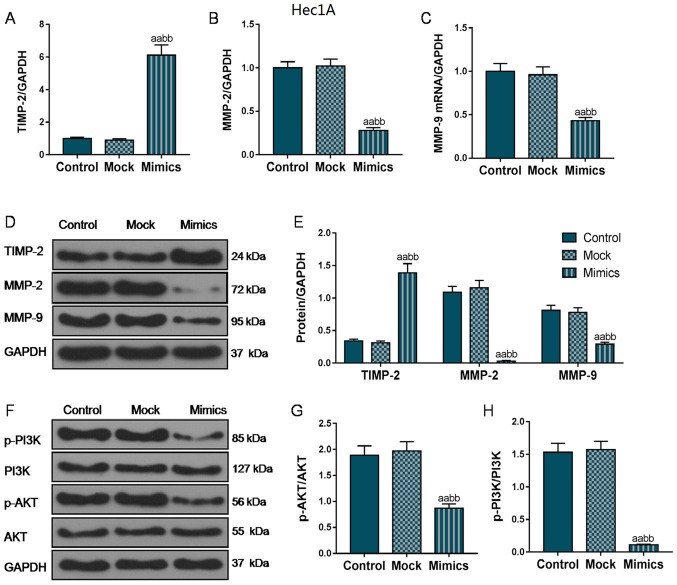
To further investigate the mechanisms through which miR-195 overexpression suppressed the migratory and invasive ability of the EC cells, RT-qPCR and western blot analysis were performed to analyze the expression of several EMT-associated genes. As shown in Fig. 4A, D and E, in the AN3-CA cells, TIMP2 mRNA and protein expression markedly increased in the Mimics group compared with that in the Control and Mock groups. In addition, the MMP-2 and MMP-9 expression levels were also markedly decreased in the Mimics group compared with the Control and Mock groups (P<0.01; Fig. 4B-E). Furthermore, we also examined the changes in the expression levels of MMPs and TIMP-2 in the Hec1A cells, and the changes in the expression trends of these EMT-related proteins in the Hec1A cells was basically consistent with that in the AN3-CA cells (Fig. 5A-E), indicating that miR-195 overexpression suppressed the invasion and metastasis of EC cells by inhibiting the expression of these EMT-associated genes.
Figure 4.
Overexpression of miR-195 inhibits the expression of metastasis-associated gene and the activation of the PI3K/AKT signaling pathway in AN3-CA cells. We further examined the effects of miR-195 on the EMT process by measuring the expression of several EMT-associated genes. (A-C) Relative mRNA levels of TIMP-2, MMP-2 and MMP-9 were detected by RT-qPCR. (D and E) Relative protein levels were measured by western blot analysis. (F-H) The protein and phosphorylation levels of PI3K and AKT were detected by western blot analysis. Each value represents the mean ± SEM (n=3). GAPDH was detected as an internal control. aP<0.05, aaP<0.01 vs. Control group; bP<0.05, bbP<0.01 vs. Mock group. TIMP-2, tissue inhibitor of metalloproteinase 2; MMP, matrix metalloproteinase.
Figure 5.
Overexpression of miR-195 inhibits the expression of metastasis-associated gene and the activation of the PI3K/AKT signaling pathway in Hec1A cells. (A-C) Relative mRNA levels of TIMP-2, MMP-2 and MMP-9 were detected by RT-qPCR. (D and E) Relative protein levels were measured by western blot analysis. (F-H) The protein and phosphorylation levels of PI3K and AKT were detected by western blot analysis. Each value represents the mean ± SEM (n=3). GAPDH was detected as an internal control. aaP<0.01 vs. Control group; bbP<0.01 vs. Mock group. TIMP-2, tissue inhibitor of metalloproteinase 2; MMP, matrix metalloproteinase.
Furthermore, we also measured the phosphorylation levels of PI3K and AKT. As shown in Figs. 4F-H and 5F-H, the protein levels of p-PI3K and p-AKT were markedly decreased in both the AN3-CA and Hec1A cells in which miR-195 was overexpressed, compared with those in the Control and Mock groups (P<0.01), indicating that the overexpression of miR-195 exerted its functional effects partly through the regulation of the PI3K/AKT signaling pathway.
GPER is a predicted target of miR-195
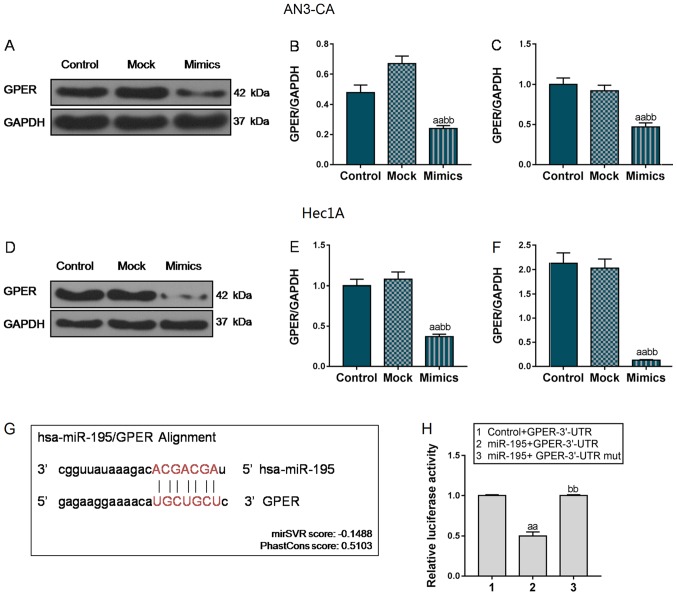
Following transfection with miR-195 overexpression vector, GPER mRNA and protein expression was markedly decreased reduced in the AN3-CA and Hec1A cells (P<0.01, Fig. 6A-F). According to the results, we hypothesized that miR-195 could specifically target GPER. To further verify the association between miR-195 and GPER, luciferase plasmids were compared and transfected into 293T cells. Using TargetScan prediction software, the GPER 3′-UTR was predicted to contain 7 nucleotides (5′-UGCUGCU-′3) that were completely paired with miR-195 (Fig. 6G). As shown in Fig. 6H, in the Control + GPER-3′-UTR group, the luciferase activity was significantly higher than that in the 293T cells co-transfected with miR-195 and GPER-3′-UTR plasmids. However, when miR-195 was co-transfected with the GPER-3′-UTR mut plasmid, the luciferase activity was notably enhanced, compared with that in the miR-195 + GPER-3′-UTR group (P<0.01). Collectively, these data indicate that miR-195 can directly regulate GPER expression.
Figure 6.
GPER was a predicted target of miR-195. In order to investigate mechanisms underlying the inhibition of EC progression by miR-195, we identified the potential target gene of miR-195 in EC cells. (A-C) The transcriptional and translational levels of GPER in AN3-CA cells were detected when cells were transfected with miR-195 mimics. (D-F) The changes in GPER mRNA and protein levels were detected in miR-195 overexpressed Hec1A cells. (G) The fragment of GPER 3′-UTR contains a binding site of miR-195. (H) Dual-luciferase reporter assay was used to reveal whether GPER was a potential target of miR-195. Each value represents the mean ± SEM (n=3). GAPDH was detected as an internal control. aaP<0.01 vs. Control or miR-195 + GPER-3′-UTR group; bbP<0.01 vs. Mock or miR-195 + GPER-3′-UTR mut group. GPER, G protein-coupled estrogen receptor 1.
Discussion
In the present study, we observed miR-195 overexpression exerted an inhibitory effect on the viability of AN3-CA and Hec1A cells, and induced the upregulation of TIMP-2 and the downregulation of MMP-2 and MMP-9 expression. miR-195 overexpression also suppressed the migratory and invasive abilities of the AN3-CA and Hec1A cells. Moreover, the suppression of the PI3K/AKT signaling pathway was found to contribute to the functional effects of miR-195. It was also found that GPER was a predicted target of miR-195. Hence, these findings suggest that the suppressive effects of miR-195 overexpression on the EMT process of EC cells are associated with the inhibition of PI3K/AKT signaling and GPER expression.
It is known that the EMT process is an essential step for the invasion and metastases formation of a tumor cell, as during this process, activated MMPs participate in the degradation of extracellular matrix elements, the reduction of cell adhesion and the promotion of neoplastic cell migration (25). TIMPs have been reported to function as natural inhibitors, of which TIMP-2 is the most effective inhibitor of MMP-2 than other TIMP members (26). miR-195, a member of miR-15, has been demonstrated to function as a tumor suppressor in a number of types of cancer (27–29). In 2013, miR-195 was reported to exert an inhibitory effect on the proliferation and development of endometrial stromal cells by blocking fractalkine expression (30). Moreover, Kong et al found that the restoration of miR-195 expression by blocking plasmacytoma variant translocation 1 (PVT1) not only inhibited the upregulation of cell proliferation, migration and invasion-associated proteins in both type I and II EC cell lines, but also induced EC tumor regression in vivo (31), which was consistent with the results of this study. It can thus be concluded that miR-195 overexpression can rebalance the expression of TIMP-2 and MMPs, which may protect the extracellular matrix from matrixins, and subsequently inhibit the migratory and invasive ability of AN3-CA and Hec1A cells, and ultimately contribute to impeding the progression of EMT in EC.
Furthermore, we also observed the significant downregulation of p-PI3K and p-AKT in the cells transfected with the miR-195 mimics vector. The PI3K/AKT pathway has been reported to participate in the metastasis of numerous cancer types (32,33). Kong et al revealed that both acidic fibroblast growth factor receptor (FGFR1) and basic fibroblast growth factor (FGF2) were effective targets of miR-195, and their inhibition by miR-195 suppressed the activities of the PI3K/AKT and MAPK/ERK pathways, ultimately resulting in the inhibition of malignancy in EC (31). In this study, miR-195 also exerted significantly inhibitory effects on the phosphorylation of PI3K and AKT, which indicated that the involvement of the PI3K/AKT signaling pathway may be required for the anticancer effects of miR-195 on EC development.
Previous studies have suggested that EC can be separated into two types (34); type I EC represents mostly low-grade endometrioid tumors driven by estrogen, and type II EC is categorized as estrogen non-dependent due to a lack of ER expression (35). However, Petrie et al revealed that Hec50 cells, a typical type II EC cell line, did not express the classical nERs, but GPER (17), indicating type II EC also strongly depended on estrogen in vivo that was attributed to GPER (17). In 2016, GPER was reported to promote ovarian cancer cell migration and invasion by upregulating the protein abundancy and functional activities of MMP-2 and MMP-9 (33). More importantly, it was also reported that GPER was involved in the regulation of several signaling pathways, including the MEK/ERK mitogen-activated protein kinase (MAPK) (36) and PI3K/AKT pathways (37). A previous study indicated that the activation of GPER notably enhanced the expression levels of MMPs and promoted cell migration and invasion in renal cell carcinoma (RCC) by regulating MAPK and PI3K/AKT; however, only the inhibitor of the PI3K/AKT pathway could effectively counteract the effects of GPER by downregulating the expression of MMPs and inhibiting the migratory and invasion ability of RCC cells (38,39). Therefore, miR-195 binds to the 3′-UTR of GPER and markedly reduces GPER protein levels and subsequently inhibits the positive effects of GPER on PI3K/AKT activation, leading to a significantly reduction in the levels of p-PI3K and p-AKT. Taken together, the results of this study indicate that the suppressive effects of miR-195 on EC cell migration and invasion are closely associated with the PI3K/AKT signaling pathway and GPER expression.
There are some limitations to this study. For example, although we have demonstrated miR-195 is able to directly target GPER, whether the negative correlation of miR-195 with GPER also applies to clinical EC samples remains to be investigated. This study found that the inhibitory effects of miR-195 on EC progression by targeting GPER involved the regulation of PI3K/AKT signaling; however, the detailed underlying mechanisms remain unclear. In addition, our data suggested that miR-195 overexpression notably suppressed the migration and invasion of EC cells through GPER; however, whether the knockdown or overexpression of GPER could cancel or enhance the suppressive effects of miR-195 on EC cells warrants further investigation.
In additions, some other limitations are that, for instance, although we have explored the association of miR-195 and the EMT process through the expression levels of MMPs and TIMPs, the effects of miR-195 on the expression levels of key EMT-related markers, including E-cadherin, Vimentin, FN and Slug need to be investigated.
In conclusion, this study found that the overexpression of miR-195 significantly suppressed cell migration and invasion, which may subsequently impede the EMT progress of EC cells, and that the PI3K/AKT signaling pathway was involved in this process. In addition, GPER was identified as a target of miR-195. Based on the essential role of GPER in type II EC cell development, we hypothesized that it also played an important role in the suppressive effects of miR-195 on EC progression; however, the underlying molecular mechanisms remain to be further elucidated. This study suggests that miR-195 may prove to be a promising therapeutic target for the treatment of EC.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
JD and WW made substantial contributions to the conception and design of the study, and were also involved in the drafting of the article or critically revising it for important intellectual content. GY was involved in data acquisition, data analysis and interpretation. XM was involved in all experiments. All authors have read and approved the final version of the manuscript. All authors agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of the work are appropriately investigated and resolved.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Zhang G, Cheng Y, Zhang Q, Li X, Zhou J, Wang J, Wei L. ATX-LPA axis facilitates estrogen-induced endometrial cancer cell proliferation via MAPK/ERK signaling pathway. Mol Med Rep. 2018;17:4245–4252. doi: 10.3892/mmr.2018.8392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Jemal A. Cancer Statistics, 2017. CA Cancer J Clin. 2017;67:7–30. doi: 10.3322/caac.21387. [DOI] [PubMed] [Google Scholar]
- 3.Ward KK, Shah NR, Saenz CC, McHale MT, Alvarez EA, Plaxe SC. Cardiovascular disease is the leading cause of death among endometrial cancer patients. Gynecol Oncol. 2012;126:176–179. doi: 10.1016/j.ygyno.2012.04.013. [DOI] [PubMed] [Google Scholar]
- 4.Elit L, Hirte H. Novel strategies for systemic treatment of endometrial cancer. Expert Opin Investig Drugs. 2000;9:2831–2853. doi: 10.1517/13543784.9.12.2831. [DOI] [PubMed] [Google Scholar]
- 5.Dong P, Kaneuchi M, Konno Y, Watari H, Sudo S, Sakuragi N. Emerging therapeutic biomarkers in endometrial cancer. Biomed Res Int. 2013;2013:130362. doi: 10.1155/2013/130362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gong B, Yue Y, Wang R, Zhang Y, Jin Q, Zhou X. Overexpression of microRNA-194 suppresses the epithelial-mesenchymal transition in targeting stem cell transcription factor Sox3 in endometrial carcinoma stem cells. Tumour Biol. 2017;39:1010428317706217. doi: 10.1177/1010428317706217. [DOI] [PubMed] [Google Scholar]
- 7.Oyanadel C, Holmes C, Pardo E, Retamal C, Shaughnessy R, Smith P, Cortés P, Bravo-Zehnder M, Metz C, Feuerhake T, et al. Galectin-8 induces partial epithelial-mesenchymal transition with invasive tumorigenic capabilities involving a FAK/EGFR/proteasome pathway in Madin-Darby canine kidney cells. Mol Biol Cell. 2018;29:557–574. doi: 10.1091/mbc.E16-05-0301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Meng X, Kong DH, Li N, Zong ZH, Liu BQ, Du ZX, Guan Y, Cao L, Wang HQ. Knockdown of BAG3 induces epithelial-mesenchymal transition in thyroid cancer cells through ZEB1 activation. Cell Death Dis. 2014;5:e1092. doi: 10.1038/cddis.2014.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Mitra A, Mishra L, Li S. EMT, CTCs and CSCs in tumor relapse and drug-resistance. Oncotarget. 2015;6:10697–10711. doi: 10.18632/oncotarget.4037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Huszar M, Pfeifer M, Schirmer U, Kiefel H, Konecny GE, Ben-Arie A, Edler L, Munch M, Muller-Holzner E, Jerabek-Klestil S, et al. Up-regulation of L1CAM is linked to loss of hormone receptors and E-cadherin in aggressive subtypes of endometrial carcinomas. J Pathol. 2010;220:551–561. doi: 10.1002/path.2673. [DOI] [PubMed] [Google Scholar]
- 11.Kent CN, Guttilla Reed IK. Regulation of epithelial-mesenchymal transition in endometrial cancer: Connecting PI3K, estrogen signaling, and microRNAs. Clin Transl Oncol. 2016;18:1056–1061. doi: 10.1007/s12094-016-1492-2. [DOI] [PubMed] [Google Scholar]
- 12.Prossnitz ER, Barton M. The G-protein-coupled estrogen receptor GPER in health and disease. Nat Rev Endocrinol. 2011;7:715–726. doi: 10.1038/nrendo.2011.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Molina L, Figueroa CD, Bhoola KD, Ehrenfeld P. GPER-1/GPR30 a novel estrogen receptor sited in the cell membrane: Therapeutic coupling to breast cancer. Expert Opin Ther Targets. 2017;21:755–766. doi: 10.1080/14728222.2017.1350264. [DOI] [PubMed] [Google Scholar]
- 14.Zhu P, Liao LY, Zhao TT, Mo XM, Chen GG, Liu ZM. GPER/ERK&AKT/NF-κB pathway is involved in cadmium-induced proliferation, invasion and migration of GPER-positive thyroid cancer cells. Mol Cell Endocrinol. 2017;442:68–80. doi: 10.1016/j.mce.2016.12.007. [DOI] [PubMed] [Google Scholar]
- 15.Yan Y, Jiang X, Zhao Y, Wen H, Liu G. Role of GPER on proliferation, migration and invasion in ligand-independent manner in human ovarian cancer cell line SKOV3. Cell Biochem Funct. 2015;33:552–559. doi: 10.1002/cbf.3154. [DOI] [PubMed] [Google Scholar]
- 16.Zhang L, Li Y, Lan L, Liu R, Wu Y, Qu Q, Wen K. Tamoxifen has a proliferative effect in endometrial carcinoma mediated via the GPER/EGFR/ERK/cyclin D1 pathway: A retrospective study and an in vitro study. Mol Cell Endocrinol. 2016;437:51–61. doi: 10.1016/j.mce.2016.08.011. [DOI] [PubMed] [Google Scholar]
- 17.Petrie WK, Dennis MK, Hu C, Dai D, Arterburn JB, Smith HO, Hathaway HJ, Prossnitz ER. G protein-coupled estrogen receptor-selective ligands modulate endometrial tumor growth. Obstet Gynecol Int. 2013;2013:472720. doi: 10.1155/2013/472720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.He YY, Cai B, Yang YX, Liu XL, Wan XP. Estrogenic G protein-coupled receptor 30 signaling is involved in regulation of endometrial carcinoma by promoting proliferation, invasion potential, and interleukin-6 secretion via the MEK/ERK mitogen-activated protein kinase pathway. Cancer Sci. 2009;100:1051–1061. doi: 10.1111/j.1349-7006.2009.01148.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Smith HO, Leslie KK, Singh M, Qualls CR, Revankar CM, Joste NE, Prossnitz ER. GPR30: A novel indicator of poor survival for endometrial carcinoma. Am J Obstet Gynecol. 2007;196:386.e1–e9.e11. doi: 10.1016/j.ajog.2007.01.004. [DOI] [PubMed] [Google Scholar]
- 20.Fan DX, Yang XH, Li YN, Guo L. 17β-estradiol on the expression of G-protein coupled estrogen receptor (GPER/GPR30) mitophagy, and the PI3K/Akt signaling pathway in ATDC5 chondrocytes in vitro. Med Sci Monit. 2018;24:1936–1947. doi: 10.12659/MSM.909365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang L, Zhao Y, Guo L. 17β-estradiol protects INS-1 insulinoma cells from mitophagy via G protein-coupled estrogen receptors and the PI3K/Akt signaling pathway. Int J Mol Med. 2018;41:2839–2846. doi: 10.3892/ijmm.2018.3470. [DOI] [PubMed] [Google Scholar]
- 22.Bhaskaran M, Mohan M. MicroRNAs: History, biogenesis, and their evolving role in animal development and disease. Vet Pathol. 2014;51:759–774. doi: 10.1177/0300985813502820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tsukamoto O, Miura K, Mishima H, Abe S, Kaneuchi M, Higashijima A, Miura S, Kinoshita A, Yoshiura K, Masuzaki H. Identification of endometrioid endometrial carcinoma-associated microRNAs in tissue and plasma. Gynecol Oncol. 2014;132:715–721. doi: 10.1016/j.ygyno.2014.01.029. [DOI] [PubMed] [Google Scholar]
- 24.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 25.Drzewiecka-Jędrzejczyk M, Wlazeł R, Terlecka M, Jabłoński S. Serum metalloproteinase-2 and tissue inhibitor of metalloproteinase-2 in lung carcinoma patients. J Thorac Dis. 2017;9:5306–5313. doi: 10.21037/jtd.2017.11.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Bourboulia D, Stetler-Stevenson WG. Matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs): Positive and negative regulators intumor cell adhesion. Semin Cancer Biol. 2010;20:161–168. doi: 10.1016/j.semcancer.2010.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Tang T, Shan G, Zeng F. Knockdown of DGCR5 enhances the radiosensitivity of human laryngeal carcinoma cells via inducing miR-195. J Cell Physiol. 2019;234:12918–12925. doi: 10.1002/jcp.27958. [DOI] [PubMed] [Google Scholar]
- 28.Yu W, Liang X, Li X, Zhang Y, Sun Z, Liu Y, Wang J. MicroRNA-195: A review of its role in cancers. Onco Targets Ther. 2018;11:7109–7123. doi: 10.2147/OTT.S183600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang J, Li W. Long noncoding RNA CYTOR sponges miR-195 to modulate proliferation, migration, invasion and radiosensitivity in nonsmall cell lung cancer cells. Biosci Rep. 2018;38(pii):BSR20181599. doi: 10.1042/BSR20181599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang Y, Chen H, Fu Y, Ai A, Xue S, Lyu Q, Kuang Y. MiR-195 inhibits proliferation and growth and induces apoptosis of endometrial stromal cells by targeting FKN. Int J Clin Exp Pathol. 2013;6:2824–2834. [PMC free article] [PubMed] [Google Scholar]
- 31.Kong F, Ma J, Yang H, Yang D, Wang C, Ma X. Long non-coding RNA PVT1 promotes malignancy in human endometrial carcinoma cells through negative regulation of miR-195-5p. Biochim Biophys Acta Mol Cell Res. 2018 Jul 19; doi: 10.1016/j.bbamcr.2018.07.008. doi: 10.1016/j.bbamcr.2018.07.008 (Epub ahead of print) [DOI] [PubMed] [Google Scholar]
- 32.Segarra J, Balenci L, Drenth T, Maina F, Lamballe F. Combined signaling through ERK, PI3K/AKT, and RAC1/p38 is required for met-triggered cortical neuron migration. J Biol Chem. 2006;281:4771–4778. doi: 10.1074/jbc.M508298200. [DOI] [PubMed] [Google Scholar]
- 33.Tian PC, Wang HL, Chen GH, Luo Q, Chen Z, Wang Y, Liu YF. 2,2′,4,4′-Tetrabromodiphenyl ether promotes human neuroblastoma SH-SY5Y cells migration via the GPER/PI3K/Akt signal pathway. Hum Exp Toxicol. 2016;35:124–134. doi: 10.1177/0960327115578974. [DOI] [PubMed] [Google Scholar]
- 34.Ulrich LS. Endometrial cancer, types, prognosis, female hormones and antihormones. Climacteric. 2011;14:418–425. doi: 10.3109/13697137.2010.550974. [DOI] [PubMed] [Google Scholar]
- 35.Geletina NS, Kobelev VS, Babayants EV, Feng L, Pustylnyak VO, Gulyaeva LF. PTEN negative correlates with miR-181a in tumour tissues of non-obese endometrial cancer patients. Gene. 2018;655:20–24. doi: 10.1016/j.gene.2018.02.051. [DOI] [PubMed] [Google Scholar]
- 36.Yin H, Zhu Q, Liu M, Tu G, Li Q, Yuan J, Wen S, Yang G. GPER promotes tamoxifen-resistance in ER+ breast cancer cells by reduced Bim proteins through MAPK/Erk-TRIM2 signaling axis. Int J Oncol. 2017;51:1191–1198. doi: 10.3892/ijo.2017.4117. [DOI] [PubMed] [Google Scholar]
- 37.Li Y, Birnbaumer L, Teng CT. Regulation of ERRalpha gene expression by estrogen receptor agonists and antagonists in SKBR3 breast cancer cells: Differential molecular mechanisms mediated by g protein-coupled receptor GPR30/GPER-1. Mol Endocrinol. 2010;24:969–980. doi: 10.1210/me.2009-0148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.He YY, Du GQ, Cai B, Yan Q, Zhou L, Chen XY, Lu W, Yang YX, Wan XP. Estrogenic transmembrane receptor of GPR30 mediates invasion and carcinogenesis by endometrial cancer cell line RL95-2. J Cancer Res Clin Oncol. 2012;138:775–783. doi: 10.1007/s00432-011-1133-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Guan BZ, Yan RL, Huang JW, Li FL, Zhong YX, Chen Y, Liu FN, Hu B, Huang SB, Yin LH. Activation of G protein coupled estrogen receptor (GPER) promotes the migration of renal cell carcinoma via the PI3K/AKT/MMP-9 signals. Cell Adh Migr. 2018;12:109–117. doi: 10.4161/19336918.2014.990781. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.