Abstract
Objective:
A major limitation of comparing the efficacy of videostroboscopy (VS) and high-speed videoendoscopy (HSV) is the lack of an objective reference by which to compare the functional assessment ratings of the two techniques. For patients with vocal fold mass lesions, intraoperative measures of lesion size and depth may serve as this objective reference. This study compared the relationships between the pre- to postoperative change in VS and HSV visual-perceptual ratings to intraoperative measures of lesion size and depth.
Design:
Prospective visual-perceptual study with intraoperative measures of lesion size and depth.
Methods:
VS and HSV samples were obtained preoperatively and postoperatively from 28 patients with vocal fold lesions and from 17 vocally-healthy controls. Two experienced clinicians rated amplitude, mucosal wave, vertical phase difference, left-right phase asymmetry, and vocal fold edge on a visual-analog scale using both imaging techniques. The change in perioperative ratings from VS and HSV was compared between groups and correlated to intraoperative measures of lesion size and depth.
Results:
HSV was as reliable as VS for ratings of amplitude and edge, and substantially more reliable for ratings of mucosal wave and left-right phase asymmetry. Both VS and HSV had mild-moderate correlations between change in perioperative ratings and intraoperative measures of lesion area. Change in function could be obtained in more patients and for more parameters using HSV than VS. Group differences were noted for postoperative ratings of amplitude and edge; however, these differences were within one level of the visual-perceptual rating scale. The presence of asynchronicity in VS recordings renders vibratory features either uninterpretable or potentially distorted and thus should not be rated.
Conclusions:
Amplitude and edge are robust vibratory measures for perioperative functional assessment, regardless of imaging modality. HSV is indicated for evaluation of subepithelial lesions or if asynchronicity is present in the VS image sequence.
1. Introduction
Laryngologists and speech pathologists have access to a variety of diagnostic techniques to directly and indirectly assess phonatory function;1 however, visualization of vocal fold vibratory characteristics during phonation is necessary for a complete assessment of tissue health and pliability. This information is critical for accurate diagnosis2 and best informs treatment options, including therapeutic and surgical interventions. Several laryngeal imaging techniques are available to assess vocal fold vibratory function, including videostroboscopy (VS),3 videokymography,4–7 and high-speed videoendoscopy (HSV).8–10 Further, HSV yields multiple playback techniques to facilitate detailed analysis, including phonovibrograms,11 digital kymography (DKG),12,13 stroboscopy derived from HSV,9,14 and mucosal wave kymography.15–17
VS is the clinical gold standard for laryngeal imaging, which synchronizes a stroboscopic light source to the speaker’s fundamental frequency to sample images of consecutive phases of vibration across multiple glottal cycles. The resulting image sequence provides a slow-motion illusion of vocal fold vibration which provides functional information regarding glottal closure patterns, amplitude of vibration and mucosal wave.18 Studies comparing VS to indirect laryngoscopy showed that the added functional data provided by VS was not only salient in up to one-third of patients, but also resulted in a change in diagnosis or clinical outcome in up to 14% of patients.19–23 Currently, VS is the ubiquitous functional laryngeal imaging tool in laryngology clinics;24 however, for patients with even moderate levels of dysphonia, technological limitations inherent in the sampling methodology prevent accurate visualization of vocal fold vibratory function.25 Specifically, the acoustic signal is perturbed to such an extent that the fundamental frequency cannot be calculated,26 the strobe is unable to accurately predict the next phase of the vibratory cycle, and the result is a series of images that do not follow the sequential phases of the glottal cycle. The resulting asynchronous image sequence violates one of the two main principles of stroboscopy, the perception of apparent motion from sampled images,27 and may not represent the underlying vibratory cycle. This sampling limitation has been estimated to affect between 17% and 63% of patients with voice disorders in general,21,25 and up to 58% of patients with epithelial or subepithelial lesions in particular.25
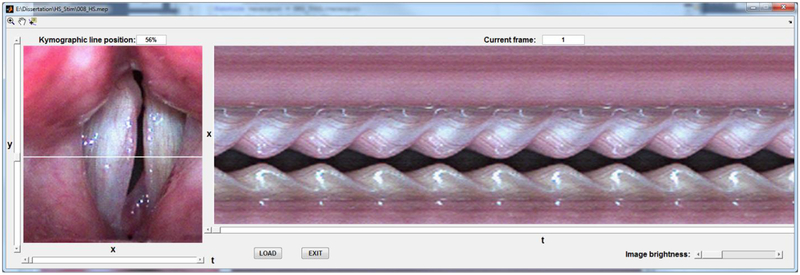
High-speed photography has been used to visualize the vocal folds since the late 1930s28,29 but has received significant attention in the past two decades with the development of HSV. HSV captures images of the vocal folds independent of the acoustic signal, thereby overcoming the sampling methodology limitation inherent to VS.9 As long as systems provide a capture rate of at least 5,333 frames per second (fps), HSV provides enough intra-cycle information to facilitate accurate clinical assessment of vibratory function using visual perception.30 Currently, HSV can also be dynamically interlinked with DKG. The combination of these two playback techniques takes advantage of DKG’s ability to show subtle changes in vibratory function over time by presenting multiple vibratory cycles within a single static image (Figure 1, right), while also giving the DKG line scans spatial context by simultaneously presenting the sequence of full HSV images (Figure 1, left). This dynamically interlinked HSV+DKG playback provides maximal information for the analysis of temporally-sensitive vibratory features. Multiple studies have reported that the increased temporal resolution of HSV provides valuable information regarding the presence and magnitude of mucosal wave and phase asymmetry,15–17,31,32 particularly when diplophonia or aperiodicity is present.10,33–35 Despite these findings, technical, methodological, and practical challenges related to storing and processing large data files for interpretation have limited the duration of phonation samples which can be collected and evaluated. In addition to these limitations, the ongoing scientific and clinical research evidence on the efficacy of HSV has not yet reached the point of developing a well-integrated HSV protocol. As a result, HSV is still viewed as a technique complementary to videostroboscopy. Therefore, the equipment costs associated with maintaining a second imaging system, the lack of standardized clinical protocols, and the inability to bill for additional services have precluded widespread implementation of HSV into clinical use.9
Figure 1:
Custom software for dynamically interlinked HSV+DKG playback. The left side displays the full HSV frames, which can be scrolled through consecutively along the time axis; the right side displays the DKG line scan, which can be scrolled through along the posterior-anterior axis. Raters were instructed to select the DKG line scan at the point of maximum amplitude along the anterior-posterior axis to perform ratings.
The relative advantages and disadvantages of each imaging technique have led to a series of comparison studies. Olthoff, Woywod, & Kruse highlighted the methodological advantages of HSV to provide substantially more interpretable data than VS.36 These findings were corroborated by Patel, Dailey, & Bless, who reported that for patients with mass lesions, HSV was superior to VS in mapping the tissue regions affected by the lesion.25 The relative diagnostic value of using these imaging techniques on patient care has also been investigated with varying results. Mortenson & Woo provided a case study of a patient with intermittent voice loss whose stroboscopic examination was grossly normal, but HSV revealed the vocal folds were vibrating at slightly different frequencies leading to a diplophonic voice quality.37 In contrast, Mendelsohn, and colleagues reported that high-speed imaging did not significantly improve the accuracy of diagnosis over VS alone in 28 patients.38 However, Zacharias et al. reported that HSV did provide salient diagnostic information sufficient to alter diagnosis or course of treatment in 7% of 151 adult patients with a variety of voice disorders.39
A major limitation of these studies is the lack of a truly objective standard by which to compare the two techniques. The imaging systems are simply compared without establishing the efficacy of either technique to accurately assess vibratory function. However, for patients with vocal fold mass lesions, an objective standard for comparison exists in the form of lesion measurements from calibrated intraoperative images. Vocal fold mass lesions are the most common type of vocal fold pathology for individuals with dysphonia.40 Hirano’s body-cover theory proposes that the integrity of the structured layers of the vocal folds directly relates to the vibratory patterns,41 and disruption of these delicate layers of the vocal folds due to trauma or other disease process produces mass lesions. The anatomical result of these lesions is typically an increase in the mass and stiffness of the vocal folds; the functional result is a decrease in the laryngeal system’s ability to modulate the airflow due to altered glottal closure patterns and inefficient vibratory function. These altered anatomical and functional characteristics ultimately contribute to a degraded acoustic output, perceived as dysphonia.42,43 Phonomicrosurgery exists on the theoretical foundation that excising the lesion will help restore the architectural microstructure of the vocal folds, and as a result, tissue pliability, vibratory function, and vocal quality will improve.44 In theory, the larger the lesion, the greater the expected change (i.e. improvement) in vibratory function following phonomicrosurgery. Therefore, correlating change in ratings of vibratory function following phonomicrosurgery to intraoperative measures of lesion size and depth (epithelial vs subepithelial) would contribute to the growing body of evidence for the efficacy of these laryngeal imaging techniques.
The purpose of this study was to determine whether the increased temporal resolution of HSV+DKG would allow for more accurate and reliable clinical ratings of vocal fold vibratory function before and after surgical intervention compared to VS. It was hypothesized that visual-perceptual ratings of vibratory function would be more reliably rated using HSV+DKG image sequences than VS image sequences. It was also hypothesized that the change in perioperative HSV+DKG ratings would correlate to intraoperative measures of lesion size and depth (epithelial vs subepithelial) better than the change in VS ratings.
2. Methods
2.1. Participants
Twenty-eight adults with dysphonia (15 male/13 female, ages 17–90, mean=46 years) and 17 vocally-healthy speakers (7 male/10 female, ages 24–65, mean=41 years) participated in this study. Patients were recruited from the Massachusetts General Hospital Voice Center (Boston, MA), and a normative control group of vocally-healthy participants was recruited from Charlotte, Eye, Ear, Nose and Throat Associates (Charlotte, NC). All patients underwent microlaryngeal surgery to remove unilateral or bilateral vocal fold mass lesions. Surgery was performed under general anesthesia using cold instruments and/or 532-nm pulsed potassium titanyl phosphate (KTP) laser photoblation (for malignant lesions). Patients were recorded using both VS and HSV during their preoperative appointment and again during their postoperative follow-up, an average of 3.5 weeks after surgery. The normative control group was recorded using VS and HSV during one session only.
2.2. Instrumentation and Post-Processing
2.2.1. High-Speed Videoendoscopy Recordings
HSV examinations were performed using a color Phantom v7.3 high-speed camera by Vision Research, (Vision Research, Inc., Wayne, NJ) equipped with a 300-W xenon light source (Model 7152A, PENTAX Medical Company, Montvale, NJ) containing 3 glass infrared filters for thermal energy reduction. HSV sampling rates were 6,250 fps for patients and 5,512.5 fps for the normative group, both of which exceeded the recommended minimum recommended frame rate for visual-perceptual assessment of vibratory function.30 The camera was set at maximum integration time. The camera was coupled to a 70-degree 10-mm Rigid Laryngoscopes (Model 49–4072, JEDMED Instrument Co, St. Louis, MO) with a resulting spatial resolution of 320 horizontal × 352 vertical pixels. HSV samples were selected using automated segmentation based on tracking the dispersion of the fundamental frequency contour as computed from the glottal area waveform.9 This technique identified the most stable 160-ms segment in each phonation sample in order to minimize the effect of changing frequency on vocal fold vibratory characteristics and exclude any onsets, offsets, or phonatory breaks. Following segmentation, a subpixel technique was applied to compensate for endoscopic motion during data collection and render the glottis fixed in a vertical position.12 HSV recordings were then upsampled to 17,500 fps and interpolated to a 420 × 380 pixel spatial resolution. Using previously-developed custom software (MathWorks, Natick, MA), the middle 750 frames (43 ms) of the HSV images were dynamically interlinked with the corresponding DKG line scans along the anterior-posterior axis. This HSV+DKG graphic user interface (as seen in Figure 1) allowed users to visualize both the full HSV spatial image sequence over time by scrolling along the time axis (left-right) and the full DKG anterior-posterior line scan image sequence at any point along the glottis by scrolling along the posterior-anterior axis (top-bottom). Raters were instructed, after reviewing the full HSV and DKG image sequences, to set the DKG line scan for the point of maximum amplitude along the anterior-posterior axis and used information from both the full HSV image sequence (Figure 1, left) and the DKG line scan (Figure 1, right) to perform ratings. A minimum of 3 vibratory cycles were displayed for analysis.
2.2.2. Videostroboscopy Recordings
VS examinations were performed using a KayPENTAX digital stroboscopy system (RLS 9100B, PENTAX Medical, Montvale, NJ) with a 120-W xenon light source. The camera was connected to a 70-degree transoral rigid endoscope (Model 9106, PENTAX Medical, Montvale, NJ). VS recordings were manually reviewed and a representative 2–4 second sample of habitual pitch (3–6 vibratory cycles) excluding onset and offset of phonation was selected from each recording. Recordings were saved as .avi files with a default playback speed of 30 fps; however, users had the ability to slow down the playback speed or advance frame by frame, per their preference. Although video with synchronized audio playback is feasible with VS, it is not feasible with HSV; therefore, all recordings from both imaging modalities were evaluated without audio.
2.3. Visual-Perceptual Ratings of Vocal Fold Vibratory Features
Two certified speech-language pathologists, each with at least 3 years of experience using VS, were asked to rate the following clinically-salient vibratory features using visual-analog scales: amplitude of vibration, mucosal wave, left-right phase asymmetry, and vocal fold edge.3 Additionally, using categorical scales, the raters were asked to determine the presence or absence of a vertical phase difference between the upper and lower edges of the vocal folds during the closing phase of vibration. Information regarding the development of these scales are presented elsewhere.14 In brief, raters underwent approximately three hours of consensus training, which resulted in operative definitions for each vibratory feature, as well as anatomically-defined thresholds for each level within the feature-specific scale. These feature-specific scales were developed based on the vibratory features outlined in the Stroboscopy Evaluation Rating Form (SERF);45 however, they were tailored to assess specific vibratory features common to patients with vocal fold mass lesions, while providing adequate precision to allow for comparison of pre- and postoperative ratings. Table 1 presents details for each feature-specific scale.
Table 1:
Vocal fold vibratory features were rated based on feature definitions and scale levels.
| Feature | Definition | Rating Scale | Range of Scalar Level |
|---|---|---|---|
| Mucosal Wave | Vocal fold deformation along the lateral plane during the opening phase |
75-pt visual-analog scale
in % with 6 anchoring
points Absent---20%---35%---50%---65%---≥80% |
15 percentage points |
| Amplitude | Maximum lateral excursion of each vocal fold from the most medial position |
75-pt visual-analog scale
in % with 6 anchoring
points Absent---20%---35%---50%---65%--->80% |
15 percentage points |
|
Left-Right Phase Asymmetry |
Phase difference between left and right vocal fold vibration |
100-pt visual-analog scale
with 4 anchoring
points Absent: left and right vocal folds reach both maximal amplitude and the midline at the same time Mild: one vocal fold lags slightly behind the other either during the lateral-to-medial or medial-to-lateral transition Moderate: one vocal fold has already transitioned and begun to move medially before the other vocal fold has hit maximal amplitude Severe: one vocal fold has hit midline while the other vocal fold is at maximal amplitude |
33 points |
| Vocal Fold Edge | Irregularity of the vibrating vocal fold edges |
100-pt visual-analog scale
with 4 anchoring
points Regular: Vocal fold edge is straight with a sharp superior edge Mildly irregular: Vocal fold edge may be rounded and/or a mild swelling may be present. A sulcus may be present on the medial edge of the fold. Glottal closure may still be complete or a small anterior or posterior gap may be present. Moderately irregular: Vocal fold edge is clearly nonlinear, but does not impede the vibratory function of the contralateral fold. Glottal closure may form an hour-glass configuration. Severely irregular: Vocal fold edge is non-linear with a large protrusion, or multiple areas of irregularity. The lesion may impede the vibratory function of the contralateral fold. |
33 points |
| Vertical Phase Difference | Vocal fold deformation along the vertical plane during the closing phase |
Dichotic
choice Present: inferior edge of the vocal fold is visible during closing Absent: inferior edge of the vocal fold is not visible during closing |
1 point |
The total number of unique recordings was 146: 73 VS recordings (28 preoperative, 28 postoperative, and 17 normal controls) and 73 HSV+DKG recordings (28 preoperative, 28 postoperative, and 17 normal controls). These recordings were compiled into two experimental sets (VS and HSV+DKG). Ten percent redundancy (8 recordings) was added, resulting in 81 recordings in the VS experimental set and 81 recordings in the HSV+DKG experimental set. All recordings were de-identified and randomized. Raters were instructed to rate all vibratory features within a single experimental set before starting the second experimental set. Experiments were also counterbalanced across raters: Rater 1 rated HSV+DKG then VS, whereas Rater 2 rated VS then HSV+DKG.
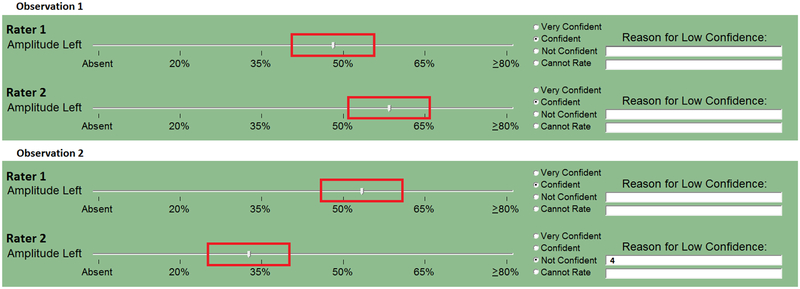
Ratings were collected using custom-designed software for the Alvin2 environment (Figure 2).46 In addition to rating the vibratory features using the visual analog or categorical scales, raters were also instructed to indicate their internal level of confidence for their ratings using a 4-point scale: (1) very confident, (2) confident, (3) not confident, or (4) cannot rate. If raters selected “not confident” or “cannot rate”, they were instructed to provide one or more reasons for this lack of confidence.
Figure 2:
A priori established range for agreement: +/− 1/2 scalar level. In Observation 1, the ranges of error do overlap; therefore, these ratings would be considered “in agreement.” In Observation 2, the ranges of error between raters do not overlap; therefore, these ratings would not be considered “in agreement.” Note Rater 2’s lack of confidence is indicated to the right.
2.4. Calibrated Intraoperative Images of Lesions
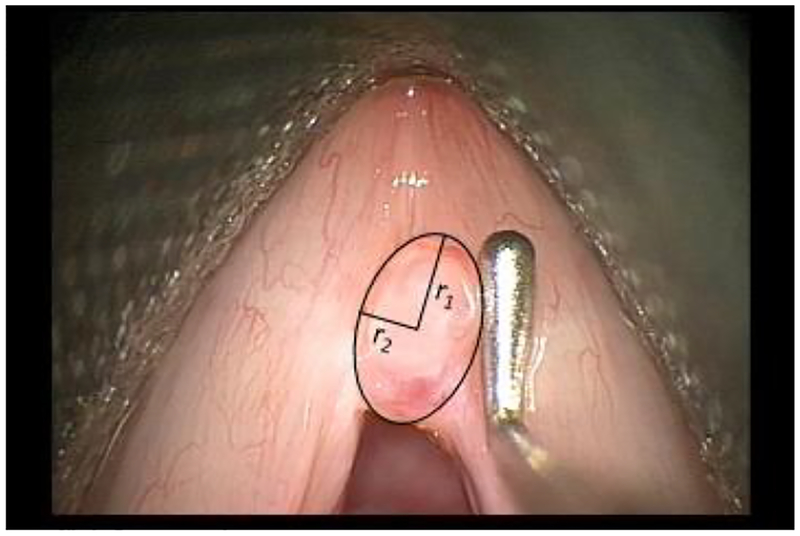
Intraoperative images of vocal fold lesions were captured using ImageJ software (ImageJ 1.33u, NIH). Frames were selected from the intraoperative video recordings showing the lesion next to a calibrated right-angle probe (Figure 3). From these frames, the anterior-posterior length and medio-lateral width were measured to the 1/100 mm using a Cobalt electronic caliper (model #53247). Total area (TA) in mm2 was calculated by using the standard area formula most appropriate for the lesion’s geometric shape. Lesions were measured as an ellipse: TA = πr1r2, half ellipse: rectangle: TA = bh, or a combination of these, where r1=radius along the anterior-posterior axis, r2=radius along the medio-lateral axis, b=length of lesion along anterior-posterior axis, and h=width of lesion along the medio-lateral axis. Lesion depth (epithelial vs subepithelial lesion) was determined by postoperative diagnosis obtained from the patient’s medical record (Table 2).
Figure 3:
Image capture from the intraoperative recording showing the lesion next to the calibrated right-angle probe. Length (along the anterior-posterior axis) and width (along the medio-lateral axis) were measured using a Cobalt micrometer caliper. Total area for this lesion was calculated based on the standard algebraic equation for an ellipse:
Table 2:
Description and distribution of lesion types included in the calibrated intraoperative images.
| Lesion Depth | Diagnosis | Count |
|---|---|---|
| Epithelial | Polyp | 7 |
| Epithelial | Hemorrhagic polyp | 7 |
| Epithelial | Mucoid polyp | 1 |
| Epithelial | Phonotraumatic vocal fold lesion | 1 |
| Subepithelial | Fibrovascular mass | 2 |
| Subepithelial | Subepithelial mass with multiple varices and ectasias | 2 |
| Subepithelial | Fibrovascular contact lesion | 1 |
| Subepithelial | Hemorrhagic cyst | 1 |
| Subepithelial | Keratin cyst, fibrovascular polyp, and residual sulcus | 1 |
| Subepithelial | Phonotraumatic vocal fold lesion | 1 |
| Subepithelial | T1 glottic cancer | 1 |
2.5. Statistical Analysis
Intra- and inter-rater agreement was calculated as a percent agreement, where ratings were considered “in agreement” if they fell within +/− 1/2 the range of one level of the feature-specific scale. See Table 1 for details on feature-specific scales. For variables resulting in continuous data (mucosal wave, amplitude, left-right phase asymmetry, and vocal fold edge), inter-rater reliability was calculated using both Spearman’s ρ and the intraclass correlation coefficient (ICC). For categorical, “vertical phase difference” data, inter-rater reliability was calculated using Cohen’s κ.
Group differences were tested using a linear mixed effects model. Tukey’s post hoc analysis was calculated to analyze all possible combinations of means, with an alpha level of 0.05. Correlations between vibratory features and intraoperative measures of lesion size were calculated using Spearman’s ρ. Intraoperative images were only available for 20 of the 28 patients; therefore, these correlations were conducted on this subset of data. Correlations were reported as mild with ρ>0.25, moderate with ρ>0.50, strong with ρ>0.70, and very strong with ρ>0.90, and considered statistically significant with p<0.05.
3. Results
A total of 1458 ratings were attempted for each imaging modality. Table 3 shows the breakdown of confidence ratings for each vibratory feature using VS or HSV+DKG. Raters indicated confidence in ratings for 54.3% of VS observations and 78.5% of HSV+DKG ratings. Conversely, 14.5% of VS observations, and 2.95% of HSV+DKG observations were unable to be rated. Any individual observations for which the raters indicated “Cannot Rate” were excluded from the following statistical analyses and utilized for descriptive purposes only.
Table 3:
Percentage (%) breakdown of confidence ratings for each vibratory feature. Total number of observations for each vibratory feature is 162, and the total number of observations for each imaging modality is 1,458.
| Vibratory Feature | VS | HSV+DKG | ||||
|---|---|---|---|---|---|---|
| Confident % (n) | Not Confident % (n) | Cannot Rate % (n) | Confident % (n) | Not Confident % (n) | Cannot Rate % (n) | |
| Left Mucosal Wave | 44 (71) | 36 (59) | 20 (32) | 60 (98) | 35 (56) | 5 (8) |
| Right Mucosal Wave | 49 (79) | 32 (52) | 19 (31) | 71 (115) | 26 (42) | 3 (5) |
| Left Amplitude | 54 (88) | 38 (61) | 8 (13) | 78 (126) | 22 (35) | 1 (1) |
| Right Amplitude | 54 (87) | 38 (61) | 9 (14) | 85 (138) | 14 (23) | 1 (1) |
| Left-Right Phase Asymmetry | 42 (68) | 31 (51) | 27 (43) | 72 (117) | 20 (33) | 7 (12) |
| Left Vocal Fold Edge | 64 (104) | 28 (46) | 7 (12) | 81 (132) | 16 (26) | 2 (4) |
| Right Vocal Fold Edge | 60 (97) | 33 (54) | 7 (11) | 85 (138) | 14 (22) | 1 (2) |
| Left Vertical Phase Difference | 65 (105) | 19 (30) | 17 (27) | 86 (140) | 10 (17) | 3 (5) |
| Right Vertical Phase Difference | 57 (92) | 26 (42) | 17 (28) | 87 (141) | 10 (16) | 3 (5) |
| Total % (n) | 54.3 (791) | 31.3 (456) | 14.5 (211) | 78.5 (1145) | 18.5 (270) | 2.9 (43) |
VS=videostroboscopy; HSV+DKG=high-speed videoendoscopy dynamically interlinked with digital kymography
3.1. Rater Agreement and Reliability
Intra-rater agreement for Rater 1 ranged from 50%–100% for VS and 80%–100% for HSV+DKG. Intra-rater agreement for Rater 2 ranged from 83%–100% for VS and 63%–100% for HSV+DKG (see Table 4 for feature-specific intra-rater agreement results). Inter-rater agreement (Table 5) was high for all parameters rated using HSV+DKG, ranging from 84%–100%. Inter-rater agreement for parameters rated using VS ranged from 60%–94%.
Table 4:
Intra-rater agreement was calculated as % agreement within +/− 1/2 the range of one level of the scale. For example, ratings of mucosal wave must have been within 15 points of each other (the range of 1 level of the scale) to be considered “in agreement.”
| Vibratory Feature | Range | Rater | ||
|---|---|---|---|---|
| VS | HSV+DKG | |||
| Left Mucosal Wave | 15 pts | 1 2 |
75 100 |
71 86 |
| Right Mucosal Wave | 15 pts | 1 2 |
50 83 |
88 100 |
| Left Amplitude | 15 pts | 1 2 |
100 100 |
100 100 |
| Right Amplitude | 15 pts | 1 2 |
100 100 |
100 100 |
| Left-Right Phase Asymmetry | 33 pts | 1 2 |
67 100 |
100 100 |
| Left Vocal Fold Edge | 33 pts | 1 2 |
100 100 |
100 100 |
| Right Vocal Fold Edge | 33 pts | 1 2 |
100 100 |
100 100 |
| Left Vertical Phase Difference | 1 pt | 1 2 |
100 100 |
100 75 |
| Right Vertical Phase Difference | 1 pt | 1 2 |
100 86 |
88 63 |
VS=videostroboscopy; HSV+DKG=high-speed videoendoscopy dynamically interlinked with digital kymography
Table 5:
Inter-rater agreement and reliability using videostroboscopy and high-speed videoendoscopy dynamically interlinked with digital kymography. Agreement is reported as the percentage of direct agreement within the range of one level of the feature-specific scale. Reliability is reported by Spearman correlations, which are statistically significant at p<0.0001 (*), Cohen’s Kappa (κ), and the intraclass correlation as well as the 95% confidence intervals for κ and the ICC.
| Vibratory Feature | % Agree (n) | Spearman’s p | ICC (95% CI) | |||
|---|---|---|---|---|---|---|
| VS | HSV+DKG | VS | HSV+DKG | VS | HSV+DKG | |
| Left Mucosal Wave | 61 | 84 | 0.72* | 0.85* | 0.31 (0.10–0.49) | 0.60 (0.45–0.71) |
| Right Mucosal Wave | 60 | 91 | 0.84* | 0.90* | 0.55 (0.37–0.68) | 0.84 (0.77–0.88) |
| Left Amplitude | 96 | 94 | 0.84* | 0.81* | 0.57 (0.41–0.69) | 0.47 (0.30–0.60) |
| Right Amplitude | 96 | 96 | 0.86* | 0.84* | 0.64 (0.50–0.74) | 0.63 (0.49–0.73) |
| Left-Right Phase Asymmetry | 86 | 90 | 0.70* | 0.77* | 0.33 (0.10–0.51) | 0.60 (0.45–0.71) |
| Left Vocal Fold Edge | 93 | 100 | 0.75* | 0.86* | 0.58 (0.42–0.71) | 0.53 (0.37–0.65) |
| Right Vocal Fold Edge | 92 | 99 | 0.72* | 0.84* | 0.51 (0.34–0.64) | 0.57 (0.42–0.68) |
| Cohen’s κ (95% CI) | ||||||
| Left Vertical Phase Difference | 88 | 87 | -- | -- | 0.12 (0.05–0.24) | 0.08 (0.04–0.21) |
| Right Vertical Phase Difference | 85 | 88 | -- | -- | 0.06 (0.01–0.15) | 0.07 (0.05–0.32) |
VS=videostroboscopy; HSV+DKG=high-speed videoendoscopy dynamically interlinked with digital kymography; ICC=intraclass correlation; CI=confidence interval
Inter-rater reliability (Table 5), measured using Spearman’s ρ, showed strong to very strong correlations between HSV+DKG ratings for mucosal wave, amplitude, left-right phase asymmetry, and vocal fold edge, with ρ values ranging from 0.77–0.90. Spearman’s ρ for VS ratings ranged from 0.70– 0.86. ICCs for ratings made using HSV+DKG ranged from 0.47–0.84, and ICCs for ratings made using VS ranged from 0.31–0.64. Vertical phase difference was found to have acceptable levels of agreement using strict percent agreement (between 85% and 88%); however, this particular parameter involves a simple dichotic choice (present/absent), thus high levels of agreement would be expected simply by chance. Kappa values for vertical phase indicated very low levels of reliability when chance was taken into account (0.06–0.12). Therefore, vertical phase difference was determined to be unreliably rated using both VS and HSV+DKG and was excluded from additional analysis.
3.2. Functional Outcomes Derived from Perioperative Ratings
Table 6 details the mean differences in feature ratings between normal control, preoperative, and postoperative groups. Ratings for normal controls differed significantly from preoperative patients for all vibratory features except left mucosal wave using VS and left-right phase asymmetry using HSV+DKG (Table 6, Normal vs PreOp). Postoperatively, HSV+DKG ratings for patients were significantly improved for left/right amplitude and left/right vocal fold edge. In comparison, VS ratings for patients improved for left vocal fold edge only (Table 6, PreOp vs PostOp). Finally, postoperatively, patient ratings were significantly different from normal controls for all vibratory features except left mucosal wave and left-right phase asymmetry using VS and left amplitude and left-right phase asymmetry using HSV+DKG (Table 6 Normal vs PostOp).
Table 6:
Statistical results from the group comparison using videostroboscopy and high-speed videoendoscopy dynamically interlinked with digital kymography for: normal vs PreOp, PreOp vs PostOp, and PostOp vs normal. The values presented are the differences between the means within each pair of groups. Positive Mean Δ values for mucosal wave and amplitude, and negative Mean Δ values for asymmetry and edge, indicate better function in the first group comparing to the second. Tukey’s adjusted p values are statistically significant at p<0.05 (*).
| Vibratory Measure | Imaging Technique | Normal vs PreOp | PreOp vs PostOp | Normal vs PostOp | |||
|---|---|---|---|---|---|---|---|
| Mean Δ | p Value | Mean A | p Value | Mean Δ | p Value | ||
| Left Mucosal Wave | VS | 13 | 0.2134 | −4 | 0.9623 | 10 | 0.5647 |
| HSV+DKG | 28 | 0.0009* | −3 | 0.9903 | 25 | 0.0037* | |
| Right Mucosal Wave | VS | 23 | 0.0066* | −3 | 0.9269 | 20 | 0.0489* |
| HSV+DKG | 36 | <0.0001* | −6 | 0.5393 | 29 | <0.0001* | |
| Left Amplitude | VS | 12 | <0.0001* | −6 | 0.0752 | 6 | 0.0269* |
| HSV+DKG | 17 | <0.0001* | −12 | <0.0001* | 5 | 0.1254 | |
| Right Amplitude | VS | 12 | <0.0001* | −3 | 0.1506 | 9 | 0.0002* |
| HSV+DKG | 14 | <0.0001* | −8 | <0.0001* | 7 | 0.0172* | |
| Left Right Phase Asymmetry | VS | −17 | 0.0366* | 13 | 0.0759 | −4 | 0.8918 |
| HSV+DKG | −14 | 0.1524 | 6 | 0.6196 | −8 | 0.7277 | |
| Left Vocal Fold Edge | VS | −36 | <0.0001* | 10 | 0.0262* | −28 | <0.0001* |
| HSV+DKG | −39 | <0.0001* | 19 | <0.0001* | −21 | <0.0001* | |
| Right Vocal Fold Edge | VS | −32 | <0.0001* | 6 | 0.4083 | −26 | <0.0001* |
| HSV+DKG | −35 | <0.0001* | 17 | <0.0001* | −18 | 0.0004* | |
VS=videostroboscopy; HSV+DKG= high-speed videoendoscopy dynamically interlinked with digital kymography; PreOp=preoperative ratings; PostOp=postoperative ratings; Mean Δ=difference between the means
3.3. Relationship of Perioperative Outcomes to Intraoperative Measures of Lesion Size
Eight of the 28 patients did not have calibrated intraoperative images of the vocal folds and were therefore excluded from this portion of the study. Five of the 20 remaining patients presented with bilateral mass lesions; therefore, the total number of individual vocal folds analyzed for this portion of the study was 25. Sixteen vocal fold masses were classified as epithelial lesions, and 9 masses were classified as subepithelial lesions (Table 2). Lesion sizes ranged from 0.47 mm2 to 32.04 mm2. Table 7 shows the Spearman correlations between the change in perioperative functional ratings made using HSV+DKG and VS and both the anterior-posterior length and total area of the lesion. The amount of change in perioperative ratings (perioperative outcomes) from both VS and HSV+DKG showed stronger correlations with total area of the lesion compared to the anterior-posterior lesion length. For both imaging modalities, changes in mucosal wave, left-right phase asymmetry, and vocal fold edge ratings showed statistically significant mild-to-moderate correlations to the total area of the lesion.
Table 7:
Spearman correlations between the calculated change in feature rating pre- to postoperative and the length and total area of the lesion. Correlations were considered mild with ρ>0.25, moderate with ρ>0.50, strong with ρ>0.70, and very strong with ρ>0.90. Findings are statistically significant at p<0.05 (*).
| Vibratory Feature | VS Spearman p |
HSV+DKG Spearman p |
|
|---|---|---|---|
| Anterior-Posterior Length (mm) | Mucosal Wave | −0.45* | −0.52* |
| Amplitude | −0.07 | −0.11 | |
| Left-Right Phase Asymmetry | −0.44* | −0.37* | |
| Vocal Fold Edge | −0.19 | −0.22 | |
| Total Area (mm2) | Mucosal Wave | −0.51* | −0.48* |
| Amplitude | −0.24 | 0.05 | |
| Left-Right Phase Asymmetry | −0.53* | −0.60* | |
| Vocal Fold Edge | −0.37* | −0.37* |
VS=videostroboscopy; HSV+DKG=high-speed videoendoscopy dynamically interlinked with digital kymography
4. Discussion
This study sought to compare the efficacy of VS and HSV+DKG imaging modalities to facilitate accurate and reliable visual-perceptual ratings of vibratory function before and after surgical intervention to remove vocal fold mass lesions. In the current study, more vibratory features could be rated using HSV+DKG (~97%) compared to VS (~86%). Conversely, approximately 3% of HSV+DKG observations and approximately 14% of VS observations could not be rated (Table 3). This finding is similar to those reported by Woo et al.21 but is substantially lower than those reported by Patel et al.25 If a vibratory feature cannot be rated both before and after surgery, the change in functional ratings—or perioperative outcome—cannot be obtained; clinicians are therefore unable to determine how and to what extent vibratory function was changed due to surgical or therapeutic intervention. Due to these unrateable observations, perioperative outcomes could not be obtained for one or more vibratory features in 46% of patients using VS (13/28) and 11% of patients using HSV (3/28).
Additional descriptive analysis of the reasons raters “Cannot Rate” specific vibratory features revealed that the majority of the VS samples that could not be rated were due to the presence of asynchronicity in the image sequence (84% (n=178), data not shown). Asynchronicity is a technical limitation unique to VS resulting from its inability to extract the fundamental frequency from overly-perturbed acoustic signals and synchronize the image capture to the consecutive phases of the underlying vibratory cycle. Multiple studies suggest this methodological limitation represents a significant clinical issue.21,25,39 Continuously asynchronous image sequences should not be subjected to interpretation because they represent a violation of one of the two main principles of VS, the perception of apparent motion from sampled images.29 In the current study, left-right phase asymmetry outcomes could not be obtained from VS recordings in 43% of patients (12/28) due to the presence of asynchronicity in either preoperative or postoperative conditions. Mucosal wave outcomes could not be determined in 36% of patients (10/28); amplitude outcomes could not be determined in approximately 18% of patients (5/28); and edge outcomes could not be determined in 11% of patients (3/28) for the same reason.
For HSV+DKG, the primary reason samples could not be rated was due to the vocal folds being obscured by an anatomical structure, precluding visualization of the target feature (74% (n=32), data not shown). Left-right phase asymmetry and mucosal wave were most affected by unrateable observations; outcomes for these vibratory features could not be obtained in 11% of patients (3/28) due to anatomical structures obscuring vocal fold vibration. Amplitude outcomes could not be determined in 4% of patients (1/28), and edge outcomes could not be determined in 7% of patients (2/28) due to anatomical obstructions as well.
The key difference between vibratory features which could not be rated using VS and HSV+DKG is that with VS, the primary reason was due to asynchronous image sequences, which provide little information regarding the underlying laryngeal function. In contrast, anatomical obstruction was the leading cause for unrated HSV+DKG images, which is an objective physical presentation by the patient and is inherent to rigid endoscopy regardless of the imaging modality.
4.1. Rater Agreement and Reliability
Spearman coefficients fell above the top end of the 95% confidence intervals for the ICC for all rated parameters, suggesting that there were systematic differences in the way both raters used the same scale. However, percent agreement was high for amplitude, mucosal wave, left-right phase asymmetry, and edge using VS, and very high to near perfect for these parameters using HSV+DKG, indicating that the raters were calibrated to each other within the error of the scale (+/− 1/2 scalar level-see Table 1 for details of feature-specific scales). These three reliability measures, taken together suggest that raters were able to reliably differentiate between scalar levels (severity levels), but they lack calibration within the level of the scale. Future studies may benefit from the application of well-defined and standardized thresholds for severity levels for feature-specific scales47 in order to further improve inter-rater reliability.48
Given these considerations for interpreting reliability, several clinically-significant findings emerge. The reliability of visual perceptual ratings of amplitude and vocal fold edge were similar for both HSV+DKG and VS. However, the ICC’s 95% confidence intervals for left and right mucosal wave and left-right phase asymmetry show minimal overlap between VS and HSV+DKG, indicating that these vibratory features are more reliably rated using HSV+DKG than VS. Thus, HSV+DKG was found to be as reliable as VS for visual perceptual ratings of amplitude and vocal fold edge and measurably more reliable for ratings of mucosal wave and left-right phase asymmetry.
Vertical phase difference was not found to be a reliably rated feature using either imaging modality. The presence of a vertical phase difference (related to divergent angle) during the closing phase of the vibratory cycle has been shown to increase the maximum flow declination rate during vocal fold closing,49 resulting in increased energy in the higher harmonics and increased intensity of the acoustic output as a whole.50,51 Despite the potential clinical significance of this vibratory feature, clinicians do not typically include vertical phase in their standard assessment. Thus, at the time of this study, even identifying the presence or absence of a vertical phase difference was a novel task, and raters frequently indicated decreased confidence differentiating mucosal wave and vertical phase using both VS and HSV+DKG. It is possible that with refinement of feature definition and scale criteria, along with further training to identify the presence/absence of vertical phase difference, this vibratory feature may eventually be more reliably rated using both imaging modalities.
The relative novelty of the HSV+DKG playback for rating these vibratory features should also be considered when interpreting reliability findings. Nawka and Konerding found that variability in reliability related to the rater’s experience using VS plateaued after 2 years.52 While both raters in this study had at least 2 years’ experience using VS, they had substantially less experience rating these specific vibratory features using HSV and even less using DKG. This lack of experience rating HSV+DKG did affect rater confidence. Specifically, raters noted that for mucosal wave, amplitude and vertical phase difference, the features occasionally presented differently depending on whether they were looking at the DKG line scan or the full HSV image sequence. Given the significant differences in the process for interpreting VS versus HSV+DKG, it is likely that HSV+DKG will require modality-specific training, and experience rating with VS will not generalize to HSV+DKG for all parameters.
4.2. Functional Outcomes Derived from Perioperative Ratings
Ratings for vibratory features were compared between groups (normal control, preoperative, and postoperative) to determine differences between a) normal controls and patients in the preoperative condition, b) patients pre-and postoperatively, and c) normal controls and patients in the postoperative condition (Table 6). The classic trajectory for patients undergoing surgical removal of vocal fold mass lesions would be that they present preoperatively with significant differences in vibratory function compared to normal controls. Following surgery, they show significant improvement, such that postoperatively they are no longer significantly different from normal controls. Lack of statistical significance in the third case would suggest that following surgery, patients resembled their vocally-healthy counterparts.
VS ratings revealed significant differences between the normative group and patients in the preoperative condition for all parameters except left mucosal wave and were also able to show a statistically significant improvement preoperative to post for left vocal fold edge. Left vocal fold edge ratings improved by less than 1/2 scalar (or severity) level, and following surgery, patients were still distinguishable from the normative group by an average of 28 points on the visual-analog scale, meaning the average postoperative rating fell within the mild severity range.
HSV+DKG ratings differentiated normal controls and preoperative patients for all parameters except left-right phase asymmetry and were also able to show improvement following surgery for both left and right vocal fold edge and left and right amplitude. Edge improved by a mean of 16–18 points (approximately 1/2 scalar level, or the equivalent of an improvement from severe to moderately severe, moderate to mild-moderate, etc.), and was not statistically significantly different from normal controls following surgery, meaning the average rating fell within the range of normal. Amplitude increased by 14–17 percentage points (pp), or an increase in lateral movement equivalent to 14%−17% of the width of the vocal fold. Following surgery left amplitude was not statistically distinguishable from the normative group, showing that for this feature, postoperative vocal function was similar to that of their vocally healthy counterparts. The underlying cause for the persistent difference in right amplitude after surgery is unknown and warrants further investigation; however, current findings are consistent with previous reports of greater right arytenoid asymmetries in patients with voice disorders.53
These findings indicate that HSV+DKG ratings showed greater change in function following phonomicrosurgery (perioperative outcomes) for more parameters (including left and right amplitude and left and right vocal fold edge) compared to VS ratings (left vocal fold edge only). Group comparisons for these two vibratory features indicated that VS ratings of vocal fold edge were rated 9 points more severe on the visual analog scale postoperatively compared to HSV+DKG ratings. VS ratings of amplitude were also an average of 6 pp (6% of the width of the vocal fold) lower than HSV+DKG ratings postoperatively. Although these differences in group means are statistically significant, the clinical significance of these findings translates to differences within one level (severity level) of the rating scale.
4.3. Relationship of Perioperative Outcomes to Intraoperative Measures of Lesion Size
Change in functional ratings made using both VS and HSV+DKG correlated better to the lesion’s total area than to the lesion’s length. Although this finding is peripheral to the focus of the current study, it is clinically salient because lesion sizes are traditionally reported by the unidimensional length or width of the lesion, rather than the two-dimensional area of the lesion.54 Theoretically, total area is a better representation of the potential mass effect on vibratory function than length or width alone. Although measuring the area of the lesion by hand has been found to be less reliable than measuring length or width, technological advances with calibrated endoscopes offer increased precision and accuracy in measuring lesion area55 and current findings indicate that lesion area may be a better clinical predictor of change in vibratory function following phonomicrosurgery, regardless of the imaging modality used for functional ratings.
Both imaging modalities showed similar mild-moderate correlations between change in perioperative visual-perceptual ratings of vocal fold function and intraoperative measures of lesion area. The timing of the postoperative evaluation may provide insight to these results. Patients’ postoperative evaluations occurred approximately 3.5 weeks following surgery. Branski, et al. found that in rabbit models, elevated levels of the biochemical marker PGE-2 at 21 days post phonotrauma suggest ongoing contraction of the wound, as well as continued resolution of inflammatory processes.56 Therefore, it is possible that healing is ongoing and edema may still be present at 3.5 weeks, reducing amplitude and mucosal wave. This ongoing healing process would likely be more pronounced with larger lesions and lesions embedded in the subepithelium or deeper towards the vocal ligament, which required more extensive dissection.42,56–58 Ongoing healing could reasonably explain why larger lesions showed little improvement in amplitude and mucosal wave, while smaller lesions showed greater gains. With both imaging modalities, larger lesions did show improvement in vocal fold edge beyond the error of the scale, suggesting that vocal fold edge is a better outcome measure immediately following surgery, particularly for larger lesions. Amplitude or mucosal wave may be better outcome measures for smaller lesions, or for larger lesions once edema has resolved.
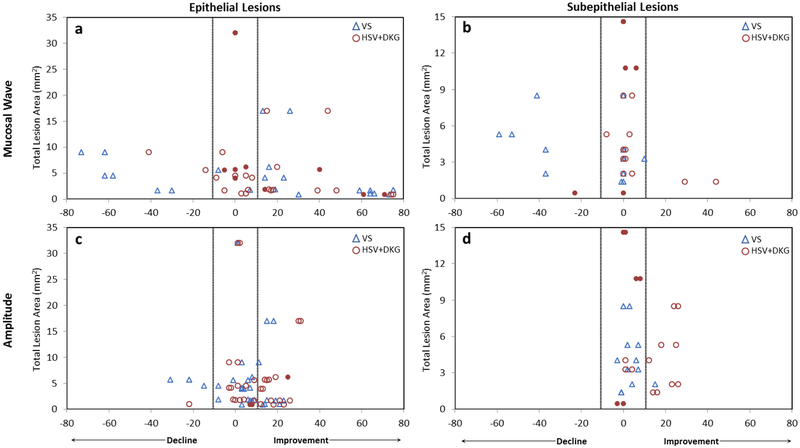
Post-hoc comparison of the scatter plots for these correlations reveals clinically salient differences in ratings specifically related to lesion depth (Figures 4–5). Figure 4a–b shows the post-hoc comparison of the change in mucosal wave ratings from VS and HSV+DKG. For both epithelial and subepithelial lesions, HSV+DKG more frequently showed no effective change (change less than one scalar level, or 15 pp) or improvement greater than one scalar level, whereas VS frequently showed either no effective change or a reduction in mucosal wave (greater than 15 pp) following lesion removal. Only one HSV+DKG rating showed a decrease in mucosal wave greater than 15 pp. In this instance, both raters had confidence in their ratings, and yet they differed by 23 pp for this individual, indicating poor reliability for these ratings. The VS ratings which showed a decrease in mucosal wave greater than 15 pp represented 7 lesioned vocal folds rated by one or more raters: 3 epithelial lesions and 4 subepithelial lesions (Figure 4a–b). Five of these 7 vocal folds (71%) were rated with low confidence for either preoperative or postoperative conditions due to the presence of intermittent asynchronicity in the recording. The HSV+DKG ratings for these same patients showed effectively no change in mucosal wave. These findings suggest that not only does the presence of asynchronicity render features uninterpretable in some instances, but it also has the potential to distort vibratory features, resulting in inaccurate/unreliable visual-perceptual ratings using VS.
Figure 4:
Scatterplots showing the relationship between lesion size and change in functional ratings for mucosal wave (a,b) and amplitude (c,d) following phonomicrosurgery. Epithelial (a,c) and subepithelial lesions (b,d) are presented separately. Positive numbers on the x-axis indicate an increase in function following surgery, negative numbers indicate a decrease in function following surgery, and vertical lines indicate the effective error of the visual-perceptual scale. Solid symbols represent patients measured using high-speed dynamically interlinked with digital kymography line scans (HSV+DKG) that could not be measured using videostroboscopy (VS).
Figure 5:
Scatterplots showing the relationship between lesion size and change in functional ratings for left-right phase asymmetry (a,b) and vocal fold edge (c,d) following phonomicrosurgery. Epithelial (a,c) and subepithelial lesions (b,d) are presented separately. Negative numbers on the x-axis indicate an improvement in function following surgery, positive numbers indicate a decline in function following surgery, and vertical lines indicate the effective error of the visual-perceptual scale. Solid symbols represent patients measured using high-speed dynamically interlinked with digital kymography line scans (HSV+DKG) that could not be measured using videostroboscopy (VS).
As with mucosal wave, amplitude ratings (Figure 4c–d) from HSV+DKG image sequences showed either no effective change or improvement in postoperative function, whereas perioperative ratings from VS image sequences more frequently showed no effective change. Four subepithelial lesions showed improvements of greater than 15 pp using HSV+DKG, whereas only one lesion showed improvement greater than the error of the scale using VS. These HSV+DKG ratings were highly reliable (as shown by the close proximity or overlap of ratings) and were made with confidence (Figure 4d). One epithelial lesion that was rated by both raters as showing decreased amplitude by greater than 15 pp (using VS) was rated with low confidence due to the presence of intermittent asynchronicity in the recording (Figure 4c). The HSV+DKG ratings for the same patient showed no effective change in amplitude. Another epithelial lesion showed a decrease in amplitude greater than 15 pp using HSV+DKG. Ratings for this lesion were unreliable (raters differed by ~30 pp), and raters indicated decreased confidence in their ratings due to poor image quality. This finding suggests that ratings performed with low confidence should be interpreted cautiously, regardless of which imaging modality is used.
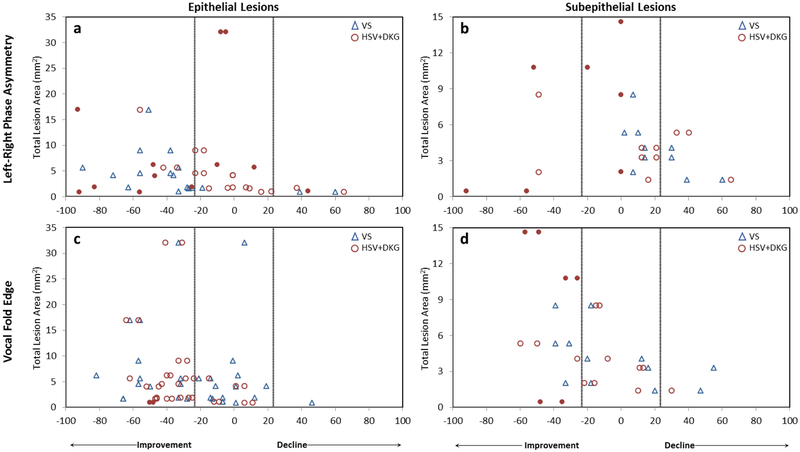
As shown in Figure 5a–b, when rated using VS, epithelial lesions showed a decrease in asymmetry (improvement in function greater than one scalar level, or 33 points), whereas subepithelial lesions showed increased left-right phase asymmetry (decline in function). However, this trend was not mirrored in change measured using HSV+DKG. Substantial improvements in left-right phase asymmetry ratings were noted on several subepithelial lesions, and the patients who showed the greatest amount of change when rated using HSV+DKG were unable to be rated for either preoperative or postoperative conditions using VS (indicated as “●” in the HSV+DKG scatterplot in Figure 5b).
Vocal fold edge showed the most change in ratings as a function of lesion size using both VS and HSV+DKG image sequences (Figure 5c–d). Using HSV+DKG, three subepithelial lesions showed improvement of at least one scalar or severity level for vocal fold edge; importantly, vocal fold edge could not be rated for either preoperative or postoperative conditions for two of these patients using VS (indicated as “●” in the HSV+DKG scatterplot in Figure 5d). Additionally, the reason why the change in function for the patient with the small epithelial lesion (Figure 5c) could not be measured using VS was due to asynchronous images in the postoperative condition. The consistent findings that HSV+DKG produced interpretable image sequences for subepithelial lesions when VS could not suggest that HSV+DKG may be more appropriate for the evaluation of subepithelial lesions.
4.4. Limitations of the Study
Although great care was taken to obtain VS and HSV recordings under similar conditions, the phonation samples obtained using each technique are different. Therefore, it is possible that the differences noted are due to phonation inter-sample differences rather than imaging technique. Additionally, the sample duration represented by VS and HSV+DKG are significantly different. VS image sequences presented 2–4 seconds of phonation, whereas the HSV+DKG image sequences presented only 43 ms. Thus, the HSV+DKG samples may not capture intermittent vibratory irregularities over time, which are more easily captured using VS. There is currently no literature to quantify the effect of sample duration on vibratory assessment; however, since stroboscopy can be derived from HSV,14 future studies should investigate the extent to which sample duration and stroboscopic sampling alters visual-perceptual ratings of the underlying true vibratory function.
5. Conclusion
HSV+DKG resulted in more ratings for more vibratory features than VS. As a result, more participants could be evaluated both preoperatively and postoperatively using HSV+DKG, and therefore, perioperative vibratory outcomes could be obtained for more participants and more features using HSV+DKG compared to VS. HSV+DKG was found to be as reliable as VS for visual perceptual ratings of amplitude and vocal fold edge and significantly more reliable for ratings of mucosal wave and left-right phase asymmetry. When VS image sequences are synchronous, VS and HSV+DKG amplitude and edge ratings are likely to be similar (within one level of the rating scale). Thus, amplitude and edge are robust vibratory measures for perioperative assessment, regardless of imaging modality. The presence of asynchronicity in VS recordings is the defining methodological limitation of this imaging technique. Continuously or intermittently asynchronous image sequences render vibratory features to be either uninterpretable or potentially distorted, and ratings should not be attempted. This methodological limitation impacted VS ratings of subepithelial lesions more than epithelial lesions; therefore, if a subepithelial lesion is suspected, and/or asynchronicity is present in the VS recording, HSV+DKG assessment is indicated. Together, these findings add to the growing body of evidence supporting the efficacy of HSV along with DKG for the evaluation of patients with voice disorders and better define the respective roles of VS and HSV+DKG within the clinical battery.
6. Acknowledgements
This study was fully supported by the National Institute of Health – NIDCD: R01-DC007640 “Efficacy of Laryngeal High-Speed Videoendoscopy.” The authors wish to thank Dr. Stephanie Zacharias and Resmi Gupta for their contributions to the execution of the study.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Portions of this study have been presented at the 44th Symposium of The Voice Foundation: Care of the Professional Voice. Philadelphia, PA, May 26–31, 2015.
7. References
- 1.Hillman RE, Montgomery WW, Zeitels SM. Appropriate use of objective measures of vocal function in the multidisciplinary management of voice disorders. Curr Opin Otolaryngol Head Neck Surg. 1997;5(3):172. [Google Scholar]
- 2.Paul BC, Chen S, Sridharan S, Fang Y, Amin MR, Branski RC. Diagnostic accuracy of history, laryngoscopy, and stroboscopy. The Laryngoscope. 2013;123(1):215–219. [DOI] [PubMed] [Google Scholar]
- 3.Hirano M, Bless DM. Videostroboscopic Examination of the Larynx. San Diego, Calif: Singular; 1993. [Google Scholar]
- 4.Švec JG, Schutte HK. Videokymography: High-speed line scanning of vocal fold vibration. J Voice. 1996;10(2):201–205. [DOI] [PubMed] [Google Scholar]
- 5.Švec J, Šram F, Schutte HK. Videokymography: A new high-speed method for the examination of vocal-fold vibrations. Otorinolaryngol Foniatr. 1999;48:155–162. [Google Scholar]
- 6.Qiu Q, Schutte HK. A new generation videokymography for routine clinical vocal fold examination. The Laryngoscope. 2006;116(10):1824–1828. [DOI] [PubMed] [Google Scholar]
- 7.Švec JG, Šram F, Schutte HK. Videokymography in voice disorders: What to look for? Ann Otol Rhinol Laryngol. 2007;116(3):172–180. [DOI] [PubMed] [Google Scholar]
- 8.Hertegård S What have we learned about laryngeal physiology from high-speed digital videoendoscopy? Curr Opin Otolaryngol Head Neck Surg. 2005;13(3):152–156. [DOI] [PubMed] [Google Scholar]
- 9.Deliyski DD, Petrushev PP, Bonilha HS, Gerlach TT, Martin-Harris B, Hillman RE. Clinical implementation of laryngeal high-speed videoendoscopy: Challenges and evolution. Folia Phoniatr Logop. 2008;60(1):33–44. [DOI] [PubMed] [Google Scholar]
- 10.Eysholdt U, Tigges M, Wittenberg T, Pröschel U. Direct evaluation of high-speed recordings of vocal fold vibrations. Folia Phoniatr Logop. 1996;48(4):163–170. [DOI] [PubMed] [Google Scholar]
- 11.Lohscheller J, Eysholdt U. Phonovibrogram visualization of entire vocal fold dynamics. The Laryngoscope. 2008;118(4):753–758. [DOI] [PubMed] [Google Scholar]
- 12.Deliyski DD. Endoscope motion compensation for laryngeal high-speed videoendoscopy. J Voice. 2005;19(3):485–496. [DOI] [PubMed] [Google Scholar]
- 13.Deliyski DD. Laryngeal high-speed videoendoscopy In: Kendall KA, Leonard RJ, eds. Laryngeal Evaluation: Indirect Laryngoscopy to High-Speed Digital Imaging. Thieme; 2010:245–270. [Google Scholar]
- 14.Powell ME, Deliyski DD, Hillman RE, Zeitels SM, Burns JA, Mehta DD. Comparison of videostroboscopy to stroboscopy derived from high-speed videoendoscopy for evaluating patients with vocal fold mass lesions. Am J Speech Lang Pathol. 2016;25(4):576–589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bonilha HS, Deliyski DD. Period and glottal width irregularities in vocally normal speakers. J Voice. 2008;22(6):699–708. [DOI] [PubMed] [Google Scholar]
- 16.Bonilha HS, Deliyski DD, Whiteside JP, Gerlach TT. Vocal fold phase asymmetries in patients with voice disorders: A study across visualization techniques. Am J Speech Lang Pathol. 2012;21(1):3–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shaw HS, Deliyski DD. Mucosal wave: A normophonic study across visualization techniques. J Voice. 2008;22(1):23–33. [DOI] [PubMed] [Google Scholar]
- 18.Hillman RE, Mehta DD. The science of stroboscopic imaging In: Kendall KA, Leonard RJ, eds. Laryngeal Evaluation: Indirect Laryngoscopy to High-Speed Digital Imaging. Thieme; 2010. [Google Scholar]
- 19.Sataloff RT, Spiegel JR, Carroll LM, Schiebel B-R, Darby KS, Rulnick R. Strobovideolaryngoscopy in professional voice users: Results and clinical value. J Voice. 1988;1(4):359–364. [Google Scholar]
- 20.Kitzing P Stroboscopy: A pertinent laryngological examination. J Otolaryngol. 1985;14(3):151–157. [PubMed] [Google Scholar]
- 21.Woo P, Colton R, Casper J, Brewer D. Diagnostic value of stroboscopic examination in hoarse patients. J Voice. 1991;5(3):231–238. [Google Scholar]
- 22.Casiano RR, Zaveri V, Lundy DS. Efficacy of Videostroboscopy in the Diagnosis of Voice Disorders. Otolaryngol Neck Surg. 1992;107(1):95–100. doi: 10.1177/019459989210700115 [DOI] [PubMed] [Google Scholar]
- 23.Printza A, Triaridis S, Themelis C, Constantinidis J. Stroboscopy for benign laryngeal pathology in evidence based health care. Hippokratia. 2012;16(4):324–328. [PMC free article] [PubMed] [Google Scholar]
- 24.Mehta DD, Hillman RE. Current role of stroboscopy in laryngeal imaging. Curr Opin Otolaryngol Head Neck Surg. 2012;20(6):429–436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Patel R, Dailey S, Bless D. Comparison of high-speed digital imaging with stroboscopy for laryngeal imaging of glottal disorders. Ann Otol Rhinol Laryngol. 2008;117(6):413–424. [DOI] [PubMed] [Google Scholar]
- 26.Titze IR. Workshop on Acoustic Voice Analysis. Denver, CO: National Center for Voice and Speech; 1994. [Google Scholar]
- 27.Mehta DD, Deliyski DD, Hillman RE. Commentary on why laryngeal stroboscopy really works: Clarifying misconceptions surrounding Talbot’s Law and the Persistence of Vision. J Speech Lang Hear Res. 2010;53(5):1263–1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Herriott W, Farnsworth DW. High speed motion pictures of the vocal cords. J Acoust Soc Am. 1938;9(3):274–274. [Google Scholar]
- 29.Farnsworth D High-speed motion picture of the human vocal cords. Bell Lab Rec. 1940;18:203–208. [Google Scholar]
- 30.Deliyski DD, Powell MEG, Zacharias SRC, Gerlach TT, de Alarcon A. Experimental investigation on minimum frame rate requirements of high-speed videoendoscopy for clinical voice assessment. Biomed Signal Process Control. 2015;17:21–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Krausert CR, Olszewski AE, Taylor LN, McMurray JS, Dailey SH, Jiang JJ. Mucosal wave measurement and visualization techniques. J Voice. 2011;25(4):395–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Eysholdt U, Rosanowski F, Hoppe U. Vocal fold vibration irregularities caused by different types of laryngeal asymmetry. Eur Arch Otorhinolaryngol. 2003;260(8):412–417. [DOI] [PubMed] [Google Scholar]
- 33.Kiritani S, Hirose H, Imagawa H. High-speed digital image analysis of vocal cord vibration in diplophonia. Speech Commun. 1993;13(1):23–32. [Google Scholar]
- 34.Wittenberg T, Tigges M, Mergell P, Eysholdt U. Functional imaging of vocal fold vibration: Digital multislice high-speed kymography. J Voice. 2000;14(3):422–442. [DOI] [PubMed] [Google Scholar]
- 35.Hertegård S, Larsson H, Wittenberg T. High-speed imaging: Applications and development. Logoped Phoniatr Vocol. 2003;28(3):133–139. [DOI] [PubMed] [Google Scholar]
- 36.Olthoff A, Woywod C, Kruse E. Stroboscopy versus high-speed glottography: A comparative study. The Laryngoscope. 2007;117(6):1123–1126. [DOI] [PubMed] [Google Scholar]
- 37.Mortensen M, Woo P. High-speed imaging used to detect vocal fold paresis: A case report. Ann Otol Rhinol Laryngol. 2008;117(9):684–687. [DOI] [PubMed] [Google Scholar]
- 38.Mendelsohn A, Remacle M, Courey MS, Gerhard F, Postma GN. The Diagnostic Role of High-Speed Vocal Fold Vibratory Imaging. J Voice. 2013;27(5):627–631. [DOI] [PubMed] [Google Scholar]
- 39.Zacharias SR, Gerlach TT, Deliyski DD. Clinical evidence of the efficacy of high-speed videoendoscopy in the evaluation of vocal fold vibration In: Deliyski DD, ed. Proceedings of the 10th International Conference on Advances in Quantitative Laryngology, Voice and Speech Research. Vol 10 Cincinnati, OH: AQL Press; 2013:29–30. [Google Scholar]
- 40.Herrington-Hall BL, Lee L, Stemple JC, Niemi KR, McHone MM. Description of laryngeal pathologies by age, sex, and occupation in a treatment-seeking sample. J Speech Hear Disord. 1988;53(1):57–64. [DOI] [PubMed] [Google Scholar]
- 41.Hirano M Clinical Examination of Voice. Springer London, Limited; 1981. [Google Scholar]
- 42.Altman KW. Vocal fold masses. Otolaryngol Clin North Am. 2007;40(5):1091–1108. [DOI] [PubMed] [Google Scholar]
- 43.Altman KW. Vocal fold polyps and cysts In: Kendall KA, Leonard RJ, eds. Laryngeal Evaluation: Indirect Laryngoscopy to High-Speed Digital Imaging. Thieme; 2011:191–199. [Google Scholar]
- 44.Zeitels SM, Healy GB. Laryngology and phonosurgery. N Engl J Med. 2003;349(9):882–892. [DOI] [PubMed] [Google Scholar]
- 45.Poburka BJ. A new stroboscopy rating form. J Voice. 1999;13(3):403–413. [DOI] [PubMed] [Google Scholar]
- 46.Hillenbrand JM, Gayvert RT. Open source software for experiment design and control. J Speech Lang Hear Res. 2005;48(1):45–60. [DOI] [PubMed] [Google Scholar]
- 47.Poburka BJ, Patel RR, Bless DM. Voice-Vibratory Assessment with Laryngeal Imaging (VALI) form: Reliability of rating stroboscopy and high-speed videoendoscopy. J Voice. 2017;31(4):513.e1–513.e14. [DOI] [PubMed] [Google Scholar]
- 48.Bonilha HS, Focht KL, Martin-Harris B. Rater methodology for stroboscopy: A systematic review. J Voice Off J Voice Found. 2015;29(1):101–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Titze IR. Principles of Voice Production. Englewood Cliffs, N.J: Prentice Hall; 1994. [Google Scholar]
- 50.Khosla S, Muruguppan S, Gutmark E, Scherer R. Vortical flow field during phonation in an excised canine larynx model. Ann Otol Rhinol Laryngol. 2007;116(3):217–228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Khosla S, Murugappan S, Gutmark E. What can vortices tell us about vocal fold vibration and voice production. Curr Opin Otolaryngol Head Neck Surg. 2008;16(3):183–187. [DOI] [PubMed] [Google Scholar]
- 52.Nawka T, Konerding U. The interrater reliability of stroboscopy evaluations. J Voice. 2012;26(6):812.e1–812.e10. [DOI] [PubMed] [Google Scholar]
- 53.Bonilha HS, O’Shields M, Gerlach TT, Deliyski DD. Arytenoid adduction asymmetries in persons with and without voice disorders. Logoped Phoniatr Vocol. 2009;34(3):128–134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wallis L, Jackson-Menaldi C, Holland W, Giraldo A. Vocal fold nodule vs. vocal fold polyp: Answer from surgical pathologist and voice pathologist point of view. J Voice. 2004;18(1):125–129. [DOI] [PubMed] [Google Scholar]
- 55.Kobler JB, Rosen DI, Burns JA, et al. Comparison of a flexible laryngoscope with calibrated sizing function to intraoperative measurements. Ann Otol Rhinol Laryngol. 2006;115(10):733–740. [DOI] [PubMed] [Google Scholar]
- 56.Branski RC, Rosen CA, Verdolini K, Hebda PA. Biochemical markers associated with acute vocal fold wound healing: A rabbit model. J Voice. 2005;19(2):283–289. [DOI] [PubMed] [Google Scholar]
- 57.Johns MM, Garrett CG, Hwang J, Ossoff RH, Courey MS. Quality-of-life outcomes following laryngeal endoscopic surgery for nonneoplastic vocal fold lesions. Ann Otol Rhinol Laryngol. 2004;113(8):597–601. [DOI] [PubMed] [Google Scholar]
- 58.Nerurkar NK, Shukla SC. Subepithelial vocal fold cyst: A pearl on a string? Murthy P, ed. Int J Phonosurgery Laryngol. 2012;2(2):53–56. [Google Scholar]