Abstract
Chemoattractant receptor-homologous molecule expressed on T helper type 2 cells (CRTH2) is a second prostaglandin D2 receptor involved in mediating the allergic response; however, its central function is not yet known. Here, we demonstrate that central CRTH2 mediates emotional impairment. Lipopolysaccharide (LPS)-induced decreases in social interaction and novel exploratory behavior were observed in wild-type (CRTH2+/+) mice but not CRTH2-deficient (CRTH2−/−) mice, but both genotypes showed hypolocomotion and anorexia following LPS injection. Tumor (colon 26) inoculation, a more pathologically relevant model, induced decreases in social interaction and novel exploratory behavior in CRTH2+/+, but not CRTH2−/− mice. In addition, the CRTH2 antagonists including clinically available ramatroban reversed impaired social interaction and novel exploratory behavior after either LPS or tumor inoculation in CRTH2+/+ mice. Finally, LPS-induced c-Fos expression in the hypothalamic paraventricular nucleus (PVN) and central amygdala (CeA) was selectively abolished in CRTH2−/− mice. These results show that CRTH2 participates in LPS-induced emotional changes and activation in the PVN and CeA. Our study provides the first evidence that central CRTH2 regulates specific emotional behaviors, and that CRTH2 antagonism has potential as a therapeutic target for behavioral symptoms associated with tumors and infectious diseases.
Keywords: c-Fos, cyclooxygenase, lipopolysaccharide, prostaglandin D2 receptor, social interaction, tumor
Introduction
Chemoattractant receptor-homologous molecule expressed on T helper type 2 cells (CRTH2), also known as GPR44, was initially identified as a G-protein-coupled receptor expressed on human Th2 cells (Abe et al., 1999; Marchese et al., 1999; Nagata et al., 1999). CRTH2 is a receptor for prostaglandin D2 (PGD2; Hirai et al., 2001), and is found in Th2 cells, basophils, eosinophils, and monocytes. CRTH2 plays a pro-inflammatory role, and its activation causes leukocyte chemotaxis, which can lead to asthma development and atopic dermatitis (Spik et al., 2005; Satoh et al., 2006). The D prostanoid receptor (DP), a classical PGD2 receptor, also plays both pro-inflammatory and anti-inflammatory roles in asthma and leukocyte chemotaxis (Matsuoka et al., 2000; Angeli et al., 2004; Spik et al., 2005; Kostenis and Ulven, 2006).
In the CNS, PGD2-DP signaling inhibits prostaglandin E2 (PGE2)-induced allodynia, regulates the sleep–wake cycle, protects neurons against glutamate-induced toxicity, and increases food intake (Minami et al., 1996; Mizoguchi et al., 2001; Liang et al., 2005; Ohinata et al., 2008). In contrast, the role of CRTH2 in the CNS is virtually unknown. Recently, we observed altered expression of PGD synthase in the suprachiasmatic nucleus of pituitary adenylate cyclase-activating polypeptide-deficient mice (N.S., H. Hashimoto, A.B., unpublished observation), with impairments in circadian rhythm entrainment (Kawaguchi et al., 2010) and other behaviors (Hashimoto et al., 2001, 2009; Tanaka et al., 2006; Hattori et al., 2012). Moreover, we found that 15-deoxyΔ[12, 14] prostaglandin J2 (15d-PGJ2), enhances nerve growth factor (NGF)-induced neurite outgrowth in vitro, through activation of CRTH2 and its downstream target p38 MAPK (Hatanaka et al., 2010).
Infectious and autoimmune diseases, diabetes, and tumors result in systemic inflammation, evoking a number of responses including fatigue, fever, anorexia, depression, and apathy, which are collectively known as sickness behavior (Kronfol and Remick, 2000; Johnson et al., 2005; Dantzer et al., 2008). In rodents, lipopolysaccharide (LPS) or interleukin (IL)-1β-injected animals serve as models of sickness behavior (Yirmiya, 1996; Bluthé et al., 2000; Frenois et al., 2007; Haba et al., 2012). The cyclooxygenase (COX) and prostaglandin (PG) pathways are responsible for the behavioral changes observed in animal models of sickness behavior (Fishkin and Winslow, 1997; Naoi et al., 2006; Pecchi et al., 2008; Teeling et al., 2010; de Paiva et al., 2010).
The neural basis of LPS-induced sickness behavior has been investigated. LPS-induced activation in brain regions, including the nucleus of the solitary tract (NTS) and central amygdala (CeA), is associated with a decrease in social behavior (Konsman et al., 2000; Marvel et al., 2004). Two major pathways mediate this effect, namely the vagus nerve-dependent and humoral pathways. In the latter pathway, LPS (and IL-1β) upregulates COX-2 and PG production in brain endothelial and perivascular cells, resulting in activation of neighboring neurons (Schiltz and Sawchenko, 2002; Serrats et al., 2010).
In this study, we investigated CRTH2 involvement in LPS-induced sickness behavior and its consequent underlying neuroanatomical basis. In addition, we examined the role of CRTH2 in tumor inoculation-induced behavioral changes.
Materials and Methods
Animals and reagents.
All animal care and handling procedures were performed according to the Guiding Principles for the Care and Use of Laboratory Animals, approved by the Japanese Pharmacological Society and the Animal Care and Use Committee of Graduate School of Pharmaceutical Sciences, Osaka University. Generation of CRTH2-deficient (CRTH2−/−) mice by gene targeting has been reported previously (Satoh et al., 2006). Male 7-week-old BALB/c (BALB/cCrSlc) mice were purchased from Shimizu Laboratory Supplies. Conspecific male wild-type (CRTH2+/+) and CRTH2−/− mice were obtained from intercrossing CRTH2 heterozygous mice. Mice were kept on a 12 h light/dark cycle (light on from 8:00 A.M. to 8:00 P.M.), at a controlled room temperature. Each mouse (8–12 weeks old) was housed individually for 1 d before performing experiments. Pelleted food (CMF; Oriental Yeast) and water were available ad libitum.
LPS (Escherichia coli serotype 0127:B8; Sigma-Aldrich) was dissolved in sterile saline before use. Ramatroban, an orally bioavailable small molecule antagonist of CRTH2, was provided from Bayer Yakuhin and Nippon Shinyaku. Ramatroban was resuspended in 0.5% (w/v) carboxymethylcellulose for intraperitoneal injections, or dissolved in artificial CSF containing 0.1% (v/v) dimethylsulfoxide (DMSO; Sigma-Aldrich) for intracerebroventricular injections. A CRTH2-selective antagonist, CAY10471, and a DP-selective antagonist, BWA868C, were obtained from Cayman Chemical, and dissolved in artificial CSF containing 0.1% (v/v) DMSO before use. The nonselective COX inhibitor, indomethacin (Wako Pure Chemical Industries), was dissolved in saline. A COX-1-selective inhibitor, SC-560, and a COX-2-selective inhibitor, NS-398 (Cayman Chemical), were dissolved in saline containing 20% (v/v) DMSO.
Behavioral analysis.
Initial behavioral and physical screening were performed using SHIRPA (SmithKline Beecham; Harwell, Imperial College, Royal London Hospital, phenotype assessment; Rogers et al., 1997; Kato et al., 2010). Other behavioral analyses were performed as described previously (Haba et al., 2012). Briefly, locomotor activity was measured for 30 min immediately after each mouse was placed in an observation cage (28 cm length × 20 cm width × 12 cm height), using a digital counter system with an infrared sensor (Supermex; Muromachi Kikai). Food intake was measured by subtracting the weight of any uneaten pellets (which remained on the cage lid and fell into the cage) from the premeasured weight of pellets. In social interaction and object exploration tests, each mouse was placed in the observation cage and allowed 15 min habituation under dim light (40 lux). After habituation, a juvenile (4-week-old) male BALB/c mouse (Shimizu Laboratory Supplies) or a novel object (a wooden ball; diameter 5 cm) was placed in the center of the cage, and the behavior of the mouse was recorded for 5 min. Duration of social interaction (sniffing, licking, or following the juvenile) or object exploratory behavior (sniffing or licking the wooden ball), were measured from recordings by a trained and blinded observer.
Intracerebroventricular injections.
CRTH2+/+ mice were anesthetized with sodium pentobarbital (50 mg/kg, i.p.), and placed in a stereotaxic instrument (Narishige). A G-4 cannula (Eicom) was implanted, –0.4 mm posterior, 1.0 mm lateral, and 2.3 mm ventral from the bregma. After cannula implantation, each mouse was given 1 mg/kg buprenorphine (Sigma-Aldrich) to relieve the pain, and housed individually for at least 1 week before performing experiments. Drugs were injected in 2 μl volumes, at an infusion rate of 1 μl/min, using a microinjection pump (KD Scientific). The day after behavioral experiments, each mouse was intracerebroventricularly injected with 3 μl of 1% (w/v) Evans blue solution (Sigma-Aldrich), and a coronal section of the brain made. The intracerebroventricular injection was judged successful if the third ventricle was stained by Evans blue.
Semiquantitative reverse-transcription PCR.
The brains were rapidly removed, and the amygdala and hypothalamus dissected from coronal brain slices (between –0.94 and 1.94 mm from the bregma) on an ice-cold glass plate. Total RNA was extracted by the guanidine thiocyanate method as described previously (Chomczynski and Sacchi, 1987). Total RNA (1 μg) was reverse transcribed using MMLV reverse transcriptase (Invitrogen). Real-time semiquantitative reverse-transcription PCR was performed using GoTaq qPCR Master Mix (Promega), according to the manufacturer's instructions, with primers specific for mouse glyceraldehyde-3-phosphate dehydrogenase (GAPDH; 5′-GTG TTC CCT ACC CCC AAT GTG-3′ and 5′-TAC CAG GAA ATG AGC TTG AC-3′), mouse COX-1 (5′-TGT TCA GCT TCT GGC CCA ACA GCT-3′ and 5′-AGC GCA TCA ACA CGG ACG CCT GTT-3′), mouse COX-2 (5′-GGG TTG CTG GGG GAA GAA ATG TG-3′ and 5′-GGT GGC TGT TTT GGT AGG CTG TG-3′), lipocalin-type prostaglandin D synthase (L-PGDS, 5′-TCA ACA AGA CAA GTT CCT GG-3′ and 5′-TGA ATT TCT CCT TCA GCT CG-3′), and hematopoietic prostaglandin D synthase (H-PGDS; 5′-GAA TAG AAC AAG CTG ACT GGC-3′ and 5′-AGC CAA ATC TGT GTT TTT GG-3′). Reactions were cycled 40 times with denaturation at 95°C for 20 s, annealing for 20 s at 57°C (H-PGDS), 62°C (COX-1), or 65°C (GAPDH, COX-2, and L-PGDS), and extension at 72°C for 20 s. All PCR products were sequenced to confirm their identity.
Immunohistochemistry.
Mice were intraperitoneally injected with LPS and placed back into their home cages. At 2 h, 6 h, 12 h, or 24 h after injection, mice were deeply anesthetized with 50 mg/kg sodium pentobarbital, and perfused transcardially with saline and 4% (w/v) paraformaldehyde in PBS. Whole brains were dissected and postfixed in the same fixative overnight at 4°C. The brains were then cryoprotected in 20% (w/v) sucrose in PBS for 48 h at 4°C. For c-Fos staining, coronal brain sections (20 μm), containing the bed nucleus of stria terminalis (BNST; ∼0.26 mm from the bregma), hypothalamic paraventricular nucleus (PVN; approximately –0.94 mm from the bregma), CeA (approximately –1.06 mm from the bregma), and NTS (approximately –7.20 mm from the bregma) were prepared and processed for immunohistochemistry with the rabbit anti-c-Fos polyclonal primary antibody (sc-52; Santa Cruz Biotechnology; 1:2000) and biotin-labeled anti-rabbit IgG secondary antibody (Vector Laboratories; 1:2000) as described previously (Haba et al., 2012). As shown in Figure 6A, images of the corresponding brain regions were photographed using light microscopy (50× magnification), and c-Fos-positive cells in a 1 mm × 1 mm area in 3–5 slices from each animal were manually counted by experienced observers blinded to the treatment and averaged. In our previous study we confirmed that this method is highly reproducible and quantitative (Haba et al., 2012). For COX-2 staining, and subsequent identification of COX-2-expressing cell types, rabbit anti-COX-2 polyclonal antibody (sc-1747; Santa Cruz Biotechnology; 1:1000), rat anti-CD31 monoclonal antibody (sc-18916; Santa Cruz Biotechnology; 1:100), mouse anti-NeuN monoclonal antibody (MAB377; Millipore Bioscience Research Reagents; 1:500), mouse anti-glial fibrillary acidic protein (GFAP) monoclonal antibody (MAB360; Millipore Bioscience Research Reagents; 1:500), and rat anti-CD11b monoclonal antibody (550282; BD PharMingen; 1:500) were used followed by Alexa 594-conjugated anti-rabbit IgG (A-21207; Invitrogen; 1:500), Alexa 488-conjugated anti-rat IgG (A-21206; Invitrogen; 1:500), or Alexa 488-conjugated anti-mouse IgG (A-212021; Invitrogen; 1:500), and 4′, 6-diamino-2-phenylindole (DAPI; D3571; Invitrogen; 1:2000). For dual staining of COX-2 and c-Fos, rabbit anti-COX-2 antibody (1:2000) and goat anti-c-Fos antibody (sc-52-G; Santa Cruz Biotechnology; 1:1000) were used followed by Alexa 594-conjugated anti-rabbit IgG (1:500), Alexa 488-conjugated anti-goat IgG (A-11055; Invitrogen; 1:500), and DAPI (1:2000).
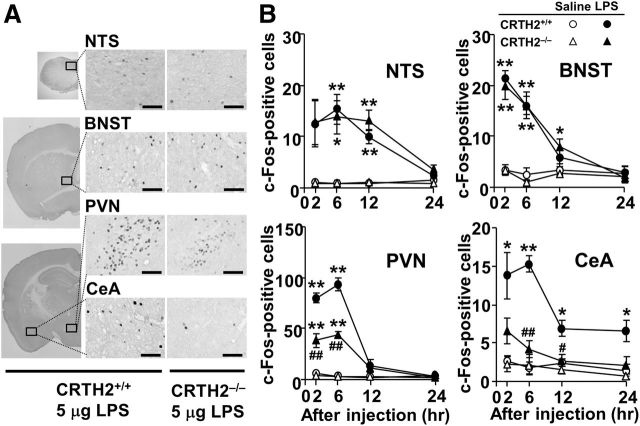
Figure 6.
LPS-induced c-Fos expression is attenuated in the PVN and CeA in CRTH2−/− mice. A, Representative photomicrographs showing c-Fos labeling in the NTS, BNST, PVN, and CeA of CRTH2+/+ (left and middle, lower and higher magnification images, respectively) and CRTH2−/− mice (right, higher magnification images), 6 h after LPS injection. Scale bars: 100 μm. B, The number of c-Fos-positive cells in the NTS, BNST, PVN, and CeA of CRTH2−/− (triangles) and CRTH2+/+ (circles) mice intraperitoneally injected with 5 μg LPS (closed symbols) or saline (open symbols). Data are expressed as the mean ± SEM (n = 3); *p < 0.05, **p < 0.01 versus saline, #p < 0.05, ##p < 0.01 versus CRTH2+/+ mice by three-way ANOVA followed by Tukey–Kramer test.
Tumor inoculation.
The murine colon 26 adenocarcinoma cell line was kindly supplied by Drs. Shinsaku Nakagawa and Naoki Okada from the Graduate School of Pharmaceutical Sciences, Osaka University. Colon 26 cells were maintained in RPMI-1640 medium (Wako Pure Chemicals) containing 10% (v/v) fetal bovine serum (FBS; Nichirei Bioscience) in a humidified atmosphere with 5% (v/v) CO2 at 37°C. Subconfluent cells were treated with 0.25% (w/v) trypsin and 1 mm EDTA, and suspended at a concentration of 1 × 107 cells/ml in RPMI-1640 medium containing 10% (v/v) FBS. Cell suspension (1 × 106 cells/0.1 ml) or cell-free medium was inoculated subcutaneously into the abdomen.
Statistical analysis
Statistical evaluation was performed using StatView (SAS Institute). Significant differences were determined by Mann–Whitney U tests, or two-way or three-way ANOVA with intervention (genotype or pretreatment with antagonists or inhibitors), challenge (LPS injection or tumor inoculation), and time as factors. Subsequently, data were analyzed using a post hoc Tukey–Kramer test when a significant main effect or interaction was observed. The threshold for statistical significance was defined as p < 0.05.
Results
Physical characterization and behavioral profiles of CRTH2−/− mice
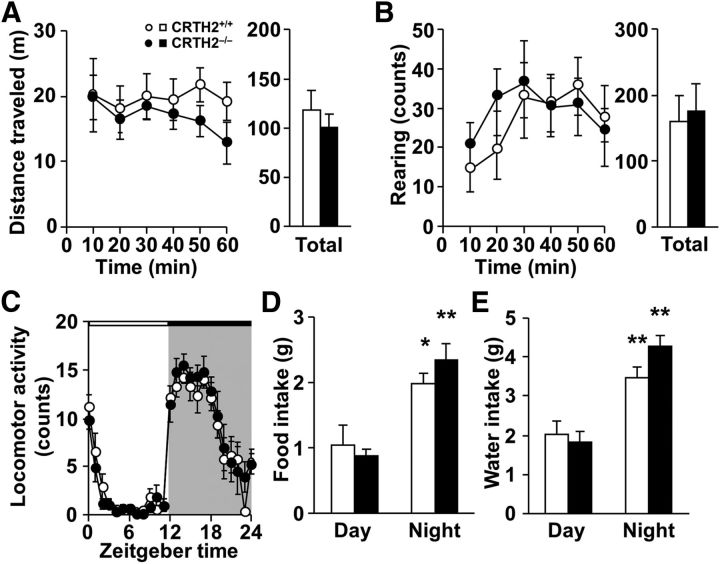
Physical malfunctions in sensory ability and motor function influence performance scores in behavioral tests. As the physical and behavioral profiles of CRTH2−/− mice have not yet been evaluated, we used the standardized SHIRPA screening program (Rogers et al., 1997) to characterize the mice. In the primary SHIRPA screening, there were no differences in appearance, motor and sensory functions, anxiety levels, or aggression between CRTH2−/− and CRTH2+/+ mice (Table 1). In the secondary screening, there were no genotype differences in locomotor activity in an open-field or circadian rhythm (Fig. 1). These results suggest that CRTH2−/− mice have no physical dysfunction, and their behavioral profiles are comparable to CRTH2+/+ mice under normal conditions.
Table 1.
SHIRPA primary screening in CRTH2+/+ and CRTH2−/− mice
| Paradigm | Examination | CRTH2+/+ | CRTH2−/− | p value |
|---|---|---|---|---|
| Appearance | Body weight (g) | 27.4 ± 0.7 | 26.4 ± 0.7 | 0.207 |
| Body position | 3.67 ± 0.2 | 4.00 ± 0.0 | 0.083 | |
| Respiration rate | 1.92 ± 0.1 | 1.91 ± 0.1 | 0.950 | |
| Heart rate | 0.50 ± 0.2 | 0.45 ± 0.2 | 0.831 | |
| Tremor | 0.10 ± 0.1 | 0.00 ± 0.0 | 0.338 | |
| Palpebral closure | 0.00 ± 0.0 | 0.00 ± 0.0 | N.D. | |
| Piloerection | 0.00 ± 0.0 | 0.00 ± 0.0 | N.D. | |
| Skin color | 1.00 ± 0.0 | 1.00 ± 0.0 | N.D. | |
| Lacrimation | 0.10 ± 0.1 | 0.00 ± 0.0 | 0.361 | |
| Salivation | 0.67 ± 0.1 | 0.60 ± 0.2 | 0.827 | |
| Motor functions | Spontaneous activity | 1.50 ± 0.3 | 2.00 ± 0.3 | 0.196 |
| Transfer arousal | 2.50 ± 0.4 | 3.09 ± 0.3 | 0.205 | |
| Gait | 0.00 ± 0.2 | 0.00 ± 0.0 | 0.166 | |
| Pelvic elevation | 1.58 ± 0.2 | 1.91 ± 0.1 | 0.158 | |
| Tail elevation | 0.92 ± 0.2 | 0.82 ± 0.1 | 1.000 | |
| Trunk curl | 0.00 ± 0.0 | 0.00 ± 0.0 | N.D. | |
| Limb grasping | 0.00 ± 0.0 | 0.00 ± 0.0 | N.D. | |
| Grip strength | 2.33 ± 0.1 | 2.18 ± 0.2 | 0.555 | |
| Righting reflex | 0.00 ± 0.0 | 0.00 ± 0.0 | N.D. | |
| Contact righting reflex | 0.83 ± 0.1 | 0.91 ± 0.1 | 0.658 | |
| Negative geotaxis | 0.25 ± 0.1 | 0.10 ± 0.1 | 0.325 | |
| Sensory functions | Touch escape | 2.25 ± 0.3 | 2.18 ± 0.2 | 0.642 |
| Positional passivity | 0.33 ± 0.1 | 0.18 ± 0.1 | 0.419 | |
| Visual placing | 2.08 ± 0.1 | 1.82 ± 0.2 | 0.166 | |
| Pinna reflex | 0.42 ± 0.1 | 0.36 ± 0.2 | 0.799 | |
| Corneal reflex | 0.83 ± 0.1 | 1.00 ± 0.0 | 0.166 | |
| Toe pinch | 2.83 ± 0.1 | 2.73 ± 0.2 | 0.852 | |
| Anxiety level | Fear | 0.00 ± 0.0 | 0.00 ± 0.0 | N.D. |
| Urination | 0.29 ± 0.2 | 0.45 ± 0.2 | 0.643 | |
| Defecation | 5.17 ± 1.1 | 5.91 ± 0.9 | 0.709 | |
| Vocalization | 0.00 ± 0.0 | 0.10 ± 0.1 | 0.296 | |
| Aggression | Provoked biting | 0.10 ± 0.1 | 0.27 ± 0.1 | 0.242 |
Data are expressed as the mean ± SEM (n = 11–12). p values were calculated using the Mann-Whitney U test. N.D., not detected.
Figure 1.
SHIRPA secondary screening in CRTH2−/− mice. A, Distance traveled in open-field test. Bar graph shows total distance traveled during 60 min (n = 9–10). B, Rearing counts in open-field test. Bar graph shows total counts of rearing during 60 min (n = 9–10). C–E, Circadian changes in locomotor activity (C; n = 8), food intake (D; n = 12), and water intake (E; n = 12). White and black horizontal bars indicate day and night, respectively (C). Open symbols and columns, CRTH2+/+ mice; closed symbols and columns, CRTH2−/− mice. Data are expressed as means ± SEM; *p < 0.05, **p < 0.01 versus day time by two-way ANOVA followed by Tukey–Kramer test.
CRTH2 is involved in LPS-induced decreases in social and object exploratory behavior
In CRTH2−/− mice, we first examined the effects of LPS (5 μg) on social interaction, object exploration, locomotor activity, and food intake (Fig. 2A,E,I,M) at each appropriate time point (Haba et al., 2012). Two-way ANOVA revealed significant main effects of LPS on all these four measures (F(1,28) = 11.5, p = 0.0021 for object exploration, p < 0.0001 for the others) and genotype on social interaction (F(1,30) = 7.37, p = 0.011) and object exploration (F(1,28) = 9.49, p = 0.0046), as well as significant interactions between LPS and genotype for social interaction (F(1,30) = 7.25, p = 0.012) and object exploration (F(1,28) = 4.76, p = 0.038). Post hoc Tukey–Kramer multiple-comparison test revealed that LPS did not change social interaction and object exploratory behavior in CRTH2−/− mice (Fig. 2A,E; p > 0.05), although LPS significantly decreased them in CRTH2+/+ mice (p < 0.01). LPS decreased locomotor activity and food intake similarly in CRTH2−/− and CRTH2+/+ mice (Fig. 2I,M; p < 0.01 in both genotypes). Systemic administration of ramatroban, a clinically available CRTH2 antagonist, in CRTH2+/+ mice produced the same effects as seen in CRTH2 deficiency (Fig. 2B,F,J,N). Two-way ANOVA revealed significant main effects of LPS on all the four measures (F(1,24) = 18.1, p = 0.0003 for social interaction; F(1,32) = 5.57, p = 0.025 for object exploration; F(1,25) = 85.9, p < 0.0001 for locomotor activity; and F(1,32) = 222, p < 0.0001 for food intake) and ramatroban on social interaction (F(1,24) = 13.7, p = 0.0011) and object exploration (F(1,32) = 18.4, p = 0.0002), as well as significant interactions between LPS and ramatroban for social interaction (F(1,24) = 11.9, p = 0.0021) and object exploration (F(1,32) = 16.5, p = 0.0003). The post hoc test revealed that ramatroban completely blocked LPS-induced decreases in social and object exploratory behavior (Fig. 2B,F; p < 0.01), although ramatroban had no significant effect on LPS-induced decreases in locomotor activity and food intake (Fig. 2J,N; p > 0.05).
Figure 2.
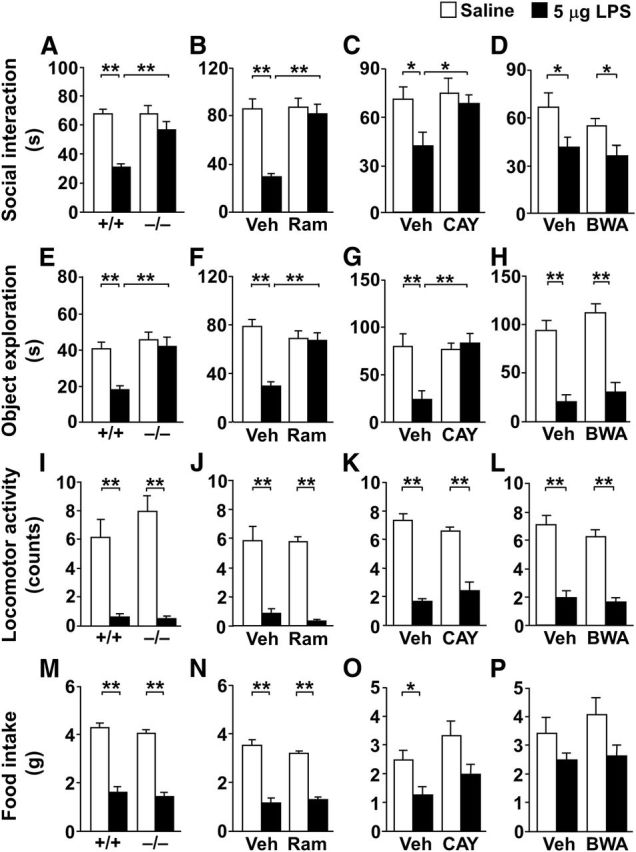
CRTH2 deficiency and the CRTH2 antagonists reverse LPS-induced decreases in social and object exploratory behavior. Five micrograms of LPS (closed columns) or saline (open columns) were intraperitoneally injected into CRTH2+/+ (+/+) and CRTH2−/− (−/−) mice (A, E, I, M; n = 6–13), and CRTH2+/+ mice were pretreated intraperitoneally for 1 h with 30 mg/kg ramatroban (Ram; B, F, J, N) and pretreated intracerebroventricularly for 30 min with 1 nmol CAY10471 (CAY; C, G, K, O; n = 5–9) or 1 nmol BWA868C (BWA; D, H, L, P; n = 4–9). After LPS injection, the duration engaged in social interaction (A–D) and object exploratory behavior (E–H) at 10 h, locomotor activity at 2 h (I–L), and food intake for 24 h (M–P) were measured. Veh, vehicle. Data are expressed as the mean ± SEM; *p < 0.05, **p < 0.01 by two-way ANOVA followed by Tukey—Kramer test.
CRTH2 antagonists injected intracerebroventricularly reverse LPS-induced decreases in social and object exploratory behavior
To examine whether brain-expressed CRTH2 is involved in LPS-induced decreases in social and object exploratory behavior, we injected the CRTH2-selective antagonist CAY10471 intracerebroventricularly to CRTH2+/+ mice (Fig. 2C,G,K,O). Two-way ANOVA revealed significant main effects of LPS in all of the four measures (F(1,27) = 4.41, p = 0.045 for social interaction, p < 0.01 for the others) and CAY10471 on object exploration (F(1,22) = 6.52, p = 0.018), as well as a significant interaction between LPS and CAY10471 on object exploration (F(1,22) = 10.4, p = 0.0039). The post hoc test revealed that CAY10471 blocked LPS-induced decreases in social interaction (Fig. 2C; p < 0.05) and object exploratory behavior (Fig. 2G; p < 0.01). In contrast, BWA868C, a DP-selective antagonist, had no effect on LPS-induced decreases in social and object exploratory behavior (Fig. 2D,H). Two-way ANOVA revealed no significant main effect of BWA868C on social interaction (F(1,28) = 0.363, p = 0.55) or object exploration (F(1,14) = 2.39, p = 0.14). Both CAY10471 and BWA868C had no effect on LPS-induced decreases in locomotor activity and food intake (Fig. 2K,L,O,P).
COX-2 mediates LPS-induced decreases in object exploratory behavior
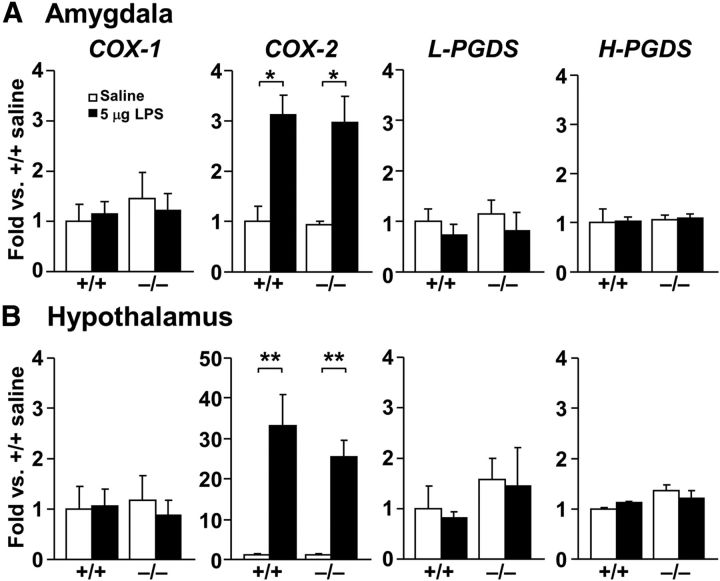
LPS-induced sickness behavior involves the COX–PG pathway (Fishkin and Winslow, 1997; Naoi et al., 2006; Teeling et al., 2010; de Paiva et al., 2010). However, it is not known if this pathway is involved in LPS-induced decreases in object exploratory behavior. Therefore, we examined mRNA expression of COX-1, COX-2, L-PGDS, and H-PGDS in brain regions known to regulate emotional behaviors (Konsman et al., 2000; Marvel et al., 2004; Frenois et al., 2007) in CRTH2−/− and CRTH2+/+ mice that received either saline or LPS (5 μg). Only COX-2 was upregulated after LPS injection in the amygdala and hypothalamus in both CRTH2−/− and CRTH2+/+ mice (Fig. 3).
Figure 3.
LPS selectively induces expression of COX-2 mRNA. mRNA levels of COX-1, COX-2, L-PGDS, and H-PGDS were determined in the amygdala (A; n = 3) and hypothalamus (B; n = 3) in CRTH2+/+ (+/+) and CRTH2−/− (−/−) mice, 2 h after intraperitoneal injection of 5 μg LPS (closed columns) or saline (open columns). Data are expressed as the mean ± SEM; *p < 0.05, **p < 0.01 by two-way ANOVA followed by Tukey—Kramer test.
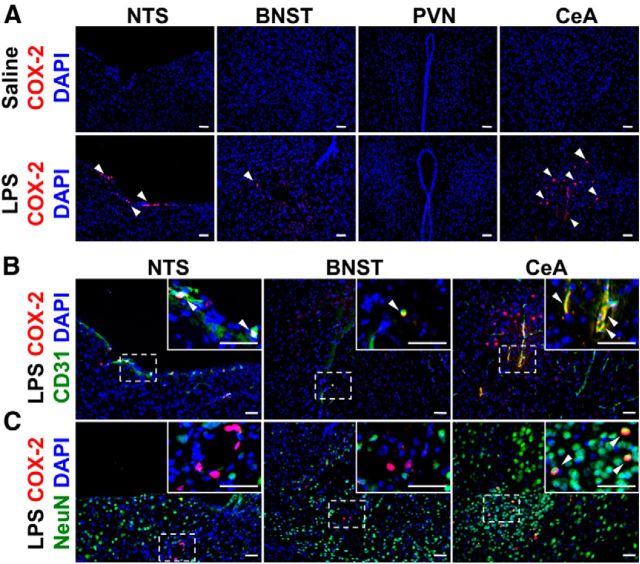
As COX-2 in brain endothelial cells plays a key role in humoral regulation of the brain (Schiltz and Sawchenko, 2002; Serrats et al., 2010), we examined cellular localization of COX-2 in the NTS, BNST, PVN, and CeA, brain regions in which LPS-impaired social behavior is mediated (Konsman et al., 2000; Marvel et al., 2004). LPS induced COX-2 expression in the NTS, BNST, and most prominently the CeA, 2 h after LPS injection (Fig. 4A). Double immunostaining of COX-2 and cell-specific markers showed COX-2 was localized in CD31-positive endothelial cells in the NTS, BNST, and CeA (Fig. 4B, arrowheads). In addition, COX-2 was localized in NeuN-positive neurons in the CeA; however, there were no COX-2-positive neurons in the NTS and BNST (Fig. 4C). COX-2 immunoreactivity was not observed in GFAP-positive astrocytes and CD11b-positive microglia (data not shown).
Figure 4.
Cellular localization of COX-2 in the NTS, BNST, PVN, and CeA after LPS injection. A, Fluorescent microscopy for COX-2 (red; arrowheads) and DAPI (blue) in the NTS, BNST, PVN, and CeA, 2 h after 5 μg LPS or saline injection in CRTH2+/+ mice. B, C, Fluorescent microscopy for COX-2 (red), the endothelial marker CD31 (green; B), the neuronal marker NeuN (green; C), and DAPI (blue) in the NTS, BNST, and CeA, 2 h after 5 μg LPS injection. Arrowheads indicate colocalization of COX-2 with CD31 or NeuN. Scale bars: 50 μm.
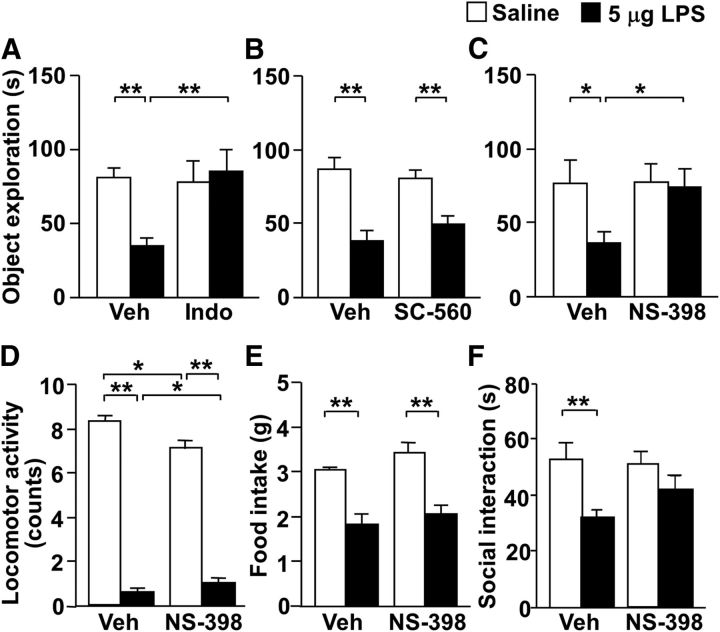
To determine the behavioral consequences of COX-2 inhibition, we examined the effect of COX inhibitors on LPS-induced behavioral changes (Fig. 5). Pretreatment with indomethacin, a COX-1 and COX-2 nonselective inhibitor, completely reversed LPS-induced decreases in object exploration (Fig. 5A). In addition, NS-398, a COX-2-selective inhibitor, but not SC-560, a COX-1-selective inhibitor, completely reversed LPS-induced decreases in object exploration (Fig. 5B,C). In NS-398-treated mice, LPS-induced decreases in social interaction were not observed; however, this effect was not statistically significant when compared with vehicle and LPS-treated mice (Fig. 5F). NS-398 had no effect on LPS-induced decreases in locomotor activity and food intake (Fig. 5D,E).
Figure 5.
Inhibitors of COX-2, but not COX-1, selectively reverse LPS-induced decreases in object exploratory behavior. A–C, CRTH2+/+ mice were intraperitoneally injected with vehicle (Veh) or 10 mg/kg indomethacin (Indo; A; n = 6–7), 10 mg/kg SC-560 (B; n = 5–6), or 5 mg/kg NS-398 (C; n = 7–8), 1 h before intraperitoneal injection of 5 μg LPS (closed columns) or saline (open columns). Ten hours after LPS injection, object exploratory behavior was examined. D–F, One hour before intraperitoneal injection of 5 μg LPS (closed columns) or saline (open columns), CRTH2+/+ mice were intraperitoneally injected with Veh or NS-398, and their locomotor activity at 2 h (D), food intake for 24 h (E), and duration engaged in social interaction at 10 h (F) after LPS injection were measured in the same animals (n = 12). Data are expressed as the mean ± SEM; *p < 0.05, **p < 0.01 by two-way ANOVA followed by Tukey—Kramer test.
These results suggest that LPS-induced decreases at least in object exploration are dependent on the COX-2–PGD2–CRTH2 signaling pathway.
CRTH2 is selectively involved in LPS-induced neural activation of the PVN and CeA
In the sickness behavior model, stimulation of the vagus nerve terminal is known to transfer peripheral inflammatory signals to the NTS, which extends to various brain nuclei. Thus, we next examined the involvement of CRTH2 in activation of brain nuclei that are associated with sickness behavior (Konsman et al., 2000; Marvel et al., 2004). In accordance with our previous observation (Haba et al., 2012), the number of c-Fos-positive cells markedly increased in the NTS, BNST, PVN, and CeA, within 6 h after LPS injection in CRTH2+/+ mice (Fig. 6; Table 2). Twenty-four hours after LPS injection, the number of c-Fos-positive cells in the NTS, BNST, and PVN returned to basal levels; however, higher levels remained in the CeA (Table 2; Haba et al., 2012). Therefore, the present study examined the number of c-Fos-positive cells in the NTS, BNST, PVN, and CeA in CRTH2−/− mice (Fig. 6). Three-way ANOVA revealed significant main effects of LPS and time (p < 0.005), as well as a significant interaction between LPS and time (p < 0.01) in all the nuclei in CRTH2−/− and CRTH2+/+ mice. In the PVN and CeA, there was a significant main effect of genotype (F(1,32) = 56.5, p < 0.0001 for the PVN; F(1,32) = 34.2, p < 0.0001 for the CeA) and a significant interaction between LPS and genotype (F(1,32) = 49.7, p < 0.0001 for the PVN; F(1,32) = 26.5, p < 0.0001 for the CeA). The Tukey–Kramer multiple-comparison test revealed that the number of c-Fos-positive cells was significantly decreased in the PVN (p < 0.01 at 2 and 6 h) and CeA (p < 0.01 at 6 h, p < 0.05 at 12 h) in CRTH2−/− mice compared with CRTH2+/+ mice. No significant difference in the number of c-Fos-positive cells was seen in the NTS and BNST between LPS-injected CRTH2−/− and CRTH2+/+ mice (Fig. 6B; p > 0.05 at all times examined). Furthermore, in CRTH2+/+ mice, the LPS-induced increase in the number of c-Fos-positive cells in the PVN and CeA was attenuated by ramatroban (Table 2). At 6 h after LPS injection, there were significant main effects of LPS in all four nuclei (F(1,16) = 50.1, p < 0.00001 for the NTS; F(1,16) = 57.5, p < 0.00001 for the BNST; F(1,16) = 35.0, p < 0.00001 for the PVN; and F(1,16) = 5.47, p = 0.033 for the CeA) and genotype in the CeA (F(1,16) = 22.0, p = 0.0002), as well as a significant interaction between LPS and ramatroban in the PVN and CeA (F(1,16) = 6.97, p = 0.018 for the PVN; F(1,16) = 7.73, p = 0.013 for the CeA). In contrast, at 24 h after LPS injection, significant main effects of LPS and genotype as well as a significant interaction between them were revealed only in the CeA (LPS, F(1,16) = 4.58, p = 0.048; genotype, F(1,16) = 8.14, p = 0.012; LPS × genotype, F(1,16) = 5.08, p = 0.039). These results suggest that CRTH2 selectively mediates LPS-induced activation of the PVN and CeA.
Table 2.
Effect of ramatroban on LPS-induced c-Fos expression in the brain
| Time | Region | Saline + vehicle | LPS + vehicle | Saline + ramatroban | LPS + ramatroban |
|---|---|---|---|---|---|
| 6 h | NTS | 3.8 ± 1.1 | 36.1 ± 5.9** | 3.1 ± 1.4 | 26.3 ± 4.9** |
| BNST | 6.3 ± 2.1 | 22.5 ± 3.8** | 3.2 ± 0.9 | 29.6 ± 3.4** | |
| PVN | 9.8 ± 2.8 | 79.8 ± 14** | 14.3 ± 3.6 | 41.1 ± 6.7**,# | |
| CeA | 2.4 ± 0.9 | 13.9 ± 2.3** | 3.1 ± 1.3 | 6.0 ± 1.5# | |
| 24 h | NTS | 11.3 ± 2.8 | 6.9 ± 2.5 | 10.9 ± 2.7 | 7.4 ± 2.9 |
| BNST | 11.4 ± 2.8 | 12.6 ± 4.0 | 8.2 ± 2.3 | 11.8 ± 3.7 | |
| PVN | 16.7 ± 4.2 | 12.1 ± 4.1 | 12.2 ± 3.4 | 10.3 ± 2.2 | |
| CeA | 7.5 ± 2.4 | 20.3 ± 4.8* | 5.8 ± 1.6 | 5.5 ± 1.6# |
CRTH2+/+ mice were intraperitoneally injected with vehicle or ramatroban (30 mg/kg), 1 h before saline or LPS (5 μg) injection. The number of c-Fos-positive cells in the NTS, BNST, PVN, and CeA, 6 and 24 h after the last injection are shown. Data are expressed as the mean ± SEM (n = 5).
*p < 0.05,
**p < 0.01 versus saline,
#p < 0.05 versus vehicle, by two-way ANOVA followed by Tukey–Kramer test.
CRTH2 participates in tumor-induced decreases in social and object exploratory behavior
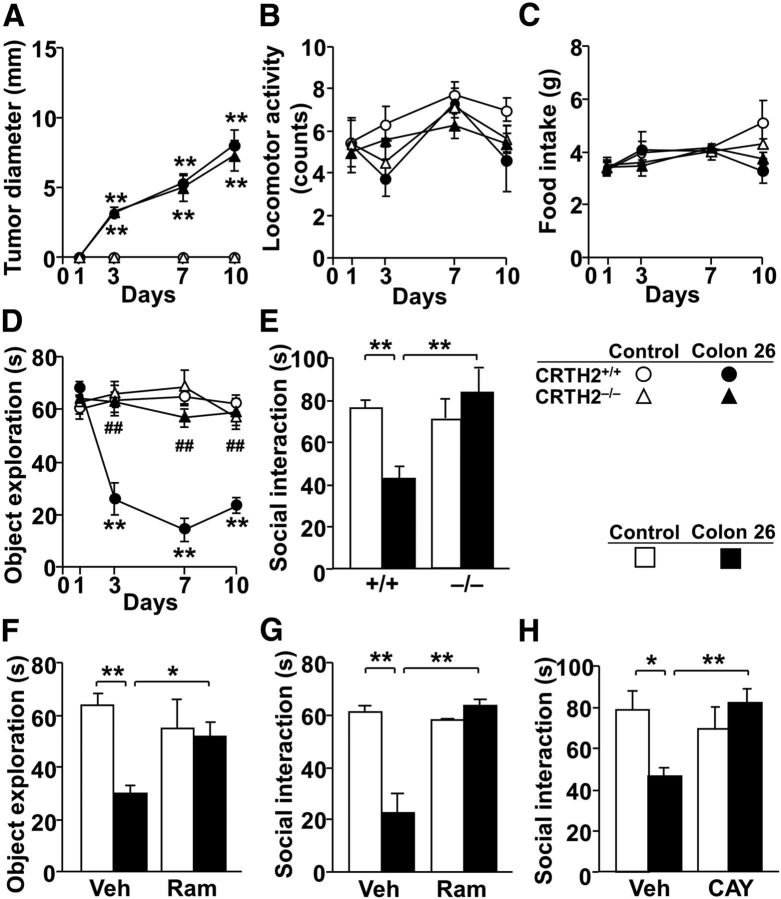
Sickness behavior is observed with tumors (Kronfol and Remick, 2000). Therefore, to gain additional pathological insight into the role of CRTH2, we determined whether CRTH2 deficiency or antagonism can improve tumor-induced behavioral deficits (Fig. 7). Inoculation of murine colon 26 adenocarcinoma cells into CRTH2+/+ mice impaired object exploratory behavior and social interaction, but not locomotor activity and food intake (Fig. 7B–D). Interestingly, although the tumor grew similarly in CRTH2−/− and CRTH2+/+ mice, impairments in object exploratory behavior and social interaction were not observed in CRTH2−/− mice (Fig. 7A,D,E). Three-way ANOVA revealed significant main effects of tumor (p < 0.0001) and time (p < 0.001), a significant interaction between them for tumor diameter and object exploration, as well as a significant main effect of genotype (p < 0.0001) and significant interactions among genotype, tumor, and day for object exploration (tumor × genotype, F(1,54) = 36.3, p < 0.0001; genotype × day, F(3,54) = 5.41, p = 0.0025; tumor × genotype × day, F(3,54) = 6.09, p = 0.0012; Fig. 7A–D). For social interaction, two-way ANOVA revealed no significant effect of tumor (F(1,20) = 2.48, p = 0.13), but a significant main effect of genotype (F(1,20) = 8.11, p = 0.010) and a significant interaction between tumor and genotype (F(1,20) = 13.8, p = 0.0014; Fig. 7E). In addition, tumor-impaired social interaction and object exploratory behavior in CRTH2+/+ mice were completely reversed by a single injection of ramatroban, even when the tumor was enlarged (Fig. 7F,G). Two-way ANOVA revealed significant main effects (p < 0.05) for all factors and significant interactions (p < 0.05) except for genotype effect in object exploration (F(1,12) = 1.09, p = 0.31). Intraperitoneal injection of the specific CRTH2 antagonist CAY10471 (30 mg/kg) also blocked tumor-impaired social interaction (Fig. 7H; tumor, F(1,44) = 1.56, p = 0.21; CAY10471, F(1,44) = 3.04, p = 0.088; tumor × CAY10471, F(1,44) = 8.38, p = 0.0059, two-way ANOVA), which further strengthens the crucial and selective role of CRTH2 in tumor-induced sickness behaviors.
Figure 7.
CRTH2 deficiency and the CRTH2 antagonists reverse tumor (colon 26)-induced decreases in social behavior. CRTH2+/+ and CRTH2−/− mice were subcutaneously injected with 1 × 106 colon 26 cells or cell-free medium. A–E, Tumor diameter (A), locomotor activity (B), food intake (C), duration engaged in object exploratory behavior (D), and social interaction (E) were determined at the indicated days (E, at day 7) after the inoculation. F–H, At day 7, 30 min after CRTH2+/+ mice were intraperitoneally injected with vehicle (Veh), 30 mg/kg ramatroban (Ram; F and G) or 30 mg/kg CAY10471 (CAY; H), the duration engaged in object exploratory behavior (F) and social interaction (G, H) was analyzed. Data are expressed as the mean ± SEM (A–E, n = 4–8; F, G, n = 4–5; H, n = 8–9). *p < 0.05, **p < 0.01 versus control, ##p < 0.01 versus CRTH2+/+ mice by three-way (A–D) or two-way (E–H) ANOVA followed by Tukey—Kramer test.
Discussion
In this study, we sought to determine the role of CRTH2 in the brain, and consequently have identified a number of novel findings. (1) CRTH2 deficiency in mice completely reverses LPS-impaired social interaction and object exploration behaviors, although it has no effect on physical and behavioral performance under normal conditions. (2) This behavioral effect can also be obtained by systemic (intraperitoneal) injection of the CRTH2 antagonist, ramatroban, as well as by intracerebroventricular injection of CAY10471, a more selective CRTH2 antagonist. In addition, we show that (3) in LPS-injected mice, COX-2 is selectively upregulated in several brain regions including the amygdala and hypothalamus; (4) COX-2 inhibitors, but not a COX-1-selective inhibitor, reverse LPS-impaired object exploration behavior selectively; and (5) LPS-induced c-Fos expression in the PVN and CeA is markedly decreased in CRTH2−/− mice and CRTH2+/+ mice that received ramatroban. Finally, we also show that (6) inoculation of tumor (colon 26) cells impairs social interaction and object exploration behaviors, (7) which can be completely reversed by CRTH2 inhibition. These results suggest that under the conditions modeled by LPS injection or tumor inoculation, CRTH2 activation by PGs produced by COX-2 is crucial for the specific activation of brain regions, including the PVN and CeA, and for the selective impairments in emotional aspects of sickness behavior, such as social interaction and object exploration behaviors. The results also suggest that CRTH2 antagonism has potential as a therapeutic target for behavioral symptoms resulting from infectious diseases and tumors.
Recently, we demonstrated that social and object exploration behavior is a core phenotype in LPS-induced sickness in terms of the duration of the LPS action. LPS-induced hypolocomotion is ameliorated within 10 h, but social interaction and novel object exploratory behavior remain decreased for at least 24 h after LPS injection (Haba et al., 2012). These results suggest that LPS affects exploratory behavior, which is likely independent from spontaneous locomotor behaviors (Haba et al., 2012). In the present study, we show that tumor cell inoculation impairs social interaction and novel object exploratory behavior, but not locomotor activity or food intake. CRTH2 deficiency and CRTH2 antagonism are able to reverse the behavioral impairments induced by LPS or tumor growth. These results suggest that social interaction and novel object exploratory behavior are distinct emotional aspects of sickness behavior, critically mediated by the COX2–PG–CRTH2 pathway, and are therefore treatable by CRTH2 inhibition. In NS-398-treated mice, LPS-induced decreases in social interaction were not observed; however, this effect was not statistically significant when compared with vehicle and LPS-treated mice (Fig. 5F). Our previous study showed that decreased object exploration behavior is seen at a dose as low as 0.3 μg of LPS while LPS-impaired social interaction required higher doses (1 μg LPS), suggesting LPS most prominently affects object exploratory behaviors (Haba et al., 2012). This may explain the relatively weak effect of NS-398 to reverse LPS-induced decreases in social interaction.
There is considerable evidence indicating that PGs are involved in LPS-induced sickness behavior (Fishkin and Winslow, 1997; Naoi et al., 2006; Teeling et al., 2010; de Paiva et al., 2010; Saper et al., 2012). In particular, PGE2-E prostanoid receptor (EP) signaling is involved in LPS-induced anorexia and release of adrenocorticotropic hormone (Matsuoka et al., 2003; Elander et al., 2007). In addition, PGE2-EP1 signaling is critical for susceptibility to repeated social defeat stress in mice through attenuation of the mesocortical dopaminergic pathway (Tanaka et al., 2012). Because PGD2 is known to act as a major sleep-promoting factor through the D prostanoid receptor DP1 (Urade and Hayaishi, 2011), PGD2 is believed to mediate sleepiness, a major aspect of the sickness syndrome; however, no function has previously been attributed to CRTH2 in the brain (Saper et al., 2012). Future studies should address if some of the behavioral changes observed in the present study could be influenced by PGD2-mediated sleepiness. Together with our present results, it appears that the PGE2 receptor (EP) and PGD2 receptors (DP1 and CRTH2) have specific neural circuits that are ascribed to distinct aspects of the sickness syndrome.
In the present study, we show that CRTH2 is involved in LPS-induced transient activation of the PVN, and more sustained activation of the CeA. Although the PVN is involved in the control of food intake (Parker and Bloom, 2012), the reduced c-Fos expression in PVN neurons in LPS-injected CRTH2−/− mice was not associated with differences in food intake. This may be because a rather strong inhibition of food intake by LPS might mask the difference in PVN activation. On the other hand, both the PVN and CeA are known to be crucial for the stress response, anxiety-like behavior, and cue-dependent fear conditioning (Herman and Cullinan, 1997; LeDoux, 2000; Tye et al., 2011). Therefore, it is possible that CRTH2 controls these emotional functions and is consequently critically involved in LPS-impaired social and object exploratory behavior. Future studies addressing the effects of local injection of the CRTH2 antagonists as well as studies using mice with ectopic deficiency of CRTH2 will greatly help to precisely understand the actions of CRTH2 in specific brain functions.
Recently, we reported that CRTH2 potentiates NGF-induced neurite outgrowth in PC12 cells, by enhancement of p38 MAPK phosphorylation (Hatanaka et al., 2010). In particular, a CRTH2-selective agonist, 13, 14-dihydro-15-keto PGD2 (DK-PGD2), activated p38 MAPK for 24 h in PC12 cells. p38 MAPK has been implicated in the induction of LPS-induced sickness behavior through serotonin transporter activation in mice (Zhu et al., 2010). A similar signaling mechanism may underlie CRTH2-dependent behavioral regulation, and this possibility warrants further investigation.
The observation that CRTH2 deficiency and CRTH2 antagonism normalized tumor cell inoculation-induced decreases in social and object exploratory behavior provides further evidence that CRTH2 is implicated in emotional aspects of sickness behavior, and implicates tumor cell inoculated mice as a new animal model to reflect psychiatric changes in tumor patients. It has been reported that depression, social withdrawal, loss of interest, and cognitive difficulties are accompanying symptoms in tumor patients (Kronfol and Remick, 2000; Dantzer et al., 2008). The involvement of CRTH2 in various psychiatric disturbances found in tumor patients, as well as in psychiatric disorders such as depression and schizophrenia, needs to be addressed in future studies to determine the role of CRTH2 as a possible therapeutic target.
Footnotes
This work was supported in part by Grants in Aid for Scientific Research (A) and (B), Young Scientists (B), Research Fellowships for Young Scientists from the Japan Society for the Promotion of Science (R. Haba and Y.O.), and the Funding Program for Next Generation World-Leading Researchers (H. Hashimoto). We thank Drs. Shinsaku Nakagawa and Naoki Okada at the Laboratory of Biotechnology and Therapeutics, Graduate School of Pharmaceutical Sciences, Osaka University, for supplying the colon 26 cells.
The authors declare no competing financial interests.
References
- Abe H, Takeshita T, Nagata K, Arita T, Endo Y, Fujita T, Takayama H, Kubo M, Sugamura K. Molecular cloning, chromosome mapping and characterization of the mouse CRTH2 gene, a putative member of the leukocyte chemoattractant receptor family. Gene. 1999;227:71–77. doi: 10.1016/S0378-1119(98)00599-X. [DOI] [PubMed] [Google Scholar]
- Angeli V, Staumont D, Charbonnier AS, Hammad H, Gosset P, Pichavant M, Lambrecht BN, Capron M, Dombrowicz D, Trottein F. Activation of the D prostanoid receptor 1 regulates immune and skin allergic responses. J Immunol. 2004;172:3822–3829. doi: 10.4049/jimmunol.172.6.3822. [DOI] [PubMed] [Google Scholar]
- Bluthé RM, Layé S, Michaud B, Combe C, Dantzer R, Parnet P. Role of interleukin-1beta and tumour necrosis factor-alpha in lipopolysaccharide-induced sickness behaviour: a study with interleukin-1 type I receptor-deficient mice. Eur J Neurosci. 2000;12:4447–4456. doi: 10.1111/j.1460-9568.2000.01348.x. [DOI] [PubMed] [Google Scholar]
- Chomczynski P, Sacchi N. Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem. 1987;162:156–159. doi: 10.1016/0003-2697(87)90021-2. [DOI] [PubMed] [Google Scholar]
- Dantzer R, O'Connor JC, Freund GG, Johnson RW, Kelley KW. From inflammation to sickness and depression: when the immune system subjugates the brain. Nat Rev Neurosci. 2008;9:46–56. doi: 10.1038/nrn2297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Paiva VN, Lima SN, Fernandes MM, Soncini R, Andrade CA, Giusti-Paiva A. Prostaglandins mediate depressive-like behaviour induced by endotoxin in mice. Behav Brain Res. 2010;215:146–151. doi: 10.1016/j.bbr.2010.07.015. [DOI] [PubMed] [Google Scholar]
- Elander L, Engström L, Hallbeck M, Blomqvist A. IL-1beta and LPS induce anorexia by distinct mechanisms differentially dependent on microsomal prostaglandin E synthase-1. Am J Physiol Regul Integr Comp Physiol. 2007;292:R258–R267. doi: 10.1152/ajpregu.00511.2006. [DOI] [PubMed] [Google Scholar]
- Fishkin RJ, Winslow JT. Endotoxin-induced reduction of social investigation by mice: interaction with amphetamine and anti-inflammatory drugs. Psychopharmacology. 1997;132:335–341. doi: 10.1007/s002130050353. [DOI] [PubMed] [Google Scholar]
- Frenois F, Moreau M, O'Connor J, Lawson M, Micon C, Lestage J, Kelley KW, Dantzer R, Castanon N. Lipopolysaccharide induces delayed FosB/DeltaFosB immunostaining within the mouse extended amygdala, hippocampus and hypothalamus, that parallel the expression of depressive-like behavior. Psychoneuroendocrinology. 2007;32:516–531. doi: 10.1016/j.psyneuen.2007.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haba R, Shintani N, Onaka Y, Wang H, Takenaga R, Hayata A, Baba A, Hashimoto H. Lipopolysaccharide affects exploratory behaviors toward novel objects by impairing cognition and/or motivation in mice: possible role of activation of the central amygdala. Behav Brain Res. 2012;228:423–431. doi: 10.1016/j.bbr.2011.12.027. [DOI] [PubMed] [Google Scholar]
- Hashimoto H, Shintani N, Tanaka K, Mori W, Hirose M, Matsuda T, Sakaue M, Miyazaki J, Niwa H, Tashiro F, Yamamoto K, Koga K, Tomimoto S, Kunugi A, Suetake S, Baba A. Altered psychomotor behaviors in mice lacking pituitary adenylate cyclase-activating polypeptide (PACAP) Proc Natl Acad Sci U S A. 2001;98:13355–13360. doi: 10.1073/pnas.231094498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hashimoto H, Hashimoto R, Shintani N, Tanaka K, Yamamoto A, Hatanaka M, Guo X, Morita Y, Tanida M, Nagai K, Takeda M, Baba A. Depression-like behavior in the forced swimming test in PACAP-deficient mice: amelioration by the atypical antipsychotic risperidone. J Neurochem. 2009;110:595–602. doi: 10.1111/j.1471-4159.2009.06168.x. [DOI] [PubMed] [Google Scholar]
- Hatanaka M, Shibata N, Shintani N, Haba R, Hayata A, Hashimoto H, Baba A. 15d-prostaglandin J2 enhancement of nerve growth factor-induced neurite outgrowth is blocked by the chemoattractant receptor- homologous molecule expressed on T-helper type 2 cells (CRTH2) antagonist CAY10471 in PC12 cells. J Pharmacol Sci. 2010;113:89–93. doi: 10.1254/jphs.10001SC. [DOI] [PubMed] [Google Scholar]
- Hattori S, Takao K, Tanda K, Toyama K, Shintani N, Baba A, Hashimoto H, Miyakawa T. Comprehensive behavioral analysis of pituitary adenylate cyclase-activating polypeptide (PACAP) knockout mice. Front Behav Neurosci. 2012;6:58. doi: 10.3389/fnbeh.2012.00058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herman JP, Cullinan WE. Neurocircuitry of stress: central control of the hypothalamo-pituitary-adrenocortical axis. Trends Neurosci. 1997;20:78–84. doi: 10.1016/S0166-2236(96)10069-2. [DOI] [PubMed] [Google Scholar]
- Hirai H, Tanaka K, Yoshie O, Ogawa K, Kenmotsu K, Takamori Y, Ichimasa M, Sugamura K, Nakamura M, Takano S, Nagata K. Prostaglandin D2 selectively induces chemotaxis in T helper type 2 cells, eosinophils, and basophils via seven-transmembrane receptor CRTH2. J Exp Med. 2001;193:255–261. doi: 10.1084/jem.193.2.255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson DR, O'Connor JC, Dantzer R, Freund GG. Inhibition of vagally mediated immune-to-brain signaling by vanadyl sulfate speeds recovery from sickness. Proc Natl Acad Sci U S A. 2005;102:15184–15189. doi: 10.1073/pnas.0507191102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato T, Kasai A, Mizuno M, Fengyi L, Shintani N, Maeda S, Yokoyama M, Ozaki M, Nawa H. Phenotypic characterization of transgenic mice overexpressing neuregulin-1. PLoS One. 2010;5:e14185. doi: 10.1371/journal.pone.0014185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawaguchi C, Isojima Y, Shintani N, Hatanaka M, Guo X, Okumura N, Nagai K, Hashimoto H, Baba A. PACAP-deficient mice exhibit light parameter-dependent abnormalities on nonvisual photoreception and early activity onset. PLoS One. 2010;5:e9286. doi: 10.1371/journal.pone.0009286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Konsman JP, Luheshi GN, Bluthé RM, Dantzer R. The vagus nerve mediates behavioural depression, but not fever, in response to peripheral immune signals; a functional anatomical analysis. Eur J Neurosci. 2000;12:4434–4446. doi: 10.1046/j.0953-816X.2000.01319.x. [DOI] [PubMed] [Google Scholar]
- Kostenis E, Ulven T. Emerging roles of DP and CRTH2 in allergic inflammation. Trends Mol Med. 2006;12:148–158. doi: 10.1016/j.molmed.2006.02.005. [DOI] [PubMed] [Google Scholar]
- Kronfol Z, Remick DG. Cytokines and the brain: implications for clinical psychiatry. Am J Psychiatry. 2000;157:683–694. doi: 10.1176/appi.ajp.157.5.683. [DOI] [PubMed] [Google Scholar]
- LeDoux JE. Emotion circuits in the brain. Annu Rev Neurosci. 2000;23:155–184. doi: 10.1146/annurev.neuro.23.1.155. [DOI] [PubMed] [Google Scholar]
- Liang X, Wu L, Hand T, Andreasson K. Prostaglandin D2 mediates neuronal protection via the DP1 receptor. J Neurochem. 2005;92:477–486. doi: 10.1111/j.1471-4159.2004.02870.x. [DOI] [PubMed] [Google Scholar]
- Marchese A, Sawzdargo M, Nguyen T, Cheng R, Heng HH, Nowak T, Im DS, Lynch KR, George SR, O'dowd BF. Discovery of three novel orphan G-protein-coupled receptors. Genomics. 1999;56:12–21. doi: 10.1006/geno.1998.5655. [DOI] [PubMed] [Google Scholar]
- Marvel FA, Chen CC, Badr N, Gaykema RP, Goehler LE. Reversible inactivation of the dorsal vagal complex blocks lipopolysaccharide-induced social withdrawal and c-Fos expression in central autonomic nuclei. Brain Behav Immun. 2004;18:123–134. doi: 10.1016/j.bbi.2003.09.004. [DOI] [PubMed] [Google Scholar]
- Matsuoka T, Hirata M, Tanaka H, Takahashi Y, Murata T, Kabashima K, Sugimoto Y, Kobayashi T, Ushikubi F, Aze Y, Eguchi N, Urade Y, Yoshida N, Kimura K, Mizoguchi A, Honda Y, Nagai H, Narumiya S. Prostaglandin D2 as a mediator of allergic asthma. Science. 2000;287:2013–2017. doi: 10.1126/science.287.5460.2013. [DOI] [PubMed] [Google Scholar]
- Matsuoka Y, Furuyashiki T, Bito H, Ushikubi F, Tanaka Y, Kobayashi T, Muro S, Satoh N, Kayahara T, Higashi M, Mizoguchi A, Shichi H, Fukuda Y, Nakao K, Narumiya S. Impaired adrenocorticotropic hormone response to bacterial endotoxin in mice deficient in prostaglandin E receptor EP1 and EP3 subtypes. Proc Natl Acad Sci U S A. 2003;100:4132–4137. doi: 10.1073/pnas.0633341100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minami T, Okuda-Ashitaka E, Mori H, Ito S, Hayaishi O. Prostaglandin D2 inhibits prostaglandin E2-induced allodynia in conscious mice. J Pharmacol Exp Ther. 1996;278:1146–1152. [PubMed] [Google Scholar]
- Mizoguchi A, Eguchi N, Kimura K, Kiyohara Y, Qu WM, Huang ZL, Mochizuki T, Lazarus M, Kobayashi T, Kaneko T, Narumiya S, Urade Y, Hayaishi O. Dominant localization of prostaglandin D receptors on arachnoid trabecular cells in mouse basal forebrain and their involvement in the regulation of non-rapid eye movement sleep. Proc Natl Acad Sci U S A. 2001;98:11674–11679. doi: 10.1073/pnas.201398898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagata K, Tanaka K, Ogawa K, Kemmotsu K, Imai T, Yoshie O, Abe H, Tada K, Nakamura M, Sugamura K, Takano S. Selective expression of a novel surface molecule by human Th2 cells in vivo. J Immunol. 1999;162:1278–1286. [PubMed] [Google Scholar]
- Naoi K, Kogure S, Saito M, Hamazaki T, Watanabe S. Differential effects of selective cyclooxygenase (COX)-1 and COX-2 inhibitors on anorexic response and prostaglandin generation in various tissues induced by zymosan. Biol Pharm Bull. 2006;29:1319–1324. doi: 10.1248/bpb.29.1319. [DOI] [PubMed] [Google Scholar]
- Ohinata K, Takagi K, Biyajima K, Fujiwara Y, Fukumoto S, Eguchi N, Urade Y, Asakawa A, Fujimiya M, Inui A, Yoshikawa M. Central prostaglandin D(2) stimulates food intake via the neuropeptide Y system in mice. FEBS Lett. 2008;582:679–684. doi: 10.1016/j.febslet.2008.01.050. [DOI] [PubMed] [Google Scholar]
- Parker JA, Bloom SR. Hypothalamic neuropeptides and the regulation of appetite. Neuropharmacology. 2012;63:18–30. doi: 10.1016/j.neuropharm.2012.02.004. [DOI] [PubMed] [Google Scholar]
- Pecchi E, Dallaporta M, Jean A, Thirion S, Troadec JD. mPGES-1 knock-out mice are resistant to cancer-induced anorexia despite the absence of central mPGES-1 up-regulation in wild-type anorexic mice. J Neuroimmunol. 2008;199:104–114. doi: 10.1016/j.jneuroim.2008.05.012. [DOI] [PubMed] [Google Scholar]
- Rogers DC, Fisher EM, Brown SD, Peters J, Hunter AJ, Martin JE. Behavioral and functional analysis of mouse phenotype: SHIRPA, a proposed protocol for comprehensive phenotype assessment. Mamm Genome. 1997;8:711–713. doi: 10.1007/s003359900551. [DOI] [PubMed] [Google Scholar]
- Saper CB, Romanovsky AA, Scammell TE. Neural circuitry engaged by prostaglandins during the sickness syndrome. Nat Neurosci. 2012;15:1088–1095. doi: 10.1038/nn.3159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satoh T, Moroi R, Aritake K, Urade Y, Kanai Y, Sumi K, Yokozeki H, Hirai H, Nagata K, Hara T, Utsuyama M, Hirokawa K, Sugamura K, Nishioka K, Nakamura M. Prostaglandin D2 plays an essential role in chronic allergic inflammation of the skin via CRTH2 receptor. J Immunol. 2006;177:2621–2629. doi: 10.4049/jimmunol.177.4.2621. [DOI] [PubMed] [Google Scholar]
- Schiltz JC, Sawchenko PE. Distinct brain vascular cell types manifest inducible cyclooxygenase expression as a function of the strength and nature of immune insults. J Neurosci. 2002;22:5606–5618. doi: 10.1523/JNEUROSCI.22-13-05606.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serrats J, Schiltz JC, García-Bueno B, van Rooijen N, Reyes TM, Sawchenko PE. Dual roles for perivascular macrophages in immune-to-brain signaling. Neuron. 2010;65:94–106. doi: 10.1016/j.neuron.2009.11.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spik I, Brénuchon C, Angéli V, Staumont D, Fleury S, Capron M, Trottein F, Dombrowicz D. Activation of the prostaglandin D2 receptor DP2/CRTH2 increases allergic inflammation in mouse. J Immunol. 2005;174:3703–3708. doi: 10.4049/jimmunol.174.6.3703. [DOI] [PubMed] [Google Scholar]
- Tanaka K, Shintani N, Hashimoto H, Kawagishi N, Ago Y, Matsuda T, Hashimoto R, Kunugi H, Yamamoto A, Kawaguchi C, Shimada T, Baba A. Psychostimulant-induced attenuation of hyperactivity and prepulse inhibition deficits in Adcyap1-deficient mice. J Neurosci. 2006;26:5091–5097. doi: 10.1523/JNEUROSCI.4376-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka K, Furuyashiki T, Kitaoka S, Senzai Y, Imoto Y, Segi-Nishida E, Deguchi Y, Breyer RM, Breyer MD, Narumiya S. Prostaglandin E2-mediated attenuation of mesocortical dopaminergic pathway is critical for susceptibility to repeated social defeat stress in mice. J Neurosci. 2012;32:4319–4329. doi: 10.1523/JNEUROSCI.5952-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teeling JL, Cunningham C, Newman TA, Perry VH. The effect of non-steroidal anti-inflammatory agents on behavioural changes and cytokine production following systemic inflammation: implications for a role of COX-1. Brain Behav Immun. 2010;24:409–419. doi: 10.1016/j.bbi.2009.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tye KM, Prakash R, Kim SY, Fenno LE, Grosenick L, Zarabi H, Thompson KR, Gradinaru V, Ramakrishnan C, Deisseroth K. Amygdala circuitry mediating reversible and bidirectional control of anxiety. Nature. 2011;471:358–362. doi: 10.1038/nature09820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urade Y, Hayaishi O. Prostaglandin D2 and sleep/wake regulation. Sleep Med Rev. 2011;15:411–418. doi: 10.1016/j.smrv.2011.08.003. [DOI] [PubMed] [Google Scholar]
- Yirmiya R. Endotoxin produces a depressive-like episode in rats. Brain Res. 1996;711:163–174. doi: 10.1016/0006-8993(95)01415-2. [DOI] [PubMed] [Google Scholar]
- Zhu CB, Lindler KM, Owens AW, Daws LC, Blakely RD, Hewlett WA. Interleukin-1 receptor activation by systemic lipopolysaccharide induces behavioral despair linked to MAPK regulation of CNS serotonin transporters. Neuropsychopharmacology. 2010;35:2510–2520. doi: 10.1038/npp.2010.116. [DOI] [PMC free article] [PubMed] [Google Scholar]