Abstract
Background
Postoperative cerebral complications (PCC) are common and serious postoperative complications for patients with Stanford type A aortic dissection (AAD). The aim of this study was to evaluate the risk factors for PCC in these patients and to provide a scientific basis for effective prevention of PCC.
Methods
In this retrospective case-control study, 125 patients with AAD who underwent thoracotomy in our department from October 2017 to October 2018 in the department of cardiovascular surgery, Fujian Medical University Union Hospital were divided into two groups: patients with PCC (n = 12), and patients without PCC (n = 113). The general clinical data, the types of corrective surgeries, the intraoperative situations, the postoperative complications, and the midterm outcomes of the patients were analyzed.
Results
The patients with PCC were significantly older than the patients without PCC (P = 0.016), and the incidence of the preoperative cerebral disease history in the patients with PCC was significantly higher than those of the PCC (−) group (P = 0.024). The Euro SCORE II of patients with PCC was dramatically higher than the patients without PCC (P = 0.005). There were significant differences between the two groups in terms of the duration of cardiopulmonary bypass (CPB) (P = 0.010) and the length of moderate hypothermic circulatory arrest (MHCA) combined with selective cerebral perfusion (SCP) (P = 0.000). The monitoring of rcSO2 indicated that there was significant difference between the two groups in terms of the bilateral baseline (P = 0.000). Patients with PCC were observed to have experienced significantly longer intubation times (P = 0.000), ICU stays (P = 0.001), and postoperative hospital stays (P = 0.009), and they also had dramatically higher rates of pulmonary infection (P = 0.000), multiple organ dysfunction syndrome (P = 0.041) and tracheotomy (P = 0.022) after surgeries. The duration of MHCA+SCP (OR:9.009, P = 0.034) and the average baseline value of rcSO2 (OR:0.080, P = 0.009) were ultimately identified as significant risk factors.
Conclusions
PCC has a serious influence on the prognoses of patients following surgical treatment with AAD. The duration of MHCA+SCP and the average baseline value of rcSO2 were the independent risk factors for PCC.
Keywords: Postoperative complications; Circulatory arrest, deep hypothermia induced; Oximetry; Aneurysm, dissecting
Background
Postoperative cerebral dysfunction is the most common complication after the surgery for Stanford type A aortic dissection (AAD), and it has a negative effect that cannot be ignored in the postoperative rehabilitation of patients with AAD. Although the intraoperative cerebral protection strategy has been modified in recent decades, the morbidity due to postoperative cerebral complications (PCC) have been reported to be 6.4%~ 16.9% in cardiovascular surgical centers around the world [1–3]. Therefore, it is extremely important for cardiovascular surgeons and ICU physicians to explore the pathogenesis of PCC. The aim of this study was to evaluate the risk factors of postoperative cerebral dysfunction in these patients and to provide a scientific basis for the effective prevention of PCC.
Methods
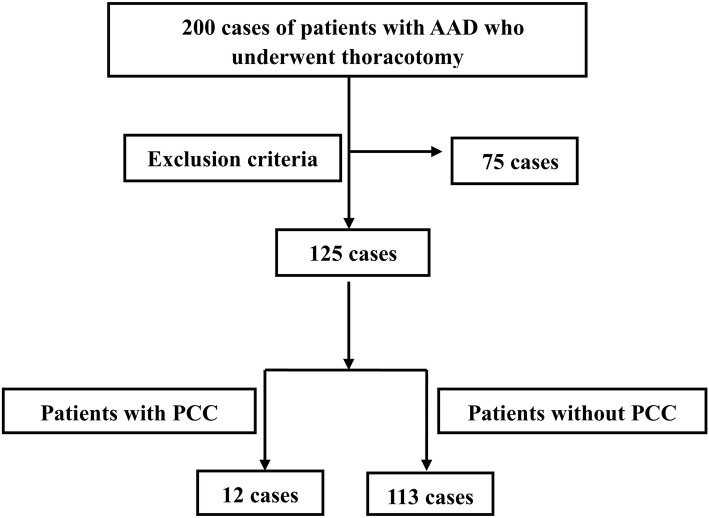
Two hundred consecutive patients with AAD who underwent thoracotomy in our department from October 2017 to October 2018 were enrolled in this retrospective case-control study. Then, the patients were divided into two groups: patients with PCC and patients without (Fig. 1). The general clinical data, types of corrective surgeries, intraoperative situations, postoperative complications, and midterm outcomes (12 months) of the patients were analyzed by telephone follow-up or out-patient review.
Fig. 1.
Flow Chart. One hundred and twenty-five patients with Stanford type A aortic dissection who underwent thoracotomy were selected from 200 patients based on the exclusion criteria, and they were divided into two groups: patients with PCC (n = 12), and patients without PCC (n = 113). Seventy-five patients were excluded from this study according to the exclusion criteria. AAD: Stanford type A aortic dissection; PCC: postoperative cerebral complications
Exclusion criteria
Patients who were younger than 18 years old.
Patients who were died before surgeries.
Patients with an abnormal preoperative consciousness.
Patients and/or their relatives who did not agree to participate in this clinical study.
Patients who were lost within the 12-month follow-up period.
Diagnostic criteria of the postoperative cerebral complications
The PCC included new-onset stroke, syncope, delirium, postoperative cognitive dysfunction (POCD), delayed emergence from anesthesia (DEA) and coma. The diagnosis of a stroke was based on the National Institutes of Health Stroke Scale (NIHSS) [4]. Syncope was defined as a transient loss of consciousness and was characterized by a rapid onset, short duration, and spontaneous complete recovery [5]. The confusion assessment method for the intensive care unit (CAM-ICU) [6] was applied for the evaluation of postoperative delirium (POD) and POCD. The Glasgow Coma Scale (GCS) [7] was used to objectively define coma in the two groups of patients after their surgeries. The patients whose response to stimulation occurred more than 60~90 min after the surgeries were recognized as having a delayed emergence from anesthesia [8], and this diagnosis should exclude the possibility of the other types of cerebral dysfunction mentioned above.
All of the diagnoses of PCC were confirmed by two experienced neurologists. Deeply sedated patients, confirmed by the monitoring sedation status system on the Richmond Agitation Sedation Scale (RASS) [9], did not receive an evaluation of cerebral function until they recovered from anesthesia.
Protocols of anesthesia
Combined intravenous-inhalation anesthesia was applied in the patients. The nasopharyngeal temperature and the rectal temperature were also monitored. Transesophageal echocardiography (TEE) was applied to monitor the intraoperative hemodynamics. Autologous blood transfusion was used to reduce the need for allogeneic blood transfusion. The balances of cerebral oxygen metabolism of the patients were measured by regional cerebral oxygen saturation (rcSO2) with the Regional Oximetry System (INVOS™ 5100C, Medtronic, USA). The bispectral index (BIS), which was measured by the BIS Monitoring System (VISTA, USA), was used to measure the depth of anesthesia.
Mechanical ventilation was performed after endotracheal intubation and was applied to provide end tidal CO2 volumes (PetCO2) of 35~40 mmHg, a FiO2 of 50~100%, a tidal volume of 6~8 ml/kg, a respiratory rate of 10~12 breaths/min and an adequate positive end-expiratory pressure (PEEP). The maintenance of anesthesia was implemented by an intravenous injection of 4~8 mg/kg/h propofol and 1~2 μg/kg/h sufentanil along with intermittent intravenous injections of cisatracurium.
During the surgery, the values of scSO2 were maintained at or above 70% of the baseline threshold. Cerebral desaturation was defined as a decrease in the saturation value below the absolute value of 50% or 70% of the baseline for 15 s. The mean and minimum values of the rcSO2, as well as the area under the curve (AUC) of the rcSO2 values below the line of the previously mentioned cerebral desaturation values, were collected for further analysis.
Surgical procedures
During surgery, A sternal incision was performed. To establish cardiopulmonary bypass (CPB), the arterial cannula was placed in the right femoral artery and/or the right axillary artery, and the drainage tube was placed in the right atrium. The CPB flow rate was 2.4~2.6 L/kg/min. The intermittent cold blood cardioplegia was perfused through the left and right coronary arteries for myocardial protection. The following procedures, including the triple-branched stent graft implantation [10] and the Sun’s procedures were described in the previous literature [11].
Protocol to address intraoperative cerebral desaturation
The positions of the patients were checked first to exclude the compression of the cervical great vessels when the intraoperative cerebral desaturation occurred. Then, the parameters of the mechanical ventilator (before or after CPB) or the oxygenator (during CPB) were adjusted to maintain the arterial pressure of CO2 (PaCO2) above 40 mmHg. Metaraminol or noradrenaline was used to elevate the mean arterial pressure (MAP) to above 60 mmHg. A cardiotonic was administered if there was a poor cardiac index (below 2.0 L/m2/min), and adequate blood volume was confirmed by TEE. The BIS value was maintained at or below 50 during the CPB to ensure that patients were in a state of deep anesthesia. Other methods to prevent cerebral hypoxia included increasing the pump flow, increasing the FiO2 and performing allogeneic blood transfusion.
Follow-up
Telephone contact or out-patient review with the patients was maintained after discharge. Every month in the first year, the patients received echocardiographies, chest radiographies, and bilateral carotid artery Doppler examinations. At 1 month and 3 months after surgery, the patients received aortic computed tomography angiography (CTA) examinations, which were then performed annually.
Statistical analysis
SPSS software (version 19.0, IBM, USA) was used for the statistical analyses. Descriptive statistical analyses, as well as Wilcoxon rank sum tests, were used to analyze the measurement data. The chi-square test or Fisher’s exact test were used to analyze the numerical data. Multivariate logistic regression analysis was used to analyze the count data. Statistical significance was defined as P < 0.05.
Results
General clinical data
Seventy-five patients were excluded from this study after meeting the exclusion criteria described above, including 9 cases who were younger than 18 years old, 42 cases with an abnormal preoperative consciousness, 8 cases died before surgeries, 13 cases who did not agree to participate in this clinical study, 3 cases who were lost within the 12-month follow-up period. One hundred twenty-five cases were included after applying the exclusion criteria. Then, the patients were divided into two groups: patients with PCC (n = 12) and patients without (n = 113).
The primary analyses revealed that the patients in the PCC (+) group were significantly older than the patients in the PCC (−) group (58.4 ± 8.4 years vs 49.4 ± 13.4 years, P = 0.016), and the incidence of preoperative cerebral disease history in the patients of the PCC (+) group was significantly higher than that of the PCC (−) group (16.7% vs 0.9%, P = 0.024). The Euro SCORE II of patients in the PCC (+) was dramatically higher than the patients of the PCC (−) group (11.9 ± 2.7 vs 9.0 ± 3.4, P = 0.005). There were no significant differences between the patients in the two groups in terms of gender, body mass index (BMI), personal history, underlying diseases (except for preoperative cerebral disease history), New York Heart Association (NYHA) class, etiologies, ultrasound cardiogram (UCG) results, preoperative complications due to aortic dissection, scale of aortic dissection and American Society of Anesthesiologists (ASA) class (Table 1).
Table 1.
Clinical data
| Category of clinical data | PCC (+) n = 12 |
PCC (−) n = 113 |
P |
|---|---|---|---|
| Age | 58.4 ± 8.4 | 49.4 ± 13.4 | 0.016 |
| Gender | 1.000 | ||
| Male | 11 (91.7%) | 96(85.0%) | |
| Female | 1(8.3%) | 17(15.0%) | |
| BMI | 24.1 ± 2.8 | 24.4 ± 3.3 | 0.987 |
| Active smoking | 6(50.0%) | 48(42.5%) | 0.761 |
| Alcoholism | 1(8.3%) | 11(9.7%) | 1.000 |
| Underlying diseases | |||
| Diabetes | 0(0.0%) | 2(1.8%) | 1.000 |
| CAD | 0(0.0%) | 1(0.9%) | 1.000 |
| Cardiac reoperation | 1(8.3%) | 2(1.8%) | 0.263 |
| Renal dysfunction | 1(8.3%) | 3(2.7%) | 0.336 |
| History of cerebral diseases | 2(16.7%) | 1(0.9%) | 0.024 |
| History of anemia | 1(8.3%) | 7(6.2%) | 0.565 |
| NYHA class | |||
| I | 0(0.0%) | 29(25.7%) | 0.078 |
| II | 10(83.3%) | 65(57.5%) | |
| III | 1(8.3%) | 16(14.2%) | |
| IV | 1(8.3%) | 3(2.7%) | |
| Etiologies | |||
| Hypertension | 7(58.3%) | 87(77.0%) | 0.291 |
| Others | 5(41.7%) | 26(23.0%) | |
| UCG | |||
| EF (%) | 61.6 ± 8.3 | 60.8 ± 7.9 | 0.725 |
| Pericardial effusiona | 0(0.0%) | 5(4.4%) | 1.000 |
| Aortic regurgitationb | 1(8.3%) | 17(15.0%) | 1.000 |
| Preoperative complications | |||
| AMIc | 1(8.3%) | 4(3.5%) | 0.402 |
| Lower limb ischemia | 0(0.0%) | 16(14.2%) | 0.361 |
| Mesenteric artery infarctiond | 0(0.0%) | 7(6.2%) | 1.000 |
| The scale of aortic dissection | |||
| Ascending aorta | 1(8.3%) | 4(3.5%) | 0.402 |
| Aortic arch | 5(41.7%) | 48(42.5%) | 1.000 |
| Innominate artery | 5(41.7%) | 23(20.4%) | 0.138 |
| Right common carotid artery | 4(33.3%) | 14(12.4%) | 0.071 |
| Left subclavian artery | 2(16.7%) | 10(8.8%) | 0.323 |
| ASA status | |||
| I | 0(0.0%) | 0(0.0%) | 1.000 |
| II | 0(0.0%) | 0(0.0%) | |
| III | 0(0.0%) | 0(0.0%) | |
| IV | 11(91.7%) | 10.5(92.9%) | |
| V | 1(8.3%) | 8(7.1%) | |
| VI | 0(0.0%) | 0(0.0%) | |
| Euro SCORE II | 11.9 ± 2.7 | 9.0 ± 3.4 | 0.005 |
The results demonstrated that the patients in the PCC(+) group were significantly older than the patients in the PCC(−) group, and the incidence of the preoperative cerebral disease history in the patients of the PCC(+) group was significantly higher than those of the PCC(−) group. The Euro SCORE II of patients in the PCC(+) group was dramatically higher than the patients of the PCC(−) group
PCC Postoperative cerebral complications, BMI Body mass index, CAD Coronary artery disease, NYHA New York Heart Association, UCG Ultracardiography, EF Ejection fraction, AMI Acute myocardial infarction, ASA American Society of Anesthesiologists
aserious pericardial effusion; b serious aortic regurgitation; c clinical manifestations, ECG, and contents of creatine kinase and troponin in the serum that were consistent with the diagnostic criteria for acute myocardial infarction; d confirmed by superior mesenteric artery angiography
Surgical and perioperative treatments
The chi-square test revealed that there were no significant differences in the types of surgical procedures between the patients in the two groups. The Wilcoxon rank sum tests also indicated that there were significant differences between the two groups in terms of the duration of CPB (165.4 ± 42.5 min vs 134.8 ± 21.5 min, P = 0.010) and the length of moderate hypothermic circulatory arrest combined with selective cerebral perfusion (MHCA + SCP) (20.3 ± 2.1 min vs 15.5 ± 2.7 min, P = 0.000). The monitoring of rcSO2 indicated that there were no significant differences between the two groups in terms of intraoperative rcSO2 except for the average value of bilateral rcSO2 at baseline (48.2 ± 3.3% vs 66.7 ± 11.7%, P = 0.000). However, the duration of surgery, aortic cross-clamping, the mean value of the intraoperative BIS index, the volumes of blood loss and perioperative allogeneic transfusion were not significantly different in the patients in these two groups (Table 2).
Table 2.
Surgical and perioperative treatments
| Category of perioperative treatments | PCC (+) n = 12 |
PCC (−) n = 113 |
P |
|---|---|---|---|
| Types of surgical correction | |||
| Aortic sinus reconstruction | 3(25.0%) | 17(15.0%) | 0.406 |
| Bentall | 1(8.3%) | 11(9.7%) | 1.000 |
| Wheat | 0 (0.0%) | 2(1.8%) | 1.000 |
| Hemiarch replacement | 12(100%) | 106(93.8%) | 1.000 |
| Ascending aorta replacement | 12(100%) | 105(92.9%) | 1.000 |
| CABG | 0(0.0%) | 1(0.9%) | 1.000 |
| Intraoperative conditions | |||
| Surgery (min) | 303.6 ± 43.0 | 282.4 ± 55.9 | 0.113 |
| CPB (min) | 165.4 ± 42.5 | 134.8 ± 21.5 | 0.010 |
| Aortic cross-clamping (min) | 50.8 ± 7.6 | 46.8 ± 7.9 | 0.064 |
| MHCA+ SCP (min) | 20.3 ± 2.1 | 15.5 ± 2.7 | 0.000 |
| Blood loss (ml) | 475.0 ± 256.3 | 374.8 ± 166.5 | 0.059 |
| Avg rcSO2 baseline (%)a | 48.2 ± 3.3 | 66.7 ± 11.7 | 0.000 |
| Avg rcSO2 (%)(L) | 56.5 ± 8.2 | 58.7 ± 9.9 | 0.505 |
| Avg rcSO2 (%)(R) | 57.3 ± 7.1 | 59.6 ± 11.7 | 0.560 |
| rcSO2 minimum (%) | 50.6 ± 7.8 | 54.7 ± 7.3 | 0.077 |
| Total time of rcSO2 < 70% baseline and > 15 s (n) | 4.6 ± 1.8 | 3.6 ± 2.4 | 0.174 |
| Total time of rcSO2 < 50% and > 15 s (n) | 1.7 ± 1.0 | 1.3 ± 1.6 | 0.099 |
| AUC of rcSO2 < 70% (%min)(L) | 150.3 ± 63.0 | 121.2 ± 75.5 | 0.227 |
| AUC of rcSO2 < 70% (%min)(R) | 120.3 ± 117.0 | 98.5 ± 63.5 | 0.917 |
| AUC of rcSO2 < 50% (%min)(L) | 86.6 ± 62.7 | 61.4 ± 64.1 | 0.145 |
| AUC of rcSO2 < 50% (%min)(R) | 52.7 ± 32.3 | 45.9 ± 49.7 | 0.303 |
| Avg BIS index | 36.8 ± 10.3 | 39.8 ± 9.3 | 0.320 |
| Perioperative allogeneic transfusion | |||
| RBC (u) | 2.7 ± 1.8 | 3.1 ± 2.8 | 0.727 |
| PLT (u) | 0.8 ± 1.2 | 1.2 ± 1.9 | 0.201 |
| FFP (ml) | 366.7 ± 602.0 | 313.7 ± 345.8 | 0.666 |
| CP (U) | 1.0 ± 2.3 | 1.5 ± 2.8 | 0.528 |
The results demonstrated that there were significant differences between the two groups in terms of the duration of CPB and MHCA+SCP. The monitoring of rcSO2 indicated that there were no significant differences between the two groups in terms of intraoperative rcSO2 except for the average value of bilateral rcSO2 at baseline
PCC Postoperative cerebral complications, CABG Coronary artery bypass grafting, CPB Cardiopulmonary bypass, MHCA Moderate hypothermic circulatory arrest, SCP Selective cerebral perfusion, rcSO2 regional cerebral oxygen saturation, AUC Area under the curve, RBC Red blood cell, PLT Platelet, FFP Fresh frozen plasma, CP Cryoprecipitation
athe baseline value of rcSO2 from the bilateral brain before anesthesia induction
Postoperational situation
Twelve (12/125, 9.6%) patients with postoperative cerebral complications were observed. The Wilcoxon rank sum tests showed that the anesthesia recovery periods of the patients in the PCC (+) group were shorter than those of the patients in the PCC (−) group, but there was no significant difference. Two cases of newly onset stroke confirmed by computed tomography (CT) or magnetic resonance imaging (MRI) (2/125, 1.6%), 1 case of syncope (1/125, 0.8%), 2 cases of POD (2/125,1.6%), 3 cases of POCD (3/125, 2.4%), 2 cases of DEA (2/125, 1.6%), 2 cases of coma (2/125, 1.6%) and 2 cases of postoperative paraplegia were observed in this study. Patients from the PCC (+) group were observed to have experienced significantly longer intubation times (69.3 ± 28.8 h vs 33.3 ± 24.2 h, P = 0.000), ICU stays (127.3 ± 72.0 h vs 63.5 ± 51.3 h, P = 0.001), and postoperative hospital stays (32.0 ± 16.7 d vs 21.2 ± 13.0 d, P = 0.009), and they also had dramatically higher rates of pulmonary infection (50.0% vs 7.1%, P = 0.000), multiple organ dysfunction syndrome (MODS) (25.0% vs 5.3%, P = 0.041), gastrointestinal complications (25% vs 5.3%, P = 0.041) and tracheotomy (33.3% vs 8.0%, P = 0.022) after surgery. However, no significant differences were observed in the non-neurological complications, including reoperations for bleeding, heart dysfunction, myocardial infarction, lethal arrhythmia, renal insufficiency, wound infection, or sepsis between the patients in these two groups. The rates of the use of extracorporeal membrane oxygenation (ECMO) were confirmed to be approximately equal between the two groups (Table 3).
Table 3.
Short term outcomes and hospital costs
| Category | PCC (+) n = 12 |
PCC (−) n = 113 |
P |
|---|---|---|---|
| Anesthesia recovery period (h)a | 19.5 ± 33.0 | 10.2 ± 4.9 | 0.072 |
| New onset stroke | 2(16.7%) | 0(0.0%) | / |
| Syncope | 1(8.3%) | 0(0.0%) | / |
| POD | 2(16.7%) | 0(0.0%) | / |
| POCD | 3(25.0%) | 0(0.0%) | / |
| DEA | 2(16.7%) | 0(0.0%) | / |
| Coma | 2(16.7%) | 0(0.0%) | / |
| Total of cerebral complications | 12(100.0%) | 0(0.0%) | / |
| Paraplegia | 1(8.3%) | 1(0.9%) | 0.183 |
| Reoperation for bleeding | 0(0.0%) | 2(1.8%) | 1.000 |
| Heart dysfunction b | 0(0.0%) | 7(6.2%) | 1.000 |
| Myocardial infarction | 0(0.0%) | 2(1.8%) | 1.000 |
| Lethal arrhythmia | 0(0.0%) | 1(0.9%) | 1.000 |
| Renal insufficiencyc | 1(8.3%) | 12(10.6%) | 1.000 |
| Pulmonary infection | 6(50.0%) | 8(7.1%) | 0.000 |
| Gastrointestinal complicationsd | 3(25.0%) | 6(5.3%) | 0.041 |
| Wound infection | 0(0.0%) | 2(1.8%) | 1.000 |
| Sepsis | 1(8.3%) | 5(4.4%) | 0.461 |
| ARDS | 1(8.3%) | 3(2.7%) | 0.336 |
| MODS | 3(25.0%) | 6(5.3%) | 0.041 |
| ECMO assistance | 0(0.0%) | 2(1.8%) | 1.000 |
| Thoracic drainagee | 534.2 ± 435.9 | 471.9 ± 504.3 | 0.574 |
| Intubation time(h) | 69.3 ± 28.8 | 33.3 ± 24.2 | 0.000 |
| Tracheotomy | 4(33.3%) | 9(8.0%) | 0.022 |
| Length of ICU stay(h) | 127.3 ± 72.0 | 63.5 ± 51.3 | 0.001 |
| Length of hospital stay(d) | 32.0 ± 16.7 | 21.2 ± 13.0 | 0.009 |
| Mortality in hospital | 1(8.3%) | 3(2.7%) | 0.336 |
| Mortality after discharge | 1(8.3%) | 0(0.0%) | 0.096 |
| Mortality after surgeryf | 2(16.7%) | 3(2.7%) | 0.072 |
| Hospital costs (RMB) | 272,911.0 ± 60,495.8 | 224,651.5 ± 61,219.9 | 0.015 |
Patients from the PCC(+) group were observed to have experienced significantly longer durations of intubation times, ICU stays, and postoperative hospital stays, and they also had dramatically higher rates of pulmonary infection, MODS and tracheotomy after surgery. The postoperative mortalities of the PCC(+) group had a trend of increasing, but there were no significant differences between the patients in these two groups. Patients from the PCC(+) group spent more money compared with the patients in the PCC(−) group
PCC Postoperative cerebral complications, POD Postoperative delirium, POCD Postoperative cognitive dysfunction, DEA Delayed emergence from anesthesia, ARDS Acute respiratory distress syndrome, MODS Multiple organ dysfunction syndrome, ECMO Extracorporeal membrane oxygenation
atwo patients with postoperative comas were not enrolled in the analysis of recovery times; b severe heart failure reached NYHA grades III-IV; c required renal replacement therapy; d included meteorism, nausea, vomiting, abdominal pain, diarrhea, constipation, and gastrointestinal hemorrhage; e within 48 h after surgery; f up to the end of the follow-up period
Five cases of death (5/125, 4.0%) were observed in this study, including 4 cases that occurred in the hospital and 1 case that occurred after discharge. Three of these deaths were attributed to multiple organ dysfunction syndrome (MODS) and 2 were attributed to low cardiac output syndrome (LCOS). Fisher’s exact test showed that there was no significant difference in the mortalities of the patients in the two groups, but we observed an ascending trend in mortality after the surgeries in the patients of the PCC (+) group compared with the patients in the PCC (−) group (16.7% vs 2.7%, P = 0.072). Wilcoxon rank sum tests also showed that patients in the PCC (+) group spent remarkably more money than the patients in the PCC (−) group (272,911.0 ± 60,495.8 RMB vs 224,651.5 ± 61,219.9 RMB, P = 0.015) (Table 3).
The results of multivariate logistic regression analysis
A total of 6 variables including age, history of the cerebral diseases, Euro SCORE II, duration of CPB, duration of MHCA+SCP and average rcSO2 bilateral baseline were analyzed in the stepwise logistic regression model to identify the independent risk factors for PCC. The multivariate logistic regression analysis showed that the duration of MHCA+SCP and the average baseline value of rcSO2 from the bilateral brain before anesthesia induction were the independent risk factors for PCC (OR: 9.009, P = 0.034 and OR: 0.080, P = 0.009) (Table 4).
Table 4.
Multivariate logistic regression analysis
| Risk factors | B | S.E. | Wald | df | Sig. | Exp(B) |
|---|---|---|---|---|---|---|
| Age | 0.454 | 0.652 | 0.484 | 1 | 0.486 | 1.574 |
| History of cerebral diseases | −7.529 | 23.720 | 0.101 | 1 | 0.751 | 0.001 |
| Euro SCORE II | 1.376 | 0.704 | 3.822 | 1 | 0.051 | 3.958 |
| CPB (min) | 0.498 | 0.518 | 0.925 | 1 | 0.336 | 1.646 |
| MHCA + SCP (min) | 2.198 | 1.038 | 4.483 | 1 | 0.034 | 9.009 |
| Avg rcSO2 at baselinea | −2.527 | 0.963 | 6.889 | 1 | 0.009 | 0.080 |
The duration of MHCA+SCP and the average value of rcSO2 at baseline were ultimately identified as significant risk factors (OR: 9.009, P = 0.034 and OR: 0.080, P = 0.009)
CPB Cardiopulmonary bypass, MHCA Moderate hypothermic circulatory arrest, SCP Selective cerebral perfusion, rcSO2 regional cerebral oxygen saturation
athe average baseline value of rcSO2 from the bilateral brain before anesthesia induction
Follow-up
Three patients were lost within the 12-month follow-up period and were excluded from this study. One incidence of death after discharge was observed in the PCC (+) group and that was attributed to recurrence of the aortic dissection.
Discussion
Although the technology of cerebral protection has been improved in the last few decades, the morbidity of PCC after surgery for AAD occurs at such a high frequency that we cannot ignore the harmful impact of it. In this study, patients with PCC had dramatically higher rates of pulmonary infection and tracheotomy after surgery, and this was relevant with the longer duration of postoperative intubation. Considering the impaired mental state and the poor spontaneous respiration of the patients, the physicians in the ICU prefer the prolonged assistance of mechanical ventilation in these patients after the surgeries for AAD. Therefore, it is almost inevitable that the incidences of postoperative ventilator-associated pneumonia (VAP) and tracheotomy would increase.
A prolonged duration of mechanical ventilation assistance would certainly result in a prolonged ICU and hospital stays. A longer duration of ICU stay and ventilatory support would lead to a higher risk of multiple drug-resistant bacterial infection and multiple organ dysfunction. In addition, it has been reported that the incidence of MODS in patients with severe stroke reached up to 34% [12]. Moreover, more elderly patients were observed in the PCC (+) group, which could be another risk factor for postoperative MODS. Therefore, it made sense that patients with PCC had a high rate of MODS.
A higher incidence of gastrointestinal complications was also observed in the patients in the PCC (+) group after surgery in this study. The probable reasons are: 1. Neuromodulatory dysfunction as a result of cerebral ischemia, which could impair the hypothalamic-pituitary-adrenal axis and the sympathetic nervous system and finally destroy the gastrointestinal barrier function [13–15]; 2. Postoperative therapy of antiplatelet agents, anticoagulants and steroids, which are frequently applied to the patients after surgery for AAD; 3. Pulmonary infection, which was reported to be associated with development of gastrointestinal bleeding after acute stroke [16]; 4. Older age, which is another important risk factor for upper gastrointestinal bleeding according to one report [17]. Furthermore, because of the higher mortality due to pulmonary infection, MODS and GI complications which dramatically prolong the duration of ICU and hospital stay, the patients with PCC logically spent more money in hospital compared with the patients without PCC.
The survival rates of the patients after the surgeries between the two groups had no significant differences at the time of hospitalization and discharge. Nevertheless, we could find an increasing trend in mortality rates of the patients in the PCC (+) group. Therefore, we speculated that PCC plays an important role in the prognosis of patients after surgery for AAD, and this could be attributed to the high rates of postoperative pulmonary infection, gastrointestinal complications and MODS in the patients with PCC. Therefore, it is vital to evaluate the risk factors for PCC in these patients and to provide a scientific basis for an effective preventive strategy.
As we know, the main mechanism of postoperative cerebral dysfunction is an imbalance of oxygen metabolism in the brain which results from microemboli, hypothermia, hypoperfusion, hyperglycemia, reperfusion injury (RI), narcotics and internal environment disturbance. Moreover, a majority (73%) of the patients in China were reported to be affected by a lack of an integral circle of Willis [18], and the intracranial artery atherosclerosis caused by uncontrolled hypertension, hyperglycemia, hypercholesterolemia, and obesity, which frequently occur in elderly patients and patients with a history of cerebral disease, may further impair the insufficient cerebrovascular cross-circulation [19–21]. Therefore, SCP will not meet the oxygen demands of the different regions of the brain.
The analysis in this study confirmed that the age, history of the cerebral disease, duration of CPB, duration of HCA + SCP and rcSO2 baseline were the potential risk factors of PCC during the preliminary statistical analysis in this study. Moreover, the multivariate logistic regression analysis revealed that the duration of MHCA+SCP and the average baseline value of rcSO2 from the bilateral brain before anesthesia induction were the independent risk factors for PCC. This indicated that anoxia, hypothermia, hypoperfusion and RI could conceivably play significant roles in the occurrence of PCC. Since the branches of the aortic arch are frequently implicated, the low value of rcO2 at baseline was considered to be a part of the systematic malperfusion in the patients with AAD. It was reported that a low value of rcO2 at baseline is an important predictive factor for the prognosis of patients who undergo cardiovascular surgeries. Although the rcO2 at baseline has a weak relationship (OR = 0.080) with PCC in our study, surgeons and anesthesiologists should pay more attention to patients with a low value of rcO2 before induction. Currently, it is nearly impossible to doubt that cerebral malperfusion during the circulatory arrest seriously damages the function of the central nervous system (CNS) and the RI following the MHCA will make the damage worse because of the widely disseminated inflammation in the brain. To alleviate ischemic injury, the surgeons always try their best to shorten the duration of circulatory arrest by modifying the surgical procedures and reducing the patient’s core body temperature to an appropriate range. Eight years ago, our department first reported a novel surgical procedure, the triple-branched stented graft implantation, which could be applied to cure AAD with a relatively short duration of systemic circulatory arrest and without a large amount of intraoperative blood loss [22].
However, there are some keypoints which should not be ignored by surgeons: first, the hypothermia itself and CPB methods may depress the regulatory functions of blood flow in the brain and may induce cerebral ischemia during the process of the SCP. A normal level of perfusion pressure or blood flow rate may mask the malperfusion of the CNS [23]; second, in addition to the side effects of the anesthetics, intraoperative stress, and internal environment imbalance, we should also consider the disadvantage of the monitoring of rcSO2 in that this monitoring only focuses on the imbalance of oxygen metabolism in the bilateral frontal lobe, and it is not sensitive to the abnormal statuses in the other regions of the brain outside of the frontal lobe. It would be much more appropriate to use a whole-brain oxygen saturation monitoring method or a real-time detection of the bilateral cerebrovascular blood flow by transcranial Doppler sonography (TCD) during the surgery [24]; and Third, cerebral hyperperfusion should be avoided.
Intraoperative monitoring of rcSO2 was traditionally considered to be a fairly reliable forecasting and preventive measure of PCC, and in this study, the average value of rcSO2 baselines was found to be significantly lower in the patients with PCC compared with the patients in the PCC(−) group. However, the same tendency of rcSO2 did not occur during the surgeries in this study. Does that mean that the monitoring of rcSO2 during the aortic surgeries is unnecessary? Obviously, the answer is no. In the course of aortic surgeries, we have observed several dramatic drops of rcSO2, and an emergency plan was initiated immediately to deal with the anoxia in the brain. Afterwards, the cerebral oxygenation would greatly improve within 5~10 min for approximately 70% patients who underwent aortic surgeries. This revealed that the intraoperative monitoring of rcSO2 could provide a much earlier warning indication of cerebral hypoxia comparing with conventional monitoring methods. Therefore, rcSO2 is considered to be an essential monitoring means during surgeries for AAD.
This study has several limitations. First, this retrospective case-control and single-center study could not ensure that the patients from the two groups received uniform therapeutic strategies. The selection bias from the subjective judgment of the physicians should not be ignored. Second, due to the relatively small sample size, the absolute quantity of the patients with PCC was relatively small and might reduce the statistical efficiency. Third, we could not exclude the possibility of underestimated mortalities, which were attributed to the relatively short duration of the follow-up period. Therefore, larger sample sizes and more randomized, controlled tests are required for further validation of these procedures.
Conclusions
PCC has a serious influence on the prognoses of patients who undergo surgical treatment for AAD. The duration of MHCA+SCP and the average baseline value of rcSO2 from the bilateral brain before anesthesia induction were the risk factors for PCC. Surgeons and anesthesiologists should pay more attention to the patients with low values of rcO2 before induction, and shortening the length of MHCA+SCP may be an effectively prophylactic measure to reduce the morbidity of PCC in the patients with AAD. Larger sample sizes and more randomized, controlled tests are required for further validation of these procedures.
Acknowledgments
The authors thank the patients who participated in the study and the research assistants and study coordinators who assisted with data collection and management of the study, including Qian-Zhen Li, Guan-Hua Fang, Liang-Liang Yan, Sheng Chen, Yun-Nan Hu, Ling-Fen Li, Jun-Jia He and Jia-Xing Zhang.
Abbreviations
- AAD
Stanford type A aortic dissection
- AMI
Acute myocardial infarction
- ARDS
Acute respiratory distress syndrome
- ASA
American Society of Anesthesiologists
- AUC
Area under the curve
- BIS
Bispectral index
- BMI
Body mass index
- CAD
Coronary artery disease
- CAM-ICU
Confusion assessment method for the intensive care unit
- CNS
Central nervous system
- CP
Cryoprecipitation
- CPB
Cardiopulmonary bypass
- DEA
Delayed emergence from anesthesia
- ECMO
Extracorporeal membrane oxygenation
- EF
Ejection fraction
- FFP
Fresh frozen plasma
- GCS
Glasgow Coma Scale
- LCOS
Low cardiac output syndrome
- MAP
Mean arterial pressure
- MHCA
Moderate hypothermic circulatory arrest
- MODS
Multiple organ dysfunction syndrome
- NIHSS
National Institutes of Health Stroke Scale
- NYHA
New York Heart Association
- PCC
Postoperative cerebral complications
- PEEP
Positive end-expiratory pressure
- PetCO2
End tidal CO2 volumes
- PLT
Platelet
- POCD
Postoperative cognitive dysfunction
- POD
Postoperative delirium
- RASS
Richmond Agitation Sedation Scale
- RBC
Red blood cell
- rcSO2
regional cerebral oxygen saturation
- RI
Reperfusion injury
- SCP
Selective cerebral perfusion
- TCD
Transcranial Doppler sonography
- TEE
Transesophageal echocardiography
- UCG
Ultrasound cardiogram
- VAP
Ventilator-associated pneumonia
Authors’ contributions
MF-C and YL performed the conception and design. LW-C supported the administration. YL and RM-L provided the study materials and patients’ informations. MF-C and YL collected and assembled the data of patients. MF-C and YL analysed the data and performed the interpretation. YL wrote the manuscript. All authors read and approved the final manuscript.
Funding
Not applicable.
Availability of data and materials
The datasets generated and/or analysed during the current study are not publicly available due to the confidentiality agreement of our institution but are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The Ethics Committees of Fujian Medical University Union Hospital approved this retrospective study, and written informed consent was obtained from all patients.
Consent for publication
Written informed consent for publication was obtained from all participants.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Di Bartolomeo R, Berretta P, Pantaleo A, Murana G, Cefarelli M, Alfonsi J, et al. Long-term outcomes of open arch repair after a prior aortic operation: our experience in 154 patients. Ann Thorac Surg. 2017;103:1406–1412. doi: 10.1016/j.athoracsur.2016.08.090. [DOI] [PubMed] [Google Scholar]
- 2.Cefarelli M, Murana G, Surace GG, Castrovinci S, Jafrancesco G, Kelder JC, et al. Elective aortic arch repair: factors influencing neurologic outcome in 791 patients. Ann Thorac Surg. 2017;104:2016–2023. doi: 10.1016/j.athoracsur.2017.05.009. [DOI] [PubMed] [Google Scholar]
- 3.Czerny M, Krähenbühl E, Reineke D, Sodeck G, Englberger L, Weber A, et al. Mortality and neurologic injury after surgical repair with hypothermic circulatory arrest in acute and chronic proximal thoracic aortic pathology: effect of age on outcome. Circulation. 2011;124:1407–1413. doi: 10.1161/CIRCULATIONAHA.110.010124. [DOI] [PubMed] [Google Scholar]
- 4.Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, et al. 2018 guidelines for the early Management of Patients with Acute Ischemic Stroke: a guideline for healthcare professionals from the American Heart Association/ American Stroke Association. Stroke. 2018;49:e46–e110. doi: 10.1161/STR.0000000000000158. [DOI] [PubMed] [Google Scholar]
- 5.Brignole M, Moya A, de Lange FJ, Deharo JC, Elliott PM, et al. 2018 ESC guidelines for the diagnosis and management of syncope. Eur Heart J. 2018;39:1883–1948. doi: 10.5603/KP.2018.0161. [DOI] [PubMed] [Google Scholar]
- 6.Ely EW, Margolin R, Francis J, May L, Truman B, Speroff T, et al. Evaluation of delirium in critically ill patients: validation of the confusion assessment method for the intensive care unit (CAM-ICU) Crit Care Med. 2001;29:1370–1379. doi: 10.1097/00003246-200107000-00012. [DOI] [PubMed] [Google Scholar]
- 7.Teasdale G, Jennett B. Assessment of coma and impaired consciousness. A practical scale. Lancet. 1974;2:81–84. doi: 10.1016/S0140-6736(74)91639-0. [DOI] [PubMed] [Google Scholar]
- 8.Tzabazis A, Miller C, Dobrow MF, Zheng K. Delayed emergence after anesthesia. J Clin Anesth. 2015;27:353–360. doi: 10.1016/j.jclinane.2015.03.023. [DOI] [PubMed] [Google Scholar]
- 9.Ely EW, Truman B, Shintani A, Thomason JW, Wheeler AP, Gordon S, et al. Monitoring sedation status over time in ICU patients: reliability and validity of the Richmond agitation-sedation scale (RASS) JAMA. 2003;289:2983–2991. doi: 10.1001/jama.289.22.2983. [DOI] [PubMed] [Google Scholar]
- 10.Chen LW, Dai XF, Wu XJ, Liao DS, Hu YN, Zhang H, et al. Ascending aorta and Hemiarch replacement combined with modified triple-branched stent graft implantation for repair of acute DeBakey type I aortic dissection. Ann Thorac Surg. 2017;103:595–601. doi: 10.1016/j.athoracsur.2016.06.017. [DOI] [PubMed] [Google Scholar]
- 11.Sun L, Qi R, Zhu J, Liu Y, Chang Q, Zheng J. Repair of acute type a dissection: our experiences and results. Ann Thorac Surg. 2011;91:1147–1152. doi: 10.1016/j.athoracsur.2010.12.005. [DOI] [PubMed] [Google Scholar]
- 12.Qin W, Zhang X, Yang S, Li Y, Yuan J, Yang L, et al. Risk factors for multiple organ dysfunction syndrome in severe stroke patients. PLoS One. 2016;11:e0167189. doi: 10.1371/journal.pone.0167189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Liu YC, Qi ZW, Guo SG, Wang Z, Yu XZ, Ma S, et al. Role of corticotrophin releasing hormone in cerebral infarction-related gastrointestinal barrier dysfunction. World J Emerg Med. 2011;2:59–65. doi: 10.5847/wjem.j.1920-8642.2011.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lin CJ, Hung JW, Cho CY, Tseng CY, Chen HY, Lin FC, et al. Poststroke constipation in the rehabilitation ward: incidence, clinical course and associated factors. Singapore Med J. 2013;54:624–629. doi: 10.11622/smedj.2013222. [DOI] [PubMed] [Google Scholar]
- 15.Schaller BJ, Graf R, Jacobs AH. Pathophysiological changes of the gastrointestinal tract in ischemic stroke. Am J Gastroenterol. 2006;101:1655–1665. doi: 10.1111/j.1572-0241.2006.00540.x. [DOI] [PubMed] [Google Scholar]
- 16.Ji R, Wang D, Shen H, Pan Y, Liu G, et al. Interrelationship among common medical complications after acute stroke: pneumonia plays an important role. Stroke. 2013;44:3436–3444. doi: 10.1111/j.1572-0241.2006.00540.x. [DOI] [PubMed] [Google Scholar]
- 17.Chen CM, Hsu HC, Chuang YW, Chang CH, Lin CH, Hong CZ. Study on factors affecting the occurrence of upper gastrointestinal bleeding in elderly acute stroke patients undergoing rehabilitation. J Nutr Health Aging. 2011;15:632–636. doi: 10.1007/s12603-011-0052-2. [DOI] [PubMed] [Google Scholar]
- 18.Li Q, Li J, Lv F, Li K, Luo T, Xie P. A multidetector CT angiography study of variations in the circle of Willis in a Chinese population. J Clin Neurosci. 2011;18:379–383. doi: 10.1016/j.jocn.2010.07.137. [DOI] [PubMed] [Google Scholar]
- 19.Bornfeldt KE, Tabas I. Insulin resistance, hyperglycemia, and atherosclerosis. Cell Metab. 2011;14:575–585. doi: 10.1016/j.cmet.2011.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bos D, van der Rijk MJ, Geeraedts TE, Hofman A, Krestin GP, Witteman JC, et al. Intracranial carotid artery atherosclerosis: prevalence and risk factors in the general population. Stroke. 2012;43:1878–1884. doi: 10.1161/STROKEAHA.111.648667. [DOI] [PubMed] [Google Scholar]
- 21.van Rooy MJ, Pretorius E. Obesity, hypertension and hypercholesterolemia as risk factors for atherosclerosis leading to ischemic events. Curr Med Chem. 2014;21:2121–2129. doi: 10.2174/0929867321666131227162950. [DOI] [PubMed] [Google Scholar]
- 22.Chen LW, Dai XF, Lu L, Zhang GC, Cao H. Extensive primary repair of the thoracic aorta in acute type a aortic dissection by means of ascending aorta replacement combined with open placement of triple-branched stent graft: early results. Circulation. 2010;122:1373–1378. doi: 10.1161/CIRCULATIONAHA.110.946012. [DOI] [PubMed] [Google Scholar]
- 23.Yu H, Wang L, Zhang H, Wei W, Chen Y, Tang W, et al. Effect of mild hypothermia on cerebral microcirculation in a murine cardiopulmonary resuscitation model. Microcirculation. 2019. 10.1111/micc.12537. [DOI] [PubMed]
- 24.Wang X, Ji B, Yang B, Liu G, Miao N, Yang J, et al. Real-time continuous neuromonitoring combines transcranial cerebral Doppler with near-infrared spectroscopy cerebral oxygen saturation during total aortic arch replacement procedure: a pilot study. ASAIO J. 2012;58:122–126. doi: 10.1097/MAT.0b013e318241abd3. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and/or analysed during the current study are not publicly available due to the confidentiality agreement of our institution but are available from the corresponding author on reasonable request.