Abstract
O-Linked α-N-acetylgalactosamine (O-Gal-NAc) glycans constitute a major part of the human glycome. They are difficult to study because of the complex interplay of 20 distinct glycosyltransferase isoenzymes that initiate this form of glycosylation, the polypeptide N-acetylgalactosaminyl-transferases (GalNAc-Ts). Despite proven disease relevance, correlating the activity of individual GalNAc-Ts with bio-logical function remains challenging due to a lack of tools to probe their substrate specificity in a complex biological environment. Here, we develop a “bump–hole” chemical reporter system for studying GalNAc-T activity in vitro. Individual GalNAc-Ts were rationally engineered to contain an enlarged active site (hole) and probed with a newly synthesized collection of 20 (bumped) uridine diphosphate N-acetylgalactosamine (UDP-GalNAc) analogs to identify enzyme–substrate pairs that retain peptide specificities but are otherwise completely orthogonal to native enzyme–substrate pairs. The approach was applicable to multiple GalNAc-T isoenzymes, including GalNAc-T1 and -T2 that prefer nonglycosylated peptide substrates and GalNAcT-10 that prefers a preglycosylated peptide substrate. A detailed investigation of enzyme kinetics and specificities revealed the robustness of the approach to faithfully report on GalNAc-T activity and paves the way for studying substrate specificities in living systems.
Graphical Abstract
INTRODUCTION
Glycosylation with O-linked α-N-acetylgalactosamine (O-GalNAc), historically called mucin-type O-glycosylation, is one of the most abundant forms of post-translational modification in higher eukaryotes1 and is essential for normal embryonic development,2 immune cell function,3 and metabolic homeostasis.4 Aberrations in O-GalNAc glycosylation are associated with tumor progression.5 Accordingly, the enzymes that initiate O-GalNAc glycosylation, the polypeptide N-acetylgalactosaminyltransferases (GalNAc-Ts), are differentially regulated, and their expression has been associated with various disease states.6–9 The human genome encodes 20 GalNAc-T paralogs, constituting one of the largest and arguably most complex glycosyltransferase families.10 Gal-NAc-Ts are membrane-associated, type II Golgi-resident enzymes with adjacent catalytic and lectin domains that face the Golgi lumen. Some isoenzymes seem to share functional redundancies, as reflected in subtle animal knockout phenotypes,11,12 whereas others are essential for viability or specific organ functions.2,13
O-GalNAc glycans frequently decorate proteins in clusters called mucin glycodomains.14 These densely glycosylated regions are built by sequential action of GalNAc-Ts that glycosylate unmodified or sparsely O-GalNAc glycosylated peptide sequences (e.g., GalNAc-T1 and -T2) and GalNAc-Ts that follow-up initial glycosylation by other GalNAc-Ts and act adjacent to existing O-GalNAc glycans (e.g., GalNAc-T10).10,15 The substrate specificities of the various isoenzymes collectively create an O-GalNAc glycoproteome with discrete biological consequences.
Despite their participation in many facets of human biology, our understanding of GalNAc-T substrate selection is limited. GalNAc-Ts transfer a GalNAc residue from the nucleotide sugar donor uridine diphosphate N-acetylgalactosamine (UDP-GalNAc) to the hydroxyl groups of Ser, Thr, and possibly Tyr residues of proteins in the secretory pathway and supposedly the nucleus.16,17 GalNAc-Ts contain an N-terminal catalytic domain and a C-terminal lectin domain, and the interplay between these two domains determines the acceptor substrate specificity of different GalNAc-T isoenzymes.18,19 Peptide-preferring GalNAc-Ts utilize the lectin domain to install a new GalNAc residue away from an existing GalNAc on a glycopeptide (6–17 residues in the N- or C-terminal direction).18 In addition to this long-range GalNAc recognition by the lectin domain, glycopeptide-preferring GalNAc-Ts can install a GalNAc adjacent to nearby GalNAc residues on a glycopeptide, which is attributed to an additional GalNAc binding site in the catalytic domain.20
Due to the complex mechanism modulating glycosite specificities, the protein substrates and particular glycosylation sites modified by individual GalNAc-Ts are not well described, hampering our understanding of their biological function and disease causality. Classical genetic approaches toward understanding GalNAc-T function are complicated by protein substrate redundancies and the existence of overlapping protein target sites.12,21 More targeted genetic studies have validated roles for GalNAc-T2 glycosylation of ApoCIII and ANGPTL3 in high-density lipoprotein metabolism,9 GalNAc-T3 glycosylation of FGF23 in familial tumoral calcinosis,22 and GalNAc-T11 glycosylation of Notch in heterotaxy, associated with congenital heart disease.13 Despite these forays, the primary source of information about specific amino acid preferences of individual GalNAc-Ts is still the performance of in vitro assays with peptide substrates.18,23,24
Clausen and co-workers made a major breakthrough in discerning GalNAc-T substrate specificity using cell lines engineered to generate O-GalNAc glycoproteins displaying truncated glycans with limited elaborations.25 Glycoproteomic analyses of these so-called Simple—Cells with and without a single GalNAc-T knockout—identified isoenzyme-specific glycosylated proteins and sites.21,26–28 However, given the interdependencies and substrate redundancies of various GalNAc-Ts, it is likely that the absence of a single GalNAc-T employed in this loss of function approach does not provide a complete picture of the roles of individual enzymes in this family. Furthermore, due to the truncated glycan structures found on SimpleCells and cancer cells and the associated oncogenic phenotype, the substrate specificities of GalNAc-Ts in this context may not be fully representative of their behavior in noncancerous cells.5,28 A powerful addition to the field would be a method that enables the identification of the protein substrates of an individual GalNAc-T in the presence of the full repertoire of glycosyltransferases and GalNAc-Ts, including the GalNAc-T of interest, that are normally found in the cell or model system.
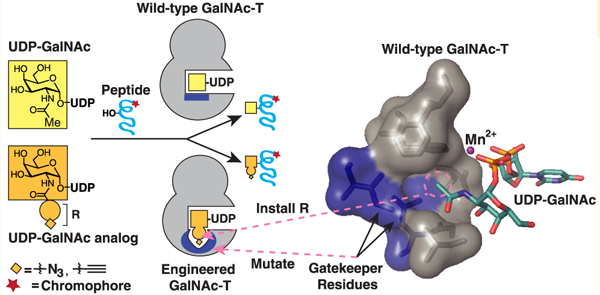
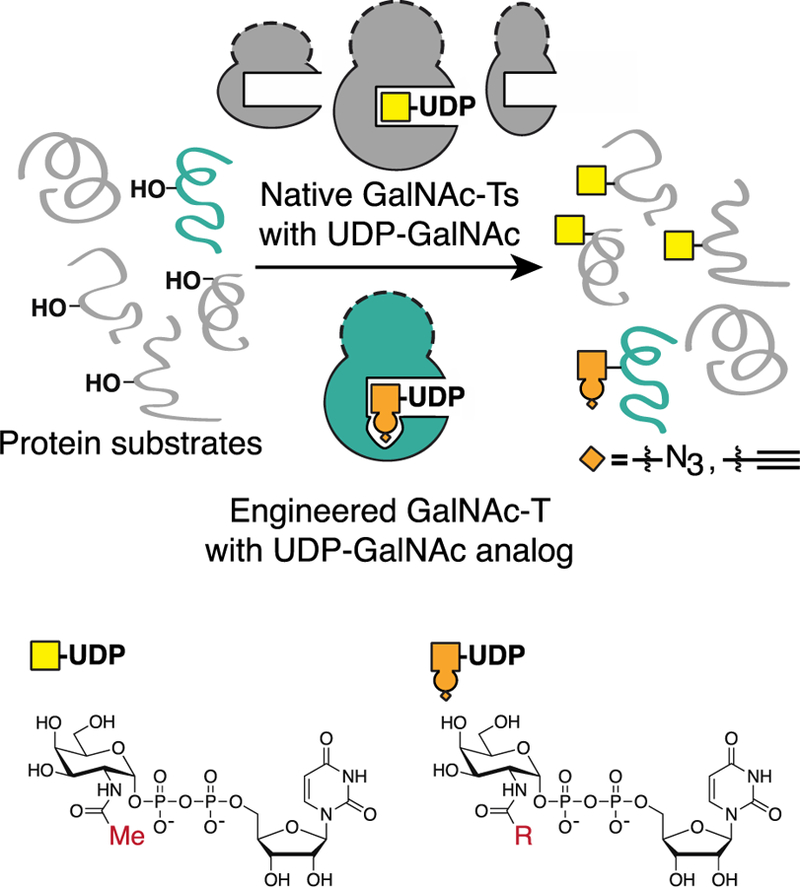
We envisioned a complementary approach in which individual GalNAc-T isoenzymes are engineered to acquire a gain of function—the ability to accept a substrate analog that marks their specific protein substrates and glycosites with a chemical handle. Key to this “bump–hole” engineering strategy is the identification of gatekeeper residues that can be mutated to alter the active site, such that it accommodates a modified substrate.29 Critically, this “bumped” substrate should not be recognized by native members of the enzyme family so as to remove false-positive signals generated by other endogenous GalNAc-T isoenzymes. A fully orthogonal System—in which the engineered enzyme utilizes the bumped substrate analog with high efficiency, while no longer recognizing the native substrate—may also benefit from a high signal-to-noise ratio. The bump–hole strategy has been successful with other enzyme families, including kinases,30,31 acetyltransferases,32 methyltransferases,33 and ADP-ribosyl-transferases.34
Previous studies have demonstrated that glycosyltransferases are amenable to enzyme–substrate engineering, wherein the active site is modified to accept a non-native substrate.35 Of particular relevance, the enzymes β-1,4-galactosyltransferase 1 (β4Gal-T1) and O-linked β-N-acetylglucosamine transferase (O-GlcNAc transferase) have been rationally designed to accommodate nucleotide sugars bearing chemical handles.36–38 Both of these engineered glycosyltransferases demonstrate expanded substrate specificity, as they still use their native substrates, UDP-galactose and UDP-GlcNAc, respectively.36,37 These engineered enzyme–substrate pairs have been successfully employed to study proteins modified with O-GlcNAc.36,38 Application of orthogonal bump–hole engineering across glycosyltransferase families has the potential to yield new insights into critical biology.
Here, we report a strategy for engineering orthogonal GalNAc-T and UDP-sugar pairs that can, in principle, be applied across the enzyme family (Scheme 1). We identified a combination of active site point mutations that caused loss of function with UDP-GalNAc but gain of function with synthetic UDP-GalNAc analogs that contain a chemical handle. We found orthogonal enzyme–substrate pairs that retained peptide glycosylation site preferences and catalytic parameters comparable to their native counterparts. The generality of the strategy was demonstrated with bump–hole pairs for the peptide-preferring GalNAc-Ts, -T1 and -T2, and a glycopeptide-preferring GalNAc-T, -T10.
Scheme 1. Bump–Hole Approacha.
aMutagenesis of key gatekeeper residues in the active site of a GalNAc-T introduces a “hole” in the catalytic domain of the engineered GalNAc-T that accommodates an enlarged UDP-GalNAc analog modified with a “bump” (orange circle) and chemical handle (orange diamond). The N-acyl side chain of UDP-GalNAc contains a methyl group (red Me) that is modified on the UDP-GalNAc analog to an R-group (red R), representing the bump and chemical handle. Monosaccharides are represented with colored boxes: GalNAc (yellow) and GalNAc analog (orange). The lectin domain of a GalNAc-T is represented as semicircle (dashed line).
RESULTS AND DISCUSSION
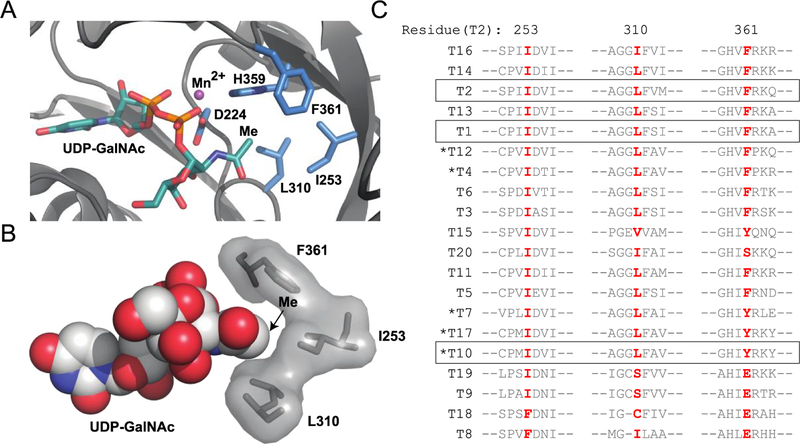
To identify potential gatekeeper residues to target for mutagenesis, we analyzed the available crystal structures of the catalytic and lectin domains of GalNAc-T1,39 -T2,40–42 and -T10,43 some of which contained bound GalNAc or UDP-GalNAc in the active site. We considered different positions around GalNAc when selecting a location in the active site for mutagenesis. Notably, we previously found that virtually all GalNAc-T isoenzymes can use UDP-N-azidoacetylgalactosamine (UDP-GalNAz) as a substrate both in vitro and in living cells or organisms, albeit with reduced efficiency compared to UDP-GalNAc.44–46 Thus, we anticipated that larger N-acyl substituents would be required to avoid recognition by wild-type GalNAc-T isoenzymes, and these would have to be accommodated by an engineered active site hole. We noted a high degree of conservation of three hydrophobic residues within 5 Å of the methyl group of GalNAc that create a binding pocket (Figure 1). We hypothesized that some combination of I253, L310, and F361 could serve as active site gatekeeper residues across the enzyme family (Figures 1 and S1). To test these hypotheses, we initially focused on GalNAc-T2, an isoenzyme that prefers to modify unglycosylated peptide substrates and is one of the best characterized of the family.40–42
Figure 1.
Identification of gatekeeper residues. (A) Residues within 5 Å of GalNAc methyl carbon for GalNAc-T2 (PDB ID 4D0T). Five of seven amino acids in close proximity to the GalNAc methyl contain side chains; of these, H359 and D224 coordinate Mn2+, while I253, L310, and F361 are promising hydrophobic residues. (B) Space-filling model of gatekeeper residues within 5 Å of GalNAc methyl in GalNAc-T2 (PDB ID 4D0T). (C) Amino acid sequences of human GalNAc-T1–GalNAc-T20 surrounding potential gatekeeper residues demonstrate a high degree of conservation with 13 isoenzymes containing Ile/Leu and 18 containing Ile at residues homologous to GalNAc-T2 positions 253/310 or 253, respectively. Only GalNAc-T8 and -T18 have dramatically different residues at positions corresponding to 253 and around 310 of GalNAc-T2. GalNAc-Ts used in this study are boxed. Clustal Omega was used to generate a multiple sequence alignment of the amino acid sequences corresponding to the full-length genes of human GalNAc-T1-GalNAc-T20 (Figure S2; Table S1). GalNAc-Ts are ordered based on homology, and GalNAc-Ts that predominantly prefer GalNAc-peptides are denoted with an asterisk.10
Prior to generating the bump–hole pair, we considered the risk that our proposed GalNAc modifications would interfere with the ability of downstream GalNAc-Ts to glycosylate GalNAc analog-modified proteins. The lectin domain is critical to most follow-up GalNAc-T glycosylation, so we evaluated the binding mode of the GalNAc-T2 lectin domain with a GalNAc-peptide, MUC5AC-13.42 We observed that the methyl group of GalNAc extends out of a pocket—created by the acceptor peptide and the lectin domain—and into a large, solvent-exposed cleft (Figure S3). This crystal structure suggests that N-acyl-modified GalNAc analogs might not experience a steric clash in the lectin domain of GalNAc-Ts with a similar binding mode for GalNAc-peptides.
We generated four single point mutants (I253A, L310A, F361A, and F361S) and one double mutant (I253A/L310A) of a soluble, FLAG epitope-tagged GalNAc-T2 construct containing the catalytic and lectin domains and secreted from mammalian cells.47 Preliminary screens with purified proteins and the known peptide substrate EA2-biotin (PTTDSTTPAPTTKK(biotin))46 showed minimal activity from either F361 mutant with UDP-GalNAc (1) or UDP-GalNAz (2),46,48 in line with mutagenesis data that were published thereafter.42 The remaining three mutants (I253A, L310A, and I253A/L310A) and wild-type GalNAc-T2 were investigated further.
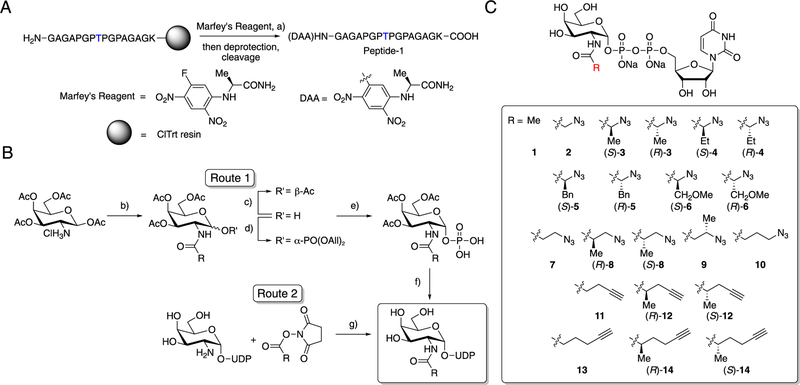
We next designed a quantitative enzymatic assay with the sensitivity and flexibility necessary to accommodate chemically diverse UDP-GalNAc analog libraries and measure kinetic parameters of mutated GalNAc-Ts. Widely used glycosyl-transferase assays rely on the detection of UDP as a side product of the glycosylation reaction.49 Although we used this method for initial screens (Figures S4 and S5), we deemed it of limited utility, as background hydrolysis of UDP-sugars under these conditions produces UDP that limits sensitivity. The assay had to be independent of the nature of the chemical handle, precluding the use of our previously developed azido-ELISA (enzyme-linked immunosorbent assay).50 We therefore used the chromophore 2,4-dinitrophenyl-5-l-alanine amide, which was installed on peptide substrates using Marfey’s reagent and is convenient to monitor by UV detection during high-performance liquid chromatography (HPLC). We prepared an artificial, labeled peptide via solid-phase peptide synthesis as an optimal GalNAc-T2 substrate (Peptide-1) based on previously identified amino acid preferences (Figure 2A).23
Figure 2.
Synthesis of a peptide substrate and a panel of UDP-GalNAc analogs. (A) Synthetic route for Peptide-1. Blue T indicates the Thr glycosylation site used by GalNAc-T2. (B) Synthetic routes for UDP-GalNAc analogs. Route 1 was used to synthesize UDP-sugars ((S)-3, (R)-3, (S)-4, (R)-4, (S)-5, (R)-5, (S)-6, (R)-6, (S)-8, (R)-8, (S)-12, (R)-12, (S)-14, (R)-14), and Route 2 was used to synthesize UDP-sugars (2, 7, 9, 10,11, 13). (C) Panel of UDP-GalNAc derivatives with azide or alkyne chemical handles. Compounds 1 and 2 are the natural substrate UDP-GalNAc and known analog UDP-GalNAz, respectively. Reagents and conditions: (a) NEt(i-Pr)2, DMF, rt; (b) R-COOH, COMU, NEt(i-Pr)2, DMF, 0 °C to rt; (c) N,N′-dimethyl-1,3-diaminopropane, THF, rt; (d) i-Pr2NPO(OAll)2, 1H-tetrazole, CH2Cl2, 0 °C, then m-CPBA, −78 °C; (e) Pd(PPh3)4, sodium p-toluenesulfinate, THF/MeOH, rt; (f) (i) uridine 5′-monophosphomorpholidate 4-morpholine-N,N′-dicyclohexylcarboxamidine salt, 1-methylimidazole hydrochloride, NEt3, DMF, rt; (ii) MeOH/water/NEt3, rt; (g) HEPES buffer (pH = 8.0), 0 °C to rt.
We next synthesized a collection of UDP-GalNAc analogs chemically diversified at the C2-acylamide moiety and including bioorthogonal azide or alkyne groups. Synthesis of nucleotide sugars is nontrivial, and both chemical and enzymatic routes have been developed.50–54 As UDP-GalNAc biosynthetic enzymes are unlikely to accommodate all envisaged modifications,51,55 we chose to chemically synthesize 20 bumped UDP-GalNAc analogs by two routes adapted from literature procedures (Figure 2B).50,53,56 Installation of the acylamide side chain onto a UDP-galactosamine scaffold via active ester chemistry proceeded smoothly to provide compounds 2, 7, 9, 10, 11, and 13 (Figure 2B, Route 2). For UDP-GalNAc analogs containing chiral acylamide α-carbon atoms, an alternate route was selected to minimize the risk of acylamide epimerization during installation. Thus, acylamides were introduced at an early stage in the syntheses of compounds ((S)-3, (R)-3, (S)-4, (R)-4, (S)-5, (R)-5, (S)-6, (R)-6, (S)-8, (R)-8, (S)-12, (R)-12, (S)-14, (R)-14) (Figure 2B, Route 1) using COMU as a highly efficient coupling reagent that precluded epimerization. Together, these two routes and an optimized purification procedure enabled us to generate a structurally diverse panel of synthetic UDP-GalNAc analogs (Figure 2C).
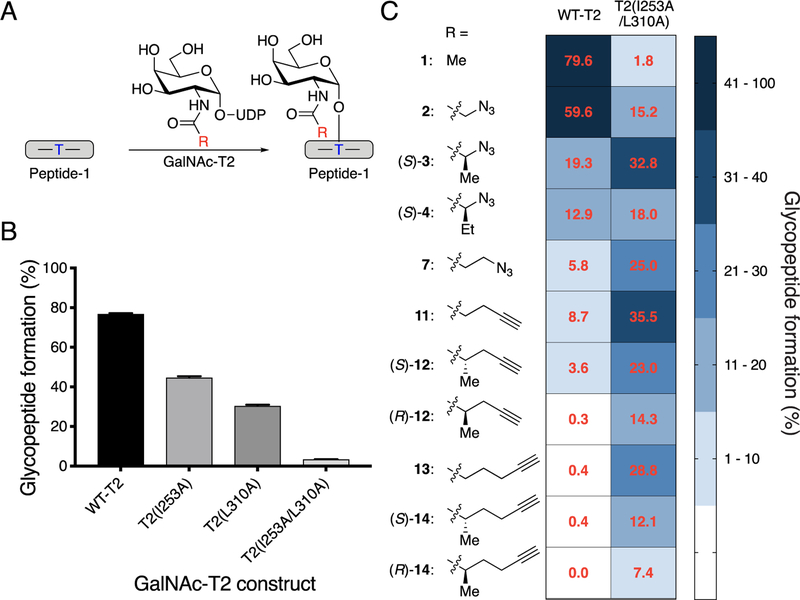
With purified enzymes, synthetic peptide substrate, and UDP-GalNAc analogs in hand, we assessed the viability of GalNAc-T bump–hole engineering in a combinatorial fashion using an HPLC-based enzymatic assay (Figure 3A). Wild-type GalNAc-T2 efficiently transferred a GalNAc residue from UDP-GalNAc (1) to the acceptor peptide, and the single mutants T2(I253A) and T2(L310A) still utilized UDP-GalNAc as a donor substrate, although with reduced activity (Figure 3B). In contrast, UDP-GalNAc was a poor substrate for the double-mutant enzyme T2(I253A/L310A), which we felt might allow for the development of an orthogonal GalNAc-T and UDP-sugar pair.
Figure 3.
Screening GalNAc-T2 for an orthogonal enzyme−substrate pair. (A) Scheme for glycosylation reaction with Peptide-1, GalNAc-T2, and UDP-GalNAc or UDP-GalNAc analog to form glycosylated Peptide-1. Blue T indicates the Thr glycosylation site used by GalNAc-T2. (B) Glycopeptide formation by wild-type and mutant GalNAc-T2. UDP-GalNAc and Peptide-1 were incubated with GalNAc-T2 at 37 °C for 1 h, and reaction was quenched by addition of aqueous EDTA (150 mM, pH = 8.0). Percent conversion to glycopeptide product was quantified by HPLC separation and peak integration. All data represent the mean of technical triplicates, and error bars represent the standard deviation. (C) Bump−hole pair optimization for GalNAc-T2. Glycosylation by wild-type and double-mutant GalNAc-T2 was compared for UDP-GalNAc (1) and UDP-GalNAc analogs with Peptide-1. Reactions were performed and quantified as in B. Heat map (blue shading) shows percent glycosylated Peptide-1 formed by wild-type or double-mutant GalNAc-T2 with UDP-GalNAc or analogs. Red values represent the mean of technical triplicates.
We investigated how enlargement of the GalNAc side chain alters substrate activity for the wild-type and double-mutant enzymes (Figures 3C and S6 for selected UDP-GalNAc analogs and Figure S4 for additional analogs). UDP-GalNAz (2) was a better substrate for wild-type GalNAc-T2 than for the double-mutant T2(I253A/L310A). Strikingly, however, some UDP-GalNAc analogs with longer or branched N-acyl chains were better substrates for T2(I253A/L310A) than for wild-type GalNAc-T2. These included UDP-GalNAc analogs with additional alkyl substituents α to the amide as in (S)-3 and (S)-4, longer side chains as in 7, 11 (“UDP-GalNAlk”),57 and 13, or both branches and length as in (S)-12, (R)-12, (S)-14, and (R)-14. For analogs 11–14, we observed that as the size of the alkyne-containing bump increased, wild-type enzyme activity plummeted. In contrast, the double-mutant enzyme maintained activity with these larger alkyne-functionalized UDP-GalNAc analogs. In the extreme case of (R)-14, glycopeptide formation by the wild-type enzyme was undetectable, while the double mutant still produced glycopeptide product.
An orthogonal enzyme−substrate pair should ideally retain the catalytic efficiency of the native enzyme−substrate pair to appropriately emulate biological function. Michaelis−Menten kinetic analysis revealed that both native and bump−hole enzyme−substrate pairs had comparable kinetic parameters (Table 1). Catalytic constants (kcat) of enzyme−substrate pairs T2(I253A/L310A) and (S)-3, 7, 11, or (S)-12 differed less than 2-fold from the wild-type GalNAc-T2/1 pair. The kcat of the engineered pair T2(I253A/L310A)/13 was approximately 5-fold lower. Concomitantly, this pair displayed a 10-fold lower Km compared to the wild-type pair, suggesting that 13 benefitted from increased contacts within the active site leading to stronger substrate binding. Thus, the catalytic efficiency (kcat/Km) of T2(I253A/L310A)/13 is 2-fold higher than wild-type GalNAc-T2/1. In contrast, (S)-12 exhibited the weakest interaction with T2(I253A/L310A) among all enzyme−substrate pairs tested, with a 10-fold higher Km than the wild-type pair, suggesting that the methyl branch at the acylamide α-position experiences a steric clash with the enzyme.
Table 1.
Kinetic Parameters of Wild-Type and Engineered GalNAc-T2 and UDP-Sugar Pairsa
| T2/UDP-Sugar | kcat (s−1) | Km (μM) | kcat/Km (mM−1S−1) |
|---|---|---|---|
| WT-T2/1 | 0.813 ± 0.017 | 30 ± 2 | 28 |
| T2(I253A/L310A)/(S)-3 | 0.566 ± 0.014 | 43 ± 4 | 13 |
| T2(I253A/L310A)/7 | 0.61 ± 0.03 | 160 ± 20 | 3.8 |
| T2(I253A/L310A)/11 | 0.68 ± 0.02 | 56 ± 6 | 12 |
| T2(I253A/L310A)/(S)-12 | 0.84 ± 0.05 | 430 ± 50 | 2.0 |
| T2(I253A/L310A)/13 | 0.158 ± 0.003 | 2.6 ± 0.8 | 61 |
To determine Km and kcat values for UDP-GalNAc and UDP-GalNAc analogs, initial rates were measured by incubating wild-type or double mutant GalNAc-T2 with concentrations of UDP-sugars varying from 15.6 to 500 μM and with a constant concentration of acceptor peptide (Peptide-1 = 267 μM for 1, (S)-3, 11; Peptide-1 = 250 μM for 7, (S)-12, 13). The glycosylation was conducted at 37 °C, and three aliquots were taken within 15 min and quenched by addition of aqueous EDTA (150 mM, pH = 8.0). Products were quantified by HPLC separation and peak integration. Enzymatic kinetic parameters were obtained by nonlinear regression fitting using GraphPad Prism. All data represent the mean of technical triplicates, and error depicts the standard deviation.
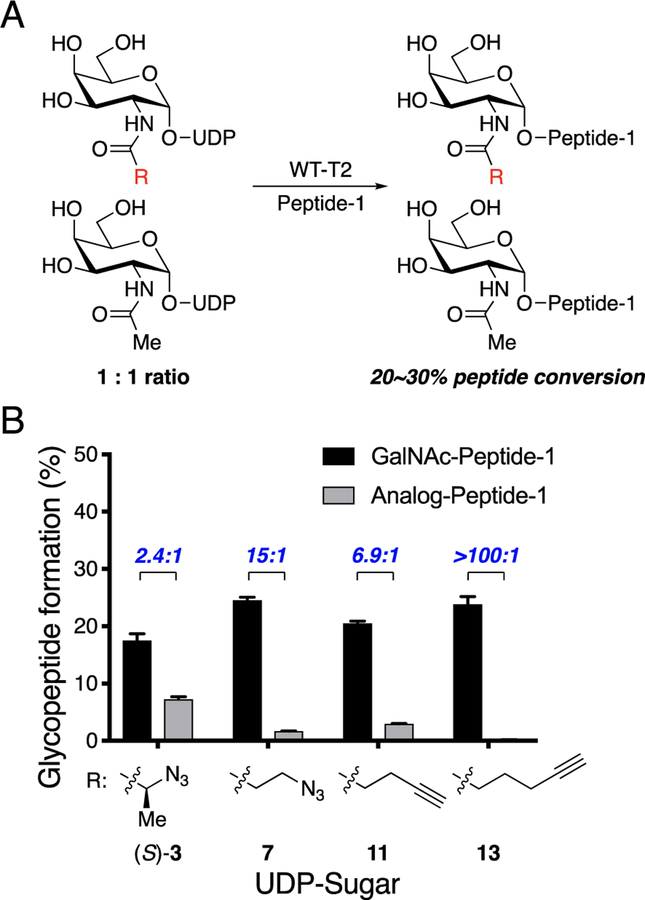
To enable potential application in biological systems, wild-type GalNAc-Ts must not accept bumped UDP-GalNAc analogs in the presence of UDP-GalNAc. We examined the relative selectivity of wild-type GalNAc-T2 toward UDP-GalNAc compared to selected analogs (Figure 4). Competition experiments revealed that in the presence of a 1:1 mixture of UDP-GalNAc and either UDP-GalNAc analog (S)-3, 7, 11, or 13, wild-type GalNAc-T2 indeed preferentially transferred GalNAc to Peptide-1. In the reaction that included analog 13, the glycopeptide product observed was almost exclusively that derived from UDP-GalNAc.
Figure 4.
Selectivity of wild-type GalNAc-T2 for UDP-GalNAc relative to UDP-GalNAc analogs. (A) Scheme for competition experiment between UDP-GalNAc and UDP-GalNAc analog. Wild-type GalNAc-T2 was treated with Peptide-1 and an equal ratio of UDP-GalNAc and UDP-GalNAc analog in a competition experiment, and glycosylation reactions were terminated at 20−30% glycopeptide formation. (B) Selectivity of wild-type GalNAc-T2 for UDP-GalNAc (1) over UDP-GalNAc analog ((S)-3, 7, 11, or 13) in a competition experiment. Reactions were performed as in A. UDP-sugars and Peptide-1 were incubated with GalNAc-T2 at 37 °C for 30 min, and reaction was quenched by addition of aqueous EDTA (150 mM, pH = 8.0). Percent conversion to glycopeptide product was quantified by HPLC separation and peak integration. Percent of Peptide-1 modified with GalNAc or GalNAc analog was measured, and selectivity ratio is shown in blue. All data represent the mean of technical triplicates, and error bars represent the standard deviation.
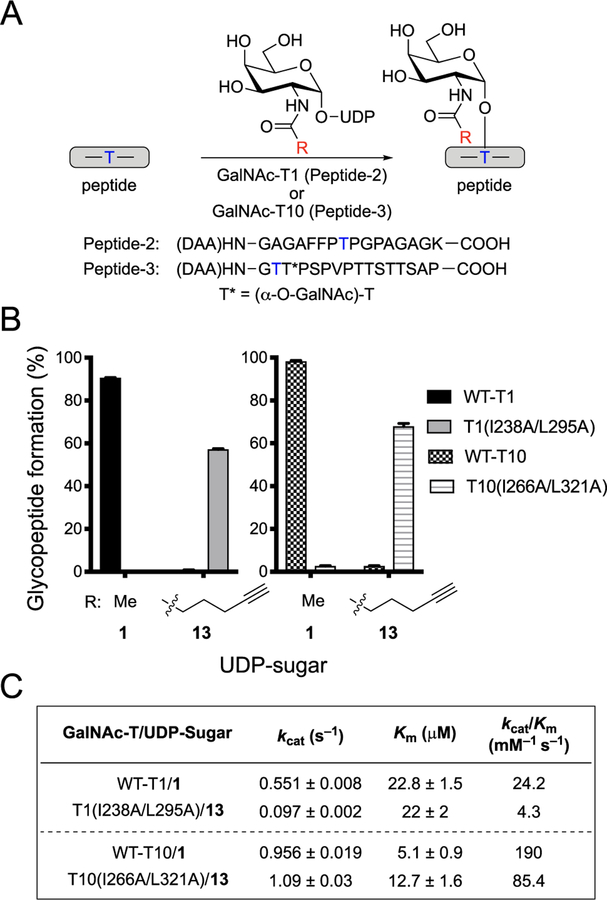
We next sought to determine if our bump−hole strategy could be generalized to study other members of the GalNAc-T family. We thus extended the approach to GalNAc-T1, a peptide-preferring isoenzyme that is phylogenetically distant from GalNAc-T2, and GalNAc-T10, an isoenzyme that prefers glycosylated substrates and, as such, is particularly difficult to study in vivo.10,28 We generated wild-type and double-mutant versions of GalNAc-T1 and -T10 using the same methods and gatekeeper residues homologous to GalNAc-T2 (I238A/L295A for -T1, I266A/L321A for -T10). We designed and prepared acceptor substrates labeled with 2,4-dinitrophenyl-5-L-alanine amide in the same fashion as Peptide-1. Similar to GalNAc-T2, amino acid preferences of GalNAc-T1 have been studied, and we used an optimized sequence for Peptide-2, which contains a single threonine for glycosylation (Figure 5A).23 GalNAc-T10, as a glycopeptide-preferring isoenzyme, required a preinstalled GalNAc directly adjacent C-terminally to the glycosylation site. To this end, MUC5AC-3, a known GalNAc-T10 glycopeptide substrate, was used as the sequence for Peptide-3 (Figure 5A).12,58
Figure 5.
Orthogonal GalNAc-T and UDP-sugar pairs for GalNAc-T1 and GalNAc-T10. (A) Scheme for glycosylation reaction with GalNAc-T, peptide, and UDP-GalNAc or UDP-GalNAc analog to form glycosylated peptide. Glycosylation reactions with GalNAc-T1 utilized Peptide-2, and reactions with GalNAc-T10 utilized Peptide-3. Blue T indicates the Thr glycosylation site used by the GalNAc-T of interest. (B) Glycopeptide formation by wild-type or double-mutant GalNAc-T1 or GalNAc-T10 with UDP-GalNAc (1) or 13. Reactions were performed as in A. GalNAc-T, UDP-sugar, and peptide were incubated at 37 °C for 1 h (-T10) or 2 h (-T1), and reaction was quenched with aqueous EDTA (150 mM, pH = 8.0). All data represent the mean of technical triplicates, and error bars represent the standard deviation. (C) Kinetic parameters of wild-type and orthogonal GalNAc-T and UDP-sugar pairs. To determine Km and kcat values for UDP-GalNAc and UDP-GalNAc analogs, initial rates were measured by incubating wild-type or double-mutant GalNAc-Ts with varying concentrations of UDP-sugars and a constant concentration of acceptor peptide. For GalNAc-T1, the concentration of UDP-sugars varied from 15.6 to 500 μM, and the concentration of acceptor Peptide-2 was held at 250 μM. For GalNAc-T10, the concentration of UDP-sugars varied from 15.6 to 250 μM, and the concentration of acceptor Peptide-3 was held at 266 μM. Glycosylation was conducted at 37 °C, and three aliquots were taken within 15 min and quenched by addition of aqueous EDTA (150 mM, pH = 8.0). Products were quantified by HPLC separation and peak integration. Enzymatic kinetic parameters were obtained by nonlinear regression fitting using GraphPad Prism. All data represent the mean of technical triplicates, and error depicts the standard deviation.
Gratifyingly, the bump−hole approach developed for GalNAc-T2 was directly transferrable to both GalNAc-T isoenzymes, despite their distinct substrate preferences. Wild-type GalNAc-T1 efficiently catalyzed glycosylation with UDP-GalNAc, whereas minimal activity was observed with UDP-GalNAc analog 13 (Figure 5B). In contrast, T1(I238A/L295A) did not utilize UDP-GalNAc as a substrate but efficiently transferred the modified GalNAc residue from analog 13 to Peptide-2 (Figures 5B and S5). Similar to GalNAc-T2 engineering, the kcat of T1(I238A/L295A) with 13 was approximately 5-fold lower than the kcat of wild-type GalNAc-T1 with UDP-GalNAc (Figure 5C). In contrast, the Km value was unaltered, resulting in a reduction of catalytic efficiency by less than an order of magnitude (Figure 5C). The selectivity of GalNAc-T10 showed identical trends; the preference of wild-type GalNAc-T10 for UDP-GalNAc over 13 was reversed in the double-mutant T10(I266A/L321A), highlighting the universality of our bump−hole approach for multiple members of the GalNAc-T family with diverse peptide and glycopeptide substrate preferences (Figure 5B). Mutation of I266 and L321 to alanine residues had no effect on the kcat and conferred only a 2-fold higher Km with the UDP-sugar analog, despite a switch in specificity from UDP-GalNAc to 13 (Figure 5C).
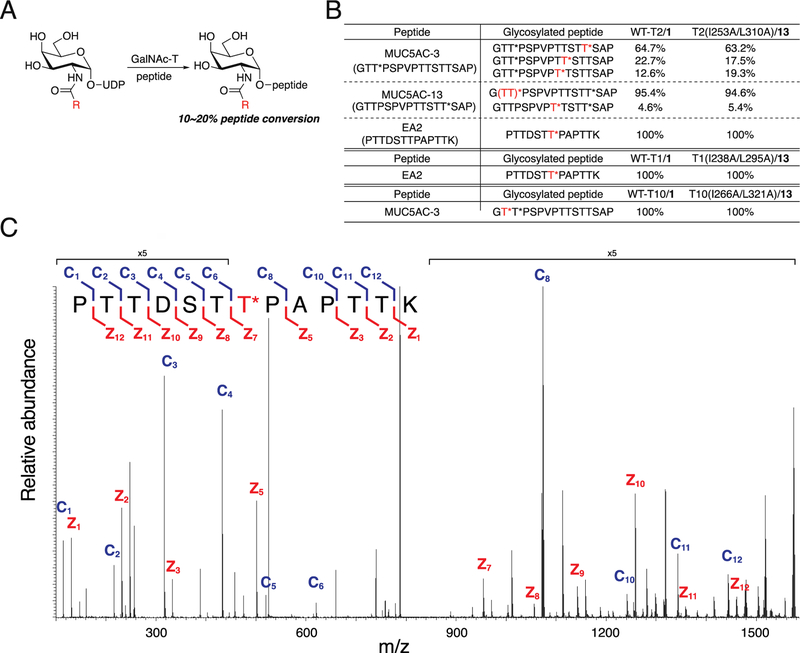
Finally, we explored whether the glycosylation site specificity of all three GalNAc-T bump−hole pairs was altered due to the active site mutations we introduced (Figure 6). In contrast to protein substrates, the specificities of GalNAc-T isoenzymes toward synthetic peptide substrates have been exhaustively mapped and are the basis for recent mechanistic and structural studies.15,18,23,46,59 We performed glycosylation reactions with known peptide and/or glycopeptide substrates of GalNAc-T1, -T2, and -T10, fragmented the resulting mono-(GalNAc-T1 and -T2) or diglycopeptides (GalNAc-T10) by tandem mass spectrometry, and manually sequenced the spectra to identify site preferences. The glycopeptide MUC5AC-3 contains multiple potential glycosylation sites, three of which are glycosylated at different frequencies by wild-type GalNAc-T2 and UDP-GalNAc.60 We observed similar fine specificities for these sites with the T2(I253A/L310A)/13 pair, with a preference for Thr13 over Thr9 and Thr10 (Figure 6B). A similar retention of site specificity was found using other known GalNAc-T2 substrates that contain multiple potential glycosylation sites, the glycopeptide MUC5AC-13 and the peptide EA2 (Figure 6B).41,60 Because the potential Thr acceptor sites on MUC5AC-3 and/or -13 are between 1 and 11 residues from the existing GalNAc residue, these glycopeptides enabled us to evaluate the influences of both the catalytic and the lectin domains on glycosite determination. The glycosylation patterns of MUC5AC-3 and MUC5AC-13 by wild-type and double-mutant GalNAc-T2 demonstrated similar glycosite specificities for lectin domain-assisted glycosylation activity in both the N- and the C-terminal directions.
Figure 6.
Glycosylation of natural peptide substrates by wild-type and engineered GalNAc-T isoenzyme−substrate pairs. (A) Scheme for glycosylation reaction with GalNAc-T, natural peptide substrate, and UDP-GalNAc or UDP-GalNAc analog to form glycosylated peptide. Glycosylation reactions were terminated at 10−20% glycopeptide formation. (B) Percent of glycosylated peptide formed out of total glycosylated peptide formed. Reactions were performed as in A at 37 °C and quenched by addition of aqueous EDTA (150 mM, pH = 8.0). Naturally occurring glycopeptides MUC5AC-3 and MUC5AC-13 each contain a single GalNAc-O-Thr (T*). Red T* indicates the site of glycosylation by the GalNAc-T of interest. Glycosylation of MUC5AC-3 by GalNAc-T2 yielded a major product that was glycosylated at Thr3 by wild-type GalNAc-T2/1. The glycosite from T2(I253A/L310A)/13 could not be unambiguously assigned and was either Thr2 or Thr3, labeled (TT)*. (C) Representative MS/MS spectrum of EA2 glycosylated by T2(1253A/L310A)/13 upon fragmentation and sequencing. Fragmentation pattern of EA2 amino acid sequence to generate c ions (blue) and z ions (red) is shown.
Glycosylation site specificity was also retained for GalNAc-T1 and -T10 bump−hole pairs using EA2 and MUC5AC-3 substrates, respectively (Figure 6B).58,61 Of note, GalNAc-T10 contains a GalNAc binding pocket in the catalytic domain directly adjacent to the active site, conferring selectivity toward preglycosylated substrates.20 The double-mutant T10(I266A/L321A) maintains specificity for Thr2 of MUC5AC-3, the only Thr adjacent to an existing glycosite, and exhibits minimally altered kinetic parameters. Our experiments demonstrate that T10(I266A/L321A) retains its identity as a glycopeptide-preferring isoenzyme with the glycopeptide MUC5AC-3, and we anticipate that this result will extend to other substrates.
CONCLUSIONS
Taken together, these data indicate that GalNAc-T bump−hole pairs retain the glycosylation site fine specificities and general kinetic parameters of the native enzyme−substrate pairs among the acceptor substrates tested. Such an outcome is promising for application of this technology to the discovery of new protein substrates and glycosylation sites in living systems. It is particularly notable that this strategy translates successfully to GalNAc-T10, which, as an isoenzyme that prefers glycosylated substrates, is notoriously difficult to study. In fact, the first O-GalNAc glycoproteomic analysis of GalNAc-T10 was recently reported using the SimpleCell technique.28 However, due to the action of GalNAc-T10 adjacent to existing glycosites, the authors did not identify any specific glycoproteins or glycosites that could be unambiguously attributed to -T10.28 Due to the presence of a chemical handle on the GalNAc analogs described here, the bump−hole system may prove powerful in efforts to assign glycosites on densely O-GalNAcylated peptides that arise from follow-up GalNAc-Ts such as GalNAc-T10.
In an effort to establish that bump−hole engineering of GalNAc-Ts minimally alters their acceptor substrate specificity, we used a small panel of peptides and glycopeptides. Totaled among the three GalNAc-Ts studied, we used 5 peptides containing 4 unique amino acid sequences and 2 glycopeptide variants of MUC5AC. We found that the wild-type and bump−hole pairs perform very similarly with these peptides and glycopeptides. However, because this subset of acceptor substrates represents a fraction of the total protein substrates that these enzymes glycosylate in living systems, further work will be necessary to validate the performance of these pairs in vivo. Additionally, it will be important to uncover whether downstream activity by GalNAc-Ts and elaborating glycosyl-transferases is observed on glycopeptides modified with these GalNAc analogs. Although GalNAz is known to be recognized by glycosyltransferases that elaborate the glycan structure,62 the effect of the bumped N-acyl group is unknown. Toward these ends, we are currently exploring the application of the engineered enzyme−substrate pairs presented here in cells.
This work represents a robust orthogonal bump−hole system for a glycosyltransferase family, wherein the engineered enzymes exclusively use a synthetic substrate analog but do not accept the native substrate. Significantly, the modified substrate is not utilized by native GalNAc-T isoenzymes. Such an achievement would not have been possible without structural data, particularly the first structure of human GalNAc-T10 bound to GalNAc and UDP reported in 2006.43 Currently, six total GalNAc-T family members have been structurally characterized with the addition of several recent crystals,15,63 establishing a foundation for the identification of promising gatekeeper residues to target for mutagenesis. Further forays into structural characterization of other glycosyltransferase families are warranted to expand this approach across the larger enzyme superfamily.
It should be noted that while structure data formed a cornerstone to this study, the ability to synthesize and rapidly screen many substrate analogs proved critical to identifying orthogonal bump−hole pairs. By exploring chemical space around the N-acyl position, we identified substrates with exquisite specificity for double-mutant GalNAc-Ts. We envision that bump−hole pairs such as those described here could be generally applicable across the GalNAc-T family, given their well-conserved UDP-GalNAc binding site.10,15 The bump−hole enzyme−substrate pairs we developed here set the stage for a new approach to the identification of biological substrates of GalNAc-Ts in living systems, analogous to work done with other enzyme families.29,64,65
METHODS
Expression of GalNAc-Ts.
The soluble domains of GalNAc-T2, -T1, and -T10 include both the catalytic and the lectin domains, and the design of our mammalian secretion constructs was based on published constructs.47 These truncated constructs were cloned into pFLAG-myc-CMV19, containing an N-terminal preprotrypsin leader sequence and an N-terminal FLAG tag. Mutations in GalNAc-T2 and -T1 were introduced using site-directed mutagenesis, and mutations in GalNAc-T10 were introduced during gene synthesis (Table S2).
Truncated GalNAc-Ts were expressed in HEK-293T cells and purified from the culture medium by FLAG affinity chromatography. Glycerol was added to purified proteins to a final concentration of 25% (v/v). Proteins were quantified by densitometry of Coomassie-stained SDS-PAGE gel bands, aliquoted, and stored at −80 °C. Western blot confirmed the identity of FLAG-tagged GalNAc-Ts (Figure S7).
Representative Experimental Procedure for the Preparation of UDP-N-Acetyl-α-d-galactosamine Derivatives (Figure 2B).
Route 1.
1,3,4,6-Tetra-O-acetyl-2-amino-2-deoxy-β-d-galactopyranose hydrochloride and azido acids were prepared according to literature procedures. A mixture of 1,3,4,6-tetra-O-acetyl-2-amino-2-deoxy-β-d-galactopyranose (192 mg, 0.500 mmol), the azido acid (0.500 mmol), and Hünig’s base (0.261 mL, 1.50 mmol) in DMF (4.00 mL) in a 25 mL round-bottom flask was cooled to 0 °C. COMU was added, and the reaction mixture was stirred at 0 °C for 1 h. The solution was allowed to warm to rt and stirred for 3 h. The mixture was diluted by the addition of ethyl acetate (50 mL), rinsed with HCl (1 M; 2 × 10 mL), saturated aqueous NaHCO3 (2 × 10 mL), and brine (20 mL), dried over Na2SO4, and concentrated. The product was purified by column chromatography.
Diallyl galactosyl 1-phosphates were prepared and deallylated according to a literature procedure.66 Tri-O-acetylated UDP-sugars were prepared by treating sugar 1-phosphates (0.200 mmol) with the uridine 5′-monophosphomorpholidate 4-morpholine-N,N′-dicyclo-hexylcarboxamidine salt (224 mg, 0.326 mmol), 1-methylimidazole hydrochloride (128 mg, 1.08 mmol), and NEt3 (55.8 μL, 0.400 mmol) in DMF (3.92 mL) in a 25 mL round-bottom flask at rt for 12 h.56 The tri-O-acetylated UDP-sugar was purified by column chromatography on C-18 silica gel and then preparative HPLC on C-18 silica gel. The compound was dissolved in MeOH/water/NEt3 (5 mL, 5:2:1) in a 25 mL round-bottom flask, and the reaction mixture was stirred at rt overnight. The product was purified by preparative HPLC on C-18 silica gel. Finally, the purified compound was passed through a Bio-Rad AG 50W-X8 resin (Na+ form) and lyophilized.
Route 2.
N-Hydroxysuccinimide (NHS) esters were prepared according to a literature procedure.67 A solution of the NHS ester (0.150 mmol) in DMF (1.08 mL) was added to a mixture of UDP-d-galactosamine disodium salt (30.5 mg, 0.0500 mmol) in HEPES buffer (0.1 M, pH = 8.0; 1.08 mL) at 0 °C. The reaction mixture was allowed to warm to rt and stirred overnight. Next, the mixture was purified by column chromatography on C-18 silica gel and then preparative HPLC on C-18 silica gel. Finally, the purified compound was passed through a Bio-Rad AG 50W-X8 resin (Na+ form) and lyophilized.
Experimental Procedure for the Preparation of Peptides (Figure 2A).
Peptide-1 and -2.
Peptides were synthesized on 2-chlorotrityl chloride resin by solid-phase peptide synthesis using N-Fmoc-protected amino acids. Each coupling step was performed with N-Fmoc-protected amino acid (10 equiv), COMU (10 equiv), and N,N-diisopropylethylamine (20 equiv) in DMF at rt for 30 min under N2 agitation. Fmoc deprotection was conducted with 20% piperidine in DMF at rt for 20 min under N2 agitation. The N-terminus was reacted with (S)-2-((5-fluoro-2,4-dinitrophenyl)amino)propanamide (10 equiv) and N,N-diisopropylethylamine (10 equiv) in DMF at rt overnight under N2 agitation. Peptides were cleaved and deprotected by a mixture of trifluoroacetic acid (88% v/v), triisopropylsilane (2% v/v), 1,4-dithiothreitol (5% w/v), and water (5% v/v) at rt for 1.5 h. The mixture was concentrated, triturated with cold Et2O, redissolved in water, and lyophilized. The desired peptide was purified by preparative HPLC on C-18 silica gel.
Peptide-3.
N-(9-Fluorenylmethyloxycarbonyl)-O-(2-acetamido-3,4,6-tri-O-acetyl-2-deoxy-α-d-galactopyranosyl)-l-threonine was prepared according to literature procedures.68–70 Peptides were synthesized on 2-chlorotrityl chloride resin by solid-phase peptide synthesis using N-Fmoc-protected amino acids. Each coupling step was performed with N-Fmoc-protected amino acid (10 equiv), COMU (10 equiv), and N,N-diisopropylethylamine (20 equiv) in DMF at rt for 30 min under N2 agitation. For the reaction with the glycosylated amino acid, the coupling reaction was conducted with N-(9-fluorenylmethyloxycarbonyl)-O-(2-acetamido-3,4,6-tri-O-acetyl-2-deoxy-α-d-galactopyranosyl)-l-threonine (2 equiv), COMU (2 equiv), and N,N-diisopropylethylamine (4 equiv) in DMF at rt overnight under N2 agitation. Fmoc deprotection was conducted with 20% piperidine in DMF at rt for 20 min under N2 agitation. The N-terminus was reacted with (S)-2-((5-fluoro-2,4-dinitrophenyl)amino)-propanamide (10 equiv) and N,N-diisopropylethylamine (10 equiv) in DMF at rt overnight under N2 agitation. Peptides were cleaved and deprotected by a mixture of trifluoroacetic acid (88% v/v), triisopropylsilane (2% v/v), 1,4-dithiothreitol (5% w/v), and water (5% v/v) at rt for 1.5 h. The mixture was concentrated, triturated with cold Et2O, redissolved in water, and lyophilized. The crude product was purified by preparative HPLC on C-18 silica gel. For the deacetylation of the sugar moiety, purified glycopeptides were treated with aqueous hydrazine (5%) for 1 h. The deprotected glycopeptides were purified by preparative HPLC on C-18 silica gel.
Representative Procedure for the Glycosylation by GalNAc-Ts with UDP-GalNAc or UDP-GalNAc Analogs (Figures 3, S6, 5A, and 5B).
The glycosylation reaction was initiated by the addition of wild-type or mutant GalNAc-T (-T2 = 50.0 nM; -T1 = 160 nM; or -T10 = 120 nM) in Tris-HCl buffer (16.7 mM Tris-HCl, 100 mM NaCl, 25% glycerol, pH = 7.4; 25.0 μL) to the mixture of UDP-sugar (500 μM) and peptide (Peptide-1, Peptide-2, or Peptide-3; 100 μM) in Tris-HCl buffer (25 mM Tris-HCl, 20 mM MnCl2; 25.0 μL) at 0 °C, resulting in a final reaction mixture containing GalNAc-T (-T2 = 25.0 nM; -T1 = 80.0 nM; -T10 = 60.0 nM), peptide (Peptide-1, Peptide-2, or Peptide-3; 50.0 μM), UDP-sugar (250 μM) in Tris-HCl buffer (20.8 mM Tris-HCl, 10 mM MnCl2, 50 mM NaCl, 12.5% glycerol, pH = 7.4; 50.0 μL). The glycosylation was conducted at 37 °C for 1 h (-T2; -T10) or 2 h (-T1) and quenched by the addition of aqueous EDTA (150 mM, pH = 8.0; 25.0 μL). Glycopeptide formation was determined by HPLC and peak integration.
Michaelis−Menten kinetics, UDP-sugar competition experiments, and glycosylation of natural peptide substrates including mass spectrometry-based sequencing are described in the Supporting Information.
Further details can be found in the Supporting Information.
Safety Statement.
No unexpected or unusually high safety hazards were encountered.
Supplementary Material
ACKNOWLEDGMENTS
We thank Lawrence Tabak (National Institutes of Health, Bethesda, MD) for the kind gift of the gene for full-length human GalNAc-T2 in the plasmid pCMV-NTAP. This work was supported by the NIH (R01 CA200423). J.C. was supported by the Korea Institute of Science and Technology (KIST). S.B.P.E.T. was supported by the Nora Baart Foundation and the Stichting Jo Kolk Studiefonds. S.A.M. was supported by a National Institute of General Medical Sciences F32 Postdoctoral Fellowship (F32-GM126663–01). B.S. was supported by a Feodor Lynen Fellowship by the Alexander von Humboldt Foundation. M.A.G. was supported by the National Science Foundation Graduate Research Fellowship (NSF GRFP) and the Stanford ChEM-H Chemistry/Biology Interface Predoctoral Training Program. M.F.D. was supported by a NWO Rubicon Postdoctoral Fellowship. J.C. thanks Dr. Mason Appel and Dr. Neil G. Rumachik for critical discussions.
Footnotes
ASSOCIATED CONTENT
Supporting Information
The Supporting Information is available free of charge on the ACS Publications website at DOI: 10.1021/jacs.9b04695.
Additional experimental details, data, and figures and tables including crystal structure modeling, GalNAc-T multiple sequence alignment, enzymatic activity screens, GalNAc-T cloning and expression data, kinetics graphs, MS/MS spectra, synthetic schemes, and NMR spectra (PDF)
The authors declare no competing financial interest.
REFERENCES
- (1).Vakhrushev SY; Steentoft C; Vester-Christensen MB; Bennett EP; Clausen H; Levery SB Enhanced Mass Spectrometric Mapping of the Human GalNAc-Type O-Glycoproteome with SimpleCells. Mol. Cell. Proteomics 2013, 12 (4), 932–944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (2).Tran DT; Zhang L; Zhang Y; Tian E; Earl LA; Ten Hagen KG Multiple Members of the UDP-GalNAc: Polypeptide N-Acetylgalactosaminyltransferase Family Are Essential for Viability in Drosophila. J. Biol. Chem 2012, 287 (8), 5243–5252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (3).Xu Z; Weiss A Negative Regulation of CD45 by Differential Homodimerization of the Alternatively Spliced Isoforms. Nat. Immunol 2002, 3 (8), 764–771. [DOI] [PubMed] [Google Scholar]
- (4).Schjoldager KT-BG; Vester-Christensen MB; Bennett EP; Levery SB; Schwientek T; Yin W; Blixt O; Clausen H O-Glycosylation Modulates Proprotein Convertase Activation of Angiopoietin-like Protein 3. J. Biol. Chem 2010, 285 (47), 36293–36303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (5).Radhakrishnan P; Dabelsteen S; Madsen FB; Francavilla C; Kopp KL; Steentoft C; Vakhrushev SY; Olsen JV; Hansen L; Bennett EP; Woetmann A; Yin G; Chen L; Song H; Bak M; Hlady RA; Peters SL; Opavsky R; Thode C; Qvortrup K; Schjoldager KT-BG; Clausen H; Hollingsworth MA; Wandall HH Immature Truncated O-Glycophenotype of Cancer Directly Induces Oncogenic Features. Proc. Natl. Acad. Sci. U. S. A 2014, 111 (39), E4066–E4075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Kingsley PD; Ten Hagen KG; Maltby KM; Zara J; Tabak LA Diverse Spatial Expression Patterns of UDP-GalNAc: Polypeptide N-Acetylgalactosaminyl-Transferase Family Member mRNAs during Mouse Development. Glycobiology 2000, 10 (12), 1317–1323. [DOI] [PubMed] [Google Scholar]
- (7).Peng R-Q; Wan H-Y; Li H-F; Liu M; Li X; Tang H MicroRNA-214 Suppresses Growth and Invasiveness of Cervical Cancer Cells by Targeting UDP-N-Acetyl-α-D-Galactosamine:Polypeptide N-Acetylgalactosaminyltransferase 7. J. Biol. Chem 2012, 287 (17), 14301–14309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (8).Lavrsen K; Dabelsteen S; Vakhrushev SY; Levann AMR; Haue AD; Dylander A; Mandel U; Hansen L; Frödin M; Bennett EP; Wandall HH De Novo Expression of Human Polypeptide N-Acetylgalactosaminyltransferase 6 (GalNAc-T6) in Colon Adenocarcinoma Inhibits the Differentiation of Colonic Epithelium. J. Biol. Chem 2018, 293 (4), 1298–1314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (9).Khetarpal SA; Schjoldager KT; Christoffersen C; Raghavan A; Edmondson AC; Reutter HM; Ahmed B; Ouazzani R; Peloso GM; Vitali C; Zhao W; Somasundara AVH; Millar JS; Park Y; Fernando G; Livanov V; Choi S; Noé E; Patel P; Ho SP; Kirchgessner TG; Wandall HH; Hansen L; Bennett EP; Vakhrushev SY; Saleheen D; Kathiresan S; Brown CD; Abou Jamra R; LeGuern E; Clausen H; Rader DJ Loss of Function of GALNT2 Lowers High-Density Lipoproteins in Humans, Nonhuman Primates, and Rodents. Cell Metab 2016, 24 (2), 234–245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (10).Bennett EP; Mandel U; Clausen H; Gerken TA; Fritz TA; Tabak LA Control of Mucin-Type O-Glycosylation: A Classification of the Polypeptide GalNAc-Transferase Gene Family. Glycobiology 2012, 22 (6), 736–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (11).Block H; Ley K; Zarbock A Severe Impairment of Leukocyte Recruitment in ppGalNAcT-1-Deficient Mice. J. Immunol 2012, 188 (11), 5674–5681. [DOI] [PubMed] [Google Scholar]
- (12).Ten Hagen KG; Fritz TA; Tabak LA All in the Family: The UDP-GalNAc:Polypeptide N-Acetylgalactosaminyltransferases. Glycobiology 2003, 13 (1), 1R–16R. [DOI] [PubMed] [Google Scholar]
- (13).Boskovski MT; Yuan S; Pedersen NB; Goth CK; Makova S; Clausen H; Brueckner M; Khokha MK The Heterotaxy Gene GALNT11 Glycosylates Notch to Orchestrate Cilia Type and Laterality. Nature 2013, 504, 456–459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (14).Woods EC; Kai F; Barnes JM; Pedram K; Pickup MW; Hollander MJ; Weaver VM; Bertozzi CR A Bulky Glycocalyx Fosters Metastasis Formation by Promoting G1 Cell Cycle Progression. eLife 2017, 6, e25752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (15).de las Rivas M; Lira-Navarrete E; Gerken TA; Hurtado-Guerrero R Polypeptide GalNAc-Ts: From Redundancy to Specificity. Curr. Opin. Struct. Biol 2019, 56, 87–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (16).Steentoft C; Vakhrushev SY; Joshi HJ; Kong Y; Vester-Christensen MB; Schjoldager KT-BG; Lavrsen K; Dabelsteen S; Pedersen NB; Marcos-Silva L; Gupta R; Bennett EP; Mandel U; Brunak S; Wandall HH; Levery SB; Clausen H Precision Mapping of the Human O-GalNAc Glycoproteome through SimpleCell Technology. EMBO J 2013, 32, 1478–1488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (17).Cejas RB; Lorenz V; Garay YC; Irazoqui FJ Biosynthesis of O-N-Acetylgalactosamine Glycans in the Human Cell Nucleus. J. Biol. Chem 2019, 294 (9), 2997–3011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (18).Revoredo L; Wang S; Bennett EP; Clausen H; Moremen W; Jarvis DL; Ten Hagen KG; Tabak LA; Gerken TA Mucin-Type O-Glycosylation Is Controlled by Short- and Long-Range Glycopeptide Substrate Recognition that Varies among Members of the Polypeptide GalNAc Transferase Family. Glycobiology 2016, 26 (4), 360–376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (19).de las Rivas M; Daniel EJP; Coelho H; Lira-Navarrete E; Raich L; Compañón I; Diniz A; Lagartera L; Jiménez-Barbero J; Clausen H; Rovira C; Marcelo F; Corzana F; Gerken TA; Hurtado-Guerrero R Structural and Mechanistic Insights into the Catalytic-Domain-Mediated Short-Range Glycosylation Preferences of GalNAc-T4. ACS Cent. Sci 2018, 4, 1274–1290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (20).Perrine CL; Ganguli A; Wu P; Bertozzi CR; Fritz TA; Raman J; Tabak LA; Gerken TA Glycopeptide-Preferring Polypeptide GalNAc Transferase 10 (ppGalNAc T10), Involved in Mucin-Type O-Glycosylation, Has a Unique GalNAc-O-Ser/Thr-Binding Site in Its Catalytic Domain Not Found in ppGalNAc T1 or T2. J. Biol. Chem 2009, 284 (30), 20387–20397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (21).Schjoldager KT; Joshi HJ; Kong Y; Goth CK; King SL; Wandall HH; Bennett EP; Vakhrushev SY; Clausen H Deconstruction of O-Glycosylation−GalNAc-T Isoforms Direct Distinct Subsets of the O-Glycoproteome. EMBO Rep 2015, 16 (12), 1713–1722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (22).Kato K; Jeanneau C; Tarp MA; Benet-Pagès A; Lorenz-Depiereux B; Bennett EP; Mandel U; Strom TM; Clausen H Polypeptide GalNAc-Transferase T3 and Familial Tumoral Calcinosis. J. Biol. Chem 2006, 281 (27), 18370–18377. [DOI] [PubMed] [Google Scholar]
- (23).Gerken TA; Raman J; Fritz TA; Jamison O Identification of Common and Unique Peptide Substrate Preferences for the UDP-GalNAc:Polypeptide α-N-Acetylgalactosaminyltransferases T1 and T2 Derived from Oriented Random Peptide Substrates. J. Biol. Chem 2006, 281 (43), 32403–32416. [DOI] [PubMed] [Google Scholar]
- (24).Gerken TA; Jamison O; Perrine CL; Collette JC; Moinova H; Ravi L; Markowitz SD; Shen W; Patel H; Tabak LA Emerging Paradigms for the Initiation of Mucin-Type Protein O-Glycosylation by the Polypeptide GalNAc Transferase Family of Glycosyltransferases. J. Biol. Chem 2011, 286 (16), 14493–14507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (25).Steentoft C; Vakhrushev SY; Vester-Christensen MB; Schjoldager KT-BG; Kong Y; Bennett EP; Mandel U; Wandall H; Levery SB; Clausen H Mining the O-Glycoproteome Using Zinc-Finger Nuclease−Glycoengineered SimpleCell Lines. Nat. Methods 2011, 8 (11), 977–982. [DOI] [PubMed] [Google Scholar]
- (26).Hintze J; Ye Z; Narimatsu Y; Madsen TD; Joshi HJ; Goth CK; Linstedt A; Bachert C; Mandel U; Bennett EP; Vakhrushev SY; Schjoldager KT Probing the Contribution of Individual Polypeptide GalNAc-Transferase Isoforms to the O-Glycoproteome by Inducible Expression in Isogenic Cell Lines. J. Biol. Chem 2018, 293 (49), 19064–19077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (27).Schjoldager KT-BG; Vakhrushev SY; Kong Y; Steentoft C; Nudelman AS; Pedersen NB; Wandall HH; Mandel U; Bennett EP; Levery SB; Clausen H Probing Isoform-Specific Functions of Polypeptide GalNAc-Transferases Using Zinc Finger Nuclease Glycoengineered SimpleCells. Proc. Natl. Acad. Sci. U. S. A 2012, 109 (25), 9893–9898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (28).Narimatsu Y; Joshi HJ; Schjoldager KT; Hintze J; Halim A; Steentoft C; Nason R; Mandel U; Bennett EP; Clausen H; Vakhrushev SY Exploring Regulation of Protein O-Glycosylation in Isogenic Human HEK293 Cells by Differential O-Glycoproteomics. Mol. Cell. Proteomics 2019, 18 (7), 1396–1409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (29).Islam K The Bump-and-Hole Tactic: Expanding the Scope of Chemical Genetics. Cell Chem. Biol 2018, 25 (10), 1171–1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (30).Fouda AE; Pflum MKH A Cell-Permeable ATP Analogue for Kinase-Catalyzed Biotinylation. Angew. Chem., Int. Ed 2015, 54 (33), 9618–9621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (31).Blethrow JD; Glavy JS; Morgan DO; Shokat KM Covalent Capture of Kinase-Specific Phosphopeptides Reveals Cdk1-Cyclin B Substrates. Proc. Natl. Acad. Sci. U. S. A 2008, 105 (5), 1442–1447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (32).Yang C; Mi J; Feng Y; Ngo L; Gao T; Yan L; Zheng YG Labeling Lysine Acetyltransferase Substrates with Engineered Enzymes and Functionalized Cofactor Surrogates. J. Am. Chem. Soc 2013, 135 (21), 7791–7794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (33).Islam K; Zheng W; Yu H; Deng H; Luo M Expanding Cofactor Repertoire of Protein Lysine Methyltransferase. ACS Chem. Biol 2011, 6 (7), 679–684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (34).Carter-O’Connell I; Jin H; Morgan RK; David LL; Cohen MS Engineering the Substrate Specificity of ADP-Ribosyltransferases for Identifying Direct Protein Targets. J. Am. Chem. Soc 2014, 136 (14), 5201–5204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (35).McArthur JB; Chen X Glycosyltransferase Engineering for Carbohydrate Synthesis. Biochem. Soc. Trans 2016, 44 (1), 129–142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (36).Rodriguez AC; Yu S-H; Li B; Zegzouti H; Kohler JJ Enhanced Transfer of a Photocross-Linking N-Acetylglucosamine (GlcNAc) Analog by an O-GlcNAc Transferase Mutant with Converted Substrate Specificity. J. Biol. Chem 2015, 290 (37), 22638–22648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (37).Ramakrishnan B; Qasba PK Structure-Based Design of β1,4-Galactosyltransferase I (β4Gal-T1) with Equally Efficient N-Acetylgalactosaminyltransferase Activity. J. Biol. Chem 2002, 277 (23), 20833–20839. [DOI] [PubMed] [Google Scholar]
- (38).Clark PM; Dweck JF; Mason DE; Hart CR; Buck SB; Peters EC; Agnew BJ; Hsieh-Wilson LC Direct In-Gel Fluorescence Detection and Cellular Imaging of O-GlcNAc-Modified Proteins. J. Am. Chem. Soc 2008, 130 (35), 11576–11577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (39).Fritz TA; Hurley JH; Trinh L-B; Shiloach J; Tabak LA The Beginnings of Mucin Biosynthesis: The Crystal Structure of UDP-GalNAc:Polypeptide α-N-Acetylgalactosaminyltransferase-T1. Proc. Natl. Acad. Sci. U. S. A 2004, 101 (43), 15307–15312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (40).Fritz TA; Raman J; Tabak LA Dynamic Association between the Catalytic and Lectin Domains of Human UDP-GalNAc:Polypeptide α-N-Acetylgalactosaminyltransferase-2. J. Biol. Chem 2006, 281 (13), 8613–8619. [DOI] [PubMed] [Google Scholar]
- (41).Lira-Navarrete E; Iglesias-Fernández J; Zandberg WF; Compañón I; Kong Y; Corzana F; Pinto BM; Clausen H; Peregrina JM; Vocadlo DJ; Rovira C; Hurtado-Guerrero R Substrate-Guided Front-Face Reaction Revealed by Combined Structural Snapshots and Metadynamics for the Polypeptide N-Acetylgalactosaminyltransferase 2. Angew. Chem., Int. Ed 2014, 53 (31), 8206–8210. [DOI] [PubMed] [Google Scholar]
- (42).Lira-Navarrete E; de las Rivas M; Compañón I; Pallarés MC; Kong Y; Iglesias-Fernández J; Bernardes GJL; Peregrina JM; Rovira C; Bernadó P; Bruscolini P; Clausen H; Lostao A; Corzana F; Hurtado-Guerrero R Dynamic Interplay between Catalytic and Lectin Domains of GalNAc-Transferases Modulates Protein O-Glycosylation. Nat. Commun 2015, 6, 6937. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (43).Kubota T; Shiba T; Sugioka S; Furukawa S; Sawaki H; Kato R; Wakatsuki S; Narimatsu H Structural Basis of Carbohydrate Transfer Activity by Human UDP-GalNAc: Polypeptide α-N-Acetylgalactosaminyltransferase (pp-GalNAc-T10). J. Mol. Biol 2006, 359 (3), 708–727. [DOI] [PubMed] [Google Scholar]
- (44).Hang HC; Yu C; Kato DL; Bertozzi CR A Metabolic Labeling Approach toward Proteomic Analysis of Mucin-Type O-Linked Glycosylation. Proc. Natl. Acad. Sci. U. S. A 2003, 100 (25), 14846–14851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (45).Prescher JA; Dube DH; Bertozzi CR Chemical Remodelling of Cell Surfaces in Living Animals. Nature 2004, 430, 873–877. [DOI] [PubMed] [Google Scholar]
- (46).Pratt MR; Hang HC; Ten Hagen KG; Rarick J; Gerken TA; Tabak LA; Bertozzi CR Deconvoluting the Functions of Polypeptide N-α-Acetylgalactosaminyltransferase Family Members by Glycopeptide Substrate Profiling. Chem. Biol 2004, 11 (7), 1009–1016. [DOI] [PubMed] [Google Scholar]
- (47).Hagen FK; Ten Hagen KG; Beres TM; Balys MM; VanWuyckhuyse BC; Tabak LA cDNA Cloning and Expression of a Novel UDP-N-Acetyl-d-Galactosamine:Polypeptide N-Acetylga-lactosaminyltransferase. J. Biol. Chem 1997, 272 (21), 13843–13848. [DOI] [PubMed] [Google Scholar]
- (48).Wagner LJS Engineering an Orthogonal O-Glycosyl-transferase and Donor Sugar Pair Ph.D. Dissertation, University of California, Berkeley, 2015. [Google Scholar]
- (49).Sheikh MO; Halmo SM; Patel S; Middleton D; Takeuchi H; Schafer CM; West CM; Haltiwanger RS; Avci FY; Moremen KW; Wells L Rapid Screening of Sugar-Nucleotide Donor Specificities of Putative Glycosyltransferases. Glycobiology 2017, 27 (3), 206–212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (50).Hang HC; Yu C; Pratt MR; Bertozzi CR Probing Glycosyltransferase Activities with the Staudinger Ligation. J. Am. Chem. Soc 2004, 126 (1), 6–7. [DOI] [PubMed] [Google Scholar]
- (51).Guan W; Cai L; Wang PG Highly Efficient Synthesis of UDP-GalNAc/GlcNAc Analogues with Promiscuous Recombinant Human UDP-GalNAc Pyrophosphorylase AGX1. Chem. - Eur. J 2010, 16 (45), 13343–13345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (52).Wolf S; Zismann T; Lunau N; Meier C Reliable Synthesis of Various Nucleoside Diphosphate Glycopyranoses. Chem. - Eur. J 2009, 15 (31), 7656–7664. [DOI] [PubMed] [Google Scholar]
- (53).Heidlas JE; Lees WJ; Whitesides GM Practical Enzyme-Based Syntheses of Uridine 5′-Diphosphogalactose and Uridine 5′-Diphospho-N-Acetylgalactosamine on a Gram Scale. J. Org. Chem 1992, 57 (1), 152–157. [Google Scholar]
- (54).Muthana MM; Qu J; Li Y; Zhang L; Yu H; Ding L; Malekan H; Chen X Efficient One-Pot Multienzyme Synthesis of UDP-Sugars Using a Promiscuous UDP-Sugar Pyrophosphorylase from Bifidobacterium Longum (BLUSP). Chem. Commun 2012, 48 (21), 2728–2730. [DOI] [PubMed] [Google Scholar]
- (55).Pouilly S; Bourgeaux V; Piller F; Piller V Evaluation of Analogues of GalNAc as Substrates for Enzymes of the Mammalian GalNAc Salvage Pathway. ACS Chem. Biol 2012, 7 (4), 753–760. [DOI] [PubMed] [Google Scholar]
- (56).Tsukamoto H; Kahne D N-Methylimidazolium Chloride-Catalyzed Pyrophosphate Formation: Application to the Synthesis of Lipid I and NDP-Sugar Donors. Bioorg. Med. Chem. Lett 2011, 21 (17), 5050–5053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (57).Batt AR; Zaro BW; Navarro MX; Pratt MR Metabolic Chemical Reporters of Glycans Exhibit Cell-Type-Selective Metabolism and Glycoprotein Labeling. ChemBioChem 2017, 18 (13), 1177–1182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (58).Cheng L; Tachibana K; Zhang Y; Guo J.-m.; Kahori Tachibana K; Kameyama A; Wang H; Hiruma T; Iwasaki H; Togayachi A; Kudo T; Narimatsu H Characterization of a Novel Human UDP-GalNAc Transferase, pp-GalNAc-T10. FEBS Lett 2002, 531 (2), 115–121. [DOI] [PubMed] [Google Scholar]
- (59).Kightlinger W; Lin L; Rosztoczy M; Li W; DeLisa MP; Mrksich M; Jewett MC Design of Glycosylation Sites by Rapid Synthesis and Analysis of Glycosyltransferases. Nat. Chem. Biol 2018, 14 (6), 627–635. [DOI] [PubMed] [Google Scholar]
- (60).Raman J; Fritz TA; Gerken TA; Jamison O; Live D; Liu M; Tabak LA The Catalytic and Lectin Domains of UDP-GalNAc:Polypeptide α-N-Acetylgalactosaminyltransferase Function in Concert to Direct Glycosylation Site Selection. J. Biol. Chem 2008, 283 (34), 22942–22951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (61).Tenno M; Ohtsubo K; Hagen FK; Ditto D; Zarbock A; Schaerli P; von Andrian UH; Ley K; Le D; Tabak LA; Marth JD Initiation of Protein O Glycosylation by the Polypeptide GalNAcT-1 in Vascular Biology and Humoral Immunity. Mol. Cell. Biol 2007, 27 (24), 8783–8796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (62).Woo CM; Iavarone AT; Spiciarich DR; Palaniappan KK; Bertozzi CR Isotope-Targeted Glycoproteomics (IsoTaG): A Mass-Independent Platform for Intact N- and O-Glycopeptide Discovery and Analysis. Nat. Methods 2015, 12 (6), 561–567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (63).Yu C; Liang L; Yin Y Structural Basis of Carbohydrate Transfer Activity of UDP-GalNAc: Polypeptide N-Acetylgalactosaminyltransferase 7. Biochem. Biophys. Res. Commun 2019, 510 (2), 266–271. [DOI] [PubMed] [Google Scholar]
- (64).Banko MR; Allen JJ; Schaffer BE; Wilker EW; Tsou P; White JL; Villén J; Wang B; Kim SR; Sakamoto K; Gygi SP; Cantley LC; Yaffe MB; Shokat KM; Brunet Chemical Genetic Screen for AMPKα2 Substrates Uncovers a Network of Proteins Involved in Mitosis. Mol. Cell 2011, 44 (6), 878–892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (65).Wang R; Islam K; Liu Y; Zheng W; Tang H; Lailler N; Blum G; Deng H; Luo M Profiling Genome-Wide Chromatin Methylation with Engineered Posttranslation Apparatus within Living Cells. J. Am. Chem. Soc 2013, 135 (3), 1048–1056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (66).Beahm BJ; Dehnert KW; Derr NL; Kuhn J; Eberhart JK; Spillmann D; Amacher SL; Bertozzi CR A Visualizable Chain-Terminating Inhibitor of Glycosaminoglycan Biosynthesis in Developing Zebrafish. Angew. Chem., Int. Ed 2014, 53 (13), 3347–3352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (67).Zhang Z; Hejesen C; Kjelstrup MB; Birkedal V; Gothelf KV A DNA-Mediated Homogeneous Binding Assay for Proteins and Small Molecules. J. Am. Chem. Soc 2014, 136 (31), 11115–11120. [DOI] [PubMed] [Google Scholar]
- (68).Plattner C; Höfener M; Sewald N One-Pot Azidochlorination of Glycals. Org. Lett 2011, 13 (4), 545–547. [DOI] [PubMed] [Google Scholar]
- (69).Winans KA; King DS; Rao VR; Bertozzi CR A Chemically Synthesized Version of the Insect Antibacterial Glycopeptide, Diptericin, Disrupts Bacterial Membrane Integrity. Biochemistry 1999, 38 (36), 11700–11710. [DOI] [PubMed] [Google Scholar]
- (70).Miermont A; Barnhill H; Strable E; Lu X; Wall KA; Wang Q; Finn MG; Huang X Cowpea Mosaic Virus Capsid: A Promising Carrier for the Development of Carbohydrate Based Antitumor Vaccines. Chem. - Eur. J 2008, 14 (16), 4939–4947. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.