Abstract
BACKGROUND
Ectopic expression of miRNAs promotes tumor development and progression. miRNA (miR)-320a is downregulated in many cancers, including gastric cancer (GC). However, the mechanism underlying its downregulation and the role of miR-320a in GC are unknown.
AIM
To determine expression and biological functions of miR-320a in GC and investigate the underlying molecular mechanisms.
METHODS
Quantitative real-time polymerase chain reaction (PCR) was used to determine expression of miR-320a in GC cell lines and tissues. TargetScanHuman7.1, miRDB, and microRNA.org were used to predict the possible targets of miR-320a, and a dual luciferase assay was used to confirm the findings. Western blotting was used to detect the protein levels of pre-B-cell leukemia homeobox 3 (PBX3) in GC cells and tissue samples. Cell Counting Kit-8 proliferation, Transwell, wound healing, and apoptosis assays were performed to analyze the biological functions of miR-320a in GC cells. Methylation-specific PCR was used to analyze the methylation level of the miR-320a promoter CpG islands. 5-Aza-2’-deoxycytidine (5-Aza-CdR) and trichostatin A (TSA) were used to treat GC cells.
RESULTS
miR-320a expression was lower in GC cell lines and tissues than in the normal gastric mucosa cell line GES-1 and matched adjacent normal tissues. miR-320a overexpression suppressed GC cell proliferation, invasion and migration, and induced apoptosis. PBX3 was a target of miR-320a in GC. The methylation level of the miR-320a promoter CpG islands was elevated and this was partly reversed by 5-Aza-CdR and TSA.
CONCLUSION
miR-320a acts as a tumor suppressor and inhibits malignant behavior of GC cells, partly by targeting PBX3. DNA methylation is an important mechanism associated with low expression of miR-320a.
Keywords: Gastric cancer, miRNA-320a, DNA methylation, Pre-B-cell leukemia homeobox 3, Tumor suppressor
Core tip: miRNA (miR)-320a functioned as a tumor suppressor and was downregulated in gastric cancer (GC). miR-320a overexpression suppressed proliferation, migration and invasion, and induced apoptosis through targeting Pre-B-cell leukemia homeobox 3 in GC cells. miR-320a depletion showed the opposite results. The potential mechanism of miR-320a deficiency in GC was the increased methylation level of the miR-320a promoter CpG islands.
INTRODUCTION
Gastric cancer (GC) is a common malignant tumor. Although the incidence and mortality of GC have substantially decreased over the last decade in most countries worldwide, the prevalence of GC is still ranked fifth and GC mortality is ranked third in the world[1]. The occurrence and development of advanced GC involve a complex process. Efficient methods for early diagnosis and curative treatment for GC are currently lacking. As precision medicine is the future direction in cancer therapy, identifying an early diagnostic or therapeutic biomarker for GC is essential.
MicroRNAs (miRNAs) are short [19–25 nucleotides (nt)] noncoding RNAs that recognize specific target mRNAs and participate in the post-transcriptional regulation of gene expression by promoting degradation and inhibiting translation of mRNAs[2]. It was previously thought that only regulatory proteins encoded by genes could control physiological functions. However, in recent years, researchers have found that miRNAs play an important role in regulating cell differentiation and cell fate decisions[3]. They can act as oncogenes or tumor suppressors according to their target genes[4]. For example, miR-98 is expressed at low levels and acts as a tumor suppressor in hepatocellular carcinoma by targeting Sal-like protein 4, and the highly expressed miR-27a-3p functions as an oncogene in GC by targeting the B-cell translocation gene 2[5,6]. Abnormal expression of miRNAs occurs in many types of cancer, and miRNAs are involved in tumorigenesis and tumor progression[7]. miR-212 is significantly downregulated in GC and is involved in GC development and progression by regulating expression of oncogene Myc[8]. miRNAs also play an important role in regulating cancer cell proliferation, invasion, migration and apoptosis[9,10].
The function of miR-320a, located on chromosome 8p21.3, was investigated previously. miR-320a acts as a tumor-suppressive miRNA in many cancers, including blood malignancies and solid tumors. Xishan et al[11] showed that miR-320a is downregulated in chronic myeloid leukemia (CML) and inhibits CML cell migration, invasion and proliferation, and promotes apoptosis by targeting the BCR/ABL oncogene. miR-320a inhibits multiple myeloma cell proliferation and induces apoptosis by targeting pre-B-cell leukemia homeobox 3 (PBX3)[12]. miR-320a is downregulated in many solid tumors and has important functions. mir-320a plays a tumor-suppressing role in colorectal cancer, nasopharyngeal carcinoma, breast cancer, and bladder carcinoma, and overexpression of miR-320a partly inhibits tumor malignant behavior[13-16]. However, the mechanism underlying the downregulation of these miRNAs is unknown.
Epigenetic regulation plays a crucial role in the development and progression of tumors, and DNA methylation is an important part of this process. The expression of miRNAs is regulated by DNA methylation, and abnormal DNA hypermethylation can lead to cancer suppressor gene silencing and promotion of tumor progression. Ayala-Ortega et al[17] showed that DNA hypermethylation at the miR-181c promoter region results in low miR-181c expression in glioblastoma cell lines compared with normal brain tissues. High methylation levels of the miR-27b promoter region downregulate expression of miR-27b, whereas demethylation restores miR-27b expression in breast cancer[18]. Downregulation of miR-320a is associated with regulation by methylation in breast cancer[19].
miRNAs are expressed in a tissue-specific manner. However, whether miR-320a acts as a tumor suppressor in GC is unknown. Wang et al[20] found that the expression of miR-320a was reduced in GC tissues. However, the biological function and epigenetic regulatory mechanism of miR-320a in GC remains unknown.
In this study, we identified the biological role of miR-320a in GC and clarified the relationship between miR-320a expression and DNA methylation. miR-320a expression was reduced in GC cell lines and tissues. miR-320a interacted with the 3′ untranslated region (UTR) of the oncogene PBX3 in GC. miR-320a overexpression inhibited malignant biological actions in GC cells. These results suggest that miR-320a acted as a tumor suppressor by regulating PBX3 expression in GC. The promoter CpG islands of miR-320a showed abnormal hypermethylation, and the methylation inhibitor 5-aza-2′-deoxycytidine (5-Aza-dC) partially reversed miR-320a expression. These findings demonstrated that methylation-associated silencing of miR-320a suppressed tumor progression by targeting PBX3 in GC.
MATERIALS AND METHODS
Clinical GC samples
This study was approved by the Ethics Committee of the Fourth Affiliated Hospital, China Medical University (Shenyang, China). We obtained 84 GC tissues and matched adjacent normal tissues (located > 5 cm from the tumor) from patients who had a diagnosis of GC confirmed by histopathology at the Cancer Research Institute of China Medical University (Shenyang, China) between 2013 and 2014. These patients did not receive chemotherapy before surgical resection, and all tissues were immediately frozen in liquid nitrogen after surgery until DNA or RNA extraction. The basic patient data are listed in Table 1.
Table 1.
Clinicopathological characteristics of patients with gastric cancer
| Clinicopathological characteristics | No. of patients | miR-320a low expression | miR-320a high expression | χ2 | P value |
| Age (yr) | |||||
| ≤ 60 | 25 | 15 | 10 | 0.285 | 0.594 |
| > 60 | 59 | 39 | 20 | ||
| Gender | |||||
| Female | 20 | 11 | 9 | 0.986 | 0.321 |
| Male | 64 | 43 | 21 | ||
| Location | |||||
| Gastric body | 20 | 15 | 5 | 1.524 | 0.467 |
| Gastric antrum | 52 | 31 | 21 | ||
| Gastric fundus | 12 | 8 | 4 | ||
| Tumor size (cm) | |||||
| < 5 | 25 | 15 | 10 | 0.285 | 0.594 |
| ≥ 5 | 59 | 39 | 20 | ||
| Differentiation | |||||
| Poor | 55 | 40 | 15 | 4.945 | 0.026 |
| Well and moderate | 29 | 14 | 15 | ||
| TNM stage | |||||
| I+II | 34 | 15 | 19 | 10.120 | 0.001 |
| III+ IV | 50 | 39 | 11 | ||
| Lymph node metastasis | |||||
| No | 18 | 7 | 11 | 6.436 | 0.011 |
| Yes | 66 | 47 | 19 |
Cell types and source and cell culture
We used five cell lines, including one normal gastric mucosa cell line (GES-1) and four GC cell lines (BGC-823, MGC-803, SGC-7901 and MKN-45). All cell lines were purchased from the Institute of Biochemistry and Cell Biology, China Academy of Science (Shanghai, China). Cells were cultured in RPMI 1640 medium (Invitrogen, Carlsbad, CA, USA) containing 10% fetal bovine serum (FBS; Gibco/BRL, Waltham, MA, USA) at 37°C in 5% CO2.
Cell transfection
The miR-320a mimics and miR-320a scramble mimics were designed and produced by GenePharma (Shanghai, China) and transfected into MKN-45 cells. The final concentration for transfection was 50 nmol/L. The miR-320a inhibitor and miR-320a scramble inhibitor were designed and produced by GenePharma and transfected into BGC-823 cells. The final concentration for transfection was 100 nmol/L. Transfection was performed using Lipofectamine 3000 Transfection Reagent (Invitrogen) according to the manufacturer’s instructions. Experiments were carried out in triplicate.
RNA extraction and quantitative real-time polymerase chain reaction (PCR)
TRIzol reagent (Invitrogen) was used to extract total RNA from cell lines, GC tissues, and matched adjacent normal tissues. RNA concentration and purity were determined spectroscopically, and samples were stored at -80°C long term or -20°C short term. The miRNA RT-PCR Quantitation Kit (GenePharma) was used for miR-320a reverse transcription using the following protocol: 30 min at 25°C, 30 min at 42°C, and 5 min at 85°C. The cDNA product was subjected to quantitative real-time PCR using the following program: 3 min at 95°C, 40 cycles of 12 s at 95°C, and 40 s at 62°C. All mRNA quantification data were normalized to U6. The primers were designed and produced by GenePharma and the primer sequences are shown in Table 2.
Table 2.
Primer/mimic sequences
| Name | Sequence (5’–3’) |
| miR-320a reverse transcription primer | |
| miR-320a | GTCTGTATGGTTGTTCTCGACTCCTTCACATCCCTATCCAACCATACAGACTCGCCCTC |
| U6 snRNA | GGAACGCTTCACGAATTTG |
| Real-time PCR primer sequence | |
| miR-320a | FO:AAGGGATCGCGGGCG |
| RE:TGCGTGTCGTGGAGTC | |
| U6 snRNA | FO:ATTGGAACGATACAGAGAAGATT |
| RE:GGAACGCTTCACGAATTTG | |
| MSP primer sequence | |
| Methylated | FO:ACGTCGTAATGTGAGGATTC |
| RE:CGCAAACAAAACTCGATATAA | |
| Nonmethylated | FO:ATTATGTTGTAATGTGAGGATTT |
| RE:CACAAACAAAACTCAATATAACCC | |
| miR-320a mimics sequence | |
| miR-320a | Sense: AAAAGCUGGGUUGAGAGGGCGA |
| Antisense: GCCCUCUCAACCCAGCUUUUUU | |
| Negative control | Sense: UUCUUCGAACGUGUCACGUTT |
| Antisense: ACGUGACACGUUCGGAGAATT | |
| PBX3 primer sequence | FO:GCCAAATTGACCCAGATCAGAC |
| RE:GAAATGGGACGTGTTCTACTCTGTT | |
PCR: Polymerase chain reaction.
Cell viability and apoptosis assay
GC cells (MKN-45 and BGC-823) were inoculated into 96-well plastic dishes containing 10% CCK-8 (Dojindo, Kumamoto, Japan) diluted in culture medium and incubated at 37°C in 5% CO2 after transfection with miR-320a mimics, scramble mimics, miR-320a inhibitor, scramble inhibitor, or no transfection. The OD value at 0, 12, 24, 48, and 72 h after transfection was measured using an enzyme assay. All experiments were performed three times. GC cells (n = 106) transfected with miR-320a mimics, scramble mimics, miR-320a inhibitor, scramble inhibitor, or no transfection were inoculated into six-well plates after transfection for 48 h, and all cells were harvested at 72 h. The Annexin V-PE/7AAD Apoptosis Detection Kit (KeyGen, Jiangsu, China) was used to determine apoptosis.
Cell migration and invasion assays
GC cells (MKN-45 and BGC-823) were inoculated into six-well plastic dishes and transfected with miR-320a mimics, scramble mimics, miR-320a inhibitor, scramble inhibitor or no transfection for 36 h. Cuts were then made using a 200-μL pipette tip. The wound healing percentage was measured at 0, 24, and 48 h after transfection using ImageJ software to evaluate the migration ability of GC cells. A Transwell assay was used to determine the invasion ability of GC cells (MKN-45 and BGC-823). A Matrigel-coated membrane matrix (UnivBio, Shanghai, China) was used to simulate a physiological matrix membrane between the upper and lower chambers. GC cells (n = 105) transfected with miR-320a mimics, scramble mimics, miR-320a inhibitor, scramble inhibitor, or no transfection were inoculated into the upper chamber filled with serum-free medium. Medium containing 10% FBS was added to the lower chamber. After 24 h, cotton wool was used to remove the noninvading cells in the upper chamber, and crystal violet (Tiangen Biotech, Beijing, China) was used to stain the invasive cells on the lower surface of the chamber. The number of invasive cells was counted under an inverted microscope. All experiments were repeated three times.
5-Aza-CdR and trichostatin A (TSA) treatments
MKN-45 cells were inoculated into six-well plates. Cells were treated with 5-Aza-CdR (Sigma–Aldrich, St Louis, MO, USA) at 0.5, 1, or 1.5 μmol/L for 72 h, or with TSA (Beyotime, Beijing, China) at 300 nmol/L for 24 h. TSA was also added to the medium for the final 24 h of the 72 h 5-Aza-CdR (0.5 μmol/L) treatment period. To maintain drug effectiveness, the medium containing the drug was replaced every 24 h. Cells were then harvested, and RNA from MKN-45 cells was purified using the TRIzol reagent. cDNA synthesis was performed as described previously, and 1 mL of diluted cDNA from each sample was amplified by quantitative PCR using a previously described protocol.
DNA isolation and bisulfite modification
Genomic DNA was isolated from primary GC samples (cancer tissues and matched adjacent normal tissues) and GC cells, and harvested after transfection for 48 h with miR-320a mimics, scramble mimics, or no transfection. The MiniBEST Universal Genomic DNA Extraction Kit (TaKaRa, Shiga, Japan) was used for DNA isolation. The genomic DNA underwent bisulfite modification using the EZ DNA Methylation-Gold Kit (TaKaRa). DNA (400 ng) was diluted to 20 μL, and 130 μL of the conversion reagent (900 μL water, 50 μL M-dissolving buffer, 300 μL M-dilution buffer) was added. Bisulfite conversion was carried out as follows: 98°C for 10 min, 64°C for 150 min, and 4°C for 5 min, and DNA was recovered using a Zymo-Spin column (Zymo Research, Irvine, CA, USA). Bisulfite-treated DNA was used for methylation analyses.
Methylation analysis
The methylation status of the miR-320a promoter CpG islands in cell lines and tissues was analyzed using methylation-specific PCR (MSP). The MSP primers were designed using MethPrimer (http://www.urogene.org/methprimer) and are listed in Table 2. The PCR product length was 149 bp. The amplification reaction volume was 25 μL, and the reaction procedure was as follows: One cycle at 94°C for 5 min, 40 cycles at 94°C for 30 min, 58°C for 30 min, 72°C for 30 min, and one cycle at 72°C for 10 min. Agarose gel electrophoresis was performed to analyze the MSP products.
Dual luciferase assay
Luciferase activity was analyzed using a dual luciferase reporter assay system (Promega, Shanghai, China). miR-320a mimics, miR-320a negative control miRNA, wild-type PBX3 (ACAGCUUUA), or mutant PBX3 (ACACCCUUA) was transfected into MKN-45 cells for 48 h using Lipofectamine 3000 (Invitrogen). Experiments were repeated three times.
Protein isolation and Western blotting
Total protein was extracted from GC tissues and cells (MKN-45 and BGC-823) after transfection for 48 h with miR-320a mimics, scramble mimics, miR-320a inhibitor, scramble inhibitor, or no transfection using RIPA buffer (Beyotime, Shanghai, China), and protein concentration was determined using a BCA Protein Assay Kit (TaKaRa, Dalian, China). Appropriate protein samples were separated by SDS-PAGE (TaKaRa, Dalian, China), and electrophoresis was carried out for 100 min at 120 V. The proteins were then transferred to polyvinylidene difluoride membranes (Millipore, Shanghai, China), which were blocked in bovine serum albumin (BSA) for 2 h at room temperature. Rabbit anti-PBX3 monoclonal antibody (1:3000; Abcam, Cambridge, UK; #Ab109173) was added overnight at 4°C. Rabbit anti-GAPDH monoclonal antibody (1:3000; ABclonal, Boston, MA, USA) was used as the internal reference. Goat anti-rabbit secondary antibody was added to the polyvinylidene difluoride membranes for 2 h the following day. Then, a chemiluminescence instrument (Tanon, Shanghai, China) was used to determine the MSP product levels.
Histology
All samples were incubated overnight in buffered formalin, after which they were embedded in paraffin, cut into 3-μm-thick sections, and stained with hematoxylin–eosin (HE).
Bioinformatics and statistical analysis
The miR-320a target gene was predicted by TargetScanHuman7.1 (http://www.targetscan.org/vert_71/), miRDB (http://www.mirdb.org/miRDB/), and microrna.org (http://www.microrna.org/microrna/home.do). MethPrimer (http://www.urogene.org/methprimer2/index.html) was used to predict the miR-320a promoter CpG islands.
Statistical analysis was performed using SPSS version 17.0 (SPSS, Chicago, IL, USA). X2 test or Student’s t test (two-tailed) were used. P < 0.05 was considered statistically significant.
RESULTS
miR-320a expression in GC tissues and its associations with clinicopathological characteristics
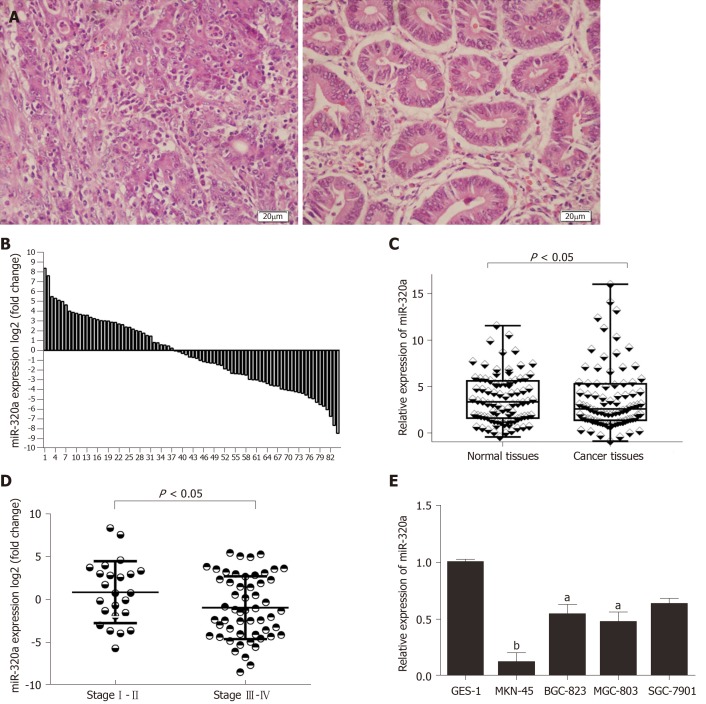
miR-320a expression in GC tissues was measured by quantitative real-time PCR. Assessment of miR-320a expression in 84 GC tissues and matched adjacent normal tissues (Figure 1A) showed that miR-320a was downregulated in 54 samples (54/84, 64%) compared with matched adjacent normal tissues, and the difference was significant (Figure 1B, 1C). To assess the association between miR-320a expression and clinicopathological characteristics, the tissues were separated into two groups: upregulated and downregulated miR-320a groups. miR-320a expression was significantly associated with TNM stage, tumor differentiation, and lymph node metastasis (Table 1 and Figure 1D). These results suggest that miR-320a is involved in GC progression.
Figure 1.
Expression of miR-320a in gastric cancer samples and cell lines, and miR-320a downregulation is correlated with tumor TNM stage. A: Representative tissue slices from gastric cancer (GC) patients were stained using HE; scale bars 20 μm; B: miR-320a expression in 84 pairs of GC tissues and adjacent nontumor tissues is presented as log2 of fold change. Expression of miR-320a in these 84 pairs of samples was determined by quantitative PCR; C: Comparison of expression of miR-320a in GC tissues and matched normal tissues (P < 0.05); D: mir-320a expression in stage I/II (n = 23) and stage III/IV (n = 54) and the results are shown as log2 of fold change in GC tissues relative to normal tissues (P < 0.05); E: Expression of miR-320a in GES-1 and GC cell lines (MKN-45, BGC-823, MGC-803 and SGC-7901). aP < 0.05, bP < 0.01. The reference genes were set using U6 snRNA. All the experiments were conducted in triplicate. Data were analyzed by ANOVA or t test. The data are reported as means ± SD.
miR-320a is downregulated in GC cell lines
miR-320a expression was assessed in four GC cell lines (BGC-823, MGC-803, SGC-7901 and MKN-45) and one normal gastric mucosa cell line (GES-1). miR-320a expression was lower in GC than in GES-1 cells, and particularly downregulated in MKN-45 cells (Figure 1E). These data suggest that miR-320a expression is associated with gastric carcinoma. MKN-45 and BGC-823 cells were used for subsequent miR-320a mimic and inhibitor transfection experiments, respectively.
miR-320a overexpression inhibits GC cell viability and induces apoptosis in vitro
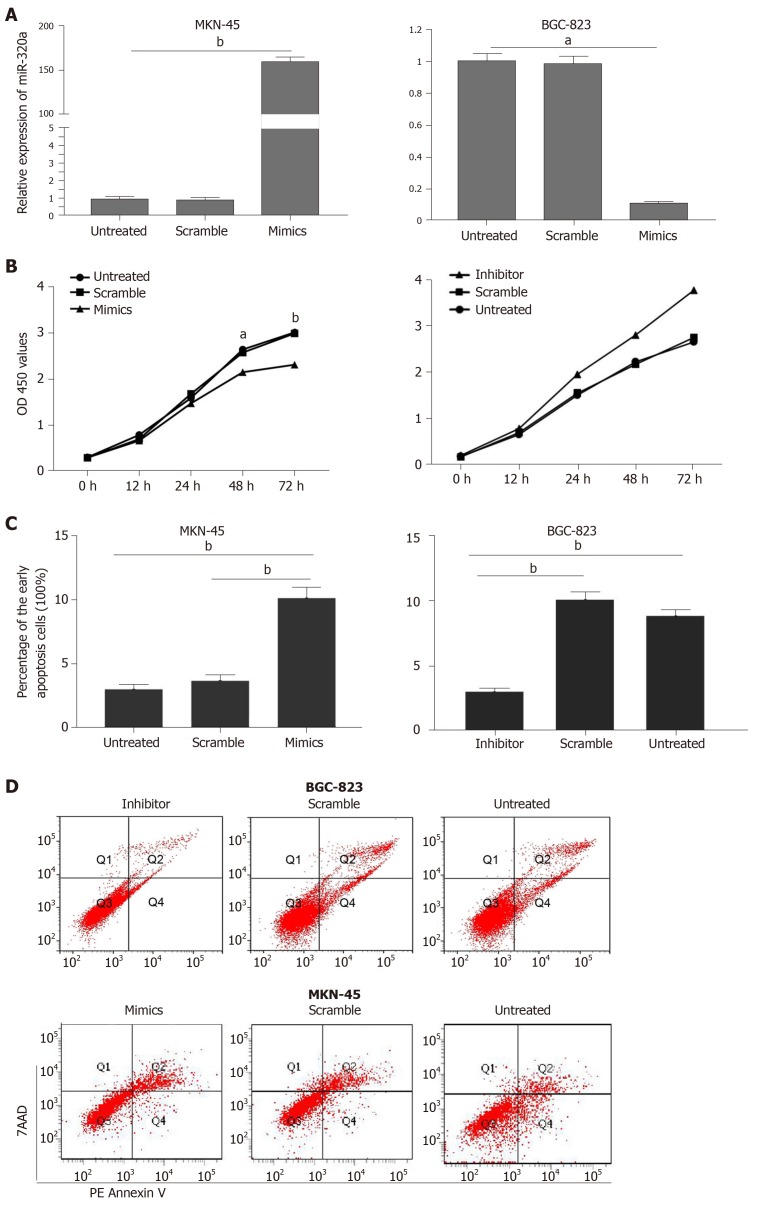
To determine the role and potential biological function of miR-320a in GC, GC cells were transfected with miR-320a mimics and miR-320a inhibitor, and the effects of miR-320a up- and downregulation on cell function were evaluated. miR-320a mimics transfection significantly upregulated, whereas miR-320a inhibitor significantly downregulated miR-320a expression compared with that in the other groups (Figure 2A). The CCK-8 assay was performed to estimate the effect of miR-320a on the viability of GC cells. We found that overexpression of miR-320a inhibited MKN-45 cell viability, whereas miR-320a depletion promoted BGC-823 cell viability (Figure 2B). Because apoptosis is an important part of the cell life cycle, we determined the effect of miR-320a on the early apoptosis rate of GC cells. miR-320a mimics accelerated MKN-45 cell apoptosis, whereas miR-320a inhibitor suppressed BGC-823 cell apoptosis, compared with that in the scramble and untreated groups (Figure 2C, 2D).
Figure 2.
Overexpression of miR-320a inhibited gastric cancer cell viability and promoted apoptosis. A: Expression of miR-320a in gastric cancer (GC) cells was determined by quantitative PCR after transfection with miR-320a mimics (50 nmol/L), scramble mimics, miR-320a inhibitor (100 nmol/L), scramble inhibitor or no transfection; B: CCK8 assay was performed to determine cell viability after transfection at 0, 12, 24 and 72 h; C: Early apoptosis rate in the three groups is shown in the histogram; D: Flow cytometry was used to determine apoptosis after transfection for 48 h. The data are reported as means ± SD. aP < 0.05, bP < 0.01.
miR-320a overexpression inhibits GC cell migration and invasion in vitro
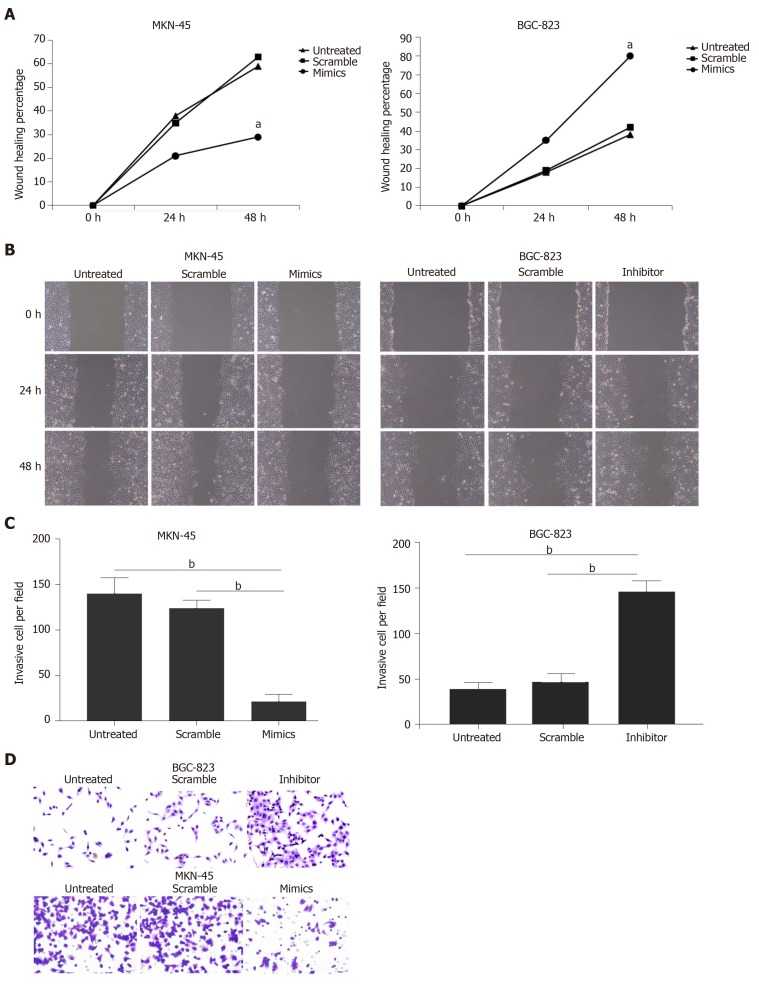
Wound scratch and Transwell invasion assays were used to determine the effects of miR-320a on the metastatic capacity and invasiveness of GC cells, which are important malignant activities of tumor cells. GC cells were divided into three treatment groups: miR-320a mimics/inhibitor, scramble mimics/inhibitor, and untreated. Wound healing was suppressed in cells treated with miR-320a mimics, whereas it was accelerated in cells treated with miR-320a inhibitor compared with that in the other two groups at 24 and 48 h after scratching (Figure 3A and 3B). The number of cells traversing the Matrigel matrix was lower in cells treated with miR-320a mimics, whereas it was higher in groups treated with miR-320a inhibitor than in the other two groups (Figure 3C and 3D). These data suggest that miR-320a overexpression inhibits GC cell migration and invasion to overcome the malignant properties of GC cells.
Figure 3.
Overexpression of miR-320a suppressed gastric cancer cell migration and invasion. A, B: Wound scratch assay was used to assess migration of MKN-45 cells. Wound healing percentage was measured at 0, 24 and 48 h after transfection with miR-320a mimics (50 nmol/L), scramble mimics, miR-320a inhibitor (100 nmol/L), scramble inhibitor or no transfection; C, D: Transwell invasion assay was carried out to determine gastric cancer cell invasion after transfection with miR-320a mimics/inhibitor, scramble mimics/inhibitor or no transfection for 48 h. Cell invasion was assessed at 24 h. aP < 0.05, bP < 0.01.
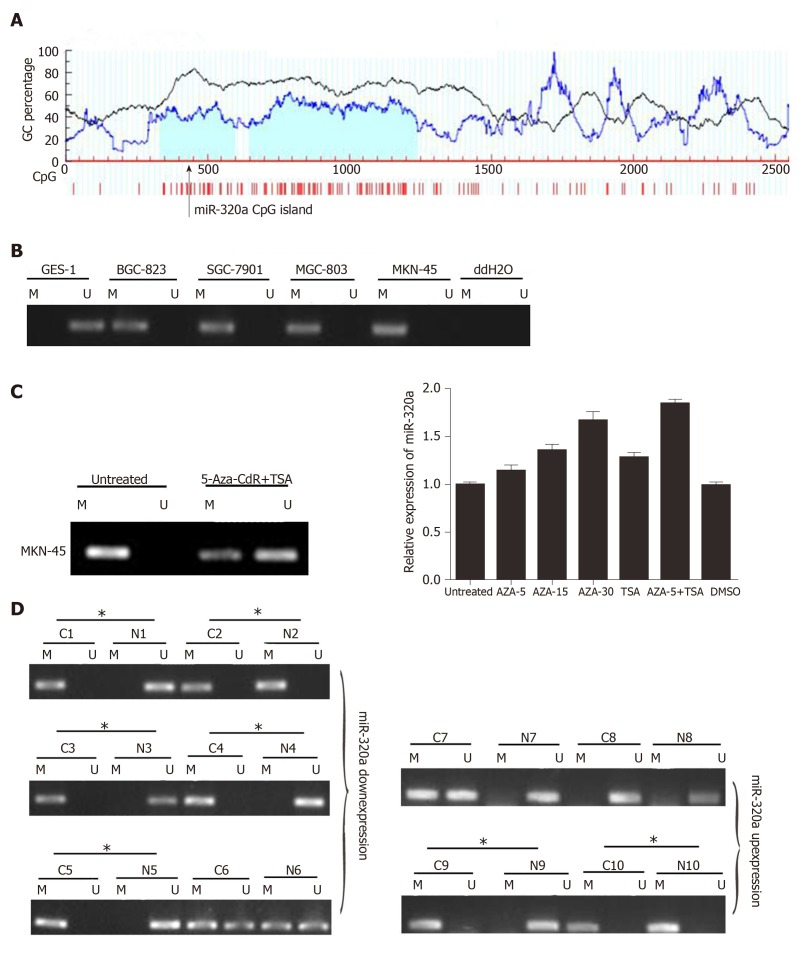
miR-320a expression is regulated by DNA methylation
To determine the cause of miR-320a downregulation, we analyzed the relationship between DNA methylation and miR-320a expression. A search of the human genome database identified CpG islands around the miR-320a promoter (Figure 4A). MSP was used to detect the methylation level of miR-320a in the four GC cell lines and normal GES-1 cell line. miR-320a promoter CpG islands were hypermethylated in GC cells, whereas no methylation was observed in GES-1 cells (Figure 4B). To examine further the effect of methylation, MKN-45 cells were treated with 5-Aza-CdR (DNA methylation inhibitor) and TSA (histone deacetylase inhibitor), and the expression and methylation levels of miR-320a were determined by quantitative real-time PCR and MSP, respectively. miR-320a expression was partially reversed by 5-Aza-CdR and TSA, particularly by the combination of 5-Aza-CdR and TSA, and the methylation level of the miR-320a promoter CpG islands was markedly reduced (Figure 4C). To determine the status of miR-320a CpG islands in GC samples, methylation levels were measured in six pairs of GC samples (primary cancer tissues and matched adjacent normal tissues) with low miR-320a expression and four pairs of GC samples with high miR-320a expression levels. miR-320a CpG islands tended to be hypermethylated in the miR-320a downregulated group (83%, 5/6) compared with the miR-320a upregulated group (50%, 2/4) (Figure 4D). These findings indicated that DNA methylation plays an important role in the downregulation of miR-320a expression in GC.
Figure 4.
Expression of miR-320a and methylation status of its promoter CpG islands in gastric cancer cell lines and tissue samples. A: Schematic of the location of miR-320a promoter CpG islands obtained from the human genome database and MethPrimer 2.0 (online); B: Methylation level of miR-320a in GES-1 and four gastric cancer (GC) cell lines; C: miR-320a expression and its promoter CpG island methylation level after 5-Aza-CdR and/or TSA treatment of MKN-45 cells; D: Representative MSP results of miR-320a promoter CPG islands in GC samples (cancer tissues and matched adjacent normal tissues). Cases with hypermethylation are marked. M: Methylated; U: Unmethylated; C: Cancer tissues; N: Normal tissues; *: Hypermethylation.
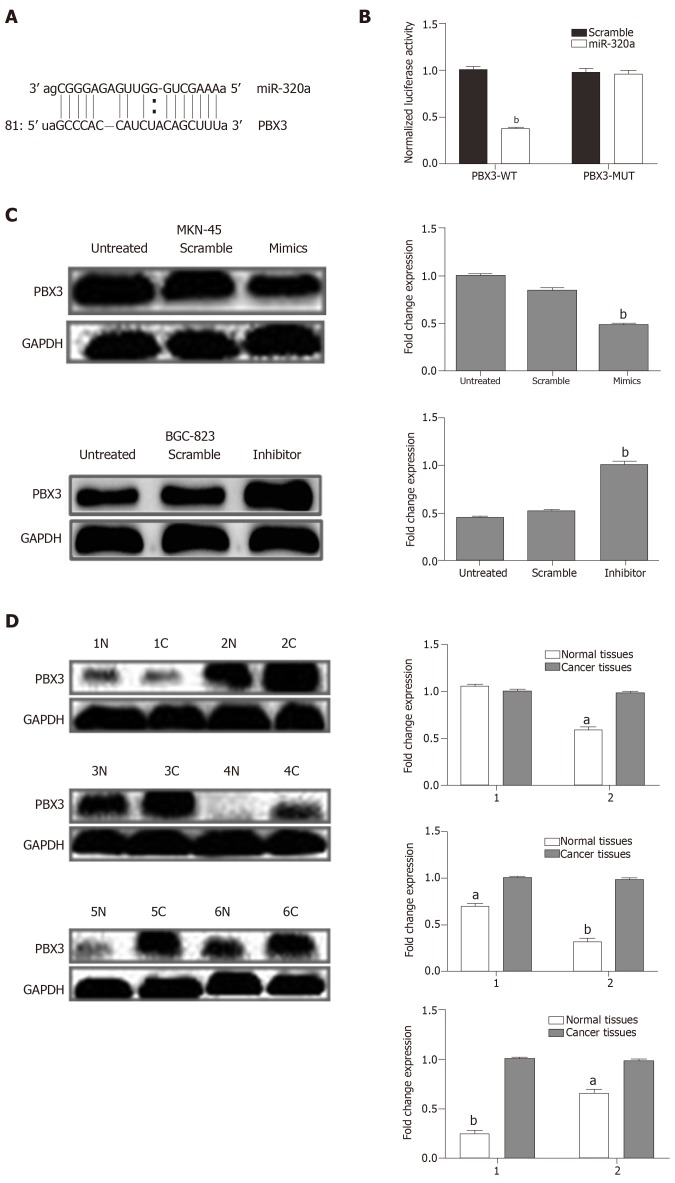
miR-320a directly targets the oncogene PBX3 in GC
To examine the mechanism underlying the effect of miR-320a in GC, online software programs (TargetScanHuman7.1, miRDB, and microRNA.org) were used to predict the target gene of miR-320a. The three databases predicted the interaction of miR-320a with the 3′-UTR of PBX3 (Figure 5A). The dual luciferase assay confirmed that PBX3 was a direct target gene of miR-320a, and the regulatory activities are shown in Figure 5B. To determine whether miR-320a modulates PBX3 protein expression, six pairs of samples (GC tissues and matched adjacent normal tissues) with low miR-320a expression were analyzed by western blotting using anti-PBX3 antibodies. The results showed that PBX3 was significantly upregulated in five pairs of GC tissues compared with matched adjacent normal tissues (Figure 5D). Assessment of PBX3 protein expression in MKN-45 cells transfected with miR-320a mimics, scramble mimics, or no transfection showed that PBX3 was significantly downregulated in cells transfected with miR-320a mimics compared with the other two groups (Figure 5C).
Figure 5.
PBX3 was a direct target of miR-320a. A: Banding region between miR-320a and PBX3 was predicted by microRNA.org; B: Relative luciferase activities of PBX3 in wild type and mutant; C: PBX3 protein level in gastric cancer (GC) cells after transfection with miR-320a mimics, scramble mimics, miR-320a inhibitor, scramble inhibitor or no transfection for 48 h; D: Protein level of PBX3 in GC tissues and matched adjacent normal tissues. Reference protein was set using GAPDH. C: Cancer tissues; N: Normal tissues; aP < 0.05; bP < 0.01.
DISCUSSION
miRNAs act as oncogenes or tumor suppressor genes and have important functions in the progression of many types of cancers[4]. In the present study, we found that miR-320a was significantly downregulated in GC cell lines and tissues. miR-320a overexpression inhibited GC cell proliferation, invasion and migration, and induced apoptosis in MKN-45 cells. Further studies demonstrated that low miR-320a expression was partly due to hypermethylation of the promoter CpG islands of miR-320a. PBX3 was identified as a direct target gene of miR-320a. Our findings demonstrated that miR-320a plays a tumor-suppressing role in GC, and DNA methylation is a crucial mechanism underlying miR-320a silencing.
Recent studies show that miR-320a is downregulated in many types of cancers, including breast, colorectal and ovarian cancers, and is involved in several biological functions by regulating specific target genes[11-16]. Xie et al[21] showed that miR-320a is markedly decreased in hepatocellular carcinoma, and the lack of miR-320a promotes cell migration and invasion. Decreased expression of miR-320a promotes proliferation and invasion of non-small cell lung cancer by targeting voltage dependent anion channel 1[22]. These data demonstrate the involvement of miR-320a in cancer. In this study, miR-320a expression was significantly lower in GC cells and tissues than in the normal GES-1 cell line and matched adjacent normal tissues. Assessment of the effect of miR-320a on the malignant biological behavior of GC cells showed that miR-320a overexpression prevented GC cell proliferation, invasion and migration, and promoted apoptosis compared with those in control MKN-45 cells. These data demonstrated that miR-320a acted as a tumor suppressor in GC. However, these findings need to be confirmed using a larger data sample.
PBX3, located on chromosome 9q33.3, belongs to the highly conserved PBX family of proteins, which are members of the PBX class of three-amino-acid loop extension superclass of homeodomain proteins[23]. PBX3 is overexpressed in many types of cancers[24-26], and plays a crucial role by activating various signaling pathways including the MAPK/ERK signaling pathway[24,26]. These data showed the powerful function of PBX3. PBX3 can act as an oncogene, promoting cell proliferation and colony formation, and its expression is associated with the clinicopathological characteristics of GC patients[27]. In the present study, Western blot analysis showed that PBX3 was upregulated in GC tissues and cells. miRNAs function by inhibiting translation or promoting degradation of target mRNAs[2]. PBX3 is regulated by multiple miRNAs in cancer, including let-7d, let-7c, miR-200b, miR-222, miR-424, and miR-181[25,26,28]. In multiple myeloma, PBX3 is regulated by miR-320a and involved in disease progression[12]. However, whether PBX3 is regulated by miR-320a in GC remains unknown. Our findings showed that miR-320a directly interacted with the 3′-UTR of PBX3 and downregulated PBX3 protein expression in GC, suggesting a mechanism underlying the post-transcriptional control of PBX3 by miR-320a.
DNA methylation plays an important role in the expression of tumor suppressor miRNAs[29]. Many miRNAs show hypermethylation of promoter CpG islands, including classic miRNAs such as miR-335 in GC, miR-181 in glioblastoma, and miR-342 in colorectal cancer[17,30,31]. However, the mechanism underlying the downregulation of miR-320a in GC is unknown. We found that CpG islands were present around miR-320a by searching the human genome database. He et al[19] reported that miR-320a was hypermethylated in colorectal cancer, which led us to hypothesize that low miR-320a expression is associated with DNA methylation in GC. MSP showed that, compared with normal GES-1 cells and matched adjacent normal tissues, miR-320a promoter CpG islands were hypermethylated in GC cell lines and GC tissues in which miR-320a was downregulated. Furthermore, the level of miR-320a hypermethylation was partially reversed by the demethylating agent 5-Aza-CdR. Overall, these data demonstrate that the expression of miR-320a is regulated by DNA methylation in GC.
There were some limitations in the present study. First, our study was a small, retrospective, single-institution study, and further larger, multicenter studies are required to validate our results. Second, although the expression of miR-320a is regulated by DNA methylation, the specific mechanisms remain largely undefined. Further detailed studies are currently underway to explore the participation of miR-320a in this process, to provide more comprehensive information about the functions of miR-320a in cancer.
In conclusion, we showed that miR-320a was downregulated and played a tumor- suppressing role by regulating the oncogene PBX3 in GC. miR-320a overexpression inhibited cell proliferation, invasion and migration, and induced cell apoptosis. miR-320a expression was regulated by DNA methylation and was partially reversed by 5-Aza-CdR and TSA. These data indicate that miR-320a may be a potential therapeutic target in GC.
ARTICLE HIGHLIGHTS
Research background
Gastric cancer (GC) is a common type of malignant tumor with poor prognosis and presents a serious threat to human health. In February 2018 , the latest statistics from the Chinese National Cancer Center showed that although the overall incidence of GC is declining, it remains second in terms of incidence among all malignancies in China, just below lung cancer. At present, there is no efficient early diagnosis and curative treatment strategy for GC. As precision medicine is the future direction in cancer therapy, it is important to identify an early diagnostic or therapeutic biomarker of GC.
Research motivation
To explore the expression pattern, biological function and potential mechanism of miRNA (miR)-320a in GC, and to determine whether miR-320a functions as an early diagnostic or therapeutic biomarker in GCaccording to its expression and biological functions.
Research objectives
miR-320a mimic and inhibitor were transfected into GC cells for bidirectional regulation of the expression level of miR-320a. The effect of miR-320a on cell viability, migration, invasion and apoptosis was determined through a series of functional experiments. These results would provide scientific evidence for clinical treatment of GC.
Research methods
Quantitative real-time polymerase chain reaction (PCR) and Western blotting were performed to determine the levels of related factors. Methylation-specific PCR was applied for analysis of methylation status of the miR-320a promoter CpG islands in GC cell lines and tissues. CCK8, flow cytometry, Transwell invasion and wound healing assays were performed to determine the effect of miR-320a on cell behavior. Dual luciferase assay was performed to identify whether pre-B-cell leukemia homeobox (PBX) 3 was the target gene of miR-320a. TargetScanHuman7.1, miRDB, microrna.org and MethPrimer were used for bioinformatics analysis. Student’s t test (two-tailed) and analysis of variance (ANOVA) were used for statistical analysis.
Research results
miR-320a was downregulated in GC, and its expression deficiency was partly due to hypermethylation of the promoter CpG islands. miR-320a overexpression inhibited cell viability, migration and invasion, and induced apoptosis through targeting PBX3 in GC cells. miR-320a functioned as a tumor suppressor in vitro. However, its biological effect in a preclinical model should be further determined.
Research conclusions
miR-320a was downregulated in GC tissues and cells, and its abnormal expression was related to GC cell behavior. We confirmed that miR-320a overexpression inhibited cell viability, migration and invasion, and induced apoptosis in GC cells. miR-320a deficiency showed the opposite results. Consistent with previous research, miR-320a functioned as a tumor suppressor. miR-320a could be a biomarker for GC diagnosis and treatment. We found that miR-320a downregulation in GC was due to the elevated methylation level of the miR-320a promoter CpG islands. The elevation was partly reversed by 5-Aza-2’-deoxycytidine and trichostatin A. In addition, PBX3 was firstly identified as the target gene of miR-320a in GC.
Research perspectives
For future research, we will focus on the related signaling pathways through which miR-320a regulates GC cell biological behaviors. Screening for related signaling pathways of GC will be performed using the Cancer Genome Atlas database, followed by related factor detection using Western blotting and quantitative real-time PCR. Activator and inhibitor for target signaling will be applied to GC cells, and cell viability, cell cycle, cell migration, invasion and apoptosis will be determined.
ACKNOWLEDGEMENTS
We are grateful to the surgeons and general practitioners for their participation and patient contacts.
Footnotes
Institutional review board statement: Institutional review board approval of our hospital was obtained for this study.
Conflict-of-interest statement: The authors have no conflicts of interest to declare.
Data sharing statement: No additional data are available.
Open-Access: This article is an open-access article which was selected by an in-house editor and fully peer-reviewed by external reviewers. It is distributed in accordance with the Creative Commons Attribution Non Commercial (CC BY-NC 4.0) license, which permits others to distribute, remix, adapt, build upon this work non-commercially, and license their derivative works on different terms, provided the original work is properly cited and the use is non-commercial. See: http://creativecommons.org/licenses/by-nc/4.0/
Manuscript source: Unsolicited manuscript
Peer-review started: May 7, 2019
First decision: June 4, 2019
Article in press: July 28, 2019
Specialty type: Oncology
Country of origin: China
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Lo GH, Mathur A, Ko E S-Editor: Wang JL L-Editor: A E-Editor: Qi LL
Contributor Information
Yong-Shuang Li, Department of Gastrointestinal Surgery, the Fourth Affiliated Hospital of China Medical University, Shenyang 110032, Liaoning Province, China.
Ying Zou, Department of Gastrointestinal Surgery, the Fourth Affiliated Hospital of China Medical University, Shenyang 110032, Liaoning Province, China.
Dong-Qiu Dai, Department of Gastrointestinal Surgery, the Fourth Affiliated Hospital of China Medical University, Shenyang 110032, Liaoning Province, China. daidq63@163.com.
References
- 1.Ferro A, Peleteiro B, Malvezzi M, Bosetti C, Bertuccio P, Levi F, Negri E, La Vecchia C, Lunet N. Worldwide trends in gastric cancer mortality (1980-2011), with predictions to 2015, and incidence by subtype. Eur J Cancer. 2014;50:1330–1344. doi: 10.1016/j.ejca.2014.01.029. [DOI] [PubMed] [Google Scholar]
- 2.Tang H, Liu X, Wang Z, She X, Zeng X, Deng M, Liao Q, Guo X, Wang R, Li X, Zeng F, Wu M, Li G. Interaction of hsa-miR-381 and glioma suppressor LRRC4 is involved in glioma growth. Brain Res. 2011;1390:21–32. doi: 10.1016/j.brainres.2011.03.034. [DOI] [PubMed] [Google Scholar]
- 3.Ivey KN, Srivastava D. MicroRNAs as regulators of differentiation and cell fate decisions. Cell Stem Cell. 2010;7:36–41. doi: 10.1016/j.stem.2010.06.012. [DOI] [PubMed] [Google Scholar]
- 4.Chen CZ. MicroRNAs as oncogenes and tumor suppressors. N Engl J Med. 2005;353:1768–1771. doi: 10.1056/NEJMp058190. [DOI] [PubMed] [Google Scholar]
- 5.Zhou W, Zou B, Liu L, Cui K, Gao J, Yuan S, Cong N. MicroRNA-98 acts as a tumor suppressor in hepatocellular carcinoma via targeting SALL4. Oncotarget. 2016;7:74059–74073. doi: 10.18632/oncotarget.12190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhou L, Liang X, Zhang L, Yang L, Nagao N, Wu H, Liu C, Lin S, Cai G, Liu J. MiR-27a-3p functions as an oncogene in gastric cancer by targeting BTG2. Oncotarget. 2016;7:51943–51954. doi: 10.18632/oncotarget.10460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Calin GA, Croce CM. MicroRNA signatures in human cancers. Nat Rev Cancer. 2006;6:857–866. doi: 10.1038/nrc1997. [DOI] [PubMed] [Google Scholar]
- 8.Xu L, Wang F, Xu XF, Mo WH, Xia YJ, Wan R, Wang XP, Guo CY. Down-regulation of miR-212 expression by DNA hypermethylation in human gastric cancer cells. Med Oncol. 2011;28 Suppl 1:S189–S196. doi: 10.1007/s12032-010-9691-0. [DOI] [PubMed] [Google Scholar]
- 9.Zhang S, Zhang C, Liu W, Zheng W, Zhang Y, Wang S, Huang D, Liu X, Bai Z. MicroRNA-24 upregulation inhibits proliferation, metastasis and induces apoptosis in bladder cancer cells by targeting CARMA3. Int J Oncol. 2015;47:1351–1360. doi: 10.3892/ijo.2015.3117. [DOI] [PubMed] [Google Scholar]
- 10.Li Z, Lei H, Luo M, Wang Y, Dong L, Ma Y, Liu C, Song W, Wang F, Zhang J, Shen J, Yu J. DNA methylation downregulated mir-10b acts as a tumor suppressor in gastric cancer. Gastric Cancer. 2015;18:43–54. doi: 10.1007/s10120-014-0340-8. [DOI] [PubMed] [Google Scholar]
- 11.Xishan Z, Ziying L, Jing D, Gang L. MicroRNA-320a acts as a tumor suppressor by targeting BCR/ABL oncogene in chronic myeloid leukemia. Sci Rep. 2015;5:12460. doi: 10.1038/srep12460. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 12.Lu Y, Wu D, Wang J, Li Y, Chai X, Kang Q. miR-320a regulates cell proliferation and apoptosis in multiple myeloma by targeting pre-B-cell leukemia transcription factor 3. Biochem Biophys Res Commun. 2016;473:1315–1320. doi: 10.1016/j.bbrc.2016.04.069. [DOI] [PubMed] [Google Scholar]
- 13.Zhao H, Dong T, Zhou H, Wang L, Huang A, Feng B, Quan Y, Jin R, Zhang W, Sun J, Zhang D, Zheng M. miR-320a suppresses colorectal cancer progression by targeting Rac1. Carcinogenesis. 2014;35:886–895. doi: 10.1093/carcin/bgt378. [DOI] [PubMed] [Google Scholar]
- 14.Qi X, Li J, Zhou C, Lv C, Tian M. MicroRNA-320a inhibits cell proliferation, migration and invasion by targeting BMI-1 in nasopharyngeal carcinoma. FEBS Lett. 2014;588:3732–3738. doi: 10.1016/j.febslet.2014.08.021. [DOI] [PubMed] [Google Scholar]
- 15.Wang B, Yang Z, Wang H, Cao Z, Zhao Y, Gong C, Ma L, Wang X, Hu X, Chen S. MicroRNA-320a inhibits proliferation and invasion of breast cancer cells by targeting RAB11A. Am J Cancer Res. 2015;5:2719–2729. [PMC free article] [PubMed] [Google Scholar]
- 16.Shang C, Zhang H, Guo Y, Hong Y, Liu Y, Xue Y. MiR-320a down-regulation mediates bladder carcinoma invasion by targeting ITGB3. Mol Biol Rep. 2014;41:2521–2527. doi: 10.1007/s11033-014-3110-0. [DOI] [PubMed] [Google Scholar]
- 17.Ayala-Ortega E, Arzate-Mejía R, Pérez-Molina R, González-Buendía E, Meier K, Guerrero G, Recillas-Targa F. Epigenetic silencing of miR-181c by DNA methylation in glioblastoma cell lines. BMC Cancer. 2016;16:226. doi: 10.1186/s12885-016-2273-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li X, Wu Y, Liu A, Tang X. MiR-27b is epigenetically downregulated in tamoxifen resistant breast cancer cells due to promoter methylation and regulates tamoxifen sensitivity by targeting HMGB3. Biochem Biophys Res Commun. 2016;477:768–773. doi: 10.1016/j.bbrc.2016.06.133. [DOI] [PubMed] [Google Scholar]
- 19.He DX, Gu XT, Jiang L, Jin J, Ma X. A methylation-based regulatory network for microRNA 320a in chemoresistant breast cancer. Mol Pharmacol. 2014;86:536–547. doi: 10.1124/mol.114.092759. [DOI] [PubMed] [Google Scholar]
- 20.Wang Y, Zeng J, Pan J, Geng X, Li L, Wu J, Song P, Wang Y, Liu J, Wang L. MiR-320a inhibits gastric carcinoma by targeting activity in the FoxM1-P27KIP1 axis. Oncotarget. 2016;7:29275–29286. doi: 10.18632/oncotarget.8676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xie N, Wang C, Zhuang Z, Hou J, Liu X, Wu Y, Liu H, Huang H. Decreased miR-320a promotes invasion and metastasis of tumor budding cells in tongue squamous cell carcinoma. Oncotarget. 2016;7:65744–65757. doi: 10.18632/oncotarget.11612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhang G, Jiang G, Wang C, Zhong K, Zhang J, Xue Q, Li X, Jin H, Li B. Decreased expression of microRNA-320a promotes proliferation and invasion of non-small cell lung cancer cells by increasing VDAC1 expression. Oncotarget. 2016;7:49470–49480. doi: 10.18632/oncotarget.9943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Milech N, Kees UR, Watt PM. Novel alternative PBX3 isoforms in leukemia cells with distinct interaction specificities. Genes Chromosomes Cancer. 2001;32:275–280. doi: 10.1002/gcc.1190. [DOI] [PubMed] [Google Scholar]
- 24.Han HB, Gu J, Ji DB, Li ZW, Zhang Y, Zhao W, Wang LM, Zhang ZQ. PBX3 promotes migration and invasion of colorectal cancer cells via activation of MAPK/ERK signaling pathway. World J Gastroenterol. 2014;20:18260–18270. doi: 10.3748/wjg.v20.i48.18260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Ramberg H, Alshbib A, Berge V, Svindland A, Taskén KA. Regulation of PBX3 expression by androgen and Let-7d in prostate cancer. Mol Cancer. 2011;10:50. doi: 10.1186/1476-4598-10-50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Han H, Du Y, Zhao W, Li S, Chen D, Zhang J, Liu J, Suo Z, Bian X, Xing B, Zhang Z. PBX3 is targeted by multiple miRNAs and is essential for liver tumour-initiating cells. Nat Commun. 2015;6:8271. doi: 10.1038/ncomms9271. [DOI] [PubMed] [Google Scholar]
- 27.Li Y, Sun Z, Zhu Z, Zhang J, Sun X, Xu H. PBX3 is overexpressed in gastric cancer and regulates cell proliferation. Tumour Biol. 2014;35:4363–4368. doi: 10.1007/s13277-013-1573-6. [DOI] [PubMed] [Google Scholar]
- 28.Li Z, Huang H, Li Y, Jiang X, Chen P, Arnovitz S, Radmacher MD, Maharry K, Elkahloun A, Yang X, He C, He M, Zhang Z, Dohner K, Neilly MB, Price C, Lussier YA, Zhang Y, Larson RA, Le Beau MM, Caligiuri MA, Bullinger L, Valk PJ, Delwel R, Lowenberg B, Liu PP, Marcucci G, Bloomfield CD, Rowley JD, Chen J. Up-regulation of a HOXA-PBX3 homeobox-gene signature following down-regulation of miR-181 is associated with adverse prognosis in patients with cytogenetically abnormal AML. Blood. 2012;119:2314–2324. doi: 10.1182/blood-2011-10-386235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lujambio A, Calin GA, Villanueva A, Ropero S, Sánchez-Céspedes M, Blanco D, Montuenga LM, Rossi S, Nicoloso MS, Faller WJ, Gallagher WM, Eccles SA, Croce CM, Esteller M. A microRNA DNA methylation signature for human cancer metastasis. Proc Natl Acad Sci U S A. 2008;105:13556–13561. doi: 10.1073/pnas.0803055105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Li Z, Li D, Zhang G, Xiong J, Jie Z, Cheng H, Cao Y, Jiang M, Lin L, Le Z, Tan S, Zou W, Gong B, Lin S, Yang K. Methylation-associated silencing of MicroRNA-335 contributes tumor cell invasion and migration by interacting with RASA1 in gastric cancer. Am J Cancer Res. 2014;4:648–662. [PMC free article] [PubMed] [Google Scholar]
- 31.Grady WM, Parkin RK, Mitchell PS, Lee JH, Kim YH, Tsuchiya KD, Washington MK, Paraskeva C, Willson JK, Kaz AM, Kroh EM, Allen A, Fritz BR, Markowitz SD, Tewari M. Epigenetic silencing of the intronic microRNA hsa-miR-342 and its host gene EVL in colorectal cancer. Oncogene. 2008;27:3880–3888. doi: 10.1038/onc.2008.10. [DOI] [PubMed] [Google Scholar]