Abstract
Microbial parasites adapted to thrive at mammalian mucosal surfaces have evolved multiple times from phylogenetically distant lineages into various extracellular and intracellular life styles. Their symbiotic relationships can range from commensalism to parasitism and more recently some host-parasites interactions are thought to have evolved into mutualistic associations too. It is increasingly appreciated that this diversity of symbiotic outcomes is the product of a complex network of parasites-microbiota-host interactions. Refinement and broader use of DNA based detection techniques are providing increasing evidence of how common some mucosal microbial parasites are and their host range, with some species being able to swap hosts, including from farm and pet animals to humans. A selection of examples will illustrate the zoonotic potential for a number of microbial parasites and how some species can be either disruptive or beneficial nodes in the complex networks of host-microbe interactions disrupting or maintaining mucosal homeostasis. It will be argued that mucosal microbial parasitic diversity will represent an important resource to help us dissect through comparative studies the role of host-microbe interactions in both human health and disease.
Keywords: mucosa, microbiota, bacteria, viruses, innate and adaptive immune responses, parasitic protozoa, parasitic protists, extracellular and intracellular parasites, pathobionts
Introduction
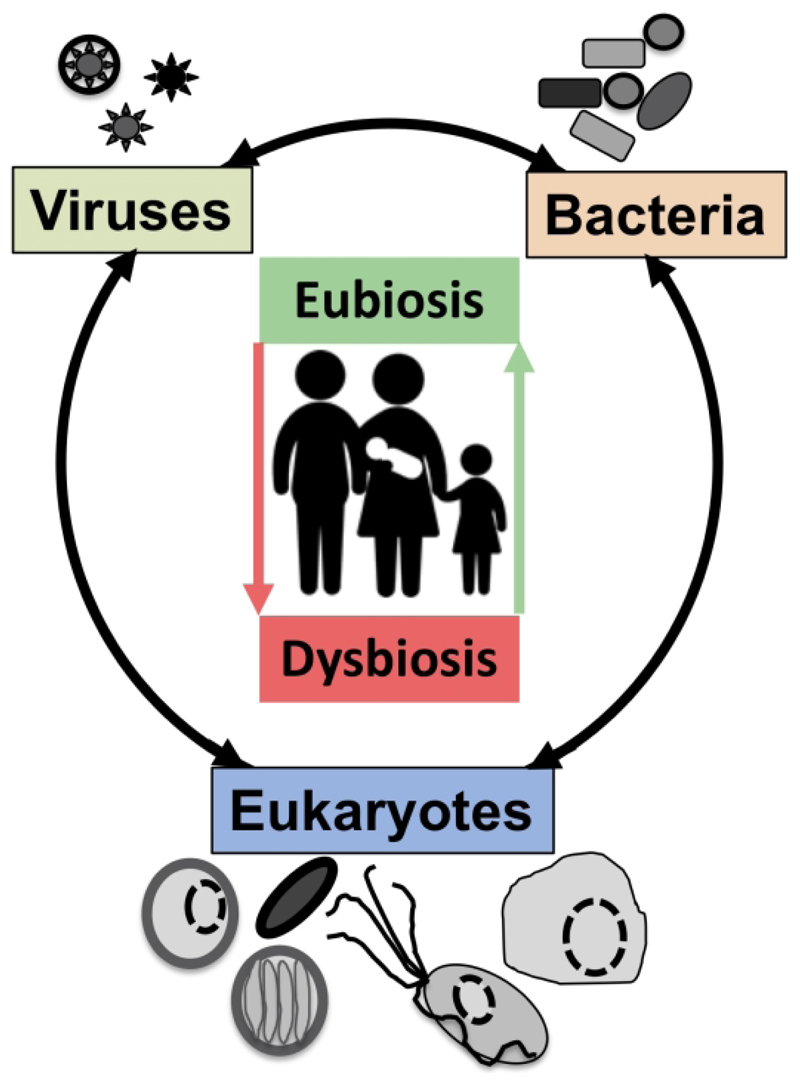
Intimately associated with the human mucosa functions and defences are the complex microbial communities, called the microbiota, which are increasingly understood to play key roles in myriads of aspects in human health and disease (Belkaid & Hand, 2014; Clemente et al., 2012). Millions of years of refinements have ensured that mammalian mucosa in a state of homeostasis are effective at mediating simultaneously two conflicting and essential functions: (i) facilitate exchanges between the outside and the inside of the body to allow optimal breathing, nutrient and water uptake and reproduction and (ii) mediate protection against physical, chemical and biological insults, with the latter being mainly microbial in nature. Many of the molecules of the mucosal innate defence system are key for mediating interactions with microbes, including receptors sensing microbes and the central components of mucus (mucins and antimicrobial peptides) can be traced back to early phases of metazoans evolution (Bakshani et al., 2018; Schroder & Bosch, 2016). In contrast, some of the effector molecules and cells characteristic of the human adaptive mucosal immune system represent more recent additions to the mucosal armoury against microbes with secretory IgAs (SIgAs), the archetypal antibody in human mucosal secretions, being only shared with reptiles and birds (Smith et al., 2013). The mucosal microbiota form extraordinarily complex microbial ecosystems where bacteria, archaea, microbial eukaryotes and viruses form intricate network of microbe-microbe and host-microbe interactions that can be broadly defined as eubiotic, associated with health, or dysbiotic, associated with disease (Levy et al., 2017; Petersen & Round, 2014)(Fig. 1). Eubiotic relationships at mucosal surfaces are dependent on the functional characteristics of the microbiota community and corresponding finely tuned mucosal innate and adaptive immune responses to microbes, that together are required for harmonious, highly dynamic and continuous host-microbes interactions at mucosal surfaces (Belkaid & Hand, 2014; Clemente et al., 2012; Levy et al., 2017). Finely choreographed host-microbiota interactions are essential to maintain mucosal homeostasis in the broadest possible range of conditions experienced by humans, including variations in diet, exposures to various environmental microbes including pathogens and an increasing range of xenobiotics (Ferreiro et al., 2018; Levy et al., 2017).
Fig. 1.
The now generally accepted new paradigm of microbe-microbe / host-microbe complex network of interactions that can contribute to health (maintaining homeostasis) or disease (inducing excessive inflammation through time and space) status of the animal/human host. The terms eubiosis and dysbiosis relate to the microbiota functional activities associated with respectively health through maintaining mucosal homeostasis (ensuring optimal mucosal functionality) or pathologies due to excess inflammation leading to damage mucosal surfaces and that can also contribute to disrupt systemic physiology and sub-optimal cognitive capacities. See main text and cited references for the conceptual limitations on the use of eubiosis and dysbiosis.
Mucosal microbial parasites (also referred to as parasitic protozoa or parasitic protists) are phylogenetically highly diverse and heterogenous that can be broadly distributed across human populations and can contribute to important pathologies but that are also often associated with asymptomatic interactions (Chabe et al., 2017; Lukes et al., 2015). Thus human-microbial parasite symbiotic relationships can range from commensalism to parasitism and more recently some host-parasites interactions are suggested to have evolved into mutualistic associations too (Chabe et al., 2017; Loke & Lim, 2016; Lukes et al., 2015; Stensvold, 2019). Hence these mucosa residents will be referred to here as “microbial eukaryote symbionts” to better capture the diversity of symbiotic interactions mediated by organisms historically typically referred to as parasites (Lukes et al., 2015; Stensvold, 2019). Notably it is increasingly appreciated that this diversity of symbiotic outcomes is the product of parasites-microbiota-host complex network of interactions (Burgess et al., 2017; Clemente et al., 2012; Rowan-Nash et al., 2019), further highlighting the importance of contextuality for the phenotypic outcome of human-microbe interactions (Belkaid & Hand, 2014; Clemente et al., 2012; Levy et al., 2017). In this editorial a selection of examples will illustrate how mucosal microbial parasites/symbionts (MMPS) can represent disruptive nodes of the host-microbes complex network of interactions underlying mucosal homeostasis and thus contribute to directly or indirectly to mucosal dysbiosis. In contrast, other examples will illustrate the potential of microbial eukaryote symbionts to contribute to eubiosis (Stensvold & van der Giezen, 2018). With these seemingly contradictory considerations in mind, it will be argued that MMPS diversity will represent an important resource to help researchers to dissect the potential causal link between eubiosis and health and dysbiosis and disease through comparative studies. This is a research topic not without controversies and important difficulties and that will require a dramatic increase in the physiological functional characterisation of members of the mucosal microbial communities (Hooks & O'Malley, 2017) including microbial eukaryotes (Chabe et al., 2017; Stensvold & van der Giezen, 2018).
Several papers associated with this Special Issue are derived from talks that were delivered at the EMBO Conference “Anaerobic protists: Integrating parasitology with mucosal microbiota and immunology” (http://meetings.embo.org/event/17-anaerobic-protists)(Dessi et al., 2019; Labruyere et al., 2017; Leitsch, 2017; Miranda-Ozuna et al., 2019; Stensvold, 2019). These are complemented by articles providing broader perspectives on the study of the MMPS (Bartley et al., 2018; Chihi et al., 2019; Deere et al., 2018; Rush et al., 2019; van Gestel et al., 2018; Vargas Rigo et al., 2018). For more in depth coverage of MMPS biology including broader taxonomic coverage (e.g. Fungi and Helminths), mucosal sites and biology (e.g. lungs, mucus) and topics including parasite genomics, parasite diagnostics and mucosal vaccine, the reader is directed to the following reviews or original papers (Baker et al., 2017; Collins & Belkaid, 2018; Corfield, 2018; Hupalo et al., 2015; Lemieux et al., 2017; Rowan-Nash et al., 2019; Ryan et al., 2017; Serradell et al., 2016).
Mucosal microbial eukaryote diversity, host range and zoonoses
What range of microbial eukaryote symbionts can thrive at our different mucosa, how broadly are they distributed across human populations and what are their host range beyond humans, how genetic diverse are they, what species/genetic lineage are associated with disease and how do these influence the mucosal microbiota and vice versa? These are some of the most basic questions for which we still have relatively limited knowledge for most species. This important knowledge gap currently limits us to properly assess the role in health and disease of microbial eukaryote symbionts and reflects the difficulty of studying mucosal associated organisms and viruses more generally through reductive approaches. A few examples will illustrate the importance of new perspectives one can gain from working on answering these basic questions in humans and animal models. New diagnostic technologies (Ryan et al., 2017) and the increasing number of microbial eukaryote symbionts genome sequence data (Hupalo et al., 2015) are all contributing at providing a better picture of the natural history of MMPS, including non-pathogenic species (Chihi et al., 2019). These in combination with metagenomics surveys (Lokmer et al., 2019) will generate more comprehensive knowledge on MMPS diversity and host range and their link with health and disease.
The relatively common gut MMPS Blastocystis spp., Dientemoeba fragilis are reviewed by Stensvold (2019)(both species) and van Gestel et al., (2019)(D. fragilis). These species are thought to be common in some populations but there are a number of contradictory datasets in relation to their potential role in both disease and health and issues with the apparent important prevalence variations between populations (van Gestel et al., 2018). Although potentially misleading detection tools can explain some variation between studies (Gough et al., 2019; van Gestel et al., 2018), a combination of environmental and biological explanations are also likely to play a role. An intriguing possibility suggested for D. fragilis higher prevalence in some countries is pig farming, which could potentially play a role in its higher prevalence in Denmark and the Netherlands where both humans and pigs cohabit in relatively higher densities (van Gestel et al., 2018). This highlights the importance of considering both human and animal prevalence and study in detail the genetic diversity and phylogeny of the microbial eukaryote symbionts to establish their origins among humans and their potential association with animal reservoirs. This is also relevant for the relatively better known species such as Giardia, including in developed countries such as the UK (Horton et al., 2019). A recent survey for Giardia duodenalis among cattle in Scotland further illustrates the importance of studying animal populations, where this species was shown to be common across surveyed beef and dairy cattle (~32%) and included genetic lineages associated with human symptomatic infections (Bartley et al., 2018). In another example, vaccination to protect dogs and cats from Giardia duodenalis infections (100% prevalence) in a peri-urban disadvantaged community in Argentina, using an elegant vaccination strategy (Rivero et al., 2010), was shown to reduce dog and cat infections with the concomitant reduction of children infections in the community associated with the vaccinated pets (Serradell et al., 2016). This example illustrates the importance of both the knowledge of the epidemiology of a potential pathogen and the molecular mechanisms underlying surface antigen variation to develop an effective vaccine for relevant hosts to eventually also control infections among humans. Similarly, the prevalence of Entamoeba spp., including Entamoeba histolytica, among humans, chimpanzees and baboon in the Greater Gombe Ecosystem in Tanzania, where the human and nonhuman primate populations overlap, demonstrated a high level of prevalence (~60% for all Entamoeba spp. and ~10% of E. histolytica) among all three species highlighting the potential for zoonotic transmission of Entamoeba species (Deere et al., 2018). Notably the presence of E. histolytica in chimpanzees was apparently never associated with symptoms in the tested population, in contrast to human infections (Deere et al., 2018).
Beyond the gut, an interesting set of data for Trichomonas vaginalis and Trichomonas tenax, infecting respectively the urogenital tract (Hirt & Sherrard, 2015) and oral cavities (Marty et al., 2017) also highlight the importance of specific and sensitive diagnostics and the knowledge of their distributions beyond humans (Maritz et al., 2014). Through carefully testing the specificity of a molecular diagnostic tool used for T. vaginalis it was discovered that some infections of the urogenital tract (three male urine samples) could be due to T. tenax rather than T. vaginalis (Brosh-Nissimov et al., 2019). A screening across dogs and cats for oral trichomonads also indicated a potential zoonotic source for T. tenax from pets (Kellerova & Tachezy, 2017). Genotyping T. tenax clinical isolates from humans also established that a subset of genetic lineages are significantly associated with periodontal patients, in addition of being common among the tested population in an affluent setting (35% among patients with periodontitis and 19% among healthy controls in the studied French cohort)(Benabdelkader et al., 2019). Notably both T. vaginalis and T. tenax are likely derived from species infecting birds (Maritz et al., 2014) as these two species are respectively more closely related phylogenetically to distinct set of species infecting birds including Trichomonas gallinae, common among pigeons, and Trichomonas gypaetinii isolated from vultures among other Trichomonas spp. isolated from various bird species (Martinez-Diaz et al., 2015). Transfer of T. gallinae from columbiform to passerines has led to important mortality rates for some passerine species dramatically illustrating the potential for a Trichomonas species to jump host and spread rapidly through populations and to become a virulent parasite in some contexts (wild finches such as the common chaffinch) whereas it is often a commensal in others (the columbiform rock pigeon)(Amin et al., 2014). The comparative study of the molecular basis of the interactions between these various Trichomonas species and mucosal landmarks required to initiate and sustain the colonisation of various hosts and mucosa will be of great interest and represent a fascinating model system to study MMPS transfers between birds and from birds to mammals, including humans (Maritz et al., 2014).
Symbiosis: from parasitism to commensalism to mutualism
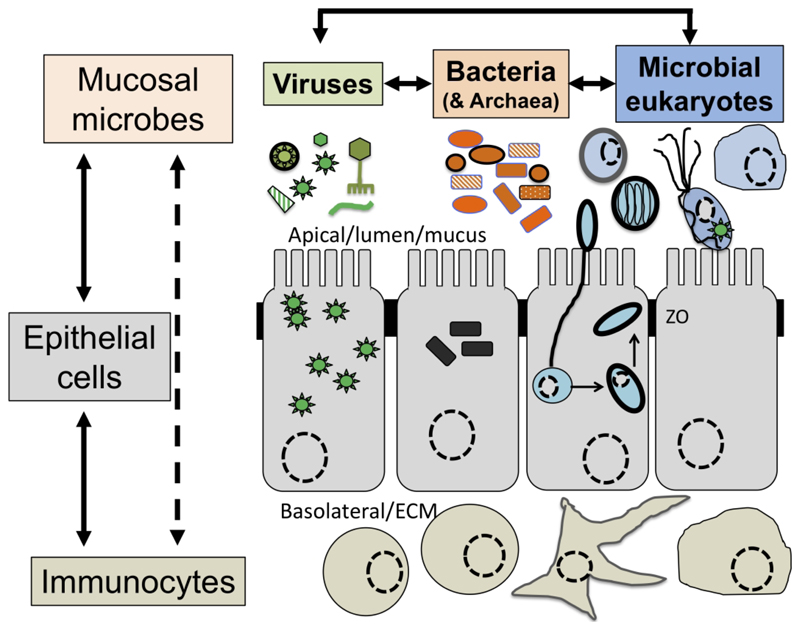
Although a number of MMPS are known to be associated with pathologies, leading to important morbidities and mortality rates in some contexts (Bar et al., 2015; Burgess et al., 2017), many infections by the same species are asymptomatic (Chabe et al., 2017; Lukes et al., 2015; Stensvold, 2019). The outcome of host-microbial eukaryote symbionts interactions are dependent on the combination of the characteristics of the host, the microbial eukaryote and the mucosa microbiota, with increasing evidence for an important role played by cross kingdoms interactions (Fig. 1)(Burgess et al., 2017; Rowan-Nash et al., 2019). Inter-kingdom interactions can modulate the inflammatory tone of the mucosa through multiple possible direct and indirect interactions between mucosal microbes, microbes and epithelial cells and microbes and immunocytes (Fig. 2). Notably the epithelial cells play key roles in both sensing microbes and orchestrating the mucosal immunological innate and adaptive responses mediated by the combination of epithelial cells and immunocytes (Fig. 2)(Levy et al., 2017; Petersen & Round, 2014). Primary immunodeficiencies, due to specific genetic background interfering with epithelial cells and/or immunocytes-microbes interactions, or secondary immunodeficiencies due to infections (e.g. HIV/AIDS) or malnutrition, can dramatically increase the susceptibility of the host to numerous infections including by those of MMPS. This is particularly marked for intracellular parasites such as Cryptosporidium and Microsporidia, with the HIV/AIDS pandemic highlighting both the importance of the adaptive immune response in controlling these parasites and the high level of human exposure to these opportunistic intracellular pathogens from divers zoonotic reservoirs (Khan et al., 2018; Stentiford et al., 2016).
Fig. 2.
The complex network of interactions at mucosal surfaces between microbes, epithelial cells and immunocytes modulating the immunological and inflammatory status of the mucosal surfaces. Optimal interactions ensure adequate responses to the presence of members of the microbiota and robust challenges to pathogens and at the same time tolerance to innocuous antigens required to maintain long term functionality of the mucosal surface underlying optimal digestion and nutrient uptake, breathing, or reproduction. Arrows indicate direct (e.g. physical contact) and indirect (e.g. metabolites or signalling molecules) interactions such as infection of epithelial cells by intracellular pathogens (viruses or Microsporidia, both illustrated) and doted arrows indicate indirect (e.g. though metabolites) interactions between illustrated cells. Note in particular the central node/role of epithelial cells that integrate, and in effect coordinate/orchestrate the complex network of interactions between microbes and immunocytes. A virus (several green “stars”) infected epithelial cell is illustrated as is a virus infected trichomonad (one green “star”, see example in the text). In addition, some viruses/phages infect bacteria also contributing to the overall functional properties of the mucosa microbial ecology. Intracellular bacteria (black rectangles) and Microsporidia (blue cell and spores) are also illustrated within epithelial cells. ZO, Zonula occludens - tight junction; ECM, extra cellular matrix. For simplicity the presence of mucus and the glycocalyx interacting with luminal microbes are not shown and only a monolayer of epithelial cells (e.g. as in the intestine) is illustrated.
In other contexts, MMPS could provide benefit to their mammalian carrier. Mice carrying the recently described gut trichomonad Tritrichomonas musculis were shown to be more resistant to challenges by the bacterial pathogen Salmonella typhimurium through enhancing mucosal defences by increasing intestinal inflammation via inflammosome activation and increase of the proinflammatory IL-18 production leading to a TH1/TH17 immune response (Chudnovskiy et al., 2016). This higher protection level to Salmonella was however associated with a cost as T. musculis colonisation was also associated with higher rate of colorectal cancer (Chudnovskiy et al., 2016). This contrasts with helminths infections that tend to inhibit gut inflammation through stimulating TH2/Treg responses (Cortes et al., 2018).
These contrasting examples illustrate the importance, and potential great value, of increasing our knowledge of the natural history of mammal-MMPS interactions and the importance of studying various microbial eukaryote symbiont species in humans and animal models to dissect the complex host-MMPS-mirobiota interactions to illuminate their influence in both health and disease (Loke & Lim, 2016). Additional examples of potentially beneficial microbial eukaryote symbionts; including Entamoeba spp. and Blastocystis are discussed in this Special Issue (Stensvold, 2019) and in other contexts (Chabe et al., 2017; Lukes et al., 2015; Stensvold & van der Giezen, 2018) and in the next section.
Microbial eukaryote symbionts/parasite-bacteria-virus interactions
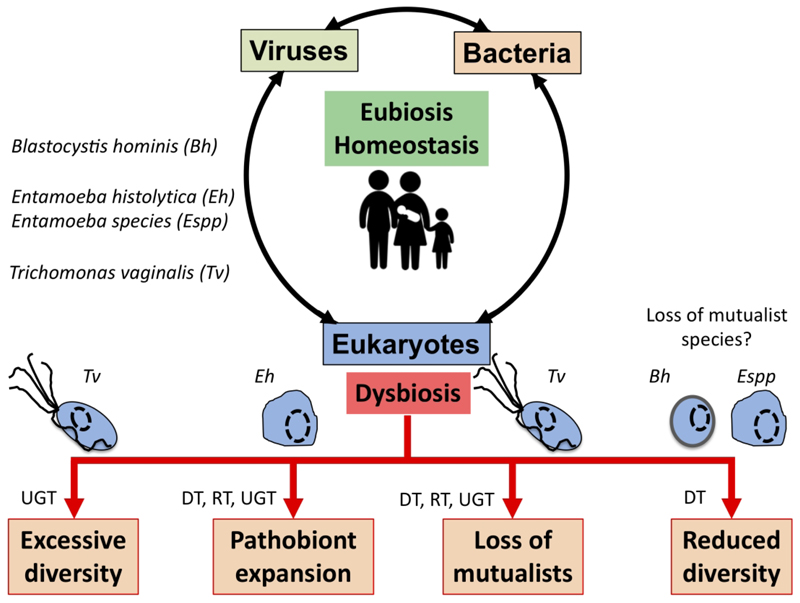
The complex interplay between MMPS, bacteria, archaea and viruses and mammalian host health and disease status is increasingly being uncovered through the study of various microbial cellular species, bacteriophages and eukaryote infecting viruses, different mucosal surfaces and mammalian species, including humans (Burgess et al., 2017; Chabe et al., 2017; Clemente et al., 2012; Rowan-Nash et al., 2019). Here a few examples illustrating the link between these interactions and health and disease are covered with MMPS potentially contributing to either eubiosis or dysbiosis depending on context of the hosts and their associated microbiota and environmental factors such as diet and xenobiotics (e.g. antibiotics) (Fig. 3).
Fig. 3.
Potential role of MMPS in inducing dysbiosis or eubiosis at mucosal surfaces. In the context of dysbiosis this would contribute to the loss of mucosal homeostasis, and by doing so to a number of potential pathologies that eventually will translate in dysfunctional mucosa leading to disease both locally, e.g. mucosa inflammation, or more distal impacts. The illustrated examples include: Trichomonas vaginalis (Tv) contributing to increasing the vaginal bacterial diversity associated with bacterial vaginosis, a form of pro-inflammatory dysbiosis of the urogenital tract (UGT). T. vaginalis infections are also associated with the loss of mutualists in the UGT. Entamoeba histolytica (Eh) can contribute to colitis, mucosa perforation and translocation of both parasites and some member of the gut microbiota into the portal vein and systemic tissues that can contribute to highly damaging systemic and local inflammations in the digestive tract (DT) and beyond. In contrast, the loss of some microbial eukaryotes, including potentially Blastocystis hominis (Bh) and some Entamoeba spp. (especially, non-histolytica species), could contribute to the gut microbiota reduced bacterial diversity associated with a dysbiotic state. Similarly, metronidazole treatments aiming at eradicating anaerobic mucosal microbial parasites such as Giardia and Trichomonas species, will also contribute at disrupting the mucosal microbiota by killing important bacterial anaerobes and can favour the expansion of bacterial pathobionts in the DT and the respiratory tract (RT). See main text for examples and citations. MMPS can also influence the RT - e.g. (Maritz et al., 2014) - but this is not covered here.
Arguably one of the most fascinating and complex example includes Trichomonas vaginalis that infect the urogenital tracts (UGT) of humans (Hirt & Sherrard, 2015). A complex set of interactions between T. vaginalis, RNA viruses infecting T. vaginalis (TVV), the bacteria Mycoplasma hominis forming symbiosis with T. vaginalis and other bacteria associated with bacterial vaginosis, are all thought to contribute in concert to symptomatic infections, adverse pregnancy outcomes and increase transmission and acquisition of human infecting viruses, including HIV, HPV and HSV-2 (Hirt & Sherrard, 2015; Kissinger, 2015). This is thought to be mediated through several mechanisms including, boosting the inflammatory tone of the UGT, increasing the population of target immunocytes for HIV and induction of microlesions disrupting the mucosal barrier (Kissinger, 2015). Furthermore, although human viruses including HIV and HSV are not known to infect T. vaginalis, HIV and HSV viral particles can be internalised by the parasite and potentially be transferred to, and infect, human cells in a new host (Pindak et al., 1989; Rendon-Maldonado et al., 2003). Although T. vaginalis can induce tissue damage and inflammation on its own, TVV and Mycoplasma hominis can act synergistically to dramatically boost inflammations associated with T. vaginalis infections as reviewed by Dessi and colleagues (Dessi et al., 2019). Dysbiosis associated with infections by T. vaginalis is also thought to contribute to the pathobiology of T. vaginalis (Fichorova et al., 2017; Mercer & Johnson, 2018). Direct targeting of bacteria peptidoglycans by the parasite through enzymes of bacterial origins (Pinheiro et al., 2018) could potentially contribute to modulate the microbiota bacterial taxonomic composition in addition to contributing to T. vaginalis capacity to colonise the mucosal surface. The combination of the parasite and several bacterial species characteristic of dysbiotic vaginal microbiota associated with trichomoniasis, were also recently shown to synergistically affect the integrity of the tight junction complex of the cervicovaginal epithelial cells (Hinderfeld et al., 2019). Notably, treating T. vaginalis infections with metronidazole can liberate from the killed parasite TVV particles and/or Mycoplasma hominis cells leading to the boosting of inflammation and to infection of human cells by M. hominis (Dessi et al., 2019; Thi Trung Thu et al., 2018). These different aspects associated with T. vaginalis infections illustrates the intricate associations of the parasite with bacterial (Mycoplasma) and viral (TVV) endosymbionts, the bacterial members of the UGT microbiota and how these interactions can influence the parasite pathobiology including increasing human infecting virus transmission rates. These considerations will be important to complement more traditional investigations focusing on the study of specific aspects of host-parasite interactions, such as the potential role of environmental glucose concentration variation (Miranda-Ozuna et al., 2019) and cell surface and secreted factors such as exosomes (Mercer & Johnson, 2018), in modulating the virulence of the parasite. These examples illustrate dramatically the importance to investigate host-MMPS-microbiota-virus interactions in an integrative manner to develop more refined diagnostics and novel prophylactic and therapeutic strategies to eventually promote reproductive and sexual health more efficiently. It will also be of interest to investigate the possibility that related endosymbionts (to TVV and Mycoplasma) are also present in other Trichomonas species including bird infecting species and T. tenax associated with periodontitis (described in the previous section).
The MMPS Giardia, Entamoeba and Cryptosporidium are also known to be infected by RNA viruses (Gomez-Arreaza et al., 2017). Cryptosporidium infected virus are associated with higher rate of the parasite propagation capacity; however, it is not clear if this increases the virulence of such infections. Similarly there is currently no evidence for Giardia and Entamoeba that their RNA viruses can contribute to boosting the pathobiology of these parasites (Gomez-Arreaza et al., 2017). Complex interplay between Giardia, Entamoeba and Cryptosporidium with bacteria members of the microbiota have also been shown to influence the virulence of these parasites in both negative (e.g. inhibiting infections) or positive ways (e.g. promoting virulence)(Burgess et al., 2017; Rowan-Nash et al., 2019). A remarkable example illustrating the importance of the microbiota in playing a role in reducing the impact of Cryptosporidium infection was uncovered when investigating two candidate drugs to treat the parasite. Two novel drugs that had promising properties in initial in vitro tests had opposite effect on Cryptosporidium infections in a mouse model (Gorla et al., 2014). Although one of the drugs was potent in controlling the parasite, the other drug was shown to actually boost infection levels, which was associated with a significant change in the bacterial taxonomic composition of the gut microbiota, with in particular a dramatic increase of the population of the mucin loving gut bacteria Akkermansia muciniphila (2,800-fold increase compared to the pre-treatment state), suggesting a dysbiotic microbiota (Gorla et al., 2014). This was rationalised as an off-target impact of the drug on members of the gut microbiota. Although A. muciniphila is considered to be an important mutualist associated with human health (Cani & de Vos, 2017), the significant boost in Cryptosporidium infection level could be explained by an excessive degradation by A. muciniphila of the mucus protective layer in the gut facilitating access to, and eventual infection of, epithelial cells by Cryptosporidium. An apparently similar outcome was observed in a mouse model with a humanised gut microbiota fed with a diet depleted from plant fibbers, which led to the depletion of the mucus protective layers by the microbiota and higher susceptibility to pathogens (Desai et al., 2016). These examples illustrate how environmental factors, including xenobiotics (an antibiotic in the example above) and diet, can influence the mucosal microbial ecology and by doing so modulate the host susceptible to infections by potential pathogens, including MMPS.
Antibiotics and vaccines for mucosal parasites/symbionts
In contrast to the availability of a broad range of antibiotic treatment regiments for bacteria, there are far less efficient options to treat with drugs symptomatic infections due to microbial parasites (Farthing, 2006; Leitsch, 2017). As for bacteria, there is also the issue of microbial parasites developing resistance to existing drugs regiments and for off-target effects on the microbiota (Wypych & Marsland, 2018). Furthermore some patients can develop strong reactions to some drugs including to the commonly used metronidazole targeting anaerobic parasites (Leitsch, 2017). These considerations stimulate continuous research efforts to identify new drugs to treat microbial parasites, either based on modifying existing well established drugs such as 5-nitroimidazole (Leitsch, 2017), or new drugs such as plant derived phenanthrenes (Vargas Rigo et al., 2018). Irrespective of the drug, it is increasingly appreciated necessary to consider their broad impact on the host microbiota, with increasing evidence that antibiotic treatments are being associated with dysbiosis favouring opportunistic pathogens, including pathobionts, and/or leading to a difunctional immune response to microbial and other antigens that can lead to debilitating conditions such as allergies and asthma (Wypych & Marsland, 2018). In the case of the treatment of anaerobic mucosal parasites (such as Trichomonas and Giardia) with metronidazole/imidazole the anaerobic members of the microbiota will also be affected (Leitsch, 2017). This can contribute to dysbiosis in the gut microbiota in particular where anaerobes are known to play important roles (Wypych & Marsland, 2018)(Fig. 3).
In comparison to drug treatments options, vaccines for MMPS are even less well developed. This is due to the combination of the inherent difficulties in developing effective mucosal vaccines (Lycke, 2012) and the complex biology of MMPS, including their capacity to mediate cell surface antigen variation (Deitsch et al., 2009; Gargantini et al., 2016) and the little knowledge we have on the nature of the host immune response to eradicate MMPS (Chapwanya et al., 2016; Farthing, 2006). One promising strategy that takes advantage of the properties of VSP proteins from Giardia (Gargantini et al., 2016) and viral-like particles has great potential to develop novel oral vaccines for various pathogens (Serradell et al., 2019), including a broad range of MMPS in addition to Giardia (Serradell et al., 2016).
Conclusion and some speculations
From the examples covered here and in cited publications one can conclude that it might be more appropriate to refer to many extracellular microbial eukaryotic symbionts with various pathogenic potential as pathobionts that is, they are members of the mucosal microbial ecosystems that can become pathogenic in some contexts where host genetic, environment and properties of the microbial community as a whole all play a role (Chow et al., 2011). Acquired immunodeficiencies or transfer of MMPS between different hosts species can lead to sub-optimal interactions with some species becoming pathogenic (Farthing, 2006; Price et al., 2017). In contrast intracellular parasites, including the Apicomplexa Cryptosporidium and the Microsporidia, are typically thought to be primarily gut pathogens (Farthing, 2006), as they must directly exploit their host cell energy and metabolites to proceed through their life cycle and in the process compromise the integrity of the epithelial monolayer of the gut (Dean et al., 2016; Farthing, 2006). One aim of this editorial was to illustrate specific aspects of the intricate and complex interactions taking place between MMPS, the other members of the microbiota and their animal or human hosts. These highlight the importance of collaborative research projects integrating parasitology, microbiology, virology, pharmacology and mucosal immunology in the context of both basic and medical and veterinary research on the factors influencing mucosa health and disease. Generating more comprehensive knowledge on the link between these microbial interactions and mucosal and systemic health and disease is undoubtedly one of “the most difficult and challenging scientific endeavour of our time”(Birchenough & Hansson, 2017), as it will need to identify and characterise key aspects of thousands of highly dynamic interactions mediated by a complex cocktail of metabolites, cell-virus and cell-cell interactions involving complex microbial communities, epithelial cells and immunocytes. The knowledge derived from the study of these complex network of interactions will be required to eventually develop much needed novel prophylactic, including mucosal vaccines for overt pathogens, and therapeutic strategies (including highly specific drugs, prebiotics, faecal transplants), to regenerate, maintain and promote human and animal health at mucosal surfaces. It is also suggested that considering microbial eukaryote symbionts/parasites will provide important opportunities for much required comparative studies to delicately dissect key nodes orchestrating mucosal-microbes interactions and how these are causally linked to the specific phenotypic outcomes in their human and animal hosts. Contextualisation of the diversity of both MMPS, the microbiota at large (bacteria, archaea and viruses) and their host within an evolutionary and ecological framework will also likely be important at helping building a more predictive theoretical framework for the outcome of host-microbes interactions (Amato, 2016; Davenport et al., 2017; Ferreiro et al., 2018; Rook et al., 2017).
Acknowledgments
EMBO and Newcastle University were the main sponsors of the EMBO conference: “Anaerobic protists: Integrating parasitology with mucosal microbiota and immunology” and Cambridge University Press, for providing the opportunity to put together this Special Issue.
References
- Amato KR. Incorporating the gut microbiota into models of human and non-human primate ecology and evolution. Am J Phys Anthropol. 2016;159:S196–215. doi: 10.1002/ajpa.22908. [DOI] [PubMed] [Google Scholar]
- Amin A, Bilic I, Liebhart D, Hess M. Trichomonads in birds--a review. Parasitology. 2014;141:733–747. doi: 10.1017/S0031182013002096. [DOI] [PubMed] [Google Scholar]
- Baker JL, Bor B, Agnello M, Shi W, He X. Ecology of the Oral Microbiome: Beyond Bacteria. Trends Microbiol. 2017;25:362–374. doi: 10.1016/j.tim.2016.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bakshani CR, Morales-Garcia AL, Althaus M, Wilcox MD, Pearson JP, Bythell JC, Burgess JG. Evolutionary conservation of the antimicrobial function of mucus: a first defence against infection. NPJ Biofilms Microbiomes. 2018;4:14. doi: 10.1038/s41522-018-0057-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bar AK, Phukan N, Pinheiro J, Simoes-Barbosa A. The Interplay of Host Microbiota and Parasitic Protozoans at Mucosal Interfaces: Implications for the Outcomes of Infections and Diseases. PLoS Negl Trop Dis. 2015;9:e0004176. doi: 10.1371/journal.pntd.0004176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartley PM, Roehe BK, Thomson S, Shaw HJ, Peto F, Innes EA, Katzer F. Detection of potentially human infectious assemblages of Giardia duodenalis in fecal samples from beef and dairy cattle in Scotland. Parasitology. 2018:1–8. doi: 10.1017/S0031182018001117. [DOI] [PubMed] [Google Scholar]
- Belkaid Y, Hand TW. Role of the microbiota in immunity and inflammation. Cell. 2014;157:121–141. doi: 10.1016/j.cell.2014.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benabdelkader S, Andreani J, Gillet A, Terrer E, Pignoly M, Chaudet H, Aboudharam G, La Scola B. Specific clones of Trichomonas tenax are associated with periodontitis. PLoS One. 2019;14:e0213338. doi: 10.1371/journal.pone.0213338. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Birchenough G, Hansson GC. Bacteria tell us how to protect our intestine. Cell Host Microbe. 2017;22:3–4. doi: 10.1016/j.chom.2017.06.011. [DOI] [PubMed] [Google Scholar]
- Brosh-Nissimov T, Hindiyeh M, Azar R, Smollan G, Belausov N, Mandelboim M, Rahav G, Keller N, Gefen-Halevi S. A false-positive Trichomonas vaginalis result due to Trichomonas tenax presence in clinical specimens may reveal a possible T. tenax urogenital infection. Clin Microbiol Infect. 2019;25:123–124. doi: 10.1016/j.cmi.2018.09.011. [DOI] [PubMed] [Google Scholar]
- Burgess SL, Gilchrist CA, Lynn TC, Petri WA Jr. Parasitic Protozoa and Interactions with the Host Intestinal Microbiota. Infect Immun. 2017;85 doi: 10.1128/IAI.00101-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cani PD, de Vos WM. Next-Generation Beneficial Microbes: The Case of Akkermansia muciniphila. Front Microbiol. 2017;8:1765. doi: 10.3389/fmicb.2017.01765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chabe M, Lokmer A, Segurel L. Gut Protozoa: Friends or Foes of the Human Gut Microbiota? Trends Parasitol. 2017;33:925–934. doi: 10.1016/j.pt.2017.08.005. [DOI] [PubMed] [Google Scholar]
- Chapwanya A, Usman AY, Irons PC. Comparative aspects of immunity and vaccination in human and bovine trichomoniasis: a review. Trop Anim Health Prod. 2016;48:1–7. doi: 10.1007/s11250-015-0909-1. [DOI] [PubMed] [Google Scholar]
- Chihi A, Stensvold CR, Ben-Abda I, Ben-Romdhane R, Aoun K, Siala E, Bouratbine A. Development and evaluation of molecular tools for detecting and differentiating intestinal amoebae in healthy individuals. Parasitology. 2019:1–7. doi: 10.1017/S0031182018002196. [DOI] [PubMed] [Google Scholar]
- Chow J, Tang H, Mazmanian SK. Pathobionts of the gastrointestinal microbiota and inflammatory disease. Curr Opin Immunol. 2011;23:473–480. doi: 10.1016/j.coi.2011.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chudnovskiy A, Mortha A, Kana V, Kennard A, Ramirez JD, Rahman A, Remark R, Mogno I, Ng R, Gnjatic S, Amir ED, et al. Host-Protozoan Interactions Protect from Mucosal Infections through Activation of the Inflammasome. Cell. 2016;167:444–456 e414. doi: 10.1016/j.cell.2016.08.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clemente JC, Ursell LK, Parfrey LW, Knight R. The impact of the gut microbiota on human health: an integrative view. Cell. 2012;148:1258–1270. doi: 10.1016/j.cell.2012.01.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins N, Belkaid Y. Do the Microbiota Influence Vaccines and Protective Immunity to Pathogens? Engaging Our Endogenous Adjuvants. Cold Spring Harb Perspect Biol. 2018;10 doi: 10.1101/cshperspect.a028860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corfield AP. The Interaction of the Gut Microbiota with the Mucus Barrier in Health and Disease in Human. Microorganisms. 2018;6 doi: 10.3390/microorganisms6030078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cortes A, Toledo R, Cantacessi C. Classic Models for New Perspectives: Delving into Helminth-Microbiota-Immune System Interactions. Trends Parasitol. 2018;34:640–654. doi: 10.1016/j.pt.2018.05.009. [DOI] [PubMed] [Google Scholar]
- Davenport ER, Sanders JG, Song SJ, Amato KR, Clark AG, Knight R. The human microbiome in evolution. BMC Biol. 2017;15:127. doi: 10.1186/s12915-017-0454-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dean P, Hirt RP, Embley TM. Microsporidia: Why Make Nucleotides if You Can Steal Them? PLoS Pathog. 2016;12:e1005870. doi: 10.1371/journal.ppat.1005870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deere JR, Parsons MB, Lonsdorf EV, Lipende I, Kamenya S, Collins DA, Travis DA, Gillespie TR. Entamoeba histolytica infection in humans, chimpanzees and baboons in the Greater Gombe Ecosystem, Tanzania. Parasitology. 2018:1–7. doi: 10.1017/S0031182018001397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deitsch KW, Lukehart SA, Stringer JR. Common strategies for antigenic variation by bacterial, fungal and protozoan pathogens. Nat Rev Microbiol. 2009;7:493–503. doi: 10.1038/nrmicro2145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desai MS, Seekatz AM, Koropatkin NM, Kamada N, Hickey CA, Wolter M, Pudlo NA, Kitamoto S, Terrapon N, Muller A, Young VB, et al. A Dietary Fiber-Deprived Gut Microbiota Degrades the Colonic Mucus Barrier and Enhances Pathogen Susceptibility. Cell. 2016;167:1339–1353 e1321. doi: 10.1016/j.cell.2016.10.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dessi D, Margarita V, Cocco AR, Marongiu A, Fiori PL, Rappelli P. Trichomonas vaginalis and Mycoplasma hominis: new tales of two old friends. Parasitology. 2019:1–6. doi: 10.1017/S0031182018002135. [DOI] [PubMed] [Google Scholar]
- Farthing MJ. Treatment options for the eradication of intestinal protozoa. Nat Clin Pract Gastroenterol Hepatol. 2006;3:436–445. doi: 10.1038/ncpgasthep0557. [DOI] [PubMed] [Google Scholar]
- Ferreiro A, Crook N, Gasparrini AJ, Dantas G. Multiscale Evolutionary Dynamics of Host-Associated Microbiomes. Cell. 2018;172:1216–1227. doi: 10.1016/j.cell.2018.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fichorova R, Fraga J, Rappelli P, Fiori PL. Trichomonas vaginalis infection in symbiosis with Trichomonasvirus and Mycoplasma. Res Microbiol. 2017;168:882–891. doi: 10.1016/j.resmic.2017.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gargantini PR, Serradell MDC, Rios DN, Tenaglia AH, Lujan HD. Antigenic variation in the intestinal parasite Giardia lamblia. Curr Opin Microbiol. 2016;32:52–58. doi: 10.1016/j.mib.2016.04.017. [DOI] [PubMed] [Google Scholar]
- Gomez-Arreaza A, Haenni AL, Dunia I, Avilan L. Viruses of parasites as actors in the parasite-host relationship: “A menage a trois”. Acta Trop. 2017;166:126–132. doi: 10.1016/j.actatropica.2016.11.028. [DOI] [PubMed] [Google Scholar]
- Gorla SK, McNair NN, Yang G, Gao S, Hu M, Jala VR, Haribabu B, Striepen B, Cuny GD, Mead JR, Hedstrom L. Validation of IMP dehydrogenase inhibitors in a mouse model of cryptosporidiosis. Antimicrob Agents Chemother. 2014;58:1603–1614. doi: 10.1128/AAC.02075-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gough R, Ellis J, Stark D. Comparison and Recommendations for the use of Dientamoeba fragilis Real-Time PCR assays. J Clin Microbiol. 2019 doi: 10.1128/JCM.01466-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinderfeld AS, Phukan N, Bar AK, Roberton AM, Simoes-Barbosa A. Cooperative interactions between Trichomonas vaginalis and associated bacteria enhance paracellular permeability of the cervicovaginal epithelium by dysregulating tight junctions. Infect Immun. 2019 doi: 10.1128/IAI.00141-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirt RP, Sherrard J. Trichomonas vaginalis origins, molecular pathobiology and clinical considerations. Curr Opin Infect Dis. 2015;28:72–79. doi: 10.1097/QCO.0000000000000128. [DOI] [PubMed] [Google Scholar]
- Hooks KB, O'Malley MA. Dysbiosis and Its Discontents. MBio. 2017;8 doi: 10.1128/mBio.01492-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horton B, Bridle H, Alexander CL, Katzer F. Giardia duodenalis in the UK: current knowledge of risk factors and public health implications. Parasitology. 2019;146:413–424. doi: 10.1017/S0031182018001683. [DOI] [PubMed] [Google Scholar]
- Hupalo DN, Bradic M, Carlton JM. The impact of genomics on population genetics of parasitic diseases. Curr Opin Microbiol. 2015;23:49–54. doi: 10.1016/j.mib.2014.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kellerova P, Tachezy J. Zoonotic Trichomonas tenax and a new trichomonad species, Trichomonas brixi n. sp., from the oral cavities of dogs and cats. Int J Parasitol. 2017;47:247–255. doi: 10.1016/j.ijpara.2016.12.006. [DOI] [PubMed] [Google Scholar]
- Khan A, Shaik JS, Grigg ME. Genomics and molecular epidemiology of Cryptosporidium species. Acta Trop. 2018;184:1–14. doi: 10.1016/j.actatropica.2017.10.023. [DOI] [PubMed] [Google Scholar]
- Kissinger P. Trichomonas vaginalis: a review of epidemiologic, clinical and treatment issues. BMC Infect Dis. 2015;15:307. doi: 10.1186/s12879-015-1055-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Labruyere E, Thibeaux R, Olivo-Marin JC, Guillen N. Crosstalk between Entamoeba histolytica and the human intestinal tract during amoebiasis. Parasitology. 2017:1–10. doi: 10.1017/S0031182017002190. [DOI] [PubMed] [Google Scholar]
- Leitsch D. A review on metronidazole: an old warhorse in antimicrobial chemotherapy. Parasitology. 2017:1–12. doi: 10.1017/S0031182017002025. [DOI] [PubMed] [Google Scholar]
- Lemieux MW, Sonzogni-Desautels K, Ndao M. Lessons learned from protective immune responses to optimize vaccines against cryptosporidiosis. Pathogens. 2017:7. doi: 10.3390/pathogens7010002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levy M, Kolodziejczyk AA, Thaiss CA, Elinav E. Dysbiosis and the immune system. Nat Rev Immunol. 2017;17:219–232. doi: 10.1038/nri.2017.7. [DOI] [PubMed] [Google Scholar]
- Loke P, Lim YAL. A Commensal Protozoan Strikes a Balance in the Gut. Cell Host Microbe. 2016;20:417–419. doi: 10.1016/j.chom.2016.09.016. [DOI] [PubMed] [Google Scholar]
- Lokmer A, Cian A, Froment A, Gantois N, Viscogliosi E, Chabe M, Segurel L. Use of shotgun metagenomics for the identification of protozoa in the gut microbiota of healthy individuals from worldwide populations with various industrialization levels. PLoS One. 2019;14:e0211139. doi: 10.1371/journal.pone.0211139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lukes J, Stensvold CR, Jirku-Pomajbikova K, Wegener Parfrey L. Are Human Intestinal Eukaryotes Beneficial or Commensals? PLoS Pathog. 2015;11:e1005039. doi: 10.1371/journal.ppat.1005039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lycke N. Recent progress in mucosal vaccine development: potential and limitations. Nat Rev Immunol. 2012;12:592–605. doi: 10.1038/nri3251. [DOI] [PubMed] [Google Scholar]
- Maritz JM, Land KM, Carlton JM, Hirt RP. What is the importance of zoonotic trichomonads for human health? Trends Parasitol. 2014;30:333–341. doi: 10.1016/j.pt.2014.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martinez-Diaz RA, Ponce-Gordo F, Rodriguez-Arce I, del Martinez-Herrero MC, Gonzalez FG, Molina-Lopez RA, Gomez-Munoz MT. Trichomonas gypaetinii n. sp., a new trichomonad from the upper gastrointestinal tract of scavenging birds of prey. Parasitol Res. 2015;114:101–112. doi: 10.1007/s00436-014-4165-5. [DOI] [PubMed] [Google Scholar]
- Marty M, Lemaitre M, Kemoun P, Morrier JJ, Monsarrat P. Trichomonas tenax and periodontal diseases: a concise review. Parasitology. 2017;144:1417–1425. doi: 10.1017/S0031182017000701. [DOI] [PubMed] [Google Scholar]
- Mercer F, Johnson PJ. Trichomonas vaginalis: Pathogenesis, Symbiont Interactions, and Host Cell Immune Responses. Trends Parasitol. 2018;34:683–693. doi: 10.1016/j.pt.2018.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miranda-Ozuna JFT, Rivera-Rivas LA, Cardenas-Guerra RE, Hernandez-Garcia MS, Rodriguez-Cruz S, Gonzalez-Robles A, Chavez-Munguia B, Arroyo R. Glucose-restriction increases Trichomonas vaginalis cellular damage towards HeLa cells and proteolytic activity of cysteine proteinases (CPs), such as TvCP2. Parasitology. 2019:1–11. doi: 10.1017/S0031182019000209. [DOI] [PubMed] [Google Scholar]
- Petersen C, Round JL. Defining dysbiosis and its influence on host immunity and disease. Cell Microbiol. 2014;16:1024–1033. doi: 10.1111/cmi.12308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pindak FF, Mora de Pindak M, Hyde BM, Gardner WA., Jr Acquisition and retention of viruses by Trichomonas vaginalis. Genitourin Med. 1989;65:366–371. doi: 10.1136/sti.65.6.366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinheiro J, Biboy J, Vollmer W, Hirt RP, Keown JR, Artuyants A, Black MM, Goldstone DC, Simoes-Barbosa A. The Protozoan Trichomonas vaginalis Targets Bacteria with Laterally Acquired NlpC/P60 Peptidoglycan Hydrolases. MBio. 2018;9 doi: 10.1128/mBio.01784-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Price LB, Hungate BA, Koch BJ, Davis GS, Liu CM. Colonizing opportunistic pathogens (COPs): The beasts in all of us. PLoS Pathog. 2017;13:e1006369. doi: 10.1371/journal.ppat.1006369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rendon-Maldonado J, Espinosa-Cantellano M, Soler C, Torres JV, Martinez-Palomo A. Trichomonas vaginalis: in vitro attachment and internalization of HIV-1 and HIV-1-infected lymphocytes. J Eukaryot Microbiol. 2003;50:43–48. doi: 10.1111/j.1550-7408.2003.tb00104.x. [DOI] [PubMed] [Google Scholar]
- Rivero FD, Saura A, Prucca CG, Carranza PG, Torri A, Lujan HD. Disruption of antigenic variation is crucial for effective parasite vaccine. Nat Med. 2010;16:551–557. doi: 10.1038/nm.2141. 551p following 557. [DOI] [PubMed] [Google Scholar]
- Rook G, Backhed F, Levin BR, McFall-Ngai MJ, McLean AR. Evolution, human-microbe interactions, and life history plasticity. Lancet. 2017;390:521–530. doi: 10.1016/S0140-6736(17)30566-4. [DOI] [PubMed] [Google Scholar]
- Rowan-Nash AD, Korry BJ, Mylonakis E, Belenky P. Cross-Domain and Viral Interactions in the Microbiome. Microbiol Mol Biol Rev. 2019;83 doi: 10.1128/MMBR.00044-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rush G, Reynolds MW, Calvani NED, Slapeta J. Addressing the constraints of Tritrichomonas foetus sample collection in remote areas: lyophilized modified Diamond's media as a substitute for liquid medium. Parasitology. 2019:1–4. doi: 10.1017/S0031182019000258. [DOI] [PubMed] [Google Scholar]
- Ryan U, Paparini A, Oskam C. New Technologies for Detection of Enteric Parasites. Trends Parasitol. 2017;33:532–546. doi: 10.1016/j.pt.2017.03.005. [DOI] [PubMed] [Google Scholar]
- Schroder K, Bosch TC. The Origin of Mucosal Immunity: Lessons from the Holobiont Hydra. MBio. 2016;7 doi: 10.1128/mBio.01184-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serradell MC, Rupil LL, Martino RA, Prucca CG, Carranza PG, Saura A, Fernandez EA, Gargantini PR, Tenaglia AH, Petiti JP, Tonelli RR, et al. Efficient oral vaccination by bioengineering virus-like particles with protozoan surface proteins. Nat Commun. 2019;10 doi: 10.1038/s41467-018-08265-9. 361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Serradell MC, Saura A, Rupil LL, Gargantini PR, Faya MI, Furlan PJ, Lujan HD. Vaccination of domestic animals with a novel oral vaccine prevents Giardia infections, alleviates signs of giardiasis and reduces transmission to humans. NPJ Vaccines. 2016;1 doi: 10.1038/npjvaccines.2016.18. 16018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith PD, MacDonald TT, Blumberg RS. Phylogeny of the mucosal immune system. In: Smith PD, MacDonald TT, Blumberg RS, editors. Principles of mucosal immunology. Garland Science/Taylor & Francis Group; London: 2013. pp. 19–26. [Google Scholar]
- Stensvold CR. Pinning down the role of common luminal intestinal parasitic protists in human health and disease - status and challenges. Parasitology. 2019:1–7. doi: 10.1017/S0031182019000039. [DOI] [PubMed] [Google Scholar]
- Stensvold CR, van der Giezen M. Associations between Gut Microbiota and Common Luminal Intestinal Parasites. Trends Parasitol. 2018;34:369–377. doi: 10.1016/j.pt.2018.02.004. [DOI] [PubMed] [Google Scholar]
- Stentiford GD, Becnel J, Weiss LM, Keeling PJ, Didier ES, Williams BP, Bjornson S, Kent ML, Freeman MA, Brown MJF, Troemel ER, et al. Microsporidia - Emergent Pathogens in the Global Food Chain. Trends Parasitol. 2016;32:336–348. doi: 10.1016/j.pt.2015.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thi Trung Thu T, Margarita V, Cocco AR, Marongiu A, Dessi D, Rappelli P, Fiori PL. Trichomonas vaginalis transports virulent Mycoplasma hominis and transmits the infection to human cells after Metronidazole treatment: a potential role in bacterial Invasion of fetal membranes and amniotic fluid. J Pregnancy. 2018;2018 doi: 10.1155/2018/5037181. 5037181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Gestel RS, Kusters JG, Monkelbaan JF. A clinical guideline on Dientamoeba fragilis infections. Parasitology. 2018:1–9. doi: 10.1017/S0031182018001385. [DOI] [PubMed] [Google Scholar]
- Vargas Rigo G, Petro-Silveira B, Devereux M, McCann M, Souza Dos Santos AL, Tasca T. Anti-Trichomonas vaginalis activity of 1,10-phenanthroline-5,6-dione-based metallodrugs and synergistic effect with metronidazole. Parasitology. 2018:1–5. doi: 10.1017/S003118201800152X. [DOI] [PubMed] [Google Scholar]
- Wypych TP, Marsland BJ. Antibiotics as Instigators of Microbial Dysbiosis: Implications for Asthma and Allergy. Trends Immunol. 2018;39:697–711. doi: 10.1016/j.it.2018.02.008. [DOI] [PubMed] [Google Scholar]