Abstract
Thrombotic microangiopathies include hemolytic uremic syndrome (HUS) and thrombotic thrombocytopenic purpura (TTP). Measurement of plasma levels of “a disintegrin-like and metalloproteinase with thrombospondin type 1 motifs 13” (ADAMTS13) activity can distinguish HUS from TTP. Reduced plasma ADAMTS13 activity (< 10% normal range) is atypical for HUS, but not for TTP. However, we detected reduced ADAMTS13 activity in a patient with Shiga toxin-producing Escherichia coli-associated HUS caused by non-IgG anti-ADMTS13 autoantibodies. Furthermore, the patient exhibited possible genetic abnormalities associated with atypical HUS. The patient fully recovered after administration of supportive therapy. To the best of our knowledge, very few cases of STEC-HUS with reduced ADAMTS13 activity have been reported; thus far, none have described the presence of non-IgG anti-ADMTS13 autoantibodies. Therefore, we suggest that anti-ADAMTS13 analyses should be performed in patients diagnosed with STEC-HUS, especially in those who present with prolonged healing or unexpected clinical symptoms.
Keywords: A disintegrin-like and metalloproteinase with thrombospondin type 1 motifs 13, Atypical hemolytic uremic syndrome, Shiga toxin-producing Escherichia coli-associated hemolytic uremic syndrome, Thrombotic microangiopathy, Thrombotic thrombocytopenic purpura
Introduction
It is difficult to distinguish hemolytic uremic syndrome (HUS) from thrombotic thrombocytopenic purpura (TTP) because of their clinical similarities [1]. Acute renal injury, microangiopathic hemolytic anemia, and thrombocytopenia characterize HUS [2]. In addition to these three symptoms, neurological symptoms are present in patients with TTP, but are undetectable in 35% of affected patients; thus, it is difficult to distinguish TTP and HUS solely on the basis of a patient’s symptoms [3]. However, this distinction can be made by measurement of ADAMTS13 activity [4]: ADAMTS13 activity is generally reduced in patients with TTP, compared with that in patients with HUS. HUS has been observed in patients with lower ADAMTS13 activities, although the values are not significantly different from the normal range [4]. If ADAMTS13 activity is reduced to < 10% of the normal range (considered a significant reduction), the patient is diagnosed with TTP [4]. Furthermore, if an ADAMTS13 inhibitor is detected, the patient is diagnosed with acquired TTP [4].
Shiga toxin-producing Escherichia coli (STEC)-HUS occurs after hemorrhagic enteritis caused by enterohemorrhagic E. coli [5]. In STEC-HUS, Shiga toxin (Stx) is released in the intestine and circulates in the bloodstream [6]. In vitro, Stx interacts with the surfaces of human endothelial cells and leukocytes, which suggests that a similar mechanism is present in the bloodstream. Vascular damage induced by Stx promotes cytokine and chemokine secretion by endothelial cells [7]. The primary target organ is the kidney, in which various cells express high levels of surface receptors targeted by Stx; these cells are, therefore, susceptible to toxin-mediated injury. [7]. Notably, Stx directly impairs the endothelium and changes endothelial function through production of inflammatory cytokines, chemokines, and adhesion factors, as well as by induction of hypercoagulability [7]. Thus, reduction of ADAMTS13 activity, caused by anti-ADAMTS13 autoantibodies, is not detected in patients with STEC-HUS [6].
Antibiotics are not recommended in the United States for treatment of patients with STEC-HUS with infectious gastroenteritis [8]. Therefore, we do not use antibiotics for treatment of such patients in our hospital, as this may cause the release of toxins and lytic phages [9]. In these patients, appropriate early volume expansion with intravenous fluid, including sodium, may avoid oliguria, anuria, and the requirement for dialysis [10].
Here, we describe treatment of a patient with STEC-HUS, in whom ADAMTS13 activity was reduced by non-IgG anti-ADAMTS13 autoantibodies. In addition, the patient exhibited possible genetic abnormalities caused by a rare mutation of membrane cofactor protein (MCP), which was detected in analysis of atypical HUS (aHUS).
Case report
A 21-year-old woman exhibited fever, lower abdominal pain, and diarrhea after ingesting raw meat. She was prescribed an antibiotic and an antiflatulent at another clinic. She reported bloody diarrhea ten times daily and experienced increasing fatigue. Laboratory analysis revealed severe anemia [hemoglobin (Hgb), 6.0 g/dL], reduced platelet (Plt) count (3.1 × 104/µL), and renal injury (serum creatinine, 6.17 mg/dL). She was admitted to our hospital 10 days after the onset of symptoms. She reported no notable medical history, no family history of disease, no medications, and no allergic disorders.
She lost 1.0 kg of body weight and exhibited palpebral conjunctiva, as well as mild jaundice without edema. She maintained consciousness and had no fever or abdominal pain; bloody diarrhea resolved upon hospitalization. Her laboratory findings were as follows: Hgb, 4.9 g/dL; Plt count, 1.9 × 104 µL; lactate dehydrogenase, 3302 U/L; haptoglobin, < 10 mg/dL; negative direct Coombs test; and liver and renal injury (Table 1). A peripheral blood smear showed numerous crushed erythrocytes. The patient exhibited the essential diagnostic criteria of microangiopathic hemolytic anemia and thrombocytopenia (TMA) with severe renal failure without neurological abnormalities. Although neurologic abnormalities are commonly considered to be characteristic of TTP and renal dysfunction is considered to be characteristic of HUS, we regarded the patient’s symptoms as indicative of a typical clinical course of HUS.
Table 1.
Laboratory values
| (Urine) | |||
| Density | 1.014 | 1.005–1.030 | |
| pH | 5.0 | ↓ | 6.0–6.5 |
| Protein | 3 + | ↑ | Negative |
| Blood | 3 + | ↑ | Negative |
| Red blood cell (/HPF) | 10–19 | ↑ | 0–4 |
| Hyaline cast (/HPF) | 10–19 | ↑ | Negative |
| Granular cast (/LPF) | 0–1 | ↑ | Negative |
| Epithelial cast (/LPF) | 1–4 | ↑ | Negative |
| Waxy cas t (/LPF) | 1–4 | ↑ | Negative |
| Pro/Cre ratio (g/g gCr) | 3.79 | ↑ | < 0.15 |
| NAG (U/L) | 44.6 | ↑ | 1.0–6.3 |
| β2MG (mg/L) | 0.31 | 0.9–1.9 | |
| Gestation test | Negative | Negative | |
| (Blood) | |||
| Leukocyte count (/µg) | 8830 | 3040–8540 | |
| Erythrocyte count (× 103/μL) | 1720 | ↓ | 3780–4990 |
| Hemoglobin (g/dL) | 4.9 | ↓ | 10.8–14.9 |
| Hematocrit (%) | 14.0 | ↓ | 35.6–45.4 |
| MCV (fL) | 90.9 | 85.0–101.0 | |
| MCHC (g/dL) | 33.1 | 30.7–34.0 | |
| Platelet count (× 103/μL) | 19 | ↓ | 150–360 |
| Neutrophil (%) | 64.0 | 38.3–71.1 | |
| Lymphocyte (%) | 23.5 | ↓ | 25.0–45.0 |
| Monocyte (%) | 11.6 | ↑ | 4.0–7.0 |
| PT-INR | 1.03 | ||
| APTT (s) | 32.1 | 26.9–38.1 | |
| Fibrinogen (mg/dL) | 195.7 | ↓ | 200.0–400.0 |
| FDP (µg/mL) | 21.1 | ↑ | ≤ 5.0 |
| d-Dimer (µg/mL) | 6.2 | ↑ | ≤ 1.0 |
| Direct Coombs | Negative | Negative | |
| Ferritin (ng/ml) | 1716.4 | ↑ | 11.0–128.9 |
| TSAT (%) | 41.7 | ↑ | 35 |
| Haptoglobin (mg/dL) | < 10 | ↓ | 19–170 |
| Total bilirubin (mg/dL) | 2.3 | ↑ | 0.3–1.2 |
| Direct bilirubin (mg/dL) | 0.4 | ↑ | 0.1–0.3 |
| AST (U/L) | 128 | ↑ | 13–33 |
| ALT (U/L) | 70 | ↑ | 8–42 |
| LDH (U/L) | 3202 | ↑ | 119–229 |
| Alkaline phosphatase (U/L) | 138 | 115–359 | |
| γ-GTP (U/L) | 10 | 9–32 | |
| Amylase (U/L) | 76 | 44–132 | |
| Creatine kinase (U/L) | 159 | 45–163 | |
| Sodium (mEq/L) | 130 | ↓ | 138–146 |
| Potassium (mEq/L) | 3.3 | ↓ | 3.6–4.9 |
| Chloride (mEq/L) | 99 | 99–109 | |
| Corrected serum calcium (mg/dL) | 9.2 | 8.6–10.4 | |
| Phosphate (mg/dL) | 3.7 | 2.5–4.7 | |
| Total protein (g/dL) | 5.4 | ↓ | 6.7–8.3 |
| Albumin (g/dL) | 2.6 | ↓ | 4.0–5.0 |
| Uric acid (mg/dL) | 12.8 | ↑ | 2.3–7.0 |
| Urea nitrogen (mg/dL) | 92.7 | ↑ | 8.0–22.0 |
| Creatine (mg/dL) | 6.17 | ↑ | 0.40–0.70 |
| eGFR (mL/min/1.73 m2) | 8.2 | ↓ | ≥ 90 |
| CRP (mg/dL) | 1.11 | ↑ | ≤ 0.20 |
| Procalcitonin (ng/mL) | 0.67 | ↑ | ≤ 0.49 |
| IgG (mg/dL) | 929 | 870–1700 | |
| IgA (mg/dL) | 210 | 110–410 | |
| IgM (mg/dL) | 142 | 46–260 | |
| CH50 (U/mL) | 39.0 | 30.0–46.0 | |
| C3 (mg/dL) | 100 | 86–160 | |
| C4 (mg/dL) | 19 | 17–45 | |
| PR3-ANCA (U/mL) | < 3.5 | < 3.5 | |
| MPO-ANCA (U/mL) | < 3.5 | < 3.5 | |
| Anti-nuclear antibody | < × 80 | < × 80 | |
| Other antibodies | Negative | Negative | |
| Hepatitis B surface antigen | Negative | Negative | |
| Hepatitis B virus antibody | Negative | Negative | |
| Hepatitis B core antigen | Negative | Negative | |
| Hepatitis C antibody | Negative | Negative | |
| HIV antibody | Negative | Negative | |
| CMV (CF) | Negative | Negative | |
| HSV | Negative | Negative | |
| Epstein–Barr virus | Negative | Negative | |
| Parvo virus | Negative | Negative | |
| ADAMTS13 activity (%) | 9.3 | ↓ | 50–150 |
| Anti-ADAMTS13 inhibitor (BU/mL) | 4.2 | ↑ | < 0.5 |
| Anti-ADAMTS13 IgG | Negative | Negative | |
HPF high-power field, LPF low-power field, Pro/Cre Ratio protein/creatinine ratio, NAGN-acetyl-β-d-glucosaminidase, β2MG β2-microglobulin, FDP fibrinogen degradation product, MCV mean corpuscular volume, MCHC mean corpuscular hemoglobin, PT-INR international normalized ratio of prothrombin time, APTT activated partial thromboplastin time, FDP fibrin degradation products, TSAT transferrin saturation, AST aspartate aminotransferase, ALT alanine aminotransferase, LDH lactase dehydrogenase, γ-GTP γ-glutamyl transpeptidase, eGFR estimated glomerular filtration rate, IgG immunoglobulin G, IgA immunoglobulin A, IgM immunoglobulin M, PR3-ANCA proteinase 3-anti-neutrophil cytoplasmic antibody, MPO-ANCA, myeloperoxidase-anti-neutrophil cytoplasmic antibody, HIV human immunodeficiency virus, CFH complement factor H
The patient’s stool was cultured after she had been prescribed an antibiotic. Thus, enterohemorrhagic E. coli was not detected. However, the patient had two positive results for anti-O157 E. coli lipopolysaccharide antibody. Therefore, we diagnosed her with STEC-HUS. The patient was hospitalized and infusion therapy was implemented; erythrocytes were transfused to treat severe anemia. Antibiotics were not used, and the patient underwent volume expansion with intravenous fluid; she was instructed to fast. These treatments are generally regarded as conventional supportive therapy [10].
We next measured the level of plasma ADAMTS13 activity in the patient, as well as the level of an ADAMTS13 inhibitor at admission. Using an ADAMTS13-act-ELISA kit (Technoclone, Vienna, Austria). This kit was able to detect the activity of whole ADAMTS13 inhibitor molecules. The plasma level of ADAMTS13 activity was 9.3% of normal, whereas that of the ADAMTS13 inhibitor was 4.2 Bethesda Units/mL. These data led us to a diagnosis of acquired TTP, with STEC infection as the underlying disease. Therefore, the patient’s final diagnosis was STEC-HUS, although we considered the possibility of acquired TTP. We noted that STEC-HUS and acquired TTP were present in the patient, although these appeared to be inconsistent diagnoses; thus, we confirmed the ADAMTS13 inhibitor properties. Anti-ADAMTS13 IgG antibodies were measured using the Technozym ADAMTS-13 INH kit (Technoclone). However, we did not detect IgG antibodies against ADAMTS13. Therefore, the ADAMTS13 inhibitor that we initially detected was judged to be a non-IgG antibody against ADAMTS13.
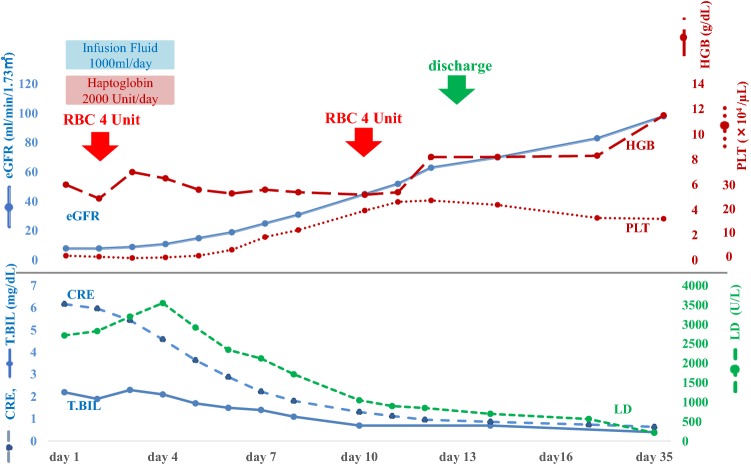
Conventional supportive treatment (fluid therapy and RBC transfusion) improved the patient’s kidney function, and she was discharged after 13 days (Fig. 1). Immediately before discharge, her ADAMTS13 activity had increased to > 100%, and the titer of ADAMTS13 inhibitor had decreased below the limit of detection. Moreover, the patient lacked detectable symptoms, and her Plt count and renal function had recovered to normal.
Fig. 1.
Patient’s clinical course
After discharge, she underwent screening to determine whether HUS had been induced by an infectious disease. Plasma complement factor H (CFH) autoantibody was not detected using a CFH IgG ELISA Kit (KA1477; Abnova, Taipei City, Taiwan). We performed whole exome sequencing of genes associated with aHUS as follows: CFH; membrane cofactor protein (MCP); complement factor I (CFI), C3; complement factor B (CFB); diacylglycerol kinase epsilon (DGKE); and thrombomodulin (THBD). These analyses detected a rare mutation, p.Ala311Val (c.932C>T), in exon 8 of MCP; this mutation had been previously detected. [11, 12].
Discussion
Here, we described a patient with STEC-HUS who exhibited reduced ADAMTS13 activity and an increased titer of non-IgG anti-ADAMTS13 autoantibodies during hospitalization. Furthermore, the patient harbored a rare p.Ala311Val (c.932C>T) mutation in MCP. To the best of our knowledge, there have been no other reports of patients with these particular comorbidities.
The patient’s coagulation markers increased upon hospitalization (Table 1). Disseminated intravascular coagulation (DIC) and TMA can be distinguished as follows: DIC exhibits extreme abnormalities in clotting markers, whereas TMA exhibits only mild abnormalities in these markers [13]. However, DIC can be evoked by TMA, such as STEC-HUS [13]. Therefore, it is often difficult to distinguish DIC during early TMA.
Most patients with acquired TTP with severe ADAMTS13 deficiency have circulating anti-ADAMTS13 IgG autoantibodies that can neutralize ADAMTS13 activity [14]. Certain infectious diseases, including STEC, cause an autoimmune reaction to ADAMTS13 [15]. When acquired TTP occurs as a result of infection, cross-reactivity of CD4-positive T cells is suspected, mediated by a protein derived from a microorganism that produces a protein homologous to ADAMTS13 [16]. Moreover, some TTP patients with severe ADAMTS13 deficiency lack ADAMTS13-neutralizing autoantibodies [17]. In general, in patients with acquired TTP, ADAMTS13-neutralizing autoantibodies reduce the activity of ADAMTS13 [16]. However, in the present case, the patient’s autoantibodies were not IgGs, but may have been IgA or IgM, which may explain the inhibition of ADAMTS13 activity. Unfortunately, we were limited by the volumes of blood collected from the patient at the early and severe clinical stages, and could not conduct further analyses to precisely identify the antibody.
The patient’s pathophysiological condition did not strictly meet the accepted definition of acquired TTP. When ADAMTS13 activity is undetectable, abnormally large von Willebrand factor multimers in plasma can exhibit greater reactivity with Plt and cause disseminated platelet thrombi that are characteristic of TTP [18]. Alternatively, proteases may be activated due to STEC infection; in the present case, this may have led to the remarkable reduction (< 10%) of ADAMTS13 activity, relative to that detected in patients with TTP. This may explain the DIC-like disease course, which resulted from the reduction in ADAMTS13 activity. However, we were unable to conduct further analyses of these putative inhibitors because of the limited blood sample, as indicated in the prior paragraph. Contrary to expectations, the patient’s ADAMTS13 activity had decreased to the level present in patients with acquired TTP; therefore, we re-measured ADAMTS13 with the same sample. The findings showed good reproducibility of the patient’s ADAMTS13 reduced activity. Next, we assessed whether the ADAMTS13 inhibitor in that sample was IgG, and found that it was not. Because of these repeated examinations at early and severe clinical phases, the samples were insufficient for further analyses, as noted in a prior paragraph.
The results of aHUS analysis revealed a rare mutation, p.Ala311Val, in exon 8 of MCP. Although this mutation has been found in patients with aHUS, its pathological significance is unknown [11, 12]. Therefore, it is difficult to determine whether this mutation is pathologically associated with the onset of aHUS.
In summary, we have described a patient who was diagnosed with STEC-HUS, which resulted in a pathology similar to that of acquired TTP. Our patient was successfully treated with the conventional supportive therapy. IgG autoantibodies against ADAMTS13 were not detected, which impaired our ability to diagnose clinical TTP. Furthermore, we identified a rare mutation of MCP, which has been associated with the onset of aHUS. It was difficult to determine whether this mutation was associated with disease in this patient, and we speculate that it may have led to exacerbation of disease. Notably, we were also unable to confirm a diagnosis of aHUS. This case was highly intriguing because of the involvement of distinct concurrent pathologies associated with TMA. In this case, STEC-HUS, DIC, reduced levels of non-IgG anti-ADAMTS13 autoantibodies, and the effect of an aHUS-like mutation combined to create a complicated pathological condition. Even in patients with apparently simple STEC-HUS, we consider it to be necessary to perform sufficient evaluation when healing is prolonged or unexpected clinical symptoms are observed.
Acknowledgements
We would like to thank Yuka Sugawara, Yoichiro Ikeda, Hideki Kato, and Masaomi Nangaku (The University of Tokyo Hospital, Japan) for genetic screening of aHUS and Ryan Chastain-Gross, PhD, from Edanz Group (http://www.edanzediting.com/ac) for editing a draft of this manuscript.
Compliance with ethical standards
Conflict of interest
All the authors have declared no competing interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee at which the studies were conducted (IRB approval number Hi-204) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Shinya Nakayama, Email: socr1@hiroshima-u.ac.jp.
Shuma Hirashio, Email: shumah@hiroshima-u.ac.jp.
Haruka Yorishima, Email: yorishima-hma@umin.ac.jp.
Toshiki Doi, Email: doitoshi@hiroshima-u.ac.jp.
Yoko Yoshida, Email: yoyoshida-tky@umin.ac.jp.
Masanori Matsumoto, Email: mmatsumo@naramed-u.ac.jp.
Takao Masaki, Email: masakit@hiroshima-u.ac.jp.
References
- 1.Furlan M, Robles R, Galbusera M, Remuzzi G, Kyrle PA, Brenner B, Krause M, Scharrer I, Aumann V, Mittler U, Solenthaler M, Lämmle B. von Willebrand factor-cleaving protease in thrombotic thrombocytopenic purpura and the hemolytic-uremic syndrome. N Engl J Med. 1998;339:1578–1584. doi: 10.1056/NEJM199811263392202. [DOI] [PubMed] [Google Scholar]
- 2.Birlutiu V, Birlutiu RM. Haemolytic-uremic syndrome due to infection with adenovirus: a case report and literature review. Medicine. 2018;97:e9895. doi: 10.1097/MD.0000000000009895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Rock GA, Shumak KH, Buskard NA, Blanchette VS, Kelton JG, Nair RC, Spasoff RA. Comparison of plasma exchange with plasma infusion in the treatment of thrombotic thrombocytopenic purpura. Canadian Apheresis Study Group. N Engl J Med. 1991;325:393–397. doi: 10.1056/NEJM199108083250604. [DOI] [PubMed] [Google Scholar]
- 4.Vesely SK, George JN, Lämmle B, Studt JD, Alberio L, El-Harake MA, Raskob GE. ADAMTS13 activity in thrombotic thrombocytopenic purpura-hemolytic uremic syndrome: relation to presenting features and clinical outcomes in a prospective cohort of 142 patients. Blood. 2003;102:60–68. doi: 10.1182/blood-2003-01-0193. [DOI] [PubMed] [Google Scholar]
- 5.Karmali MA, Steele BT, Petric M, Lim C. Sporadic cases of haemolytic-uraemic syndrome associated with faecal cytotoxin and cytotoxin-producing Escherichia coli in stools. Lancet. 1983;1(8325):619–620. doi: 10.1016/S0140-6736(83)91795-6. [DOI] [PubMed] [Google Scholar]
- 6.Trachtman H, Austin C, Lewinski M, Stahl RAK. Renal and neurological involvement in typical Shiga toxin-associated HUS. Nat Rev Nephrol. 2012;8:658–669. doi: 10.1038/nrneph.2012.196. [DOI] [PubMed] [Google Scholar]
- 7.Petruzziello-Pellegrini TN, Marsden PA. Shiga toxin-associated hemolytic uremic syndrome: advance in pathogenesis and therapeutics. Curr Opin Nephrol Hypertens. 2012;21:433–440. doi: 10.1097/MNH.0b013e328354a62e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Guerrant RL, Van Gilder T, Steiner TS, Thielman NM, Slutsker L, Tauxe RV, Hennessy T, Griffin PM, DuPont H, Sack RB, Tarr P, Neill M, Nachamkin I, Reller LB, Osterholm MT, Bennish ML, Pickering LK, Infectious Diseases Society of America Practice guidelines for the management of infectious diarrhea. Clin Infect Dis. 2001;32:331–351. doi: 10.1086/318514. [DOI] [PubMed] [Google Scholar]
- 9.Thielman NM, Guerrant RL. Clinical practice. Acute infectious diarrhea. N Engl J Med. 2004;350:38–47. doi: 10.1056/NEJMcp031534. [DOI] [PubMed] [Google Scholar]
- 10.Hickey CA, Beattie TJ, Cowieson J, Miyashita Y, Strife CF, Frem JC, Peterson JM, Butani L, Jones DP, Havens PL, Patel HP, Wong CS, Andreoli SP, Rothbaum RJ, Beck AM, Tarr PI. Early volume expansion during diarrhea and relative nephroprotection during subsequent hemolytic uremic syndrome. Arch Pediatr Adolesc Med. 2011;165:884–889. doi: 10.1001/archpediatrics.2011.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yoshida Y, Miyata T, Matsumoto M, Shirotani-Ikejima H, Uchida Y, Ohyama Y, Kokubo T, Fujimura Y. A novel quantitative hemolytic assay coupled with restriction fragment length polymorphisms analysis enabled early diagnosis of atypical hemolytic uremic syndrome and identified unique predisposing mutations in Japan. PLoS One. 2015;10:e0124655. doi: 10.1371/journal.pone.0124655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Omura T, Watanabe E, Otsuka Y, Yoshida Y, Kato H, Nangaku M, Miyata T, Oda S. Complete remission of thrombotic microangiopathy after treatment with eculizumab in a patient with non-Shiga toxin-associated bacterial enteritis: a case report. Medicine (Baltimore). 2016;95:e4104. doi: 10.1097/MD.0000000000004104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wada H, Wakita Y, Nakase T, Shimura M, Hiyoyama K, Nagaya S, Deguchi H, Mori Y, Kaneko T, Deguchi K, Fujii J, Shiku H. Increased plasma-soluble fibrin monomer levels in patients with disseminated intravascular coagulation. Am J Hematol. 1996;51:255–260. doi: 10.1002/(SICI)1096-8652(199604)51:4<255::AID-AJH1>3.0.CO;2-V. [DOI] [PubMed] [Google Scholar]
- 14.Scully M, Hunt BJ, Benjamin S. Guidelines on the diagnosis and management of thrombotic thrombocytopenic purpura and other thrombotic microangiopathies. Br J Haematol. 2012;158:323–335. doi: 10.1111/j.1365-2141.2012.09167.x. [DOI] [PubMed] [Google Scholar]
- 15.Fujimura Y, Matsumoto M. Registry of 919 patients with thrombotic microangiopathies across Japan: database of Nara Medical University during 1998–2008. Intern Med. 2010;49:7–15. doi: 10.2169/internalmedicine.49.2706. [DOI] [PubMed] [Google Scholar]
- 16.Verbij FC, Fijnheer R, Voorberg J, Sorvillo N. Acquired TTP: ADAMTS13 meets the immune system. Blood Rev. 2014;28:227–234. doi: 10.1016/j.blre.2014.07.004. [DOI] [PubMed] [Google Scholar]
- 17.Knecht ME, Mayr M, Ferrari S, Scheiflinger F, Trendelenburg M. A patient with SLE-associated thrombotic microangiopathy and non-neutralizing antibodies against ADAMTS13. Nephrol Dial Transplant. 2010;25:1720–1722. doi: 10.1093/ndt/gfq021. [DOI] [PubMed] [Google Scholar]
- 18.George JN. Clinical practice. Thrombotic thrombocytopenic purpura. N Engl J Med. 2006;354:1927–1935. doi: 10.1056/NEJMcp053024. [DOI] [PubMed] [Google Scholar]