Abstract
Introduction
Pegaspargase, a pegylated asparaginase, is a core component in the treatment of acute lymphoblastic leukemia. Pegaspargase in liquid form has a limited shelf life of 8 months due to depegylation, leading to changes in purity and potency over time. Lyophilization is an approach that can improve the stability of biological drug conjugates.
Methods
Here we describe the development of a lyophilized formulation of pegaspargase and present results of a series of tests demonstrating that the lyophilized form has comparable physicochemical properties to the liquid form.
Results
Stability tests of critical quality attributes, including purity, potency, aggregates and total free polyethylene glycol, demonstrate that lyophilized pegaspargase remains stable for at least 3 years, with optimum stability achieved with storage under refrigerated conditions (2–8 °C).
Conclusions
Lyophilization improved the stability of pegaspargase without altering other physicochemical properties, permitting a prolonged shelf life of at least 2 years when stored at 2–8 °C. This may enable greater storage flexibility and allow for better management of pegaspargase.
Funding
Study Sponsor: Baxalta (now part of Takeda). Publication Sponsor: Servier Affaires Médicales.
Electronic supplementary material
The online version of this article (10.1007/s12325-019-00988-5) contains supplementary material, which is available to authorized users.
Keywords: Cancer chemotherapy, Enzymes, Formulation, Lyophilization, Pediatric, Pegylation, Stability, Stabilization
Introduction
The treatment of acute lymphoblastic leukemia (ALL) involves the administration of multiple chemotherapeutic agents, typically including an asparaginase agent to deplete serum l-asparagine levels [1–3]. l-Asparaginase may be derived from Escherichia coli (E. coli asparaginase) or isolated from Erwinia chrysanthemi (Erwinase®, Jazz Pharmaceuticals; Erwinia asparaginase; crisantaspase) [4–10]. Modified forms of E. coli asparaginase with improved pharmacological properties have been developed, including pegylated E. coli asparaginase (Oncaspar® [pegaspargase], Servier) [11]. A recombinant asparaginase (Spectrila®, medac Pharma) is also available, and pegylated forms of recombinant enzymes are in development (MC0609; pegcrisantaspase) [12–14]. Pegaspargase is used as a first-line treatment for ALL in clinical practice, whereas crisantaspase is generally employed following failure of E. coli-derived asparaginases due to immunological reactions [15].
Pegylation of E. coli asparaginase confers advantages over the native enzyme, including prolonged half-life [16–18], which leads to reduced immunogenicity [17, 19] and reduced frequency of administration (every 2 weeks versus twice to three times weekly). In a comparative study of native E. coli asparaginase versus pegaspargase in children with ALL, significantly lower proportions of patients treated with pegaspargase had elevated anti-asparaginase antibody ratios compared with those receiving native E. coli asparaginase. The difference was most notable during the first delayed intensification phase (following previous exposure to treatment during the induction phase): over 40% of patients treated with native E. coli asparaginase, compared with 11% of those receiving pegaspargase, had antibody ratios greater than 1.5 times the negative control. High antibody titers had been associated with low asparaginase activity in previous studies, and antibody ratios greater than 1.5 were associated with low asparaginase activity in patients receiving native E. coli asparaginase [19]. However, in patients receiving pegaspargase, none of the samples with antibody ratios of 1.5 or greater were associated with low asparaginase activity, suggesting that pegaspargase was not neutralized or cleared more rapidly when antibody levels were elevated [19].
Pegaspargase is a conjugated enzyme, with polyethylene glycol (PEG) covalently bound to l-asparaginase via an ester linkage. Hydrolytic instability has been observed in vitro [20], leading to potential for depegylation if the ester bond undergoes hydrolysis. Similar to other pegylated pharmaceuticals [21, 22], pegylation increases steric hindrance of the active site on l-asparaginase; consequently, the native or depegylated product has greater enzymatic activity than the pegylated product, as well as a shorter half-life [20]. The shelf life of pegaspargase is limited to 8 months [23].
It is therefore important to understand the stability of new pegylated asparaginase preparations, as this may impact many factors including shelf life [20]. Since depegylation results from hydrolysis of the ester bond linking PEG to l-asparaginase [20], removal of water from the formulation by lyophilization (freeze-drying) is a rational approach to improve stability of the pegylated product and is a widely used method to improve the long-term storage stability of biopharmaceuticals [24].
The freezing and drying processes involved in lyophilization can themselves impose stresses on proteins [24]. One potential consequence of such stress is protein aggregation [25], forming sub-visible particles in the reconstituted solution, which can lead to increased immunogenicity [26, 27]. Stabilizing agents, such as sucrose or sorbitol, have been shown to reduce the sub-visible particle load in reconstituted lyophilized solutions of immunoglobulins [25, 28], and are typically included among excipients used in the production of lyophilized drug products [24].
Product quality testing is therefore important to ensure that the lyophilized product gains the desired property of improved stability, without incurring detrimental effects, such as changes in activity or increased propensity to form protein aggregates. A comprehensive series of analytical methods must be employed to determine properties of the lyophilized cake and behavior of the protein upon reconstitution as a solution for injection/administration [24]. This article describes the development process that has been used to produce a lyophilized form of pegaspargase, and provides a comparative assessment of structure, potency, quality and purity of the lyophilized and liquid formulations, in addition to exploring optimum storage conditions for lyophilized pegaspargase.
Methods
Compliance with Ethics Guidelines
This article does not contain any studies with human participants or animals.
Drug Substance Composition
Lyophilized pegaspargase contains largely the same constituents as liquid pegaspargase, although quantities of some excipients are reduced in the lyophilized formulation, including dibasic sodium phosphate, monobasic sodium phosphate and sodium chloride, and sucrose is added to the lyophilized formulation as a stabilizing agent (Table 1). Both formulations contain 750 U/mL pegaspargase, following reconstitution of the lyophilized formulation with 5.2 mL sterile water for injection per vial.
Table 1.
Constituents of liquid and lyophilized formulations of pegaspargase
| Component | Function | Pegaspargase formulation/amount per mL | |
|---|---|---|---|
| Liquid | Lyophilizeda | ||
| Pegaspargase drug substance | Active ingredient | 750 U | 750 U |
| Dibasic sodium phosphate | Buffering agent | 5.58 mg | 2.79 mg |
| Monobasic sodium phosphate | Buffering agent | 1.20 mg | 0.60 mg |
| Sodium chloride | Tonicity | 8.50 mg | 4.25 mg |
| Sucrose | Lyoprotectant (stabilizing agent) | N/A | 45 mg |
| Water for injection | Solvent | QS to 1.0 g | QS to 1.0 g |
QS quantify sufficient to produce total mass of 1.0 g, N/A not applicable
aValues following reconstitution with water for injection
Development Process
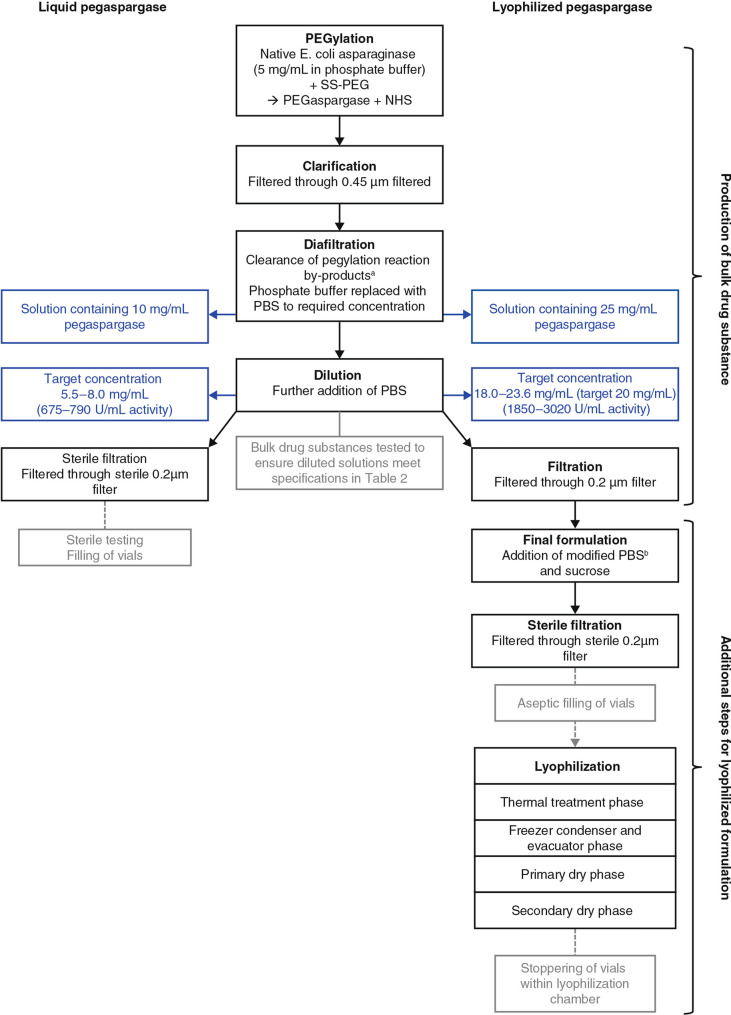
The development process for the bulk drug substance, for both formulations, comprises the following stages: pegylation, clarification, diafiltration, dilution and filtration/sterile filtration (Fig. 1). Initially, l-asparaginase solution is prepared in phosphate buffer at a concentration of 5 mg/mL. In the pegylation step, the native enzyme is chemically modified by reaction of monomethoxypolyethylene glycol succinimidyl succinate (SS–PEG) with primary amines on the protein, producing a pegylated protein with N-hydroxysuccinimide (NHS) as a by-product. The impurities, NHS and any unbound PEG, are removed during the diafiltration step, in which the solution is passed through a membrane with a nominal molecular weight cut-off of 100 kDa, allowing impurities through while retaining the pegylated protein product.
Fig. 1.
Development process for liquid and lyophilized pegaspargase. Black boxes represent core process steps; grey boxes show intermediate steps and tests; blue boxes highlight different pegaspargase concentrations in drug substance for liquid and lyophilized pegaspargase. a Free PEG, 10K PEG, NHS. b Dibasic and monobasic sodium phosphate and sodium chloride at half the concentrations of PBS used for production of bulk drug substance/liquid pegaspargase. NHS N-hydroxysuccinimide, PBS phosphate buffered saline, SS–PEG monomethoxypolyethylene glycol succinimidyl succinate
Modification of the liquid pegaspargase development process leads to production of a concentrated solution for lyophilization, which allows for final formulation with sucrose to the correct concentration. This modification occurs during the diafiltration step, during which the pegaspargase bulk solution for lyophilization is concentrated to a protein level (20 mg/mL) three times higher than in the liquid form (6.5 mg/mL). Following dilution, there is a filtration step and plastic bag fill for the lyophilization bulk solution (with a hold time of ≤ 60 days before lyophilized product manufacture, determined by the time required for transport to another facility for downstream processing) compared with a direct stainless steel tank fill for the liquid product (hold time of ≤ 72 h before liquid formulation manufacture). Specifications for the drug substance produced for each formulation are shown in Table 2.
Table 2.
Batch analysis and stability testing specifications for concentrated drug substance of lyophilized pegaspargase
| Test | Acceptance criteria | Lots of lyophilized pegaspargase | ||||||
|---|---|---|---|---|---|---|---|---|
| Lot 1 | Lot 2 | Lot 3 | ||||||
| Drug substance for liquid pegaspargase | Drug substance for lyophilized pegaspargasea | Initial test | Week 12 | Initial test | Week 12 | Initial test | Week 12 | |
| Protein concentration | 5.5–8.0 mg/mL | 18.0–23.6 mg/mL (target 20.0 mg/mL) | 18.4 | 18.5 | 20.9 | 21.3 | 19.7b | 20.1 |
| Potency (activity) | 675–790 U/mL | 1850–3020 U/mL | 2091 | 2290 | 2305 | 2538 | 2360b | 2426 |
| Appearance | Colourless solution | Colourless solution | Complies | Complies | Complies | Complies | Complies | Complies |
| Clarity | Clear, no visible particles | Clear, no visible particles | Complies | Complies | Complies | Complies | Complies | Complies |
| pH | 7.2–7.4 | 7.2–7.4 | 7.2 | 7.3 | 7.2 | 7.3 | 7.2 | 7.2 |
| Specific activity | ≥ 85 U/mg of protein | ≥ 85 U/mg of protein | 114 | 124 | 110 | 119 | 116 | 128 |
| Purity by GF-HPLC | ≥ 95% active components | ≥ 95% active components | 98 | 98 | 98 | 97 | 98 | 98 |
| ≤ 5% aggregates | ≤ 5% aggregates | 1 | 0 | 5 | 3 | 5 | 4 | |
| Free 10K PEG by RP-HPLC | ≤ 0.2 mg/mL | ≤ 0.6 mg/mL | < 0.1 | < 0.1 | < 0.1 | 0.3 | < 0.1 | 0.5 |
| Total free PEG by RP-HPLC | ≤ 2.0 mg/mL | ≤ 6.0 mg/mL | 0.7 | 5.0 | < 0.1 | 4.2 | < 0.1 | 4.5 |
| NHS | ≤ 1.3 ppm | ≤ 4.0 ppm | 0.7 | 0.6 | NT | 2.3 | 1.9 | 2.8 |
| Modification by TNBS | 69–82 mol PEG per mol protein | 69–82 mol PEG per mol protein | 73 | 76 | NT | 73 | 76 | 76 |
| Endotoxin | ≤ 35 EU/mL | ≤ 35 EU/mL | < 5 | NT | < 5 | NT | < 5 | NT |
| Bioburden | N/A | ≤ 2 CFU/20 mL | 0 | NT | 0 | NT | 0 | NT |
| Sterilityc | Pass USP | Pass USP | Complies | NT | NT | NT | NT | NT |
CFU colony forming units, EU endotoxin units, GF-HPLC gel filtration high-performance liquid chromatography, mol mole, N/A not applicable, NHS N-hydroxysuccinimide, PEG polyethylene glycol, ppm parts per million, RP-HPLC reversed-phase high-performance liquid chromatography, TNBS 2,4,6-trinitrobenzene sulfonic acid, NT not tested, USP United States Pharmacopoeia
aParameters that differ for lyophilized versus liquid pegaspargase drug substance are shown in italics
bValues taken from testing of samples post-diafiltration/pre-dilution
cSterility testing performed after filtration (liquid)/bioburden reduction (lyophilized)
As well as threefold higher protein concentration and enzyme activity in the bulk drug substance for lyophilized pegaspargase, threefold higher levels of impurities (free 10K PEG, total free PEG [the sum of 5K and 10K PEG] and NHS) are permitted in the more concentrated drug substance. In the drug product, prefill target weight for the lyophilized cake is 2.5 g, which is reconstituted with 5.2 mL of sterile water for injections. This restores impurities to the concentrations permitted in liquid pegaspargase following reconstitution as a solution for administration.
For liquid pegaspargase, the final step in the development process is sterile filtration—the drug substance is filtered through a sterile 0.2 μm filter into a jacketed stainless steel vessel and stored at 2–8 °C for a maximum of 72 h before it is aliquoted into sterile vials. For lyophilized pegaspargase, the concentrated drug substance is filtered through a 0.2 μm filter into a pre-sterilized 20 L bulk solution bag, in which it is stored at 2–8 °C for a maximum of 60 days, prior to the final formulation, sterile filtration and lyophilization process steps (Fig. 1). Samples were monitored at 0, 2, 4, 6, 8 and 12 weeks to assess the stability of pegaspargase during refrigerated storage as liquid concentrated bulk drug substance.
The final formulation process for lyophilized pegaspargase involves addition of a buffer containing sucrose as a lyoprotectant/stabilizing agent, plus dibasic sodium phosphate, monobasic sodium phosphate and sodium chloride (all buffer components apart from sucrose are present in the standard phosphate buffered saline [PBS] used for liquid pegaspargase but in different concentrations) (Table 1). The resulting solution contains pegaspargase at two times the concentration of liquid pegaspargase, as the higher concentration (lower water content) reduces the time required for freeze/drying cycles during the lyophilization process. This solution undergoes sterile filtration, after which vials are filled in aseptic conditions prior to lyophilization.
The lyophilization process comprises a thermal treatment phase and a freeze condenser and evacuator phase. On completion of the lyophilization process, the evacuated lyophilization chamber is backfilled with nitrogen gas and the vials are stoppered and crimp sealed. Lyophilization process cycle parameters are shown in Supplementary Table 1.
Comparability Assessment
A series of comparability tests were performed on samples of liquid pegaspargase and reconstituted lyophilized pegaspargase. Prior to testing, vials of lyophilized pegaspargase were reconstituted with a 21-gauge needle by dispensing 5.2 mL of sterile water for injection into each vial and mixing for 5 min or until particles of lyophilized cake were no longer visible by visual inspection at approximately 50 rotations per minute (rpm) on a roller mixer. Comparability tests were conducted on three lots each of liquid and lyophilized pegaspargase, to ensure that consistent results were replicated and permit statistical analysis.
Assessment of Physiochemical Properties and Critical Quality Attributes
Physicochemical properties and critical quality attributes of each lot were assessed to determine if batches of liquid pegaspargase and reconstituted lyophilized pegaspargase met predefined acceptance criteria (as detailed in Table 3). Acceptance criteria for the liquid product, including control limits for appearance, pH, protein concentration, activity, free PEG, etc. were established on the basis of a large number of historical lots of liquid pegaspargase, and were similar to the specifications for bulk drug substance (as detailed in Table 2). Similarly, acceptance criteria suitable for a lyophilized product were established on the basis of those for liquid pegaspargase to assess and determine the suitability of such a product based on physicochemical properties and critical quality attributes.
Table 3.
Assessment of physicochemical properties and critical quality attributes
| Test | Acceptance criteria | Liquid pegaspargasea | Reconstituted lyophilized pegaspargasea |
|---|---|---|---|
| Appearance | Colourless solutionb | Complies | Complies |
| Clarity | Clear, no visible particles | Complies | Complies |
| Fill volume | To deliver 5.0 mL | 5.1–5.2 mL | 5.2–5.5 mL |
| pH | 7.2–7.4 | 7.2–7.3 | 7.4 |
| Protein concentration | 4.5–8.5 mg/mL | 6.2–7.3 mg/mL | 6.5–7.1 mg/mL |
| Potency (activity) | 600–900 U/mL | 737–778 U/mL | 718–805 U/mL |
| Specific activity | ≥ 85 U/mg protein | 101–125 U/mg protein | 111–114 U/mg protein |
| Purity by GF-HPLC | |||
| Active components | ≥ 95% | 98–99% | 97% |
| Aggregates | ≤ 8% | 1–4% | 1–5% |
| Free 10K PEG | ≤ 0.2 mg/mL | < 0.07 mg/mL | 0.1–0.2 mg/mL |
| Total free PEG | ≤ 2.0 mg/mL | 0.2–0.6 mg/mL | 0.5–1.3 mg/mL |
| NHS | ≤ 2.0 ppm | 0.2–0.6 ppm | 0.2–1.0 ppm |
| Modification by TNBS | 69–82 mol PEG/mole protein | 73–75 mol PEG/mole protein | 75–77 mol PEG/mole protein |
| Particulate matter | Per container | ||
| ≥ 2 µm | ≤ 27,000 particles | 1961–3204 | 530–5834 |
| ≥ 10 µm | ≤ 6000 particles | 13–46 | 12–116 |
| ≥ 25 µm | ≤ 600 particles | 0 | 1–2 |
| Sterility | Pass USP sterility test | Complies | Compliesc |
| General safety | Pass USP tests in guinea pigs and mice | Conforms | Conformsc |
| Endotoxin by LAL | ≤ 35 EU/mL | < 15 EU/mL | < 4 EU/mL |
| Content uniformity | – | N/A | Complies |
| Reconstitution time | ≤ 3 min | N/A | < 1 min |
| Water (by KF) | ≤ 3.0% | N/A | 0.1–0.2% |
GF-HPLC gel filtration high-performance liquid chromatography, KF Karl Fischer titration, LAL Limulus amebocyte lysate, N/A not applicable, NHS N-hydroxysuccinimide, PEG polyethylene glycol, ppm parts per million, TNBS 2,4,6-trinitrobenzene sulfonic acid, USP United States Pharmacopeia Convention
aRange of values for three lots tested
bLyophilized product has appearance of white to off-white cake prior to reconsitution
cSterility and general safety tests not performed for one of the three lots tested
Characterization Tests
A series of tests was performed to characterize higher-order protein structure and other aspects of liquid and lyophilized pegaspargase formulations. In most cases, characterization tests were conducted side-by-side on paired lots of liquid and lyophilized pegaspargase to permit direct comparison. Tests included the following: characterization of molecular mass, using matrix-assisted laser desorption ionization time-of-flight (MALDI-TOF); extent of pegylation, measured using size-exclusion chromatography with multi-angle static light scattering (SEC-MALS) and high-performance liquid chromatography–mass spectrometry (HPLC–MS); purity, assessed by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) profiles; calorimetric enthalpy and transition peak midpoint (melting temperature), assessed by differential scanning calorimetry (DSC); and secondary and tertiary protein structure motifs and thermal unfolding characteristics, investigated by performing circular dichroism (CD) spectroscopy at various wavelengths. Further details of the analytical methods used in the characterization tests are detailed in the Supplementary Methods section.
In vitro enzyme kinetics were also evaluated. Pegaspargase was added to asparagine, diluted in buffer at pH 7.3 to a range of concentrations (5–250 μM). After addition of the derivatization reagent, and a second dilution step, the samples were incubated at 37 °C for 15 min to allow the enzymatic reaction to proceed. The released aspartic acid was derivatized before measurement by reversed-phase HPLC (RP-HPLC). The results obtained from this range of concentrations were used to determine enzyme kinetic parameters including Michaelis constant catalytic rate constant and maximum velocity.
Forced Degradation Studies
Degradation profiles for each formulation were determined in forced degradation studies in order to evaluate the comparative stress stability behavior of each pegaspargase formulation. The effect of heat stress was evaluated in samples stored at 40 ± 2 °C (in 75 ± 5% relative humidity [RH]) for 6 months. The effects of changes in pH were tested by applying acid/base stress sufficient to induce 5–25% degradation. For acid stress, 200 µL 0.8 M HCl or 200 µL 0.4 M HCl were added to 20 mL liquid or reconstituted lyophilized pegaspargase, respectively, and samples were incubated at 37 °C for 90 min. For base stress, 200 µL 0.8 M NaOH or 200 µL 0.4 M NaOH were added to 20 mL liquid or reconstituted lyophilized pegaspargase, respectively, and samples were incubated at 37 °C for 20 min. The concentration of HCl and NaOH used in the stress tests were different for the liquid and reconstituted lyophilized pegaspargase because of the constitution of the different formulations. As the lyophilized cake is unstable at commercial liquid pegaspargase concentrations, the filled product for lyophilization is approximately double that of commercial pegaspargase, resulting in reduced concentration of stabilizing salts upon reconstitution. Therefore, half the concentrations of HCl and NaOH are required for the same pH to be reached because only half the stabilizing buffer is present in reconstituted lyophilized pegaspargase. Oxidative stress was applied by adding 200 µL 30% H2O2 to 20 mL pegaspargase and incubating at 40 °C for 30 min. Agitation stress was applied to inverted vials shaken at 120 rpm, using an orbital shaker, for 8 h at room temperature. Light (photo) stress involved exposure to 1.2 million Lux h cool white or 200 W h ultraviolet (UV) light. Critical quality attributes including purity, potency, aggregates and total free PEG were tested at various time points during or after exposure to stresses.
Stability Tests
Long-term stability of lyophilized pegaspargase was assessed for at least 3 years for samples stored under refrigerated (5 ± 3 °C) and accelerated (25 ± 2 °C; 60 ± 5% RH) conditions. Purity, potency and aggregates were measured in reconstituted samples (every 3 months for the first year and every 6 months thereafter). One method was used to determine the purity of active non-aggregated proteins, measured by gel-filtration HPLC (GF-HPLC), and the number of aggregated non-active proteins. Activity was measured as an indicator of potency by RP-HPLC. This is assessed by incubating the sample with l-asparagine to generate l-aspartic acid by enzymatic reaction. The amount of released l-aspartic acid is analysed by RP-HPLC fitted with a UV detector set at 265 nm. This method is also used to determine the identity of the drug substance. Residual water was measured by Karl Fischer titration in samples of lyophilized cake at the same time points.
Results
Pre-Lyophilization Tests on Bulk Drug Substance
In this comparability study, all batches of bulk drug substance (concentrated liquid) used in the development process of lyophilized pegaspargase met the required pre-specified criteria detailed in Table 2, including protein concentration, potency, appearance, pH and purity. Stability testing showed that storage of bulk drug substance for lyophilized pegaspargase at 2–8 °C for up to 12 weeks prior to lyophilization met the acceptance criteria approved for liquid pegaspargase (Table 2 and additional time point data shown in Supplementary Tables 2–4). No significant changes in purity were detected, although higher potency, total free PEG and 10K PEG were observed as a result of expected depegylation. However, these elevated results were still within specification limits (Table 2).
Post-Lyophilization Tests on Lyophilized and Reconstituted Lyophilized Pegaspargase
Tests investigating the physiochemical properties and critical quality attributes of each formulation showed no significant differences between liquid and lyophilized pegaspargase. All parameters, including measures of potency and purity, were within acceptance criteria limits (Table 3).
Characterization tests indicated no significant differences in molecular weight, extent and site of pegylation, and enzyme kinetics. The results of these methods met predefined acceptance criteria (Table 4). However, results of DSC and CD spectroscopy demonstrated that lyophilized pegaspargase has a trend towards lower calorimetric enthalpy required for the transition to an unfolded state, relative to liquid pegaspargase (Table 4 footnotes).
Table 4.
Characterization tests: summary of similarities/differences between liquid and lyophilized pegaspargase
| Characteristic | Test | Acceptance criteriaa | Observed differencesb |
|---|---|---|---|
| Molecular mass | MALDI-TOF | ≤ 3 kDa per monomer | 0.4–0.8 kDa |
| Extent of pegylation (1) | SEC-MALS | ≤ 3 mol PEG per mole protein | 1.7–2.4 mol |
| Extent of pegylation (2) | HPLC–MSc | ≤ 10% difference in pegylation at each potential binding site | Comparable, despite differences in peptides containing multiple lysine groupsd |
| Calorimetry | DSC |
≤ 2 °C for Tm ≤ 15% for calorimetric enthalpy |
Tm 0.06–0.54 °C Calorimetric enthalpy 4–19%e |
| Secondary structure |
CD spectroscopy: Far-UV scan (190–250 nm) |
≤ 10% difference in each secondary structure motiff |
≤ 2% difference in proportions of any motif |
| Tertiary structure |
CD spectroscopy: Near-UV scan (250–350 nm) |
No obvious differences when CD spectra overlaid | No obvious differences in CD spectroscopy scans |
| Thermal unfolding | CD spectroscopy at fixed wavelength of 220 nm, over a range of temperatures | No obvious differences when CD spectra overlaid |
No significant differences in CD spectroscopy scansg Consistent melting temperatures |
| Purity | SDS-PAGE | Similar banding profiles; no additional bands (representing new impurities) | Similar banding profiles with no new impurities |
| Enzyme kinetics |
Km: 10–20 µM Vmax: 0.10–0.15 µmol/min/µg Kcat: 58–88 s−1 |
Within defined rangesh |
CD circular dichroism, DSC differential scanning calorimetry, HPLC–MS high-performance liquid chromatography–mass spectrometry, Kcat catalytic rate constant, Km Michaelis constant, MALDI-TOF matrix assisted laser desorption ionization time-of-flight, PEG polyethylene glycol, SDS-PAGE sodium dodecyl sulfate polyacrylamide gel electrophoresis, SEC-MALS size-exclusion chromatography with multi-angle static light scattering, Tm transition peak midpoint (melting temperature), UV ultraviolet, Vmax maximum velocity
aMaximum difference between lyophilized and liquid pegaspargase to conclude equivalence between formulations
bRange for differences between paired lots of lyophilized and liquid pegaspargase
cl-Asparaginase was reacted with succinic anhydride under similar conditions to those used for pegylation, resulting in a conformation similar to that found during the manufacturing pegylation step. Therefore, the exposed lysine residues on the protein surface were succinylated. The samples were then denatured using a high concentration of urea to expose lysine residues that had been buried inside the three-dimensional structure of the protein. These had been protected from initial succinylation, and also from pegylation during the development process. A second derivatization step using isotopically labelled d4-succinic anhydride led to the modification of the remaining free lysine residues. Thus, all lysine residues were succinylated: the exposed lysine residues were modified with unlabelled succinate and the buried lysine residues were modified with isotopically labelled succinate, consequently distinguishable by the 4-Da mass difference of the label using HPLC-MS
dDifferences in extent of pegylation (> 10%) were observed for some peptides containing multiple lysine groups. However, these differences were attributed to uncertainties in precision of the complex method, and do not represent significant differences in the extent of pegylation, since there were no corresponding differences in molecular mass or extent of pegylation in other characterization tests
eLyophilized pegaspargase trended toward lower enthalpy required for the transition to an unfolded state relative to liquid pegaspargase, exceeding the acceptance criteria of 15% difference in one case (from three pairs of lots). This appeared to relate to higher enthalpy of liquid pegaspargase in that pair of lots, while the lyophilized pegaspargase lot had consistent enthalpy with other lyophilized lots
fα-helix, anti-parallel and parallel β-sheet, β-turn and random coil
gSlight shift in thermal melt curves, consistent with slight differences in Tm on DCS
hLots were analysed individually and not paired for side-by-side analysis of enzyme kinetics. Results for each lot were within the defined ranges; differences between the means for three lots each of the two formulations (15% for Km, 8.4% for Vmax and 7.2% for Kcat) were within the expected intra-assay variability
The degradation profile of lyophilized pegaspargase under conditions of heat stress was decelerated compared with liquid pegaspargase. Deleterious changes in purity, potency, aggregates and free PEG were observed to be more rapid in liquid than lyophilized pegaspargase (Fig. 2). Additional stress conditions tested (photo, agitation and acidic, basic and oxidative stress) showed no significant differences in degradation of liquid and lyophilized pegaspargase (Supplementary Tables 5–7).
Fig. 2.
Heat stress profiles for liquid (Lot A) and lyophilized (Lot 1) pegaspargase: purity (a), potency (b), aggregates (c) and total free PEG (d) of/in liquid and lyophilized pegaspargase stored at 40 °C for 6 months. a Determined by GF-HPLC. b Determined by RP-HPLC. Results from a representative lot of liquid and lyophilized pegaspargase are presented. Each lot was tested once at each time point. Liquid and lyophilized lots were tested using validated methods. GF-HPLC gel filtration high-performance liquid chromatography, LSL lower specification limit, PEG polyethylene glycol, RP-HPLC reversed-phase high-performance liquid chromatography, USL upper specification limit
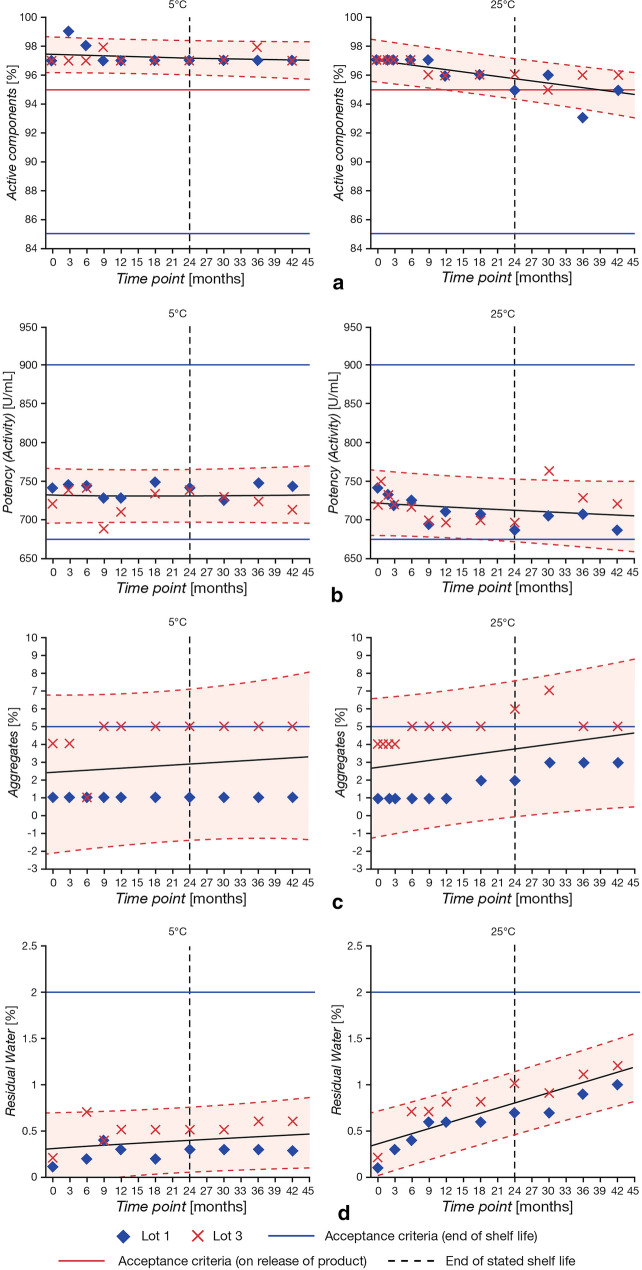
Stability tests showed that purity, potency, aggregates and residual water in lyophilized cake remained within acceptance criteria limits for samples stored at both 5 °C and at 25 °C for up to 3 years. Refrigerated conditions were more favorable for maintained stability of lyophilized pegaspargase. Stability profiles over 45 months showed an expected gradual decline in purity that was slower for samples stored at 5 °C than 25 °C (Fig. 3a). Potency (activity) remained constant beyond 3 years in samples at 5 °C, while the decline in purity at 25 °C corresponded with a decrease in specific activity at this temperature (Fig. 3b). Aggregates remained at a constantly low level in samples at 5 °C, whereas increased aggregation was observed in samples at 25 °C (Fig. 3c). There was minimal increase in residual water in the lyophilized cake at 5 °C, while increased residual water was observed at 25 °C (RH 60%) (Fig. 3d). On the basis of these long-term stability results, the shelf life of lyophilized pegaspargase was set as 24 months at 5 ± 3 °C.
Fig. 3.
Stability of lyophilized pegaspargase stored at 5 °C and 25 °C (60% RH) over 45 months: trend analyses for purity (a), potency (b), aggregates (c) and residual water (d) substance for lyophilized pegaspargase. Lots stored in an inverted orientation. Results from a representative lot of liquid and lyophilized pegaspargase are presented. Each lot was tested once at each time point. Liquid and lyophilized lots were tested using validated methods. Acceptance criteria for purity (% active components) are lower at end of shelf life than on release; acceptance criteria for other quality attributes (aggregates, potency, residual water) are the same at release and end of shelf life. RH relative humidity
Discussion
The lyophilization process described here produced a pegaspargase formulation with comparable physicochemical properties, purity and potency profiles to those of liquid pegaspargase. Ongoing process validation showed no significant differences in quality attributes between pre- and post-lyophilized drug product, indicating that the lyophilization process used had no negative impact on product quality. Characterization tests showed that lyophilized pegaspargase also remained comparable to the liquid formulation, in terms of structure, extent and site of pegylation, and enzyme kinetics. DSC and CD spectroscopic investigation of thermal unfolding revealed slight differences in enthalpy and melting point for the two formulations, which may relate to differences in salt and/or sucrose concentrations in excipients. Forced degradation tests demonstrated improved stability of lyophilized compared with liquid pegaspargase under conditions of heat stress.
The primary aim of developing the lyophilized formulation of pegaspargase was to improve stability of the pegylated protein, allowing for a prolonged shelf life. From the long-term stability tests performed, we believe that this goal has been achieved. These tests demonstrated that lyophilized pegaspargase met acceptance criteria for various quality attributes, including purity, potency and protein aggregation, during a 45-month storage period under refrigerated conditions (2–8 °C). Samples stored at 25 °C (RH 60%) had a greater tendency to absorb moisture, resulting in residual water in the lyophilized cake and potentially increasing the likelihood of cake collapse. In addition, these samples demonstrated a trend for slightly decreasing values in purity and potency and increasing aggregation over time. Active asparaginase is a tetramer, which becomes inactive when it disassembles into dimers or monomers over time and at higher temperatures, thereby resulting in a gradual decrease in potency [9]. It is therefore recommended that lyophilized pegaspargase should be refrigerated at 2–8 °C during long-term storage for optimal stability and maintenance of protein activity at the intended potency.
In light of the improved stability of lyophilized pegaspargase, while maintaining comparability with the liquid formulation in terms of physicochemical properties, purity and potency, the European Medicines Agency granted approval of lyophilized pegaspargase following a positive opinion from the Committee for Medicinal Products for Human Use [23]. On the basis of our results, the shelf life of lyophilized pegaspargase was set as 24 months at 2–8 °C [23]. This is a considerable improvement on the 8-month shelf life of liquid pegaspargase. As long-term stability has been demonstrated for test lots of lyophilized pegaspargase within a 45-month period, and tests are ongoing, it may be possible to extend this shelf life further in the future.
Lyophilization has been used to improve stability of other recombinant protein therapeutics, e.g. IgG1 monoclonal antibodies (mAbs) [25, 28, 29], with potential applications including improved stability during shipping [29]. Tests of lyophilized IgG1 mAbs under simulated shipping conditions (agitation stress) suggested that lyophilized formulations can remain vulnerable to physical degradation on shaking [29]. However, we found no instability upon agitation for liquid or lyophilized pegaspargase.
One of the limitations of this study is that it did not investigate the stability of lyophilized pegaspargase following reconstitution. In a separate study, chemical and physical in-use stability has been demonstrated for 24 h for reconstituted solutions stored below 25 °C [23]. Stability, in terms of enzyme activity based on catalysis of l-asparagine to l-aspartic acid and ammonia, has been reported for up to 7 days following reconstitution of a lyophilized formulation of native E. coli asparaginase in different parenteral solutions refrigerated at 2–8 °C [30]. It is important that sterility is maintained, so unless precautions are taken to preclude any risk of microbial contamination on reconstitution, it is recommended that the solution should be used immediately [23]. Another limitation is that each lot of liquid and lyophilized pegaspargase was tested once at each time point for each comparability assessment, using validated test methods. Characterization of liquid and lyophilized pegaspargase lots was performed in a side-by-side fashion, however, to minimize variability between assays. Finally, beyond demonstrating an effective example of stabilization, it could be argued that the information reported here may not be of broad applicability to the community, particularly without associated preclinical or clinical data for the lyophilized formulation.
Despite these limitations, the prolonged shelf life of lyophilized pegaspargase is expected to improve product supply management in pharmacy formulary settings through enabling greater flexibility and longer-term planning. The prolonged shelf life may also lead to improvements in access to pegaspargase for patients with ALL in countries where liquid pegaspargase is currently not available.
Conclusion
Lyophilization has been demonstrated to improve the stability of pegaspargase without altering other physicochemical properties, permitting a prolonged shelf life of 24 months at 2–8 °C.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Acknowledgements
This study was conducted by Sigma-Tau Pharmasource (now Exelead, Indianapolis, IN, US), reported by Sigma-Tau Pharmaceuticals (now Leadiant Biosciences, Inc Gaithersburg, MD, US), and funded by the Sponsor, Baxalta (now part of Takeda).
Funding
Publication costs, including article processing charges and the Open Access fee were funded by Servier Affaires Médicales, Suresnes, France. All authors had full access to all of the data in this study and take complete responsibility for the integrity of the data and accuracy of the data analysis.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Medical Writing, Editorial and Additional Assistance
David Bollag (Servier) and Kim Brown, Alicia Jeannotte, Susan Wieting, Bruce Tangarone and Anna Wijatyk (Shire employees at the time of the study) provided valuable advice and assistance during the preparation of this manuscript. Chris Lockey, a former employee of Baxalta (now part of Takeda) and Sigma-Tau Pharmasource (now Exelead, Indianapolis, IN, US), was instrumental in leading the development process detailed in this manuscript. Medical writing support was provided by Laura McMahon of Physicians World Europe GmbH (Mannheim, Germany), and funded by Shire, Zurich, Switzerland (now part of Takeda) and Servier Affaires Médicales, Suresnes, France.
Disclosures
Alexander Faschinger was an employee of Shire (now part of Takeda) at the time of the reported analyses and is currently an employee of Servier; Nicole Sessler is an employee of Baxalta Innovations Gmbh (now part of Takeda).
Compliance with Ethics Guidelines
This article does not contain any studies with human participants or animals.
Data Availability
Data are the proprietary property of Shire and Servier and access may be requested by contacting Servier’s legal representative.
Footnotes
Enhanced Digital Features
To view enhanced digital features for this article go to 10.6084/m9.figshare.8101064.
References
- 1.Alvarnas JC, Brown PA, Aoun P, et al. Acute lymphoblastic leukemia, version 2.2015. J Natl Compr Cancer Netw. 2015;13(10):1240–1279. doi: 10.6004/jnccn.2015.0153. [DOI] [PubMed] [Google Scholar]
- 2.Hoelzer D, Bassan R, Dombret H, et al. Acute lymphoblastic leukaemia in adult patients: ESMO clinical practice guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2016;27(suppl 5):v69–v82. doi: 10.1093/annonc/mdw025. [DOI] [PubMed] [Google Scholar]
- 3.Piatkowska-Jakubas B, Krawczyk-Kulis M, Giebel S, et al. Use of l-asparaginase in acute lymphoblastic leukemia: recommendations of the Polish Adult Leukemia Group. Pol Arch Med Wewn. 2008;118(11):664–669. [PubMed] [Google Scholar]
- 4.Pieters R, Hunger SP, Boos J, et al. l-Asparaginase treatment in acute lymphoblastic leukemia: a focus on Erwinia asparaginase. Cancer. 2011;117(2):238–249. doi: 10.1002/cncr.25489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Jazz Pharmaceuticals Inc. Erwinase® Highlights of Prescribing Information 2016. https://erwinaze.com/ERWINAZEPI.pdf. Accessed Dec 2017.
- 6.Barnes WR, Dorn GL, Vela GR. Effect of culture conditions on synthesis of l-asparaginase by Escherichia coli A-1. Appl Environ Microbiol. 1977;33(2):257–261. doi: 10.1128/aem.33.2.257-261.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jennings MP, Beacham IR. Co-dependent positive regulation of the ansB promoter of Escherichia coli by CRP and the FNR protein: a molecular analysis. Mol Microbiol. 1993;9(1):155–164. doi: 10.1111/j.1365-2958.1993.tb01677.x. [DOI] [PubMed] [Google Scholar]
- 8.Roberts J, Burson G, Hill JM. New procedures for purification of l-asparaginase with high yield from Escherichia coli. J Bacteriol. 1968;95(6):2117–2123. doi: 10.1128/jb.95.6.2117-2123.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wriston JC, Jr, Yellin TO. l-Asparaginase: a review. Adv Enzymol Relat Areas Mol Biol. 1973;39:185–248. doi: 10.1002/9780470122846.ch3. [DOI] [PubMed] [Google Scholar]
- 10.Netrval J. Effect of organic and amino acids on l-asparaginase production by Escherichia coli. Arch Mikrobiol. 1973;92(4):345–352. doi: 10.1007/BF00409287. [DOI] [PubMed] [Google Scholar]
- 11.Dinndorf PA, Gootenberg J, Cohen MH, Keegan P, Pazdur R. FDA drug approval summary: pegaspargase (Oncaspar) for the first-line treatment of children with acute lymphoblastic leukemia (ALL) Oncologist. 2007;12(8):991–998. doi: 10.1634/theoncologist.12-8-991. [DOI] [PubMed] [Google Scholar]
- 12.Borghorst S, Hempel G, Poppenborg S, Franke D, Konig T, Baumgart J. Comparative pharmacokinetic/pharmacodynamic characterisation of a new pegylated recombinant E. coli l-asparaginase preparation (MC0609) in Beagle dog. Cancer Chemother Pharmacol. 2014;74(2):367–378. doi: 10.1007/s00280-014-2506-9. [DOI] [PubMed] [Google Scholar]
- 13.Rau RE, Dreyer Z, Choi MR, et al. Outcome of pediatric patients with acute lymphoblastic leukemia/lymphoblastic lymphoma with hypersensitivity to pegaspargase treated with PEGylated Erwinia asparaginase, pegcrisantaspase: a report from the Children’s Oncology Group. Pediatr Blood Cancer. 2018;65(3):e26873. [DOI] [PMC free article] [PubMed]
- 14.Chien WW, Allas S, Rachinel N, et al. Pharmacology, immunogenicity, and efficacy of a novel pegylated recombinant Erwinia chrysanthemi-derived l-asparaginase. Invest New Drugs. 2014;32(5):795–805. doi: 10.1007/s10637-014-0102-9. [DOI] [PubMed] [Google Scholar]
- 15.Rizzari C, Conter V, Stary J, Colombini A, Moericke A, Schrappe M. Optimizing asparaginase therapy for acute lymphoblastic leukemia. Curr Opin Oncol. 2013;25(Suppl 1):S1–S9. doi: 10.1097/CCO.0b013e32835d7d85. [DOI] [PubMed] [Google Scholar]
- 16.Asselin BL, Whitin JC, Coppola DJ, Rupp IP, Sallan SE, Cohen HJ. Comparative pharmacokinetic studies of three asparaginase preparations. J Clin Oncol. 1993;11(9):1780–1786. doi: 10.1200/JCO.1993.11.9.1780. [DOI] [PubMed] [Google Scholar]
- 17.Park YK, Abuchowski A, Davis S, Davis F. Pharmacology of Escherichia coli-l-asparaginase polyethylene glycol adduct. Anticancer Res. 1981;1(6):373–376. [PubMed] [Google Scholar]
- 18.Patel N, Krishnan S, Offman MN, et al. A dyad of lymphoblastic lysosomal cysteine proteases degrades the antileukemic drug l-asparaginase. J Clin Invest. 2009;119(7):1964–1973. doi: 10.1172/JCI37977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Avramis VI, Sencer S, Periclou AP, et al. A randomized comparison of native Escherichia coli asparaginase and polyethylene glycol conjugated asparaginase for treatment of children with newly diagnosed standard-risk acute lymphoblastic leukemia: a Children’s Cancer Group study. Blood. 2002;99(6):1986–1994. doi: 10.1182/blood.V99.6.1986. [DOI] [PubMed] [Google Scholar]
- 20.Würthwein G, Lanvers-Kaminsky C, Hempel G, et al. Population pharmacokinetics to model the time-varying clearance of the PEGylated asparaginase Oncaspar® in children with acute lymphoblastic leukemia. Eur J Drug Metab Pharmacokinet. 2017;42(6):955–963. doi: 10.1007/s13318-017-0410-5. [DOI] [PubMed] [Google Scholar]
- 21.Turecek PL, Bossard MJ, Schoetens F, Ivens IA. PEGylation of biopharmaceuticals: a review of chemistry and nonclinical safety information of approved drugs. J Pharm Sci. 2016;105(2):460–475. doi: 10.1016/j.xphs.2015.11.015. [DOI] [PubMed] [Google Scholar]
- 22.Dozier JK, Distefano MD. Site-specific PEGylation of therapeutic proteins. Int J Mol Sci. 2015;16(10):25831–25864. doi: 10.3390/ijms161025831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shire Pharmaceuticals. Oncaspar summary of product characteristics 2017. http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/003789/WC500200735.pdf. Accessed December 2017.
- 24.Remmele RL, Krishnan S, Callahan WJ. Development of stable lyophilized protein drug products. Curr Pharm Biotechnol. 2012;13(3):471–496. doi: 10.2174/138920112799361990. [DOI] [PubMed] [Google Scholar]
- 25.Davis JM, Zhang N, Payne RW, et al. Stability of lyophilized sucrose formulations of an IgG1: subvisible particle formation. Pharm Dev Technol. 2013;18(4):883–896. doi: 10.3109/10837450.2012.705295. [DOI] [PubMed] [Google Scholar]
- 26.Carpenter JF, Randolph TW, Jiskoot W, et al. Overlooking subvisible particles in therapeutic protein products: gaps that may compromise product quality. J Pharm Sci. 2009;98(4):1201–1205. doi: 10.1002/jps.21530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Braun A, Kwee L, Labow MA, Alsenz J. Protein aggregates seem to play a key role among the parameters influencing the antigenicity of interferon alpha (IFN-alpha) in normal and transgenic mice. Pharm Res. 1997;14(10):1472–1478. doi: 10.1023/A:1012193326789. [DOI] [PubMed] [Google Scholar]
- 28.Murphy BM, Zhang N, Payne RW, et al. Structure, stability, and mobility of a lyophilized IgG1 monoclonal antibody as determined using second-derivative infrared spectroscopy. J Pharm Sci. 2012;101(1):81–91. doi: 10.1002/jps.22753. [DOI] [PubMed] [Google Scholar]
- 29.Telikepalli S, Kumru OS, Kim JH, et al. Characterization of the physical stability of a lyophilized IgG1 mAb after accelerated shipping-like stress. J Pharm Sci. 2015;104(2):495–507. doi: 10.1002/jps.24242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Stecher AL, de Deus PM, Polikarpov I, Abrahão-Neto J. Stability of l-asparaginase: an enzyme used in leukemia treatment. Pharm Acta Helv. 1999;74(1):1–9. doi: 10.1016/S0031-6865(99)00009-6. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data are the proprietary property of Shire and Servier and access may be requested by contacting Servier’s legal representative.