Abstract
In adult songbirds the telencephalic song nucleus HVC and its efferent target RA undergo pronounced seasonal changes in morphology. In breeding birds, there are increases in HVC volume and total neuron number, and RA neuronal soma area compared to nonbreeding birds. At the end of breeding, HVC neurons die through caspase-dependent apoptosis, and RA neuron size decreases. Changes in HVC and RA are driven by seasonal changes in circulating testosterone (T) levels. Infusing T, or its metabolites 5α-dihydrotestosterone (DHT) and 17 β-estradiol (E2), intracerebrally into HVC (but not RA) protects HVC neurons from death, and RA neuron size, in nonbreeding birds. The phosphoinositide 3-kinase (PI3K)-Akt (a serine/threonine kinase)-mechanistic target of rapamycin (mTOR) signaling pathway is a point of convergence for neuroprotective effects of sex steroids and other trophic factors. We asked if mTOR activation is necessary for protective effect of hormones in HVC and RA of adult male Gambel’s white-crowned sparrows (Zonotrichia leucophrys gambelii). We transferred sparrows from breeding to nonbreeding hormonal and photoperiod conditions to induce regression of HVC neurons by cell death, and decrease of RA neuron size. We infused either DHT+E2, DHT+E2 plus the mTOR inhibitor rapamycin, or vehicle alone in HVC. Infusion of DHT+E2 protected both HVC and RA neurons. Co-infusion of rapamycin with DHT+E2, however, blocked the protective effect of hormones on HVC volume and neuron number, and RA neuron size. These results suggest that activation of mTOR is an essential downstream step in the neuroprotective cascade initiated by sex steroid hormones in the forebrain.
Keywords: neuroprotection, mechanistic target of rapamycin, rapamycin, songbird, birdsong, testosterone, estrogen, hormone, protein phosphorylation, signaling pathway, trophic support, trophic factors, neurotrophins
1. Introduction
Sex steroids such as testosterone (T) and its metabolites 17 β-estradiol (E2) and 5α-dihydrotestosterone (DHT) can protect neurons in adult brains from neurodegenerative insult, ischemia, and traumatic brain injury (Brann, Dhandapani, Wakade, Mahesh, & Khan, 2007; Cardona-Gómez, Mendez, DonCarlos, Azcoitia, & Garcia-Segura, 2001; Garcia-Segura, Azcoitia, & DonCarlos, 2001; Liu, Kelley, Herson, & Hurn, 2010; Pike, Carroll, Rosario, & Barron, 2009; Sohrabji, 2015). Steroids can exert neuroprotective effects by both genomic and nongenomic mechanisms (Marin, Guerra, Alonso, Ramirez, & Diaz, 2005; Prokai & Simpkins, 2007). Nongenomic mechanisms include activation of kinase signaling pathways (Honda et al., 2000; Quesada, Lee, & Micevych, 2008).
The contributions of kinase signaling cascades to hormonal neuroprotection, especially in vivo, are not fully understood. Seasonal plasticity of the telencephalic circuit that regulates the motor production of learned song in adult songbirds provides an excellent model for investigating nongenomic mechanisms of hormonal protection in the context of naturally occurring neurodegeneration (Figure 1a). In male Gambel’s white-crowned sparrows (Zonotrichia leucophrys gambelii) the number of neurons within the telencephalic song control nucleus HVC (proper name, Reiner et al., 2004) changes seasonally in response to changes in circulating levels of T (E. A. Brenowitz, 2008). Each breeding season, as T levels increase, HVC incorporates nearly 68,000 new neurons (Tramontin, Hartman, & Brenowitz, 2000), most of which project to the robust nucleus of the arcopallium (RA) (Scott & Lois, 2007; Tokarev, Boender, Claßen, & Scharff, 2016). RA neuronal soma area is larger in breeding than nonbreeding condition birds (E. A. Brenowitz, 2008). The increase in RA neuron size is mediated entirely by changes in transsynaptic trophic support of RA neurons from HVC neurons (Eliot A. Brenowitz, 2015; E. A. Brenowitz & Larson, 2015). As sparrows transition from breeding to nonbreeding conditions, T drops to basal levels and an equal number of HVC neurons die rapidly through apoptosis (C. K. Thompson, Bentley, & Brenowitz, 2007; C. K. Thompson & Brenowitz, 2008), and RA neuronal soma area decreases. The loss of HVC neurons can be reduced by infusing T or DHT+E2 locally into HVC (Meitzen, Moore, Lent, Brenowitz, & Perkel, 2007; C.K. Thompson & Brenowitz, 2010). The molecular mechanisms by which T and its metabolites exert their neuroprotective effect in HVC (and indirectly in RA), and whether nongenomic actions are involved, are largely unknown.
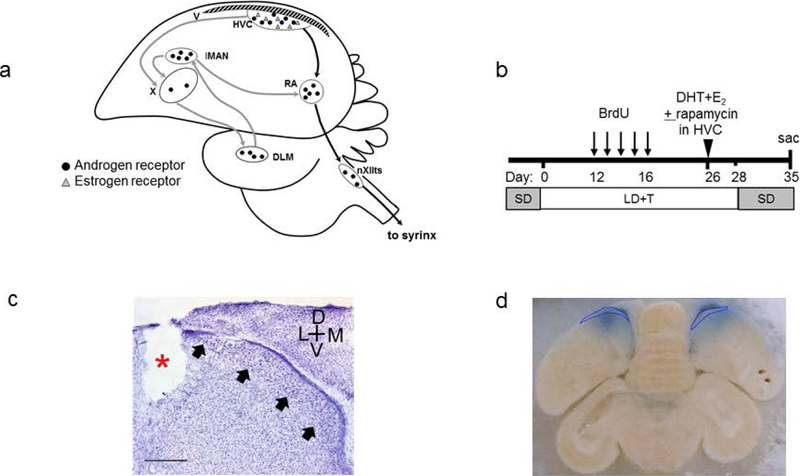
Figure 1.
(a) Schematic sagittal drawing of the songbird brain showing projections of major nuclei in the song system and the distribution of androgen and estrogen receptors. Abbreviations: DLM, medial portion of the dorsolateral nucleus of the medial thalamus; HVC, acronym used as proper name; LMAN, lateral portion of the magnocellular nucleus of the anterior nidopallium; RA, robust nucleus of the acropallium; V, lateral ventricle; X, Area X of the medial striatum; nXIIts, tracheosyringeal part of the hypoglossal nucleus. (b) Schematic timeline of the experimental procedures. SD, short-day photoperiod; LD + T, long-day photoperiod plus systemic testosterone (T) implant. Birds were infused in HVC with 5-α dihydrotestosterone (DHT) and 17-β estradiol (E2), vehicle alone, or DHT+E2+rapamycin, starting on day 26 and were killed on day 35. (c) A representative image of a Nissl stained section showing the borders of HVC (black arrows) and cannula track (red asterisk). Cross-hairs indicate orientation. Scale bar = 500 μm. (d) Coronal view of brain at level of HVC (outlined in blue), showing the spread of Evan’s blue dye, contained in infused solution of treatment agent, from tip of in-dwelling cannulae. Note that the blue dye diffused throughout full extent of HVC.
Sex steroids like testosterone activate phosphoinositide 3-kinase (PI3K) and Akt (a serine/threonine kinase) via phosphorylation (Quesada et al., 2008). Activation of the mechanistic target of rapamycin (mTOR) canonical pathway, in turn, is regulated by phosphorylated Akt (pAkt (serine 473)) (Martini, De Santis, Braccini, Gulluni, & Hirsch, 2014; Memmott & Dennis, 2009). The PI3K-Akt-mTOR signaling pathway is a point of convergence for the neuroprotective effects of steroid hormones, growth factors (e.g., vascular endothelial growth factor, VEGF), and neurotrophins (e.g., brain-derived neurotrophic factor, BDNF) (Chang, Poser, & Xia, 2004; Quesada & Micevych, 2008; Z. Zhang et al., 2010).
mTOR is an evolutionarily conserved serine-threonine kinase that is the catalytic subunit of the mTORC1 and mTORC2 complexes. mTORC1 and mTORC 2 are inhibited by chronic treatment with rapamycin (reviewed in Sabatini, 2017). mTOR and its downstream effectors, ribosomal protein S6K and the 40S subunit ribosomal protein S6, are present in cells of the songbird forebrain (Ahmadiantehrani & London, 2017). Ahmadiantehrani and London showed in male zebra finches (Taenopygia guttata) that 1) hearing novel song activated mTOR in cells of two forebrain auditory nuclei, as measured by immunoreactivity for phosphorylated S6, 2) infusion of rapamycin into the forebrain decreased mTOR activation as measured by pS6+ cells, and 3) infusion of rapamycin into the forebrain impaired song learning by juveniles (Ahmadiantehrani, Gores, & London, 2018; Ahmadiantehrani & London, 2017).
The proliferative and cell protective effects of steroid hormones in various organs and cell lines require activation of mTOR and are blocked by rapamycin. Rapamycin blocked the effect of estrogen on MCF-7 cell survival (Rodrik, Zheng, Harrow, Chen, & Foster, 2005). In uterine myocytes estrogen stimulated proliferation indirectly through induction of insulin-like growth factor 1 (IGF-1) in ovariectomized rats, and rapamycin blocked the proliferative effect of the hormone (Jaffer, Shynlova, & Lye, 2009). It is less well understood, however, whether rapamycin also blocks hormonal protection of neurons in the brain (Sohrabji, 2015).
We hypothesized that rapamycin might block the neuroprotective effects of sex steroids in the birdsong system. To test this hypothesis, we co-infused rapamycin with DHT and E2 in HVC in adult male white-crowned sparrows from breeding (high systemic T levels) to nonbreeding (basal systemic T) conditions. We found that rapamycin disrupted the neuroprotective effects of steroids in HVC and RA, suggesting the mTOR pathway plays a role in the protective effects of steroid hormones.
2. Materials and Methods
Animals
All animal procedures were performed in accordance with relevant institutional and national guidelines, and were approved by the Institutional Animal Care and Use Committee of the University of Washington. We captured 42 adult male Gambel’s white-crowned sparrows in eastern Washington during their post-breeding season migration. We housed the birds in indoor group aviaries for at least 12 weeks on a short day photoperiod (SD; 8 h light: 16 h dark), typical of their wintering grounds, before the start of the experiment. This housing ensured that the song system and reproductive system were fully regressed and sensitive to the stimulatory effects of T and long-day photoperiods (Tramontin et al., 2000). Food and water were available ad libitum.
Experimental design
At the beginning of the experiment (day 0) we transferred the birds overnight from SD to a long day photoperiod (LD; 20 h light:4 h dark) typical of their Alaskan breeding grounds (Figure 1b). We implanted each bird subcutaneously with a 12 mm Silastic capsule (inner diameter 1.47 mm × outer diameter 1.96 mm) filled with crystalline T (Steraloids, Newport, Rhode Island). Birds were housed individually in visual and auditory contact with other birds. We kept all the birds on LD+T for 28 days. This time period is sufficient to induce full growth of the song control system under these conditions (Tramontin et al., 2000). To induce the death of HVC neurons, we shifted birds to SD and removed the Silastic T implant on day 28, and killed them on day 35; seven days of basal T levels is sufficient for full regression of HVC to its nonbreeding size (Larson, Thatra, Lee, & Brenowitz, 2014; C. K. Thompson et al., 2007).
After 21–23 days of LD+T conditions, we implanted two cannulae bilaterally near HVC in each bird. We anesthetized the bird with isoflurane and placed it into a stereotaxic head holder. We made an incision in the scalp, removed the portion of the skull overlying the midsagittal sinus, and used the bifurcation of the sinus as the zero point. We lowered the cannulae 0.7 mm into the telencephalon just lateral to HVC (lateral: 3.1 mm, posterior: 0.0 mm). We fixed the cannulae to the skull with dental cement and attached both cannulae to the same osmotic pump with a “Y” connector (Plastics One, Roanoke, VA). The osmotic pumps (Alzet, Inc., model 1002, Cupertino, CA) were filled with avian saline. On Day 26, we changed the pumps to new ones containing 100 μl of one of three solutions (n= 14 birds for each infusion group): 1) DHT and E2 (0.01 mg/ml each, dissolved in 40% DMSO, 43% PEG-300, 10% avian saline (0.9% NaCl), and 7% ethanol, as in (Meitzen et al., 2007.). Each pump contained Evans Blue (0.08 mg/ml) to measure diffusion of agents from the cannulae (Yen, Wei, Kuo, & Lai, 2013). We used DHT and E2, instead of T, to directly and selectively activate androgen receptors and estrogen receptors, and because the efficiency of T metabolism to DHT and/or E2 may vary with photoperiod (Forlano, Schlinger, & Bass, 2006; Riters et al., 2001; Soma, Bindra, Gee, Wingfield, & Schlinger, 1999; Soma, Schlinger, Wingfield, & Saldanha, 2003); 2) rapamycin (diluted to a final concentration of 9.16 ng/ml) combined with DHT and E2 (0.01 mg/ml each) in the same vehicle as above. Pilot studies showed that in birds infused with this dose of rapamycin, HVC was smaller and had fewer neurons; 3) vehicle alone as a control. We placed the osmotic pumps into microcentrifuge tubes filled with avian saline, sealed the tubes with Parafilm (Bemis Flexible Packaging, Neena, WI) and quick-drying cement, and mounted the tubes into “backpacks” custom-made for white-crowned sparrows, as previously described (Larson et al., 2013; Meitzen et al., 2007). The osmotic pump rested between the bird’s wings and allowed free movement.
Perfusion and histology
Five birds from each of the three treatment groups were deeply anesthetized and perfused with saline followed by 4% paraformaldehyde on day 35 (Figure 1b). The brains were collected, post-fixed in 4% paraformaldehyde, soaked in sucrose, and stored at 4°C until processing. Tissue was sectioned coronally at 40 μm on a freezing microtome into three series and stored at 4°C until further processing.
Analysis of rapamycin effect on protein phosphorylation
Nine birds from each of the three treatment groups were anesthetized with isoflurane (2%) and decapitated. The brains were removed rapidly (within 1 min) and Vibratome sectioned at 300 μm in ice-cold, oxygenated artificial cerebral spinal fluid (ACSF; 119 mM NaCl, 2.5 mM KCl, 1.3 mM MgSO4, 2.5 mM CaCl2, 1 mM NaH2PO4, 16.2 mM NaHCO3, 11 mM D-glucose, and 10 mM HEPES). From these slices, punches of tissue containing HVC were collected from both hemispheres. Tissue punch location was verified post-hoc by Nissl-staining of re-sectioned, fixed tissue. HVC punches included the proliferative ventricular zone just dorsal to HVC. Only punches taken entirely within the Nissl-defined borders of HVC were used for protein analysis (see Larson et al., 2015; C. K. Thompson et al., 2012). All tissue punches were flash frozen on dry ice, and stored at −80 degrees Celsius until processed for Western blots. We wished to limit our protein analysis to cells within HVC. Due to the low amount of total protein per HVC from one bird, it was necessary to pool tissue punches collected from three birds to obtain the minimum of 10 μg of total protein per replicate required for Western blot analysis. For each treatment group we therefore used three replicates of pooled punches (i.e., a total of nine birds in three pooled replicates).
Proteins were extracted from HVC using a RIPA buffer [150 mM NaCL, 1% SDS, 1% Triton X-100, 2 mM EDTA], supplemented with proteinase inhibitor cocktail (complete mini tablet, Roche Diagnostics) and phosphatase inhibitors (PhoSTOP, Roche diagnostics) as well as a proteasome inhibitor (MG132, Sigma). Each sample, representing a pool of HVC punches, was added with 100 ul of RIPA buffer and aspirated 10 times with a 28G syringe. After aspiration, the samples were left on ice for 10 minutes for incubation. After incubation, the samples were centrifuged for 10 min at 17,900xg to precipiate cell membranes. The supernatant was transferred to a fresh tube and the protein concentration was measured using a Bradford assay. Samples were processed with 4x Laemmli buffer at a concentration of 10 μg and boiled for two min at 95 degrees Celsius for denaturation. The gel electrophoresis was carried out on an 8% Bis Acrylamid gel and the subsequent protein transfer to polyvinylidene fluoride (PVDF) membrane was carried out overnight at four degrees Celsius. We tested a set of primary antibodies raised in rabbit, mouse, or goat targeting proteins up- or downstream of the mTOR signaling pathway [total and phospho mTOR, total and phospho Akt, total and phospho Erk1/2, total and phospho S6 ribosomal protein, Rac1, RhoA], but only total and phospho mTOR yielded a sufficient and interpretable Western blot signal.
Western blots were incubated with a rabbit polyclonal primary antibody against phospho mTOR (Cell Signaling, Cat# 2971, RRID:AB_330970), followed by incubation with a secondary HRP-labeled antibody. The primary antibody was produced by immunizing rabbits with a synthetic phosphopeptide encompassing the sequence around Ser2448 of the human mTOR sequence. The specificity of the antibody was previously confirmed (Bolster, Crozier, Kimball, & Jefferson, 2002; Hasumi et al., 2009). The phosphoprotein was visualized using enhanced chemiluminescence. Another set of Western blots was incubated with a rabbit polyclonal primary antibody against total mTOR (Cell Signaling, Cat# 2972, RRID:AB_330978), followed by a secondary HRP-labeled antibody. The total mTOR antibody was produced by immunizing rabbits with a synthetic peptide encompassing the sequence around Ser2481 of the human mTOR sequence. The antibody specificity was previously confirmed (Schafer et al., 2017; Stuttfeld et al., 2018). Both primary antibodies are listed in the Antibody Registry (http://antibodyregistry.org/). We validated these antibodies for our study using protein samples extracted from cortical tissue of mice, rats, and Gambeĺs White-Crowned Sparrows.
The immunoreactive bands on X-ray films were scanned at 600 dpi and their intensity was densitometrically quantified using Image J (Schneider, Rasband, & Eliceiri, 2012). Each phospho-immunoreactive band was normalized against the individual total protein signal for quantification of the amount of phosphorylation. For normalization, the complete immunoreactive signal between 150 and 300 kDa was taken for each band and condition. Whereas the immunoreactive signals for the mammalian samples appeared at the expected size of 289 kDa, the immunoreactive signal for the white-crowned sparrow samples consistently appeared as a wide band between 150kDa and 300 kDa, with the strongest signal at the expected size of 289 kDa. In order to maintain comparability and reproducibility of data analysis, we included the complete immunoreactive signal among the Gambeĺs White-Crowned Sparrow samples for densitometric analysis.
Measurement of neural attributes of HVC
To measure HVC volume, we used an overhead projector to trace the borders in both hemispheres throughout the full rostral-caudal extent. We scanned these tracings into a microcomputer and measured the surface area of each cross section using a customized module of Image J (W. Rasband, NIH). We determined the volume of HVC by summing the estimated volume between sections calculated with the formula for the volume of a cone frustum (Tramontin, Smith, Breuner, & Brenowitz, 1998).
Total neuron number in HVC was estimated for each treatment group. We used a random, systematic sampling protocol that has been previously described and validated (Tramontin et al., 1998), which yields estimates of neuronal density that do not differ from those obtained using the stereological optical dissector method. We counted the number of neurons in both hemispheres of HVC of each bird in fields chosen randomly by computer in every 3rd section containing HVC (8 sections per hemisphere on average). Four boxes (each 125 μm2) were randomly placed in each sampled section of HVC and all neurons were counted. We distinguished neurons from glia by their single nucleolus and uniform, nongranular cytoplasm. We sampled at least 150 HVC neurons throughout the rostral-caudal extent of the nucleus in each brain. This sample size is sufficient to encompass the entire range of variability in neuron density (Tramontin et al., 1998). Neuron number was estimated by multiplying density by HVC volume for each bird.
Measurement of neuronal size in RA
Neuron size in RA is a reliable and sensitive measure of seasonal plasticity of the song control system in songbirds (E. A. Brenowitz, 2008). For the present study, RA neuron size is an interesting phenotypic measure because the changes in this trait are mediated entirely by changes in transsynaptic trophic support of RA neurons from HVC neurons (Eliot A. Brenowitz, 2015); any changes observed in RA reflect effects of presynaptic manipulations in HVC.
We measured RA neuronal soma area in the same brains used for the HVC anatomical measurements described above. We distinguished neurons from glia in Nissl stained sections by their single nucleolus and uniform, nongranular cytoplasm. We used the same random, systematic sampling protocol as in HVC. We measured the soma area of at least 100 RA neurons per bird in fields chosen randomly by computer in each section. This sample size is sufficient to encompass the entire range of variability in soma area in RA (Tramontin et al., 1998).
Data analysis
All measurements were made blind to treatment for each bird. Comparisons were made using one-way ANOVAs, with infusion treatment as the factor, and Holm-Sidak tests for post-hoc pairwise comparisons between treatment groups with P level adjusted for multiple comparisons. For all statistical tests the alpha level was 0.05. Hedge’s g was used to calculate effect size (Hedges & Olkin, 1985). g values ≥ 0.8 indicate large effects (Cohen, 1992; Hedges & Olkin, 1985). Hedges’ g corrects for biases due to small sample sizes, and is therefore appropriate for measuring effect size in a pairwise contrast for a multiple comparison procedure following an ANOVA test (Hedges & Olkin, 1985).
3. Results:
Effect of rapamycin on protein phosphorylation
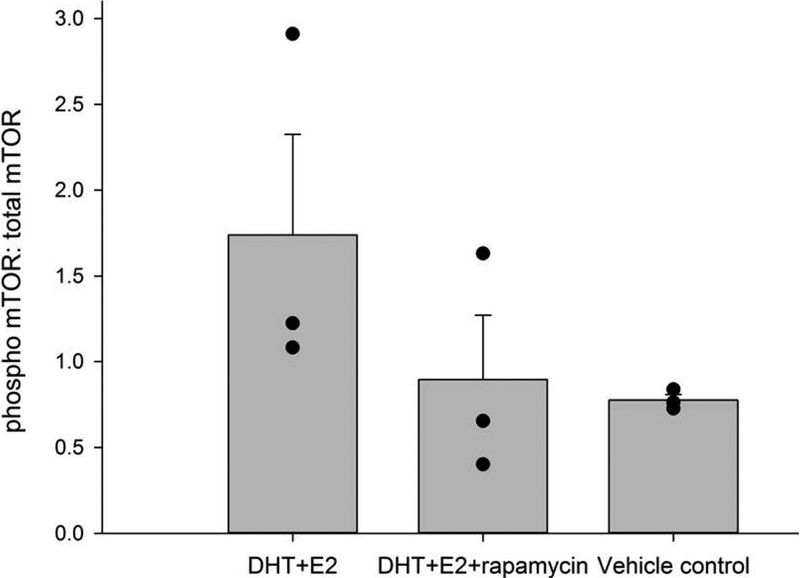
To assess the effects of rapamycin, birds were treated with DHT and E2 using osmotic pumps directed to the HVC (Figure 1). The ratio of phosphorylated mTOR (p-mTOR) to total mTOR, normalized for background for each replicate, was measured in punches taken in both brain hemispheres. Birds infused in the HVC with rapamycin+DHT+E2 displayed a lower ratio of p-mTOR/total mTOR than in birds infused with either DHT+E2 or vehicle alone (Figure 2). These differences were not significant in the ANOVA (F = 1.697, df =2,6 p = 0.261, power = 0.119), for reasons discussed below. However, the effect sizes for the comparison between the rapamycin+DHT+E2 and DHT+E2 groups, and between the vehicle control and DHT+E2 groups, were large (Hedge’s g = 0.791 and 1.069, respectively). Values of d ≥ 0.8 are considered to indicate large effects (Cohen, 1992; Sullivan & Feinn, 2012). These effect sizes comparisons suggest that rapamycin decreased phosporylation of mTOR in HVC cells.
Figure 2.
Infusing rapamycin in HVC decreased hormonal activation of mTOR. The ratio of phosphorylated mTOR (p-mTOR) to total mTOR in punches taken from HVC of birds infused with DHT+E2, DHT+E2 + rapamycin, or vehicle control. Data from individual birds represented as black dots, and bars show group mean ± SEM.
Effect of rapamycin on HVC neural attributes
Visual inspection of cannula tracks showed that all cannulae were adjacent to the borders of, and did not damage, HVC in all birds (Fig 1c). As indicated by Evans blue tissue staining, the osmotic pumps released their contents, which then diffused throughout the full extent of HVC, in all birds (Figure 1d).
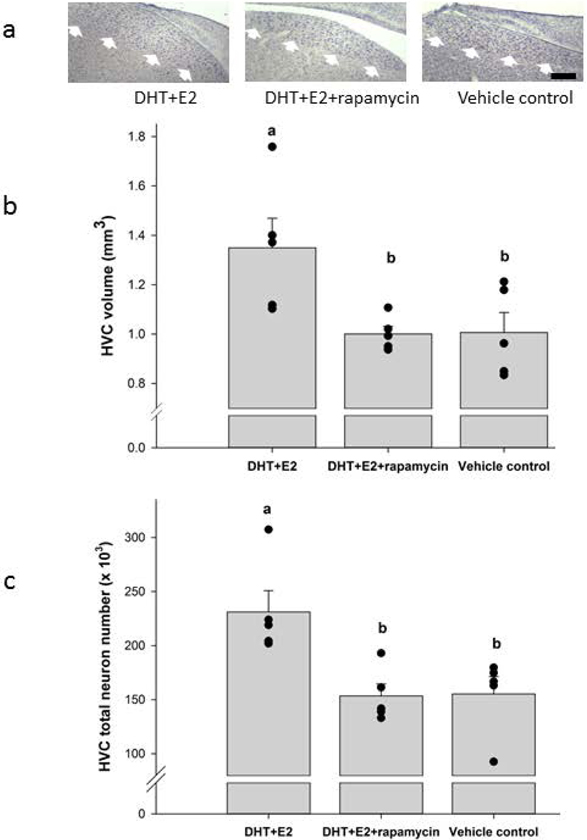
Rapamycin blocked the neuroprotective effect of steroid hormones on the size of HVC (ANOVA F = 5.54, df = 2,12, P = 0.020, power = 0.895; Figure 3a,b). The volume of HVC was significantly larger in the DHT+E2 infused birds than in either the DHT+E2+rapamycin birds (post-hoc t = 2.903, df= 8, P = 0.039) or the vehicle group (t = 2.86, df = 8, P = 0.029). Effect sizes for these comparisons were large (Hedge’s g = 1.681 and 1.369, respectively). HVC volume did not differ between the DHT+E2+rapamycin and vehicle groups (t = 0.048, df = 8, P = 0.962).
Figure 3.
(a) Coronal sections showing the borders of HVC (white arrows) in birds infused bilaterally in HVC with DHT+E2, DHT+E2 + rapamycin, or vehicle control. Scale bar, 250 μm. (b) HVC volume (mm3) in birds in each treatment group. (c) Total neuron number in HVC of birds in each treatment group. Data from individual birds represented as black dots, and bars show group mean ± SEM. Letters above bars indicate significant differences among treatment groups (one-way ANOVA, Holms-Sidak post hoc pairwise comparisons).
The differences in HVC volume between treatment groups reflected changes in total neuron number (ANOVA df = 2,12, P = 0.007, power = 0.843; Figure 3c). HVC had significantly more neurons in the DHT+E2 infused birds than in either the DHT+E2+rapamycin birds (post-hoc t = 3.468, df= 8, P = 0.014) or the vehicle group (t = 3.388, df = 8, P = 0.011). Effect sizes for these comparisons were large (Hedge’s g = 1.989 and 1.764, respectively). HVC total neuron number did not differ between the DHT+E2+rapamycin and vehicle groups (t = 0.08, df = 8, P = 0.938).
Effect of rapamycin on neuron size in RA
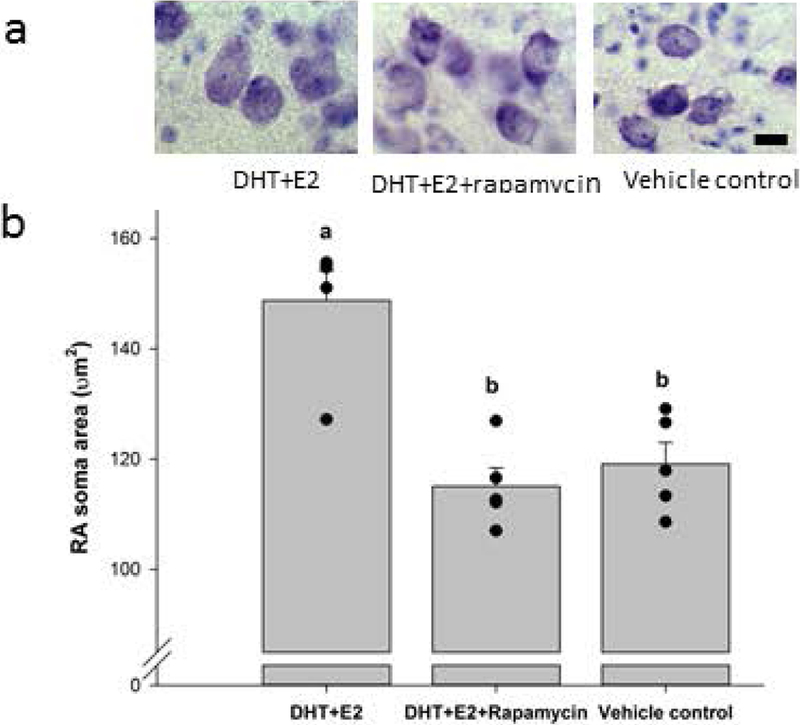
Rapamycin blocked the transsynaptic neuroprotective effect of steroid hormones infused in HVC on the size of neurons in RA (ANOVA df = 2,14, P = < 0.001, power = 1.00; Figure 4). Neuronal soma area in birds infused with DHT+E2 was significantly larger than in either birds infused with DHT+E2+rapamycin (post-hoc t = 6.115, df=8, P < 0.001) or vehicle (post-hoc t = 5.159, df = 8, P < 0.001). Effect sizes for these comparisons were large (Hedge’s g = 3.423 and 2.529, respectively). The high values of the ANOVA test power and effect sizes indicate that infusion of rapamycin in HVC had a strong transsynaptic effect on neuron size in RA. Neuronal soma area did not differ between the DHT+E2+rapamycin and vehicle groups (post-hoc t = 0.956, df = 8, P = 0.358). We did not observe Evans blue dye in sections containing RA, nor in the surrounding arcopallium, in any brain. This observation indicates that agents released from the cannula tip did not diffuse to RA, and the observed differences in RA neuron size therefore cannot be explained by the direct action of rapamycin on RA neurons.
Figure 4.
a) Representative neurons within RA of birds infused bilaterally in HVC with DHT+E2, DHT+E2 + rapamycin, or vehicle control. Scale bar, 10 μm. b) Soma area (μm2) of neurons in RA of birds in each treatment group. Data from individual birds represented as black dots, and bars show group mean ± SEM. Letters above bars indicate significant differences among treatment groups (one-way ANOVA, Holms-Sidak post hoc pairwise comparisons).
4. Discussion:
The results show that rapamycin infusion blocked the neuroprotective effects of sex steroids on the song nuclei HVC and RA in adult sparrow brains. HVC volume and neuron number, and RA neuron size, decreased considerably when vehicle-infused sparrows were transferred from breeding (high systemic T levels) to nonbreeding (basal systemic T) conditions, as previously reported (E. A. Brenowitz, 2008; Larson et al., 2014; C. K. Thompson et al., 2007; C. K. Thompson & Brenowitz, 2008; C.K. Thompson & Brenowitz, 2010). Intracerebral infusion of DHT+E2 near HVC preserved a large HVC with a high number of neurons, and large neurons in RA, similar to the effects of intracerebral infusion of T in HVC (C.K. Thompson & Brenowitz, 2010). Infusing the mTOR inhibitor rapamycin together with DHT+E2 blocked the neuroprotective effects of DHT+E2 on HVC and RA (Sato et al., 2010).
The decrease in neuronal size in RA in rapamycin-infused birds is noteworthy. Seasonal changes in soma area and spontaneous firing rate of RA neurons are entirely mediated by changes in transsynaptic trophic support from HVC neurons (Eliot A. Brenowitz, 2015). Local infusion of T or its metabolites in HVC of nonbreeding condition birds increases RA neuron size and firing rate, and protects RA neurons in sparrows transferred from breeding to nonbreeding condition (E. A. Brenowitz & Lent, 2001, 2002; Meitzen et al., 2007; C.K. Thompson & Brenowitz, 2010). Direct manipulations of sex steroid metabolites in RA, however, have no effect on either neuron size or firing rate (Meitzen et al., 2007). Brain-derived neurotrophic factor (BDNF) mediates the transsynaptic trophic effect from HVC to RA. BDNF synthesis is increased in HVC neurons by sex steroids, and this protein is released from HVC axons on to RA neurons (Eliot A. Brenowitz, 2015; Wissman & Brenowitz, 2009). Local infusion of BDNF protein in RA increases neuron size and firing rate in nonbreeding condition birds. Blockade in RA of tyrosine kinase B, the high affinity receptor for BDNF, with a fusion antibody or small molecule antagonists prevents the increases in neuron size and firing rate in breeding condition birds (E. A. Brenowitz, 2013, Miller, Wood, Lent, Perkel, and Brenowitz, unpublished observation; Wissman & Brenowitz, 2009).
We believe that the most parsimonious explanation for our results is that rapamycin acted by inhibiting mTOR activation in response to DHT+E2 treatment. In this scenario, mTOR activation in HVC neurons is a critical step in the transsynaptic trophic support cascade intiated by sex steroids. We attempted to test this hypothesis by extracting protein from tissue punches of HVC collected from birds in each treatment group, and using Western blots to measure levels of total and phosphorylated mTOR. Analysis of the protein data derived from the Western blots with ANOVA did not yield a significant test result. There was, however, a large effect size for the comparison of pmTOR:total mTOR between the rapamycin+DHT+E2 and DHT+E2 groups, and this suggests that the rapamycin treatment was effective in inhibiting mTOR phosphorylation.
We believe that the non-significant ANOVA test resulted from the challenge of finding antibodies that are immunoreactive to avian phospho-proteins. We tested several commercial and custom antibodies against phosphorylated mTOR, but none yielded clear labeling of protein samples derived from avian tissue, in contrast to strong labeling of protein from mouse brains. The small sample sizes used for the protein analysis yielded inadequate statistical power, and resulted from the need to pool HVC tissue punches from three birds per replicate to obtain an adequate amount of total protein. A post-hoc power analysis of the protein data for the rapamycin+DHT+E2 and DHT+E2 groups indicated that to achieve a significant test result with alpha = 0.05 and power = 0.8, a total of 21 samples, or 7 samples per treatment group, would be required. Given that each protein sample for the Western blot analysis required the pooling of HVC tissue punches from 3 birds, this would have required the use of 63 birds (3 birds per sample × 21 samples) for just this protein analysis. We believed that use of this many wild-caught White-crowned sparrows for this one analysis was not justified, given the consistency of the effects of rapamycin treatment on neural attributes of HVC and RA. The pronounced significant decreases in volume and total neuron number in HVC, and of neuron size in RA, in rapamycin-treated birds compared with the DHT+E2-treated birds supports the efficacy of rapamycin infusions. The use of wild-caught sparrows constrains the total number of birds that can be used for this type of protein analysis, but these birds offer the important advantage of showing naturally occurring adult brain plasticity in an elaborated manner not seen in domesticated species like the Zebra Finch and Canary, nor in mammalian models (see E. A. Brenowitz, 2008). In the absence of robust protein measurements, we can only tentatively suggest that rapamycin acted by inihibiting mTOR activation.
Rather than indicating the importance of mTOR activation for steroid neuroprotection, our results might be explained by increased local estrogen synthesis resulting from the rapid expression and translation of estrogen synthase (i.e., aromatase) in response to injured brain tissue caused by insertion of the cannulae (Saldanha, Duncan, & Walters, 2009; Wynne, Maas, & Saldanha, 2008). However, since all birds received cannula implants and would therefore have experienced the same injury, it seems unlikely that this alone could have produced the observed group differences in HVC and RA neural traits. This alternative hypothesis therefore cannot explain the results.
A second alternative hypothesis is that rapamycin is neurotoxic and killed HVC neurons. If this was the case, then we might expect to observe more pyknotic cells, and fewer intact neurons, in HVC of the rapamycin-infused birds. Close inspection of sections containing HVC, however, did not reveal more (or many) pyknotic cells in rapamycin-infused birds than the other treatment groups. Also, as reported above, HVC neuron number did not differ between the rapamycin and vehicle control birds. It seems very unlikely that a generalized toxicity effect would reduce HVC neuron number by the exact same magnitude in the rapamycin-treated birds as the lack of neuroprotective hormones in the vehicle control birds. In fact, rapamycin has actually been reported to be neuroprotective in different models (Cai & Yan, 2013; Su, Li, Jia, & Zhuo, 2014). Ahmadiantehrani and London(2017) found that the number of cells immunoreactive to phosphorylated S6, an mTOR target protein, in forebrain auditory regions of juvenile male zebra finches returned to baseline levels by 15 hours following infusion of rapamycin, which further suggests that it is not toxic to cells. Rapamycin is being used in clinical anti-aging studies in humans, without the adverse side effects that would be expected if it is neurotoxic (Kraig et al., 2018). We therefore believe that it is unlikely that our results were due to a generalized toxicity of rapamycin treatment.
Rather, the results suggest that the neuroprotective effect of sex steroids on HVC and RA requires activation of mTOR in the PI3K-Akt signaling pathway. Hormone protection in various mammalian preparations is also known to involve activation of this signaling pathway (D’Astous, Mendez, Morissette, Garcia-Segura, & Di Paolo, 2006; Pike et al., 2008; Sato et al., 2010; L. Zhang et al., 2010). The confidence with which we can reach this conclusion for HVC is tempered by our inability to measure significant reductions in phosphorylated levels of mTOR from Western blots, as discussed above.
In conclusion, we showed that infusing androgenic and estrogenic hormones in sparrow HVC protected neurons in both HVC and its efferent target RA. Rapamycin blocked the neuroprotective effects of these sex steroids. We suggest that rapamycin acted by inactivating mTOR, but this needs to be tested directly. Together with the results of Ahmadiantehrani and London (2017), our results show that the PI3K-Akt-mTOR signaling pathway plays an important role in mediating plasticity in the avian forebrain. Our study demonstrates that the avian song system provides an excellent model for investigating nongenomic mechanisms by which sex steroids have neuroprotective effects in the context of naturally occurring neurodegeneration in adult brains.
Acknowledgements:
Supported by NIH R01 NS075331 to EAB and R56 NS021072 to MVC. The authors declare no conflicts of interest.
References:
- Ahmadiantehrani S, Gores EO, & London SE (2018). A complex mTOR response in habituation paradigms for a social signal in adult songbirds. Learning & memory (Cold Spring Harbor, N.Y.), 25(6), 273–282. doi: 10.1101/lm.046417.117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmadiantehrani S, & London SE (2017). Bidirectional manipulation of mTOR signaling disrupts socially mediated vocal learning in juvenile songbirds. Proceedings of the National Academy of Sciences, 114(35), 9463–9468. doi: 10.1073/pnas.1701829114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolster DR, Crozier SJ, Kimball SR, & Jefferson LS (2002). AMP-activated Protein Kinase Suppresses Protein Synthesis in Rat Skeletal Muscle through Down-regulated Mammalian Target of Rapamycin (mTOR) Signaling. Journal of Biological Chemistry, 277(27), 23977–23980. doi: 10.1074/jbc.C200171200 [DOI] [PubMed] [Google Scholar]
- Brann DW, Dhandapani K, Wakade C, Mahesh VB, & Khan MM (2007). Neurotrophic and neuroprotective actions of estrogen: Basic mechanisms and clinical implications. Steroids, 72(5), 381–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenowitz EA (2008). Plasticity of the song control system in adult birds In Zeigler HP & Marler P (Eds.), Neuroscience of birdsong (pp. 332–349). Cambridge: Cambridge University Press. [Google Scholar]
- Brenowitz EA (2013). Testosterone and brain-derived neurotrophic factor interactions in the avian song control system. Neuroscience, 239(0), 115–123. doi: 10.1016/j.neuroscience.2012.09.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenowitz EA (2015). Transsynaptic trophic effects of steroid hormones in an avian model of adult brain plasticity. Frontiers in Neuroendocrinology, 37(0), 119–128. doi: 10.1016/j.yfrne.2014.09.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenowitz EA, & Larson TA (2015). Neurogenesis in the adult avian song-control system. Cold Spring Harb Perspect Biol, 7(6). doi: 10.1101/cshperspect.a019000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenowitz EA, & Lent K (2001). Afferent input is necessary for seasonal growth and maintenance of adult avian song control circuits. J Neurosci, 21(7), 2320–2329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenowitz EA, & Lent K (2002). Act locally and think globally: intracerebral testosterone implants induce seasonal-like growth of adult avian song control circuits. Proc Natl Acad Sci U S A, 99(19), 12421–12426. doi: 10.1073/pnas.192308799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai Z, & Yan L-J (2013). Rapamycin, Autophagy, and Alzheimer’s Disease. Journal of biochemical and pharmacological research, 1(2), 84–90. [PMC free article] [PubMed] [Google Scholar]
- Cardona-Gómez GP, Mendez P, DonCarlos LL, Azcoitia I, & Garcia-Segura LM (2001). Interactions of estrogens and insulin-like growth factor-I in the brain: implications for neuroprotection. Brain Research Reviews, 37(1–3), 320–334. doi: 10.1016/S0165-0173(01)00137-0 [DOI] [PubMed] [Google Scholar]
- Chang SH, Poser S, & Xia Z (2004). A Novel Role For Serum Response Factor in Neuronal Survival. The Journal of Neuroscience, 24(9), 2277–2285. doi: 10.1523/jneurosci.4868-03.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen J (1992). A power primer. Psychological Bulletin, 112(1), 155–159. doi: 10.1037/0033-2909.112.1.155 [DOI] [PubMed] [Google Scholar]
- D’Astous M, Mendez P, Morissette M, Garcia-Segura LM, & Di Paolo T. r. s. (2006). Implication of the Phosphatidylinositol-3 Kinase/Protein Kinase B Signaling Pathway in the Neuroprotective Effect of Estradiol in the Striatum of 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine Mice. Molecular Pharmacology, 69(4), 1492–1498. doi: 10.1124/mol.105.018671 [DOI] [PubMed] [Google Scholar]
- Forlano PM, Schlinger BA, & Bass AH (2006). Brain aromatase: New lessons from non-mammalian model systems. [DOI] [PubMed] [Google Scholar]
- Garcia-Segura LM, Azcoitia I, & DonCarlos LL (2001). Neuroprotection by estradiol. Prog Neurobiol, 63(1), 29–60. [DOI] [PubMed] [Google Scholar]
- Hasumi Y, Baba M, Ajima R, Hasumi H, Valera VA, Klein ME, … Linehan WM (2009). Homozygous loss of BHD causes early embryonic lethality and kidney tumor development with activation of mTORC1 and mTORC2. Proceedings of the National Academy of Sciences, 106(44), 18722–18727. doi: 10.1073/pnas.0908853106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedges LV, & Olkin I (1985). Statistical methods for meta-analysis. San Diego: Academic Press. [Google Scholar]
- Honda K, Sawada H, Kihara T, Urushitani M, Nakamizo T, Akaike A, & Shimohama S (2000). Phosphatidylinositol 3-kinase mediates neuroprotection by estrogen in cultured cortical neurons. J Neurosci Res, 60(3), 321–327. doi: [pii] [DOI] [PubMed] [Google Scholar]
- Jaffer S, Shynlova O, & Lye S (2009). Mammalian Target of Rapamycin Is Activated in Association with Myometrial Proliferation during Pregnancy. Endocrinology, 150(10), 4672–4680. doi: 10.1210/en.2009-0419 [DOI] [PubMed] [Google Scholar]
- Kraig E, Linehan LA, Liang H, Romo TQ, Liu Q, Wu Y, … Kellogg DL (2018). A randomized control trial to establish the feasibility and safety of rapamycin treatment in an older human cohort: Immunological, physical performance, and cognitive effects. Experimental Gerontology, 105, 53–69. doi: 10.1016/j.exger.2017.12.026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larson TA, Lent KL, Bammler TK, MacDonald JW, Wood WE, Caras ML, … Brenowitz EA (2015). Network analysis of microRNA and mRNA seasonal dynamics in a highly plastic sensorimotor neural circuit. BMC Genomics, 16(1), 905. doi: 10.1186/s12864-015-2175-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larson TA, Thatra NM, Lee BH, & Brenowitz EA (2014). Reactive neurogenesis in response to naturally occurring apoptosis in an adult brain. J Neurosci, 34(39), 13066–13076. doi: 10.1523/JNEUROSCI.3316-13.2014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larson TA, Wang TW, Gale SD, Miller KE, Thatra NM, Caras ML, … Brenowitz EA (2013). Postsynaptic neural activity regulates neuronal addition in the adult avian song control system. Proc Natl Acad Sci U S A, 110(41), 16640–16644. doi: 10.1073/pnas.1310237110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu M, Kelley MH, Herson PS, & Hurn PD (2010). Neuroprotection of sex steroids. Minerva Endocrinologica, 35(2), 127–143. [PMC free article] [PubMed] [Google Scholar]
- Marin R, Guerra B, Alonso R, Ramirez CM, & Diaz M (2005). Estrogen activates classical and alternative mechanisms to orchestrate neuroprotection. Curr Neurovasc Res, 2(4), 287–301. [DOI] [PubMed] [Google Scholar]
- Martini M, De Santis MC, Braccini L, Gulluni F, & Hirsch E (2014). PI3K/AKT signaling pathway and cancer: an updated review. Annals of Medicine, 46(6), 372–383. doi: 10.3109/07853890.2014.912836 [DOI] [PubMed] [Google Scholar]
- Meitzen J, Moore IT, Lent K, Brenowitz EA, & Perkel DJ (2007). Steroid hormones act transsynaptically within the forebrain to regulate neuronal phenotype and song stereotypy. J Neurosci, 27(44), 12045–12057. doi: 10.1523/JNEUROSCI.3289-07.2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Memmott RM, & Dennis PA (2009). Akt-dependent and -independent mechanisms of mTOR regulation in cancer. Cellular Signalling, 21(5), 656–664. doi: 10.1016/j.cellsig.2009.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pike CJ, Carroll JC, Rosario ER, & Barron AM (2009). Protective actions of sex steroid hormones in Alzheimer’s disease. Frontiers in Neuroendocrinology, 30(2), 239–258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pike CJ, Nguyen T-VV, Ramsden M, Yao M, Murphy MP, & Rosario ER (2008). Androgen cell signaling pathways involved in neuroprotective actions. Hormones and Behavior, 53(5), 693–705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prokai L, & Simpkins JW (2007). Structure-nongenomic neuroprotection relationship of estrogens and estrogen-derived compounds. Pharmacology & therapeutics, 114(1), 1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quesada A, Lee BY, & Micevych PE (2008). PI3 kinase/Akt activation mediates estrogen and IGF-1 nigral DA neuronal neuroprotection against a unilateral rat model of Parkinson’s disease. Dev Neurobiol, 68(5), 632–644. doi: 10.1002/dneu.20609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quesada A, & Micevych P (2008). Estrogen and progesterone modulate [35S]GTPgammaS binding to nociceptin receptors. Neuroendocrinology, 88(1), 35–42. doi:000113933 [pii] 10.1159/000113933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reiner A, Perkel DJ, Bruce LL, Butler AB, Csillag A, Kuenzel W, … Avian Brain Nomenclature, F. (2004). Revised nomenclature for avian telencephalon and some related brainstem nuclei. J Comp Neurol, 473(3), 377–414. doi: 10.1002/cne.20118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riters LV, Baillien M, Eens M, Pinxten R, Foidart A, Ball GF, & Balthazart J (2001). Seasonal variation in androgen-metabolizing enzymes in the diencephalon and telencephalon of the male European starling (Sturnus vulgaris). J Neuroendocrinol, 13(11), 985–997. [DOI] [PubMed] [Google Scholar]
- Rodrik V, Zheng Y, Harrow F, Chen Y, & Foster DA (2005). Survival signals generated by estrogen and phospholipase D in MCF-7 breast cancer cells are dependent on Myc. Molecular and cellular biology, 25(17), 7917–7925. doi: 10.1128/mcb.25.17.7917-7925.2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabatini DM (2017). Twenty-five years of mTOR: Uncovering the link from nutrients to growth. Proceedings of the National Academy of Sciences. doi: 10.1073/pnas.1716173114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saldanha CJ, Duncan KA, & Walters BJ (2009). Neuroprotective actions of brain aromatase. Frontiers in Neuroendocrinology, 30(2), 106–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sato A, Sunayama J, Matsuda K, Tachibana K, Sakurada K, Tomiyama A, … Kitanaka C (2010). Regulation of neural stem/progenitor cell maintenance by PI3K and mTOR. Neurosci Lett, 470(2), 115–120. doi:S0304–3940(09)01671–1 [pii] 10.1016/j.neulet.2009.12.067 [DOI] [PubMed] [Google Scholar]
- Schafer S, Viswanathan S, Widjaja AA, Lim W-W, Moreno-Moral A, DeLaughter DM, … Cook SA (2017). IL-11 is a crucial determinant of cardiovascular fibrosis. Nature, 552, 110. doi:10.1038/nature2467610.1038/nature24676https://www.nature.com/articles/nature24676#supplementary-informationhttps://www.nature.com/articles/nature24676#supplementary-information [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schneider CA, Rasband WS, & Eliceiri KW (2012). NIH Image to ImageJ: 25 years of image analysis. Nature Methods, 9, 671. doi: 10.1038/nmeth.2089 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scott BB, & Lois C (2007). Developmental origin and identity of song system neurons born during vocal learning in songbirds. J Comp Neurol, 502(2), 202–214. [DOI] [PubMed] [Google Scholar]
- Sohrabji F (2015). Estrogen-IGF-1 interactions in neuroprotection: Ischemic stroke as a case study. Frontiers in Neuroendocrinology, 36(0), 1–14. doi: 10.1016/j.yfrne.2014.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soma KK, Bindra RK, Gee J, Wingfield JC, & Schlinger BA (1999). Androgen-metabolizing enzymes show region-specific changes across the breeding season in the brain of a wild songbird. Journal of Neurobiology, 41(2), 176–188. [PubMed] [Google Scholar]
- Soma KK, Schlinger BA, Wingfield JC, & Saldanha CJ (2003). Brain aromatase, 5 alpha-reductase, and 5 beta-reductase change seasonally in wild male song sparrows: relationship to aggressive and sexual behavior. J Neurobiol, 56(3), 209–221. doi: 10.1002/neu.10225 [DOI] [PubMed] [Google Scholar]
- Stuttfeld E, Aylett CHS, Imseng S, Boehringer D, Scaiola A, Sauer E, … Ban N (2018). Architecture of the human mTORC2 core complex. eLife, 7, e33101. doi: 10.7554/eLife.33101 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su W, Li Z, Jia Y, & Zhuo Y (2014). Rapamycin Is Neuroprotective in a Rat Chronic Hypertensive Glaucoma Model. PLoS ONE, 9(6), e99719. doi: 10.1371/journal.pone.0099719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan GM, & Feinn R (2012). Using Effect Size—or Why the P Value Is Not Enough. Journal of Graduate Medical Education, 4(3), 279–282. doi: 10.4300/JGME-D-12-00156.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson CK, Bentley GE, & Brenowitz EA (2007). Rapid seasonal-like regression of the adult avian song control system. Proc Natl Acad Sci U S A, 104(39), 15520–15525. doi:0707239104 [pii] 10.1073/pnas.0707239104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson CK, & Brenowitz EA (2008). Caspase inhibitor infusion protects an avian song control circuit from seasonal-like neurodegeneration. J Neurosci, 28(28), 7130–7136. doi: 10.1523/JNEUROSCI.0663-08.2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson CK, & Brenowitz EA (2010). Neuroprotective effects of testosterone in a naturally-occurring model of neurodegeneration in the adult avian song control system. Journal of Comparative Neurology, 518, 4760–4770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson CK, Meitzen J, Replogle K, Drnevich J, Lent KL, Wissman AM, … Brenowitz EA (2012). Seasonal changes in patterns of gene expression in avian song control brain regions. PLoS ONE, 7(4), e35119. doi: 10.1371/journal.pone.0035119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tokarev K, Boender AJ, Claßen GAE, & Scharff C (2016). Young, active and well-connected: adult-born neurons in the zebra finch are activated during singing. Brain Structure and Function, 221(4), 1833–1843. doi: 10.1007/s00429-015-1006-y [DOI] [PubMed] [Google Scholar]
- Tramontin AD, Hartman VN, & Brenowitz EA (2000). Breeding conditions induce rapid and sequential growth in adult avian song control circuits: a model of seasonal plasticity in the brain. J Neurosci, 20(2), 854–861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tramontin AD, Smith GT, Breuner CW, & Brenowitz EA (1998). Seasonal plasticity and sexual dimorphism in the avian song control system: stereological measurement of neuron density and number. J Comp Neurol, 396(2), 186–192. [DOI] [PubMed] [Google Scholar]
- Wissman AM, & Brenowitz EA (2009). The role of neurotrophins in the seasonal-like growth of the avian song control system. J Neurosci, 29(20), 6461–6471. doi: 10.1523/JNEUROSCI.0638-09.2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wynne RD, Maas S, & Saldanha CJ (2008). Molecular characterization of the injury-induced aromatase transcript in the adult zebra finch brain. Journal of Neurochemistry, 105(5), 1613–1624. doi: 10.1111/j.1471-4159.2008.05256.x [DOI] [PubMed] [Google Scholar]
- Yen LF, Wei VC, Kuo EY, & Lai TW (2013). Distinct Patterns of Cerebral Extravasation by Evans Blue and Sodium Fluorescein in Rats. PLoS ONE, 8(7), e68595. doi: 10.1371/journal.pone.0068595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L, Qu Y, Tang J, Chen D, Fu X, Mao M, & Mu D (2010). PI3K/Akt signaling pathway is required for neuroprotection of thalidomide on hypoxic-ischemic cortical neurons in vitro. Brain Res, 1357, 157–165. doi: 10.1016/j.brainres.2010.08.007 [DOI] [PubMed] [Google Scholar]
- Zhang Z, Yang R, Zhou R, Li L, Sokabe M, & Chen L (2010). Progesterone Promotes the Survival of Newborn Neurons in the Dentate Gyrus of Adult Male Mice. Hippocampus, 20(3), 402–412. doi: 10.1002/hipo.20642 [DOI] [PubMed] [Google Scholar]