Abstract
Background
We evaluated the effects of neurofeedback as an augmentation treatment on depressive symptoms and functional recovery in patients with treatment-resistant depression (TRD).
Methods
We included 24 adult patients with TRD and 12 healthy adults. 24 TRD patients were assigned to the neurofeedback augmentation group (n = 12) and the medication-only (treatment as usual [TAU]) group (n = 12). The neurofeedback augmentation group underwent combined therapy comprising medication and 12–24 sessions of neurofeedback training for 12 weeks. To assess the serum levels of brain-derived neurotrophic factor (BDNF) in both groups, pre- and post-treatment blood samples were obtained. Patients were evaluated using the Hamilton Depression Rating Scale (HAM-D), Beck Depression Inventory (BDI), Clinical Global Impression-Severity (CGI-S), 5-level version of European Quality of Life Questionnaire 5-Dimensional Classification (EQ-5D-5L), and Sheehan Disability Scale (SDS) at baseline, and at the 1-, 4-, and 12-week.
Results
From baseline to week 12, neurofeedback training reduced mean scores on HAM-D, BDI-II, CGI-S, and SDS, and increased mean EQ-5D-5L tariff score. In the neurofeedback augmentation group, the response and remission rates were 58.3% and 50.0%, respectively, at week 12. Changes in HAM-D, EQ-5D-5L tariff score, and SDS were significantly larger in the neurofeedback group than in the medication-only (TAU) group. No significant difference in BDNF level was found pre- vs. post-treatment in any of the groups.
Conclusion
Despite the small sample size, these results suggest that neurofeedback treatment may be effective as an augmentation treatment, not only for depressive symptoms, but also for functional recovery, in patients with TRD.
Trial Registration
Clinical Research Information Service Identifier: KCT0004183
ClinicalTrials.gov Identifier: NCT04078438
Keywords: Treatment-Resistant Depression, Neurofeedback, Functional Recovery
Graphical Abstract
INTRODUCTION
Major depressive disorder (MDD) is a severely disabling disorder resulting in the deterioration of daily function and lowering quality of life.1 The lifetime prevalence of depression is 10%–15%,2 and the annual prevalence of MDD in the United States is approximately 7%.3 The World Health Organization has reported that MDD is expected to be the top disease in terms of global burden by 2030.4 Fifty percent of patients with depressive disorder have a chronic disease course, and 20% of such patients have insufficient responses to treatment despite the use of antidepressant medication.5 In addition, although antidepressants have been shown to be effective, residual symptoms may continue by stopping the medication early due to inconvenient side effects of the medication. Sixty percent of patients with depressive disorder have poorer executive function. Patients with depressive disorder are continuously affected by deficits in social functioning, such as interpersonal relationships and job adaptation, even if some of their symptoms are improved by medication.6 Therefore, various additional treatments other than antidepressant treatment have been attempted to improve residual depressive symptoms and the remission rate.
Brain waves have been used to measure brain activity7,8 and previous studies have reported that different brain waves reflect different brain states, including moods.9,10,11,12 Neurofeedback is a type of electroencephalography (EEG) training that allows individuals to change the levels of particular types of brain waves displayed on a computer by operational conditioning.13,14,15 EEG studies showed that the neurofeedback is capable of generating long term changes in the spectral EEG topography,16 while neuroimaging studies represented the neuroplastic effects from neurofeedback treatment.17,18,19
Neurofeedback is a noninvasive, safe, and simple treatment method without adverse effects associated with medication use.20 However, there is a disadvantage in that it is relatively more expensive than drug treatment. Another neuromodulation treatment, repetitive transcranial magnetic stimulation (rTMS), has indications for treatment in patients who are resistant to more than 1 mediation. Neurofeedback may be considered a new augmentation treatment for patients with treatment-resistant depression (TRD), even after the use of antidepressants. Some studies have reported improvements in both depressive symptoms and executive function following neurofeedback treatment.15,21 A recent article insisted that neurofeedback treatment for depression as having “revealed promising effects in recent clinical trials.”22 However, most studies have been case reports or uncontrolled studies,15 and the mechanism underlying the treatment effects of neurofeedback are still unclear. In addition, there has been no study of neurofeedback on depressive symptoms and functional recovery in patients with TRD.
Brain-derived neurotrophic factor (BDNF) acts on certain neurons of the central nervous system and the peripheral nervous system. It helps support the survival of existing neurons and encourages the growth and differentiation of new neurons and synapses. Previous studies have suggested the presence of an etiological link between the development of depression and BDNF.23,24,25 However, no studies have examined the association between neurofeedback and changes in BDNF level.
The purpose of this pilot study was to evaluate the effects of neurofeedback as an augmentation treatment on depressive symptoms and functional recovery in patients with TRD. We also aimed to identify the usefulness of BDNF as a biomarker for neurofeedback by examining changes in the BDNF level before vs. after treatment in the neurofeedback treatment and medication-only (treatment as usual, TAU) groups.
METHODS
Participants
Thirty-six subjects were recruited from our university hospital in Korea from September 2014 to April 2017. All participants enrolled in this study were aged 18 years or older. Twelve of the subjects were healthy controls without any kind of axis I diagnosis according to the Diagnostic and Statistical Manual of Mental Disorders, 4th edition, text-revised (DSM-IV-TR), neurological illness, or medical illness. The healthy control subjects were not under any maintenance medications. The other 24 subjects were diagnosed with MDD according to the DSM-IV-TR criteria for MDD, and had especially treatment-resistant MDD. We defined TRD as the failure to achieve remission with remaining persistent depressive symptoms (Hamilton Rating Scale for Depression [HAM-D] score ≥ 14) despite adequate antidepressant therapy (2 or more antidepressant trials involving effective doses and sufficient duration to produce a robust therapeutic effect [e.g., 12 weeks]).26,27,28 Patients with psychosis, bipolar disorder, brain injury, clinically diagnosed neurological disorder, convulsive disorder, or pregnancy were excluded from participation. Participants with TRD maintained the same medication use from 1 month before the study until the end of the study. If a participant was absent from 4 or more treatment sessions, he or she was dropped from the study.
Study design
This was a small, prospective, open-labeled controlled study. Twenty-four patients with TRD were assigned to the neurofeedback augmentation group (n = 12) or the medication-only (TAU) group (n = 12), and 12 healthy adults were assigned to the healthy control group.
The neurofeedback augmentation group was asked to participate in 12 weeks of combined therapy of medication and 12–24 sessions of neurofeedback training. The neurofeedback protocol was determined considering the patient's main symptoms.29 Patients in the neurofeedback augmentation group received sensorimotor rhythm (SMR) beta or beta training for 30 minutes, and then alpha/theta (A/T) training for 30 minutes in each session.
To reduce the impact of confounding factors, the medication-only (TAU) group visited at the same schedule as neurofeedback augmentation group and received psychotherapy placebo sessions instead of neurofeedback training sessions.21 These sessions included psychological assessment and supportive psychotherapy. The medication-only (TAU) group maintained the same medication use as that before the study.
All patients were evaluated using the HAM-D, Beck Depression Inventory-II (BDI-II), Clinical Global Impression-Severity (CGI-S), 5-level version of European Quality of Life Questionnaire 5-Dimensional Classification (EQ-5D-5L), and Sheehan Disability Scale (SDS) at baseline, and at the 1-, 4-, and 12-week time points. In addition, pre- and post-treatment blood samples were obtained between 11 am and 12 am for evaluation of the serum level of BDNF in both patient groups. The healthy controls provided blood samples using the same procedure at baseline only. These samples were used to determine the serum BDNF level.
Neurofeedback procedure
Neurofeedback training was performed using a Neurocybernetics EEG Biofeedback system (Neurocybernetics Inc., Encino, CA, USA). The neurofeedback protocol was determined by the neurofeedback team, which included 3 psychiatrists, in consideration of the patient's main symptoms, as in a prior study by Cheon et al.29 One individual was assigned to beta training at T3 and then A/T training, 5 individuals were assigned to beta training at F3 and then A/T training, and the other 6 individuals in the neurofeedback augmentation group were assigned to SMR training at T4 and then A/T training. The assigned neurofeedback protocol was performed once or twice per week for both beta or SMR beta and A/T for 12 weeks. In each session, the participants received SMR training at T4 or beta training at F3 or T3 for 30 minutes, and then A/T training at Pz for 30 minutes.
During the SMR or beta training, the participants played a computer game. The brain electrical activity of the participants was displayed on a monitor in the form of an audio-visual exercise. Achievement scores and graphs were provided as reward feedback. The reward bands for SMR and beta were 12–15 Hz and 15–18 Hz, respectively. During the A/T neurofeedback training, the participants were provided with only auditory feedback while sitting in a comfortable chair with eyes closed. They were trained to reduce the alpha amplitude and increase the theta amplitude so that the theta amplitude was dominant over the alpha amplitude.30 The reward bands for theta and alpha were 4–8 Hz and 8–13 Hz, respectively.
BDNF measurement
After sufficient stabilization for about 30 minutes, the participants provided venous blood, which was collected in an anticoagulant tube between 11:00 a.m. and noon. The specimens were left at room temperature for 1 hour and stored at 4°C or lower for 1 hour before serum separation. The serum was stored at −70°C until specimen analysis. Serum BDNF levels were measured using an enzyme-linked immunosorbent assay (ELISA). We used the Human BDNF Immunoassay ELISA kit (Quantikine Human BDNF, R&D Systems, Minneapolis, MN, USA) and a microplate reader (Molecular Devices, Sunnyvale, CA, USA).
Baseline and follow-up assessments
The HAM-D is an observer rating scale and one of the most widely used measures of depressive disorder.31,32 This scale is composed of 17 items, and it has been used to assess the therapeutic effect as well as the severity of depression. The total possible score is 52, where higher scores indicate more severe depression. Hamilton considered a total score of 8–13 mild range, 14–18 moderate, 18–22 severe, and 23 or higher very severe.
The BDI is a self-report scale designed to evaluate the presence and severity of depressive symptoms.33 It consists of 21 items including cognitive, emotional, motivative, and physical symptoms of depression. Each question is scored from 0 to 3 points, and the total score ranges from 0 to 63, indicating the higher the total scores, the more severe the depression. It considers a total score of 0–9 minimal range, 10–15 mild, 16–23 moderate, and 24–63 severe.34,35
The CGI-S is a single-item scale composed of 7 levels ranging from maximum score of 7 to normal state of 1.36 This scale is a measure by which the evaluator comprehensively assesses the severity of mental illness regardless of diagnosis. It is relatively easy and quick to assess the illness so has been used in many clinical studies.37
The EQ-5D-5L was developed by the EuroQol Group, and is used to assess 5 dimensions: mobility, self care, usual activity, pain/discomfort, and anxiety/depression. Each dimension consists of 5 levels: no problems, slight problems, moderate problems, severe problems, and extreme problems; so a total of 3,125 health states can be evaluated.38,39 The valuation of EQ-5D-5L (converted tariff score) is an index score calculated by applying weight to each of the 5 EQ-5D-5L questionnaires to provide a comprehensive summary of health-related quality of life. The resulting set of tariff is widely used to calculate preferences for EQ-5D-5L health states. As EQ-5D-5L is translated according to the culture and situation of each country, EQ-5D-5L tariffs may differ at each country. The index score is valued from −0.066 to 1, and the higher scores indicate the higher quality of life for the patients.40,41,42,43 In this study, EQ-5D-5L tariffs (index values) were calculated according to the ‘The EQ-5D-5L valuation study in Korea.’43
The SDS is a self-report scale designed by Sheehan to assess the severity of functional impairment.44 This scale consists of 3 items, and each item is divided into 11 levels from 0 to 10 points. It is considered 0 point: none, 1–3 points: mild, 4-6 points: moderate, and 7–10 points: severe. Total score ranges from 0 to 30 and means that the higher the total scores, the more severe the functional impairment.45
The primary endpoint was determined a priori to be a change in the HAM-D score. Secondary endpoints included change from baseline to study endpoint on the HAM-D, response and remission rates, and BDI-II, CGI-S, SDS, and EQ-5D-5L tariff scores. The results were compared among the groups. The HAM-D, BDI-II, and CGI-S were used to evaluate depression. Remission was defined as achieving a HAM-D score below 7, and response was defined as a 50% or greater reduction in HAM-D score from baseline.46 The SDS and EQ-5D-5L were used to measure daily function and quality of life.
The HAM-D, BDI-II, CGI-S, EQ-5D-5L, and SDS were evaluated in all study participants at baseline and at the 1-, 4-, and 12-week time points. We also measured changes in the serum BDNF level before vs. after treatment in the TRD groups.
Statistical analysis
The demographic and clinical data of the participants were analyzed using frequency, descriptive statistics and Mann-Whitney test. The efficacy parameters of the HAM-D, CGI-S, BDI-II, EQ-5D-5L tariff score, and SDS were compared using independent-sample t-tests, and changes in the HAM-D, CGI-S, BDI-II, EQ-5D-5L tariff score, and SDS scores were assessed using paired t-tests adjusted for multiple comparisons using the Bonferroni method. Mean changes in the HAM-D, BDI-II, EQ-5D-5L tariff, and SDS scores, as well as those in the BDNF values, were compared using non-parametric tests not adjusted for multiple comparisons. Response and remission rates were compared using the frequency test (χ2 test or Fisher exact test). All statistical analyses were performed using IBM SPSS version 23.0 (IBM Co., Armonk, NY, USA). Statistical significance was considered at P < 0.05.
Ethics statement
All participants were fully informed regarding the study and provided written informed consent. There was no monetary or any other kind of reward for participation in all participants. This study was reviewed and approved by the Hospital Institutional Review Board (approval No. YUMC 2014-01-389). In addition, this study was registered at Clinical Research Information Service (CRIS) (approval No. KCT0004183) and ClinicalTrials.gov (approval No. NCT04078438).
RESULTS
Demographics and baseline clinical characteristics
Thirty-six participants entered this study between September 2014 and April 2017. Twelve individuals each were assigned to the neurofeedback augmentation group, the medication-only (TAU) group, and the normal control group. Twenty-four patients with TRD selected their group. No participant was dropped from the study. The 24 participants in the neurofeedback augmentation group and the medication-only (TAU) group completed the trial. The 12 healthy controls provided blood samples at baseline only and they were found to have no mental illness in face-to-face interviews with a psychiatrist and had apparently well-maintained daily functioning.
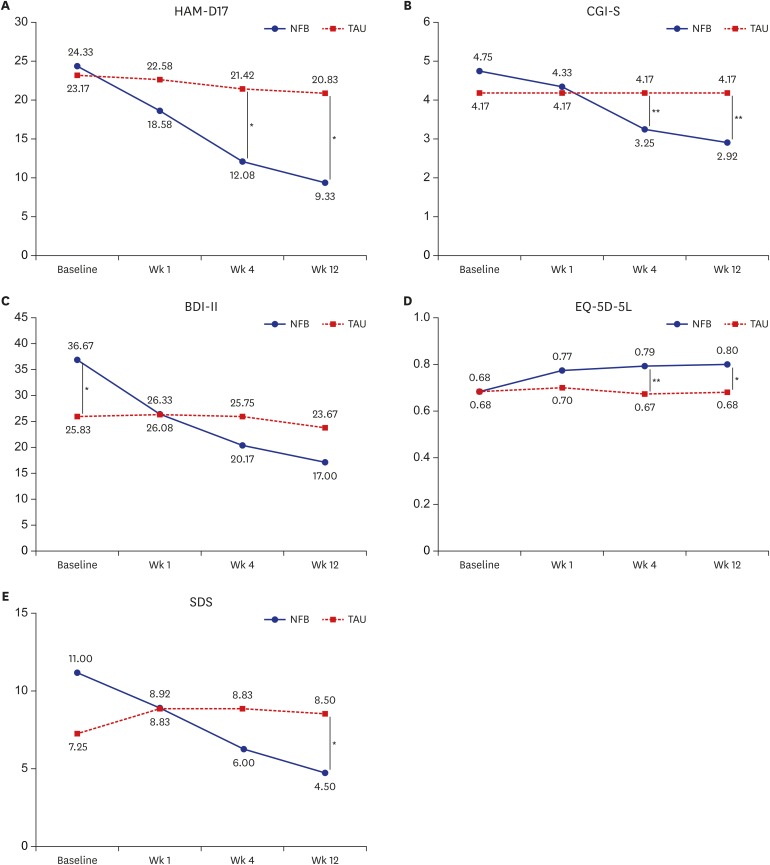
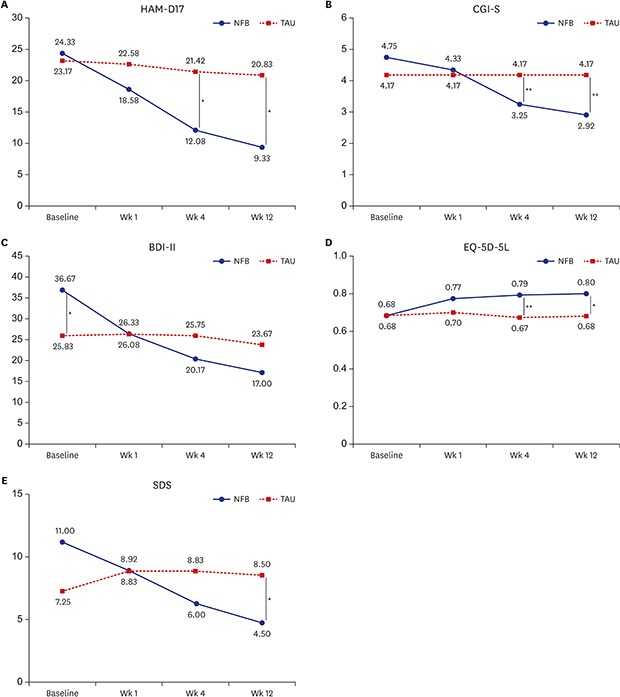
Table 1 shows the characteristics of the participants. There were no significant differences in any of the demographic characteristics between the neurofeedback augmentation group and the medication-only (TAU) group. The baseline mean HAM-D, CGI-S, EQ-5D-5L tariff, and SDS scores were not significantly different between the 2 groups, although there was a difference in the mean BDI-II score (P < 0.05). The mean BDI-II score was significantly higher in the neurofeedback augmentation group than in the medication-only (TAU) group at baseline (Fig. 1).
Table 1. Demographic characteristics of the different groups.
| Variables | NFB (n = 12) | TAU (n = 12) | NOR (n = 12) | F | χ2 | P value | |
|---|---|---|---|---|---|---|---|
| Age, yr | 48.25 ± 14.44 | 54.33 ± 12.67 | 43.50 ± 13.80 | 1.896 | - | 0.166 | |
| Education, yr | 13.58 ± 2.39 | 12.17 ± 4.13 | 15.08 ± 2.46 | 2.653 | - | 0.850 | |
| Gender | - | 0.811 | 0.903 | ||||
| Men | 3 (25.0) | 4 (33.3) | 5 (41.7) | ||||
| Women | 9 (75.0) | 8 (66.7) | 7 (58.3) | ||||
| Residential type | - | 2.860 | 0.314 | ||||
| Alone | 0 | 2 (16.7) | 0 | ||||
| With family | 12 (100.0) | 10 (83.3) | 12 (100) | ||||
| Marital status | - | 3.888 | 0.481 | ||||
| Single | 4 (44.4) | 1 (11.1) | 4 (33.3) | ||||
| Married | 7 (58.3) | 10 (83.3) | 8 (66.7) | ||||
| Separation | 0 | 0 | 0 | ||||
| Divorced | 1 (8.3) | 1 (8.3) | 0 | ||||
| Live together | 0 | 0 | 0 | ||||
| Occupation | - | 11.700 | 0.003 | ||||
| Employed | 2 (16.7) | 4 (33.3) | 10 (83.3) | ||||
| Unemployed | 10 (83.3) | 8 (66.7) | 2 (16.7) | ||||
| Socioeconomic status | - | 1.484 | 0.911 | ||||
| Upper | 3 (25.0) | 4 (33.3) | 3 (25.0) | ||||
| Middle | 5 (41.7) | 5 (41.7) | 7 (58.3) | ||||
| Lower | 4 (33.3) | 3 (25.0) | 2 (16.7) | ||||
| Familial psychiatric illness | - | 4.839 | 0.124 | ||||
| No | 3 (25.0) | 4 (33.3) | 0 | ||||
| Yes | 9 (75.0) | 8 (66.7) | 12 (100.0) | ||||
Values are presented as mean ± standard deviation or number (%).
NFB = neurofeedback augmentation group, TAU = treatment as usual, NOR = normal control.
Fig. 1. Mean HAM-D, CGI-S, BDI-II, EQ-5D-5L tariff, and SDS scores during the study.
HAM-D = Hamilton Depression Rating Scale, NFB = neurofeedback augmentation group, TAU = treatment as usual, CGI-S = Clinical Global Impression-Severity, BDI-II = Beck Depression Inventory-II, EQ-5D-5L = 5-level version of European Quality of Life Questionnaire 5-Dimensional Classification, SDS = Sheehan Disability Scale.
*P < 0.05; **P < 0.01.
Treatment effects
Symptomatic and functional changes
The responses of the depressed patients over time is shown in Fig. 1. The mean HAM-D scores gradually decreased after neurofeedback treatment and the changes of HAM-D score from baseline between the 2 group showed significant differences beginning at week 1 (P < 0.05) (Tables 2 and 3).
Table 2. Changes in HAM-D, CGI-S, BDI-II, EQ-5D-5L, and SDS scores during the study.
| Variables | Visit | NFB (n = 12) | TAU (n = 12) | P valuea | ||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | |||
| HAM-D | Week 1 | −5.75 | 7.15 | −0.58 | 4.25 | 0.043 |
| Week 4 | −12.25 | 12.64 | −1.75 | 7.39 | 0.021 | |
| Week 12 | −15.00 | 8.89 | −2.33 | 7.78 | 0.001 | |
| CGI-S | Week 1 | −0.42 | 0.51 | 0.00 | 0.74 | 0.123 |
| Week 4 | −1.50 | 0.52 | 0.00 | 0.85 | < 0.001 | |
| Week 12 | −1.83 | 0.72 | 0.00 | 0.85 | < 0.001 | |
| BDI-II | Week 1 | −16.83 | 17.77 | 0.25 | 1.76 | 0.003 |
| Week 4 | −19.00 | 12.98 | −0.08 | 3.78 | < 0.001 | |
| Week 12 | −23.17 | 15.16 | −2.17 | 7.03 | < 0.001 | |
| EQ-5D-5L | Week 1 | 0.09 | 0.10 | −0.03 | 0.07 | 0.004 |
| Week 4 | 0.11 | 0.11 | −0.02 | 0.05 | 0.001 | |
| Week 12 | 0.12 | 0.14 | −0.01 | 0.07 | 0.007 | |
| SDS | Week 1 | −3.92 | 5.57 | 1.58 | 2.02 | 0.004 |
| Week 4 | −5.17 | 5.51 | 1.58 | 2.47 | 0.001 | |
| Week 12 | −6.67 | 6.46 | 1.25 | 2.34 | 0.001 | |
HAM-D = Hamilton depression rating scale, CGI-S = clinical global impression-severity, BDI-II = Beck depression inventory-II, EQ-5D-5L = 5-level version of European Quality of Life Questionnaire 5-Dimensional Classification, NFB = neurofeedback augmentation group, TAU = treatment as usual, SD = standard deviation, SDS = Sheehan disability scale.
aP values corrected for multiple comparisons using Bonferroni test.
Table 3. Mean changes in HAM-D, CGI-S, BDI-II, EQ-5D-5L, and SDS scores during the study.
| Variables | Baseline score mean (SD) | LS mean change (SE) | |||
|---|---|---|---|---|---|
| NFB (n = 12) | TAU (n = 12) | NFB (n = 12) | TAU (n = 12) | P valuea | |
| HAM-D | 24.33 (5.77) | 23.17 (5.42) | −15.00 (2.57) | −2.33 (2.25) | 0.001 |
| CGI-S | 4.75 (0.62) | 4.17 (0.83) | −1.83 (0.21) | 0.00 (0.25) | < 0.001 |
| BDI-II | 36.67 (14.79) | 25.83 (7.99) | −23.17 (4.38) | −2.17 (2.03) | < 0.001 |
| EQ-5D-5L | 0.68 (0.16) | 0.68 (0.16) | 0.12 (0.14) | −0.01 (0.07) | 0.008 |
| SDS | 11.17 (7.09) | 7.25 (4.47) | −6.67 (1.86) | 1.25 (0.68) | < 0.001 |
HAM-D = Hamilton depression rating scale, CGI-S = clinical global impression-severity, BDI-II = Beck depression inventory-II, EQ-5D-5L = 5-level version of European Quality of Life Questionnaire 5-Dimensional Classification, SDS = Sheehan disability scale, SD = standard deviation, LS = least square, SE = standard error, NFB = neurofeedback augmentation group, TAU = treatment as usual, SE = standard error.
aNonparametric test used without adjustment of P values for multiple comparisons.
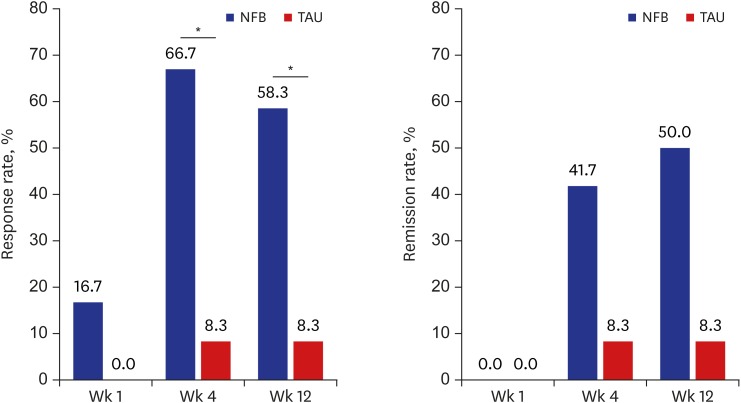
Seven of the 12 (58.3%) neurofeedback-treated individuals and 1 of the 12 (8.3%) medication-only (TAU)-treated individuals were rated as having a “response” at study endpoint (week 12) based on the total HAM-D score. There was a significant difference in the response rate between the 2 groups beginning at week 4 (P < 0.05) (Fig. 2).
Fig. 2. Response and remission rates at each visit obtained using the HAM-D 17 total score during treatment with NFB or TAU.
NFB = neurofeedback augmentation group, TAU = treatment as usual, HAM-D = Hamilton Depression Rating Scale.
*P < 0.05.
There were gradual improvements from baseline in the CGI-S score and the BDI-II score, only in the neurofeedback augmentation group. There were significant differences in the changes in the BDI-II score (beginning at week 1) and in the CGI-S score (beginning at week 4) between the 2 groups (Tables 2, 3 and Fig. 1).
There was a significant separation of the neurofeedback augmentation group and the medication-only (TAU) group in terms of the EQ-5D-5L tariff score beginning at week 4 and the SDS score at week 12 (P < 0.05) (Fig. 1). These changes indicate improvements in quality of life and daily function. There were significant differences in the changes in the EQ-5D-5L tariff and SDS scores between the neurofeedback augmentation group and the medication-only (TAU) group beginning at week 1 (P < 0.05) (Tables 2 and 3).
BDNF
There were no significant differences in the baseline serum BDNF level among the three groups (the neurofeedback augmentation group, medication-only group, and healthy controls). In the neurofeedback augmentation group, the average serum BDNF level was decreased from 27,083.61 pg/mL (standard error [SE], 2,363.12) at baseline to 23,311.92 pg/mL (SE, 2,620.16) at week 12. In the medication-only group, the average serum BDNF level was increased from 23,681.72 pg/mL (SE, 2,701.51) at baseline to 27,593.64 pg/mL (SE, 3,702.28) at week 12. No significant difference was found between the pre- and post-treatment serum BDNF levels in the different groups (Table 4).
Table 4. Comparisons of BDNF level between baseline and the 12-week time point among groups.
| Groups | Baseline score | 12-week score | LS mean change | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean | SE | P valuea | P valueb | Mean | SE | P valueb | Mean | SE | P valueb | |
| NFB (n = 12) | 27,083.61 | 2,363.12 | 0.099 | 0.170 | 23,311.92 | 2,620.16 | 0.296 | −3,771.69 | 2,017.56 | 0.022 |
| TAU (n = 7) | 23,681.72 | 2,701.51 | 0.484 | 0.170 | 27,593.64 | 3,702.28 | 0.296 | 3,911.93 | 1,909.04 | 0.022 |
| NOR (n = 12) | 23,492.47 | 1,375.38 | - | - | - | - | - | - | - | - |
BDNF = brain-derived neurotropic factor, SE = standard error, NFB = neurofeedback augmentation group, TAU = treatment as usual, NOR = normal control.
aP value of nonparametric test among NFB and NOR; bP value of nonparametric test between NFB and TAU (P values not adjusted for multiple comparisons).
Safety
There were no serious adverse events. In the medication-only (TAU) group, 6 patients reported discomfort due to dry mouth (4 patients), headache (2), dizziness (2), fatigue (2), constipation (1), and nausea (1). In neurofeedback augmentation group, 2 patients reported discomfort due to tinnitus and urinary incontinence. There was no significant difference in the occurrence of side effects between the 2 groups.
DISCUSSION
This study is the first prospective controlled study to investigate the therapeutic effects of and functional recovery due to neurofeedback in patients with TRD. It is also the first study to investigate the relationship between serum BDNF as a biomarker and therapeutic response following neurofeedback.
We found subjective and objective depressive symptom relief and functional improvement after 12 weeks of neurofeedback training in patients with TRD. The HAM-D and CGI-S scores were significantly decreased in the neurofeedback augmentation group when compared to those in the medication-only (TAU) group. This indicates that objective depressive symptoms and depression severity were further improved using neurofeedback when compared to medication-only (TAU). The response and remission rates for medications used for the treatment of depressive disorder are 50%–70% and 30%, respectively. The 12- and 24-month response rates of TRD have been reported to be 11.6% and 18.4%, respectively, and the 12- and 24-month remission rates of TRD have been reported to be 3.6% and 7.8%, respectively.47 However, in this study, the response and remission rates were 58.3% and 50.0%, respectively, in the neurofeedback augmentation group. These rates were significantly higher than those in the medication-only (TAU) group. These results support the finding that neurofeedback was effective in improving depressive symptoms in a preliminary study conducted by Cheon et al.29
As indicators of quality of life and functional recovery, the SDS and EQ-5D-5L tariff scores were significantly lower in the neurofeedback augmentation group than in the medication-only (TAU) group. Our findings may indicate that neurofeedback is effective not only for symptom improvement, but also for functional recovery. Neurofeedback has been shown to improve executive function in depressed patients21 and has been shown to improve performance (peak performance) in healthy individuals.48 It is also an active training program in which the individual can restore the regulation of the brain network, spontaneously, and it might be helpful for patients to improve their self efficacy.49 Mitigating depressive symptoms itself may have facilitated recovery of function. Further research is needed to determine the mechanism by which neurofeedback helped to restore function in depressed patients.
There was no significant difference between the 2 groups in the occurrence of adverse events. All reported adverse events in the neurofeedback augmentation group were identified as unrelated to neurofeedback and were presumed to be related to the medication used or the patient’s medical condition. There were no dropouts among the 12 patients in the neurofeedback augmentation group or the 12 in the medication-only (TAU) group. Neurofeedback combined with medication thus seems to be beneficial in terms of adverse effects and compliance, as well as its therapeutic effects.
This study was based on the neurofeedback protocol of Cheon et al.29,50 The patients were evaluated during weekly neurofeedback team meetings with 3 psychiatrists and a neurofeedback therapist. The neurofeedback protocol was determined by psychiatrists certified in neurofeedback during the neurofeedback team meeting for each patient. In the current study, the neurofeedback protocol was not uniform, but individualized. The patient's most serious symptoms were considered for preferential treatment, and the protocol was discussed and adjusted during weekly neurofeedback meetings. For depressed patients, correcting any abnormal pattern of asymmetric activity in the frontal regions and recovering balance in their brain activity played important roles in reducing depressive symptoms regardless of whether which treatment method was employed.
Our previous study showed that left frontal beta training and alpha/theta training could improve depressive symptoms.29 Individualized symptom targeted protocol (left frontal beta or right frontal SMR) also improved depression and anxiety symptoms.50 A recent review article19 categorized the neurofeedback treatment protocol of depression into 3 categories. The first protocol is the asymmetric left frontal activation method.51,52,53,54,55,56 In EEG studies of depression, an abnormal pattern of asymmetric activity in frontal regions resulting from relative hyperactivity over the right frontal regions and/or relative hypoactivity over the left frontal regions has frequently been observed.52 Choi et al.21 reported that the therapeutic effects of neurofeedback may be due to relative weakening of right frontal activity and strengthening of asymmetric left frontal activity.21,48,57,58,59 The second protocol is the reduction of theta activity (4–8 Hz) in relation to beta (15–28) in the left prefrontal cortex. This approach is consistent with other neuromodulation treatment modalities, rTMS, which has a therapeutic effect by controlling frontal function in depression. In other words, high-frequency rTMS applied to the left dorsolateral prefrontal cortex, is an effective treatment for patients with major depressive disorder. Other forms of stimulation, such as low-frequency stimulation applied to the right prefrontal cortex is also proved as effective treatment.60 Beta activity is related to executive and motivational functions, both of which are negatively affected in depression, possibly in association with prefrontal hypo-activation.22
Several studies have reported that left frontal beta training had a more direct effect in brain areas related to depressive disorder, and that beta or SMR training combined with A/T training was effective in alleviating depressive symptoms.61,62 The preliminary study conducted by Cheon et al.,29 reported that beta training in the F3 (T3) region or SMR training in the T4 region and A/T training resulted in a reduction in depressive symptoms. Those results are consistent with the results of the current study. The treatment methods belonging to the third category is alpha/theta protocol which is indirectly related to the treatment of depression. The efficacy of alpha theta neurofeedback may lie in its ability to allow participants to deal with anxiety and anxiety-eliciting situations,63 It has also been suggested that neurofeedback targeting lower frequencies such as alpha/theta may directly affect core neurocognitive networks, and thereby produce widespread symptom improvements,14 Neuroanatomical circuitry involves the ascending mescencephalic-cortical arousal system, and limbic circuits subserving cognitive as well as affective/motivational functions, and including coupling between frontal and posterior cortices, exemplifying a role for theta and alpha waves in mediating the interaction between distal and widely distributed connections.64
We hypothesized that the clinical improvement due to neurofeedback was related to brain differentiation, and expected that the serum BDNF level would be normalized after neurofeedback treatment. However, there was no significant difference in BDNF level pre- vs. post-treatment in either group. The BDNF results should be considered in light of the fact that they were obtained in 3 small samples of 12 patients. The therapeutic effects of neurofeedback were confirmed even though we used 3 different protocols for training. However, we were unable to obtain serum BDNF results consistent with the above finding due to differences in brain activity regulation in the different brain regions. Because brain BDNF is mainly produced in the hippocampus, serum BDNF might be inappropriate as a biomarker of depression if the depressive symptoms were improved by frontal activation through neurofeedback. In other words, it is possible that the improvements in depressive symptoms due to neurofeedback were not directly related to brain plasticity. We should thus consider whether BDNF is appropriate as a biomarker of depression. Bus et al.65 have also reported that changes in the levels of serum, plasma, or whole blood BDNF may not reflect changes in the levels of BDNF in the brain. In animals, the level of BDNF in the brain has been reported to be positively associated with that of BDNF in the blood,66 However, in humans, such an association is as yet unclear. Whether BDNF in peripheral blood samples (plasma, serum, etc.) reflects the pathophysiology of psychological illness remains controversial. BDNF signaling is significantly reduced in the hippocampus and prefrontal cortex in depressed persons. However, it is increased in the nucleus accumbens and amygdala in such individuals,67 Therefore, peripheral BDNF levels may not reflect the levels of BDNF in all regions of the brain.
This study has several limitations. First, it had a small sample size. Inclusion of more participants would allow the discovery of more clinically important findings. Second, confounding factors influencing the BDNF level, such as use of various medications, stress, and durations of the depressive episode, depression, and treatment were not adequately controlled for. Additional studies on the relation between neurofeedback and BDNF as a biomarker in patients with TRD are warranted.
Third, performing the study completely double-blind was difficult because 2 different treatment modalities were used. We did not design sham neurofeedback training as an ethical issue. By allowing the patient to choose the treatment method, we may have affected the study results. Additional studies with a sham randomized controlled double-blind design are recommended to overcome this limitation.
Despite these limitations, our results suggest that neurofeedback might be a beneficial augmentation treatment to make nonresponding patients with depression feel well again and successfully engage with life. In the future, additional studies in large populations will be required to replicate these results. In addition, long-term follow-up studies of patients with improvements and remission after neurofeedback treatment will be needed to evaluate persistence, safety, and relapse in patients administered neurofeedback treatment.
ACKNOWLEDGMENTS
This study was presented at the poster section of the 30th European College of Neuropsychopharmacology (Paris, 3rd September, 2017).
Footnotes
Funding: This work was supported by the 2016 Yeungnam University Research Grant.
Disclosure: The authors have no potential conflicts of interest to disclose.
- Conceptualization: Cheon EJ.
- Data curation: Lee YJ, Lee GW, Seo WS, Koo BH, Kim HG, Cheon EJ.
- Formal analysis: Lee YJ, Lee GW.
- Investigation: Lee GW.
- Methodology: Lee YJ, Cheon EJ.
- Project administration: Cheon EJ.
- Software: Lee YJ, Lee GW.
- Validation: Lee YJ.
- Writing - original draft: Lee YJ, Lee GW.
- Writing - review & editing: Lee YJ, Kim HG, Seo WS, Koo BH, Cheon EJ.
References
- 1.American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-IV-TR. Washington, DC: American Psychiatric Pub; 2000. [Google Scholar]
- 2.Lépine JP, Briley M. The increasing burden of depression. Neuropsychiatr Dis Treat. 2011;7(Suppl 1):3–7. doi: 10.2147/NDT.S19617. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®) Washington, DC: American Psychiatric Pub; 2013. [Google Scholar]
- 4.Henderson M, Harvey SB, Øverland S, Mykletun A, Hotopf M. Work and common psychiatric disorders. J R Soc Med. 2011;104(5):198–207. doi: 10.1258/jrsm.2011.100231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Crown WH, Finkelstein S, Berndt ER, Ling D, Poret AW, Rush AJ, et al. The impact of treatment-resistant depression on health care utilization and costs. J Clin Psychiatry. 2002;63(11):963–971. doi: 10.4088/jcp.v63n1102. [DOI] [PubMed] [Google Scholar]
- 6.McKnight PE, Kashdan TB. The importance of functional impairment to mental health outcomes: a case for reassessing our goals in depression treatment research. Clin Psychol Rev. 2009;29(3):243–259. doi: 10.1016/j.cpr.2009.01.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shagass G. Electrical activity of the brain. In: Greenfield NS, Stembach RA, editors. Handbook of Psychophysiology. New York, NY: Holt, Rinehart and Winston; 1972. pp. 263–328. [Google Scholar]
- 8.Hughes JR, John ER. Conventional and quantitative electroencephalography in psychiatry. J Neuropsychiatry Clin Neurosci. 1999;11(2):190–208. doi: 10.1176/jnp.11.2.190. [DOI] [PubMed] [Google Scholar]
- 9.Lubar JF. Discourse on the development of EEG diagnostics and biofeedback for attention-deficit/hyperactivity disorders. Biofeedback Self Regul. 1991;16(3):201–225. doi: 10.1007/BF01000016. [DOI] [PubMed] [Google Scholar]
- 10.Cowan J, Allen T. Using brainwave biofeedback to train the sequence of concentration and relaxation in athletic activities; Proceedings of 15th Association for the Advancement of Applied Sport Psychology; 2000. p. 95. [Google Scholar]
- 11.Aftanas LI, Golocheikine SA. Human anterior and frontal midline theta and lower alpha reflect emotionally positive state and internalized attention: high-resolution EEG investigation of meditation. Neurosci Lett. 2001;310(1):57–60. doi: 10.1016/s0304-3940(01)02094-8. [DOI] [PubMed] [Google Scholar]
- 12.Vernon D, Egner T, Cooper N, Compton T, Neilands C, Sheri A, et al. The effect of training distinct neurofeedback protocols on aspects of cognitive performance. Int J Psychophysiol. 2003;47(1):75–85. doi: 10.1016/s0167-8760(02)00091-0. [DOI] [PubMed] [Google Scholar]
- 13.Hammond DC. Neurofeedback with anxiety and affective disorders. Child Adolesc Psychiatr Clin N Am. 2005;14(1):105–123. doi: 10.1016/j.chc.2004.07.008. [DOI] [PubMed] [Google Scholar]
- 14.Niv S. Clinical efficacy and potential mechanisms of neurofeedback. Pers Individ Dif. 2013;54(6):676–686. [Google Scholar]
- 15.Cheon E. Neurofeedback treatment in adult psychiatric patient: focusing on depressive and anxiety disorder. J Korean Soc Biol Ther Psychiatry. 2013;19(2):85–92. [Google Scholar]
- 16.Egner T, Zech TF, Gruzelier JH. The effects of neurofeedback training on the spectral topography of the electroencephalogram. Clin Neurophysiol. 2004;115(11):2452–2460. doi: 10.1016/j.clinph.2004.05.033. [DOI] [PubMed] [Google Scholar]
- 17.Lévesque J, Beauregard M, Mensour B. Effect of neurofeedback training on the neural substrates of selective attention in children with attention-deficit/hyperactivity disorder: a functional magnetic resonance imaging study. Neurosci Lett. 2006;394(3):216–221. doi: 10.1016/j.neulet.2005.10.100. [DOI] [PubMed] [Google Scholar]
- 18.Ros T, Munneke MA, Ruge D, Gruzelier JH, Rothwell JC. Endogenous control of waking brain rhythms induces neuroplasticity in humans. Eur J Neurosci. 2010;31(4):770–778. doi: 10.1111/j.1460-9568.2010.07100.x. [DOI] [PubMed] [Google Scholar]
- 19.Dias AM, van Deusen A. A new neurofeedback protocol for depression. Span J Psychol. 2011;14(1):374–384. doi: 10.5209/rev_sjop.2011.v14.n1.34. [DOI] [PubMed] [Google Scholar]
- 20.Rogel A, Guez J, Getter N, Keha E, Cohen T, Amor T, et al. Transient adverse side effects during neurofeedback training: a randomized, sham-controlled, double blind study. Appl Psychophysiol Biofeedback. 2015;40(3):209–218. doi: 10.1007/s10484-015-9289-6. [DOI] [PubMed] [Google Scholar]
- 21.Choi SW, Chi SE, Chung SY, Kim JW, Ahn CY, Kim HT. Is alpha wave neurofeedback effective with randomized clinical trials in depression? A pilot study. Neuropsychobiology. 2011;63(1):43–51. doi: 10.1159/000322290. [DOI] [PubMed] [Google Scholar]
- 22.DeRubeis RJ, Siegle GJ, Hollon SD. Cognitive therapy versus medication for depression: treatment outcomes and neural mechanisms. Nat Rev Neurosci. 2008;9(10):788–796. doi: 10.1038/nrn2345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Karege F, Perret G, Bondolfi G, Schwald M, Bertschy G, Aubry JM. Decreased serum brain-derived neurotrophic factor levels in major depressed patients. Psychiatry Res. 2002;109(2):143–148. doi: 10.1016/s0165-1781(02)00005-7. [DOI] [PubMed] [Google Scholar]
- 24.Shimizu E, Hashimoto K, Okamura N, Koike K, Komatsu N, Kumakiri C, et al. Alterations of serum levels of brain-derived neurotrophic factor (BDNF) in depressed patients with or without antidepressants. Biol Psychiatry. 2003;54(1):70–75. doi: 10.1016/s0006-3223(03)00181-1. [DOI] [PubMed] [Google Scholar]
- 25.Zhou Z, Lu T, Xu G, Yue X, Zhu W, Ma M, et al. Decreased serum brain-derived neurotrophic factor (BDNF) is associated with post-stroke depression but not with BDNF gene Val66Met polymorphism. Clin Chem Lab Med. 2011;49(2):185–189. doi: 10.1515/CCLM.2011.039. [DOI] [PubMed] [Google Scholar]
- 26.Fava M, Davidson KG. Definition and epidemiology of treatment-resistant depression. Psychiatr Clin North Am. 1996;19(2):179–200. doi: 10.1016/s0193-953x(05)70283-5. [DOI] [PubMed] [Google Scholar]
- 27.Fava M. Diagnosis and definition of treatment-resistant depression. Biol Psychiatry. 2003;53(8):649–659. doi: 10.1016/s0006-3223(03)00231-2. [DOI] [PubMed] [Google Scholar]
- 28.Quitkin FM, Rabkin JG, Stewart JW, McGrath PJ, Harrison W. Study duration in antidepressant research: advantages of a 12-week trial. J Psychiatr Res. 1986;20(3):211–216. doi: 10.1016/0022-3956(86)90004-x. [DOI] [PubMed] [Google Scholar]
- 29.Cheon EJ, Koo BH, Choi JH. The efficacy of neurofeedback in patients with major depressive disorder: an open labeled prospective study. Appl Psychophysiol Biofeedback. 2016;41(1):103–110. doi: 10.1007/s10484-015-9315-8. [DOI] [PubMed] [Google Scholar]
- 30.Raymond J, Varney C, Parkinson LA, Gruzelier JH. The effects of alpha/theta neurofeedback on personality and mood. Brain Res Cogn Brain Res. 2005;23(2-3):287–292. doi: 10.1016/j.cogbrainres.2004.10.023. [DOI] [PubMed] [Google Scholar]
- 31.Hamilton M. The hamilton depression rating scale. J Neurol Neurosurg Psychiatry. 1960;23:56–62. doi: 10.1136/jnnp.23.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yi JS, Bae SO, Ahn YM, Park DB, Noh KS, Shin HK, et al. Validity and reliability of the Korean version of the Hamilton Depression Rating Scale (K-HDRS) J Korean Neuropsychiatr Assoc. 2005;44(4):456–465. [Google Scholar]
- 33.Beck AT, Steer RA, Carbin MG. Psychometric properties of the Beck Depression Inventory: twenty-five years of evaluation. Clin Psychol Rev. 1988;8(1):77–100. [Google Scholar]
- 34.Beck A, Ward C, Mendelson M, Mock J, Erbaugh J. An inventory for measuring depression. Arch Gen Psychiatry. 1961;4:561–571. doi: 10.1001/archpsyc.1961.01710120031004. [DOI] [PubMed] [Google Scholar]
- 35.Rhee M, Lee Y, Park S, Sohn C, Jung Y, Hong S, et al. A standardization study of beck depression inventory (I): Korean version (K-BDI): reliability land factor analysis. Korean J Psychopathol. 1995;4(1):77–95. [Google Scholar]
- 36.Busner J, Targum SD. The clinical global impressions scale: applying a research tool in clinical practice. Psychiatry (Edgmont Pa) 2007;4(7):28–37. [PMC free article] [PubMed] [Google Scholar]
- 37.Busner J, Targum SD, Miller DS. The Clinical Global Impressions scale: errors in understanding and use. Compr Psychiatry. 2009;50(3):257–262. doi: 10.1016/j.comppsych.2008.08.005. [DOI] [PubMed] [Google Scholar]
- 38.EuroQol Group. EuroQol--a new facility for the measurement of health-related quality of life. Health Policy. 1990;16(3):199–208. doi: 10.1016/0168-8510(90)90421-9. [DOI] [PubMed] [Google Scholar]
- 39.Kim MH, Cho YS, Uhm WS, Kim S, Bae SC. Cross-cultural adaptation and validation of the Korean version of the EQ-5D in patients with rheumatic diseases. Qual Life Res. 2005;14(5):1401–1406. doi: 10.1007/s11136-004-5681-z. [DOI] [PubMed] [Google Scholar]
- 40.van Reenen M, Janssen B. EQ-5D-5L User Guide: Basic Information on How to Use the EQ-5D-5L Instrument. Rotterdam: EuroQol Research Foundation; 2015. [Google Scholar]
- 41.Lamers LM, McDonnell J, Stalmeier PF, Krabbe PF, Busschbach JJ. The Dutch tariff: results and arguments for an effective design for national EQ-5D valuation studies. Health Econ. 2006;15(10):1121–1132. doi: 10.1002/hec.1124. [DOI] [PubMed] [Google Scholar]
- 42.Brooks R, Rabin R, De Charro F. The Measurement and Valuation of Health Status Using EQ-5D: a European Perspective: Evidence from the EuroQol BIOMED Research Programme. Berlin: Springer Science & Business Media; 2013. [Google Scholar]
- 43.Kim SH, Ahn J, Ock M, Shin S, Park J, Luo N, et al. The EQ-5D-5L valuation study in Korea. Qual Life Res. 2016;25(7):1845–1852. doi: 10.1007/s11136-015-1205-2. [DOI] [PubMed] [Google Scholar]
- 44.Sheehan DV. The Anxiety Disease: New Hope for the Millions Who Suffer from Anxiety. New York, NY: Scribner; 1983. [Google Scholar]
- 45.Park J, Kim J. Korean version of the Sheehan disability scale (SDS): reilability and validity. Korean J Clin Psychol. 2010;29(1):73–81. [Google Scholar]
- 46.Zimmerman M, Chelminski I, Posternak M. A review of studies of the Montgomery-Asberg Depression Rating Scale in controls: implications for the definition of remission in treatment studies of depression. Int Clin Psychopharmacol. 2004;19(1):1–7. doi: 10.1097/00004850-200401000-00001. [DOI] [PubMed] [Google Scholar]
- 47.Dunner DL, Rush AJ, Russell JM, Burke M, Woodard S, Wingard P, et al. Prospective, long-term, multicenter study of the naturalistic outcomes of patients with treatment-resistant depression. J Clin Psychiatry. 2006;67(5):688–695. doi: 10.4088/jcp.v67n0501. [DOI] [PubMed] [Google Scholar]
- 48.Vernon DJ. Can neurofeedback training enhance performance? An evaluation of the evidence with implications for future research. Appl Psychophysiol Biofeedback. 2005;30(4):347–364. doi: 10.1007/s10484-005-8421-4. [DOI] [PubMed] [Google Scholar]
- 49.French DJ, Gauthier JG, Roberge C, Bouchard S, Nouwen A. Self-efficacy in the thermal biofeedback treatment of migraine sufferers. Behav Ther. 1997;28(1):109–125. [Google Scholar]
- 50.Cheon EJ, Koo BH, Seo WS, Lee JY, Choi JH, Song SH. Effects of neurofeedback on adult patients with psychiatric disorders in a naturalistic setting. Appl Psychophysiol Biofeedback. 2015;40(1):17–24. doi: 10.1007/s10484-015-9269-x. [DOI] [PubMed] [Google Scholar]
- 51.Reischies FM, Hedde JP, Drochner R. Clinical correlates of cerebral blood flow in depression. Psychiatry Res. 1989;29(3):323–326. doi: 10.1016/0165-1781(89)90078-4. [DOI] [PubMed] [Google Scholar]
- 52.Henriques JB, Davidson RJ. Regional brain electrical asymmetries discriminate between previously depressed and healthy control subjects. J Abnorm Psychol. 1990;99(1):22–31. doi: 10.1037//0021-843x.99.1.22. [DOI] [PubMed] [Google Scholar]
- 53.Tomarken AJ, Davidson RJ, Henriques JB. Resting frontal brain asymmetry predicts affective responses to films. J Pers Soc Psychol. 1990;59(4):791–801. doi: 10.1037//0022-3514.59.4.791. [DOI] [PubMed] [Google Scholar]
- 54.Bench CJ, Friston KJ, Brown RG, Scott LC, Frackowiak RS, Dolan RJ. The anatomy of melancholia--focal abnormalities of cerebral blood flow in major depression. Psychol Med. 1992;22(3):607–615. doi: 10.1017/s003329170003806x. [DOI] [PubMed] [Google Scholar]
- 55.Mayberg HS, Liotti M, Brannan SK, McGinnis S, Mahurin RK, Jerabek PA, et al. Reciprocal limbic-cortical function and negative mood: converging PET findings in depression and normal sadness. Am J Psychiatry. 1999;156(5):675–682. doi: 10.1176/ajp.156.5.675. [DOI] [PubMed] [Google Scholar]
- 56.Weiskopf N, Scharnowski F, Veit R, Goebel R, Birbaumer N, Mathiak K. Self-regulation of local brain activity using real-time functional magnetic resonance imaging (fMRI) J Physiol Paris. 2004;98(4-6):357–373. doi: 10.1016/j.jphysparis.2005.09.019. [DOI] [PubMed] [Google Scholar]
- 57.Plotkin WP, Rice KM. Biofeedback as a placebo: anxiety reduction facilitated by training in either suppression or enhancement of alpha brainwaves. J Consult Clin Psychol. 1981;49(4):590–596. doi: 10.1037//0022-006x.49.4.590. [DOI] [PubMed] [Google Scholar]
- 58.Thornton K. Improvement/rehabilitation of memory functioning with neurotherapy/QEEG biofeedback. J Head Trauma Rehabil. 2000;15(6):1285–1296. doi: 10.1097/00001199-200012000-00008. [DOI] [PubMed] [Google Scholar]
- 59.Linden DE. Neurofeedback and networks of depression. Dialogues Clin Neurosci. 2014;16(1):103–112. doi: 10.31887/DCNS.2014.16.1/dlinden. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Fitzgerald PB, Daskalakis ZJ. A practical guide to the use of repetitive transcranial magnetic stimulation in the treatment of depression. Brain Stimulat. 2012;5(3):287–296. doi: 10.1016/j.brs.2011.03.006. [DOI] [PubMed] [Google Scholar]
- 61.Scott WC, Kaiser D, Othmer S, Sideroff SI. Effects of an EEG biofeedback protocol on a mixed substance abusing population. Am J Drug Alcohol Abuse. 2005;31(3):455–469. doi: 10.1081/ada-200056807. [DOI] [PubMed] [Google Scholar]
- 62.Walker J, Lawson R, Kozlowski G. Current status of QEEG and neurofeedback in the treatment of depression. In: Evans J, editor. Handbook of Neurofeedback. Binghampton, NY: Haworth Medical Press; 2007. p. 343. [Google Scholar]
- 63.Peniston EG, Kulkosky PJ. Alpha-theta brainwave training and beta-endorphin levels in alcoholics. Alcohol Clin Exp Res. 1989;13(2):271–279. doi: 10.1111/j.1530-0277.1989.tb00325.x. [DOI] [PubMed] [Google Scholar]
- 64.Gruzelier J. A theory of alpha/theta neurofeedback, creative performance enhancement, long distance functional connectivity and psychological integration. Cogn Process. 2009;10(Suppl 1):S101–S109. doi: 10.1007/s10339-008-0248-5. [DOI] [PubMed] [Google Scholar]
- 65.Bus BA, Molendijk ML, Penninx BJ, Buitelaar JK, Kenis G, Prickaerts J, et al. Determinants of serum brain-derived neurotrophic factor. Psychoneuroendocrinology. 2011;36(2):228–239. doi: 10.1016/j.psyneuen.2010.07.013. [DOI] [PubMed] [Google Scholar]
- 66.Karege F, Schwald M, Cisse M. Postnatal developmental profile of brain-derived neurotrophic factor in rat brain and platelets. Neurosci Lett. 2002;328(3):261–264. doi: 10.1016/s0304-3940(02)00529-3. [DOI] [PubMed] [Google Scholar]
- 67.Yu H, Chen ZY. The role of BDNF in depression on the basis of its location in the neural circuitry. Acta Pharmacol Sin. 2011;32(1):3–11. doi: 10.1038/aps.2010.184. [DOI] [PMC free article] [PubMed] [Google Scholar]