Abstract
Diabetes mellitus is a rising epidemic in most part of the world and is often associated with multiple organ disorders such as kidney, liver, and cardiovascular diseases. Liver is a major metabolic hub, and the metabolic disorders associated with diabetes result in liver dysfunctions culminating in spectrum of liver diseases such as fatty liver disorders, cirrhosis, and hepatocellular carcinoma. The intervention strategies to prevent diabetes-associated liver injury require an overall understanding of the key factors and molecular pathways which can be strategically targeted. The present review focuses on some of the key aspects of fatty acid metabolism, fetuin-A regulation, inflammatory pathways, and genetic factors associated with insulin resistance, dyslipidemia, hyperglycemia, oxidative stress, and so on involved in the nexus between diabetes and liver injury. Further recent interventions, pharmacological target, and newer therapeutic agents are discussed briefly for the better clinical management of diabetes-associated hepatic disorders.
Keywords: free fatty acid, fetuin-A, inflammatory mediators, diabetic liver injury, diabetes mellitus
Abbreviations: AP-1, Activator protein 1; DLI, diabetic liver injury; DM, diabetes mellitus; DMPs, Damage-associated molecular patterns; FetA, fetuin-A; FFA, free fatty acid; FOXO1, Forkhead box protein O1; G6Pase, Glucose-6-phosphatase; HCC, hepatocellular carcinoma; IκB, Inhibitor of Kb; IKK, IκB kinase; IL, interleukin; IRS2, Insulin receptor substrate-2; LPS, Lipopolysaccharide; MD2, Myeloid differentiation protein-2; MMP, matrix metalloproteinase; MyD88, Myeloid differentiation factor 88; NAFLD, nonalcoholic fatty liver disease; NASH, nonalcoholic steatohepatitis; NIDDM, noninsulin dependent diabetes mellitus; NFe κB, Nuclear factor-κB; PC, Pyruvate carboxylase; PEPCK, Phosphoenolpyruvate carboxykinase; PIP3, Phosphatidyl inositol (3, 4, 5)-triphosphate; T2DM, type 2 diabetes mellitus; TNF, tumor necrosis factor; Th 17, T helper 17 cells; TLR4, Toll-like receptor; VLDL, very low–density lipoprotein
Graphical abstract
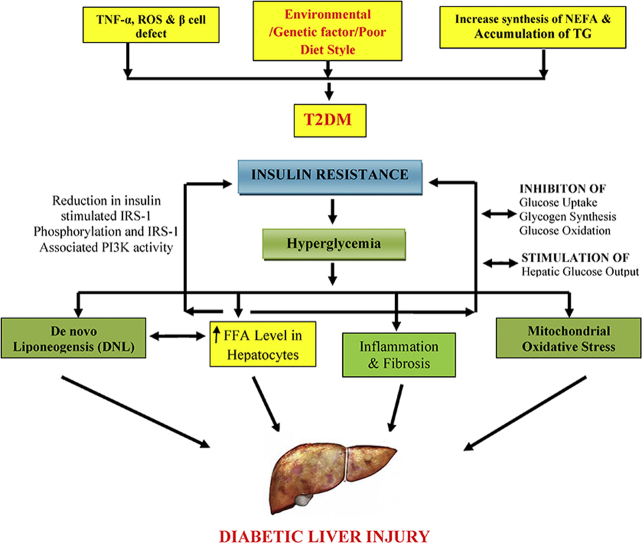
Illustration of linking between free fatty acid with diabetic liver injury [FFA: free fatty acid; ROS: reactive oxygen species; TNF: tumor necrosis factor; IRS-1: insulin receptor substrate 1; PI3K: phosphoinositide 3-kinase; DNL: de novo liponeogensis (conversion of excess carbohydrate into fatty acids); TG: triglycerides; T2DM: type 2 diabetes mellitus; NEFA: nonesterified fatty acid; +: positive stimulation].
Highlights
-
•
The vicious cycle of diabetic liver injury.
-
•
Elevated levels of plasma Free Fatty Acid play a key role in the pathogenesis of diabetes.
-
•
Insulin resistance causes hyperglycemia and hyperinsulinemia which leads to complications of liver injury.
-
•
Other highlight of this review is about recent interventions which combat diabetes- associated hepatic disorders.
Diabetes mellitus (DM) is a growing epidemic worldwide. The World Health Organization (WHO) reported the burden of adults with diabetes to be 422 million in 2016 as compared with 108 million in 1980 globally. The global prevalence (age-standardized) of diabetes has nearly doubled since 1980, rising from 4.7% to 8.5% in the adult population. Similar pattern was observed all over Asia with an increase in frequency of diabetic adults from 4.1% to 8.6% due to changes in life style in the span of 20 years. The WHO also reported that blood glucose age-standardized mortality rates per 100000 in the Southeast Asian region were 115.3 times higher irrespective of gender bias.1 Other associated disorders such as end-stage renal diseases, retinopathy, cardiovascular, and liver disorders were found to be 12%–55%, 2.6%, 40%, and 12.3%–57%, respectively in diabetic patients. It is reported that the global prevalence of nonalcoholic fatty liver disease (NAFLD) ranges from 50% to 75% in patients with type 2 diabetes mellitus (T2DM),2, 3 whereas in India, this range is in fact wider ranging from 12.5% to 87.5%.4 Furthermore, more than 90% of obese patients with T2DM are strongly associated with NAFLD.5
The metabolic defects associated with diabetes often result in altered liver metabolism resulting in hepatotoxicity and cell death. Although liver has a good regenerative potential, long-standing diabetes can manifest in a spectrum of liver diseases which include altered liver functions, nonalcoholic fatty liver, end-stage liver disease or cirrhosis, liver failure, and even hepatocellular carcinoma (HCC).6, 7
NAFLD has been recognized as a common complication in patients with T2DM.5, 8 However, the pathogenesis of T2DM involves strong association with a liver injury which is collectively termed as diabetic liver injury (DLI) with reference to insulin resistance.9 Insulin resistance, hepatocytes injury, and free fatty acid (FFA) are the major culprits which are involved in the pathogeneses of DLI. Most of the ingested or synthesized fatty acids have two fates, either they incorporate into triacylglycerols for the storage of metabolic energy or get incorporated into the phospholipid component of the membrane depending upon the organism's need.10 Once there is a constant elevation of FFA, it leads to inhibition of glucose uptake, glycogen synthesis, glucose oxidation, and increase in hepatic glucose output which produces the state of insulin resistance resulting in reduction of insulin-stimulated insulin receptor substrate 1(IRS-1) phosphorylation and its associated PI3K activity.11 This, in turn, leads to suppression of hepatic gluconeogenesis by promoting Forkhead box protein O1 (FOXO1) translocation to the nucleus. Thus, stimulation and accumulation of FFA is the main leading cause to the progression of T2DM and associated liver injury (Figure 1).12
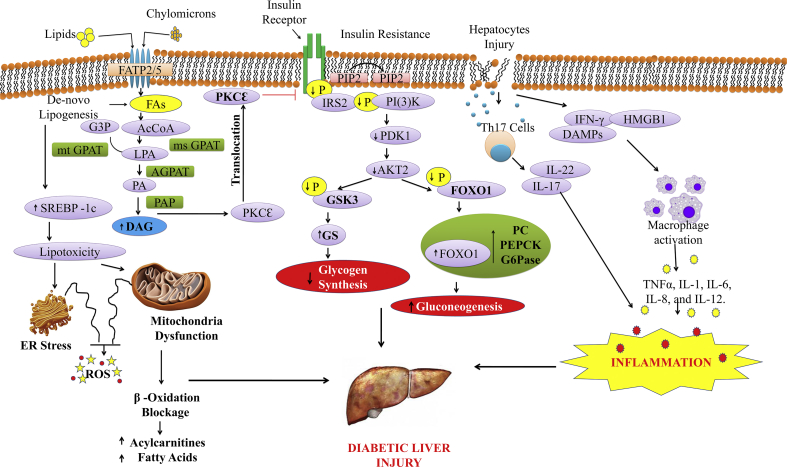
Figure 1.
Molecular mechanisms involved in the pathogenesis of diabetic-associated fatty liver disorders via hepatic insulin resistance and hyperglycaemia. Fatty-acyl coenzymes-A (AcCoAs) (shown here as phosphatidic acid, PA) formed by 1-acylglycerol-3-phosphate O-acyltransferase-2 (AGPAT2) are then added to the glycerol backbone by phosphatidic acid phosphatase (PAP) to generate diacylglycerol (DAG) and by diacylglycerol acyltransferases (DGAT) to generate triacylglycerol (TAG). Increased DAG causes protein kinase Cε (PKCε) translocation to the cell membrane, which inhibits insulin signaling. Reduced phosphorylation of insulin receptor substrate-2 (IRS-2) and PI(3)K impairs AKT2 activity by reductions in 3-phosphoinositide-dependent protein kinase-1 (PDK-1) activity, suppressing glycogen synthase kinase-3 (GSK-3) phosphorylation and reducing insulin-stimulated liver glycogen synthesis through reduced glycogen synthase (GS) activity. Fatty acids (FAs) derived from lipolysis and from chylomicron remnants are taken up through fatty-acid transport proteins (FATPs), mainly FATP2 and FATP5 in the liver. Fatty acids can also be reesterified to lysophosphatidic acid (LPA) by AcCoA and the conversion of glycerol 3-phosphate (G3P) by either mitochondrial glycerol-3-phosphate acyltransferase (mtGPAT) or microsomal GPAT (msGPAT). In case of hepatocytes injury, Th17 cells and DAMPs production lead to the secretion of cytokines and chemokines which cause inflammation. FOXO1, Forkhead box protein O1; PC, pyruvate carboxylase; PEPCK, phosphoenolpyruvate carboxykinase; G6Pase, glucose-6-phosphatase; PIP3, phosphatidyl inositol (3, 4, 5)-triphosphate; Th17, T helper 17 cells; DMPs, damage-associated molecular patterns.
Pathological events associated with diabetic liver injury
Dyslipidemia
The primary risk factors for T2DM and the metabolic syndrome include dyslipidemia, obesity, and hypertension.13 Dyslipidemia is directly or indirectly involved in the pathogenesis of DLI by inducing insulin resistance and hepatic steatosis. Insulin resistance is also associated with an increase of FFA flux that contributes to increased triglycerides production that, in turn, stimulates assembly and secretion of very low–density lipoprotein (VLDL) in hepatocytes.14 Preclinical animal model study revealed that apigenin treatment reversed the effect of high fat diet–induced metabolic disturbances such as dyslipidemia, hepatic steatosis, and insulin resistance in C57BL/6J diabetic mice.15 Similarly, Harriman et al16 in 2016 also demonstrated that ND-630 inhibits the acetyl-CoA carboxylase and reduces hepatic steatosis, improves insulin sensitivity, and modulates dyslipidemia in diabetic rats. It is also demonstrated that fatty liver and dyslipidemia with insulin resistance is relatively common in overweight and obese volunteers with T2DM.17 In fact, dyslipidemia is linked with high plasma levels of triglycerides, FFAs, cholesterol, and low plasma high density lipoproteins /low density lipoproteins (HDL/LDL) ratio in obesity-associated hepatic steatosis patients. Similar associations of dyslipidemia were also observed in obese Zucker fa/fa rats.18
Oxidative Stress
Diabetes induces oxidative stress in the liver, which is characterized by an increased concentration of reactive oxygen species in tissue leading to significant reduction in antioxidants and increment in lipid peroxidation and protein oxidation. In the background of diabetes, this oxidative distortion in the liver determines the severity of pathogenesis from NAFLD to steatohepatitis leading to cirrhosis.19 Animal studies have evidenced that treatment with biotin-(15 mg/kg) attenuated hepatotoxicity and oxidative stress in streptozotocin (STZ)-induced type 1 diabetes in Swiss albino mice.20 Another study has reported that Hypoxis hemerocallidea significantly reduced hyperglycemia and hyperglycemic-induced oxidative stress in the liver and kidney tissues of streptozotocin-induced diabetic wistar rats.21 Preclinical study from our group reported that there was significant reduction in hypoxia-inducible factor-1 alpha (HIF-1alpha), catalase enzyme levels and increase in renal tissue malonaldehyde levels in the diabetic group in comparison with the carbohydrate restriction administered group. This study directly revealed that oxidative stress and HIF-1alpha play a major role in the pathogenesis of diabetic-associated renal injury.22 There are numerous supportive reports available which evidently proved that there is the possible role of oxidative stress in the development of the diabetes-associated metabolic syndrome.
Insulin Resistance
In many liver diseases, insulin resistance has been recognized as an independent predictor and risk factor for the development of alcoholic and nonalcoholic steatohepatitis, chronic viral hepatitis, and HCC.23, 24, 25, 26, 27 Insulin resistance causes activation of hormone-sensitive lipase which results in increased fat mobilization. This leads to increased levels of circulating FFA in the serum and liver, with the formation of a large amount triglyceride deposition in the liver which contributes to hepatocytes degeneration and fatty liver disease.28 Insulin resistance is often associated with fibrosis in NAFLD patients.29 Siddiqui et al30 demonstrated that decrease of β-cell functioning results in insulin resistance in patients suffering from nonalcoholic steatohepatitis (NASH).
Hyperglycemia
Hyperglycemia and insulin resistance has an important correlation with respect to the diabetes and liver disorders. It is reported that individuals with high HbA1c had higher risk of hepatic steatosis. Another scientific evidence reported that hyperglycemia directly or indirectly concerns with DLI in rodent model of high-fat/ethanol diet–induced liver injury.31 Qingpu et al in 2016 demonstrated that 1-deoxynojirimycin can increase hepatic insulin sensitivity in db/db mice via modulating glucose metabolic enzymes and insulin-stimulated protein kinase B/glycogen synthase kinase (PKB/GSK)-3β signal pathway. Moreover, 1-Deoxynojirimycin also improved lipid homeostasis and attenuated hepatic steatosis in db/db mice.32 In another study, it was reported that canine fibroblast growth factor 21 also ameliorated hyperglycemia by inhibiting hepatic gluconeogenesis via regulating signal transducer and activator of transcription 3 signaling pathway. This indirectly improved pancreatic beta-cell survival in diabetic mice and dogs.33
Dysbalance in Intestinal Microbiota
Intestinal microbiota plays a major role in maintaining the physiological, metabolic, and enzymatic homeostasis. Probiotics are well-known nutraceuticals which provide useful health effects by acting on intestinal microbial ecology and immunity. Probiotics also have a direct or an indirect role in the treatment of obesity, insulin resistance syndrome (IRS), type 2 diabetes, and NAFLD.34 One of the study demonstrated that there is a higher levels of Lactobacillus spp. in T2DM patients in comparison with healthy controls. Similarly, Chinese and European studies showed an enhanced concentration of Lactobacillus gasseri, Streptococcus mutans, and certain Clostridiales, and lower levels of Roseburia intestinalis and Faecalibacterium prausnittzii, in the diabetic cohort.35 In fact, few drugs also change the flora of intestine, as metformin administration resulted in increased levels of Akkermansia muciniphila which leads to improvement of glucose tolerance and reduced systemic inflammation.36 Along with lifestyle modifications, administration of Bifidobacterium longum with fructooligosaccharides were able to significantly reduce the serum aspartate transaminase levels, tumor necrosis factor (TNF-α), C-reactive protein, homeostasis assessment model-insulin resistance, serum endotoxin, steatosis, and the NASH activity index compared with lifestyle modification alone.37 Another study documented beneficial effect of probiotics VSL#3, administration of VSL#3 that improved insulin resistance and reduced the aortic plaque extension, mesenteric adipose tissue inflammation, and steatohepatitis in ApoE−/− mice.38, 39 Therefore, it seems that intestinal microbiota may play a key role in the management of diabetes and liver disorders.
Molecular mechanism of diabetic liver injury
Free Fatty Acid
An increased production of nonesterified fatty acids (NEFA) from adipose tissue might be responsible for insulin resistance which is the main risk factor for the development of noninsulin dependent diabetes mellitus (NIDDM).40 Elevated level of plasma FFA produces peripheral and hepatic insulin resistance, which in normal patients is compensated by FFA-induced potentiation of glucose-stimulated insulin secretions. It is reported that in the development of NIDDM, FFAs fail to stimulate insulin secretion, which leaves hepatic and peripheral insulin resistance unchecked resulting in hepatic overproduction and underutilization of peripheral glucose.41 The postulated mechanism has been reported that increased FFA oxidation causes elevation of the intramitochondrial acetyl-CoA/CoA and NADH/NAD 1 ratios with subsequent inactivation of pyruvate dehydrogenase. This, in turn, causes increased citrate concentrations which lead to inhibition of phosphofructokinase and subsequent accumulation of glucose-6-phosphate. Finally, increased concentrations of glucose-6-phosphate cause inhibition of hexokinase II resulting in decreased glucose uptake.2, 3, 42
Wu et al43 in 2011 demonstrated that db/db mice fed on high-fructose diet had significant increase in liver triglyceride, glucose levels, insulin sensitivity, dysregulation of hepatic de novo lipogenesis, fatty acid oxidation, and glucose oxidation compared with the control group of mice. This provided a proof of concept for the involvement of FFA in the pathogenesis of diabetes and metabolic disorders in rodent models. NAFLD not only increases the risk of T2DM but also worsens glycemic control levels and contribute to the pathogenesis of chronic complications of diabetes.43
Circulating FFA derived from adipocytes are elevated in many insulin-resistant states and have been suggested to play a key role in the pathogenesis of insulin resistance occurring in diabetes and obesity by inhibiting glucose uptake, glycogen synthesis, glucose oxidation, and by increasing hepatic glucose output shown in Figure 1.44, 45, 46 So it may be clearly stated that insulin resistance leads to increase in the FFA flux, hyperglycemia, and hyperinsulinemia followed by more fat accumulation which further enhances the oxidative and inflammatory burden to the liver, finally produces NASH in diabetic patients.
Similarly, elevated circulating FFA levels has been found to be partly associated with diminished suppression of adipose tissue lipolysis by insulin, resulting in increased delivery of FFAs to the liver which is further exacerbated by impaired hepatic fatty acid oxidation secondary to insulin resistance. Once glucose levels are elevated in the perspective of prediabetes or overt diabetes, this provides an auxiliary substrate for triglyceride synthesis. Moreover, impaired VLDL secretion, due to insulin resistance, further contributes to hepatic fat accumulation and aggravates the pathological conditions associated with diabetic and non-DLI.47, 48
Fetuin-A (α2HS-glycoprotein)
It is a liver secretory glycoprotein, and also known as transforming growth factor-β1 signaling inhibitor. Alteration in serum fetuin-A (FetA) concentration is associated with NAFLD and cardiovascular diseases. One of the clinical trials suggested that FetA levels were elevated in NAFLD patients and hepatic expression of FetA level correlated with key enzymes in glucose and lipid metabolism.49 Therefore, circulating FetA could be a useful serum biomarker for predicting liver and vascular fibrosis progression in NAFLD patients.50 FetA has been implicated in the impairment of insulin receptor signaling, toll-like receptor 4 activation, macrophage migration and polarization, adipocytes dysfunction, hepatocytes triacylglycerols accumulation, and liver inflammation and fibrosis (Figure 2). Trepanowski et al51 in 2015 has documented that weight loss, aerobic exercise, and usage of metformin and pioglitazone are effective in reducing FetA level.
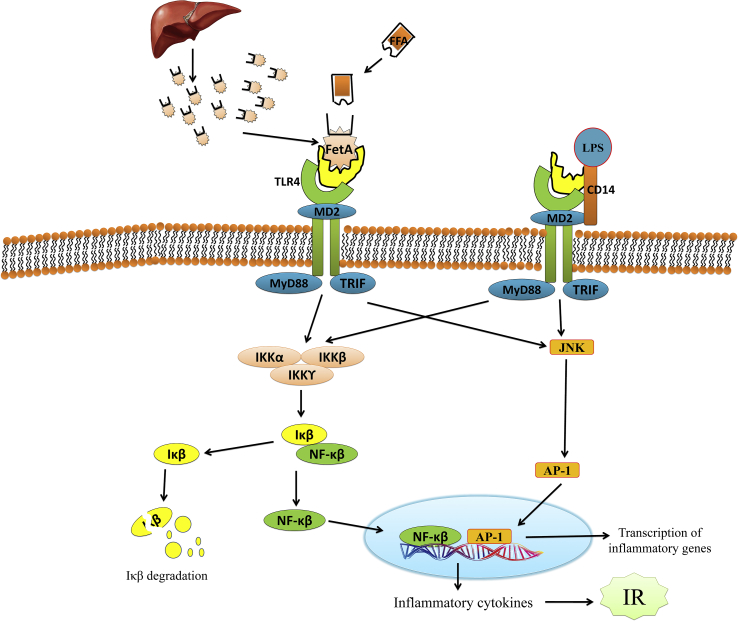
Figure 2.
Molecular mechanism of Fetuin-A involved in the pathogenesis of diabetic associated fatty liver disorders via suppression of insulin signaling resulting in insulin resistance. FetA, a glycoprotein produced by the liver, could provide the elusive link between FFAs and the TLR4 pathway. FFAs binds to FetA, which then binds TLR4. TLR4 signaling leads to the activation of the transcription factors, nuclear factor-κB (NF-κB) and AP-1, which can then upregulate the transcription of inflammatory genes, resulting in the production of inflammatory cytokines that can lead to insulin resistance. NF-κB, nuclear factor-κB; MyD88, myeloid differentiation factor 88; LPS, lipopolysaccharide; TLR4, toll-like receptor; AP-1, activator protein 1; IKK, IκB kinase; TIR-domain-containing adapter-inducing interferon-β; MD2, myeloid differentiation protein-2; IκB, inhibitor of Κb; JNK, Jun N-terminal kinases.
Proinflammatory Cytokines
Inflammation is one of the pivotal mechanisms of liver injury in diabetes.52 The rise in proinflammatory cytokines favors diabetes-related glucose toxicity, leading to mitochondrial dysfunction, oxidative stress, and hepatocellular death. The epidemiological, genetic, and experimental evidence demonstrated a significant role of interleukin-6 (IL-6) in the pathogenesis of inflammation, insulin resistance, diabetes and associated complications.53, 54 The mixed inflammatory cells (mononuclear cells and a few neutrophils) infiltration into the lobule and portal area produces evident inflammation in the diabetic liver.55 Hyperlipidemia and hyperglycemia in diabetes induce transcription of proinflammatory cytokines, TNF-α, monocyte chemotactic protein-1 (MCP-1), adipokines, and fatty acid-binding protein 4 which lead to hepatic injury and insulin resistance.56, 57 Recently, vitamin D3 was used to regulate IL-6 and osteopontin expression in C56Bl/J6 diabetic mice liver.58
Chemokines
Chemokines are signaling proteins secreted by cells and have an ability to induce directed chemotaxis in nearby responsive cells in response to bacterial infection, viruses, and agents that cause physical damage to the body organs. However, their release is often stimulated by pro-inflammatory cytokines.59 Numerous chemokines (CXCL8, 11 and CCL2, 3, 4, 5, and 11) play a crucial role in the pathophysiology of various devastating disorders including hepatotoxicity, diabetes, and cardiovascular disorders. Fractalkine (CX3CL1) is involved in the development of numerous inflammatory conditions including metabolic diseases. In the recent study, it is demonstrated that plasma CX3CL1 levels were significantly higher (P = 0.005) in T2D patients than in nondiabetics.60 Wehr et al61 in 2014 suggested that monocyte chemoattractant protein-1 (MCP-1, CCL2), the primary ligand for chemokines receptor C—C chemokines receptor 2, is increased in the livers of patients with NASH and murine models of steatohepatitis and fibrosis. From the above scientific evidences, it is concluded that various chemokines play vital role in the pathogenesis of diabetes and liver disorders, but still there is a requirement for exploring the role of chemokines in the dual disease condition of DLI.
Adipokines
The adipocytokines are cytokines (cell-signaling proteins) secreted by the adipose tissue which includes leptin, adiponectin, visfatin, apelin, vaspin, hepcidin, RBP4, and inflammatory cytokines, such as TNF-α, IL-1β, and so on. These adipocytokines play a vital role in the pathogenesis of diabetes and other metabolic disorders.62 Inflammatory adipokines such as TNF-α, IL-6, LPS, and saturated FFA activate inhibitory molecules such as protein tyrosine phosphatase 1B (PTP1B), suppressor of cytokine signalling (SOCS), and Jun N-terminal kinases to suppress insulin signaling resulting in insulin resistance as shown in Figure 3. Adipokines are associated with deteriorating liver function in a complex manner in patients with alcoholic liver cirrhosis. The alcohol-induced liver injury is related to a variety of molecular factors including cytokines, adipokines, chemokines, and neurotransmitters.63 It is suggested that there is an alteration in the levels of different adipokines in cirrhosis patients,64 although most studies did not exclude patients with baseline diabetes which in itself is associated with altered adipokines.65
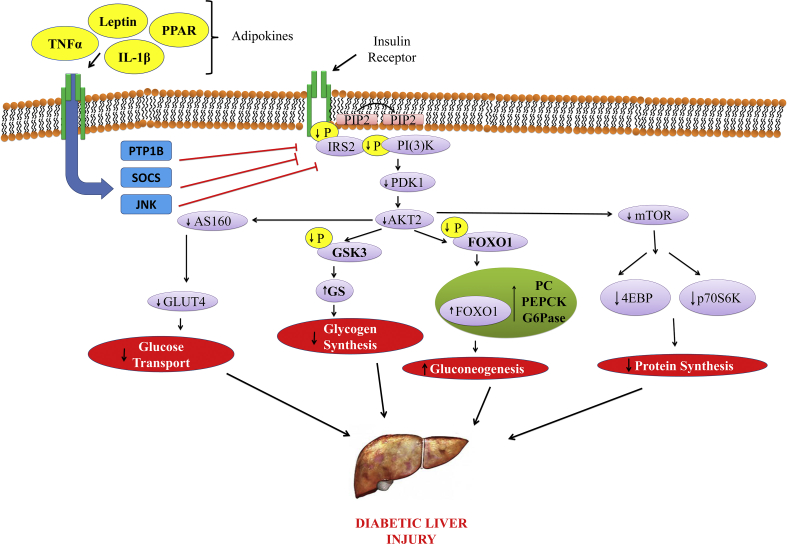
Figure 3.
Molecular mechanism of inflammatory adipokines involved in the pathogenesis of diabetic associated fatty liver disorders via suppression of insulin signaling resulting in insulin resistance. PI3K-AKT signaling pathway regulates metabolic processes such as glucose uptake (muscle and adipocytes), glycogen synthesis (muscle and liver), protein synthesis (muscle and liver), and gluconeogenesis (liver). Inflammatory adipokines, TNF-a, IL-6, LPS, and saturated free fatty acid, activate inhibitory molecules such as SOCS and JNK to suppress insulin signaling resulting in insulin resistance. PI3K dependent PDK1 activation is negatively regulated by phosphor lipid phosphatase such as phosphatase and tensin homolog (PTEN) that degrade PIP3. IRS2, insulin receptor substrate-2; FOXO1, Forkhead box protein O1; PC, pyruvate carboxylase; PEPCK, phosphoenolpyruvate carboxykinase; G6Pase, glucose-6-phosphatase; PIP3, phosphatidyl inositol (3, 4, 5)-triphosphate; Th17, T helper 17 cells; DMPs, damage-associated molecular patterns; MAPK, Ras-mitogen-activated protein kinase; PI3K-AKT, phosphatidyl inositol 3-kinase; PKB, protein kinase B.
Hyperinsulinemia may down regulate adiponectin, leading to hepatic lipogenesis and further decreasing FFA oxidation. Thus, hyperinsulinemia may act as a major contributor to the progression of liver damage through stimulation of adipokines pathways. It is postulated that inflamed and necrotic hepatocytes releases several types of chemical mediators and adipocytokines which activates the stellate cells to produce connective tissue growth factor and collagen and cause an accumulation in the extracellular matrix, thereby favoring fibrosis.3 It is well described that adiponectin and its receptors play an important role in hepatic insulin resistance,66 and it is exhibited that high molecular weight (HMW) oligomers help in attenuation of hepatic insulin resistance. Within the same context, the synthesis of the HMW oligomers is necessary to mediate the insulin-sensitizing effects of adiponectin on the suppression of hepatic gluconeogenesis in primary rat hepatocytes.67
Linglin et al68 in 2006 reported that adiponectin knockout mice were more insulin resistant than controls. Similarly, one of the clinical trials suggested that increased adiponectin level could help restore the hepatic insulin resistance in severely obese women.69, 70 From the above evidence, it may be concluded that adiponectin might prove as a promising target for hepatic insulin resistance.
Matrix Metalloproteinase
Matrix metalloproteinase (MMP) plays an important role in atherosclerosis, but very little is explored related to the effects of hyperglycemia on MMP regulation in vascular cells. In a recent scientific evidence, gelatin zymography and western blot analysis revealed that the activity and expression of 92-kDa (MMP-9) gelatinase, but not of 72 kDa (MMP-2) gelatinase, were significantly increased in vascular tissue and plasma of two distinct rodent models of DM.71 A recent study also documented that matrix metalloproteinase (MMP-2 & 9) levels of the hepatic tissue were significantly increased in the rodent model of acute liver injury.72 Similarly, another study reported that racemic gossypol with a dose of 15 mg/kg/day for 4 weeks followed by 15 mg/kg/week for additional 8 weeks ameliorate messenger mRNA levels of glucocorticoid receptor (Nr3c1), phosphoenolpyruvate carboxykinase, glucose-6-phosphatase, collagen I, collagen III, fibronectin, tissue inhibitor of metalloproteinase 1, and 2 liver fibrosis in diabetic rats induced by high-fat diet and streptozocin.73 Hou et al74 in 2014 suggested that insulin resistance–induced alteration of MMP-9 to TIMP-1 ratio may play a role in the elevation of collagen-IV, which may participate in the development of hepatic fibrosis in the later course of insulin resistance. However, there are number of studies which suggested the role of MMP in diabetes and liver disorders individually but very less contribution is there which predict the role of MMP in dual DLI condition.
Genetic Factors
There are numerous genes which regulate various physiological processes and play a pivotal role in the normal functioning of liver and other major organs. The physiological functioning such as insulin secretion, fatty acid synthesis and oxidation, glucose uptake, and fatty acid metabolism involve various genes such as sterol regulatory element-binding protein (SREBP), fatty acid synthase (FAS), and acetyl-CoA carboxylase (ACC1), and so on. Insulin resistance is the key factor in NAFLD pathophysiology and is deeply entangled with the progression of liver disease through upregulation of various pathological pathways,75 but the causal relationship between insulin resistance and fibrogenesis remains unclear. Functional common single nucleotide polymorphisms (SNPs) of genes included in the insulin-signaling pathway influence insulin resistance and the susceptibility to type 2 diabetes. In 2010, it was demonstrated that the combination of ENPP1, 121Gln, and IRS-1 (insulin receptor substrate-1) 972Arg alleles were associated with decreased activation of the insulin-signaling pathway in the liver and influenced fibrosis severity in a large multicentre series of NAFLD patients.76 On the basis of scientific literature, it is reported that dyslipidemia and fatty acid are the major culprits of liver-associated disorders which directly enhance the fatty acids accumulation in hepatocytes, and up-regulate de novo synthesis and uptake in association with increased expression of ACC1, FAS, SREBP, and adipophilin (ADRP) genes. Fatty acid oxidation–related genes, long-chain acyl-CoA dehydrogenase (LCAD), long-chain L-3-hydroxyacylcoenzyme A dehydrogenase alpha (HADHα), uncoupling protein 2 (UCP2), straight-chain acyl-CoA oxidase (ACOX), branched-chain acyl-CoA oxidase (BOX), cytochrome P450 2E1 (CYP2E1), CYP4A11, and carnitine palmitoyltransferase 1a (CPT1a) were also over expressed, indicating that oxidation was enhanced in NAFLD and worsen the pathological conditions.77 One of the studies suggested that transcriptional induction of fatty acid synthase (FASN) gene expression in hepatic steatosis is impaired in NASH, whereas hepatic inflammation in the absence of steatosis does not affect FASN expression, suggesting that FASN may serve as a new diagnostic marker or therapeutic target for the progression of NAFLD.78 The related gene mutations involved in the diabetes-associated liver injury are summarized in Table 1.
Table 1.
List of Related Gene Mutations Involved in the Diabetes-Associated Liver Injury.
| S. No. | Gene | Mutation | Pathology associated | Reference |
|---|---|---|---|---|
| 1 | ENPP1 | ENPP1: 121Gln | Insulin resistance associated with NAFLD | 76 |
| 2 | IRS-1 | IRS-1: 972Arg | Insulin resistance associated with NAFLD | 76 |
| 3 | PNPLA3 | PNPLA3:148M | Hepatic inflammation and NAFLD | 79 |
| 4 | FOXO1 | FOXO1: 253S | Insulin resistance with increased hepatic glucose production | 80 |
| 5 | IRS-1 | IRS-1:G972R | Insulin resistance | 81 |
| 6 | SREBP-1c | SREBP-1c: Ser372 | Insulin resistance, dyslipidemia, | 82 |
| 7 | CAPN10 | Missense mutation Thr504Ala | Insulin resistance, T2DM | 83 |
| 8 | HADHα, ACOX, BOX, CYP2E1, and CYP4A11 | Overexpressed | Fatty acid metabolism dysregulation, NAFLD | 77 |
ENPP1, ectoenzyme nucleotide pyrophosphate phosphodiesterase 1; FOXO1, Forkhead box protein O1; NAFLD, nonalcoholic fatty liver disease; IRS, insulin receptor substrate; DM, diabetes mellitus; SREBP, sterol regulatory element-binding protein; PNPLA3, patatin like phospholipase domain-containing protein-3; CAPN10, calpain10.
Current Therapies for the Management of Diabetes-Associated Liver Disorders
Diabetes is classified into two types: T1DM and T2DM; however, it is suggested that T2DM is mostly linked with the metabolic disorder which is associated with liver dysfunction. Most of the clinical hepatologists prescribed oral antihyperglycemic for the management of diabetic-associated liver disorder. The dietary interventions are the first choice of treatment for the management of diabetes-associated liver disorder; however, recent study by Krishan et al22 in 2018 suggested that carbohydrate restriction ameliorates diabetes associated complications by reducing oxidative stress and upregulating HIF-1alpha levels in type-1diabetic rats. However, the oral antihyperglycemic such as thiazolidinediones, biguanide, sulfonylureas, alpha-glucosidase inhibitors, dipeptidyl peptidase 4 (DPP-4) inhibitors, glitazones, and meglitinides proved to be pleiotropic and targeted various pharmacological targets and helped to control the progression of DLI.84 Recently, in 2013, Armstrong et al85 documented that liraglutide was safe, well tolerated, and improved liver enzymes in patients with type 2 diabetes. It is also reported that administration of liraglutide improved T2DM but also leading to improvement of liver inflammation, alteration of liver fibrosis, and reduction of body weight.86 Similarly, glucagon-like peptide 1 agonists and DPP-4 inhibitors show promising preliminary results in patients of NAFLD with diabetes.87 In 2016, study demonstrated that Ipragliflozin (SGLT2 inhibitors) improves NAFLD in T2DM patients.88 Still there is a need to explore more about the clinical interventions and new pharmacological interventions for the management of diabetes associated liver disorder.
Recent advances and novel interventions in the treatment of diabetes-associated liver disorders
There are numerous scientific reports which demonstrated the novel therapeutic targets for the treatment of various metabolic disorders that are directly or indirectly concerned with NAFLD. Recently, Azam et al89 in 2017 suggested that Naltrexone has a considerable role in attenuation of ER stress-induced liver injury. Another report demonstrated that carboxylesterase 2 prevents liver steatosis by modulating lipolysis, endoplasmic reticulum stress, and lipogenesis via regulation of hepatocyte nuclear factor 4 α in mice.90 Newberry et al in 2017 reported that perturbing exogenous hepatic fatty acid use modulates both hepatic steatosis and fibrosis in the setting of hepatic microsomal triglyceride transfer protein deletion, adding new insight into the pathophysiological mechanisms and consequences of very low–density lipoprotein.91 One of the scientific reports suggested that administration of fibroblast growth factor 21 (FGF21) in mice inhibits mammalian target of rapamycin complex 1 (mTORC1) in the liver, whereas FGF21-deficient mice display pronounced insulin-stimulated mTORC1 activation and exacerbated hepatic insulin resistance. FGF21 inhibits insulin- or nutrient-stimulated activation of mTORC1 to enhance phosphorylation of protein kinase B in HepG2 cells at both normal and insulin resistance condition.92 Moreover, Mazzotti et al in 2016 documented that drugs acting on the incretin axis and on Na-glucose co-transport at renal tubular level offer new hopes for a treatment to reduce the burden of hepatic triglyceride accumulation and progression of liver disease.93 One of the recent randomized controlled trial proved that empagliflozin reduces liver fat and improves ALT levels in patients with type 2 diabetes and NAFLD.94 However, the major recent advances with new goals are summarized in Table 2.
Table 2.
Recent Advances and Novel Interventions in the Treatment of Diabetes-Associated Liver Disorders.
| S. No. | New approaches and outcomes | Intervention and drug used | Diabetes and associated liver disorder | Reference |
|---|---|---|---|---|
| 1 | Hepatoprotective effects of the dual peroxisome proliferator-activated receptor alpha/delta agonist, Elafibranor (GFT505), in rodent models of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis | Proliferator-activated receptor alpha/delta agonist (Elafibranor GFT505) | NAFLD and IR | 95 |
| 2 | The probiotics mixture showed promise as a treatment for NAFLD pathogenesis, and may improve HFSD-induced steatosis through its effects on leptin, resistin, inflammatory biomarkers, and hepatic function markers in rodents. | Probiotics mixture | NAFLD and inflammation | 96 |
| 3 | One of scientific data suggested that Rosuvastatin has potential for use as a preventive drug for the development of HCC associated with NAFLD in mice | Competitive inhibitor of the enzyme HMG-CoA reductase (rosuvastatin) | NAFLD and HCC | 97 |
| 4 | Novel Thiazolidinedione (Lobeglitazone) improves Non-Alcoholic Fatty Liver Disease in Type 2 Diabetes. | Thiazolidinedione class of drugs and act as an agonist for both PPARα and PPARγ (lobeglitazone) | NAFLD and T2DM | 98 |
| 5 | Rimonabant reduces obesity-associated hepatic steatosis and features of metabolic syndrome in obese Zucker fa/fa rats | Inverse agonist for the cannabinoid receptor CB1 (rimonabant) | NAFLD and obesity | 16 |
| 6 | Farnesoid X nuclear receptor ligand obeticholic acid for noncirrhotic, nonalcoholic steatohepatitis (FLINT): a multicentre, randomized, placebo-controlled trial. | Farnesoid X nuclear receptor ligand and semisynthetic bile acid analog (Obeticholic acid) | Obeticholic acid improved the histological features of nonalcoholic steatohepatitis | 99 |
| 7 | Vitamin E therapy changes serum alanine aminotransferase levels in patients with non-alcoholic steatohepatitis. | Act as a fat-soluble antioxidant (vitamin E) | Nonalcoholic steatohepatitis | 100 |
| 8 | Saroglitazar improves diabetic dyslipidemia, in patients with diabetic dyslipidemia and liver dysfunction- an observational study | Saroglitazar (peroxisome proliferator–activated receptor (PPAR) agonist at the subtypes α and γ) | NAFLD and T2DM | 101 |
NAFLD, nonalcoholic fatty liver disease; T2DM, type 2 diabetes mellitus; HCC, hepatocellular carcinoma; IR, insulin resistance; PPAR, peroxisome proliferator–activated receptor; HMG, 3-hydroxy-3-methyl-glutaryl.
Lay summary
From the above scientific discussion, it is clear that insulin resistance and FFA are the central culprits of DLI and correlated with each other. As insulin resistance occurs, there is a high influx of FFA, hyperglycemia, and hyperinsulinemia which produce fatty liver- and diabetes-like complications. However, the vicious cycle of DLI is still under revilement and needs more exploration through biological approaches. There are certain genes such as SREBP-1c, FAS, ACC1, and phosphoenolpyruvate carboxykinase genes which when altered causes activation of various pathological pathways. Other side of the coin includes pathological connection such as dyslipidemia, hyperglycemia, hyperinsulinemia, and oxidative stress which leads to the progression of dual disorder of DLI. So, by validating these molecular and pathological pathways, there is a chance to find the therapeutic targets for the treatment of DLI. Thus, novel intervention and approaches will generate new insights for the development of the new pharmacological basis.
Conclusion
It is concluded that insulin resistance, hepatocytes injury, and FFA are the major culprits for the initiation of oxidative stress, inflammation, dyslipidemia, hyperglycemia, and so on which are involved in the pathogenesis of devastating DLI.
Conflicts of interest
The authors have none to declare.
Acknowledgments
The authors are thankful to Department of Science and Technology - Science and Engineering Research Board (DST-SERB), New Delhi for providing financial assistance under the project no: (EMR/2014/000438). The authors also acknowledge the Department of Pharmaceutical Sciences & Drug Research, Punjabi University, Patiala and UGC, New Delhi for extending necessary research facilities for our work.
References
- 1.World Health Organization . World Health Organization; 2016. Global Report on Diabetes. [Google Scholar]
- 2.McGarry J.D. What if Minkowski had been ageusic? An alternative angle on diabetes. Science (Wash. DC) 1992;258:766–770. doi: 10.1126/science.1439783. [DOI] [PubMed] [Google Scholar]
- 3.Randle P.J., Garland P.B., Hales C.N., Newsholme E.A. The glucose fatty-acid cycle: its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet. 1963;i:785–789. doi: 10.1016/s0140-6736(63)91500-9. [DOI] [PubMed] [Google Scholar]
- 4.Kalra S., Vithalani M., Gulati G. Study of prevalence of nonalcoholic fatty liver disease (NAFLD) in type 2 diabetes patients in India (SPRINT) J Assoc Phys India. 2013;61(7):448–453. [PubMed] [Google Scholar]
- 5.Tolman K.G., Fonseca V., Dalpiaz A., Tan M.H. Spectrum of liver disease in type 2 diabetes and management of patients with diabetes and liver disease. Diabetes Care. 2007;30:734–743. doi: 10.2337/dc06-1539. [DOI] [PubMed] [Google Scholar]
- 6.Chapman M., Crockett S., Purvis T. Macrovascular disease in the elderly with type 1 diabetes. J Diabetes Metabol. 2013;4:299. [Google Scholar]
- 7.Trombetta M., Spiazzi G., Zoppini G., Muggeo M. Review article: type 2 diabetes and chronic liver disease in the Verona diabetes study. Aliment Pharmacol Ther. 2005;22(suppl 2):24–27. doi: 10.1111/j.1365-2036.2005.02590.x. [DOI] [PubMed] [Google Scholar]
- 8.Fruci B., Giuliano S., Mazza A., Malaguarnera R., Belfiore A. Nonalcoholic Fatty liver: a possible new target for type 2 diabetes prevention and treatment. Int J Mol Sci. 2013;14:22933–22966. doi: 10.3390/ijms141122933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.E1 Bugianesi, McCullough A.J., Marchesini G. Insulin resistance: a metabolic pathway to chronic liver disease. Hepatology. 2005;42(5):987–1000. doi: 10.1002/hep.20920. [DOI] [PubMed] [Google Scholar]
- 10.Lehninger A., Nelson D.L., Cox M.M. W. H Freeman; 2005. Lehninger's Principles of Biochemistry. [Google Scholar]
- 11.Saltiel A.R., Kahn C.R. Insulin signalling and the regulation of glucose and lipid metabolism. Nature. 2001;414(6865):799–806. doi: 10.1038/414799a. [DOI] [PubMed] [Google Scholar]
- 12.Perry R.J., Samuel V.T., Petersen K.F., Shulman G.I. The role of hepatic lipids in hepatic insulin resistance and type 2 diabetes. Nature. 2014;510(7503):84–91. doi: 10.1038/nature13478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Angulo P. GI epidemiology: nonalcoholic fatty liver disease. Aliment Pharmacol Ther. 2007;25:883–889. doi: 10.1111/j.1365-2036.2007.03246.x. [DOI] [PubMed] [Google Scholar]
- 14.Gaggini M., Morelli M., Buzzigoli E., DeFronzo R.A., Bugianesi E., Gastaldelli A. Non-alcoholic fatty liver disease (NAFLD) and its connection with insulin resistance, dyslipidemia, atherosclerosis and coronary heart disease. Nutrients. 2013;5(5):1544–1560. doi: 10.3390/nu5051544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jung U.J., Cho Y.Y., Choi M.S. Apigenin ameliorates dyslipidemia, hepatic steatosis and insulin resistance by modulating metabolic and transcriptional profiles in the liver of high-fat diet-induced obese mice. Nutrients. 2016;8(5):305. doi: 10.3390/nu8050305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Harriman G., Greenwood J., Bhat S. Acetyl-CoA carboxylase inhibition by ND-630 reduces hepatic steatosis, improves insulin sensitivity, and modulates dyslipidemia in rats. Proc Natl Acad Sci Unit States Am. 2016;113(13):E1796–E1805. doi: 10.1073/pnas.1520686113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kelley D.E., McKolanis T.M., Hegazi R.A., Kuller L.H., Kalhan S.C. Fatty liver in type 2 diabetes mellitus: relation to regional adiposity, fatty acids, and insulin resistance. Am J Physiol Endocrinol Metab. 2003;285(4):E906–E916. doi: 10.1152/ajpendo.00117.2003. [DOI] [PubMed] [Google Scholar]
- 18.Gary-Bobo M., Elachouri G., Gallas J.F. Rimonabant reduces obesity-associated hepatic steatosis and features of metabolic syndrome in obese Zucker fa/fa rats. Hepatology. 2007;46(1):122–129. doi: 10.1002/hep.21641. [DOI] [PubMed] [Google Scholar]
- 19.Lucchesi A.N., Freitas N.T., Cassettari L.L., Marques S.F., Spadella C.T. Diabetes mellitus triggers oxidative stress in the liver of alloxan-treated rats: a mechanism for diabetic chronic liver disease. Acta Cir Bras. 2013;28(7):502–508. doi: 10.1590/s0102-86502013000700005. [DOI] [PubMed] [Google Scholar]
- 20.Aldahmash B.A., El-Nagar D.M., Ibrahim K.E. Attenuation of hepatotoxicity and oxidative stress in diabetes STZ-induced type 1 by biotin in Swiss albino mice. Saudi J Biol Sci. 2016;23(2):311–317. doi: 10.1016/j.sjbs.2015.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oguntibeju O.O., Meyer S., Aboua Y.G., Goboza M. Hypoxis hemerocallidea significantly reduced hyperglycaemia and hyperglycaemic-induced oxidative stress in the liver and kidney tissues of streptozotocin-induced diabetic male wistar rats. Evid Base Compl Alternative Med. 2016:1–10. doi: 10.1155/2016/8934362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Krishan P., Singh G., Bedi O. Carbohydrate restriction ameliorates nephropathy by reducing oxidative stress and upregulating HIF-1α levels in type-1 diabetic rats. J Diabetes Metab Disord. 2017;16(1):47. doi: 10.1186/s40200-017-0331-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Larsen N., Vogensen F.K., van den Berg F.W. Gut microbiota in human adults with type 2 diabetes differs from non-diabetic adults. PLoS One. 2010;5:e9085. doi: 10.1371/journal.pone.0009085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Takamatsu S., Noguchi N., Kudoh A. Influence of risk factors for metabolic syndrome and non-alcoholic fatty liver disease on the progression and prognosis of hepatocellular carcinoma. Hepatogastroenterology. 2008;55:609–614. [PubMed] [Google Scholar]
- 25.Harrison S.A. Insulin resistance among patients with chronic hepatitis C: etiology and impact on treatment. Clin Gastroenterol Hepatol. 2008;6:864–876. doi: 10.1016/j.cgh.2008.03.024. [DOI] [PubMed] [Google Scholar]
- 26.Marchesini G., Marzocchi R. Metabolic syndrome and NASH. Clin Liver Dis. 2007;11:105–117. doi: 10.1016/j.cld.2007.02.013. [DOI] [PubMed] [Google Scholar]
- 27.Wan Y., Garner J., Wu N. Role of stem cells during diabetic liver injury. J Cell Mol Med. 2016;20(2):195–203. doi: 10.1111/jcmm.12723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fiorentino T.V., Andreozzi F., Mannino G.C. One-hour post-load hyperglycemia confers higher risk of hepatic steatosis to HbA1c-defined pre-diabetic subjects. J Clin Endocrinol Metab. 2016;101(11):4030–4038. doi: 10.1210/jc.2016-1856. [DOI] [PubMed] [Google Scholar]
- 29.Bambha K., Wilson L.A., Unalp A. Coffee consumption in NAFLD patients with lower insulin resistance is associated with lower risk of severe fibrosis. Liver Int. 2014;34(8):1250–1258. doi: 10.1111/liv.12379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Siddiqui M.S., Cheang K.L., Luketic V.A. Nonalcoholic steatohepatitis (nash) is associated with a decline in pancreatic beta cell (β-cell) function. Dig Dis Sci. 2015;60(8):2529–2537. doi: 10.1007/s10620-015-3627-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tai C.J., Choong C.Y., Shi Y.C. Solanum nigrum protects against hepatic fibrosis via suppression of hyperglycemia in high-fat/ethanol diet-induced rats. Molecules. 2016;21(3):269. doi: 10.3390/molecules21030269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Liu Q., Li X., Li C. 1-Deoxynojirimycin alleviates liver injury and improves hepatic glucose metabolism in db/db mice. Molecules. 2016;21(3):279. doi: 10.3390/molecules21030279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Xu P., Zhang Y., Jiang X. Canine Fibroblast growth factor 21 ameliorates hyperglycemia associated with inhibiting hepatic gluconeogenesis and improving pancreatic beta-cell survival in diabetic mice and dogs. PLoS One. 2016;11(5):e0155598. doi: 10.1371/journal.pone.0155598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sáez-Lara M.J., Robles-Sanchez C., Ruiz-Ojeda F.J., Plaza-Diaz J., Gil A. Effects of probiotics and synbiotics on obesity, insulin resistance syndrome, type 2 diabetes and non-alcoholic fatty liver disease: a review of human clinical trials. Int J Mol Sci. 2016;17(6):928. doi: 10.3390/ijms17060928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Everard A., Belzer C., Geurts L. Cross-talk between Akkermansia muciniphila and intestinal epithelium controls diet-induced obesity. Proc Natl Acad Sci USA. 2013;110:9066–9071. doi: 10.1073/pnas.1219451110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shin N.R., Lee J.C., Lee H.Y. An increase in the Akkermansia spp. population induced by metformin treatment improves glucose homeostasis in diet-induced obese mice. Gut. 2014;63:727–735. doi: 10.1136/gutjnl-2012-303839. [DOI] [PubMed] [Google Scholar]
- 37.Malaguarnera M., Vacante M., Antic T. Bifidobacterium longum with fructo-oligosaccharides in patients with non alcoholic steatohepatitis. Dig Dis Sci. 2012;57:545–553. doi: 10.1007/s10620-011-1887-4. [DOI] [PubMed] [Google Scholar]
- 38.Mencarelli A., Cipriani S., Renga B. VSL#3 resets insulin signaling and protects against NASH and atherosclerosis in a model of genetic dyslipidemia and intestinal inflammation. PLoS One. 2012;7:e45425. doi: 10.1371/journal.pone.0045425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sanduzzi Zamparelli M., Compare D., Coccoli P. The metabolic role of gut microbiota in the development of nonalcoholic fatty liver disease and cardiovascular disease. Int J Mol Sci. 2016;17(8):1225. doi: 10.3390/ijms17081225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lillioja S., Mott D., Spraul M. Insulin resistance as precursor of non-insulin dependent diabetes mellitus. Prospective studies of Pima Indians. N Engl J Med. 1993;329:1988–1992. doi: 10.1056/NEJM199312303292703. [DOI] [PubMed] [Google Scholar]
- 41.Boden G. Role of fatty acids in the pathogenesis of insulin resistance and NIDDM. Diabetes. 1997;46(1):3–10. [PubMed] [Google Scholar]
- 42.Randle P.J., Garland P.B., Newsholme E.A., Hales C.N. The glucose fatty acid cycle in obesity and maturity onset diabetes mellitus. Ann NY Acad Sci. 1965;131:324–333. doi: 10.1111/j.1749-6632.1965.tb34800.x. [DOI] [PubMed] [Google Scholar]
- 43.Wu M., Singh S.B., Wang J. Antidiabetic and antisteatotic effects of the selective fatty acid synthase (FAS) inhibitor platensimycin in mouse models of diabetes. Proc Natl Acad Sci USA. 2011;108(13):5378–5383. doi: 10.1073/pnas.1002588108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Williamson R.M., Price J.F., Glancy S. Prevalence of and risk factors for hepatic steatosis and nonalcoholic fatty liver disease in people with type 2 diabetes: the Edinburgh type 2 diabetes study. Diabetes Care. 2011;34(5):1139–1144. doi: 10.2337/dc10-2229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bergman R.N., Ader M. Free fatty acids and pathogenesis of type 2 diabetes mellitus. Trends Endocrinol Metabol. 2000;11:351–356. doi: 10.1016/s1043-2760(00)00323-4. [DOI] [PubMed] [Google Scholar]
- 46.Shulman G.I. Cellular mechanisms of insulin resistance. J Clin Invest. 2000;106:171–176. doi: 10.1172/JCI10583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bhatt Harikrashna B., Smith Robert J. Fatty liver disease in diabetes mellitus. Hepatobiliary Surg Nutr. 2015;4(2):101–108. doi: 10.3978/j.issn.2304-3881.2015.01.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Bugianesi E., Gastaldelli A., Vanni E. Insulin resistance in non-diabetic patients with non-alcoholic fatty liver disease: sites and mechanisms. Diabetologia. 2005;48:634–642. doi: 10.1007/s00125-005-1682-x. [DOI] [PubMed] [Google Scholar]
- 49.Haukeland J.W., Dahl T.B., Yndestad A. Fetuin A in nonalcoholic fatty liver disease: in vivo and in vitro studies. Eur J Endocrinol. 2012;166(3):503–510. doi: 10.1530/EJE-11-0864. [DOI] [PubMed] [Google Scholar]
- 50.Sato M., Kamada Y., Takeda Y. Fetuin-A negatively correlates with liver and vascular fibrosis in nonalcoholic fatty liver disease subjects. Liver Int. 2015;35(3):925–935. doi: 10.1111/liv.12478. [DOI] [PubMed] [Google Scholar]
- 51.Trepanowski J.F., Mey J., Varady K.A. Fetuin-A: a novel link between obesity and related complications. IJO. 2015;39(5):734–741. doi: 10.1038/ijo.2014.203. [DOI] [PubMed] [Google Scholar]
- 52.Ban C.R., Twigg S.M. Fibrosis in diabetes complications: pathogenic mechanisms and circulating and urinary markers. Vasc Health Risk Manag. 2008;4:575–596. doi: 10.2147/vhrm.s1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bastard J.P., Maachi M., Lagathu C. Recent advances in the relationship between obesity, inflammation, and insulin resistance. Eur Cytokine Netw. 2006;17:4–12. [PubMed] [Google Scholar]
- 54.Kristiansen O.P., Mandrup-Poulsen T. Interleukin-6 and diabetes: the good, the bad, or the indifferent? Diabetes. 2005;54:114–124. doi: 10.2337/diabetes.54.suppl_2.s114. [DOI] [PubMed] [Google Scholar]
- 55.Wan Y., Garner J., Wu N. Role of stem cells during diabetic liver injury. J Cell Mol Med. 2016;20(2):195–203. doi: 10.1111/jcmm.12723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Tilg H. The role of cytokines in non-alcoholic fatty liver disease. Dig Dis. 2010;28:179–185. doi: 10.1159/000282083. [DOI] [PubMed] [Google Scholar]
- 57.Solomon S.S., Odunusi O., Carrigan D. TNF-alpha inhibits insulin action in liver and adipose tissue: a model of metabolic syndrome. Horm Metab Res. 2010;42:115–121. doi: 10.1055/s-0029-1241834. [DOI] [PubMed] [Google Scholar]
- 58.Labudzynskyi D., Shymanskyy I., Veliky M. Role of vitamin D3 in regulation of interleukin-6 and osteopontin expression in liver of diabetic mice. Diabetes. 2016;37(2.12):22–31. [PubMed] [Google Scholar]
- 59.Le Y., Zhou Y., Iribarren P., Wang J. Chemokines and chemokine receptors: their manifold roles in homeostasis and disease" (PDF) Cell Mol Immunol. 2004;1(2):95–104. [PubMed] [Google Scholar]
- 60.Sindhu S., Akhter N., Arefanian H. Increased circulatory levels of fractalkine (CX3CL1) are associated with inflammatory chemokines and cytokines in individuals with type-2 diabetes. J Diabetes Metab Disord. 2017;16(1):15. doi: 10.1186/s40200-017-0297-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wehr A., Baeck C., Ulmer F. Pharmacological inhibition of the chemokine CXCL16 diminishes liver macrophage infiltration and steatohepatitis in chronic hepatic injury. PLoS One. 2014;9(11):e112327. doi: 10.1371/journal.pone.0112327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Andrade-Oliveira V., Câmara N.O., Moraes-Vieira P.M. Adipokines as drug targets in diabetes and underlying disturbances. J Diabetes Res. 2015:1–11. doi: 10.1155/2015/681612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Diehl A.M. Obesity and alcoholic liver disease. Alcohol. 2004;34:81–87. doi: 10.1016/j.alcohol.2004.07.010. [DOI] [PubMed] [Google Scholar]
- 64.Marra F., Navari N., Vivoli E., Galastri S., Provenzano A. Modulation of liver fibrosis by adipokines. Dig Dis. 2011;29:371–376. doi: 10.1159/000329799. [DOI] [PubMed] [Google Scholar]
- 65.Novo E., Cannito S., Paternostro C., Bocca C., Miglietta A., Parola M. Cellular and molecular mechanisms in liver fibrogenesis. Arch Biochem Biophys. 2014;548:20–37. doi: 10.1016/j.abb.2014.02.015. [DOI] [PubMed] [Google Scholar]
- 66.Matsuzawa Y. Adiponectin: a key player in obesity related disorders. Curr Pharmaceut Des. 2010;16(17):1896–1901. doi: 10.2174/138161210791208893. [DOI] [PubMed] [Google Scholar]
- 67.Liu Y., Michael M.D., Kash S. Deficiency of adiponectin receptor 2 reduces diet-induced insulin resistance but promotes type 2 diabetes. Endocrinology. 2007;148(2):683–692. doi: 10.1210/en.2006-0708. [DOI] [PubMed] [Google Scholar]
- 68.Xie L., Boyle D., Sanford D., Scherer P.E., Pessin J.E., Mora S. Intracellular trafficking and secretion of adiponectin is dependent on GGA-coated vesicles. J Biol Chem. 2006;281(11):7253–7259. doi: 10.1074/jbc.M511313200. [DOI] [PubMed] [Google Scholar]
- 69.Halaas J.L., Gajiwala K.S., Maffei M. Weight-reducing effects of the plasma protein encoded by the obese gene. Science. 1995;269(5223):543–546. doi: 10.1126/science.7624777. [DOI] [PubMed] [Google Scholar]
- 70.Li Y., Ding L., Hassan W., Abdelkader D., Shang J. Adipokines and hepatic insulin resistance. J Diabetes Res. 2013:1–8. doi: 10.1155/2013/170532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Uemura S., Matsushita H., Li W. Diabetes mellitus enhances vascular matrix metalloproteinase activity role of oxidative stress. Circ Res. 2001;88(12):1291–1298. doi: 10.1161/hh1201.092042. [DOI] [PubMed] [Google Scholar]
- 72.Koneru M., Sahu B.D., Gudem S. Polydatin alleviates alcohol-induced acute liver injury in mice: relevance of matrix metalloproteinases (MMPs) and hepatic antioxidants. Phytomedicine. 2017;27:23–32. doi: 10.1016/j.phymed.2017.01.013. [DOI] [PubMed] [Google Scholar]
- 73.Chen G., Wang R., Chen H., Wu L., Ge R.S., Wang Y. Gossypol ameliorates liver fibrosis in diabetic rats induced by high-fat diet and streptozocin. Life Sci. 2016;149:58–64. doi: 10.1016/j.lfs.2016.02.044. [DOI] [PubMed] [Google Scholar]
- 74.Hou J.F., Wang X.G., Wei J., Jiao K. Frontier and Future Development of Information Technology in Medicine and Education. Springer; Dordrecht: 2014. Alteration of liver MMP-9/TIMP-1 and plasma type IV collagen in the development of rat insulin resistance; pp. 531–543. [Google Scholar]
- 75.Fracanzani A.L., Valenti L., Bugianesi E. The risk of severe liver disease in NAFLD with normal aminotransferase levels: a role for insulin resistance and diabetes. Hepatology. 2008;48:792–798. doi: 10.1002/hep.22429. [DOI] [PubMed] [Google Scholar]
- 76.Dongiovanni P., Valenti L., Rametta R. Genetic variants regulating Insulin Receptor signaling are associated with the severity of liver damage in patients with nonalcoholic fatty liver disease. Gut. 2010;59:267–273. doi: 10.1136/gut.2009.190801. [DOI] [PubMed] [Google Scholar]
- 77.Kohjima M., Enjoji M., Higuchi N. Re-evaluation of fatty acid metabolism-related gene expression in nonalcoholic fatty liver disease. Int J Mol Med. 2007;20(3):351–358. [PubMed] [Google Scholar]
- 78.Dorn C., Riener M.O., Kirovski G. Expression of fatty acid synthase in nonalcoholic fatty liver. Int J Clin Exp Pathol. 2010;3(5):505–514. [PMC free article] [PubMed] [Google Scholar]
- 79.Romeo S., Kozlitina J., Xing C. Genetic variation in PNPLA3 confers susceptibility to nonalcoholic fatty liver disease. Nat Genet. 2008;40(12):1461. doi: 10.1038/ng.257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Nakae J., Park B.C., Accili D. Insulin stimulates phosphorylation of the forkhead transcription factor FKHR on serine 253 through a Wortmannin-sensitive pathway. J Biol Chem. 1999;274(23):15982–15985. doi: 10.1074/jbc.274.23.15982. [DOI] [PubMed] [Google Scholar]
- 81.McGettrick A.J., Feener E.P., Kahn C.R. Human insulin receptor substrate-1 (IRS-1) polymorphism G972R causes IRS-1 to associate with the insulin receptor and inhibit receptor autophosphorylation. J Biol Chem. 2005;280(8):6441–6446. doi: 10.1074/jbc.M412300200. [DOI] [PubMed] [Google Scholar]
- 82.Li Y., Xu S., Mihaylova M.M. AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell Metabol. 2011;13(4):376–388. doi: 10.1016/j.cmet.2011.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Weedon M.N., Schwarz P.E., Horikawa Y. Meta-analysis and a large association study confirm a role for calpain-10 variation in type 2 diabetes susceptibility. Am J Hum Genet. 2003;73:1208–1212. doi: 10.1086/379285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Hamed A.E., Abas B., Shaltout I., Esmt G., Gomez R. Managing diabetes and liver disease association, guidelines (consensus) development. J Endocrinol Diabetes Obes. 2015;3:1–9. [Google Scholar]
- 85.Armstrong M.J., Houlihan D.D., Rowe I.A. Safety and efficacy of liraglutide in patients with type 2 diabetes and elevated liver enzymes: individual patient data meta-analysis of the LEAD program. Aliment Pharmacol Ther. 2013;37(2):234–242. doi: 10.1111/apt.12149. [DOI] [PubMed] [Google Scholar]
- 86.Ohki T., Isogawa A., Iwamoto M. The effectiveness of liraglutide in nonalcoholic fatty liver disease patients with type 2 diabetes mellitus compared to sitagliptin and pioglitazone. Sci World J. 2012:1–8. doi: 10.1100/2012/496453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gouni-Berthold I., Papanas N., Maltezos E. The role of oral antidiabetic agents and incretin mimetics in type 2 diabetic patients with non-alcoholic fatty liver disease. Curr Pharmaceut Des. 2014;20(22):3705–3715. doi: 10.2174/13816128113196660676. [DOI] [PubMed] [Google Scholar]
- 88.Komiya C., Tsuchiya K., Shiba K. Ipragliflozin improves hepatic steatosis in obese mice and liver dysfunction in type 2 diabetic patients irrespective of body weight reduction. PLoS One. 2016;11(3):e0151511. doi: 10.1371/journal.pone.0151511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Moslehi A., Nabavizadeh F., Zekri A., Amiri F. Naltrexone changes the expression of lipid metabolism-related proteins in the endoplasmic reticulum (ER) stress induced hepatic steatosis in mice. Clin Exp Pharmacol Physiol. 2016;44(2):207–212. doi: 10.1111/1440-1681.12695. [DOI] [PubMed] [Google Scholar]
- 90.Li Y., Zalzala M., Jadhav K. Carboxylesterase 2 prevents liver steatosis by modulating lipolysis, ER stress and lipogenesis and is regulated by HNF4α. Hepatology. 2016;63(6):1860–1874. doi: 10.1002/hep.28472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Newberry E.P., Xie Y., Kennedy S.M. Prevention of hepatic fibrosis with liver microsomal triglyceride transfer protein deletion in Liver fatty acid binding protein null mice. Hepatology. 2016;65(3):836–852. doi: 10.1002/hep.28941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Gong Q., Hu Z., Zhang F. Fibroblast growth factor 21 improves hepatic insulin sensitivity by inhibiting mammalian target of rapamycin complex 1. Hepatology. 2016;64(2):425–438. doi: 10.1002/hep.28523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Mazzotti A., Caletti M.T., Marchignoli F., Forlani G., Marchesini G. Which treatment for type 2 diabetes associated with non-alcoholic fatty liver disease? Dig Liver Dis. 2016;49(3):235–240. doi: 10.1016/j.dld.2016.12.028. [DOI] [PubMed] [Google Scholar]
- 94.Kuchay M.S., Krishan S., Mishra S.K. Effect of empagliflozin on liver fat in patients with type 2 diabetes and nonalcoholic fatty liver disease: a randomized controlled trial (E-LIFT trial) Diabetes Care. 2018:1–8. doi: 10.2337/dc18-0165. [DOI] [PubMed] [Google Scholar]
- 95.Staels B., Rubenstrunk A., Noel B. Hepatoprotective effects of the dual peroxisome proliferator-activated receptor alpha/delta agonist, GFT505, in rodent models of nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. Hepatology. 2013;58(6):1941–1952. doi: 10.1002/hep.26461. [DOI] [PubMed] [Google Scholar]
- 96.Al-muzafar H.M., Amin K.A. Probiotic mixture improves fatty liver disease by virtue of its action on lipid profiles, leptin, and inflammatory biomarkers. BMC Compl Alternative Med. 2017;17(1):43. doi: 10.1186/s12906-016-1540-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Yokohama K., Fukunishi S., Ii M. Rosuvastatin as a potential preventive drug for the development of hepatocellular carcinoma associated with non-alcoholic fatty liver disease in mice. Int J Mol Med. 2016;38(5):1499–1506. doi: 10.3892/ijmm.2016.2766. [DOI] [PubMed] [Google Scholar]
- 98.Lee Y.H., Kim J.H., Kim S.R. Lobeglitazone, a novel thiazolidinedione, improves non-alcoholic fatty liver disease in type 2 diabetes: its efficacy and predictive factors related to responsiveness. J Kor Med Sci. 2017;32(1):60–69. doi: 10.3346/jkms.2017.32.1.60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Neuschwander-Tetri B.A., Loomba R., Sanyal A.J. Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): a multicentre, randomised, placebo-controlled trial. Lancet. 2015;385(9972):956–965. doi: 10.1016/S0140-6736(14)61933-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Hoofnagle J.H., Natta M.L., Kleiner D.E. Vitamin E and changes in serum alanine aminotransferase levels in patients with non-alcoholic steatohepatitis. Aliment Pharmacol Ther. 2013;38(2):134–143. doi: 10.1111/apt.12352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Hemant Thacker M.D., Shrimanker R., Tulle R. Novel action of saroglitazar in patients with diabetic dyslipidemia-an observational study. Endod Pract. 2016;22:183. [Google Scholar]