Abstract
Background
Stroke is a significant burden in Saudi Arabia and the Saudi Ministry of Health's stroke committee has identified an urgent need to improve care.
Aim
The purpose of this study was to undertake a health-economic analysis to quantify the impact of developing stroke care in the country.
Methods
An economic model was developed to assess the costs and clinical outcomes associated with an ischemic stroke care development program compared with current stroke care. Based on Saudi epidemiological data, cohorts of ischemic stroke patients enter the model each year for the first 10 years based on increasing incidence. Four treatment options were modeled including reperfusion and non-reperfusion treatments. The development scenario estimates the impact of gradually increasing uptake of more effective treatments over 10 years. Changes in the stroke care organization are considered along with resources required to increase capacity, allowing more patients to be admitted to stroke hospitals and access effective treatments.
Results
The stroke care development program is associated with an increase in functionally independent patients and a decrease in disabling strokes compared with current stroke care. Additionally, the development program is associated with estimated cost savings of $602 million over 15 years ($255 million direct costs, $348 million indirect costs).
Conclusions
The model predicts that the stroke care development program is associated with improved patient outcomes and lower overall costs compared with the current stroke care program.
Keywords: Cost analysis, economic model, Saudi Arabia, cost-effectiveness, ischemic stroke, stroke care
Introduction
Stroke is a major cause of morbidity and disability throughout the world.1 Across Saudi Arabia, stroke prevalence rates are estimated at 0.67%.2 Two decades ago, the rate of first stroke incidence was reported at 29.8 per 100,000 person-years, which when updated to the current demographic distribution leads to an estimated rate of 50.9/100,000.3 Similar rates were reported in a recent study in the Aseer region (57.64/100,000).4 The combination of an aging population and increased risk of stroke with age is expected to lead to a growing stroke burden from 16,900 first strokes up to 28,400 in the next 10 years.3
Treatment guidelines recommend reperfusion therapy for ischemic stroke (IS). Intravenous tissue plasminogen activator (IV-tPA) is a treatment with moderate benefit that can be administered up to 4.5 h after symptom onset. Mechanical thrombectomy (MT) is a treatment with substantial benefit that can be administered up to 6–8 h after symptom onset, and up to 24 h in imaging-selected patients.5 A challenge for stroke care systems is to achieve optimal patient flow, quickly and correctly identifying patients eligible for reperfusion treatments and transferring them to the appropriate center.6
Stroke care in Saudi Arabia lags behind other developed countries.7 Approximately 95% of patients are cared for at non-specialized stroke hospitals and receive non-reperfusion supportive care for both large vessel occlusion (LVO) and non-large vessel occlusion (non-LVO). Around 5% of patients are admitted to stroke units, among whom approximately 3.6% receive IV-tPA; fewer than 100 patients receive MT annually. This is broadly due to a lack of organization of care and inconsistent allocation of resources.7 Furthermore, a lack of care standards and incentives for patient management strategies leads to sub-optimal patient outcomes.
An urgent need for stroke care development was identified by the Saudi Arabian Ministry of Health's stroke committee, which coordinated efforts to analyze the implications of improving care, to support the development of a plan.
Aim
The study aimed to undertake a health-economic analysis, comparing current stroke care in Saudi Arabia with a proposed stroke care development program, and to assess the associated clinical and cost outcomes. This included modeling the establishment of specialized stroke units to increase utilization of IV-tPA and MT, and changing the current organization of care and patient flow to provide quicker access to treatment.
Method
Extensive research was undertaken to determine current stroke care provision in Saudi Arabia, and international stroke services were examined to identify the optimal model for developing stroke care. The economic analysis was conducted from a societal perspective. The model was designed in collaboration with a panel of leading Saudi stroke consultants, which was consulted to confirm differences in patient pathways, treatment rates, length of stay and discharge destinations.
Model structure
The model structure was based on a published cost-effectiveness model comparing reperfusion treatments.8 The model uses a Markov structure split into an acute phase from stroke onset to 90 days, and a rest-of-life phase from 91 days until death. The modified Rankin Scale (mRS) was used to characterize seven health states,9 and all treatment effects were assumed to occur within the acute phase. In the rest-of-life phase, patients remained in the same mRS as at 90 days until either a recurrent stroke or death.8 This structure was modified to calculate mRS scores, costs and quality-adjusted life-years (QALYs) for each IS patient.
Ten cohorts of IS patients entered the model over 10 years and we estimated lifetime costs of these patients. The patient numbers in each new cohort were based on increasing incidence figures to account for the effect of an aging population. A decision tree structure (Figure 1) was used to separate each cohort into each program and stroke type (LVO or non-LVO). The population was also separated into Saudi and non-Saudi because one-third of the population is non-native. According to local specialists, many non-Saudis return to their home countries after a disabling stroke since their main purpose for being in Saudi Arabia was employment (all patients have equivalent access to care, so this aspect is unaffected). However, it can result in different long-term and societal costs, arising only from their non-permanence in the country, and not from the care that they would receive if they were to remain. Treatment options included MT+IV-tPA, IV-tPA alone, MT alone, and non-reperfusion treatment and were available depending on stroke type. A gradual uptake of reperfusion treatments was assumed in the development program, which was estimated based on international evidence.10–15
Figure 1.
Stroke care programs overview.
Progressive investment costs and resource allocation were considered to model changes to the stroke care pathway for the development program.
The base case considers a 15-year time horizon to allow some benefits to accrue for the final cohort entering the model in year 10. Future health outcomes and costs were discounted at 3% per year.16 All costs are presented in US dollars ($1 = 3.75 Saudi Riyals, January 2019).
Patient information
Total populations of 20,768,627 Saudi and 12,645,033 non-Saudi individuals were included and used age- and gender-specific stroke incidence figures to calculate the annual number of strokes over 10 years. Incidence figures ranged from 50.9/100,000 people to 75.4/100,000 of the projected population.3,17 Ischemic strokes and LVO strokes were assumed to make up 85% and 34% of all strokes, respectively.15 The mean age of stroke for the base-case scenario was 62.4 Current treatment utilization was estimated using local data validated by the consultant panel (Supplement Table I).
Clinical effectiveness
The impact of a learning curve is often overlooked in the economic evaluation of medical devices, and can affect short- and long-term patient outcomes and complication rates.18 MT is a highly specialized procedure, requiring operator skills developed over time. Given the current facility shortage, lack of organization of stroke care and patient access to IV-tPA and MT, two different data sources were used for short-term LVO patient outcomes for the current and development scenarios.
For the current program, outcomes data were applied from MR CLEAN, a study undertaken in the Netherlands in centers with variable experience with MT and with less restrictive patient selection compared with other randomized controlled trials (RCTs).19 Patient outcomes were used as a baseline to reflect the current experience level in Saudi Arabia. For the development program, data from the HERMES collaboration meta-analysis were applied, representing studies involving centers with more homogeneous advanced experience using MT.20 Given the effort towards creating specialized stroke units in Saudi Arabia, these data may better represent future patient outcomes as experience and skill level increase. Clinical efficacy data for non-LVO strokes were taken from a subgroup analysis of a pooled analysis of nine RCTs comparing alteplase with placebo/open control (Supplement Table II).21 Adverse event rates and recurrent stroke probabilities are detailed in Supplement Table II.20,22,23
Mortality
Age-specific other-cause mortality was applied in the rest-of-life phase to model deaths unrelated to stroke.24 Other-cause mortality was adjusted to reflect the higher mortality risk observed in stroke survivors.25
Unit costs and resource use
Treatment costs included devices, drugs, staff, diagnostics and triage. They were estimated by analyzing resource use from Ministry of Health (MOH) hospitals. (Supplement Table III).
Acute costs were dependent on each program. Two types of hospitals were considered: non-specialized stroke hospital and stroke hospitals. In the current program, the majority of patients (95%) were admitted to non-specialized stroke hospitals. In the development program, a gradual increase of patients cared for at stroke hospitals was projected, up to a maximum of 70%. Additionally, stroke hospitals in the development program were assumed to have more efficient inpatient diagnostic and rehabilitation care, resulting in shorter length of stay (LOS) and earlier discharge.26 All acute and long-term costs were calculated based on LOS data for different wards and facility types. These data were based on local data from MOH hospitals and validated by the consultant panel. Cost per bed, tests, and hospital visits were based on the official price list from MOH 2018, adjusted for hospital specialization level. (Supplement Table III)
Three discharge destinations were included within acute costs: home with outpatient rehabilitation, inpatient rehabilitation, and long-term care facility (nursing home). Patients were routed to different discharge destinations and had differing LOS depending on their mRS score.
The same discharge destinations were used for the post-acute phase costs (90 days to one year), but were dependent on the stroke care program, rather than the hospital type. After one year, patients with outpatient rehabilitation and inpatient rehabilitation were assumed return home and have neurologist follow-up appointments only. Longer term care was included as a yearly cost for the rest-of-life for patients with more disabling strokes.
In the base-case, it was assumed that all non-Saudi patients with mRS≤2 would remain in Saudi Arabia and incur longer term care costs. However, 70% of mRS 3 patients, and 20% of mRS 4 to 5 patients would remain in the country.
Recurrent stroke costs included treatment and acute care. Patients were assumed to receive the same treatment as for their previous stroke except those with an mRS ≥3, who were assumed to receive non-reperfusion management.5,27
Productivity losses were included to reflect the burden on society using the human capital approach. The population was split by Saudi and non-Saudi, gender, and age to reflect differences in wage and labour participation (Supplement Tables IV to VII).17,28,29
Investment in an additional biplane angiosuite was included in the development program, for which the cost provided by the consultant panel reflected the official purchasing cost of this equipment by MOH hospitals (Supplement Table VIII). The full cost of each investment was only applied for the first 10 years; after this time, new patients stop entering the model, therefore it would benefit future patients whose outcomes are not captured within the model. After the 10-year period, the annual costs are weighted by the proportion of recurrent stroke patients, as they would continue to benefit from investments.
Model outcomes
The primary model outcomes are the incremental costs and QALYs with the development program. A deterministic sensitivity analysis was performed to explore uncertainty around input parameters. Probabilistic sensitivity analysis was also undertaken to assess overall model uncertainty, varying all model parameters simultaneously to generate 1,000 sampled sets of inputs and outputs.30 Internal validity was confirmed by a health economist not involved in the model development.
Results
With 192,474 IS patients modeled over 10 years, the development program is associated with overall cost savings of $602 million over a 15 year time horizon, with a higher estimate of around $629 million when lifetime costs are considered. An estimated $255 million is saved in direct costs to the healthcare system, with further savings of $348 million in indirect costs to society. The key reductions in cost result from decreased acute hospital and long-term nursing costs, which outweigh the cost of developing stroke care provision (Figure 2). A reduction is also predicted for post-90-day inpatient/outpatient rehabilitation and societal costs. Other categories show a cost increase (Supplement Table IX).
Figure 2.
Cost breakdown by stroke care program.
The development program was associated with better health outcomes showing higher QALYs (0.29) and life years (LYs) (0.25) gained per patient at 15 years compared with the current program (Table 1). The effect was greater when only LVO strokes were considered, with a predicted incremental 0.79 QALYs and 0.71 LYs over a lifetime horizon.
Table 1.
Base case results
| Category | Current stroke care program | Stroke care development program | Incremental |
|---|---|---|---|
| Total costs | $9,244,395,513 | $8,641,826,427 | −$602,569,086 |
| Costs/patient | $48,029 | $44,899 | $−3,131 |
| QALYs/patient | 2.08 | 2.36 | 0.29 |
| Life-years/patient | 3.50 | 3.75 | 0.25 |
| ICER | Dominant |
QALYs: quality-adjusted life-years. ICER: incremental cost-effectiveness ratio.
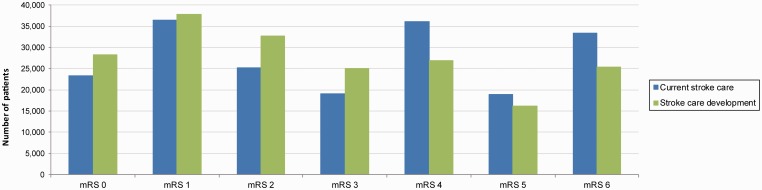
Modified Rankin scores are estimated at 90 days for both programs and stroke types (Figure 3). Approximately 30% of LVO patients have an mRS ≤2 in the development program compared with 12% in the current program, equalling an increase of 18% in functionally independent patients and a decrease in severe disability.
Figure 3.
Number of patients at each mRS at 90 days by program.
Sensitivity analysis
Deterministic sensitivity analysis indicates the development program remains cost-saving across changes in a large range of parameters. From 1,000 probabilistic simulations, the development program was associated with greater QALYs in all estimates and is predicted to have cost savings in 77.9% of cases (Figure 4).
Figure 4.
Probabilistic sensitivity analysis (a) Percentage of iterations for incremental cost. (b) Percentage of iterations for proportion of patients with mRS≤2 for each program.
Discussion
This study goes beyond traditional cost-effectiveness and budget impact analysis to provide a framework for evaluating costs and outcomes in a region where the development of integrated and organized stroke care programs is critical to meet the need for quality care. The flexibility and novelty of the model allow different scenarios to be explored according to the gradual expected changes in treatment patterns for all IS patients, investments required, and the combination of features not previously observed. The development program is predicted to be cost-saving with estimated savings of US$602 million and a QALY gain of 0.29 (per patient) over 15 years compared to the current program. The key drivers as identified in sensitivity analyses are the mean age of stroke, total acute hospital costs, and total long-term nursing home costs. Varying these parameters individually did not change the direction of the results. Probabilistic sensitivity analysis also indicated that the model results are robust to changes in input parameters.
As with all economic models, various assumptions were made. As local published clinical data were lacking, the data used in the current program came from an RCT conducted in the Netherlands.19 This was chosen to represent current stroke care because it was undertaken in centers with limited prior experience of treating patients with MT. Data from the HERMES meta-analysis, used to represent clinical outcomes in the development scenario, were also based on trials from countries other than Saudi Arabia. Further, combining the two different data sources in this way assumes the differences in mRS outcomes were due to differences in medical practitioner experience and stroke care, whereas such differences could have occurred for many reasons. Therefore, factors unrelated to treatment could have caused additional variation in mRS scores.
Stroke incidence rates are based on a Saudi study from the 1990s as this was the only source to report separate values by age group, which was necessary to accurately reflect changes in stroke risk with age. The recent study in the Aseer region reported similar incidence rates; however, it showed an unusual pattern in the two most elderly groups of patients, where the stroke rate was considerably higher for males than females, which may not accurately reflect the stroke numbers amongst the projected population of women aged 65 years and over.
Adapting the model from an existing model led to the assumption that all patients have the same age at first stroke. This was adjusted for the inclusion of societal costs, but for other parameters, an average starting age was assumed for all patients to calculate healthcare costs. Likewise, patients are assumed to stay at the same mRS after their first stroke until either recurrent stroke or death. Recurrent strokes are also assumed to occur after the first 90 days and can occur once per year.8
The organization of care considered in the model includes at least one comprehensive stroke hospital (CSH) in each region (13 in total), with more CSHs in more densely populated areas to cover higher stroke care demands. The CSH will be served by an integrated care stroke network with urgent referral systems from primary stroke hospitals (PSH) and acute stroke ready hospitals (ASRH). This organization has been included in the stroke standards developed by the Stroke Clinical Advisory Committee, chaired by the first author.
The model considers most of the resources required to implement the changes in the organization of care, which in several cases will imply a reallocation of resources such as beds and ambulances. The inclusion of additional technologies such as telestroke, imaging or prenotification software could be possible due to the model flexibility; nonetheless, the costs of their inclusion are unlikely to alter the model results. Additional inpatient rehabilitation and long-term services required for the development program will be outsourced by the private sector, and will therefore not be an MOH investment.
A strength of the analysis was the extensive input from local stroke specialists, who provided all local data and validated the model. The model also reflects the impact of gradual implementation of changes in the development program until it reaches an optimal operation/functional level. Moreover, this model is a useful tool to estimate the future state of stroke care and can be replicated in other regions to assess stroke care organization alternatives to inform better resource allocation from healthcare decision-makers.
Conclusion
Ischemic stroke provides a suitable case study for economic models given the complexity of the care pathway, facilities, and expertise required to provide specific interventions. Saudi Arabia's demographic situation, coupled with the current sub-optimal care available for stroke, make it a prime candidate to explore future needs and the impact of infrastructure and patient management changes upon cost and patient outcomes. The development of stroke care could improve patient outcomes and lower overall costs compared with the current stroke care provision. The model outcomes enabled the decision-making process towards the improvement of stroke care in Saudi Arabia.
Supplemental Material
Supplemental Material for A national economic and clinical model for ischemic stroke care development in Saudi Arabia: A call for change by Fahmi Al-Senani, Mohammed Al-Johani, Mohammad Salawati, Souda ElSheikh, Maha AlQahtani, Jamal Muthana, Saeed AlZahrani, Judith Shore, Matthew Taylor, Valeska S Ravest, Simon Eggington, Matthieu Cuche, Heather Davies, Kyriakos Lobotesis and Jeffrey L Saver in International Journal of Stroke
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JLS is an employee of the University of California, which has patent rights in stroke retrieval devices. He serves as an unpaid consultant to Genentech advising on PRISMS trial. JLS has received contracted hourly payments and travel reimbursement for services as a scientific consultant advising on trial design and conduct to Medtronic/Covidien, Stryker, Neuravi/Cerenovus, BrainsGate, Boehringer Ingelheim, and Diffusion Medical. JLS has received contracted stock options for services as a scientific consultant advising on trial design and conduct to Rapid Medical. FA-S, MS and MA-J have received reimbursement from Medtronic for travel costs for a workshop related to this analysis. MT, JS, and HD are employed by YHEC. MC, SE, and VSR are employees of Medtronic. No other authors have any declarations.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: York Health Economics Consortium (YHEC) received funding from Medtronic for the model development.
References
- 1.Robert AA, Mohamed Zamzami M. Stroke in Saudi Arabia: a review of the recent literature. Pan Afr Med J 2014; 17: 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Alahmari K, Paul SS. Prevalence of stroke in kingdom of Saudi Arabia – through a physiotherapist diary. Mediterr J Soc Sci 2016; 7: 228–233. [Google Scholar]
- 3.Al-Rajeh S, Larbi EB, Bademosi O , et al. Stroke register: experience from the eastern province of Saudi Arabia. Cerebrovasc Dis 1998; 8: 86–89. [DOI] [PubMed] [Google Scholar]
- 4.Alhazzani, AA Mahfouz AA, Abolyazid AY , et al. Study of stroke incidence in the aseer region, southwestern Saudi Arabia. Int J Environ Res Public Health 2018; 15: 215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Powers WJ, Rabinstein AA, Ackerson T , et al. Guidelines for the early management of patients with acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2018; 49: e46–110. [DOI] [PubMed] [Google Scholar]
- 6.Fiehler J, Cognard C, Gallitelli M , et al. European recommendations on organisation of interventional care in acute stroke (EROICAS). Eur Stroke J 2016; 11: 701–716. [DOI] [PubMed] [Google Scholar]
- 7.Alanazy MH, Barakeh RB, Asiri A , et al. Practice patterns and barriers for intravenous thrombolysis: a survey of neurologists in Saudi Arabia. Neurol Res Int 2018; 8: 1–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lobotesis K, Veltkamp R, Carpenter IH, Claxton LM, Saver JL, Hodgson R. Cost-effectiveness of stent-retriever thrombectomy in combination with IV t-PA compared with IV t-PA alone for acute ischemic stroke in the UK. J Med Econ 2016; 19: 785–794. [DOI] [PubMed] [Google Scholar]
- 9.Van Swieten JC, Koudstaal PJ, Visser MC, Schouten H, Van Gijn J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke 1988; 19: 604–607. [DOI] [PubMed] [Google Scholar]
- 10.Rai AT, Seldon AE, Boo S , et al. A population-based incidence of acute large vessel occlusions and thrombectomy eligible patients indicates significant potential for growth of endovascular stroke therapy in the USA. J Neurointerv Surg 2017; 9: 722–726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tawil SE, Cheripelli B, Huang X , et al. How many stroke patients might be eligible for mechanical thrombectomy? Eur Stroke J 2016; 1: 264–271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nogueira RG, Jadhav AP, Haussen DC , et al. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med 2017; 378: 11–21. [DOI] [PubMed] [Google Scholar]
- 13.Rai AT, Domico JR, Buseman C , et al. A population-based incidence of M2 strokes indicates potential expansion of large vessel occlusions amenable to endovascular therapy. J Neurointerv Surg 2017; 10: 510–515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chia NH, Leyden JM, Newbury J, Jannes J, Kleinig TJ. Determining the number of ischemic strokes potentially eligible for endovascular thrombectomy: a population-based study. Stroke 2016; 47: 1377–1380. [DOI] [PubMed] [Google Scholar]
- 15.McMeekin P, White P, James MA, Price CI, Flynn D, Ford GA. Estimating the number of UK stroke patients eligible for endovascular thrombectomy. Eur Stroke J 2017; 2: 319–326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.World Health Organization. Making choices in health: WHO guide to cost-effectiveness analysis. Geneva: Global Program Evidence Health Policy, World Health Organization, 2003.
- 17.General Authority for Statistics (GASTAT). Demographic survey, www.stats.gov.sa/en/43 (2019, accessed 28 January 2019).
- 18.Tarricone R, Callea G, Ogorevc M, Prevolnik Rupel V. Improving the methods for the economic evaluation of medical devices. Health Econ 2017; 26(Suppl 1): 70–92. [DOI] [PubMed] [Google Scholar]
- 19.Berkhemer OA, Fransen PSS, Beumer D , et al. A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med 2015; 372: 11–20. [DOI] [PubMed] [Google Scholar]
- 20.Goyal M, Menon BK, Van Zwam WH , et al. Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. Lancet 2016; 387: 1723–1731. [DOI] [PubMed] [Google Scholar]
- 21.Lees KR, Emberson J, Blackwell L , et al. Effects of alteplase for acute stroke on the distribution of functional outcomes: a pooled analysis of 9 trials. Stroke 2016; 47: 2373–2379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Whiteley WN, Emberson J, Lees KR , et al. Risk of intracerebral haemorrhage with alteplase after acute ischaemic stroke: a secondary analysis of an individual patient data meta-analysis. Lancet Neurol 2016; 15: 925–933. [DOI] [PubMed] [Google Scholar]
- 23.Mohan KM, Wolfe CDA, Rudd AG, Heuschmann PU, Kolominsky-Rabas PL, Grieve AP. Risk and cumulative risk of stroke recurrence: a systematic review and meta-analysis. Stroke 2011; 42: 1489–1494. [DOI] [PubMed] [Google Scholar]
- 24.World Health Organization. Global health observatory data repository: Saudi arabia, www.who.int/countries/sau/en/ (2018, accessed 28 January 2018).
- 25.Slot KB, Berge E, Sandercock P, Lewis SC, Dorman P, Dennis M. Causes of death by level of dependency at 6 months after ischemic stroke in 3 large cohorts. Stroke 2009; 40: 1585–1589. [DOI] [PubMed] [Google Scholar]
- 26.Bindawas SM, Vennu VS. Stroke rehabilitation: a call to action in Saudi Arabia. Neurosciences 2016; 21: 297–305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.NHS England. Clinical commissioning policy: mechanical thrombectomy for acute ischaemic stroke (all ages). England: Author, 2018.
- 28.General Authority for Statistics (GASTAT). Labor force survey, www.stats.gov.sa/en/814 (2017, accessed 28 January 2018).
- 29.Steen Carlsson K, Andsberg G, Petersson J, Norrving B , et al. Long-term cost-effectiveness of thrombectomy for acute ischaemic stroke in real life: an analysis based on data from the Swedish Stroke Register (Riksstroke). Int J Stroke 2017; 12: 802–814. [DOI] [PubMed] [Google Scholar]
- 30.Briggs AH, Claxton K, Sculpher MJ. Decision modelling for health economic evaluation. (Oxford handbooks in health economic evaluation). Oxford: Oxford University Press, 2006, p.237.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Material for A national economic and clinical model for ischemic stroke care development in Saudi Arabia: A call for change by Fahmi Al-Senani, Mohammed Al-Johani, Mohammad Salawati, Souda ElSheikh, Maha AlQahtani, Jamal Muthana, Saeed AlZahrani, Judith Shore, Matthew Taylor, Valeska S Ravest, Simon Eggington, Matthieu Cuche, Heather Davies, Kyriakos Lobotesis and Jeffrey L Saver in International Journal of Stroke