Abstract
Background
While Zika virus (ZIKV) infection is typically self-limited, congenital birth defects and Guillain-Barré syndrome are well-described. There are no approved ZIKV vaccines.
Methods
ZIKA-001, a phase I, open label, clinical trial evaluated the safety and immunogenicity of synthetic, consensus DNA vaccine (GLS-5700) encoding the ZIKV pre-membrane+envelope proteins (prME). Two groups of 20 participants received either 1 or 2 mg of vaccine intradermally at 0, 4, and 12 weeks followed by electroporation with the CELLECTRA®-3P device
Results
Participants’ median age was 38 years; 60% female 30% Latino and 78% Caucasian. No serious adverse events (SAEs) have been reported through week 14 interim analysis. Local minor reactions including injection site pain, redness, swelling and itching, were experienced by half of participants. Systemic AEs were rare.
After the third vaccination, binding antibodies, measured by ELISA, were detected in all participants with GMTs of 1642 and 2871 for 1 and 2 mg vaccine recipients, respectively. 62% of participants developed neutralizing antibodies in a Vero cell assay. In a neuronal cell assay, 70% and 95% of participants’ sera blocked infection by 90% and 50% respectively. Passive transfer of post-vaccination sera protected 92% of interferon (IFN) α/β-receptor knockout (IFNAR−/−) mice challenged with a lethal dose of ZIKV-PR209 strain whereas none of the mice receiving baseline sera survived. Survival was independent of neutralization titer. These data suggest that vaccine-induced humoral responses are protective.
Conclusion
Our trial shows for the first time in humans the safety and immunogenicity of an engineered DNA vaccine against ZIKV. Future studies will evaluate its efficacy.
INTRODUCTION
Zika virus (ZIKV) is a flavivirus originally discovered in a sentinel rhesus macaque in Uganda in 19471 and is endemic in Africa and Asia. Following outbreaks in Yap Island and French Polynesia2,3, ZIKV was identified in Brazil in 20154 and has spread rapidly throughout the Americas5. ZIKV infection is typically self-limited, presenting with fever, rash, conjunctivitis, arthralgias6 and, uncommonly, neurologic syndromes such as Guillain-Barré syndrome7. ZIKV infection during pregnancy is associated with severe congenital birth defects8. Recent studies suggest that ZIKV can persist in bodily fluids, particularly semen, for up to 6 months after infection9.
ZIKV is generally transmitted by the bite of infected mosquitoes (Aedes aegypti and other members of the family). ZIKV has also been transmitted via sexual contact10,11, blood transfusion12,13 and laboratory exposures14.
There are no approved ZIKV-specific therapies or vaccines.
Our novel, synthetic, DNA vaccine targeting the ZIKV pre-membrane and envelope proteins (prME) delivered by the CELLECTRA®-3P electroporation (EP) device has been shown pre-clinically to generate cellular and humoral immunity, including neutralizing antibodies in mice and non-human primates, to protect against infection in interferon (IFN) α/β receptor knockout (IFNAR−/−) mice, and protect non-human primates from challenge15. Here, we conducted a phase I study to evaluate the safety and immunogenicity of this novel, synthetic, ZIKV prME DNA vaccine, GLS-5700, delivered by EP-enhanced intradermal (ID) injection.
METHODS
Study Design and Participants
ZIKA-001 is a phase I, dose-ranging, open label, clinical trial designed to evaluate the safety, side effect profile, and immunogenicity of an investigational ZIKV vaccine, GLS-5700,15 administered by intradermal injection followed by EP. Eligible participants were healthy Dengue virus (DENV) seronegative adults, 18-65 years of age. Full details of inclusion and exclusion criteria and the schedule of events can be found in the protocol, available with the full-length article at NEJM.org. Participants were recruited at 3 locations in the US and Canada: The University of Pennsylvania Clinical Trials Unit, Philadelphia, PA; QPS Miami Research Associates, Miami, FL and Université Laval, Québec City, Canada. This study was reviewed and approved by institutional review boards at each institution. Participants provided written informed consent before enrollment. The study was sponsored by GeneOne Life Science, Inc. and co-developed with Inovio Pharmaceuticals, Inc. (ClinicalTrials.gov # NCT02809443).
Vaccine
The vaccine was manufactured under cGMP by VGXI (The Woodlands, TX). GLS-5700 contains plasmid pGX7201 at a 10 mg/ml concentration in sodium salt citrate buffer. Plasmid pGX7201 encodes for the ZIKV pre-membrane (prM) and envelope (E) proteins, generated as a consensus of pre-2016 human infectious ZIKV strain sequences available in GenBank, and cloned into the modified pVax1 expression vector, pGX000115.
Study Procedures
Two participant groups (n=20/group) received GLS-5700 at one of two dose levels: 1 mg or 2 mg DNA/dose. Vaccine was administered as 0.1 ml (1 mg) ID injections followed by EP at the site of inoculation, in order to increase the immunogenicity of the vaccine16,17. Participants received one or two injections into the deltoid region during vaccinations at 0, 4, and 12 weeks. EP with CELLECTRA-3P used four 52 millisecond pulses at 0.2 amps (40-200 volts depending on tissue resistance) per session. The first 2 pulses were spaced 0.2 seconds apart then a 3 second dwell before the final 2 pulses spaced 0.2 seconds apart. EP needle arrays were 3 mm long. Dose escalation from 1 mg to 2 mg and recruitment of the remaining 1 mg cohort occurred following safety evaluation by the study Safety Monitoring Committee following initial dosing of the first five 1 mg group participants.
Safety Evaluations
Participants recorded post-treatment local and systemic reactions using a Post-Vaccination Memory Aid for 7 days following each dose. Local administration site reactions and systemic events recorded in the participant’s memory aid were captured by study staff on clinical report forms. Adverse events (AE) were graded according to the “Toxicity Grading Scale for Healthy Adult and Adolescent Volunteers Enrolled in Preventive Vaccine Clinical Trials”, issued by the FDA in September 2007. Screening and safety laboratory evaluations included complete blood counts, comprehensive metabolic panels, AST, ALT and CPK. Participants with an abnormal baseline electrocardiogram were excluded from the study because of a theoretical arrhythmogenicity associated with the EP, a procedure that generates a small electric impulse. However, EP-associated arrhythmias have not been observed in trials17.
Assessment of ZIKV Specific Antibody Responses
Blood samples to measure vaccine specific immune responses were scheduled for collection at weeks 0 (baseline), 1, 4, 8, 12, 14, 24, 36, and 60. Serum was used in an ELISA to measure binding antibody responses to recombinant vaccine matched ZIKV envelope (rZIKV-E) protein15 and reported as endpoint titer. We measured neutralizing antibody against ZIKV in participant serum using two different assays. First, a 50% micro-neutralization (MN50) assay in Vero cells(for details about this assay see Table S1 in the Supplementary Appendix, available at NEJM.org) Additionally, an immunofluorescence-based neutralization assay using human glioblastoma cells (U87MG), a model for ZIKV infection of neural progenitor cells19. For this assay, ZIKV-MR766 was pre-incubated with a single 1:25 dilution of either week 0 or week 14 sera for 1.5 hours and then added to monolayers of U87MG cells. Four days postinfection, cells were fixed and subjected to indirect immunofluorescence with a pan-flavivirus antibody to detect virus infected cells. The proportion and 95% CI of participants’ week 14 sera that inhibited infection relative to baseline by 50 and 90% was quantified by two independent reviewer’s analyses of images.
Assessment of ZIKV Specific T-Cell Responses
Whole blood was processed to obtain peripheral blood mononuclear cells (PBMC) which were frozen for subsequent ELISpot analysis. (for details about this assay see Table S1 in the Supplementary Appendix, available at NEJM.org) The average number of spot forming units (SFU) counted in media control wells was subtracted from the average in individual ZIKV peptide wells and then adjusted to 1×106 PBMCs for each ZIKV peptide pool. The total ZIKV specific response is the sum of the 3 individual peptide pools.
Assessment of in vivo Protection of Post-Vaccination Serum
We evaluated the induction of protective capability of vaccine-induced antibodies against ZIKV infection in the IFNAR−/−murine model20. Immunocompetent mice develop a non-lethal short lived infection to ZIKV21, whereas IFNAR−/− mice develop lethal infection within 6–7 days22. IFNAR−/− mice were intraperitoneally (IP) administered 0.1 ml of either phosphate buffered saline (PBS, control) or serum collected from participants at week 0 or week-14 (after 3rd dose of the vaccine), and challenged intraperitoneally one hour later with 1×105 plaque-forming units (PFU) of Puerto Rico ZIKV clinical strain PR209. The animals were followed for clinical signs of disease twice daily up to 14 days15. Animal challenge studies were conducted in accordance with the Wistar Institute IACUC guidelines.
Statistical Analysis
The antibody binding response assessed by ELISA is reported as the proportion of individuals (95% CI) developing an antibody response at a given time point and as the geometric mean titer with the 95% CI. Between groups comparisons were performed using Fisher exact test of positive response rates and Student t test to compare the magnitude of the antibody response between groups and within individuals and baseline after log transformation. Spearman correlation was used to evaluate the correlation between titers for binding and neutralizing antibody responses. The Mann Whitney test was used for comparisons of the magnitude of the T-cell responses between the two dose groups.
RESULTS
Forty participants were enrolled at 3 clinical research sites in Philadelphia, Miami and Québec City between August and September 2016 (Figure 1). Participant demographic characteristics are summarized in Table S2 (Supplementary Appendix, available at NEJM.org). The median age was 38 (interquartile range 30-54) years. Participants were 60% female, 30% Latino and 78% Caucasian.
Figure 1. Screening, Enrollment, Vaccinations, and Follow-up.

The 1 and 2 mg groups were enrolled sequentially according to a dose-escalation protocol. After the first 5 participants received the 1 mg dose, a safety committee reviewed the safety profile and agreed to continue with enrollment at 1 mg dose and proceed with 2 mg dose escalation enrollment. The rest of the participants were allocated to the 1 or 2 mg dose sequentially at each site. All available study data and samples were used for study analyses. All participants but 1 completed the study-injection regimen and 24 weeks of follow-up. One participant was lost to follow-up after the second dose of the vaccine.
Vaccine Safety
All but 1 participant completed the 3-injection series. One participant in the 2 mg group was lost to follow-up after the 2nd vaccination. No SAEs were reported. The most frequent solicited local AEs were injection site pain (any level), redness, swelling and itching that occurred in approximately half of the participants (Figure 2 and Table S3, Supplementary Appendix, available at NEJM.org). Systemic AEs were uncommon and included headache, myalgias, upper respiratory infections, fatigue/malaise and nausea. Except for injection site reactions, most AEs were considered as not related to vaccine administration. There were no laboratory abnormalities except 1 case each of hyperkalemia, anemia, neutrophilia and mild neutropenia in four different participants that were transient and considered not related to vaccine administration.
Figure 2.
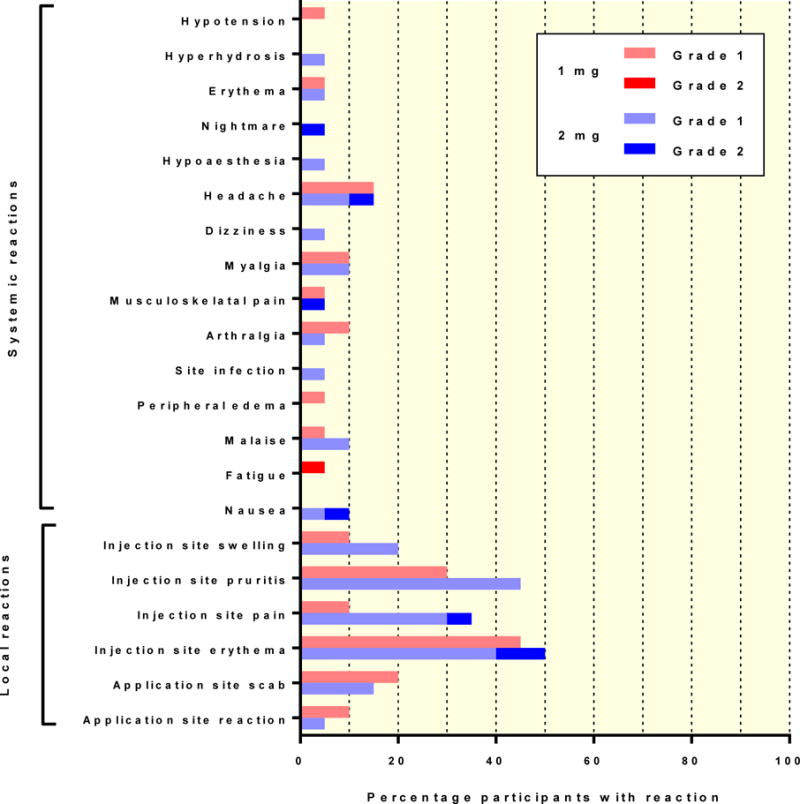
Local and systemic elicited treatment-emergent adverse events related to the vaccine administration. Table S3 (in the Supplementary Appendix, available at NEJM.org) list includes all treatment-emergent events without regard to relationship to study drug administration.
Antibody responses
At baseline, none of the participants had measurable antibody responses against ZIKV by ELISA. Four weeks after the first dose, 41% of study participants had detectable binding antibody responses: 25% and 60% in the 1 mg and 2 mg groups, respectively. At week 6, two weeks after the second dose, the antibody response was 74% overall; 65% and 84% in the two groups, respectively, increasing to 70% and 95% for each group on week 12, just prior to the third vaccination. Two weeks after the third dose, at week 14, all (100%) participants developed ZIKV-specific binding antibodies (Figure 3A).
Figure 3. Antibody response.


A. Geometric mean (95% CI) binding antibody titers at weeks 0, 1, 4, 6, 12 and 14 in the 1 mg (red) and the 2 mg (blue) dose groups as measured by ELISA. The antibody responses were statistically different at week 6 (p=0.039) (2 weeks after the second vaccine administration).
B. Geometric mean (95% CI) neutralizing antibody titers and proportion of participants (95% CI) with a neutralizing antibody response at each time point by standard 50% micro neutralization (MN50) assay methodology in Vero cells 18.
C. Proportion and 95% CI of week 14 individual sera (1:25 dilution) that would produce a 50 and 90% inhibition of cell fluorescence quantified by two independent analyses in glioblastoma cells (U87MG).
Geometric mean antibody titers (GMT) by ELISA trended higher for the 2 mg dose group at all time points, but only for the week 6 time point was there a statistically significant difference between groups (p=0.039) (Figure 3A and Table S4 in the Supplementary Appendix).
At the end of the vaccination period approximately 60% of the participants developed significant neutralizing antibody titers against ZIKV using the Vero cell assay. Neutralizing titers ranged between 1:18 to 1:317 and did not correlate with vaccine dose (Figure 3B). Additionally, there was not a significant correlation between the titers of binding and neutralizing antibodies. More than 95% of participants’ week 14 sera (diluted 1:25) neutralized infection of glioblastoma cells (U87MG) by 50% while more than 70% of participants’ week 14 sera neutralized infection of U87MG cells by 90%. (Figure 3C).
T cell responses
Pre-immunization PBMCs were compared to PBMCs obtained at weeks 4, 6 and 14 by ELISPOT to detect IFN-γ-secreting cells in response to stimulation with ZIKV-prME peptides. The number of IFN-γ producing cells obtained per million PBMCs against three peptide pools encompassing the entire prME protein is shown in Figure 4 and Table S5 in the Supplementary Appendix. The values represent median and IQR responses in each group. Participants vaccinated with the 2 mg dose had significantly higher median responses after the second vaccine dose (week 6; Mann-Whitney exact p=0.006); after the third dose the responses were similar in the two dosing groups.
Figure 4. Cellular response.

Pre-immunization PBMCs were compared to PBMCs obtained at weeks 0, 4, 6 and 14 by ELISpot to detect IFN-γ-secreting cells in response to stimulation with ZIKV-prME peptides. The number of IFN-γ producing cells obtained per million PBMCs against three peptide pools encompassing the entire prME protein is shown. The values represent median and IQR responses in each group.
In vivo protection against ZIKV infection
The protective efficacy of post-vaccination serum was evaluated in the IFNAR−/− mouse challenge model (Reference). One hour after intraperitoneal administration of 0.1 ml of either week 0 or week 14 serum or PBS (7 mice per participant), animals were challenged with 105 PFUs of ZIKV-PR209 isolate administered intraperitoneally. All animals treated with PBS or week 0 serum succumbed after 7-9 days, whereas 91.6% of animals pretreated with week 14 serum survived (Figure 5 A-B), suggesting that the antibody response generated by the vaccine is protective in this infection model. Protection was independent of neutralization titer as mice injected with serum from five individuals who, post-vaccination, had binding antibody responses but no neutralizing antibody responses demonstrated 91.4% survival post-challenge. There were no differences in survival observed between male and female mice.
Figure 5. Protection of IFNAR−/− mice following passive transfer of immune sera.

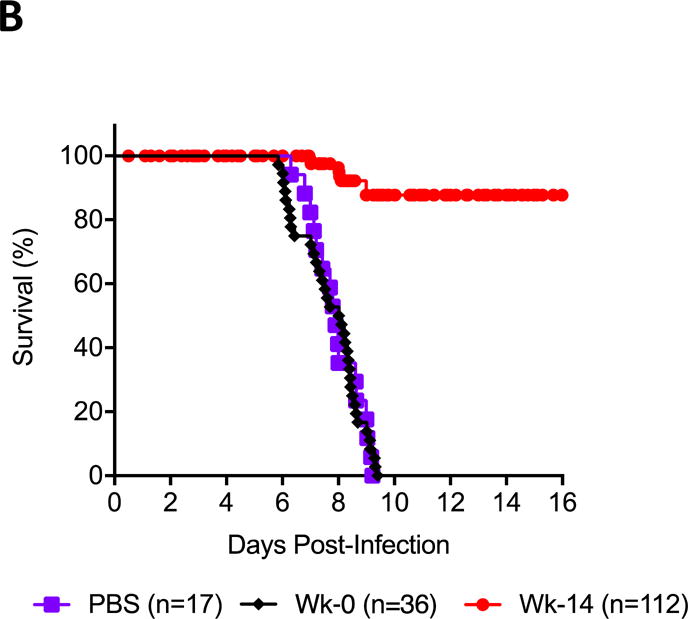
Survival of IFNAR−/− mice following passive transfer of participant’s immune sera. Week 14 sera from participants were administered intraperitoneally (0.1ml/mouse; 7 mice per participant) one hour before an intraperitoneal challenge with ZIKV (105 PFUs/mouse of ZIKV PR209 strain). (A) Proportions of mice surviving at day 14 post challenge given either sera collected pre-vaccination (Week 0) lanes 1-9; or with sera collected post-vaccination (Week 14), lanes 10-25. Participants P4, P8, P25, P37 and P39 had binding antibody titers but no neutralizing titers at week 14 (* bolded in the X axis) (B) Kaplan-Meier curves of survival of mice included in the challenge study and injected with PBS, pre-vaccination (Week 0) sera, or post-vaccination (Week 14) sera. There were no differences between male and female mice.
DISCUSSION
Currently there are no licensed vaccines against ZIKV. The data from this clinical trial supports the safety and the immunogenicity of the GLS-5700 ZIKV vaccine. GLS-5700 was able to induce high levels of binding antibodies in 100% of participants after a 3-dose vaccination regimen and 95% after 2 immunizations. Neutralizing antibodies were found in 60% of individuals in a Vero cell assay and in greater than 95% when assayed on neuronal cell targets. Immune serum obtained from vaccinated study participants both prevented ZIKV infection in in vitro cellular models and, importantly, prevented mortality in an in vivo mouse model.
GLS-5700 is one of a number of ZIKV vaccines that have shown promising results in animal models23–25. These include nucleic acid (DNA and mRNA) based vaccines, viral vectored vaccines, inactivated and live-attenuated vaccines. GLS-5700 is a wholly synthetic DNA vaccine designed to express a novel consensus ZIKV-prME antigen.
Synthetic DNA vaccines are ideally suited to address emerging infectious diseases through rapid design for novel antigens26. Vaccines can be rapidly designed using a common platform expressing relevant antigens from an emergent pathogen. Other platforms share this ability for rapid alteration with varying benefits and challenges26. The development time from initial design to initiation of this first-in-man clinical trial was 7 months for GLS-5700. Synthetic DNA vaccines administered by CELLECTRA® EP have been shown to elicit cellular and humoral immune responses in preclinical and clinical studies far greater than naked injection of DNA alone16,27 and can impact human infection and pathogen clearance28 29.This study further advances EP enhancement approaches by focusing on ID injection of a smaller delivery volume and lower energy output during EP. GLS-5700 was well tolerated with rates of local and systemic side effects similar to other DNA vaccines delivered by EP29.
Both antibodies and T cell responses are postulated as important in the protection from ZIKV infection in humans. Our study shows that GLS-5700 is capable of inducing binding antibodies in all participants after 3 doses, in each of the dose levels evaluated. GLS-5700 also induces ZIKV neutralizing antibodies in approximately two thirds of the participants by the PRNT50 Vero assay, and higher levels in a neuronal cell target assay, as well as inducing moderate T-cell responses against ZIKV. The magnitude of the T-cell responses should be interpreted with caution as there was a PBMC shipping problem identified which impacted cell viability.
Our study was not designed to address efficacy of this ZIKV vaccine in humans; that will require larger randomized trials in a ZIKV-endemic region. However, we showed that GLS-5700 induces immune responses that are protective in both in vitro and in vivo models of ZIKV infection, suggesting that vaccine-induced antibodies may be clinically relevant to prevent infection, the primary criteria for ZIKV vaccine development designated by the World Health Organization30. Protection of IFNAR−/− mice was >91% regardless of whether passively transferred post-vaccination sera had detectable neutralizing antibodies or not. This phenomenon has also been observed in other flavivirus infections, like West Nile Virus (WNV), for which passive transfer of poorly neutralizing antibodies against WNV protected naïve mice from challenge31. Our study confirms this observation in humans and highlights the limitations of current neutralizing assay utility as functional measurements for the development of ZIKV or other flavivirus vaccines.
Consensus DNA plasmids have been demonstrated in multiple pre-clinical studies to provide broad protection in animal models against a number of viruses including Ebola32, MERS33, and Zika15. We show here that the GLS-5700 vaccine generates a protective response against multiple ZIKV isolates, the African lineage MR766 ZIKV strain in a neuronal cell neutralization assay and the PR209 Caribbean sub-lineage of Asian ZIKV in a passive transfer IFNAR−/− mouse challenge model.
The development of significant levels of protective antibodies against ZIKV by GLS-5700 raises the potential of using hyper-immune serum from vaccinees as a preventive and/or therapeutic agent through passive administration. Whether hyper-immune serum would have utility against ZIKV infection during pregnancy, as it has for other infectious disease, like CMV34 or VZV35, or as a prophylaxis in high risk situations, such as following rabies exposures, is unknown and requires independent investigation.
In conclusion, our trial shows, for the first time, the safety and immunogenicity of a novel engineered DNA vaccine encoding a consensus ZIKV-prME antigen delivered simply by CELLECTRA® EP-enhanced intradermal inoculation that can potentially be used for the prevention of this devastating infection. Future studies will evaluate the efficacy of the vaccine.
Supplementary Material
Acknowledgments
We would like to thank the following people that make this study possible:
GeneOne acknowledges the contributions of Deborah Kane, Sally Sturm, the Clinical Research Monitors, and the Morristown Medical Center medical library and library staff. The significant effort of the following site personnel is also gratefully acknowledged: at the University of Pennsylvania, Maryann Najdzinowicz, BSN, RN, Deborah Kim RPh; Yan Jiang, RN; at QPS- Miami Research, Meredith Arguelles LPN, Maria Soto, RN, Manhui Pang MD and Terri Piedra; and at Université Laval, Isabelle Chabot, Nathalie Breton, RN, and Dany Poulin, RN. Most importantly, we thank the study participants for the time they donated and their dedication.
This work was sponsored by GeneOne Life Science, Inc. and supported in part by the following NIH grants: AI069534 “Clinical Trials Unit, University of Pennsylvania”. D.B.W. was supported in part by the Intramural Research Program, National Institute of Allergy and Infectious Diseases, National Institutes of Health (grant R01-AI092843), as well as through the WW. Smith Chair in Cancer Research.
References
- 1.Dick G, Kitchen S, Haddow A. Zika virus (I). Isolations and serological specificity. Transactions of the Royal Society of Tropical Medicine and Hygiene. 1952;46:509–20. doi: 10.1016/0035-9203(52)90042-4. [DOI] [PubMed] [Google Scholar]
- 2.Duffy MR, Chen TH, Hancock WT, et al. Zika virus outbreak on Yap Island, Federated States of Micronesia. The New England journal of medicine. 2009;360:2536–43. doi: 10.1056/NEJMoa0805715. [DOI] [PubMed] [Google Scholar]
- 3.Cao-Lormeau VM, Roche C, Teissier A, et al. Zika virus, French polynesia, South pacific, 2013. Emerg Infect Dis. 2014;20:1085–6. doi: 10.3201/eid2006.140138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Campos GS, Bandeira AC, Sardi SI. Zika Virus Outbreak, Bahia, Brazil. Emerg Infect Dis. 2015;21:1885–6. doi: 10.3201/eid2110.150847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Fauci AS, Morens DM. Zika virus in the Americas—yet another arbovirus threat. New England Journal of Medicine. 2016;374:601–4. doi: 10.1056/NEJMp1600297. [DOI] [PubMed] [Google Scholar]
- 6.Brasil P, Calvet GA, Siqueira AM, et al. Zika virus outbreak in Rio de Janeiro, Brazil: clinical characterization, epidemiological and virological aspects. PLoS Negl Trop Dis. 2016;10:e0004636. doi: 10.1371/journal.pntd.0004636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Parra B, Lizarazo J, Jimenez-Arango JA, et al. Guillain-Barre Syndrome Associated with Zika Virus Infection in Colombia. The New England journal of medicine. 2016;375:1513–23. doi: 10.1056/NEJMoa1605564. [DOI] [PubMed] [Google Scholar]
- 8.Mlakar J, Korva M, Tul N, et al. Zika Virus Associated with Microcephaly. The New England journal of medicine. 2016;374:951–8. doi: 10.1056/NEJMoa1600651. [DOI] [PubMed] [Google Scholar]
- 9.Paz-Bailey G, Rosenberg ES, Doyle K, et al. Persistence of Zika Virus in Body Fluids - Preliminary Report. The New England journal of medicine. 2017 doi: 10.1056/NEJMc1814416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Foy BD. Probable Non–Vector-borne Transmission of Zika Virus, Colorado, USA-Volume 17, Number 5—May 2011. Emerging Infectious Disease journal-CDC. 2011 doi: 10.3201/eid1705.101939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.D’Ortenzio E, Matheron S, Yazdanpanah Y, et al. Evidence of Sexual Transmission of Zika Virus. The New England journal of medicine. 2016;374:2195–8. doi: 10.1056/NEJMc1604449. [DOI] [PubMed] [Google Scholar]
- 12.Musso D, Nhan T, Robin E, et al. Potential for Zika virus transmission through blood transfusion demonstrated during an outbreak in French Polynesia, November 2013 to February 2014. Euro surveillance : bulletin Europeen sur les maladies transmissibles = European communicable disease bulletin. 2014;19:20761. doi: 10.2807/1560-7917.es2014.19.14.20761. [DOI] [PubMed] [Google Scholar]
- 13.Motta IJ, Spencer BR, Cordeiro da Silva SG, et al. Evidence for Transmission of Zika Virus by Platelet Transfusion. The New England journal of medicine. 2016;375:1101–3. doi: 10.1056/NEJMc1607262. [DOI] [PubMed] [Google Scholar]
- 14.Filipe A, Martins C, Rocha H. Laboratory infection with Zika virus after vaccination against yellow fever. Archiv für die gesamte Virusforschung. 1973;43:315–9. doi: 10.1007/BF01556147. [DOI] [PubMed] [Google Scholar]
- 15.Muthumani K, Griffin BD, Agarwal S, et al. In vivo protection against ZIKV infection and pathogenesis through passive antibody transfer and active immunisation with a prMEnv DNA vaccine. Npj Vaccines. 2016;1:16021. doi: 10.1038/npjvaccines.2016.21. https://www.nature.com/articles/npjvaccines201621 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kalams SA, Parker SD, Elizaga M, et al. Safety and comparative immunogenicity of an HIV-1 DNA vaccine in combination with plasmid interleukin 12 and impact of intramuscular electroporation for delivery. J Infect Dis. 2013;208:818–29. doi: 10.1093/infdis/jit236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Trimble CL, Morrow MP, Kraynyak KA, et al. Safety, efficacy, and immunogenicity of VGX-3100, a therapeutic synthetic DNA vaccine targeting human papillomavirus 16 and 18 E6 and E7 proteins for cervical intraepithelial neoplasia 2/3: a randomised, double-blind, placebo-controlled phase 2b trial. The Lancet. 2015;386:2078–88. doi: 10.1016/S0140-6736(15)00239-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Davis BS, Chang G-JJ, Cropp B, et al. West Nile virus recombinant DNA vaccine protects mouse and horse from virus challenge and expresses in vitro a noninfectious recombinant antigen that can be used in enzyme-linked immunosorbent assays. Journal of virology. 2001;75:4040–7. doi: 10.1128/JVI.75.9.4040-4047.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Anfasa F, Siegers JY, van der Kroeg M, et al. Phenotypic Differences between Asian and African Lineage Zika Viruses in Human Neural Progenitor Cells. mSphere. 2017;2 doi: 10.1128/mSphere.00292-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lazear HM, Govero J, Smith AM, et al. A mouse model of Zika virus pathogenesis. Cell host & microbe. 2016;19:720–30. doi: 10.1016/j.chom.2016.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhang NN, Tian M, Deng YQ, et al. Characterization of the contemporary Zika virus in immunocompetent mice. Hum Vaccin Immunother. 2016;12:3107–9. doi: 10.1080/21645515.2016.1219004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lazear HM, Govero J, Smith AM, et al. A Mouse Model of Zika Virus Pathogenesis. Cell Host Microbe. 2016;19:720–30. doi: 10.1016/j.chom.2016.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Abbink P, Larocca RA, Rafael A, et al. Protective efficacy of multiple vaccine platforms against Zika virus challenge in rhesus monkeys. Science (New York, NY) 2016;353:1129–32. doi: 10.1126/science.aah6157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Larocca RA, Abbink P, Peron JPS, et al. Vaccine protection against Zika virus from Brazil. Nature. 2016;536:474–8. doi: 10.1038/nature18952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pardi N, Hogan MJ, Pelc RS, et al. Zika virus protection by a single low-dose nucleoside-modified mRNA vaccination. Nature. 2017;543:248–51. doi: 10.1038/nature21428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Maslow JN. Vaccine development for emerging virulent infectious diseases. Vaccine. 2017 Feb 16; doi: 10.1016/j.vaccine.2017.02.015. pii: S0264-410X(17)30196-2. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hirao LA, Draghia-Akli R, Prigge JT, et al. Multivalent smallpox DNA vaccine delivered by intradermal electroporation drives protective immunity in nonhuman primates against lethal monkeypox challenge. J Infect Dis. 2011;203:95–102. doi: 10.1093/infdis/jiq017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bagarazzi ML, Yan J, Morrow MP, et al. Immunotherapy against HPV16/18 generates potent TH1 and cytotoxic cellular immune responses. Sci Transl Med. 2012;4:155ra38. doi: 10.1126/scitranslmed.3004414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Trimble CL, Morrow MP, Kraynyak KA, et al. Safety, efficacy, and immunogenicity of VGX-3100, a therapeutic synthetic DNA vaccine targeting human papillomavirus 16 and 18 E6 and E7 proteins for cervical intraepithelial neoplasia 2/3: a randomised, double-blind, placebo-controlled phase 2b trial. Lancet. 2015;386:2078–88. doi: 10.1016/S0140-6736(15)00239-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.WHO/UNICEF Zika Virus (ZIKV) Vaccine Target Product Profile (TPP): Vaccine to protect against congenital Zika syndrome for use during an emergency. 2017. at http://www.who.int/immunization/research/development/WHO_UNICEF_Zikavac_TPP_Feb2017.pdf.
- 31.Vogt MR, Dowd KA, Engle M, et al. Poorly neutralizing cross-reactive antibodies against the fusion loop of West Nile virus envelope protein protect in vivo via Fcγ receptor and complement-dependent effector mechanisms. Journal of virology. 2011;85:11567–80. doi: 10.1128/JVI.05859-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shedlock DJ, Aviles J, Talbott KT, et al. Induction of broad cytotoxic T cells by protective DNA vaccination against Marburg and Ebola. Mol Ther. 2013;21:1432–44. doi: 10.1038/mt.2013.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Muthumani K, Falzarano D, Reuschel EL, et al. A synthetic consensus anti-spike protein DNA vaccine induces protective immunity against Middle East respiratory syndrome coronavirus in nonhuman primates. Sci Transl Med. 2015;7:301ra132. doi: 10.1126/scitranslmed.aac7462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Nigro G, Adler SP, La Torre R, Best AM. Passive immunization during pregnancy for congenital cytomegalovirus infection. New England Journal of Medicine. 2005;353:1350–62. doi: 10.1056/NEJMoa043337. [DOI] [PubMed] [Google Scholar]
- 35.Enders G, Bolley I, Miller E, Cradock-Watson J, Ridehalgh M. Consequences of varicella and herpes zoster in pregnancy: prospective study of 1739 cases. The Lancet. 1994;343:1548–51. doi: 10.1016/s0140-6736(94)92943-2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


