Abstract
Background
In limited retrospective series, infliximab, adalimumab and vedolizumab have demonstrated efficacy in chronic antibiotic-refractory pouchitis. Here, we report single-centre data of all biological therapies in refractory pouchitis.
Methods
We retrospectively assessed all records from patients with ulcerative colitis and ileal pouch -anal anastomosis who received infliximab, adalimumab or vedolizumab for pouchitis. Clinically relevant remission, defined as a modified Pouchitis Disease Activity Index <5 and a reduction of modified Pouchitis Disease Activity Index ≥2 points from baseline, was assessed at week 14.
Results
Thirty-three unique patients were identified. Prior to colectomy, patients had been exposed to cyclosporine (n = 14), infliximab (n = 12), adalimumab (n = 3), and/or vedolizumab (n = 3). All developed chronic antibiotic-refractory pouchitis, for which they received infliximab (n = 23), adalimumab (n = 13) or vedolizumab (n = 15). Clinically relevant remission was observed in 43.5% of patients in the infliximab group, and in 38.5% and 60.0% in the adalimumab and vedolizumab group, respectively. In the long-term, significantly more patients continued vedolizumab compared to anti-tumour necrosis factor (anti-TNF) therapy (hazard ratio 3.0, p = 0.04). Adverse events (mainly infusion reactions) explained 40.7% of the patients discontinuing anti-TNF therapy, whereas discontinuation of vedolizumab was only related to insufficient efficacy. Four patients eventually required a permanent ileostomy.
Conclusion
In this case series of chronic antibiotic-refractory pouchitis, biological therapy was effective in the majority of patients and only a minority eventually required a permanent ileostomy. The use of anti-TNF agents was hampered by a high rate of adverse events, partly related to immunogenicity as some patients had been exposed to anti-TNF prior to colectomy. Vedolizumab was also efficacious and may provide a safe alternative in these chronic antibiotic-refractory pouchitis patients.
Keywords: Chronic pouchitis, ulcerative colitis, biological therapy, vedolizumab, infliximab, adalimumab
Key summary
What is known about this topic?
Chronic antibiotic refractory pouchitis is a debilitating condition with largely empirical treatment regimens due to the lack of randomised trials.
Biologicals have been used to treat chronic pouchitis, but the use of infliximab (IFX), adalimumab (ADA) and vedolizumab (VDZ) remains limited to very small case series focusing on a single drug.
What are the significant findings in this study?
This is the largest single-centre study in chronic antibiotic-refractory pouchitis, reporting on the efficacy and safety of IFX, ADA and VDZ.
The use of anti-TNF agents is hampered by a high rate of adverse events, partially related to immunogenicity as a consequence of pre-colectomy exposure to anti-TNF and lack of concomitant immunomodulatory therapy.
VDZ might be an efficacious and safe alternative in chronic pouchitis.
Introduction
Despite the advent of biological therapies and small molecules, 25–30% of patients with ulcerative colitis (UC) eventually require a colectomy.1 Restorative proctocolectomy with ileal pouch-anal anastomosis (IPAA) is the surgical treatment of choice for patients with UC, refractory to medical therapy or those with a (pre)malignant lesion.2
Surgery comprises a one-, two- or three-stage procedure and, traditionally, the two-stage procedure is most frequently performed. Another, probably safer, option is the modified two-stage procedure, which comprises a subtotal colectomy with end ileostomy at initial surgery, followed by completion proctectomy and construction of IPAA, with closure of ileostomy during secondary surgery some months later.3,4
Although IPAA improves health-related quality of life,5 patients are prone to develop pouch-related complications, including pouchitis as the most common complication in up to 50% of patients over the first 10 years after surgery.1,6,7 Due to the broad differential diagnosis, an endoscopy with biopsies is necessary to establish a diagnosis of pouchitis,8 with the 18-point Pouchitis Disease Activity Index (PDAI) as the most commonly used diagnostic tool.9 However, the diagnostic criteria for pouchitis can be simplified with similar sensitivity and specificity, using the modified pouchitis disease activity index (mPDAI).10 In contrast to the PDAI, the mPDAI does not include a histological analysis.
Acute pouchitis can be treated successfully with antibiotic therapy (two weeks of metronidazole and/or ciprofloxacin).2 However, some patients have frequent episodes of pouchitis (≥4 per year, relapsing pouchitis), requiring longer periods of non-stop antibiotic therapy, and are considered antibiotic-dependent. In the case of antibiotic-refractory pouchitis, patients have persistent symptoms despite a four-week antibiotic course.1
Of patients with acute pouchitis, 5–19% will eventually develop chronic antibiotic-refractory pouchitis.1 Due to the lack of randomised trials, therapy is largely empirical, including prolonged antibiotic therapy, mesalamine, corticosteroids, thiopurines and probiotics.11–13 Over the recent years, biologicals have been introduced in the treatment of chronic antibiotic-refractory pouchitis, similar to the mainstay UC treatment strategies and based on evidence that the production of TNF-alpha and cell adhesion molecules are increased in patients with pouchitis.14 In the literature, the use of infliximab (IFX) in chronic antibiotic-refractory pouchitis has demonstrated efficacy in small case series,15–20 whereas data on adalimumab (ADM) and vedolizumab (VDZ) are scarce.21–28 Due to the limited data currently available, we aimed to evaluate the real-life efficacy of IFX, ADM and VDZ in patients with chronic antibiotic-refractory pouchitis in a single tertiary referral centre.
Methods
Study design and patients
We conducted this retrospective, open-label cohort study at the University Hospitals Leuven (Leuven, Belgium). All patients included in the analysis had given written consent to participate in the Institutional Review Board approved inflammatory bowel disease (IBD) Biobank (B322201213950/S53684). The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institution's human research committee.
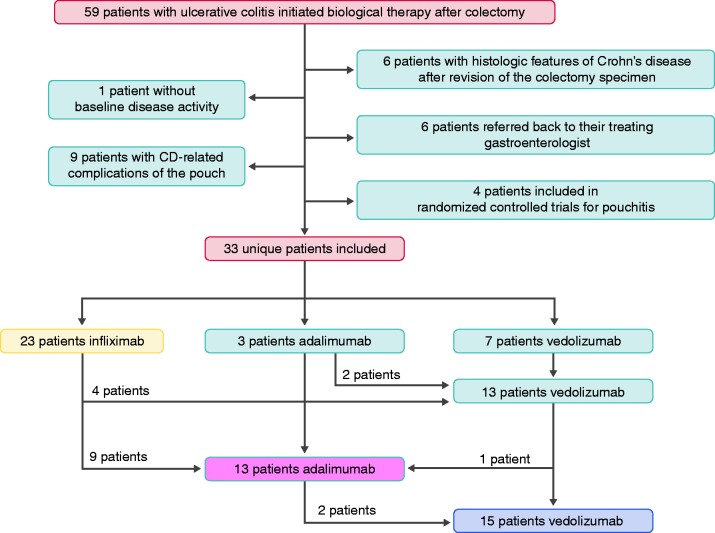
We screened the medical files of all UC patients who initiated biological therapy in our centre after colectomy with IPAA. Colectomy specimens were carefully reviewed, and all patients with histological features of Crohn’s disease (CD) were excluded (Figure 1). Similarly, patients with CD-related complications of the pouch (fistula, strictures, pre-pouch ileitis)20 without any pouchitis were excluded. All included patients had to be refractory to antibiotic therapy, with persistent pouchitis symptoms despite a minimal four-week course of antibiotics. Patient who did not complete induction therapy at our centre but were referred back to their treating physician were excluded, as well as patients included in placebo-controlled randomised trials for pouchitis. Finally, patients without baseline disease activity (mPDAI score of <5) were excluded. Baseline disease characteristics, as well as postoperative and pouch-related complications were retrieved from the medical files of all included patients.
Figure 1.
Flowchart. CD: Crohn's disease.
Depending on the discretion of the treating physician, patients with chronic antibiotic-refractory pouchitis were treated with either IFX (5 mg/kg administered intravenously (IV) at weeks 0, 2 and 6, and thereafter maintenance therapy was continued at 5 mg/kg IFX every eight weeks), ADM (160 mg/80 mg subcutaneously (SC) at baseline and week 2 respectively, and thereafter maintenance therapy with ADM 40 mg SC every other week was continued) or VDZ (300 mg IV at weeks 0, 2 and 6 and thereafter maintenance therapy with 300 mg VDZ IV was continued every eight weeks). Concomitant therapy, including antibiotics, was recorded.
Outcomes
The primary endpoint, short-term clinically relevant remission was assessed at week 14, and defined as mPDAI <5 and reduction in the mPDAI score with ≥2 points from baseline, similar to the primary endpoint of an ongoing randomised controlled trial in chronic pouchitis (NCT02790138).29 Primary non-response (PNR) was defined as the absence of any clinical improvement by week 14, whereas secondary loss-of-response (LOR) was considered in those patients who lost response after initial clinical and/or endoscopic improvement. Long-term outcome was assessed by the continuation of the drug at final follow-up. In case pouch biopsies were available before and after therapy, polymorphic nuclear leukocyte infiltration and mean ulcerations per low-power field were assessed by an IBD-experienced histopathologist (GDH), according to the initial PDAI scoring system.9
Statistical analysis
All analyses were performed using IBM SPSS Statistics 25 (IBM SPSS, Costa Mesa, California, USA) and GraphPad Prism version 6.0 (GraphPad Software, La Jolla, California, USA). A non-responder imputation was applied, as well as a last-observation carried forward approach in patients discontinuing therapy and thus resulting in missing data at later time points. Continuous variables are expressed as medians and interquartile ranges (IQRs). Unpaired data were compared using the Mann-Whitney U-test for continuous variables, and Fisher’s exact test for categorical variables. Continuous paired data were compared using the Wilcoxon signed-rank test. Time to event data were analysed using the Kaplan-Meier method, with comparisons between groups made using the Wilcoxon (Breslow) test. A Cox proportional hazards model was used to study the influence of therapy on biological therapy discontinuation-free survival. A two-tailed p-value <0.05 was considered significant.
Results
Patient characteristics
Thirty-three unique patients with chronic antibiotic-refractory pouchitis were included (Table 1). The majority of the patients (90.9%) required colectomy due to refractory colitis, with 51.5% of all procedures performed electively. All but one patients were cases of J-pouches (97.0%), and most were constructed in a two-stage (51.5%) or modified two-stage procedure (24.2%). Patients developed a first episode of acute pouchitis a median of 0.8 (IQR 0.4–2.9) years after restoration of the faecal stream, which evolved to chronic antibiotic-refractory pouchitis a median of 3.1 (IQR 1.2–5.1) years after restoration of the faecal stream.
Table 1.
Surgical characteristics of the 33 included patients.
| Characteristic | n = 33 |
|---|---|
| Women, n (%) | 10 (30.3) |
| Primary sclerosing cholangitis, n (%) | 3 (9.1) |
| Smoking at primary surgery, n (%) | |
| - Never | 19 (57.6) |
| - Current | 2 (6.1) |
| - Ex-smoker | 12 (36.3) |
| Age at primary surgery, years, median (IQR) | 34.4 (28.3–39.6) |
| Duration between initial diagnosis and primary surgery, years, median (IQR) | 4.8 (2.0–9.2) |
| Previous therapies prior to colectomy, n (%) | |
| - Infliximab | 12 (36.4) |
| - Adalimumab | 3 (9.1) |
| - Vedolizumab | 3 (9.1) |
| - Cyclosporine | 14 (42.4) |
| Backwash ileitis, n (%) | |
| Indication for colectomy, n (%) | 4 (12.1) |
| - Refractory colitis | 30 (90.9) |
| - Dysplasia | 3 (9.1) |
| Timing colectomy, n (%) | |
| - Urgent (because of acute severe colitis) | 16 (48.5) |
| - Elective | 17 (51.5) |
| Laparoscopic assisted colectomy, n (%) | 19 (57.8) |
| Laparoscopic assisted pouch construction, n (%) | 17 (51.5) |
| Stapled anastomosis, n (%) | 29 (87.9) |
| J pouch, n (%) | 32 (97.0) |
| Number of stages for pouch surgery, n (%) | |
| - One stage | 3 (9.1) |
| - Two stage | 17 (51.5) |
| - Modified two stage | 8 (24.2) |
| - Three stage | 5 (15.2) |
| Duration between restauration faecal stream and first pouchitis episode, years, median (IQR) | 0.8 (0.4–2.9) |
| Duration between restauration faecal stream and chronic pouchitis, years, median (IQR) | 3.1 (1.2–5.1) |
| Age at onset of chronic pouchitis, years, median (IQR) | 37.6 (31.2–44.0) |
| Pouch failure, n (%) | 7 (21.2) |
| Permanent ileostomy, n (%) | 4 (12.1) |
IQR: interquartile range; UC: ulcerative colitis.
Pouch failure defined as need for reconstruction of a second pouch or construction of permanent ileostomy.
Due to the chronic antibiotic-refractory pouchitis, patients initiated IFX (n = 23), ADM (n = 13) or VDZ (n = 15) therapy (Table 2). Prior to colectomy, 36.4%, 9.1% and 9.1% had been exposed to IFX, ADM and VDZ, respectively. Furthermore, 42.4% had failed cyclosporine therapy. One-fifth (21.7%) of patients treated with IFX for chronic pouchitis, had been exposed to IFX prior to colectomy. Similarly, 61.5% ADM-treated chronic pouchitis patients previously failed (or were intolerant to) IFX prior to colectomy, and additionally 76.9% of them were also treated with IFX for their chronic pouchitis, prior to ADM therapy. Finally, 46.7% of VDZ patients failed (or were intolerant to) anti-TNF therapy prior to colectomy and 60.0% of them were also exposed to anti-TNF for their chronic pouchitis, prior to VDZ therapy. Three patients in the VDZ cohort had failed VDZ prior to colectomy.
Table 2.
Baseline disease characteristics all different biological cohorts.
| Characteristic | Infliximab (n = 23) | Adalimumab (n = 13) | Vedolizumab (n = 15) |
|---|---|---|---|
| Women, n (%) | 6 (26.1) | 5 (38.5) | 5 (33.3) |
| Age at treatment initiation, years, median (IQR) | 41.0 (37.2–47.1) | 39.3 (33.7–43.2) | 49.8 (30.1–58.9) |
| Time since colectomy, years, median (IQR) | 5.4 (4.2–8.1) | 6.0 (4.1–8.1) | 6.2 (2.0–12.8) |
| Time since restoration faecal stream, years, median (IQR) | 5.2 (3.7–7.9) | 5.2 (3.8–10.6) | 6.0 (1.8–12.6) |
| Previous therapies prior to colectomy, n (%) | |||
| - Infliximab | 5 (21.7) | 8 (61.5) | 7 (46.7) |
| - Adalimumab | 0 (0.0) | 0 (0.0) | 3 (20.0) |
| - Vedolizumab | 0 (0.0) | 0 (0.0) | 3 (20.0) |
| - Cyclosporine | 12 (54.5) | 8 (61.5) | 4 (26.7) |
| Previous therapies for pouchitis, n (%) | |||
| - Antibiotics | 23 (100) | 13 (100) | 15 (100.0) |
| - Pre- and probiotics | 5 (21.7) | 5 (38.5) | 5 (33.3) |
| - 5ASA (topical/systemic) | 3 (13.0) | 3 (23.1) | 3 (20.0) |
| - Steroids (topical/systemic) | 18 (78.3) | 11 (84.6) | 11 (73.3) |
| - Immunomodulators | 5 (21.7) | 3 (23.1) | 2 (13.3) |
| - Infliximab | 0 (0.0) | 10 (76.9) | 6 (40.0) |
| - Adalimumab | 0 (0.0) | 0 (0.0) | 4 (26.7) |
| - Vedolizumab | 0 (0.0) | 1 (7.7) | 0 (0.0) |
| Previous therapies prior to index therapy, n (%) | |||
| Infliximab | 5 (21.7) | 13 (100.0) | 12 (80.0) |
| Adalimumab | 0 (0.0) | 0 (0.0) | 7 (46.7) |
| Vedolizumab | 0 (0.0) | 1 (7.7) | 3 (20.0) |
| Pre-pouch ileitis, n (%) | 9 (39.1) | 4 (30.8) | 7 (46.7) |
| Cuffitis, n (%) | 8 (34.8) | 5 (38.5) | 9 (60.0) |
| C-reactive protein, mg/l, median (IQR) | 9.1 (2.1–12.7) | 8.3 (3.1–13.3) | 3.9 (2.8–8.3) |
| Haemoglobin, g/dl, median (IQR) | 13.4 (12.3–14.2) | 13.8 (12.4–14.2) | 13.7 (12.8–14.5) |
| Serum albumin, g/l, median (IQR) | 40.0 (38.3–42.6) | 43.3 (41.9–44.7) | 41.7 (38.1–45.7) |
| Modified PDAI, median (IQR) | 8.0 (6.0–9.0) | 7.0 (6.0–9.0) | 8.0 (7.0–8.5) |
| Concomitant therapy during induction, n (%) | |||
| - Topical steroids | 4 (17.4) | 1 (7.7) | 3 (20.0) |
| - Systemic steroids | 1 (4.3) | 1 (7.7) | 1 (6.7) |
| - Immunomodulators | 5 (21.7) | 0 (0.0) | 0 (0.0) |
| - Antibiotics | 7 (30.4) | 2 (15.4) | 6 (40.0) |
IQR: interquartile range; PDAI: pouch disease activity index; 5ASA: 5-aminosalicylates.
Outcome of infliximab therapy
IFX induced a significant drop in mPDAI (8.0 (6.0–9.0) at baseline down to 6.0 (4.5–8.0) at week 14, p = 0.005) (Figure 2(a)), resulting in an overall clinically relevant remission rate of 43.5% (Figure 3). However, nine patients (39.1%) discontinued IFX therapy during induction, mainly due to acute infusion reactions (n = 5) or delayed hypersensitivity (n = 1). Of note, five out of those six patients had been exposed to IFX prior to colectomy and developed an allergic reaction despite prophylactic therapy.
Figure 2.
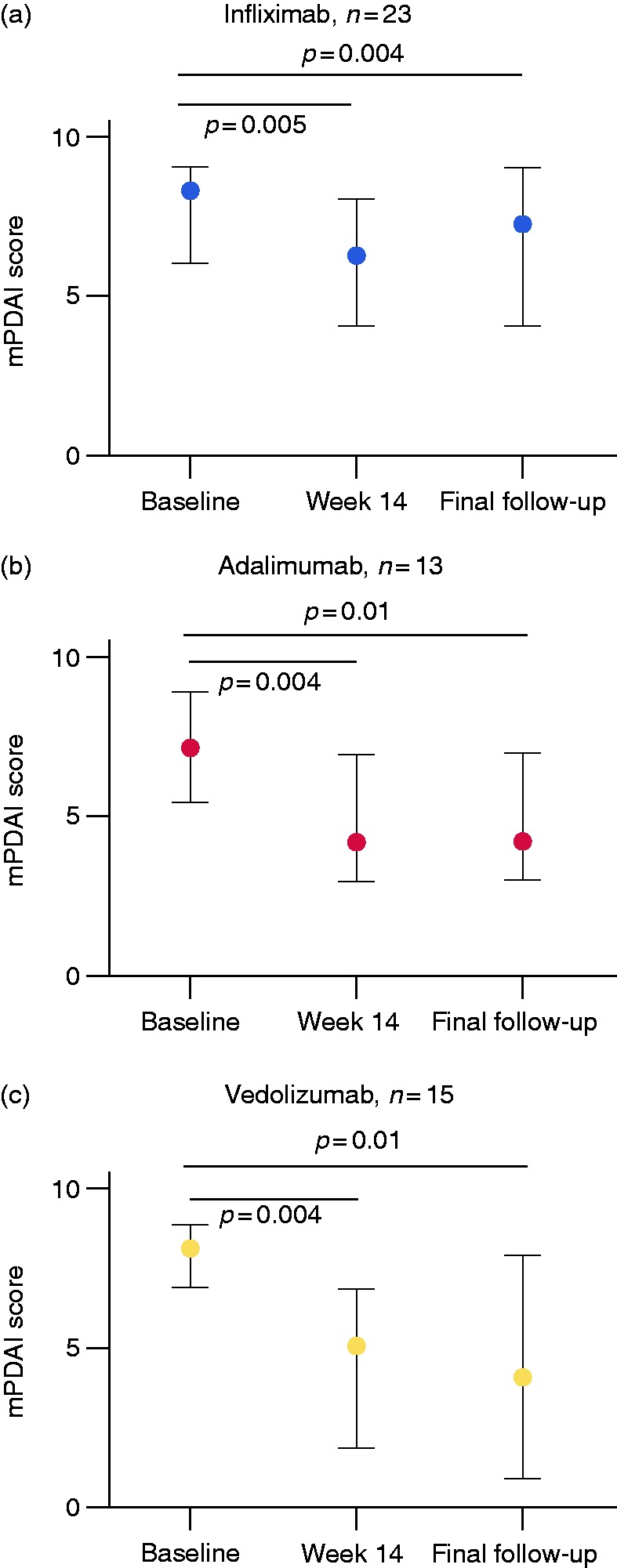
Evolution of the median (interquartile range) modified pouch disease activity index (mPDAI) during infliximab (a), adalimumab (b) and vedolizumab (c) therapy.
Figure 3.
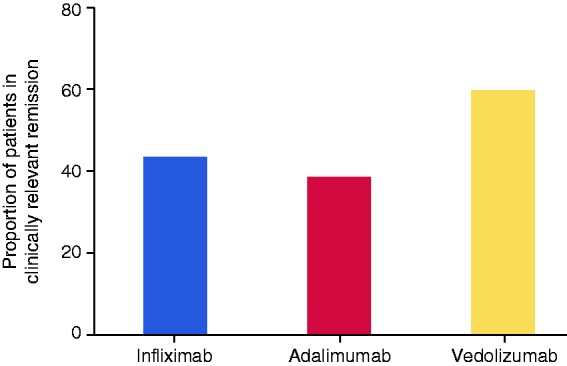
Clinically relevant remission rates by week 14 in infliximab (n = 23), adalimumab (n = 13) and vedolizumab (n = 15) treated patients with chronic antibiotic-refractory disease.
Although IFX induced and maintained response in some patients, even at the histological level (Figure 4(a) and (b)), the overall mPDAI score at final follow-up only moderately differed from baseline (8.0 (6.0–9.0) at baseline vs 7.0 (4.5–8.5) at final follow-up after 30.7 (8.4–82.5) weeks, p = 0.04). This limited sustained response, together with a further increase of patients suffering from infusion reactions, resulted in a 73.9% IFX discontinuation rate (Table 3), with only four patients (17.4%) in clinically relevant remission at final follow-up. In six patients, reactive drug monitoring and subsequent treatment intensification was applied, which turned out to be successful in three (50.0%) of them.
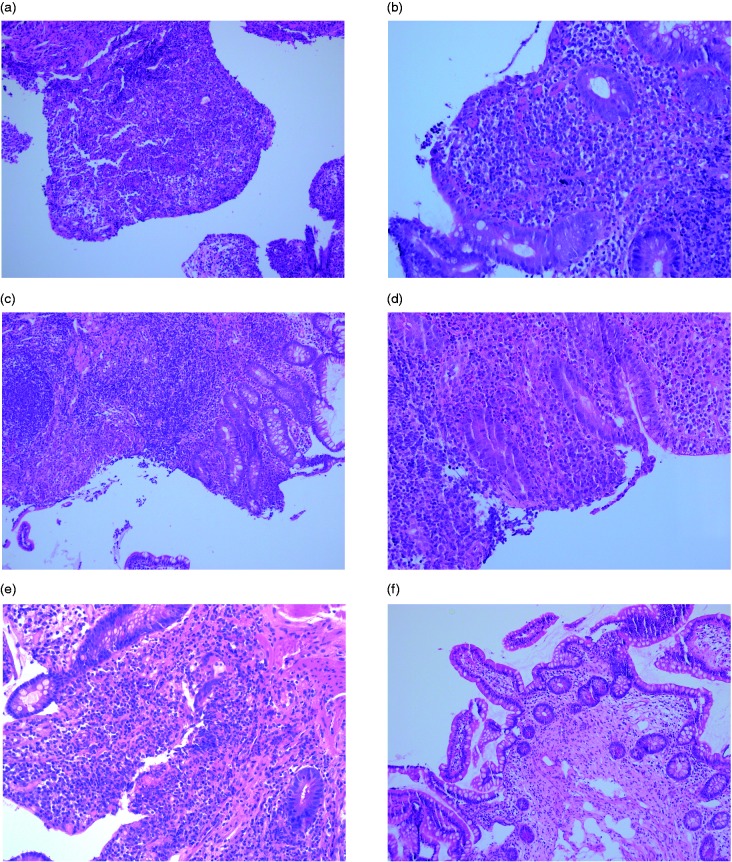
Figure 4.
Histological changes in pouch biopsies of ulcerative colitis (UC) patients with chronic antibiotic-refractory pouchitis before and after biological therapy (H&E stain; original magnification (OM) ×100–200). (a) H&E stainings from a pouchitis patient before infliximab therapy: active pouchitis (polymorphonuclear infiltration 3, ulceration per low power field 3). (b) H&E stainings from a pouchitis patient after infliximab therapy: week 14, epithelial restoration (decrease of the polymorphonuclear infiltration 0, ulceration per low power field 0). (c) H&E stainings from a pouchitis patient before adalimumab therapy: active pouchitis (polymorphonuclear infiltration 2, ulceration per low power field 2). (d) H&E stainings from a pouchitis patient after adalimumab therapy: week 13, partial epithelial restoration (decrease of the polymorphonuclear infiltration 1, ulceration per low power field 1). (e) H&E stainings from a pouchitis patient before vedolizumab therapy: active pouchitis (polymorphonuclear infiltration 2, ulceration per low power field 1). (f) H&E stainings from a pouchitis patient after vedolizumab therapy: week 14, complete epithelial restoration (decrease of the polymorphonuclear infiltration 0, ulceration per low power field 0). H&E: heamtoxylin and eosin.
Table 3.
Long-term outcome after biological therapy in chronic antibiotic-refractory pouchitis.
| Outcome | Infliximab (n = 23) | Adalimumab (n = 13) | Vedolizumab (n = 15) |
|---|---|---|---|
| Treatment discontinuation, n (%) | 17 (73.9) | 10 (77.0) | 4 (26.7) |
| Time to treatment discontinuation, weeks, median (IQR) | 9.0 (4.0–28.0) | 29.0 (22.3–102.8) | 47.6 (12.0–86.1) |
| Reason for treatment discontinuation, n (%) | |||
| - Primary non-response | 3 (17.6) | 4 (36.4) | 2 (13.3) |
| - Secondary loss-of-response | 5 (29.4) | 2 (18.2) | 2 (13.3) |
| - Infusion/injection site reaction | 8 (47.1) | 0 (0.0) | 0 (0.0) |
| - Delayed hypersensitivity | 1 (5.9) | N.A. | 0 (0.0) |
| - Adverse event | 0 (0.0) | 2 (18.2) | 0 (0.0) |
| - Undetectable serum levels | 0 (0.0) | 1 (9.1) | 0 (0.0) |
| - Oncologic comorbidity | 0 (0.0) | 1 (9.1) | 0 (0.0) |
| Permanent ileostomy, n (%) | 4 (17.0) | 2 (15.4) | 0 (0.0) |
IQR: interquartile range.
Outcome of adalimumab therapy
ADM also induced a significant drop in mPDAI (7.0 (6.0–7.0) at baseline down to 4.0 (3.0–4.0) at week 14, p = 0.004) (Figure 2(b)), resulting in an overall clinically relevant remission rate of 38.5% (Figure 3). In contrast to IFX, no allergic reactions were observed during induction. However, none of the ADM-treated patients had previously been exposed to ADM. Despite initial therapeutic success in some patients, including histological improvement (Figure 4(c) and (d)), 77.0% of ADM-treated patients discontinued therapy after a median of 29.0 (22.3–102.8) weeks. Importantly, only two patients lost their initial response to ADM therapy, while all other patients discontinued therapy due to PNR (n = 4), adverse events (n = 2), comorbidities (n = 1) or pharmacokinetic reasons (n = 1) (Table 3). All ADM-treated patients received standard dosage, according to the label.
Outcome of vedolizumab therapy
VDZ induced a significant drop in mPDAI (8.0 (7.0–8.5) at baseline down to 5.0 (2.0–7.0) at week 14, p = 0.004) (Figure 2(c)), resulting in an overall clinically relevant remission rate of 60.0% (Figure 3). At the histological level, VDZ could induce a decrease in the polymorphonuclear infiltration with associated resolution of the ulcerated mucosa (Figure 4(e) and (f)). Eight patients (53.3%) had been exposed to anti-TNF therapy for the treatment of their chronic antibiotic-refractory pouchitis before. In contrast to both anti-TNF agents, only four patients (26.7%) discontinued therapy (two PNR, two LOR of whom just one previously had been exposed to VDZ), resulting in a clinically relevant remission rate of 53.3% after 63.0 (19.6–117.4) weeks of follow-up with a mPDAI score of 4.0 (1.5–7.0) (Table 3). All VDZ-treated patients received standard dosage, according to the label.
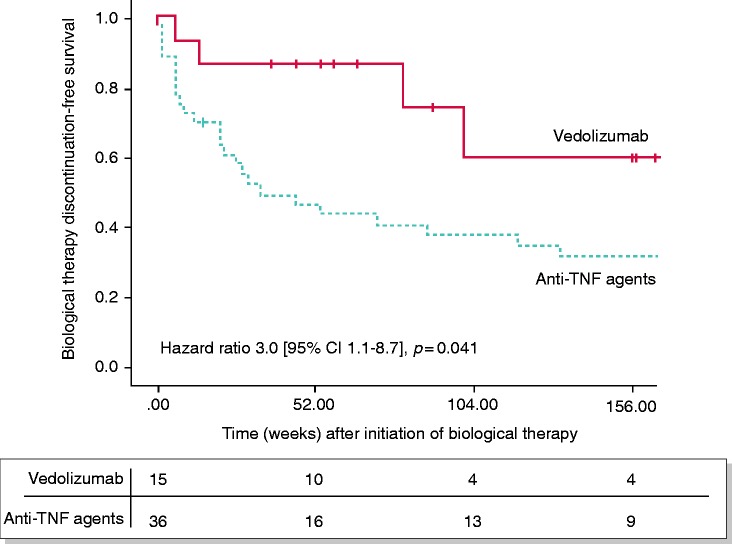
By week 14, both VDZ and anti-TNF induced a decrease in mPDAI (p = 0.004, p <0.0001 respectively), including a significant decrease in the endoscopic component of the mPDAI (p = 0.004, p = 0.0008 respectively) (Supplementary Material Table 1). Nevertheless, by the time of first assessment (week 14) numerically more patients had discontinued anti-TNF therapy compared to VDZ therapy (36.1% vs 13.3%, p = 0.10). In the long-term, significantly more patients needed to discontinue anti-TNF therapy as compared to VDZ (hazard ratio (HR) 3.0 95% confidence interval (CI) 1.1–8.7), p = 0.04) (Figure 5, Supplementary Material Figure 1). Overall, four patients (12.1%) required a permanent ileostomy: all failed or were intolerant to IFX, with two patients subsequently failing ADM. None of the VDZ treated patients required ileostomy.
Figure 5.
Biological therapy discontinuation-free survival in patients with chronic antibiotic-refractory pouchitis, grouped by either anti-TNF or vedolizumab therapy. CI: confidence interval.
Discussion
To our knowledge, this is the largest, single-centre retrospective series in patients with chronic antibiotic-refractory pouchitis, treated with IFX, ADM or VDZ. Chronic antibiotic-refractory pouchitis is a debilitating condition, largely empirically treated due to the lack of randomised, double-blind placebo-controlled trials.2,28,30 Efficacy data in the biological era are derived from small (n <10) or multi-centre case series, without any direct comparison between drugs. Although patients were not randomised in the current study, the single-centre methodology provides a unique opportunity to compare outcomes of different drugs, as patients were all treated and monitored in a similar manner. Nevertheless, data should be interpreted with caution given the lack of randomization or propensity matching.
Similar to previous series,15–17,21,22,26,27 we could confirm that IFX, ADM and VDZ can all induce a clinically relevant remission, including a significant drop in the endoscopic PDAI sub score. Although clinical remission was assessed differently, the 60.0% VDZ-induced remission rate in our cohort confirms two recent multi-centre studies which also reported a VDZ response rate above 50% in chronic pouchitis.26,27 A meta-analysis of all available observational studies reported that anti-TNF medications are associated with clinical remission in 50% of chronic pouchitis patients,30 which approximates roughly to the anti-TNF clinical remission rate of 41.6% that we observed. As anti-TNF agents have a clear trend to have higher and faster efficacy in CD-like complications of the pouch compared to refractory pouchitis,20 this could potentially also contribute to lower anti-TNF remission rates in the current cohort which mainly focuses on refractory pouchitis (despite some patients having pre-pouch ileitis).
Although no significant differences in the initial remission rates could be observed between the different classes of drugs, more patients discontinued anti-TNF therapy during induction compared to VDZ. In the long-term this phenomenon further persisted, reflected by a HR of 3.0 (95% CI 1.1–8.7, p=0.04) in favour of VDZ therapy. Apart from the primary and secondary non-responders, inherent to any biological agent, anti-TNF therapy was mainly discontinued due to adverse events, including several allergic reactions, especially in patients who received anti-TNF therapy prior to colectomy. However, only a minority of IFX-treated patients initiated concomitant immunomodulators, which also may have contributed to the higher rate of allergic reactions. However, as the majority of UC patients who come to colectomy for refractory colitis nowadays have already been treated with anti-TNF therapy, the treatment of chronic pouchitis with anti-TNF agents can always be hampered due to immunogenicity after a drug holiday. This may ultimately result in infusion reactions, PNR or LOR, as highlighted in the current study. Additionally, a recent British study focusing on the long-term outcome of IFX for inflammatory conditions in the ileoanal pouch also reported an overall IFX failure rate of just over half of all patients, although 97.1% of their cohort had never been exposed to IFX before.19 This suggests that, regardless of previous anti-TNF exposure, IFX (and to a larger extent anti-TNF agents as a whole based on our data) cannot maintain a durable response for chronic antibiotic-refractory pouchitis. In contrast, the low immunogenicity rates linked to VDZ,31 together with our findings on a durable response in chronic antibiotic-refractory pouchitis, even in the case of previous VDZ exposure, may prioritise VDZ in chronic pouchitis. The results of the ongoing phase IV Earnest trial (NCT02790138), studying VDZ in chronic pouchitis in a double-blind placebo-controlled manner, are therefore eagerly awaited.29 Additionally, the efficacy of faecal microbiota transplantation, tofacitinib and ustekinumab in chronic pouchitis still has to be explored,32–34 and may further change treatment algorithms.
Being a retrospective analysis, this study has its own inherent limitations, including a relatively small sample size and the lack of histological data in all patients. However, the single-centre design and the data on different biological agents makes it different from other published small series. Nevertheless, the lack of randomization in the current design did not allow for matched groups and the limited sample size did not support propensity matching either. Hence, data should be interpreted with caution, especially as, for instance, baseline inflammatory markers were higher in anti-TNF treated patients as compared to VDZ-treated patients, despite similar mPDAI scores. Additionally, previous anti-TNF failure in two-thirds of the VDZ-treated cohort makes VDZ the last resort in those patients, and hence may decrease the likelihood of treatment discontinuation and thus bias our findings. However, the absence of any patient requiring a permanent ileostomy in the VDZ treatment arm, is a strong argument against the latter potential bias. Furthermore, more than 50% of VDZ-treated patients were in clinical remission at final follow-up, suggesting that the drug was not continued purely because of being the final resort prior to a diverting ostomy.
In conclusion, IFX, ADM and VDZ therapy can induce clinically relevant remission in patients with chronic antibiotic-refractory pouchitis. However, the use of anti-TNF agents is hampered by adverse events (mainly linked to previous anti-TNF exposure) and by limited efficacy on the long-term. Therefore, VDZ seems an efficacious and safe alternative in the treatment of chronic pouchitis.
Supplemental Material
Supplemental Material for Outcome of biological therapies in chronic antibiotic-refractory pouchitis: A retrospective single-centre experience by Bram Verstockt, Charlotte Claeys, Gert De Hertogh, Gert Van Assche, Albert Wolthuis, André D’Hoore, Séverine Vermeire and Marc Ferrante in United European Gastroenterology Journal
Acknowledgements
The authors would like to thank Vera Ballet and Eline Vandeput for an excellent job in maintaining the Leuven IBD patient database; Sophie Organe, Nooshin A Davani, Helene Blevi, Tamara Coopmans and Willem-Jan Wollants for an excellent job in maintaining the Biobank database.
The following author contributions were made: BV: study design, data acquisition and interpretation, statistical analysis and drafting of the manuscript. CC: data acquisition and critical revision of the manuscript. GDH: assessing histological slices and critical revision of the manuscript. GVA: patient recruitment and critical revision of the manuscript. AW: surgery and critical revision of the manuscript. ADH: surgery and critical revision of the manuscript. SV and MF: study design, data interpretation, supervision and critical revision of the manuscript. All authors agreed with the final version of the manuscript prior to submission. Guarantor of the manuscript: Marc Ferrante.
Declaration of conflicting interests
B Verstockt is a doctoral fellow, and S Vermeire and M Ferrante are senior clinical investigators of the Research Foundation Flanders (FWO), Belgium. B Verstockt has also received research grants from the Belgium Week of Gastroenterology, the Belgian IBD Research and Development Group (BIRD) and the IBD Patient’s Association Flanders (CCV VZW); and received support from the Vlaamse Vereniging voor Gastro-enterologie (VVGE). B Verstockt reports financial support for research from Pfizer; lecture fees from Abbvie, Ferring, Takeda Pharmaceuticals, Janssen and R Biopharm; consultancy fees from Janssen and Sandoz. G De Hertogh received fees for his activities as central pathology reader in clinical trials from Centocor, Takeda and Genentech. G Van Assche reports financial support for research from Abbott and Ferring Pharmaceuticals; lecture fees from Janssen, MSD and Abbott; consultancy fees from PDL BioPharma, UCB Pharma, Sanofi-Aventis, Abbott, Abbvie, Ferring, Novartis, Biogen Idec, Janssen Biologics, NovoNordisk, Zealand Pharma A/S, Millenium/Takeda, Shire, Novartis and Bristol Mayer Squibb. A D'Hoore reports lecture fees from Takeda Pharmaceuticals and research grants from J&J. S Vermeire reports financial support for research: MSD, AbbVie, Takeda, Pfizer, J&J; Lecture fees: MSD, AbbVie, Takeda, Ferring, Centocor, Hospira, Pfizer, J&J, Genentech/Roche; Consultancy: MSD, AbbVie, Takeda, Ferring, Centocor, Hospira, Pfizer, J&J, Genentech/Roche, Celgene, Mundipharma, Celltrion, SecondGenome, Prometheus, Shire, Prodigest, Gilead, Galapagos. M Ferrante reports financial support for research: Janssen, Pfizer, Takeda, Consultancy: Abbvie, Boehringer-Ingelheim, Celltrion, Ferring, Janssen, Lilly, Mitsubishi Tanabe, MSD, Pfizer, Takeda; Speakers fee: Abbvie, Amgen, Biogen, Boehringer-Ingelheim, Chiesi, Falk, Ferring, Janssen, Lamepro, Mitsubishi Tanabe, MSD, Pfizer, Takeda, Tramedico, Tillotts, Zeria. C Claeys and A Wolthuis declare no conflicts of interest.
Ethics approval
All patients included in the analysis had given written consent to participate in the Institutional Review Board approved IBD Biobank (B322201213950/S53684), collecting serum and clinical characteristics among other items. The study protocol conforms to the ethical guidelines of the 1975 Declaration of Helsinki as reflected in a prior approval by the institution s human research committee.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Informed consent
All patients included in the analysis had given written consent.
References
- 1.Shen B. Acute and chronic pouchitis–pathogenesis, diagnosis and treatment. Nat Rev Gastroenterol Hepatol 2012; 9: 323–333. [DOI] [PubMed] [Google Scholar]
- 2.Magro F, Gionchetti P, Eliakim R, et al. Third European Evidence-Based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 1: Definitions, diagnosis, extra-intestinal manifestations, pregnancy, cancer surveillance, surgery, and ileo-anal pouch disorders. J Crohns Colitis 2017. Jun 1; 1(6): 649–670. doi: 10.1093/ecco-jcc/jjx008. [DOI] [PubMed] [Google Scholar]
- 3.Zittan E, Wong-Chong N, Ma GW, et al. Modified two-stage ileal pouch-anal anastomosis results in lower rate of anastomotic leak compared with traditional two-stage surgery for ulcerative colitis. J Crohns Colitis 2016; 10: 766–772. [DOI] [PubMed] [Google Scholar]
- 4.Germain A, de Buck van Overstraeten A, Wolthuis A, et al. Outcome of restorative proctocolectomy with an ileo-anal pouch for ulcerative colitis: Effect of changes in clinical practice. Colorectal Dis 2018; 20: O30–O38. [DOI] [PubMed] [Google Scholar]
- 5.de Buck van Overstraeten A, Wolthuis AM, Vermeire S, et al. Long-term functional outcome after ileal pouch anal anastomosis in 191 patients with ulcerative colitis. J Crohns Colitis 2014; 8: 1261–1266. [DOI] [PubMed] [Google Scholar]
- 6.Ferrante M, Declerck S, De Hertogh G, et al. Outcome after proctocolectomy with ileal pouch-anal anastomosis for ulcerative colitis. Inflamm Bowel Dis 2008; 14: 20–28. [DOI] [PubMed] [Google Scholar]
- 7.Lightner AL, Mathis KL, Dozois EJ, et al. Results at up to 30 years after ileal pouch-anal anastomosis for chronic ulcerative colitis. Inflamm Bowel Dis 2017; 23: 781–790. [DOI] [PubMed] [Google Scholar]
- 8.Maaser C, Sturm A, Vavricka SR, et al. ECCO-ESGAR guideline for diagnostic assessment in IBD Part 1: Initial diagnosis, monitoring of known IBD, detection of complications. J Crohns Colitis 2019; 13: 144–164. [DOI] [PubMed] [Google Scholar]
- 9.Sandborn WJ, Tremaine WJ, Batts KP, et al. Pouchitis after ileal pouch-anal anastomosis: A Pouchitis Disease Activity Index. Mayo Clin Proc 1994; 69: 409–415. [DOI] [PubMed] [Google Scholar]
- 10.Shen B, Achkar JP, Connor JT, et al. Modified pouchitis disease activity index: A simplified approach to the diagnosis of pouchitis. Dis Colon Rectum 2003; 46: 748–753. [DOI] [PubMed] [Google Scholar]
- 11.Gionchetti P, Rizzello F, Venturi A, et al. Antibiotic combination therapy in patients with chronic, treatment-resistant pouchitis. Aliment Pharmacol Ther 1999; 13: 713–718. [DOI] [PubMed] [Google Scholar]
- 12.Gionchetti P, Calabrese C, Calafiore A, et al. Oral beclomethasone dipropionate in chronic refractory pouchitis. J Crohns Colitis 2014; 8: 649–653. [DOI] [PubMed] [Google Scholar]
- 13.Gionchetti P, Calabrese C, Lauri A, et al. The therapeutic potential of antibiotics and probiotics in the treatment of pouchitis. Expert Rev Gastroenterol Hepatol 2015; 9: 1175–1181. [DOI] [PubMed] [Google Scholar]
- 14.Patel RT, Bain I, Youngs D, et al. Cytokine production in pouchitis is similar to that in ulcerative colitis. Dis Colon Rectum 1995; 38: 831–837. [DOI] [PubMed] [Google Scholar]
- 15.Calabrese C, Gionchetti P, Rizzello F, et al. Short-term treatment with infliximab in chronic refractory pouchitis and ileitis. Aliment Pharmacol Ther 2008; 27: 759–764. [DOI] [PubMed] [Google Scholar]
- 16.Ferrante M, D'Haens G, Dewit O, et al. Efficacy of infliximab in refractory pouchitis and Crohn's disease-related complications of the pouch: A Belgian case series. Inflamm Bowel Dis 2010; 16: 243–249. [DOI] [PubMed] [Google Scholar]
- 17.Barreiro-de Acosta M, Garcia-Bosch O, Souto R, et al. Efficacy of infliximab rescue therapy in patients with chronic refractory pouchitis: A multicenter study. Inflamm Bowel Dis 2012; 18: 812–817. [DOI] [PubMed] [Google Scholar]
- 18.Viazis N, Giakoumis M, Koukouratos T, et al. Long term benefit of one year infliximab administration for the treatment of chronic refractory pouchitis. J Crohns Colitis 2013; 7: e457–e460. [DOI] [PubMed] [Google Scholar]
- 19.Segal JP, Penez L, Mohsen Elkady S, et al. Long term outcomes of initial infliximab therapy for inflammatory pouch pathology: A multi-centre retrospective study. Scand J Gastroenterol 2018; 53: 1051–1058. [DOI] [PubMed] [Google Scholar]
- 20.Huguet M, Pereira B, Goutte M, et al. Systematic review with meta-analysis: Anti-TNF Therapy in refractory pouchitis and crohn's disease-like complications of the pouch after ileal pouch-anal anastomosis following colectomy for ulcerative colitis. Inflamm Bowel Dis 2018; 24: 261–268. [DOI] [PubMed] [Google Scholar]
- 21.Barreiro-de Acosta M, Garcia-Bosch O, Gordillo J, et al. Efficacy of adalimumab rescue therapy in patients with chronic refractory pouchitis previously treated with infliximab: A case series. Eur J Gastroenterol Hepatol 2012; 24: 756–758. [DOI] [PubMed] [Google Scholar]
- 22.Philpott J, Ashburn J, Shen B. Efficacy of vedolizumab in patients with antibiotic and anti-tumor necrosis alpha refractory pouchitis. Inflamm Bowel Dis 2017; 23: E5–E6. [DOI] [PubMed] [Google Scholar]
- 23.Schmid M, Frick JS, Malek N, et al. Successful treatment of pouchitis with Vedolizumab, but not fecal microbiota transfer (FMT), after proctocolectomy in ulcerative colitis. Int J Colorectal Dis 2017; 32: 597–598. [DOI] [PubMed] [Google Scholar]
- 24.Mir F, Yousef MH, Partyka EK, et al. Successful treatment of chronic refractory pouchitis with vedolizumab. Int J Colorectal Dis 2017; 32: 1517–1518. [DOI] [PubMed] [Google Scholar]
- 25.Coletta M, Paroni M, Caprioli F. Successful treatment with vedolizumab in a patient with chronic refractory pouchitis and primary sclerosing cholangitis. J Crohns Colitis 2017; 11: 1507–1508. [DOI] [PubMed] [Google Scholar]
- 26.Bar F, Kuhbacher T, Dietrich NA, et al. Vedolizumab in the treatment of chronic, antibiotic-dependent or refractory pouchitis. Aliment Pharmacol Ther 2018; 47: 581–587. [DOI] [PubMed] [Google Scholar]
- 27.Gregory M, Weaver KN, Hoversten P, et al. Efficacy of vedolizumab for refractory pouchitis of the ileo-anal pouch: Results from a multicenter US cohort. Inflamm Bowel Dis 2019. Feb 27.; pii: izz030. doi: 10.1093/ibd/izz030. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kjaer MD, Qvist N, Nordgaard-Lassen I, et al. Adalimumab in the treatment of chronic pouchitis. A randomized double-blind, placebo-controlled trial. Scand J Gastroenterol 2019; 54: 188–193. [DOI] [PubMed] [Google Scholar]
- 29.ClinicalTrials.gov. Phase 4 study to evaluate the efficacy and safety of vedolizumab in the treatment of chronic pouchitis (EARNEST), 2016, https://www.clinicaltrials.gov/ct2/show/NCT02790138?term=NCT02790138&rank=1 (accessed 2 June 2019).
- 30.Segal JP, Ding NS, Worley G, et al. Systematic review with meta-analysis: The management of chronic refractory pouchitis with an evidence-based treatment algorithm. Aliment Pharmacol Ther 2017; 45: 581–592. [DOI] [PubMed] [Google Scholar]
- 31.Van den Berghe N, Verstockt B, Tops S, et al. Immunogenicity is not the driving force of treatment failure in vedolizumab-treated inflammatory bowel disease patients. J Gastroenterol Hepatol 2019. Jul; 34(7): 1175–1181. doi: 10.1111/jgh.14584. [DOI] [PubMed] [Google Scholar]
- 32.Herfarth H, Barnes EL, Long MD, et al. Combined endoscopic and oral fecal microbiota transplantation in patients with antibiotic-dependent pouchitis: Low clinical efficacy due to low donor microbial engraftment. Inflamm Intest Dis 2019; 4: 1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Selvig D, Piceno Y, Terdiman J, et al. Fecal Microbiota transplantation in pouchitis: Clinical, endoscopic, histologic, and microbiota results from a pilot study. Dig Dis Sci 2019. Jul 13.; doi: 10.1007/s10620-019-05715-2. [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 34.Ollech JE, Rubin DT, Glick L, et al. Ustekinumab Is effective for the treatment of chronic antibiotic-refractory pouchitis. Dig Dis Sci 2019. Jun 11.; doi: 10.1007/s10620-019-05697-1. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Material for Outcome of biological therapies in chronic antibiotic-refractory pouchitis: A retrospective single-centre experience by Bram Verstockt, Charlotte Claeys, Gert De Hertogh, Gert Van Assche, Albert Wolthuis, André D’Hoore, Séverine Vermeire and Marc Ferrante in United European Gastroenterology Journal