Abstract
Objective
Accelerated atherosclerosis remains the major cause of late death (after 5 years) in SLE. Yet, the ‘traditional’ cardiovascular risk equations (such as Framingham) consistently underestimate the risk. We sought to construct a data-driven formula for cardiovascular risk in SLE, based on data collected during the first year in a longitudinal cohort, for research purposes.
Methods
Two risk formulas were derived: one was for a broad set of cardiovascular outcomes (myocardial infarction, stroke, onset of angina, coronary procedures such as bypass or stent, claudication, peripheral artery disease or congestive heart failure); and the other for hard outcomes (myocardial infarction or stroke). Traditional and SLE-specific risk factors for cardiovascular disease were measured during the first year of cohort participation. Using Cox proportional hazards modelling, SLE formulas to calculate the 10-year risk of a subsequent cardiovascular event were derived and compared with the Framingham (for the broader outcome) and American College of Cardiology formulas (for the hard outcomes).
Results
SLE-related risk factors for each model included mean disease activity score (as measured by the SELENA revision of the SLE Disease Activity Index), low C3 and history of lupus anticoagulant. In those with SLE-related risk factors, the estimated 10-year risk based on our formula was substantially higher than the risk estimated based on the formulas for the general population.
Conclusions
The excess cardiovascular risk among patients with SLE varies substantially depending on the SLE-related risk factors, age and traditional risk factors. Cardiovascular risk formulas based on individual data from patients with SLE may better estimate 10-year cardiovascular risk among patients with SLE than the Framingham or American College of Cardiology equations.
Keywords: systemic lupus erythematosus (SLE), cardiovascular, risk assessment
Introduction
The risk of a cardiovascular event in SLE is substantially increased in SLE over controls. In the Hopkins Lupus Cohort, we showed that the risk was increased by a factor of 2.661 over the expectation based on the Framingham risk score. Surrogate markers of coronary atherosclerosis are similarly increased, with a prevalence rate of coronary artery calcium of 1.9 over community controls, after adjusting for traditional cardiovascular risk factors.2
The pathogenesis of accelerated atherosclerosis in SLE is complex and multifactorial. SLE can itself affect the endothelium of the coronary arteries.3 Antiphospholipid antibodies, particularly the lupus anticoagulant, can increase cardiovascular events through hypercoagulability. Traditional cardiovascular risk factors can be caused or worsened by SLE, such as lupus nephritis increasing hypertension and hyperlipidaemia, and worsened by treatment with prednisone.4 Traditional cardiovascular risk factors were the major predictors of 2-year progression of subclinical atherosclerosis in the SLE statin intervention randomised clinical trial.5
In the general population, the most frequently employed cardiovascular risk score is Framingham.6 Other equations include Reynolds, which includes high-sensitivity C-reactive protein (hsCRP),7 and the American College of Cardiology/American Heart Association (ACC/AHA).8 However, these risk scores are insufficient to ascertain risk in SLE, as the SLE risk is affected by SLE-related factors. A similar dilemma was faced in rheumatoid arthritis, in which cardiovascular disease is also the major cause of death. In rheumatoid arthritis, risk score models have been adapted for patients by introducing a 1.5 multiplication criteria, if the patient met two out of three criteria.9 10
SLE, however, is a heterogeneous disease, and certain disease manifestations and treatments have been specifically linked with increased risk of cardiovascular events. Therefore, the excess risk of a cardiovascular event among patients with SLE varies between patients. Rather than apply a constant factor to estimate the increased risk in specific patients,11 we sought to develop a formula for estimating cardiovascular risk based on both a patient’s traditional and SLE risk factors. In the Hopkins Lupus Cohort, we have a protocol of quarterly visits to ascertain SLE and traditional cardiovascular risk factors and thousands of years of follow-up. We used the database from this cohort to derive two formulas for cardiovascular risk and compared them with either Framingham or ACC/AHA scores.
Patients and methods
Patients with SLE seen at Hopkins since 1987 were invited to join the cohort. All patients gave informed written consent. Patients in the cohort are seen quarterly. Patients met the Systemic Lupus International Collaborating Clinics (SLICC) classification criteria for SLE.12 The authors had full access to all the data in the study and take responsibility for the integrity and analysis of the data.
The analysis for this study was based on cohort data through 2018. Since the goal was to estimate risk of an incident cardiovascular event, patients with a history of a cardiovascular event before cohort entry were excluded. Because the risk factors used in the analysis were those measured over the first year of cohort participation (as described in ascertainment of risk factors), we excluded patients with a cardiovascular event during the first year, and those who left the cohort within the first year.
Ascertainment of cardiovascular events
Patients were followed to ascertain incident stroke or myocardial infarction, angina or coronary procedures, claudication or congestive heart failure. Myocardial infarction diagnosis was based on patient symptoms, electrocardiographic findings, cardiac echocardiogram and/or cardiac biomarker levels. Thrombotic stroke was defined as rapid onset of neurological deficit not secondary to brain trauma (closed head injury), tumour, infection (eg, encephalitis or meningitis) or other non-vascular cause. In addition, there had to be either a clinically relevant lesion on brain imaging or duration >24 hours or death within 24 hours. Angina, coronary procedures, claudication and congestive heart failure were defined based on the SLICC/American College of Rheumatology Damage Index.13
Ascertainment of risk factors
Traditional cardiovascular predictors included as candidates for our prediction model were those used in the Framingham and ACC/AHA risk scores. These included age, sex, ethnicity, systolic blood pressure, total cholesterol, smoking and diabetes mellitus. High-density lipoprotein cholesterol was not included because it was unknown for a substantial number of cohort members during the first year of follow-up. We did not include statin therapy as we had previously shown that statin intervention did not prevent progression of atherosclerosis in SLE.14
SLE-related risk factors included as candidates for our model were time since SLE diagnosis, corticosteroid use, hydroxychloroquine use, low C3 and C4, anti-dsDNA, proteinuria, the SELENA revision of the SLE Disease Activity Index (SELENA-SLEDAI) score,15 estimated glomerular filtration rate based on the chronic kidney disease epidemiology serum creatinine formula and history of lupus anticoagulant and anticardiolipin. These were chosen based on our previous study.1 These were measured at quarterly cohort clinic visits.
For quantitative variables that changed from visit to visit (eg, blood pressure, SLEDAI), we used the average value of these measures over the first year of cohort participation as predictors in our models. We chose to average them (rather than simply use the first measure observed in the cohort) because some of the traditional risk factors (eg, blood pressure) and SLE-related predictors (SLEDAI, anti-dsDNA, low C3) vary over time, especially initially, and a single baseline measure may not represent the general condition of a patient with SLE. For variables that were not assessed at each clinic visit, we used both cohort and precohort medical review data.
Statistical methods
To derive our risk equations, we estimated the degree to which these predictors were associated with subsequent cardiovascular events, using Cox proportional hazards regression. To build our model, we started with the traditional risk factors included in the Framingham and ACC/AHA risk scores and then added the SLE risk factors. The relationship between quantitative variables and the log HRs was assessed for linearity and, in some cases, quantitative predictors were dichotomised for simplicity based on our findings. If two predictors were highly correlated (eg, C3 and C4), we determined which was the most important to include. Informed by these preliminary analyses, final multivariable models were developed. Using the results of the Cox model, formulas to calculate the risk of a stroke and myocardial infarction within the next 10 years were derived using the Breslow method as implemented in SAS V.9.4.
The discrimination of the models was quantified using Harrell’s C-statistic. To assess calibration of the models, we divided the samples into five groups based on the 10-year risk predicted by the model and compared the average risk in each group with the 10-year risk estimated by a Kaplan-Meier approach applied to each group.
We developed two risk formulas. First, we developed a formula for the 10-year risk of a general cardiovascular event. This included myocardial infarction, stroke, onset of angina, coronary procedures such as bypass or stent, claudication, peripheral arterial disease or congestive heart failure. Second, we developed a risk formula for ‘hard’ cardiovascular events which included myocardial infarction or stroke.
We compared our risk estimates with estimates of cardiovascular risk in the general population based on previously published formulas. Specifically, for the broader cardiovascular outcome we compared our findings with a formula based on the Framingham cardiovascular risk score.6 In addition, we compared our estimates of the risk of hard cardiovascular events (myocardial infarction or stroke) with that found using the ACC/AHA risk score.8
Results
There were 2557 participants in the Hopkins Lupus Cohort at the time of this analysis. Of these, 267 had a cardiovascular event (based on the broader definition) prior to cohort entry or during the first year of cohort participation. These patients were excluded from the development of the risk model for the broad outcomes. In addition, 670 patients were followed for <1 year in the cohort and were also excluded (because the analysis was based on follow-up that occurred after 1 year of cohort participation). Finally, 94 patients were excluded due to missing values for one of the main predictors. The derivation of the rule for the broader definition of cardiovascular events was based on the remaining 1721 patients. Table 1 shows the characteristics of those patients.
Table 1.
Characteristics of patients with SLE used to derive the risk formula for the broader definition of cardiovascular events
| Patient Characteristic | Number (%) |
| Sex | |
| Female | 1587 (92%) |
| Male | 134 (8%) |
| Race | |
| White | 898 (52%) |
| Black | 688 (40%) |
| Other | 135 (8%) |
| Age group (years) | |
| 20–29 | 519 (30%) |
| 30–39 | 528 (31%) |
| 40–49 | 356 (21%) |
| 50–59 | 222 (13%) |
| 60+ | 96 (6%) |
| Years since SLE diagnosis at cohort entry | |
| <2 | 682 (40%) |
| 2–4 | 289 (17%) |
| 4–7 | 242 (14%) |
| 7–10 | 167 (10%) |
| 10+ | 339 (20%) |
| Duration of follow-up (years) | |
| 2–5 | 749 (44%) |
| 5–10 | 425 (25%) |
| 10–15 | 271 (16%) |
| 15+ | 276 (16%) |
| Mean Systolic Blood Pressure (mmHg)* | |
| <120 | 802 (47%) |
| 120–129 | 461 (27%) |
| 130–139 | 261 (15%) |
| 140+ | 197 (11%) |
| Treated for hypertension | 596 (35%) |
| Current or Past Smoking | 599 (35%) |
| Mean Total Serum Cholesterol (mg/dl)* | |
| <140 | 181 (11%) |
| 140-199 | 945 (56%) |
| 200+ | 576 (34%) |
| Diabetes | 151 (9%) |
| Mean SLEDAI* | |
| 0 | 186 (11%) |
| >0–2 | 757 (44%) |
| 3+ | 778 (45%) |
| History of Lupus Anticoagulant | 345 (20%) |
| Low mean C3* | 403 (23%) |
| Mean eGFR* | |
| ≥90 | 1173 (69%) |
| 60–89 | 385 (23%) |
| <60 | 130 (8%) |
*Mean during the first year on the cohort.
eGFR, estimated glomerular filtration rate; SLEDAI, SLE Disease Activity Index.
Of the 1721 patients, 168 had a cardiovascular event after the first year of cohort participation, and during cohort participation. Of these 168 events, there were 70 strokes, 27 myocardial infarctions, 27 patients with angina or requiring coronary procedures, 14 patients with heart failure, 13 with peripheral artery disease, 11 with claudication and 6 with more than one type of event.
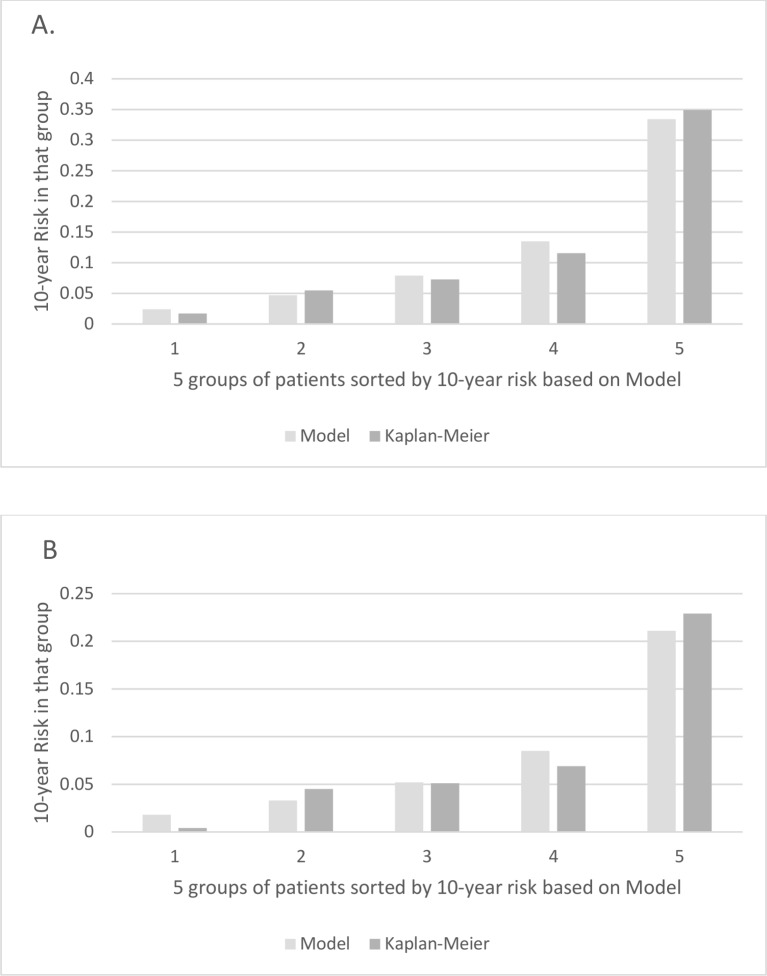
Table 2 shows the HRs for rates of the broader definition of cardiovascular events based on a multivariable Cox model including both traditional and SLE-related risk factors. The C-statistic for this model was 0.78. Figure 1A provides the calibration plot which shows the degree to which the average 10-year risks calculated from the model agree with the Kaplan-Meier estimates of the risk.
Table 2.
Association between predictors measured in the first year of follow-up and subsequent risk of a cardiovascular event based on the broad definition of a cardiovascular event*
| HR (95% CI) | P value | |
| Age (per decade) | 1.39 (1.22 to 1.60) | <0.0001 |
| Male (vs female) | 1.48 (0.95 to 2.32) | 0.084 |
| Systolic blood pressure (per 10 mm Hg, treated) | 1.23 (1.10 to 1.38) | 0.0003 |
| Systolic blood pressure (per 10 mm Hg, not treated) | 1.21 (1.07 to 1.37) | 0.0029 |
| Current or past smoking | 1.76 (1.29 to 2.40) | 0.0004 |
| Total serum cholesterol >140 mg/dL | 2.35 (1.15 to 4.80) | 0.020 |
| Diabetes mellitus | 2.40 (1.66 to 3.49) | <0.0001 |
| SLEDAI (per unit increase) | 1.07 (1.00 to 1.14) | 0.041 |
| History of lupus anticoagulant | 1.71 (1.23 to 2.37) | 0.0013 |
| Low mean C3 (<79) | 2.35 (1.62 to 3.42) | <0.0001 |
*The broad definition includes coronary death, myocardial infarction, coronary insufficiency, angina, ischaemic stroke, haemorrhagic stroke, transient ischaemic attack, peripheral artery disease, heart failure.
SLEDAI, SLE Disease Activity Index.
Figure 1.
Calibration plots for risk scores: model bars indicate the average 10-year risk in each group. Grey bars indicate the Kaplan-Meier estimates of the 10-year risk in each group. (A) Calibration plot of model for broad outcomes risk score. (B) Calibration plot for hard outcome risk score.
The HRs in table 2 can be used to calculate the 10-year risk for a patient with any risk factor profile using the formula:
Risk=1−0.950HR
where HR is the hazard ratio for that patient relative to a non-smoking female aged 50 years with untreated systolic blood pressure=120 mm Hg, and no other traditional or SLE-related risk factors. For example, for a male aged 70 years with systolic blood pressure equal to 120 mm Hg and no other traditional or SLE risk factors, the HR would be increased by a factor of 1.484 (because the HR for males over females is 1.48, table 2) and by a factor of 1.3922 (due to 70 being two decades older than 50). Thus, the overall HR for a male aged 70 years is (1.484)(1.3922), which equals 2.88. The 10-year risk is therefore equal to 1−0.9502.88, which computes to 13.7%.
Table 3 shows the implications of this formula for the 10-year cardiovascular risk among selected subsets of patients with SLE. From table 3, based on the point estimates, it can be seen that those with no SLE-related risk factors have comparable risk to the Framingham population. However, among those with SLE-related risk factors, the estimated 10-year risk is substantially higher than that in the general population based on the Framingham formula.
Table 3.
Estimated 10-year risk of a cardiovascular event (broad definition*) based on the formulas from the Hopkins Lupus Cohort and the Framingham cohort, given various risk factors
| Traditional risk factors† | SLE-related risk factors | Estimated 10-year risk | |||||||
| Sex | Age | SBP | Chol | HDL | Mean SLEDAI | Low C3 | Lupus anticoagulant | Hopkins Lupus Cohort (% risk, 95% CI) | Framingham‡ (% risk) |
| Low risk from traditional risk factors, no SLE risk factors | |||||||||
| F | 50 | 120 | 150 | 50 | 0 | No | No | 5.0 (3.1 to 6.8) | 2.8 |
| M | 50 | 120 | 150 | 50 | 0 | No | No | 7.3 (3.3 to 11.2) | 5.3 |
| F | 70 | 120 | 150 | 50 | 0 | No | No | 9.4 (4.7 to 13.9) | 6.1 |
| M | 70 | 120 | 150 | 50 | 0 | No | No | 13.7 (4.9 to 21.7) | 14.1 |
| Moderate risk from traditional risk factors, no SLE risk factors | |||||||||
| F | 50 | 145 | 240 | 40 | 0 | No | No | 7.9 (4.2 to 11.4) | 9.6 |
| M | 50 | 145 | 240 | 40 | 0 | No | No | 11.5 (4.6 to 17.8) | 15.1 |
| F | 70 | 145 | 240 | 40 | 0 | No | No | 14.7 (6.6 to 22.1) | 19.7 |
| M | 70 | 145 | 240 | 40 | 0 | No | No | 21.0 (7.0 to 32.8) | 36.8 |
| Low risk from traditional risk factors, SLE risk factors | |||||||||
| F | 50 | 120 | 150 | 50 | 3 | Yes | Yes | 22.3 (12.0 to 31.4) | 2.8 |
| M | 50 | 120 | 150 | 50 | 3 | Yes | Yes | 31.2 (13.3 to 45.4) | 5.3 |
| F | 70 | 120 | 150 | 50 | 3 | Yes | Yes | 38.7 (16.1 to 55.1) | 6.1 |
| M | 70 | 120 | 150 | 50 | 3 | Yes | Yes | 51.6 (17.1 to 71.7) | 14.1 |
| Moderate risk from traditional risk factors, SLE risk factors | |||||||||
| F | 50 | 145 | 240 | 40 | 3 | Yes | Yes | 33.3 (15.7 to 47.1) | 9.6 |
| M | 50 | 145 | 240 | 40 | 3 | Yes | Yes | 45.1 (18.1 to 63.2) | 15.1 |
| F | 70 | 145 | 240 | 40 | 3 | Yes | Yes | 54.3 (21.7 to 73.4) | 19.7 |
| M | 70 | 145 | 240 | 40 | 3 | Yes | Yes | 68.7 (23.5 to 87.2) | 36.8 |
*Risk of a broad cardiovascular outcome, that is, coronary death, myocardial infarction, coronary insufficiency, angina, ischaemic stroke, haemorrhagic stroke, transient ischaemic attack, peripheral artery disease, heart failure.
†Also assuming no smoking, no diabetes and no treatment for hypertension.
‡Calculated at: https://www.framinghamheartstudy.org/fhs-risk-functions/cardiovascular-disease-10-year-risk/.
Chol, total cholesterol; HDL, high-density lipoprotein; SBP, systolic blood pressure; SLEDAI, SLE Disease Activity Index.
Our analysis of ‘hard’ cardiovascular outcomes was based on slightly more patients (1777) because we removed fewer patients with a history of events prior to the start of follow-up. Of these patients, 121 had a hard cardiovascular event after the first year of cohort participation, and during cohort participation. Of the 121 there were 79 strokes, 38 myocardial infarctions and 4 patients listed as both stroke and myocardial infarction.
Table 4 shows the HRs for rates of hard cardiovascular events based on a multivariable Cox model including both traditional and SLE-related risk factors. The C-statistic for this model was 0.77. Figure 1B provides the calibration plot which shows the degree to which the average 10-year risks calculated from the model agree with the Kaplan-Meier estimates of the risk.
Table 4.
Association between predictors measured in the first year of follow-up and subsequent risk of a hard cardiovascular event*
| HR (95% CI) | P value | |
| Age (per decade) | 1.26 (1.08 to 1.47) | 0.0038 |
| African-American | 1.22 (0.84 to 1.78) | 0.30 |
| Male (vs female) | 1.26 (0.72 to 2.19) | 0.42 |
| Systolic blood pressure (per 10 mm Hg, treated) | 1.29 (1.12 to 1.48) | 0.0003 |
| Systolic blood pressure (per 10 mm Hg, not treated) | 1.27 (1.10 to 1.48) | 0.0015 |
| Cholesterol (per 25 mg/dL increase) | 1.08 (0.99 to 1.16) | 0.074 |
| Current or past smoking | 1.64 (1.13 to 2.38) | 0.0094 |
| Diabetes | 1.70 (1.08 to 2.68) | 0.023 |
| SLEDAI (per unit increase) | 1.08 (1.00 to 1.16) | 0.059 |
| History of lupus anticoagulant | 2.08 (1.44 to 3.02) | 0.0001 |
| Low mean C3 | 1.98 (1.28 to 3.07) | 0.0023 |
*A hard cardiovascular event means a stroke or myocardial infarction.
SLEDAI, SLE Disease Activity Index.
The HRs in table 4 can be used to calculate the 10-year risk for a patient with any risk factor profile using the formula:
Risk=1−0.975HR
where HR is the hazard ratio for that patient relative to a non-smoking female aged 50 years with systolic blood pressure=120 mm Hg, total serum cholesterol exceeding 150 mg/dl, and no diabetes and no SLE risk factors.
Table 5 shows the implications of this formula for 10-year risk of hard cardiovascular events among selected subsets of patients with SLE. From table 5, based on the point estimates, it can be seen that for lupus patients without SLE risk factors there is a slight excess risk in the younger patients and a decreased risk in the older patients. However, among those with SLE risk factors, the estimated 10-year risk is substantially higher than that in the general population based on the ACC/AHA formula, especially for younger patients.
Table 5.
Estimated 10-year risk of a hard cardiovascular event* based on the formulas from the Hopkins Lupus Cohort and the ACC/AHA model, given various risk factors
| Traditional risk factors† | SLE-related risk factors | Estimated 10-year risk* | |||||||
| Sex | Age | SBP | Chol | HDL | Mean SLEDAI | Low C3 | Lupus anticoagulant | Hopkins Lupus Cohort (% risk, 95% CI) | ACC/AHA‡ (% risk) |
| Low risk from traditional risk factors, no SLE risk factors | |||||||||
| F | 50 | 120 | 150 | 50 | 0 | No | No | 2.5 (1.3 to 3.6) | 0.8 |
| M | 50 | 120 | 150 | 50 | 0 | No | No | 3.1 (1.0 to 5.2) | 2.1 |
| F | 70 | 120 | 150 | 50 | 0 | No | No | 3.8 (1.5 to 6.2) | 7.9 |
| M | 70 | 120 | 150 | 50 | 0 | No | No | 4.8 (1.0 to 8.5) | 14.1 |
| Moderate risk from traditional risk factors, no SLE risk factors | |||||||||
| F | 50 | 145 | 240 | 40 | 0 | No | No | 5.7 (2.3 to 9.0) | 3.0 |
| M | 50 | 145 | 240 | 40 | 0 | No | No | 7.1 (1.9 to 12.1) | 7.0 |
| F | 70 | 145 | 240 | 40 | 0 | No | No | 8.9 (2.7 to 14.7) | 13.4 |
| M | 70 | 145 | 240 | 40 | 0 | No | No | 11.0 (1.9 to 19.3) | 26.2 |
| Low risk from traditional risk factors, SLE risk factors | |||||||||
| F | 50 | 120 | 150 | 50 | 3 | Yes | Yes | 12.0 (5.3 to 18.1) | 0.8 |
| M | 50 | 120 | 150 | 50 | 3 | Yes | Yes | 14.8 (4.0 to 24.4) | 2.1 |
| F | 70 | 120 | 150 | 50 | 3 | Yes | Yes | 18.2 (5.2 to 29.4) | 7.9 |
| M | 70 | 120 | 150 | 50 | 3 | Yes | Yes | 22.3 (3.0 to 37.8) | 14.1 |
| Moderate risk from traditional risk factors, SLE risk factors | |||||||||
| F | 50 | 145 | 240 | 40 | 3 | Yes | Yes | 26.0 (8.8 to 40.0) | 3.0 |
| M | 50 | 145 | 240 | 40 | 3 | Yes | Yes | 31.6 (7.0 to 49.7) | 7.0 |
| F | 70 | 145 | 240 | 40 | 3 | Yes | Yes | 37.8 (8.2 to 57.9) | 13.4 |
| M | 70 | 145 | 240 | 40 | 3 | Yes | Yes | 45.0 (4.5 to 68.3) | 26.2 |
*Risk of a hard event (cardiovascular death, myocardial infarction or stroke).
†Also assuming female, no smoking, no diabetes mellitus and no treatment for hypertension.
‡As calculated at: http://www.cvriskcalculator.com.
ACC, American College of Cardiology; AHA, American Heart Association; Chol, total cholesterol; HDL, high-density lipoprotein; SBP, systolic blood pressure; SLEDAI, SLE Disease Activity Index.
Discussion
We have derived two SLE cardiovascular risk scores from SLE patient data over the first year of cohort follow-up to predict actual cardiovascular events following that period. Based on our analysis, the excess risk for cardiovascular events differs substantially between patients with SLE and depends on both their traditional risk factors and SLE-related risk factors.
The SLE cardiovascular risk score is instructive in that it clearly identified both SLE-related and traditional cardiovascular risk factors. The three SLE-specific risk factors were a global activity score (the SELENA-SLEDAI score), low C3 and the lupus anticoagulant. Disease activity as a risk factor is no surprise, as SLE activity affects the vascular endothelium.3 In addition, those with higher disease activity are more likely to be prescribed corticosteroids which we and others have shown to be associated with increased risk of cardiovascular events.1 16 Low C3 is already a known risk factor for some poor SLE outcomes, such as end-stage renal disease.17 Low C3 is particularly important, as even though it is a component in the SLEDAI, it had a ‘stand alone’ independent role, as well. Finally, the lupus anticoagulant is the antiphospholipid antibody most strongly associated with thrombosis in SLE,18 including myocardial infarction.19 Although renal insufficience is not specific for SLE, it certainly occurs at a younger age in patients with SLE. The traditional cardiovascular risk factors of age, male gender, systolic blood pressure, cholesterol, smoking and diabetes were also important in the SLE risk score.
A number of other SLE-related variables were considered for inclusion in the model including anticardiolipin, time since diagnosis of SLE, anti-dsDNA, C4, use of corticosteroids and use of hydroxychloroquine. These variables did not significantly improve the prediction of the model after inclusion of the other variables, and were therefore not included in the final risk scores. We were surprised that corticosteroid use was not significantly predictive of cardiovascular events, because in our previous analyses of data from our cohort, we observed a strong association between corticosteroid exposure and cardiovascular disease risk.1 However, in those analyses, we considered corticosteroid use throughout the follow-up, whereas in this analysis, we only considered corticosteroid use in the first year of cohort participation. And, it should also be noted that corticosteroid treatment increases blood pressure, cholesterol and diabetes which were included in the model.
Previously, Urowitz et al proposed a risk score for the broad class of cardiovascular events derived by simply multiplying the components of the Framingham risk score by 2.11 A strength of their approach is that it is based on a score derived from the relatively large Framingham cohort data set. However, a disadvantage of their approach is that it ignores the heterogeneity of risk among patients with SLE due to different severity or manifestations of SLE. Our data suggest that some patients with SLE are not at much higher risk than indicated by the Framingham score, whereas others who have SLE-related risk factors such as lupus anticoagulant are at substantially higher risk (table 3).
Limitations of our study include that patients came from one geographic area with one provider. The cohort, however, is ethnically balanced in terms of African-Americans and Caucasians. Another limitation is that our data reflect care from 1987 to the present. Patients with SLE diagnosed today might experience a different risk due to changes in treatment. A third limitation is that the risk estimates are based on a statistical model that makes some smoothing assumptions (such as linearity and lack of effect modification) that are likely to be only approximately true. A fourth limitation is that, as indicated by the CIs in our tables, there is some imprecision in our estimates of the exact level of risk. This is especially true for estimates of risk for males since 92% of the subjects were females.
The SLE cardiovascular risk score we derived requires independent external validation. Until that time, it should be considered a research tool. However, our findings highlight the importance of distinguishing different subsets of patients with SLE and show that the risk and excess risk of cardiovascular events varies greatly depending on both traditional and SLE-related risk factors. These findings may be helpful in the future in making decisions about treatment interventions as well as ordering imaging studies such as cardiac CT.
Footnotes
Contributors: All authors have made substantial contributions to the conception, design, drafting, analysis, interpretation of data and revision of the work. All authors have given final approval of the version published and agree to be accountable for all aspects of the work.
Funding: The Hopkins Lupus Cohort is supported by NIH Grants AR043727 and AR069572.
Disclaimer: The authors did not receive financial support or other benefits from commercial sources for the work reported in the manuscript, nor do the authors have any financial interests which could create a potential conflict of interest or the appearance of a conflict of interest with regard to this work.
Competing interests: None declared.
Patient consent for publication: Not required.
Ethics approval: The Hopkins Lupus Cohort was approved on a yearly basis by the Johns Hopkins University School of Medicine Institutional Review Board (Study number NA_00039294).
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: Data are available on reasonable request.
References
- 1.Magder LS, Petri M. Incidence of and risk factors for adverse cardiovascular events among patients with systemic lupus erythematosus. Am J Epidemiol 2012;176:708–19. 10.1093/aje/kws130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kiani AN, Magder LS, Post WS, et al. . Coronary calcification in SLE: comparison with the multi-ethnic study of atherosclerosis. Rheumatology 2015;54:1976–81. 10.1093/rheumatology/kev198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Belmont HM, Abramson SB, Lie JT. Pathology and pathogenesis of vascular injury in systemic lupus erythematosus. interactions of inflammatory cells and activated endothelium. Arthritis Rheum 1996;39:9–22. 10.1002/art.1780390103 [DOI] [PubMed] [Google Scholar]
- 4.Petri M, Lakatta C, Magder L, et al. . Effect of prednisone and hydroxychloroquine on coronary artery disease risk factors in systemic lupus erythematosus: a longitudinal data analysis. Am J Med 1994;96:254–9. 10.1016/0002-9343(94)90151-1 [DOI] [PubMed] [Google Scholar]
- 5.Kiani AN, Post WS, Magder LS, et al. . Predictors of progression in atherosclerosis over 2 years in systemic lupus erythematosus. Rheumatology 2011;50:2071–9. 10.1093/rheumatology/ker285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.D'Agostino RB, Vasan RS, Pencina MJ, et al. . General cardiovascular risk profile for use in primary care: the Framingham heart study. Circulation 2008;117:743–53. 10.1161/CIRCULATIONAHA.107.699579 [DOI] [PubMed] [Google Scholar]
- 7.Tattersall MC, Gangnon RE, Karmali KN, et al. . Women up, men down: the clinical impact of replacing the Framingham risk score with the Reynolds risk score in the United States population. PLoS One 2012;7:e44347 10.1371/journal.pone.0044347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Goff DC, Lloyd-Jones DM, Bennett G, et al. . 2013 ACC/AHA Guideline on the Assessment of Cardiovascular Risk. J Am Coll Cardiol 2014;63:2935–59. 10.1016/j.jacc.2013.11.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Agca R, Heslinga SC, Rollefstad S, et al. . EULAR recommendations for cardiovascular disease risk management in patients with rheumatoid arthritis and other forms of inflammatory joint disorders: 2015/2016 update. Ann Rheum Dis 2017;76:17–28. 10.1136/annrheumdis-2016-209775 [DOI] [PubMed] [Google Scholar]
- 10.Wahlin B, Innala L, Magnusson S, et al. . Performance of the expanded cardiovascular risk prediction score for rheumatoid arthritis is not superior to the ACC/AHA risk calculator. J Rheumatol 2019;46:130–7. 10.3899/jrheum.171008 [DOI] [PubMed] [Google Scholar]
- 11.Urowitz MB, Ibañez D, Su J, et al. . Modified Framingham risk factor score for systemic lupus erythematosus. J Rheumatol 2016;43:875–9. 10.3899/jrheum.150983 [DOI] [PubMed] [Google Scholar]
- 12.Petri M, Orbai A-M, Alarcón GS, et al. . Derivation and validation of the systemic lupus international collaborating clinics classification criteria for systemic lupus erythematosus. Arthritis Rheum 2012;64:2677–86. 10.1002/art.34473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gladman D, Ginzler E, Goldsmith C, et al. . The development and initial validation of the systemic lupus international collaborating Clinics/American College of rheumatology damage index for systemic lupus erythematosus. Arthritis Rheum 1996;39:363–9. 10.1002/art.1780390303 [DOI] [PubMed] [Google Scholar]
- 14.Petri MA, Kiani AN, Post W, et al. . Lupus atherosclerosis prevention study (LAPS). Ann Rheum Dis 2011;70:760–5. 10.1136/ard.2010.136762 [DOI] [PubMed] [Google Scholar]
- 15.Petri M, Kim MY, Kalunian KC, et al. . Combined oral contraceptives in women with systemic lupus erythematosus. N Engl J Med 2005;353:2550–8. 10.1056/NEJMoa051135 [DOI] [PubMed] [Google Scholar]
- 16.Souverein PC, Berard A, Van Staa TP, et al. . Use of oral glucocorticoids and risk of cardiovascular and cerebrovascular disease in a population based case-control study. Heart 2004;90:859–65. 10.1136/hrt.2003.020180 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mok CC, Ying KY, Tang S, et al. . Predictors and outcome of renal flares after successful cyclophosphamide treatment for diffuse proliferative lupus glomerulonephritis. Arthritis Rheum 2004;50:2559–68. 10.1002/art.20364 [DOI] [PubMed] [Google Scholar]
- 18.Petri M, Rheinschmidt M, Whiting-O'Keefe Q, et al. . The frequency of lupus anticoagulant in systemic lupus erythematosus. A study of sixty consecutive patients by activated partial thromboplastin time, Russell viper venom time, and anticardiolipin antibody level. Ann Intern Med 1987;106:524–31. 10.7326/0003-4819-106-4-524 [DOI] [PubMed] [Google Scholar]
- 19.Petri M. The lupus anticoagulant is a risk factor for myocardial infarction (but not atherosclerosis): Hopkins lupus cohort. Thromb Res 2004;114:593–5. 10.1016/j.thromres.2004.07.012 [DOI] [PubMed] [Google Scholar]