Abstract
Background
Diagnosis of Tuberculosis in children is challenging. Hereby we report a case series of 4 patients where Cartridge Based Nucleic Acid Amplification Test (CB NAAT) helped us in early diagnosis.
Case characteristics
Case 1 was a 14-year old girl who was on anti-tuberculous therapy (ATT) for 2 months without any improvement; sputum CBNAAT detected multi-drug resistant tuberculosis (MDR-TB). Case 2 was a 9-year old boy with history and examination findings suggestive of meningitis; CB-NAAT of the cerebrospinal fluid (CSF) was positive. Case 3 was a 1-year old child having fever, cough of 1 month duration with Mantoux positive. CB-NAAT of gastric lavage was positive. Case 4 was a 3- month old child who presented with severe respiratory distress of 11 days duration. Chest x-ray showed miliary mottling; CB-NAAT of gastric lavage was positive.
Conclusion
CB-NAAT testing was very useful in making an early and definitive diagnosis of tuberculosis, including MDR-TB.
Keywords: CB-NAAT, Pediatric, Tuberculosis, GeneXpert
Introduction
Tuberculosis (TB) remains a major global health problem. The TB epidemic is larger than previously estimated with an incidence of 10.4 million new TB cases worldwide in 2015, of which 5.9 million (56%) were among men, 3.5 million (34%) among women and 1.0 million (10%) among children. [1] India accounts for one fourth of the global TB burden. In 2015, an estimated 2,800,000 cases occurred and 480,000 people died due to TB. India has the highest burden of both TB and MDR-TB, based on estimates reported in Global TB Report 2016 (0.13 million of 0.48 million of MDR cases) [2].
Due to the paucibacillary nature of pediatric TB, the yield of identifying TB bacilli using the traditional method of sputum microscopy is very low. Because of the problem of AFB-smear-negative cases, clinical diagnosis of TB is the norm. According to the latest Revised National TB Control Program (RNTCP) and Indian Academy of Pediatrics (IAP) guidelines, all attempts should be made to obtain a bacteriological diagnosis. Early detection is the key to successful treatment and reduction of the disease transmission.
Cartridge based nucleic acid amplification test (CB-NAAT, GeneXpert,) is an automated cartridge-based molecular technique which not only detects Mycobacterium Tuberculosis but also rifampicin resistance within two hours and has been endorsed by WHO as an initial diagnostic test in children suspected of having tuberculosis both in pulmonary and specific forms of extra pulmonary tuberculosis. Globally the use of rapid molecular tests is increasing. Despite advances in diagnostics, a considerable proportion of the TB cases reported to WHO is still clinically diagnosed rather than bacteriological confirmation. So hereby we are reporting case series where CB-NAAT helped us in early diagnosis and effective management.
Our hospital is a tertiary care hospital and a designated medical centre for Directly observed treatment, short-course (DOTS) but CB- NAAT testing facility is not available in our centre. We transported the samples to the nearest MDR treatment centre in Mangalore where CB-NAAT testing is done free of cost by the government.
Case report
CASE NO. 1. A 14 year old girl who was on INH, pyrazinamide, ethambutol, rifampicin but at a suboptimal dose (not according to DOTS) presented with a history of high-grade fever, cough, breathlessness, loss of appetite and loss of weight for the last 2 months. Her initial diagnosis of TB was made based on clinical symptoms and supportive investigations but no sputum examination had been done. On examination of the Respiratory system, there was diminished movement on right infra scapular, infra axillary & mammary regions. Bronchial breath sounds was heard in the same areas. Complete blood count showed leucocytosis with raised ESR (106 mm/h). Chest x ray and CT chest was done (Fig. 1). Expectorated Sputum sample was sent for AFB and was found to be positive (3+). CB-NAAT test detected Mycobacterium Tuberculosis with rifampicin resistance, a surrogate marker for MDR tuberculosis. That was later confirmed by Line probe assay through RNTCP. The girl was treated as per RNTCP & IAP guidelines for MDR Tuberculosis.
Fig. 1.
Case 1. CT chest showing the Consolidation with breakdown cavitations of apico-posterior lobe in right lung, areas of consolidation with cavitation interspersed left lung, multiple centric lobar nodules with tree in bud appearance, multiple pre & para tracheal lymph nodes.
CASE NO. 2. A 9 yr old boy was admitted with history of high grade fever, headache and vomiting for 10 days. Meningeal signs were positive. No induration was noted in the Mantoux test, erythrocyte sedimentation rate (ESR) was 13 mm/ h with normal chest x ray. Gastric lavage sample was negative for AFB. CSF analysis showed cells count of 140 cells, predominately lymphocytes, protein of 138 mg/dl, sugar of 99 mg/dl. CSF culture showed no growth, but CSF CB - NAAT was positive for MTB, sensitive to rifampicin. CT brain showed mild enhancement of meninges suggestive of meningitis. The boy was started on anti-tuberculous therapy (ATT) and steroids as per guidelines. He was improved and doing well on follow up.
CASE NO. 3 A 1-year old boy presented with fever and cough for 1 month and a strong history of contact with an active case of TB. Tuberculin skin test measured 18 mm. Gastric lavage sample was CB-NAAT positive for TB, sensitive to rifampicin. The boy was started on ATT and doing well on follow up.
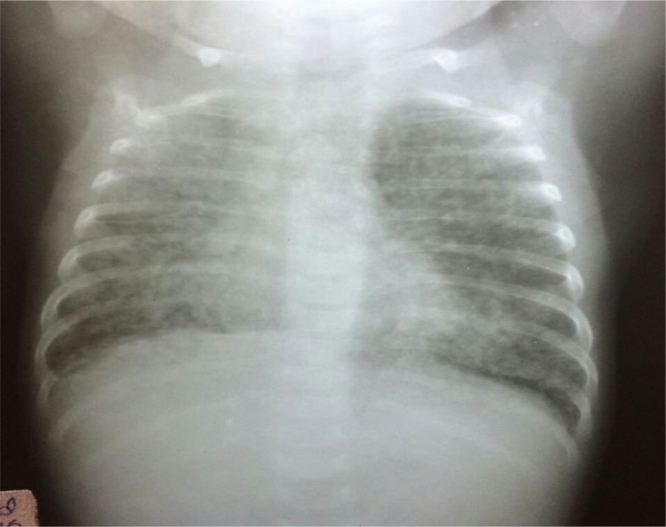
CASE NO. 4 A 3-month old male infant presented with cough, fever and breathing difficulty for 11 days. He was in severe respiratory distress with a respiratory rate of 88/min, intercostal and sub costal retractions with bilateral crepitations and rhonchi. Head nodding and nasal flaring with mottling of the skin were present requiring oxygen supplementation. X-ray showed miliary mottling (Fig. 2). Gastric lavage aspirate was CB-NAAT positive for TB (rifampicin sensitive). The infant was started on ATT.
Fig. 2.
Case 2. Chest x-ray showing bilateral miliary mottling.
For all the above cases HIV testing was negative; CB-NAAT Report was received within 24 hours. But the limitation of this study is that, except for the MDR case, culture was not done for the other 3 cases as it was not required for starting ATT.
Discussion
Though plenty of the literature is available on CB-NAAT in the recent years, use in routine pediatric practice is still limited. Thus, the purpose of reporting the above four case series was to show that CB-NAAT can be useful in day-to-day practice and to re-emphasise that not only expectorated sputum samples but also gastric lavage and other non-sputum specimens such as CSF can also be submitted for CB-NAAT testing.
Out of the total 67 million children with TB, according to a recent WHO report, 5 Million have INH resistance; 2 Million have MDR and 100,000 with XDR. Every year 25, 000 children develop MDR and 1200 XDR TB [3]. Looking at the increasing incidence and prevalence of drug resistance, CB-NAAT test will be a valuable screening tool, though with minor limitations.
The first study in India on the use of Xpert MTB/RIF assay in children was done by Sarman Singh et al. They reported on the utility of this test on archived gastric lavage and induced sputum samples with intrathoracic TB. Even in the samples stored up to 4 years, the assay remained a useful tool with a sensitivity that was superior to that of smear microscopy [4].
In a study done by Sharma SK et al, Xpert MTB/RIF had high sensitivity of 95.7% and specificity of 99.3% for detecting MTB in pulmonary samples of patients with TB. The sensitivity of Xpert MTB/RIF for detecting smear-negative culture positive samples was 77.7%; its sensitivity for detecting smear-positive culture-positive samples was 99.2%. The sensitivity and specificity for detecting rifampicin resistance was 94.5% and 97.7% respectively with respect to culture as reference standard [5]. Xpert MTB/RIF gives a diagnostic edge as pediatric tuberculosis is smear negative in most of the cases.
Specimens that can be sent for testing include respiratory specimens such as sputum, bronchial or tracheal aspirates, broncho-alveolar lavage and gastric lavage as well as extra pulmonary specimens like tissue biopsy including lymph node, pus from abscess, CSF, ascitic and pericardial fluid, pleural fluid [6]. CB-NAAT testing for TB on other samples such as stool, urine and blood is not recommended [7]. According to WHO, a positive yield of 60–70% of culture positive cases, availability of results in 2 hours, detection threshold of 130–150 cfu/ml, and increasing number of other specimens that can be included along with respiratory specimens [8].
Though Xpert MTB/RIF test is a useful tool for rapid identification of rifampicin resistant M. tuberculosis, results must always be confirmed by culture and drug susceptibility testing (DST) to detect additional drug resistance [1], [5]. Culture-based methods currently remain the reference standard for drug susceptibility testing. Conventional microscopy and culture are essential for monitoring therapy and for performing DST for anti-TB agents other than rifampicin (including for isoniazid and second-line anti-TB drugs.) [8]. Specimens of any type can be used for culture; the threshold for detection is low (10–100 cfu/ml compared to 130–150 cfu/ml for CB-NAAT). Patients with a negative GeneXpert result can still have TB with MTB or mycobacteria other than tuberculosis (MOTT) species. Additionally, the PCR test amplifies any DNA, whether of live or dead bacilli. Obtaining a clear history of treatment with ATT is required to avoid false positive results [9].
Limitations of CB-NAAT test include a requirement of stable electrical power supply, temperature control and annual calibration of instrument. Sufficient measures must be taken so that power supply remains uninterrupted (with additional batteries, a generator or solar panels), cartridges must be stored at the recommended temperature range (2–28 °C), and the equipment itself between 15 °C and 30 °C [7]. Regardless of all these limitations, Xpert MTB/RIF remains a very valuable tool because of its unambiguous, rapid results, and high sensitivity and specificity.
Therefore, there is a need for adaptation of this molecular test in regular pediatric practice. It is provided free of cost by the Government of India at selected treatment centres.
References
- 1.World health Organisation (WHO) 2016. Global Tuberculosis Report.http://apps.who.int/iris/bitstream/10665/250441/1/9789241565394-eng.pdf?ua=14 [Google Scholar]
- 2.Central TB division . New Delhi. Directorate General of Health services, Ministry of Health and Family Welfare, Government of India, New Delhi; 2011. TB division: Revised National TB control Programme Annual Status Report.http://tbcindia.nic.in/WriteReadData/TB%20India%202017.pdf [Google Scholar]
- 3.World health Organisation (WHO). Updated WHO MDR-TB treatment guidelines and the use of new drugs in children. 2016. who.int/entity/tb/areas-of-work/children/MalgosiaGrzemska_MDRTBguidelines.pdf.
- 4.Singh S, Singh A, Prajapathi S, Kabra SK, Lodha R, Mukherjee A. Xpert MTB/RIF assay can be used on archived gastric aspirate and induced sputum samples for sensitive diagnosis of paediatric tuberculosis. BMC Microbiol. 2015;15:191. doi: 10.1186/s12866-015-0528-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sharma SK, Kohli M, Yadav RN, Chaubey J, Bhasin D, Sreenivas V. Evaluating the diagnostic accuracy of xpert MTB/RIF assay in pulmonary tuberculosis. PLoS ONE. 2015;10(10) doi: 10.1371/journal.pone.0141011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Shah I, Gupta Y. Xpert MTB/RIF for diagnosis of tuberculosis and drug resistance in indian children. Indian Pediatr. 2016;53:837–838. doi: 10.1007/s13312-016-0943-8. [DOI] [PubMed] [Google Scholar]
- 7.World health Organisation (WHO). Xpert MTB/RIF implementation manual. Technical and operational ‘how-to’: Practical Considerations. 2014 apps.who.int/iris/bitstream/10665/112469/1/9789241506700_eng.pdf.
- 8.World health Organisation (WHO). Prevention and management of MDR-TB in children. 2016. who.int/entity/tb/areas-of-work/children/SimonSchaaf_MDRTB.pdf.
- 9.Agrawal M, Bajaj A, Bhatia V. Comparative study of Gene Xpert with ZN stain and culture in samples of suspected pulmonary tuberculosis. J Clin Diagn Res. 2016;10(5):DC09–DC12. doi: 10.7860/JCDR/2016/18837.7755. [DOI] [PMC free article] [PubMed] [Google Scholar]