Abstract
Fetal cardiovascular malformations is widely focused and screened, but the accuracy of screening is not satisfactory. In this study, we compared the types of congenital heart malformation, accompanying diseases and fetal outcomes in the first and second trimesters of pregnancy to clarify the advantage of early screening.
From January 2013 to June 2018, 230 fetuses were diagnosed with congenital heart malformations using ultrasound method in Qilu Hospital of Shandong University, and divided into 2 groups:the first trimester fetuses (group A) and the second trimester fetuses (group B). In addition, we collected and organized medical data of 347 cases diagnosed with congenital heart disease during 1998 to 2005 (group C). We compared the spectrum of congenital heart disease, associated comorbidities and outcome of fetuses diagnosed with congenital heart disease.
There were differences in the types and incidence of cardiac malformations between the first and second trimesters of pregnancy. The number of cases of non-cardiac malformation, congenital heart disease with single ventricular circulation, fetal intrauterine death and premature pregnancy termination was significantly lower in the late stage (group A and group B) than that in the early stage (group C). More patients were screened for trisomy 21, 18, 13 syndromes and Turner syndrome in group A than group B (P <.001). More fetuses with a 22q11 deletion were screened in group B than group C.
Early pregnancy screening using ultrasound diagnosis is very important for fetuses with congenital heart disease.
Keywords: fetal cardiovascular malformation, fetus, first trimester, screening, second trimester
1. Introduction
The incidence of fetal cardiovascular malformation has increased in recent years, and the prognosis is extremely poor. Early diagnosis is crucial, the methods for diagnosing fetal malformation mainly include computed tomography (CT), magnetic resonance imaging (MRI), and ultrasonography.[1] Among them, ultrasound has become the preferred method for diagnosing fetal congenital cardiovascular malformation because of its simple operation, low cost, no ionizing radiation, and real-time dynamics.[2,3] Ultrasound screening of fetal cardiovascular malformations in early pregnancy gradually plays a very important role in the regulation of pregnancy.[4] Combined with considerations such as maternal age, serum biochemistry, ultrasound abnormalities, and other ultrasound markers, ultrasound screening can identify most chromosomal abnormalities or abnormalities in the body.[5,6] In the early pregnancy examination, if the transparent layer of the fetal neck is found to be increased and has normal karyotype, the risk of congenital heart disease is significantly increased.[7,8,9] A foreign study also showed that fetuses with tricuspid regurgitation in early pregnancy screening also have a significantly increased risk of chromosomal and congenital heart disease.[10,11] This study analyzed the results of ultrasound diagnosis in the first trimester and the second trimester of fetuses diagnosed with congenital heart malformations, and compared the types of congenital heart malformations, accompanying diseases and fetal outcomes, in order to clarify the advantages of early screening.
2. Materials and methods
2.1. General Information
From January 2013 to June 2018, pregnant women were under ultrasound screening in Qilu Hospital of Shandong University, and 230 fetuses were diagnosed with congenital heart malformations. According to the time of initial ultrasound screening and disease diagnosis, fetuses were divided into 2 groups: Group A (early pregnancy group) including 70 cases of fetal diagnosed with congenital heart disease in early pregnancy (11+0 weeks–13+6 weeks). Group B (second trimester of pregnancy group) including 160 cases diagnosed with congenital heart disease in the second trimester (14+0 weeks–28+0 weeks). In addition, 347 fetuses that underwent a mid-pregnancy check and diagnosed with congenital heart disease during the period 1998 to 2005 were collected as group C. The study was approved by the Ethics Committee of Qilu Hospital of Shandong University. Informed consent was signed before the study began.
2.2. Research methods
The GEE8 color Doppler ultrasound system (probe frequency 3–5 MHz) was used to change the probe angle based on the 4-chamber view (4CV) to obtain the 3-vessel plane (3VV), the 3-vessel-tracheal plane (3VVT), the aortic root-axial section (AR-SAV), and left and right ventricular outflow tract (VOTV). The heart chamber, ventricular septum, atrial septum, cardiac outflow tract, heart valve morphology, and cardiac blood flow characteristics were observed, and various cardiovascular malformations were identified.
2.3. Test indicators
The success rate of different ultrasound section showing fetal cardiovascular malformation was observed. The types of congenital heart malformations, concomitant diseases, and fetal outcomes were compared. 66 cases of pregnancies diagnosed with congenital heart disease in the first trimester completed genetic consultation and karyotype analysis before delivery, while 164 cases diagnosed in the second trimester completed genetic testing after delivery. Fetal cells derived from chorionic villus or amniocentesis was used for analyzing prenatal cytology and molecular genetics.
2.4. Statistical methods
The data were analyzed by Statistical Package for the Social Sciences (SPSS) version 20.0 software (SPSS, Inc., Chicago, IL). The t test was used for the analysis of the measurement data between the groups. The analysis of the measurement data was performed by χ2 test. P <.05 showed that the difference was statistically significant.
3. Results
3.1. The display rate of different ultrasound section to the fetal cardiovascular malformation
As demonstrated in Figure 1, the 4-chamber view had the lowest display rate of fetal cardiovascular malformation (56.5%), while the 4-chamber + 3-vessel-tracheal plane showed the highest display rate of fetal cardiovascular malformation (95.6%). The difference between the various sections was statistically significant (P <.05).
Figure 1.
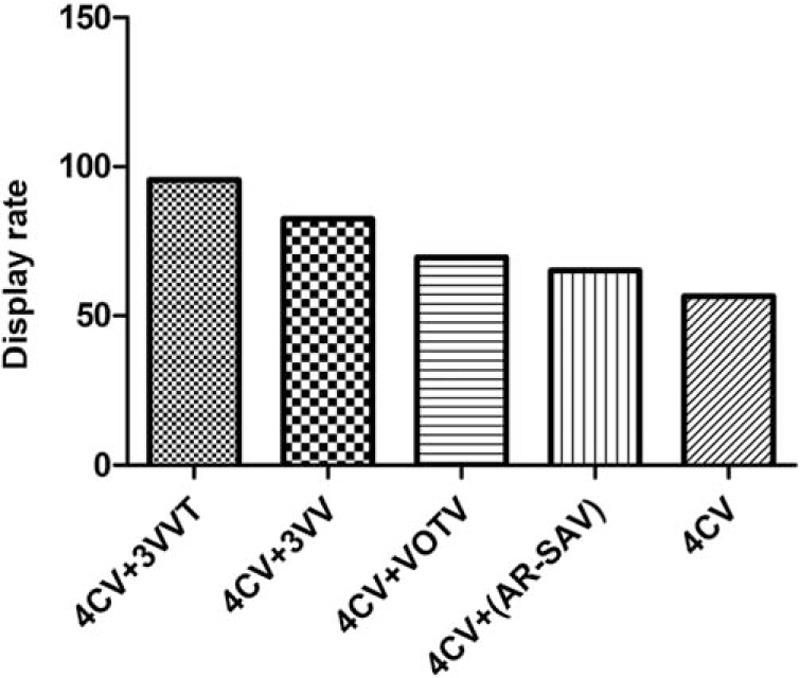
Display rate of different ultrasound section in fetal cardiovascular malformations.
3.2. Prenatal ultrasound diagnosis of fetal heart malformation
It was suggested that compared with the 2 groups, the percentage of cases with atrioventricular septal defect (AVSD) (P = .045), left ventricular dysplasia (HLHS) (P <.001), pulmonary atresia (PA) (P = .032), and tricuspid atresia (TA) (P = .021) diagnosed in group A was significantly higher than that in group B, the difference was statistically significant (P <.05). The percentage of transposition of the great arteries (TGA) (P = .024) and pulmonary stenosis (PS) (P <.001) screened in the second trimester was significantly different from that in the early pregnancy group. In addition, more fetuses were diagnosed with a right ventricular dual outflow tract, aortic coarctation, and vascular annulus in the second trimester (Table 1).
Table 1.
Comparison of the types and quantities of 3 groups of prenatal ultrasound diagnosis of congenital heart disease.

In addition, the mid-pregnancy detection rate of AVSD, HLHS, PA, and ventricular double inlet (DIV) before 2005 was significantly upregulated after 2013. On the other hand, the detection rate of TGA, tetralogy of Fallot (TOF), aortic atresia (AS) (P = .040), aortic coarctation (CoA), PS, and vascular ring (VR) was significantly higher in Group B than that in Group C (Table 1).
3.3. Comparison of associated diseases and fetal outcomes
Compared with group A, the percentage of cases with the chromosomal abnormalities and/or non-cardiac malformations, simple chromosomal abnormalities, structural abnormalities associated with non-cardiac, congenital heart disease with single ventricular circulation, intrauterine death, and termination of pregnancy was significantly decreased in group B (P <.05) (Table 2). Furthermore, compared with group B, the proportion of non-cardiac structural malformation diseases, congenital heart disease with single ventricular circulation, intrauterine death and termination of pregnancy in group C was significantly increased (P <.05). However, there was no difference in accompanying disease, chromosomal abnormality between group B and group C (P >.05).
Table 2.
Comparison of prenatal ultrasound diagnosis of concomitant diseases, cardiac cycle types and fetal outcomes in 3 groups.

3.4. Comparison of the types and numbers of chromosomal abnormalities in 3 groups
Results in Table 3 indicated that, compared with group A, less fetuses with chromosomal abnormality trisomy 21, 18, 13 syndromes and Turner syndrome were screened in group B, and the difference was significant (P <.001). Moreover, the percentage of fetuses with 22q11 deletion showed no difference between group A and group B (P >.05). Compared with group B, less fetuses with 22q11 deletion were screened in group C (P = .014), while the percentage of fetuses with chromosomal abnormality, trisomy 21, 18, 13 syndromes, and Turner syndrome showed no difference between 2 groups (P >.05).
Table 3.
Comparison of the types and quantities of chromosomal abnormalities in 3 groups.

4. Discussion
In the screening for fetal malformations including congenital heart malformations, early pregnancy screening has an important impact on mid-pregnancy screening and an effect on the outcome of congenital heart disease after birth that cannot be ignored.[12] Combining the special medical history of pregnant women, ultrasound examinations, and serum biochemical indicators can prompt screening for some major chromosomal abnormalities at 11 to 13 weeks of pregnancy, including some structural abnormalities, cardiac abnormalities, and some complications.
The 4-chamber view is the basic level of the heart examination and can show the shape and structure of the left and right atrium and ventricle. [13,14] This study showed that early pregnancy ultrasound screening plays a crucial role in the diagnosis of cardiovascular malformation. It was also proved that the display rate of fetal cardiovascular malformation detected by the 4-chamber heart + 3-vessel-tracheal plane is higher than that of the typical 4-chamber view.
Previous studies have reported that the spectrum and incidence of disease found in prenatal and postnatal screening are different.[15,16] This study also confirmed that the types and incidence of cardiac malformations screened in early pregnancy and mid-pregnancy are also inconsistent. The reason is because in the early pregnancy, the incidence of severe congenital heart disease is higher, and often accompanied by other system abnormalities, eventually leading to an increased probability of intrauterine death. The difference in the overall detection rate of different types of cardiac malformations also affects the type of disease detected during the first trimester and the second trimester.[17] The most common types of congenital heart disease detected during the first trimester are some morphological abnormalities that can be clearly seen during prenatal screening. Similar to the study of Matalon et al[18], the rate of AVSD and HLHS screened in early pregnancy reaches 44.3% in this study. In an early pregnancy study, Chameides et al found that 69% of the fetuses have AVSD or HLHS.[19] In the second trimester study, in addition to AVSD and HLHS, the diagnosis of other cardiac malformations requires observational assessment of large vessels (TGA, PS, right aortic arch, CoA, and AS) because blood vessels are constantly developing during pregnancy. Before 2005, early pregnancy screening was not yet popular. After 2013, early routine screening of pregnant women with high-risk pregnancies had a great impact on mid-pregnancy screening. More AVSD, HLHS, and PA were detected in 1998 to 2005 compared to the mid-pregnancy screenings of 2013 to 2018. Consistently, early pregnancy screening during the period 2013 to 2018 found more AVSD, HLHS, and PA than mid-pregnancy screening. Further studies found that in the mid-pregnancy screening during different periods, the number of non-cardiac malformations, congenital heart disease with single ventricular circulation, intrauterine death, and premature termination of pregnancy was significantly decreased in the later stages compared to the early stages due to the high screening rate of severe congenital heart disease and the high early termination of pregnancy rate in early pregnancy. In previous reports, the detection rate of fetal chromosomal abnormalities is 16% to 50%.[20,21,22] In the 2013 to 2018 screening, the study found that the risk of cardiac malformation is significantly promoted in the early stages of pregnancy in comparison with other periods, and often accompanied by chromosomal abnormalities. In the early pregnancy, 47.1% of congenital heart fetuses are associated with chromosomal abnormalities, while in foreign reports, the proportion is as high as 72.09%.[12] In the mid-pregnancy screening of the first 2 periods, the detection rates are 17.5% and 17.3%, respectively. Above findings indicate that the chromosomal abnormalities diagnosed in early pregnancy are significantly higher than those in the second trimester.
5. Conclusions and limitations
In summary, the spectrum and incidence of congenital heart disease detected in early pregnancy and mid-pregnancy screening are different. Early pregnancy screening has an important impact on the outcome of a fetus with congenital heart disease, because at this stage, severe heart malformations, chromosomal abnormalities, and more complications are more likely to be discovered, resulting in more termination of pregnancy in early pregnancy. This study is beneficial to improve health problems of children in society. However, there are a few limitations: the small sample size, no information on the health status of the parents, and no analysis in correlations of congenital heart disease with the job of the parents.
Declarations
Author contributions
Conceptualization: Bing Han.
Data curation: Bing Han, Ying Li, Haibo Song, Yinhui Xu.
Formal analysis: Ying Li, Yi Tang, Haibo Song, Yinhui Xu.
Investigation: Bing Han, Ying Li, Yi Tang, Haibo Song, Yinhui Xu.
Methodology: Bing Han, Xueling Qu.
Project administration: Xueling Qu.
Resources: Yi Tang.
Supervision: Bing Han.
Writing – original draft: Bing Han, Ying Li, Fen Wang.
Writing – review & editing: Bing Han.
Footnotes
Abbreviations: SPSS = Statistical Package for the Social Sciences, AVSD = atrioventricular septal defect, HLHS = left ventricular dysplasia, PA = pulmonary atresia, CoA = aortic coarctation, TGA = transposition of the great arteries, PS = pulmonary stenosis.
The datasets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
The study was approved by the Ethics Committee of Qilu Hospital of Shandong University. Signed written informed consents were obtained from the guardians.
The authors have no conflicts of interest to disclose.
References
- [1]. Wang] L, Wu QQ, Chen Y, et al. Ultrasound screening of fetal structural abnormalities by standard ultrasound views during the first trimester. Chin Med J (Engl) 2013;126:986–7. [PubMed] [Google Scholar]
- [2]. Finch AD. The importance of fetal echocardiography in the detection and management of congenital cardiac malformations. Rev Esp Cardiol 2006;3:23–8. [DOI] [PubMed] [Google Scholar]
- [3]. El Guindi W, Dreyfus M, Carles G, et al. 3D ultrasound and Doppler angiography for evaluation of fetal cardiovascular anomalies. Int J Gynaecol Obstet 2013;120:173–7. [DOI] [PubMed] [Google Scholar]
- [4]. Miller E, Hare JW, Cloherty JP, et al. Elevated maternal hemoglobin A1c in early pregnancy and major congenital anomalies in infants of diabetic mothers. N Engl J Med 1981;304:1331–4. [DOI] [PubMed] [Google Scholar]
- [5]. Oyen N, Poulsen G, Boyd HA, et al. Recurrence of congenital heart defects in families. Circulation 2009;120:295–301. [DOI] [PubMed] [Google Scholar]
- [6]. Tegnander E, Williams W, Johansen OJ, et al. Prenatal detection of heart defects in a non-selected population of 30,149 fetuses--detection rates and outcome. Ultrasound Obstet Gynecol 2006;27:252–65. [DOI] [PubMed] [Google Scholar]
- [7]. Wren C, Richmond S, Donaldson L. Temporal variability in birth prevalence of cardiovascular malformations. Heart 2000;83:414–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8]. Moons P, Sluysmans T, De Wolf D, et al. Congenital heart disease in 111 225 births in Belgium: birth prevalence, treatment and survival in the 21st century. Acta Paediatr 2009;98:472–7. [DOI] [PubMed] [Google Scholar]
- [9]. Souma ML. Maternal transport: behind the drama. Am J Obstet Gynecol 1979;134:904–10. [DOI] [PubMed] [Google Scholar]
- [10]. Abu-Sulaiman RM, Subaih B. Congenital heart disease in infants of diabetic mothers: echocardiographic study. Pediatr Cardiol 2004;25:137–40. [DOI] [PubMed] [Google Scholar]
- [11]. Ylinen K, Aula P, Stenman UH. Kesaniemi-Kuokkanen T and Teramo K: Risk of minor and major fetal malformations in diabetics with high haemoglobin A1c values in early pregnancy. Br Med J (Clin Res Ed) 1984;289:345–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12]. Pilania R, Sikka P, Rohit MK, et al. Fetal cardiodynamics by echocardiography in insulin dependent maternal diabetes and its correlation with pregnancy outcome. J Clin Diagn Res 2016;10:Qc01–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13]. Dong SZ, Zhu M. MR imaging of fetal cardiac malposition and congenital cardiovascular anomalies on the four-chamber view. Springerplus 2016;5:1214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14]. Steinhard J, Entenmann A, van der Valk E, et al. The transverse four-chamber view for the assessment of atrial tissue deformation in the fetus. PLoS One 2018;13:e0199581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15]. Buist NR, Tuerck J, Lis E, et al. Effects of untreated maternal phenylketonuria and hyperphenylalaninemia on the fetus. N Engl J Med 1984;311:52–3. [DOI] [PubMed] [Google Scholar]
- [16]. Lenke RR, Levy HL. Maternal phenylketonuria and hyperphenylalaninemia. An international survey of the outcome of untreated and treated pregnancies. N Engl J Med 1980;303:1202–8. [DOI] [PubMed] [Google Scholar]
- [17]. Platt LD, Koch R, Hanley WB, et al. The international study of pregnancy outcome in women with maternal phenylketonuria: report of a 12-year study. Am J Obstet Gynecol 2000;182:326–33. [DOI] [PubMed] [Google Scholar]
- [18]. Matalon KM, Acosta PB, Azen C. Role of nutrition in pregnancy with phenylketonuria and birth defects. Pediatrics 2003;112:1534–6. [PubMed] [Google Scholar]
- [19]. Chameides L, Truex RC, Vetter V, et al. Association of maternal systemic lupus erythematosus with congenital complete heart block. N Engl J Med 2003;297:1204–7. [DOI] [PubMed] [Google Scholar]
- [20]. Brucato A, Frassi M, Franceschini F, et al. Risk of congenital complete heart block in newborns of mothers with anti-Ro/SSA antibodies detected by counterimmunoelectrophoresis: a prospective study of 100 women. Arthritis Rheum 2001;44:1832–5. [DOI] [PubMed] [Google Scholar]
- [21]. Buyon JP, Hiebert R, Copel J, et al. Autoimmune-associated congenital heart block: demographics, mortality, morbidity and recurrence rates obtained from a national neonatal lupus registry. J Am Coll Cardiol 1998;31:1658–66. [DOI] [PubMed] [Google Scholar]
- [22]. Costedoat-Chalumeau N, Amoura Z, Lupoglazoff JM, et al. Outcome of pregnancies in patients with anti-SSA/Ro antibodies: a study of 165 pregnancies, with special focus on electrocardiographic variations in the children and comparison with a control group. Arthritis Rheum 2004;50:3187–94. [DOI] [PubMed] [Google Scholar]


