Abstract
Objectives:
Refugee children are known to have a high prevalence of elevated blood lead levels (EBLLs). We sought to determine trends in EBLLs among refugee children during an 18-year period and examine relationships between descriptive characteristics and EBLLs by using 10 µg/dL and 5 µg/dL levels of concern.
Methods:
We retrospectively evaluated refugee health screening data from Massachusetts for 1998 through 2015 for refugee children aged <7 years. We performed bivariate and multivariate analyses of variables including sex, age, region of origin, anemia, intestinal parasites, tuberculosis test results, and anthropometric measurements and used both 5 µg/dL and 10 µg/dL levels of concern for EBLLs.
Results:
Of 3421 eligible refugee children, 3054 (88.2%) were tested. Using 5 µg/dL and 10 µg/dL levels of concern, 1279 (41.9%) and 241 (7.9%) children, respectively, had EBLLs. Mean BLLs declined steadily from 7.58 µg/dL in 2004 to 4.03 µg/dL in 2015. African (adjusted odds ratio [aOR] = 2.49; 95% confidence interval [CI], 1.81-3.43), East Asian and Pacific (aOR = 1.98; 95% CI, 1.35-2.91), and South-Central Asian (aOR = 2.47; 95% CI, 1.53-4.01) regions of origin and anemia (aOR = 1.50; 95% CI, 1.14 -1.97) were significantly associated with BLLs ≥5 µg/dL.
Conclusions:
The prevalence of EBLLs among refugees compared with US-born children is high. Because EBBLs increase the risk for neurocognitive impairment in children, public health professionals, policy makers, researchers, refugee resettlement staff members, and health care providers must remain vigilant in screening for lead poisoning and educating refugees about the hazards posed to young children by lead.
Keywords: anthropometrics, lead poisoning, malnutrition, neurocognitive, refugee health
In 2016, the United States resettled 84 994 refugees, many of whom were fleeing strife overseas.1 These refugees have been resettled in almost every US state. Depending on their nation of origin, refugee populations can comprise >50% children.
The first major report to identify elevated blood lead levels (EBLLs) as a health concern for refugee children resettled in the United States was published in 2001.2 Subsequent reports have confirmed this health concern for refugee populations.3-8 For example, an investigation by the Centers for Disease Control and Prevention (CDC), published in 2004, determined that African refugee children with histories of malnourishment who were resettled in New Hampshire were more susceptible to environmental lead exposure than US-born children living in the same housing development.3 A 2011 report on Burmese refugee children resettled in Indiana showed a high prevalence of EBLLs that was associated with culturally traditional cosmetic use in the home.6 In 2011, a report from Massachusetts identified an association between EBLLs in refugee children and living in older housing (which presumably contained lead paint).7 A 2018 study of refugee children resettled in Kentucky also showed that EBLLs were associated with older housing, and that although blood lead levels (BLLs) were initially high, they did decline in some refugees after resettlement in the United States.8
Although CDC specifies that no amount of lead in the blood is safe or acceptable, historically it had defined EBLLs as being ≥10 µg/dL. Before 2012, however, research suggested that lower BLLs could cause irreversible harm to young children, particularly to their neurocognitive development.9-11 As a result, in 2012 CDC adopted the recommendations of its Advisory Committee for Childhood Lead Poisoning Prevention to lower the blood lead “level of concern” to 5 µg/dL.12
The primary objective of our study was to determine trends in BLLs among refugee children during an 18-year period. A secondary objective was to examine the relationships between the demographic, health, and nutritional characteristics of refugee children that we used in our 2001 publication1 and EBLLs using both the older 10 µg/dL and the newer 5 µg/dL levels of concern.
Methods
Study Population
For the purposes of this study, we used the term “refugee” as it is defined by US law (Immigration and Nationality Act, as amended by the Refugee Act of 1980).13 The term encompasses persons with a number of related humanitarian immigration statuses who are eligible for the benefits and services of the US Refugee Resettlement Program, including refugees, recipients of political asylum, Cuban and Haitian entrants, and others (eg, victims of trafficking and special immigrant visa holders from Iraq and Afghanistan). Some refugees, such as unaccompanied minors arriving at the southern US border, may not be eligible for health assessment and other support services because of their visa categorization. The Office of Refugee Resettlement recommends that all eligible refugees undergo health assessments within 90 days after arrival in the United States. During the years of the study, the content and rate of completion of refugee health assessments have varied widely from state to state. In Massachusetts, nearly all refugees have received health assessments, most of which are completed within 30 days of arrival.
We obtained the data for this study from a database of health information collected during these health assessments and maintained by the Massachusetts Department of Public Health (MDPH), Bureau of Infectious Disease and Laboratory Sciences, Division of Global Populations and Infectious Disease Prevention (unpublished data, MDPH). For this study, we included children who had arrived in Massachusetts from 1998 through 2015, were entitled to a refugee health assessment, and were aged <7 years at the time of their health assessment and blood tests. From that group of eligible children, we included only those who had had a blood lead test drawn within 90 days of arrival. We conducted the analyses and presentation of de-identified public health screening data according to the human studies regulations of the MDPH. The MDPH approved this article for public release.
Dependent Variable: BLLs
Massachusetts state regulations (105 CMR §460.070)14 require reporting of blood test results to MDPH. All specimens of children included in the study were collected as part of refugee medical screening at designated screening sites and tested at laboratories certified under the Clinical Laboratories Improvement Act for blood lead analyses. All blood lead specimens were obtained by venipuncture, which is less prone than capillary sampling to contamination from skin.15 Most samples were analyzed by graphite furnace absorption spectroscopy at the Massachusetts Public Health Laboratory. One site, which joined the Refugee Health Assessment Program network midway through the study period, used a Magellan Diagnostics’ LeadCare Testing Systems analyzer. The lower limit of detection for both techniques was 2 µg/dL. For this study, we defined EBLLs as both ≥5 µg/dL and ≥10 µg/dL.
Independent Variables: Clinical and Demographic Characteristics
We abstracted data from the MDPH refugee health database on the following variables: sex, age at time of testing, country and region of origin, blood hemoglobin level, pathogenic intestinal parasite test results (from stool microscopy for ova and parasites and from fluorescent antibody staining for giardia antigen in stool), tuberculosis test results (from either tuberculin skin test using purified protein derivative or interferon gamma release assay), and anthropometric measurements (height, weight). In abstracting data on age, we were aware that children for whom precise birth dates were not available at the time of processing were typically assigned a January 1 birthday.16 However, we were unable to determine a method to correct this date assignment. Consistent with US Department of State practice,17 we defined world regions as sub-Saharan Africa, East Asia and the Pacific, Europe and Eurasia (mainly countries of the former USSR and Yugoslavia), Near East (North Africa and Middle East), South and Central Asia, and Western Hemisphere.
We defined anemia according to CDC age-specific cut-off values for hemoglobin concentration. We considered children to be anemic if their hemoglobin values were <11.0 g/dL for children aged 0-23 months, <11.1 g/dL for children aged 24-60 months, and <11.5 g/dL for children aged >60 months.18 Heights had been measured as recommended by CDC: supine for children aged <2 years and upright for children aged ≥2 years. We used z scores (ie, number of standard deviations from the mean of the numerator for any given denominator, as determined from a similar reference population) for anthropometric categorization. We used weight-for-height z scores to identify acute undernutrition, with wasting defined as a z score <–2. We used height-for-age z scores to identify chronic undernutrition, with stunting defined as a z score <–2. We used weight-for-age z scores to identify being underweight, which was defined as having a z score <–2.
Data Analyses
We were unable to directly calculate mean BLLs because the lead analyzers used during the study period had a lower limit of detection of 2 µg/dL. We estimated mean BLLs for each year and overall, averaging the means that were calculated in 2 ways: (1) all BLLs ≤2 µg/dL were assigned a value of 0 µg/dL, and (2) all BLLs ≤2 µg/dL were assigned a value of 1.9.
We performed bivariate analyses on all variables to examine the relationships between the independent variables and the dependent variable, EBLL. We calculated crude odds ratios (ORs), and we used 95% confidence intervals (CIs) and P values based on Wald χ2 testing to determine significance. We then included the independent variables that yielded significant ORs (P < .05) in logistic regression models. We ran models separately for the 2 dependent variable outcomes (ie, BLLs ≥5 µg/dL and ≥10 µg/dL) and reported adjusted odds ratios (aORs) and 95% CIs.
We performed statistical analyses using SAS version 9.3.19 We determined anthropometric calculations using the SAS program for the 2000 CDC Growth Charts,20 and we based percentiles and z scores for weight and height on CDC growth charts.21
Results
During the 18-year period, of 3461 eligible children, 3054 (88.2%) had lead testing completed (Table 1). The largest region of origin of refugees was Africa (n = 1025, 33.6%), followed by Europe and Eurasia (n = 671, 22.0%) and the Near East (n = 617, 20.2%). The age distribution of refugees by annual increments was relatively uniform; however, the sample included fewer infants aged <1 year and children aged 6 years than other ages.
Table 1.
Descriptive characteristics of 3461 refugee children aged <7 years resettled in Massachusetts, by lead testing status, 1998-2015a
| Characteristics | Tested, No. (%), (n = 3054) | Not Tested, No. (%), (n = 407) |
|---|---|---|
| Sex | ||
| Female | 1489 (48.8) | 207 (50.9) |
| Male | 1565 (51.2) | 200 (49.1) |
| Region of origin | ||
| Sub-Saharan Africa | 1025 (33.6) | 122 (30.0) |
| East Asia and Pacific | 337 (11.0) | 54 (13.3) |
| Europe and Eurasia | 671 (22.0) | 153 (37.6) |
| Near East (North Africa and Middle East) | 617 (20.2) | 47 (11.5) |
| South and Central Asia | 342 (11.2) | 19 (4.7) |
| Western Hemisphere | 62 (2.0) | 12 (2.9) |
| Age, y | ||
| <1 | 248 (8.1) | 58 (14.3) |
| 1 | 496 (16.2) | 64 (15.7) |
| 2 | 517 (16.9) | 81 (19.9) |
| 3 | 540 (17.7) | 48 (11.8) |
| 4 | 523 (17.1) | 63 (15.5) |
| 5 | 512 (16.8) | 74 (18.2) |
| 6 | 218 (7.1) | 19 (4.7) |
| Anemiab | ||
| Yes | 670 (26.0) | 52 (23.2) |
| No | 1907 (74.0) | 172 (76.8) |
| Pathogenic intestinal parasitesc | ||
| Yes | 363 (19.9) | 43 (17.1) |
| No | 1464 (80.1) | 209 (82.9) |
| Positive tuberculosis test resultd | ||
| Yes | 296 (9.7) | 47 (11.5) |
| No | 2758 (90.3) | 360 (88.5) |
| Weight-for-height z scoree <−2 | ||
| Yes | 252 (8.3) | 23 (6.3) |
| No | 2802 (91.7) | 340 (93.7) |
| Height-for-age z scoref <−2 | ||
| Yes | 299 (9.8) | 35 (9.6) |
| No | 2755 (90.2) | 331 (90.4) |
| Weight-for-age z scoreg <−2 | ||
| Yes | 308 (10.1) | 25 (6.3) |
| No | 2746 (89.9) | 371 (93.7) |
a Data source: Unpublished data from the Massachusetts Department of Public Health Refugee Health Assessment Program database.
b Hemoglobin values <11.0 g/dL for children aged 0-23 months, <11.1 g/dL for children aged 24-60 months, and <11.5 g/dL for children aged >60 months. The denominator is the total number of children tested.
c Stool microscopy for ova and parasites and fluorescent antibody staining for giardia antigen in stool. The denominator is the total number of children tested.
d Tuberculin skin test performed by using purified protein derivative or interferon gamma release assay. The denominator is the total number of children tested.
eWeight-for-height z scores used to identify acute undernutrition, with wasting defined as a z score <–2. The denominator is the total number of children measured.
fHeight-for-age z scores used to identify chronic undernutrition, with stunting defined as a z score <–2. The denominator is the total number of children measured.
gWeight-for-age z scores used to identify weight status, with underweight defined as a z score <–2. The denominator is the total number of children measured.
Characteristics of tested children were similar to characteristics of nontested children (n = 407) for sex but not for world region of origin or age (Table 1). The largest region of origin of refugees among the nontested sample was Europe and Eurasia (n = 153, 37.6%), a higher proportion than in the tested sample. Similarly, 58 (14.3%) refugees in the nontested sample were aged <1 year, which was higher than in the tested sample (n = 248, 8.1%).
Compared with children from all other regions of origin, African children had the highest prevalence of EBLLs when using the ≥10 µg/dL level of concern (n = 132 of 1025, 12.9%), whereas children from South and Central Asia (n = 181 of 342, 52.9%) and East Asia and Pacific (n = 176 of 337, 52.2%) regions had the highest prevalence when using the ≥5 µg/dL level of concern (Table 2). The Near East region had the lowest prevalence using either level of concern (n = 22 of 617 [3.6%] for ≥10 µg/dL; n = 148 of 617 [24.0%] for ≥5 µg/dL).
Table 2.
Frequencya of elevated blood lead levels, using the ≥5 µg/dL and ≥10 µg/dL levels of concern, in 3054 tested refugee children aged >7 years resettled in Massachusetts, by most common countries of origin in each world region, 1998-2015b
| Region and Country | No. of Children Tested | Blood Lead Level ≥5 µg/dL, No. (%) | Blood Lead Level ≥10 µg/dL, No. (%) |
|---|---|---|---|
| Sub-Saharan Africa | |||
| Somalia | 484 | 220 (45.5) | 52 (10.7) |
| Democratic Republic of the Congo | 134 | 35 (26.1) | 5 (3.7) |
| Region total | 1025 | 491 (47.9) | 132 (12.9) |
| East Asia and Pacificc | |||
| Burma | 254 | 124 (48.8) | 12 (4.7) |
| Region total | 337 | 176 (52.2) | 20 (5.9) |
| Europe and Eurasia | |||
| Republic of Moldova | 22 | <5 (<22.7) | <5 (<22.7) |
| Ukraine | 184 | 67 (36.4) | <5 (<2.7) |
| Region total | 671 | 259 (38.6) | 36 (5.4) |
| Near East (North Africa and Middle East) | |||
| Iraq | 583 | 145 (24.9) | 21 (3.6) |
| Syria | 22 | <5 (<22.7) | <5 (<22.7) |
| Region total | 617 | 148 (24.0) | 22 (3.6) |
| South and Central Asia | |||
| Bhutan | 267 | 128 (47.9) | 11 (4.1) |
| Afghanistan | 64 | 47 (73.4) | 13 (20.3) |
| Region total | 342 | 181 (52.9) | 25 (7.3) |
| Western Hemisphere | |||
| Haiti | 28 | 14 (50.0) | <5 (<17.9) |
| Cuba | 27 | 9 (33.3) | <5 (<18.5) |
| Region total | 62 | 24 (38.7) | 6 (9.7) |
| Total | 3054 | 1279 (41.9) | 241 (7.9) |
a Exact numbers in cells with <5 children are suppressed, in accordance with data privacy rules of the Massachusetts Department of Public Health.
b Data source: Unpublished data from the Massachusetts Department of Public Health Refugee Health Assessment Program database.
c Single country reported because no other countries in this region reached the absolute number of refugee arrivals that would meet Massachusetts Department of Public Health criteria for public reporting.
Mean BLLs ranged from 3.55 µg/dL in 2013 to 7.58 µg/dL in 2004 (Table 3). The overall mean BLL for all tested refugees was 4.79 µg/dL.
Table 3.
Mean blood lead levels in 3054 tested refugee children aged <7 years resettled in Massachusetts, based on test with 2 µg/dL level of concern, and setting all blood lead levels of ≤2 µg/dL at 0 µg/dL and at 1.9 µg/dL, 1998-2015a
| Year | Total No. of Children | No. of Children Tested (%) | Blood Lead Level ≤2 µg/dL, No. (% of Tested) | Blood Lead Level With ≤2 µg/dL Set to 0 µg/dL, Mean (SD), µg/dL | Blood Lead Level With ≤2 µg/dL Set to 1.9 µg/dL, Mean (SD), µg/dL | True Mean, µg/dLb |
|---|---|---|---|---|---|---|
| 1998 | 237 | 182 (76.8) | 18 (9.9) | 5.38 (3.27) | 5.57 (2.99) | 5.48 |
| 1999 | 201 | 140 (69.7) | 23 (16.4) | 4.77 (3.25) | 5.08 (2.84) | 4.93 |
| 2000 | 194 | 165 (85.1) | 18 (10.9) | 6.42 (5.95) | 6.63 (5.75) | 6.53 |
| 2001 | 129 | 110 (85.3) | 17 (15.5) | 5.16 (3.73) | 5.46 (3.37) | 5.31 |
| 2002 | 78 | 68 (87.2) | 11 (16.2) | 5.15 (4.02) | 5.45 (3.66) | 5.30 |
| 2003 | 100 | 83 (83.0) | 8 (9.6) | 7.25 (6.79) | 7.44 (6.61) | 7.34 |
| 2004 | 249 | 201 (80.7) | 15 (7.5) | 7.51 (7.22) | 7.65 (7.09) | 7.58 |
| 2005 | 186 | 153 (82.3) | 31 (20.3) | 4.94 (4.04) | 5.33 (3.62) | 5.13 |
| 2006 | 87 | 73 (83.9) | 12 (16.4) | 4.92 (3.70) | 5.23 (3.33) | 5.07 |
| 2007 | 96 | 88 (91.7) | 12 (13.6) | 4.38 (3.32) | 4.63 (3.03) | 4.50 |
| 2008 | 178 | 157 (88.2) | 24 (15.3) | 4.56 (4.73) | 4.85 (4.49) | 4.71 |
| 2009 | 225 | 185 (82.2) | 49 (26.5) | 3.97 (3.95) | 4.48 (3.51) | 4.22 |
| 2010 | 222 | 210 (94.6) | 46 (21.9) | 4.13 (3.25) | 4.55 (2.78) | 4.34 |
| 2011 | 181 | 178 (98.3) | 33 (18.5) | 3.84 (2.48) | 4.19 (1.99) | 4.01 |
| 2012 | 277 | 269 (97.1) | 58 (21.6) | 3.87 (2.84) | 4.28 (2.34) | 4.07 |
| 2013 | 255 | 245 (96.1) | 62 (25.3) | 3.31 (2.60) | 3.79 (2.06) | 3.55 |
| 2014 | 335 | 328 (97.9) | 89 (27.1) | 3.54 (3.43) | 4.06 (2.97) | 3.80 |
| 2015 | 231 | 219 (94.8) | 53 (24.2) | 3.80 (3.66) | 4.26 (3.25) | 4.03 |
| Total | 3461 | 3054 (88.2) | 579 (19.0) | 4.61 (4.24) | 4.97 (3.90) | 4.79 |
a Data source: Unpublished data from the Massachusetts Department of Public Health Refugee Health Assessment Program database.
b It was not possible to directly calculate mean blood lead levels (BLLs) because the lead analyzers used during the study period had a lower 2 µg/dL level of concern. We estimated the mean BLLs for each year and overall by averaging the means that were calculated in 2 ways: (1) all BLLs ≤2 µg/dL were assigned a value of 0 µg/dL, and (2) all BLLs ≤2 µg/dL were assigned a value of 1.9 μg/dL.
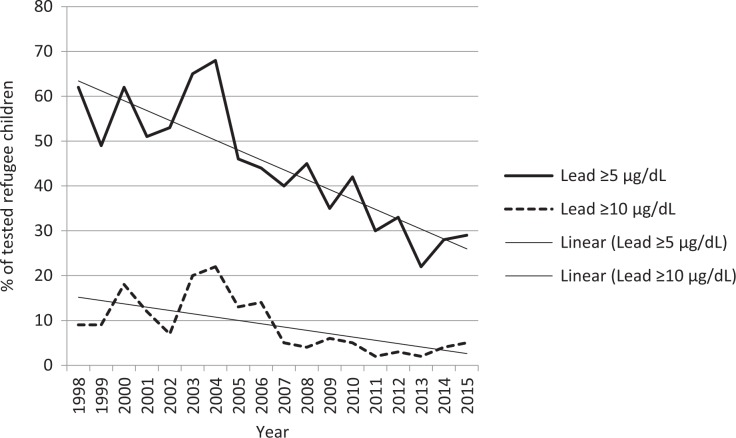
The prevalence of EBLLs in refugees peaked in 2004 at 22% (using the ≥10 µg/dL level of concern) and 68% (using the ≥5 µg/dL level of concern) (Figure). After 2004, prevalence declined steadily to 5% (using the ≥10 µg/dL level of concern) and 29% (using the ≥5 µg/dL level of concern) in 2015.
Figure.
Trends in proportion of 3054 tested refugee children aged <7 years resettled in Massachusetts with blood lead levels ≥5 µg/dL and ≥10 µg/dL, from the Massachusetts Department of Public Health Refugee Health Assessment Program database, 1998-2015. The linear best-fit trend line was calculated automatically by Excel.
Applying the ≥10 µg/dL and ≥5 µg/dL levels of concern for EBLLs to the 3054 children tested, we classified 241 (7.9%) and 1279 (41.9%) children, respectively, as having EBLLs (Table 4). Thus, of the 1279 children with EBLLs, 1038 (81.2%) had levels in the 5-10 µg/dL range.
Table 4.
Crude and adjusteda odds of having blood lead levels ≥5 µg/dL and ≥10 µg/dL in 3054 tested refugee children aged <7 years resettled in Massachusetts, 1998-2015b
| Characteristic | No. of Children Tested | Blood Lead Level ≥5 µg/dL | Blood Lead Level ≥10 µg/dL | ||||
|---|---|---|---|---|---|---|---|
| No (%) | Crude OR (95% CI) | Adjusted OR (95% CI)a,c | No. (%) | Crude OR (95% CI) | Adjusted OR (95% CI)a,c | ||
| Total | 3054 | 1279 (41.9) | — | — | 241 (7.9) | — | — |
| Sex | |||||||
| Female | 1489 | 593 (39.8) | 0.85 (0.73-0.98) | 0.90 (0.71-1.12) | 128 (8.6) | 1.21 (0.93-1.57) | NA |
| Male | 1565 | 686 (43.8) | 1 [Reference] | 1 [Reference] | 113 (7.2) | 1 [Reference] | 1 [Reference] |
| Region | |||||||
| Sub-Saharan Africa | 1025 | 491 (47.9) | 2.09 (1.67-2.62) | 2.49 (1.81-3.43) | 132 (12.9) | 3.63 (2.46-5.38) | 2.49 (1.42-4.35) |
| East Asia and Pacific | 337 | 176 (52.2) | 1.97 (1.45-2.69) | 1.98 (1.35-2.91) | 20 (5.9) | 1.46 (0.82-2.61) | 0.95 (0.45-2.01) |
| Europe and Eurasia | 671 | 259 (38.6) | 1 [Reference] | 1 [Reference] | 36 (5.4) | 1 [Reference] | 1 [Reference] |
| Near East (North Africa and Middle East) | 617 | 148 (24.0) | 0.74 (0.53-1.03) | 0.83 (0.55-1.24) | 22 (3.6) | 0.88 (0.43-1.81) | 0.57 (0.23-1.41) |
| South and Central Asia | 342 | 181 (52.9) | 2.04 (1.34-3.12) | 2.47 (1.53-4.01) | 25 (7.3) | 1.71 (0.82-3.56) | 1.52 (0.66-3.47) |
| Western Hemisphere | 62 | 24 (38.7) | 1.64 (0.83-3.26) | 2.09 (0.92-4.72) | 6 (9.7) | 3.33 (1.30-8.53) | 2.51 (0.77-8.22) |
| Age group, y | |||||||
| <1 | 248 | 74 (29.8) | 0.68 (0.50-0.93) | 0.35 (0.22-0.55) | 12 (4.8) | 0.74 (0.39-1.42) | 0.39 (0.18-0.87) |
| 1 | 496 | 235 (47.4) | 1.44 (1.14-1.81) | 1.24 (0.84-1.82) | 48 (9.7) | 1.56 (1.02-2.37) | 1.19 (0.68-2.09) |
| 2 | 517 | 221 (42.7) | 1.19 (0.95-1.50) | 0.95 (0.66-1.38) | 54 (10.4) | 1.70 (1.13-2.55) | 1.05 (0.61-1.82) |
| 3 | 540 | 239 (44.3) | 1.27 (1.01-1.59) | 1.29 (0.90-1.85) | 49 (9.1) | 1.45 (0.96-2.20) | 1.22 (0.70-2.10) |
| 4 | 523 | 229 (43.8) | 1.25 (0.99-1.56) | 1.09 (0.76-1.57) | 31 (5.9) | 0.92 (0.57-1.46) | 0.61 (0.33-1.14) |
| 5 or 6d | 730 | 281 (38.5) | 1 [Reference] | 1 [Reference] | 47 (6.4) | 1 [Reference] | 1 [Reference] |
| Anemiae | |||||||
| Yes | 670 | 331 (49.4) | 1.75 (1.47-2.10) | 1.50 (1.14-1.97) | 93 (13.9) | 2.88 (2.14-3.88) | 2.22 (1.50-3.28) |
| No | 1907 | 682 (35.8) | 1 [Reference] | 1 [Reference] | 101 (5.3) | 1 [Reference] | 1 [Reference] |
| Pathogenic intestinal parasitef | |||||||
| Yes | 363 | 171 (47.1) | 1.20 (0.95-1.51) | NA | 32 (8.8) | 1.09 (0.73-1.64) | NA |
| No | 1464 | 623 (42.6) | 1 [Reference] | NA | 119 (8.1) | 1 [Reference] | NA |
| Positive tuberculosis testg | |||||||
| Yes | 296 | 141 (47.6) | 1.30 (1.02-1.65) | 1.26 (0.87-1.83) | 32 (10.8) | 1.48 (1.00-2.19) | NA |
| No | 2758 | 1138 (41.3) | 1 [Reference] | 1 [Reference] | 209 (7.6) | 1 [Reference] | 1 [Reference] |
| Weight-for-height z scoreh <−2 | |||||||
| Yes | 252 | 103 (40.9) | 0.96 (0.74-1.24) | NA | 21 (8.3) | 1.07 (0.67-1.70) | NA |
| No | 2802 | 1176 (42.0) | 1 [Reference] | NA | 220 (7.9) | 1 [Reference] | NA |
| Height-for-age z scorei <−2 | |||||||
| Yes | 299 | 161 (53.8) | 1.71 (1.34-2.17) | 1.03 (0.71-1.50) | 42 (14.0) | 2.10 (1.47-3.00) | 1.01 (0.61-1.68) |
| No | 2755 | 1118 (40.6) | 1 [Reference] | 1 [Reference] | 199 (7.2) | 1 [Reference] | 1 [Reference] |
| Weight-for-age z scorej <−2 | |||||||
| Yes | 308 | 157 (51.0) | 1.50 (1.19-1.91) | 0.90 (0.61-1.33) | 39 (12.7) | 1.83 (1.27-2.63) | 0.98 (0.57-1.68) |
| No | 2746 | 1122 (40.9) | 1 [Reference] | 1 [Reference] | 202 (7.4) | 1 [Reference] | 1 [Reference] |
Abbreviations: NA, not applicable; OR, odds ratio.
a Adjusted for region of origin, age group, anemia, and low weight-for-age z scores and height-for-age z scores.
b Data source: Unpublished data from the Massachusetts Department of Public Health Refugee Health Assessment Program database.
cNA indicates where independent variables did not yield significant ORs (P < .05) on bivariate analysis and were not included in the multivariate logistic regression model.
d Results for children aged 5 or 6 years consolidated because of insufficient data for reporting results for 6-year-olds.
e Hemoglobin values <11.0 g/dL for children aged 0-23 months, <11.1 g/dL for children aged 24-60 months, and <11.5 g/dL for children aged >60 months.
f Stool microscopy for ova and parasites and fluorescent antibody staining for giardia antigen in stool.
g Tuberculin skin test performed by using purified protein derivative or interferon gamma release assay.
hWeight-for-height z scores used to identify acute undernutrition, with wasting defined as a z score <–2.
iHeight-for-age z scores used to identify chronic undernutrition, with stunting defined as a z score <–2.
jWeight-for-age z scores used to identify weight status, with underweight defined as a z score <–2.
Using the ≥10 µg/dL level of concern in bivariate analyses, refugee children from Africa were significantly more likely than children from Europe and Eurasia to have EBLLs (OR = 3.63; 95% CI, 2.46-5.38); children aged 1 year (OR = 1.56; 95% CI, 1.02-2.37) or 2 years (OR = 1.70; 95% CI, 1.13-2.55) were significantly more likely than children aged 5 or 6 years to have EBLLs; and children with anemia were significantly more likely than children without anemia to have EBLLs (OR = 2.88; 95% CI, 2.14-3.88) (Table 4).
Using the ≥5 µg/dL level of concern in bivariate analyses, compared with children from Europe and Eurasia, children from sub-Saharan Africa (OR = 2.09; 95% CI, 1.67-2.62), East Asia and Pacific (OR = 1.97; 95% CI, 1.45-2.69), and South and Central Asia (OR = 2.04; 95% CI, 1.34-3.12) regions were significantly more likely to have EBLLs (Table 4). Furthermore, refugee children with anemia (compared with children without anemia) (OR = 1.75; 95% CI, 1.47-2.10) and positive tuberculin skin test results (compared with children without positive tuberculin skin test results) (OR = 1.30; 95% CI, 1.02 -1.65) were significantly more likely to have EBLLs. Children with a low height-for-age were significantly more likely than children with normal height-for-age to have EBLLs (OR = 1.71; 95% CI, 1.34-2.17), and children with low weight-for-age were significantly more likely than children with normal weight-for-age to have EBLLs (OR = 1.50; 95% CI, 1.19 -1.91).
When using the ≥10 µg/dL level of concern in multivariate analysis, only children from sub-Saharan Africa (compared with children from Europe and Eurasia) (aOR = 2.49; 95% CI, 1.42-4.35) and children with anemia (compared with children without anemia) (aOR = 2.22; 95% CI, 1.50-3.28) were more likely to have EBLLs (Table 4).
When using the ≥5 µg/dL level of concern in multivariate analysis, some of the unadjusted associations held up. Compared with children from Europe and Eurasia, children from sub-Saharan Africa (aOR = 2.49; 95% CI, 1.81-3.43), East Asia and Pacific (aOR = 1.98; 95% CI, 1.35-2.91), and South and Central Asia (aOR = 2.47; 95% CI, 1.53-4.01) regions were significantly more likely to have EBLLs (Table 4). Children with anemia (aOR = 2.22; 95% CI, 1.50-3.28) were significantly more likely than children without anemia to have BLLs ≥10 µg/dL.
Discussion
In 2012, CDC adopted a change in the level of concern used to define EBLLs, lowering it from ≥10 µg/dL to ≥5 µg/dL.12 In this study, when we applied the current ≥5 µg/dL level of concern retrospectively to an 18-year cohort of refugee children aged <7 years, we found a high prevalence (42%) of EBLLs. Similarly, when we looked only at 2012 (the first year the new level of concern was used), we found a prevalence of EBLLs of 33% in our study population. This finding is especially concerning when considering that the prevalence of BLLs ≥5 µg/dL that year was 5.0% in the United States and 3.0% in Massachusetts.22
Elevated blood lead levels ≥10 µg/dL may cause a variety of physical symptoms.23 However, we found that most (81%) refugee children with EBLLs were in the 5-10 µg/dL range. Blood lead level elevations in this lower range typically lack overt physical symptoms, yet evidence supports an association between BLLs in this range and neurocognitive impairment.24 Before 2012, when CDC officially changed the level of concern used to define EBLLs to reflect this new research, children with levels in this range would not have been considered at risk of adverse clinical outcomes.
When we used ≥10 µg/dL to determine EBLLs and compared refugee children coming from Europe and Eurasia (predominantly from the former socialist countries of Eastern Europe), only children from sub-Saharan Africa had higher adjusted odds of having EBLLs. However, at the ≥5 µg/dL level of concern, refugee children from sub-Saharan Africa, East Asia and the Pacific, and South and Central Asia regions also had higher adjusted odds of having EBLLs. These findings highlight the broad environmental risks faced by refugee children from a multitude of countries of origin.
Similarly, when we used the ≥10 µg/dL level of concern, refugee children from sub-Saharan Africa had the highest prevalence of EBLLs. However, when we used the ≥5 µg/dL level of concern, refugee children from the Near East and South and Central Asia regions had the highest prevalence of EBLLs (>50%). These results are supported by a 2009 CDC investigation of US-bound Burmese refugees, which found a high risk of previous lead exposure in this population.4 These findings are concerning because in the past decade, increasing numbers of East Asians have arrived in the United States as refugees.25 The findings are also concerning because of conclusive research showing the negative neurocognitive effect on children of BLLs in the 5-10 µg/dL range.9-11
Although we found that region of origin could be a risk factor for EBLLs in many refugee children, anemia was also a significant risk factor. In the United States, CDC has identified other risk factors for EBLLs among children, including living in poverty, living in older housing, and having Medicaid health insurance.26 Refugee children typically have all 3 risk factors. However, the risk related to older housing may vary depending on the state in which the child lives. Another factor that may contribute to EBLLs in some refugee populations, as identified in a 2009 study from Indiana, is certain native cultural practices, such as the use of culturally traditional cosmetics and medications that contain lead.6 Finally, malnutrition, which is more prevalent among newly arrived refugee children than among US-born children, is also a risk factor for EBLLs.27
Not surprisingly, CDC identified refugee children as one of the groups in the United States at highest risk for lead poisoning and developed materials for preventing lead exposure in refugee communities.28 As part of these materials, CDC issued guidelines in 2006 for BLL testing in refugee children that emphasize that this testing be conducted shortly after arrival in the United States. These guidelines further recommend retesting the BLLs of refugee children aged 3-6 months after placement in permanent housing, regardless of initial results. Although refugee families are typically initially placed in lead-safe housing by sponsoring agencies, they may subsequently move to more affordable housing that may not be lead-safe.
Although our study did not find significant associations between various measures of undernutrition and EBLLs, CDC recommends that all refugee children have nutritional evaluations and receive appropriate nutritional and vitamin supplements.28 This recommendation is consistent with a study from New Hampshire that revealed that children with acute malnutrition as evidenced by low weight-for-height before arriving in the United States had greater increases in BLLs after resettlement than children without acute malnutrition.3,27 This study also demonstrated that a disproportionate number of refugee children had EBLLs compared with US-born children living in the same housing development, which had previously undergone lead abatement but was still not lead-free. CDC postulated that refugee children were more likely to develop EBLLs when exposed to environmental lead exposure because many arrive in the United States with deficiencies in nutritional elements, such as iron, calcium, and vitamin D.3
Although refugee children continue to arrive in the United States with a relatively high prevalence of EBLLs, we identified a steady improvement in BLLs among refugee children, particularly since 2004, and this decline mirrors a similar decline in the BLLs of US-born children.29 Also, the study demonstrated a 50% reduction, from 1998 to 2015, in the proportion of tested refugee children who had EBLLs, using either the ≥5 µg/dL or the ≥10 µg/dL level of concern. Nevertheless, the most recent year of data in this study showed that the prevalence of children with BLLs ≥5 µg/dL in 2015 (29%) was nearly 10 times that of children aged <6 years in the general US population in 2015 (3%).22
The reduction in the proportion of refugee children with EBLLs and in the mean BLLs in these children may be the result of several factors. A primary factor is the global cessation of the use of leaded gasoline, which, in the years leading up to our previous study in 2001, was still being widely used, particularly throughout Africa.30 In the United States, a more recent emphasis on having “healthy homes” includes a focus on lead abatement and removal in both the home and play environments.31 The ethics of conducting controlled studies of such interventions limit the ability to accrue evidence of their efficacy.32 However, it can still be argued that the dramatic decline in the prevalence of lead poisoning in US children is sufficient evidence for the fact that the broad public health approach taken to reduce the effect of this environmental hazard has been successful. Despite this success, efforts overseas and domestically are still warranted to target other known environmental lead hazards, such as informal mining operations,33 batteries,34 lead ammunition,35 and some traditional cosmetics and medications.36
In response to the higher prevalence of EBLLs in refugee children compared with US-born children, public health professionals and policy makers, researchers, refugee resettlement agency staff members, and health care providers still have work to do. For refugee families with young children, resettlement in lead-safe housing, education, and screening for lead poisoning risk factors in the home and care environments should be a priority. Given that refugee children are often exposed to lead before resettlement and continue to be exposed to lead through cultural and traditional practices, health care providers should be diligent about screening for EBLLs in this population, as recommended by CDC, even in parts of the country where the use of lead paint is less common. Finally, research into the refugee populations may offer insights into refugee children who come from similar world regions and, thus, may share risk factors for EBLLs.37,38
Limitations
This study had several limitations. First, one clinical site that provided lead testing for 27 refugee children reported using a LeadCare analyzer that was identified in a 2017 US Food and Drug Administration and CDC alert as having the potential to yield inaccurately low BLL test results when used with venous blood samples.39 Because the number of refugee children tested at this site amounted to <1% of the study sample, we opted not to redo the analyses. Including data from these children may have led to an underestimation of the true prevalence of EBLLs in our study. Second, our use of data from multiple laboratories and a lower limit of detection of 2 µg/dL may have affected the accuracy of mean BLLs that we calculated. However, we took additional steps to provide an accurate approximation of the true mean. Lastly, children for whom precise birth dates were not available at the time of their processing were assigned a January 1 birthday. This potential imprecision of dates of birth may have skewed the age distribution of EBLLs. However, only 1% of tested children and 0.5% of nontested children were assigned January 1 birthdays.
Conclusions
Almost 2 decades after lead poisoning was identified as a serious health condition for refugee children arriving in the United States, EBLLs are still a public health concern for this population. Our results suggest that although mean BLLs among refugee children have dropped since 2004, and the prevalence of EBLLs in this population has declined substantially since 1998, the prevalence of BLLs in the 5-10 µg/dL range is high. Although often lacking overt symptoms, children with these moderate EBLLs are at risk for developing neurocognitive impairment. Consequently, public health professionals and policy makers, researchers, refugee resettlement agency staff members, and health care providers must remain vigilant in screening for lead poisoning and educating refugees about the hazards posed to young children by lead.
Footnotes
Declaration of Conflicting Interests: The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding: The authors declared receipt of the following financial support for the research, authorship, and/or publication of this article: National Center for Emerging and Zoonotic Infectious Diseases (5U50CK000297).
ORCID iD: Paul L. Geltman, MD, MPH  https://orcid.org/0000-0002-9704-7782
https://orcid.org/0000-0002-9704-7782
References
- 1. US Department of State Bureau of Population, Refugees, and Migration Refugee Processing Center. Refugee admissions by region, fiscal year 1975 through May 31, 2019. http://www.wrapsnet.org/s/Graph-Refugee-Admissions-since-1975-6-5-19.xls. Accessed June 24, 2019.
- 2. Geltman PL, Brown MJ, Cochran J. Lead poisoning among refugee children resettled in Massachusetts, 1995 to 1999. Pediatrics. 2001;108(1):158–162. [DOI] [PubMed] [Google Scholar]
- 3. Plotinsky RN, Straetemans M, Wong LY, et al. Risk factors for elevated blood lead levels among African refugee children in New Hampshire, 2004. Environ Res. 2008;108(3):404–412. doi:10.1016/j.envres.2008.08.002 [DOI] [PubMed] [Google Scholar]
- 4. Trepka MJ, Pekovic V, Santana JC, Zhang G. Risk factors for lead poisoning among Cuban refugee children. Public Health Rep. 2005;120(2):179–185. doi:10.1177/003335490512000212 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Mitchell T, Jentes E, Ortega L, et al. Lead poisoning in United States-bound refugee children: Thailand-Burma border, 2009. Pediatrics. 2012;129(2):e392–e399. doi:10.1542/peds.2011-1218 [DOI] [PubMed] [Google Scholar]
- 6. Ritchey MD, Scalia Sucosky M, Jefferies T, et al. Lead poisoning among Burmese refugee children—Indiana, 2009. Clin Pediatr (Phila). 2011;50(7):648–656. doi:10.1177/0009922811398958 [DOI] [PubMed] [Google Scholar]
- 7. Eisenberg KW, van Wijngaarden E, Fisher SG, et al. Blood lead levels among refugee children resettled in Massachusetts, 2000 to 2007. Am J Public Health. 2011;101(1):48–54. doi:10.2015/AJPH.2009.184408 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kotey S, Carrico R, Wiemken TL, et al. Elevated blood lead levels by length of time from resettlement to health screening in Kentucky refugee children. Am J Public Health. 2018;108(2):270–276. doi:10.2105/AJPH.2017.304115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Bellinger DC, Needleman HL. Intellectual impairment and blood lead levels. N Engl J Med. 2003;349(5):500–502. doi:10.1056/NEJM200307313490515 [DOI] [PubMed] [Google Scholar]
- 10. Needleman HL, Schell A, Bellinger D, Leviton A, Allred EN. The long-term effects of exposure to low doses of lead in childhood: an 11-year follow-up report. N Engl J Med. 1990;322(2):83–88. doi:10.1056/NEJM199001113220203 [DOI] [PubMed] [Google Scholar]
- 11. Canfield RL, Henderson CR, Jr, Cory-Slechta DA, Cox C, Jusko TA, Lanphear BP. Intellectual impairment in children with blood lead concentrations below 10 microg per deciliter. N Engl J Med. 2003;348(16):1517–1526. doi:10.1056/NEJMoa022848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Centers for Disease Control and Prevention. CDC response to Advisory Committee on Childhood Lead Poisoning Prevention. Recommendations in “low level lead exposure harms children: a renewed call of primary prevention.” 2012. https://www.cdc.gov/nceh/lead/acclpp/cdc_response_lead_exposure_recs.pdf. Accessed July 20, 2016.
- 13. The Refugee Act. Pub L No 106-104 (1980).
- 14. Commonwealth of Massachusetts. Code of Massachusetts Regulations. Lead poisoning prevention and control. 2017. https://www.mass.gov/files/documents/2017/12/06/105cmr460.pdf. Accessed July 18, 2019.
- 15. Sargent JD, Dalton MA. Rethinking the threshold for an abnormal capillary blood lead screening test. Arch Pediatr Adolesc Med. 1996;150(10):1084–1088. [DOI] [PubMed] [Google Scholar]
- 16. Geltman PL, Radin M, Zhang Z, Cochran J, Meyers AF. Growth status and related medical conditions among refugee children in Massachusetts, 1995-1998. Am J Public Health. 2001;91(11):1800–1805. doi:10.2105/ajph.91.11.1800 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. US Department of State. Proposed refugee admissions for fiscal year 2019. https://www.state.gov/wp-content/uploads/2018/12/Proposed-Refugee-Admissions-for-Fiscal-Year-2019.pdf. Accessed June 24, 2019.
- 18. Centers for Disease Control and Prevention. Recommendations to prevent and control iron deficiency in the United States. MMWR Recomm Rep. 1998;47(RR-3):1–29. [PubMed] [Google Scholar]
- 19. SAS [statistical software]. Version 9.3. Cary, NC: SAS Institute, Inc; 2011. [Google Scholar]
- 20. Centers for Disease Control and Prevention. Growth chart training: a SAS program for the 2000 CDC growth charts (ages 0 to <20 years). Reviewed February 2019. http://www.cdc.gov/nccdphp/dnpao/growthcharts/resources/sas.htm. Accessed May 28, 2019.
- 21. Kuczmarski RJ, Ogden CL, Guo SS, et al. 2000 CDC growth charts for the United States: methods and development. Vital Health Stat 11. 2002;246:1–190. [PubMed] [Google Scholar]
- 22. Centers for Disease Control and Prevention. CDC’s national surveillance data (2012-2017). April 30, 2019. https://www.cdc.gov/nceh/lead/data/national.htm. Accessed June 26, 2017.
- 23. US Department of Health and Human Services, Agency for Toxic Substances and Disease Registry. Toxicological profile for lead: draft for public comment. 2019. https://www.atsdr.cdc.gov/toxprofiles/tp13.pdf. Accessed May 28, 2019.
- 24. Warniment C, Tsang K, Galazka SS. Lead poisoning in children. Am Fam Physician. 2010;81(6):751–757. [PubMed] [Google Scholar]
- 25. US Department of State Bureau of Population, Refugees, and Migration Refugee Processing Center. Refugee admissions by region, fiscal year 1975 through 30-Apr-2019. http://www.wrapsnet.org/s/Graph-Refugee-Admissions-since-1975-5-6-19.xls. Accessed June 24, 2019.
- 26. Wengrovitz AM, Brown MJ. Recommendations for blood lead screening of Medicaid-eligible children aged 1-5 years: an updated approach to targeting a group at high risk. MMWR Recomm Rep. 2009;58(RR-9):1–11. [PubMed] [Google Scholar]
- 27. Kellenberg J, DiPentima R, Maruyama M, et al. Elevated blood lead levels in refugee children—New Hampshire, 2003-2004 [published erratum appears in MMWR Morb Mortal Wkly Rep. 2005;54(3):76]. MMWR Morb Mortal Wkly Rep. 2005;54(2):42–46. [PubMed] [Google Scholar]
- 28. Centers for Disease Control and Prevention. Lead poisoning prevention in newly arrived refugee children: tool kit. 2006. www.cdc.gov/nceh/lead/Publications/RefugeeToolKit/Refugee_Tool_Kit.htm. Accessed March 12, 2018.
- 29. Raymond J, Wheeler W, Brown MJ. Lead screening and prevalence of blood lead levels in children aged 1-2 years—Child Blood Lead Surveillance System, United States, 2002-2010 and National Health and Nutrition Examination Survey, United States, 1999-2010. MMWR Morb Mortal Wkly Rep. 2014;63(2):36–42. [PubMed] [Google Scholar]
- 30. US Environmental Protection Agency, Office of Policy. Partnership for Clean Fuels and Vehicles: Evaluation of the Design and Implementation of the Lead Campaign: Final Report. Washington, DC: US Environmental Protection Agency; 2011. https://nepis.epa.gov/Exe/ZyPDF.cgi/P100EDFQ.PDF?Dockey=P100EDFQ.PDF. Accessed June 24, 2019. [Google Scholar]
- 31. Centers for Disease Control and Prevention, US Department of Housing and Urban Development Health Housing Reference Manual. Atlanta, GA: US Department of Health and Human Services; 2006. https://www.cdc.gov/nceh/publications/books/housing/housing_ref_manual_2012.pdf. Accessed June 25, 2019. [Google Scholar]
- 32. Nussbaumer-Streit B, Yeoh B, Griebler U, et al. Household interventions for preventing domestic lead exposure in children. Cochrane Database Syst Rev. 2016;10:CD006047 doi:10.1002/14651858.CD006047.pub5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Bashir M, Umar-Tsafe N, Getso K, et al. Assessment of blood lead levels among children aged ≤5—Zamfara State, Nigeria, June-July 2012. MMWR Morb Mortal Wkly Rep. 2014;63(15):325–327. [PMC free article] [PubMed] [Google Scholar]
- 34. Rivera García B, Rullán J, O’Neill M, et al. Take-home lead exposure among children with relatives employed at a battery recycling facility—Puerto Rico, 2011 [published erratum appears in MMWR Morb Mortal Wkly Rep. 2012;61(49):1012]. MMWR Morb Mortal Wkly Rep. 2012;61(47):967–970. [PubMed] [Google Scholar]
- 35. Bodeau-Livinec F, Glorennec P, Cot M, et al. Elevated blood lead levels in infants and mothers in Benin and potential sources of exposure. Int J Environ Res Public Health. 2016;13(3):e316 doi:10.3390/ijerph13030316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Nasidi A, Karwowski M, Woolf A, et al. Infant lead poisoning associated with use of tiro, an eye cosmetic from Nigeria—Boston, Massachusetts, 2011. MMWR Morb Mortal Wkly Rep. 2012;61(30):574–576. [PubMed] [Google Scholar]
- 37. Tehranifar P, Leighton J, Auchincloss AH, et al. Immigration and risk of childhood lead poisoning: findings from a case-control study of New York City children. Am J Public Health. 2008;98(1):92–97. doi:10.2105/AJPH.2006.093229 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Kaplowitz SA, Perlstadt H, Dziura JD, Post LA. Behavioral and environmental explanations of elevated blood lead levels in immigrant children and children of immigrants. J Immigr Minor Health. 2016;18(5):979–986. [DOI] [PubMed] [Google Scholar]
- 39. US Food and Drug Administration. FDA warns against using Magellan Diagnostics LeadCare Testing Systems with blood obtained from a vein: FDA safety communication. 2017. https://www.fda.gov/medical-devices/safety-communications/fda-warns-against-using-magellan-diagnostics-leadcare-testing-systems-blood-obtained-vein-fda-safety. Accessed May 28, 2019.