Abstract
Background: Recurrent keratoconus (RKC) develops as a progressive thinning of the peripheral and the inferior cornea after keratoplasty, in both graft and host, causing secondary astigmatism, refractive instability, and reduced visual acuity. We evaluated the effectiveness of corneal cross-linking (CXL) in patients diagnosed with RKC. Methods: Accelerated-CXL via the epi-off technique was performed in15 patients (18 eyes) diagnosed with RKC. Topographic and biomechanical changes were assessed at 12 months. Results: Differences in maximum keratometry, thinnest corneal thickness, and biomechanical parameters (deformation amplituderatio, inverse concave radius, applanation 1 velocity, and applanation 2 velocity, stiffness A1) versus baseline were statistically significant (p < 0.05).Best corrected visual acuity was improved in 13 eyes and unchanged in 4;manifest refractive spherical equivalent was reduced in 13 eyes, increased in 3,and unchanged in 1 eye; topographic astigmatism was reduced in 9 eyes, remained stable in 1 eye, and increased in 7 eyes. Conclusions: Improved topographic and biomechanic indexes at 1 year after CXL suggest it‘s potential as first-line therapy for RKC, as it is for KC.
Keywords: recurrent keratoconus, corneal cross-linking, corneal ectasia, corvis
1. Introduction
Recurrent keratoconus (RKC) develops as a progressive thinning of the peripheral and the inferior cornea after keratoplasty. Corneal thinning occurs in both the graft and the host cornea, resulting in high secondary astigmatism, refractive instability, and reduced visual acuity [1].It is one of the main complications of keratoplasty, with a mean occurrence at17.9 years (range 7–40) and an estimated prevalence of 7%at 20 years and 12% at 25 years [2,3].
Currently, there is no consensus on the topographic criteria for RKC: An increase in maximum keratometry (Kmax) combined with a reduction in best-corrected visual acuity (BCVA) [4], and an increase in both steepest keratometry and cylinder [5] have been proposed as diagnostic criteria.
The first step in the diagnosis of RKC is topography to detect the increased curvature and secondary astigmatism, and then tomography to evaluate thinning of the ectatic cornea. But these processes cannot valuate corneal stiffness or mechanical stability. For these reasons, and for gaining a better understanding of the development of corneal ectasia, instruments that measure corneal biomechanics in vivo have attracted increasing interest.
Non-surgical management of RKC relies on the use of personalized contact lenses [6], but tolerance to wearing them may be lost as the disease progresses [7]. Surgical treatment includes laser in situ keratomileusis, astigmatic keratotomy (AK), and wedge resections [8,9]. Repeating penetrating keratoplasty (PK) at an increased diameter could also be an option, although approaching the limbus increases the risk for iatrogenic limbal stem cell damage and graft rejection [10,11]. These approaches yield unsatisfactory results, however, because they act on the graft cornea, for refractive purposes, and not on the host cornea, which is the site of disease.
Riboflavin/ultraviolet A corneal cross-linking (CXL) of corneal collagen has proven effective in halting progression of ectasias and deterioration of visual acuity in patients with progressive KC and post-refractive corneal ectasia [12,13], but little is known about the effectiveness of CXL on RKC. Richoz et al. reported that corneal CXL in three patients (three eyes) with RKC halted the progression of ectasias up to one year and suggested it as an additional treatment option for such patients [4]. The aim of the present study was to evaluate the feasibility and effectiveness of accelerated CXL in patients with RKC and transparent graft.
2. Materials and Methods
We enrolled consecutive patients with a history of keratoplasty performed for keratoconus and diagnosed with RKC. The study was conducted from September 2017 to February 2019at the Eye Clinic of the University of Verona in accordance with the tenets of the Declaration of Helsinki and the principles of good clinical practice. No approval of Ethics Committee was required for retrospective studies with anonymized data. Written, informed consent was obtained from all patients. Data collection and management were carried out in compliance with Italian data privacy law (Legislative decree 196 of 30 August 2003).
2.1. Ophthalmic Examination
For the purposes of this study, all patients were followed up for at least 10 years and evaluated with topography every 6 months. Diagnosis of RCK by topography (Eye Top, Costruzione, Strumenti Oftalmici, Scandici/Florence, IT) showed an increase in Kmax of at least 1 diopter (D) in the last 12 months, and changes in visual acuity.
Follow-up visits were scheduled at 4 and 8 days and at 1, 2, 3, 6, and 12 months after the procedure. Examination included slit lamp biomicroscopy, corneal pachymetry (Galilei G4, Ziemer Ophtalmic Systems, Port, CH), topography (Eye Top), biomechanical assessment (non-contact tonometer Corvis® ST, Oculus Optikgeräte GmbH, Wetzlar, GE) and BCVA (CSO Vision Chart, CSO) under controlled lighting conditions at 4 meters using a decimal scale (Snellen chart).
2.2. CXL Procedure and Care
CXL epi-off technique was performed after topical anesthesia in sterile conditions. The corneal epithelium was removed over the keratoplasty flap (approximately 1mm) in the host cornea using a blunt spatula, and then a solution containing riboflavin (0.1% riboflavin) was instilled for 15 min. A total of 9.0 mm of cornea was exposed to UVA light (power 9 mW/cm2) for 10 min (Q. Light Stativ, Q. Products, CH) [14]. A hyposmotic 0.5% riboflavin solution was imbibed in eyes with a corneal thickness < 440 μm. At the end of the procedure, a therapeutic contact lens was applied to the cornea. Post-operative therapy comprised: Netilmicin 3 mg/mL + dexamethasone 1 mg/mL eye drop four times a day for 10 days (tapered by one drop per day every 15 days thereafter), ophthalmic solution based on sodium, hyaluronate, amino acids, and vitamin B2 six times per-day, and oral supplementation of amino acids for 3 months. The therapeutic contact lens was removed when re-epithelialization was complete.
2.3. Biomechanics
Corneal assessment with the Corvis®-ST allows non-contact imaging acquired via a high-speed camera of dynamic corneal response (DCR) to an air pulse, and acquisition of data on corneal stiffness and viscoelastic properties, standard tonometry, and central corneal thickness (CCT). In detail, during the deformation response, a precisely metered air pulse causes the cornea to move inward or to flatten (first applanation). The cornea continues to move inward until it reaches a point of highest concavity. Because of the cornea’s viscoelastic nature, it rebounds from this concavity to another point of applanation (second applanation), and then back to its normal convex curvature [15].
The DCR parameters evaluated in this study were: Speed of corneal apex at first and second applanation (Appl1 and Appl2 velocity), distance between the two bending peaks created in the cornea at the maximum concavity state (highest concavity peak distance, HCPD), inverse concave radius (1/R), that represents the radius of curvature at highest concavity, deformation amplitude ratio (DA Ratio) at 1 or 2 millimeters from the center of cornea, stiffness parameter A1 (SP-A1), and biomechanical intraocular pressure (bIOP) compensating for CCT, age, and biomechanics.
2.4. Statistical Analyses
Results of descriptive analyses are expressed as means ±standard deviation (SD), median and range for quantitative variables, and as a count and percentage for categorical variables. All parameters were evaluated as changes within individual eyes. The main outcome measures to assess the effects of CXL were topographic Kmax, pachymetric thinnest corneal thickness (TCT), and DCR. Statistical differences in values of Kmax and TCT were evaluated by Steel Multiple Comparison Wilcoxon test (R statistical package, version 3.5.1). Bonferroni correction was applied for multiple comparisons. A paired-sample Wilcoxon test was performed to compare baseline values of DCR to those ofa 1-year follow up (STATA 13.0 statistical package (Stata Corp. LP, College Station, TX, USA).
A p-value < 0.05 was considered statistically significant. The distribution of the outcome parameters are presented as box-whiskers plots in the style of Tukey.
3. Results
The study sample was 15 patients (18 eyes, four women and 11men, mean age 48.5 ± 9.2 years, range 34–61).Penetrating keratoplasty (PK) had been performed in 16 eyes and deep anterior lamellar keratoplasty (DALK) in two at 16.2 (5.5; 12–29) and 8.5 (12.1; 7–10)years before the diagnosis of RKC, respectively. A bilateral graft was present in three patients with previous PK.
The diameter of the donor graft was between 8.00 and 8.50 mm, and the diameter of host cornea between 7.75 and 8.25 mm. The residual stromal beds of the two eyes that had undergone DALK were approximately 100µm.All grafts were clear, and none of the patients reported ocular surface inflammation, corneal neovascularization or rejection episodes during the follow-up period after corneal transplantation. Five patients (six eyes) wore rigid gas permeable (RGP) contact lenses before enrollment in the study.
Complete closure of the corneal epithelium occurred within eight days after CXL. Corneal haze was noted up to the end of the first month, and complete recovery of corneal transparency by the end of the second month, after which combination corticosteroid and antibiotic eye drop therapy was suspended. Graft diastasis over 360° developed in one eye two days after the operation. The graft was sutured and the patient was withdrawn the study.
3.1. Topography and Pachymetry Results
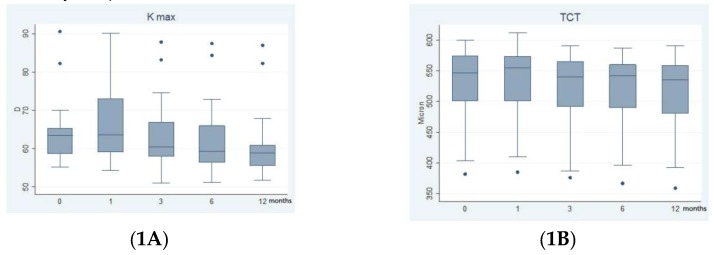
Figure 1A shows the results of CXL on maximum keratometry (Kmax). Kmax was 64.49 D (8.67; 55.14–90.56) before surgery, 66.23 D (11.00; 54.37–90.07) at 1 month, 63.54 D (9.62; 51.04–87.93) at three months, 62.90 D (9.71; 51.18–87.39) at six months, and 61.63 D (9.13; 51.81–86.94) at one year after surgery. The differences in Kmax before and after CXL were statistically not significant using the Steel Multiple Comparison Wilcoxon test: p = 0.1305. After applying the Bonferroni correction differences in Kmax before and after CXL were statistically not significant at one month (p = 1.00) and three months (p = 0.32), significant at six months (p = 0.04), and 12 months (p < 0.004).
Figure 1.
Effect of corneal cross-linking (CXL) on maximum keratometry (Kmax) and thinnest corneal thickness (TCT); (1A): Changes in Kmax were statistically significant at 6 months (p = 0.04) and 12 months (p = 0.004) versus baseline. (1B): Changes in TCT were statistically significant at 6 month (p = 0.04) and 12 months (p = 0.04) versus baseline.
Note: The boxes represent the 25th and 75th percentiles; the whiskers are the lines extending from each end of the box to the minimum or maximum or the lowest datum within 1.5-fold the inter quartile range (IQR) of the lower quartile or the highest datum within 1.5-fold IQR of the upper quartile. The median is the line that bisects the boxes and the circles are the outlier values. Follow-up time in months is shown on the x axis. D = diopters.
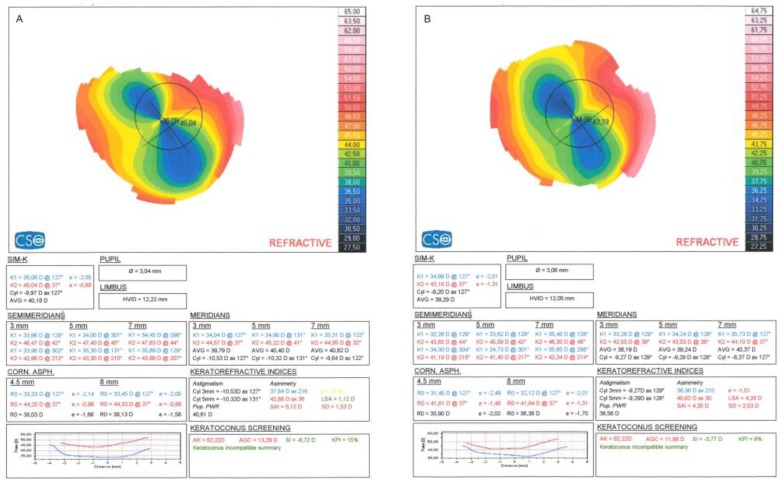
Figure 2 shows CXL topography results for a representative case in which Kmax was unchanged, whereas a reduction was noted in simulated keratometry dioptric mean of the steepest meridian (K1), simulated keratometry dioptric mean of the flattest meridian perpendicular to K1 (K2), topographic cylinder (Cyl), apical gradient curvature (AGC), average keratometry (AVG), and symmetry index (SI) at 1 year.
Figure 2.
Corneal topography results before surgery (A) and at 12 months (B).
Note: The ectatic area corresponds to the corneal periphery, i.e., to the graft-host junction, confirming our choice of CXL rather than keratoplasty (KP) to stabilize the host cornea. In this eye, the Kmax (AK, apical keratometry) remained stable at 12 months, whereas the other topographic indexes (K1; K2; topographic cylinder (Cyl); average keratometry (AVG); apical gradient curvature (AGC); SI; KPI, keratoconus prediction index) all improved.
TCT was observed in the host cornea in proximity to the graft-host junction in all eyes.Figure 1B shows the results of CXL on TCT.TCT before surgery was 534 μm (64.38; 382–600), 546 μm (64.15; 385–612) at one month, 529 μm (65.84; 376–590) at three months, 530.57 μm (64.04; 367–587) at 6months, and 518.20 μm (66.54; 393–590) at one year. The difference in TCT before and after CXL was statistically not significant using the Steel Multiple Comparison Wilcoxon test: p =0.3475. After applying the Bonferroni correction difference in TCT before and after CXL was statistically not significant at one month (p = 0.16) and three months (p = 0.08), statistically significant six months (p = 0.04), and one year (p = 0.04).
3.2. Biomechanical Results
Deformation amplitude (HCDA) was reduced from 1.16 (0.10; 1.01–1.36) mm before surgery to 1.12 (1.10; 0.98–1.32) mm at 12 months; Appl1 velocity was reduced from 0.14 (0.03; 0.1–0.24) m/s before surgery to 0.13 (0.03; 0.1–0.22) m/s at 12 months; Appl2 velocity was increased from −0.28 (0.05; −0.21–(−0.44)) m/s before surgery to −0.26 (0.05; −0.19–(−0.42)) at 12 months; highest concavity radius (HCR) was increased from 6.10 (0.87; 7.79–4.48) mm to 6.16 (0.87; 7.83–4.52) mm−1.
Stiffness A1 (SP-A1) was increased from 51.28 (7.54; 34.8–63.3) mmHg/mm to 52.04 (7.65; 35.4–63.6) mmHg/mm.
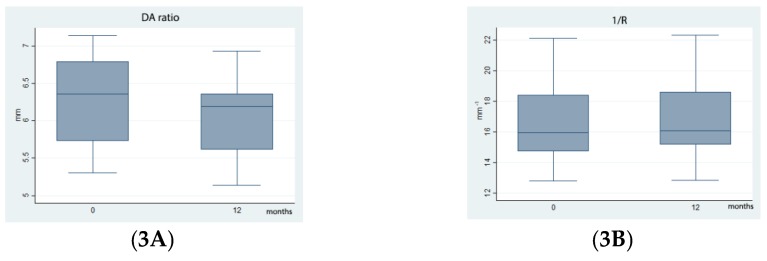
Statistically significant differences were noted for the distribution of DA Ratio, 1/R, SP-A1, Appl1 velocity, and Appl2 velocity before and after CXL (p < 0.001) (Figure 3), whereas no statistically significant differences were noted in Appl 1 length, Appl 2 length, and HCPD at one year.
Figure 3.
Biomechanical changes after CXL.
Note: A statistically significant difference was noted between baseline and post-surgical distribution of DA Ratio(3A), 1/R (3B), Appl 1 Velocity (3C),Appl 2 Velocity (3D) andSP-A1 (3E) (p < 0.05).The boxes represent the 25th and 75th percentiles; the whiskers are the lines extending from each end of the box to the minimum or maximum or the lowest datum within 1.5-fold the inter quartile range (IQR) of the lower quartile or the highest datum within 1.5-fold IQR of the upper quartile. The median value is the line that bisects the boxes and the circles are the outlier values.Follow-up time in months on the horizontal axis. mm = millimeters
bIOP and CCT, as measured with the Corvis® ST tonometer, remained unchanged from baseline to 12-month evaluation: 13.27 mm Hg (2.0; 10.5–16.8) and 13.12 mm Hg (1.73; 10.3–16.2), 529.76 microns (34.39; 408–650) and 527.88 microns (63.20; 398–597), respectively.
3.3. Refraction Results
A reduction in visual acuity due to de-epithelialization and haze during the first week after CXL was recorded for all patients. A progressive increase in BCVA was noted at 12months in 13 eyes, with an improvement of three Snellen lines in three eyes, two Snellen lines in four, and one Snellen line in six. BCVA was unchanged in four.
Table 1 presents the refraction data. At one year, topographic astigmatism was reduced in 9eyes, increased in seven, and remained unchanged in one eye; the manifest refraction spherical equivalent (MRSE) was reduced in 13 eyes, increased in three, and remained unchanged in one eye.
Table 1.
Topographic astigmatism and manifest refraction spherical equivalent (MRSE).
| Eye | Topographic Astigmatism before CXL | Topographic Astigmatism at 1 year | MRSE before CXL | MRSE at 1 year |
|---|---|---|---|---|
| 1 | 5.46 | 5.70 | −1.25 | −0.75 |
| 2 | 5.93 | 6.56 | −5.25 | −3.00 |
| 3 | 4.66 | 5.07 | −4.75 | −3.25 |
| 4 | 8.21 | 10.64 | −4.50 | −3.50 |
| 5 | 9.97 | 8.20 | +6.50 | +8 |
| 6 | 2.5 | 2.17 | −4.25 | −2.25 |
| 7 | 9.43 | 9.75 | −10.50 | −11.25 |
| 9 | 16.81 | 15.29 | −6.00 | −4.25 |
| 10 | 20.94 | 21.97 | −4.00 | −3.25 |
| 11 | 5.96 | 5.58 | −2.25 | −1.50 |
| 12 | 3.2 | 2.80 | +0.50 | +1.75 |
| 13 | 3.26 | 3.37 | −3.50 | −1.50 |
| 14 | 8.15 | 8.15 | −5.00 | −4.25 |
| 15 | 3.48 | 3.25 | −1.25 | −1.00 |
| 16 | 5.46 | 4.89 | −5.25 | −5.00 |
| 17 | 4.88 | 4.16 | +3.25 | +2.75 |
| 18 | 10.26 | 9.57 | −1.75 | −1.75 |
The topographic cylinder value before CXL was 7.47 D (4.67; 2.5–20.94), 7.66 D (4.60; 3.32–21.89) at 1 month, 7.42 D (4.68; 2.91–21.75) at 3 months, 7.38 D (5.00; 3.10–23.56) at 6 months, and 7.02 D (4.76; 2.80–21.97) at one year. Optical correction with glasses were prescribed in 11 eyes, while the use of RGP contact lenses was continued in six.
3.4. Safety Evaluation
No infectious events were recorded during the follow-up period. Diastasis of the donor button over 360° developed in one patienttwo days after the procedure; he subsequently underwent graft suture and was withdrawn the study.
4. Discussion
Patients with previous keratoplasty (KP) for keratoconus (KC) and later diagnosed with recurrent KC (RKC) are treated by re-transplantation or other surgical options performed on the corneal graft for refractive purposes. The effectiveness of CXL in KC is amply documented, but the literature is scant on CXL for RKC.
Rabinowitz suggested that RKC results from the persistence of the peripheral corneal ring after KP, or from the incomplete removal of peripheral ectasia during surgery or due to new onset from the diseased peripheral stroma. Based on this assumption, RKC can be considered a pancorneal disease [16]. The presence of corneal ectasia in both the central and the peripheral host cornea demonstrates that KC is a pancorneal disease and that KP, even at large diameter, may not be enough to completely remove the disease from the host eye.
In our study, preoperative topography showed that ectasia recurred in the peripheral host cornea and then involved the graft cornea, demonstrating that RKC has its onset from the residual stromal ring. Although CXL performed over an 8-mm zone can effectively stabilize the peripheral cornea in KC [17], the ectasia onset in RKC starts from the peripheral cornea. Richoz performed epi-off CXL in patients with RKC, applying irradiation at 1 mm within the limbus. In this study, CXL over a 9-mm zone, including both the graft and the host cornea strengthened and stabilized the whole cornea, as confirmed by the topographic and biomechanical findings. The reduction in Kmax indicated not only a halting but also a regression of the ectatic process, while the changes in DCR parameters were correlated with stiffening of the corneal structure, with a significant decrease in DA Ratio, 1/R and Appl1 velocity at one year after CXL, and a significant increase in Appl2 velocity and SP-A1 at one year after CXL.
The reduction in DA Ratio, in 1/R and the increase in SP-A1 we noted at 12 months demonstrate an improvement in corneal stress resistance and stiffness after CXL. Furthermore 1/R and DA Ratio are useful parameters of DCR by their demonstrated independence on intraocular pression [18].
Values of bIOP and CCT measured by Corvis resulted unchanged from baseline to 12 months. It should be noted that the bIOP equation was not developed for patients with KC, and especially for those who had undergone keratoplasty. Moreover the equation assumes that the cornea’s age and patient’s age coincide, however, this is not factual for the eyes included in this study.
We found that the biomechanical behavior (both at diagnosis and after CXL) of RKC was similar to KC [19]. However, the lack of studies on RKC, none of which to our knowledge have assessed biomechanical behavior, precludes comparison with previous studies.
We hypothesize that the reduction in TCT observed at one year stems from keratocyte apoptosis and compression and contraction of collagen fibers, as expected after CXL for KC. The literature reports no statistically significant reduction in TCT after accelerated CXL; however, these studies were conducted on KC and not on RKC [20,21].
5. Conclusions
An improvement in topographic and biomechanic indexes at one year after accelerated CXL for RKC was observed. These observations suggest CXL as first-line therapy for RKC, as it is for KC. Further studies including an appropriate control group and longer follow-up are desirable to determine whether CXL for RKC is effective to delay or obviate the need for KP.
Author Contributions
Each author contributed to the intellectual content of the manuscript: Conceptualization, E.P.; methodology, E.P.; A.F.; validation, E.P.; formal analysis, N.G.; investigation, G.C.; resources, E.P. and G.C..; data curation, G.C.; writing—original draft preparation, E.P.; G.C. and E.B.; writing—review and editing, E.P.; A.D.G. and A.F.; visualization, G.C.; supervision, E.P. and G.M.; project administration, E.P.
Funding
This study received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Ilari L., Daya S.M. Corneal wedge resection to treat progressive keratoconus in the host cornea after penetrating keratoplasty. J. Cataract Refract. Surg. 2003;29:395–401. doi: 10.1016/S0886-3350(02)01487-6. [DOI] [PubMed] [Google Scholar]
- 2.Pramanik S., Musch D.C., Sutphin J.E., Farjo A.A. Extended long-term outcomes of penetrating keratoplasty for keratoconus. Ophthalmology. 2006;113:1633–1638. doi: 10.1016/j.ophtha.2006.02.058. [DOI] [PubMed] [Google Scholar]
- 3.de Toledo J.A., de la Paz M.F., Barraquer R.I., Barraquer J. Long-term progression of astigmatism after penetrating keratoplasty for keratoconus: evidence of late recurrence Long-term progression of astigmatism after penetrating keratoplasty for keratoconus: evidence of late recurrence. Cornea. 2003;22:317–323. doi: 10.1097/00003226-200305000-00008. [DOI] [PubMed] [Google Scholar]
- 4.Richoz O., Schutz J.S., Pajic B., Coskunseven E., Hafezi F. Crosslinking for recurrent keratoconus. Ophthalmology. 2012;119:878. doi: 10.1016/j.ophtha.2011.11.007. [DOI] [PubMed] [Google Scholar]
- 5.Yoshida J., Murata H., Miyai T., Shirakawa R., Toyono T., Yamagami S., Usui T. Characteristics and risk factors of recurrent keratoconus over the long term after penetrating keratoplasty. Graefes Arch. Clin. Exp. Ophthalmol. 2018;256:2377–2383. doi: 10.1007/s00417-018-4131-5. [DOI] [PubMed] [Google Scholar]
- 6.Rico-Del-Viejo L., Garcia-Montero M., Hernández-Verdejo J.L., García-Lázaro S., Gómez-Sanz F.J., Lorente-Velázquez A. Nonsurgical Procedures for Keratoconus Management. J. Ophthalmol. 2017;2017:9707650. doi: 10.1155/2017/9707650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rathi V.M., Mandathara P.S., Dumpati S. Contact lens in keratoconus. Indian J. Ophthalmol. 2013;61:410–415. doi: 10.4103/0301-4738.116066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cohen K.L., Tripoli N.K., Noecker R.J. Prospective analysis of photokeratoscopy for arcuate keratotomy to reduce postkeratoplasty astigmatism. Refract. Corneal Surg. 1989;5:388–393. [PubMed] [Google Scholar]
- 9.Donnenfeld E.D., Kornstein H.S., Amin A., Speaker M.D., Seedor J.A., Sforza P.D., Landrio L.M., Perry H.D. Laser in situ keratomileusis for correction of myopia and astigmatism after penetrating keratoplasty. Ophthalmology. 1999;106:1966–1975. doi: 10.1016/S0161-6420(99)90410-4. [DOI] [PubMed] [Google Scholar]
- 10.Bersudsky V., Blum-Hareuveni T., Rehany U., Rumelt S. The profile of repeated corneal transplantation. Ophthalmology. 2001;108:461–469. doi: 10.1016/S0161-6420(00)00544-3. [DOI] [PubMed] [Google Scholar]
- 11.Holland E.J., Schwartz G.S. Iatrogenic limbal stem cell deficiency. Trans. Am. Ophthalmol. Soc. 1997;95:95–110. doi: 10.1007/0-387-21570-0_10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Razmjoo H., Peyman A., Rahimi A., Modrek H.J. Cornea Collagen Cross-linking for Keratoconus: A Comparison between Accelerated and Conventional Methods. Adv. Biomed. Res. 2017;6:10. doi: 10.4103/2277-9175.200785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Medeiros C.S., Giacomin N.T., Bueno R.L., Ghanem R.C., Moraes H.V., Jr., Santhiago M.R. Accelerated corneal collagen crosslinking: Technique, efficacy, safety, and applications. J. Cataract Refract. Surg. 2016;42:1826–1835. doi: 10.1016/j.jcrs.2016.11.028. [DOI] [PubMed] [Google Scholar]
- 14.Elbaz U., Shen C., Lichtinger A., Zauberman N.A., Goldich Y., Chan C.C., Slomovic A.R., Rootman D.S. Accelerated (9-mW/cm2) corneal collagen crosslinking for keratoconus-A 1-year follow-up. Cornea. 2014;33:369–773. doi: 10.1097/ICO.0000000000000154. [DOI] [PubMed] [Google Scholar]
- 15.Vinciguerra R., Ambrósio R., Elsheikh A., Roberts C.J., Lopes B., Morenghi E., Azzolini C., Vinciguerra P. Detection of Keratoconus With a New Biomechanical Index. J. Refract. Surg. 2016;32:803–810. doi: 10.3928/1081597X-20160629-01. [DOI] [PubMed] [Google Scholar]
- 16.Rabinowitz Y.S. Keratoconus. Surv. Ophthalmol. 1998;42:297–319. doi: 10.1016/S0039-6257(97)00119-7. [DOI] [PubMed] [Google Scholar]
- 17.Cagini C., Di Lascio G., Messina M., Riccitelli F., Dua H.S. Correlation of central and peripheral keratometric parameters after corneal collagen cross-linking in keratoconus patients. Int. Ophthalmol. 2019;39:2041–2048. doi: 10.1007/s10792-018-1041-9. [DOI] [PubMed] [Google Scholar]
- 18.Karimi A., Meimani N., Razaghi R., Rahmati S.M., Jadidi K., Rostami M. Biomechanics of the Healthy and Keratoconic Corneas: A Combination of the Clinical Data, Finite Element Analysis, and Artificial Neural Network. Curr. Pharm. Des. 2018;24:4474–4483. doi: 10.2174/1381612825666181224123939. [DOI] [PubMed] [Google Scholar]
- 19.Vinciguerra R., Elsheikh A., Roberts C.J., Kang D.S.Y., Lopes B.T., Morenghi E., Azzolini C., Vinciguerra P. Influence of Pachymetry and Intraocular Pressure on Dynamic Corneal Response Parameters in Healthy Patients. J. Refract. Surg. 2016;32:550–561. doi: 10.3928/1081597X-20160524-01. [DOI] [PubMed] [Google Scholar]
- 20.Shetty R., Pahuja N.K., Nuijts R.M., Ajani A., Jayadev C., Sharma C., Nagaraja H. Current Protocols of Corneal Collagen Cross-Linking: Visual, Refractive, and Tomographic Outcomes. Am. J. Ophthalmol. 2015;160:243–249. doi: 10.1016/j.ajo.2015.05.019. [DOI] [PubMed] [Google Scholar]
- 21.Vounotrypidis E., Athanasiou A., Kortüm K., Kook D., Shajari M., Priglinger S., Mayer W.J. Long-term database analysis of conventional and accelerated crosslinked keratoconic mid-European eyes. Graefes Arch. Clin. Exp. Ophthalmol. 2018;256:1165–1172. doi: 10.1007/s00417-018-3955-3. [DOI] [PubMed] [Google Scholar]