Abstract
Background
To assess the maternal characteristics and nutritional status according to body mass index (BMI) at 6–14 weeks of gestation and to examine the relationship between maternal nutritional status in early pregnancy and its impact on neonatal birth weight.
Methods
The investigation was conducted from April 2011 to June 2012 in Dhaka, Bangladesh. A total of 498 primigravida pregnant women participated in the study; women with known diabetes or previous gestational diabetes (GDM) were excluded. Maternal demographic details, pregnancy history and anthropometric measurements were obtained from the mother at the recruitment (6–14 weeks), 2nd visit between 24 and 28 week of gestation and 3rd visit at delivery. Cord venous blood samples of newborns (n = 138) were collected immediately after delivery for blood glucose, insulin, lipid profile, leptin and micronutrients including serum folate, ferritin, homocysteine, vitamin D, and vitamin B12.
Results
The prevalence at 6–14 weeks of pregnancy of anemia (Hb, < 11 g/dl), vitamin D deficiency (< 30 nmol/l), vitamin B12 deficiency (< 200 pg/ml), high homocysteine level (> 15 μmol/l), folate deficiency (< 3 ng/ml) and iron deficiency (ferritin < 13 ng/ml) were 19.5, 46.4, 15.1, 1.2, 0.4, and 12.7% respectively. GDM was found in 18.4% women. The prevalence of GDM was higher in overweight women (28.1%) than underweight (16.7%) and normal weight women (16.0%: p < 0.05). The incidence of low birth weight (LBW) and preterm delivery were 11.6 and 5.8% respectively and was not related to maternal BMI at 6–14 weeks of pregnancy. Maternal height was positively (p = 0.02), and homocysteine was negatively associated with neonatal birth weight (p = 0.02). In addition, the newborn’s cord serum folate was positively (p = 0.03) and cord triglyceride was negatively (p = 0.03) associated with neonatal birth weight.
Conclusion
Multiple maternal micronutrient deficiencies were present in early pregnancy. Maternal BMI in early pregnancy was not related to preterm deliveries or LBW. LBW was associated with lower folate, elevated cord triglyceride concentrations of the neonates and mother’s height and increase in maternal homocysteine levels. The data has important implications for pregnancy care in Bangladesh and other similar communities.
Keywords: Maternal nutrition, Early pregnancy, Cardio-metabolic status, Newborns at birth
Background
Maternal nutrition plays an important role in placental-fetal growth and development. Maternal undernutrition during pregnancy results in intrauterine growth restriction (IUGR) which is associated with increased perinatal morbidity and mortality. These children have an increased risk for development of metabolic syndrome in adult life [1, 2].
Evidence shows that the animal embryo comprises two group of cells in the early embryonic stage. The inner cell mass which develops as the fetus and the outer cell mass become the placenta. These developmental process is influenced by nutrition and hormones [3, 4]. Maternal undernutrition at the time of conception have shown fewer cells in the inner cell mass in experimental animal studies, which is associated with reduced birth weight and postnatal growth, altered organ/body weight ratios and the development of chronic diseases as type 2 diabetes (T2DM), hypertension (HTN), coronary artery disease (CAD) etc. [5] Evidence has also suggested that disturbances during critical periods of fetal development alter the structure or function of distinct cells, organ systems or homoeostatic pathways. Therefore, the subjects will have an increased risk of developing cardiovascular disease and T2DM in later life [6]. Studies in different time points have found the association between maternal malnutrition in early pregnancy and the occurrence of CAD in the offspring. They have shown that exposure to malnutrition, especially in late gestation, is linked to impaired glucose tolerance, while exposure in early gestation is linked to atherogenic lipid disorders and obesity [7–9].
Low birth weight (LBW) is considered an indicator of undernutrition in utero. Epidemiological studies have shown that it is associated with a wide range of adverse outcomes in later life, including shorter stature, lower cognitive performance, increased risk factors for later chronic non-communicable disease including, T2DM, HTN, CAD, chronic lung and kidney disease [10].
Even though, the obesity level is rising across the globe, yet more than 20 million infants (15.5% of all births) are born each year at LBW (< 2.5 kg). LBW remains a problem focused in developing countries, and half of all LBW babies are born in South and central Asia [11]. Although fetal growth and birth weight are known to be influenced by genetic factors, it is influenced most significantly by mothers’ health and nutritional status before and during pregnancy [12]. In economical transitioning countries, rapid changes in lifestyle and nutrition supply within a short time can create a “mismatch”. A study has shown that adults who had a coronary event had been small at birth and thin at 2 years of age and thereafter put on weight rapidly. This type of growth pattern is also related to insulin resistance in later life [13].
Data from Bangladesh in this issue is still scarce. As dietary patterns vary from country to country, especially regarding vegetarian and non-vegetarian components in Bangladesh and its neighboring country Indi. Data from India may not be directly applicable to people living in Bangladesh [14].
The purpose of the exploratory study was to examine the relationship between maternal nutritional status as assessed by body mass index (BMI) in early pregnancy and its impact on pregnancy outcomes and neonatal birth weight. In addition, we examined the relationship between neonatal birth weight with neonatal metabolic and micronutrients characteristics.
Methods
Study design and population
This is one of several sub studies of a prospective multi-center EU FP7 project GIFTS (Genomic and lifestyle predictors of fetal outcome relevant to diabetes and obesity and their relevance to prevention strategies in South Asian peoples) project conducted from April 2011 and June 2012 in Dhaka, Bangladesh. Other GIFTS studies included analysis of other populations in Pakistan [15] and Bangladeshi’s in the UK, a micronutrient intervention study in pregnancy in Dhaka (ISRCTN Number: 83599025), genomic predictors of birthweight and gestational diabetes (GDM) in South Asian populations, analysis of body composition in Bangladeshi children aged 2–4 in whose mothers were exposed to a community mobilization program [16], studies in GDM [17–19] and social and cultural influences of South Asian women during pregnancy [20].
Study participants were recruited from five areas (Azimpur, Lalbag, Hazaribag, Kamrangirchar and Keraniganj) in the Dhaka city, Bangladesh through the monthly household visit by Government Family Welfare Visitors (FWV). These areas are from both urban and semi-urban areas. During these visits, pregnant women were identified by a history of a missed menstrual cycle. Upon confirmation of pregnancy by urine pregnancy test women were invited to visit the Maternal and Child Health Training Institute (MCHTI), a tertiary Government hospital for antenatal care and registration.
A given error margin (α) of 5% with 95% confidence from a population of 100,000 and with a prevalence of undernutrition 30% [21], the required sample size is 322 (two-sided test).
Pregnant women were recruited in first trimester of their pregnancy (≤14-week gestation). Women who were primigravida, singleton, conceiving without treatment of fertility and were willing to participate in the study were included after written consent (n = 498). Women who had a history of T2DM, or history of GDM or pregnancy induced HTN were excluded from this study.
Date of last menstrual period and ultra-sonographic examination was performed at the time of recruitment to register the duration of gestation. Ultra-sonographic estimation was used as the criterion in case of disparity.
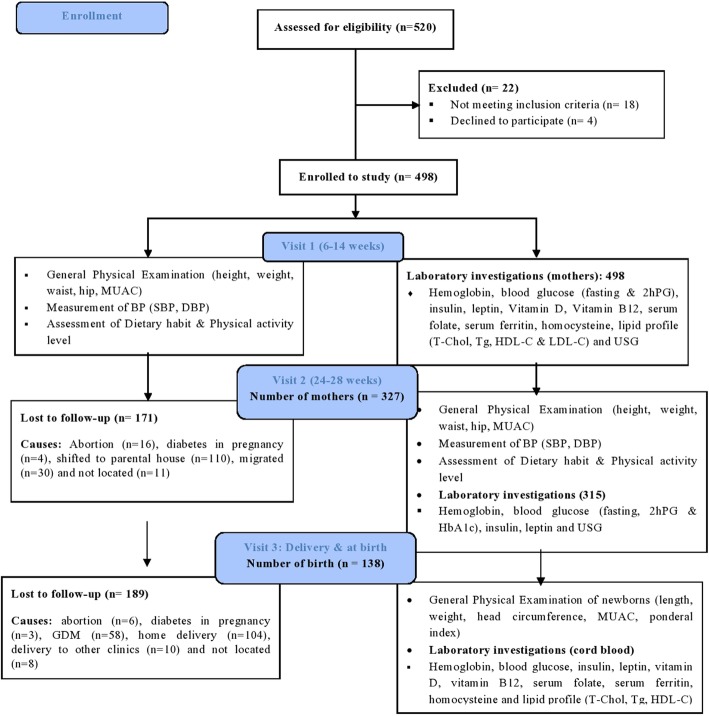
All records from each participant were kept during the study period (1st visit in first trimester at the time of registration (between 6 and 14 weeks, n = 498), 2nd visit between 24 and 28 week of gestation (n = 327) and 3rd visit at delivery (n = 138). Figure 1 is the study flow diagram detailing the dropouts at each stage of the process. Each study visit was performed after an overnight fast of 8–12 h.
Fig. 1.
Study flow diagram
GDM was diagnosed following a slight modification of International Association of Diabetes and Pregnancy Study Group (IADPSG) criteria in the 2nd trimester (24–28 weeks of gestation). Diagnosis was made by fasting and 2 hours after ingesting 75 g glucose. This was done to avoid high dropout rate because of 3 blood measures in pregnancy. GDM was defined as a fasting plasma glucose (FPG) ≥5.1 mmol/L or plasma glucose ≥8.5 mmol/L 2 hours after ingesting 75 g glucose (2hPG) [15]. Diabetes in pregnancy was defined as a FPG ≥7.0 mmol/L or 2hPG ≥11.1 mmol/L [22]. Women diagnosed with GDM and diabetes in pregnancy in visit 2 were not followed-up to birth as they gave birth in Bangladesh Institute of Research and Rehabilitation in Diabetes, Endocrine and Metabolic Disorders (BIRDEM 2), a specialist Hospital of Diabetic Association of Bangladesh.
Maternal data collection
Data were collected using questionnaire, physical examination and laboratory investigations. Four trained personnel were involved in data collection. A structured pre-formulated questionnaire was used to collect information on age, place of residence, education, type of occupation, socio-economic status (assessed by total family monthly expenditure), family history of chronic diseases including T2DM, HTN, CAD, and stroke, history of participants’ past illness or any chronic disease, history of addiction, physical activity level, nutritional status, and pregnancy-related history from the mother at the baseline visit. Maternal anthropometry including height, weight, waist, hip and mid upper arm circumference (MUAC) were measured (using standardized measurement protocols) at the time of registration and repeated at the second visit between 24 and 28 weeks’ gestation. BMI was calculated as weight divided by height squared (kg/m2). Waist circumference was measured by placing a plastic tape at the midpoint between the lower rib margin and the iliac crest. Similarly, hip was measured by taking the extreme end posteriorly and the symphysis pubis anteriorly. Mid arm circumference (MAC) of the left upper arm, measured at the mid-point between the tip of the shoulder and the tip of the elbow. In addition, blood pressure was measured in right hand in sitting position. Prior to the measurement, 10-min rest was assured and using standard cuffs fitted with sphygmomanometer minimized variation in measurement. Maternal food and nutrient intake were assessed at each trimester through Food Frequency Questionnaires (FFQs). The FFQ used in this study had a food list of 135 items. The FFQ consisted of frequency categories (daily, weekly and monthly). Local household utensils including glasses, spoons, cups and plates were used for estimating the amount of foods and beverages consumed by the respondents. These local utensils acted as visual aids to increase the accuracy of portion size estimations. Food Photo Manual was used, which converts food items of different sizes and composition to gram equivalents. Physical activity levels of participants at work, at home and during leisure time (minutes/day) was recorded following a structured questionnaire used in Bangladesh previously [23].
Maternal blood sample collection and storage
Blood samples were collected for hemoglobin, lipid profile, circulating serum folate, ferritin, homocysteine, vitamin D (25-Hydroxyvitamin D), and vitamin B12 at 1st visits and glucose (Fasting and 2-h after 75 g oral glucose), insulin and leptin at both visits (1st and 2nd). Serum samples were used for the estimation of serum glucose, lipid profile, insulin, leptin, vitamin D, vitamin B12, folate, ferritin, homocysteine and whole blood used for DNA.
Serum glucose was measured by glucose oxidase (Randox, Laboratories Ltd., UK), serum triglyceride (Tg) by GPO-PAP method (Randox Laboratories Ltd., UK), serum cholesterol (Chol) by enzymatic endpoint method (CHOD-PAP- Randox Laboratories Ltd., UK) and serum high density lipoprotein cholesterol (HDL-C) by differential precipitation, enzymatic colorimetric test (Linear Chemicals, Spain), and low density lipoprotein cholesterol (LDL-C) was estimated by Friedewald’s formula. All serum glucose and lipid profile were measured by using biochemistry auto analyzer (HITACHI 704 Japan), serum insulin, leptin, vitamin D were measured using ELISA kits (DRG Instruments GmbH, Germany). Serum folate, ferritin and vitamin B12 were measured using chemiluminescence immunoassay (Immulite-1000 / Siemens, USA). Serum total homocysteine was measured using Microparticle Enzyme Immunoassay (Abbott AxSYM/ produced by Axis-Shield, Oslo, Norway). Serum was not allowed to be thawed until the assay is performed at the Reference Laboratory in Bangladesh University of Health Sciences (BUHS), Dhaka, Bangladesh. Homeostasis model assessment of insulin resistance (HOMA-IR) was calculated using the method of Matthews et al [24] Intra assay and inter assay coefficient of variations for the measurement of insulin (5.5 and 4.9), B12 (3.1 and 3.74); folate (4.8 and 6.42), homocysteine (3.75 and 1.10) and Vit D (1.7 and 5) were in acceptable ranges.
Newborn data collection
Newborn characteristics including sex and gestational age at birth were recorded at the time of delivery (n = 138). Trained midwives examined all newborns within 24 h of birth. Newborn outcomes of interest included anthropometric characteristics (length, birth weight, head circumference, and mid-upper arm circumference (MUAC)). Newborns’ weight was measured with a digital scale (Model EN-20. MEA/CTN, China) and length was measured using a locally manufactured wooden length board. Head and mid upper arm were measured using a non-stretchable tape.
Umbilical venous cord blood samples of newborns were collected immediately following birth and analyzed for newborn’s serum glucose, insulin, lipids, vitamin D, vitamin B12, serum ferritin, serum folate, homocysteine and leptin. Methods and storage for the analysis of cord blood were same as used for the analysis of maternal blood.
Statistical analysis
Descriptive statistics in the form of frequencies (n) and percentages (%) were used to describe the maternal BMI at 6–14 weeks of pregnancy, which we categorized into underweight (BMI < 18.5 kg/m2), normal weight (BM 18.5–22.9 kg/m2) and overweight (BMI ≥23 kg/m2) [25]. Similar descriptive statistics were applied to neonatal birth weights, which we categorized as low (< 2500 g) and normal weight (≥2500 g) [26]. We used Analysis of variance (ANOVA) to compare differences of means between mothers with different maternal nutrition status and Independent T-test to compare differences in neonates’ birth weights. Pairwise comparisons between the groups were performed and corrected for multiple testing using Tukey’s HSD. Further, we have used the medians with interquartile range and other related non-parametric tests including Dunn test and Kruskal-Wallis test for skewed variables. Chi-square test was used to test for association between categorical variables. Both simple and multiple linear regression analyses were done for prediction of neonatal birth weight by selected maternal and neonatal parameters. The modeling process proceeded in two steps; first simple generalized linear regression models were fitted to the data in order to find independent variables with P < 0.20. In the second phase, the variables with P < 0.20 and the variables with P < 0.05 were then used to fit separate multiple generalized linear regression models. We also checked the correlation between different variables before starting multiple generalized linear regression models. Maternal BMI, LDL-C and HOMA-IR were not included due to their high correlation with maternal weight (r = 0.91, p < 0.001), total cholesterol (r = 0.80, p < 0.001) and insulin (r = 0.98, p < 0.001) respectively. At each stage of the model development, the Akaike Information Criteria (AIC) was used to check if the inclusion of a variable or variables improved the model fit [27]. The model with the smallest AIC value was considered as a better fit. All analyses were done using Stata SE 14 and IBM SPSS 24 and the significance level was two sided and set at α = 0.05.
Results
Table 1 showed the baseline characteristics of the pregnant women based on their BMI measured at their first visit at 10.1 ± 2.2 weeks gestation. Among the 498 women, 154 (30.9%) were underweight, 241 (48.4%) had normal weight and 103 (20.7%) were overweight. The mean age of the women at baseline were 20 ± 2.6 years. As expected, all the anthropometric indices (including weight, BMI, waist, waist-hip ratio, and MUAC), systolic and diastolic blood pressure were significantly lower in underweight women. Underweight mothers (compared with normal weight and overweight counterparts) consumed lower fat, whilst ingesting similar levels of carbohydrates and protein; furthermore, they were more physically active.
Table 1.
Baseline characteristics of pregnant women based on their BMI (kg/m2)
| Variables | Overall | Underweight (BMI < 18.5) |
Normal weight (BMI 18.5- < 23) |
Overweight (BMI ≥23) |
|---|---|---|---|---|
| Number, % | 498 | 154 (30.9) | 241 (48.4) | 103 (20.7) |
| Age (years) | 20.0 (2.6) | 19.4 (2.0) | 19.7 (2.4) | 21.5 (3.4) *† |
| Gestational age (weeks) | 10.1 (2.2) | 10.2 (2.2) | 10.1 (2.3) | 9.9 (2.3) |
| High SES (> 10,000 BDT), % | 160 (32.1) | 48 (31.2) | 81 (33.6) | 31 (30.1) |
| Weight (Kg) | 46.1 (8.5) | 38.5 (3.3) | 46.0 (4.6) * | 57.8 (7.7) *† |
| Height (cm) | 149.7 (5.4) | 149.7 (5.3) | 149.7 (5.5) | 149.6 (5.5) |
| BMI (kg/m2) | 20.6 (3.4) | 17.2 (0.99) | 20.5 (1.2) * | 25.8 (2.9) *† |
| Waist (cm) | 76.5 (9.2) | 68.8 (5.0) | 76.5 (5.9) * | 88.1 (8.4) *† |
| Hip (cm) | 90.5 (7.9) | 83.4 (4.9) | 90.7 (4.2) * | 100.8 (6.7) *† |
| WHR | 0.85 (0.13) | 0.83 (0.07) | 0.84 (0.05) | 0.90 (0.25) *† |
| MUAC (cm) | 23.8 (2.9) | 21.3 (1.4) | 24.0 (1.8) * | 27.2 (2.9) *† |
| Systolic BP (mmHg) | 92.8 (9.3) | 90.2 (8.5) | 92.9 (9.0) * | 96.4 (10.3) *† |
| Diastolic BP (mmHg) | 63.2 (7.2) | 61.4 (6.0) | 62.7 (6.9) | 67.0 (7.9) *† |
| Physical inactivity, % | 35 (7.0) | 3 (1.9) | 21 (8.7) * | 11 (10.7) * |
| Energy (Kcal/day) | 1489.7 (175.9) | 1473.2 (175.6) | 1487.2 (173.3) | 1520.9 (180.0) |
| Carbohydrate (gm) | 215.0 (35.1) | 213.7 (34.5) | 214.6 (34.0) | 217.6 (38.7) |
| Protein (gm) | 52.3 (13.8) | 52.8 (13.6) | 51.8 (13.8) | 53.0 (14.3) |
| Fat (gm) | 45.8 (9.4) | 44.4 (9.4) | 46.0 (9.2) | 47.8 (8.7) * |
Data are presented as mean (SD) or number (%)
*p-value < 0.05 compared with underweight; †p-value < 0.05 compared with normal. Abbreviation: BMI body mass index, SES socio-economic condition, BDT Bangladeshi Taka (1 UDS = 80 BDT), WHR waist-hip ratio, MUAC mid upper arm circumference, BP blood pressure
Table 2 showed the baseline biochemical parameters of the pregnant women based on their BMI. Significantly lower levels of fasting plasma glucose, 2 h plasma glucose and triglyceride at baseline were observed among underweight compared to overweight mothers (p < 0.05). In addition, insulin, HOMA-IR, cholesterol, LDL-C and leptin levels were significantly lower in underweight women than normal weight and overweight women. No significant difference was observed in vitamin D, vitamin B12, homocysteine, folate and ferritin levels between three groups. The prevalence of anemia (Hb, < 11 g/dl), vitamin D deficiency (< 30 nmol/l), vitamin B12 deficiency (< 200 pg/ml), high homocysteine level (> 15 μmol/l), folate deficiency (< 3 ng/ml) and iron deficiency (ferritin < 13 ng/ml) were 19.5, 46.4, 15.1, 1.2, 0.4, and 12.7% respectively. Anemia was significantly higher in underweight women than in women with normal weight (27.3 vs 15.4%, p = 0.004). GDM was found in 18.4% women (58 out of 315 at 24–28 weeks). The prevalence of GDM was significantly higher in overweight women (28.1%) than underweight (16.7%) and normal weight women (16.0%: p < 0.05). Only three women (0.95%) were diagnosed with diabetes in pregnancy.
Table 2.
Baseline biochemical parameters of pregnant women based on their BMI (kg/m2)
| Variables | Overall | Underweight (BMI < 18.5) |
Normal weight (BMI 18.5- < 23) |
Overweight (BMI ≥23) |
|---|---|---|---|---|
| Number, % | 498 | 154 (30.9) | 241 (48.4) | 103 (20.7) |
| Hb (g/dl) | 12.1 (11.3, 12.9) | 12.1 (10.9, 12.9) | 12.1 (11.3, 12.9) | 12.1 (11.3, 13.3) |
| Anemia (Hb < 11 g/dl), % | 97 (19.5) | 42 (27.3) | 37 (15.4) * | 18 (17.5) |
| FPG (mmol/l) | 4.7 (4.5, 5.0) | 4.7 (4.5, 4.9) | 4.7 (4.5, 5.1) | 4.8 (4.6, 5.2) *† |
| 2hPG (mmol/l) | 6.2 (5.3, 7.0) | 6.3 (5.4, 7.6) | 6.4 (5.4, 7.3) | 7.0 (6.1, 8.0) *† |
| GDM, % a | 58 (18.4) | 16 (16.7) | 26 (16.0) | 16 (28.1) *† |
| Insulin (μIU/ml) | 7.6 (5.4, 10.3) | 7.1 (5.0, 8.9) | 7.6 (5.4, 10.0) * | 9.7 (7.5, 15.2) *† |
| HOMA-IR | 1.6 (1.1, 2.2) | 1.5 (1.0, 1.9) | 1.6 (1.1, 2.2) * | 2.1 (1.6, 3.2) *† |
| Cholesterol (mg/dl) | 163.0 (150.0, 180.0) | 156.0 (150.0, 170.3) | 162.0 (150.0, 181.0) * | 172.0 (156, 200) *† |
| Triglycerides (mg/dl) | 102.5 (80.0, 132.8) | 91.5 (75.0, 124.5) | 100.5 (78.0, 136.8) | 122.0 (92, 147.0) *† |
| HDL-C (mg/dl) | 43.0 (38.0, 48.8) | 43.0 (38.8, 49.0) | 42.0 (37.0, 52.0) | 42.0 (36.0, 48.0) |
| LDL-C (mg/dl) | 97.0 (88.0, 112.0) | 94.0 (84.8, 104.0) | 99.0 (90.0, 114.0) * | 104.0 (92.0, 125.0) * |
| Vitamin D (nmol/l) | 30.2 (18.6, 41.8) | 30.5 (18.1, 42.5) | 31.2 (17.7, 43.8) | 32.8 (20.3, 47.2) |
| Deficiency (< 30 nmol/l), % | 231 (46.4) | 75 (48.7) | 112 (46.5) | 44 (42.7) |
| Vitamin B12 (pg/ml) | 296.5 (221.5, 389.8) | 295.0 (222.3, 432.8) | 304.0 (226.3, 406.5) | 295.0 (251.3, 370.5) |
| Deficiency (< 200 pg/ml), % | 76 (15.1) | 26 (16.9) | 40 (16.6) | 10 (9.7) |
| Homocysteine (μmol/l) | 7.0 (5.9, 8.8) | 7.0 (5.7, 8.8) | 7.0 (5.7, 8.6) | 6.8 (5.7, 8.1) |
| High (> 15 μmol/l), % | 6 (1.2) | 3 (1.9) | 3 (1.3) | – |
| Folate (ng/ml) | 10.5 (7.7, 18.5) | 11.3 (7.6, 18.7) | 10.3 (7.6, 20.1) | 9.8 (7.6, 14.3) |
| Deficiency (< 3 ng/ml), % | 2 (0.4) | – | – | 2 (1.9) |
| Ferritin (ng/ml) | 45.6 (23.3, 75.2) | 45.4 (25.9, 84.2) | 45.9 (22.6, 72.7) | 49.0 (28.7, 75.6) |
| Deficiency (< 13 ng/ml), % | 63 (12.7) | 16 (10.4) | 31 (12.9) | 16 (15.7) |
| Leptin (ng/ml) | 7.3 (4.6, 12.2) | 4.6 (3.4, 6.6) | 7.3 (5.4, 11.1) * | 16.2 (11.4, 23.2) *† |
Data are presented as median (interquartile range) or number (%). Biochemical assays were all measured in serum. a from 2nd visit
*p-value < 0.05 compared with underweight; †p-value < 0.05 compared with normal. Abbreviation: BMI body mass index, FPG fasting plasma glucose, 2hPG 2 h plasma glucose, GDM gestational diabetes, HOMA-IR homeostasis model assessment of insulin resistance, HDL-C high density lipoprotein cholesterol, LDL-C low density lipoprotein cholesterol
As seen in Fig. 138 of 498 mothers were followed-up until the birth of their child. Those mothers lost to follow up differed at their first visit (14–18 weeks) in that they had a lower weight (47.0 vs 45.2 kg; p < 0.01), reduced height (151 vs 149 cm; p < 001), increased 2 h glucose (6.2 vs 6.8 mmol/l; p < 001) and increased vitamin D levels (31 vs 35.3 pg/ml; p = 0.01).
Table 3 showed the demographic and biochemical characteristics of neonates by birth weight category. Among the 138 neonates, 16 (11.6%) had LBW (< 2500 g) and only 8 (5.8%) were preterm (< 37 weeks). Mean weight (grams), length (cm), weight for length (kg/m2), head circumference (cm), MUAC (cm) and ponderal index was 2814 ± 404, 45.9 ± 2.7, 13.4 ± 1.7, 33.1 ± 1.4, 9.8 ± 0.8 and 2.9 ± 0.5, respectively. LBW babies had significantly lower length, weight for height, head circumference, MUAC and ponderal index.
Table 3.
Demographic and biochemical characteristics of neonates at birth, categorized by birth weight
| Variables | Overall | Low birth weight (weight < 2500 g) |
Normal birth weight (weight ≥ 2500 g) |
|---|---|---|---|
| Number, % a | 138 | 16 (11.6) | 122 (88.4) |
| Sex (male), n, % a | 74 (53.6) | 5 (31.3) | 69 (56.6) |
| Preterm (< 37 weeks), n, % a | 8 (5.8) | 3 (18.8) | 5 (4.1) * |
| Delivery (caesarian section), n, % a | 90 (65.2) | 8 (50.0) | 82 (67.2) |
| Gestational age at delivery (weeks) b | 40.2 (1.6) | 40.4 (2.6) | 40.2 (1.4) |
| Weight (gram) b | 2814 (404) | 2146 (166) | 2899 (342) * |
| Length (cm) b | 45.9 (2.7) | 43.6 (2.8) | 46.3 (2.5) * |
| Weight for height (kg/m2) b | 13.4 (1.7) | 11.3 (1.0) | 13.6 (1.6) * |
| Head circumference (cm) b | 33.1 (1.4) | 31.4 (1.4) | 33.3 (1.3) * |
| MUAC (cm) b | 9.8 (0.8) | 9.1 (0.7) | 9.9 (0.8) * |
| Ponderal index b | 2.9 (0.5) | 2.6 (0.4) | 3.0 (0.5) * |
| Biochemical parameters | |||
| Blood glucose (mmol/l) c | 3.8 (3.3, 5.1) | 4.3 (3.2, 5.1) | 3.8 (3.3, 5.1) |
| Insulin (μIU/ml) c | 6.5 (5.0, 11.5) | 5.6 (3.5, 15.1) | 6.6 (4.6, 11.4) |
| HOMA-IR c | 1.18 (0.74, 2.30) | 1.23 (0.56, 2.85) | 1.18 (0.74, 2.27) |
| Glucose to insulin ratio c | 10.6 (7.1, 15.6) | 10.9 (6.1, 17.9) | 10.5 (7.4, 15.2) |
| Cholesterol (mg/dl) c | 66.0 (56.0, 78.0) | 64.0 (47.0, 77.0) | 66.0 (56.3, 78.8) |
| Triglycerides (mg/dl) c | 35.0 (26.0, 50.0) | 48.0 (28.0, 76.0) | 34.5 (25.3, 48.0) |
| HDL-C (mg/dl) c | 23.0 (19.0, 30.0) | 19.0 (16.0, 29.0) | 24.0 (20.0, 30.0) |
| LDL-C (mg/dl) c | 33.0 (25.0, 41.0) | 32.0 (17.0, 39.0) | 33.0 (25.0, 41.0) |
| Vitamin D (nmol/l) c | 33.9 (28.3, 45.2) | 31.2 (27.8, 45.3) | 34.5 (28.7, 45.2) |
| Vitamin B12 (pg/ml) c | 417.0 (329.3, 640.3) | 414.0 (258.5, 669.0) | 421.0 (340.0, 629.5) |
| Homocysteine (μmol/l) c | 5.0 (4.0, 6.4) | 5.5 (4.5, 6.9) | 5.0 (4.0, 6.3) |
| Folate (ng/ml) c | 16.8 (14.2, 21.8) | 13.7 (12.3, 20.3) | 16.8 (14.4, 22.2) |
| Ferritin (ng/ml) c | 119.0 (58.8, 197.0) | 112.0 (61.1, 155.0) | 127.0 (59.3, 200.0) |
| Leptin (ng/ml) c | 5.1 (3.0, 7.8) | 5.3 (2.9, 7.1) | 5.1 (3.0, 8.1) |
Data are presented as a number (%) or b mean (SD) or c median (interquartile range) where appropriate. Biochemical assays were all measured in serum. *p-value < 0.05 compared with LBW. Abbreviation: LBW low birth weight, MUAC mid upper arm circumference, HOMA-IR homeostasis model assessment of insulin resistance, HDL-C high density lipoprotein cholesterol, LDL-C low density lipoprotein cholesterol
No significant difference was observed for any of the following parameters (blood glucose, insulin, HOMA-IR, glucose to insulin ratio, cholesterol, triglyceride, HDL-C, LDL-C, vitamin D, vitamin B12, folate, ferritin, homocysteine and leptin birth weight levels) in LBW babies compared to normal weight infants.
Table 4 showed the anthropometric and laboratory parameters of neonates categorized by their mothers’ BMI. No significant difference for anthropometric indices (birth weight, birth length, head circumference, and MUAC) was observed among neonates born to underweight mothers at inclusion than normal weight mothers. Significant higher cord blood glucose (4.3 vs 3.7 mmol/L), blood cholesterol (67.0 vs 60.5 mg/dl), HDL-C (25.0 vs 21.5 mg/dl) and lower vitamin B12 (372.0 vs 514.5 pg/ml) values were observed in neonates born to underweight mothers than overweight/obese mothers.
Table 4.
Anthropometric and laboratory parameters of neonates at birth, categorized by maternal BMI (kg/m2)
| Underweight (BMI < 18.5) |
Normal weight (BMI 18.5- < 23) |
Overweight (BMI ≥23) |
|
|---|---|---|---|
| Total number (n = 138) | 38 | 67 | 33 |
| Anthropometric indices | |||
| Birth weight (gram)a | 2700 (400) | 2800 (400) | 2900 (400) |
| Birth length (cm) a | 45.4 (3.1) | 46.3 (2.6) | 45.9 (2.3) |
| Head circumference (cm) a | 32.9 (1.3) | 33.0 (1.5) | 33.4 (2.7) |
| MUAC (cm) a | 9.7 (0.8) | 9.8 (0.8) | 9.8 (0.9) |
| Laboratory parameters | |||
| Blood glucose (mmol/l) b | 4.3 (3.4, 5.2) | 3.7 (3.3, 5.1) | 3.7 (3.2, 4.2) * |
| Insulin (μIU/ml) b | 6.0 (4.3, 13.5) | 7.3 (3.2, 12.1) | 6.1 (4.6, 9.4) |
| HOMA-IR b | 1.1 (0.7, 2.7) | 1.4 (0.8, 2.6) | 1.1 (0.7, 1.5) |
| Glucose to insulin ratio b | 11.5 (8.3, 16.0) | 10.0 (6.5, 16.1) | 10.3 (7.2, 15.1) |
| Cholesterol (mg/dl) b | 67.0 (58.5, 85.0) | 67.5 (56.3, 75.8) | 60.5 (50.8, 66.3) * |
| Triglycerides (mg/dl) b | 40.0 (28.5, 48.5) | 35.5 (24.0, 52.0) | 34.0 (28.0, 53.0) |
| HDL-C (mg/dl) b | 25.0 (20.0, 34.0) | 23.0 (19.3, 31.0) | 21.5 (17.5, 27.0) * |
| LDL-C (mg/dl) b | 36.0 (25.0, 43.0) | 35.5 (26.5, 41.8) | 30.0 (24.8, 35.0) |
| Vitamin D (nmol/l) b | 31.7 (27.1, 42.7) | 33.6 (28.4, 43.7) | 38.2 (31.5, 46.6) |
| Vitamin B12 (pg/ml) b | 372.0 (297.3, 496.8) | 424.5 (297.0, 641.5) | 514.5 (369.0, 751.0) *† |
| Homocysteine (μmol/l) b | 5.4 (4.6, 7.2) | 5.0 (4.0, 6.0) | 4.7 (3.6, 6.4) |
| Folate (ng/ml) b | 15.8 (13.2, 24.8) | 17.3 (13.8, 22.4) | 16.5 (14.5, 18.7) |
| Ferritin (ng/ml) b | 103.8 (61.5, 253.8) | 142.5 (64.4, 203.8) | 81.6 (43.5, 161.0) |
| Leptin (ng/ml) b | 4.3 (2.7, 8.1) | 5.1 (2.9, 8.8) | 5.7 (4.0, 7.1) |
Data are presented as a mean (SD) or b median (interquartile range)
*p-value < 0.05 compared with underweight; †p-value < 0.05 compared with normal. Abbreviation: BMI body mass index, MUAC mid upper arm circumference, HOMA-IR homeostasis model assessment of insulin resistance, HDL-C high density lipoprotein cholesterol, LDL-C low density lipoprotein cholesterol
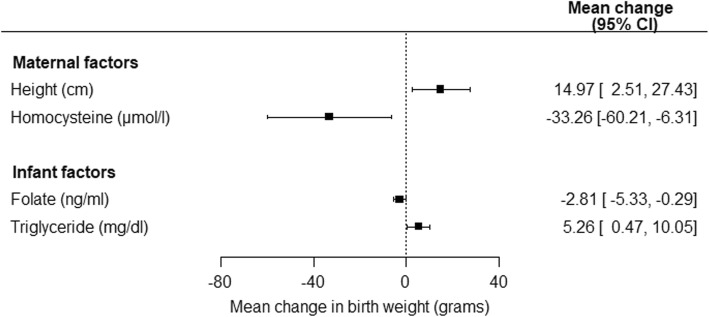
Table 5 showed the associations between neonatal birth weight and maternal characteristics in early pregnancy. Maternal height and weight at baseline were positively associated with neonatal birth weight in the univariate analysis. In the adjusted analysis (model 2), maternal height was positively associated with neonatal birth weight (p = 0.02) while homocysteine was negatively associated with neonatal birth weight (p = 0.02). Model 2 showed that a unit increase in homocysteine was associated with a 33.26 g decrease in mean birth weight (p = 0.02).
Table 5.
Generalized linear regression analysis showing the associations between neonatal birth weight and maternal measures. Using the Akaike information criteria (AIC), model 2 was selected because it has the smallest AIC value
| Maternal variables | Univariate (unadjusted) |
1Full model (n = 138) AIC = 2071.32 |
2Model 2 (n = 138) AIC = 2053.40 |
3Model 3 (n = 138) AIC = 2066.53 |
||||
|---|---|---|---|---|---|---|---|---|
| β (95% CI) | P-value | β (95% CI) | P-value | β (95% CI) | P-value | β (95% CI) | P-value | |
| Height (cm) | 17.42 (5.82, 29.01) | < 0.01 | 20.32 (5.65, 34.99) | 0.01 | 14.97 (2.51, 27.43) | 0.02 | 13.1 (0.76, 25.42) | 0.04 |
| Weight (kg) | 11.15 (3.20, 19.09) | 0.01 | 3.80 (−6.84, 14.44) | 0.48 | 6.86 (−1.62, 15.35) | 0.11 | 7.75 (−0.67, 16.16) | 0.07 |
| Homocysteine (μmol/l) | −26.67 (−54.78, 1.44) | 0.06 | −32.99 (− 62.03, −3.95) | 0.03 | −33.26 (− 60.21, − 6.31) | 0.02 | ||
| 2hPG (mmol/l) | 44.93 (− 15.76, 105.62) | 0.15 | 51.54 (− 11.97, 115.06) | 0.11 | 51.31 (−8.00, 110.63) | 0.09 | ||
| Triglycerides (mg/dl) | −0.93 (−2.21, 0.34) | 0.15 | −0.96 (−2.48, 0.56) | 0.22 | −1.09 (− 2.31, 0.14) | 0.08 | ||
| Hemoglobin | −24.88 (− 80.13, 30.38) | 0.38 | −5.75 (−68.68, 57.18) | 0.86 | ||||
| FPG (mmol/l) | 70.77 (−70.28, 211.82) | 0.32 | 46.92 (−97.80, 191.65) | 0.53 | ||||
| Insulin (μIU/ml) | 7.38 (−6.76, 21.51) | 0.30 | 11.07 (−4.59, 26.74) | 0.17 | ||||
| Cholesterol (mg/dl) | −0.43 (−3.30, 2.44) | 0.77 | −0.94 (− 4.36, 2.48) | 0.59 | ||||
| HDL-C (mg/dl) | 5.59 (−4.88, 16.06) | 0.29 | 6.01 (−5.43, 17.44) | 0.30 | ||||
| Vitamin D (nmol/l) | −0.96 (− 5.27, 3.35) | 0.66 | −0.45 (− 5.01, 4.11) | 0.85 | ||||
| Vitamin B12 (pg/ml) | 0.10 (−0.40, 0.60) | 0.69 | −0.07 (− 0.60, 0.46) | 0.80 | ||||
| Ferritin (ng/ml) | 0.56 (−0.98, 2.11) | 0.47 | 0.45 (−1.15, 2.04) | 0.58 | ||||
| Folate (ng/ml) | −2.72 (−10.88, 5.43) | 0.51 | −0.51 (−9.00, 7.97) | 0.91 | ||||
| Age (years) | 12.69 (−10.50, 35.88) | 0.28 | −6.08 (−32.70, 20.55) | 0.66 | ||||
| Gestational age in weeks | −11.83 (−55.64, 31.98) | 0.60 | −24.20 (−70.14, 21.74) | 0.30 | ||||
| BMI (kg/m2) | 16.28 (−3.01, 35.58) | 0.10 | ||||||
| LDL-C (mg/dl) | −0.27 (−3.29, 2.75) | 0.86 | ||||||
| HOMA-IR | 41.48 (−18.76, 101.72) | 0.18 | ||||||
1Model 1 is adjusted for all maternal variables 2Model 2 was fitted by selecting variables with P ≤ 0.2 from the univariate analysis
3Model 3 was fitted by selecting variables with P ≤ 0.05 from the univariate analysis
Abbreviation: 2hPG 2 h plasma glucose, FPG fasting plasma glucose, HDL-C high density lipoprotein cholesterol, BMI body mass index, LDL-C low density lipoprotein cholesterol, HOMA-IR homeostasis model assessment of insulin resistance
The associations between neonatal birth weight and neonatal cardiometabolic and micronutrients characteristics were presented in Table 6. The univariate analysis showed that folate was positively associated with neonatal birth weight while triglyceride was negatively associated with neonatal birth weight. In the adjusted analysis (model 2), a unit increase in triglycerides was associated with a decrease in birth weight of 2.81 g (p = 0.02). However, a unit increase in folate significantly increase birth weight by 5.26 g (p = 0.03).
Table 6.
Generalized linear regression analysis showing the associations between neonatal birth weight and neonatal cardiometabolic and micronutrient variables. Using the Akaike information criteria (AIC), model 2 was selected because it has the smallest AIC value
| Neonatal variables | Univariate (unadjusted) |
1Full Model (n = 138) AIC = 1903.11 |
2Model 2 (n = 138) AIC = 1713.23 |
3Model 3 (n = 138) AIC = 1810.955 |
||||
|---|---|---|---|---|---|---|---|---|
| β (95% CI) | P-value | β (95% CI) | P-value | β (95% CI) | P-value | β (95% CI) | P-value | |
| Triglyceride (mg/dl) | −3.60 (−6.11, −1.08) | 0.01 | −2.18 (−5.27, 0.91) | 0.17 | −2.81 (− 5.33, −0.29) | 0.03 | − 3.25 (− 5.75, − 0.75) | 0.01 |
| Folate (ng/ml) | 5.15 (0.72, 9.57) | 0.02 | 5.41 (0.23, 10.59) | 0.04 | 5.26 (0.47, 10.05) | 0.03 | 4.77 (0.45, 9.09) | 0.03 |
| Blood glucose (mmol/l) | −41.98 (−88.39, 4.43) | 0.08 | −29.54 (−97.25, 38.16) | 0.39 | −38.39 (−90.27, 13.50) | 0.15 | ||
| Glucose insulin ratio (GIR) | −5.38 (−12.60, 1.84) | 0.14 | −2.98 (−16.77, 10.81) | 0.67 | −0.28 (− 8.66, 8.10) | 0.95 | ||
| Homocysteine (μmol/l) | −27.39 (−59.44, 4.67) | 0.09 | −21.43 (−55.87, 13.01) | 0.22 | −18.34 (− 50.38, 13.70) | 0.26 | ||
| Insulin (μIU/ml) | 3.04 (−8.85, 14.94) | 0.61 | −8.32 (−29.48, 12.83) | 0.44 | ||||
| Cholesterol (mg/dl) | −1.62 (−4.99, 1.74) | 0.34 | −6.15 (−13.55, 1.25) | 0.10 | ||||
| HDL- C (mg/dl) | 3.18 (−3.83, 10.19) | 0.37 | 9.91 (−1.52, 21.34) | 0.09 | ||||
| LDL-C (mg/dl) | 0.06 (−4.92, 5.04) | 0.98 | 8.57 (−0.79, 17.92) | 0.07 | ||||
| Vitamin D (nmol/l) | 1.10 (−4.41, 6.61) | 0.69 | 1.45 (−4.01, 6.91) | 0.60 | ||||
| Vitamin B12 (pg/ml) | 0.02 (−0.30, 0.35) | 0.89 | −0.03 (− 0.38, 0.33) | 0.87 | ||||
| Ferritin (ng/ml) | 0.06 (−0.62, 0.74) | 0.86 | −0.06 (− 0.77, 0.65) | 0.87 | ||||
| Leptin (ng/ml) | 4.81 (−9.24, 18.87) | 0.50 | −0.75 (−14.71, 13.20) | 0.92 | ||||
| Gestational age in weeks | −11.83 (−55.64, 31.98) | 0.60 | − 19.19 (−68.13, 29.74) | 0.44 | ||||
1Model 1 is adjusted for all neonatal variables 2Model 2 was fitted by selecting variables with P ≤ 0.2 from the univariate analysis
3Model 3 was fitted by selecting variables with P ≤ 0.05 from the univariate analysis
Abbreviation: HDL-C high density lipoprotein cholesterol, LDL-C low density lipoprotein cholesterol
Figure 2 illustrated maternal (including height and homocysteine) and neonatal (including cord triglyceride and folate) factors that were significantly associated with neonatal birth weight.
Fig. 2.
Maternal and neonatal factors that were significantly associated with neonatal birth weight
Discussion
Our data reports the maternal characteristics and nutritional status in early pregnancy (6–14 weeks) and its impact on neonatal birth weight. High prevalence of both underweight (< 18.5 kg/m2) and overweight (BMI ≥23 kg/m2) were observed in our study. Our findings are in line with 2007 Bangladesh Demographic and Health Survey (BDHS) [28]. As our assessment occurred in early pregnancy, the observed weight gain is likely related to the pre-pregnancy status. Our findings are consistent with other rural and urban studies in Bangladesh who also identified similar prevalence of women with low BMI in early pregnancy, but the rate of overweight/obesity was lower. This is likely to be a consequence of applying the newer WHO defined cutoff levels for BMI in South Asian populations [29–31].
In our study, we assessed the level of micronutrient status in early pregnancy (gestational age 10.1 ± 2.2 week), therefore the chances of confounding effect of hemodilution which is common in late pregnancy was unlikely. Our data showed a high prevalence of vitamin D deficiency and insufficiency in pregnant women in early pregnancy. Studies in Pakistan with identical definition for vitamin D deficiency with similar inclusion criteria had shown similar results [15, 32]. Similar findings were also observed in other studies conducted in Bangladesh [33] and India [34].
Limited dietary sources, dark skin pigmentation, and religious or cultural practices that limit skin exposure to sunlight may prevent endogenous production of vitamin D, as illustrated by El-Sonbaty and Abdul-Ghaffar et al. [35] and Guzel et al. [36] may in part explain the higher occurrence of hypovitaminosis in a population with abundant exposure to sunlight year-round. The average sun exposure in Dhaka was 12 h and above during the months of recruitment (April – September) and therefore it is less likely that vitamin D levels may have been impacted by sunlight exposure. Most importantly daily dietary intake of vitamin D was low (normal 15 μgm vs 4.34 μgm) in our study population. This may suggest that dietary intervention with vitamin D rich foods may be an appropriate strategy to address the issue.
Increasing evidence suggest that vitamin B12 deficiency is highly prevalent in women of reproductive age, particularly amongst populations with limited intake of animal source foods [32, 37]. Evidence also suggests that vitamin B12 concentration gradually decline throughout gestation, therefore, it is challenging to assess the prevalence of deficiency in pregnant women. Studies have shown that based on gestational week, prevalence of vitamin B12 deficiency may vary from 5% (< 28 days gestation) to 72% (immediately prior to delivery) worldwide [38]. The JiVitA study in rural north-western Bangladesh found a prevalence of 20% deficiency in early pregnancy [39]. Another urban study conducted in similar settings of present study in Dhaka city found 26% deficiency in early pregnancy [40]. Our finding is in line with national micronutrient status survey in Bangladesh [41] and Amsterdam Born Children and their Development (ABCD) study, Netherland [42]. However, our results are in contrast with Pune, Indian study [37] and Maternal and Infant Nutrition Interventions in Matlab (MINIMAT) trial in Bangladesh where reported B12 status (46% deficiency) early in their third trimester [31]. One-third of women in Pune study were vegetarian and they consumed less protein (44.9 vs 52.3 g) and fat (32.7 vs 45.8 g) than our participated women. However, average daily dietary intake of vitamin B12 in our study population need to be increased as this was observed low (1.62 mcg) compare to recommended daily intake (2.6 mcg) for Bangladeshi population. Further, Bangladesh diet contains high level of fish compared to many societies with non-vegetarian diet.
Regarding iron and folate deficiency, our findings are consistent with other previous urban study in Bangladesh [40]. Lower levels of folate deficiency (0.2%) was also observed in Pune, India study. Further, it was reported that higher maternal folate concentrations predict greater adiposity and higher insulin resistance in neonates [43]. Furthermore, universal distribution of folate and iron to all pregnant mothers in Bangladesh may have reduced folate deficiency.
The prevalence of GDM in our study was 18.4%. Two similar studies in India and China with identical definition for GDM reported 18.5 and 19.9% prevalence respectively [44, 45]. Bangladesh still has high maternal mortality rate (340/100,000), which is one of the highest in the world [46]. It is alarming though to observe the higher occurrence of GDM in women defined as under or normal weight, considered as low risk. Further investigation is required to define the disposition of the phenotype and prognosis particularly at a time when the upswing of diabetes is threatening the health system in poor countries.
Like many other developing countries, millions of Bangladeshi children suffer from nutritional deficiencies due to their mothers’ poor nutritional status prior to and during pregnancy. Children are often born with LBW. According to National LBW Survey 2003–2004 reports, 36% babies were born with LBW. The mean birth weight of infants in Bangladesh is 2632 g and the mean birth length is 48.5 cm [47]. In our study, incidence of LBW and preterm delivery were 11.6 and 5.8% respectively in our study. Preterm birth rate found higher in LBW babies. Bangladesh Demographic and Health Survey (BDHS) 2011 and MINIMAT trail reported 16 and 20% LBW respectively [41, 31]. They had significantly lower head circumference but there was no significant difference in gestation age or with mother’s BMI status in early pregnancy.
Our results showed that neonates born to underweight mothers had significantly higher blood glucose, higher cholesterol, but higher HDL-C, and lower vitamin B12 than neonates born to overweight mothers irrespective of their birth weight. Mechanisms underlying these associations are still poorly understood. However, our findings reflect the possible interaction of environmental factors and fetal growth and the in-utero glucose and lipid metabolism. Future long-term longitudinal studies in different ethnicities would help to explore the underlying mechanisms.
Finally, we investigated both maternal and neonatal nutritional and cardiometabolic factors on newborn birth weight. In our study, maternal height and weight at inclusion were positively associated with neonatal birth weight in univariate analysis. In the adjusted analysis, maternal height was positively, and homocysteine was negatively associated with neonatal birth weight. A series of studies investigated the relationship between maternal height and birth weight showed that shorter maternal height was associated with reduced fetal growth and low birth weight [31, 48–51]. They concluded that the primary reason for this association was undernutrition/malnutrition. Negative association of maternal homocysteine concentrations with newborn birth weight is consistent with studies conducted in India [52, 53] and Norway [54]. We observed a unit increase in homocysteine was associated with a 33.3 g decrease in mean birth weight in our study. Interestingly, a similar finding has also been observed in a systemic review and meta-analysis conducted by Hogeveen M et al [55] They reported that a one standard deviation (SD) increase in maternal homocysteine corresponded to a decrease in birth weight of 31 g. Homocysteine is a well-recognized risk factor for endothelial dysfunction and cardiovascular disease. Homocysteine concentrations are influenced by vitamin B status, mainly by folate and Vit B12. Therefore, vitamin B supplementation should be introduced to the pregnant women for lowering maternal homocysteine level and reducing its wide range of adverse pregnancy outcomes, including recurrent early pregnancy loss, preeclampsia, LBW, fetal loss and future risk of cardiovascular disease.
Among the neonatal variable, cord folate was positively, and triglyceride was negatively associated with birth weight. Literature has shown that folic acid supplement during the last 12 to 16 weeks of pregnancy delivered heavier babies than those born to mothers receiving iron alone. Further, it was found that the effect of the folic acid supplements was more marked on first born children [ 56].
As in other studies, we have found that lower birth weight was associated with elevated cord triglyceride concentrations [57–59]. Reduced lipoprotein lipase (LPL) activity might be the theoretical explanation for the inverse correlation between fetal cord triglyceride and birth weight. It is known that the expression of LPL is strongly associated with the development of adipose tissue. LBW infants appear to have impaired utilization of circulating triglyceride, because of lacking peripheral adipose tissue [60].
Among the strengths of the study were interviewer training for interview technique, questionnaires, recall of the participants, anthropometric measurements, collection of blood, and its subsequent processing (centrifugation and storage). Primigravida mothers were recruited in their 1st trimester to avoid any noise interfering with the analysis. All the maternal and cord blood parameters were determined in the same laboratory, should reduce the chance of error due to different analysis technique. This study also creates an opportunity for long time follow-up of study participants and their offspring which will serve as a relevant data bank for future studies. The limitations of the present study include a higher number of dropouts and thereby limited number of neonatal samples especially those with very low birthweight (n = 16). It should be noted that the exclusion of GDM mothers in their second visit, political violence during the study period were the major causes of participants’ dropout. In addition, the local cultural practice in Bangladesh is that primigravida mothers deliver the child in their parent’s house; this caused a major undertaking to trace all the participants. Because of limited project budget funds and the logistics of cord blood collection and sample processing, we could not collect samples from the majority of home deliveries; analysis of the neonates was therefore mainly restricted to hospital deliveries to ensure data integrity. Reassuringly, with respect to analysis of the neonate data, there is no significant difference in terms of the nutrition status in those mothers who were recruited initially (n = 498) and those who delivered in the hospital (n = 138). At recruitment, 30.9% of the women were categorized as having low BMI (< 18.5), 48.4% normal BMI (18.5–22.9), and 23.9% (overweight and obese). The samples from the newborns were collected from (n = 138) deliveries from the hospitals. Those mothers who delivered were categorized as low BMI were 27.5%, normal 48.6% and over-weight and obese were 23.7%. The difference was not statistically significant (p = 0.43). Therefore, it is less likely that the results were influenced by the high rate of dropouts.
Even though areas of residence were selected based on a random sampling procedure but the population were selected from five areas in and around Dhaka city. The people in Bangladesh (98–99%) belong to one ethnic group and have similar body structure. Therefore, results may be interpreted with caution to the Indian Bengali population.
However, we could not find any significant differences in the baseline-data for mothers delivered in the hospital or home related to BMI, and therefore, it is less likely to have impacted the results. Home delivery is common in Bangladesh especially in rural and semi-urban population [40]. Micronutrients were assessed only at early gestation in this study, whereas they might also be important for fetal growth at the later gestational period [7–9]. This could be another potential limitation of this study.
Conclusion
Maternal malnutrition including both micro (iron deficiency, vitamin D deficiency, vitamin B12 deficiency, high homocysteine level and folate deficiency) and undernutrition as measured by BMI was highly prevalent in early pregnancy. LBW was associated with lower folate, elevated cord triglyceride concentrations of the neonates and mother’s height and increase in maternal homocysteine levels. Neonates born to underweight mothers exhibited significantly higher blood glucose, cholesterol, higher HDL-C and lower vitamin B12 irrespective of their birth weight although this conclusion should be tempered by the numbers studied. The results may call for closer monitoring of pregnant women by the antenatal clinics. The data has also important implications for pregnancy care in Bangladesh and other similar communities.
Acknowledgements
We gratefully acknowledge the contribution of the participating women, hospitals staffs and authorities, Government family welfare visitors, midwives and research staffs involved in GIFTs study. We thank Dr. Sarah Finer (Blizard Institute, Barts and The London School of Medicine and Dentistry, Queen Mary University of London), Virgina Govoni, Project Coordinator GIFTs, Professor Caroline Fall (Human Development and Health, University of Southampton, UK) for advice on anthropometric measurements in the newborn and authorities of Diabetic Association of Bangladesh for their administrative support. We also acknowledge European Union for their financial support (FP7 EU grant: 83599025).
Abbreviations
- AIC
Akaike Information Criteria
- ANOVA
Analysis of variance
- BDT
Bangladeshi Taka
- BIRDEM
Bangladesh Institute of Research and Rehabilitation in Diabetes, Endocrine and Metabolic Disorders
- BMI
Body mass index
- BP
Blood pressure
- CAD
Coronary artery disease
- FFQs
Food Frequency Questionnaires
- FPG
Fasting plasma glucose
- FWV
Family Welfare Visitors
- GDM
Gestational diabetes
- GIFTS
Genomic and lifestyle predictors of fetal outcome relevant to diabetes and obesity and their relevance to prevention strategies in South Asian peoples
- HDL-C
High density lipoprotein cholesterol
- HOMA-IR
Homeostasis model assessment of insulin resistance
- HTN
Hypertension
- 2hPG
2 h plasma glucose
- IADPSG
International Association of Diabetes and Pregnancy Study Group
- IUGR
Intrauterine growth restriction
- T2DM
Type 2 diabetes
- LBW
Low birth weight
- LDL-C
low density lipoprotein cholesterol
- MCHTI
Maternal and Child Health Training Institute
- MUAC
Mid upper arm circumference
- SES
Socio-economic condition
- WHR
Waist-hip ratio
Authors’ contributions
BB involved in designed the protocol, carried out the field work, performed the statistical analysis, data management and writing the manuscript. TS and AM carried out the field work, collected the data and helped in writing the manuscript. IAM performed the statistical analysis and data management. IAH and ZH performed laboratory measurements. IJ, NC, AA and AB helped in writing the manuscript. AK, GAH and AH planned the study, designed the protocol and edited the manuscript. All authors have read and approved the final manuscript.
Authors’ information
Not applicable.
Funding
Financial support from European Union (FP7 EU grant: 83599025). The Funder had no role in the design, conduct or interpretation of the study and decision to publish results.
Availability of data and materials
The datasets during and / or analyzed during the current study available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The protocol was approved by Ethical Review Committee of Diabetic Association of Bangladesh (7 May 2011), and research participation, confidentiality and consent followed the Helsinki declaration, with local adaptation to allow both verbal and written instructions. We obtained written consent from all participants.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Bishwajit Bhowmik, Email: doctorbiplob@yahoo.com, Email: doctorbiplob@gmail.com.
Tasnima Siddique, Email: tasnimasiddiquee08@yahoo.com.
Anindita Majumder, Email: majumderanindita.rpmc@gmail.com.
Ibrahimu Mdala, Email: ibramdala@gmail.com.
Israt A. Hossain, Email: israt.ru84@gmail.com
Zahid Hassan, Email: mzhassan@buhs.ac.bd.
Ishrat Jahan, Email: mchtidhaka@gmail.com.
Nayla Cristina do V. Moreira, Email: naylacristinam@yahoo.com.br
Abdul Alim, Email: alimprodhan21@gmail.com.
Abdul Basit, Email: abdulbasit@bide.edu.pk.
Graham A. Hitman, Email: g.a.hitman@qmul.ac.uk
Abul Kalam A. Khan, Email: president@dab-bd.org
Akhtar Hussain, Email: hussain.akhtar@nord.no.
References
- 1.Marsal K. Intrauterine growth restriction. Curr Opin Obstet Gynecol. 2002;14:127–135. doi: 10.1097/00001703-200204000-00005. [DOI] [PubMed] [Google Scholar]
- 2.Barker DJP, Clark PM. Fetal undernutrition and disease in later life. Rev Reprod. 1997;2:105–112. doi: 10.1530/ror.0.0020105. [DOI] [PubMed] [Google Scholar]
- 3.Kleeman DO, Walker SK, Seamark RF. Enhanced fetal growth in sheep administered progesterone during the first three days of pregnancy. J Reprod Fertil. 1994;102:411–417. doi: 10.1530/jrf.0.1020411. [DOI] [PubMed] [Google Scholar]
- 4.Walker SK, Hartwich KM, Seamark RF. The production of unusually large offspring following embryo manipulation: concepts and challenges. Theriogenology. 1996;45:111–120. doi: 10.1016/0093-691X(95)00360-K. [DOI] [Google Scholar]
- 5.Kwong WY, Wild A, Roberts P, Willis AC, Fleming TP. Maternal undernutrition during the preimplantation period of rat development causes blastocyst abnormalities and programming of postnatal hypertension. Development. 2000;127:4195–4202. doi: 10.1242/dev.127.19.4195. [DOI] [PubMed] [Google Scholar]
- 6.Gluckman PD, Hanson MA. The developmental origins of the metabolic syndrome. Trends Endocrinol Metab. 2004;15:183–187. doi: 10.1016/j.tem.2004.03.002. [DOI] [PubMed] [Google Scholar]
- 7.Barker DJ, Winter PD, Osmond C, Margetts B, Simmonds SJ. Weight in infancy and death from ischaemic heart disease. Lancet. 1989;2:577–580. doi: 10.1016/S0140-6736(89)90710-1. [DOI] [PubMed] [Google Scholar]
- 8.Roseboom TJ, van der Meulen JH, Osmond C, Barker DJ, Ravelli AC, Schroeder-Tanka JM. Coronary heart disease after prenatal exposure to the Dutch famine, 1944–45. Heart. 2000;84:595–598. doi: 10.1136/heart.84.6.595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Eriksson JG, Forsén T, Tuomilehto J, Osmond C, Barker DJ. Early growth and coronary heart disease in later life: longitudinal study. BMJ. 2001;322:949–953. doi: 10.1136/bmj.322.7292.949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Victora CG, Adair L. Fall C, et al, the maternal and child Undernutrition study group: maternal and child undernutrition: consequences for adult health and human capital. Lancet. 2008;371:340–357. doi: 10.1016/S0140-6736(07)61692-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.United Nations Children’s Fund and WHO: Low Birth Weight country, regional and global estimates. New York; 2004. [http://www.unicef.org/publications/index_24840.html]. Last accessed 1 April 2017.
- 12.Freathy RM, Mook-Kanamori DO, Sovio U, Prokopenko I, Timpson NJ, Berry DJ. Variants in ADCY5 and near CCNL1 are associated with fetal growth and birth weight. Nat Genet. 2010;42(5):430–435. doi: 10.1038/ng.567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Barker DJ, Osmond C, Forsén TJ, Kajantie E, Eriksson JG. Trajectories of growth among children who have coronary events as adults. N Engl J Med. 2005;353:1802–1809. doi: 10.1056/NEJMoa044160. [DOI] [PubMed] [Google Scholar]
- 14.Ehtisham S, Crabtree N, Clark P, Shaw N, Barrett T. Ethnic differences in insulin resistance and body composition in United Kingdom adolescents. J Clin Endocrinol Metab. 2005;90:3963–3969. doi: 10.1210/jc.2004-2001. [DOI] [PubMed] [Google Scholar]
- 15.Riaz M, Shaikh F, Fawwad A, Hakeem R, Shera AS, Hitman GA, et al. Maternal nutrition during early pregnancy and Cardiometabolic status of neonates at birth. J Diabetes Res. 2018;24:7382946. doi: 10.1155/2018/7382946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Fottrell E, Ahmed N, Nahar B, Shaha SK, Kuddus A, Hitman GA, et al. Growth and body composition of children aged 2-4 years after exposure to community mobilisation women's groups in Bangladesh. J Epidemiol Community Health. 2018;72:888–895. doi: 10.1136/jech-2017-210134. [DOI] [PubMed] [Google Scholar]
- 17.Rayanagoudar G, Moore M, Zamora J, Hanson P, Huda MS, Hitman GA, Thangaratinam S. Postpartum care of women with gestational diabetes: survey of healthcare professionals. Eur J Obstet Gynecol Reprod Biol. 2015;194:236–240. doi: 10.1016/j.ejogrb.2015.09.019. [DOI] [PubMed] [Google Scholar]
- 18.Rayanagoudar G, Hashi AA, Zamora J, Khan KS, Hitman GA, Thangaratinam S. Quantification of the type 2 diabetes risk in women with gestational diabetes: a systematic review and meta-analysis of 95,750 women. Diabetologia. 2016;59:1403–1411. doi: 10.1007/s00125-016-3927-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Greenhalgh T, Clinch M, Afsar N, Choudhury Y, Sudra R, Campbell-Richards D, et al. Socio-cultural influences on the behaviour of south Asian women with diabetes in pregnancy: qualitative study using a multi-level theoretical approach. BMC Med. 2015;13:120. doi: 10.1186/s12916-015-0360-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rogozińska E, Chamillard M, Hitman GA, Khan KS, Thangaratinam S. Nutritional manipulation for the primary prevention of gestational diabetes mellitus: a meta-analysis of randomised studies. PLoS One. 2015;10:e0115526. doi: 10.1371/journal.pone.0115526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.NIPORT et al. Bangladesh demographic and health survey 2011. Dhaka: NIPORT, Mitra and Associates, and ICF International; 2013. [Google Scholar]
- 22.International Association of Diabetes and Pregnancy Study Groups Consensus Panel. Metzger BE, Gabbe SG, Persson B, Buchanan TA, Catalano PA, et al. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care. 2010;33:676–682. doi: 10.2337/dc09-1848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Siddiquee T, Bhowmik B, Da Vale Moreira NC, Mujumder A, Mahtab H, Khan AKA. Akhtar Hussain. Prevalence of obesity in a rural Asian Indian (Bangladeshi) population and its determinants. BMC Public Health. 2015;15:860. doi: 10.1186/s12889-015-2193-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Matthews DR, Hosker JP, Rudenski AS, et al. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- 25.World Health Organization, Western Pacific Region . The Asia–Pacific Perspective: Redefining Obesity and its Treatment. Sydney: Health Communications Australia Pty Limited; 2000. The International Association for the Study of Obesity and the International Obesity Task Force. [Google Scholar]
- 26.Organization WH. International statistical classification of diseases and related health problems, tenth revision, 2nd ed: World Health Organization; 2004. [PubMed]
- 27.Snipes M, Taylor DC. Model selection and Akaike information criteria: an example from wine ratings and prices. Wine Econ Policy Vol. 2014;3:3–9. doi: 10.1016/j.wep.2014.03.001. [DOI] [Google Scholar]
- 28.National Institute of Population Research and Training . Bangladesh demographic and health survey 2007. Dhaka: National Institute of Population Research and Training; 2009. p. 346p. [Google Scholar]
- 29.Hossain B, Sarwar T, Reja S, Akter MN. Nutritional status of pregnant women in selected rural and urban area of Bangladesh. J Nutr Food Sci. 2013;3:219. doi: 10.4172/2155-9600.1000219. [DOI] [Google Scholar]
- 30.Milton AH, Smith W, Rahman B, Ahmed B, Shahidullah SM, et al. Prevalence and determinants of malnutrition among reproductive aged women of rural Bangladesh. Asia Pac J Public Health. 2010;22:110–117. doi: 10.1177/1010539509350913. [DOI] [PubMed] [Google Scholar]
- 31.Lindström E, Hossain MB, Lönnerdal B, Raqib R, El Arifeen S, Ekström EC. Prevalence of anemia and micronutrient deficiencies in early pregnancy in rural Bangladesh, the MINIMat trial. Acta Obstet Gynecol Scand. 2011;90:47–56. doi: 10.1111/j.1600-0412.2010.01014.x. [DOI] [PubMed] [Google Scholar]
- 32.Hossain N, Khanani R, Hussain-Kanani F, Shah T, Arif S, Pal L. High prevalence of vitamin D deficiency in Pakistani mothers and their newborns. Int J Gynecol Obstet. 2011;112:229–233. doi: 10.1016/j.ijgo.2010.09.017. [DOI] [PubMed] [Google Scholar]
- 33.Roth DE, Al Mahmud A, Raqib R, Akhtar E, Perumal N, Pezzack B, et al. Randomized placebo-controlled trial of high-dose prenatal third-trimester vitamin D3 supplementation in Bangladesh: the AViDD trial. Nutr J. 2013;12:47. doi: 10.1186/1475-2891-12-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Marwaha RK, Tandon N, Chopra S, Agarwal N, Garg MK, Sharma B, et al. Vitamin D status in pregnant Indian women across trimesters and different seasons and its correlation with neonatal serum 25-hydroxyvitamin D levels. Br J Nutr. 2011;106:1383–1389. doi: 10.1017/S000711451100170X. [DOI] [PubMed] [Google Scholar]
- 35.El-Sonbaty MR, Abdul-Ghaffar NU. Vitamin D deficiency in veiled Kuwaiti women. Eur J Clin Nutr. 1996;50:315–320. [PubMed] [Google Scholar]
- 36.Guzel R, Kozanoglu E, Guler-Uysal F, Soyupack S, Sarpel T. Vitamin D status and bone mineral density of veiled and unveiled Turkish women. J Womens Health Gend Based Med. 2001;10(8):765–770. doi: 10.1089/15246090152636523. [DOI] [PubMed] [Google Scholar]
- 37.Stabler SP, Allen RH. Vitamin B12 deficiency as a worldwide problem. Annu Rev Nutr. 2004;24:299–326. doi: 10.1146/annurev.nutr.24.012003.132440. [DOI] [PubMed] [Google Scholar]
- 38.Siddiqua JT, Allen HL, Raqib R, Ahmed T. Vitamin B12 deficiency in pregnancy and lactation: is there a need for pre and postnatal supplementation? J Nutr Disorders Ther. 2014;4:142. doi: 10.4172/2161-0509.1000142. [DOI] [Google Scholar]
- 39.Shamim A, Kabir A, Merrill R, Ali H, Rashid M, Schulze K, et al. Plasma zinc, vitamin B12 and α-tocopherol are positively and plasma γ-tocopherol is negatively associated with Hb concentration in early pregnancy in north-West Bangladesh. Public Health Nutr. 2013;16:1354–1361. doi: 10.1017/S1368980013000475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Siddiqua TJ, Ahmad SM, Ahsan KB, Rashid M, Roy A, Rahman SM, et al. Vitamin B12 supplementation during pregnancy and postpartum improves B12 status of both mothers and infants but vaccine response in mothers only: a randomized clinical trial in Bangladesh. Eur J Nutr. 2016;55:281–293. doi: 10.1007/s00394-015-0845-x. [DOI] [PubMed] [Google Scholar]
- 41.National Institute of Population Research and Training, Mitra and Associates, & ICF International . Bangladesh demographic and health survey 2011. Dhaka: NIPORT, Mitra and Associates, and ICF International; 2013. [Google Scholar]
- 42.Krikke GG, Grooten IJ, Vrijkotte TG, van Eijsden M. Roseboom TJ1, painter RC. Vitamin B12 and folate status in early pregnancy and cardiometabolic risk factors in the offspring at age 5-6 years: findings from the ABCD multi-ethnic birth cohort. BJOG. 2016;123:384–392. doi: 10.1111/1471-0528.13574. [DOI] [PubMed] [Google Scholar]
- 43.Yajnik CS, Deshpande SS, Jackson AA, Refsum H, Rao S, Fisher DJ. Vitamin B12 and folate concentrations during pregnancy and insulin resistance in the offspring: the Pune maternal nutrition study. Diabetologia. 2008;51:29–38. doi: 10.1007/s00125-007-0793-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bhavadharini B, Mahalakshmi MM, Anjana RM, Maheswari K, Uma R, Deepa M, et al. Prevalence of gestational diabetes mellitus in urban and rural Tamil Nadu using IADPSG and WHO 1999 criteria (WINGS 6) Clin Diabetes Endocrinol. 2016;2:8. doi: 10.1186/s40842-016-0028-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shang M, Lin L. IADPSG criteria for diagnosing gestational diabetes mellitus and predicting adverse pregnancy outcomes. J Perinatol. 2014;34:100–104. doi: 10.1038/jp.2013.143. [DOI] [PubMed] [Google Scholar]
- 46.World Health Organization . Trends in maternal mortality: 1990 to 2008: estimates developed by WHO, UNICEF, UNFPA and the World Bank. Geneva: World Health Organization; 2010. [Google Scholar]
- 47.Bangladesh Bureau of Statistics (BBS) and UNICEF (2005) National Low Birth Weight Survey of Bangladesh . Planning division, Ministryof planning. Dhaka: Government of People’s Republic of Bangladesh; 2003. [Google Scholar]
- 48.Jananthan R, Wijesinghe DG, Sivananthewerl T. Maternal anthropometry as a predictor of birth weight. Trop Agric Res. 2009;21:89–98. doi: 10.4038/tar.v21i1.2590. [DOI] [Google Scholar]
- 49.Kramer MS. The epidemiology of adverse pregnancy outcomes: an overview. J Nutr. 2003;133:1592S–15926S. doi: 10.1093/jn/133.5.1592S. [DOI] [PubMed] [Google Scholar]
- 50.Ozaltin E, Hill K, Subramanian SV. Association of maternal stature with offspring mortality, underweight, and stunting in low- to middle-income countries. JAMA. 2010;303:1507–1516. doi: 10.1001/jama.2010.450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wills AK, Chinchwadkar MC, Joglekar CV, Natekar AS, Yajnik CS, Fall CH, Kinare AS. Maternal and paternal height and BMI and patterns of fetal growth: the Pune maternal nutrition study. Early Hum Dev. 2010;86:535–540. doi: 10.1016/j.earlhumdev.2010.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yajnik CS, Chandak GR, Joglekar C, Katre P, Bhat DS, Singh SN, et al. Maternal homocysteine in pregnancy and offspring birthweight: epidemiological associations and Mendelian randomization analysis. Int J Epidemiol. 2014;43:1487–1497. doi: 10.1093/ije/dyu132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yajnik CS, Deshpande SS, Panchanadikar AV, Naik SS, Deshpande JA, Coyaji KJ, et al. Maternal total homocysteine concentration and neonatal size in India. Asia Pac J Clin Nutr. 2005;14:179–181. [PubMed] [Google Scholar]
- 54.Vollset SE, Refsum H, Irgens LM, et al. Plasma total homocysteine, pregnancy complications, and adverse pregnancy outcomes: the Hordaland Homocysteine study. Am J Clin Nutr. 2000;71:962–968. doi: 10.1093/ajcn/71.4.962. [DOI] [PubMed] [Google Scholar]
- 55.Hogeveen M, Blom HJ, den Heijer M. Maternal homocysteine and small-for-gestational-age offspring: systematic review and meta-analysis. Am J Clin Nutr. 2012;95:130–136. doi: 10.3945/ajcn.111.016212. [DOI] [PubMed] [Google Scholar]
- 56.Iyengar L, Rajalakshmi K. Effect of folic acid supplement on birth weights of infants. Am J Obstet Gynecol. 1975;122:332–336. doi: 10.1016/0002-9378(75)90179-9. [DOI] [PubMed] [Google Scholar]
- 57.Schaefer G, Kristof G, Irina K, Siri LK, Joachim D, Klaus V, Emilio H. Maternal Lipids as Strong Determinants of Fetal Environment and Growth in Pregnancies With Gestational Diabetes Mellitus Ute M. Diabetes Care. 2008;31:1858–1863. doi: 10.2337/dc08-0039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rodie VA, Caslake MJ, Stewart F, et al. Fetal cord plasma lipoprotein status in uncomplicated human pregnancies and in pregnancies complicated by pre-eclampsia and intrauterine growth restriction. Atherosclerosis. 2004;176:181–187. doi: 10.1016/j.atherosclerosis.2004.04.026. [DOI] [PubMed] [Google Scholar]
- 59.Yajnik CS, Fall CH, Coyaji KJ, Hirve SS, Rao S, Barker DJ, et al. Neonatal anthropometry: the thin-fat Indian baby. The Pune maternal nutrition study. Int J Obes Relat Metab Disord. 2003;27:173–180. doi: 10.1038/sj.ijo.802219. [DOI] [PubMed] [Google Scholar]
- 60.Schaefer-Graf UM1. Graf K, Kulbacka I, Kjos SL, Dudenhausen J, Vetter K, Herrera E. Maternal lipids as strong determinants of fetal environment and growth in pregnancies with gestational diabetes mellitus. Diabetes Care. 2008;31:1858–1863. doi: 10.2337/dc08-0039. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets during and / or analyzed during the current study available from the corresponding author on reasonable request.