Abstract
Iryanthera polyneura (Myristicaceae) is popularly known as cumala-colorada, and can be found in the Amazon rain forest. The present study aimed the evaluation of the chemical composition of the essential oils (EOs) obtained from the leaves of I. polyneura throughout a two-year period in order to correlate chemical findings with seasonality and climatic variation. Leaves from I. polyneura were collected 15 times from three different individuals, identified as 22EO, 80EO and 530EO, between the years of 2009 and 2011. The EOs were obtained and tested by gas-chromatography-mass spectrometry techniques. ANOVA and multivariate analyses were used to assess the relationship between the percentage of terpenes and seasonal/climatic variations. Fifty-nine compounds were detected in the EOs, of which 44 were identified (74.5%). The main components of the EOs were spathulenol (6.42 ± 1.02%), α-cadinol (5.82 ± 0.40%) and τ-muurolol (5.24 ± 0.03%). Higher levels of spathulenol were observed in 22EO during rainy season, while τ-muurolol occurred in high amounts during the dry season, as did α-cadinol in 22EO and 80EO. Correspondence analysis revealed a distinction between two groups of EOs based on seasonality, whereas a canonic correspondence analysis and ANOVA determined how the major compounds were related to both seasonality and climatic factors. Non-metric multidimensional scaling in association with an analysis of similarities showed that the abundance and composition of terpenes in the samples varied moderately among the three Iryanthera individual plants. Present findings have shown that there is variation in the occurrence of the major compounds spathulenol, τ-muurolol and α-cadinol produced by I. polyneura and that the pattern of variation is related to season and climatic changes.
Introduction
Brazil is the richest country in terms of biodiversity in the world. Currently, the Brazilian flora harbors at least 46,500 species, of which 4,754 are algae, 33,158 are angiosperms, 1,567 bryophytes, 5,719 fungi, 30 gymnosperms and 1,345 are ferns and lycophytes [1]. As living organisms, plants communicate with the environment, and some of their strategies are related to defense [2] and others to reproduction [3].
EOs are an organic mixture of compounds occurring in specialized plant cells or glands, that play a fundamental role in plant-biotic/abiotic environment interactions [4]. They originate from intermediate products of the main metabolic pathways involved in the production of sugars, lipids and proteins. Characteristically, EOs have high genetic plasticity and wide chemical diversity, which allows the plants to adapt to the environmental demands, which are in continuously changing [5]. Among the functions exerted by EOs in plants, defense against bacteria, virus and fungi, as well as herbivorous insects or animals have been well-described [6].
Plants within the Myristicaceae have been known to produce EOs. Ethnopharmacological interest in this family emerged in the 1950’s, when Richard Schultes disclosed the discovery that some species of the genus Virola were sources of snuffs yakee, parika or epena, a powerful hallucinogenic narcotic used by Indian people from the Amazon region [7]. Myristicaceae species are still used for many purposes by native Amazonians, for example, in the preparation of arrows and for the treatment of dermatological or stomach infections, rheumatism, tumors, among others [8, 9, 10]. Some species of the genus Iryanthera have been popularly used as hallucinogenic, such as the bark of I. macrophylla (Benth.) Warb., or used as antidiarrhoeic, as the bark of I. tessmanii Markgr.; moreover, the wood of several species has been used in construction [11]. A phytochemical study made with the fruits of I. lancifolia Ducke revealed the presence of dihydrochalcones and flavonolignans which showed higher antioxidant activity in relation to α-tocopherol and quercetin [12]. I. polyneura were chemically accessed [13], and diarylpropanoids were isolated from the wood of their trunks.
Due to the lack of chemical and biological information regarding I. polyneura, the aim of the present work was to assess the chemical composition of the EOs obtained from the leaves of I. polyneura throughout a two-year span. Further, researchers aimed at the comprehension of how climatic factors such as the relative humidity, precipitation, minimum, average and maximum daily temperatures, insolation, wind and evaporation, and the seasonal variation might affect the chemical composition of the oils.
Material and methods
Collection of botanical material
The collection of botanical material was carried out in a terra-firme forest, within the Amazon Basin. The collections were made under licenses # 14895 (ICMBio/MMA/Brazil) and 12A/2008 (IBAMA/CGen/MMA), as required by the Brazilian Government. A voucher from each individual was collected as described in Table 1. The present study was based on a previous one made with Osteophloeum platyspermum (Spruce ex A.DC.) Warb., Myristicaceae [14].
Table 1. Indexation of botanical material collected from Iryanthera polyneura.
DBH = diameter at breast height.
| Individual (Field Number) | Herbarium Voucher | Determined by | DBH (cm) | Universal Transverse Marcator (UTM Zone 20) | |
|---|---|---|---|---|---|
| Longitude (X) | Latitude (Y) | ||||
| 22 | Oliveira A.A. 4064; UNIP 5170 |
Paciencia, M. | 14.1 | 780103.21 | 9686375.79 |
| 80 | Oliveira 4144; UNIP 5279 |
Paciencia, M. | 17.4 | 780073.14 | 9686558.12 |
| 530 | Paciencia, M. 3609; UNIP 8566 |
Paciencia, M. | 14.5 | 780141.24 | 9686608.38 |
Collections were made from trees having DBH≥10cm (diameter at breast height), closely located in the same hectare plot, containing enough leaf biomass to allow for a periodical collection spanning two years, from October 2009 to December 2011. The individuals were identified as 22EO, 80EO and 530EO. Individuals 22EO and 530EO provided 15 material collections, while 80EO provided 14. The collection dates were determined in a random way, depending on the expedition logistics.
Samples of the collected material for botanical research were deposited at the UNIP Herbarium (Table 1), São Paulo, Brazil. The material was cleaned so as to remove contaminants such as insects or other animals, other organs of the same plant, other plants, sand and earth. The cleaned material was kept in a cold room until it was used to obtain the volatile oils.
Preparation of the EOs
Fresh leaves of I. polyneura were periodically collected from the same individuals in order to obtain EOs. EOs were obtained by hydrodistillation in a Clevenger apparatus [15] for four hours. The oils were collected from the Clevenger apparatus with the aid of pentane, and were evaporated using a rotary evaporator apparatuses (Buchi, Switzerland). The remaining water was then removed with the addition of anhydrous sodium sulfate. The yields (mg/mg) of all EO samples (15 samples obtained from individuals 22EO, and, 14 samples obtained from individual 80EO and 530EO) were evaluated and then stored at -10°C, until use. To perform gas chromatography coupled to mass spectrometer analyses (GC-MS), 20μL of each oil were diluted in 980μL of acetone.
Analysis of volatile oils by gas chromatography coupled to mass spectrometry (GC-MS)
GC-MS analyses were performed on a Shimadzu 14B/QP5050A apparatus, with quadrupole type analyzer of the same brand. The column used was BPX5 (non-polar 5% Phenyl Polisylphenlene Siloxane), 30m, with internal diameter of 0.25mm. The running conditions of the gas chromatography were: initial oven temperature 60°C (6 min), final temperature 320°C (8 min), temperature increase of 10°C/min and a total running time of 40 min, column pressure was 150.0kPa with a column flow of 2.5mL/min, linear velocity of 58, split ratio of 9 and total flow of 30.0mL/min. Each of the 44 acetone-diluted samples were injected at a volume of 1μL. The substances were identified by comparing the obtained mass spectra with libraries, such as Willey229, NIST107, SHIM1607 and NIST21, and with a reference books and retention indices [16].
The retention indices (RI) were obtained from the injection of a mixture of n-alcanes (C9H20–C25H52, Sigma Aldrich, 99%) using Van den Dool and Kratz equation [17].
Experimental design
With three exceptions, yields (mg/mg) of all EO samples were obtained. Fifteen samples were obtained from individuals 22EO and 530EO and 14 samples obtained from individual 80EO.
The chemical and statistical variation were calculated from 41 samples of EOs (15 from 22EO, 12 from 80EO and 14 from 530EO) and were based on terpene percentages. Two EOs from individual 80EO and one EO from individual 530EO were discarded because the number of terpenes detected after GC-MS analysis was very low. Therefore, the samples were considered outliers, since they created trends in the analyses in a very significant way. For the multivariate analyses, 41 EOs were considered as the cases and the 59 terpenes as the variables. Climatic variables used for running the analyses were relative humidity, precipitation, daily temperatures (minimum, average and maximum), insolation, wind speed and evaporation, where data obtained from the day of collection was considered (www.inpe.gov.br). Seasons in the Amazon rain forest are defined according to the rain intensity, so the period of the year when the rain is more intense (accumulated precipitation as high as 3,500mm/year) is defined as the rainy season (RS), popularly known as the “winter”, and spans from January to July. The period of the year known as “summer”, or dry season (DS), is a period in which the precipitation drops down to 900mm/year and spans from July to December [18, 19].
Statistical analyses
One-way ANOVAs and t-tests were performed in order to compare the occurrence of the terpene levels among individuals and between seasons. Differences were considered significant if α<0.05 [20]. NMDS analysis was performed in the association with ANOSIM test of hypotheses, which is similar to the non-parametric analysis of variance based on similarities among samples [21]. In the present case we used ANOSIM to test two different null hypotheses (H0). The first was that there were no differences in the percentual-abundance and the composition of terpenes of the three Iryanthera individual plants (22EO, 80EO, and 530EO). The second was that there was no difference between EO samples obtained during dry and rainy seasons. Correspondence analysis (CA) and canonical correspondence analysis (CCA) were also conducted using the software MVSP, Multivariate Statistical Package version 3.22 (Kovach Computing Services) and Primer 6 version 6.1.6 (Primer-E Ltd.).
Results and discussion
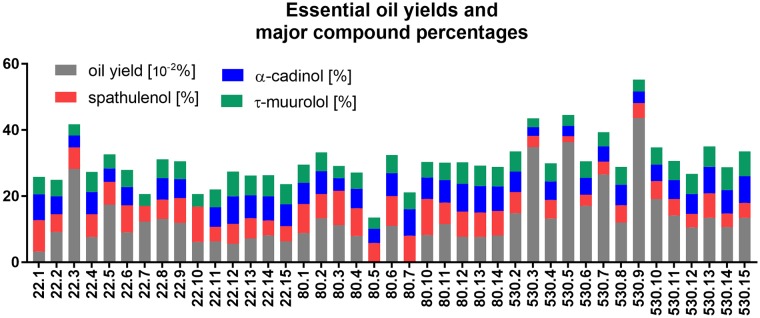
Yields of the volatile oils obtained from the leaves of three individuals of Iryanthera polyneura spanned from 0.03% to 0.43%, with the lowest yield observed for 22EO1 and the highest for the 530EO9 (Table 2, Fig 1). The yields of the oils from individuals 80EO and 530EO were significantly higher than the oils from the individual 22EO (F(2,39) = 5.238; P = 0.0097).
Table 2. Yields (%) obtained for the essential oils obtained from the leaves of three different Iryanthera polyneura individual plants.
#22, #80 and #530 identifies different individuals, while EO1 toEO15 identifies dates of collection. Essential oil yield means for each individual was compared using one-way ANOVA, followed by Tukey post-test, considered significant if α<0.05.
| Essential oil ID | Yield (%) | Essential oil ID | Yield (%) | Essential oil ID | Yield (%) | Date of collection |
|---|---|---|---|---|---|---|
| 22EO1 | 0.033 | 80EO1 | 0.0889 | 530EO1 | 0.1041 | 10/01/2009 |
| 22EO2 | 0.0916 | 80EO2 | 0.1332 | 530EO2 | 0.1487 | 11/01/2009 |
| 22EO3 | 0.2821 | 80EO3 | 0.1124 | 530EO3 | 0.3483 | 02/03/2010 |
| 22EO4 | 0.0769 | 80EO4 | 0.0799 | 530EO4 | 0.1325 | 03/12/2010 |
| 22EO5 | 0.1745 | 80EO5 | NT | 530EO5 | 0.3635 | 05/15/2010 |
| 22EO6 | 0.0907 | 80EO6 | 0.1104 | 530EO6 | 0.1708 | 05/28/2010 |
| 22EO7 | 0.1232 | 80EO7 | NT | 530EO7 | 0.2657 | 08/29/2010 |
| 22EO8 | 0.1313 | 80EO8 | 0.0767 | 530EO8 | 0.1201 | 11/05/2010 |
| 22EO9 | 0.1195 | 80EO9 | 0.3378 | 530EO9 | 0.4362 | 12/14/2010 |
| 22EO10 | 0.0617 | 80EO10 | 0.0828 | 530EO10 | 0.191 | 02/11/2011 |
| 22EO11 | 0.0631 | 80EO11 | 0.115 | 530EO11 | 0.1419 | 04/15/2011 |
| 22EO12 | 0.0561 | 80EO12 | 0.0774 | 530EO12 | 0.1058 | 07/08/2011 |
| 22EO13 | 0.0723 | 80EO13 | 0.0767 | 530EO13 | 0.1346 | 08/20/2011 |
| 22EO14 | 0.0817 | 80EO14 | 0.0814 | 530EO14 | 0.107 | 10/21/2011 |
| 22EO15 | 0.0633 | 530EO15 | 0.1348 | 12/16/2011 | ||
| mean±SD | 0.10±0,02A | mean±SD | 0.11±0,02 A | mean±SD | 0.19±0,03B | |
| Essential oil yield comparison | ||||||
| Barlett’s statistic corrected | 4.479 | P = 0.1065 | ||||
| One-way ANOVA/Tukey | F(2,39) = 5.238 | P = 0.0097 | ||||
| Multiple comparisons | 22EO vs. 80EO | P = 0.9154 | ||||
| 22EO vs. 530EO | P = 0.0120 | |||||
| 80EO vs. 530EO | P = 0.0487 | |||||
A = no differences among means;
B = differences among means. NT = not tested.
Fig 1. Yields from the essential oils obtained from the Iryanthera polyneura leaves and the percentage of the major compounds spathulenol, α-cadinol and τ-muurolol present.
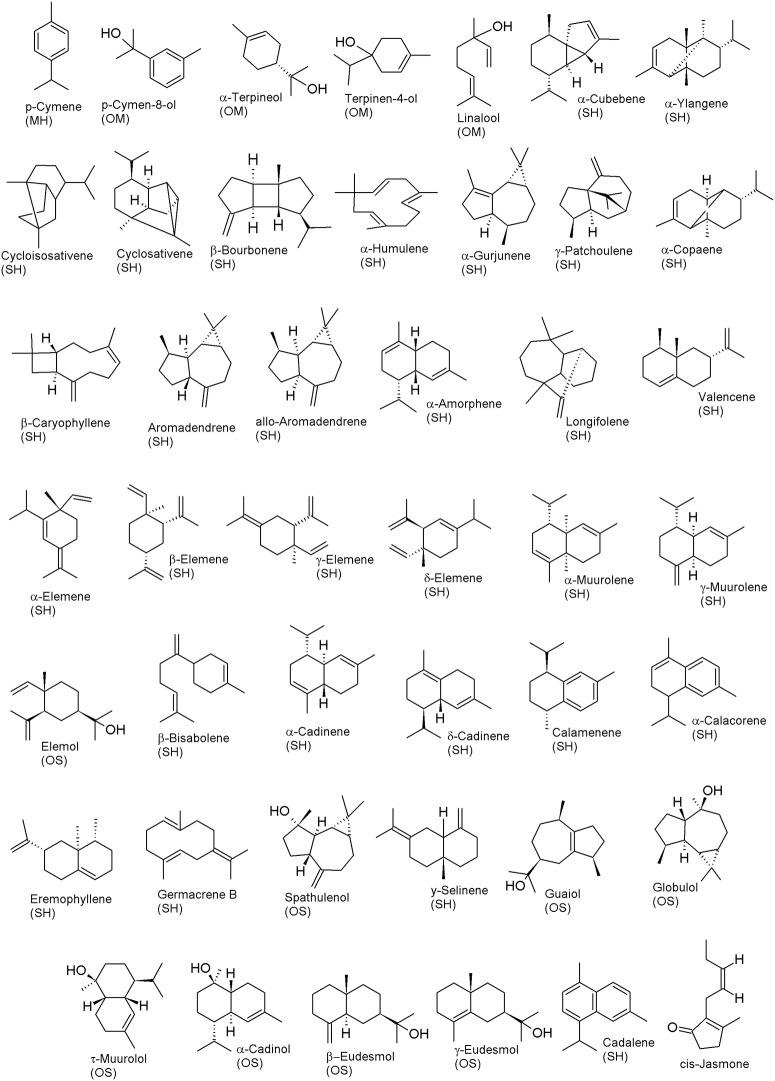
GC-MS analyses of the 44 EOs showed that terpenes were the only type of compound that was identified for this species in the present analysis (Table 3, Fig 2), unlike the possible occurrence of phenylpropanoids in some species of Myristicaceae, such as Myristica fragrans [22]. Sesquiterpene hydrocarbons prevailed in the oils of the three I. polyneura individual plants, followed by the presence of oxygenated sesquiterpenes. Forty four terpenes, or 74.5%, were identified, and 15 compounds were not determined (25.4%). Spathulenol (6.42 ± 1.02%), α-cadinol (5.82 ± 0.40%) and τ-muurolol (5.24 ± 0.03%) were the major compounds found in the EOs (N = 44). Suffredini et al. (2016) has already reported the highest percentages of spathulenol and α-cadinol in EOs from another species of Myristicaceae, such as Osteophloeum platyspermum Warb.
Table 3. Chemical composition (in percentage, %) of essential oils obtained from individual I. polyneura leaves from 22EO, 80EO and 530EO individuals and a summary of the class of compounds identified.
| 22EO | 80EO | 530EO | ||||||
|---|---|---|---|---|---|---|---|---|
| Compound | RI | RI ref. | Mean | Standard error | Mean | Standard error | Mean | Standard error |
| p-Cymene | 1027 | * | 0.01 | 0.01 | 0.02 | 0.01 | 0.01 | 0.01 |
| Linalool | 1109 | * | 0.06 | 0.03 | 0.03 | 0.03 | ||
| Terpinen-4-ol | 1182 | * | 0.03 | 0.03 | 0.02 | 0.02 | ||
| p-Cimen-8-ol | 1190 | * | 0.02 | 0.01 | ||||
| α-Terpineol | 1195 | * | 0.06 | 0.02 | 0.03 | 0.02 | 0.01 | 0.01 |
| δ-Elemene | 1337 | 1335 | 1.08 | 0.17 | 0.97 | 0.10 | 1.44 | 0.22 |
| α-Cubebene | 1349 | 1345 | 0.10 | 0.03 | 0.21 | 0.15 | 0.17 | 0.04 |
| Cycloisosativene | 1364 | * | 0.35 | 0.10 | 0.45 | 0.09 | 0.52 | 0.09 |
| Cyclosativene | 1366 | 1369 | 0.98 | 0.26 | 0.46 | 0.10 | 1.20 | 0.25 |
| α-Yllangene | 1370 | 1373 | 0.12 | 0.04 | 0.06 | 0.03 | 0.24 | 0.06 |
| α-Copaene | 1375 | 1374 | 0.43 | 0.09 | 0.51 | 0.05 | 0.68 | 0.12 |
| β-Bourbonene | 1383 | 1387 | 0.63 | 0.14 | 0.33 | 0.06 | 0.37 | 0.07 |
| β-Elemene | 1391 | 1389 | 2.56 | 0.28 | 3.84 | 0.25 | 3.35 | 0.18 |
| β-Caryophyllene | 1417 | 1417 | 0.64 | 0.09 | 0.63 | 0.10 | 0.81 | 0.11 |
| Sesq1 | 1428 | * | 0.72 | 0.13 | 1.26 | 0.20 | 0.98 | 0.19 |
| γ-Elemene | 1433 | 1434 | 0.31 | 0.05 | 0.50 | 0.08 | 0.59 | 0.08 |
| Aromadendrene | 1437 | 1439 | 0.65 | 0.08 | 1.36 | 0.11 | 1.03 | 0.10 |
| α-Humulene | 1452 | 1452 | 0.13 | 0.04 | 0.16 | 0.05 | 0.17 | 0.03 |
| α-Gurjunene | 1408 | 1409 | 0.01 | 0.01 | 0.06 | 0.03 | 0.08 | 0.03 |
| α-Patchoulene | 1454 | 1454 | 0.01 | 0.01 | 0.08 | 0.07 | ||
| Allo-Aromadendrene | 1459 | 1458 | 0.12 | 0.03 | 0.30 | 0.05 | 0.26 | 0.04 |
| γ-Muurolene | 1475 | 1478 | 2.85 | 0.28 | 2.90 | 0.15 | 3.90 | 0.33 |
| α-Amorphene | 1479 | 1483 | 1.25 | 0.15 | 1.28 | 0.12 | 1.85 | 0.23 |
| Sesq2 | 1484 | * | 0.65 | 0.09 | 0.98 | 0.05 | 0.93 | 0.07 |
| α-Elemene | 1493 | * | 1.36 | 0.12 | 1.70 | 0.11 | 1.94 | 0.15 |
| α-Muurolene | 1499 | 1500 | 2.80 | 0.30 | 3.12 | 0.28 | 4.03 | 0.41 |
| β-Bisabolene | 1508 | 1505 | 0.01 | 0.01 | ||||
| α -Cadinene | 1513 | 1513 | 1.53 | 0.32 | 1.73 | 0.07 | 2.10 | 0.23 |
| Sesq3 | 1514 | * | 0.69 | 0.27 | 0.09 | 0.09 | 0.33 | 0.21 |
| Calamenene | 1522 | 1521 | 1.92 | 0.15 | 1.25 | 0.14 | 2.41 | 0.27 |
| Valencene | 1533 | 1532 | 0.61 | 0.07 | 0.85 | 0.06 | 1.00 | 0.07 |
| α-Cadinene | 1536 | 1537 | 0.10 | 0.06 | ||||
| α-Calacorene | 1541 | 1544 | 0.60 | 0.14 | 0.31 | 0.08 | 0.30 | 0.10 |
| Elemol | 1549 | 1548 | 2.08 | 0.24 | 1.13 | 0.16 | 1.11 | 0.12 |
| Germacrene B | 1555 | 1559 | 0.72 | 0.13 | 1.07 | 0.28 | 0.97 | 0.15 |
| Longifolene | 1558 | 1555 | 0.35 | 0.03 | 0.80 | 0.08 | 0.41 | 0.07 |
| Sesq4 | 1565 | * | 3.00 | 0.22 | 4.15 | 0.34 | 4.17 | 0.32 |
| Spathulenol | 1577 | 1577 | 6.51 | 0.47 | 8.13 | 0.42 | 4.61 | 0.37 |
| Sesq5 | 1582 | * | 4.79 | 0.21 | 7.14 | 0.40 | 5.09 | 0.43 |
| γ-Selinene | 1590 | * | 1.03 | 0.13 | 2.00 | 0.17 | 1.04 | 0.14 |
| Guaiol | 1597 | 1600 | 0.79 | 0.13 | 0.13 | 0.04 | 0.18 | 0.03 |
| Globulol | 1601 | 1590 | 1.30 | 0.15 | 2.62 | 0.21 | 0.74 | 0.24 |
| Eremophyllene | 1601 | * | 0.05 | 0.05 | 1.05 | 0.21 | ||
| Sesq6 | 1611 | 0.78 | 0.11 | |||||
| Sesq7 | 1624 | * | 1.87 | 0.26 | 1.89 | 0.15 | 1.91 | 0.22 |
| Sesq8 | 1628 | * | 5.19 | 0.47 | 3.58 | 0.30 | 4.33 | 0.25 |
| Sesq9 | 1631 | * | 1.82 | 0.26 | 1.29 | 0.06 | 1.37 | 0.12 |
| τ-Muurololl | 1641 | * | 5.28 | 0.29 | 5.19 | 0.26 | 5.26 | 0.37 |
| Sesq10 | 1646 | * | 2.46 | 0.25 | 2.45 | 0.11 | 2.87 | 0.19 |
| β-Eudesmol | 1649 | 1649 | 1.28 | 0.22 | 0.89 | 0.04 | 0.76 | 0.05 |
| α-Cadinol | 1654 | 1654 | 5.35 | 0.65 | 6.61 | 0.41 | 5.49 | 0.45 |
| γ-Eudesmol | 1664 | 1630 | 0.07 | 0.04 | 0.16 | 0.06 | ||
| Sesq11 | 1667 | * | 0.19 | 0.13 | 0.74 | 0.17 | 1.16 | 0.18 |
| Cadalene | 1673 | 1675 | 1.13 | 0.12 | 0.98 | 0.09 | 1.41 | 0.09 |
| Sesq12 | 1693 | * | 0.15 | 0.06 | 0.81 | 0.10 | 0.86 | 0.11 |
| Sesq13 | 1706 | * | 0.23 | 0.08 | 0.46 | 0.02 | 0.31 | 0.03 |
| Sesq14 | 1716 | * | 0.67 | 0.16 | 0.88 | 0.15 | 0.70 | 0.16 |
| cis-Jasmone | 1725 | * | 0.01 | 0.01 | ||||
| Sesq15 | 1748 | * | 0.09 | 0.05 | 0.30 | 0.03 | 0.24 | 0.03 |
| Monoterpene hydrocarbon (%, MH) | 0.69 | 0.85 | 0.47 | |||||
| Oxigenated monoterpene (%, OM) | 3.70 | 1.42 | 0.23 | |||||
| Sesquiterpene hydrocarbon (%, SM) | 78.24 | 80.74 | 81.82 | |||||
| Oxigenated sesquiterpene (%, OS) | 17.36 | 17.00 | 17.48 | |||||
| MH OM SH OS amounts | ||||||||
| MH (A) | 0.67 | 0.1102 | F (3,8) = 2580 | <0.0001 | ||||
| OM (B) | 1.783 | 1.018 | A-B | 0.7197 | ||||
| SH (C) | 80.27 | 1.06 | A-C | <0.0001 | ||||
| OS (D) | 17.28 | 0.1442 | A-D | <0.0001 | ||||
| B-C | <0.0001 | |||||||
| B-D | <0.0001 | |||||||
| C-D | <0.0001 | |||||||
Sesq = unidentified sesquiterpenes;
* = not found
Fig 2. Structure of the compounds identified in the essential oils from the leaves of Iryanthera polyneura.
MH = monoterpene hydrocarbon; OM = oxygenated monoterpene; SH = sesquiterpene hydrocarbon; OS = oxygenated sesquiterpene.
Table 3 shows the qualitative and quantitative terpene variation in individuals 22EO, 80EO and 530EO. Cis-jasmone and α-cadinene were solely found in 22EO, while sesq6 exclusively occurred in 80EO and β-bisabolene in 530EO. This difference may suggest that each individual has genetic variability or that each one may be in a particular physiological phase influenced by intrinsic or extrinsic factors. Spathulenol, elemol, α-muurolene, τ-muurolene, sesq2, sesq3, and τ-muurolol were commonly found in all individuals. Hydrogenated and oxygenated monoterpenes (MH and OM) and sesquiterpenes (SH and OS) were identified in the oils obtained from the three individuals. The terpene content in each of the four groups was summed and used in the statistical evaluation. According to the ANOVA, the SH-group was significantly more prevalent in the oils, in relation to the other three groups (F(3,8) = 2.580; P < 0.001), as was the group OS.
Results obtained from the statistical analyses of the chemical composition percentage and seasonal variation were assessed. A t-test was performed in order to evaluate variation between terpene levels in relation to seasonality, and a F test to compare variances was applied to all t-tests (P > 0.05). For that analysis, the total terpene percentages were summed for each of the 41 oils, which produced two groups, those EOs from the DS and those from the RS. The analyses were made for each set of oils, 22EO, 80EO and 530EO. The total terpene percentage of oils occurring in 22EO was significantly lower in DS samples (t0.05(2),12 = 3.603; P = 0.0036), but higher for the oils from 80EO (t0.05(2),9 = 2.305; P = 0.0467), while no differences were observed in the terpenes from 530EO (t0.05(2),12 = 1.295; P = 0.2197). When considering the total amount of terpenes of all individuals, which were pooled together to evaluate a variation for the species, no differences were observed between the RS and DS (t0.05(2),13 = 0.6985; P = 0.4972).
Statistical analyses were also made considering only the terpenes that were common to all of the 41 EOs, τ-muurolene, α-muurolene, elemol, spathulenol, τ-muurolol, sesq 2 and sesq 3. The analysis revealed that the common terpenes occurred in higher amounts in the DS (t0.05(2),37 = 2.078; P = 0.0447).
Spathulenol, τ-muurolol and α-cadinol were the major compounds identified. As seen in Table 4, the occurrence of spathulenol was measured in each individual, according to the seasonality. It was observed that spathulenol was higher in the RS for individual 22EO (t0.05(2),11 = 2.345; P = 0.0388), but in individuals 80EO and 530EO, levels were not affected by the time of year of collection. The occurrence of spathulenol among the three individuals and the differences between the RS and DS were evaluated. In the analyses, the content of spathulenol was significantly higher in both the RS (F(2,19) = 10.82; P = 0.0007) and the DS (F(2,16) = 5.911; P = 0.012), in the oils derived from individual 80EO. The occurrence of τ-muurolol was also evaluated for each individual according to the seasonality, and it was observed that the occurrence of this terpene was significantly higher during DS, for 22EO (t0.05(2),11 = 2.311; P = 0.0412), 80EO (t0.05(2),10 = 3.012; P = 0.0131) and 530EO (t0.05(2),11 = 2.349; P = 0.0385). The total amount of τ-muurolol calculated for the three individuals together was seasonally compared, and a significantly higher level of production of the terpene during the DS vs the RS was confirmed (t0.05(2),46 = 2.22; P = 0.0314). Finally, the seasonal variation of α-cadinol differed from what was observed for spathulenol and for τ-muurolol. The percentage of α-cadinol was higher in the DS for individuals 22EO (t0.05(2),11 = 2.436; P = 0.0331) and 80EO (t0.05(2),10 = 2.912; P = 0.0155), while the compound was predominant in RS in individual 530EO (t0.05(2),11 = 2.257; P = 0.0453). The total amount of α-cadinol produced by all three individuals together, when seasonally compared, resulted in a significant higher occurrence of α-cadinol in DS (t0.05(2),39 = 2.674; P = 0.0109).
Table 4. Statistical analyses based on the chemical composition (in percentage, %) of the essential oils obtained from the leaves of individuals 22EO, 80EO and 530EO of Iryanthera polyneura in either the dry season (DS) or the rainy season (RS).
| Analysis | factors | mean | SE | F / t | comparison | P/adjusted P | Analysis | factors | mean | SE | F / t | comparison | P/adjusted P | Analysis | factors | mean | SE | F / t | comparison | P/adjusted P |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Comparison of the percentage means of the major compounds occurring in the rainy and the dry seasons of each individual plant | ||||||||||||||||||||
| spathulenol | α-cadinol | τ-muurolol | ||||||||||||||||||
| t-tests | 22EO (A) | t = 2.345 df = 11 |
0.0388 | t-tests | 22EO (A) | t = 2.436 df = 11 |
0.0331 | t-tests | 22EO (A) | t = 2.311 df = 11 |
0.0412 | |||||||||
| RS | 7.307 | 0.6993 | RS | 5.466 | 0.4597 | RS | 4.793 | 0.3727 | ||||||||||||
| DS | 5.428 | 0.2749 | DS | 7.012 | 0.4258 | DS | 5.993 | 0.3538 | ||||||||||||
| 80EO (B) | t = 0.8917 df = 10 |
0.3935 | 80EO (B) | t = 2.912 df = 10 |
0.0155 | 80EO (B) | t = 3.012 df = 10 |
0.0131 | ||||||||||||
| RS | 8.51 | 0.8227 | RS | 5.695 | 0.5491 | RS | 4.593 | 0.3467 | ||||||||||||
| DS | 7.748 | 0.2299 | DS | 7.533 | 0.3114 | DS | 5.79 | 0.194 | ||||||||||||
| 530EO (C) | t = 1.314 df = 12 |
0.2135 | 530EO (C) | t = 2.257 df = 11 |
0.0453 | 530EO (C) | t = 2.349 df = 11 |
0.0385 | ||||||||||||
| RS | 4.199 | 0.4534 | RS | 4.83 | 0.6215 | RS | 4.439 | 0.46 | ||||||||||||
| DS | 5.162 | 0.5943 | DS | 2.948 | 0.309 | DS | 5.833 | 0.3482 | ||||||||||||
| Comparison of the percentage means of major compounds occurring in the rainy season among the three individual plants | ||||||||||||||||||||
| spathulenol | α-cadinol | τ-muurolol | ||||||||||||||||||
| ANOVA 1-factor | F (2,19) = 10.82 | 0.0007 | ANOVA 1-factor | F (2,17) = 2.068 | 0.1571 | ANOVA 1-factor | F (2,19) = 0.1554 | 0.8572 | ||||||||||||
| 22EO (A) | 6.948 | 0.7043 | A-B | 0.2564 | 22EO (A) | 5.466 | 0.4597 | A-B | 0.9444 | 22EO (A) | 4.959 | 0.3629 | A-B | 0.8446 | ||||||
| 80EO (B) | 8.51 | 0.8227 | A-C | 0.0154 | 80EO (B) | 5.695 | 0.5491 | A-C | 0.2702 | 80EO (B) | 4.593 | 0.3467 | A-C | 0.9728 | ||||||
| 530EO (C) | 4.199 | 0.4534 | B-C | 0.0007 | 530EO (C) | 4.37 | 0.4825 | B-C | 0.179 | 530EO (C) | 4.823 | 0.5533 | B-C | 0.9354 | ||||||
| Comparison of the percentage means of major compounds occurring in the dry season among the three individual plants | ||||||||||||||||||||
| spathulenol | α-cadinol | τ-muurolol | ||||||||||||||||||
| ANOVA 1-factor | F (2,16) = 5.911 | 0.012 | ANOVA 1-factor | F (2,12) = 7.055 | 0.0094 | ANOVA 1-factor | F (2,16) = 0.06998 | 0.9327 | ||||||||||||
| 22EO (A) | 6.004 | 0.621 | A-B | 0.0767 | 22EO (A) | 7.334 | 0.3408 | A-B | 0.6579 | 22EO (A) | 5.653 | 0.4532 | A-B | 0.9609 | ||||||
| 80EO (B) | 7.748 | 0.2299 | A-C | 0.506 | 80EO (B) | 7.76 | 0.2616 | A-C | 0.0458 | 80EO (B) | 5.79 | 0.194 | A-C | 0.9333 | ||||||
| 530EO (C) | 5.162 | 0.5943 | B-C | 0.0104 | 530EO (C) | 6.03 | 0.4011 | B-C | 0.0094 | 530EO (C) | 5.833 | 0.3482 | B-C | 0.9963 | ||||||
| Comparison of the percentage means of the major compounds occurring in the rainy and the dry seasons of the three individual plants | ||||||||||||||||||||
| spathulenol | α-cadinol | τ-muurolol | ||||||||||||||||||
| t-tests | t = 0.1277 df = 39 |
0.8991 | t-tests | t = 2.674 df = 39 |
0.0109 | t-tests | t = 2.22 df = 46 |
0.0314 | ||||||||||||
| Individuals RS | 6.374 | 0.5252 | Individuals RS | 5.049 | 0.3869 | Individuals RS | 4.838 | 0.2323 | ||||||||||||
| Individuals DS | 6.289 | 0.3799 | Individuals DS | 6.602 | 0.4359 | Individuals DS | 5.695 | 0.3173 | ||||||||||||
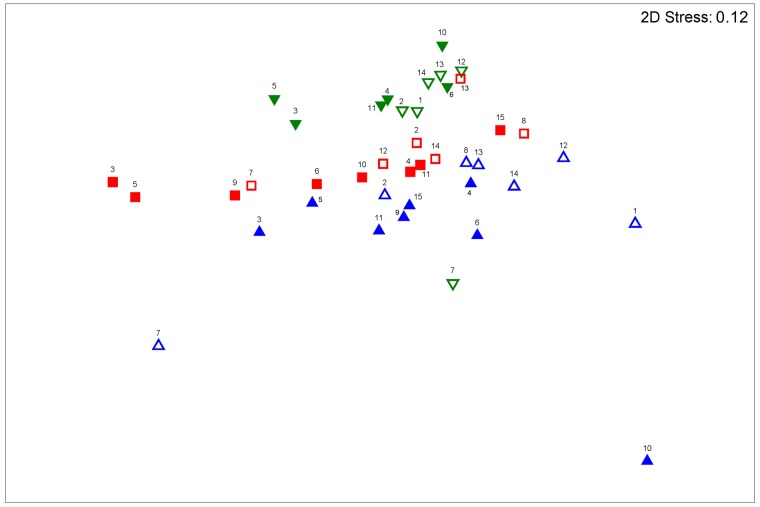
NMDS (stress = 0.12), in association with an ANOSIM, showed that the abundance and composition of terpenes in the samples varied significantly among the three Iryanthera individuals (RGlobal = 0.284, p = 0.001) and also between DS and RS (RDS x RS = 0.185; p = 0.038). Variation was determined according to calculations, because the values of the R statistic computed by ANOSIM vary from 0 to 1, with values closer to 0 representing indistinguishable groups of data. There were significant differences between abundance and the composition of terpenes between the two tested cases (all analyses were significant). However, the distinction between groups was not very evident. Considering only the three Iryanthera individuals tested, the groups that could be best distinguished (with respect to composition and abundance of terpenes) corresponded to individuals 80EO and 530EO (R80EO vs. 530EO = 0.373, p = 0.001), followed by individuals 80EO and 22EO (R80EO vs. 220EO = 0.342, p = 0.001). On the other hand, the group of samples of individuals 22EO and 530EO could scarcely by distinguished from each other (R22EO vs. 530EO = 0.183, p = 0.008). These results are shown in Fig 3.
Fig 3. Ordination (NMDS) analysis of the 41 essential oils derived from three Iryanthera polyneura individual plants, using a Bray-Curtis Index (upward triangles = 22EO; downward-facing triangles = 80EO; squares = 530EO; shaded figures = rain season; open figures = dry season).
The matrix used for the analysis was based on the percentages of the 59 terpenes found in the essential oils.
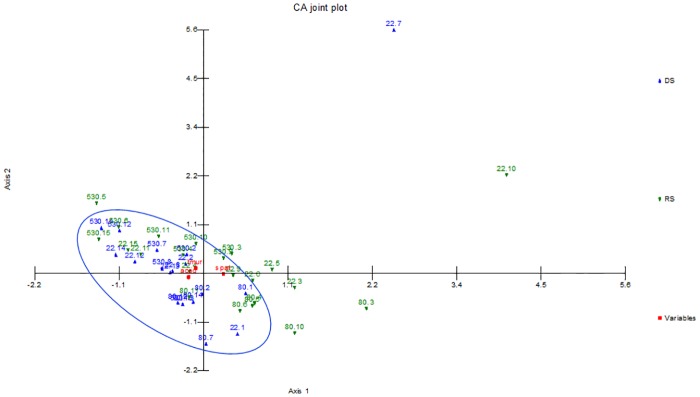
CA and CCA were performed to characterize oil composition according to the percentage of the major compounds spathulenol, τ-muurolol and α-cadinol. The utilization of such statistical tools makes it possible to observe spatial and temporal variations in the chemical composition of the oils considered [14, 23]. Further analyses allowed researchers to correlate geographical and environmental factors [24–26].
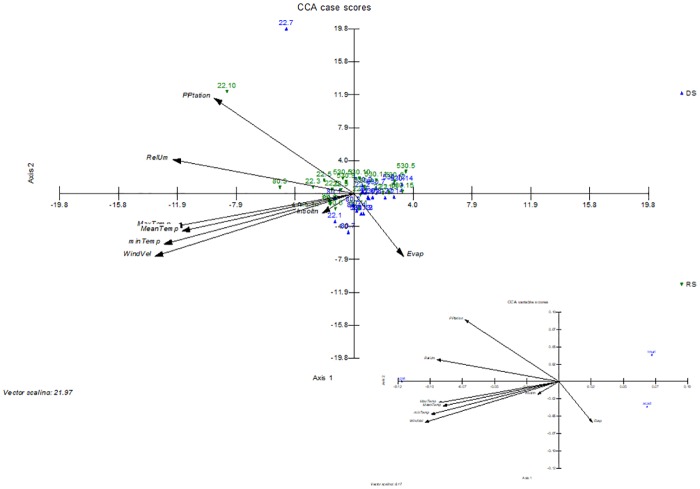
Fig 4 shows a CA analysis made with the EOs from the three individuals (22EO, 80EO and 530EO) that has been based on the major compounds present on the oils. A cumulative percentage of 100% is shown on the second axis. The analysis revealed that the major compounds from the EOs varied based on whether they were obtained from leaves collected in the DS or the RS. The CCA analysis, shown in Fig 5, resulted in a cumulative constraint percentage of 100% on the second axis, and also showed a gradient, which was formed on the first axis with the samples of EOs obtained from the leaves collected in the DS and RS. Also, the CCA (Fig 5) showed that τ-muurolol and α-cadinol, which were more present in higher concentrations in the DS, according to the ANOVA first performed, are particularly influenced by the evaporation, rather than other climatic factors. Although the total amount of spathulenol does not change between the DS and RS (Table 4), the slight tendency of individual 22EO to have higher amounts of spathulenol during RS seemed to have been influenced by the precipitation and relative humidity (which are usually frequent during the RS), temperature and wind velocity.
Fig 4. Correspondence analysis made with 41 essential oils (cases) obtained from the leaves of three Iryanthera polyneura individual plants (22EO, 80EO and 530EO).
The major compounds spathulenol, τ-muurolol and α-cadinol are the variables. The numerical sequence (1 to 15) corresponds to different collection dates for each individual, fand increases according to chronology. Ellipses highlight the discriminated groups.
Fig 5. Canonical correspondence analysis made using 41 essential oils (cases) and three major compounds spathulenol, τ-muurolol and α-cadinol (variables).
Oils were derived from the leaves of three Iryanthera polyneura individual plants (22EO, 80EO and 530EO). The numerical sequence (1 to 15) corresponds to different collection dates for each individual, following a chronological order.
Conclusions
The findings presented have shown that there is a variation in the occurrence of the major compounds comprising EO from I. polyneura individual plants. The compounds spathulenol, τ-muurolol and α-cadinol varied among individuals and that the percentage variation was related to season and climatic changes.
Supporting information
(XLSX)
(XLSX)
(XLSX)
Data Availability
All relevant data are within the manuscript and its Supporting Information files.
Funding Statement
The work was partially funded by Fundação de Amparo à Pesquisa do Estado de São Paulo, grant #2008/58704-6 awarded to Ivana Barbosa Suffredini. A scholarship (grant#001) was granted to Erika Ramos Martins, by Universidade Paulista – UNIP. The funders did not play any role in the study, data collection, analyses, decision on publishing or preparation of the manuscript. There was no additional external funding received for this study.
References
- 1.Flora do Brasil. www.floradobrasil.jbrj.gov.br. Accessed in 02/02/2019.
- 2.Burow M, Halkier BA. How does a plant orchestrate defense in time and space? Using glucosinolates in Arabidopsis as case study. Curr Opin Plant Biol. 2017;38(1): 142–147, 2017. 10.1016/j.pbi.2017.04.009. [DOI] [PubMed] [Google Scholar]
- 3.Friberg M, Schwind C, Roark LC, Raguso RA, Thompson JN. Floral scent contributes to interaction specificity in coevolving plants and their insect pollinators. J Chem Ecol. 2014;40(9): 955–65. 10.1007/s10886-014-0497-y [DOI] [PubMed] [Google Scholar]
- 4.Verpoorte R. Secondary metabolism In: Verpoorte R. & Alfermann A.W. (eds.). Metabolic engineering of plant secondary metabolism, 1 ed Netherlands: Kluwer Academic Publishers, p. 1–29, 2000. [Google Scholar]
- 5.Hartmann T. From waste products to echochemicals: Fifty years research of plant secondary metabolism. Phytochemistry. 2007;68(22–24): 2831–2846. 10.1016/j.phytochem.2007.09.017 [DOI] [PubMed] [Google Scholar]
- 6.Fürstenberg-Hägg J, Zagrobelny M, Bak S. Plant defense against insect herbivores. Int J Mol Sci. 2013;14(5): 10242–10297. 10.3390/ijms140510242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rodrigues WA. Revisão taxonômica das espécies de Virola aublet (Myristicaceae) do Brasil. Acta Amaz. 1980;10(1Suppl.1): 5–127. [Google Scholar]
- 8.Gottlieb OR. Chemical studies on medicinal Myristicaceae from Amazonia. J Ethnopharmacol. 1979;1(4): 309–323. 10.1016/s0378-8741(79)80001-x [DOI] [PubMed] [Google Scholar]
- 9.Shultes RE, Holmsted B. De plantis toxicariis e Mundo Novo tropicale commentationes. VIII. Miscellaneous notes on Myristicaceous Plants of South America. Lloydia. 1971;34(1): 61–78. [PubMed] [Google Scholar]
- 10.Shultes RE. De plantis toxicariis e Mundo Novo tropicale commentationes XXXV: Miscellaneous notes on biodynamic Plants of the Northwest Amazon. J Ethnophamarcol. 1985;14(2–3): 125–158. 10.1016/0378-8741(85)90083-2 [DOI] [PubMed] [Google Scholar]
- 11.Revilla J. Plantas Úteis da Bacia Amazônica. vol. 1 Manaus: Sebrae-AM/INPA; p.350–3252. 2002. [Google Scholar]
- 12.Silva DH, Davino SC, Barros SB, Yoshida M. Dihydrochalcones and flavonolignans from Iryanthera lancifolia. J. Nat. Prod., 1999; 62(11): 1475–8. 10.1021/np980509w [DOI] [PubMed] [Google Scholar]
- 13.Almeida MEL, Braz Filho R, Von Bulow MV, Corrêa JJL, Gottlieb OR, Maia JGS, et al. Diarylpropanoids from Iryanthera polyneura. Phytochemistry. 1979;18: 1015–1016. 10.1111/j.1523-1739.2005.00705.x [DOI] [Google Scholar]
- 14.Suffredini IB, Sousa SRN, Frana AS, Diaz IEC, Suffredini HB, Paciencia MLB. Multivariate Analysis of the Terpene Composition of Iryanthera ulei Warb. (Myristicaceae) and Its Relationship to Seasonal Variation Over a Two-Year Period. J Essent Oil-Bear Plant. 2016; 19(6): 1380–1393. 10.1080/0972060X.2016.1215264 [DOI] [Google Scholar]
- 15.Santos PRD, Moreira DL, Guimarães EF, Kaplan MAC. Essential oil analysis of 10 Piperaceae species from the Atlantic forest. Phytochemistry. 2001;58(4): 547–551. 10.1016/S0031-9422(01)00290-4 [DOI] [PubMed] [Google Scholar]
- 16.Adams RP. Identification of essential oil components by gas chromatography/mass spectrometry. 4ed Illinois: Allured Publishing Corporation, 804 p. 2007. [Google Scholar]
- 17.Van Den Dool HKD. A generalization of the relation index system including liner temperature programmed gas-liquid partition chromatography. J Chromatogr A. 1963;11: 463–467. 10.1016/S0021-9673(01)80947-X [DOI] [PubMed] [Google Scholar]
- 18.Fisch G, Marengo JA, Nobre CA. Clima da Amazônia. At: http://climanalise.cptec.inpe.br/~rclimanl/boletim/cliesp10a/fish.html, accessed in 03/07/2016.
- 19.Ab’Sáber A. Dossiê Amazônia brasileira I. In: Ab’Sáber A.: Problemas da Amazônia brasileira. Estudos Avançados. 2005;19(53): 5–35. [Google Scholar]
- 20.Zar JH. Biostatistical Analysis, Fourth ed Prentice-Hall Inc, New Jersey: 929 p. 1999. [Google Scholar]
- 21.Clarke KR. Non-parametric multivariate analyses of changes in community structure. Austr J Ecol 1993;18(1): 117–143. 10.1111/j.1442-9993.1993.tb00438.x [DOI] [Google Scholar]
- 22.Chiu S, Wang T, Belski M, Abourashed EA. HPLC-Guided Isolation, Purification and Characterization of Phenylpropanoid and Phenolic Constituents of Nutmeg Kernel (Myristica fragrans). Nat Prod Commun. 2016;11(4): 483–488. [PMC free article] [PubMed] [Google Scholar]
- 23.Pinheiro CG, Machado CM, Amaral LP, Silva DT, Almeida CAA, Longhi SJ, et al. Seasonal variability of the essential oil of Hesperozygis ringens (Benth.) Epling. Braz J Biol. 2016;76(1): 176–184. 10.1590/1519-6984.16314 [DOI] [PubMed] [Google Scholar]
- 24.Hanlidou E, Karousoua R, Lazarib D. Essential-Oil Diversity of Salvia tomentosa Mill. in Greece. Chem Biodivers. 2014;11(8): 1205–1215. 10.1002/cbdv.201300408 [DOI] [PubMed] [Google Scholar]
- 25.Pitarokilia D, Constantinidis T, Saitanis C, Tzakou O. Volatile Compounds in Thymus sect. Teucrioides (Lamiaceae): Intraspecific and Interspecific Diversity, Chemotaxonomic Significance and Exploitation Potential. Chem Biodivers. 2014;11(4): 593–618. 10.1002/cbdv.201300333 [DOI] [PubMed] [Google Scholar]
- 26.Ragusa A, Centonze C, Grasso ME, Latronico MF, Mastrangelo PF, Fanizzi FP, et al. Composition and Statistical Analysis of Biophenols in Apulian Italian EVOOs. Foods, 2017;6(10): 90 10.3390/foods6100090 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(XLSX)
(XLSX)
(XLSX)
Data Availability Statement
All relevant data are within the manuscript and its Supporting Information files.