Abstract
The aim of the study was the evaluation of a three-step method for the selection of bacterial strains capable of producing surfactin. The procedure consisted of the following steps: 1.blood agar test, 2. measurement of the surface tension (ST) of the medium using the du Nouy method before and after submerged culture, 3. qualitative and quantitative assessment of surfactin by HPLC. Forty five Bacillus subtilis natto strains producing haemolysis zones (≥3mm) were selected. Nineten of them reduced ST of the medium to ≤ 40 mN/m; in six cases, the reduction was as much as 50%. All indicated strains produced surfactin. Positive correlations (p <0.5) between the percentage reduction of ST of the medium and surfactin concentration (r = 0.44), indicate that this parameter is determinant of the ability to synthesize this compound. The blood agar test has been shown to be useful only as a pre-selection criterion for surfactin producers (18 strains selected by this method reduced ST by only ≤30%). The proposed selection strategy proved effective and made it possible to select the BS15 strain that reduced the ST of the medium to 30.56 ± 0.15 mN/m and simultaneously provided a high concentration of surfactin compared to other strains.
Introduction
Surfactants (surface active agents) are a group of compounds widely used in industry, agriculture and in households as a component of cleaning agents, medicines and cosmetics [1]. The global market for surfactants has been estimated at approximately USD 30.65 billion in 2015. It is forecasted that due to the continuous upward trend (around 4.4% per year) it may reach USD 39.69 billion by 2021 [2]. The majority of synthetic surfactants are produced using chemical methods from petrochemical raw materials, which poses a toxicological threat to living organisms, especially in aquatic ecosystems [3–5]. An alternative production method is biosynthesis carried out with the use of specialized microorganisms capable of producing biosurfactants with lower toxicity, high resistance to extreme pH, temperature or salinity, and, most importantly, increased biodegradability [6–11]. Due to their ability to reduce surface tension (at the water-air interface) and interfacial tension (at the water-oil interface), biosurfactants are among the most versatile process chemicals [12,13]. Their surface-active properties result from the amphiphilic structure of the molecule, which favors the aggregation of these compounds at the interface [14,15]. The decrease in surface tension is caused by the increase in biosurfactant concentration and the formation of aggregated amphipathic molecules, so-called micelles. The surfactant concentration at which the micelles are formed is referred to as the critical micellization concentration (CMC) [11,16]. This parameter corresponds to the minimum surfactant concentration that is necessary to achieve the maximum reduction of surface tension [11,16,17]. The CMC value indicates the efficiency of the surface active compound, while its effectiveness is related to the measurement of surface tension and interfacial tension [16]. Surfactin is one of the most effective surfactants with a high application potential [18–21]. This compound is an interesting bioproduct due to its properties. Surfactin shows antibacterial activity towards Mycoplasma sp. [22], it is an antineoplastic agent for breast, colon cancer and leukemia [23]. It also exhibits emulsifying effect and thus accelerates biodegradation of hydrocarbons in oil contaminated areas [24]. Therefore, surfactin, a lipopeptide, can successfully replace synthetic compounds in many branches of the economy. This biosurfactant reduces the surface water tension from 72 to 27 mN/m and the interfacial tension of water/n-hexane from 43 mN/m to 1 mN/m [10,25,26]. In addition, the CMC of surfactin is very low, i.e. 0.017 g/L in water [27]. The surfactin properties listed above result from its chemical structure [28]. This compound consists of a peptide loop of seven amino acids (L-Glu, L-Leu, D-Leu, L-Val, L-Asp, D-Leu, L-Leu), and a hydrophobic fatty acid chain thirteen to fifteen carbons long. L-Glu, D-Leu, L-Asp, D-Leu amino acids are the molecular backbone of the compound while others can be exchanged for L-Val L-Ile, L-Leu or L-Ala [29]. Hence, surfactin is produced by microorganisms as a mixture of closely related isoforms that differ in the length and branching of the fatty acid side chains as well as in the configuration of amino acids in the peptide ring. The structure of surfactin, which depends on the composition of the culture medium and the specifics of the producer himself, largely determines the surface-active properties of this compound [20,30,31]. Surfactin, like other biosurfactants, is produced as a secondary metabolite in the stationary phase of microbial growth [28,32]. It is not necessary for the growth and development of microorganisms, but its synthesis may increase their adaptability to given environmental conditions and provide an alternative defense mechanism [33].
A special role in the biosynthesis of lipopeptide biosurfactants is played by bacteria of the genus Bacillus (B. subtilis, B. pumilus, B. myvensis, B. licheniformis, B. amyloliquefaciens). These bacteria produce peptide synthetases and/or polyketide synthetases and synthesise surfactin derivatives with different chemical structure and properties [20,34,35]. It should be noted that most reports on surfactin biosynthesis refer to Bacillus subtilis as its main producer [20,36,37]. Despite numerous studies on surfactin biosynthesis since 1968, the production of this compound on an industrial scale still involves large financial expenses. The costs of media components can account for up to 50% of production expenditure while the efficiency of microbiological biosynthesis is still low [38–40]. This is a significant obstacle limiting the spread of this technology and commercial use of biosurfactants [10]. In order to develop a highly efficient surfactin biosynthesis process using Bacillus sp., effective selection strategies should first be developed to obtain strains capable of biosynthesis of isoforms that significantly reduce surface tension. So far several producers have been identified, mainly within the Bacillus subtilis species, including ATCC 21332, DSM 3256, DSM 3258 strains. The surfactin yield (g/L) reported for these strains cultivated on synthetic media was ca. 2.39 ± 0.9; 1.79 ± 0.8, 1.6 ± 0.11, respectively [29]. However, the yield of surfactin produced by a strain depends to a large extent on the culture conditions and methods, and the availability of nutrients in the medium [12]. It should be emphasized that the strains deposited in collections account for only 1% of the total microflora living in the natural environment [41]. The search for new native strains, acquiring and developing knowledge about their physiology, metabolism and genetics is the key to an efficient microbial bioconversion of food substrates (especially by-products of agro-food processing) to surfactin isoforms [12,28,34,36,42,43]. Effective screening of microorganisms requires well-chosen experimental and analytical methods that guarantee a quick and efficient assessment of metabolic characteristics of isolated strains. So far, many methods have been developed for the qualitative and quantitative selection of microorganisms in the production of biosurfactants, including the measurement of surface, interfacial and emulsifying activity of bioproducts, oil spreading method, drop collapse test or more advanced, verifying the products, such as high performance liquid chromatography [44,45]. It should be noted, however, that only their proper combination at subsequent stages of the selection enables efficient and low-budget estimation of the ability of surfactin biosynthesis by native strains.
The goal of this study was to evaluate the effectiveness of selected methods for screening Bacillus subtilis strains isolated from fermented food products for the production of surfactin isoforms. The ability of surfactin biosynthesis was verified during three-stage selection. First, the isolated strains were evaluated for the intensity of erythrocyte lysis on solid media containing defibrinated sheep blood (blood agar test), which indicated the likely biosurfactant biosynthesis capabilities. Next, strains forming hemolysis zones were used for the biosynthesis of surfactants on model liquid medium (SmF), and the surfactin biosynthesis ability was evaluated on the basis of the degree of reduction of the surface tension (ST) of the medium after cultivation. The final step in strain selection, which verified whether screening was successful, was confirmation of the ability to synthesize surfactin isoforms using high performance liquid chromatography. During the study, an attempt was made to assess the relationship between the concentration of surfactin produced by isolated strains and the percentage of surface tension reduction in the post-culture medium.
Materials and methods
Isolation of Bacillus subtilis natto strains. Blood agar test
The bacteria were isolated from a natto food product (Ton Color, Poland), whose basic ingredient is soya beans subjected to prior fermentation by Bacillus subtilis natto strains. The material (approximately 5 g) was homogenized in a Stomacher bag, equipped with a side membrane, with the addition of 20 ml of sterile 0.9% NaCl. One ml of the filtrate was taken and a series of 10-fold dilutions were performed in sterile 0.9% NaCl in the range of 10−1 to 10−7. Samples of 10−6 and 10−7 dilutions were plated on agar medium supplemented with 5% defibrinated sheep blood (Columbia Blood Agar, Poland). The medium was prepared by dissolving 43 g of dry base mixture (Columbia Agar Base, Grasso Biotech, Poland) containing casein hydrolyzate (5.0g), meat extract (8.0g), yeast extract (10.0g), sodium chloride (5.0g), corn starch (1.0g) and agar (14.0g) in 1L distilled water, while stirring and heating the solution to 90°C. Once the components have completely dissolved, the medium was sterilized in an autoclave for 15 min at 121°C, and then enriched with 50 ml of defibrinated sheep blood under aseptic conditions. The pH of the medium was 7.3 ± 0.2. The plates after inoculation were incubated for 96 h at 30°C. The strains showing the ability to erythrocyte lysis, manifested by the occurrence of clear zones (> 3mm) around the colonies, had been deposited on agar slopes (bacteriological tryptone peptone 5g/L, 2.5 g/L yeast extract, 1g/L glucose, 15g/L agar, pH 7.2–7.4) and subsequently incubated for 96 h at 30°C. All isolated strains were deposited in cryobanks (Grasso Biotech, Poland) and stored at -20°C until analysis. Differences between isolated B. subtilis strains were confirmed by analysis of allele sequence variability in the pta (phosphate acetyltransferase) locus using the MLST (multilocus sequence typing) method.
Submerged fermentation (SmF) conditions
The SmF of the isolated strains was carried out in two stages. In the first step, an inoculum was obtained, which was transferred to the mineral medium [46] enabling the biosynthesis of surfactin. In order to obtain an inoculum, 100 ml of nutrient broth (bacteriological tryptone peptone 5 g/L, yeast extract 2.5 g/L, glucose 1 g/L, pH 7.2–7.4) was inoculated under aseptic conditions with selected bacterial strains. Then the culture was grown for 24 h with shaking (70 rpm) in a water bath at 37°C. Next, 2.5 mg of inoculum obtained in this way was transferred to Cooper's medium (glucose 40g/L; NH4NO3 4g/L; KH2PO4 4.08 g/L; Na2HPO4 x2H2O 7.12g/L; MgSO4 x7H2O 0.2 g/L; CaCl2 0.0008g/L; FeSO4 x 7H2O 0.0011g/L; EDTA 0.0012 g/L, pH 7.0). SmF cultures with shaking (70 rpm) were carried out in triplicate in 250 ml flasks for 120 h at 37°C [46].
Measurement of biomass concentration
In order to determine the biomass concentration of selected Bacillus subtilis natto strains after the culture, 5 ml of cell suspension was taken and centrifuged twice (10 min, 8000 g, MPW-260R centrifuge, MPW-Med Instruments, Poland); the precipitate was rinsed with 5 ml of 0.9% NaCl. Finally, cell biomass was suspended in 5 ml of 0.9% NaCl and the optical density (OD600) was measured using an UV-VIS spectrophotometer (Pharo 300, Merck) [40]. The biomass concentration of the analyzed strains [mg/ml] was determined using a curve showing the dependence of the optical density (OD) of liquid test culture (Bacillus subtilis no. ŁO820; from the Collection of pure cultures of industrial strains, Łódź University of Technology, Poland) on the amount of dry cell mass. The biomass was dried to a constant mass using a weighting dryer (RADWAG, WPS-30S) at 105°C and 20 s sampling time.
Measurement of surface tension
The surface tension of the culture medium before and after cultivation was measured by the Du-Nouy-Ring method [44] using a tensiometer (model PI-MT1M, Donserv, Poland). This method measured the force required to detach the platinum ring of radius 20.5 mm from the surface of the culture medium at room temperature. Between the measurements, the platinum ring was rinsed with ethanol and then allowed to dry. All measurements were taken in the post-culture medium after biomass removal by centrifugation (2400 g, 15 min, 4⁰C). In order to increase the representativeness of the results, the measurements were repeated five times.
Extraction and determination of surfactin concentration
Qualitative and quantitative assessments of surfactin isoforms were preceded by extraction using affinity chromatography with the solid-phase extraction (SPE) system. For extraction we used Bond Elut C18 columns (Agilent Technologies), which are retentive for non-polar compounds. Surfactin was extracted from the post-culture medium after removing the biomass of bacteria (2400g, 15 min, 20°C). Prior to the extraction, the SPE column was conditioned with methanol and stabilized with distilled water (as recommended by the manufacturer). Surfactin was extracted by applying 20 mL of culture medium to the SPE column. Isolation was carried out at a flow rate of 1 drop per second. The column bed was then rinsed with 5% methanol and dried thoroughly under reduced pressure (SPE system, Agilent Technologies). Elution of surfactin was carried out using HPLC grade methanol. Before injection, the solution was filtered through a membrane filter (pore size 0.22 μm). The chromatographic separation was performed by HPLC (Model 1260 chromatograph, Agilent Technologies) with DAD detection, according to the method presented by [29]. The Poroshell 120 EC-C18 column (4.5 x 150 mm, 2.7 μm) equipped with a guard column (Poroshell 120 EC-C18, 3.0 x 5 mm, 2.7 μm) was used. Acetonitrile and 3.8 mM trifluoroacetic acid (80:20, v/v) were used as the mobile phase with isocratic flow of 1 mL/min at 25°C. Surfactin isoforms were detected at 205 nm (peptide bond absorption wavelength). Quantitative calculations were performed using the external standard method (ESTD) with methanolic surfactin solution obtained from Bacillus subtilis (Sigma-Aldrich).
Statistical analysis
Experiments were performed in triplicate (n = 3). The results are presented as mean ± standard deviation (SD). Data analysis was carried out using the Statistica software ver. 13.3.
Results and discussion
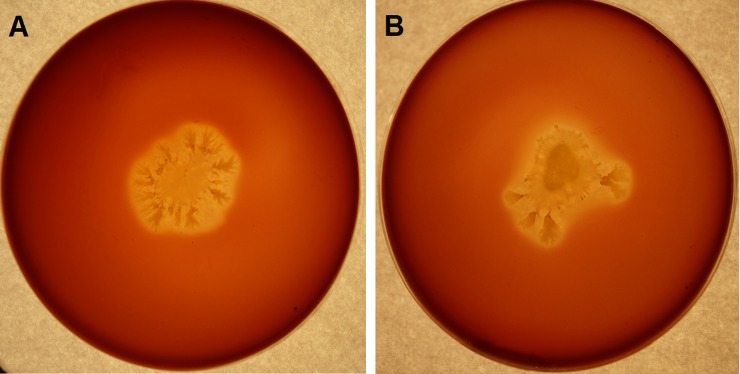
The interest in lipopeptide surfactants, due to their wide application potential, forced the development of many screening methods that could verify potential producer strains. Because these methods have various constraints, e.g. low selectivity or specificity for a selected group of biosurfactants, only their proper combination in a specific strategy can be successful, i.e. allow for acquisition of completely new strains capable of producing biosurfactants [44,45]. The strategy for selection of Bacillus subtilis natto strains synthesizing surfactin proposed in this paper consisted of three key stages whose main objective was a fast and effective screening. In the first stage, we investigated the ability of isolated strains to produce hemolysis zones on a solid medium enriched with defibrinated sheep blood after 96 h incubation at 30°C, because the lysis of erythrocytes by the strain being analyzed is considered to give an indication on biosurfactant production [47]. At this stage, 45 strains having clear zones (> 3 mm) around the colonies were selected (Fig 1).
Fig 1.
Hemolysis zone around a single BS22 (A) and BS25 (B) colony on a solid medium with 5% defibrinated sheep blood (Columbia Blood Agar).
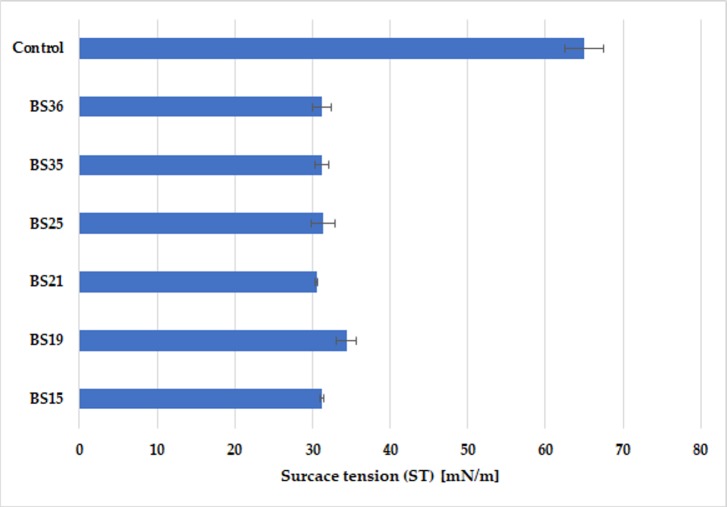
The next step in verifying selected strains as potential surfactin producers was the measurement of surface tension (ST) of Cooper's medium, before and after the submerged culture (SmF). This method was widely used in many studies as a measure of the surface-active properties of microbiologically synthesized biosurfactants [43,48,49]. Surface tension measurements performed in our study clearly indicated a decrease in the value of this parameter by about 50%, i.e. to ca. 30 mN/m, in post-culture media of as many as 6 strains: BS15, BS19, BS 21, BS25, BS35, BS36 (Table 1,Fig 2).
Table 1. Average biomass concentration of selected strains after submerged culture (SmF).
Average percentage decrease in the surface tension of the culture medium during the cultivation.
| Strains | Biomass concentration [mg/ml] | Surface tension decrease [%] | ||||
|---|---|---|---|---|---|---|
| Mean | ± | SD | Mean | ± | SD | |
| BS1 | 0.37 | ± | 0.081 | 13.74 | ± | 7.130 |
| BS2 | 1.20 | ± | 0.053 | 21.23 | ± | 0.335 |
| BS3 | 0.63 | ± | 0.189 | 40.54 | ± | 10.784 |
| BS4 | 0.80 | ± | 0.050 | 22.37 | ± | 0.598 |
| BS5 | 0.84 | ± | 0.171 | 15.73 | ± | 0.179 |
| BS6 | 0.84 | ± | 0.177 | 18.56 | ± | 2.994 |
| BS7 | 0.95 | ± | 0.246 | 36.84 | ± | 7.956 |
| BS8 | 0.80 | ± | 0.080 | 4.53 | ± | 1.102 |
| BS9 | 0.44 | ± | 0.013 | 30.82 | ± | 1.256 |
| BS10 | 0.44 | ± | 0.007 | 33.75 | ± | 0.478 |
| BS11 | 0.80 | ± | 0.007 | 32.26 | ± | 1.854 |
| BS12 | 1.02 | ± | 0.181 | 29.27 | ± | 2.346 |
| BS13 | 1.10 | ± | 0.017 | 14.82 | ± | 1.242 |
| BS14 | 0.58 | ± | 0.089 | 32.39 | ± | 2.843 |
| BS15 | 0.46 | ± | 0.008 | 52.77 | ± | 0.404 |
| BS16 | 0.81 | ± | 0.031 | 28.01 | ± | 0.311 |
| BS17 | 0.51 | ± | 0.044 | 32.78 | ± | 1.048 |
| BS18 | 0.98 | ± | 0.027 | 24.93 | ± | 2.905 |
| BS19 | 0.78 | ± | 0.058 | 49.34 | ± | 1.756 |
| BS20 | 0.75 | ± | 0.075 | 31.18 | ± | 13.444 |
| BS21 | 0.76 | ± | 0.002 | 54.12 | ± | 0.229 |
| BS22 | 1.01 | ± | 0.084 | 35.81 | ± | 1.373 |
| BS23 | 0.57 | ± | 0.011 | 41.99 | ± | 6.636 |
| BS24 | 0.60 | ± | 0.062 | 42.75 | ± | 1.931 |
| BS25 | 1.00 | ± | 0.381 | 53.69 | ± | 0.750 |
| BS26 | 0.55 | ± | 0.045 | 28.34 | ± | 1.733 |
| BS27 | 0.41 | ± | 0.007 | 28.34 | ± | 3.837 |
| BS28 | 0.44 | ± | 0.062 | 10.76 | ± | 3.121 |
| BS29 | 0.61 | ± | 0.012 | 13.66 | ± | 7.577 |
| BS30 | 0.26 | ± | 0.028 | 35.29 | ± | 5.478 |
| BS31 | 0.32 | ± | 0.004 | 3.69 | ± | 3.694 |
| BS32 | 0.72 | ± | 0.030 | 42.39 | ± | 2.042 |
| BS33 | 1.33 | ± | 0.086 | 20.89 | ± | 4.331 |
| BS34 | 1.01 | ± | 0.187 | 40.22 | ± | 6.064 |
| BS35 | 0.67 | ± | 0.001 | 49.44 | ± | 1.420 |
| BS36 | 0.75 | ± | 0.079 | 49.44 | ± | 2.037 |
| BS37 | 0.69 | ± | 0.021 | 46.23 | ± | 0.185 |
| BS38 | 0.71 | ± | 0.031 | 45.99 | ± | 2.284 |
| BS39 | 1.10 | ± | 0.277 | 46.25 | ± | 0.939 |
| BS40 | 1.34 | ± | 0.047 | 31.17 | ± | 4.535 |
| BS41 | 1.13 | ± | 0.035 | 29.05 | ± | 7.118 |
| BS42 | 0.64 | ± | 0.033 | 45.49 | ± | 1.314 |
| BS43 | 0.52 | ± | 0.062 | 43.49 | ± | 3.066 |
| BS44 | 0.52 | ± | 0.006 | 21.30 | ± | 4.305 |
| BS45 | 0.89 | ± | 0.109 | 44.37 | ± | 1.815 |
Data presented as mean ± SD for each strain (n = 3).
Fig 2. Surface tension (ST) [mN/m] of Cooper’s mineral medium measured before (Control) and after submerged cultivation (SmF) of selected Bacillus subtilis natto strains (BS15, BS19, BS21, BS25, BS35, BS36).
Earlier reports [44,50] suggested that strains that have the ability to reduce the medium surface tension to ≤ 40 mN/m are considered potential biosurfactant producers. Based on this assumption, it should be concluded that approximately 38% of strains selected by the authors of the present study met this criterion. The surface tension of the post-culture medium of the six selected strains was close to 30 mN/m. De Faria et al. [51], Bezza and Chirwa [52], Al Wahaibi et al. [53], Jha et al. [54] also confirmed the ability of the studied microorganisms to lower the ST of the culture medium to this level. On the other hand, about 18 strains of the 45 strains selected in our study showed a decrease in surface tension of the culture medium by only ≤ 30%. This indicates that the ability to form hemolysis zones can not be the only determinant taken into account when assessing the capability to produce biosurfactants by Bacillus subtilis natto strains. Youssef et al. [55] demonstrated that as much as 60% of 205 strains showing erythrolytic capacity reduced ST only to > 60 mN/m. Meanwhile, despite the negative blood agar test, 38% of strains reduced the ST even to ca. 35 mN/m. It was also believed that low correlation (r = -0.15) between surface tension and the occurrence of haemolysis zones proved poor efficiency of this method in the verification of strains capable of producing surfactin. Joshi et al. [43], Hsieh et al. [47], Mulligan et al. [56] concluded that blood agar test can only be used as a preliminary screening of strains, enabling identification of potential surfactant producers. This was due to the limitations of this method resulting from its low specificity associated, eg, with the diffusion of biosurfactants in the agar medium or the action of lytic enzymes (including proteases), which might suggest false negative or false positive results [44]. However, its advantages, i.e. speed (96 h) and low costs, speak for its use in the initial stage of selection.
The final stage of the screening was verification of earlier stages of the selection, i.e. determination of the concentration of surfactin and its isoforms in the medium. HPLC is considered one of the best methods for qualitative and quantitative characterization of surfactin, and therefore it has been used in many studies on microbiological surfactin biosynthesis [29,32,36,40,57]. In our study, 19 strains were selected for the assessment. The selection criterion was the ability to reduce the surface tension by ≥ 35%. The strains BS14 and BS20, which reduced the ST of the medium to a lesser extent (by ca. 32%) were also taken into account. They were a reference point for confirming the hypothesis that there is a relationship between the concentration of surfactin and the percentage decrease in surface tension of the culture medium. Due to the need to compare the analyzed strains for the ability to surfactin biosynthesis, the final results, i.e. the concentration of this compound, are expressed in μg/mg of biomass. Although the average biomass concentration of the tested strains after cultivation was 0.75 ± 0.073 mg/ml, the differences in this parameter between the strains were considerable. For example, while the biomass concentration in the post-culture medium of the BS1 strain was 0.37 ± 0.081 mg/ml, for the BS40 strain it was 1.37 ± 0.047 mg/ml (Table 1). The rate of biomass production is related to the differences in the assimilation of nutrients and results from the adaptability of particular strains to the given culture conditions.
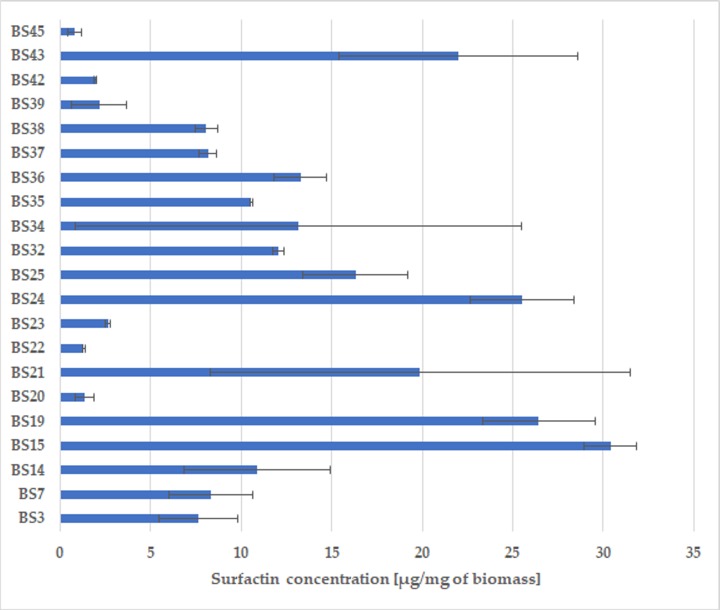
We observed considerable differences between the assessed strains in the surfactin producing abilities. The concentration of the compound produced ranged from 0.82 ± 0.40 μg/mg (BS45) to 30.40 ± 1.44 μg/mg (BS15) (Fig 3).
Fig 3. Average concentration of surfactin [μg/mg] synthesized by selected strains (n = 3).
Error bars represent standard deviation (SD).
Particularly high concentration of synthesized surfactin (apart from the BS15 strain) was also found for the following strains: BS19 (26.45 ± 3.08 μg/mg); BS24 (25.54 ± 2.88 μg/mg) and BS43 (22.00 ± 6.60 μg/mg). However, the standard deviation (SD) of surfactin productivity for BS15 did not exceed 5%, which indicated a good repeatability of the results and distinguished this strain from the others. Thus, the application of the solid phase extraction (SPE) system along with the affinity chromatography and HPLC analysis of the eluate undoubtedly enabled rapid verification of the surfactin biosynthesis capacity. Thanks to the use of SPE columns for the extraction of hydrophobic compounds, the analysis time was significantly shortened and the risk of incorrect results that might occur during the multi-stage isolation procedures was minimized.
Because the main goal of this study was to evaluate the methods that enable the selection of potential surfactin producers, at this stage of the research we have abandoned the assessment of the maximum surfactin production efficiency of the obtained strains, which could be achieved by optimizing the culture parameters. Therefore, it should be emphasized that surfactin yields resulting from the production optimization reported by other authors, should not be directly compared with the surfactin production efficiency observed in this study. In order to maximize surfactin production, many authors Abdel-Mawgoud et al. [8], Amani et al. [58], Jokari et al. [59] adapted the culture conditions (pH, temperature, and especially dissolved oxygen concentration) and the composition of the medium specifically for a given Bacillus subtilis strain. Willenbacher et al. [21] showed that the modification of the mineral Cooper’s medium composition by changing the glucose concentration (8 g/L), supplementing with sodium citrate (0.008 mM) and replacing the nitrogen source with (NH4)SO4 significantly improved the yield of surfactin produced by Bacillus subtilis DSM10T from 0.7 to 1.1g/L. In turn, Hsieh et al. [47] observed that the reference strain ATCC 21332, considered to be an outstanding surfactant producer (even 800 mg/L), was able to produce only 109.5 mg/L on the mineral medium if the fermentation products were constantly removed and the medium was enriched with metal cations. Jajor et al. [40], after testing two strains of Bacillus subtilis, #309 and KB1 natto for the production of surfactin under varying oxygen availability, demonstrated that increased oxygenation reduced surfactin biosynthesis in the culture of strain #309, while this factor acted as a stimulant on the strain isolated from natto.
The results indicate a significant (p <0.05) positive correlation between the percentage reduction of surface tension and the total concentration of surfactin and its isoforms in the post-culture medium of selected strains (Table 2).
Table 2. Relationship between the total concentration of surfactin and its isoforms A-F [μg/mg] and the surface tension decrease [%] of the medium after cultivation of Bacillus subtilis natto strains (n = 63).
| Surfactin concentration [μg/mg] | Surface tension decrease [%] |
|
|---|---|---|
| r | p | |
| Total | 0.44 | 0.000 |
| Isoform A | 0.42 | 0.001 |
| Isoform B | 0.51 | 0.000 |
| Isoform C | 0.37 | 0.003 |
| Isoform D | 0.42 | 0.001 |
| Isoform E | 0.43 | 0.001 |
| Isoform F | 0.46 | 0.000 |
Thus, the percentage decrease in ST can be used as a reliable indicator of the ability of strains to produce surfactin, which would significantly accelerate the selection of potential producers. There are, however, some limitations in the measurement range, as ST decreases with the increase of the biosurfactant concentration only until it reaches the critical micellization concentration (CMC). Once this value is reached, it is not possible to determine the increase in the concentration of surfactant. As a consequence, two strains may reduce ST to critical level and at the same time may differ significantly in terms of surfactin production efficiency [44], which is also evidenced by the results presented here. For example, BS15 and BS25 reduced the ST of the medium by about 53%, whereas the concentration of surfactin synthesized by them was 30.44 ± 1.44 μg/mg and 16.33 ± 2.9 μg/mg, respectively (Table 1, Fig 3).
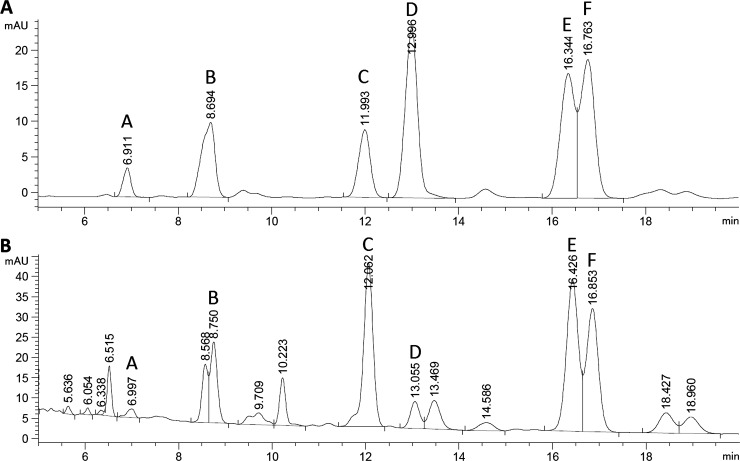
Reports show that surfactin may exist in the form of five [40], six [60], eight [61] and even nine isoforms [62], differing in physicochemical properties. A chromatogram of the surfactin standard (≥95%) produced by Bacillus subtilis (Sigma-Aldrich) indicates the presence of 6 isoforms: A-F (Fig 4). Over 80% of selected strains having the ability to lower the ST of the medium by ≥ 35% (including BS15) synthesized all these isoforms (Table 3, Fig 5).
Fig 4. Chromatogram of surfactin standard (from Bacillus subtilis, ≥95%, HPLC, Sigma-Aldrich, dissolved in methanol) against the chromatogram of surfactin synthesized by strain BS15.
Table 3. Average concentration of surfactin A-F isoforms [μg/mg].
Data presented as mean ± SD for each strain (n = 3).
| Strains | Isoform A | Isoform B | Isoform C | Isoform D | Isoform E | Isoform F |
|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | |
| BS3 | 0.14±0.03 | 0.66±0.26 | 2.34±0.56 | 0.16±0.14 | 2.67±0.52 | 1.67±0.73 |
| BS7 | 0.06±0.02 | 1.26±0.35 | 2.63±0.72 | 0.21±0.06 | 2.90±0.80 | 1.30±0.36 |
| BS14 | 0.13±0.01 | 0.84±0.17 | 3.79±0.99 | 0.28±0.25 | 3.81±1.59 | 2.06±1.06 |
| BS15 | 0.34±0.01 | 2.85±0.03 | 8.37±0.44 | 0.95±0.05 | 9.88±0.56 | 8.00±0.36 |
| BS19 | 0.10±0.01 | 1.67±0.15 | 8.79±0.97 | 1.30±0.18 | 8.53±1.35 | 6.04±0.50 |
| BS20 | 0.04±0.00 | 0.37±0.09 | 0.59±0.28 | 0.00±0.00 | 0.36±0.15 | 0.00±0.00 |
| BS21 | 0.14±0.09 | 2.75±0.89 | 5.35±2.76 | 0.76±0.61 | 7.06±4.80 | 3.83±2.44 |
| BS22 | 0.00±0.00 | 0.30±0.01 | 0.61±0.06 | 0.00±0.00 | 0.35±0.02 | 0.07±0.01 |
| BS23 | 0.03±0.03 | 0.70±0.08 | 0.86±0.06 | 0.00±0.00 | 1.05±0.02 | 0.00±0.00 |
| BS24 | 0.18±0.02 | 1.75±0.26 | 10.13±1.55 | 0.97±0.24 | 8.35±1.07 | 4.15±0.31 |
| BS25 | 0.18±0.07 | 1.18±0.43 | 5.48±0.97 | 0.61±0.06 | 5.55±0.90 | 3.31±0.47 |
| BS32 | 0.21±0.01 | 1.28±0.05 | 4.87±0.04 | 0.32±0.01 | 3.28±0.12 | 2.08±0.07 |
| BS34 | 0.14±0.05 | 0.82±0.68 | 5.15±4.67 | 0.55±0.59 | 3.77±3.51 | 2.75±2.79 |
| BS35 | 0.22±0.01 | 1.16±0.07 | 4.05±0.14 | 0.26±0.01 | 3.15±0.08 | 1.72±0.03 |
| BS36 | 0.21±0.00 | 1.41±0.17 | 5.13±0.42 | 0.36±0.10 | 3.78±0.40 | 2.39±0.34 |
| BS37 | 0.22±0.01 | 1.02±0.05 | 3.37±0.20 | 0.18±0.03 | 2.24±0.10 | 1.15±0.09 |
| BS38 | 0.17±0.01 | 1.02±0.05 | 3.35±0.14 | 0.17±0.03 | 2.24±0.25 | 1.14±0.15 |
| BS39 | 0.07±0.02 | 0.29±0.12 | 0.34±0.31 | 0.00±0.00 | 0.89±0.83 | 0.57±0.53 |
| BS42 | 0.09±0.02 | 1.61±0.16 | 0.26±0.07 | 0.00±0.00 | 0.00±0.00 | 0.00±0.00 |
| BS43 | 0.30±0.06 | 2.39±0.53 | 8.44±2.59 | 0.67±0.26 | 6.00±1.80 | 4.20±1.36 |
| BS45 | 0.06±0.00 | 0.69±0.33 | 0.07±0.07 | 0.00±0.00 | 0.00±0.00 | 0.00±0.00 |
Fig 5. Average percentage of individual isoforms of surfactin produced by selected Bacillus subtilis natto strains.
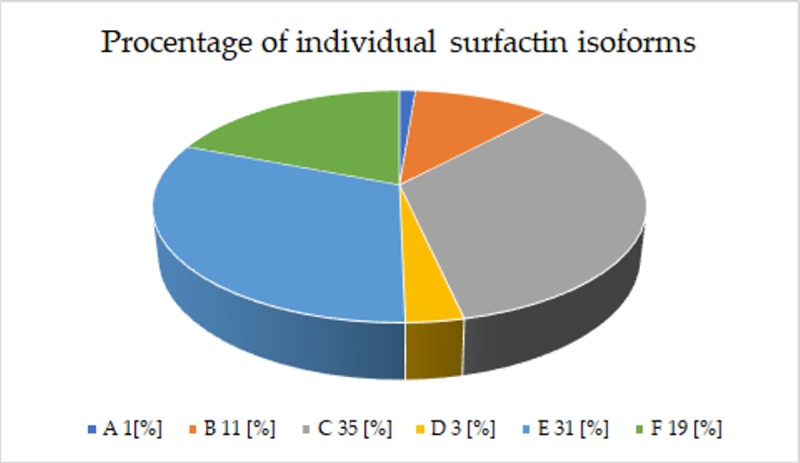
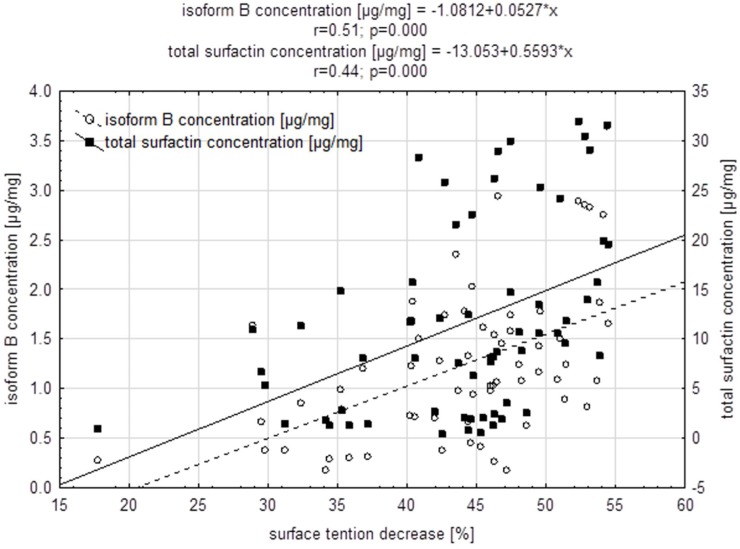
However, the percentages of the individual isoforms of the standard and of those produced by the analyzed Bacillus subtilis natto strains were not identical. The C isoform had the highest percentage (35%) of total concentration of surfactin produced by selected B. subtilis natto strains, followed by E (31%), F (19%), B (11%), D (3%),and A (1%) (Fig 5). The chromatogram of the standard showed that the D isoform had the largest percentage, followed by the isoforms F, E, B, C, and A. Interestingly, of the remaining 20% of the 19 selected strains, only BS20 and BS23 did not synthesize D and F isoforms; BS22 did not synthetise A,D, BS39 -D, and BS42 as well as BS45—D,E,F, which was probably related to the increased production of the isoforms B (BS42, BS45), C (BS20, BS22) and E (BS23, BS39); (Table 3). It should be noted, however, that strains characterized by the highest efficiency of surfactin biosynthesis, BS15 and BS19, reducing ST by ca. 53, 49% respectively, synthesized all 6 isoforms (Fig 5). A significant high correlation (r = 0.51) between the decrease in ST [%] of the medium and the concentration of the B isoform [μg/mg] (Fig 6, Table 2) suggests that its biosynthesis may significantly affect the reduction of surface tension.
Fig 6. Relationship between the decrease in surface tension [%] and the concentration of surfactin and its B isoform [μg/mg] in post-culture media of Bacillus subtilis natto strains (n = 63).
This conclusion is also supported by the fact that all strains tested for surfactin production and its ability to reduce surface tension synthesized the B isoform, as opposed to the isoforms D, E or F. In addition, a very high percentage of this isoform, 82% and 84%, was found in the total concentration of surfactin produced by strains BS42 and BS45, respectively. Although these strains synthesized only three (A, B, C) of the six isoforms, they had the ability to lower the ST by as much as 45% (Table 1). Studies confirmed that the increased concentration of an individual isoform in total surfactin concentration can significantly determine the properties of the surfactant, including surface tension reduction [63]. The biosynthesis of an isoform containing 15 carbon atoms is particularly desirable, which has a CMC of 14.8 mg/L and can reduce water ST to 27.1 mN/m (25°C) [64]. The surface and interfacial activity of surfactin increases with the length of the alkyl chain, thereby enhancing agregation of micelles. Liu et al. [65] also observed that an increase in the concentration of the isoform containing 15 carbon atoms in the alkyl chain increased oil-washing efficiency and oil displacement efficiency by surfactin synthesized by Bacillus subtilis BS-37. Razafindralambo et al. [66] reported a relationship between the isoform consisting of 14 carbon atoms and the increased foam formation of the lipopetide, both in terms of foam density and liquid stability in foam. Thus, the structure of the peptide translates into its properties. Differences in the amount and concentration of synthesized isoforms may be genetically determined but also dependent on the composition of the culture medium and culture conditions (including pH, temperature, oxygenation) that affect the fermentation process [8,67]. For example, Jajor et al. [40] observed that reduced aeration of Bacillus subtilis KB1 and #309 cultures reduced the amount of analogues containing 15 carbon atoms and increased the amount of analogues with 12 atoms. This study can be extended to assess the impact of compound concentration in a two-phase (hydrophobic/hydrophilic) system on the shape of the surfactin peptide ring. It would also be possible to calculate the effect of biosurfactant addition on the interfacial tension in a two-phase system and to estimate lateral and rotational diffusion of the peptide ring [68]. Studies on the use of biomolecules such as surfactin can also be supplemented with modeling of molecular structure using density functional theory (DFT), which includes dispersion interactions and peptide bonds in peptide-based systems [69].
Conclusions
The development of effective screening methods guarantees quick verification of isolated strains in terms of their ability to produce surfactin and thus creates the opportunity to identify efficient producers of this compound. Blood agar test can only be an initial evaluation criterion due to the low specificity of this method. In contrast, the measurement of surface tension during cultivation is a key parameter translating into the concentration of surfactin until the biosurfactant reaches its critical micellization concentration. Extraction using affinity chromatography with the SPE system significantly shortens the time of surfactin isolation from the culture medium and thus allows rapid verification of potential surfactin producers using the HPLC method. The three stages of screening presented in this study enable effective selection of surfactin producers. By this method we initially selected 45 strains capable of producing hemolysis zones, and then narrowed down the group to 19 strains significantly lowering the ST of the culture medium (≥35%). The BS15 strain, which belonged to this group, reduced the surface tension of the culture medium by 52.77 ± 0.404 [%] and synthesized surfactin, including B isoform, at the highest concentration (> 30 μg/mg; 2.85 ± 0.03 μg/mg, respectively). The selection of BS15 is additionally supported by the high repeatability of the results in the experimental replicates. For surfactin concentration, the standard deviation was 4.7%, and for the B isoform concentration standard deviation was only 1.05% of the mean. Thus, BS15 is a promising material for further research on the optimization of surfactin production.
Data Availability
All relevant data are within the paper.
Funding Statement
This study was funded by the Polish Minister of Science and Higher Education, under the program "Regional Initiative of Excellence" in 2019–2022 (Grant No. 008/RID/2018/19). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Knepper TP, Berna JL, Surfactants: Properties, production, and environmental Aspects, Analysis and Fate of Surfactants and the Aquatic Environment in: Comprehensive Analytical Chemistry 2003, pp. 1–49. [Google Scholar]
- 2.Acmite Market Intelligence Market Report: World Surfactant Market. Archived from the original on April 2016. 4th edition.
- 3.Banat IM, Makkar RS, Cameotra SS. Potential commercial applications of microbial surfactants. Appl Microbiol Biotechnol. 2000; 53: 495–508. 10.1007/s002530051648 [DOI] [PubMed] [Google Scholar]
- 4.Ivanković T, Hrenović J. Surfactants in the environment. Arh Hig Rada Toksikol. 2010; 61: 95–110. 10.2478/10004-1254-61-2010-1943 [DOI] [PubMed] [Google Scholar]
- 5.Jackson SA, Borchert E, O’Gara F, Dobson ADW. Metagenomics for the discovery of novel biosurfactants of environmental interest from marine ecosystems. Curr Opin Biotechnol. 2015; 33: 176–182. 10.1016/j.copbio.2015.03.004 [DOI] [PubMed] [Google Scholar]
- 6.Shin K-H, Kim K-W, Ahn Y. Use of biosurfactant to remediate phenanthrene-contaminated soil by the combined solubilization-biodegradation process. J Hazard Mate. 2006; 137: 1831–1837. [DOI] [PubMed] [Google Scholar]
- 7.Mukherjee S, Das P, Sen R. Towards commercial production of microbial surfactants. Trends Biotechnol 2006; 24: 509–515. 10.1016/j.tibtech.2006.09.005 [DOI] [PubMed] [Google Scholar]
- 8.Abdel-Mawgoud AM, Aboulwafa MM, Hassouna NA-H. Characterization of surfactin produced by Bacillus subtilis isolate BS5. Appl Biochem Biotechnol. 2008; 150: 289–303. 10.1007/s12010-008-8153-z [DOI] [PubMed] [Google Scholar]
- 9.Al-Bahry SN, Al-Wahaibi YM, Elshafie AE, Al-Bemani AS, Joshi SJ, Al-Makhmari HS, et al. Biosurfactant production by Bacillus subtilis B20 using date molasses and its possible application in enhanced oil recovery. Int Biodeter Biodegr. 2016; 81: 141–146. [Google Scholar]
- 10.Gudiña EJ, Fernandes EC, Rodrigues AI, Teixeira JA, Rodrigues LR. Biosurfactant production by Bacillus subtilis using corn steep liquor as culture medium. Front Microbiol. 2015; 6: 59 10.3389/fmicb.2015.00059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Santos DKF, Rufino RD, Luna JM, Santos VA, Sarubbo LA. Biosurfactants: Multifunctional Biomolecules of the 21st Century. Int J Mol Sci 2016; 17: 401 10.3390/ijms17030401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Das K, Mukherjee AK. Comparison of lipopeptide biosurfactants production by Bacillus subtilis strains in submerged and solid state fermentation systems using a cheap carbon source: Some industrial applications of biosurfactants. Process Biochem. 2007; 42: 1191–1199. [Google Scholar]
- 13.Wei Y-H, Wang L-Ch, Chen W-Ch, Chen S-Y. Production and characterization of fengycin by indigenous Bacillus subtilis F29-3 originating from a potato farm. Int J Mol Sci. 2010; 11: 4526–4538. 10.3390/ijms11114526 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gudiña EJ, Rangarajan V, Sen R, Rodrigues LR. Potential therapeutic applications of biosurfactants. Trends Pharmacol Sci. 2013; 34: 667–675. 10.1016/j.tips.2013.10.002 [DOI] [PubMed] [Google Scholar]
- 15.Banat IM, Satpute SK, Cameotra SS, Patil R, Nyayanit NV. Cost effective technologies and renewable substrates for biosurfactants’ production. Front Microbiol. 2014; 5: 697 10.3389/fmicb.2014.00697 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sobrinho HB, Luna JM, Rufino RD, Porto ALF, Sarubbo LA. Biosurfactants: classification, properties and environmental applications. Biotechnology 2014; 11: 1–29. [Google Scholar]
- 17.Chaprão MJ, Ferreira INS, Correa PF, Rufino RD, Luna JM, Silva EJ, et al. Application of bacterial and yeast biosurfactants for enhanced removal and biodegradation of motor oil from contaminated sand. Electron J Biotechn. 2015; 18: 471–479. [Google Scholar]
- 18.Bonmatin J-M, Laprevote O, Peypoux F. Diversity among microbial cyclic lipopeptides: iturins and surfactins. Activity-structure relationships to design new bioactive agents. Comb Chem High T Scr. 2003; 6: 541–556. [DOI] [PubMed] [Google Scholar]
- 19.Jacques P. Surfactin and other lipopeptides from Bacillus spp In Biosurfactants. Microbiology Monographs; Soberón-Chávez G. Springer, Berlin, Heidelberg; 2011; pp.57–91. [Google Scholar]
- 20.Chen W-Ch, Juang R-S, Wei Y-H. Applications of a lipopeptide biosurfactant, surfactin, produced by microorganisms. Biochem Eng J. 2015; 103: 158–169. [Google Scholar]
- 21.Willenbacher J, Yeremchuk W, Mohr T, Syldatk Ch, Hausmann R. Enhancement of surfactin yield by improving the medium composition and fermentation process. AMB Express 2015; 5: 57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vollenbroich D, Özel M, Vater J, Kamp RM, Pauli G. Mechanism of inactivation of enveloped viruses by the biosurfactant surfactin from Bacillus subtilis. Biologicals 1997; 25: 289–297. 10.1006/biol.1997.0099 [DOI] [PubMed] [Google Scholar]
- 23.Wu Y-S, Ngai S-C, Goh B-H, Chan K-G, Lee L-H, Chuah L-H. Anticancer activities of surfactin and potential application of nanotechnology assisted surfactin delivery. Front Pharmacol. 2017; 8: 761 10.3389/fphar.2017.00761 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lai CC, Huang YC, Wei YH, Chang JS. Biosurfactant-enhanced removal of total petroleum hydrocarbons from contaminated soil. J Hazard Mater. 2009; 167: 609–614. 10.1016/j.jhazmat.2009.01.017 [DOI] [PubMed] [Google Scholar]
- 25.Peypoux F, Bonmatin J, Wallach J. Recent trends in the biochemistry of surfactin. Appl Microbiol Biotechnol. 1999; 51: 553–563. 10.1007/s002530051432 [DOI] [PubMed] [Google Scholar]
- 26.Liu J-F, Mbadinga SM, Yang S-Z, Gu J-D, Mu B-Z. Chemical structure, property and potential applications of biosurfactants produced by Bacillus subtilis in petroleum recovery and spill mitigation. Int J Mol Sci 2015; 16: 4814–4837. 10.3390/ijms16034814 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sen R, Swaminathan T. Characterization of concentration and purification parameters and operating conditions for the small-scale recovery of surfactin. Process Biochem. 2005; 40: 2953–2958. [Google Scholar]
- 28.Vedaraman N, Venkatesh N. Production of surfactin by Bacillus subtilis MTCC 2423 from waste frying oils. Braz J Chem Eng. 2011; 28: 175–180. [Google Scholar]
- 29.Willenbacher J, Zwick M, Mohr T, Schmid F, Syldatk C, Hausmann R. Evaluation of different Bacillus strains in respect of their ability to produce surfactin in a model fermentation process with integrated foam fractionation. Appl Microbiol Biotechnol. 2014; 98: 9623–9632. 10.1007/s00253-014-6010-2 [DOI] [PubMed] [Google Scholar]
- 30.Nitschke M, Pastore GM. Biosurfactant production by Bacillus subtilis using cassava-processing effluent. Appl Biochem Biotechnol. 2004; 112: 163–172. 10.1385/abab:112:3:163 [DOI] [PubMed] [Google Scholar]
- 31.Zhao F, Shi R, Cui Q, Han S, Dong H, Zhang Y. Biosurfactant production under diverse conditions by two kinds of biosurfactant-producing bacteria for microbial enhanced oil recovery. J Pet Sci Eng. 2017; 157: 124–130. [Google Scholar]
- 32.Mubarak MQE, Hassan AR, Hamid AA,. Khalil S, Isa MHM. A simple and effective isocratic HPLC method for fast identification and quantification of surfactin. Sains Malays. 2015; 44: 115–120. [Google Scholar]
- 33.Czaczyk K, Marciniak A, Białas W, Mueller A, Myszka K. The effect of environmental factors influencing lipopeptide biosurfactants biosynthesis by Bacillus spp. Food. Science. Technology. Quality 2007; 1: 140–149. [Google Scholar]
- 34.Zhu Z, Zhang F, Wei Z, Ran W, Shen Q. The usage of rice straw as a major substrate for the production of surfactin by Bacillus amyloliquefaciens XZ-173 in solid-state fermentation. J Environ Manage. 2013; 127: 30, 96–102. [DOI] [PubMed] [Google Scholar]
- 35.Pathak KV, Keharia H. Application of extracellular lipopeptide biosurfactant produced by endophytic Bacillus subtilis K1 isolated from aerial roots of banyan (Ficus benghalensis) in microbially enhanced oil recovery (MEOR). 3 Biotech. 2014; 4: 41–48. 10.1007/s13205-013-0119-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gurjar J, Sengupta B. Production of surfactin from rice mill polishing residue by submerged fermentation using Bacillus subtilis MTCC 2423. Bioresour Technol. 2015; 189: 243–249. 10.1016/j.biortech.2015.04.013 [DOI] [PubMed] [Google Scholar]
- 37.Alonso S, Martin PJ. Impact of foaming on surfactin production by Bacillus subtilis: implications on the development of integrated in situ foam fractionation removal systems. Biochem Eng J. 2016; 110: 125–133. [Google Scholar]
- 38.Taskin M, Kurbanoglu EB. Evaluation of waste chicken feathers as peptone source for bacterial growth. J Appl Microbiol. 2011; 111: 4, 826–834. 10.1111/j.1365-2672.2011.05103.x [DOI] [PubMed] [Google Scholar]
- 39.Slivinsky CT, Mallmann E, de Araújo JM, Mitchell DA, Krieger N. Production of surfactin by Bacillus pumilus UFPEDA 448 in solid-state fermentation using a medium based on okara with sugarcane bagasse as a bulking agent. Process Biochem. 2012; 47: 1848–1855. [Google Scholar]
- 40.Jajor P, Piłakowska-Pietras D, Krasowska A, Łukaszewicz M. Surfactin analogues produced by Bacillus subtilis strains grown on rapeseed cake. J Mol Struct. 2016; 1126: 141–146. [Google Scholar]
- 41.Ratledge C, Kristiansen B. Basic Biotechnology. Third edition Cambridge University Press; 2006. 10.1186/1472-6750-6-31 [DOI] [Google Scholar]
- 42.Nitschke M, Pastore GM. Production and properties of a surfactant obtained from Bacillus subtilis grown on cassava wastewater. Bioresour Technol. 2006; 97: 336–341. 10.1016/j.biortech.2005.02.044 [DOI] [PubMed] [Google Scholar]
- 43.Joshi S, Bharucha C, Jha S, Yadav S, Nerurkar A, Desai AJ. Biosurfactant production using molasses and whey under thermophilic conditions. Bioresour Technol. 2008; 99: 195–199. 10.1016/j.biortech.2006.12.010 [DOI] [PubMed] [Google Scholar]
- 44.Walter V, Syldatk C, Hausmann R. Screening concepts for the isolation of biosurfactant producing microorganisms. In Biosurfactants Advances in Experimental Medicine and Biology, Sen R. Springer, New York, NY, 2010; 672, pp.1–13. 10.1007/978-1-4419-5979-9_1 [DOI] [PubMed] [Google Scholar]
- 45.Dhiman R, Meena KR, Sharma A, Kanwar SS. Biosurfactants and their screening methods. Res J Recent Sci. 2016; 5: 39–43. [Google Scholar]
- 46.Cooper DG, Macdonald CR, Duff SJB, Kosaric N. Enhanced production of surfactin from Bacillus subtilis by continuous product removal and metal cation additions. Appl Environ Microb. 1981; 42: 408–412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hsieh FC, Li MC, Lin TC. Rapid detection and characterization of surfactin-producing Bacillus subtilis and closely related species based on PCR. Curr Microbiol. 2004; 49: 186–191. 10.1007/s00284-004-4314-7 [DOI] [PubMed] [Google Scholar]
- 48.Ismail W, Al-Rowaihi IS, Al-Humam AA, Hamza RY, El Nayal AM, Bououdina M. Characterization of a lipopeptide biosurfactant produced by a crude-oil-emulsifying Bacillus sp. I-15. Int. Biodeter Biodegr. 2013; 84: 168–178. [Google Scholar]
- 49.de Franҫa IWL, Lima AP, Lemos JAM, Lemos CGF, Melo VMM, De Santana HB. Production of a biosurfactant by Bacillus subtilis ICA56 aiming bioremediation of impacted soils. Catal Today 2015; 255: 10–15. [Google Scholar]
- 50.Cooper DG. Biosurfactants. Mikrobiol. Sci. 1986; 3: 145–149. [PubMed] [Google Scholar]
- 51.de Faria AF, Teodoro-Martinez DS, de Oliveira Barbosa GN, Vaz BG, Silva ÍS, Garcia JS, et al. Production and structural characterization of surfactin (C14/Leu7) produced by Bacillus subtilis isolate LSFM-05 grown on raw glycerol from the biodiesel industry. Process Biochem. 2011; 46: 1951–1957. [Google Scholar]
- 52.Bezza FA, Chirwa EMN. Production and applications of lipopeptide biosurfactant for bioremediation and oil recovery by Bacillus subtilis CN2. Biochem Eng J. 2015; 101: 168–178. [Google Scholar]
- 53.Al-Wahaibi Y, Joshi S, Al-Bahry S, Elshafie A, Al-Bermani A, Shibula B. Biosurfactant production by Bacillus subtilis B30 and its application in enhancing oil recovery. Colloid BioSurf B Biointerfaces 2014; 114: 324–333. [DOI] [PubMed] [Google Scholar]
- 54.Jha SS, Joshi SJ, Geetha SJ. Lipopeptide production by Bacillus subtilis R1 and its possible applications. Braz J Microb 2016; 47: 955–964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Youssef NH, Duncan KE, Nagle DP, Savage KN, Knapp RM, McInerney MJ. Comparison of methods to detect biosurfactant production by diverse microorganisms. J Microbiol Methods 2004; 56: 339–347. 10.1016/j.mimet.2003.11.001 [DOI] [PubMed] [Google Scholar]
- 56.Mulligan CN, Cooper DG, Neufeld RJ. Selection of microbes producing biosurfactants in media without hydrocarbons. J Ferment Technol. 1984; 62: 311–314. [Google Scholar]
- 57.Oliveira DWF, Sousa JR, França IWL, Felix AKN, Martins JJL, Gonçalves LRB. Kinetic study of biosurfactant production by Bacillus subtilis LAMI005 grown in clarified cashew apple juice. Colloids Surf. B Biointerfaces 2013; 101: 34–43. 10.1016/j.colsurfb.2012.06.011 [DOI] [PubMed] [Google Scholar]
- 58.Amani H, Haghighi M, Keshtkar MJ. Production and optimization of microbial surfactin by Bacillus subtilis for ex situ enhanced oil recovery. Petrol Sci Technol. 2013; 31: 1249–1258. [Google Scholar]
- 59.Jokari S, Rashedi H, Amoabediny Gh, Naghizadeh Dilmaghani S, Mazaheri Assadi M. Optimization of surfactin production by Bacillus subtilis ATCC 6633 in a miniaturized bioreactor. Int J Environ Res. 2013; 7: 851–858. [Google Scholar]
- 60.Wei Y-H, Wang L-F, Changy J-S, Kung S-S. Identification of induced acidification in iron-enriched cultures of Bacillus subtilis during biosurfactant fermentation. J Biosci Bioeng. 2003; 96: 174–178. 10.1016/s1389-1723(03)90121-6 [DOI] [PubMed] [Google Scholar]
- 61.Haddad NIA, Liu X, Yang S, Mu B. Surfactin isoforms from Bacillus subtilis HSO121: separation and characterization. Protein Peptide Lett. 2008; 15: 265–269. [DOI] [PubMed] [Google Scholar]
- 62.Zhao Y, Yang SZ, Mu BZ. Quantitative analyses of the isoforms of surfactin produced by Bacillus subtilis HSO 121 using GC-MS. Anal Sci. 2012; 28: 789–793. 10.2116/analsci.28.789 [DOI] [PubMed] [Google Scholar]
- 63.Nayarisseri A, Singh P, Singh SK. Screening, isolation and characterization of biosurfactant-producing Bacillus tequilensis strain ANSKLAB04 from brackish river water. Int J Environ Sci Technol. 2018; 1–10. [Google Scholar]
- 64.Wang Q, Yu H, Wang M, Yang H, Shen Z. Enhanced biosynthesis and characterization of surfactin isoforms with engineered Bacillus subtilis through promoter replacement and Vitreoscilla hemoglobin co-expression. Process Biochem. 2018; 70: 36–44. [Google Scholar]
- 65.Liu Q, Lin J, Wang W, Huang H, Li S. Production of surfactin isoforms by Bacillus subtilis BS-37 and its applicability to enhanced oil recovery under laboratory conditions. Biochem Eng J. 2015; 93: 31–37. [Google Scholar]
- 66.Razafindralambo H, Popineau Y, Deleu M, Hbid C, Jacques P, Thonart P, et al. Foaming properties of lipopeptides produced by Bacillus subtilis. Effect of lipid and peptide structural attributes. Agric Food Chem. 1998; 46: 911–916. [Google Scholar]
- 67.Liu JF, Yang J, Yang SZ, Ye RQ, Mu BZ. Effects of different amino acids in culture media on surfactin variants produced by Bacillus subtilis TD7. Appl Biochem Biotechnol. 2012; 166: 2091–2100. 10.1007/s12010-012-9636-5 [DOI] [PubMed] [Google Scholar]
- 68.Nicolas JP. Molecular Dynamics simulation of surfactin molecules at the water-hexane interface. Biophys J. 2003; 85: 1377–1391. 10.1016/S0006-3495(03)74571-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ilawe NV, Schweitzer-Stenner R, DiGuiseppi D, Wong M. Is a cross-β-sheet structure of low molecular weight peptides necessary for the formation of fibrils and peptide hydrogel? Phys. Chem. Chem. Phys. 2018; 20: 18158–18168. 10.1039/c8cp00691a [DOI] [PubMed] [Google Scholar]