Abstract
Low heart rate variability (HRV) has strongly been associated with an increased risk for cardiovascular disease. With cardiovascular disease being the number one cause of global deaths, factors that influence its development are relevant to understand. Season of birth has been suggested as one of the factors influencing the development of HRV. The current study was set up to replicate the finding that men born in winter have higher HRV later in life compared to those born in other seasons. To this end, we studied a sample of 1,871 healthy participants from the Brain Resource International Database during rest and during task. Furthermore, sex and age differences and associations with personality traits and psychiatric symptoms were explored. We replicated the earlier finding that men born in winter have a lower ratio of low frequency (LF) power to high frequency (HF) power during rest compared to summer and fall, and, although less pronounced, higher HF compared to summer. A difference between summer and winter for LF/HF in men was internally replicated using data recorded during task. Additionally, for both sexes, LF/HF ratio increased with age, and LF and HF both decreased. In general, LF/HF was lower in women, but heart rate was higher. In men, low HRV was associated with depression and the personality trait openness. In conclusion, results from a large multicenter data set covering the entire lifespan demonstrate that HRV changes with age in both sexes and confirm that season of birth influences HRV later in life in men.
Keywords: aging, heart rate variability, season of birth, sex differences
Short abstract
This is a replication study, showing heart rate variability (HRV) to vary with season of birth (SoB). Since both HRV and SoB have been related to cardiovascular disease, the relationship is relevant to understand. We used an almost five times larger sample than the original sample, covering wider geographic areas and the full lifespan. Additionally, age and sex differences in HRV were found. Replicating earlier findings provides solid evidence for a relationship between SoB and HRV later in life, thereby providing insights on how HRV develops and ultimately how increased risk for cardiovascular disease can be reduced.
1. INTRODUCTION
Season of birth (SOB) has been associated with many psychiatric disorders such as schizophrenia, major depression disorder (MDD; Torrey, Rawlings, Ennis, Merrill, & Flores, 1996), autism spectrum disorder (ASD), and attention‐deficit hyperactivity disorder (ADHD), as well as with physical disorders such as epilepsy and cardiovascular disease (Nonaka & Imaizumi, 2000; Reffelmann, Ittermann, Empen, Dörr, & Felix, 2011). With cardiovascular disease being the number one cause of global deaths, factors that influence its development are relevant to understand. So far, being born in winter seems to be cardio‐protective (Sohn, 2016), whereas men born in spring had lower blood pressure (Banegas et al., 2000). Also, the variability in heart rate has been related to SOB (Huang et al., 2015). Heart rate variability (HRV) is the fluctuation around the mean heart rate and is generally regarded as an index for autonomic functioning or sympathovagal balance, representing the flexible shift between sympathetic and parasympathetic activity (Thayer & Lane, 2000). It is often reflected by the ratio of low frequency to high frequency power (LF/HF) in frequency domain analyses of the electrocardiogram (ECG). Low HRV has strongly been associated with an elevated risk for heart‐related problems (Hillebrand et al., 2013; Thayer, Yamamoto & Brosschot, 2010). Moreover, dysregulated HRV has been related to the prevalence of psychiatric disorders such as MDD (Kemp et al., 2010), ADHD (Griffiths et al., 2017), ASD (Wang et al., 2015), psychosis (Alvares, Quintana, Hickie, & Guastella, 2016), and anxiety (Chalmers, Quintana, Abbott, & Kemp, 2014) and is even related to personality (Huang et al., 2013; Shepherd, Mulgrew, & Hautus, 2015). Thus, HRV seems to contribute to the development of emotional and physical well‐being, which in turn could be influenced by SOB. A relation between HRV and SOB implies the influence of environmental factors associated with seasonality on HRV during the prenatal and postnatal period. These factors may include climatic factors such as sunlight, humidity, temperature, but also nutrition, exercise (being outdoors), and prenatal stress. Although a relationship between HRV and SOB has been suggested by one study, this study was rather small and conducted at a research site with a specific (Taiwanese) climate (Huang et al., 2015). In a sample of 382 school children (aged 6–10 years), SOB was related to both sympathetic and parasympathetic activity. They found that (a) boys born in winter have higher HF power than boys born in other seasons, (b) boys born in winter have a lower LF/HF power than boys born in other seasons, (c) temperature of SOB and age were predictive factors for HF among boys, and (d) humidity during SOB was predictive of normalized low frequency power (LF%) and LF/HF among girls.
The aim of the current study was to conceptually replicate the findings of Huang et al. (2015). We extended their findings to a lifespan perspective as well as using a multicentric approach to rule out whether effects are driven by specific local and/or cultural aspects rather than season. To this end, an existing data set of 1,871 healthy participants between the ages of 6 to 87 years was used. Additionally, this study investigated age and sex differences in HRV and correlations between personality, depression, and anxiety scores with HRV.
2. METHOD
2.1. Participants
Data of participants were extracted from the Brain Resource International Database (BRID). This database contains data from multiple laboratories (New York, Rhode Island, Nijmegen, Sydney, Adelaide) that have been acquired using standardized data acquisition techniques (identical amplifiers, standardization of other hardware, audio calibration, paradigm details, software acquisition, task instructions). Interlab reliability and test‐retest reliability measures are high and have been reported elsewhere (Clark et al., 2006; Paul et al., 2007; Williams et al., 2005). The database consists of healthy participants between 6 and 87 years old. Database exclusion criteria included a personal or family history of mental illness, brain injury, neurological disorder, a serious medical condition, drug/alcohol addiction, first degree relative with bipolar disorder, schizophrenia, or genetic disorder. Data from participants that did not follow task instructions were rejected from analyses.
After selecting data from participants for whom ECG data were available, using data from sites that contributed at least N = 20 due to our interest in seasonality that geographically varies, the sample consisted of 1,871 participants (956 men; mean age = 28.31 ± 19.92). Participants were required to refrain from caffeine and smoking (2 hr), and alcohol (6 hr) prior to testing. All participants provided written informed consent. Since only an existing data set was used, no Institutional Review Board approval was obtained. Demographic characteristics can be found in Table 1.
Table 1.
Comparison of the HRV parameters across a sample born in four seasons
| Men | Spring N = 257 | Summer N = 224 | Fall N = 231 | Winter N = 244 | F test (ANOVA) | p value | Comparison |
|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | ||||
| Age in years | 25.493 (17.834) | 26.807 (19.814) | 26.370 (19.283) | 25.615 (19.631) | 0.253 | .859 | |
| RR. ms* (N = 904) | 841.595 (140.725) | 833.592 (132.684) | 843.330 (143.182) | 826.018 (139.163) | 0.747 | .524 | |
| HF. ln (ms2) | 6.383 (1.460) | 6.101 (1.413) | 6.203 (1.495) | 6.395 (1.528) | 2.222 | .084 | Spring‐winter: p = .931 Summer‐winter: p = .032 Fall‐winter: p = .168 |
| LF. ln (ms2) | 6.559 (1.165) | 6.418 (1.272) | 6.398 (1.271) | 6.395 (1.287) | 0.988 | .398 | |
| LF/ HF [ln (ratio)] | 0.176 (1.057) | 0.317 (1.067) | 0.195 (1.057) | <0.001 (1.074) | 3.548 | .014 | Spring‐winter: p = .066 Summer‐winter: p = .001 Fall‐winter: p = .047 |
| VLF. ln (ms2) | 5.951 (1.270) | 5.803 (1.196) | 5.893 (1.272) | 5.839 (1.125) | 0.681 | .564 | |
| LF %. nu* (N = 904) | 9.746 (11.026) | 9.881 (14.407) | 10.290 (13.289) | 9.449 (9.964) | 0.183 | .908 | |
| Spring N = 217 | Summer N = 220 | Fall N = 241 | Winter N = 237 | ||||
| Women | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | F test (ANOVA) | p value | Comparison |
| Age in years | 29.431 (19.544) | 31.936 (21.058) | 31.063 (20.792) | 30.291 (20.515) | 0.601 | .615 | |
| RR. ms* (N = 866) | 809.534 (132.657) | 825.032 (132.001) | 828.269 (119.491) | 825.660 (136.921) | 0.905 | .438 | |
| HF. ln (ms2) | 6.198 (1.311) | 6.116 (1.443) | 6.178 (1.384) | 6.181 (1.366) | 0.148 | .931 | |
| LF. ln (ms2) | 6.109 (1.251) | 6.170 (1.256) | 6.100 (1.174) | 6.132 (1.184) | 0.149 | .931 | |
| LF/HF [ln (ratio)] | −0.887 (0.949) | 0.054 (1.042) | −0.078 (0.910) | −0.048 (1.000) | 0.986 | .399 | |
| VLF. ln (ms2) | 5.703 (1.182) | 5.637 (1.196) | 5.695 (1.065) | 5.743 (1.155) | 0.328 | .805 | |
| LF%. nu* (N = 866) | 7.487 (8.979) | 7.819 (10.732) | 7.194 (7.683) | 7.836 (14.007) | 0.181 | .909 |
Descriptives represent the resting condition; task data is not shown.
For RR and LF%, the sample was smaller.
2.2. Psychological data acquisition
Preceding the ECG measurement, participants completed the NEO Five‐Factor Inventory (NEO‐FFI). The NEO‐FFI was used to examine the Big Five personality traits. The NEO‐FFI is a 60‐item, self‐report instrument that measures five personality traits: neuroticism, extraversion, openness, agreeableness, and conscientiousness. These domains have shown good internal consistency (Cronbach's alpha range 0.87–0.92; Renner, Penninx, Peeters, Cuijpers, & Huibers, 2013). To measure anxiety and stress, the Depression, Anxiety and Stress Scale (DASS; Henry & Crawford, 2005) was used.
2.3. Experimental design
ECG was recorded during an EEG test battery. Of this test battery, ECG recordings during eyes open at rest (EO) and auditory oddball (ODB) were used for HRV analysis. These tasks were recorded separately and independent from each other. During EO (2 min), the participant was instructed to sit relaxed and fixate on the red dot displayed on the screen for 2 min, in essence similar to the 5‐min recording in Huang et al. (2015) where participants sat quietly awake. During ODB (6 min), the participant was presented with high‐pitched and low‐pitched tones and instructed to press a button when hearing high‐pitched tones.
2.4. Physiological data acquisition
Two ECG electrode channels were combined to create the ECG data series, with one positioned on the inside of the nondominant wrist, directly above the radial pulse, and the other at Erb's point (located two thirds distal from midline on the clavicle). Both of these channels were referenced to a separate common reference channel at C7 (the 7th cervical vertebra's most pronounced transverse process). The Erb's point and C7 recording sites are both positioned directly above the bone to serve as relatively muscle‐free data recordings. The data were sampled at 500 Hz, and a low‐pass filter of 100 Hz was applied prior to digitization (Griffiths et al., 2017).
2.5. Data processing
Data processing was performed according to Griffiths et al. (2017): Data reduction was performed using software from Brain Resource Ltd. and the standard methods for the BRID (Kozlowska et al., 2015). R waves (i.e., the main spikes observed in the graphical deflections observed in an ECG) were detected in the ECG and converted to a RR tachograph, which is a graph of the numerical value of the RR interval (i.e., the interval between two R peaks) and time. The combined ECG and Erb's data (ECG − 5 * Erb's) underwent a 5–15 Hz Tukey band‐pass filter. Data cleaning was performed using semiautomated methods to identify and remove any tacho series in which R waves were not reliably detected or scored, series that where ectopic, where arrhythmic beats were present, or where the voltage was too extreme (low or high) to be scored. Beats were considered missing when the interbeat interval exceeded 1.2 times the moving average (using a forgetting factor of 0.4), and an ectopic beat was classified as an interval of less than 0.8 of the moving average. Intervals around these beats were also removed from the tacho series.
The RR interval signal was interpolated by a cubic spine interpolation algorithm. Absolute very low frequency power (VLF: 0.0033–0.04 Hz), low frequency power (LF: 0.04–0.15 Hz), and high frequency power (HF: 0.15–0.4 Hz) were calculated, as well as the ratio of LF/HF. Frequency domain measures were measured in power (ms2) by use of a Welch's periodogram.
The natural logarithm (ln) of VLF, LF, HF and LF/HF was taken, consistent with the analyses of Huang et al. (2015).
2.6. Climatic data
The data set was divided into four seasons: spring (March to May), summer (June to August), fall (September to November), and winter (December to February) for the northern hemisphere, identical to Huang et al. (2015) and spring (September to November), summer (December to February), fall (March to May), and winter (June to August) for the southern hemisphere.
For the different sites, dew point, temperature, and solar irradiance (SI) were calculated per month using Meteonorm 7.2.2 (http://www.meteonorm.com/en/downloads). Per site, interpolated city was used as location, interpolating data from different weather stations. Data were based on the month averages over the years 1991–2010 using standard Meteonorm output. In addition, the difference between 2‐month averages was calculated (SI change). Dew point was used as an indicator of humidity (studied in Huang et al., 2015).
2.7. Statistical analysis
The data set was split a priori into men and women. Statistical analyses identical to Huang et al. (2015) were performed: HRV metrics among the four seasons were compared with univariate analyses, in order to test the following hypotheses one‐tailed (i.e., a significance threshold of p ≤ .1): (a) boys born in winter had lower LF/HF than when born in other seasons, and (b) boys born in winter had higher HF power than when born in other seasons. These analyses were conducted separately for rest and task conditions. For significant effects, independent samples t tests were performed to directly compare seasons. Effect sizes were calculated using Cohen's d. Being an international multicenter study, we verified post hoc whether location had a significant effect on outcome, using site as an additional factor in univariate analysis.
Additionally, influences of climatic factors such as temperature, dew point, and SI were tested for using correlation analyses. Because climate metrics were not normally distributed, Spearman correlation was used. For correlations for which we did not expect a significant effect based on Huang et al. (2015) Bonferroni correction was applied, setting the significance level at p ≤ .013.
Next, age and HRV were correlated and sex differences between HRV measures (meanRR, LF, HF, and LF/HF, VLF) were tested using univariate analyses with sex as fixed factor and the different HRV metrics as dependent variables. For this analysis, men and women were combined. Since VLF power is unreliable in the shorter EO recordings because any frequencies under 0.0085 Hz will not be reliably scored, VLF power was investigated only during the longer oddball recordings.
Finally, correlation analysis was performed between HRV measures: LF/HF, VLF, LF, HF, meanRR, and the Big Five Personality traits, and also between the HRV measures and DASS scores for depression, anxiety, and stress, both for HRV measures at rest and at task. For these analyses, Bonferroni correction was applied, setting the significance level at p ≤ .006.
3. RESULTS
3.1. Replication
Data were divided based on sex. No significant age differences were observed between seasons (men: F(3) = 0.253; p = .859; women: F(3) = 0.601; p = .615). Similar to Huang et al. (2015) a comparison of HRV parameters in men revealed significant differences for HF and LF/HF among the four seasons, during the resting condition. A significant main effect of SOB was found for LF/HF ratio, F(3) = 3.548, p = .014, and for HF, F(3) = 2.222, p = .084, in men. Direct comparison between seasons revealed that LF/HF was lower in winter, consistent with the findings from Huang et al. (winter‐spring: t(499) = −1.843, p = .066; winter‐summer: t(466) = −3.200, p = .001, Cohen's d = 0.296; winter‐fall: t(473) = −1.991, p = .047, Cohen's d = 0.183; Figure 1). As in Huang et al., this effect was lacking in women. Furthermore, higher HF power in men born in winter was also replicated (winter‐spring: t(499) = −0.087, p = .931; winter‐summer: t(466) = −2.157, p = .032, Cohen's d = −0.200; winter‐fall: t(473) = −1.380, p = .168; Table 1, Figure 1). Post hoc analysis showed no indications for interaction effects with location, for both LF/HF and HF power.
Figure 1.
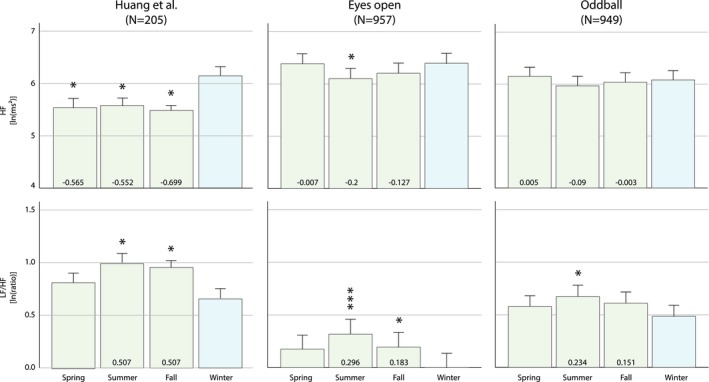
Heart rate variability plotted for different seasons of birth. Top: Variation in high frequency (HF [ln (ms2)]). Bottom: Variation in low frequency relative to high frequency (LF/HF [ln (ratio)]). Left: Original results from Huang et al. (2015). Middle and right: Results from the current study for the eyes open resting state and oddball performance conditions, respectively. Significance between each season (green) in relation to winter (blue) are indicated. Cohen's d' effect sizes are provided in the columns for the significant comparisons found in Huang et al. *p ≤ .05; ***p ≤ .001
During task condition, there was an association of SOB with LF/HF ratio, F(3) = 2.156, p = .092, in men but not women. Direct comparison between seasons revealed that this effect was mainly driven by a difference in LF/HF between those born in summer and those born in winter (winter‐spring: t(488) = −1.301, p = .194; winter‐summer: t(458) = −2.505, p = .013, Cohen's d = 0.234; winter‐fall: t(470) = −1.638, p = .102; Figure 1).
3.2. Mediating factors
As expected, temperature was significantly different between seasons, F(3) = 1617.87, p < .001, as well as dew point, F(3) = 727.55, p < .001, SI, F(3) = 2340.10, p < .001, and SI change, F(3) = 3701.54, p < .001, as seen in Table 2. A small significant correlation between dew point and LF/HF was found in men (r = .081; p = .012) but not in women. In women, meanRR shows a significant yet small correlation to SI (r = −.085; p = .012).
Table 2.
Climatic data of the four seasons during SOB of participants
| Spring | Summer | Fall | Winter | p value | |
|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | ||
| Temperature (°C) | 15.723 (3.615) | 21.730 (1.541) | 16.668 (3.477) | 10.469 (3.319) | <.001 |
| Dew point | 7.213 (3.012) | 11.623 (3.306) | 9.492 (2.765) | 5.046 (3.439) | <.001 |
| Solar intensity (SI) | 158.328 (34.502) | 203.448 (29.710) | 116.308 (39.842) | 75.699 (20.944) | <.001 |
| Change in SI | 29.702 (11.720) | −17.325 (17.804) | −32.066 (14.108) | 22.656 (11.193) | <.001 |
3.3. Sex differences
Because there was a significant age difference between sexes (men: M = 25.834, SD = 19.094, women: M = 30.336, SD = 20.513, F(1) = 25.816, p < .001), sex analyses were covaried by age. Univariate analyses revealed that there were significant sex differences found for most HRV metrics, most pronounced in LF, F(1) = 12.718, p < .001, LF/HF, F(1) = 32.376, p < .001, meanRR, F(1) = 17.959, p < .001, during rest. No sex differences were found for HF.
During task, this was similar for LF, F(1) = 48.398; p < .001, LF/HF, F(1) = 49.274; p < .001, VLF, F(1) = 17.483, p < .001, and meanRR, F(1) = 17.919; p < .001. Men had higher RR interval lengths than women and more LF, resulting in higher LF/HF ratios compared to women.
3.4. HRV across the lifespan
Since the currently used sample consists of participants across the entire lifespan, we investigated the influence of age on HRV metrics. Using Spearman's correlations, strong correlations with age were found for both men and women (see Figure 2). MeanRR increased (men: r = .441; p < .001, women: r = .459; p < .001), especially until 20 years, reflecting a heart rate decrease with age. Conversely, LF (r = −.281, p < .001) and HF (r = −.553, p < .001) power decreased with age (Figure 2, Table 3) resulting in a positive relationship between LF/HF and age (r = .423; p < .001) in men as well as in women (LF: r = −.488, p < .001; HF: r = −.513, p < .001; LF/HF: r = .117, p < .001). These results were replicated during task (results not shown), and, in addition, a significant correlation with VLF was also obtained (men: r = −.314, p < .001; women: r = −.408, p < .001).
Figure 2.
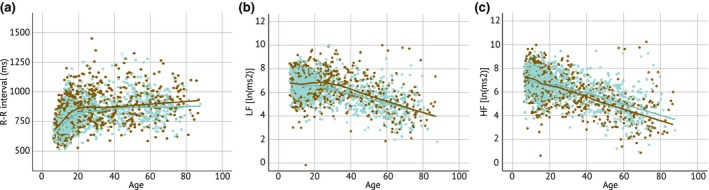
HRV across the lifespan, separated for men (brown dots and line) and women (blue dots and line). Depicted HRV metrics are (a) meanRR interval, (b) LF, and (c) HF
Table 3.
HRV across the lifespan
| Age | ||
|---|---|---|
| Male | Female | |
| R‐R interval (ms) | r = .441, p < .001, N = 194 | r = .459, p < .001, N = 866 |
| Low frequency (LF [LN (ms2)] | r = −.281, p < .001, N = 956 | r = −.488, p < .001, N = 915 |
| High frequency (HF [LN (ms2)] | r = −.553, p < .001, N = 956 | r = −.513, p < .001, N = 915 |
3.5. Depression, anxiety, and stress
Although the current sample consists of healthy participants, variation within the healthy range can be observed in the DASS. There was no interaction between SOB, DASS scores, and HRV. However, HRV measures did correlate to DASS scores: In men, age was significantly correlated to depression (r = .086; p = .007), stress (r = .148, p < .001), and anxiety (r = .071; p = .025), thus analyses were covaried by age. In women, age correlated only to stress levels (r = .112; p < .001). To correct for multiple testing, a strict p value of .006 was set.
In men, LF/HF correlated to depression severity (r = .141; p < .001) as well as to stress (r = .111; p = .001), indicating lower HRV when depression and stress scores are higher. However, this was not found during task.
In women, there were significant correlations found between meanRR length and depression (r = .120; p < .001) and meanRR and stress (r = .113; p = .001) during rest. This was replicated during task (depression: r = .102; p = .004, stress: r = .114; p = .001), but when covarying for age, these effects where no longer visible. When controlling for age, a significant correlation was found between stress and LF (r = .110; p = .004), showing that sympathetic activity is increased when stress levels are high.
3.6. Personality–HRV
No effect of SOB was found on personality. Although personality is often assumed to be stable across time, correlations with age were found in both men and women. Neuroticism and extraversion decreased with age (neuroticism: r = −.193, p < .001; extraversion: r = −.214, p < .001), whereas openness, agreeableness, and conscientiousness increased with age (openness: r = .188, p < .001; agreeableness: r = .202, p < .001; conscientiousness: r = .358, p < .001). For this reason, personality–HRV analyses were covaried by age. Doing so, openness was positively correlated with LF (r = .173; p < .001) and LF/HF (r = .167; p < .001) in men, which was replicated during task (LF: r = .157; p < .001 and LF/HF: r = .140; p < .001). In women, openness was positively correlated to meanRR, both at rest (r = .196; p < .001) and during task (r = .211; p < .001), but not to LF or LF/HF.
Neuroticism in men was significantly related to lower meanRR (r = −.109; p = .004) at rest, but not during task (p = .009; Bonferroni‐corrected threshold was set at p = .006). However, during task, neuroticism was related to VLF power (r = −.115; p = .003), and extraversion was significantly related to meanRR during task (r = .116; p = .002) but not Bonferroni‐corrected significant at rest (r = .101; p = .008).
4. DISCUSSION
The aim of the current study was to conceptually replicate the findings of Huang et al. (2015). The replication was conceptual (Schmidt, 2009) because we studied different and wider geographic areas, and a wider—lifelong—age range. Additionally, this study investigated age and sex differences in HRV and correlations between personality, depression, and anxiety scores with HRV. We replicated that men born in winter had lower LF/HF. They also expressed higher HF power, indicating parasympathetic activity, although this effect was less pronounced. For LF/HF, this finding was internally replicated using HRV data during task performance. Furthermore, we found that HRV was different between men and women, and HRV changed with aging. Finally, we found a correlation of LF/HF ratio with depression and stress scores, as well as with the personality trait openness.
Regarding mediating factors, a small association of LF/HF with dew point was found in men, whereas Huang et al. (2015) found an association with humidity in girls. Although dew point and humidity are closely related, they are not the same. Note that Huang et al. conducted their study in Taiwan, characterized by a subtropical climate and tested participants only between April and October.
In rest, we found only LF/HF and HF to be significantly different between SOB. This makes sense since it has previously been suggested that parasympathetic activity is the main driver of LF/HF ratio in rest, also reflected in HF (Billman, 2013; Uijtdehaage & Thayer, 2000). We used a rest and task condition in order to validate the findings, both requiring the participant to remain stationary, distinguished by cognitive demands. Although LF/HF was found to be significantly different during both rest and task conditions, HF differed significantly only during the rest condition. It is not fully understood why. Task demands may have led to decreased parasympathetic activation, resulting in diminished HF power.
There are several hypotheses for why HRV would be higher when born in winter. First of all, vitamin D has been associated with HRV (Canpolat et al., 2015; Mann et al., 2013). The sex difference may arise from hormonal differences. Previous findings suggest that higher levels of testosterone were associated with higher levels of HRV, while there was no association between estrogen and HRV (Wranicz et al., 2004) and that the level of testosterone is also driven by vitamin D levels (Nimptsch, Platz, Willett, & Giovannucci, 2012). Another explanation could lie in the levels of serotonin and dopamine metabolites, of which seasonal variation has been reported (Brewerton, Putnam, Lewine, & Risch, 2018; Losonczy, Mohs, & Davis, 1984). That is, serotonin and dopamine metabolite levels are higher in winter. Sex differences have been reported, but a theory as to how these came about has not been suggested (Brewerton et al., 2018). Furthermore, it has yet to be established how seasonality in these metabolites relates to HRV. To explore the influence of estrogen and testosterone, it may be useful to include information about the menstrual cycle for women, as it has been previously shown to have an effect (Bai, Zhou, & Li, 2009). Unfortunately, for the current sample these data were not available.
We found that women expressed, in general, lower RR intervals, while having lower LF/HFs, compared to men. This was similar to a previous meta‐analysis (Koenig & Thayer, 2016). However, another study showed that LF/HF was higher in women (De Meersman & Stein, 2007). Nonetheless, sex differences in HRV seem to be age and measure dependent, and age is an important modulator of HRV. It was shown that sex differences decrease with age, starting from 30 years old, and disappear around the age of 50, dependent on what measure for HRV is used (Umetani, Singer, McCraty, & Atkinson, 1998). This may be attributed to the level of estrogen (Liu, Kuo, & Yang, 2003). Since our sample consisted of participants across the whole lifespan, we correlated HRV metrics with age. Age was found to have a substantial impact on HRV, where meanRR interval increased with age, while VLF, LF, and HF power decreased with age, in line with previous work showing a negative correlation between age and HF (Abhishekh et al., 2013) and increasing heart rates (decreasing meanRR; Umetani et al., 1998). No indications of the proposed disappearance of sex differences by the age of 50 were observed in the current sample, but this may be due to the fact that we—in contrast to Umetani et al. (1998)—have investigated the frequency domain HRV rather than the time domain. Our correlations with age were reversed from the results presented by Huang et al. (2015) which is probably a consequence of their small age range. In order to see if age would have any influence on the primary analyses, we replicated the main analysis with age as covariate, in order to control for any age‐related HRV differences. Age did not seem to be a modulator of the effect of SOB on HRV, since the results were similar to the earlier obtained results.
Previous research suggests that both depression (Disanto et al., 2012; Torrey et al., 1996) and HRV (Kristal‐Boneh, Froom, Harari, Malik, & Ribak, 2000) show seasonal variation. As mentioned before, dopamine and serotonin metabolite levels also show such variation, and serotonin levels are associated with depression and anxiety (Deakin, 1998). In the current study, LF/HF was positively correlated with depression and stress in men. Previous studies pointed to HRV as a marker for depression (Brunoni et al., 2013; Ehrenthal, Herrmann‐Lingen, Fey, & Schauenburg, 2010; Kemp et al., 2010; Licht et al., 2008; Udupa et al., 2007). The latter has been found in the current study in men, but a seasonal effect was not detected. It is important to note that, although the sample consisted of healthy participants without diagnosed depression, anxiety, or stress, we did find similar results to previous studies, albeit only for men. For women, only stress was associated with higher LF, which reflects, as can be expected, higher sympathetic tone.
Few studies have examined the relationship between HRV and personality, and findings have been inconsistent. Furthermore, different studies have used different personality questionnaires/factors. In the current study, both in men and women (although for different HRV metrics), positive associations were found for openness with LF, LF/HF ratio, and meanRR intervals. An association with openness has been reported in a few other studies, in which LF and LF/HF were shown to be positively associated with levels of openness (Čukić & Bates, 2014) and HF negatively, which is believed to be the result of activation of reward/motivation, emotion, and arousal pathways in the brain. It has previously been shown that lower HRV was associated with greater difficulties in emotion regulation, but this was measured with RMSSD (root mean square of successive differences), which is a HRV variable in the time domain (Williams et al., 2015). This may suggest that men born in winter have decreased emotion regulation than when born in other seasons, but we were not able to test this with the current data.
When interpreting the meaning of the current results, it is important to note that LF/HFs might not truly reflect the balance between sympathetic and parasympathetic activity—HRV is the fluctuation around the mean heart rate, largely under influence of respiration but is also affected by blood pressure and arterial fluctuations and is often used as an index for autonomic functioning (Thayer & Lane, 2000). LF has been associated with sympathetic activity and HF with parasympathetic activity. Consequently, the LF/HF reflects the interaction between the two (McCraty & Shaffer, 2015). HF has been related to respiratory sinus arrhythmia (RSA) and is a measure of the natural variation occurring in the HR during a breathing cycle (Porges, 1995). However, during slow respiration, vagal activity can easily generate oscillations that cross over into the LF band, for example, during slow‐paced breathing. Thus, some research suggests that LF is not merely a reflection of sympathetic activity (Billman, 2013; Reyes del Paso, Langewitz, Mulder, van Roon, & Duschek, 2013) and that LF/HF does not adequately reflect the balance between sympathetic and parasympathetic activity. Still, during task, one would not expect slow‐paced breathing, and the results of ODB replicated the resting state EO data, thus (para)sympathetic balance would be the most sensible explanation within this study. In addition, the standard recording length for HRV is 5 min, whereas we had recordings of 120 s for the rest condition. Recent studies, however, suggest that these shorter time frames are still reliable, although it has been suggested that VLF data require a minimum of 270 s (Shaffer & Ginsberg, 2017). Nevertheless, one study showed that shorter VLF recordings still highly correlate with 5‐min recordings, thus the shorter VLF recordings may provide a good indication (Baek, Cho, Cho, & Woo, 2015).
One limitation of the study is that, to correct for ectopic heartbeats and artifacts, phases of beat‐to‐beat intervals were cut out instead of using an interpolation method, possibly having unwanted influences on the frequency spectra. Since we received the HRV metrics from an existing database, we had no influence on this method. Using such a correction method may have had influences on frequency spectra. However, since Huang et al. (2015) did use an interpolation method and the current study does replicate these earlier data, it can be assumed that in this case there was no major influence. Other limitations are that there are many factors influencing HRV, such as body mass index, sleep deprivation, and menstrual cycle but also acute effects of having a meal or drink, and these are not controlled for (Bai et al., 2009; Monnard & Grasser, 2017; Takase et al., 2004). Furthermore, recent developments point to using nonlinear measures such as entropy to analyze HRV (Voss, Schulz, Schroeder, Baumert, & Caminal, 2009; Young & Benton, 2015). Since this was a replication study, we did not use such methods, but it would certainly be of interest to do so in the future.
The currently used sample was a worldwide multicenter study and tested people across the lifespan throughout the whole year, resulting in a high generalizability of the current results. Future studies could focus on unraveling the underlying reason of a relationship between season of birth and HRV, thereby possibly unraveling the etiology of sex‐specific problems associated with certain HRV patterns. Furthermore, the newly found relationships between HRV and depressive symptoms and personality traits should be replicated and investigated further.
In summary, we replicated the findings of Huang et al. (2015) while extending the study to a larger age range. Men born in winter had lower LF/HF compared to other seasons. This difference was not found in women. Sex differences were widely observed in HRV metrics as well as age differences. Women expressed higher heart rates but also higher HRV than men. LF/HF increased with age. RR increased with age, and LF and HF power decreased. Furthermore, LF/HF was related to depressive symptoms and personality trait openness in men. In conclusion, with this study we have replicated the earlier study by Huang et al., extending the findings to the full age lifespan, a wide geographic distribution, HRV measurements throughout the year, and additional measures that could be of interest to better understand the relationship between HRV and seasonality.
ACKNOWLEDGMENTS
We acknowledge the data and support provided by BRAINnet; http://www.brainnet.net, under the governance of the BRAINnet Foundation. BRAINnet is the scientific network that coordinates access to the Brain Resource International Database for independent scientific purposes. We express our particular gratitude to Taylor Braund for his contribution. M.A. reports options from Brain Resource (Sydney, Australia); he is director and owner of Research Institute Brainclinics and a minority shareholder in neuroCare Group (Munich, Germany).
Iseger TA, Vollebregt MA, Krepel N, Arns M. Heart rate variability related to season of birth: A replication study. Psychophysiology. 2019;56:e13419 10.1111/psyp.13419
REFERENCES
- Abhishekh, H. A. , Nisarga, P. , Kisan, R. , Meghana, A. , Chandran, S. , Trichur, R. , & Sathyaprabha, T. N. (2013). Influence of age and gender on autonomic regulation of heart. Journal of Clinical Monitoring and Computing, 27(3), 259–264. 10.1007/s10877-012-9424-3 [DOI] [PubMed] [Google Scholar]
- Alvares, G. A. , Quintana, D. S. , Hickie, I. B. , & Guastella, A. J. (2016). Autonomic nervous system dysfunction in psychiatric disorders and the impact of psychotropic medications: A systematic review and meta‐analysis. Journal of Psychiatry & Neuroscience, 41(2), 89–104. 10.1503/jpn.140217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baek, H. J. , Cho, C.‐H. , Cho, J. , & Woo, J.‐M. (2015). Reliability of ultra‐short‐term analysis as a surrogate of standard 5‐min analysis of heart rate variability. Telemedicine Journal and e‐Health, 21(5), 404–414. 10.1089/tmj.2014.0104 [DOI] [PubMed] [Google Scholar]
- Bai, X. , Li, J. , Zhou, L. , & Li, X. (2009). Influence of the menstrual cycle on nonlinear properties of heart rate variability in young women. American Journal of Physiology. Heart and Circulatory Physiology, 297(2), H765–H774. 10.1152/ajpheart.01283.2008 [DOI] [PubMed] [Google Scholar]
- Banegas, J. R. , Rodríguez‐Artalejo, F. , de la Cruz, J. J. , Graciani, A. , Villar, F. , & del Rey‐Calero, J. (2000). Adult men born in spring have lower blood pressure. Journal of Hypertension, 18(12), 1763–1766. [DOI] [PubMed] [Google Scholar]
- Billman, G. E. (2013). The LF/HF ratio does not accurately measure cardiac sympatho‐vagal balance. Frontiers in Physiology, 4, 26 10.3389/fphys.2013.00026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brewerton, T. D. , Putnam, K. T. , Lewine, R. R. J. , & Risch, S. C. (2018). Seasonality of cerebrospinal fluid monoamine metabolite concentrations and their associations with meteorological variables in humans. Journal of Psychiatric Research, 99, 76–82. 10.1016/j.jpsychires.2018.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunoni, A. R. , Kemp, A. H. , Dantas, E. M. , Goulart, A. C. , Nunes, M. A. , Boggio, P. S. , … Benseñor, I. M. (2013). Heart rate variability is a trait marker of major depressive disorder: Evidence from the sertraline vs. electric current therapy to treat depression clinical study. International Journal of Neuropsychopharmacology, 16(9), 1937–1949. 10.1017/S1461145713000497 [DOI] [PubMed] [Google Scholar]
- Canpolat, U. , Özcan, F. , Özeke, Ö. , Turak, O. , Yayla, Ç. , Açıkgöz, S. K. , … Aydoğdu, S. (2015). Impaired cardiac autonomic functions in apparently healthy subjects with vitamin D deficiency. Annals of Noninvasive Electrocardiology, 20(4), 378–385. 10.1111/anec.12233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chalmers, J. A. , Quintana, D. S. , Abbott, M. J.‐A. , & Kemp, A. H. (2014). Anxiety disorders are associated with reduced heart rate variability: A meta‐analysis. Frontiers in Psychiatry, 5, 80 10.3389/fpsyt.2014.00080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark, C. R. , Paul, R. H. , Williams, L. M. , Arns, M. , Fallahpour, K. , Handmer, C. , & Gordon, E. (2006). Standardized assessment of cognitive functioning during development and aging using an automated touchscreen battery. Archives of Clinical Neuropsychology, 21(5), 449–467. 10.1016/j.acn.2006.06.005 [DOI] [PubMed] [Google Scholar]
- Čukić, I. , & Bates, T. (2014). Openness to experience and aesthetic chills: Links to heart rate sympathetic activity. Personality and Individual Differences, 64, 152–156. 10.1016/j.paid.2014.02.012 [DOI] [Google Scholar]
- Deakin, J. (1998). The role of serotonin in depression and anxiety. European Psychiatry, 13(Suppl 2), 57s–63s. 10.1016/S0924-9338(98)80015-1 [DOI] [PubMed] [Google Scholar]
- De Meersman, R. E. , & Stein, P. K. (2007). Vagal modulation and aging. Biological Psychology, 74(2), 165–173. 10.1016/j.biopsycho.2006.04.008 [DOI] [PubMed] [Google Scholar]
- Disanto, G. , Morahan, J. M. , Lacey, M. V. , DeLuca, G. C. , Giovannoni, G. , Ebers, G. C. , & Ramagopalan, S. V. (2012). Seasonal distribution of psychiatric births in England. PLOS One, 7(4), e34866 10.1371/journal.pone.0034866 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehrenthal, J. C. , Herrmann‐Lingen, C. , Fey, M. , & Schauenburg, H. (2010). Altered cardiovascular adaptability in depressed patients without heart disease. World Journal of Biological Psychiatry, 11(3), 586–593. 10.3109/15622970903397714 [DOI] [PubMed] [Google Scholar]
- Griffiths, K. R. , Quintana, D. S. , Hermens, D. F. , Spooner, C. , Tsang, T. W. , Clarke, S. , & Kohn, M. R. (2017). Sustained attention and heart rate variability in children and adolescents with ADHD. Biological Psychology, 124, 11–20. 10.1016/j.biopsycho.2017.01.004 [DOI] [PubMed] [Google Scholar]
- Henry, J. D. , & Crawford, J. R. (2005). The short‐form version of the Depression Anxiety Stress Scales (DASS‐21): Construct validity and normative data in a large non‐clinical sample. British Journal of Clinical Psychology, 44(Pt 2), 227–239. 10.1348/014466505X29657 [DOI] [PubMed] [Google Scholar]
- Hillebrand, S. , Gast, K. B. , de Mutsert, R. , Swenne, C. A. , Jukema, J. W. , Middeldorp, S. , … Dekkers, O. M. (2013). Heart rate variability and first cardiovascular event in populations without known cardiovascular disease: Meta‐analysis and dose‐response meta‐regression. Europace, 15(5), 742–749. 10.1093/europace/eus341 [DOI] [PubMed] [Google Scholar]
- Huang, W.‐L. , Chang, L.‐R. , Kuo, T. B. J. , Lin, Y.‐H. , Chen, Y.‐Z. , & Yang, C. C. H. (2013). Gender differences in personality and heart‐rate variability. Psychiatry Research, 209(3), 652–657. 10.1016/j.psychres.2013.01.031 [DOI] [PubMed] [Google Scholar]
- Huang, W.‐L. , Hwang, B.‐T. , Lai, C.‐T. , Li, J.‐Y. , Kuo, T. B. J. , & Yang, C. C. H. (2015). Is heart rate variability related to season of birth? Clinical Cardiology, 38(7), 407–412. 10.1002/clc.22410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kemp, A. H. , Quintana, D. S. , Gray, M. A. , Felmingham, K. L. , Brown, K. , & Gatt, J. M. (2010). Impact of depression and antidepressant treatment on heart rate variability: A review and meta‐analysis. Biological Psychiatry, 67(11), 1067–1074. 10.1016/j.biopsych.2009.12.012 [DOI] [PubMed] [Google Scholar]
- Koenig, J. , & Thayer, J. F. (2016). Sex differences in healthy human heart rate variability: A meta‐analysis. Neuroscience and Biobehavioral Reviews, 64, 288–310. 10.1016/j.neubiorev.2016.03.007 [DOI] [PubMed] [Google Scholar]
- Kozlowska, K. , Palmer, D. M. , Brown, K. J. , Scher, S. , Chudleigh, C. , Davies, F. , & Williams, L. M. (2015). Conversion disorder in children and adolescents: A disorder of cognitive control. Journal of Neuropsychology, 9(1), 87–108. 10.1111/jnp.12037 [DOI] [PubMed] [Google Scholar]
- Kristal‐Boneh, E. , Froom, P. , Harari, G. , Malik, M. , & Ribak, J. (2000). Summer‐winter differences in 24 h variability of heart rate. Journal of Cardiovascular Risk, 7(2), 141–146. 10.1177/204748730000700209 [DOI] [PubMed] [Google Scholar]
- Licht, C. M. , de Geus, E. J. , Zitman, F. G. , Hoogendijk, W. J. , van Dyck, R. , & Penninx, B. W. (2008). Association between major depressive disorder and heart rate variability in the Netherlands study of depression and anxiety (NESDA). Archives of General Psychiatry, 65(12), 1358–1367. 10.1001/archpsyc.65.12.1358 [DOI] [PubMed] [Google Scholar]
- Liu, C. C. , Kuo, T. B. J. , & Yang, C. C. H. (2003). Effects of estrogen on gender‐related autonomic differences in humans. American Journal of Physiology. Heart and Circulatory Physiology, 285(5), H2188–H2193. 10.1152/ajpheart.00256.2003 [DOI] [PubMed] [Google Scholar]
- Losonczy, M. F. , Mohs, R. C. , & Davis, K. L. (1984). Seasonal variations of human lumbar CSF neurotransmitter metabolite concentrations. Psychiatry Research, 12(1), 79–87. 10.1016/0165-1781(84)90140-9 [DOI] [PubMed] [Google Scholar]
- Mann, M. C. , Exner, D. V. , Hemmelgarn, B. R. , Sola, D. Y. , Turin, T. C. , Ellis, L. , & Ahmed, S. B. (2013). Vitamin D levels are associated with cardiac autonomic activity in healthy humans. Nutrients, 5(6), 2114–2127. 10.3390/nu5062114 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCraty, R. , & Shaffer, F. (2015). Heart rate variability: New perspectives on physiological mechanisms, assessment of self‐regulatory capacity, and health risk. Global Advances in Health and Medicine, 4(1), 46–61. 10.7453/gahmj.2014.073 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monnard, C. R. , & Grasser, E. K. (2017). Water ingestion decreases cardiac workload time‐dependent in healthy adults with no effect of gender. Scientific Reports, 7(1), 7939 10.1038/s41598-017-08446-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nimptsch, K. , Platz, E. A. , Willett, W. C. , & Giovannucci, E. (2012). Association between plasma 25‐OH vitamin D and testosterone levels in men. Clinical Endocrinology, 77(1), 106–112. 10.1111/j.1365-2265.2012.04332.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nonaka, K. , & Imaizumi, Y. (2000). Deaths from cerebrovascular diseases correlated to month of birth: Elevated risk of death from subarachnoid hemorrhage among summer‐born. International Journal of Biometeorology, 44(4), 182–185. 10.1007/s004840000068 [DOI] [PubMed] [Google Scholar]
- Paul, R. H. , Gunstad, J. , Cooper, N. , Williams, L. M. , Clark, C. R. , Cohen, R. A. , … Gordon, E. (2007). Cross‐cultural assessment of neuropsychological performance and electrical brain function measures: Additional validation of an international brain database. International Journal of Neuroscience, 117(4), 549–568. 10.1080/00207450600773665 [DOI] [PubMed] [Google Scholar]
- Porges, S. W. (1995). Orienting in a defensive world: Mammalian modifications of our evolutionary heritage. A polyvagal theory. Psychophysiology, 32(4), 301–318. 10.1111/j.1469-8986.1995.tb01213.x [DOI] [PubMed] [Google Scholar]
- Reffelmann, T. , Ittermann, T. , Empen, K. , Dörr, M. , & Felix, S. B. (2011). Is cardiovascular mortality related to the season of birth? Evidence from more than 6 million cardiovascular deaths between 1992 and 2007. Journal of the American College of Cardiology, 57(7), 887–888. 10.1016/j.jacc.2010.10.021 [DOI] [PubMed] [Google Scholar]
- Renner, F. , Penninx, B. W. J. H. , Peeters, F. , Cuijpers, P. , & Huibers, M. J. H. (2013). Two‐year stability and change of neuroticism and extraversion in treated and untreated persons with depression: Findings from the Netherlands study of depression and anxiety (NESDA). Journal of Affective Disorders, 150(2), 201–208. 10.1016/j.jad.2013.03.022 [DOI] [PubMed] [Google Scholar]
- Reyes del Paso, G. A. , Langewitz, W. , Mulder, L. J. , van Roon, A. , & Duschek, S. (2013). The utility of low frequency heart rate variability as an index of sympathetic cardiac tone: A review with emphasis on a reanalysis of previous studies. Psychophysiology, 50(5), 477–487. 10.1111/psyp.12027 [DOI] [PubMed] [Google Scholar]
- Schmidt, S. (2009). Shall we really do it again? The powerful concept of replication is neglected in the social science. Review of General Psychology, 13(2), 90–100. 10.1037/a0015108 [DOI] [Google Scholar]
- Shaffer, F. , & Ginsberg, J. P. (2017). An overview of heart rate variability metrics and norms. Frontiers in Public Health, 5, 258 10.3389/fpubh.2017.00258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shepherd, D. , Mulgrew, J. , & Hautus, M. J. (2015). Exploring the autonomic correlates of personality. Autonomic Neuroscience: Basic & Clinical, 193, 127–131. 10.1016/j.autneu.2015.05.004 [DOI] [PubMed] [Google Scholar]
- Sohn, K. (2016). The influence of birth season on mortality in the United States. American Journal of Human Biology, 28(5), 662–670. 10.1002/ajhb.22848 [DOI] [PubMed] [Google Scholar]
- Takase, B. , Akima, T. , Satomura, K. , Ohsuzu, F. , Mastui, T. , Ishihara, M. , & Kurita, A. (2004). Effects of chronic sleep deprivation on autonomic activity by examining heart rate variability, plasma catecholamine, and intracellular magnesium levels. Biomedicine & Pharmacotherapy, 58(Suppl 1), S35–S39. 10.1016/S0753-3322(04)80007-6 [DOI] [PubMed] [Google Scholar]
- Thayer, J. F. , & Lane, R. D. (2000). A model of neurovisceral integration in emotion regulation and dysregulation. Journal of Affective Disorders, 61(3), 201–216. 10.1016/S0165-0327(00)00338-4 [DOI] [PubMed] [Google Scholar]
- Thayer, J. F. , Yamamoto, S. S. , & Brosschot, J. F. (2010). The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. International Journal of Cardiology, 141(2), 122–131. 10.1016/j.ijcard.2009.09.543 [DOI] [PubMed] [Google Scholar]
- Torrey, E. F. , Rawlings, R. R. , Ennis, J. M. , Merrill, D. D. , & Flores, D. S. (1996). Birth seasonality in bipolar disorder, schizophrenia, schizoaffective disorder and stillbirths. Schizophrenia Research, 21(3), 141–149. 10.1016/0920-9964(96)00022-9 [DOI] [PubMed] [Google Scholar]
- Udupa, K. , Sathyaprabha, T. N. , Thirthalli, J. , Kishore, K. R. , Lavekar, G. S. , Raju, T. R. , & Gangadhar, B. N. (2007). Alteration of cardiac autonomic functions in patients with major depression: A study using heart rate variability measures. Journal of Affective Disorders, 100(1–3), 137–141. 10.1016/j.jad.2006.10.007 [DOI] [PubMed] [Google Scholar]
- Uijtdehaage, S. H. , & Thayer, J. F. (2000). Accentuated antagonism in the control of human heart rate. Clinical Autonomic Research, 10(3), 107–110. 10.1007/BF02278013 [DOI] [PubMed] [Google Scholar]
- Umetani, K. , Singer, D. H. , McCraty, R. , & Atkinson, M. (1998). Twenty‐four hour time domain heart rate variability and heart rate: Relations to age and gender over nine decades. Journal of the American College of Cardiology, 31(3), 593–601. 10.1016/S0735-1097(97)00554-8 [DOI] [PubMed] [Google Scholar]
- Voss, A. , Schulz, S. , Schroeder, R. , Baumert, M. , & Caminal, P. (2009). Methods derived from nonlinear dynamics for analysing heart rate variability. Philosophical Transactions. Series A, Mathematical, Physical, and Engineering Sciences, 367(1887), 277–296. 10.1098/rsta.2008.0232 [DOI] [PubMed] [Google Scholar]
- Wang, Y. , Hensley, M. K. , Tasman, A. , Sears, L. , Casanova, M. F. , & Sokhadze, E. M. (2015). Heart rate variability and skin conductance during repetitive TMS course in children with autism. Applied Psychophysiology and Biofeedback, 41(1), 47–60. 10.1007/s10484-015-9311-z [DOI] [PubMed] [Google Scholar]
- Williams, D. P. , Cash, C. , Rankin, C. , Bernardi, A. , Koenig, J. , & Thayer, J. F. (2015). Resting heart rate variability predicts self‐reported difficulties in emotion regulation: A focus on different facets of emotion regulation. Frontiers in Psychology, 6, 261 10.3389/fpsyg.2015.00261 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams, L. M. , Simms, E. , Clark, C. R. , Paul, R. H. , Rowe, D. , & Gordon, E. (2005). The test‐retest reliability of a standardized neurocognitive and neurophysiological test battery: "Neuromarker". International Journal of Neuroscience, 115(12), 1605–1630. 10.1080/00207450590958475 [DOI] [PubMed] [Google Scholar]
- Wranicz, J. K. , Rosiak, M. , Cygankiewicz, I. , Kula, P. , Kula, K. , & Zareba, W. (2004). Sex steroids and heart rate variability in patients after myocardial infarction. Annals of Noninvasive Electrocardiology, 9(2), 156–161. 10.1111/j.1542-474X.2004.92539.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young, H. , & Benton, D. (2015). We should be using nonlinear indices when relating heart‐rate dynamics to cognition and mood. Scientific Reports, 5, 16619 10.1038/srep16619 [DOI] [PMC free article] [PubMed] [Google Scholar]


