Abstract
Vascular calcification (VC) is highly prevalent in patients with advanced age, or those with chronic kidney disease and diabetes, accounting for substantial global cardiovascular burden. The pathophysiology of VC involves active mineral deposition by transdifferentiated vascular smooth muscle cells exhibiting osteoblast-like behavior, building upon cores with or without apoptotic bodies. Oxidative stress drives the progression of the cellular phenotypic switch and calcium deposition in the vascular wall. In this review, we discuss potential compounds that shield these cells from the detrimental influences of reactive oxygen species as promising treatment options for VC. A comprehensive summary of the current literature regarding antioxidants for VC is important, as no effective therapy is currently available for this disease. We systematically searched through the existing literature to identify original articles investigating traditional antioxidants and novel compounds with antioxidant properties with regard to their effectiveness against VC in experimental or clinical settings. We uncovered 36 compounds with antioxidant properties against VC pathology, involving mechanisms such as suppression of NADPH oxidase, BMP-2, and Wnt/β-catenin; anti-inflammation; and activation of Nrf2 pathways. Only two compounds have been tested clinically. These findings suggest that a considerable opportunity exists to harness these antioxidants for therapeutic use for VC. In order to achieve this goal, more translational studies are needed.
Subject terms: Calcification, Cell biology
Facts
Anti-oxidants are compounds that counteract oxidative stress within cellular microenvironments, divided broadly into those of natural and non-natural origin.
Reactive oxygen species are vital pathogenic players in atherosclerotic cardiovascular diseases, against which anti-oxidants can be excellent sources of treatment candidates.
Vascular calcification (VC) contributes significantly to cardiovascular morbidity and mortality in at-risk population including those with diabetes and kidney diseases, but effective therapies for VC remain in question.
Open questions
To what extent anti-oxidative therapies have been utilized to treat VC experimentally and clinically?
What molecular mechanisms are involved in the observed therapeutic efficacy of antioxidants against VC?
How to choose the optimal dose and duration of different anti-oxidants for the treatment of VC in light of the existing literature?
Introduction
Cardiovascular diseases are the leading cause of mortality worldwide, accounting for nearly one-fourth of total deaths globally, while real-world data suggest that the burden is likely higher than we deduce based on trial information1. Despite the substantial benefit conferred by the widespread use of statin for cholesterol reduction as well as the promotion of guideline adherence, the global mortality due to cardiovascular diseases continued to increase from the 1990s to 2013 (ref. 2). This trend has been partly attributed to population aging, but risk factors other than demographic influences likely contribute as well, such as atypical factors including chronic inflammation, mineral dysregulation, and excess pathogenic hormonal milieu3. Findings from previous studies suggested vascular calcification (VC) as an important phenotype arising from the influences of atypical cardiovascular risk factors4. Indeed, VC is highly prevalent in certain patient populations, including those with chronic kidney disease (CKD) or diabetes mellitus, and the presence of VC is predictive of an increased risk of cardiovascular diseases, overall mortality, and hospitalization independent of multiple confounders5. This detrimental influence of VC is mainly related to enhanced arterial stiffness, ventricular pressure load with hypertrophy and coronary ischemia. However, effective therapies for VC remain scant, and even the promising ones involve the optimization of divalent ions and interplay between hormones within the scope of mineral bone disorder without proven efficacy6. Consequently, novel therapies that tackle alternative aspects of VC pathophysiology are urgently needed.
The central role of oxidative stress in VC
The pathophysiology of VC is complex, but most agree that active osteoid matrix-like deposition by transdifferentiated vascular smooth muscle cells (VSMCs) or infrequently by other vascular wall constituent cells plays an important role. This highly regulated cellular phenotypic switch, accompanied by the loss of contractile function (for VSMCs) and the gain of synthetic function, can be stimulated by disturbed mineral homeostasis (especially a high phosphate environment), hyperglycemia with advanced glycation endproduct (AGE) accumulation, or inflammatory cytokines, which coincide with the clinical features of the high-risk population for developing VC7. The process of active mineralization is further augmented by the loss of calcification inhibitors such as matrix Gla protein (MGP), pyrophosphate, or fetuin-A8.
A core component that drives the phenotypic change of VSMCs during VC is believed to be premature vascular aging and VSMC senescence9. Oxidative stress (OS), manifested by the production of reactive oxygen species (ROS) and counteracted by antioxidant mechanisms, is a major inducer of cellular senescence. OS presumably induces damages to vital cellular substructures including DNA, protein, and membrane/cytoplasmic lipids. The sources of ROS in calcified VSMCs include cellular enzymes such as xanthine oxidase, nicotinamide adenine dinucleotide phosphate oxidase (NADPH oxidase, Nox), and the cytochrome P450 systems. The downstream events of redox imbalance may include telomere shortening, mitochondrial DNA damage, and direct mutagenic influences on gene fragments involved in contractile functions, thereby predisposing VSMCs to aging and the loss of smooth muscle cell phenotypes. These events are followed by osteogenic transcription factor activation10,11. In addition, mitochondrial damage can be accompanied by further leakage of ROS from inner mitochondrial membrane, perpetuating a vicious cycle of ROS accumulation and a greater degree of apoptosis. Inflammation following cytokine release triggered by OS can further set the stage for VC development.
In light of the importance of ROS in the pathogenesis of VC, it is expected that antioxidants can serve as a promising therapeutic approach for managing VC, a disorder currently without effective treatments12. We thus performed a literature-based review to summarize the existing knowledge with regard to the utility of antioxidants against VC, in experimental models and in clinical studies.
Strategy of literature search
We systematically searched through the existing literature, using the following combinations of keywords and MESH terms: “vascular smooth muscle cells”, “calcification”, or “vascular calcification” and “reactive oxygen species”, “oxidative stress”, “antioxidant”, or “anti-oxidant” in the PUBMED and MEDLINE databases up to 7 June 2019 to retrieve relevant articles for analysis; 310 articles were identified, of which 101 were excluded as duplicate entries (Fig. 1). We subsequently reviewed the abstracts of the remaining 209 articles in depth, and excluded review articles (n = 53) and those for which the full text was unavailable (n = 1), after which these articles were classified into clinical (n = 47) and experimental (n = 108) research studies. For clinically oriented studies, we further excluded those whose focuses were not therapeutic, therapeutic but not against VC, or case reports; for experimental ones, we excluded those not examining chemicals, examining pathogenesis only, examining non-VC pathologies, or those examining aggravating factors (Fig. 1).
Fig. 1. The algorithm of study retrieval from the literature and the application of selection criteria.

VC vascular calcification
The remaining 52 articles with their full-texts were finally reviewed with data extracted for description. Among these, 5 (9.6%) involved clinical studies that examined antioxidants for treatment of VC, while 47 (90.4%) involved experimental studies. For clinical therapeutic studies, four (80%) used parenteral sodium thiosulfate (STS), while one (20%) used topical cerium nitrate13–17. For experimental approaches, totally 36 compounds with antioxidant property have been tested against VC, including 12 natural dietary substances, 5 natural non-dietary substances, and 19 pharmaceuticals or synthetic chemicals (Fig. 2)18–64. Among compounds from each category, they are further divided into those with and without ROS scavenging ability.
Fig. 2. The proportion of identified antioxidant compounds within each category, stratified based on the presence or absence of ROS scavenging ability.
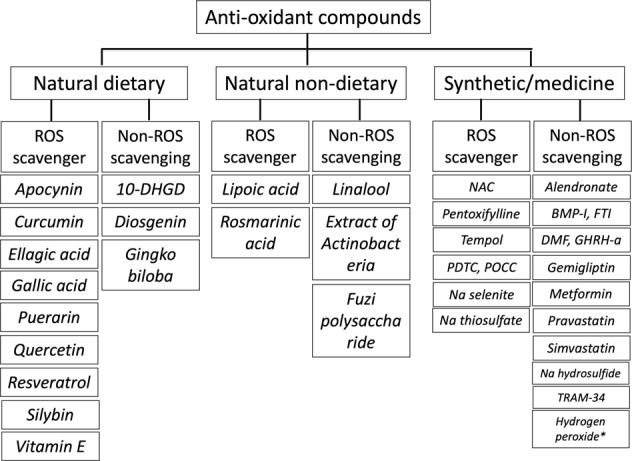
10-DHGD 10-dehydrogingerdione, BMP-I bone morphogenetic protein inhibitor, DMF dimethylfumarate, FTI farnesyl transferase inhibitor, GHRH-a growth hormone-releasing hormone receptor agonist, NAC N-acetylcysteine, PDTC pyrrolidine dithiocarbamate, POCC poly(1,8-octamethylene-citrate-co-cysteine), ROS reactive oxygen species. * Very low dose (0.01 mM)
We examined candidate antioxidants with regard to their effects on VC and the potential mechanisms involved if the articles used experimental approaches, and the antioxidants that have been tested for therapeutic efficacy against VC in clinical settings, in the following sections. Compounds which have been tested in animal models are tabulated separately to outline the dosage and models through which anti-calcification effect are shown separately (Table 1).
Table 1.
Summary of animal studies investigating the effect of anti-oxidants on vascular calcification
| VC models | Animal | Antioxidants | Doses | Effect | Reference |
|---|---|---|---|---|---|
| Adenine-induced CKD | Rat | Sodium thiosulfate | 0.4 g/kg thrice weekly IP | Reduced aortic calcification, serum Ca2+, induced metabolic acidosis and calciuria | 36 |
| High cholesterol diet + VitD | Rabbit | Lipoic acid | 120 µmol/kg/day PO | Decrease AV calcification | 22 |
| ApoE−/− with nephrectomy | Mouse | Simvastatin | 100 mg/kg/day PO | Decrease aortic plaque calcification; neutral serum cholesterol levels | 32 |
| LDLR−/− + high fat diet | Mouse | BMP antagonist | 2.5 mg/kg/day IP | Reduced aortic atheroma, calcification, and cholesterol levels | 39 |
| Adenine-induced CKD | Rat | Tempol | 3 mM in drinking water PO | Reduced aortic calcification; neutral serum Ca2+/PO4−, vitamin D, PTH levels | 40 |
| High dose VitD | Mouse | Lipoic acid | 40 mg/kg/day IP | Reduced aortic calcification, apoptosis, and improved mitochondrial function | 23 |
| ApoE−/− + partial renal ablation | Mouse | Farnesyltransferase inhibitor | 50 mg/kg/day IP | Reduced aortic atheroma, calcification, and plaque collagen amount | 41 |
| Warfarin-induced VC | Rat | Quercetin | 10 mg/kg/day PO | Reduced aortic calcification and lower SBP/PP | 26 |
| MGP−/− | Mouse | Quercetin | 0.02% in drinking water PO | Reduced aortic calcification | 28 |
| Adenine-induced CKD | Rat | Diosgenin | 10–40 mg/kg/day PO | Reduced aortic calcification; neutral serum Ca2+/PO4−, uric acid, creatinine levels | 20 |
| High fat diet + STZ + warfarin | Rat | Apocynin | 2.5 mg/kg/day PO | Reduced femoral artery calcification and tissue AGEs | 18 |
| High cholesterol diet + VitD | Rabbit | Lipoic acid | 0.12 mmol/kg/day PO | Reduced aortic calcification, stiffness, and preserved vasoconstrictor responses | 24 |
| Adenine-induced CKD | Rat | Diosgenin | 40 mg/kg/day PO | Reduced aortic RUNX2 expression and decreased coronary resistance | 21 |
| Zucker obese animals + 5/6 nephrectomy + VitD | Rat | Vitamin E | 30,000 mg/kg PO | Reduced aortic calcification | 35 |
| High fat diet + 5/6 nephrectomy | Rat | Vitamin E | 30,000 mg/kg PO | Reduced aortic, gastric, and pulmonary calcium content | 55 |
| Adenine-induced CKD | Rat | Quercetin | 25 mg/kg/day PO | Reduced aortic calcification and lower phosphate, creatinine, and uric acid levels | 29 |
| l-NAME-induced hypertension | Rat | Ellagic acid | 10–30 mg/kg/day PO | Reduced aortic calcification, aortic thickness, SBP, and restored vasoconstrictor/dilator responses | 56 |
| Adenine-induced CKD | Rat | Gemigliptin | 10–20 mg/kg/day IP | Reduced aortic calcification | 57 |
| Adenine-induced CKD | Rat | Quercetin | 100 mg/kg/day PO | Reduced aortic calcification and restore aortic mitochondrial integrity | 30 |
| OPG−/− | Mouse | GHRH agonist | 5 µg/kg/day SC | Reduced aortic calcification | 58 |
| Adenine-induced CKD | Rat | Linalool | 100–150 mg/kg/day PO | Reduced aortic calcification, aortic ROS scavenging | 61 |
| 5/6 nephrectomy + high Ca/P diet + VitD | Rat | Puerarin | 400 mg/kg/day PO | Reduced aortic calcification and lower inflammatory cytokines | 62 |
| High cholesterol diet | Rabbit | 10-DHGD, Pentoxifylline | 10-DHGD (10 mg/kg/day PO) Pentoxifylline (40 mg/kg/day PO) | Reduced aortic calcification, lower serum total/LDL/HDL cholesterol, triglyceride | 64 |
10-DHGD 10-dehydrogingerdione, AGE advanced glycation endproducts, AV aortic valve, BMP bone morphogenic factor, CKD chronic kidney disease, GHRH growth hormone-releasing hormone, HDL high-density lipoprotein, IP intraperitoneal, LDLR low-density lipoprotein receptor, l-NAME N(ω)-nitro-l-arginine methyl ester, MGP matrix Gla protein, OPG osteoprotegerin, PO per oral, PP pulse pressure, PTH parathyroid hormone, ROS reactive oxygen species, SBP systolic blood pressure, SC subcutaneous, STZ streptozotocin, VC vascular calcification, VitD vitamin D
Antioxidants from natural and dietary sources for treatment of VC
To date, in vitro and in vivo experiments have tested 12 natural antioxidants that are also dietary ingredients, including 10-dehydrogingerdione (10-DHGD)64, apocynin18,19, curcumin38, diosgenin20,21, ellagic acid56, gallic acid48, gingko biloba extracts50, puerarin62, quercetin25–31, resveratrol52, silybin38, and vitamin E34,35. Among these compounds, apocynin, diosgenin, quercetin, and vitamin E have been shown by multiple studies to have anti-VC properties. Nine (75%) of these compounds possess ROS scavenging ability, while others do not (Fig. 2).
Apocynin is isolated from the plant Apocynum, also known as dogbane, and has been used as a herbal medicine by Native Americans and as herbal tea in several Asian countries65. Through inhibition of Nox, apocynin has been found to reduce superoxide generation and ameliorate inflammation in various animal models66. Brodeur et al. found that the administration of apocynin to streptozotocin-induced diabetic rats with warfarin-induced arterial calcification could substantially improve the severity of femoral artery VC, although the mechanism involved has not been tested18. Other studies further disclosed that apocynin ameliorated VC by suppressing bone morphogenetic protein-2 (BMP-2) expression and attenuating the phosphorylation of ERK1/2, thereby reducing RUNX2 and osteopontin levels based on an angiotensin II-induced osteogenic transformed VSMCs model19. Diosgenin is a phytosteroidal saponin purified from fenugreek (Trigonella foenum-graecum) seeds and can be biosynthesized from cholesterol. Previous studies demonstrated that diosgenin possessed lipid and glucose-lowering effects in addition to anti-inflammatory and antioxidant properties67. Manivannan et al. found that diosgenin could reduce aortic calcification in CKD rats in a dose-dependent manner by increasing antioxidant enzyme levels, lowering lipid peroxidation20, and stimulating endothelial nitric oxide synthase (eNOS) activities, thereby improving coronary reserves21. Vitamin E, including lipophilic tocopherols and tocotrienols, is universally found in green vegetables, nuts, eggs, and milk, and has long been known to exhibit strong antioxidant properties. Mody et al., in a pilot study nearly two decades ago, found that a vitamin E analog could halt the osteoblastic differentiation of aortic VSMCs induced by oxidized low-density lipoprotein (ox-LDL)34. Vitamin E has subsequently been shown to restore plasma antioxidant levels, reduce AGEs, and suppress aortic calcification severity in obese CKD rats and in human VSMCs35.
Quercetin, a naturally occurring flavanol and a secondary plant metabolite, is enriched in many fruits and vegetables and is also used as a dietary supplement68. It has been extensively investigated with respect to its effect against VC based on our literature search results. Lu et al. disclosed that quercetin abolished heat shock protein 72 induced anti-calcification effect in vitro and ex vivo25; however, another group later found that quercetin completely nullified warfarin-induced aortic calcification and aortic medial cartilaginous metaplasia by inhibiting transglutaminase 2 and β-catenin expression in vitro26–28. Alternatively, quercetin could reduce aortic calcification in CKD rats partially by increasing superoxide dismutase 2 (SOD2) levels and modulating the inducible NO synthase (iNOS)/MAPK pathway29. Cui et al. further showed that quercetin restored mitochondrial integrity with improved membrane potential, ATP production, reduced fragmentation/fission, attenuated Drp1 phosphorylation, and reversed the apoptotic process in calcified VSMCs30. Recently, Liang et al. showed that quercetin might be protective for VSMCs by attenuating the expression of BMP-2 and toll-like receptor (TLR)−4 in ox-LDL-induced calcification31.
On the other hand, the antioxidant properties of 10-DHGD, curcumin, ellagic acid, gallic acid, gingko biloba, puerarin, resveratrol, and silybin were described in single reports only. 10-DHGD is a biologically active component extracted from the rhizomes of ginger (Zingiber officinale), a medicinal plant that is also used as dietary spices69. 10-DHGD has been shown to exhibit anti-inflammatory and antioxidant properties, and to stimulate NO release70. Elseweidy et al. revealed that 10-DHGD could attenuate the development of aortic atherosclerotic calcifications in dyslipidemic rabbits by downregulating the expression of aortic BMP-2, Wnt3a, tumor necrosis factor-α (TNF-α), receptor activator of nuclear factor-κB (RANK), and RUNX264. Curcumin is a polyphenol extracted from the dried Rhizomes of Curcuma longa and spices such as turmeric. It is known to possess anti-inflammatory and antioxidant properties71. Silybin is the main active ingredient of Silybum marianum extracts. It exhibits antioxidant property and is mostly used for treating viral hepatitis72. Roman-Garcia et al. showed that curcumin and silybin reduced ROS levels and calcification of VSMCs38. Being a natural polyphenol with antioxidant property, gallic acid and its derivative, ellagic acid, mainly exist in fruits such as pomegranates, grapes, berries, and tea leaves73. Gallic acid has been shown to reduce osteoblastic differentiation of VSMCs by suppressing BMP-2/Smad 1/5/8 signaling, while ellagic acid has been shown to decrease calcium deposition in aortic walls and improves blood pressure in hypertensive rats48,56. Ginkgo biloba extracts are mixtures containing several flavanols, ginkgolides, and bilobalides, and are found to be vasoactive and antioxidative74. Li et al. demonstrated that Ginkgo biloba could attenuate VSMC calcification by downregulating NF-κB and reducing ROS levels50. The primary extract of Pueraria lobata, puerarin, is used as an herbal medicine for treating acute ischemic stroke. Its active ingredient is an isoflavone glycoside75. Liu et al. disclosed that puerarin inhibited ROS production, reduced NF-κB and BMP-2 expression, and IL-1β levels, resulting in reduced aortic calcification62. Finally, resveratrol, a phytoalexin and polyphenol mainly contained in plants such as grapes and its derivatives such as red wine, has been repeatedly shown to be vasculo- and cardio-protective76. Zhang et al. found that resveratrol reduced VSMC calcification by activating Nrf2 and Sirt1, upregulating Klotho expression and inhibiting fibroblast growth factor-23 (ref. 52).
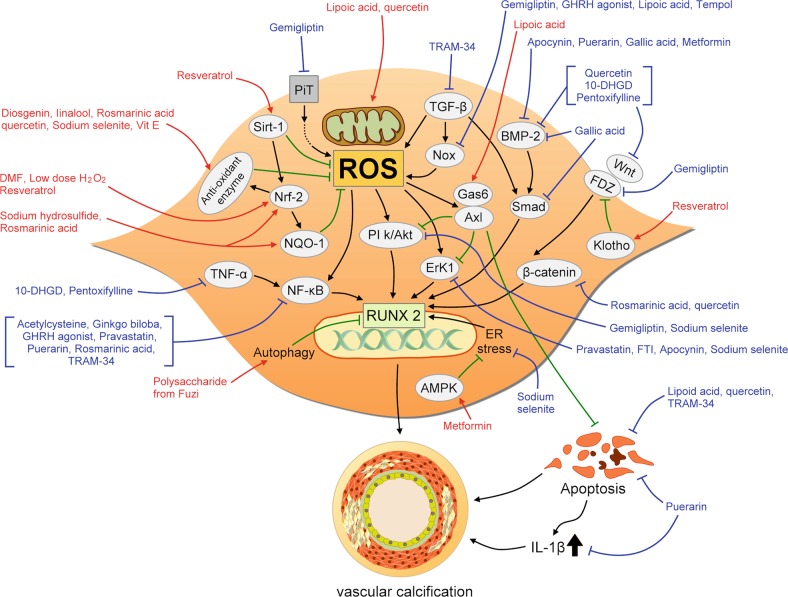
Based on the above reports, there is a high probability that multiple edible natural antioxidants can be potential candidates for managing VC, although the doses of each agent vary widely, and the mechanisms may differ depending on agent-specific or experimental model-specific issues. We have constructed an illustrative diagram to integrate the known and confirmed molecular alterations responsible for the beneficial effects of these nutraceuticals (Fig. 3).
Fig. 3. An illustrative diagram showing the plausible molecular mechanisms through which each antioxidant counteracts vascular calcification.
Red arrows indicate positive influences by antioxidants, while blue connecting lines indicate their inhibitory action. 10-DHGD 10-dehydrogingerdione, ER endoplasmic reticulum, FTI farnesyl transferase inhibitor, IL interleukin, Nox NADPH oxidase, ROS reactive oxygen species, GHRH growth hormone-releasing hormone, TGF transforming growth factor, TNF tumor necrosis factor
Antioxidants from natural non-dietary sources
We identified five types of potential antioxidants with therapeutic effects against VC based on the literature search, including linalool61, lipoic acid22–24, rosmarinic acid63, fermentation broth extracts from Actinobacteria59, and polysaccharide from Fuzi product60. Two (40%) of them have ROS scavenging ability, while the other three (60%) do not (Fig. 2).
Linalool, a natural terpene alcohol inherent to multiple types of flowers, is now a common constituent of fragrance with cosmetic and non-cosmetic applications77. Kaur et al. disclosed that linalool could scavenge ROS in VSMCs, increase antioxidant enzyme levels, and reduce the expression of PiT-1 and RUNX2 in CKD rats61. Lipoic acid, produced from octanoic acid in mitochondria, is a cofactor for multiple physiological reactions and a powerful antioxidant known to date. Several groups have proposed that lipoic acid could substantially attenuate VC in vitro and in vivo. Liberman et al. showed that lipoic acid ameliorated aortic calcification and improved vascular compliance in high cholesterol- and vitamin D-fed rabbits by inhibiting Nox4 expression and reducing ROS22,24. Similar to quercetin, lipoic acid exhibited a beneficial effect against VC by improving mitochondrial function and inhibiting apoptosis, through restoring Gas6/Axl survival signaling23. Rosmarinic acid is a phenolic compound purified from medicinal plants belonging to the families Apiaceae, Boraginaceae, and Lamiaceae with anti-inflammatory and antioxidant properties78. Ji et al. showed that rosmarinic acid reduced VSMC calcification by upregulating Nrf2, NADPH quinone dehydrogenase 1 (NQO1) expression, increasing antioxidant enzyme levels while inhibiting NF-κB and β-catenin signaling in vitro and in vivo63. Finally, extracts of bacterial and plant products are also found to counteract VC experimentally. Preliminary results from Liao et al. showed that polysaccharide extracted from Radix Aconiti Carmichaeli, a raw product with anti-oxidant activity, could reduce calcification severity of ox-LDL-treated VSMCs, partially by activating autophagy60. Salimi et al. also found that metabolites extracted from cultured Nocardia strain could attenuate protein oxidation, reduce inflammation, and reduce VSMC calcification severity59. The potential downstream effectors of these natural non-dietary compounds are summarized in Fig. 3. Although parts of the above compounds are derived by extracting botanical and bacterial metabolites and are likely heterogeneous, these findings suggest that a plentiful of natural substances can be tested as powerful treatments for VC.
Antioxidants of synthetic origin or pharmaceuticals
Multiple non-natural chemicals/medications also exhibit antioxidant properties and are effective against VC. We identified 19 types of such candidates with potential therapeutic effects against VC: acetylcysteine33, alendronate49, BMP inhibitor39, dimethyl fumarate47, farnesyltransferase inhibitor41, gemigliptin57, growth hormone-releasing hormone (GHRH) receptor agonist58, metformin42, pentoxifylline64, pravastatin33, pyrrolidine dithiocarbamate (PDTC)34, simvastatin32, sodium hydrosulfide53, sodium selenite37,45, STS36,43,44, Tempol40, TRAM-3446, topical poly(1,8-octamethylene-citrate-co-cysteine) (POCC)54, and hydrogen peroxide51. Among these, 11 (57.9%) were medications with specific clinical indications, while 8 (42.1%) were non-medicinal chemicals. Seven of them (36.8%) have ROS scavenging ability, while the other 12 (63.2%) do not (Fig. 2). We have briefly summarized the action of each agent in the following section.
STS is first such chemical being tested for its activity against VC and reaps the most attention by researchers in this field. Originally used for the treatment of cyanide toxicity, STS is also found to have strong antioxidant properties and a calcium-chelating tendency79. Pasch et al. showed that STS significantly reduced VC severity in uremic rats, and they proposed that STS acted by inducing acidosis and urinary excretion of calcium36. Subsequent studies suggested that STS could suppress BMP-2 expression and upregulate MGP in calcified VSMCs43, indicating that STS may interfere directly with the pathophysiology of VC in addition to its ability to bind calcium. With the assistance from adipocytes, STS may attenuate the calcification of arterial walls to a greater extent44.
The most renowned antioxidant medication is probably acetylcysteine, which acts through its conversion into substrates for hepatic glutathione synthesis, and is used clinically for managing acetaminophen overdose and for prophylaxis of contrast-induced kidney injury80. While examining the vasculopathic effect of protease inhibitors, Afonso et al. found that acetylcysteine could retard the progression of VSMC calcification through its antioxidant and anti-inflammatory capability33. Sodium selenite, a formula that delivers the trace element selenium to host individuals, has been tested in two studies for its anti-calcification ability, since selenium restoration has been shown to raise glutathione peroxidase activity and to attenuate ROS production in patients with different illnesses81. Liu et al. revealed that sodium selenite could inhibit the ERK1/2 pathway, leading to a lower severity of calcification in a model of H2O2-induced VSMC phenotypic switch37. They also found that sodium selenite suppresses OS by elevating glutathione peroxidase activity, inhibiting PI3K/Akt activity and endoplasmic reticulum stress in VSMCs45, supporting its use as a treatment for VC.
Other medications, including anti-osteoporotic medication (alendronate), oral hypoglycemic agent (gemigliptin and metformin), hypolipidemic agents (pravastatin and simvastatin), medication for multiple sclerosis (dimethyl fumarate), microcirculatory insufficiency (pentoxifylline), and growth hormone deficiency (GHRH receptor agonist) have all been associated with suppressed ROS production and attenuated VC in different experimental models. Cutini et al. showed that alendronate increased NO production and activated MAPK signaling pathway, while it attenuated inflammation and reduced VC severity49. Ha et al. discovered that dimethyl fumarate administration led to Nrf2 activation, BMP-2 downregulation, and less severe VC in a calcified VSMC model and in animals with vitamin D-induced aortic calcification47. Pentoxifylline is also shown to decrease the expression of BMP-2, Wnt3a, and RANK in addition to its anti-inflammatory and anti-calcific properties64. Gemigliptin belongs to the dipeptidyl peptidase-4 inhibitor class of antidiabetic agents, which exhibit a pleiotropic effect through glucagon-like peptide-1 (GLP-1)-dependent and -independent mechanisms including insulin sensitization and the inhibition of oxidation and apoptosis82. Choi et al. reported that gemigliptin attenuated VC through a variety of beneficial molecular alterations, including decreased expressions of PiT-1 and Nox4 with reduced ROS production, and suppressed PI3K-Akt and Wnt-FDZ pathway activities57. On the other hand, metformin has been shown to ameliorate VSMC calcification through AMPK-independent (BMP-2 inhibition) and AMPK-dependent (AMPK and eNOS activation) mechanisms42. Statins also exhibit anti-inflammatory and antioxidant properties, apart from their cholesterol-lowering effect. Ivanovski et al. first showed that simvastatin was capable of decreasing aortic OS markers and reducing aortic calcification in ApoE knockout mice with CKD, independent of cholesterol levels32. This is further supplemented by Afonso et al.’s work, which suggested that pravastatin could decrease local OS by inhibiting farnesyl transferase in calcified VSMCs33. GHRH receptor agonists, a hormonal agent not previously investigated for its vascular action, was shown to alleviate aortic calcification via NF-κB downregulation and the reduction in inflammation58. It seems that many of these antioxidant medications exert their actions by modulating NF-κB expression and lowering the contribution of inflammation to VC pathogenesis, although some positively influence vascular health through multiple mechanisms (Fig. 1). Judging from their efficacy against VC, it is expected that part of this medication list may eventually gain momentum as an effective therapy for VC treatment.
Several non-medicinal chemicals with antioxidant property are also found to protect against VC, serving as potential treatment candidates. It was expected that BMP inhibitors LDN-193189 would retard VC progression in LDL receptor knockout mice; pharmacological BMP inhibition additionally attenuated the production of ROS and reduced the severity of inflammation in vascular tissues, as shown by Derwall et al.39. The inhibition of protein prenylation, which markedly influences isoprenoid synthesis and mevalonate metabolism, using farnesyl transferase inhibitor R115777 has also been shown to reduce aortic tissue OS levels and ameliorate calcium deposition by inhibiting Ras-Raf signaling41. Interestingly, calcium-activated potassium channel blocker TRAM-34, which helps maintain VSMC membrane potential and affects intracellular Ca2+ signaling, was found to decrease VSMC calcification by inducing NO release, attenuating TGF-β signaling, and inhibiting NF-κB activation with reduced apoptosis46. The administration of sodium hydrosulfide, which strengthens endogenous antioxidant hydrogen sulfide action, was similarly found to reduce VC severity by decreasing H2O2 production, inhibiting TNF-α expression, and activating Nrf2-NQO1 pathway53. Dithiocarbamate compounds, such as PDTC, have been reported to exhibit an even stronger antioxidant property than N-acetylcysteine by inhibiting NF-κB activation83. Mody et al. showed that PDTC could substantially halt the process of calcification in vascular cells, although the mechanisms involved have not been explored34.
Synthetic compounds exhibiting antioxidant properties are also being used to fight VC. Poly(1,8-octamethylene citrate) (POC), an elastomer constructed as a biodegradable material, has been shown previously to decrease ROS production in neighboring tissues when coated onto vascular grafts84. Its derivative, POCC, when coated, was also found to attenuate local oxidized DNA and lipid damages, accompanied by a reduction in graft calcification54.
The confirmed effector mechanisms of these medicinal and non-medicinal chemicals with antioxidant properties against VC are summarized in an integrative way in Fig. 3.
Antioxidants with contradictory findings regarding VC
Some of these non-medicinal compounds have shown contradictory findings in the pathogenesis of VC. For example, tempol, a frequently used experimental superoxide dismutase mimetic, has been controversial regarding its effect on the progress of VC. Yamada et al. showed that tempol could reverse the osteoblastic transdifferentiation of VSMCs and improve uremic VC in CKD rats through ROS reduction and Nox4 inhibition40. However, Liberman et al. showed that in a rabbit model of atherosclerotic calcification, although tempol lowered aortic ROS levels, it paradoxically increased medial calcification and plaque burden24. This can be attributed to the species-dependent or calcification model-dependent actions of tempol, but further studies are needed before arriving at a definite conclusion. In addition, hydrogen peroxide is traditionally deemed an endogenous OS mediator and used exogenously as a ROS inducer. Intriguingly, Zhang et al. showed that a very low concentration (0.01 mM) of hydrogen peroxide could on the contrary reduce intracellular ROS generation and stimulate Nrf2 activity, decreasing the severity of VSMC calcification in vitro without inducing apoptosis51. This phenomenon is reminiscent of the physiological role of “preconditioning” maneuver, which protects specific organs from severe damage if they are subjected to mild insult beforehand. Nonetheless, their findings are still preliminary and need replication.
Clinically tested antioxidants for treatment of VC
We identified five clinical studies testing one parenteral and the other topical agent with an antioxidative ability regarding their efficacy in treating VC (Table 2)13–17. STS, which has been repeatedly tested in experimental models for inhibiting VC progression, is also used clinically to treat calcification involving arteries from different anatomical locations. However, it is evident that these studies are anecdotal, enrolling few patients, and the results appear heterogeneous with some showing calcification regression14,15 while others reporting no interval changes13,16. A topical chemical, cerium nitrate, has been shown to improve the overall outcome of patients with calciphylaxis when used for wound dressing, presumably through ROS scavenging and chelating local calcium17. Further studies are still needed to confirm these findings.
Table 2.
Clinical studies investigating the therapeutic effect of anti-oxidants in patients with clinical VC
| Medications | Sample size | doses | Disease type | Response | Reference |
|---|---|---|---|---|---|
| Sodium thiosulfate | 22 HD | 25 g thrice weekly IV for 3 months | Carotid, coronary, and aortic calcification | Mean annual VC changes did not increase | 13 |
| Sodium thiosulfate | 6 HD | 10 g thrice weekly in dialysate for 6 months | Coronary calcification | 66% had calcification reduction vs. 0% in control | 14 |
| Sodium thiosulfate | 18 HD | 10 g thrice weekly IV for 6 months | Abdominal aortic calcification | Modest calcification reduction but moderate leg pain improvement | 15 |
| Sodium thiosulfate | 38 HD | 0.18 g/kg thrice weekly IV for 3 months | Coronary calcification | Treatment group had stable calcification while control group progressed | 16 |
| Cerium nitrate | 71 | Topical application to wound | Calciphylaxis | OR 0.44 (0.2–0.99) for mortality | 17 |
HD hemodialysis, IV intravenous, OR odds ratio, VC vascular calcification
Finally, we undertook a comprehensive search in the clinicaltrials.gov registry to look for any completed or ongoing clinical trials involving the use of these 36 antioxidative compounds for treating VC, up to 7 June 2019. Only two registered trials attempted to test these agents against the target phenotype, VC, including alendronate and statin, although the results have not been available. It is anticipated that more antioxidative compounds will gain momentum as anti-VC medications in the future.
Conclusion and future perspective
Natural compounds are rich sources of potential therapeutic candidates for various diseases, including neurodegenerative disorders, metabolic dysregulation, cardiovascular diseases85, and VC. Repurposing the existing medications for alternative applications is an important way of identifying novel therapies with known safety profiles86. For VC, the pivotal components in its pathogenesis are ROS production and inflammation87,88, which are targeted by many natural compounds and existing medications. Moreover, foods rich in these natural compounds may also be beneficial for patients with VC, although the ingestion amount needed to achieve the optimal result remains elusive. With the knowledge gained by the current literature, we expect that more antioxidants will emerge in the future as potential candidates in the therapeutic armamentarium against VC.
Acknowledgements
We are grateful to the staff of the second core laboratory of National Taiwan University Hospital and to Miss Ting-Yu Chen for her graphical assistance. The study is financially sponsored by Ministry of Science and Technology, Taiwan (MOST-108-2314-B-002-055-, MOST-108-2321-B-038-003-, 107-2314-B-002 -244 -MY3).
Consent for publication
Not applicable.
Data availability
Literature summary data will be available upon reasonable request.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethic approval and consent to participate
Not applicable.
Footnotes
Edited by I. D’agnano
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Cherepanov D, et al. Real-world cardiovascular disease burden in patients with atherosclerotic cardiovascular disease: a comprehensive systematic literature review. Curr. Med. Res. Opin. 2018;34:459–473. doi: 10.1080/03007995.2017.1401529. [DOI] [PubMed] [Google Scholar]
- 2.Roth GA, et al. Demographic and epidemiologic drivers of global cardiovascular mortality. N. Engl. J. Med. 2015;372:1333–1341. doi: 10.1056/NEJMoa1406656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Covic A, et al. Bone and mineral disorders in chronic kidney disease: implications for cardiovascular health and ageing in the general population. Lancet Diabetes Endocrinol. 2018;6:319–331. doi: 10.1016/S2213-8587(17)30310-8. [DOI] [PubMed] [Google Scholar]
- 4.Greif M, et al. Lipoprotein (a) is independently correlated with coronary artery calcification. Eur. J. Intern. Med. 2013;24:75–79. doi: 10.1016/j.ejim.2012.08.014. [DOI] [PubMed] [Google Scholar]
- 5.Malik S, et al. Coronary artery calcium score for long-term risk classification in individuals with type 2 diabetes and metabolic syndrome from the multi-ethnic study of atherosclerosis. JAMA Cardiol. 2017;2:1332–1340. doi: 10.1001/jamacardio.2017.4191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nitta K, Ogawa T, Hanafusa N, Tsuchiya K. Recent advances in the management of vascular calcification in patients with end-stage renal disease. Contrib. Nephrol. 2019;198:62–72. doi: 10.1159/000496532. [DOI] [PubMed] [Google Scholar]
- 7.Leopold JA. Vascular calcification: mechanisms of vascular smooth muscle cell calcification. Trends Cardiovasc. Med. 2015;25:264–274. doi: 10.1016/j.tcm.2014.10.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shanahan CM, Crouthamel MH, Kapustin A, Giachelli CM. Arterial calcification in chronic kidney disease: key roles for calcium and phosphate. Circ. Res. 2011;109:697–711. doi: 10.1161/CIRCRESAHA.110.234914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ho CY, Shanahan CM. Medial arterial calcification. Arterioscler. Thromb. Vasc. Biol. 2016;36:1475–1482. doi: 10.1161/ATVBAHA.116.306717. [DOI] [PubMed] [Google Scholar]
- 10.Byon CH, et al. Oxidative stress induces vascular calcification through modulation of the osteogenic transcription factor Runx2 by AKT signaling. J. Biol. Chem. 2008;283:15319–15327. doi: 10.1074/jbc.M800021200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Durik M, et al. Nucleotide excision DNA repair is associated with age-related vascular dysfunction. Circulation. 2012;126:468–478. doi: 10.1161/CIRCULATIONAHA.112.104380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Al-Aly Z. Phosphate, oxidative stress, and nuclear factor-κB activation in vascular calcification. Kidney Int. 2011;79:1044–1047. doi: 10.1038/ki.2010.548. [DOI] [PubMed] [Google Scholar]
- 13.Mathews SJ, et al. Effects of sodium thiosulfate on vascular calcification in end-stage renal disease: a pilot study of feasibility, safety and efficacy. Am. J. Nephrol. 2011;33:131–138. doi: 10.1159/000323550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yonova DH, et al. First impressions of cardiovascular calcification treatment in hemodialysis patients with a new dialysis fluid containing sodium thiosulphate (STS) Int. J. Artif. Organs. 2014;37:308–314. doi: 10.5301/ijao.5000309. [DOI] [PubMed] [Google Scholar]
- 15.Ghiandai G, Ralli C, Imperiali P, Zingarelli A, Duranti E. Is the sodium thiosulfate therapy useful for vascular calcification in dialysis Pts? G. Ital. Nefrol. 2015;32:gin/32.3.6. [PubMed] [Google Scholar]
- 16.Yu Y, Bi ZM, Wang Y, Chen ZQ, Xu SW. Effect of sodium thiosulfate on coronary artery calcification in maintenance hemodialysis patients. Zhonghua Yi Xue Za Zhi. 2016;96:3724–3748. doi: 10.3760/cma.j.issn.0376-2491.2016.46.007. [DOI] [PubMed] [Google Scholar]
- 17.Darres A, et al. The effectiveness of topical cerium nitrate-silver sulfadiazine application on overall outcome in patients with calciphylaxis. Dermatology. 2019;235:120–129. doi: 10.1159/000493975. [DOI] [PubMed] [Google Scholar]
- 18.Brodeur MR, et al. Reduction of advanced-glycation end products levels and inhibition of RAGE signaling decreases rat vascular calcification induced by diabetes. PLoS ONE. 2014;9:e85922. doi: 10.1371/journal.pone.0085922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Feng W, et al. Apocynin attenuates angiotensin II-induced vascular smooth muscle cells osteogenic switching via suppressing extracellular signal-regulated kinase 1/2. Oncotarget. 2016;7:83588–83600. doi: 10.18632/oncotarget.13193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Manivannan J, et al. Diosgenin attenuates vascular calcification in chronic renal failure rats. Mol. Cell. Biochem. 2013;378:9–18. doi: 10.1007/s11010-013-1588-8. [DOI] [PubMed] [Google Scholar]
- 21.Manivannan J, Shanthakumar J, Arunagiri P, Raja B, Balamurugan E. Diosgenin interferes coronary vasoconstriction and inhibits osteochondrogenic transdifferentiation of aortic VSMC in CRF rats. Biochimie. 2014;102:183–187. doi: 10.1016/j.biochi.2014.03.011. [DOI] [PubMed] [Google Scholar]
- 22.Liberman M, et al. Oxidant generation predominates around calcifying foci and enhances progression of aortic valve calcification. Arterioscler. Thromb. Vasc. Biol. 2008;28:463–470. doi: 10.1161/ATVBAHA.107.156745. [DOI] [PubMed] [Google Scholar]
- 23.Kim H, et al. α-Lipoic acid attenuates vascular calcification via reversal of mitochondrial function and restoration of Gas6/Axl/Akt survival pathway. J. Cell. Mol. Med. 2012;16:273–286. doi: 10.1111/j.1582-4934.2011.01294.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Bassi E, Liberman M, Martinatti MK, Bortolotto LA, Laurindo FRM. Lipoic acid, but not tempol, preserves vascular compliance and decreases medial calcification in a model of elastocalcinosis. Braz. J. Med. Biol. Res. 2014;47:119–127. doi: 10.1590/1414-431X20133193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lu T, et al. Induction of intracellular heat-shock protein 72 prevents the development of vascular smooth muscle cell calcification. Cardiovasc. Res. 2012;96:524–532. doi: 10.1093/cvr/cvs278. [DOI] [PubMed] [Google Scholar]
- 26.Beazley KE, et al. Transglutaminase inhibitors attenuate vascular calcification in a preclinical model. Arterioscler. Thromb. Vasc. Biol. 2013;33:43–51. doi: 10.1161/ATVBAHA.112.300260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Beazley KE, Eghtesad S, Nurminskaya MV. Quercetin attenuates warfarin-induced vascular calcification in vitro independently from matrix Gla protein. J. Biol. Chem. 2013;288:2632–2640. doi: 10.1074/jbc.M112.368639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Beazley KE, Lima F, Borras T, Nurminskaya M. Attenuation of chondrogenic transformation in vascular smooth muscle by dietary quercetin in the MGP-deficient mouse model. PLoS ONE. 2013;8:e76210. doi: 10.1371/journal.pone.0076210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chang X, et al. Quercetin attenuates vascular calcification through suppressed oxidative stress in adenine-induced chronic renal failure rats. Biomed. Res. Int. 2017;2017:5716204. doi: 10.1155/2017/5716204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Cui L, Li Z, Chang X, Cong G, Hao L. Quercetin attenuates vascular calcification by inhibiting oxidative stress and mitochondrial fission. Vasc. Pharmacol. 2017;88:21–29. doi: 10.1016/j.vph.2016.11.006. [DOI] [PubMed] [Google Scholar]
- 31.Liang Q, Chen Y, Li C, Lu L. Quercetin attenuates Ox-LDL-induced calcification in vascular smooth muscle cells by regulating ROS-TLR4 signaling pathway. Nan Fang. Yi Ke Da Xue Xue Bao. 2018;38:980–985. doi: 10.3969/j.issn.1673-4254.2018.08.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ivanovski O, et al. Effect of simvastatin in apolipoprotein E deficient mice with surgically induced chronic renal failure. J. Urol. 2008;179:1631–1636. doi: 10.1016/j.juro.2007.11.042. [DOI] [PubMed] [Google Scholar]
- 33.Afonso P, et al. LMNA mutations resulting in lipodystrophy and HIV protease inhibitors trigger vascular smooth muscle cell senescence and calcification: role of ZMPSTE24 downregulation. Atherosclerosis. 2016;245:200–211. doi: 10.1016/j.atherosclerosis.2015.12.012. [DOI] [PubMed] [Google Scholar]
- 34.Mody N, Parhami F, Sarafian TA, Demer LL. Oxidative stress modulates osteoblastic differentiation of vascular and bone cells. Free Radic. Biol. Med. 2001;31:509–519. doi: 10.1016/S0891-5849(01)00610-4. [DOI] [PubMed] [Google Scholar]
- 35.Peralta-Ramírez A, et al. Vitamin E protection of obesity-enhanced vascular calcification in uremic rats. Am. J. Physiol. Ren. Physiol. 2013;306:F422–F429. doi: 10.1152/ajprenal.00355.2013. [DOI] [PubMed] [Google Scholar]
- 36.Pasch A, et al. Sodium thiosulfate prevents vascular calcifications in uremic rats. Kidney Int. 2008;74:1444–1453. doi: 10.1038/ki.2008.455. [DOI] [PubMed] [Google Scholar]
- 37.Liu H, Lu Q, Huang K. Selenium suppressed hydrogen peroxide-induced vascular smooth muscle cells calcification through inhibiting oxidative stress and ERK activation. J. Cell. Biochem. 2010;111:1556–1564. doi: 10.1002/jcb.22887. [DOI] [PubMed] [Google Scholar]
- 38.Roman-Garcia P, Barrio-Vazquez S, Fernandez-Martin JL, Ruiz-Torres MP, Cannata-Andia JB. Natural antioxidants and vascular calcification: a possible benefit. J. Nephrol. 2011;24:669–672. doi: 10.5301/jn.5000029. [DOI] [PubMed] [Google Scholar]
- 39.Derwall M, et al. Inhibition of bone morphogenetic protein signaling reduces vascular calcification and atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2012;32:613–622. doi: 10.1161/ATVBAHA.111.242594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yamada S, et al. The antioxidant tempol ameliorates arterial medial calcification in uremic rats: Important role of oxidative stress in the pathogenesis of vascular calcification in chronic kidney disease. J. Bone Miner. Res. 2012;27:474–485. doi: 10.1002/jbmr.539. [DOI] [PubMed] [Google Scholar]
- 41.Nikolov IG, et al. Farnesyltransferase inhibitor R115777 protects against vascular disease in uremic mice. Atherosclerosis. 2013;229:42–51. doi: 10.1016/j.atherosclerosis.2013.02.041. [DOI] [PubMed] [Google Scholar]
- 42.Cao X, et al. Metformin inhibits vascular calcification in female rat aortic smooth muscle cells via the AMPK-eNOS-NO pathway. Endocrinology. 2013;154:3680–3689. doi: 10.1210/en.2013-1002. [DOI] [PubMed] [Google Scholar]
- 43.Zhong H, Liu F, Dai X, Zhou L, Fu P. Sodium thiosulfate protects human aortic smooth muscle cells from osteoblastic transdifferentiation via high-level phosphate. Kaohsiung J. Med. Sci. 2013;29:587–593. doi: 10.1016/j.kjms.2013.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Chen NX, O’Neill K, Akl NK, Moe SM. Adipocyte induced arterial calcification is prevented with sodium thiosulfate. Biochem. Biophys. Res. Commun. 2014;449:151–156. doi: 10.1016/j.bbrc.2014.05.005. [DOI] [PubMed] [Google Scholar]
- 45.Liu, H., Li, X., Qin, F. & Huang, K. Selenium suppresses oxidative-stress-enhanced vascular smooth muscle cell calcification by inhibiting the activation of the PI3K/AKT and ERK signaling pathways and endoplasmic reticulum stress. J. Biol. Inorg. Chem. 19, 375–388 (2014). [DOI] [PubMed]
- 46.Freise C, Querfeld U. Inhibition of vascular calcification by block of intermediate conductance calcium-activated potassium channels with TRAM-34. Pharmacol. Res. 2014;85:6–14. doi: 10.1016/j.phrs.2014.04.013. [DOI] [PubMed] [Google Scholar]
- 47.Ha C-M, et al. Activation of Nrf2 by dimethyl fumarate improves vascular calcification. Vasc. Pharmacol. 2014;63:29–36. doi: 10.1016/j.vph.2014.06.007. [DOI] [PubMed] [Google Scholar]
- 48.Kee HJ, et al. Gallic acid inhibits vascular calcification through the blockade of BMP2–Smad1/5/8 signaling pathway. Vasc. Pharmacol. 2014;63:71–78. doi: 10.1016/j.vph.2014.08.005. [DOI] [PubMed] [Google Scholar]
- 49.Cutini PH, Rauschemberger MB, Sandoval MJ, Massheimer VL. Vascular action of bisphosphonates: in vitro effect of alendronate on the regulation of cellular events involved in vessel pathogenesis. J. Mol. Cell. Cardiol. 2016;100:83–92. doi: 10.1016/j.yjmcc.2016.08.017. [DOI] [PubMed] [Google Scholar]
- 50.Li E-G, Tian J, Xu Z-H. Effects of Gingko biloba extract (EGb 761) on vascular smooth muscle cell calcification induced by β-glycerophosphate. Ren. Fail. 2016;38:552–557. doi: 10.3109/0886022X.2016.1148724. [DOI] [PubMed] [Google Scholar]
- 51.Zhang W, Li Y, Ding H, Du Y, Wang L. Hydrogen peroxide prevents vascular calcification induced ROS production by regulating Nrf-2 pathway. Ren. Fail. 2016;38:1099–1106. doi: 10.1080/0886022X.2016.1194143. [DOI] [PubMed] [Google Scholar]
- 52.Zhang P, et al. Resveratrol ameliorated vascular calcification by regulating Sirt-1 and Nrf2. Transplant. Proc. 2016;48:3378–3386. doi: 10.1016/j.transproceed.2016.10.023. [DOI] [PubMed] [Google Scholar]
- 53.Aghagolzadeh P, et al. Hydrogen sulfide attenuates calcification of vascular smooth muscle cells via KEAP1/NRF2/NQO1 activation. Atherosclerosis. 2017;265:78–86. doi: 10.1016/j.atherosclerosis.2017.08.012. [DOI] [PubMed] [Google Scholar]
- 54.Jiang B, et al. Vascular scaffolds with enhanced antioxidant activity inhibit graft calcification. Biomaterials. 2017;144:166–175. doi: 10.1016/j.biomaterials.2017.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rios R, et al. Vitamin E protects against extraskeletal calcification in uremic rats fed high fat diets. BMC Nephrol. 2017;18:374. doi: 10.1186/s12882-017-0790-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jordão JB, Porto HK, Lopes FM, Batista AC, Rocha ML. Protective effects of ellagic acid on cardiovascular injuries caused by hypertension in rats. Planta Med. 2017;83:830–836. doi: 10.1055/s-0043-103281. [DOI] [PubMed] [Google Scholar]
- 57.Choi S-Y, et al. Dipeptidyl peptidase-4 inhibitor gemigliptin protects against vascular calcification in an experimental chronic kidney disease and vascular smooth muscle cells. PLoS ONE. 2017;12:e0180393. doi: 10.1371/journal.pone.0180393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Shen J, et al. Regulation of vascular calcification by growth hormone–releasing hormone and its agonists. Circ. Res. 2018;122:1395–1408. doi: 10.1161/CIRCRESAHA.117.312418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Salimi F, Jafari-Nodooshan S, Zohourian N, Kolivand S, Hamedi J. Simultaneous anti-diabetic and anti-vascular calcification activity of Nocardia sp. UTMC 751. Lett. Appl. Microbiol. 2018;66:110–117. doi: 10.1111/lam.12833. [DOI] [PubMed] [Google Scholar]
- 60.Liao L, et al. Polysaccharide from Fuzi protects against Ox‑LDL‑induced calcification of human vascular smooth muscle cells by increasing autophagic activity. Mol. Med. Rep. 2018;17:5109–5115. doi: 10.3892/mmr.2018.8488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kaur T, Kaul S, Bhardwaj A. Efficacy of linalool to ameliorate uremia induced vascular calcification in wistar rats. Phytomedicine. 2018;51:191–195. doi: 10.1016/j.phymed.2018.10.007. [DOI] [PubMed] [Google Scholar]
- 62.Liu H, et al. Puerarin inhibits vascular calcification of uremic rats. Eur. J. Pharmacol. 2019;855:235–243. doi: 10.1016/j.ejphar.2019.05.023. [DOI] [PubMed] [Google Scholar]
- 63.Ji R, et al. Rosmarinic acid exerts an antagonistic effect on vascular calcification by regulating the Nrf2 signalling pathway. Free Radic. Res. 2019;53:187–197. doi: 10.1080/10715762.2018.1558447. [DOI] [PubMed] [Google Scholar]
- 64.Elseweidy MM, et al. Potential therapeutic roles of 10-dehydrogingerdione and/or pentoxifylline against calcium deposition in aortic tissues of high dietary cholesterol-fed rabbits. Mol. Cell. Biochem. 2019;453:131–142. doi: 10.1007/s11010-018-3438-1. [DOI] [PubMed] [Google Scholar]
- 65.Simonyi A, et al. The neuroprotective effects of apocynin. Front. Biosci. 2012;4:2183–2193. doi: 10.2741/e535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Stefanska J, Pawliczak R. Apocynin: molecular aptitudes. Mediators Inflamm. 2008;2008:106507. doi: 10.1155/2008/106507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Sethi G, et al. Pro-apoptotic and anti-cancer properties of diosgenin: a comprehensive and critical review. Nutrients. 2018;10:645. doi: 10.3390/nu10050645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Andres S, et al. Safety aspects of the use of quercetin as a dietary supplement. Mol. Nutr. Food Res. 2018;62:1700447. doi: 10.1002/mnfr.201700447. [DOI] [PubMed] [Google Scholar]
- 69.Choi S-Y, Park G-S, Lee S-Y, Kim J-Y, Kim Y-K. The conformation and CETP inhibitory activity of [10]-dehydrogingerdione isolated from Zingiber officinale. Arch. Pharm. Res. 2011;34:727–731. doi: 10.1007/s12272-011-0505-5. [DOI] [PubMed] [Google Scholar]
- 70.Elseweidy MM, Abdallah FR, Younis NN, Aldohmy S, Kassem HM. 10-Dehydrogingerdione raises HDL-cholesterol through a CETP inhibition and wards off oxidation and inflammation in dyslipidemic rabbits. Atherosclerosis. 2013;231:334–340. doi: 10.1016/j.atherosclerosis.2013.09.024. [DOI] [PubMed] [Google Scholar]
- 71.Mirzaei H, et al. Phytosomal curcumin: a review of pharmacokinetic, experimental and clinical studies. Biomed. Pharmacother. 2017;85:102–112. doi: 10.1016/j.biopha.2016.11.098. [DOI] [PubMed] [Google Scholar]
- 72.Federico A, Dallio M, Loguercio C. Silymarin/silybin and chronic liver disease: a marriage of many years. Molecules. 2017;22:191. doi: 10.3390/molecules22020191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ceci C, et al. Experimental evidence of the antitumor, antimetastatic and antiangiogenic activity of ellagic acid. Nutrients. 2018;10:1756. doi: 10.3390/nu10111756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Pietri S, Maurelli E, Drieu K, Culcasi M. Cardioprotective and anti-oxidant effects of the terpenoid constituents of ginkgo biloba extract (EGb 761) J. Mol. Cell. Cardiol. 1997;29:733–742. doi: 10.1006/jmcc.1996.0316. [DOI] [PubMed] [Google Scholar]
- 75.Liu B, Tan Y, Wang D, Liu M. Puerarin for acute ischaemic stroke. Cochrane Database Syst. Rev. 2016;2:CD004955. doi: 10.1002/14651858.CD004955.pub2. [DOI] [PubMed] [Google Scholar]
- 76.Baur JA, Sinclair DA. Therapeutic potential of resveratrol: the in vivo evidence. Nat. Rev. Drug Discov. 2006;5:493–506. doi: 10.1038/nrd2060. [DOI] [PubMed] [Google Scholar]
- 77.Lapczynski A, Letizia CS, Api AM. Fragrance material review on d-linalool. Food Chem. Toxicol. 2008;46:S193–S194. doi: 10.1016/j.fct.2008.06.044. [DOI] [PubMed] [Google Scholar]
- 78.Elufioye TO, Habtemariam S. Hepatoprotective effects of rosmarinic acid: Insight into its mechanisms of action. Biomed. Pharmacother. 2019;112:108600. doi: 10.1016/j.biopha.2019.108600. [DOI] [PubMed] [Google Scholar]
- 79.Pouyatos B, Gearhart C, Nelson-Miller A, Fulton S, Fechter L. Oxidative stress pathways in the potentiation of noise-induced hearing loss by acrylonitrile. Hear. Res. 2007;224:61–74. doi: 10.1016/j.heares.2006.11.009. [DOI] [PubMed] [Google Scholar]
- 80.Rushworth GF, Megson IL. Existing and potential therapeutic uses for N-acetylcysteine: the need for conversion to intracellular glutathione for antioxidant benefits. Pharmacol. Ther. 2014;141:150–159. doi: 10.1016/j.pharmthera.2013.09.006. [DOI] [PubMed] [Google Scholar]
- 81.Manzanares W, et al. High-dose selenium reduces ventilator-associated pneumonia and illness severity in critically ill patients with systemic inflammation. Intensive Care Med. 2011;37:1120–1127. doi: 10.1007/s00134-011-2212-6. [DOI] [PubMed] [Google Scholar]
- 82.Aroor AR, Sowers JR, Jia G, DeMarco VG. Pleiotropic effects of the dipeptidylpeptidase-4 inhibitors on the cardiovascular system. Am. J. Physiol. Heart Circ. Physiol. 2014;307:H477–H492. doi: 10.1152/ajpheart.00209.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Zhu B-Z, Carr AC, Frei B. Pyrrolidine dithiocarbamate is a potent antioxidant against hypochlorous acid-induced protein damage. FEBS Lett. 2002;532:80–84. doi: 10.1016/S0014-5793(02)03637-2. [DOI] [PubMed] [Google Scholar]
- 84.van Lith R, Gregory EK, Yang J, Kibbe MR, Ameer GA. Engineering biodegradable polyester elastomers with antioxidant properties to attenuate oxidative stress in tissues. Biomaterials. 2014;35:8113–8122. doi: 10.1016/j.biomaterials.2014.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kunal S, Jawed AS, Gaurav S, Rakesh M, Naibedya C. Role of phytochemicals in the prevention of menopausal bone loss: evidence from in vitro and in vivo, human interventional and pharmacokinetic studies. Curr. Med. Chem. 2009;16:1138–1157. doi: 10.2174/092986709787581806. [DOI] [PubMed] [Google Scholar]
- 86.Cha Y, et al. Drug repurposing from the perspective of pharmaceutical companies. Br. J. Pharmacol. 2018;175:168–180. doi: 10.1111/bph.13798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Chao C-T, et al. Circulating microRNA-125b predicts the presence and progression of uremic vascular calcification. Arterioscler. Thromb. Vasc. Biol. 2017;37:1402–1414. doi: 10.1161/ATVBAHA.117.309566. [DOI] [PubMed] [Google Scholar]
- 88.Chao C-T, Yeh H-Y, Yuan T-H, Chiang C-K, Chen H-W. MicroRNA-125b in vascular diseases: an updated systematic review of pathogenetic implications and clinical applications. J. Cell. Mol. Med. 2019;23:5884–5894. doi: 10.1111/jcmm.14535. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Literature summary data will be available upon reasonable request.