Abstract
Background
Disease relapse is the primary cause of death from ovarian carcinoma. Isolated lymph node relapse is a rare pattern of ovarian carcinoma recurrence, with a reported median postrelapse survival of 2.5 to 4 years. To date, investigations have not compared isolated lymph node relapse ovarian carcinoma directly to a matched extranodal relapse cohort or performed molecular characterization of cases that subsequently experience isolated lymph node relapse.
Objective
Here we seek to compare the clinical outcome, tumor-infiltrating lymphocyte burden, and frequency of known prognostic genomic events in isolated lymph node relapse ovarian carcinoma vs extranodal relapse ovarian carcinoma.
Study Design
Forty-nine isolated lymph node relapse ovarian carcinoma patients were identified and matched to 49 extranodal relapse cases using the Edinburgh Ovarian Cancer Database, from which the clinical data for identified patients were retrieved. Matching criteria were disease stage, histologic subtype and grade, extent of residual disease following surgical debulking, and age at diagnosis. Clinicopathologic factors and survival data were compared between the isolated lymph node relapse and extranodal relapse cohorts. Genomic characterization of tumor material from diagnosis was performed using panel-based high-throughput sequencing and tumor-infiltrating T cell burden was assessed using immunohistochemistry for CD3+ and CD8+ cells.
Results
Isolated lymph node relapse cases demonstrated significantly prolonged postrelapse survival and overall survival vs extranodal relapse upon multivariable analysis (HRmulti = 0.52 [0.33–0.84] and 0.51 [0.31–0.84]). Diagnostic specimens from high-grade serous ovarian carcinomas that subsequently displayed isolated lymph node relapse harbored significantly greater CD3+ and CD8+ cell infiltration compared to extranodal relapse cases (P = .001 and P = .009, Bonferroni-adjusted P = .003 and P = .019). Isolated lymph node relapse high-grade serous ovarian carcinoma cases did not show marked enrichment or depletion of cases with BRCA1/2 mutation or CCNE1 copy number gain when compared to their extranodal relapse counterparts (24.4% vs 19.4% and 18.2% vs 22.6%, P = .865 and P = .900).
Conclusion
Isolated lymph node relapse ovarian carcinoma represents a distinct clinical entity with favorable outcome compared to extranodal relapse. There was no clear enrichment or depletion of BRCA1/2 mutation or CCNE1 gain in the isolated lymph node relapse ovarian carcinoma cohort compared with extranodal relapse cases, suggesting that these known prognostic genomically defined subtypes of disease do not display markedly altered propensity for isolated lymph node relapse. Diagnostic tumor material from isolated lymph node relapse patients demonstrated greater CD3+ and CD8+ cell infiltration, indicating stronger tumor engagement by T cell populations, which may contribute to the more indolent disease course of isolated lymph node relapse.
Key words: Cancer recurrence, isolated lymph node relapse, ovarian cancer, survival, tumor-infiltrating lymphocytes
Ovarian carcinoma (OC) is the most lethal gynecologic malignancy, accounting for over 180,000 deaths per year worldwide.1 OC is now recognized to comprise 5 core histologic subtypes: high-grade serous (HGS), endometrioid, clear cell, low-grade serous, and mucinous OC—each displaying distinct molecular landscapes and clinical behavior.2 Within HGS cases, homologous recombination deficiency by virtue of BRCA1 or BRCA2 mutation has been associated with favorable outcome, greater sensitivity to platinum-based chemotherapy, and marked benefit from poly (ADP-ribose) polymerase inhibitors.3, 4, 5, 6 Conversely, CCNE1 copy number gain has been associated with chemoresistance and poorer survival in this group.3, 7
AJOG at a Glance.
Why was this study conducted?
A number of investigators have reported a relatively indolent disease course in ovarian carcinoma patients experiencing isolated lymph node relapse. However, none have systematically compared these to extranodal relapse or performed molecular characterization of patients who go on to experience this distinct pattern of recurrence.
Key findings
Isolated lymph node relapse patients demonstrated significantly prolonged overall and postrelapse survival compared with extranodal relapse cases. Isolated lymph node relapse cases demonstrated greater tumor-infiltrating lymphocyte burden at diagnosis, but did not demonstrate significant enrichment or depletion of BRCA1/2 mutation or gain of CCNE1, both known to be prognostic in ovarian carcinoma.
What does this add to what is known?
This is the first report demonstrating significantly improved clinical outcome in isolated lymph node relapse ovarian carcinoma when compared directly with extranodal relapse, and represents the first study to perform molecular characterization of patients who go on to experience isolated lymph node relapse.
Though patients in most OC cases—particularly HGS OCs—are typically sensitive to chemotherapy in the first-line setting, the majority of patients will experience disease relapse, which acquires resistance to chemotherapy.8, 9 The most common sites of recurrence are the pelvis and peritoneum.10 Involvement of lymph nodes (LNs) at relapse is common; however, recurrence confined solely to LNs is a rare event, accounting for ≤5% of relapsed OCs.11, 12 These isolated LN relapse (ILNR) cases have been described as a unique clinical disease entity and are thought to experience a relatively indolent disease course, with a reported median postrelapse survival (PRS) and overall survival (OS) of around 2.5 to 4 years and >5 years, respectively.11, 12, 13, 14, 15, 16, 17, 18
A number of previous studies have reported on the clinical outcome of apparent ILNR OC (summarized in Table 1).11, 12, 13, 14, 15, 16, 17, 18 Many of these studies have reported only a small number of cases,11, 13, 17, 18 with a minority reporting larger numbers identified from multiple centers.14, 16 To our knowledge, none of these studies have compared outcome directly to a matched extranodal relapse (ENR) cohort. Furthermore, they have not performed contemporary histologic subtyping or molecular characterization so as to identify potential subgroups of disease with a propensity to experience this distinct pattern of disease relapse.
Table 1.
Previous reports of isolated lymph node relapse ovarian carcinoma
| ILNR cases | N | Ferrero16 |
Tu15 |
Gadducci14 |
Fotiou18 |
Legge12 |
Santillan17 |
Blanchard11 |
Uzan13 |
Summary |
|---|---|---|---|---|---|---|---|---|---|---|
| 73 | 38 | 69 | 21 | 32 | 25 | 27 | 12 | Few reports of ≥40 cases | ||
| Age at diagnosis | Years | Median 54 | 24 (63%) >50; 14 (37%) ≤50 | Median 58 | Mean 50 | Median 60 | Mean 58 | Mean 59 | Median 51 | Largely unremarkable compared with unselected OC cohorts |
| Range | 29–73 | 34–78 | 36–67 | 45–76 | 41–82 | 41-85 | 42–71 | |||
| Stage at diagnosis | I | 14 (19%) | 0 | 11 (16%) | 3 (14%) | 0 | 2 (8%) | 4 (15%) | 5 (42%) | |
| II | 4 (6%) | 15 (39%) | 6 (9%) | 3 (14%) | 1 (3%) | 5 (20%) | 5 (18%) | 1 (8%) | ||
| III | 51 (70%) | 23 (61%) | 46 (67%) | 14 (67%) | 29 (91%) | 15 (60%) | 15 (56%) | 6 (50%) | ||
| IV | 4 (6%) | 0 | 6 (9%) | 1 (5%) | 2 (6%) | 3 (12%) | 3 (11%) | 0 | ||
| RD following first-line debulking | 0 cm / <0.5 cm | 57 (78%) | 17 (45%) | 22 (32%) | 8 (38%) | 14 (44%) | 18 (72%) | NA | 7 (58%) | |
| ≤1 cm | 10 (14%) | 10 (26%) | 11 (16%) | 7 (33%) | 6 (19%) | 5 (20%) | NA | 4 (33%) | ||
| <2 cm | 4 (6%) | 11 (29%) | 36 (52%) | 4 (19%) | 0 | NA | ||||
| >2 cm | 2 (3%) | 2 (10%) | 12 (38%) | 2 (8%) | NA | 1 (8%) | ||||
| Grade at diagnosis | I | 4 (6%) | 7 (18%) | 3 (4%) | 0 | 9 (32%) | 25 (100%) high grade | NA | NA | |
| II | 5 (7%) | 14 (37%) | 13 (19%) | 8 (38%) | ||||||
| III | 64 (88%) | 17 (45%) | 54 (78%) | 13 (62%) | 19 (68%) | |||||
| NA | - | - | - | - | 4 | - | ||||
| Reported histologic subtype at diagnosis | Serous | 53 (73%) | 19 (50%) | 52 (75%) | 16 (76%) | 26 (81%) | 19 (76%) | 17 (62%)a | 8 (67%) | Predominantly serous / HGS cases, as with unselected OC cohorts |
| Endometrioid | 11 (15%) | 9 (24%) | 12 (17%) | 5 (24%) | 2 (6%) | 2 (8%) | 3 (11%) | 3 (25%) | ||
| Clear cell | 0 | 0 | 1 (1%) | 0 | 0 | 0 | 0 | |||
| Mucinous | 1 (1%) | 1 (3%) | 0 | 1 (3%) | 0 | 3 (11%) | 0 | |||
| Other | 8 (11%) | 9 (24%) | 4 (6%) | 3 (9%) | 4 (16%) | 4 (15%) | 1 (8%) | |||
| DFI / time to ILNRb | Median months | 18 | 18 | 44 (62%) >12 months |
21 | 17.5 | 16 | 26 months from diagnosis | 21 | Median 1.5–2 years DFI |
| Range | 6–192 | 9-96 | 8–156 | 1–134 | 6–40 | 1–159 | 6–72 | |||
| ILNR site(s) | Para-aortic only | 37 (51%) | 10 (26%) | 23 (33%) | 8 (38%) | 14 (44%) | 15 (60%) | 9 (33%) retro. alone, 6(22%) retro. + other. supraclavicular, mediastinal, iliac, and inguinal involvement in 7 (26%), 4 (15%), 4 (15%), and 3 (11%) cases | 5 (42%) | Most commonly involves pelvic and/or para-aortic sites |
| Pelvic only | 21 (29%) | 15 (39%) | 12 (17%) | 4 (19%) | 1 (3%) | 3 (12%) | 4 (33%) | |||
| Para-aortic & pelvic | 9 (12%) | 7 (18%) | 6 (9%) | 4 (19%) | 9 (28%) | 1 (4%) | 1 (8%) | |||
| Inguinal only | 3 (4%) | 2 (5%) | 12 (17%) | 4 (19%) | 2 (6%) | 5 (20%) | 1 (8%) | |||
| Other combinations | 3 (4%) | 4 (11%) | 16 (23%) | 1 (5%) | 6 (19%) | 1 (4%) | 1 (8%) | |||
| ILNR pattern | Single region | 61 (84%) | 27 (71%) | 47 (77%) | 17 (81%) | 20 (63%) | 24 (96%) | 17 (63%) | 10 (83%) | Most commonly localized to a single region |
| Multiregion | 12 (16%) | 11 (29%) | 14 (23%) | 4 (19%) | 9 (28%) | 1 (4%) | 10 (37%) | 2 (17%) | ||
| NA | - | - | 8 | - | 3 | - | - | - | ||
| PRS | Median months | 5-y PRS 64%; 5-y OS ∼80% | 5-y PRS 66.5% | 32.1 | 47 | 37 | 37 | 26 | 5-y PRS 71% | Median 2–4 years |
| OS | Median months | 62.9 | 66 | 109 | 61 | 68 | Median >5 years | |||
| Surgery for ILNR | Yes | 73 (100%) | 19 (50%) | 24 (35%) | 21 (100%) | 12 (38%) | 25 (100%) | 8 (30%) | 12 (100%) | Heterogeneous management, typically involving chemotherapy |
| No | 0 | 19 (50%) | 45 (65%) | 0 | 20 (63%) | 0 | 19 (70%) | 0 | ||
| ILNR intervention: regime | Chemo alone | 0 | 5 (13%) | 44 (64%) | 0 | 19 (59%) | 0 | 8 (30%) | 0 | |
| Surgery alone | 3 (4%) | 0 | 1 (1%) | 0 | 1 (3%) | 2 (8%) | 2 (7%) | 0 | ||
| Surgery-chemo combination | 70 (96%) | 19 (50%) | 22 (32%) | 17 (81%) | 11 (34%) | 15 (60%) | 5 (19%) | 10 (83%) | ||
| Radio alone | 0 | 0 | 1 (1%) | 0 | 0 | 0 | 2 (7%) | 0 | ||
| No intervention | 0 | 0 | 0 | 0 | 1 (3%) | 0 | 7 (26%) | 0 | ||
| Other | 0 | 14 (37%) | 1 (1%) | 4 (19%) | 0 | 8 (32%) | 3 (11%) | 2 (17%) |
Chemo, chemotherapy; DFI, disease-free interval; ENR, extranodal relapse; ILNR, isolated lymph node relapse; NA, not available; OS, overall survival; PRS, postrelapse survival; radio, radiotherapy; RD, residual disease; retro, retroperitoneal.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Includes 5 cases described as papillary
From end of first-line chemotherapy.
Here, we report clinical and molecular characterization of a matched ILNR and ENR cohort with contemporary pathology review to compare the clinical outcome and molecular landscape of ILNR and ENR OC.
Materials and Methods
Isolated lymph node relapse patient identification
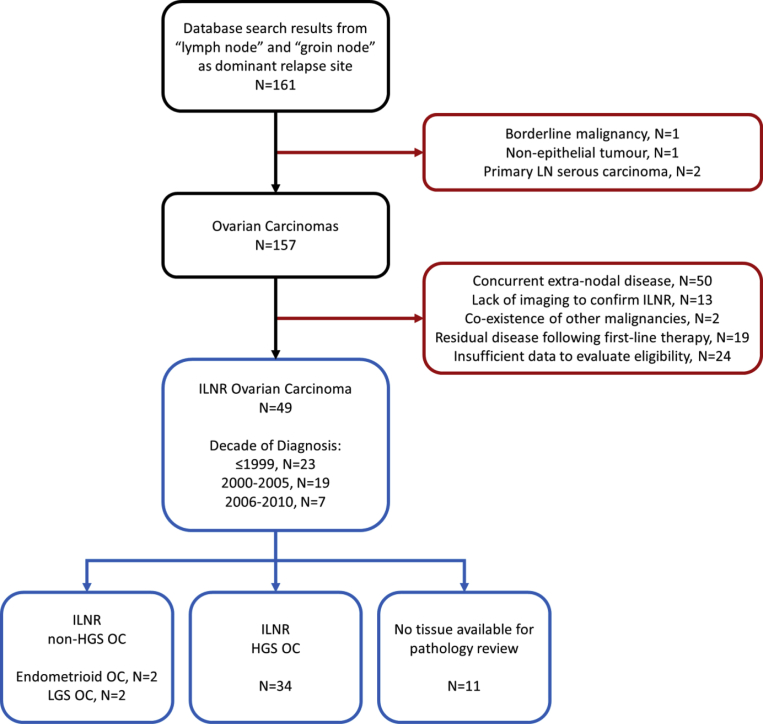
ILNR OC cases were identified from the Edinburgh Ovarian Cancer Database (Appendix: Supplementary Figure S1), wherein the clinical variables, treatment details, and follow-up data of OC patients treated within the Edinburgh Cancer Centre are collected prospectively as part of routine care. Potential ILNR cases were identified using the search terms “lymph node” or “groin node” as the dominant site of relapse, yielding 161 results. Nonepithelial tumors (n = 1), tumors of borderline malignancy (n = 1), and primary LN serous carcinomas (n = 2) were excluded. Patients with concurrent extranodal disease (n = 50), lack of cross-sectional imaging to confirm sole ILNR (n = 13), or coexistence of other malignancies leading to uncertain origin of LN disease (n = 2) were excluded. Patients with residual disease (RD) after completion of first-line treatment (n = 19) or insufficient clinical data for eligibility assessment (n = 24) were also excluded, leaving 49 ILNR cases.
Supplementary Figure S1.
Isolated lymph node relapse cohort identification from the Edinburgh Ovarian Cancer Database
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Matching of isolated lymph node relapse to extranodal relapse
ILNR cases were electronically matched to ENR cases with complete response to first-line therapy using the Edinburgh Ovarian Cancer Database. Matching criteria were as follows: (1) diagnostic histologic subtype and grade, (2) stage at diagnosis, (3) extent of RD following debulking surgery, and (4) closest age at diagnosis following matching of criteria 1–3. Criteria were relaxed to facilitate matching of all ILNR cases as detailed in Supplementary Table S1 (Appendix).
Ethical approval and tissue collection
Clinical research access and ethical approval for correlation of molecular data to clinicopathologic features and clinical outcome in OC was obtained via NHS Lothian Research and Development (reference ID 2007/W/ON/29). Ethical approval for the use of human tumor material in translational research was obtained from South East Scotland Human Annotated Bioresource (Lothian NRS Bioresource Ethics Reference 15/ES/0094-SR831). Tumor material was available for 75.5% (74 of 98) of cases (77.6%, 38 of 49 ILNR and 73.5%, 36 of 49 ENR).
Histologic subtyping of ovarian carcinomas
Contemporary pathology review of ILNR and matched ENR cases was performed by an expert gynecologic pathologist (C.S.H.). Where appropriate (n = 9), immunohistochemistry (IHC) for WT1 and p53 was performed to aid histologic subtyping.19 WT1 IHC was performed using 1:1000 dilutions of antibody M3561 clone 6F-H2 (Dako, Agilent Technologies, Santa Clara, CA). p53 staining was performed using 1:50 dilutions of antibody M7001 clone DO-7 (Dako, Agilent Technologies). Both stains were performed using the Leica Bond III Autostainer (Leica Biosystems, Milton Keynes, UK).
Nucleic acid isolation
Up to 10 10-μm formalin-fixed paraffin-embedded sections, macrodissected using marked hematoxylin–eosin-stained slides as a guide to enrich for tumor purity (Appendix: Supplementary Table S2), were used for DNA extraction. DNA was extracted using the QIAamp DNA FFPE Tissue Kit and Deparaffinization Solution (Qiagen, Venlo, the Netherlands).
Panel-based sequencing of BRCA and non-BRCA homologous recombination deficiency genes
High-throughput sequencing was performed using an 83-gene custom Integrated DNA Technologies gene capture panel with unique molecular indices, as described in the Appendix. Gene targets, centered around the homologous recombination DNA repair pathway, are detailed in Supplementary Table S3 (Appendix). The median per-sample mean target coverage achieved was 386X.
Assessment of CCNE1 copy number
Copy number variants in CCNE1 were characterized by TaqMan Genotyping qPCR Copy Number Assays (Applied Biosystems, Thermo Fisher Scientific, Waltham, MA), as detailed in the Appendix.
Assessment of tumor-infiltrating lymphocyte density
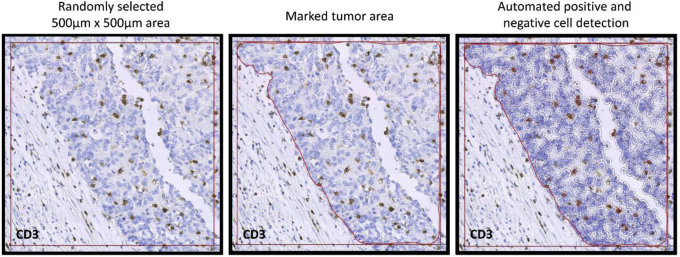
Tumor-infiltrating lymphocytes (TILs) were assessed using 4-μm FFPE sections of diagnostic tumor material from first-line cytoreductive surgery. IHC for CD3 and CD8 was performed using Bond ready-to-use CD8-4B11 and CD3-LN10 antibodies (Leica Biosystems) on the Leica Bond III Autostainer. Human tonsil was used as a positive control for both markers. Stained slides were digitized and marker-positive cells were quantified using QuPath20 in 8 randomly selected tumor-containing 500 × 500-μm fields per sample. Tumor area was marked as a region of interest (Appendix: Supplementary Figure S2) and marker-positive cells were quantified using the positive cell detection protocol as a percentage of the total cell number demonstrating marker positivity.
Supplementary Figure S2.
Automated marker-positive cell quantification by QuPath
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
TIL scoring validation was performed by manual counting of marker-positive cells by 2 human observers (R.L.H. and A.H.P.), in a randomly selected validation cohort representing 15% of samples for each marker. The correlation of marker-positive cell counts (observer 1 vs observer 2 vs QuPath) demonstrated excellent agreement for both markers (Spearman’s rho > 0.95, P < .0001 for all comparisons).
Statistical analyses
Statistical analyses were performed using R version 3.5.1 (R Foundation, Vienna, Austria). Disease-free interval (DFI) was calculated as time from end of first-line chemotherapy to disease recurrence. Comparisons of OS and PRS were conducted using Cox proportional hazards regression models within the Survival R package21 and presented as hazard ratios (HRs) alongside their 95% confidence intervals (CIs). Frequency comparisons were made using the χ2 test and Fisher exact test as appropriate. Comparisons of TIL density were made using the Mann–Whitney U test. Analyses were adjusted for multiplicity of testing using the Bonferroni correction, where specified.
Results
Cohort characteristics
Demographics of the ILNR and ENR cohorts are summarized in Table 2. There was no significant difference in age at diagnosis, RD following primary surgical debulking, histology or grade of disease at diagnosis, or disease stage at diagnosis between the ILNR and ENR groups. These data indicate good fidelity of the ILNR–ENR matching process. Patterns of ILNR are described in Table 3.
Table 2.
Demographics of isolated lymph node relapse and extranodal relapse ovarian carcinoma cohorts
| Factor | Class | ILNR, n = 49 |
ENR, n = 49 |
ILNR vs ENR |
||
|---|---|---|---|---|---|---|
| N | %/range | N | %/range | P value | ||
| Stage at diagnosis | I | 5 | 10.6 | 5 | 10.2 | 1.000a |
| II | 10 | 21.3 | 11 | 22.4 | ||
| III | 27 | 57.4 | 28 | 57.1 | ||
| IV | 5 | 10.6 | 5 | 10.2 | ||
| NA | 2 | 0 | ||||
| Histology at diagnosis | Serous | 25 | 51.0 | 33 | 67.3 | .502b |
| Endometrioid | 12 | 24.5 | 11 | 22.5 | ||
| Clear cell | 1 | 2.0 | 1 | 2.0 | ||
| Mixed histology | 8 | 16.3 | 4 | 8.2 | ||
| Unclassified adenocarcinoma | 3 | 6.1 | 0 | 0.0 | ||
| Grade at diagnosis | I | 0 | 0.0 | 1 | 2.0 | 1.000c |
| II | 6 | 13.0 | 6 | 12.2 | ||
| III | 40 | 87.0 | 42 | 75.7 | ||
| NA | 3 | 0 | ||||
| Contemporary histologic classification | HGS | 34 | 89.5 | 31 | 86.1 | .733d |
| Endometrioid | 2 | 5.3 | 3 | 8.3 | ||
| LGS | 2 | 5.3 | 1 | 2.8 | ||
| Mixed HGS / endometrioid | 0 | 0.0 | 1∗ | 2.8 | ||
| No specimen available | 11 | 13 | ||||
| Surgical debulking status | RD <2 cm | 34 | 75.6 | 33 | 70.2 | .733e |
| RD 2–5 cm | 7 | 15.6 | 8 | 17.0 | ||
| RD ≥5 cm | 4 | 8.9 | 6 | 12.8 | ||
| NA | 4 | 2 | ||||
| First-line chemotherapy | Platinum | 21 | 42.9 | 17 | 34.7 | .693f |
| Platinum combination | 25 | 51.0 | 28 | 57.1 | ||
| Other | 3 | 6.1 | 4 | 8.2 | ||
| Neoadjuvant first-line chemotherapy | Yes | 2 | 4.1 | 1 | 2.0 | 1.000g |
| No | 47 | 95.9 | 48 | 98.0 | ||
| Year of diagnosis | ≤1999 | 23 | 46.9 | 21 | 42.9 | .667f |
| 2000–2005 | 19 | 38.8 | 23 | 46.9 | ||
| 2006–2010 | 7 | 14.3 | 5 | 10.2 | ||
| Age at diagnosis | Median years | 61 | 41-80 | 62 | 41-80 | .339h |
| Specimen from diagnosis | Primary site | 33 | 91.7 | 29 | 80.6 | .307i |
| Omentum | 2 | 5.6 | 6 | 16.7 | ||
| Other | 1 | 2.8 | 1 | 2.8 | ||
| NA | 2 | 0 | ||||
| No specimen available | 11 | 13 | ||||
ENR, extranodal relapse; ILNR, isolated lymph node relapse; NA, not available; RD, residual disease.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
χ2 test, stage I/II vs stage III/IV
χ2 test, serous/mixed vs other
χ2 test, grade I/II vs grade III
Fisher exact test, high-grade serous vs non-high-grade serous
χ2 test, RD <2 cm vs ≥2 cm
χ2 test
Fisher exact test
Welch 2-sample t test
Fisher exact test, primary site vs omentum/other
This tumor had 2 morphologically distinct components with different immunophenotypes.
Table 3.
Patterns of isolated lymph node relapse ovarian carcinoma
| Cases (n) | Proportion of cases (%) | |
|---|---|---|
| ILNR pattern | ||
| Single-site | 22 | 44.9 |
| Multiregional | ||
| 2 | 17 | 34.7 |
| 3 | 8 | 16.3 |
| 4 | 2 | 4.1 |
| ILNR sites | ||
| Para-aortic only | 16 | 32.7 |
| Pelvic only | 4 | 8.2 |
| Inguinal only | 2 | 4.1 |
| Pelvic & para-aortic | 6 | 12.2 |
| Supraclavicular and other sites | 6 | 12.2 |
| Pelvic, para-aortic, and other(s) | 6 | 12.2 |
| Other combinations | 9 | 18.4 |
ILNR, isolated lymph node relapse.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Clinical outcome in isolated lymph node relapse vs extranodal relapse
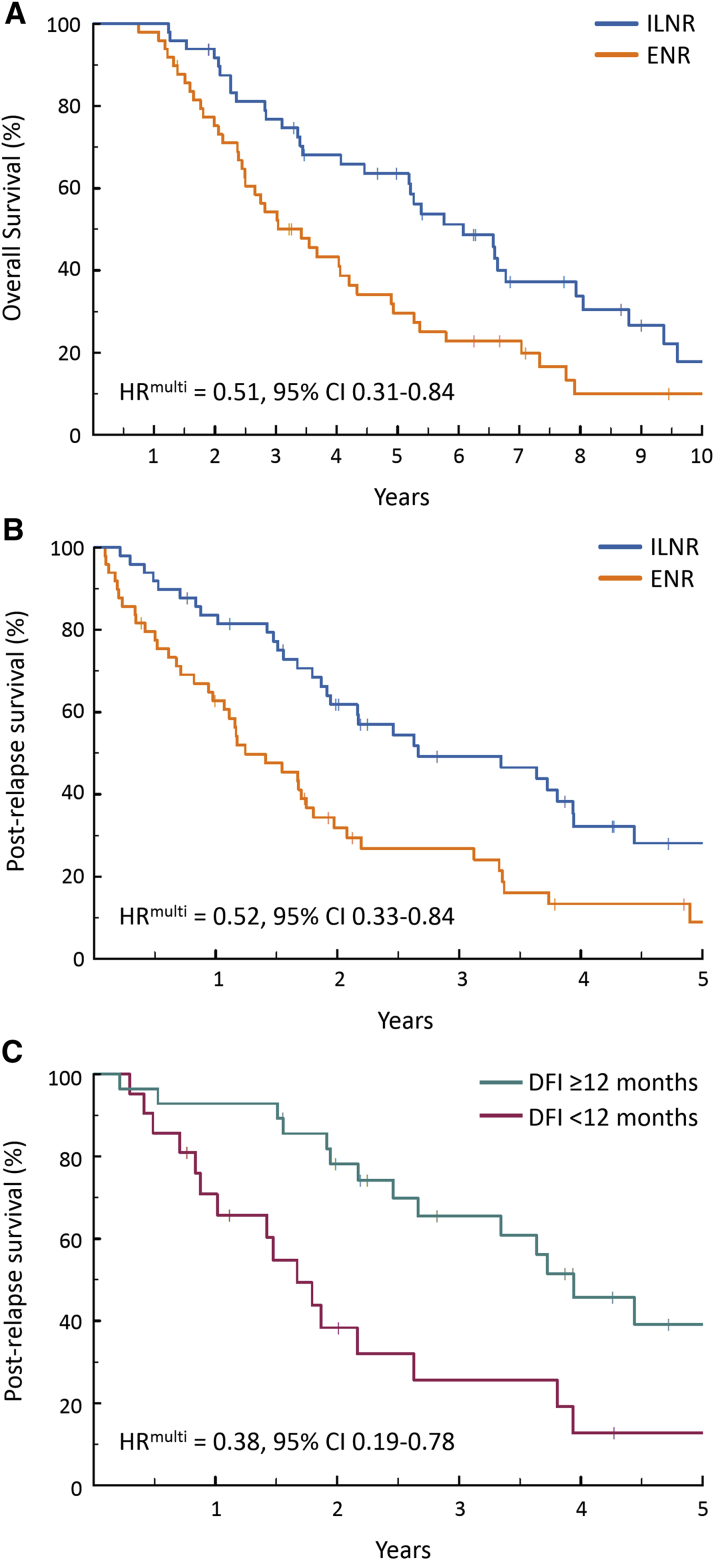
ILNR patients displayed prolonged OS and PRS compared with the ENR cohort (HR = 0.55 [0.34–0.87], P = .011 and HR = 0.50 [0.31–0.80], P = .004) (Figure 1, A and Figure 1, B). The median OS and PRS in the ILNR cohort was 72.9 (95% CI 62.2–96.5) and 32.0 (95% CI 23.3–53.3) months, compared with 41.1 (95% CI 30.0–58.8) and 14.9 (95% CI 12.9–23.7) months in the ENR cohort. The length of the DFI prior to ILNR or ENR was not significantly different between the 2 cohorts (HR = 0.86 [0.60–1.29], P = .473).
Figure 1.
Clinical outcome of isolated lymph node relapse OC
A, Overall survival in ILNR vs ENR OC. B, Postrelapse survival in ILNR vs ENR OC. C, Postrelapse survival in ILNR OC by DFI length.
CI, confidence interval; DFI, disease-free interval; ENR, extranodal relapse; HR, hazard ratio; ILNR, isolated lymph node relapse; OC, ovarian carcinoma.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Multivariable analysis for OS accounting for extent of RD following primary debulking, FIGO stage, and age at diagnosis identified significantly prolonged OS in the ILNR cohort (HRmulti = 0.51 [0.31–0.84], P = .008) (Appendix: Supplementary Table S4). Multivariable analysis of PRS, accounting for DFI and age, identified prolonged PRS in ILNR cases (HRmulti = 0.52 [0.33–0.84], P = .007) (Appendix: Supplementary Table S5).
Significantly prolonged OS (HRmulti for OS = 0.53 [0.29–0.99], P = .046) and PRS (HRmulti for PRS = 0.54 [0.31–0.96], P = .037) was demonstrated for ILNR OC when HGS cases were considered specifically (34 ILNR HGS OCs, 31 ENR HGS OCs).
Longer disease-free interval is associated with prolonged postrelapse survival in isolated lymph node relapse ovarian carcinoma
The importance of DFI on clinical outcome in ILNR OC remains controversial, with some authors reporting no association between DFI length and PRS or OS in this setting11, 16, 18 and others reporting significant associations.12, 14, 15 Within the ILNR cohort, DFI ≥12 months was associated with markedly prolonged PRS when accounting for patient age (HRmulti = 0.38 [0.19–0.78], P = .008), with median PRS of 47.3 months vs 20.1 months in those with DFI ≥12 months and DFI <12 months, respectively (Figure 1, C).
Impact of isolated lymph node relapse pattern on outcome
There was no clear differential PRS between multiregion ILNR and single-region ILNR (2 regions vs single-site HR = 1.06 [0.49–2.30], P = .890; ≥3 sites vs single-site HR = 0.94 [0.36–1.43], P = .898).
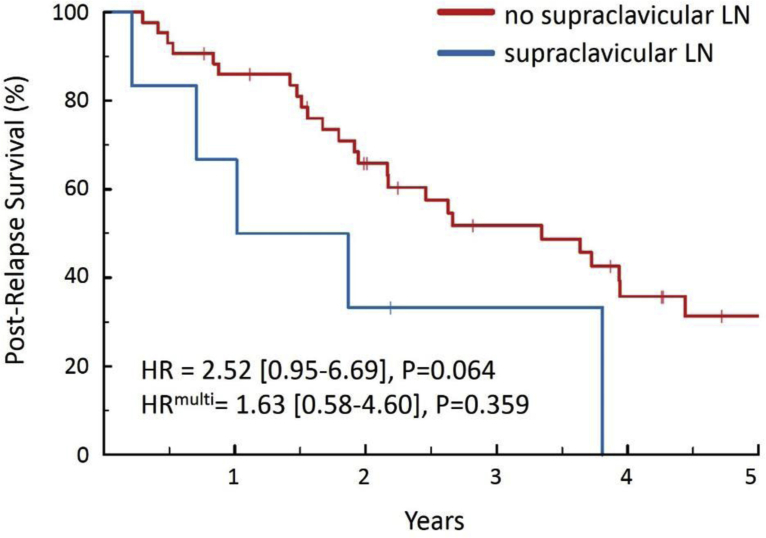
Six ILNR cases (12.2%) involved supraclavicular LN sites. Although these cases demonstrated an apparent trend for inferior PRS (HR = 2.52 [0.95–6.69], P = .064) (Appendix: Supplementary Figure S3), there was no significant difference after accounting for DFI and age (HRmulti = 1.63 [0.58–4.60], P = .359). Other specific LN sites were not associated with apparent differential PRS (Appendix: Supplementary Table S6).
Supplementary Figure S3.
Postrelapse survival of isolated lymph node relapse ovarian carcinoma with and without supraclavicular lymph node involvement
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Molecular landscape of isolated lymph node relapse high-grade serous ovarian carcinoma
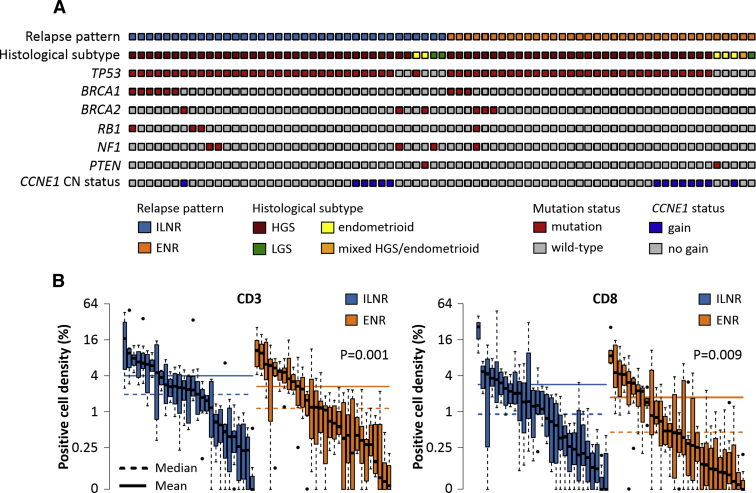
Sixty-four HGS OC cases (33 ILNR, 31 ENR) were successfully characterized for HR gene mutations and CCNE1 copy number. Frequencies of genomic abnormalities are outlined in Figure 2, A and Supplementary Table S3. Within HGS OC cases, there was no significant difference in the rate of CCNE1 copy number gain (18.2%, 6/33 vs 22.6%, 7/31, P = .900) or BRCA1/2 mutation (24.4%, 8/33 vs 19.4%, 6/31, P = .865) between the ILNR and ENR cohorts (Figure 2, A).
Figure 2.
Molecular landscape of isolated lymph node relapse and extranodal relapse ovarian carcinoma
A, Genomic events in ILNR and ENR cases. B, Tumor-infiltrating lymphocyte burden of ILNR and ENR HGS ovarian carcinomas.
ENR, extranodal relapse; HGS, high-grade serous; ILNR, isolated lymph node relapse; LGS, low-grade serous.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
The CD3+ and CD8+ TIL burden was greater in diagnostic tumor specimens from HGS OC patients who went on to experience ILNR when compared with their ENR counterparts (median CD3+ cell density 1.94% vs 1.13%, P = .001 and median CD8+ cell density 0.90% vs 0.45%, P = .009; Bonferroni-adjusted P = .003 and P = .019) (Figure 2, B).
Comment
Principal findings
The principal findings of this study are as follows: (1) ILNR represents a distinct pattern of OC relapse with prolonged survival vs ENR cases; (2) longer DFI prior to ILNR is associated with prolonged PRS in ILNR; (3) ILNR OC do not demonstrate significantly differential composition of known genomic subtypes associated with prognosis, namely BRCA1/2 mutation or gain of CCNE1; (4) cases that go on to experience ILNR demonstrate greater TIL burden at diagnosis compared with ENR cases.
Study strengths and limitations
A key strength of this study is the direct comparison of ILNR OC with matched ENR cases: a number of studies have reported ILNR as a distinct pattern of OC relapse with a relatively indolent disease course but have not systematically compared ILNR cases directly to a matched ENR cohort.11, 12, 13, 14, 15, 16, 17, 18 Moreover, these studies did not perform pathology review of identified cases, precluding the ability to characterize ILNR outcome in the context of contemporary OC histotypes, which are now known to display markedly differential clinical outcome.22 Critically, we characterize ILNR OC following contemporary histologic subtyping to facilitate investigation of ILNR in a histotype-specific manner.
The majority of previous studies investigating ILNR have identified fewer than 20 OC cases of serous histology that go on to experience this rare relapse pattern; moreover, previous reports have not performed molecular characterization of OC cases that demonstrate ILNR.11, 12, 13, 14, 15, 16, 17, 18 We identified 49 ILNR OC patients treated within the Edinburgh Cancer Centre, including 34 cases reviewed as HGS OC. This study represents the largest ILNR OC series from a single center and the only report investigating the molecular landscape of ILNR OC to date.
Though this study does represent one of the largest reported ILNR OC cohorts, case numbers were still restricted owing to the rarity of ILNR OC. In particular, power to detect differential outcome between distinct patterns of ILNR was limited, and we could not perform meaningful analysis comparing rates of rare genomic events present in both ILNR and ENR cohorts, including mutational events in RB1, NF1, and PTEN, as well as gene-specific analysis of BRCA1 and BRCA2. Other limitations of this study include heterogeneous treatment of OC patients across the time period in which these cases were diagnosed, though diagnosis periods were comparable between the ILNR and ENR cohorts (Table 2).
Clinical outcome in isolated lymph node relapse ovarian carcinoma
The median PRS and OS of ILNR cases was approximately 2.7 and 6 years, consistent with previous reports of ILNR OC.11, 12, 13, 14, 15, 16, 17, 18 ILNR cases displayed significantly prolonged OS and PRS compared to their ENR counterparts upon multivariable analysis (HRmulti = 0.51 and 0.52 for OS and PRS). Critically, this difference was maintained in a histotype-specific analysis of HGS cases, which account for the majority of OCs. To our knowledge, this is the first report directly demonstrating a significant difference in outcome between ILNR and ENR OC.
Only half of the reports investigating the impact of DFI length on ILNR outcome to date have identified associations with OS or PRS.12, 14, 15 Here, we demonstrate that DFI ≥12 months is associated with a substantial PRS benefit (median PRS approximately 3.9 vs 1.7 years), largely reflective of established associations in unselected OC cases.23 Although this contradicts reports from some investigators,11, 16, 18 2 of these studies reported specifically in the context of ILNR undergoing secondary debulking16, 18 and the other compared cases using a cut-off DFI of 24 months, rather than 12 months as described here,11 potentially explaining this discrepancy. Notably, the intervals considered in our study are akin to those used clinically to define platinum sensitivity in unselected relapsed OC.23
We show no significant difference in clinical outcome between patients with ILNR at multiple sites vs those with single-site ILNR, or between distinct patterns of ILNR. Although univariable analysis suggested that supraclavicular LN involvement may confer inferior PRS, this trend was not apparent when accounting for DFI and patient age, suggesting that this is not a genuine phenomenon of supraclavicular ILNR. Notably, the number of patients with supraclavicular LN involvement was low (n = 6). Together, these data support the consideration of ILNR OC as a single disease entity, regardless of the number and location of involved sites.
The genomic landscape of isolated lymph node relapse ovarian carcinoma
Until now, the molecular landscape of ILNR has been completely uncharacterized. It has therefore been unclear as to whether OC cases that go on to experience ILNR demonstrate enrichment of tumors belonging to known favorable genomic subgroups. Within unselected cohorts of HGS OC, inactivation of BRCA1 or BRCA2 has been associated with favorable outcome,3, 4 while copy number gain of CCNE1 has been associated with poor survival and chemoresistance.3, 7 Genomic characterization of this cohort did not identify significant depletion or enrichment of these molecular events in ILNR HGS OC cases versus their ENR counterparts. These data suggest that the survival benefit of ILNR OC is not underpinned by large-scale enrichment for BRCA1/2-mutant cases with favorable prognosis or absence of CCNE1-gained cases that have poorer prognosis, and suggest that these genomic subgroups do not display markedly differential propensity for ILNR.
Greater tumor-infiltrating lymphocyte burden at diagnosis in patients who subsequently experience isolated lymph node relapse
Intriguingly, assessment of the CD3+ and CD8+ cell burden in ILNR and ENR tumor material—reflective of whole T cell and cytotoxic T cell populations—uncovered significantly greater TIL burden in diagnostic tissue from patients who subsequently experienced ILNR (2-fold enrichment for CD8+ cells, approximately 1.7-fold enrichment for CD3+ cells). These data suggest that active engagement of the immune system at diagnosis impacts upon the nature of disease at relapse, and that immune-mediated control of cancer cells may contribute to the indolent disease course of ILNR OC. Indeed, these data may well be of interest in relation to the use of immune-directed therapies in cancer treatment.24, 25 However, though many ILNR cases displayed high TIL burden, some cases demonstrated relatively low levels of TILs, alluding to mechanisms beyond effective T-cell engagement at diagnosis underpinning some ILNR cases.
Conclusion
Collectively, the data presented here—supported by previous descriptions of apparent ILNR in the literature—demonstrate that ILNR represents a distinct pattern of OC with favorable clinical outcome when compared with ENR. Patients that go on to experience ILNR harbor greater TIL burden at diagnosis, but they do not show marked enrichment or depletion of known genomic subgroups associated with differential outcome.
Acknowledgments
We extend our thanks to the patients who contributed to this study and to the Edinburgh Ovarian Cancer Database, from which the clinical data reported here were retrieved. We are thankful to the Wellcome Trust Clinical Research Facility (Western General Hospital, Edinburgh, UK) for their support with the sequencing described here, and to the Nicola Murray Foundation for their generous support of the Nicola Murray Centre for Ovarian Cancer Research.
Footnotes
This study was supported by an MRC PhD Studentship and MRC-funded Research Fellowship awarded to R.L.H., and by funding from The Nicola Murray Foundation.
The following authors report no conflicts of interest: R.L.H., J.C., A.M.M., M.C., A.H.P., T.R., C.A.S., C.S.H. M.M. reports honoraria from Tesaro, BristolMyersSquibb, and Roche. F.N. reports nonpersonal interests in AstraZeneca and Tesaro. C.G. discloses the following: research funding from AstraZeneca, Aprea, Nucana, Tesaro, and Novartis; honoraria/consultancy fees from Roche, AstraZeneca, Tesaro, Nucana, Av Clovis, Foundation One, Cor2Ed, and Sierra Oncology; named on issued/pending patents related to predicting treatment response in ovarian cancer outside the scope of the work described here.
Cite this article as: Hollis RL, Carmichael J, Meynert AM, et al. Clinical and molecular characterization of ovarian carcinoma displaying isolated lymph node relapse. Am J Obstet Gynecol 2019;221:245.e1-15.
Appendix
Supplementary Table S1.
Tolerances for electronic matching of extranodal relapse to isolated lymph node relapse
| Feature | ILNR OC case documented diagnostic characteristic | Matched ENR OC case documented diagnostic characteristic | Patients |
|---|---|---|---|
| Stage at diagnosis | Inadequate information to stage | Stage II, n = 1 | 3 |
| Stage IIIC, n = 1 | |||
| Stage IIB | Stage IIA, n = 1 | ||
| Documented histology at diagnosis | Adenocarcinoma | Serous papillary, n = 3 | 9 |
| Endometrioid | Mixed serous/endometrioid, n = 1 | ||
| Mixed histology | Serous papillary, n = 5 | ||
| Grade at diagnosis | NA | Grade I, n = 1 | 3 |
| Grade III, n = 2 | |||
| RD following debulking | <2 cm | NA, n = 1 | 4 |
| NA | 2–5 cm, n = 1 | ||
| >5 cm, n = 2 |
The matching criteria were relaxed for 2 fields for 2 patients and 3 fields for 1 patient, as follows: 1 x stage IIB grade III mixed histology carcinoma with RD <2 cm matched to stage IIA grade III serous carcinoma with RD <2 cm; 1 x stage IV unclassified adenocarcinoma of unknown grade and RD <2 cm matched to stage IV grade III serous carcinoma with RD <2 cm; 1 x stage IV unclassified adenocarcinoma of unknown grade and RD not available matched to stage IV grade III serous carcinoma with RD >5 cm.
ENR, extranodal relapse; ILNR, isolated lymph node relapse; NA, not available; OC, ovarian carcinoma; RD, residual disease.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Supplementary Table S2.
Cellularity of specimens used for DNA extraction
| Tumor cellularity of macrodissected area | Number (%) of cases |
|---|---|
| <20% | 3 (4.1%) |
| 20–39% | 7 (9.5%) |
| 40–59% | 9 (12.2%) |
| 60–79% | 26 (35.1%) |
| ≥80% | 29 (39.2%) |
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Supplementary Table S3.
Frequency of patients with detrimental mutations in genes sequenced on Integrated DNA Technologies gene capture high throughput sequencing panel
| Gene | ILNR OC with mutation | ENR OC with mutation |
|---|---|---|
| ABCB1 | 1 | 0 |
| ARID1A | 1 | 2 |
| ATM | 2 | 0 |
| ATR | 1 | 0 |
| BRCA1 | 6 | 3 |
| BRCA2 | 3 | 3 |
| CTNNB1 | 1 | 1 |
| FANCC | 0 | 1 |
| KRAS | 1 | 1 |
| MSH2 | 1 | 1 |
| NF1 | 4 | 1 |
| PIK3CA | 2 | 1 |
| PRKDC | 1 | 0 |
| PTEN | 1 | 1 |
| RB1 | 3 | 1 |
| SLX4 | 0 | 1 |
| TP53 | 32 | 31 |
| Genes with no detected mutations: ATRX, BAP1, BARD1, BCL2L1, BLM, BRAF, BRIP1, C11orf65, CCNE1, CDK12, CHD4, CHEK1, CHEK2, EGFR, EMSY, ERBB2, ERCC4, EZH2, FANCA, FANCB, FANCD2, FANCE, FANCF, FANCG, FANCI, FANCL, FANCM, GNAS, KIT, MAD2L2, MDM2, MLH1, MRE11, MSH6, MUS81, MUTYH, NBN, NDUFB2, NF2, NRAS, PALB2, PARP1, PARP2, PAXIP1, PDGFRA, PER3, PMS2, PPP2R1A, PPP2R2A, RAD50, RAD51, RAD51B, RAD51C, RAD54L, RNASEH2A, RNASEH2B, RNASEH2C, RPA1, RUNDC3B, SHFM1, SLC25A40, SLFN11, TOE1, TP53BP1, UBE2T, VRK2 | ||
ENR, extranodal relapse; ILNR, isolated lymph node relapse; OC, ovarian carcinoma.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Supplementary Table S4.
Multivariable analysis for overall survival in isolated lymph node relapse vs extranodal relapse in ovarian carcinoma
| Factor | Class | N | HRmulti | Lower 95% CI | Upper 95% CI | P |
|---|---|---|---|---|---|---|
| Relapse type | ILNR | 49 | 0.51 | 0.31 | 0.84 | .008 |
| ENR | 49 | Ref | Ref | Ref | Ref | |
| Stage at diagnosis | Early (I/II) | 31 | 0.41 | 0.17 | 1.02 | .055 |
| III | 55 | 0.45 | 0.19 | 1.03 | .060 | |
| IV | 10 | Ref | Ref | Ref | Ref | |
| NA | 2 | - | - | - | - | |
| Surgical debulking status | RD <2 cm | 67 | 0.60 | 0.32 | 1.12 | .109 |
| RD ≥2 cm | 25 | Ref | Ref | Ref | Ref | |
| NA | 6 | - | - | - | - | |
| Age at diagnosis | Years | 1.03 | 1.00 | 1.05 | .050 |
CI, confidence interval; ENR, extranodal relapse; HR, hazard ratio; ILNR, isolated lymph node relapse; RD, residual disease.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Supplementary Table S5.
Multivariable analysis of time to isolated lymph node relapse as a predictor of postrelapse survival in isolated lymph node relapse ovarian carcinoma
| Factor | Class | N | HRmulti | Lower 95% CI | Upper 95% CI | P |
|---|---|---|---|---|---|---|
| Relapse type | ILNR | 49 | 0.53 | 0.33 | 0.84 | .007 |
| ENR | 49 | Ref | Ref | Ref | Ref | |
| DFI | ≥12 months | 46 | 0.47 | 0.29 | 0.75 | .006 |
| <12 months | 52 | Ref | Ref | Ref | Ref | |
| Age | Years | 1.03 | 1.01 | 1.06 | .006 |
CI, confidence interval; DFI, disease-free interval; HR, hazard ratio.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
Supplementary Table S6.
Univariable analyses of specific lymph node site involvement and association with postrelapse survival
| Factor | Class | N | HR | Lower 95% CI | Upper 95% CI | P |
|---|---|---|---|---|---|---|
| Supraclavicular LN involvement | Yes | 6 | 2.52 | 0.95 | 6.69 | .064 |
| No | 43 | Ref | Ref | Ref | Ref | |
| Pelvic LN involvement | Yes | 20 | 0.73 | 0.35 | 1.51 | .393 |
| No | 29 | Ref | Ref | Ref | Ref | |
| Inguinal LN involvement | Yes | 9 | 0.72 | 0.28 | 1.87 | .502 |
| No | 40 | Ref | Ref | Ref | Ref | |
| Para-aortic LN involvement | Yes | 36 | 1.10 | 0.48 | 2.56 | .818 |
| No | 13 | Ref | Ref | Ref | Ref |
CI, confidence interval; HR, hazard ratio; LN, lymph node.
Hollis et al. Clinical and molecular characterization of ILNR ovarian carcinoma. Am J Obstet Gynecol 2019.
References
- 1.Bray F., Ferlay J., Soerjomataram I., Siegel R.L., Torre L.A., Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Prat J. Ovarian carcinomas: five distinct diseases with different origins, genetic alterations, and clinicopathological features. Virchows Arch. 2012;460:237–249. doi: 10.1007/s00428-012-1203-5. [DOI] [PubMed] [Google Scholar]
- 3.The Cancer Genome Atlas Investigators Integrated genomic analyses of ovarian carcinoma. Nature. 2011;474:609–615. doi: 10.1038/nature10166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tan D.S., Rothermundt C., Thomas K. “BRCAness” syndrome in ovarian cancer: a case-control study describing the clinical features and outcome of patients with epithelial ovarian cancer associated with BRCA1 and BRCA2 mutations. J Clin Oncol. 2008;26:5530–5536. doi: 10.1200/JCO.2008.16.1703. [DOI] [PubMed] [Google Scholar]
- 5.Fong P.C., Boss D.S., Yap T.A. Inhibition of poly(ADP-ribose) polymerase in tumors from BRCA mutation carriers. N Engl J Med. 2009;361:123–134. doi: 10.1056/NEJMoa0900212. [DOI] [PubMed] [Google Scholar]
- 6.Moore K., Colombo N., Scambia G. Maintenance olaparib in patients with newly diagnosed advanced ovarian cancer. N Engl J Med. 2018;379:2495–2505. doi: 10.1056/NEJMoa1810858. [DOI] [PubMed] [Google Scholar]
- 7.Patch A.M., Christie E.L., Etemadmoghadam D. Whole-genome characterization of chemoresistant ovarian cancer. Nature. 2015;521:489–494. doi: 10.1038/nature14410. [DOI] [PubMed] [Google Scholar]
- 8.Ledermann J.A., Raja F.A., Fotopoulou C., Gonzalez-Martin A., Colombo N., Sessa C. Newly diagnosed and relapsed epithelial ovarian carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013;24(Suppl 6):vi24–vi32. doi: 10.1093/annonc/mdt333. [DOI] [PubMed] [Google Scholar]
- 9.Armstrong D.K. Relapsed ovarian cancer: challenges and management strategies for a chronic disease. Oncologist. 2002;7(Suppl 5):20–28. doi: 10.1634/theoncologist.7-suppl_5-20. [DOI] [PubMed] [Google Scholar]
- 10.Amate P., Huchon C., Dessapt A.L. Ovarian cancer: sites of recurrence. Int J Gynecol Cancer. 2013;23:1590–1596. doi: 10.1097/IGC.0000000000000007. [DOI] [PubMed] [Google Scholar]
- 11.Blanchard P., Plantade A., Pages C. Isolated lymph node relapse of epithelial ovarian carcinoma: outcomes and prognostic factors. Gynecol Oncol. 2007;104:41–45. doi: 10.1016/j.ygyno.2006.06.039. [DOI] [PubMed] [Google Scholar]
- 12.Legge F., Petrillo M., Adamo V., Pisconti S., Scambia G., Ferrandina G. Epithelial ovarian cancer relapsing as isolated lymph node disease: natural history and clinical outcome. BMC Cancer. 2008;8:367. doi: 10.1186/1471-2407-8-367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Uzan C., Morice P., Rey A. Outcomes after combined therapy including surgical resection in patients with epithelial ovarian cancer recurrence(s) exclusively in lymph nodes. Ann Surg Oncol. 2004;11:658–664. doi: 10.1245/ASO.2004.11.023. [DOI] [PubMed] [Google Scholar]
- 14.Gadducci A., Cosio S., Zola P. The clinical outcome of epithelial ovarian cancer patients with apparently isolated lymph node recurrence: a multicenter retrospective Italian study. Gynecol Oncol. 2010;116:358–363. doi: 10.1016/j.ygyno.2009.11.008. [DOI] [PubMed] [Google Scholar]
- 15.Tu H., Huang H., Huang Q.D., Li Z., Feng Y.L., Liu J.H. [Treatment and prognostic analysis of ovarian cancer patients with isolated region of lymph node recurrence] Zhonghua Fu Chan Ke Za Zhi. 2012;47:928–933. [PubMed] [Google Scholar]
- 16.Ferrero A., Ditto A., Giorda G. Secondary cytoreductive surgery for isolated lymph node recurrence of epithelial ovarian cancer: a multicenter study. Eur J Surg Oncol. 2014;40:891–898. doi: 10.1016/j.ejso.2013.11.026. [DOI] [PubMed] [Google Scholar]
- 17.Santillan A., Karam A.K., Li A.J. Secondary cytoreductive surgery for isolated nodal recurrence in patients with epithelial ovarian cancer. Gynecol Oncol. 2007;104:686–690. doi: 10.1016/j.ygyno.2006.10.020. [DOI] [PubMed] [Google Scholar]
- 18.Fotiou S., Aliki T., Petros Z. Secondary cytoreductive surgery in patients presenting with isolated nodal recurrence of epithelial ovarian cancer. Gynecol Oncol. 2009;114:178–182. doi: 10.1016/j.ygyno.2009.04.025. [DOI] [PubMed] [Google Scholar]
- 19.Kobel M., Rahimi K., Rambau P.F. An immunohistochemical algorithm for ovarian carcinoma typing. Int J Gynecol Pathol. 2016;35:430–441. doi: 10.1097/PGP.0000000000000274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bankhead P., Loughrey M.B., Fernández J.A. QuPath: Open source software for digital pathology image analysis. Sci Rep. 2017;7:16878. doi: 10.1038/s41598-017-17204-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Therneau T. A Package for Survival Analysis in S. 2015. Available at: https://CRAN.R-project.org/package=survival. Accessed June 14, 2019.
- 22.Hollis R.L., Gourley C. Genetic and molecular changes in ovarian cancer. Cancer Biol Med. 2016;13:236–247. doi: 10.20892/j.issn.2095-3941.2016.0024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ushijima K. Treatment for recurrent ovarian cancer—at first relapse. J Oncol. 2010;2010 doi: 10.1155/2010/497429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Postow M.A., Chesney J., Pavlick A.C. Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N Engl J Med. 2015;372:2006–2017. doi: 10.1056/NEJMoa1414428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Muro K., Chung H.C., Shankaran V. Pembrolizumab for patients with PD-L1-positive advanced gastric cancer (KEYNOTE-012): a multicentre, open-label, phase 1b trial. Lancet Oncol. 2016;17:717–726. doi: 10.1016/S1470-2045(16)00175-3. [DOI] [PubMed] [Google Scholar]