Abstract
This review describes in detail the phytochemistry and neurological effects of the medicinal herb Centella asiatica (L.) Urban. C. asiatica is a small perennial plant that grows in moist, tropical and sub-tropical regions throughout the world. Phytochemicals identified from C. asiatica to date include isoprenoids (sesquiterpenes, plant sterols, pentacyclic triterpenoids and saponins) and phenylpropanoid derivatives (eugenol derivatives, caffeoylquinic acids, and flavonoids). Contemporary methods for fingerprinting and characterization of compounds in C. asiatica extracts include liquid chromatography and/or ion mobility spectrometry in conjunction with high-resolution mass spectrometry. Multiple studies in rodent models, and a limited number of human studies support C. asiatica’s traditional reputation as a cognitive enhancer, as well as its anxiolytic and anticonvulsant effects. Neuroprotective effects of C.asiatica are seen in several in vitro models, for example against beta amyloid toxicity, and appear to be associated with increased mitochondrial activity, improved antioxidant status, and/or inhibition of the pro-inflammatory enzyme, phospholipase A2. Neurotropic effects of C. asiatica include increased dendritic arborization and synaptogenesis, and may be due to modulations of signal transduction pathways such as ERK1/2 and Akt. Many of these neurotropic and neuroprotective properties of C.asiatica have been associated with the triterpene compounds asiatic acid, asiaticoside and madecassoside. More recently, caffeoylquinic acids are emerging as a second important group of active compounds in C. asiatica, with the potential of enhancing the Nrf2-antioxidant response pathway. The absorption, distribution, metabolism and excretion of the triterpenes, caffeoylquinic acids and flavonoids found in C. asiatica have been studied in humans and animal models, and the compounds or their metabolites found in the brain. This review highlights the remarkable potential for C. asiatica extracts and derivatives to be used in the treatment of neurological conditions, and considers the further research needed to actualize this possibility.
1. Introduction
1.1. Taxonomy, nomenclature and synonyms:
Centella asiatica (L.) Urban is a member of the plant family Apiaceae (formerly Umbelliferae), and subfamily Mackinlayoideae (USDA 2016). The plant was reassigned from the subfamily Hydrocotyloideae as a result of molecular phylogeny studies (Nicolas and Plunkett 2009). Synonyms for C. asiatica are Hydrocotyle asiatica L. and Hydrocotyle erecta L.f. (USDA 2016). The name H. asiatica is often used in older materia medica, herbal textbooks and research publications. Almost 80 species of Centella have been recorded, many of which have older synonyms under the genus Hydrocotyle (IPNI 2016). It is therefore advisable that literature searches on Centella species encompass the equivalent Hydrocotyle nomenclature (IPNI 2016) where appropriate. Common English names for C. asiatica are Indian Pennywort and Asian Pennywort (IPNI 2016). In the United States and other Western countries, the herb of commerce, and dietary supplements derived from it, are commonly referred to by the Sri Lankan Sinhalese name “gotu kola” (Nadkarni 1976; Newall et al. 1996). Common names for C. asiatica from various parts of the world are listed in Table 1.
Table 1.
Common names for Centella asiatica in different regions or languages of the world.
| Region/language | Names | Reference |
|---|---|---|
| Arabic | Artaniyal-hindi, zarnab. | (Nadkarni 1976), (Kapoor 1990) |
| Chinese | Tungchian, luei gong gen, ji xue cao. | (Brinkhaus et al. 2000), (USDA 2016) |
| English | Indian Pennywort, Asiatic pennywort, marsh pennywort, pennyweed, sheeprot, ndian water navelwort. | (USDA 2016), (Brinkhaus et al. 2000; Newall et al. 1996), |
| French | Hydrocotyle Asiatique, écuelle d’eau. | (Brinkhaus et al. 2000), (USDA 2016) |
| German | Indischer wassernabel, Asiatisches Sumpfpfennigkraut. | (Brinkhaus et al. 2000), (USDA 2016) |
| India (language given in brackets after name) | Brahmi, mandukaparni, cheka parni (Sanskrit); khulakudi, brahma-manduki (Hindi); karivana, karinga, undri (Marathi); thulkuri, brahma-manduki (Bengali); karbrahmi (Gujarati); saraswathi aku, manduka, brahmakuraku (Telegu); vellarai, babassa (Tamil); kutakam (Malayalam); ekpanni (Konkani). | (Nadkarni 1976) |
| Indonesia | Kaki kuda, pegagan, anantan gede, gagan-gagan, gang-gagan, kirok gatok, panegowan, rending, calingan rambat, kos tekosan, pagaga, tungke-tungke, papaiduh, pepiduh, piduh, puhe beta, kaki kuta, tete karo, tete kadho. | (Brinkhaus et al. 2000) |
| Italian | Idrocotyle. | (Brinkhaus et al. 2000) |
| Japan | Tsubo-kusa, tsubokura. | (Brinkhaus et al. 2000), (Kapoor 1990) |
| Malagasy | Talapetraka. | (Rahajanirina et al. 2012) |
| Malaysia | Dawoopungah-gah, pegaga. | (Nadkarni 1976), (Maulidiani et al. 2014) |
| Mauritius | Bavilaaqua. | (Brinkhaus et al. 2000) |
| Myanmar | Minkhuabin. | (Nadkarni 1976) |
| Nepal | Kholachagya. | (Kapoor 1990) |
| Persian | Sard Turkistan. | (Kapoor 1990) |
| Spanish | Sombrerito, hierba de clavo. | (USDA 2016) |
| Sri Lanka (Sinhalese) | Gotu kola; hingolukola. | (Nadkarni 1976), (Kapoor 1990) |
| Swedish | Sallatsspikblad. | (USDA 2016) |
| Thailand | Bau-bog. | (Botanica 2017) |
| Tibetan | Sin-nmar. | (Kapoor 1990) |
1.2. Botany and geographical distribution
C. asiatica is a perennial creeping plant which propagates by producing stolons. The plants consist of shovel or spade shaped leaves with scalloped edges, borne on long petioles clustered at the stem nodes. (Figure 1). The insignificant green or pinkish-white flowers are borne in dense umbels, and the seeds are pumpkin shaped nutlets 3–5 mm in length (Floridata 2016). This plant grows in swampy areas of tropical and subtropical regions of the world (James and Dubery 2009; Long et al. 2012; USDA 2016). It is native to the Asian tropical regions of the Indian Subcontinent, South East Asia, Malaysia and the Solomon Islands, as well as some temperate regions in China, Japan, Korea and Taiwan.
Figure 1:
Centella asiatica plant, showing spade-like leaves, internode stolons and small flowers.
C. asiatica is also native to African countries south of the equator, some regions in East, West, and West Central Africa, and endogenous to New South Wales in Australia and some Pacific Islands. The herb has been naturalized to several other regions of the world including the African islands of Madagascar and Seychelles, some temperate regions of Asia (e.g. the Caucasus), and several regions of South Eastern United States and South Central America (Mexico, Ecuador, Columbia and Venezuela). It is used as a modern or traditional botanical remedy in many of these areas and also finds culinary use as a salad vegetable, or in juices, in several Asian countries (Plengmuankhae and Tantitadapitak 2015). Regional common names are listed in Table 1.
1.3. Medicinal uses of C. asiatica
History and traditional uses:
Brinkhaus et al. (2000) have reviewed in detail the long history of the medicinal applications of C. asiatica, from earliest evidence of its use by the Indian physician Sushruta (ca. 1200 BC), to traditional herbal medicine systems in Asian and African countries, its introduction to, and scientific study in Europe in the 19th and 20th centuries, and present worldwide use of the herb or its derivatives, in commercial topical and oral products (Brinkhaus et al. 2000). In several regions of the world, preparations of the herb were applied topically to treat infectious skin diseases and accelerate healing of skin ulcers and wounds, while internal preparations were used to treat severe dysentery, gastric ulcers and syphilitic lesions (Brinkhaus et al. 2000; Long et al. 2012; Nadkarni 1976). Of particular relevance to this review however, is C. asiatica’s importance in the Ayurvedic tradition as a “medhyarasayana” herb i.e. one that has rejuvenative effects, boosts memory, prevents cognitive deficits and improves brain function (Kapoor 1990; Nadkarni 1976; Shinomol et al. 2011).
Preparations:
For rejuvenatory (“rasayana”) effects, the herb is traditionally administered orally as the fresh juice “swarasam” or prepared in clarified butter as a “gritham” (Nadkarni 1976). For “mental weakness and for improving memory”, powder of the dried leaves is also given with milk (Nadkarni 1976). For other uses, topical or oral preparations include dried leaf powder, poultices of the fresh leaves, leaf juice, or decoctions of the dried leaf (Long et al. 2012; Nadkarni 1976). In modern use as a dietary supplement, C. asiatica is typically sold as capsules of the dried herb or a dried extract, or as an herbal tincture. Extracts containing standardized mixtures of the purified triterpenes, which have been used in clinical studies, include TTFCA (total triterpenic fraction of Centella asiatica) and TECA (titrated extract of Centella asiatica) (Brinkhaus et al. 2000). More recently developed commercial preparations include standardized mixtures of purified triterpenes from C. asiatica, e.g. CAST™ (Centella asiatica selected triterpenes) and Centerox™ by Indena SpA (Milan, Italy), or crude extracts which are standardized to their triterpene content, e.g. Centevita™ (Indena) and Centellin® (Sabinsa Corp, New Jersey USA).
The present review will focus on the phytochemistry of C. asiatica, and the neuroprotective and cognitive enhancing properties of C. asiatica and its constituent compounds. Preclinical studies have used crude solvent extracts of the herb, or purified compounds, while clinical studies have used a plethora of products, often not well described (Table 4). A significant body of scientific research also exists on the wound healing and gastroprotective properties of preparations of C. asiatica or its triterpenes. Those studies have been reviewed elsewhere (Brinkhaus et al. 2000) and are outside the scope of the present review.
Table 4.
Human trials using Centella asiatica to assess changes in cognition, mood and quality of life
| CA Material Given | CA Source Country | Study Location | Standardization | Dose | Study Design | Control | Duration | Subjects | Assessment | Authors’ Conclusions | Referenc e |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Tablets of shade-dried powdered whole plant | India | India | Not provided | 500 mg daily | Double-blind; randomized | Starch placebo | Three months | 30 mentally-retarded children (age 7–18 years; both genders) | Binet-Kamat Test, adapted Weschler intelligence scale for children | ▲ IQ, ▲ behavior | (Rao et al. 1973) |
| Crude stem and leaf powder by Nature’s Way | Madagascar (personal communicatio n with Nature’s Way) | Canada | None (personal communication with Nature’s Way | 12 g dissolved in grape juice | Double-blind; randomized | Grape juice placebo | Once | 40 healthy adults (age 18–45; 21 males, 19 females) | Acoustic startle response, 100-mm visual analog scale for moods, heart rate and blood pressure | ▼ startle response, ▲ self-rated energy level | (Bradwejn et al. 2000) |
| Capsules of “specialized” aerial part extract | Thailand | Thailand | Total phenolic content (tannic acid equivalents) = 29.9 mg/g; asiaticoside 1.09 mg/g and asiatic acid 28.89 mg/g of crude extract | 250, 500 or 750 mg daily | Double-blind; randomized | Placebo | Once and for two months | 28 healthy elderly (mean age 65.05 ± 3.56 years; 4 males, 25 females) | Modified CDR test battery; 16 visual analogue scales of Bond-Lader | ▼ reaction time, ▲ accuracy of working memory, ▲ alertness and calmness | (Wattanathorn et al. 2008) |
| Capsules of dried and ground leaves | India | India | Not provided | 500 mg twice daily | Open-label | None | Six months | 60 elderly with mild cognitive impairment (age >65 years; genders not reported) | MMSE, ADL scale, IADL, caregiver interviews, Yesavage GDS | ▲ MMSE score, ▼ diastolic BP, ▲ appetite and sense of well-being, ▲ sleep, ▼ anxiety, ▼ peripheral neuritis | (Tiwari et al. 2008) |
| Capsules of crude herb | Not provided | Malaysia | Not provided | 500 mg / 10kg body weight daily | Randomize d; blinding not discussed | Corn starch placebo | Two months | 41 healthy middle-aged adults (age 35–50 years; 19 males; 22 females) | Woodcock-Johnson Cognitive Abilities Test III | ▲ cognitive ability scores (3/8 tests in males; 5/8 in females) | (Dev et al. 2009) |
| Capsules of 70% hydroethanolic extract of dried aerial parts | India | India | Ursolic acid 1.5–1.89% of plant extract | 500 mg twice daily after meals | Open-label | None | Two months | 33 adults with generalized anxiety disorder (mean age 33 years; 18 males, 15 females) | SF-36 health survey questionnaire, validated stress scale, anxiety scale, depression scale, adjustment scale and attention scale | ▼ anxiety, ▲ self-perceived stress, ▼ depression index, ▲ adjustment index, ▲ attention level | (Jana et al. 2010) |
| Capsules of a blended aqueous leaf extract combined with sugar (Patent 4721; Thailand) | Thailand | Thailand | Total phenolic content (tannic acid equivalents) = 29.9 mg/g, asiaticoside 1.09 mg/g and asiatic acid 48.89 mg/g of crude extract | 250, 500, or 750 mg daily | Double-blind; randomized | Placebo | 90 days | 80 healthy elderly (mean age 65.05 ± 3.56 years; 4 males, 76 females) | 30 second chair stand test, hand grip, six minute walk test, SF-36 quality of life questionnaire | ▲ lower extremity strength and life satisfaction subscale with 500 and 750 mg doses | (Mato et al. 2011) |
| Capsules of “Ghava Satva” | Not provided | India | Not provided | 500 mg twice daily after meals with warm water | Open-label | None | Six months | 25 elderly with MMSE<25 (age >60 years; gender not reported) | MMSE, caregiver interviews, vitals | ▲ MMSE score, ▼ mean systolic and diastolic blood pressure | (Rakesh et al. 2013) |
| Capsules of dried 70% hydro-ethanolic whole herb extraction | Not provided | Indonesia | “Standardized dry form” | 1000 or 750 mg daily | Double-blind; non-randomized | 3 mg folic acid | Six weeks | 48 subjects with post-stroke vascular cognitive impairment (mean age 60.27 ±.11.83 years; 60.41% males, 39.58% females) | MoCA-lna | ▲ delayed recall memory | (Farhana et al. 2016) |
MoCA-Ina = Montreal Cognitive Assessment-Indonesian version
MMSE = Mini Mental State Examination
CDR= Clinical Dementia Rating
ADL= Activities of Daily Living
GDS = Geriatric Depression Scale
IADL = Instrumental Activities of Daily Living
CA = Centella asiatica
▲ = increased or improved; ▼ decreased
▲ = increased or improved; ▼ decreased
2. Phytochemistry
2.1. Major phytochemicals
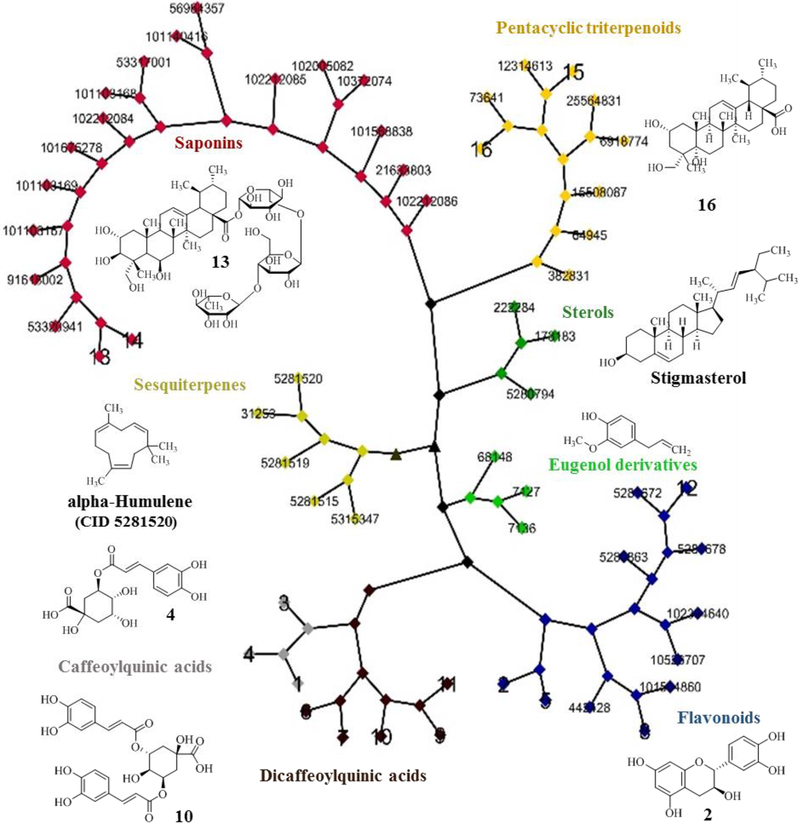
An early comprehensive overview of secondary metabolites found in C. asiatica has been provided by Brinkhaus et al. (Brinkhaus et al. 2000). These secondary metabolites are found in the aerial parts of the plants as well as in the roots and rhizomes (Brinkhaus et al. 2000). The Centella leaf transcriptome was recently published and provides a detailed map of the genes involved in the synthesis of secondary metabolites (Sangwan et al. 2013). A Cytoscape network of 57 secondary metabolites typically found in C. asiatica (Table 2, Figure 2) were clustered according to chemical 2D structure similarity using the Tanimoto method (Flowers 1998). Compounds of the isoprenoid pathway fall into four clusters: sesquiterpene, plant sterols, pentacyclic triterpenoids and saponins. Compounds that share the phenylpropenoid scaffold fall into three clusters: eugenol derivatives, caffeoylquinic acids (with sub-clusters of mono- or di- caffeoylquinic acids), and flavonoids.
Table 2.
Compounds present in Centella asiatica
| Compound | CID (PubChem) | CAS (PubChem) | Reference |
|---|---|---|---|
| alpha-Humulene | 5281520 | 6753–98–6 | (Oyedeji and Afolayan 2005) |
| Arjunolic acid | 73641 | 465–00–9 | (Azerad 2016) |
| Asiatic acid | 119034 | 464–92–6 | (Brinkhaus et al. 2000; Jamil et al. 2007) |
| Asiaticoside | 108062 | 16830–15–2 | (Brinkhaus et al. 2000) |
| Asiaticoside B | 91618002 | 125265–68–1 | (Brinkhaus et al. 2000) |
| Asiaticoside C | 101103169 | (James and Dubery 2009) | |
| Asiaticoside D | 102212084 | (Azerad 2016) | |
| Asiaticoside E | 102212085 | (Azerad 2016) | |
| Asiaticoside F | 53317001 | (Azerad 2016) | |
| Asiaticoside G | 53320941 | (Azerad 2016) | |
| β-Caryophyllene | 5281515 | 87–44–5 | (Oyedeji and Afolayan 2005) |
| Bicyclogermacrene | 5315347 | (Oyedeji and Afolayan 2005) | |
| Brahminoside B | 101504860 | (James and Dubery 2009) | |
| Campesterol | 173183 | 474–62–4 | (Jamil et al. 2007) |
| Castillicetin | 102394640 | (Chandrika and Prasad Kumarab 2015) | |
| Castilliferol | 10526707 | (Chandrika and Prasad Kumarab 2015) | |
| Catechin | 9064 | 154–23–4 | (Mustafa et al. 2010) |
| Centellasapogenol A | 15508087 | (Azerad 2016) | |
| Centellasaponin A | 21633803 | (Azerad 2016) | |
| Centellasaponin B | 101140416 | (Azerad 2016) | |
| Centellasaponin C | 101103167 | (Azerad 2016) | |
| Centellasaponin D | 101103168 | (Azerad 2016) | |
| Centelloside E | 101568838 | (Azerad 2016) | |
| Centelloside D | 56964357 | (Azerad 2016) | |
| Chavicol | 68148 | 501–92–8 | (Brinkhaus et al. 2000) |
| Chebuloside II | 102005082 | (Azerad 2016) | |
| Chlorogenic acid (3-O-Caffeoylquinic acid) | 1794427 | 327–97–9 | (Long et al. 2012) |
| Corosolic acid | 6918774 | 4547–24–4 | (Yoshida et al. 2005) |
| Cryptochlorogenic Acid, (4-O-Caffeoylquinic acid) | 9798666 | 905–99–7 | (Long et al. 2012) |
| 1,3-Dicaffeoylquinic acid | 6474640 | 19870–46–3 | (Long et al. 2012) |
| 1,5-Dicaffeoylquinic acid | 122685 | 30964–13–7 | (Long et al. 2012) |
| 3,4-Dicaffeoylquinic acid (Isochlorogenic acid B) | 5281780 | 14534–61–3 | (Long et al. 2012) |
| 3,5-Dicaffeoylquinic acid (Isochlorogenic acid A) | 6474310 | 2450–53–5 | (Long et al. 2012) |
| 4,5-Dicaffeoylquinic acid (Isochlorogenic acid C) | 6474309 | 32451–88–0 | (Long et al. 2012) |
| Epicatechin | 72276 | 490–46–0 | (Mustafa et al. 2010) |
| 3-Epimaslinic acid | 25564831 | 26563–68–8 | (Jamil et al. 2007; Yoshida et al. 2005) |
| Eugenol acetate | 7136 | 93–28–7 | (Brinkhaus et al. 2000) |
| Germacrene B | 5281519 | (Oyedeji and Afolayan 2005) | |
| Kaempferol | 5280863 | 520–18–3 | (Devkota et al. 2010) |
| Madecassic acid, brahmic acid | 73412 | 18449–41–7 | (Brinkhaus et al. 2000) |
| Madecassoside | 45356919 | 34540–22–2 | (Brinkhaus et al. 2000) |
| Methyleugenol | 7127 | 93–15–2 | (Brinkhaus et al. 2000) |
| Myrcene | 31253 | 123–35–3 | (Brinkhaus et al. 2000; Oyedeji and Afolayan 2005) |
| Myricetin | 5281672 | 529–44–2 | (Chandrika and Prasad Kumarab 2015) |
| Naringin | 442428 | 10236–47–2 | (Sangwan et al. 2013) |
| Neochlorogenic acid (5-O-Daffeoylquinic acid) | 5280633 | 906–33–2 | (Long et al. 2012) |
| Patuletin | 5281678 | 519–96–0 | (Chandrika and Prasad Kumarab 2015) |
| Pomolic Acid | 382831 | 13849–91–7 | (Yoshida et al. 2005) |
| Quadranoside IV | 10372074 | (Azerad 2016) | |
| Quercetin | 5280343 | 117–39–5 | (Sangwan et al. 2013) |
| Rutin | 5280805 | 153–18–4 | (Sangwan et al. 2013) |
| Scheffuroside B | 102212086 | (Azerad 2016) | |
| Scheffuroside F | 101675278 | (Azerad 2016) | |
| Sitosterol | 222284 | 68555–08–8 | (Yoshida et al. 2005) |
| Stigmasterol | 5280794 | 83–48–7 | (Chandrika and Prasad Kumarab 2015) |
| Terminolic acid | 12314613 | 564–13–6 | (Brinkhaus et al. 2000) |
| Ursolic acid | 64945 | 77–52–1 | (Yoshida et al. 2005) |
Figure 2.
Cytoscape network for compounds found in C. asiatica. The clustering relationship is based on structural similarities among 57 compounds using the Tanimoto algorithm. Seven main clusters are present: saponins, pentacyclic triterpenoids, sterols, sesquiterpenes, eugenol derivatives, caffeoylquinic acids (subclusters of mono- or di- caffeoylquinic acids) and flavonoids. A representative compound of each cluster is shown. The compound numbers displayed correspond to the Pubchem CIDs (Table 2), and the compound numbers 1–16 refer to the compounds given in Figure 4. ▲, indicates a node with a Tanimoto score of < 0.68. A Tanimoto score > 0.68 is statistically significant at the 95% confidence interval (Kim et al. 2012b).
Considerable variation has been reported in the volatile constituents of essential oil preparations of C. asiatica. Essential oil from the aerial parts of C. asiatica contains high levels of sesquiterpenes (C15) and monoterpenoids (C10), including α-humulene, β-caryophyllene, bicyclogermacrene, germacrene-D, and myrcene as the major constituents (Oyedeji and Afolayan 2005; Wong and Tan 1994). Other constituents have been characterized as oxygenated sesquiterpenes (e.g. humulene epoxide (C15H24O), caryophyllene oxide (C15H24O)), oxygenated monoterpenes (e.g. menthone (C10H18O), α-terpineol (C10H18O)) and a sulfide sesquiterpenoid (mintsulfide, C15H24S) (Apichartsrangkoon et al. 2009; Oyedeji and Afolayan 2005). Essential oil extracts show antibacterial activities against Gram-positive (Bacillus subtilis, Staphylococcus aureus) and Gram-negative bacteria (Escherichia coli, Pseudomonas aeruginosa, Shigella sonnei) (Oyedeji and Afolayan 2005).
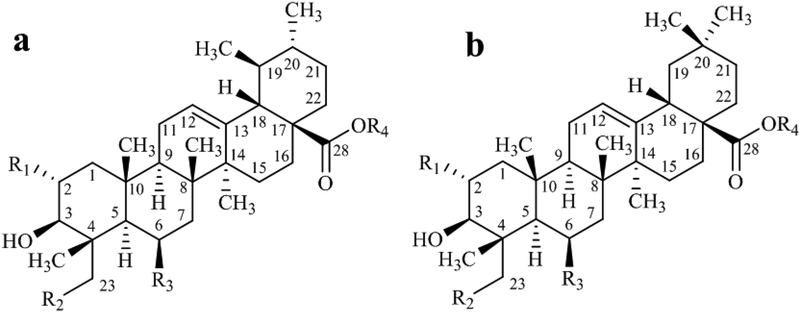
Centella asiatica is most known for its high content of pentacyclic triterpenoids (C30), collectively referred also as “centelloids”. The structural diversity of pentacyclic triterpenoids found in C. asiatica has been reviewed previously (Azerad 2016; James and Dubery 2009). The centelloids can be grouped into two structural sub-types (Figure 3), the ursane and the oleanane series, that differ by the methyl substitution pattern on C-19 and C-20 (Azerad 2016). Glycosylation of the sapogenins usually involves the carboxylic acid group at C-28 (Azerad 2016).
Figure 3.
Structural sub-types of centelloids. a- ursane family and b- oleanane family. R1-R3= OH or H, R4=H or oligosaccharide. Adapted from Azerad (Azerad 2016).
The saponins, asiaticoside and madecassoside, and their aglycones, asiatic acid and madecassic acids, are the most abundant pentacyclic triterpenoids in C.asaitica. Saponins account up to 8% of the dry mass of the herb (James and Dubery 2011). The levels of saponins and sapogenins vary widely depending on the geographical origin, genetic, environmental and growth conditions (see section 2.3). Saponins are synthesized via the isoprenoid pathway. All isoprenoid biosynthetic pathways start with the isomeric 5-carbon precursors, isopentenyl diphosphate and dimethylallyl diphosphate. Assembly of three of these C5 precursors by prenyl transferases yields farnesyl diphosphate (C15). Head-to-head condensation of two molecules farnesyl diphosphate catalyzed by squalene synthase yields squalene (C30). Squalene is converted by squalene epoxidase in the presence of O2 and NADPH to (3S)-squalene 2,3-epoxide (2,3-oxidosqualene), the common precursor of the biosynthetic pathways that lead to sterols and triterpenoids, respectively. In both biosynthetic pathways the cyclization of 2,3-oxidosqualene is facilitated by a family of 2,3-oxidosqualene cyclases. However, dependent on the conformation adopted by 2,3-oxidosqualene during cyclization the dammarenyl cation is formed leading to triterpenoids of C.asiatica via the action of specific 2,3-oxidosqualene cyclase, α-amyrin synthase, while sterols are formed via protosteryl cation and the action of cyclo-artenol synthase (Azerad 2016; Gallego et al. 2014; Mangas et al. 2008; Muller et al. 2013; Tugizimana et al. 2015).
C. asiatica is also rich in chlorogenic acids, a diverse group of compounds formed by quinic acid esterified to cinnamic acid derivatives (Ncube et al. 2016). The enzyme phenylalanine ammonia lyase catalyzes the E2 elimination of ammonia from L-phenylalanine to produce trans-cinnamic acid, which is further transformed to 4-hydroxycinnamic acid (p-coumaric acid) via the P450-dependent enzyme 4-cinnamic acid hydroxylase. Additional enzymatic hydroxylation and methylation reactions yield hydroxycinnamic acid derivatives e.g. caffeic acid with the typical ortho oxygenation pattern. Esterification of hydroxyl groups on L-quinic acid with the carboxyl function of cffeic acid yields caffeoylquinic acids. For instance, the ester of caffeoyl acid involving the 3-hydroxyl position of L-quinic acid yields 3-O-caffeoylquinic acid, also referred as chlorogenic acid. Structural isomers of caffeoyl esters involve other hydroxyl sites on the quinic acid: 4-O-caffeoylquinic acid (4-CQA or cryptochlorogenic acid) and 5-O-caffeoylquinic acid (5-CQA or neochlorogenic acid). Caffeoylquinic acids undergo positional isomerization in aqueous solution (Xie et al. 2011). Thus, in case of extraction protocols based on water or aqueous solvent mixtures the content of the different caffeoylquinic acids may not reflect the de facto contents of the different caffeoylquinic acids in the plant (Xie et al. 2011).
In C. asiatica, the following isomeric dicaffeolyl esters are commonly found: 1,3-dicaffeoylquinic acid, 1,5-dicaffeoylquinic acid, 3,4-dicaffeoylquinic acid,, and 3,5-dicaffeoylquinic acid and 4,5-dicaffeoylquinic acid (Gray et al. 2014). Some dicaffeoylquinic acids are also referred to by the trivial names isochlorogenic acid A, B or C (Table 2). Irbic acid (3,5-O-dicaffeoyl-4-O-malonylquinic acid) is a dicaffeoylquinic acid derivative with a malonyl group on the quinic acid moiety, which has been reported in C. asiatica species (Maulidiani et al. 2014) and cell cultures (Antognoni et al. 2011).
C. asiatica contains many phenolic constituents, including flavonoids, such as catechin, epicatechin, kaempferol, quercetin and related glycosides (Ncube et al. 2016). The biosynthesis of flavonoids combines the shikimate pathway with the acetate pathway using p-cinnamoyl-CoA as the starter unit and three malonyl-CoA units for chain extension. Chalcone synthase forms naringenin-chalcone via a Claisen-type reaction. An intramolecular Michael-type nucleophilic attack yields the A ring of the flavanone, naringenin. Further enzymatic transformation of naringenin leads to a range of flavones (e.g. apigenin) and flavonols, e.g. kaempferol and quercetin (Dewick 2002; Muller et al. 2013; Ncube et al. 2016; Winkel-Shirley 2001). Flavonoids esterified to hydroxycinnamic derivatives, such as castilliferol (kaempferol-3-p-coumarate) and castillicetin (quercetin-3-caffeate), have been isolated from the aqueous methanol extracts of C. asiatica (Subban et al. 2008).
2.2. Contemporary methods for the characterization and fingerprinting of C. asiatica extracts
Traditionally, the analysis of Centella plant extracts was based on thin layer chromatography (Bonfill et al. 2006; Brinkhaus et al. 2000). From ass early as 1996, the characterization and finger printing of Centella plant extracts have been mostly based on HPLC methods (Gunther and Wagner 1996). HPLC with ultraviolet (UV) detection is commonly employed for the characterization and quantification of flavonoids, mono- and dicaffeoylquinic acids and saponins. Due to the lack of strong UV-absorbing chromophores in centellosides, HPLC methods in conjunction with evaporative light scattering detection have been developed for the separation, isolation and characterization of centellosides (Zhang et al. 2008). High-speed countercurrent chromatographic techniques have been described for the efficient isolation of larger amounts of centellosides (Diallo et al. 1991; Du et al. 2004). For the structural identification of natural products, 1- and 2-dimensional Nuclear Magnetic Resonance Spectroscopy (NMR) techniques remain superior. Examples in which NMR techniques were used for the identification of centellosides are available (Antognoni et al. 2011; Subban et al. 2008).
The emergence of chromatographic techniques hyphenated to mass spectrometry (MS) techniques has gained popularity for the analysis of plant extracts in general. Modern mass spectrometry platforms offer accurate mass measurements in combination with collision-induced dissociation techniques for structural analysis and quantification of compounds in complex mixtures. LC-MS/MS-based studies of Centella extracts with detailed descriptions of experimental conditions are available (Gray et al. 2014; Jiang et al. 2016; Maulidiani et al. 2014).
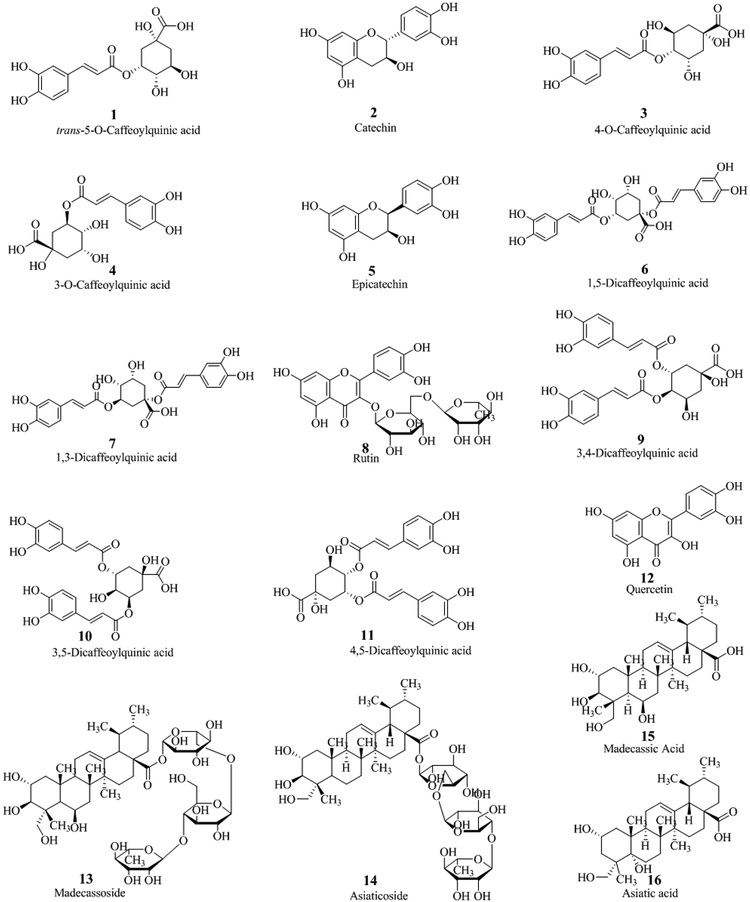
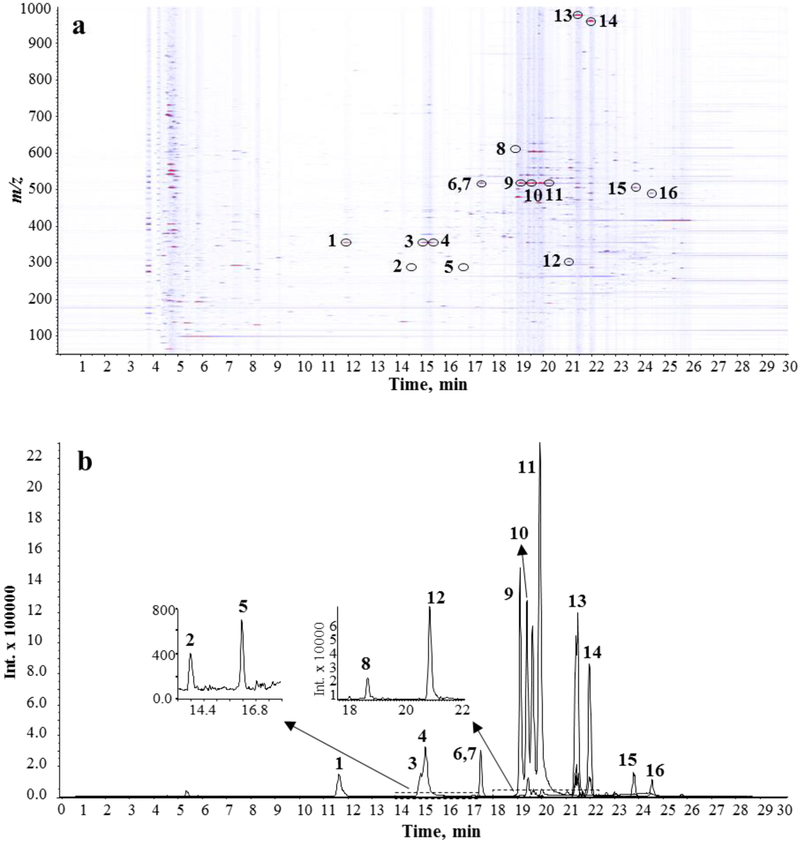
In our laboratory, we favor the use of ultra-performance liquid chromatography (UPLC) in conjunction with accurate mass high-resolution tandem mass spectrometry for the chemical analysis and characterization of plant extracts. For instance, our recent work on the bioactivity of C. asiatica constituents uses aqueous extracts of the aerial parts of the plant obtained after boiling under reflux for two hours (Gray et al. 2014). These aqueous extracts contain high levels of chlorogenic acids and dicaffeoyl acids besides minor levels of pentacyclic triterpenoids and flavonoids. Compounds present in this type of extract are shown in Figure 4 and numbered according to their elution time (experimental conditions described in Figure 5).
Figure 4.
Structures of compounds detected in aqueous extracts of C. asiatica.
Figure 5.
UPLC-MS analysis of an aqueous extract of C. asiatica. 10 μl Injection (5 mg ml−1 of dry mass resuspended in 70% aqueous methanol). a Ion intensity map (of an aqueous extract of C. asiatica. In this 2D visualization, retention time is given on the x-axis, the m/z values are shown on the y-axis, and colors represents signal intensity. b Extracted ion chromatograms for 16 commonly found compounds. [M-H]− (1,3,4: m/z 353.09; 2,5: m/z 289.07; 6,7,9–11: m/z 515.12; 8: m/z 609.15; 12: m/z 301.03; 13: m/z 973.50; 14: m/z 957.51; 15: m/z 503.34; 16: m/z 487.34). Chromatographic separation was conducted using a Shimadzu Nexera UPLC system equipped with Inertsil Phenyl-3 column (150 × 4.6 mm, 5 μm). Mobile phase A was water with 0.1 % formic acid, and mobile phase B was methanol with 0.1% formic acid. The gradient started with 5% B and was held for 1 min, followed by a 10 min linear gradient from 5 % to 30 %. The gradient was then stepped to 100% B at 23 min and held for 12 min and finally, stepped back to 5% B to equilibrate the column. The flow rate was 0.4 mL min−1, and the column temperature was maintained at 45 °C. An AB Sciex Triple TOF 5600 mass spectrometer equipped with a TurboSpray electrospray ionization sourceoperated in the negative ionization mode was used. The instrument was operated in the information-dependent acquisition (IDA) mode using a collision energy of 40 V. Compound identity is based on accurate mass, isotopic pattern, retention time, MS/MS spectra and standard addition using authentic compounds. Structures of compounds 1 to 16 are given in Figure 4.
A typical UPLC-MS chromatogram of an aqueous extract of C. asiatica is shown in Figure 5. Ethanolic extracts of C. asiatica differ markedly from the aqueous extracts in that they contain high levels of asiaticoside and madecassosides, and their corresponding aglycones, asiatic acid and madecassic acids (Figure 6). This example illustrates that standardization and detailed characterization of C. asiatica extracts are necessary to reliably and reproducibly study the biological activity of C. asiatica preparations.
Figure 6.
UPLC-MS chromatogram of an ethanolic extract of C. asiatica. 10 μl Injection (5 mg ml−1 of dry mass resuspended in methanol 70%). Shown is an overlay of extracted ion chromatograms for commonly found compounds. [M-H]− (1,3,4: m/z 353.09; 6,7,9–11: m/z 515.12; 8: m/z 609.15; 12: m/z 301.03; 13: m/z 973.50; 14: m/z 957.51; 15: m/z 503.34; 16: m/z 487.34). Experimental conditions as described in the legend of Figure 5. Structures of compounds 1 to 16 are given in Figure 4.
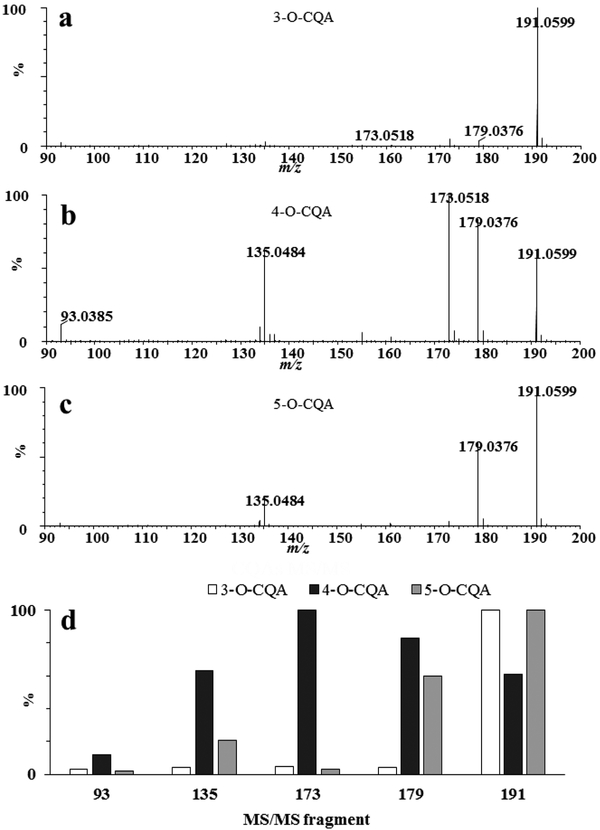
Although isomeric compounds cannot be resolved solely by mass, some of them can be differentiated by their relative intensity of fragment ions in the mass spectra. This is the case for mono-caffeoylquinic acids using collision induced dissociation (CID) fragmentation in the negative ionization mode (Figure 7). Five major products were observed m/z 191.0599, [quinic acid - H]−; m/z 179.0376, [caffeic acid - H]−; m/z 173.0518, [quinic acid -H2O - H]−; m/z 135.0484 [caffeic acid-CO2- H]−; and m/z 93.0385 [phenol - H]−. As described by Xie et al. (2011), 3-O-CQA can be distinguished from 4-O-CQA by its base peak with m/z 191.0599, whereas the base peak of 4-O-CQA appears at m/z 173.0518 (Figure 7). Similarly, Clifford et al. showed that dicaffeoyl quinic acid isomers can be discriminated using CID fragmentation (Clifford et al. 2005). In cases where isomers cannot be distinguished by their fragmentation behavior, their chromatographic retention behavior may allow discriminating between isomers. For instance, the structural isomers of isochlorogenic acids have the same accurate mass and elemental composition but differ in their retention behavior during reversed-phase chromatography (Compounds 9, 10, 11 in Figures 5 and 6).
Figure 7.
Mass spectra of isomeric mono-caffeoylquinic acids recorded in negative ionization mode showing differences in the intensity of MS fragment ions. Standards were injected by infusion (10 μl min−1 in methanol:water 70:30 V/V, 1 mg L−1). A Synapt G2 HDMS (Waters Corp., MA, USA) was used for ion detection. The cone voltage was 20 V and the transfer energy was set at 25 eV. a 3-O-caffeoylquinic acid, b 4-O-caffeoylquinic acid, c 5-O-caffeoylquinic acid, d Intensity comparison for major MS/MS product ions of CQAs. Data are in accord with Xie et al., 2011.
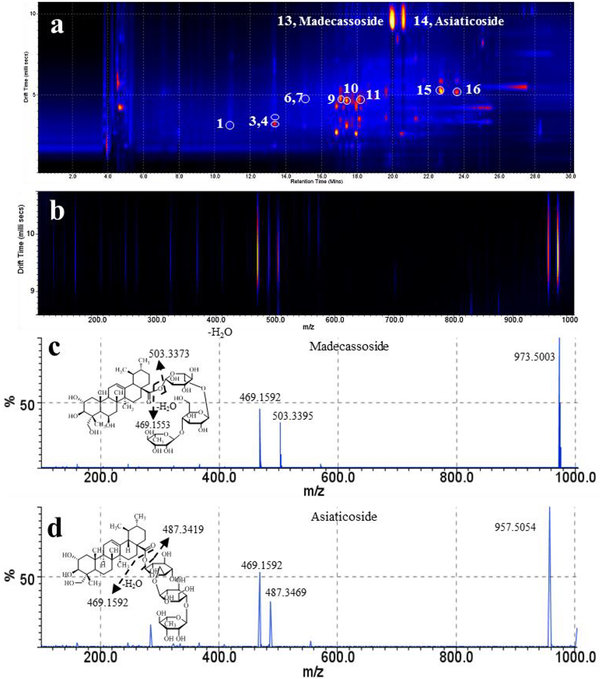
Ion mobility spectrometry (IMS) in conjunction with mass spectrometry has emerged as an additional tool for the detection and characterization of secondary metabolites in plant extracts and tissues. The addition of ion mobility spectrometry increases peak capacity on a millisecond timescale and provides insight into gas-phase ion structures. In the drift region of the ion mobility device, an ion moves according to its charge, size, shape and average collisional cross-section (between the ion and a background gas). In our laboratory we use travelling wave ion mobility spectrometry (TWIMS) coupled to a high resolution time-of-flight (TOF) analyzer, a Waters SYNAPT G2 HDMS (Waters Corp., MA, USA), for the analysis of complex biological matrices. This instrument configuration features a TWIMS cell between two collision cells, the T-wave trap (pre-IMS) and transfer cell (post-IMS). For the detection and characterization of secondary metabolites in aqueous extracts of C.asiatica we use UPLC in conjunction with electrospray ionization and the MSE acquisition mode. The MSE acquisition mode is a data independent acquisition technique with provides exact mass information by working in the low-energy regime (precursor MS) and fragment ion information in the high-energy regime for every detectable analyte. Figure 8 shows a two-dimensional map visualizing drift time versus retention time of an aqueous extract of C. asiatica. The combination of UPLC separation, nested gas phase mobility separation of ions and accurate mass measurement allows comprehensive detection of the constituents within a C. asiatica extract. In addition, fragmentation post ion mobility separation provides fragment ion information. In this mode, ions are separated in the T-wave ion mobility cell and subsequently subjected to collision-induced fragmentation in the transfer region. Applying collision energy to the transfer device (collision energy ramp from 20 to 65 V) causes fragmentation of the mobility-separated precursor ions. Because the fragment ions preserve their velocity of the precursor ion, the fragment ions align with the drift times of the precursor ions. As an example of the use of this technique, asiaticoside ([M-H]− m/z 957.5054) and madecassoside ([M-H]− m/z 973.5003), both present at low levels in the aqueous extract of C. asiatica (Gray et al. 2014), were subjected to fragmentation in the transfer region by working in the high energy regime. Both compounds produce their respective deprotonated sapogenins [madecassic acid - H]−(m/z 503.3373) and [asiatic acid - H]− (m/z 487.3419). In addition, both saponins generate a fragment ion with m/z 469.1592 attributed to the deprotonated oligosaccharide moiety. Drift time distributions for the fragment ions and precursor ions, proposed transfer dissociation, and fragmentation spectra are shown in Figure 8.
Figure 8.
ESI q-IMS-MS/MS analysis of an aqueous extract of C. asiatica (5 mg ml−1 of dry mass resuspended in methanol 70%, 10 μl injection). a, 2D map visualization of drift time versus retention time, b, drift time distribution of ions, c and d, fragmentation spectrum and proposed transfer dissociation of madecassoside ([M-H]− m/z 973.5) and asiaticoside ([M-H]− m/z 957.5), respectively. Method: Acquity UPLC chromatographic conditions were as described in Figure 5. The LC system was coupled to a Synapt G2 HDMS (Waters Corp., MA, USA) used for detection in negative ion electrospray mode. Nitrogen was used as carrier gas for ion mobility experiments. Data acquisition range was m/z 50–1200. The cone voltage was 20 V. The T-wave ion mobility cell was operated at 800 m s−1 and the wave amplitude was set at 35 V. Helium and nitrogen IMS carrier were both set at 80 ml min−1.
In depth characterization and finger printing studies of C. asiatica extracts and preparation have yet to become available; however, applications of IMS-MS focusing on the characterization of isomeric compounds that are also found in C. asiatica are available. For example, IMS-MS/MS studies have been reported for the elucidation of structural isomers of chlorogenic acids (Shinomol et al. 2011) and carotenoids (Dong et al. 2010). The combination of ion mobility spectrometry and high resolution mass spectrometry adds selectivity and specificity to the analysis of plant metabolites by increasing the separation space for complex plant extracts and matrices (Pacini et al. 2015; Stark et al. 2013) enabling the separation of isomeric metabolites and improving structure elucidation of complex natural products. The availability of commercial instruments that combine ion mobility separation with mass spectrometry will likely advance the characterization and standardization efforts of plant extract and traditional herbal medicine preparations.
Emerging analytical trends include the application of mass spectrometry-driven metabolomic studies of extracts of Centella species and cell cultures after perturbations. For instance, gas chromatography (GC)-MS and LC-MS-based chemometric studies of extracts of C. asiatica cultures have been used to investigate the effect of methyl jasmonate on metabolite composition (Tugizimana et al. 2015). Similarly, a LC-MS/MS-driven method in combination with multivariate data analysis has recently been employed for determining the effect of salicylic acid on the profile of chlorogenic acids in C. asiatica cultures (Ncube et al. 2016). The advent of commercially available IMS-MS systems has led to exciting applications in the field of plant metabolomics (Maldini et al. 2015; Zhang et al. 2014); however, IMSMS-based studies of C. asiatica preparations or cell cultures are lacking.
2.3. Chemical variation in Centella asiatica
The increasing commercial importance of C. asiatica as a source of bioactive extracts and isolated triterpenoids has led to a search for elite chemotypic strains of the herb, as well as means of optimizing the yield of the active compounds from a given strain. There have been several studies in different growing regions of C. asiatica comparing the levels of the triterpenoid and phenolic compounds in multiple accessions of the herb. In eleven C. asiatica accessions collected from the Nilgiri hill region in India, wide variations were seen in the content of asiatic acid (0.04 to 0.58%), asiaticoside (0.11 to 1.56%) and madecassoside (0.29 to 6.09%) (Srivastava et al. 2014). A comparison of 14 phenotypically distinct C. asiatica accessions collected from an unspecified region of India again showed wide variability in these triterpenes as well as madecassic acid, with total content of these four compounds varying from 0.26 to 11 mg/g fresh herb weight (Singh et al. 2015). Twelve C. asiatica accessions collected from multiple regions of South Africa had asiaticoside levels ranging from 0.75% to 2.06% dry weight (Long et al. 2012). A study in Madagascar compared triterpene levels in two C. asiatica morphotypes, reniform leaves (type A; n=32 collection sites) and round leaves (type B; n= 13 collection sites), from the Eastern and Western regions of the island respectively (Rahajanirina et al. 2012). Type A plants were found to have significantly higher levels of asiaticoside (3.7% vs 2%) and madecassoside (3.6% vs 2%) than the Type B plants, although aglycone levels did not differ significantly. In addition to the triterpenoids, large differences in total flavonoid content (18, 30 and 144 mg/g) have been observed in three accessions from the Assam region of India, although antioxidant activity and total phenolics did not differ as widely (Upadhyaya and Saikia 2012).
Large seasonal variations have been documented in levels of triterpenes, chlorogenic acids and the flavonoid kaempferol in C. asiatica plants growing in Australia (Alqahtani et al. 2015), in agreement with seasonal effects on triterpenes reported in Madagascar (Rahajanirina et al. 2012). Light and drying conditions has also been shown to affect triterpene levels (Srithongkul et al. 2011). Plants grown with full daylight exposure had higher levels of triterpenes, flavonoids chlorogenic acids and radical scavenging activity than those grown in 50% shade (Maulidiani et al. 2012). Plants dried at lower temperatures had higher yields of triterpene saponins than those dried in higher temperatures (Plengmuankhae and Tantitadapitak 2015).
Comparison of C. asiatica accessions obtained from diverse parts of India including the Andaman Islands, and propagated under identical conditions, has shown that the triterpenoid saponin content is a highly heritable trait (Lal et al. 2017), and elite strains can be identified and cultivated (Thomas et al. 2010). Zhang et al. (2009) have compared LC-UV fingerprints of extracts from 14 populations of C. asiatica collected from different regions in China (Zhang et al. 2009). Hierarchical clustering analysis of 12 common peaks (of which only three were identified as asiatic acid, asiaticoside and madecassoside) from plant extracts showed the existence of four distinct chemotypes of C. asiatica. A subsequent study in 2012 examined inter-simple sequence repeat (ISSR) markers of 162 individuals from these 14 populations, and found high genetic diversity across the species, with greater diversity in populations from some regions than others(Zhang et al. 2012b). Interestingly, the 14 populations clustered into four genetic groups which were similar to the four chemical groups found by chemical clustering in the earlier study (Zhang et al. 2009)
These studies demonstrate that genetic and geographical factors as well as cultivation, harvesting and processing practices all influence the chemical profile of C. asiatica. While this has been best researched for the triterpene compounds, the other potentially active components of C. asiatica, such as flavonoids and chlorogenic acid derivatives, are also clearly influenced by these factors. These reports highlight the importance of evaluating the chemical characteristics of C. asiatica material used in scientific studies, as well as clear reporting in publications of the geographical source, method of preparation and chemical features of the materials studied. This would not only ensure that the materials tested contained active compounds, but also allow for better comparison between studies.
3. Neurological activities of Centella asiatica and its constituents
3.1. Anxiolytic, anti-convulsant, anti-ischemic injury effects of C. asiatica
C. asiatica has many reported beneficial neurobehavioral effects. Its anxiolytic properties have been demonstrated in healthy rodents (Wijeweera et al. 2006), as well as chronically stressed mice and mice with sleep deprivation-induced anxiety (Chanana and Kumar 2016; Wanasuntronwong et al. 2012). In the sleep-deprived mice, C. asiatica treatment attenuated oxidative damage and decreased anxiety. The effect of the plant was enhanced when co-administered with nitric oxide antagonists, and decreased with nitric oxide agonists suggesting the possible involvement of nitric oxide modulating mechanisms in the anxiolytic effects (Chanana and Kumar 2016).
While early studies suggested the anxiolytic effects of C. asiatica may be related to more general sedative effects of the plant (Sakina and Dandiya 1990), recent studies have failed to confirm this (Wijeweera et al. 2006). Instead, new evidence suggests its anxiolytic effects may due to effects on the GABA system. Aqueous and ethanolic extracts of C. asiatica have been shown to substantially increase the activity of glutamate decarboxylase, a key enzyme in GABA synthesis (Awad et al. 2007). Since GABA is an inhibitory neurotransmitter, these effects on GABA synthesis may also contribute to C. asiatica’s anti-convulsant properties.
C. asiatica treatment has been shown to be protective in rodent models of seizures (Gupta et al. 2003; Sakina and Dandiya 1990). It has been suggested that effects on the acetylcholine system could mediate its anti-convulsant activity. In a rat seizure model, butanol, ethyl acetate, and hexane extracts of C. asiatica all normalized acetylcholine levels and acetylcholinesterase activity in the brains of treated animals (Visweswari et al. 2010).
The neuroprotective effects of C. asiatica have also been demonstrated in rodent models of stroke. Oral treatment with an ethanol extract of the plant decreased infarct volume and improved neurobehavioral activity resulting from middle cerebral artery occlusion in rats (Tabassum et al. 2013). In this case, the neuroprotective effects were credited to the antioxidant effects of the extract, as the improvements were accompanied by decreased levels of reactive oxygen species (ROS) and markers of lipid peroxidation, and increased activity of several antioxidant enzymes. This finding was later confirmed by Doknark et al. (2014) who found that oral treatment with an ethanolic extract improved behavioral impairments and significantly reduced markers of oxidative damage in the brain (Doknark et al. 2014).
3.2. Cognitive enhancing effects of C. asiatica
The cognitive enhancing properties of the plant have long-been recognized in the Ayurvedic tradition (Shinomol et al. 2011). Over the past decade, numerous preclinical studies have confirmed these effects, both in the context of pathological and normal, age-related, cognitive decline.
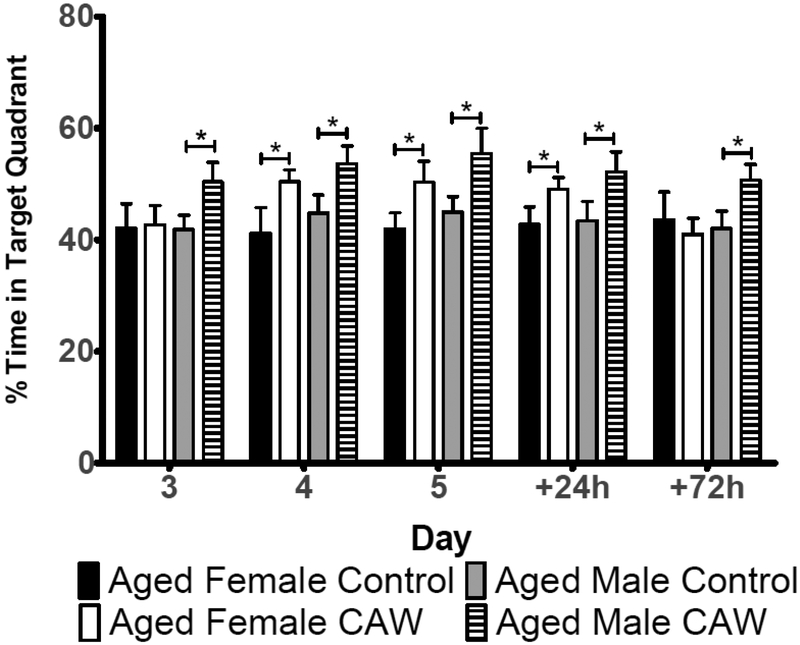
Aqueous extracts of C. asiatica seem particularly effective at improving cognitive performance. Kumar et al. (2011) showed that an aqueous extract of C. asiatica improved performance in the Morris Water Maze and the elevated plus maze in the D-galactose model of brain aging (Kumar et al. 2011). This improvement was associated with a normalization of acetylcholinesterase activity in the brains of treated animals. Our own lab has also demonstrated that a water extract of C. asiatica can improve memory retention in otherwise healthy, aged mice (Gray et al. 2016). In the probe phase of the Morris Water Maze that assesses memory retention, we found that aged, C. asiatica-treated male and female mice spent significantly more time in the quadrant that formerly held the platform than did their control, untreated counterparts (Figure 9). Interestingly, this improvement persisted for 24 hours in the females and 72 hours in the males after the final day of hidden platform testing. The enhanced memory retention was accompanied by increased expression of the synaptic genes synaptophysin and post-synaptic density protein 95 (PSD95) in the brains of these animals (Gray et al. 2016).
Figure 9.
Morris water maze probe data of healthy aged male and female mice treated with a water extract of Centella asiatica (CAW, 2 mg/mL dissolved in the drinking water) for 2 weeks prior to, and during testing. CAW increased memory retention in both male and female healthy aged mice. Extract treated animals spent significantly more time in the target quadrant than control, untreated mice. (Adapted from Gray et al. J. Ethnopharmacology 2016)
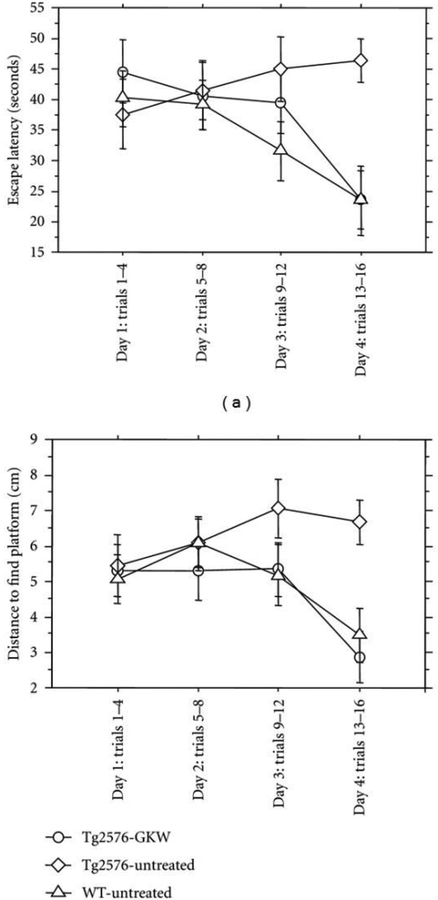
Extracts of C. asiatica have also been shown to have beneficial effects for cognitive dysfunction observed in models of Alzheimer’s disease (AD). Kumar et al. (2009) demonstrated that a water extract of C. asiatica reversed colchicine-induced deficits in elevated plus maze and the Morris Water Maze in rats (Kumar et al. 2009). This is consistent with findings from our own lab where treatment with the water extract of C. asiatica attenuated behavioral deficits normally observed in the Tg2576 mouse model of Aβ-toxicity (Soumyanath et al. 2012). We found that two weeks of treatment with the extract normalized time and distance traveled to find the hidden platform in the Morris Water Maze to that of age-matched wild-type animals (Figure 10).
Figure 10.
Morris water maze data of aged female Tg 2576 transgenic or wild type (WT) mice treated with a water extract of Centella asiatica (GKW, 2 mg/mL dissolved in the drinking water) for 2 weeks prior to, and during testing. C.asiatica treatment normalized memory deficits in the Tg2576 mouse model of Aβ accumulation, reducing the time and distance traveled by Tg2576 mice to find the hidden platform compared to untreated Tg2576 animals. (adapted from Soumyanath et al. 2012).
The neuroprotective effects of C. asiatica in the context of beta-amyloid (Aβ) toxicity have been well documented in vitro. In both PC12 and N2A neuroblastoma cells, as well as in primary rat cortical neurons, extracts of C. asiatica prevented Aβ induced cell death (Chen et al. 2015; Xu et al. 2008). Our lab has demonstrated that an aqueous extract of C. asiatica can protect MC65 and SH-SY5Y neuroblastoma cells from Aβ-induced cytotoxicity (Gray et al. 2014; Soumyanath et al. 2012) and that this protection is accompanied by a decrease in ROS and a normalization of mitochondrial function and calcium homeostasis (Gray et al. 2015). Similarly, in PC12 neuroblastoma cells, this protective effect of an ethanol extract of C. asiatica was attributed to inhibition of the production of ROS (Chen et al. 2015), while in N2A cells and cortical neurons the protective effect of the C. asiatica extract was thought to be related to enhanced phosphorylation of CREB (cyclic AMP response element binding protein) possibly through ERK (extracellular signal-related kinase) pathway (Xu et al. 2008).
3.3. Effects of C. asiatica on neuronal health
Impaired arborization and decreased synaptic density are two endpoints widely believed to represent the structural basis of cognitive dysfunction. Therefore, the cognitive enhancing effects of C. asiatica could result from its effects on neuronal differentiation and dendritic arborization. Extracts of C. asiatica have demonstrated neuritogenic properties in various neuroblastoma cell lines (Soumyanath et al. 2005; Wanakhachornkrai et al. 2013) and Mohandas Rao’s group has shown that a fresh leaf extract of C. asiatica can enhance dendritic arborization in the hippocampus and amygdala for both neonatal and adult rats (Gadahad et al. 2008; Mohandas Rao et al. 2006; Mohandas Rao et al. 2009; Mohandas Rao et al. 2012).
3.4. Potential mechanisms of C. asiatica associated with neuroprotection and cognitive enhancement
Although the molecular mechanism by which C. asiatica exerts its neuroprotective and cognitive enhancing effects is still not fully understood, there are several emerging hypotheses. Normalization of acetylcholinesterase activity has been suggested (Kumar et al. 2011) as have modulations of signal transduction pathways that regulate arborization and synaptogenesis, like ERK1/2 and Akt (Wanakhachornkrai et al. 2013). Another potential candidate mechanism is through inhibition of phospholipase A2 (PLA2). Generation of arachidonic acid by PLA2 is the rate-limiting step in the production of the eicosanoids that play critical roles in the modulation of inflammation and oxidative stress. Increased inflammation, oxidative stress, altered phospholipid metabolism, and increased PLA2 activity are common features of many neurological disorders including Alzheimer’s disease, multiple sclerosis, ischemia, epilepsy and spinal cord injury (Farooqui 2006). The water extract of C. asiatica has been shown to inhibit multiple PLA2 isoforms in isolated neurons and the brains of treated animals (Barbosa 2008; Defillipo 2012).
In fact, there is growing evidence that antioxidant effects of C. asiatica, along with its effects on mitochondrial function, may play an important role in the neuroprotective and cognitive enhancing effects of the plant. This antioxidant activity may contribute to the protection conferred by C. asiatica against Aβ toxicity. In PC12 and IMR32 neuroblastoma cells an ethanol extract of C. asiatica reduced Aβ-induced increases in reactive oxygen species and restored the activity of several antioxidant enzymes that were impaired in response to Aβ (Chen et al. 2015).
We have demonstrated in SH-SY5Y neuroblastoma cells treated with exogenous Aβ as well as in MC65 neuroblastoma cells that overexpress amyloid precursor protein that an aqueous extract of C. asiatica can reverse the increase in reactive oxygen species observed in response to Aβ (Gray et al. 2015). We found that this was accompanied by an increase in the mRNA expression of nuclear factor (erythroid-derived 2)-like 2 (NRF2) (also called NFE2L2) and its target antioxidant response genes. NRF2 is a transcription factor that regulates the endogenous antioxidant response pathway. The endogenous antioxidant response pathway protects cells from oxidative stress (Motohashi and Yamamoto 2004) by increasing transcription of cytoprotective genes (Itoh et al. 1997) through the binding of the transcription factor transcription factor Nrf2 to antioxidant response elements (AREs) in antioxidant gene promoters.
The increased mRNA expression of NRF2 and its target genes suggests that C. asiatica can in fact activate the endogenous antioxidant response pathway; however, the exact mechanism by which it does so remains to be elucidated. Further evidence of the antioxidant effects of C. asiatica were also observed in vivo following oral administration of an extract of the plant. In the PSAPP mouse model of Aβ accumulation, treatment with the extract for eight months resulted in decreased lipid peroxidation and oxidative damage to DNA in the hippocampus of treated animals (Dhanasekaran et al. 2009). Similar in vivo antioxidant effects have been observed in models of healthy aging as well. In the D-galactose model of brain aging, treatment with the aqueous extract of C. asiatica decreased lipid peroxidation and restored antioxidant enzyme activity (Kumar et al. 2011). This is consistent with our finding that an aqueous extract of C. asiatica increased the expression of NRF2 and its target genes in the brains of healthy, aged mice (Gray et al. 2016).
These antioxidant properties are also thought to contribute to the neuroprotective effects of C. asiatica in other neurodegenerative conditions beyond Alzheimer’s disease. Huntington’s disease, multiple sclerosis, Parkinson’s disease and heavy metal exposure share the common features of increased ROS, diminished ATP production and impaired mitochondrial respiration, resulting in synaptic degradation and cognitive impairment (Arun et al. 2016; Conley et al. 2000; Du et al. 2010; Hensley et al. 1994; Navarro and Boveris 2007; Rhein et al. 2009). The antioxidant and mitochondrial effects of C. asiatica have been observed in many of these contexts as well. In a rodent model of experimentally induced Parkinsons, treatment with an aqueous extract of C. asiatica decreased oxidative damage to lipids and proteins in the brains of rats treated with the mitochondrial toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) and restored levels of antioxidant enzymes (Haleagrahara and Ponnusamy 2010). In a 3- nitropropionic acid (NPA) induced model of Huntington’s disease, mice fed C. asiatica leaf powder showed lower brain levels of lipid peroxidation, protein carbonyls and ROS, as well as an increase in antioxidant enzyme activity (Shinomol and Muralidhara. 2008b), and decreased oxidative stress in isolated brain mitochondria treated with 3-NPA (Shinomol and Muralidhara. 2008a). C. asiatica also protected against brain mitochondrial swelling and partially restored electron transport chain (ETC) enzyme activity in rats (Shinomol and Muralidhara. 2008b).
Similar antioxidant and mitoprotective effects of C. asiatica have been observed in response to other neurotoxic insults. C. asiatica treatment has been shown to reverse impaired brain mitochondrial enzyme activity, reduce lipid peroxidation and restore antioxidant enzyme activity following exposure to arsenic, aluminum and D-galactose (Flora and Gupta 2007; Kumar et al. 2011; Prakash and Kumar 2013). Additionally, our lab has observed similar mitochondrial effects of an aqueous extract of C. asiatica in neuroblastoma cells exposed to Aβ. We found C. asiatica attenuated decreases in ATP production and diminished mitochondrial bioenergetics resulting from Aβ exposure. We also found that C. asiatica increased the expression of genes encoding enzymes in the ETC in cells exposed to Aβ as well as in healthy cells, suggesting a possible effect on mitochondrial biogenesis (Gray et al. 2015).
3.5. Neuroactive effects of constituent compounds from C. asiatica
Because extracts of C. asiatica are complex mixtures made up of diverse compounds, there is growing interest in identifying compounds within C. asiatica that may be responsible for the beneficial effects of entire plant extracts. While it is likely that other compounds will be identified that mediate the positive effects of C. asiatica, there is an increasing body of literature suggesting that triterpenes and caffeoylquinic acids (CQAs) found within C. asiatica might play an important role in the neuroprotective and cognitive enhancing properties of the plant.
3.5.1. Triterpenes
The triterpenes found within C. asiatica have been shown to have similar neuroactive and neuroprotective effects to the entire plant extract in many of the same experimental contexts. For instance, both asiatic acid and asiaticoside have been shown to have anxiolytic properties in rat models (Ceremuga et al. 2015; Wijeweera et al. 2006) without exhibiting sedative effects (Nasir et al. 2012; Wijeweera et al. 2006), as was seen with the C. asiatica extracts (Chanana and Kumar 2016; Wanasuntronwong et al. 2012). Asiatic acid, asiaticoside and madecassoside also have neuroprotective effects in models of stroke showing anti-inflammatory effects including reduced cytokine levels and microglial activation (Chen et al. 2014; Krishnamurthy et al. 2009; Luo et al. 2014). Both madecassoside and asiatic acid also reduced infarct volume and attenuated the neurological deficits associated with the ischemia (Krishnamurthy et al. 2009; Luo et al. 2014).
Asiatic acid and asiaticoside have also been shown to have neuroprotective properties against neurotoxic insults. In rat primary cortical neurons, asiatic acid protected the cells against C2 ceramide-induced apoptosis while asiaticoside was protective against ischemia-induced cell death (Sun et al. 2015; Zhang et al. 2012a). There is conflicting literature about whether these compounds can protect against Aβ toxicity. There are reports that both asiatic acid and asiaticoside are protective against Aβ-induced cytotoxicity in glial cells (Jew et al. 2000; Mook-Jung et al. 1999) yet in our lab none of the triterpenes found in C. asisatica showed any protective effect in MC65 cells against Aβ-induced cytotoxicity (Gray et al. 2014) Asiatic acid, asiaticoside and madecassoside also exert cognitive enhancing properties in various models of cognitive impairment. Asiaticoside improved learning and memory deficits caused by ischemia reperfusion (Chen et al. 2014), as well as those related to aging in the senescence-accelerated prone mouse strain (SAMP8) mouse model of accelerated senescence (Lin et al. 2013). In both cases, the beneficial effects of AS were thought to be related to effects of kinase signaling pathways, decreasing p38MAPK activation in the stroke model, and increasing PKA and calmodulin-dependent protein kinase (CAMK) activation in the aged mice. Madecassoside also inhibited the p38MAPK signaling pathway and attenuated cognitive impairment in the D-galactose model of brain aging (Lin et al. 2014). Both asiaticoside and madecassoside have been shown to increase expression of proteins related to synaptic plasticity (Lin et al. 2013; Lin et al. 2014).
Asiatic acid improved both valproic acid (VPA)- and glutamate- induced cognitive impairment in mice (Umka Welbat et al. 2016; Xu et al. 2012). In the VPA-treated animals this effect was attributed to reduced hippocampal histone deacetylase activity, while in the glutamate-treated mice the cognitive improvement was thought to be related to antioxidant activities of asiatic acid. Asiatic acid has also been shown to enhance cognitive performance in healthy rodents, improving learning and memory and even increasing cell proliferation in the hippocampus of adult rats (Nasir et al. 2011; Sirichoat et al. 2015). The increased proliferation was accompanied by increased Notch1 protein levels (Sirichoat et al. 2015). Asiatic acid has also been shown to be neuroprotective in the context of spinal cord injury where treatment resulted in increased expression of Nrf2 and its target genes along with decreased levels of reactive oxygen species and a more robust physical recovery (Jiang et al. 2016).
3.5.2. Caffeoylquinic acids (CQAs)
CQAs also possess many of the same neuroprotective properties that are seen with C. asiatica extracts against a variety of toxic insults. The mono-CQA chlorogenic acid has been shown to protect primary mouse cortical neurons from glutamate excitotoxicity, possibly through its ability to normalize calcium homeostasis in these cells (Mikami and Yamazawa 2015). The diCQAs 3,4-diCQA, 3,5-diCQA and 3,5-diCQA methyl ester were similarly able to prevent glutamate-induced increases in calcium, reactive oxygen species and protect against cell death in rat cortical neurons (Kim et al. 2012a). The CQA derivative 1,5-O-dicaffeoyl-3-O-[4-malic acid methyl ester]-quinic acid has also been shown to be neuroprotective in SHSY5Y neuroblastoma cells treated with NMDA. This compound inhibited NDMA-induced phosphorylation of ERK1/2, p38 MAPK and JNK1/2m while also restoring the activation of CREB, AKT and GSK3B that had been impaired by the treatment (Tian et al. 2015).
Our lab has shown that diCQAs can protect against Aβ-induced cytotoxicity. Both 1,5-diCQA and 3,5diCQA potently protected both MC65 and SH-SY5Y neuroblastoma cells from Aβ-induced cell death and induced the expression of NRF2 and its target genes. 3,5diCQA also enhanced both basal and maximal mitochondrial respiration and significantly increased the expression of genes encoding enzymes in the electron transport chain suggesting a possible effect on mitochondrial biogenesis (Gray et al. 2015).
CQAs and their metabolites can also exert cognitive enhancing effects in many of the same model systems where treatment with C. asiatica was beneficial. Caffeic acid, for instance, reduced infarct volume, attenuated neurological deficits and improved working, spatial and long-term aversive memory deficits in a mouse model of stroke (Pinheiro Fernandes et al. 2014). The same compound was found to be protective in the context of global cerebral ischemia-reperfusion injury in rats where caffeic acid treated animals had diminished memory deficits, decreased pro-inflammatory markers and lipid peroxidation, increased antioxidant enzyme activity and reduced hippocampal cell injury and death. In this model, caffeic acid also reduced 5-lipoxygenase expression leading to the hypothesis that the neuroprotective effects may have resulted from the interaction with that enzyme, which metabolizes arachidonic acid into the pro-inflammatory leukotrienes (Liang et al. 2015).
CQAs have also been shown to improve age-related cognitive impairment. In the SAMP8 mouse model of aging both a CQA-rich purple sweet potato extract and the individual compound 3,5-diCQA improved learning and memory deficits. In both cases this was accompanied by an increased expression of glucose metabolism enzymes (Han et al. 2010; Sasaki et al. 2013). The cognitive enhancing properties of CQAs have been observed in many rodent models of Alzheimer’s disease as well. Treatment with caffeic acid or a diet rich in caffeic acid improved memory in the Tg2576 mouse model of Aβ accumulation as well as in Aβ25–35 injected mice and Aβ1–40 injected rats. These cognitive improvements were associated with increased synaptophysin expression and reduced markers of inflammation, oxidative damage and p38MAPK activity (Kim et al. 2012a; Subash et al. 2015; Wang et al. 2016). Caffeic acid also restored streptozotocin-induced memory deficits in rats and acetylcholinesterase activity (Deshmukh et al. 2016). The mono-CQAs chlorogenic acid and 3-O-caffeoylquinic acid improved memory in scopolamine and Aβ1–42 treated mice respectively. Both compounds decreased lipid peroxidation in the brains of treated animals and chlorogenic acid treatment also inhibited acetylcholinestserase activity in the hippocampus and cortex of the animals (Choi et al. 2014; Kwon et al. 2010).
3.6. Neurological effects of Centella asiatica in humans
3.6.1. Cognition
Centella asiatica has been used in traditional systems of medicine for hundreds of years to treat a multiplicity of conditions including infectious diseases, insomnia, hypertension, asthma, infertility and dementia (Brinkhaus et al. 2000). Practitioners of traditional medicine use local C. asiatica plants prepared in a variety of ways, introducing processing-induced variation in the active constituents to the genetic and geographical factors noted previously (section 2.3). These variations make it challenging to derive direct correlations between scientific studies and traditional usage.
While there is historical usage and extensive preclinical literature on C. asiatica’s neurological properties, rigorous human trials are lacking. Our literature search revealed less than ten human studies, focusing on neurological disorders (Table 4). Each of these studies reported improvements in cognition (Appa Rao et al. 1973; Dev et al. 2009; Farhana et al. 2016; Rakesh et al. 2013; Tiwari et al. 2008; Wattanathorn et al. 2008), mood (Bradwejn et al. 2000; Jana et al. 2010; Wattanathorn et al. 2008) and/or quality of life (Mato et al. 2011; Tiwari et al. 2008) with C. asiatica supplementation. One of the studies noted differences in cognitive outcomes based upon gender where the males had improvements in three out of eight cognitive measures (long-term retrieval, visual spatial thinking and speed processing), whereas females had improvements in five measures (long-term retrieval, short-term memory, working memory, executive process and delayed recall) (Dev et al. 2009). In two separate clinical trials included in Table 4, the subject population was primarily female. Given the observed gender differences by Dev et al., 2009, incorporation of adequate subjects from both genders is warranted in future studies.
Unfortunately, the studies reviewed used products with a variety of preparation methods and some lack an adequate description of the product used, and/or incorporate no means of standardization, thereby limiting the ability to compare between studies. In addition, the studies were of variable quality and relevance. Some did not include a placebo or control group, and some assessed cognitive changes in a cognitively healthy elderly or middle-aged subject population, limiting clinical applicability to states of cognitive impairment (Table 4). Despite these issues, the overall positive effect of the herb as a whole is encouraging. The absence of reports of adverse effects in any of these human studies is also reassuring, and corroborates the classification of C. asiatica as a Class 1 herb (one that can safely be consumed when used appropriately) in the Botanical Safety Handbook (1997).
Clearly the extensive preclinical data and the positive results seen in the limited human studies so far warrants more rigorous human trials. Ideally, these would investigate the effects of an authenticated and chemically well-characterized C. asiatica products, that have been standardized to known active compounds and are administered at doses based on interspecies scaling from preclinical models. Clinical studies would optimally include a control group, adequate subjects from both genders and the appropriate study population for the outcome of interest.
3.6.2. Microcirculation
A separate series of human trials of Centella asiatica have focused on its dermatologic and vascular applications (Brinkhaus et al. 2000). Several placebo-controlled, randomized clinical studies demonstrated the beneficial effects of C. asiatica on peripheral microcirculation using a preparation known as TTFCA. TTFCA contains 40% asiaticoside and 60% combined asiatic acid and madecassic acid (Grimaldi et al. 1990). Doses of 60–180mg of TTFCA daily for 4–24 weeks improved microcirculation and decreased associated symptoms such as edema in hypertensive microangiopathy (Cesarone et al. 2001; Incandela et al. 2001). In addition, 60 mg of TTFCA twice daily for six months increased capillary permeability and microcirculatory parameters such as resting skin flux, venoarteriolar response, transcutaneous oxygen and carbon dioxide and rate of ankle swelling) in diabetic patients (Belcaro et al. 2011; Cesarone et al. 2001). Further studies have found that TTFCA protects the venous endothelium from oxidative damage (Cesarone et al. 2001).
While these studies examined Centella’s triterpenes’ effects on the peripheral system via endothelial restoration and anti-oxidation, the brain also possesses a complex microvasculature communicating primarily via endothelial tight junctions (Drewes 2001). To date, there have been no studies on the effects of TTFCA or other C. asiatica products on brain perfusion; however, there have been reports of brain bioavailability of the triterpenes, asiatic acid and madecassic acid in mouse models (Yin et al. 2012). Based upon this, it can be hypothesized that C. asiatica may affect the microvasculature within the brain, but this aspect requires further investigation.
4. Absorption, distribution, metabolism and elimination (ADME) of active compoundsof C. asiatica
The complex composition of botanical materials renders ADME studies of their multiple biologically active compounds in humans or animal models particularly challenging. To date, in vivo ADME studies of C. asiatica have primarily focused on the Centella-specific triterpenoids asiaticoside, madecassoside and their aglycones asiatic acid and madecassic acid. No reports were found of studies examining the ADME of caffeoylquinic acids (CQAs) or flavonoids after administration of C. asiatica; however, there is literature on the ADME of these latter groups of compounds derived from other botanical sources.
4.1. Triterpenes
Using sensitive methodologies, such as high-performance liquid chromatography (HPLC) coupled to ultraviolet detection (LC-UV) or tandem mass spectrometry (LC-MSn), many have been able to detect and quantify triterpenoids and their metabolites within human and animal biological matrices (Han et al. 2012; Leng et al. 2013; Sharma et al. 2011; Thongnopnua 2008; Wang et al. 2014; Xia et al. 2015). In humans, bioavailability studies of C. asiatica have focused on asiaticoside and asiatic acid. Pharmacokinetic studies have reported the time to reach the maximum plasma concentration (tmax) for asiatic acid was 4.0–4.6 hours post-ingestion of either purified asiatic acid or the triterpene mixture, TTFCA (Grimaldi et al. 1990; Rush et al. 1993). Rush et al. (1993) found that the separate administration of equimolar oral doses of asiatic acid (12mg) and asiaticoside (24mg) in humans resulted in a similar total concentration (area under the curve) of plasma asiatic acid over 12 hours. Of note, the peak plasma concentration was attained earlier when asiatic acid was administered compared to asiaticoside. The asiatic acid plasma profile also demonstrated more of a typical “sawtooth pattern” compared to asiaticoside suggestive of more immediate availability, compared to the delay required for in vivo hydrolysis of asiaticoside by intestinal enzymes (Rush et al. 1993). This is also seen in a separate human study by Grimaldi et al. (1990), where there was no observed difference in the AUC of asiatic acid after the administration of pure asiatic acid and asiaticoside independently. In this study, the investigators also compared a single administration to a seven-day course of treatment, in which they found the area under the curve over 24 hours was much greater for chronic treatment than single treatment despite the short half-life. They propose this is due to slow metabolism of asiaticoside from previous doses, either because of delayed absorption or delayed metabolism.
Since most C. asiatica preparations are not composed of purified compounds, further studies are needed to understand the pharmacokinetics of triterpenes when present in complex extracts. One study has been conducted in a dog model using an encapsulated water extract of C. asiatica standardized to the amount of asiaticoside. In this study, the peak concentration of asiatic acid was observed at 2.7 hours after consumption (Zheng and Wang 2009). As with the human study, this model demonstrated little asiaticoside within the plasma after oral administration due to a proposed complete biotransformation into asiatic acid and its glucuronide and sulphate conjugates (Rush et al. 1993; Zheng and Wang 2009).
Studies in rats have shown the absolute oral bioavailability of Centella’s triterpenoids is 16–30% for the glycosides and 50% for the acids (Chassaud et al. 1971; Vogel et al. 1990; Yuan et al. 2015) with primary elimination via the feces (Leng et al. 2013). These low levels are proposed to be due to poor solubility, slow absorption, dissolution within the gastrointestinal tract, and/or variations in gastrointestinal transit time (Chassaud et al. 1971; Yuan et al. 2015). Bioavailability studies in rats using pure triterpenoid compounds, or ethanolic extracts of C. asiatica aerial parts, have revealed similarities in the observed pharmacokinetic profile of madecassoside (Han et al. 2012; Leng et al. 2013; Wang et al. 2014), madecassic acid (Wang et al. 2014), and asiatic acid (Nair et al. 2012; Yuan et al. 2015). Most of the studies reported a rapid time to maximum concentration (Tmax= 0.5–1.3h) and a delayed second maximum concentration peak in plasma levels and/or tissue distribution due to a proposed enterohepatic recirculation and/or selective and differential absorption from the gastrointestinal tract (Han et al. 2012; Leng et al. 2013; Nair et al. 2012; Wang et al. 2014). Notably, unlike the human studies on asiaticoside described earlier (Grimaldi et al. 1990; Rush et al. 1993), the glycoside madecassoside was reported to be present in the plasma of rats following oral administration (Han et al. 2012; Leng et al. 2013; Wang et al. 2014). Of these studies, Wang et al. (2014) also found the amount detected in plasma was affected by the disease state. In their rat model of arthritis, they noted higher levels of madecassic acid and lower levels of madecassoside in rats with induced arthritis, compared to non-arthritic rats (Wang et al. 2014). They propose this may be due to a wider distribution of the madecassoside and decreased elimination in vivo due to the disease.
Xia et al. (2015) used the well-established zebrafish model and LC/IT (ion trap)-MSn to identify any downstream metabolites from the aglycones. Using this sensitive methodology, they detected ten different phase one metabolites from asiatic acid and nine from madecassic acid, derived from hydroxylation and /or dehydrogenation reactions in vivo (Xia et al. 2015). It is hypothesized that these metabolites may be more bioactive compared to their parent compounds. The occurrence and biological activity of these metabolites in human and other mammalian systems requires further study.
Application to neurological diseases:
Few studies have looked at the tissue distribution of the triterpenes following oral administration, in particular brain levels of these compounds. One study detected asiatic acid and madecassic acid within the brains of mice (Yin et al. 2012). Another focused only on madecassoside levels, which were reported to be below the lower limit of quantification in the brains of rats (Leng et al. 2013). Given the previously discussed observed cognitive changes and brain biochemical effects seen in animal models with C. asiatica administration, further investigation is needed to understand the brain bioavailability of these compounds and their metabolites.
4.2. Caffeoylquinic acids (CQAs)
There have been no reported studies on the ADME of CQAs following oral administration of C. asiatica, but there are multiple studies on the fate of these compounds when derived from other botanical sources (Williamson et al. 2011). Coffee, in particular, is a major source of CQAs, being rich in 3-, 4- and 5- CQA as well as the 3,4-, 3,5- and 4,5- diCQA isomers. Controversial data exists on the oral absorption of the CQAs (Williamson et al. 2011). In humans, high plasma levels of intact mono and diCQAs were reported following coffee consumption (Farah et al. 2008; Monteiro et al. 2007) (Farah et al. 2008). Both 1,5-diCQA and its methylated metabolite, 1,5-diferuloylqunic acid, were measured in human plasma following oral administration of 1,5-diCQA (Liu et al. 2010). DiCQAs were also found in rat plasma after oral administration of Ainsliaea fragrans extract containing 1,5-, 3,4-, 3,5- and 4,5 diCQA (Su et al. 2014). By contrast, intact CQAs were not seen in the plasma after oral administration of Artichoke extracts known to contain both mono and di-CQAs (Wittemer et al. 2005). It has been suggested that absorption is dose dependent; at higher doses, a proportion of the CQAs may escape metabolism or hydrolysis due to enzyme saturation (Erk et al. 2012) facilitating their appearance in the plasma. It has also been noted, that mono and di-CQAs can isomerize in vivo (Basavarajappa et al. 2014; Kahle et al. 2007). A comparison of bioavailability and pharmacokinetic data for CQAs and metabolites from several studies (Williamson et al. 2011) identifies several other discrepancies and highlights the need for dose response studies.
The metabolism of CQAs is well documented (Erk et al. 2012; Stalmach et al. 2009; Williamson et al. 2011; Wong et al. 2010) and includes hydrolytic cleavage of caffeic acid moieties from the quinic acid group, Phase I methylation of the caffeic acid to ferulic and isoferulic acid, reduction of the double bond in hydroxycinnamic acids by colonic microflora to produce dihydrocaffeic, dihydroferulic and dihydroisoferulic acids, Phase II sulfation or glucuronidation of the hydroxyl groups of any these compounds, and glycine conjugation with the free carboxylic acid group of the hydroxycinnamic acids. Lactone forms of mono CQAs have also been identified (Stalmach et al. 2009). Further breakdown of caffeic acid to ethyl, methyl and vinyl catechols by gut microflora has been proposed (Senger et al. 2016). Urinary excretion products include the CQA metabolites rather than the CQAs themselves, mostly as sulfate and glucuronide conjugates (Stalmach et al. 2009). Total urinary metabolites corresponded to approximately 30% of the CQAs intake from coffee (Stalmach et al. 2009).
An LC-MSn method capable of quantifying 56 compounds that may be derived from CQA metabolism in human plasma has been reported (Marmet et al. 2014). Using this method, Scherbel et al. (Scherbl et al. 2016) reported the pharmacokinetics in humans of monoCQAs and various metabolite groups listed earlier following consumption of coffee. Considerable inter-subject variation was noted in pharmacokinetic parameters. Peak plasma levels for the mono CQAs and hydroxycinnamic acid metabolites were generally rapidly attained (tmax ≤ 1 hour) suggesting their absorption from the upper gastrointestinal tract, whereas the dihydro metabolites of the hydroxycinnamates peaked significantly later (5.5 to 8.5 hours), supporting their later formation in the colon. Similar results were found in other studies on CQAs from coffee (Stalmach et al. 2009) and artichoke extract (Wittemer and Veit 2003). Peak urinary excretion of CQA metabolites were reported in the 6–12 hour (Scherbl et al. 2016) or 5–8 hour (Stalmach et al. 2009) sample collections.
A pharmacokinetic and tissue distribution study of mono and diCQAs from Ainsliaea fragrans in rats (Su et al. 2014) reported mono CQAs were undetectable in the plasma, but that diCQA levels peaked once at less than 30 minutes and again at around four hours, possibly due to enterohepatic circulation. Mono and diCQAs and caffeic acid were detected in multiple organs (lung, liver, muscle, brain, spleen, kidney and heart). Peak brain levels of individual diCQAs ranged from 25 ng/g to 20 μg/g following oral administration of the A. fragrans extract (46 mg/kg b.wt total diCQAs). Based on this data, the C. asiatica oral doses (2.5 mg/kg b. wt. total diCQAs) used in our in vivo studies (Soumyanath et al. 2012) (Gray et al. 2016) could potentially give brain levels around1000 ng/g of individual diCQAs. This is comparable to the minimum neuroprotective concentration (500 ng/ml) we have observed in vitro for these compounds (Gray et al. 2014). Caffeic acid and ferulic acids levels in the brain were significantly increased after injection of an intravenous bolus of these compounds to rats (Gasperotti et al. 2015). Ferulic acid but not caffeic acid was also found in rat brains after oral administration of Portulaca oleracea extract, which contains both compounds (Zhang et al. 2015). The detection of these CQAs and metabolites in the brain supports their potential to penetrate the blood brain barrier after administration of C. asiatica, and suggests that in vitro effects seen for some of these compounds on neuronal cultures may be recapitulated in vivo.
4.3. Flavonoids
The flavanols catechin, epicatechin, the flavone rutin, and flavonols quercetin and kaempferol have been detected within extracts of C. asiatica (Alqahtani et al. 2015; Chandrika and Prasad Kumarab 2015; Lu et al. 2011). To our knowledge, there have not been any studies on the ADME of these flavonoids derived specifically from C. asiatica; however, there have been multiple studies of flavonoids derived from other plant materials, such as food sources (Hollman 2004; Kumar and Pandey 2013).
With the exception of the catechins, most flavonoids are bound to sugars within plant material making them β-glycosides (Kumar and Pandey 2013). These glycosides are primarily absorbed after being deglycosylated by the small intestinal lumen and microflora (Lu et al. 2013; Németh et al. 2003; Yang et al. 2012), while the catechins, aglycones and hydrophilic compounds are absorbed via the small intestine mucosa (Hollman et al. 1999). Following absorption, the compounds are conjugated in the liver to form O-methylated, O-sulfated and O-glucuronidated conjugates, which have been detected in the plasma of rats and humans (Actis-Goretta et al. 2012; Bravo 1998; Hollman et al. 1999; Hollman 2004; Jäger and Saaby 2011).
Rats have been used to assess the brain distribution of these compounds. Quercetin and kaempferol aglycones and conjugates were found in the brain following the consumption of the botanical extract Gingko biloba (Rangel-Ordóñez et al. 2010). Similar studies are needed to assess the brain bioavailability of flavonoids from C. asiatica and to further understand the role these compounds play in its effect on cognition and mood.
5. Conclusions and Perspective
Centella asiatica demonstrates remarkable cognitive, neurotropic and neuroprotective effects. These properties support the traditional use of the herb as a memory enhancer, and highlight its potential to modulate disease processes involved in neurodegenerative disorders. Successful development of novel treatments based on C. asiatica will require a focus on at least three main areas of research. First, a more extensive understanding of the active compounds of C. asiatica is required. To date, the triterpenes asiatic acid, asiaticoside, and madecassoside, as well as caffeoylquinic acids, have been shown to contribute to the neurological effects of the herb.. At the present time, these two groups of compounds are the most relevant for standardization of C. asiatica products. However, further research, using bioassay guided fractionation approaches, or OPLS-DA activity/fingerprint correlations may identify additional active compounds within C. asiatica. Interactions between the different compounds (i.e. synergy, additivity, antagonism) also need to be explored. Second, the detailed biochemical mechanisms underlying the observed effects on mitochondrial activity, oxidative stress, neurotransmitter homeostasis, and synaptic plasticity need to be defined as does the manner in which each of these effects contributes to the broader neuroprotective and cognitive enhancing properties of the plant. This will be a challenge when dealing with complex extracts where the multiple components could simultaneously influence multiple pathways. A careful evaluation of individual active compounds and known mixtures to evaluate interactions will be required. Finally, human studies of C. asiatica extracts, or compounds derived from the botanical, will be important to confirm the clinical significance of earlier studies. Adequate chemical characterization and description of the C. asiatica test material will be vital for these studies, both to ensure the presence of adequate levels of active compounds, and to allow comparison between studies. This is particularly important given the noted variation in content of phytochemicals in C. asiatica due to genetic and geographical factors and processing methods. The use of contemporary analytical techniques such as LCHRMS and IM-HRMS will be particularly important for the fingerprinting of extracts and documentation of known and unknown compounds.
Table 3.
Summary of the effects of C. asiatica and some of its constituent compounds in preclinical neurological systems
| Test material | Neuroactive property | Potential mechanism |
|---|---|---|
| Centella asiatica extract | ||
| Triterpenes | ||
| CQAs |
(Barbosa 2008),
Acknowledgement:
The authors acknowledge the following sources of grant support: NIH-NCCIH K99 AT008831 (NG), NIH-NCCIH R01AT008099 (AS, AA, CSM, JFS, JQ, PL), NIH-NCRR S10RR027878 (JFS), NIH-NCRR S10RR025628 (CSM), NIH-NCCIH T32 AT002688 (KW), Department of Veterans Affairs Merit Review grant (JQ).
References cited
- (1997) Botanical Safety Handbook, American Herbal Products Association. CRC Press, Boca Raton, USA [Google Scholar]
- Actis-Goretta L, Lévèques A, Giuffrida F, Romanov-Michailidis F, Viton F, Barron D, Duenas-Paton M, Gonzalez-Manzano S, Santos-Buelga C, Williamson G, Dionisi F (2012) Elucidation of (−)-epicatechin metabolites after ingestion of chocolate by healthy humans. Free Radic Biol Med 53: 787–795 [DOI] [PubMed] [Google Scholar]
- Alqahtani A, Tongkao-On W, Li KM, Razmovski-Naumovski V, Chan K, Li GQ (2015) Seasonal Variation of Triterpenes and Phenolic Compounds in Australian Centella asiatica (L.) Urb. Phytochemical Analysis 26: 436–443 [DOI] [PubMed] [Google Scholar]
- Antognoni F, Perellino NC, Crippa S, Dal Toso R, Danieli B, Minghetti A, Poli F, Pressi G (2011) Irbic acid, a dicaffeoylquinic acid derivative from Centella asiatica cell cultures. Fitoterapia 82: 950–954 [DOI] [PubMed] [Google Scholar]
- Apichartsrangkoon A, Wongfhun P, Gordon M (2009) Flavor Characterization of Sugar‐Added Pennywort (Centella asiaticaL.) Juices Treated with Ultra‐High Pressure and Thermal Processes. Journal of Food Science 74: C643–C646 [DOI] [PubMed] [Google Scholar]
- Appa Rao M, Srinivasan K, Rao K (1973) The effect of Mandookaparni (Centella asiatica) on the general mental ability (Medhya) of mentally retarded children. In: Unit for Research in Indian Medicine VHSMC, Adyar Madra (ed), Jour. Res. Ind. med, pp 9–16 [Google Scholar]
- Arun S, Liu L, Donmez G (2016) Mitochondrial Biology and Neurological Diseases. Curr Neuropharmacol 14: 143–154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Awad R, Levac D, Cybulska P, Merali Z, Trudeau V, Arnason J (2007) Effects of traditionally used anxiolytic botanicals on enzymes of the gamma-aminobutyric acid (GABA) system. Can J Physiol Pharmacol 85: 933–942 [DOI] [PubMed] [Google Scholar]
- Azerad R (2016) Chemical structures, production and enzymatic transformations of sapogenins and saponins from Centella asiatica (L.) Urban. Fitoterapia 114: 168–187 [DOI] [PubMed] [Google Scholar]
- Barbosa NR, Pittella F, and Gattaz WF (2008) Centella asiatica water extract inhibits iPLA2 and cPLA2 activities in rat cerebellum Phytomedicine 15: 896–900 [DOI] [PubMed] [Google Scholar]
- Basavarajappa BS, Nagre NN, Xie S, Subbanna S (2014) Elevation of endogenous anandamide impairs LTP, learning, and memory through CB1 receptor signaling in mice. Hippocampus 24: 808–818 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belcaro G, Maquart FX, Scoccianti M, Dugall M, Hosoi M, Cesarone MR, Luzzi R, Cornelli U, Ledda A, Feragalli B (2011) TECA (Titrated Extract of Centella Asiatica): new microcirculatory, biomolecular, and vascular application in preventive and clinical medicine. A status paper. Panminerva Med 53: 105–118 [PubMed] [Google Scholar]
- Boettler U, Sommerfeld K, Volz N, Pahlke G, Teller N, Somoza V, Lang R, Hofmann T, Marko D (2010) Coffee constituents as modulators of Nrf2 nuclear translocation and ARE (EpRE)-dependent gene expression. J Nutr Biochem 22: 426–440 [DOI] [PubMed] [Google Scholar]
- Bonfill M, Mangas S, Cusido RM, Osuna L, Pinol MT, Palazon J (2006) Identification of triterpenoid compounds of Centella asiatica by thin-layer chromatography and mass spectrometry. Biomedical Chromatography 20: 151–153 [DOI] [PubMed] [Google Scholar]
- Botanica B (2017) Gotu-Kola.
- Bradwejn J, Zhou Y, Koszycki D, Shlik J (2000) A double-blind, placebo-controlled study on the effects of Gotu Kola (Centella asiatica) on acoustic startle response in healthy subjects. J Clin Psychopharmacol 20: 680–684 [DOI] [PubMed] [Google Scholar]
- Bravo L (1998) Polyphenols: chemistry, dietary sources, metabolism, and nutritional significance. Nutr Rev 56: 317–333 [DOI] [PubMed] [Google Scholar]
- Brinkhaus B, Lindner M, Schuppan D, Hahn EG (2000) Chemical, pharmacological and clinical profile of the East Asian medical plant Centella asiatica. Phytomedicine 7: 427–448 [DOI] [PubMed] [Google Scholar]
- Cao X, Xiao H, Zhang Y, Zou L, Chu Y, Chu X (2010) 1, 5-Dicaffeoylquinic acid-mediated glutathione synthesis through activation of Nrf2 protects against OGD/reperfusion-induced oxidative stress in astrocytes. Brain Res 1347: 142–148 [DOI] [PubMed] [Google Scholar]
- Ceremuga T, Valdivieso D, Kenner C, Lucia A, Lathrop K, Stailey O, Bailey H, Criss J, Linton J, Fried J, Taylor A, Padron G, Johnson A (2015) Evaluation of the anxiolytic and antidepressant effects of asiatic acid, a compound from Gotu kola or Centella asiatica, in the male Sprague Dawley rat. AANA J 83: 91–98 [PubMed] [Google Scholar]
- Cesarone MR, Incandela L, De Sanctis MT, Belcaro G, Bavera P, Bucci M, Ippolito E (2001) Evaluation of treatment of diabetic microangiopathy with total triterpenic fraction of Centella asiatica: a clinical prospective randomized trial with a microcirculatory model. Angiology 52 Suppl 2: S49–54 [PubMed] [Google Scholar]
- Chanana P, Kumar A (2016) Possible Involvement of Nitric Oxide Modulatory Mechanisms in the Neuroprotective Effect of Centella asiatica Against Sleep Deprivation Induced Anxiety Like Behaviour, Oxidative Damage and Neuroinflammation. Phytother Res 30: 671–680 [DOI] [PubMed] [Google Scholar]
- Chandrika UG, Prasad Kumarab PA (2015) Gotu Kola (Centella asiatica): Nutritional Properties and Plausible Health Benefits. Adv Food Nutr Res 76: 125–157 [DOI] [PubMed] [Google Scholar]
- Chassaud LF, Fry BJ, Hawkins DR, Lewis JD, Sword IP, Taylor T, Hathway DE (1971) The metabolism of asiatic acid,-madecassic acid and asiaticoside in the rat. Arzneimittelforschung 21: 1379–1384 [PubMed] [Google Scholar]
- Chen C, Tsai W, Chen C, TM P (2015) Centella asiatica extract protects against amyloid β1–40-induced neurotoxicity in neuronal cells by activating the antioxidative defence system. J Tradit Complement Med 6: 362–369 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S, Yin Z, Jiang C, Ma Z, Fu Q, Qu R, Ma S (2014) Asiaticoside attenuates memory impairment induced by transient cerebral ischemia-reperfusion in mice through anti-inflammatory mechanism. Pharmacol Biochem Behav 122: 7–15 [DOI] [PubMed] [Google Scholar]
- Choi S, Kim J, Suh S, Kim C, Kim H, Kim C, Park G, Park C, Shin D (2014) Ligularia fischeri extract protects against oxidative-stress-induced neurotoxicity in mice and PC12 cells. J Med Food 17: 1222–1231 [DOI] [PubMed] [Google Scholar]
- Clifford MN, Knight S, Kuhnert N (2005) Discriminating between the six isomers of dicaffeoylquinic acid by LC-MSn. Journal of Agricultural and Food Chemistry 53: 3821–3832 [DOI] [PubMed] [Google Scholar]
- Conley K, Jubrias S, Esselman P (2000) Oxidative capacity and ageing in human muscle. Journal of Physiology 52: 203–210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Defillipo PP, Raposo AH, Fedoce AG, Ferreira AS, Polonini HC, Gattaz WF, and Raposo NR (2012) Inhibition of cPLA2 and sPLA2 activities in primary cultures of rat cortical neurons by Centella asiatica water extrac. Nat Prod Commun 7: 841–843 [PubMed] [Google Scholar]
- Deshmukh R, Kaundal M, Bansal V, Samardeep. (2016) Caffeic acid attenuates oxidative stress, learning and memory deficit in intra-cerebroventricular streptozotocin induced experimental dementia in rats. Biomed pharmacother 81: 56–62 [DOI] [PubMed] [Google Scholar]
- Dev RDO, Mohamed S, Hambali Z, Samah BA (2009) Comparison on cognitive effects of Centella asiatica in healthy middle age female and male volunteers. In: Malaysia UP: (ed), European Journal of Scientific Research, pp 553–656 [Google Scholar]
- Devkota A, Dall’Acqua S, Comai S, Innocenti G, Jha PK (2010) Centella asiatica (L.) urban from Nepal: Quali-quantitative analysis of samples from several sites, and selection of high terpene containing populations for cultivation. Biochemical Systematics and Ecology 38: 12–22 [Google Scholar]
- Dewick P (2002) The Shikimate pathway: Aromatic amino acids and phenylpropanoids Meicinal Natural Products. John Wiley & Sons, Ltd, pp 121–166 [Google Scholar]
- Dhanasekaran M, Holcomb L, Hitt A, Tharakan B, Porter J, Young K, Manyam B (2009) Centella asiatica extract selectively decreases amyloid beta levels in hippocampus of Alzheimer’s disease animal model. Phytother Res 23: 14–19 [DOI] [PubMed] [Google Scholar]
- Diallo B, Vanhaelenfastre R, Vanhaelen M (1991) Direct Coupling of High-Speed Countercurrent Chromatography to Thin-Layer Chromatography - Application to the Separation of Asiaticoside and Madecassoside from Centella-Asiatica. Journal of chromatography 558: 446–450 [Google Scholar]
- Doknark S, Mingmalairak S, Vattanajun A, Tantisira B, Tantisira M (2014) Study of ameliorating effects of ethanolic extract of Centella asiatica on learning and memory deficit in animal models. J Med Assoc Thai 97: S68–76 [PubMed] [Google Scholar]
- Dong LL, Shion H, Davis RG, Terry-Penak B, Castro-Perez J, van Breemen RB (2010) Collision Cross-Section Determination and Tandem Mass Spectrometric Analysis of Isomeric Carotenoids Using Electrospray Ion Mobility Time-of-Flight Mass Spectrometry. Analytical Chemistry 82: 9014–9021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drewes LR (2001) Molecular architecture of the brain microvasculature: perspective on blood-brain transport. J Mol Neurosci 16: 93–98; discussion 151–157 [DOI] [PubMed] [Google Scholar]
- Du H, Guo L, Yan S, Sosunov A, McKhann G, Yan S (2010) Early deficits in synaptic mitochondria inan Alzheimer’s disease mouse model.. Proc Natl Acad Sci 107: 18670–18675 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Du QZ, Jerz G, Chen P, Winterhalter P (2004) Preparation of ursane triterpenoids from Centella asiatica using high speed countercurrent chromatography with step-gradient elution. Journal of Liquid Chromatography & Related Technologies 27: 2201–2215 [Google Scholar]
- Erk T, Williamson G, Renouf M, Marmet C, Steiling H, Dionisi F, Barron D, Melcher R, Richling E (2012) Dose-dependent absorption of chlorogenic acids in the small intestine assessed by coffee consumption in ileostomists. Molecular Nutrition & Food Research 56: 1488–1500 [DOI] [PubMed] [Google Scholar]
- Farah A, Monteiro M, Donangelo CM, Lafay S (2008) Chlorogenic acids from green coffee extract are highly bioavailable in humans. Journal of Nutrition 138: 2309–2315 [DOI] [PubMed] [Google Scholar]
- Farhana KM, Malueka RG, Wibowo S, Gofir A (2016) Effectiveness of Gotu Kola Extract 750 mg and 1000 mg Compared with Folic Acid 3 mg in Improving Vascular Cognitive Impairment after Stroke. Evid Based Complement Alternat Med 2016: 2795915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farooqui AA, Ong WY, and Horrocks LA (2006) Inhibitors of brain phospholipase A2 activity: their neuropharmacological effects and therapeutic importance for the treatment of neurologic disorders. Pharmacol Rev 58: 591–620 [DOI] [PubMed] [Google Scholar]
- Flora S, Gupta R (2007) Beneficial effects of Centella asiatica aqueous extract against arsenic-induced oxidative stress and essential metal status in rats. Phytother Res 21: 980–988 [DOI] [PubMed] [Google Scholar]
- Floridata (2016) Floridata Plant Encyclopedia.
- Flowers D (1998) On the Properties of Bit String-Based Measures of Chemical Similarity. J. Chem. Inf. Comput. Sci 38: 379–386 [Google Scholar]
- Gadahad M, Rao M, Rao G (2008) Enhancement of hippocampal CA3 neuronal dendritic arborization by Centella asiatica (Linn) fresh leaf extract treatment in adult rats. J Chin Med Assoc. 71: 6–13 [DOI] [PubMed] [Google Scholar]
- Gallego A, Ramirez-Estrada K, Vidal-Limon HR, Hidalgo D, Lalaleo L, Kayani WK, Cusido RM, Palazon J (2014) Biotechnological production of centellosides in cell cultures of Centella asiatica (L) Urban. Engineering in Life Sciences 14: 633–642 [Google Scholar]
- Gasperotti M, Passamonti S, Tramer F, Masuero D, Guella G, Mattivi F, Vrhovsek U (2015) Fate of Microbial Metabolites of Dietary Polyphenols in Rats: Is the Brain Their Target Destination? ACS Chemical Neuroscience 6: 1341–1352 [DOI] [PubMed] [Google Scholar]
- Gray NE, H S, JA Z, JF Q, A S (2015) Centella asiatica Attenuates Amyloid-β-Induced Oxidative Stress and Mitochondrial Dysfunction. Journal of Alzheimer’s Disease 45: 933–946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray NE, Harris C, Quinn J, Soumyanath A (2016) Centella asiatica modulates antioxidant and mitochondrial pathways and improves cognitive function in mice. Journal of Ethnopharmacol 180: 78–86 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray NE, Morré J, Kelley J, Maier CS, Stevens JF, Quinn JF, A S (2014) Caffeoylquinic Acids in Centella asiatica Protect against Amyloid-β Toxicity. J Alzheimer’s Dis 40: 359–373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimaldi R, De Ponti F, D’Angelo L, Caravaggi M, Guidi G, Lecchini S, Frigo GM, Crema A (1990) Pharmacokinetics of the total triterpenic fraction of Centella asiatica after single and multiple administrations to healthy volunteers. A new assay for asiatic acid. J Ethnopharmacol 28: 235–241 [DOI] [PubMed] [Google Scholar]
- Gunther B, Wagner H (1996) Quantitative determination of triterpenes in extracts and phytopreparations of Centella asiatica (L) Urban. Phytomedicine 3: 59–65 [DOI] [PubMed] [Google Scholar]
- Gupta Y, Veerendra Kumar M, Srivastava A (2003) Effect of Centella asiatica on pentylenetetrazole-induced kindling, cognition and oxidative stress in rats. Pharmacol Biochem Behav 74: 579–585 [DOI] [PubMed] [Google Scholar]
- Haleagrahara N, Ponnusamy K (2010) Neuroprotective effect of Centella asiatica extract (CAE) on experimentally induced parkinsonism in aged Sprague Dawley rats. J Toxicol Sci 35: 41–47 [DOI] [PubMed] [Google Scholar]
- Han J, Miyamae Y, Shigemori H, Isoda H (2010) Neuroprotective effect of 3,5-di-O-caffeoylquinic acid on SH-SY5Y cells and senescence-accelerated-prone mice 8 through the up-regulation of phosphoglycerate kinase-1. Neuroscience 169: 1039–1045 [DOI] [PubMed] [Google Scholar]
- Han WJ, Xia YF, Dai Y (2012) Development and validation of high-performance liquid chromatography/electrospray ionization mass spectrometry for assay of madecassoside in rat plasma and its application to pharmacokinetic study. Biomedical Chromatography 26: 26–32 [DOI] [PubMed] [Google Scholar]
- Hensley K, Carney J, Mattson M, Aksenova M, Harris M, Wu J, Floyd R, Butterfield D (1994) A model for betaamyloid aggregation and neurotoxicity based on free radical generation by the peptide: relevance to Alzheimer disease. Proc Natl Acad Sci 91: 3270–3274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hollman PC, Bijsman MN, van Gameren Y, Cnossen EP, de Vries JH, Katan MB (1999) The sugar moiety is a major determinant of the absorption of dietary flavonoid glycosides in man. Free Radic Res 31: 569–573 [DOI] [PubMed] [Google Scholar]
- Hollman PCH (2004) Absorption, Bioavailability, and Metabolism of Flavonoids. Pharmaceutical Biology 42: 74–83 [Google Scholar]
- Incandela L, Belcaro G, De Sanctis MT, Cesarone MR, Griffin M, Ippolito E, Bucci M, Cacchio M (2001) Total triterpenic fraction of Centella asiatica in the treatment of venous hypertension: a clinical, prospective, randomized trial using a combined microcirculatory model. Angiology 52 Suppl 2: S61–67 [PubMed] [Google Scholar]
- IPNI (2016) International Plant Names Index. Royal Botanic Gardens, Kew. [Google Scholar]
- Itoh K, T C, Takahashi S, Ishii T, Igarashi K, Katoh Y, Oyake T, Hayashi N, Satoh K, Hatayama I, Yamamoto M, Nabeshima Y (1997) An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements. Biochem Biophys Res Commun 236: 313–322 [DOI] [PubMed] [Google Scholar]
- Jäger AK, Saaby L (2011) Flavonoids and the CNS. Molecules 16: 1471–1485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- James J, Dubery I (2011) Identification and Quantification of Triterpenoid Centelloids in Centella asiatica (L.) Urban by Densitometric TLC. Jpc-Journal of Planar Chromatography-Modern Tlc 24: 82–87 [Google Scholar]
- James JT, Dubery IA (2009) Pentacyclic triterpenoids from the medicinal herb, Centella asiatica (L.) Urban. Molecules 14: 3922–3941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamil S, Nizami Q, Salam M (2007) Centella asiatica (Linn.) Urban: a review. Natural Product Radiance 6: 158–170 [Google Scholar]
- Jana U, Sur TK, Maity LN, Debnath PK, Bhattacharyya D (2010) A clinical study on the management of generalized anxiety disorder with Centella asiatica. Nepal Med Coll J 12: 8–11 [PubMed] [Google Scholar]
- Jew SS, Yoo CH, Lim DY, Kim H, MookJung I, Jung MW, Choi H, Jung YH, Kim H, Park HG (2000) Structure activity relationship study of asiatic acid derivatives against beta amyloid (A beta)induced neurotoxicity. Bioorganic & medicinal chemistry letters 10: 119–121 [DOI] [PubMed] [Google Scholar]
- Jiang W, Li M, He F, Bian Z, He Q, Wang X, Yao W, Zhu L (2016) Neuroprotective effect of asiatic acid against spinal cord injury in rats. Life Sci 157: 45–51 [DOI] [PubMed] [Google Scholar]
- Kahle K, Huemmer W, Kempf M, Scheppach W, Erk T, Richling E (2007) Polyphenols are intensively metabolized in the human gastrointestinal tract after apple juice consumption. Journal of Agricultural and Food Chemistry 55: 10605–10614 [DOI] [PubMed] [Google Scholar]
- Kapoor L (1990) Handbook of Ayurvedic medicinal plants. CRC Press, Boca Raton [Google Scholar]
- Kim J, Lee H, Hwang B, Kim S, Yoo J, Seong Y (2012a) Neuroprotection of Ilex latifolia and caffeoylquinic acid derivatives against excitotoxic and hypoxic damage of cultured rat cortical neurons. Arch Pharm Res 35: 1115–1122 [DOI] [PubMed] [Google Scholar]
- Kim S, Bolton EE, Bryant SH (2012b) Effects of multiple conformers per compound upon 3-D similarity search and bioassay data analysis. Journal of Cheminformatics 4: 28–28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krishnamurthy R, Senut M, Zemke D, Min J, Frenkel M, Greenberg E, Yu S, Ahn N, Goudreau J, Kassab M, Panickar K, Majid A (2009) Asiatic acid, a pentacyclic triterpene from Centella asiatica, is neuroprotective in a mouse model of focal cerebral ischemia. J Neurosci Res 87: 2541–2550 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar A, Dogra S, Prakash A (2009) Neuroprotective Effects of Centella asiatica against Intracerebroventricular Colchicine-Induced Cognitive Impairment and Oxidative Stress. Int J Alzheimers Dis 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar A, Prakash A, Dogra S (2011) Centella asiatica Attenuates D-Galactose-Induced Cognitive Impairment, Oxidative and Mitochondrial Dysfunction in Mice. Int J Alzheimers Dis 2011: 347569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar S, Pandey AK (2013) Chemistry and biological activities of flavonoids: an overview. ScientificWorldJournal 2013: 162750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumari S, Deori M, Elancheran R, Kotoky J, Devi R (2016) In vitro and In vivo Antioxidant, Anti-hyperlipidemic Properties and Chemical Characterization of Centella asiatica (L.) Extract. Front Pharmacol 7: 400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwon S, Lee H, Kim J, Hong S, Kim H, Jo T, Park Y, Lee C, Kim Y, Lee S, Jang C (2010) Neuroprotective effects of chlorogenic acid on scopolamine-induced amnesia via anti-acetylcholinesterase and anti-oxidative activities in mice. Eur J Pharmacol 649: 210–217 [DOI] [PubMed] [Google Scholar]
- Lal RK, Gupta P, Dubey BK (2017) Genetic variability and associations in the accessions of Manduk parni {Centella asiatica (L)}. Industrial Crops and Products 96: 173–177 [Google Scholar]
- Leng DD, Han WJ, Rui Y, Dai Y, Xia YF (2013) In vivo disposition and metabolism of madecassoside, a major bioactive constituent in Centella asiatica (L.) Urb. Journal of Ethnopharmacology 150: 601–608 [DOI] [PubMed] [Google Scholar]
- Liang G, Shi B, Luo W, Yang J (2015) The protective effect of caffeic acid on global cerebral ischemia-reperfusion injury in rats. Behav Brain Funct 11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin X, Huang R, Zhang S, Wei L, Zhuo L, Wu X, Tang A, Huang Q (2013) Beneficial effects of asiaticoside on cognitive deficits in senescence-accelerated mice. Fitoterapia 87: 69–77 [DOI] [PubMed] [Google Scholar]
- Lin X, Zhang S, Huang R, Wei L, Tan S, Liang C, Lv S, Chen Y, Liang S, Tian Y, Lu Z, Huang Q (2014) Protective effect of madecassoside against cognitive impairment induced by D-galactose in mice. Pharmacol Biochem Behav 124: 434–442 [DOI] [PubMed] [Google Scholar]
- Liu J, Dou G, Dong X, Yuan D, Ji X, Wu Z, Meng Z (2010) An improved LC-MS/MS method for simultaneous determination of 1,5-dicaffeoylquinic acid and its active metabolites in human plasma and its application to a pharmacokinetic study in patients. Biomedical Chromatography 24: 935–940 [DOI] [PubMed] [Google Scholar]
- Long HS, Stander MA, Van Wyk BE (2012) Notes on the occurrence and significance of triterpenoids (asiaticoside and related compounds) and caffeoylquinic acids in Centella species. South African Journal of Botany 82: 53–59 [Google Scholar]
- Lu L, Qian D, Yang J, Jiang S, Guo J, Shang EX, Duan JA (2013) Identification of isoquercitrin metabolites produced by human intestinal bacteria using UPLC-Q-TOF/MS. Biomed Chromatogr 27: 509–514 [DOI] [PubMed] [Google Scholar]
- Lu Q, Li WH, Hu SQ (2011) [Studies on fingerprints of Centella asiatica by HPLC]. Zhong Yao Cai 34: 46–50 [PubMed] [Google Scholar]
- Luo Y, Yang Y, Liu J, Li W, Yang J, Sui X, Yuan X, Nie Z, Liu Y, Chen D, Lin S, Wang Y (2014) Neuroprotective effects of madecassoside against focal cerebral ischemia reperfusion injury in rats. Brain Res 1565: 37–47 [DOI] [PubMed] [Google Scholar]
- Maldini M, Natella F, Baima S, Morelli G, Scaccini C, Langridge J, Astarita G (2015) Untargeted Metabolomics Reveals Predominant Alterations in Lipid Metabolism Following Light Exposure in Broccoli Sprouts. Int J Mol Sci 16: 13678–13691 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mangas S, Moyano E, Osuna L, Cusido RM, Bonfill M, Palazon J (2008) Triterpenoid saponin content and the expression level of some related genes in calli of Centella asiatica. Biotechnology Letters 30: 1853–1859 [DOI] [PubMed] [Google Scholar]
- Marmet C, Actis-Goretta L, Renouf M, Giuffrida F (2014) Quantification of phenolic acids and their methylates, glucuronides, sulfates and lactones metabolites in human plasma by LC-MS/MS after oral ingestion of soluble coffee. Journal of Pharmaceutical and Biomedical Analysis 88: 617–625 [DOI] [PubMed] [Google Scholar]
- Mato L, Wattanathorn J, Muchimapura S, Tongun T, Piyawatkul N, Yimtae K, Thanawirattananit P, Sripanidkulchai B (2011) Centella asiatica Improves Physical Performance and Health-Related Quality of Life in Healthy Elderly Volunteer. Evid. Based Complement. Alternat. Med 2011: 579467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maulidiani H, Abas F, Khatib A, Shaari K, Lajis NH (2014) Chemical characterization and antioxidant activity of three medicinal Apiaceae species. Industrial Crops and Products 55: 238–247 [Google Scholar]
- Maulidiani H, Khatib A, Shaari K, Abas F, Shitan M, Kneer R, Neto V, Lajis NH (2012) Discrimination of three pegaga (Centella) varieties and determination of growth-lighting effects on metabolites content based on the chemometry of 1H nuclear magnetic resonance spectroscopy. Journal of Agricultural and Food Chemistry 60: 410–417 [DOI] [PubMed] [Google Scholar]
- Meena H, Pandey H, Pandey P, Arya M, Ahmed Z (2012) Evaluation of antioxidant activity of two important memory enhancing medicinal plants Baccopa monnieri and Centella asiatica. Indian J Pharmacol 44: 114–117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikami Y, Yamazawa T (2015) Chlorogenic acid, a polyphenol in coffee, protects neurons against glutamate neurotoxicity. Life Sci 139: 69–74 [DOI] [PubMed] [Google Scholar]
- Mohandas Rao K, Muddanna Rao S, Gurumadhva Rao S (2006) Centella asiatica (L.) leaf extract treatment during the growth spurt period enhances hippocampal CA3 neuronal dendritic arborization in rats. Evid Based Complement Alternat Med. 3: 349–357 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohandas Rao K, Muddanna Rao S, Gurumadhva Rao S (2009) Enhancement of Amygdaloid Neuronal Dendritic Arborization by Fresh Leaf Juice of Centella asiatica (Linn) During Growth Spurt Period in Rats. Evid Based Complement Alternat Med. 6: 203–210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohandas Rao K, Rao M, Rao G (2012) Evaluation of amygdaloid neuronal dendritic arborization enhancing effect of Centella asiatica (Linn) fresh leaf extract in adult rats. Chin J Integr Med 2012 [DOI] [PubMed] [Google Scholar]
- Monteiro M, Farah A, Perrone D, Trugo LC, Donangelo C (2007) Chlorogenic acid compounds from coffee are differentially absorbed and metabolized in humans. Journal of Nutrition 137: 2196–2201 [DOI] [PubMed] [Google Scholar]
- Mook-Jung I, Shin JE, Yun SH, Huh K, Koh JY, Park HK, Jew SS, Jung MW (1999) Protective effects of asiaticoside derivatives against beta amyloid neurotoxicity. Journal of Neuroscience Research 59: 417–425 [PubMed] [Google Scholar]
- Motohashi H, Yamamoto M (2004) Nrf2-Keap1 defines a physiologically important stress response mechanism. Trends Mol Med 10: 549–557 [DOI] [PubMed] [Google Scholar]
- Muller V, Albert A, Winkler JB, Lankes C, Noga G, Hunsche M (2013) Ecologically relevant UV-B dose combined with high PAR intensity distinctly affect plant growth and accumulation of secondary metabolites in leaves of Centella asiatica L. Urban. Journal of Photochemistry and Photobiology B-Biology 127: 161–169 [DOI] [PubMed] [Google Scholar]
- Mustafa RA, AH A, Fau - Mohamed S, S M, Fau - Bakar F, Abu- Bakar FA (2010) Total phenolic compounds, flavonoids, and radical scavenging activity of 21 selected tropical plants. Journal of Food Science [DOI] [PubMed] [Google Scholar]
- Nadkarni AK (1976) Dr K.M. Nadkarni’s Indian Materia Medica. Popular Prakashan PVT Ltd [Google Scholar]
- Nair SN, Menon S, Shailajan S (2012) A liquid chromatography/electrospray ionization tandem mass spectrometric method for quantification of asiatic acid from plasma: application to pharmacokinetic study in rats. Rapid Commun Mass Spectrom 26: 1899–1908 [DOI] [PubMed] [Google Scholar]
- Nasir M, Abdullah J, Habsah M, Ghani R, R G (2012) Inhibitory effect of asiatic acid on acetylcholinesterase, excitatory post synaptic potential and locomotor activity. Phytomedicine 19: 311–316 [DOI] [PubMed] [Google Scholar]
- Nasir M, Habsah M, Zamzuri I, Rammes G, Hasnan J, Abdullah J (2011) Effects of asiatic acid on passive and active avoidance task in male Spraque-Dawley rats. J Ethnopharmacol 134: 203–209 [DOI] [PubMed] [Google Scholar]
- Navarro A, Boveris A (2007) The mitochondrial energy transduction system and the aging process.. Am J Physiol Cell Physiol 292: C670–686 [DOI] [PubMed] [Google Scholar]
- Ncube EN, Steenkamp PA, Madala NE, Dubery IA (2016) Chlorogenic Acids Biosynthesis in Centella asiatica Cells Is not Stimulated by Salicylic Acid Manipulation. Applied Biochemistry and Biotechnology 179: 685–696 [DOI] [PubMed] [Google Scholar]
- Németh K, Plumb GW, Berrin JG, Juge N, Jacob R, Naim HY, Williamson G, Swallow DM, Kroon PA (2003) Deglycosylation by small intestinal epithelial cell beta-glucosidases is a critical step in the absorption and metabolism of dietary flavonoid glycosides in humans. Eur J Nutr 42: 29–42 [DOI] [PubMed] [Google Scholar]
- Newall CA, Anderson L, Phillipson JD (1996) Herbal medicines A guide for healthcare professionals. Pharmaceutical Press, London [Google Scholar]
- Nicolas AN, Plunkett GM (2009) The demise of subfamily Hydrocotyloideae (Apiaceae) and the re-alignment of its genera across the entire order Apiales. Molecular Phylogenetics and Evolution 53: 134–151 [DOI] [PubMed] [Google Scholar]
- Oyedeji OA, Afolayan AJ (2005) Chemical composition and antibacterial activity of the essential oil of Centella asiatica growing in South Africa. Pharmaceutical Biology 43: 249–252 [Google Scholar]
- Pacini T, Fu W, Gudmundsson S, Chiaravalle AE, Brynjolfson S, Palsson BO, Astarita G, Paglia G (2015) Multidimensional analytical approach based on UHPLC-UV-ion mobility-MS for the screening of natural pigments. Anal Chem 87: 2593–2599 [DOI] [PubMed] [Google Scholar]
- Pinheiro Fernandes F, Fontenele Menezes A, de Sousa Neves J, Fonteles A, da Silva A, de Araújo Rodrigues P, Santos do Carmo M, de Souza C, de Andrade G (2014) Caffeic acid protects mice from memory deficits induced by focal cerebral ischemia. Behav Pharmacol 25: 637–647 [DOI] [PubMed] [Google Scholar]
- Pittella F, Dutra R, Junior D, Lopes M, Barbosa N (2009) Antioxidant and cytotoxic activities of Centella asiatica (L) Urb. Int J Mol Sci 10: 3713–3721 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plengmuankhae W, Tantitadapitak C (2015) Low temperature and water dehydration increase the levels of asiaticoside and madecassoside in Centella asiatica (L.) Urban. South African Journal of Botany 97: 196–203 [Google Scholar]
- Prakash A, Kumar A (2013) Mitoprotective effect of Centella asiatica against aluminum-induced neurotoxicity in rats: possible relevance to its anti-oxidant and anti-apoptosis mechanism. Neurol Sci 34: 1403–1409 [DOI] [PubMed] [Google Scholar]
- Rahajanirina V, Rakotondralambo Raoseta SO, Roger E, Razafindrazaka H, Pirotais S, Boucher M, Danthu P (2012) The influence of certain taxonomic and environmental parameters on biomass production and triterpenoid content in the leaves of Centella asiatica (L.) Urb. from Madagascar. Chemistry and Biodiversity 9: 298–308 [DOI] [PubMed] [Google Scholar]
- Rakesh R, Sushma T, Sangeeta G, Gambir I (2013) Response of Centella asiatica in the management of age related problems among elderly with special reference to cognitive problems as per Prakriti. Int. J. res. Ayurveda Pharm, pp 163–167 [Google Scholar]
- Ramesh B, Girish T, Raghavendra R, Naidu K, Rao U, Rao K (2014) Comparative study on anti-oxidant and anti-inflammatory activities of Caesalpinia crista and Centella asiatica leaf extracts. J Pharm Bioallied Sci 6: 86–91 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rangel-Ordóñez L, Nöldner M, Schubert-Zsilavecz M, Wurglics M (2010) Plasma levels and distribution of flavonoids in rat brain after single and repeated doses of standardized Ginkgo biloba extract EGb 761®. Planta Med 76: 1683–1690 [DOI] [PubMed] [Google Scholar]
- Rao MA, Srinivasan K, Rao To (1973) The effect of Mandookaparni (Centella asiatica) on the general mental ability (Medhya) of mentally retarded children In: Unit for Research in Indian Medicine VHSMC, Adyar, Madra (ed), Jour. Res. Ind. med, pp 9–16 [Google Scholar]
- Rhein V, Baysang G, Rao S, Meier F, Bonert A, Müller-Spahn F, Eckert A (2009) Amyloid-beta leads to impaired cellular respiration, energy production and mitochondrial electron chain complex activities in human neuroblastoma cells.. Cell Mol Neurobiol 29: 1063–1071 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rush WR, Murray GR, Graham DJ (1993) The comparative steady-state bioavailability of the active ingredients of Madecassol. Eur J Drug Metab Pharmacokinet 18: 323–326 [DOI] [PubMed] [Google Scholar]
- Sakina MR, Dandiya Pe (1990) A Psycho-Neuropharmacologi- cal Profile of CentellaasiaticaExtract.. Fitoterapia 61: 291–296 [Google Scholar]
- Sangwan RS, Tripathi S, Singh J, Narnoliya LK, Sangwan NS (2013) De novo sequencing and assembly of Centella asiatica leaf transcriptome for mapping of structural, functional and regulatory genes with special reference to secondary metabolism. Gene 525: 58–76 [DOI] [PubMed] [Google Scholar]
- Sasaki K, Han J, Shimozono H, Villareal M, Isoda H (2013) Caffeoylquinic acid-rich purple sweet potato extract, with or without anthocyanin, imparts neuroprotection and contributes to the improvement of spatial learning and memory of SAMP8 mouse. J Agric Food Chem. 61: 5037–5045 [DOI] [PubMed] [Google Scholar]
- Scherbl D, Renouf M, Marmet C, Poquet L, Cristiani I, Dahbane S, Emady-Azar S, Sauser J, Galan J, Dionisi F, Richling E (2016) Breakfast consumption induces retarded release of chlorogenic acid metabolites in humans. European Food Research and Technology: 1–16 [Google Scholar]
- Senger DR, Li D, Jaminet SC, Cao S (2016) Activation of the Nrf2 Cell Defense Pathway by Ancient Foods: Disease Prevention by Important Molecules and Microbes Lost from the Modern Western Diet. PlosOne Feb 17: 1–40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma PD, Surana SJ, Jadav RB, Patel PH (2011) Bioanalytical HPLC method development and validation for quantification of asiatic acid from centella asiatica linn. International Journal of Pharmaceutical Sciences Review and Research 10: 46–50 [Google Scholar]
- Shinomol G, Muralidhara MMB (2011) Exploring the role of “Brahmi” (Bocopa monnieri and Centella asiatica) in brain function and therapy. Recent Pat Endocr Metab Immune Drug Discov 5: 33–49 [DOI] [PubMed] [Google Scholar]
- Shinomol G, Muralidhara. (2008a) Effect of Centella asiatica leaf powder on oxidative markers in brain regions of prepubertal mice in vivo and its in vitro efficacy to ameliorate 3-NPA-induced oxidative stress in mitochondria. Phytomedicine. 15: 971–984 [DOI] [PubMed] [Google Scholar]
- Shinomol G, Muralidhara. (2008b) Prophylactic neuroprotective property of Centella asiatica against 3-nitropropionic acid induced oxidative stress and mitochondrial dysfunctions in brain regions of prepubertal mice. Neurotoxicology 29: 948–957 [DOI] [PubMed] [Google Scholar]
- Singh J, Singh Sangwan R, Gupta S, Saxena S, Sangwan NS (2015) Profiling of triterpenoid saponin content variation in different chemotypic accessions of Centella asiatica L. Plant Genetic Resources: Characterisation and Utilisation 13: 176–179 [Google Scholar]
- Sirichoat A, Chaijaroonkhanarak W, Prachaney P, Pannangrong W, Leksomboon R, Chaichun A, Wigmore P, Welbat J (2015) Effects of Asiatic Acid on Spatial Working Memory and Cell Proliferation in the Adult Rat Hippocampus. Nutrients 7: 8413–8423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soumyanath A, Zhang Y, Henson E, Wadsworth T, Bishop J, Gold B, Quinn J (2012) Centella asiatica Extract Improves Behavioral Deficits in a Mouse Model of Alzheimer’s Disease: Investigation of a Possible Mechanism of Action.. Int J Alzheimers Dis,: 381974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soumyanath A, Zhong YP, Gold SA, Yu X, Koop DR, Bourdette D, Gold BG (2005) Centella asiatica accelerates nerve regeneration upon oral administration and contains multiple active fractions increasing neurite elongation in vitro. Journal of Pharmacy & Pharmacology 57: 1221–1229 [DOI] [PubMed] [Google Scholar]
- Srithongkul J, Kanlayanarat S, Srilaong V, Uthairatanakij A, Chalermglin P (2011) Effects of light intensity on growth and accumulation of triterpenoids in three accessions of Asiatic pennywort (Centella asiatica (L.) Urb.). Journal of Food, Agriculture and Environment 9: 360–363 [Google Scholar]
- Srivastava S, Verma S, Gupta A, Rajan S, Rawat AKS (2014) Studies on chemotypic variation in Centella Asiatica (L.) Urban from Nilgiri Range of India. Journal of Planar Chromatography - Modern TLC 27: 454–459 [Google Scholar]
- Stalmach A, Mullen W, Barron D, Uchida K, Yokota T, Cavin C, Steiling H, Williamson G, Crozier A (2009) Metabolite profiling of hydroxycinnamate derivatives in plasma and urine after the ingestion of coffee by humans: Identification of biomarkers of coffee consumption. Drug Metabolism and Disposition 37: 1749–1758 [DOI] [PubMed] [Google Scholar]
- Stark TD, Angelov A, Hofmann M, Liebl W, Hofmann T (2013) Comparative direct infusion ion mobility mass spectrometry profiling of Thermus thermophilus wild-type and mutant cruC carotenoid extracts. Anal Bioanal Chem 405: 9843–9848 [DOI] [PubMed] [Google Scholar]
- Su D, Huang J, Song Y, Feng Y (2014) Comparative pharmacokinetics and tissue distribution study of mono-, and di-caffeoylquinic acids isomers of Ainsliaea fragrans Champ by a fast UHPLC-MS/MS method. Fitoterapia 99: 139–152 [DOI] [PubMed] [Google Scholar]
- Subash S, Essa M, Braidy N, Awlad-Thani K, Vaishnav R, Al-Adawi S, Al-Asmi A, Guillemin G (2015) Diet rich in date palm fruits improves memory, learning and reduces beta amyloid in transgenic mouse model of Alzheimer’s disease. J Ayurveda Integr Med 6: 111–120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subban R, Veerakumar A, Manimaran R, Hashim KM, Balachandran I (2008) Two new flavonoids from Centella asiatica (Linn.). Journal of Natural Medicines 62: 369–373 [DOI] [PubMed] [Google Scholar]
- Sun T, Liu B, Li P (2015) Nerve Protective Effect of Asiaticoside against Ischemia-Hypoxia in Cultured Rat Cortex Neurons. Med Sci Monit 21: 3036–3041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabassum R, Vaibhav K, Shrivastava P, Khan A, Ejaz Ahmed M, Javed H, Islam F, Ahmad S, Saeed Siddiqui M, Safhi M, Islam F (2013) Centella asiatica attenuates the neurobehavioral, neurochemical and histological changes in transient focal middle cerebral artery occlusion rats.. Neurol Sci 34: 925–933 [DOI] [PubMed] [Google Scholar]
- Thomas MT, Kurup R, Johnson AJ, Chandrika SP, Mathew PJ, Dan M, Baby S (2010) Elite genotypes/chemotypes, with high contents of madecassoside and asiaticoside, from sixty accessions of Centella asiatica of south India and the Andaman Islands: For cultivation and utility in cosmetic and herbal drug applications. Industrial Crops and Products 32: 545–550 [Google Scholar]
- Thongnopnua P (2008) High-performance liquid chromatographic determination of asiatic acid in human plasma. Thai Journal of Pharmaceutical Sciences, pp 10–16 [Google Scholar]
- Tian X, An L, Gao L, Bai J, Wang J, Meng W, Ren T, Zhao Q (2015) Compound MQA, a Caffeoylquinic Acid Derivative, Protects Against NMDA-Induced Neurotoxicity and Potential Mechanisms In Vitro. CNS Neurosci Ther 21: 575–584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tiwari S, Singh S, Patwardhan K, Ghlot S, Gambhir I (2008) Effect of Centella asiatica on mild cognitive impairment (MCI) and other common age-related clinical problems. Digst Jounral of Nanomaterials and Biostructures, pp 215–220 [Google Scholar]
- Tugizimana F, Ncube EN, Steenkamp PA, Dubery IA (2015) Metabolomics-derived insights into the manipulation of terpenoid synthesis in Centella asiatica cells by methyl jasmonate. Plant Biotechnology Reports 9: 125–136 [Google Scholar]
- Umka Welbat J, Sirichoat A, Chaijaroonkhanarak W, Prachaney P, Pannangrong W, Pakdeechote P, Sripanidkulchai B, Wigmore P (2016) Asiatic Acid Prevents the Deleterious Effects of Valproic Acid on Cognition and Hippocampal Cell Proliferation and Survival. Nutrients 8: E303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Upadhyaya S, Saikia LR (2012) Evaluation of phytochemicals, antioxidant activity and nutrient content of Centella asiatica (L.) urban leaves from different localities of Assam. International Journal of Pharma and Bio Sciences 3: 656–663 [Google Scholar]
- USDA (2016) National Plant Germplasm System, Germplasm Resources Information Network (GRIN database).
- Visweswari G, Prasad K, Chetan P, Lokanatha V, W R (2010) Evaluation of the anticonvulsant effect of Centella asiatica (gotu kola) in pentylenetetrazol-induced seizures with respect to cholinergic neurotransmission. Epilepsy Behav 17: 332–335 [DOI] [PubMed] [Google Scholar]
- Vogel H, De Souza N, D’Sa A (1990) Effect of terpenoids isolated from centella asiatica on granuloma tissue. Acta Therapeutica, pp 285–298 [Google Scholar]
- Wanakhachornkrai O, Pongrakhananon V, Chunhacha P, Wanasuntronwong A, Vattanajun A (2013) Neuritogenic effect of standardized extract of Centella asiatica ECa233 on human neuroblastoma cells. BMC Complement Altern Med 13: 204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wanasuntronwong A, Tantisira M, Tantisira B, Watanabe H (2012) Anxiolytic effects of standardized extract of Centella asiatica (ECa 233) after chronic immobilization stress in mice. J Ethnopharmacol 143: 579–585 [DOI] [PubMed] [Google Scholar]
- Wang T, Leng DD, Gao FF, Jiang CJ, Xia YF, Dai Y (2014) A LC-ESI-MS method for the simultaneous determination of madecassoside and its metabolite madecassic acid in rat plasma: Comparison pharmacokinetics in normal and collagen-induced arthritic rats. Chinese Journal of Natural Medicines 12: 0943–0951 [DOI] [PubMed] [Google Scholar]
- Wang Y, Wang Y, Li J, Hua L, Han B, Zhang Y, Yang X, Zeng Z, Bai H, Yin H, Lou J (2016) Effects of caffeic acid on learning deficits in a model of Alzheimer’s disease. Int J Mol Med 38: 869–875 [DOI] [PubMed] [Google Scholar]
- Wattanathorn J, Mator L, Muchimapura S, Tongun T, Pasuriwong O, Piyawatkul N, Yimtae K, Sripanidkulchai B, Singkhoraard J (2008) Positive modulation of cognition and mood in the healthy elderly volunteer following the administration of Centella asiatica. J. Ethnopharmacol 116: 325–332 [DOI] [PubMed] [Google Scholar]
- Wijeweera P, Arnason J, Koszycki D, Merali Z (2006) Evaluation of anxiolytic properties of Gotukola--(Centella asiatica) extracts and asiaticoside in rat behavioral models. Phytomedicine 13: 668–676 [DOI] [PubMed] [Google Scholar]
- Williamson G, Dionisi F, Renouf M (2011) Flavanols from green tea and phenolic acids from coffee: Critical quantitative evaluation of the pharmacokinetic data in humans after consumption of single doses of beverages. Molecular Nutrition and Food Research 55: 864–873 [DOI] [PubMed] [Google Scholar]
- Winkel-Shirley B (2001) Flavonoid biosynthesis. A colorful model for genetics, biochemistry, cell biology, and biotechnology. Plant Physiology 126: 485–493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wittemer SM, Ploch M, Windeck T, Muller SC, Drewelow B, Derendorf H, Veit M (2005) Bioavailability and pharmacokinetics of caffeoylquinic acids and flavonoids after oral administration of Artichoke leaf extracts in humans. Phytomedicine 12: 28–38 [DOI] [PubMed] [Google Scholar]
- Wittemer SM, Veit M (2003) Validated method for the determination of six metabolites derived from artichoke leaf extract in human plasma by high-performance liquid chromatography-coulometric-array detection. Journal of Chromatography B: Analytical Technologies in the Biomedical and Life Sciences 793: 367–375 [DOI] [PubMed] [Google Scholar]
- Wong CC, Meinl W, Glatt HR, Barron D, Stalmach A, Steiling H, Crozier A, Williamson G (2010) In vitro and in vivo conjugation of dietary hydroxycinnamic acids by UDP-glucuronosyltransferases and sulfotransferases in humans. J Nutr Biochem 21: 1060–1068 [DOI] [PubMed] [Google Scholar]
- Wong KC, Tan GL (1994) Essential Oil of Centella asiatica (L.) Urb. Journal of Essential Oil Research 6: 307–309 [Google Scholar]
- Xia B, Bai L, Li X, Xiong J, Xu P, Xue M (2015) Structural analysis of metabolites of asiatic acid and its analogue madecassic acid in zebrafish using LC/IT-MSn. Molecules 20: 3001–3019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xie C, Yu K, Zhong D, Yuan T, Ye F, Jarrell JA, Millar A, Chen X (2011) Investigation of isomeric transformations of chlorogenic acid in buffers and biological matrixes by ultraperformance liquid chromatography coupled with hybrid quadrupole/ion mobility/orthogonal acceleration time-of-flight mass spectrometry. J Agric Food Chem 59: 11078–11087 [DOI] [PubMed] [Google Scholar]
- Xu M, Xiong Y, Liu J, Qian J, Zhu L, Gao J (2012) Asiatic acid, a pentacyclic triterpene in Centella asiatica, attenuates glutamate-induced cognitive deficits in mice and apoptosis in SH-SY5Y cells. Acta Pharmacol Sin 33: 578–587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Y, Cao Z, Khan I, Luo Y (2008) Gotu Kola (Centella Asiatica) extract enhances phosphorylation of cyclic AMP response element binding protein in neuroblastoma cells expressing amyloid beta peptide. J Alzheimer’s Dis 13: 341–349 [DOI] [PubMed] [Google Scholar]
- Yang J, Qian D, Jiang S, Shang EX, Guo J, Duan JA (2012) Identification of rutin deglycosylated metabolites produced by human intestinal bacteria using UPLC-Q-TOF/MS. J Chromatogr B Analyt Technol Biomed Life Sci 898: 95–100 [DOI] [PubMed] [Google Scholar]
- Yin MC, Lin MC, Mong MC, Lin CY (2012) Bioavailability, distribution, and antioxidative effects of selected triterpenes in mice. J Agric Food Chem 60: 7697–7701 [DOI] [PubMed] [Google Scholar]
- Yoshida M, Fuchigami M, Nagao T, Okabe H, Matsunaga K, Takata J, Karube Y, Tsuchihashi R, Kinjo J, Mihashi K, Fujioka T (2005) Antiproliferative constituents from Umbelliferae plants VII. Active triterpenes and rosmarinic acid from Centella asiatica. Biol Pharm Bull 28: 173–175 [DOI] [PubMed] [Google Scholar]
- Yuan Y, Zhang H, Sun F, Sun S, Zhu Z, Chai Y (2015) Biopharmaceutical and pharmacokinetic characterization of asiatic acid in Centella asiatica as determined by a sensitive and robust HPLC-MS method. J Ethnopharmacol 163: 31–38 [DOI] [PubMed] [Google Scholar]
- Zhang FL, Wei YJ, Zhu J, Gong ZN (2008) Simultaneous quantitation of three major triterpenoid glycosides in Centella asiatica extracts by high performance liquid chromatography with evaporative light scattering detection. Biomedical Chromatography 22: 119–124 [DOI] [PubMed] [Google Scholar]
- Zhang L, Foreman DP, Grant PA, Shrestha B, Moody SA, Villiers F, Kwak JM, Vertes A (2014) In situ metabolic analysis of single plant cells by capillary microsampling and electrospray ionization mass spectrometry with ion mobility separation. Analyst 139: 5079–5085 [DOI] [PubMed] [Google Scholar]
- Zhang W, Li D, Leng A, Ai J, Du Y, Meng Y, Gao Y, Ying X (2015) Tissue distribution and excretion of the five components of Portulaca oleracea L. extract in rat assessed by UHPLC. Brazilian Journal of Pharmaceutical Sciences 51: 643–652 [Google Scholar]
- Zhang X, Wu J, Dou Y, Xia B, Rong W, Rimbach G, Lou Y (2012a) Asiatic acid protects primary neurons against C2-ceramide-induced apoptosis. Eur J Pharmacol 679: 51–59 [DOI] [PubMed] [Google Scholar]
- Zhang XG, Han T, He ZG, Zhang QY, Zhang L, Rahman K, Qin LP (2012b) Genetic diversity of Centella asiatica in China analyzed by inter-simple sequence repeat (ISSR) markers: Combination analysis with chemical diversity. Journal of Natural Medicines 66: 241–247 [DOI] [PubMed] [Google Scholar]
- Zhang XG, Han T, Zhang QY, Zhang H, Huang BK, Xu LL, Qin LP (2009) Chemical fingerprinting and hierarchical clustering analysis of Centella asiatica from different locations in China. Chromatographia 69: 51–57 [Google Scholar]
- Zheng XC, Wang SH (2009) Determination of asiatic acid in beagle dog plasma after oral administration of Centella asiatica extract by precolumn derivatization RP-HPLC. J Chromatogr B Analyt Technol Biomed Life Sci 877: 477–481 [DOI] [PubMed] [Google Scholar]