Abstract
Since chronic inflammation is associated with the pathogenesis of atherosclerosis, inflammatory cytokines might contribute to the phenotypic modulation of vascular smooth muscle cells (VSMCs). Tumor necrosis factor α (TNFα) facilitated the transformation of contractile VSMCs to the synthetic phenotype, as determined by the expression of marker proteins and a collagen gel contraction assay. Western blot analysis and a cyclooxygenase-2 (COX2) promoter assay revealed that TNFα stimulation resulted in the induction of COX2. The overexpression, silencing, or pharmacological inhibition of COX2 significantly affected TNFα-induced phenotypic conversion, and of the tested prostaglandins, only PGD2 significantly induced phenotypic conversion. ERK was significantly activated by PGD2 stimulation, and the pharmacological inhibition of ERK blocked the PGD2-induced phenotypic conversion of VSMCs. However, antagonists or agonists of PGD2 receptors did not affect VSMC conversion. In contrast, spontaneously dehydrated forms of PGD2, such as PGJ2, Δ12-PGJ2, and 15-d-PGJ2, strongly induced phenotypic conversion. A reporter gene assay showed that TNFα, PGD2, and 15-d-PGJ2 significantly activated the peroxisome proliferator-responsive element (PPRE) promoter. In addition, the overexpression or silencing of peroxisome proliferator-activated receptor δ (PPARδ) significantly influenced 15-d-PGJ2-induced phenotypic conversion. Finally, atherosclerotic neointima formation was significantly suppressed in mice lacking TNFα. In addition, mice fed celecoxib exhibited complete inhibition of carotid artery ligation-induced neointima formation. This study shows that PGD2 regulates the phenotypic conversion of VSMCs by generating an endogenous ligand of PPAR, and that this leads to neointima formation in occlusive arterial disease.
Subject terms: Lipid signalling, Atherosclerosis
Artherosclerosis: Switching mechanism revealed
A lipid compound that stimulates muscle cells to change type is instrumental in the development of arterial plaque formation in artherosclerosis. Sun Sik Bae at Pusan National University School of Medicine in Gyungnam, South Korea, and co-workers examined the role of inflammatory proteins in the development of artherosclerosis, a condition involving the build-up of scar tissue or ‘plaques’ on artery walls. The behavior of vascular smooth muscle cells (VSMCs) is crucial to plaque development because, triggered by inflammatory protein activity, the cells switch from contractile-type cells to faster proliferating VSMCs, accelerating plaque growth. The team found that a compound called prostaglandin D2, a direct by-product of inflammatory protein behavior, together with a protein regulating gene expression, are key factors triggering this VSMC change. These insights may prove valuable in developing therapies for artherosclerosis.
Introduction
Atherosclerosis is a major leading cause of sudden death in western countries1, and the pathogenesis of atherosclerosis is associated with chronic inflammation resulting from interactions between lipoproteins, immune cells, and surrounding tissues, such as the endothelium and medial layers of the arterial wall. Furthermore, the migration of monocytes into vessel walls represents the initial stage of atherosclerotic plaque development2. Thereafter, these monocytes differentiate into macrophages, which uptake oxidized low-density lipoprotein (ox-LDL) to generate fatty streaks3. In addition to forming fatty streaks, vascular smooth muscle cells (VSMCs) rapidly proliferate and migrate into arterial wall lesions where they synthesize and secrete extracellular matrix proteins to form the fibrous caps of plaques1.
VSMCs are involved in a variety of vascular physiologies, such as vasoconstriction, vascular tone, blood pressure, and blood flow. In mature blood vessels, VSMC proliferation ceases, and the expression of specialized proteins, such as myosin heavy chain (MHC), myosin light-chain kinase (MLCK), calponin, transgelin 2 (SM22α), and ion channels, is highly elevated. Unlike cardiac or skeletal muscle cells, VSMCs retain their phenotypic plasticity. For example, contractile VSMCs undergo reversible phenotypic changes in response to environmental cues and ultimately gain the ability to proliferate and carry out their contractile function4. Such phenotypic conversion and the proliferation of VSMCs are observed in vascular diseases, such as atherosclerosis, arteriosclerosis, and vascular aging5. Recently, platelet-derived growth factor (PDGF), insulin-like growth factor-1 (IGF-1), and laminin were reported to act as environmental cues that regulate VSMC phenotype6–8. However, the mechanism underlying the phenotypic conversion of VSMCs in diseased states has not been elucidated.
Much evidence supports the idea that atherosclerosis is a result of chronic inflammation. Indeed, cells of the innate and adaptive immune systems, such as monocytes, macrophages, dendritic cells (DCs), B and T cells, and mast cells, are found in atherosclerotic plaque9,10. Chronic inflammation is closely associated with lesion progression, which is characterized by VSMC migration across the internal elastic lamina into intimal or subendothelial areas1. Furthermore, lesion development at this stage is influenced by inflammatory cytokines derived from interactions between monocytes/macrophages and T cells11.
During atherosclerosis progression, inflammatory cytokines, such as interleukin-1β (IL-1β) and tumor necrosis factor α (TNFα), are involved in vascular remodeling. For example, IL-1β deficiency suppresses the progression of atherosclerosis and outward vascular remodeling in a murine model12–14. In particular, vascular remodeling is modulated by TNFα, and vascular wall thicknesses in TNFα/ApoE double-knockout mice have been reported to be thinner during the later stage of atherosclerosis15. In addition, it has been reported that atherosclerotic lesions are smaller in TNFα/ApoE double-knockout mice16,17. Thus, it appears that TNFα is an important inflammatory cytokine during the progression of atherosclerosis.
It has also been reported that TNFα can induce the expression of cyclooxygenase-2 (COX2) in a variety of cell types, such as renal outer medulla cells, dorsal root ganglion cells, carcinoma cells, endometrial cells, and chondrocytes18–22, and that the induction of COX2 by TNFα plays a key role in VSMC proliferation23. COX regulates the rate-limiting step of the production of thromboxane A2 (TXA2) and prostaglandins, such as prostaglandin D2 (PGD2), prostaglandin I2 (PGI2), prostaglandin E2 (PGE2), and prostaglandin F2α (PGF2α)24. Furthermore, prostaglandins have diverse effects on VSMCs; for example, they induce contraction, relaxation, and proliferation25–28. In particular, PGD2 is metabolized to generate additional inflammatory cytokines, such as prostaglandin J2 (PGJ2), Δ12-prostaglandin J2 (Δ12-PGJ2), 15-deoxy-Δ12,14-prostaglandin J2 (15-d-PGJ2), Δ12-prostaglandin D2 (Δ12-PGD2), and 13,14-dihydro-15-keto-PGD2 (DK-PGD2)29,30; of these metabolites, PGJ2 prostaglandins are known to be endogenous ligands of peroxisome proliferator-activated receptor (PPAR)31,32.
PPARs are members of a superfamily of nuclear receptors, which includes PPARα, PPARδ, and PPARγ33, and PPARs have been reported to play diverse roles in the vasculature. For instance, it has been reported that the smooth muscle-specific overexpression of PPARγ represses smooth muscle marker gene expression34, that PPARδ is upregulated during vascular lesion formation, and that its overexpression enhances VSMC proliferation35. Therefore, the underlying mechanisms of PPARs in the vasculature remain unclear. In the present study, we investigated critical inflammatory cytokines that determine the phenotypic switching of VSMCs and attempted to define the molecular mechanisms underlying inflammatory cytokine-mediated signaling pathways.
Materials and methods
Animals
Mice lacking TNFα (TNFα−/−, B6.129S-Tnftm1Gkl/J mice) and ApoE (ApoE−/−, C57B6.129P2-Apoetm1Unc/J mice) were purchased from The Jackson Laboratory (Bar Harbor, Maine, USA). TNFα−/− mice were crossed with ApoE-deficient mice (ApoE−/−) to generate mice heterozygous at both loci. TNFα+/−ApoE−/− mice were intercrossed to produce TNFα+/+ApoE−/− and TNFα−/−ApoE−/− littermates. Animals were housed under specific pathogen-free conditions. All animal procedures were performed in accordance with our institutional guidelines for animal research and were approved by our institutional animal care and use committee (PNU-2016-1195). The investigation conformed with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996).
Cell preparation and cell culture
VSMCs were isolated from 4-week-old male Sprague-Dawley rats by using a tissue explantation method. Briefly, rats were euthanized by intravenous ketamine (100 mg/kg) injection and perfused with phosphate-buffered saline (PBS) for 5 min. The thoracic aorta was aseptically isolated, and the surrounding fat and connective tissues were discarded. Vessels were cut longitudinally, and the lumen sides were scraped with a razor blade to remove the intima. Vessels were cut into 3–5-mm lengths and explanted the lumen side down onto collagen-coated culture dishes. Seven days after explantation, the tissue fragments were discarded, and sprouted VSMCs (referred to as P0-stage VSMCs) were collected.
Preparation of contractile phenotype VSMCs and phenotypic conversion
To prepare contractile VSMCs, P0-stage VSMCs were differentiated as described previously8. The differentiation of VSMCs was verified by western blotting for SMC marker proteins, namely, MHC, MLCK, SM22α, calponin, and smooth muscle actin (SMA). Contractile VSMCs were cultured with medium containing the indicated inflammatory cytokines for 4 days. To examine the facilitation of phenotypic conversion by PPARδ overexpression, cells were incubated with the indicated cytokines for 2 days.
Immunocytochemistry
For immunocytochemistry, cells were washed with ice-cold PBS and fixed with 4% paraformaldehyde for 10 min. The cells were permeabilized with 0.2% Triton X-100, incubated with the indicated primary antibodies for 1 h, and then treated with Cy3- or Alexa Fluor 488-conjugated secondary antibodies for 30 min. The samples were mounted with anti-fading reagent (2% n-propyl gallate in 80% glycerol/PBS solution), and images were obtained by using a confocal microscope (FV1000-ZDC, Olympus, Tokyo Japan).
Western blotting
Cell lysates were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis on 10% polyacrylamide gels under reducing conditions. The proteins were transferred to nitrocellulose membranes, which were immunoblotted by using the indicated primary antibodies and IRDye-conjugated secondary antibodies (Li-COR Biosciences, Lincoln, NE, USA). The western blots were developed by using the Odyssey system (Li-COR Biosciences).
Collagen gel contraction assay
VSMCs were isolated by trypsin digestion and resuspended in serum-free DMEM (1 × 106 cells/ml). The cell suspensions were mixed on ice with collagen gel solution (8 mg/ml collagen type I in 2× PBS, pH 8.0) to give 5 × 105 cells/ml and 4 mg/ml collagen gel solution. One hundred microliters of VSMC–collagen gel mixture was added to 12-well plates, which were then incubated for 30 min at 37 °C to enable polymerization. The gels were then floated in serum-free DMEM for 5 h, and angiotensin II (AngII, 1 μM) was added to initiate contraction, while images were captured by using a digital charge-coupled device camera. Collagen gel contraction was defined as a decrease in the gel area as determined by Scion Image software (compliments of Scion Corporation, Frederick, MD; http://www.scioncorp.com). Relative gel areas were obtained by dividing the areas by the initial areas.
Plasmids and promoter assay
pGL3-SMA and pGL3-SM22α were kindly provided by Dr. Gary K. Owens (University of Virginia). pGL3-myocardin was prepared as described previously8. pGL3-PPRE and pCDNA3.1-PPARδ were provided by Dr. Jang Hyun Choi (UNIST, Ulsan, Korea). Human COX2 cDNA was subcloned into the BamHI/EcoRI site of the pMIGR2 vector. To assess promoter activity, we used a dual-luciferase reporter assay system. VSMCs were plated in 12-well plates and cotransfected with the luciferase reporter constructs and the renilla luciferase plasmids by using Lipofectamine 2000 (Invitrogen, Carlsbad, CA, USA). Each well contained 0.88 μg of luciferase reporter plasmid, 0.8 μg of expression vector, and 80 ng of renilla luciferase plasmid. The medium was replaced with fresh medium 7 h post transfection, and the cells were lysed and assayed for luciferase activity 24 h post transfection. The protein extracts (20 μl) were analyzed by using a GlomaxTM 20/20 luminometer (Promega, WI, USA).
Short-hairpin RNA and constructs
To silence COX2 and PPARδ, shCOX2 (5′-CCG GTA AGA CAG ATC AGA AGC GAG GAC TCG AGT CCT CGC TTC TGA TCT GTC TTT TTT TG-3′) and shPPARδ (5′-CCG GTG CAA GCC CTT CAG TGA CAT CAC TCG AGT GAT GTC ACT GAA GGG CTT GCT TTT TG-3′) oligonucleotides with an AgeI site at the 5′-end site and an EcoRI site at the 3′ end were designed, and then sense and antisense oligonucleotides were synthesized (XENOTECH, Daejeon, Korea). Both complementary oligonucleotides were mixed, heated at 98 °C for 5 min, and cooled to room temperature. The annealed nucleotides were subcloned into the AgeI/EcoRI sites of the pLKO.1 lentiviral vector.
Lentiviral knockdown
For gene silencing, HEK293-FT packaging cells (Invitrogen) were grown to ~70% confluence in six-well plates. The cells were triple transfected with 6 μg of the pLKO.1 lentiviral construct, 1 μg of Δ8.9, and 1 μg of pVSV-G by using a calcium phosphate method. The medium was replaced with fresh medium 8 h post transfection. Lentiviral supernatants were harvested 24 h post transfection and passed through 0.45-μm filters. Cell-free viral culture supernatants were used to infect contractile VSMCs in the presence of 8 μg/ml polybrene (Sigma-Aldrich, St. Louis, MO, USA). Additional rounds of infection were performed 48 and 72 h post transfection. Infected cells were isolated by selection by using 10 μg/ml puromycin for 2 days.
Carotid artery ligation, induction of atherosclerosis, and immunohistochemistry
To induce neointima formation, the left common carotid artery of male mice (6 weeks old) was ligated proximal to the bifurcation, and 4 weeks later, both the left and right common carotid arteries were isolated. To treat the mice with celecoxib, the mice were fed a D12450B diet containing 1500 ppm of celecoxib (Central Lab. Animal Inc., Seoul) for 2 weeks before ligation and fed a diet containing celecoxib for an additional 4 weeks. Atherosclerosis was induced by feeding male mice (8 weeks old) a high-fat western-type diet containing 1.25% cholesterol (Research Diets, D12108) for 15 weeks. After killing, the mice were perfused with PBS, and the isolated carotid arteries and aortas were fixed in 4% paraformaldehyde at 4 °C overnight and embedded in paraffin for immunohistochemistry. Five-micrometer sections of each block were either stained with H&E or the indicated primary antibodies. Staining was visualized by using a MIRAX MIDI Versatile Digital Slide Scanner (Carl Zeiss, Jena, Germany) or by confocal microscopy (Olympus). For oil red O staining, aortic tissues were stained with oil red O solution (0.5% in isopropyl alcohol) for 2 h and washed with distilled water four times. For quantification, oil red O was extracted with isopropyl alcohol (1 ml), and the absorbance was measured at 518 nm.
Statistical analysis
The data were plotted and analyzed by using GraphPad Prism. Unpaired Student’s t test (two tails) was used to determine the significance of intergroup differences. Multiple sets of data were analyzed by analysis of variance (one-way ANOVA) and Tukey’s multiple comparison test. The results are expressed as the means ± SEMs, and P values less than 0.05 were considered significant.
Results
Phenotypic changes in VSMCs induced by TNFα
Since VSMC sprouting after the explantation of aortic tissue fragments on collagen-coated plates is indicative of the synthetic phenotype, we employed a previously described differentiation protocol8. Plating VSMCs on laminin-coated plates initiated differentiation, as shown in Fig. 1a, b. P0-stage VSMCs did not respond to AngII stimulation, whereas P4-stage VSMCs rapidly contracted in response to AngII stimulation (Fig. 1c). However, the differentiation of established cell lines, such as A10 cells, did not result in contraction in response to AngII stimulation (Supplementary Fig. S1). The treatment of contractile VSMCs (P4 stage) with TNFα significantly reduced the expression of SMC marker proteins (Fig. 1d, e) and the promoter activity of myocardin, SMA, and SM22α (Supplementary Fig. S2). Furthermore, TNFα treatment resulted in the loss of AngII-dependent contraction (Fig. 1f).
Fig. 1. Phenotypic conversion of contractile VSMCs by TNFα.
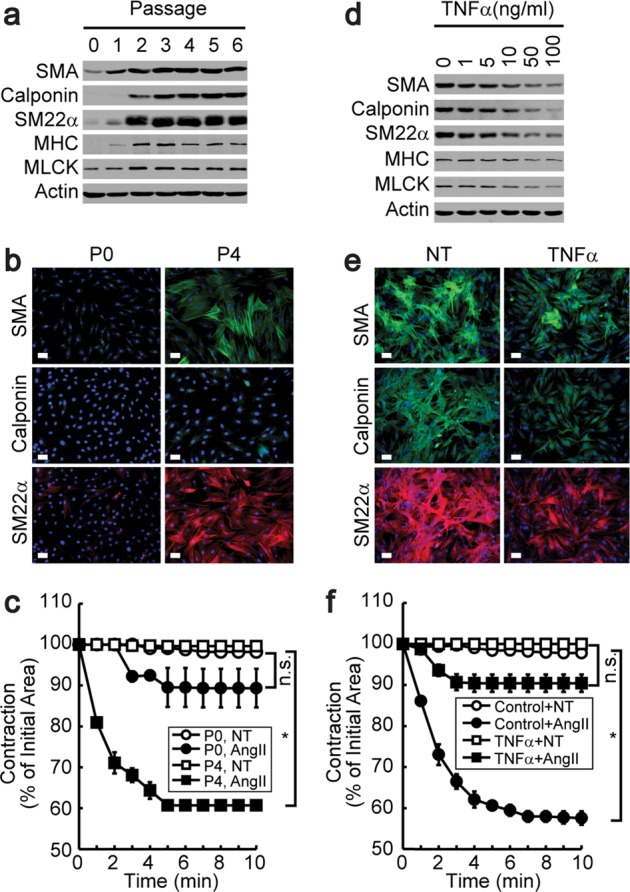
Synthetic VSMCs isolated from rat aortic tissue fragments were explanted onto laminin-coated plates. The expression of contractile marker genes was assessed by western blot analysis (a) or immunocytochemistry (b) at the indicated passages. Magnification, 40×. Bar, 100 μm. c Cells from each passage were embedded in collagen gel beads and stimulated with AngII. Time-lapse images were recorded digitally, and contractions are expressed as the percentage of the initial area (n = 3). *P < 0.05 compared with the no- treatment (NT) group. n.s., not significant. P4-stage VSMCs were stimulated with TNFα for 4 days at the indicated doses, and the expression of contractile marker genes was assessed by western blot analysis (d) and immunocytochemistry (e). Magnification, 40×. f Contractile VSMCs were stimulated with TNFα for 4 days, and AngII-induced collagen gel contraction was analyzed as described above (n = 3). *P < 0.05 compared with the no-treatment (NT) group. n.s. not significant. The results are presented as the means ± SEM. One-way ANOVA and Tukey’s multiple comparison test were used to determine the P values. The asterisks indicate statistical significance (P < 0.05).
COX2 was required for TNFα-induced phenotypic changes in VSMCs
COX2 expression was significantly reduced during the differentiation of VSMCs from the synthetic to the contractile phenotype (Supplementary Fig. S3a), whereas the stimulation of contractile VSMCs with TNFα resulted in the induction of COX2 (Fig. 2a, b). In addition, the stimulation of VSMCs with TNFα strongly enhanced the promoter activity of COX2 (Fig. 2c). Furthermore, ectopic COX2 expression significantly reduced the expression of marker proteins, such as SMA and SM22α, and the promoter activity of SM22α (Fig. 2d, e). In addition, the pharmacological inhibition of COX2 by celecoxib (10 μM) markedly inhibited the TNFα-induced phenotypic conversion of contractile VSMCs (Fig. 2f). The silencing of COX2 blunted the TNFα-induced phenotypic conversion of contractile VSMCs and preserved the expression of contractile phenotype marker proteins and AngII-dependent contraction (Fig. 2g, h, Supplementary Fig. S3b).
Fig. 2. COX2 was required for TNFα-induced VSMC phenotypic changes.
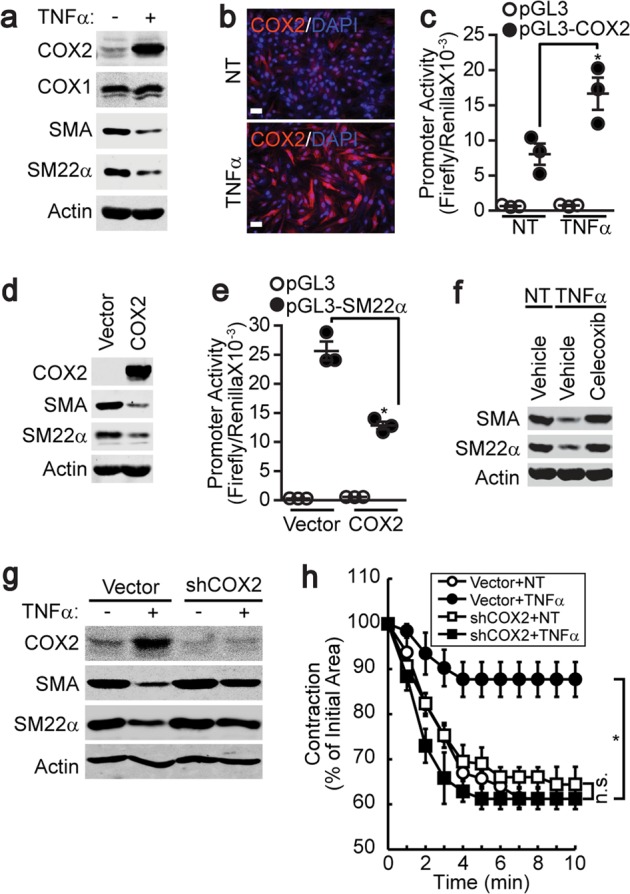
P4-stage VSMCs were stimulated with TNFα for 4 days, and the expression levels of COX2 and contractile marker genes were assessed by western blot analysis (a) and immunocytochemistry (b). Magnification, 40×. Bar, 100 μm. c The COX2 promoter was subcloned into the pGL3 vector and was transfected into P4-stage VSMCs (n = 3). The TNFα-induced activation of the COX2 promoter was assessed as described in the “Materials and methods” section. COX2 was ectopically expressed in P4-stage VSMCs, and the expression levels of COX2, SMA, and SM22α (d) and SM22α promoter activity (e) were examined (n = 3). f P4-stage VSMCs were pretreated with a selective COX2 inhibitor (celecoxib), and TNFα-dependent gene expression changes were examined. COX2 was silenced in P4-stage VSMCs, and TNFα-dependent gene expression (g) and the loss of contractility were evaluated (n = 3) (h). *P < 0.05 compared with the untreated group. n.s. not significant. The results are presented as the means ± SEM. One-way ANOVA and Tukey’s multiple comparison test were used to determine the P values. The asterisks indicate statistical significance (P < 0.05).
PGD2 regulated the phenotypic changes in VSMCs independently of the DP1 and DP2 receptor signaling pathways
Since COX2 plays a key role in the rate-limiting step of prostaglandin synthesis, we examined the effect of prostaglandins on the expression of SMC marker proteins. The stimulation of contractile VSMCs with PGD2 (10 μM) significantly downregulated the expression of SMC marker proteins, whereas other prostaglandins had no effect (Fig. 3a, Supplementary Fig. S4a, b). In addition, the stimulation of contractile VSMCs with PGD2 abrogated AngII-dependent contraction (Fig. 3b). The stimulation of contractile VSMCs with PGD2 significantly activated the ERK signaling pathway in a time-dependent manner (Supplementary Fig. S4c), and the inhibition of ERK blunted PGD2-induced VSMC phenotypic changes (Fig. 3c). DP1 and DP2, which couple to Gs and Gi, respectively, regulate cAMP production and are known to be PGD2 receptors36. However, the treatment of contractile VSMCs with PGD2, a DP1 agonist (BW245C, 10 μM), or a DP2 agonist (DK-PGD2, 10 μM) did not change cAMP production (Supplementary Fig. S5a–d). In addition, the expression of DP1 and DP2 was not observed in either synthetic or contractile VSMCs (Supplementary Fig. S5e). PGD2 did not induce calcium mobilization, which is mediated by the activation of Gq and phospholipase C-β (Supplementary Fig. S5f–h). Since the ERK signaling pathway is important for the PGD2-dependent suppression of SMC marker gene expression, we examined whether the PGD2 receptors DP1 and DP2 are involved in the activation of the ERK signaling pathway. As shown in Fig. 3d, the inhibition of MEK by PD98059 (10 μM) completely blocked the PGD2-induced activation of ERK. However, the inhibition of DP1 (BWA868C, 10 μM) or DP2 (TM-30089, 10 μM) or the simultaneous inhibition of DP1 and DP2 did not block the PGD2-induced activation of ERK. In addition, a selective agonist of DP1 (BW245C, 10 μM) or DP2 (DK-PGD2, 10 μM) did not induce the activation of ERK (Fig. 3e). Furthermore, neither agonists nor antagonists of DP1 and DP2 affected the expression of SMC marker proteins (Fig. 3f, g).
Fig. 3. DP1- and DP2-independent VSMC phenotypic changes induced by PGD2.

a P4-stage VSMCs were stimulated with the indicated prostanoids or a thromboxane analog (U46619) for 4 days. Cell lysates were examined for the expression of SMA and SM22α. b P4-stage VSMCs were stimulated with TNFα or PGD2 for 4 days, and AngII-dependent contraction was analyzed (n = 2). *P < 0.05 compared with the no-treatment (NT) group. c P4-stage VSMCs were pretreated with the indicated inhibitors (PD, PD98059; SP, SP600125; SB, SB203580), and PGD2-dependent changes in the expression of SMA and SM22α were assessed. d P4-stage VSMCs were pretreated with a MEK, DP1, or DP2 inhibitor (PD98059, BWA868C, or TM-30089) and then stimulated with PGD2. Cell lysates were analyzed for ERK activation. e P4-stage VSMCs were stimulated with a DP1 or DP2 agonist (BW245C (upper panel) or DK-PGD2 (lower panel)), and ERK activation was assessed. f P4-stage VSMCs were pretreated with a DP1 or DP2 antagonist (BWA868C or TM-30089), and PGD2-dependent gene expression changes in SMA and SM22α were examined. g P4-stage VSMCs were stimulated with a DP1 or DP2 agonist (BW245C (upper panel) or DK-PGD2 (lower panel)), and the expression levels of SMA and SM22α were assessed. The results are presented as the means ± SEM. One-way ANOVA and Tukey’s multiple comparison test were used to determine the P values. The asterisks indicate statistical significance (P < 0.05).
The PGJ2 series of PGD2 metabolites affected VSMC phenotypes
PGD2 generates inflammatory metabolites, such as Δ12-PGD2 and 15-d-PGJ229,30. Since our results showed that DP1 and/or DP2 receptors did not affect VSMC phenotypic changes, we hypothesized that PGD2 metabolites are involved in the phenotypic modulation of VSMCs. As shown in Fig. 4a, the PGJ2 series of PGD2 metabolites significantly induced VSMC phenotypic changes, whereas metabolites of other series had no effect. In addition, PGJ2 series metabolites significantly induced ERK activation, whereas other series of metabolites did not (Supplementary Fig. S6). Since PGD2 and PGJ2 are endogenous ligands of PPAR31,32, we explored the effect of PGD2 metabolites on PPRE promoter activity. As shown in Fig. 4b, TNFα, PGD2, and 15-d-PGJ2 significantly elevated PPRE promoter activity, whereas 15-d-PGD2, which did not induce VSMC phenotypic changes, had no effect. In addition, VSMCs mainly expressed PPARδ and PPARγ (Supplementary Fig. S7a, b); the selective inhibition of PPARδ (GSK3787, 10 μM) and PPARγ (GW9662, 10 μM) suppressed 15-d-PGJ2-induced phenotypic changes in contractile VSMCs, whereas the inhibition of PPARα (GW6471, 10 μM) had no effect (Fig. 4c). Since 15-d-PGJ2 significantly activated the PPARδ-dependent activation of the PPRE promoter (Fig. 4d), we investigated the effect of 15-d-PGJ2 in the presence or absence of PPARδ. The overexpression of PPARδ significantly enhanced contractile VSMC phenotypic changes in the presence of 15-d-PGJ2 (Fig. 4e). Furthermore, the silencing of PPARδ inhibited 15-d-PGJ2-induced contractile VSMC phenotypic changes (Fig. 4f). The proliferation of contractile VSMCs was significantly lower than that of synthetic VSMCs (Fig. 4g), whereas the proliferation of contractile VSMCs was enhanced by the overexpression of PPARδ (Fig. 4h). In addition, the stimulation of abdominal aortic fragments with 15-d-PGJ2 facilitated VSMC sprouting ex vivo (Fig. 4i).
Fig. 4. PPARδ was required for PGD2- and 15-d-PGJ2-dependent VSMC phenotypic changes.

a P4-stage VSMCs were stimulated with the indicated PGD2 metabolites, and the expression levels of SMA and SM22α were assessed by western blot analysis. b The promoter activity of PPRE was verified in the presence of the indicated stimuli (n = 3). c P4-stage VSMCs were pretreated with a PPARα, PPARδ, or PPARγ antagonist (GW6471, GSK3787, or GW9662) and then stimulated with 15-d-PGJ2. The expression levels of SMA and SM22α were assessed by western blot analysis. d Contractile VSMCs were infected with a retrovirus carrying either vector or the PPARδ construct, and the promoter activity of PPRE was verified in the presence of the indicated stimuli (n = 3). GW501516 (10 μM) was used as a positive control for the PPARδ agonist. e FLAG-tagged PPARδ was expressed in P4-stage VSMCs, and the expression levels of SMA and SM22α were assessed after stimulation with 15-d-PGJ2. f PPARδ was silenced in P4-stage VSMCs, and the expression of SMA and SM22α was assessed after cells were stimulated with 15-d-PGJ2. g The proliferation of P0- or P4-stage VSMCs was measured (n = 10). h Vector or PPARδ was expressed in P4-stage VSMCs, and proliferation was measured (n = 5). i Aortic tissues were isolated, the endothelial and adventitial layers were discarded, and the aortic tissue fragments were placed on collagen-coated plates and then stimulated with 15-d-PGJ2 for 7 days. Proliferating VSMCs are indicated by dashed lines. The results are presented as the means ± SEM. One-way ANOVA and Tukey’s multiple comparison test were used to determine the P values. The asterisks indicate statistical significance (P < 0.05).
Atherosclerotic neointima formation was attenuated in mice lacking TNFα
To assess the role of TNFα in the regulation of the VSMC phenotype in vivo, mice lacking both ApoE and TNFα (ApoE−/−TNFα−/−) were fed a high-fat western diet, and neointima formation was examined. As shown in Fig. 5a, mice fed a high-fat diet exhibited significantly induced COX2 expression in the inflamed medial layers and plaque lesions, whereas COX2 expression was abolished in mice lacking TNFα. Macrophages were observed only in plaque lesions in both genotypes. Neointima formation was significantly lower in ApoE−/−TNFα−/− mice than in ApoE−/−TNFα+/+ mice (Fig. 5b). To avoid inadvertent errors caused by the locations of the sections, we also examined neointima formation in the aortic sinuses. As shown in Fig. 5c, neointima formation in mice lacking TNFα was significantly reduced.
Fig. 5. Suppression of atherosclerosis in mice lacking TNFα.
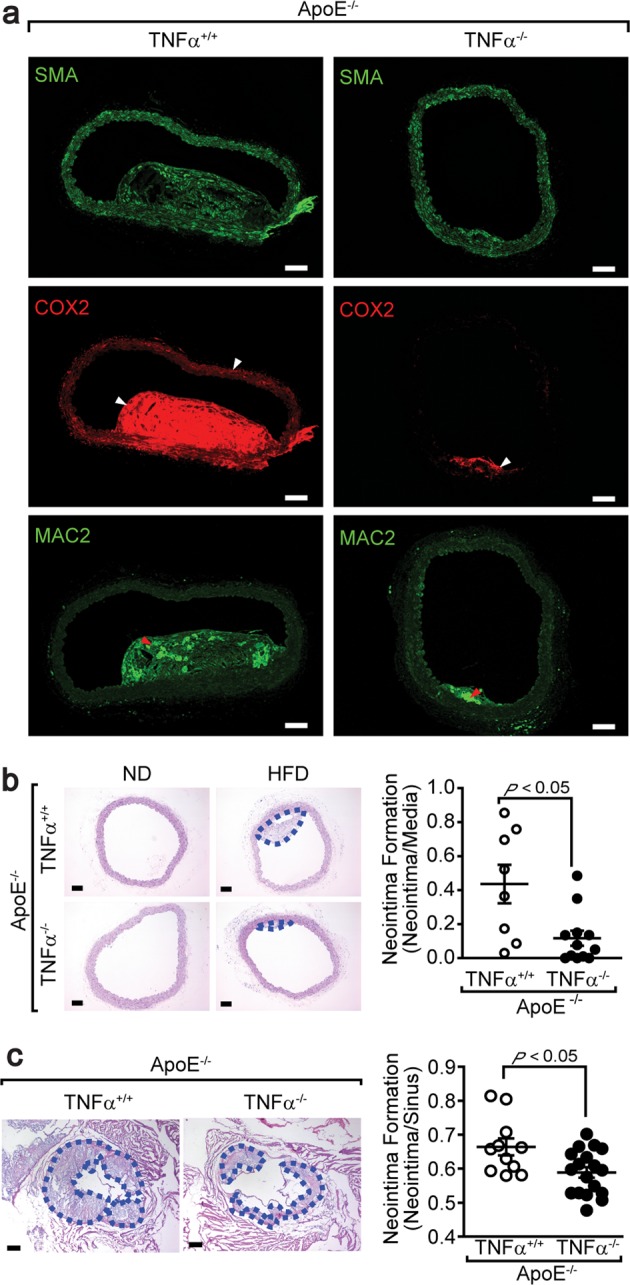
a Mice lacking both ApoE and TNFα were fed a high-fat diet for 15 weeks. Aortic tissues were isolated and stained with the indicated antibodies. The white and red arrows indicate COX2 expression and macrophages, respectively. Bar, 25 μm. The mice were fed either a normal or high-fat diet for 15 weeks. Aortas (b) (ApoE−/−TNFα+/+ mice, n = 8; ApoE−/−TNFα−/− mice, n = 12) and aortic sinuses (c) (ApoE−/−TNFα+/+ mice, n = 11; ApoE–/–TNFα–/– mice, n = 18) were stained with hematoxylin and eosin, and the neointimal areas were measured. The dashed blue lines indicate neointimal tissues. Bar, 50 μm. The results are presented as the means ± SEM. Unpaired Student’s t test (two tails) was used to determine the P values.
Carotid artery ligation-induced neointima formation was attenuated in mice lacking TNFα or fed celecoxib
We next adapted the carotid artery ligation model to determine whether TNFα and COX2 are required for neointima formation. In control mice, the ligation of the left common carotid artery distal to the aortic arch near the bifurcation markedly induced neointima formation, whereas neointima formation was significantly lower in mice lacking TNFα (Fig. 6a). In addition, mice fed the selective COX2 inhibitor celecoxib exhibited completely blocked carotid artery ligation-induced neointima formation (Fig. 6b). Overall, plaque formation was significantly decreased in mice lacking TNFα, as assessed by oil red O staining (Fig. 6c).
Fig. 6. Suppression of neointima formation by COX2 inhibition.
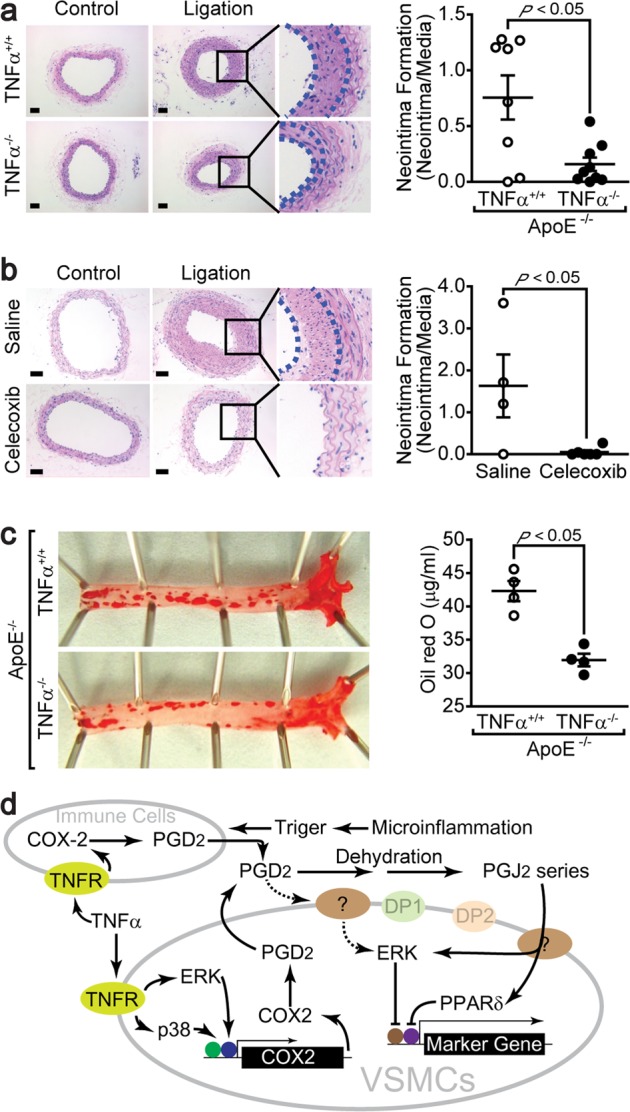
a The carotid arteries of mice lacking TNFα were ligated for 4 weeks (ApoE−/−TNFα+/+ mice, n = 8; ApoE−/−TNFα−/− mice, n = 9). Representative photomicrographs of cross sections of carotid arteries stained with hematoxylin and eosin are shown. The dashed line defines the medial and neointimal layers. Bar, 25 μm. b The mice were fed a diet containing a selective COX2 inhibitor (celecoxib) for 2 weeks before carotid artery ligation and fed a diet containing celecoxib for an additional 4 weeks (saline, n = 4; celecoxib, n = 6). Aortic tissues were isolated, and cross sections from 2 mm distal to the ligation were stained with hematoxylin and eosin. The dashed lines define the medial and neointimal layers. Bar, 25 μm. c The mice were fed a high-fat diet for 15 weeks, abdominal aortas were isolated and stained with oil red O, and lipid accumulation was quantified by extracting oil red O with isopropyl alcohol (n = 4). d A schematic representation of the PGD2-mediated phenotypic modulation of VSMCs by PPARδ. The results are presented as the means ± SEM. Unpaired Student’s t test (two tails) was used to determine the P values.
Discussion
The present study demonstrates that TNFα regulates the dedifferentiation of contractile VSMCs to the synthetic phenotype via the induction of COX2 and the production of prostaglandins, such as PGD2 and PGJ2. In addition, it shows that the loss of TNFα and the inhibition of COX2 suppresses the induction of intimal thickening in mice. Consequently, we propose that the PGD2-mediated activation of PPARδ is a potential target for the inhibition of neointima formation.
It has been reported that vascular remodeling is closely associated with chronic inflammation9,37. Therefore, it is possible that inflammatory cytokines play pivotal roles in vascular remodeling, particularly in the phenotypic switching of VSMCs. In particular, TNFα has been implicated in the pathogenesis of atherosclerosis; for example, the disruption of TNFα has been reported to delay the progression of atherosclerosis and to reduce lesion sizes15,17. The production of TNFα by cells of the hematopoietic lineage appears to be important because the transplantation of bone marrow from mice lacking TNFα has been found to reduce the severity of atherosclerosis16. Likewise, our results show that atherosclerotic lesion size was significantly reduced in mice lacking TNFα (Fig. 5). One of the major roles of TNFα seems to be involved in VSMC phenotypic changes. For instance, the stimulation of contractile VSMCs suppresses the expression of smooth muscle marker genes and results in the loss of contractile function in smooth muscle cells (Fig. 1). The consequence of TNFα-induced VSMC phenotypic changes is VSMC hyperproliferation since synthetic VSMCs grow faster than contractile VSMCs in vitro (Fig. 4g)8. Hence, we suggest that TNFα stimulates phenotypic conversion in smooth muscle, thereby enhancing VSMC proliferation and causing neointima formation.
Although the mechanisms underlying the TNFα-mediated phenotypic conversion of VSMCs remain unclear, recent evidence supports the role of COX2 induction during vascular remodeling. TNFα can induce the expression of COX2 in various cell types18–22, and likewise, we found that TNFα stimulates the promoter activity of COX2 and thereby enhances COX2 expression in contractile VSMCs (Fig. 2a–c). Several lines of evidence support the role of COX2 induction by TNFα in contractile VSMC phenotypic changes. First, the silencing or overexpression of COX2 significantly affected smooth muscle marker gene expression and contractile function in VSMCs (Fig. 2d, e, g, h, Supplementary Fig. S3b). Second, the inhibition of COX enzymatic activity suppressed TNFα-induced contractile VSMC phenotypic changes and carotid artery ligation-induced neointima formation (Figs. 2f, 6b). Third, it has been reported that the induction of COX2 by TNFα is required for VSMC proliferation23. Therefore, these results suggest that the TNFα-dependent induction of COX2 and COX2 enzymatic activity are required for VSMC phenotypic conversion.
Prostanoids, which consist of prostaglandins and thromboxanes, are produced by COX family members, and our results show that PGD2 (a prostaglandin produced by COX2) seems to be a major factor in VSMC phenotypic changes (Fig. 3a, Supplementary Fig. S4a, b). Likewise, it has been reported that lipocalin-type prostaglandin D synthase serum levels are correlated with coronary artery disease severity38. Two PGD2 receptors, namely, DP1 and DP2, have been identified thus far39, but neither of these two receptors seems to be involved in PGD2-mediated VSMC phenotypic changes. For instance, in the present study, neither the inhibition nor activation of DP1 and/or DP2 affected VSMC phenotypic change (Fig. 3f, g). In line with this, no significant difference in atherosclerosis is found in mice lacking the DP1 receptor40. In addition, the expression of DP1 and DP2 was not observed in VSMCs, and the stimulation of VSMCs with PGD2 did not alter the levels of cAMP, which is a second messenger of the DP1 and DP2 receptor signaling pathways (Supplementary Fig. S5). The ERK signaling pathway plays an important role in PGD2-induced VSMC phenotypic changes. For example, the stimulation of contractile VSMCs with PGD2 strongly activated ERK, and the inhibition of ERK signaling completely blocked PGD2-induced VSMC phenotypic changes (Fig. 3c, d), indicating that PGD2 induces VSMC phenotypic changes by a DP1- or DP2-independent mechanism.
There are two mechanistic possibilities for PGD2-induced VSMC phenotypic changes: (1) a novel receptor that activates the ERK signaling pathway and (2) the metabolic conversion of PGD2. PGD2 is metabolized to lipid mediators, such as prostaglandin J2 (PGJ2), Δ12-prostaglandin J2 (Δ12-PGJ2), 15-deoxy-Δ12,14-prostaglandin J2 (15-d-PGJ2), Δ12-prostaglandin D2 (Δ12-PGD2), and 13,14-dihydro-15-keto-PGD2 (DK-PGD2)29,30. Indeed, in the present study, PGJ-type PGD2 metabolites significantly induced VSMC phenotypic changes (Fig. 4a). PGJs have been reported to be endogenous ligands of PPARδ32, and our results also showed that PGJs significantly activated PPRE promoter activity (Fig. 4b), which was also significantly blunted by the inhibition of ERK. It is also noteworthy that PGJs significantly enhanced ERK activity (Supplementary Fig. S6). Furthermore, several of our observations support the role of PPARδ in VSMC phenotypic changes. (1) A selective PPARδ antagonist blunted VSMC phenotypic changes induced by 15-d-PGJ2 (Fig. 4c). (2) The ectopic regulation of PPARδ expression significantly affected phenotypic changes in VSMCs (Fig. 4e, f). (3) The overexpression of PPARδ enhanced VSMC proliferation (Fig. 4h). These observations encourage us to suggest that ERK-dependent PPARδ activation is involved in the regulation of VSMC phenotypes.
Since VSMCs also exhibited a decreased expression of PPARγ (Supplementary Fig. S7b), it is possible that PGD2-dependent phenotypic changes in VSMCs are mediated by PPARγ. Indeed, the PPARγ isoform showed a similar effect in terms of phenotypic changes in VSMCs (Supplementary Fig. S8). The role of PPARγ in the development of cardiovascular disease is somewhat controversial. For example, the forced expression or disruption of PPARγ has incompatible effects in terms of VSMC proliferation, cardiovascular disease progression, and the mediation of pro- or anti-inflammatory responses34,41. Currently, we do not have any direct evidence defining the exact role of PPARγ in cardiovascular disease, but strongly believe that the role of PPARγ is determined by cellular or disease contexts that might be influenced by many factors.
In the present study, we provide evidence that the COX2-dependent generation of PGD2 followed by PPARδ activation plays an important role in pathological changes in VSMC phenotypes (Fig. 6d). In this regard, our findings suggest that selective modulation of the biological activity of PGD2 might be therapeutically useful in the context of cardiovascular disease.
Supplementary information
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) funded by the Korean government (MSIP) (2014R1A2A2A01004433 and 2017R1A2B4002249). We would like to thank Professor Jang Hyun Choi at UNIST for providing plasmids (pGL3-PPRE and PPARδ).
Conflict of interests
The authors declare that they have no conflict of interest.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary information accompanies this paper at 10.1038/s12276-019-0330-3.
References
- 1.Glass CK, Witztum JL. Atherosclerosis. the road ahead. Cell. 2001;104:503–516. doi: 10.1016/s0092-8674(01)00238-0. [DOI] [PubMed] [Google Scholar]
- 2.Ross R. Rous-Whipple Award Lecture. Atherosclerosis: a defense mechanism gone awry. Am. J. Pathol. 1993;143:987–1002. [PMC free article] [PubMed] [Google Scholar]
- 3.Navab M, et al. The Yin and Yang of oxidation in the development of the fatty streak. A review based on the 1994 George Lyman Duff Memorial Lecture. Arterioscler. Thromb. Vasc. Biol. 1996;16:831–842. doi: 10.1161/01.atv.16.7.831. [DOI] [PubMed] [Google Scholar]
- 4.Owens GK. Regulation of differentiation of vascular smooth muscle cells. Physiol. Rev. 1995;75:487–517. doi: 10.1152/physrev.1995.75.3.487. [DOI] [PubMed] [Google Scholar]
- 5.Hao H, Gabbiani G, Bochaton-Piallat ML. Arterial smooth muscle cell heterogeneity: implications for atherosclerosis and restenosis development. Arterioscler. Thromb. Vasc. Biol. 2003;23:1510–1520. doi: 10.1161/01.ATV.0000090130.85752.ED. [DOI] [PubMed] [Google Scholar]
- 6.Hayashi K, et al. Insulin receptor substrate-1/SHP-2 interaction, a phenotype-dependent switching machinery of insulin-like growth factor-I signaling in vascular smooth muscle cells. J. Biol. Chem. 2004;279:40807–40818. doi: 10.1074/jbc.M405100200. [DOI] [PubMed] [Google Scholar]
- 7.Yoshida T, Gan Q, Shang Y, Owens GK. Platelet-derived growth factor-BB represses smooth muscle cell marker genes via changes in binding of MKL factors and histone deacetylases to their promoters. Am. J. Physiol. 2007;292:C886–C895. doi: 10.1152/ajpcell.00449.2006. [DOI] [PubMed] [Google Scholar]
- 8.Yun SJ, et al. Akt1 isoform modulates phenotypic conversion of vascular smooth muscle cells. Biochim. Biophys. Acta. 2014;1842:2184–2192. doi: 10.1016/j.bbadis.2014.08.014. [DOI] [PubMed] [Google Scholar]
- 9.Andersson J, Libby P, Hansson GK. Adaptive immunity and atherosclerosis. Clin. Immunol. 2010;134:33–46. doi: 10.1016/j.clim.2009.07.002. [DOI] [PubMed] [Google Scholar]
- 10.Lundberg AM, Hansson GK. Innate immune signals in atherosclerosis. Clin. Immunol. 2010;134:5–24. doi: 10.1016/j.clim.2009.07.016. [DOI] [PubMed] [Google Scholar]
- 11.Hansson GK. Cell-mediated immunity in atherosclerosis. Curr. Opin. Lipidol. 1997;8:301–311. doi: 10.1097/00041433-199710000-00009. [DOI] [PubMed] [Google Scholar]
- 12.Alexander MR, et al. Genetic inactivation of IL-1 signaling enhances atherosclerotic plaque instability and reduces outward vessel remodeling in advanced atherosclerosis in mice. J. Clin. Invest. 2012;122:70–79. doi: 10.1172/JCI43713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Alexander MR, Murgai M, Moehle CW, Owens GK. Interleukin-1beta modulates smooth muscle cell phenotype to a distinct inflammatory state relative to PDGF-DD via NF-kappaB-dependent mechanisms. Physiol. Genomics. 2012;44:417–429. doi: 10.1152/physiolgenomics.00160.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kirii H, et al. Lack of interleukin-1beta decreases the severity of atherosclerosis in ApoE-deficient mice. Arterioscler. Thromb. Vasc. Biol. 2003;23:656–660. doi: 10.1161/01.ATV.0000064374.15232.C3. [DOI] [PubMed] [Google Scholar]
- 15.Kober F, et al. MRI follow-up of TNF-dependent differential progression of atherosclerotic wall-thickening in mouse aortic arch from early to advanced stages. Atherosclerosis. 2007;195:e93–e99. doi: 10.1016/j.atherosclerosis.2007.06.015. [DOI] [PubMed] [Google Scholar]
- 16.Branen L, et al. Inhibition of tumor necrosis factor-alpha reduces atherosclerosis in apolipoprotein E knockout mice. Arterioscler. Thromb. Vasc. Biol. 2004;24:2137–2142. doi: 10.1161/01.ATV.0000143933.20616.1b. [DOI] [PubMed] [Google Scholar]
- 17.Ohta H, et al. Disruption of tumor necrosis factor-alpha gene diminishes the development of atherosclerosis in ApoE-deficient mice. Atherosclerosis. 2005;180:11–17. doi: 10.1016/j.atherosclerosis.2004.11.016. [DOI] [PubMed] [Google Scholar]
- 18.Battula S, Hao S, Pedraza PL, Stier CT, Ferreri NR. Tumor necrosis factor-alpha induces renal cyclooxygenase-2 expression in response to hypercalcemia. Prostaglandins Other Lipid Mediat. 2012;99:45–50. doi: 10.1016/j.prostaglandins.2012.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fehrenbacher JC, Burkey TH, Nicol GD, Vasko MR. Tumor necrosis factor alpha and interleukin-1beta stimulate the expression of cyclooxygenase II but do not alter prostaglandin E2 receptor mRNA levels in cultured dorsal root ganglia cells. Pain. 2005;113:113–122. doi: 10.1016/j.pain.2004.09.031. [DOI] [PubMed] [Google Scholar]
- 20.Itatsu K, et al. Cyclooxygenase-2 is involved in the up-regulation of matrix metalloproteinase-9 in cholangiocarcinoma induced by tumor necrosis factor-alpha. Am. J. Pathol. 2009;174:829–841. doi: 10.2353/ajpath.2009.080012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kim YA, et al. Tumor necrosis factor-alpha-induced cyclooxygenase-2 overexpression in eutopic endometrium of women with endometriosis by stromal cell culture through nuclear factor-kappaB activation. J. Reprod. Med. 2009;54:625–630. [PubMed] [Google Scholar]
- 22.Morisset S, Patry C, Lora M, de Brum-Fernandes AJ. Regulation of cyclooxygenase-2 expression in bovine chondrocytes in culture by interleukin 1alpha, tumor necrosis factor-alpha, glucocorticoids, and 17beta-estradiol. J. Rheumatol. 1998;25:1146–1153. [PubMed] [Google Scholar]
- 23.Young W, Mahboubi K, Haider A, Li I, Ferreri NR. Cyclooxygenase-2 is required for tumor necrosis factor-alpha- and angiotensin II-mediated proliferation of vascular smooth muscle cells. Circ. Res. 2000;86:906–914. doi: 10.1161/01.res.86.8.906. [DOI] [PubMed] [Google Scholar]
- 24.Bozza PT, Bakker-Abreu I, Navarro-Xavier RA, Bandeira-Melo C. Lipid body function in eicosanoid synthesis: an update. Prostaglandins Leukot. Essent. Fat. Acids. 2011;85:205–213. doi: 10.1016/j.plefa.2011.04.020. [DOI] [PubMed] [Google Scholar]
- 25.Ragolia L, Palaia T, Paric E, Maesaka JK. Prostaglandin D2 synthase inhibits the exaggerated growth phenotype of spontaneously hypertensive rat vascular smooth muscle cells. J. Biol. Chem. 2003;278:22175–22181. doi: 10.1074/jbc.M302769200. [DOI] [PubMed] [Google Scholar]
- 26.Ren J, Karpinski E, Benishin CG. Prostaglandin E2 contracts vascular smooth muscle and inhibits potassium currents in vascular smooth muscle cells of rat tail artery. J. Pharmacol. Exp. Ther. 1995;275:710–719. [PubMed] [Google Scholar]
- 27.Rice KM, et al. PGF2alpha-associated vascular smooth muscle hypertrophy is ROS dependent and involves the activation of mTOR, p70S6k, and PTEN. Prostaglandins Other Lipid Mediat. 2008;85:49–57. doi: 10.1016/j.prostaglandins.2007.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Williams SP, Dorn GW, 2nd, Rapoport RM. Prostaglandin I2 mediates contraction and relaxation of vascular smooth muscle. Am. J. Physiol. 1994;267:H796–H803. doi: 10.1152/ajpheart.1994.267.2.H796. [DOI] [PubMed] [Google Scholar]
- 29.Fitzpatrick FA, Wynalda MA. Albumin-catalyzed metabolism of prostaglandin D2. Identification of products formed in vitro. J. Biol. Chem. 1983;258:11713–11718. [PubMed] [Google Scholar]
- 30.Kikawa Y, Narumiya S, Fukushima M, Wakatsuka H, Hayaishi O. 9-Deoxy-delta 9, delta 12-13,14-dihydroprostaglandin D2, a metabolite of prostaglandin D2 formed in human plasma. Proc. Natl Acad. Sci. USA. 1984;81:1317–1321. doi: 10.1073/pnas.81.5.1317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Forman BM, et al. 15-Deoxy-delta 12, 14-prostaglandin J2 is a ligand for the adipocyte determination factor PPAR gamma. Cell. 1995;83:803–812. doi: 10.1016/0092-8674(95)90193-0. [DOI] [PubMed] [Google Scholar]
- 32.Reddy AT, Lakshmi SP, Banno A, Reddy RC. Identification and molecular characterization of peroxisome proliferator-activated receptor delta as a novel target for covalent modification by 15-deoxy-delta(12,14)-prostaglandin J2. ACS Chem. Biol. 2018;13:3269–3278. doi: 10.1021/acschembio.8b00584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kliewer SA, Umesono K, Noonan DJ, Heyman RA, Evans RM. Convergence of 9-cis retinoic acid and peroxisome proliferator signalling pathways through heterodimer formation of their receptors. Nature. 1992;358:771–774. doi: 10.1038/358771a0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kleinhenz JM, et al. Smooth muscle-targeted overexpression of peroxisome proliferator activated receptor-gamma disrupts vascular wall structure and function. PLoS ONE. 2015;10:e0139756. doi: 10.1371/journal.pone.0139756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhang J, et al. Peroxisome proliferator-activated receptor delta is up-regulated during vascular lesion formation and promotes post-confluent cell proliferation in vascular smooth muscle cells. J. Biol. Chem. 2002;277:11505–11512. doi: 10.1074/jbc.M110580200. [DOI] [PubMed] [Google Scholar]
- 36.Matsuoka T, Narumiya S. Prostaglandin receptor signaling in disease. Sci. World J. 2007;7:1329–1347. doi: 10.1100/tsw.2007.182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Libby P. Inflammation in atherosclerosis. Nature. 2002;420:868–874. doi: 10.1038/nature01323. [DOI] [PubMed] [Google Scholar]
- 38.Inoue T, et al. Lipocalin-type prostaglandin D synthase is a powerful biomarker for severity of stable coronary artery disease. Atherosclerosis. 2008;201:385–391. doi: 10.1016/j.atherosclerosis.2008.03.010. [DOI] [PubMed] [Google Scholar]
- 39.Pettipher R. The roles of the prostaglandin D(2) receptors DP(1) and CRTH2 in promoting allergic responses. Br. J. Pharmacol. 2008;153:S191–S199. doi: 10.1038/sj.bjp.0707488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Strack AM, et al. Nicotinic acid and DP1 blockade: studies in mouse models of atherosclerosis. J. Lipid Res. 2013;54:177–188. doi: 10.1194/jlr.M031344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hamblin M, et al. Vascular smooth muscle cell peroxisome proliferator-activated receptor-gamma deletion promotes abdominal aortic aneurysms. J. Vasc. Surg. 2010;52:984–993. doi: 10.1016/j.jvs.2010.05.089. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


