Abstract
Background
Solar ultraviolet radiation (UV) induces DNA damages in skin via direct absorption of UVB or indirectly by photosensitization mediated through UVA. Recent findings have revealed that UVA induces cyclobutane pyrimidine dimer (CPD) generation via chemiexcitation in melanocytes hours after the exposure. This UVA-induced delayed CPD (dark CPD) constitutes the majority of CPD in melanocytes. These findings indicate that sun light can damage the skin hours after the exposure, suggesting the need for skin care products post sun exposure. The main objective of this study was to investigate whether a blend of Chrysanthemum Morifolium flower extract (Chrys) and vitamin C derivative, Ascorbic Acid-2-Glucoside (AA2G), can provide protective effects against reactive oxygen species, melanin formation and UVA-induced dark CPD.
Methods
Intracellular ROS levels were measured in epidermal keratinocytes using DHR123 dye. Melanogenesis inhibition efficacy was determined using B16 cells. As for the dark CPD measurement, Melan-a cells were treated with or without actives for 6 days, then irradiated with UVA at various doses. Cells were exposed with anti-CPD mAb followed by secondary Ab. CPD levels were determined by measuring fluorescent intensity using a high content imaging analysis.
Results
Chrys, AA2G and their blend at various concentrations demonstrated ROS scavenging activity. Though Chrys alone did not show significant melanogenesis inhibition in B16 assay, the blend of Chrys with AA2G demonstrated additive effects in comparison with AA2G alone. The blend of AA2G and Chrys at various concentrations exhibited enhanced efficacy for inhibiting dark CPD compared to AA2G alone.
Conclusion
The results from this study indicate that the use of natural antioxidant, Chrys in combination with AA2G, provides protection against UVA-induced delayed CPD formation by enhancing ROS scavenging activity and melanogenesis inhibition. These findings could potentially be applied for formulating post-sun exposure skin care products, possibly extending to evening-after care products.
Keywords: cellular DNA photodamage, chrysanthemum morifolium extract, ascorbic acid-2-glucoside, AA2G, DNA damage, cyclobutane pyrimidine dimers, CPD, dark CPD, melanocytes
Introduction
Skin, the largest organ of the human body, plays an essential role of being the primary defense mechanism against external environment stressors including ultraviolet (UV) radiation, pollution, ozone, air borne chemicals, and allergens, etc. Of all the environmental factors, exposure to UV radiations, both acute and chronic, has been attributed to various skin concerns such as sunburn (erythema), pigmentation (tanning), inflammation, photo-aging and even skin cancer.1,2 UV radiation consists of UVA (320-400nm), UVB (290–320nm) and UVC (100–290nm) components. Typically, ambient sunlight is composed of 90–95% UVA, 5–10% UVB, with ozone absorbing most of solar UVC radiation.3
Both UVA and UVB play a significant role in inducing DNA damages with different mechanisms for generating DNA mutations at the cellular level. High energy UVB is site specific and absorbed directly by the nucleotides, creating the DNA alterations such as cyclobutane pyrimidine dimer (CPD), which is capable of interfering with DNA replication.4,5 On the other hand, UVA mediates the DNA damage indirectly via photosensitization. When UVA is absorbed by intracellular chromophores such as porphyrins or bilirubin,6 it results in generating reactive oxygen species (ROS). These ROS interact directly with DNA via Fenton reaction generating superoxide (O2˙−) or the hydroxyl radical (OH•) leading to developing single strand breaks or induce oxidized base formation in DNA via singlet oxygen production.7–9
CPD is the major form of DNA photolesions created by UV radiation.10 It joins two adjacent pyrimidine bases by two single bonds creating a carbon ring between them,11 which interrupts base pairing and alters the DNA helix from its normal B form.12 CPDs in skin cells have critical biological consequences, including mutagenicity that may lead to the induction of skin cancer as well as skin aging.13,14 Moreover, CPDs also have non-mutagenic consequences such as initiating cytokine release and photo-immunosuppression that are also thought to be involved in skin cancer.15,16 Until recently, CPD was believed to be formed picoseconds after a direct absorption of UVB by thymine or cytosine. However, Premi et al reported that CPDs can be continuously generated for hours after UVA exposure in melanocytes via chemiexcitiation. The resulting, so-called “Dark CPD” constitutes the majority of CPDs in melanocytes. These delayed-formed CPDs arise when UVA activates nitric oxide synthase (NOS) and NADPH oxidase (NOX), which produce nitric oxide (NO•) and O2˙−. These two radicals then combine to form the radical peroxynitrite (ONOO−), degrading melanin polymer to the melanin fragments. The melanin monomers are then oxidized by ONOO−. The fragments, acting as a molecular shuttle, transfers excited electron in the melanin fragment resulting in a triplet state intermediate. The triplet state reaction intermediate creates an unstable dioxetane and then yield two carbonyls, one of which acquires the most energy discharged to DNA where the CPD is generated.17,18
Sunscreen use has been shown to mitigate the adverse effects of sunlight primarily for UVB induced burning and damages. Although there is an increasingly recognized role of UVA in skin aging, pigmentation disorders, and even possibly melanoma development via “delayed CPD” formation, sunscreen users are minimally protected against UVA,18,19 unless using a broad spectrum sunscreen, adequately and frequently. Treating UVA-induced pigmentation disorders are tough and usually need a longer intervention time.20–22 Hence, other methods of reducing the risks of photodamage are being sought23 such as inclusion of various antioxidants from botanical extracts or naturally occurring non-enzymatic vitamins (like vitamin C), or their derivatives (like Ascorbic Acid-2-Glucoside (AA2G)) in sunscreens or other skin care formulations. Chrysanthemum Morifolium is one such botanical which is well known in the traditional Chinese medicine for antioxidant and anti-inflammatory properties.24–26 Previous chemical characterization studies indicate that Chrysanthemum Morifolium flower extract contains quercetin, acacetin 7-O-beta-D-(3”-acetyl) -glucopyranoside, luteolin 7-O-beta-D-(6”-acetyl)-glucopyranoside, hesperetin 7-O-beta-D-glucopyranoside, acacetin 7-O-beta-D-glucopyranoside, diosmetin 7-O-beta-D-glucopyranoside, apigenin 7-O-beta-D-glucopyranoside, hesperidin, linarin, luteolin 7-O-beta-D-glucopyranoside, 3,5-dicaffeoyl-epi-quinic acid and 1,3-dicaffeoyl-epi-quinic acid.25,27 The main objective of this study was to determine the protective effects of a blend of Chrysanthemum Morifolium flower extract (Chrys) and AA2G against UVA-induced dark CPD generation in melanocytes. The capacity of the actives to inhibit ROS generation and melanogenesis was also assessed since they are essential steps in the process of dark CPD formation.
Materials And Methods
Cell Culture
Primary cells and cell lines were grown in a humidified atmosphere of 5% CO2 at 37°C. Adult Primary Human Epidermal Keratinocytes (HEKa) were procured from Thermo Fisher Scientific (Waltham, MA, USA) and maintained in Epilife Medium with 60 µM calcium and supplemented 1% Human Keratinocyte Growth Supplement (Thermo Fisher Scientific) and 1% Penicillin-Streptomycin (P/S) Solution, 100x (Corning, Manassas, VA, USA). Cells were cultured in T75 flasks and passaged before 80% confluency. Experiments were performed between the fourth and sixth passages.28
B16-F10 (B16) cells (ATCC, Manassas, VA) were cultured in Dulbecco’s Modified Eagle’s Medium with 4.5 g/L glucose, L-glutamine, sodium pyruvate (DMEM) supplemented with 10% fetal bovine serum (FBS) and 1% P/S Solution, 100x. The cells were sub-cultured in T75 flasks three to four days each to sustain regular growth.29
Melan-a (Mel-a) cells were purchased from Welcome Trust Functional Genomic Cell Bank, St. George Medical School, University of London, London (United Kingdom). Cells were grown in RPMI-1640 media supplemented with 10% FBS, 1% P/S Solution, 100x, 1% amphotericin B, 2 mM L-glutamine, and 200nM tetradecanoyl phorbol acetate (TPA). Mel-a cells were used at passage 30 or less for the experiment.30
Melanogenesis Inhibition Assay
Chrys was sourced from SK-Bioland, Jiangsu Province, China. The extraction solvent was water and the carrier solvent of choice was Dipropylene Glycol. B16 cells (5000 cells/well) were pre-cultured in 96-well plates in DMEM medium supplemented with 10% FBS and 1% P/S for 24 hrs. For treatments, cells were treated with the Chrys, AA2G (Hayashibara, Okayama, Japan), or the combination in DMEM-phenol free medium supplemented with 10% FBS, 1% P/S, 2 mM L-glutamine, and 50 nM α-MSH (Sigma, St. Louis, MO, USA) for 96 hrs. The absorbance at 405 nm was measured and MTT assay21,22,31 was performed to examine the viability of cells. Relative melanin content was normalized with the % cell viability.32
Measurement Of Intracellular ROS Concentration
Measurement of intracellular ROS concentration was performed using a Dihydrorhodamine 123 (DHR123; Thermo Fisher Scientific) dye. HEKa cells were seeded on 12-well plates overnight and then incubated with the actives for three hours. Subsequently, cells were incubated with 50 µM of pyocyanin (Sigma, St. Louis, MO, USA) at 37°C for 30 mins. Cells were then trypsinized then incubated with 50 µM of DHR123 for 30 mins. The ROS production was analyzed by counting 104 events using BD AccuriTM C6 Plus Flow Cytometer.33
Cells Treatment And UVA Exposure
Mel-a cells were seeded at a density of 2 x 104 cells/well on 96-well strip-well plates and allowed to attach overnight at 37°C, 5% CO2. Cells were treated with vehicle control or the testing actives and replenished with fresh media every other day for 6 days. On the 7th day, the media were removed and replaced with warm PBS. Cells were exposed to UVA (325 nm - 400 nm) at various intensities using an Asahi Spectra Max-303 (Asahi Spectra Co., Ltd., Japan) with XUL0325 Longpass Filter/UV 325nm, 25mm dia (Asahi Spectra Co., Ltd). The filter cutoff wavelength was chosen to be 325nm to eliminate the possibility of UVB contamination. Cells not exposed to UVA served as the negative control. At appropriate time points, cell viability and CPD formation were measured.
Cell Viability Assay
Twenty-four hours after UVA exposure, cell viability was determined using both PrestoBlue assay (Thermo Fisher Scientific) and Image-IT live/dead (Thermo Fisher Scientific) staining. Briefly, PrestoBlue reagent was applied to cells at 1:10 ratio. The fluorescence was read (excitation 560 nm; emission 590 nm) at recommended time of incubation (2 h-endpoint). For the staining, Image-IT live/dead stain was applied to the cells, and the cells were counted using ArrayScan™ XTI Live High Content Platform (Thermo Fisher Scientific).
Measurement Of CPD Formation
UVA-induced DNA damage in the form of CPD were detected and measured in Mel-a cells. Briefly, at 0, 2, 4, 8, and 24 hr time point after UVA irradiation, cells were fixed in 4% performic acid (PFA) for 20 mins, permeabilized with Triton X-100, followed by blocking with normal human serum blocking buffer. The cells then were incubated with a CPD-specific monoclonal antibody (CosmoBio USA, Carlsbad, CA, USA) for 1 hr at room temperature. After washing the bound anti-CPD mAb, secondary antibody Alexa Fluor 647 (Thermo Fisher Scientific) was applied. CPD content was analyzed via ArrayScan XTI Live High Content Platform. The mean total fluorescence intensity per well, the mean area of live/dead stain per well, and the mean number of cells per field were reported in an ArrayScan internal software, from which the data were exported and analyzed further in Microsoft Excel.
Results
Suppression Of Intracellular ROS Production
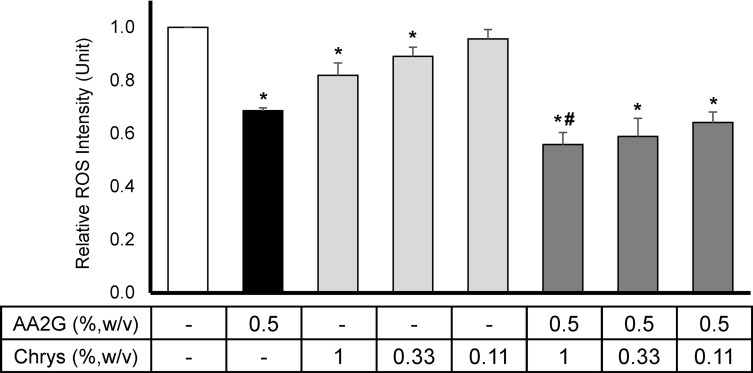
Intracellular ROS levels were measured using DHR123 dye in HEKa cell culture model. As shown in Figure 1, 0.5%w/v AA2G reduced the intracellular ROS production by 31 ± 1.1% compared to the vehicle control. Though relatively lower than the result from AA2G, Chrys demonstrated significant dose-dependent ROS suppression at 0.33% w/v (11 ± 3.5%) and 1% w/v (18 ± 4.7%), relative to the vehicle control. The combination of Chrys and AA2G at various concentrations resulted in better suppression of ROS activity. The most robust activity was seen with the 0.5% w/v AA2G + 1.0% w/v Chrys blend which reduced the ROS levels by 44 ± 5.6%. Other combination of AA2G+Chrys blend also demonstrated significant ROS scavenging activity relative to the vehicle control and AA2G (0.5% w/v) alone. No cytotoxicity was observed (data not shown).
Figure 1.
Effect of AA2G, Chrys, and AA2G + Chrys on ROS production in HEKa: HEKa cells were treated with AA2G, Chys, and AA2G + Chrys blend at various concentrations. The intracellular ROS level was induced by pyocyanin, and the measurement was conducted using the DHR123 dye. All data are expressed as Mean ± Standard Error of Mean of three independent experiments. *Significant with respect to the vehicle control (p < 0.05). #Significant with respect to the 0.5% w/v AA2G treatment (p < 0.05). Statistics were analyzed with Microsoft Excel software utilizing a Student’s one-tailed t-test assuming equal levels of variance. Statistically significant values were defined as p < 0.05.
Prevention Of Melanin Production
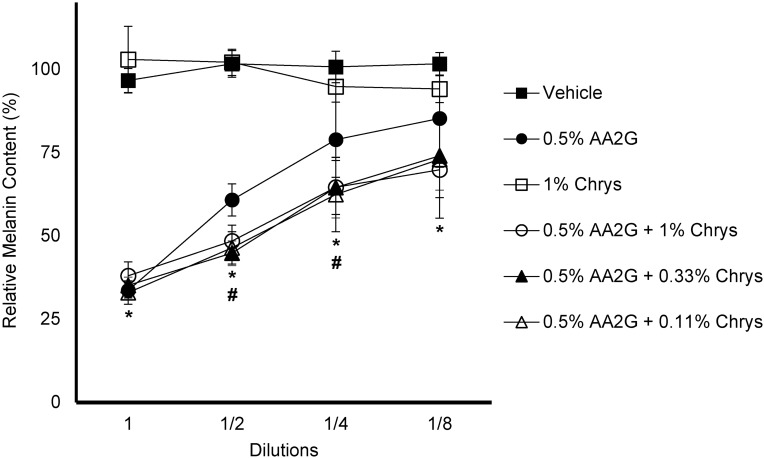
The efficacy of Chrys, AA2G and the blend of these two (at various concentrations) to inhibit melanogenesis was tested in the B16 melanoma cell line. Figure 2 represents the relative melanin content of various test concentrations and combinations. AA2G (0.5% w/v) and its various combinations with Chrys (0.11, 0.33 and 1% w/v) showed a clear dose-dependent melanogenesis inhibition, whereas none was observed with Chrys (1% w/v) alone. There were no significant inter group differences between AA2G and its various combinations with Chrys at the highest and lowest test concentrations, represented by 1 (no dilution) and 1/8th dilution, respectively. However, a significant difference (p < 0.05) was observed between AA2G (0.5% w/v) and its other test combinations with Chrys at 1/2 and 1/4th dilutions, representing additive melanogenesis inhibition activity of the AA2G +Chrys blend, relative to the use of AA2G or Chrys alone.
Figure 2.
Effect of AA2G, Chys, and AA2G + Chrys blend on Melanin production in B16 cells: B16 cells were treated with AA2G, Chys, and AA2G + Chrys blend at various concentrations for 96 hrs. The melanin synthesis was induced by α-MSH (50 nM). Relative melanin level was measured using a plate reader and normalized with the % cell viability. All data are expressed as Average ± Standard Deviation of six independent end points. *Significance of 0.5%w/v AA2G and 0.5%w/v AA2G + Chrys (0.11, 0.33, and 1% w/v) with respect to the vehicle control (p < 0.05). #Significance of 0.5%w/v AA2G + Chrys (0.11, 0.33, and 1% w/v) at 1/2 and 1/4th dilution with respect to the 0.5% w/v AA2G treatment (p < 0.05). Statistics were analyzed with Microsoft Excel software utilizing a Student’s one-tailed t-test assuming equal levels of variance. Statistically significant values were defined as p < 0.05.
UVA-Induced Dark CPD Production
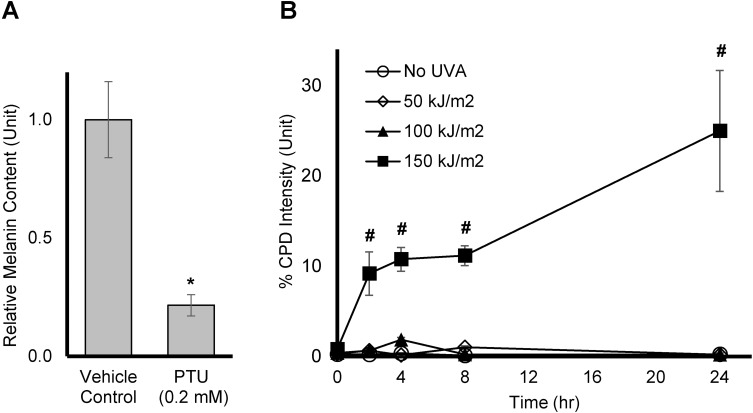
To verify that dark CPD is indeed generated by UVA in melanin producing Mel-a cells, the cells were maintained for 6 days, and then exposed to UVA dose of 50, 100, or 150 kJ/m.2 CPD levels were measured at 0, 2, 4, 8, and 24 hrs after the UVA exposure. Melanin production was confirmed by comparing the melanin level of cells treated with vehicle control or Phenylthiourea (PTU), an inhibitor of tyrosinase and melanin synthesis on Day 7, as shown in Figure 3A. In Figure 3B, no significant amount of CPD was generated when cells were exposed to UVA at 50 or 100 kJ/m2. On the other hand, a significant and sustained increase in CPD formation was observed at 150 kJ/m2 up to 24 hrs after exposure time. Thus, the dose of UVA irradiation at 150 kJ/m2 was chosen for testing the actives.
Figure 3.
Effect of UVA on dark CPD production in Mel-a cells: (A) Mel-a cells were maintained for 6 days, then melanin was extracted using 1N NaOH. The melanin content was accessed using a plate reader, read at 405 nm. All data are expressed as Average ± Standard Deviation of six independent end points. (B) Mel-a cells were maintained for 6 days, then the cells were exposed with UVA at various doses. CPD fluorescent intensity was measured at various time points. All data are expressed as Mean ± Standard Error of Mean of six independent end points. *Significance with respect to the vehicle control (p < 0.05). #Significance with respect to No UVA control at corresponding time points (p < 0.05). Statistics were analyzed with Microsoft Excel software utilizing a Student’s one-tailed t-test assuming equal levels of variance. Statistically significant values were defined as p < 0.05.
Prevention Of Dark CPD Formation
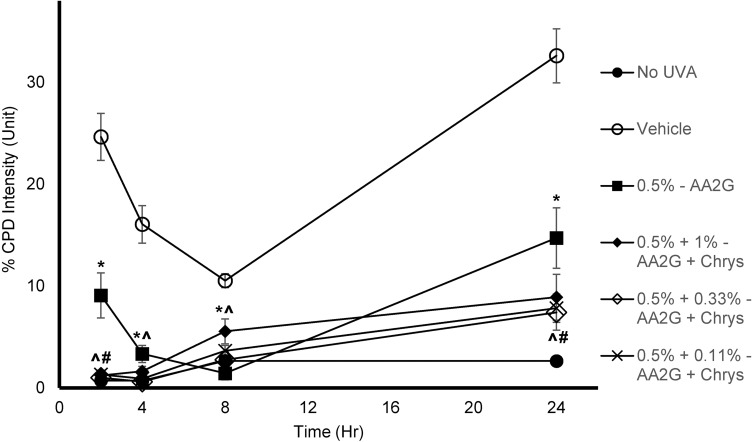
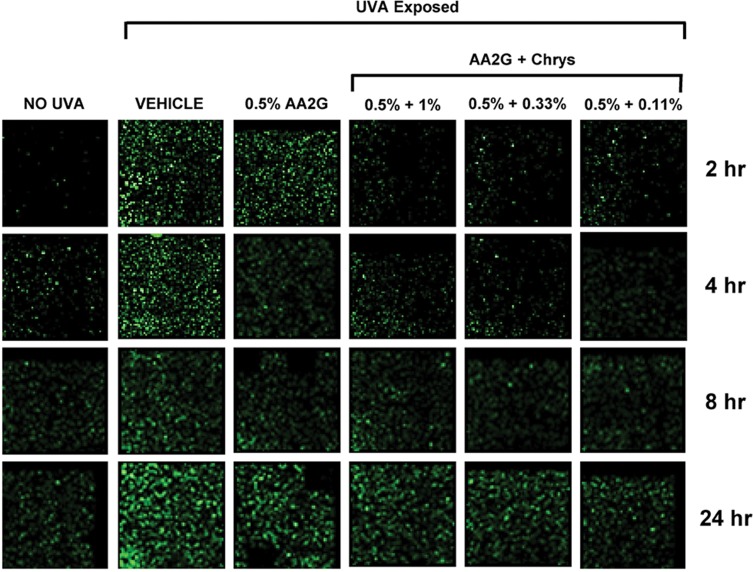
To determine whether AA2G and AA2G + Chrys combination prevent dark CPD formation, Mel-a cells were pre-treated with the actives for 6 days, then exposed to UVA (150 kJ/m2). As reflected in Table 1, cell viability was not affected by UVA exposure with or without the active treatment. Figure 4 shows that UVA exposed cells treated with vehicle control induced CPD formation hours after the exposure and peaked at the 24 hr time point. 0.5% w/v AA2G reduced CPD formation at 2, 4, 8, and 24 hr time point. 0.5% w/v AA2G + Chrys (0.11, 0.33 and 1% w/v) blends demonstrated preventive effect on CPD generation at all time points. Unlike the dose-dependent ROS inhibition activity of the Chrys + AA2G blends, no dose-dependence was observed for Chrys concentration in the AA2G + Chrys blend for prevention of Dark CPD formation. Interestingly at 2 hr time point, 0.5% w/v AA2G + Chrys (0.11, 0.33 and 1% w/v) combination prevented CPD formation approximately by 95% while 0.5% w/v AA2G reduced CPD by 63% ± 9.0% compared to the vehicle control. At the 24 hr timepoint, 0.5% w/v AA2G + Chrys (0.11, 0.33 and 1% w/v) blend decreased CPD level approximately by 75% while 0.5% w/v AA2G reduced CPD by 55% ± 9.2% compared to the vehicle control. These findings indicate that AA2G + Chrys blend exhibit an enhanced efficacy when compared to the AA2G alone. No significant induction of CPD was observed from non-UVA irradiated cells. Chrys alone did not show capacity of reducing CPD level (data not shown). The above findings are supplemented by Figure 5 which demonstrates that the AA2G + Chrys blend, at various concentrations, effectively decreases the CPD levels (represented by green florescence intensity) relative to the vehicle control.
Table 1.
Cell Viability Of Mel-A Cells 24 hrs After UVA Exposure, With Or Without Active(s) Treatment
| Treatment | Viable Cells (%) (Image-IT Live/Dead Staining) | Viable Cells (%) (PrestoBlue) |
|---|---|---|
| No UVA | 100.0 ± 3.5 | 100 ± 2.3 |
| 150 kJ/m2 - Vehicle Control | 91.2 ± 9.5 | 98.7 ± 8.0 |
| 150 kJ/m2 – 0.5% AA2G | 89.1 ± 6.5 | 104.2 ± 6.2 |
| 150 kJ/m2 – 0.5% AA2G + 1% Chrys | 85.9 ± 3.5 | 100.2 ± 2.8 |
| 150 kJ/m2 – 0.5% AA2G + 0.33% Chrys | 91.6 ± 4.7 | 102.3 ± 3.8 |
| 150 kJ/m2 – 0.5% AA2G + 0.11% Chrys | 91.8 ± 3.5 | 97.7 ± 3.0 |
Figure 4.
Effect of AA2G and AA2G + Chrys on dark CPD formation in Mel-a cells: Mel-a cells were pre-treated with or without actives every other day for 6 days, then exposed with 150 kJ/m2 of UVA dose. The cells were fixed in PFA, treated with anti-CPD mAb, followed by secondary anti-body. CPD level was obtained by analyzing the fluorescent intensity. All data are expressed as Mean ± Standard Error of Mean of six independent end points. *Significance of 0.5% w/v AA2G with respect to the vehicle control (p < 0.05). #Significance of 0.5% w/v AA2G + Chrys (0.11, 0.33, and 1% w/v) with respect to the 0.5% w/v AA2G treatment (p < 0.05). ^Significance of 0.5% w/v AA2G + Chrys (0.11, 0.33, and 1% w/v) with respect to the vehicle control (p < 0.05). Statistics were analyzed with Microsoft Excel software utilizing a Student’s one-tailed t-test assuming equal levels of variance. Statistically significant values were defined as p < 0.05.
Figure 5.
Representative images of effect of AA2G and AA2G + Chrys blend on CPD formation. Mel-a cells were pre-treated with or without actives every other day for 6 days, then exposed with 150 kJ/m2 of UVA. The cells were fixed in PFA, treated with anti-CPD mAb, secondary anti-body. Fluoresce images were taken at 2, 4, 8, and 24 hr time points. CPD level was represented by green florescence intensity.
Discussion
UV is a well-known environmental stress factor to the skin that can accelerate the skin aging process and even develop skin cancers. Sunscreens are developed to protect the skin from UV; however, its inadequate and less frequent use leaves room for UV-induced skin photodamage. The DNA mutations resulting from direct absorption of UVB and its damaging effects are well understood, however, the susceptibility of human skin particularly to UVA-induced ROS generation and DNA damage has become of increasing interest. Recently, Premi et al demonstrated that UVA can induce dark CPD formation hours after the exposure has ceased, and the UVA-induced delayed CPDs are the majority of CPDs generated in melanocytes.17,18 It has also been reported that the rate of removal of UVA-generated CPDs is lower than that produced by UVB irradiation of the skin.10 These findings indicate that UVA-induced photodamage could be more severe than previously thought as it continues at the molecular level even after UVA exposure has ceased. Thus, in addition to the use of sunscreens, new strategies for protecting skin post sun exposure is gaining interest. In this study, we aimed to evaluate the skin protection efficacy of a blend of two antioxidants (AA2G + Chrys) against molecular markers representing UVA-induced skin photodamage.
Chemiexcitation in melanocytes and subsequent formation of dark CPD are a multistep process. As the initial step of dark CPD formation, NO• and O2˙−are upregulated and generate a powerful radical ONOO− and melanin synthesis by UVA. ONOO− degrades melanin polymer to monomers which act as a molecular sensitizer to create excited dioxetane and excited state triplet carbonyl. We believe that targeting these two paths, ROS generation and melanogenesis could potentially help suppress CPD formation more effectively. Therefore, we first examined the effects of AA2G, Chrys and AA2G + Chrys on ROS generation and melanin production. ROS assay using DHR123 in HEKa cells and melanogenesis inhibition assay using B16 cells are reliable and effective methods to measure intracellular ROS and melanin levels. AA2G and Chrys demonstrated both ROS scavenging and melanogenesis inhibition effects, and the blend enhanced the efficacy (Figure 2). It is unclear how Chrys can contribute to the additive depigmenting effect in the blend with AA2G while Chrys itself showed no significant effect. We believe that the additive effect was possible as a result of ROS inhibiting effect of Chrys which reinforced the tyrosinase inhibiting activity of AA2G.
The susceptibility of human skin is substantially high for CPD damage and it does not provide efficient photoprotection against formation of CPD. This is evident from our observation that CPD formation continues over a prolonged period of time even after UVA irradiation source has ceased (Figure 3B). Similar observations were previously made by other researchers.17,18 In addition, it has been shown that the rate of removal of UVA-generated CPDs is lower than that produced by UVB irradiation of the skin,10 which indicates increased susceptibility of skin to UVA-induced DNA damage. The effect of UVA on reduced CPD repair is not yet clearly understood but could potentially be explained by an in-cell cycle arrest after irradiation34,35 or by oxidative stress mediated degradation of DNA repair protein. Since the blend of AA2G and Chrys demonstrated the suppressive effect on ROS generation and melanin production, they were further evaluated for their potential of preventing dark CPD production. Mel-a cells were used since they can produce melanin effectively (Figure 3A). Different induction and pretreatment periods were used in various experiments as ROS induction is faster in HEKa (usually in a few hours) than melanin production induction in Mel-a cells (usually several days).The pre-treatment of AA2G and AA2G + Chrys blend demonstrated the preventive effect on dark CPD formation. The blend treatment also showed enhanced activity when the CPD levels were highest (2 & 24 hrs) compared to AA2G treatment alone (Figures 4 and 5).
In summary, AA2G and Chrys combination showed promising results for promoting ROS scavenging activity and melanogenesis inhibition, therefore reducing dark CPD generation. We believe that this activity could be further enhanced by inclusion of ingredients which could act as triplet state quenchers, a downstream intermediate critical for formation of dark CPD.
Conclusions
Consumers believe that solar protection is needed only during the daytime. It has been now shown that DNA damage could occur hours after the exposure to the sun, specifically UVA. The results from this study indicate that the use of natural antioxidant, Chrysanthemum Morifolium flower extract in combination with a well-known ascorbic acid derivative, AA2G, enhances the protection against delayed DNA damage (dark CPD formation) in the skin. Such combination strategies may be applied for formulating a more effective skincare product which helps to alleviate skin damage after the solar exposure, potentially in a form of the evening skin care product.
Disclosure
This study was funded by Amway Corporation and authors SY, JL, HJ, JS, SMB are employees of the Amway Corporation. RW and JN are affiliated with Zen-Bio, Inc. The authors report no other conflicts of interest in this work.
References
- 1.Elwood JM, Jopson J. Melanoma and sun exposure: an overview of published studies. Int J Cancer. 1997;73(2):198–203. doi: 10.1002/(ISSN)1097-0215 [DOI] [PubMed] [Google Scholar]
- 2.Maini S, Fahlman BM, Krol ES. Flavonols protect against UV radiation-induced thymine dimer formation in an artificial skin mimic. J Pharm Pharm Sci. 2015;18(4):600–615. doi: 10.18433/J34W39 [DOI] [PubMed] [Google Scholar]
- 3.Amaro-Ortiz A, Yan B, D’Orazio JA. Ultraviolet radiation, aging and the skin: prevention of damage by topical cAMP manipulation. Molecules. 2014;19(5):6202–6219. doi: 10.3390/molecules19056202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Eller MS, Ostrom K, Gilchrest BA. DNA damage enhances melanogenesis. Proc Natl Acad Sci U S A. 1996;93(3):1087–1092. doi: 10.1073/pnas.93.3.1087 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Katiyar SK, Mantena SK, Meeran SM. Silymarin protects epidermal keratinocytes from ultraviolet radiation-induced apoptosis and DNA damage by nucleotide excision repair mechanism. PLoS One. 2011;6(6):e21410. doi: 10.1371/journal.pone.0021410 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wondrak GT, Jacobson MK, Jacobson EL. Endogenous UVA-photosensitizers: mediators of skin photodamage and novel targets for skin photoprotection. Photochem Photobiol Sci. 2006;5(2):215–237. doi: 10.1039/B504573H [DOI] [PubMed] [Google Scholar]
- 7.Cadet J, Douki T. Oxidatively generated damage to DNA by UVA radiation in cells and human skin. J Invest Dermatol. 2011;131(5):1005–1007. doi: 10.1038/jid.2011.51 [DOI] [PubMed] [Google Scholar]
- 8.Lyons NM, O’Brien NM. Modulatory effects of an algal extract containing astaxanthin on UVA-irradiated cells in culture. J Dermatol Sci. 2002;30(1):73–84. doi: 10.1016/S0923-1811(02)00063-4 [DOI] [PubMed] [Google Scholar]
- 9.Breimer LH. Molecular mechanisms of oxygen radical carcinogenesis and mutagenesis: the role of DNA base damage. Mol Carcinog. 1990;3(4):188–197. doi: 10.1002/mc.2940030405 [DOI] [PubMed] [Google Scholar]
- 10.Mouret S, Baudouin C, Charveron M, Favier A, Cadet J, Douki T. Cyclobutane pyrimidine dimers are predominant DNA lesions in whole human skin exposed to UVA radiation. Proc Natl Acad Sci. 2006;103(37):13765–13770. doi: 10.1073/pnas.0604213103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Setlow RB, Carrier WL. Pyrimidine dimers in ultraviolet-irradiated DNA’s. J Mol Biol. 1966;17(1):237–254. doi: 10.1016/S0022-2836(66)80105-5 [DOI] [PubMed] [Google Scholar]
- 12.Brash DE. UV signature mutations. Photochem Photobiol. 2015;91(1):15–26. doi: 10.1111/php.12377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.You YH, Lee DH, Yoon JH, Nakajima S, Yasui A, Pfeifer GP. Cyclobutane pyrimidine dimers are responsible for the vast majority of mutations induced by UVB irradiation in mammalian cells. J Biol Chem. 2001;276(48):44688–44694. doi: 10.1074/jbc.M107696200 [DOI] [PubMed] [Google Scholar]
- 14.Panich U, Sittithumcharee G, Rathviboon N, Jirawatnotai S. Ultraviolet radiation-induced skin aging: the role of DNA damage and oxidative stress in epidermal stem cell damage mediated skin aging. Stem Cells Int. 2016;2016:7370642. doi: 10.1155/2016/7370642 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Halliday GM. Inflammation, gene mutation and photoimmunosuppression in response to UVR-induced oxidative damage contributes to photocarcinogenesis. Mutat Res. 2005;571(1–2):107–120. doi: 10.1016/j.mrfmmm.2004.09.013 [DOI] [PubMed] [Google Scholar]
- 16.Delinasios GJ, Karbaschi M, Cooke MS, Young AR. Vitamin E inhibits the UVAI induction of “light” and “dark” cyclobutane pyrimidine dimers, and oxidatively generated DNA damage, in keratinocytes. Sci Rep. 2018;8(1):423. doi: 10.1038/s41598-017-18924-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Premi S, Wallisch S, Mano CM, et al. Photochemistry. Chemiexcitation of melanin derivatives induces DNA photoproducts long after UV exposure. Science. 2015;347(6224):842–847. doi: 10.1126/science.1256022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Premi S, Brash DE. Chemical excitation of electrons: a dark path to melanoma. DNA Repair (Amst). 2016;44:169–177. doi: 10.1016/j.dnarep.2016.05.023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Haywood R, Wardman P, Sanders R, Linge C. Sunscreens inadequately protect against ultraviolet-A-induced free radicals in skin: implications for skin aging and melanoma? J Invest Dermatol. 2003;121(4):862–868. doi: 10.1046/j.1523-1747.2003.12498.x [DOI] [PubMed] [Google Scholar]
- 20.Baswan SM, Leverett J, Pawelek J. Clinical evaluation of the lightening effect of cytidine on hyperpigmented skin. J Cosmet Dermatol. 2019;18(1):278–285. doi: 10.1111/jocd.12784 [DOI] [PubMed] [Google Scholar]
- 21.Baswan S, Yim S, Leverett J, Pawelek J. LB1591 In-vitro and in-vivo evaluation of skin lightening efficacy of cytidine. J Investig Dermatol. 2018;138(9):B21. doi: 10.1016/j.jid.2018.06.130 [DOI] [Google Scholar]
- 22.Baswan SM, Yim S, Leverett J, Scholten J, Pawelek J. Cytidine decreases melanin content in a reconstituted three-dimensional human epidermal model. Arch Dermatol Res. 2019;311(3):249–250. doi: 10.1007/s00403-019-01897-x [DOI] [PubMed] [Google Scholar]
- 23.Hammiller B, Karuturi BVK, Miller C, et al. Delivery of antioxidant enzymes for prevention of ultraviolet irradiation-induced epidermal damage. J Dermatol Sci. 2017;88(3):373–375. doi: 10.1016/j.jdermsci.2017.07.015 [DOI] [PubMed] [Google Scholar]
- 24.Duh P-D, Tu -Y-Y, Yen G-C. Antioxidant activity of water extract of harng jyur (Chrysanthemum morifolium Ramat). LWT - Food Sci Technol. 1999;32(5):269–277. doi: 10.1006/fstl.1999.0548 [DOI] [Google Scholar]
- 25.Kim HJ, Lee YS. Identification of new dicaffeoylquinic acids from Chrysanthemum morifolium and their antioxidant activities. Planta Med. 2005;71(9):871–876. doi: 10.1055/s-2005-873115 [DOI] [PubMed] [Google Scholar]
- 26.Song C, Liu Y, Song A, et al. The chrysanthemum nankingense genome provides insights into the evolution and diversification of chrysanthemum flowers and medicinal traits. Mol Plant. 2018;11(12):1482–1491. doi: 10.1016/j.molp.2018.10.003 [DOI] [PubMed] [Google Scholar]
- 27.Wang YJ, Yang XW, Guo QS. [Studies on chemical constituents in Huangjuhua (flowers of Chrysanthemum morifolium)]. Zhongguo Zhong Yao Za Zhi. 2008;33(5):526–530. [PubMed] [Google Scholar]
- 28.Liebmann J, Born M, Kolb-Bachofen V. Blue-light irradiation regulates proliferation and differentiation in human skin cells. J Invest Dermatol. 2010;130(1):259–269. doi: 10.1038/jid.2009.194 [DOI] [PubMed] [Google Scholar]
- 29.Chang NF, Chen YS, Lin YJ, et al. Study of hydroquinone mediated cytotoxicity and hypopigmentation effects from UVB-irradiated arbutin and deoxyarbutin. Int J Mol Sci. 2017;18(5). doi: 10.3390/ijms18050969 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rana J, Diwakar G, Saito L, Scholten JD, Mulder T. Inhibition of melanin content by punicalagins in the super fruit pomegranate (Punica granatum). J Cosmet Sci. 2013;64(6):445–453. [PubMed] [Google Scholar]
- 31.Fotakis G, Timbrell JA. In vitro cytotoxicity assays: comparison of LDH, neutral red, MTT and protein assay in hepatoma cell lines following exposure to cadmium chloride. Toxicol Lett. 2006;160(2):171–177. doi: 10.1016/j.toxlet.2005.07.001 [DOI] [PubMed] [Google Scholar]
- 32.Masuda M, Itoh K, Murata K, et al. Inhibitory effects of morinda citrifolia extract and its constituents on melanogenesis in murine B16 melanoma cells. Biol Pharm Bull. 2012;35(1):78–83. doi: 10.1248/bpb.35.78 [DOI] [PubMed] [Google Scholar]
- 33.Djiadeu P, Azzouz D, Khan MA, Kotra LP, Sweezey N, Palaniyar N. Ultraviolet irradiation increases green fluorescence of dihydrorhodamine (DHR) 123: false-positive results for reactive oxygen species generation. Pharmacol Res Perspect. 2017;5(2):e00303. doi: 10.1002/prp2.303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.de Laat A, van Tilburg M, van der Leun JC, van Vloten WA, de Gruijl FR. Cell cycle kinetics following UVA irradiation in comparison to UVB and UVC irradiation. Photochem Photobiol. 1996;63(4):492–497. doi: 10.1111/j.1751-1097.1996.tb03075.x [DOI] [PubMed] [Google Scholar]
- 35.Bånrud H, Stokke T, Moan J, Kristian B. S phase arrest and induction of multinucleated cells after exposure to ultraviolet radiation. Carcinogenesis. 1995;16(5):1087–1094. [DOI] [PubMed] [Google Scholar]